Insect Pest Incidence with the System of Rice Intensification: Results of a Multi-Location Study and a Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
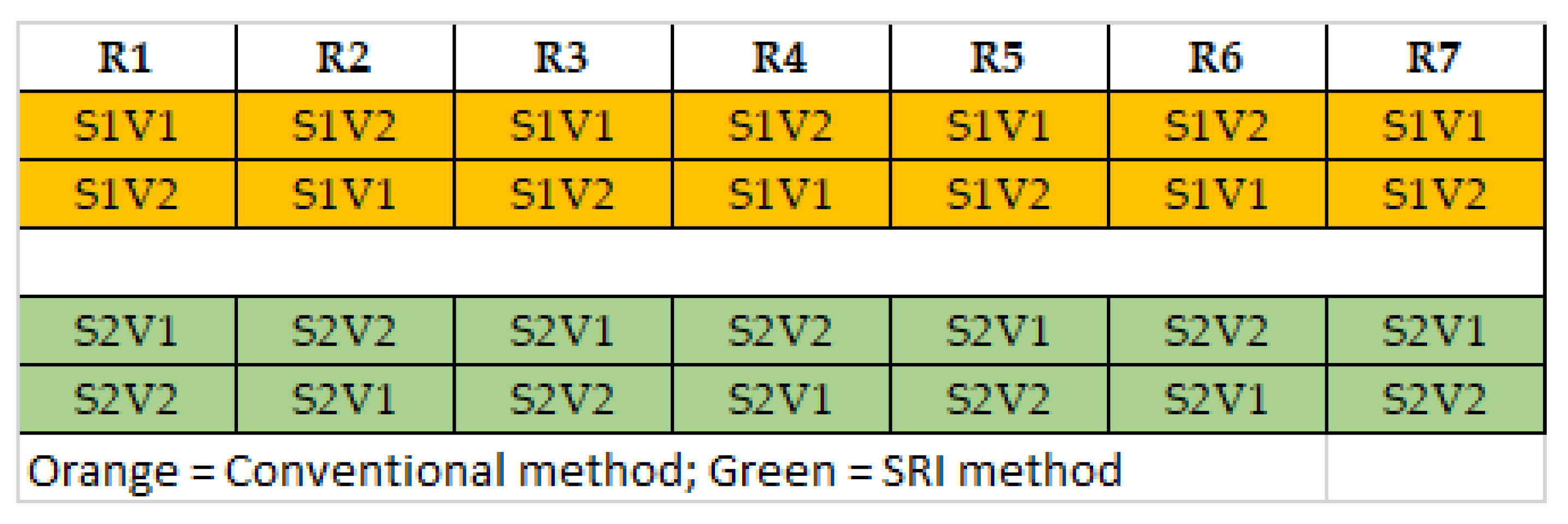
2.1. Experiment 1 at Multiple Locations—Insect Pest Incidence with SRI vs. Conventional Methods
2.2. Experiment 2 at Institute Farm—Assessment of Yield Loss, Biochemical Parameters, and Biodiversity
2.3. Statistical Analysis
2.4. Meta-Analysis
3. Results
3.1. Experiment 1 at Multiple Locations
3.2. Experiment 2 at Institute Farm
3.2.1. Stem Borer Incidence and Yield Loss in SRI and Conventional Methods
3.2.2. Effects on Biochemical Composition of Rice Plants
3.2.3. Impact of Cultivation Methods on Insect Biodiversity
3.3. Meta-Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The Future of Food and Agriculture–Trends and Challenges; UN Food and Agriculture Organization: Rome, Italy, 2017; pp. 11–16. [Google Scholar]
- Food and Agriculture Organization. Rice Market Monitor, April 2018. XXI. Available online: http://www.fao.org/economic/RMM (accessed on 20 June 2022).
- Karthikeyan, K.; Christy, M.M.; Sreeshma, M.K. Effect of system of rice cultivation on insect pest incidence in Palakkad district of Kerala. J. Biopest. 2014, 7, 122–127. [Google Scholar]
- Thakur, A.K.; Uphoff, N. How the System of Rice Intensification can contribute to climate-smart agriculture? Agron. J. 2017, 109, 1163–1182. [Google Scholar] [CrossRef]
- Cacacho, E.L.; Agcaoili, S.O.; Cacatian, S.B.; Ouano, N.B. Assessment of the organic-based system of rice intensification (SRI) under coastal rice field ecosystem. Indian J. Sci. Technol. 2020, 13, 1867–1872. [Google Scholar] [CrossRef]
- Vibhu, N.; Ravichandran, V.K. System of Rice Intensification: Optimizing production with fewer inputs. The Hindu, 4 April 2008. [Google Scholar]
- Babendreier, D.; Maolin, H.; Rui, T.; Zhang, F.; Vongsabouth, T.; Win, K.K.; Min, K.; Peng, H.; Song, K.; Annamalai, S.; et al. Biological control of Lepidopteran pests in rice: A multi-nation case study from Asia. J. Integr. Pest Manag. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Dhaliwal, G.S.; Jindal, V.; Dhawan, A.K. Insect pest problems and crop losses: Changing trends. Indian J. Ecol. 2010, 37, 1–7. [Google Scholar]
- Heinrichs, E.A.; Muniappan, R. IPM for tropical crops: Rice. CAB Rev. 2017, 12, 1–31. [Google Scholar] [CrossRef]
- Padmavathi, C.; Chitra, S.; Gururaj, K. Advances in rice IPM: Indian scenario. J. Res. PJTSAU 2017, 45, 1–13. [Google Scholar]
- Shetty, P.K. Socio-ecological implications of pesticide use in India. Econ. Polit. Wkly. 2004, 39, 5261–5267. [Google Scholar]
- Ngo, T.D. SRI Application in Vietnam. Paper Presented at Pre-Congress SRI Day on 8 November 2010. Available online: https://www.slideshare.net/SRI.CORNELL/sri-application-in-vietnam (accessed on 15 December 2021).
- Karthikeyan, K.; Smitha, R.; Balachandran, P.V.; Shanmugasundaram, B.; Purushothaman, S.M. Influence of SRI cultivation on the incidence of major pests of rice. In Proceedings of the Extended Summaries of the 2nd National Symposium on System of Rice Intensification in India: Progress and Prospects, Tripura, India, 3–5 October 2007; pp. 96–97. [Google Scholar]
- Ratnasudhakar, T.; Narasimha Reddy, P. Influence of System of Rice Intensification (SRI) on the incidence of insect pests. In Proceedings of the Extended summaries of the Second National Symposium on System of Rice Intensification in India: Progress and Prospects, Agartala, India, 3–5 October 2007; pp. 87–88. [Google Scholar]
- Padmavathi, C.; Mahendra Kumar, R.; Subba Rao, L.V.; Surekha, K.; Srinivas Prasad, M.; Ravindra Babu, V.; Pasalu, I.C. Insect-pest dynamics and arthropod diversity in SRI and conventional methods of rice cultivation. Oryza 2009, 4, 227–230. [Google Scholar]
- Glass, G.V. Integrating findings: The meta-analysis of research. Rev. Res. Educ. 1978, 5, 351–379. [Google Scholar]
- Skawsang, S.; Nagai, M.K.; Tripathi, N.; Soni, P. Predicting rice pest population occurrence with satellite-derived crop phenology: Ground meteorological observation and machine learning: A case study for the Central Plain of Thailand. Appl. Sci. 2019, 9, 4846. [Google Scholar] [CrossRef]
- Van Soest, P.J. Use of detergents in the analysis of fibrous feeds. II. A rapid method for the determination of fiber and lignin. J. AOAC 1963, 46, 829–835. [Google Scholar] [CrossRef]
- Viechtbauer, W. METAFOR: Meta-Analysis Package for R, Version 2.4.0. 2020. Available online: https://CRAN.R-project.org/package=metafor (accessed on 17 December 2020).
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H. Introduction to Meta-Analysis; John Wiley: Chichester, UK, 2009; 421p. [Google Scholar]
- Viechtbauer, W. Hypothesis tests for population heterogeneity in meta-analysis. Br. J. Math. Stat. Psychol. 2007, 60, 29–60. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. Br. Med. J. 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Dettori, J.R.; Norvell, D.C.; Chapman, J.R. Seeing the forest by looking at the trees: How to interpret a meta-analysis forest plot. Glob. Spine J. 2021, 11, 614–616. [Google Scholar] [CrossRef]
- Pathak, M.; Shakyawar, R.C.; Dinseh, S. Biodiversity of insect pests, natural enemies and diseases in SRI and traditional system of rice cultivation in northeastern region of India. Oryza 2013, 50, 370–374. [Google Scholar]
- Jayakumar, S.; Sankari, A. Spider population and their predatory efficiency in different rice establishment techniques in Aduthurai, Tamil Nadu. J. Biopest. 2010, 3, 020–027. [Google Scholar]
- Visalakshmi, V.; Rama Mohana Rao, P.; Hari Satyanarayana, N. Impact of paddy cultivation systems on insect pest incidence. J. Crop Weed. 2014, 10, 139–142. [Google Scholar]
- Yashpal Singh, N.; Sahu, C.M.; Ghirtlahre, S.K.; Painkra, K.L.; Chandrakar, G. Studies on the seasonal incidence of leaf folder, Cnaphalocrocis medinalis Guenee, in midland SRI and normal transplanted rice ecosystem. Int. J. Trop. Agric. 2015, 33, 547–551. [Google Scholar]
- Kakde, A.M.; Patel, K.G. Succession of rice pest complex and natural enemies in conventional and SRI methods of planting. Int. J. Curr. Microbiol. Appl. Sci. 2018, 6, 2181–2188. [Google Scholar]
- Kega, V.M.; Nderitu, J.H.; Kasina, M.; Olubayo, F. Influence of cropping and irrigation systems on population fluctuation of the African white stem borer (Maliapha separatella Rag) and damage on rice. J. Entomol. 2015, 12, 95–102. [Google Scholar] [CrossRef]
- Karthikeyan, K.; Sosamma, J.; Purushothaman, S.M. Incidence of insect pests and natural enemies under SRI method of rice cultivation. Oryza 2010, 47, 164–167. [Google Scholar]
- Kakde, A.M.; Patel, K.G. Seasonal incidence of rice leaf folder in relation to SRI and conventional methods of planting and its correlation with weather parameters. Plant Arch. 2015, 15, 121–126. [Google Scholar]
- Dung, N.T. SRI Application in Rice Production in Northern Ecological Areas of Vietnam. Report of National IPM Program to Ministry of Agriculture and Rural Development, Hanoi. Available online: http://sri.cals.cornell.edu/countries/vietnam/vndungipmrpt06.pdf (accessed on 20 December 2020).
- David, P.M.; Ezhilrani, M.K.; Thiyagarajan, T.M. Relative abundance of insects in SRI and conventional rice. Presented at the National Symposium on Biodiversity and Insect Pest Management, Chennai, India, 3–4 February 2005; Entomology Research Institute: Chennai, India, 2005; pp. 96–99. [Google Scholar]
- Mahender Kumar, R.; Raghuveer Rao, P.; Somasekhar, N.; Surekha, K.; Padmavathi, C.; Srinivas Prasad, M.; Ravindra Babu, V.; Subba Rao, L.V.; Sreedevi, B.; Ravichandran, S.; et al. SRI: A method for sustainable intensification of rice production with enhanced water productivity. Agrotechnology 2013, S11, 009. [Google Scholar] [CrossRef]
- Raj, R.; Kumar, A.; Solanki, I.S.; Shiva, D.; Anchal, D.; Gupta, A.K.; Vikash, K.; Singh, C.B.; Jat, R.K.; Pandey, U.C. Influence of crop establishment methods on yield, economics, and water productivity of rice cultivars under upland and lowland production ecologies of Eastern Indo-Gangetic Plains. Paddy Water Environ. 2017, 15, 861–877. [Google Scholar] [CrossRef]
- Hossain, M.; Hossain, S.M.; Anwar, M.; Sarkar, M.R.; Mamun, A. Performance of BRRI Dhan in SRI and conventional methods and their technology mixes. Pak. J. Agron. 2003, 2, 195–200. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Mahender Kumar, R.; Humayun, P.; Srinivas, V.; Ratna Kumari, B.; Vijayabharathi, R.; Singh, A.; Surekha, K.; Padmavathi, C.; Somashekar, N.; et al. Assessment of different methods of rice (Oryza sativa L.) cultivation affecting growth parameters, soil chemical, biological, and microbiological properties, water saving, and grain yield in rice–rice system. Paddy Water Environ. 2014, 12, 79–87. [Google Scholar] [CrossRef]
- Calatayud, P.A.; Njuguna, E.; Juma, G. Silica in insect-plant interactions. Entomol. Ornithol. Herpetol. 2016, 5, e125. [Google Scholar] [CrossRef]
- Alhousari, F.; Greger, M. Silicon and mechanisms of plant resistance to insect pests. Plants 2018, 7, 33. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, J.; Zhang, P.; Han, L.; Reynolds, O.L.; Zeng, R.; Wu, J.; Shao, Y.; You, M.; Gurr, G.M. Silicon Supplementation Alters the Composition of Herbivore Induced Plant Volatiles and Enhances Attraction of Parasitoids to Infested Rice Plants. Front. Plant Sci. 2017, 8, 1265. [Google Scholar] [CrossRef]
- Han, Y.; Lei, W.; Wen, L.; Hou, M. Silicon-mediated resistance in a susceptible rice variety to the rice leaf folder, Cnapholocrosis medinalis Guenée (Lepidoptera: Pyralidae). PLoS ONE 2015, 10, e0120557. [Google Scholar] [CrossRef]
- Yang, L.; Han, Y.; Li, P.; Wen, L.; Hou, M. Silicon amendment to rice plants impairs sucking behaviors and population growth in the phloem feeder Nilaparvata lugens (Hemiptera: Delphacidae). Sci. Rep. 2017, 7, 1101. [Google Scholar] [CrossRef] [PubMed]
- Gowele, G.E.; Mahoo, H.F.; Kahimba, F.C. Comparison of silicon status in rice grown under the system of rice intensification and flooding regime in Mkindo irrigation scheme, Morogoro, Tanzania. Tanzan. J. Agric. Sci. 2020, 19, 216–226. [Google Scholar]
- Devi, P.R.; Singh, K.I. Studies of insect pest diversity: Special reference to system of rice intensification and conventional method in Imphal valley, Manipur, India. Int. Res. J. Mark. Econ. 2015, 2, 19–30. [Google Scholar]












| Cultivation Methods | BPH Number per 10 Hills | WBPH Number per 10 Hills | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Hybrid Plots | Variety Plots | Hybrid Plots | Variety Plots | |||||||
| JDP | SGP | WGL | JDP | SGP | WGL | JDP | WGL | JDP | WGL | |
| Conventional | 32 a | 350 a | 68 a | 18 a | 965 a | 61 a | 19 b | 40 a | 13 a | 0 a |
| SRI | 10 b | 52 b | 0 b | 6 b | 45 b | 0 b | 16 a | 40 a | 13 a | 0 a |
| Cultivation Methods | Yield of Hybrid Varieties | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| CBT | CHN | PTB | RGL | RNC | RNR | RPR | WGL | Ave. | |
| Conventional | 4171 b | 3726 a | 3750 b | 5876 a | 5443 a | 5284 b | 2145 b | 2163 a | 4070 |
| SRI | 7193 a | 3521 a | 4901 a | 6109 a | 4564 b | 5783 a | 2874 a | 2352 a | 4662 |
| Cultivation Methods | Yield of Inbred Varieties | ||||||||
| CBT | CHN | PTB | RGL | RNC | RNR | RPR | WGL | Ave. | |
| Conventional | 3736 b | 3085 a | 3746 b | 5641 a | 4401 a | 4508 a | 1290 a | 2026 a | 3554 |
| SRI | 6136 a | 3081 a | 5707 a | 5755 a | 3643 b | 4521 a | 1555 a | 2217 a | 4076 |
| Cultivation Methods | Yield Loss (%) | |||||
|---|---|---|---|---|---|---|
| Hybrid Plots | Inbred Plots | |||||
| 1 WEH | 2 WEH | 3 WEH | 1 WEH | 2 WEH | 3 WEH | |
| Conventional | 12.9 | 23.8 | 50.8 | 17.2 | 20.0 | 34.0 |
| SRI | 10.8 | 23.5 | 34.4 | 7.2 | 15.8 | 28.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chintalapati, P.; Rathod, S.; Repalle, N.; Varma, N.R.G.; Karthikeyan, K.; Sharma, S.; Kumar, R.M.; Katti, G. Insect Pest Incidence with the System of Rice Intensification: Results of a Multi-Location Study and a Meta-Analysis. Agronomy 2023, 13, 1100. https://doi.org/10.3390/agronomy13041100
Chintalapati P, Rathod S, Repalle N, Varma NRG, Karthikeyan K, Sharma S, Kumar RM, Katti G. Insect Pest Incidence with the System of Rice Intensification: Results of a Multi-Location Study and a Meta-Analysis. Agronomy. 2023; 13(4):1100. https://doi.org/10.3390/agronomy13041100
Chicago/Turabian StyleChintalapati, Padmavathi, Santosha Rathod, Naganna Repalle, Nadimpalli Rama Gopala Varma, Kolandhaivelu Karthikeyan, Sanjay Sharma, Rapolu Mahender Kumar, and Gururaj Katti. 2023. "Insect Pest Incidence with the System of Rice Intensification: Results of a Multi-Location Study and a Meta-Analysis" Agronomy 13, no. 4: 1100. https://doi.org/10.3390/agronomy13041100
APA StyleChintalapati, P., Rathod, S., Repalle, N., Varma, N. R. G., Karthikeyan, K., Sharma, S., Kumar, R. M., & Katti, G. (2023). Insect Pest Incidence with the System of Rice Intensification: Results of a Multi-Location Study and a Meta-Analysis. Agronomy, 13(4), 1100. https://doi.org/10.3390/agronomy13041100





