Virion Secretion of Hepatitis B Virus Naturally Occurring Core Antigen Variants
Abstract
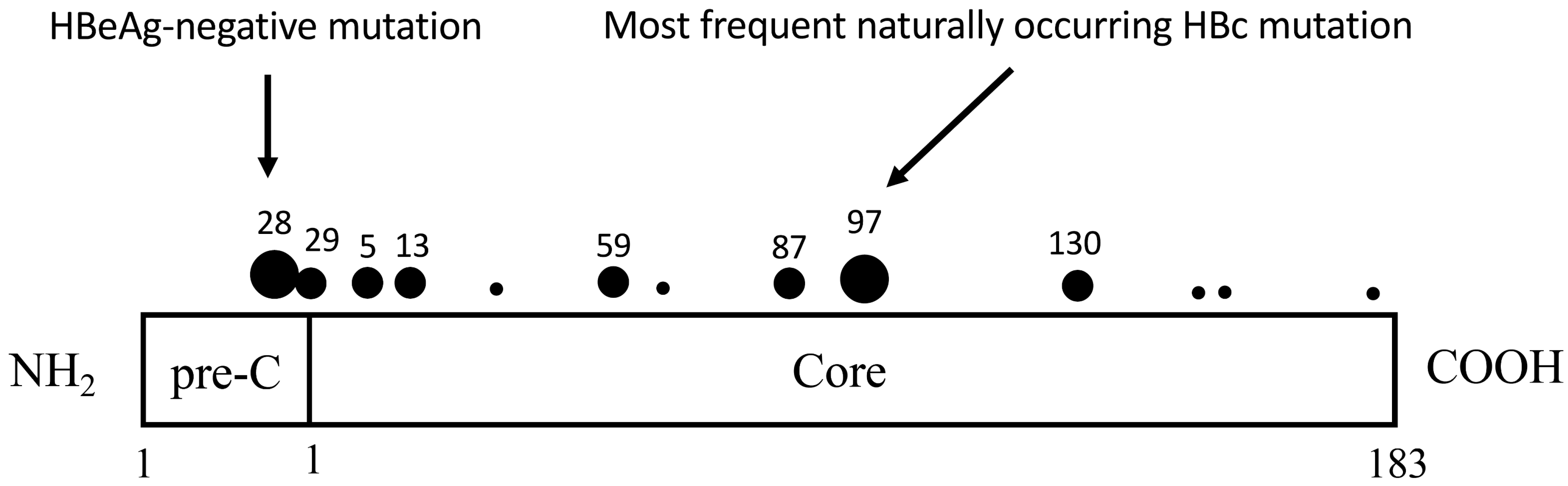
1. Introduction
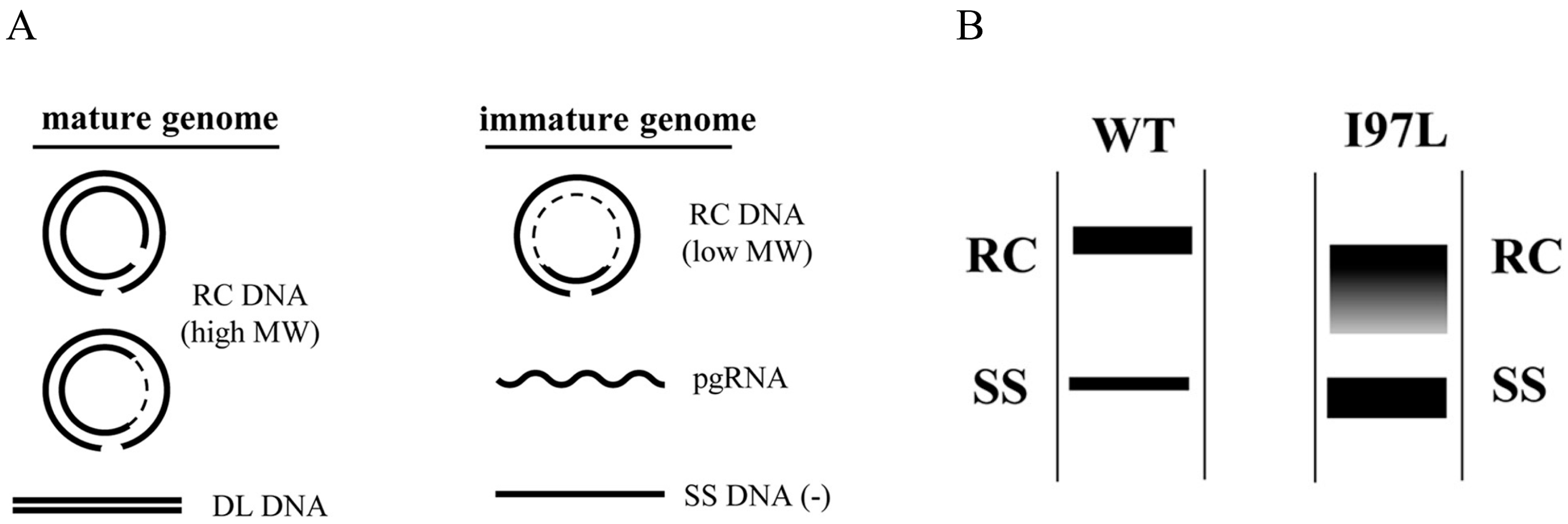
2. An Immature Secretion Phenotype of Mutant 97L
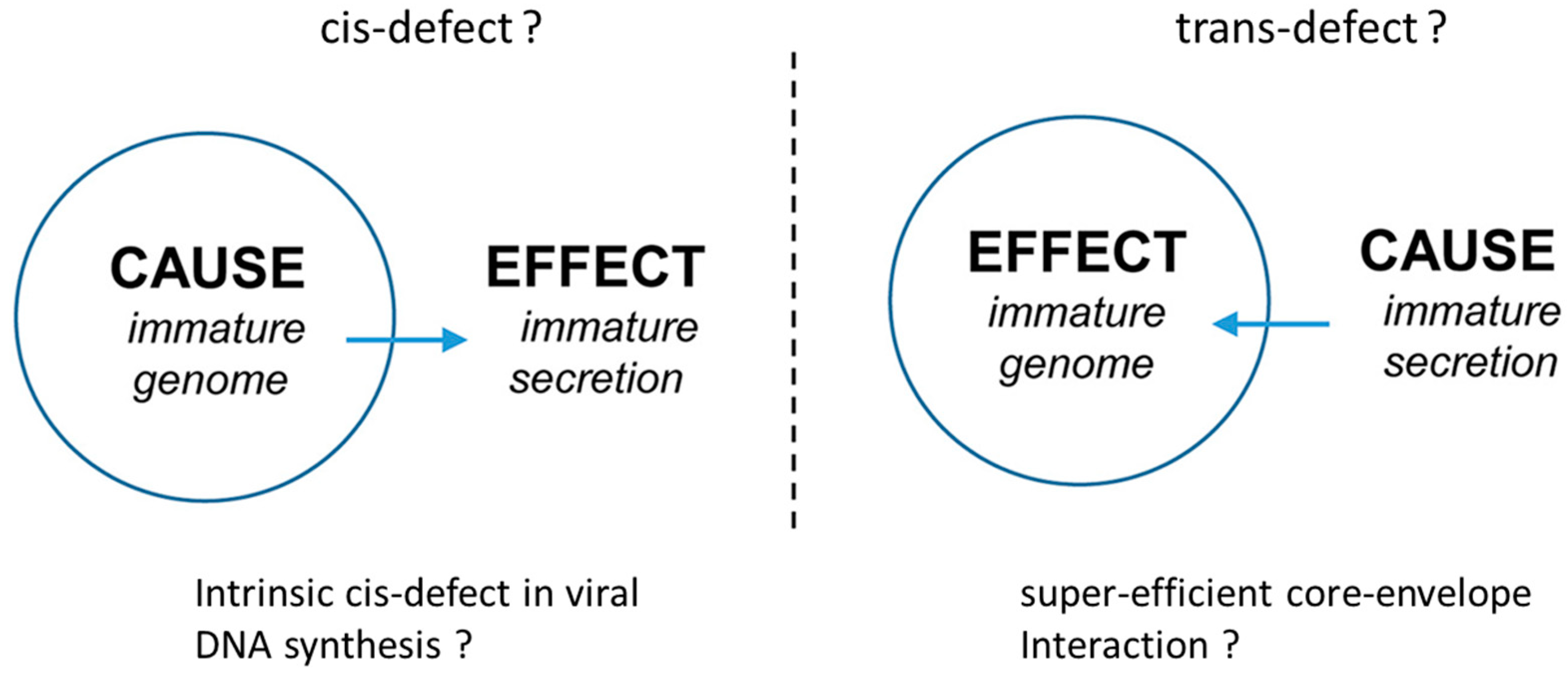
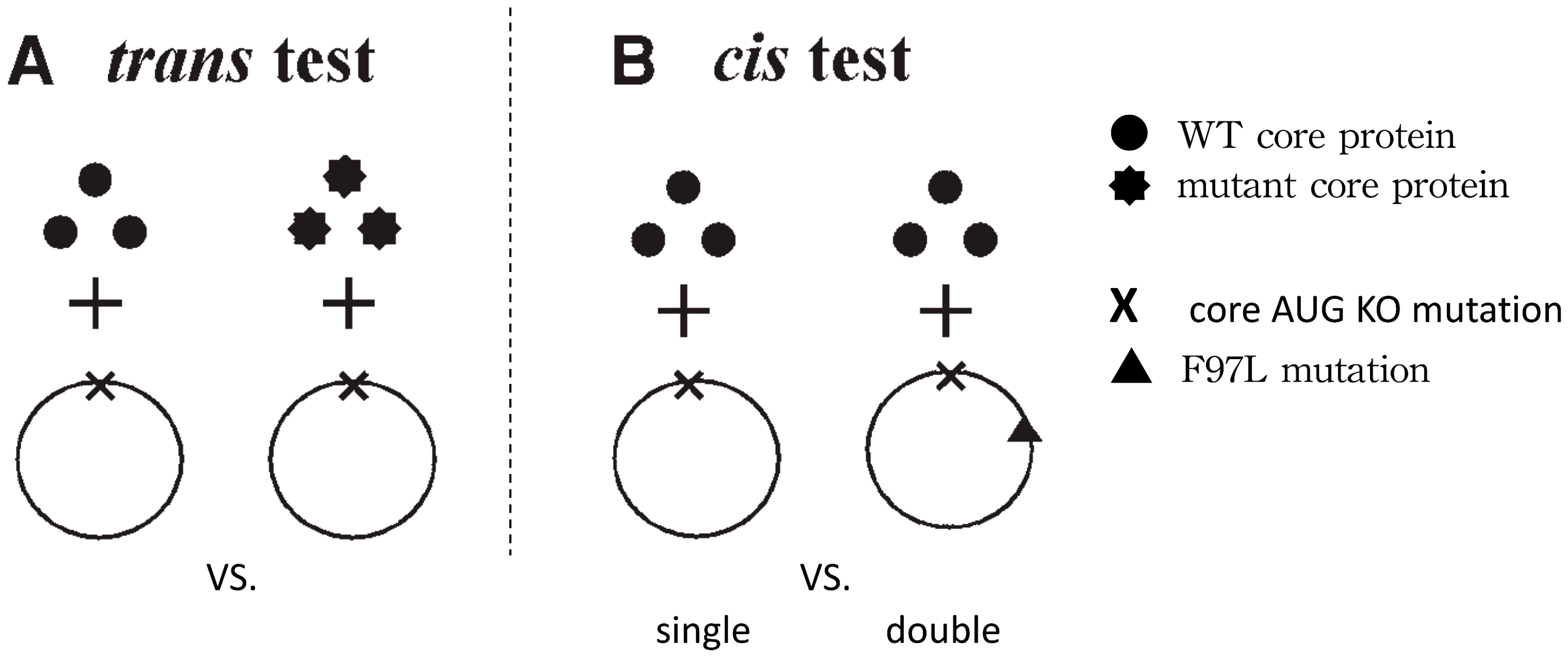
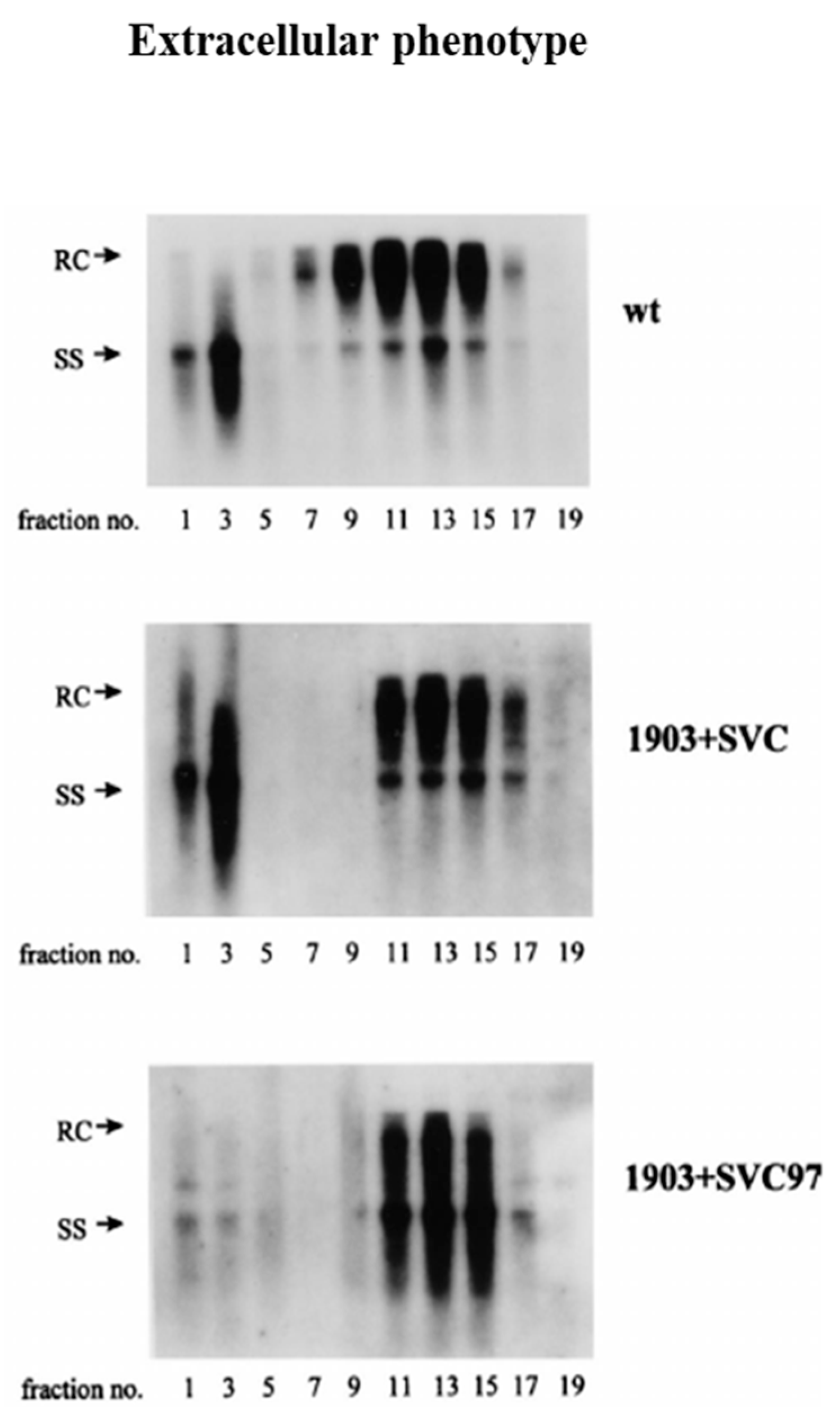
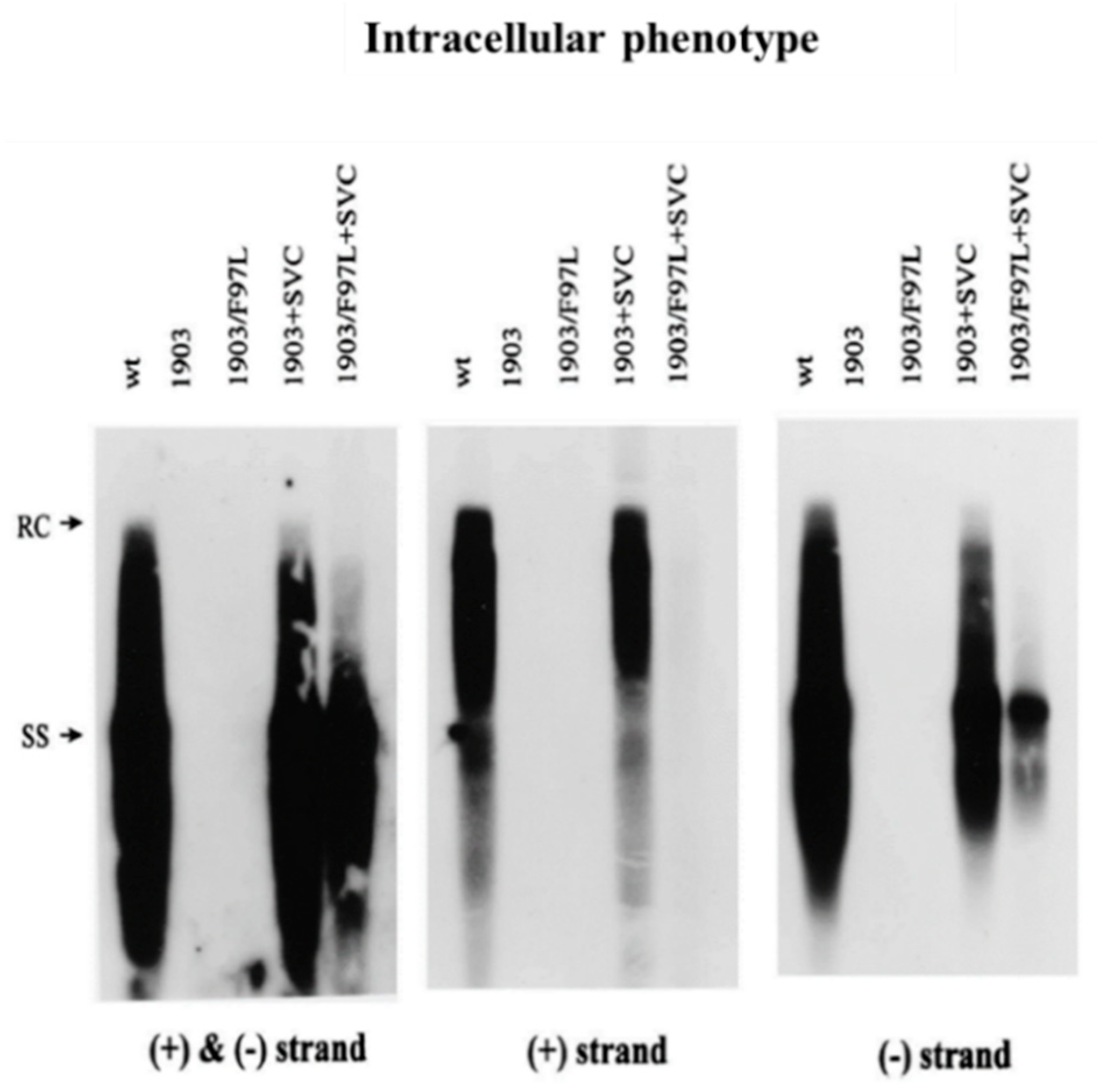
3. A Cis-Trans Genetic Complementation Test
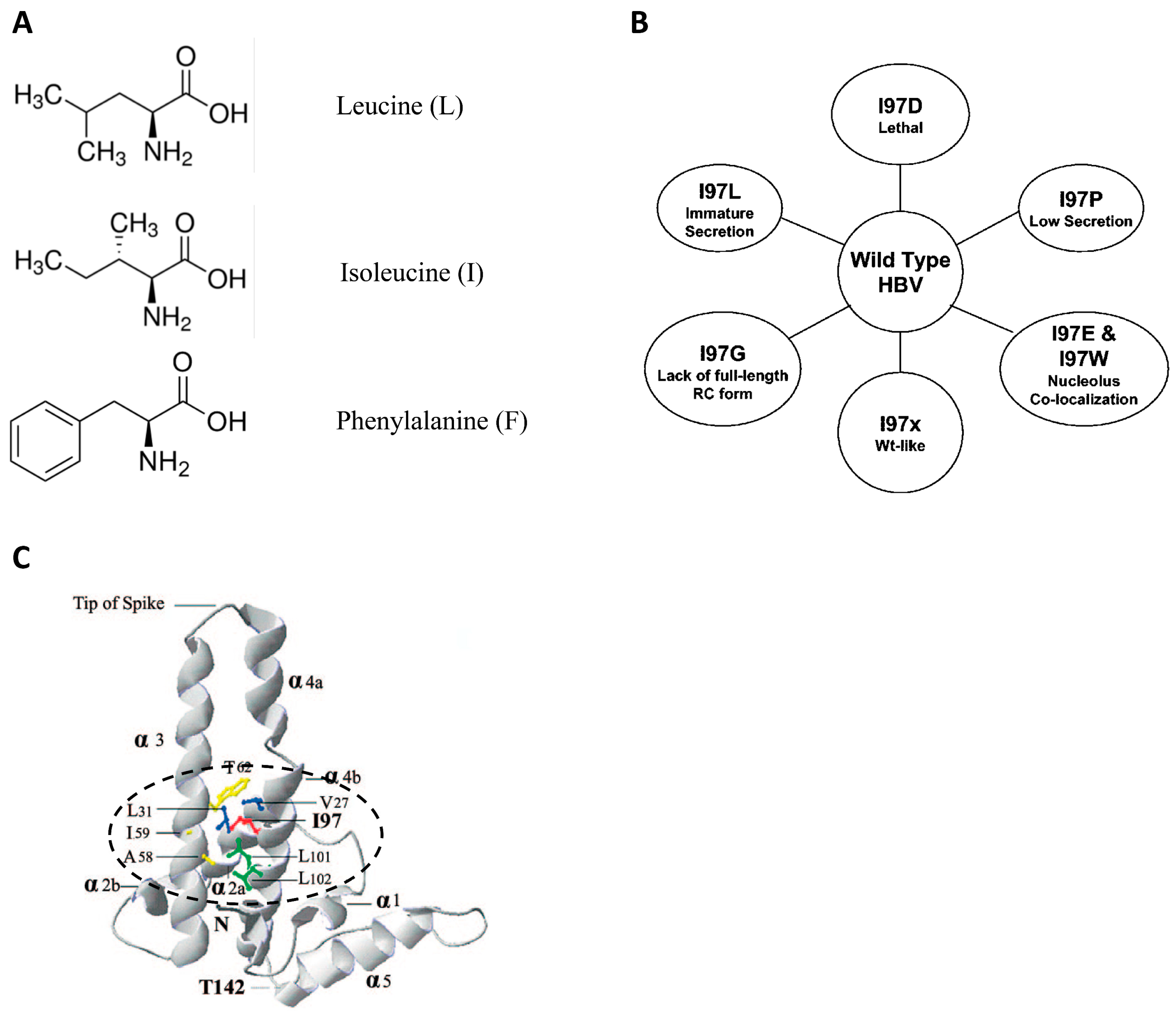
4. A Hydrophobic Pocket around Amino Acid 97 in Signal Transduction
5. Capsid Stability, Assembly, and Morphology of HBc I97L
6. Compensatory Mutations for the Immature Secretion
6.1. Low Virion Secretion and Compensatory Mutations
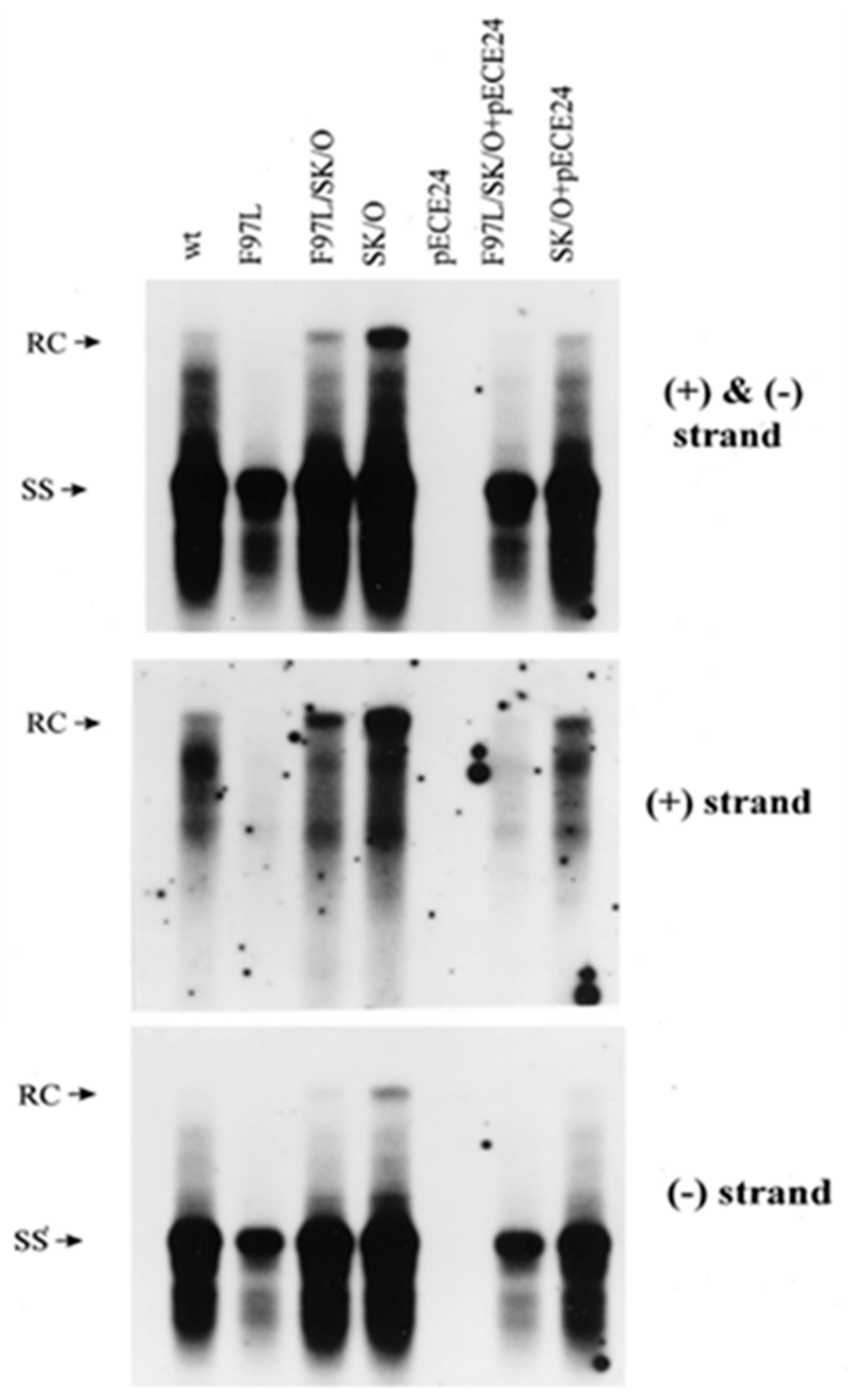
6.2. PreS1 LHBs Compensatory Mutation
7. Host Factors in Virion Secretion
7.1. Vps4 ATPase
7.2. ESCRT Machinery
7.3. Cellular Kinase and HBc Phosphorylation in Virion Secretion
7.4. Host Restriction Factor in Virion Secretion
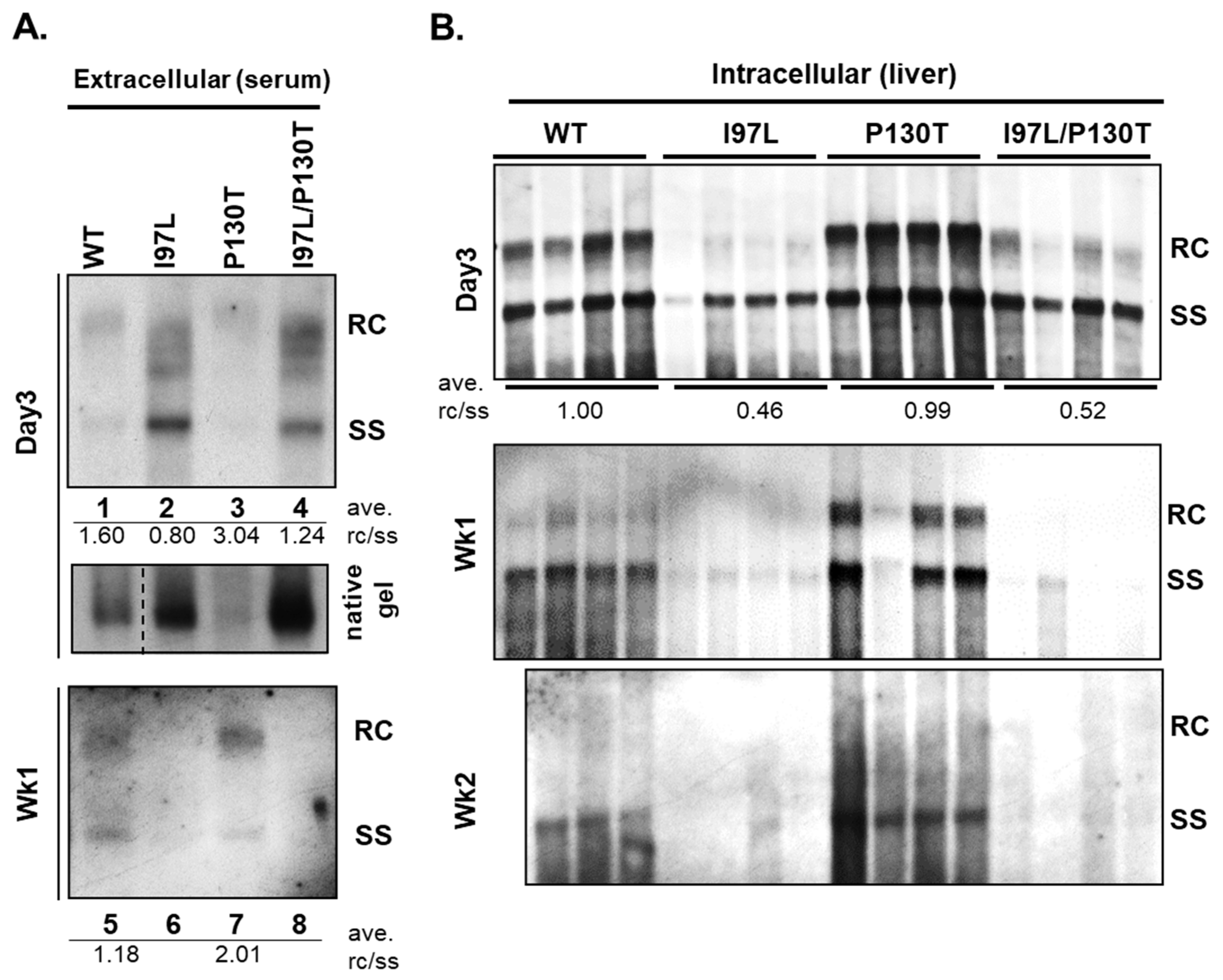
8. Persistence and Genome Maturity
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ARD | arginine-rich domain |
| BST-2 | Bone marrow stromal antigen 2, also known as tetherin |
| cccDNA | covalently closed circular DNA |
| CDK2 | cyclin-dependent kinase 2 |
| CLP | capsid-like particles |
| CTL | cytotoxic T-lymphocytes |
| DHBc | DHBV core (capsid) protein |
| DHBV | duck hepatitis B virus |
| DS DNA | double-stranded DNA |
| ESCRT | The Endosomal Sorting Complex Required for Transport |
| GPI | glycosylphosphatidylinositol |
| HBc | HBV core protein |
| HBeAg | HBV e antigen |
| HBsAg | HBV surface antigen |
| HBV | human hepatitis B virus |
| HGS | hepatocyte growth factor-regulated tyrosine kinase substrate |
| HTLV-2 | Human T Cell Leukemia Virus Type 2 |
| ISG | interferon-stimulated genes |
| NTCP | sodium taurocholate cotransporting polypeptide |
| PEG | polyethylene glycol |
| PKA | cyclic AMP dependent protein kinase A |
| PKC | Ca2+ and/or lipid-activated protein kinase C |
| PLK1 | polo-like kinase 1 |
| RC DNA | relaxed circle DNA |
| SGHBV | Snow Goose hepatitis B virus |
| SKO | HBsAg protein synthesis knockout (SKO) |
| SRPK1 and 2 | serine/arginine-rich protein kinase 1 and 2 |
| SS DNA | single-stranded DNA |
| WHV | woodchuck hepatitis B virus |
| WT | wild type |
References
- Blumberg, B.S. Australia antigen and the biology of hepatitis B. Science 1977, 197, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Purcell, R.H. Hepatitis viruses: Changing patterns of human disease. Proc. Natl. Acad. Sci. USA 1994, 91, 2401–2406. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.; Yang, C.-C.; Choijilsuren, G.; Chang, C.-H.; Liou, A.-T. Hepatitis B Virus. Trends Microbiol. 2018, 26, 386–387. [Google Scholar] [CrossRef] [PubMed]
- Yuen, M.F.; Chen, D.S.; Dusheiko, G.M.; Janssen, H.L.A.; Lau, D.T.Y.; Locarnini, S.A.; Peters, M.G.; Lai, C.L. Hepatitis B virus infection. Nat. Rev. Dis. Prim. 2018, 4, 18035. [Google Scholar] [CrossRef] [PubMed]
- Revill, P.; Testoni, B.; Locarnini, S.; Zoulim, F. Global strategies are required to cure and eliminate HBV infection. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.; Chou, S.-F.; Yang, C.-C.; Huang, J.-Y.; Choijilsuren, G.; Jhou, R.-S. Control and Eradication Strategies of Hepatitis B Virus. Trends Microbiol. 2016, 24, 739–749. [Google Scholar] [CrossRef]
- Nassal, M. HBV cccDNA: Viral persistence reservoir and key obstacle for a cure of chronic hepatitis B. Gut 2015, 64, 1972–1984. [Google Scholar] [CrossRef]
- Ehata, T.; Omata, M.; Yokosuka, O.; Hosoda, K.; Ohto, M. Variations in codons 84-101 in the core nucleotide sequence correlate with hepatocellular injury in chronic hepatitis B virus infection. J. Clin. Investig. 1992, 89, 332–338. [Google Scholar] [CrossRef]
- Ehata, T.; Omata, M.; Chuang, W.L.; Yokosuka, O.; Ito, Y.; Hosoda, K.; Ohto, M. Mutations in core nucleotide sequence of hepa-titis B virus correlate with fulminant and severe hepatitis. J. Clin. Investig. 1993, 91, 1206–1213. [Google Scholar] [CrossRef]
- Chuang, W.L.; Omata, M.; Ehata, T.; Yokosuka, O.; Ohto, M. Concentrating missense mutations in core gene of hepatitis B virus. Evidence for adaptive mutation in chronic hepatitis B virus infection. Dig. Dis. Sci. 1993, 38, 594–600. [Google Scholar] [CrossRef]
- Shih, C.; Burke, K.; Chou, M.J.; Zeldis, J.B.; Yang, C.S.; Lee, C.S.; Isselbacher, K.J.; Wands, J.R.; Goodman, H.M. Tight clustering of human hepatitis B virus integration sites in hepatomas near a triple-stranded region. J. Virol. 1987, 61, 3491–3498. [Google Scholar] [CrossRef] [PubMed]
- Hosono, S.; Tai, P.-C.; Wang, W.; Ambrose, M.; Hwang, D.G.-Y.; Yuan, T.-T.; Peng, B.-H.; Yang, C.-S.; Lee, C.-S.; Shih, C. Core Antigen Mutations of Human Hepatitis B Virus in Hepatomas Accumulate in MHC Class II-Restricted T Cell Epitopes. Virology 1995, 212, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Pollicino, T.; Campo, S.; Raimondo, G. PreS and Core Gene Heterogeneity in Hepatitis B Virus (HBV) Genomes Isolated from Patients with Long-Lasting HBV Chronic Infection. Virology 1995, 208, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Menne, S.; Maschke, J.; Tolle, T.K.; Lu, M.; Roggendorf, M. Characterization of T-cell response to woodchuck hepatitis virus core protein and protection of woodchucks from infection by immunization with peptides containing a T-cell epitope. J. Virol. 1997, 71, 65–74. [Google Scholar] [CrossRef]
- Shanmuganathan, S.; Waters, J.A.; Karayiannis, P.; Thursz, M.; Thomas, H.C. Mapping of the cellular immune responses to woodchuck hepatitis core antigen epitopes in chronically infected woodchucks. J. Med. Virol. 1997, 52, 128–135. [Google Scholar] [CrossRef]
- Summers, J.; Mason, W.S. Replication of the genome of a hepatitis B-like virus by reverse transcription of an RNA intermedi-ate. Cell 1982, 29, 403–415. [Google Scholar] [CrossRef]
- Yuan, T.T.-T.; Sahu, G.K.; Whitehead, W.E.; Greenberg, R.; Shih, C. The Mechanism of an Immature Secretion Phenotype of a Highly Frequent Naturally Occurring Missense Mutation at Codon 97 of Human Hepatitis B Virus Core Antigen. J. Virol. 1999, 73, 5731–5740. [Google Scholar] [CrossRef]
- Yuan, T.T.-T.; Tai, P.-C.; Shih, C. Subtype-Independent Immature Secretion and Subtype-Dependent Replication Deficiency of a Highly Frequent, Naturally Occurring Mutation of Human Hepatitis B Virus Core Antigen. J. Virol. 1999, 73, 10122–10128. [Google Scholar] [CrossRef]
- Schormann, W.; Kraft, A.; Ponsel, D.; Bruss, V. Hepatitis B Virus Particle Formation in the Absence of Pregenomic RNA and Reverse Transcriptase. J. Virol. 2006, 80, 4187–4190. [Google Scholar] [CrossRef]
- Chang, S.-F.; Netter, H.J.; Bruns, M.; Schneider, R.; Frölich, K.; Will, H. A New Avian Hepadnavirus Infecting Snow Geese (Anser caerulescens) Produces a Significant Fraction of Virions Containing Single-Stranded DNA. Virology 1999, 262, 39–54. [Google Scholar] [CrossRef]
- Greco, N.; Hayes, M.H.; Loeb, D.D. Snow goose hepatitis B virus (SGHBV) envelope and capsid proteins independently con-tribute to the ability of SGHBV to package capsids containing single-stranded DNA in virions. J. Virol. 2014, 88, 10705–10713. [Google Scholar] [CrossRef] [PubMed]
- Tencza, M.G.; Newbold, J.E. Heterogeneous response for a mammalian hepadnavirus infection to acyclovir: Drug-arrested intermediates of minus-strand viral DNA synthesis are enveloped and secreted from infected cells as virion-like particles. J. Med. Virol. 1997, 51, 6–16. [Google Scholar] [CrossRef]
- Wu, S.-Y.; Chang, Y.-S.; Chu, T.-H.; Shih, C. Persistence of Hepatitis B Virus DNA and the Tempos between Virion Secretion and Genome Maturation in a Mouse Model. J. Virol. 2019, 93, 01001-19. [Google Scholar] [CrossRef] [PubMed]
- Le Pogam, S.; Shih, C. Influence of a Putative Intermolecular Interaction between Core and the Pre-S1 Domain of the Large Envelope Protein on Hepatitis B Virus Secretion. J. Virol. 2002, 76, 6510–6517. [Google Scholar] [CrossRef]
- Shih, C.; Yuan, T.T. A cis/trans Genetic Test for Pleiotropic Phenotypes Associated with a Frequent Naturally Occurring Mutation at Amino Acid 97 of HBV Core Protein. Methods Mol. Med. 2004, 95, 247–257. [Google Scholar]
- Bruss, V.; Ganem, D. The role of envelope proteins in hepatitis B virus assembly. Proc. Natl. Acad. Sci. USA 1991, 88, 1059–1063. [Google Scholar] [CrossRef]
- Crowther, R.A.; Kiselev, N.A.; Böttcher, B.; Berriman, J.A.; Borisova, G.P.; Ose, V.; Pumpens, P. Three-dimensional structure of hepatitis B vi-rus core particles determined by electron cryomicroscopy. Cell 1994, 77, 943–950. [Google Scholar] [CrossRef]
- Böttcher, B.; Wynne, S.A.; Crowther, R.A. Determination of the fold of the core protein of hepatitis B virus by electron cry-omicroscopy. Nature 1997, 386, 88–91. [Google Scholar] [CrossRef]
- Conway, J.F.; Cheng, N.; Zlotnick, A.; Wingfield, P.T.; Stahl, S.J.; Steven, A.C. Visualization of a 4-helix bundle in the hepatitis B virus capsid by cryo-electron microscopy. Nat. Cell Biol. 1997, 386, 91–94. [Google Scholar] [CrossRef]
- Wynne, S.; Crowther, R.; Leslie, A. The Crystal Structure of the Human Hepatitis B Virus Capsid. Mol. Cell 1999, 3, 771–780. [Google Scholar] [CrossRef]
- Ning, B.; Shih, C. Nucleolar Localization of Human Hepatitis B Virus Capsid Protein. J. Virol. 2004, 78, 13653–13668. [Google Scholar] [CrossRef] [PubMed]
- Roseman, A.M.; Berriman, J.A.; Wynne, S.A.; Butler, P.J.G.; Crowther, R.A. A structural model for maturation of the hepatitis B virus core. Proc. Natl. Acad. Sci. USA 2005, 102, 15821–15826. [Google Scholar] [CrossRef] [PubMed]
- Böttcher, B.; Nassal, M. Structure of Mutant Hepatitis B Core Protein Capsids with Premature Secretion Phenotype. J. Mol. Biol. 2018, 430, 4941–4954. [Google Scholar] [CrossRef] [PubMed]
- Salfeld, J.; Pfaff, E.; Noah, M.; Schaller, H. Antigenic determinants and functional domains in core antigen and e antigen from hepatitis B virus. J. Virol. 1989, 63, 798–808. [Google Scholar] [CrossRef]
- Gerin, J.L.; Ford, E.C.; Purcell, R.H. Biochemical characterization of Australia antigen: Evidence for defective particles of hep-atitis B virus. Am. J. Pathol. 1975, 81, 651–668. [Google Scholar]
- Kaplan, P.M.; Ford, E.C.; Purcell, R.H.; Gerin, J.L. Demonstration of subpopulations of Dane particles. J. Virol. 1976, 17, 885–893. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Yamada, G.; Mizuno, M.; Nishihara, T.; Kinoyama, S.; Kobayashi, T.; Takahashi, T.; Nagashima, H. Full and empty particles of hepatitis B virus in hepatocytes from patients with HBsAg-positive chronic active hepatitis. Lab Investig. 1983, 48, 678–682. [Google Scholar]
- Kimura, T.; Ohno, N.; Terada, N.; Rokuhara, A.; Matsumoto, A.; Yagi, S.; Tanaka, E.; Kiyosawa, K.; Ohno, S.; Maki, N. Hepatitis B virus DNA-negative Dane particles lack core protein but contain a 22-kDa precore protein without C-terminal argi-nine-rich domain. J. Biol. Chem. 2005, 280, 21713–21719. [Google Scholar] [CrossRef]
- Ning, X.; Nguyen, D.; Mentzer, L.; Adams, C.; Lee, H.; Ashley, R.; Hafenstein, S.; Hu, J. Secretion of Genome-Free Hepatitis B Virus–Single Strand Blocking Model for Virion Morphogenesis of Para-retrovirus. PLoS Pathog. 2011, 7, e1002255. [Google Scholar] [CrossRef]
- Newman, M.; Suk, F.M.; Cajimat, M.; Chua, P.K.; Shih, C. Stability and Morphology Comparisons of Self-assembled Vi-rus-Like Particles from Wild Type and Mutant Human Hepatitis B Virus Capsid Proteins. J. Virol. 2003, 77, 12950–12960. [Google Scholar] [CrossRef]
- Heger-Stevic, J.; Zimmermann, P.; Lecoq, L.; Böttcher, B.; Nassal, M. Hepatitis B virus core protein phosphorylation: Identifi-cation of the SRPK1 target sites and impact of their occupancy on RNA binding and capsid structure. PLoS Pathog. 2018, 14, e1007488. [Google Scholar] [CrossRef] [PubMed]
- Ceres, P.; Stray, S.J.; Zlotnick, A. Hepatitis B Virus Capsid Assembly Is Enhanced by Naturally Occurring Mutation F97L. J. Virol. 2004, 78, 9538–9543. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.T.-T.; Shih, C. A Frequent, Naturally Occurring Mutation (P130T) of Human Hepatitis B Virus Core Antigen Is Compensatory for Immature Secretion Phenotype of Another Frequent Variant (I97L). J. Virol. 2000, 74, 4929–4932. [Google Scholar] [CrossRef] [PubMed]
- Le Pogam, S.; Yuan, T.T.-T.; Sahu, G.K.; Chatterjee, S.; Shih, C. Low-Level Secretion of Human Hepatitis B Virus Virions Caused by Two Independent, Naturally Occurring Mutations (P5T and L60V) in the Capsid Protein. J. Virol. 2000, 74, 9099–9105. [Google Scholar] [CrossRef]
- Ponsel, D.; Bruss, V. Mapping of Amino Acid Side Chains on the Surface of Hepatitis B Virus Capsids Required for Envelopment and Virion Formation. J. Virol. 2003, 77, 416–422. [Google Scholar] [CrossRef]
- Nassal, M. The arginine-rich domain of the hepatitis B virus core protein is required for pregenome encapsidation and pro-ductive viral positive-strand DNA synthesis but not for virus assembly. J. Virol. 1992, 66, 4107–4116. [Google Scholar] [CrossRef]
- Kock, J.; Nassal, M.; Deres, K.; Blum, H.E.; Von Weizsäcker, F. Hepatitis B Virus Nucleocapsids Formed by Carboxy-Terminally Mutated Core Proteins Contain Spliced Viral Genomes but Lack Full-Size DNA. J. Virol. 2004, 78, 13812–13818. [Google Scholar] [CrossRef]
- Le Pogam, S.; Chua, P.K.; Newman, M.; Shih, C. Exposure of RNA Templates and Encapsidation of Spliced Viral RNA Are Influenced by the Arginine-Rich Domain of Human Hepatitis B Virus Core Antigen (HBcAg 165-173). J. Virol. 2005, 79, 1871–1887. [Google Scholar] [CrossRef]
- Su, P.Y.; Yang, C.J.; Chu, T.H.; Chang, C.H.; Chiang, C.; Tang, F.M.; Lee, C.Y.; Shih, C. HBV maintains electrostatic homeosta-sis by modulating negative charges from phosphoserine and encapsidated nucleic acids. Sci. Rep. 2016, 6, 38959. [Google Scholar] [CrossRef]
- Chua, P.K.; Wen, Y.M.; Shih, C. Coexistence of two distinct secretion mutations (P5T and I97L) in hepatitis B virus core pro-duces a wild-type pattern of secretion. J. Virol. 2003, 77, 7673–7676. [Google Scholar] [CrossRef]
- Chua, P.K.; Wang, R.Y.-L.; Lin, M.-H.; Masuda, T.; Suk, F.-M.; Shih, C. Reduced Secretion of Virions and Hepatitis B Virus (HBV) Surface Antigen of a Naturally Occurring HBV Variant Correlates with the Accumulation of the Small S Envelope Protein in the Endoplasmic Reticulum and Golgi Apparatus. J. Virol. 2005, 79, 13483–13496. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.; Chua, P.K.; Tang, F.-M.; Su, P.-Y.; Shih, C. Testing an Electrostatic Interaction Hypothesis of Hepatitis B Virus Capsid Stability by Using an In Vitro Capsid Disassembly/Reassembly System. J. Virol. 2009, 83, 10616–10626. [Google Scholar] [CrossRef] [PubMed]
- Chua, P.K.; Tang, F.-M.; Huang, J.-Y.; Suen, C.-S.; Shih, C. Testing the Balanced Electrostatic Interaction Hypothesis of Hepatitis B Virus DNA Synthesis by Using an In Vivo Charge Rebalance Approach. J. Virol. 2010, 84, 2340–2351. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tai, P.C.; Banik, D.; Lin, G.I.; Pai, S.; Pai, K.; Lin, M.H.; Yuoh, G.; Che, S.; Hsu, S.H.; Chen, T.C.; et al. Novel and frequent mutations of hepatitis B virus coincide with an MHC class I-restricted T cell epitope of the sur-face antigen. J. Virol. 1997, 71, 4852–4856. [Google Scholar] [CrossRef]
- Tai, P.C.; Suk, F.M.; Gerlich, W.; Neurath, R.; Shih, C. Hypermodification and immune escape of an internally deleted middle envelope (M) protein of frequent and predominant hepatitis B virus variants. Virology 2002, 292, 44–58. [Google Scholar] [CrossRef]
- Wang, H.-C.; Wu, H.-C.; Chen, C.-F.; Fausto, N.; Lei, H.-Y.; Su, I.-J. Different Types of Ground Glass Hepatocytes in Chronic Hepatitis B Virus Infection Contain Specific Pre-S Mutants that May Induce Endoplasmic Reticulum Stress. Am. J. Pathol. 2003, 163, 2441–2449. [Google Scholar] [CrossRef]
- Sugauchi, F.; Ohno, T.; Orito, E.; Sakugawa, H.; Ichida, T.; Komatsu, M.; Kuramitsu, T.; Ueda, R.; Miyakawa, Y.; Mizokami, M. Influence of hepatitis B virus genotypes on the development of preS deletions and advanced liver disease. J. Med. Virol. 2003, 70, 537–544. [Google Scholar] [CrossRef]
- Su, I.-J.; Wang, L.H.-C.; Hsieh, W.-C.; Wu, H.-C.; Teng, C.-F.; Tsai, H.-W.; Huang, W. The emerging role of hepatitis B virus Pre-S2 deletion mutant proteins in HBV tumorigenesis. J. Biomed. Sci. 2014, 21, 1–8. [Google Scholar] [CrossRef]
- Teng, C.-F.; Li, T.; Huang, H.-Y.; Lin, J.-H.; Chen, W.-S.; Shyu, W.-C.; Wu, H.-C.; Peng, C.-Y.; Su, I.-J.; Jeng, L.-B. Next-Generation Sequencing-Based Quantitative Detection of Hepatitis B Virus Pre-S Mutants in Plasma Predicts Hepatocellular Carcinoma Recurrence. Viruses 2020, 12, 796. [Google Scholar] [CrossRef]
- Lin, W.L.; Hung, J.H.; Huang, W. Association of the Hepatitis B Virus Large Surface Protein with Viral Infectivity and Endo-plasmic Reticulum Stress-mediated Liver Carcinogenesis. Cells 2020, 9, 2052. [Google Scholar] [CrossRef]
- Chou, S.-F.; Tsai, M.-L.; Huang, J.-Y.; Chang, Y.-S.; Shih, C. The Dual Role of an ESCRT-0 Component HGS in HBV Transcription and Naked Capsid Secretion. PLoS Pathog. 2015, 11, e1005123. [Google Scholar] [CrossRef] [PubMed]
- Chua, P.K.; Lin, M.-H.; Shih, C. Potent inhibition of human Hepatitis B virus replication by a host factor Vps. Virology 2006, 354, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Lambert, C.; Döring, T.; Prange, R. Hepatitis B Virus Maturation Is Sensitive to Functional Inhibition of ESCRT-III, Vps4, and γ2-Adaptin. J. Virol. 2007, 81, 9050–9060. [Google Scholar] [CrossRef] [PubMed]
- Suk, F.-M.; Lin, M.-H.; Newman, M.; Pan, S.; Chen, S.-H.; Liu, J.-D.; Shih, C. Replication Advantage and Host Factor-Independent Phenotypes Attributable to a Common Naturally Occurring Capsid Mutation (I97L) in Human Hepatitis B Virus. J. Virol. 2002, 76, 12069–12077. [Google Scholar] [CrossRef][Green Version]
- Torii, S.; Orba, Y.; Sasaki, M.; Tabata, K.; Wada, Y.; Carr, M.; Hobson-Peters, J.; Hall, R.A.; Takada, A.; Fukuhara, T.; et al. Host ESCRT factors are recruited during chikungunya virus infection and are required for the in-tracellular viral replication cycle. J. Biol. Chem. 2020, 295, 7941–7957. [Google Scholar] [CrossRef]
- Martini, F.; Arone, C.; Hasset, A.; Hall, W.W.; Sheehy, N. The ESCRT-0 Protein HRS Interacts with the Human T Cell Leuke-mia VirusType 2 Antisense Protein APH-2 and Suppresses Viral Replication. J. Virol. 2019, 94, e01311-19. [Google Scholar] [CrossRef]
- Stieler, J.T.; Prange, R. Involvement of ESCRT-II in Hepatitis B Virus Morphogenesis. PLoS ONE 2014, 9, e91279. [Google Scholar] [CrossRef]
- Prange, R. Host factors involved in hepatitis B virus maturation, assembly, and egress. Med. Microbiol. Immunol. 2012, 201, 449–461. [Google Scholar] [CrossRef]
- Zeyen, L.; Döring, T.; Prange, R. Hepatitis B Virus Exploits ERGIC-53 in Conjunction with COPII to Exit Cells. Cells 2020, 9, 1889. [Google Scholar] [CrossRef]
- Albin, C.; Robinson, W.S. Protein Kinase Activity in Hepatitis B Virus. J. Virol. 1980, 34, 297–302. [Google Scholar] [CrossRef]
- Gerlich, W.H.; Goldmann, U.; Müller, R.; Stibbe, W.; Wolff, W. Specificity and localization of the hepatitis B virus-associated protein kinase. J. Virol. 1982, 42, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Pugh, J.; Zweidler, A.; Summers, J. Characterization of the major duck hepatitis B virus core particle protein. J. Virol. 1989, 63, 1371–1376. [Google Scholar] [CrossRef] [PubMed]
- Perlman, D.H.; Berg, E.A.; O’Connor, P.B.; Costello, C.E.; Hu, J. Reverse transcription-associated dephosphorylation of hepadnavirus nucleocapsids. Proc. Natl. Acad. Sci. USA 2005, 102, 9020–9025. [Google Scholar] [CrossRef] [PubMed]
- Ning, X.; Basagoudanavar, S.H.; Liu, K.; Luckenbaugh, L.; Wei, D.; Wang, C.; Wei, B.; Zhao, Y.; Yan, T.; Delaney, W.; et al. Capsid Phosphorylation State and Hepadnavirus Virion Secretion. J. Virol. 2017, 91, e00092-17. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.T.; Li, J.; Liao, W.; Ou, J. Roles of the three major phosphorylation sites of hepatitis B virus core protein in viral replica-tion. Virology 1999, 259, 342–348. [Google Scholar] [CrossRef]
- Gazina, E.V.; Fielding, J.E.; Lin, B.; Anderson, D.A. Core Protein Phosphorylation Modulates Pregenomic RNA Encapsidation to Different Extents in Human and Duck Hepatitis B Viruses. J. Virol. 2000, 74, 4721–4728. [Google Scholar] [CrossRef]
- Chu, T.H.; Liou, A.T.; Su, P.Y.; Wu, H.N.; Shih, C. Nucleic acid chaperone activity associated with the ARD domain of human hepatitis B virus core protein. J. Virol. 2014, 88, 2530–2543. [Google Scholar] [CrossRef]
- De Rocquigny, H.; Rat, V.; Pastor, F.; Darlix, J.-L.; Hourioux, C.; Roingeard, P. Phosphorylation of the Arginine-Rich C-Terminal Domains of the Hepatitis B Virus (HBV) Core Protein as a Fine Regulator of the Interaction between HBc and Nucleic Acid. Viruses 2020, 12, 738. [Google Scholar] [CrossRef]
- Yu, M.; Summers, J. Multiple functions of capsid protein phosphorylation in duck hepatitis B virus replication. J. Virol. 1994, 68, 4341–4348. [Google Scholar] [CrossRef]
- Köck, J.; Kann, M.; Pütz, G.; Blum, H.M.H.; Von Weizsäcker, F. Central Role of a Serine Phosphorylation Site within Duck Hepatitis B Virus Core Protein for Capsid Trafficking and Genome Release. J. Biol. Chem. 2003, 278, 28123–28129. [Google Scholar] [CrossRef]
- Daub, H.; Blencke, S.; Habenberger, P.; Kurtenbach, A.; Dennenmoser, J.; Wissing, J.; Ullrich, A.; Cotten, M. Identification of SRPK1 and SRPK2 as the Major Cellular Protein Kinases Phosphorylating Hepatitis B Virus Core Protein. J. Virol. 2002, 76, 8124–8137. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Summers, J. Phosphorylation of the duck hepatitis B virus capsid protein associated with conformational changes in the C terminus. J. Virol. 1994, 68, 2965–2969. [Google Scholar] [CrossRef] [PubMed]
- Kann, M.; Gerlich, W.H. Effect of core protein phosphorylation by protein kinase C on encapsidation of RNA within core par-ticles of hepatitis B virus. J. Virol. 1994, 68, 7993–8000. [Google Scholar] [CrossRef] [PubMed]
- Wittkop, L.; Schwarz, A.; Cassany, A.; Grun-Bernhard, S.; Delaleau, M.; Rabe, B.; Cazenave, C.; Gerlich, W.; Glebe, D.; Kann, M. Inhibition of protein kinase C phos-phorylation of hepatitis B virus capsids inhibits virion formation and causes intracellular capsid accumulation. Cell Microbiol. 2010, 12, 962–975. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J.C.-Y.; Zlotnick, A. A Kinase Chaperones Hepatitis B Virus Capsid Assembly and Captures Capsid Dynamics in vitro. PLoS Pathog. 2011, 7, e1002388. [Google Scholar] [CrossRef]
- Ludgate, L.; Ning, X.; Nguyen, D.H.; Adams, C.; Mentzer, L.; Hu, J. Cyclin-Dependent Kinase 2 Phosphorylates S/T-P Sites in the Hepadnavirus Core Protein C-Terminal Domain and Is Incorporated into Viral Capsids. J. Virol. 2012, 86, 12237–12250. [Google Scholar] [CrossRef]
- Diab, A.M.; Foca, A.; Andrisani, O.M.; Durantel, D.; Fusil, F.; Lahlali, T.; Jalaguier, P.; Amirache, F.; N’Guyen, L.; Isorce, N.; et al. Polo-like-kinase 1 is a proviral host factor for hepatitis B virus replication. Hepatology 2017, 66, 1750–1765. [Google Scholar] [CrossRef]
- Hu, Z.; Ban, H.; Zheng, H.; Liu, M.; Chang, J.; Guo, J.-T. Protein phosphatase 1 catalyzes HBV core protein dephosphorylation and is co-packaged with viral pregenomic RNA into nucleocapsids. PLoS Pathog. 2020, 16, e1008669. [Google Scholar] [CrossRef]
- Lv, M.; Zhang, B.; Shi, Y.; Han, Z.; Zhang, Y.; Zhou, Y.; Zhang, W.; Niu, J.; Yu, X.-F. Identification of BST-2/tetherin-induced hepatitis B virus restriction and hepatocyte-specific BST-2 inactivation. Sci. Rep. 2015, 5, 11736. [Google Scholar] [CrossRef]
- Miyakawa, K.; Matsunaga, S.; Watashi, K.; Sugiyama, M.; Kimura, H.; Yamamoto, N.; Mizokami, M.; Wakita, T.; Ryo, A. Mo-lecular dissection of HBV evasion from restriction factor tetherin: A new perspective for antiviral cell therapy. Oncotarget 2015, 6, 21840–21852. [Google Scholar] [CrossRef]
- Yan, R.; Zhao, X.; Cai, D.; Liu, Y.; Block, T.M.; Guo, J.-T.; Guo, H. The Interferon-Inducible Protein Tetherin Inhibits Hepatitis B Virus Virion Secretion. J. Virol. 2015, 89, 9200–9212. [Google Scholar] [CrossRef] [PubMed]
- Michailidis, E.; Pabon, J.; Xiang, K.; Park, P.; Ramanan, V.; Hoffmann, H.-H.; Schneider, W.M.; Bhatia, S.N.; De Jong, Y.P.; Shlomai, A.; et al. A robust cell culture system supporting the complete life cycle of hepatitis B virus. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-J.; Yang, H.-I. Natural history of chronic hepatitis B revealed. J. Gastroenterol. Hepatol. 2011, 26, 628–638. [Google Scholar] [CrossRef]
- Chen, C.J.; Yang, H.I.; Iloeje, U.H.; REVEAL-HBV Study Group. Hepatitis B virus DNA levels and outcomes in chronic hepatitis B. Hepatology 2009, 49 (Suppl. 5), S72–S84. [Google Scholar] [PubMed]
- Honda, T.; Ishigami, M.; Ishizu, Y.; Kuzuya, T.; Hayashi, K.; Ishikawa, T.; Murakami, Y.; Iwadate, M.; Umeyama, H.; Toyoda, H.; et al. Core I97L mutation in conjunction with P79Q is associated with persistent low HBV DNA and HBs antigen clearance in patients with chronic hepatitis B. Clin. Microbiol. Infect. 2017, 23, 407.e1–407.e7. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liaw, Y. Clinical utility of HBV surface antigen quantification in HBV e antigen-negative chronic HBV infection. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Sarin, S.K.; Kumar, M.P.; Lau, G.K.; Abbas, Z.; Chan, H.L.Y.; Chen, C.J.; Chen, D.S.; Chen, H.L.; Chien, R.N.; Dokmeci, A.; et al. Asian-Pacific clinical practice guidelines on the management of hepatitis B: A 2015 update. Hepatol. Int. 2016, 10, 1–98. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL 2017 clinical practice guidelines on the management of hepatitis B vi-rus infection. J. Hepatol. 2017, 67, 370–398. [Google Scholar] [CrossRef]
- Terrault, N.; Lok, A.S.; McMahon, B.J.; Chang, K.-M.; Hwang, J.P.; Jonas, M.M.; Brown, R.S., Jr.; Bzowej, N.H.; Wong, J.B. Update on prevention, diagnosis, and treatment of chronic hepatitis B: AASLD 2018 hepatitis B guidance. Hepatology 2018, 67, 1560–1599. [Google Scholar] [CrossRef]
- Liaw, Y. Finite nucleos(t)ide analog therapy in HBeAg-negative chronic hepatitis B: An emerging paradigm shift. Hepatol. Int. 2019, 13, 665–673. [Google Scholar] [CrossRef]
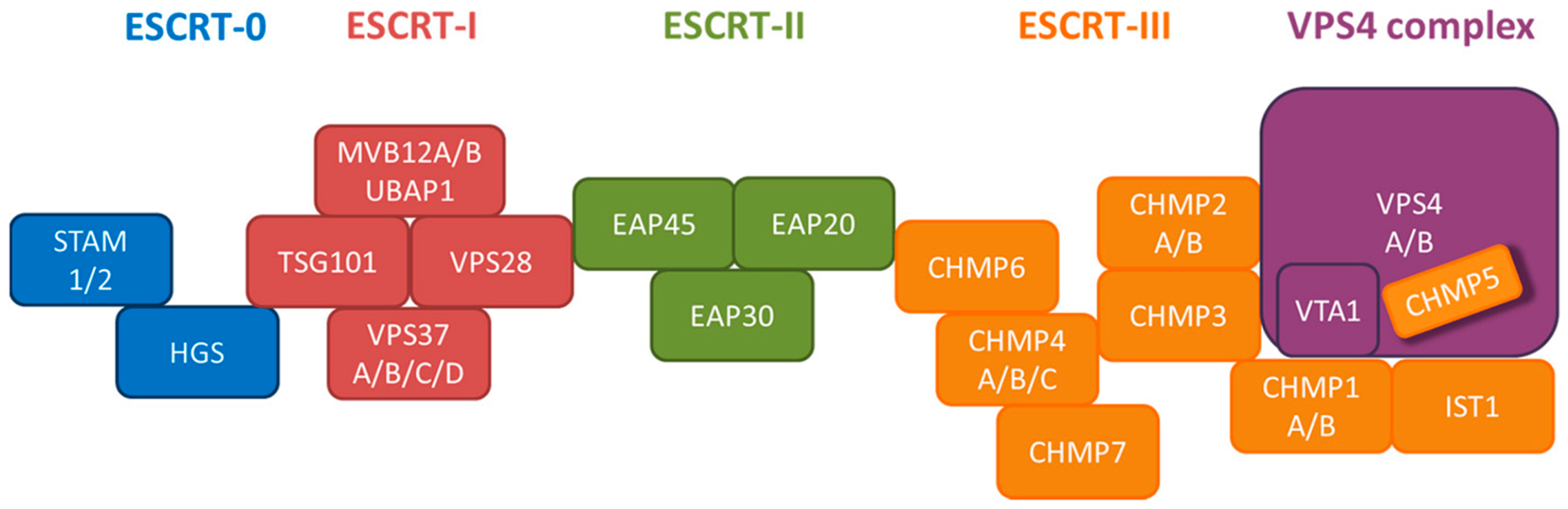
| Complex | Suppression of HBV DNA Replication |
|---|---|
| ESCRT-0 | si-HGS |
| si-STAM1, 2 | |
| ESCRT-I | si-VPS28 |
| si-VPS37B * | |
| si-UBAP1 | |
| ESCRT-II | si-EAP20 |
| si-EAP45 | |
| ESCRT-III & related factors | si-CHMP4A, B * |
| si-CHMP3 | |
| si-CHMP2A, B | |
| si-CHMP1A, B | |
| si-IST1 | |
| VPS4 complex | si-VPS4A |
| si-VTA1 * |
| HBc Variants | Genome Maturity * | Persistence # |
|---|---|---|
| I97L | + | + |
| WT (adr) | ++ | ++ |
| P130T | +++ | +++ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shih, C.; Wu, S.-Y.; Chou, S.-F.; Yuan, T.-T.T. Virion Secretion of Hepatitis B Virus Naturally Occurring Core Antigen Variants. Cells 2021, 10, 43. https://doi.org/10.3390/cells10010043
Shih C, Wu S-Y, Chou S-F, Yuan T-TT. Virion Secretion of Hepatitis B Virus Naturally Occurring Core Antigen Variants. Cells. 2021; 10(1):43. https://doi.org/10.3390/cells10010043
Chicago/Turabian StyleShih, Chiaho, Szu-Yao Wu, Shu-Fan Chou, and Ta-Tung Thomas Yuan. 2021. "Virion Secretion of Hepatitis B Virus Naturally Occurring Core Antigen Variants" Cells 10, no. 1: 43. https://doi.org/10.3390/cells10010043
APA StyleShih, C., Wu, S.-Y., Chou, S.-F., & Yuan, T.-T. T. (2021). Virion Secretion of Hepatitis B Virus Naturally Occurring Core Antigen Variants. Cells, 10(1), 43. https://doi.org/10.3390/cells10010043





