Abstract
Soil organic carbon (SOC) and mineral nitrogen (Nmin), especially nitrates (NO3−) in agroecosystems have attracted much attention over the past few decades due to their crucial roles in soil fertility, crop productivity, environmental quality, and/or climate change mitigation and adaptation. The aim of the study was to evaluate the contents of organic carbon, ammonium, and nitrate in soils under differentiated pH, texture, and fertilization rates. A large-scale environmental study was conducted in Polish arable lands. The spatial distribution of the sampling points reflected agricultural production conditions, variability of soil properties, and representativeness of textures that are characteristic of Poland. Our results indicated that SOC content was significantly affected by the soil pH and texture as well as mineral and organic fertilization. The same factors, except organic amendments, significantly supported mineral nitrogen concentration in the present study. The most important factors controlling SOC in the study were ranked as follows: soil pH > pre-crop N fertilization > crop N fertilization > N applied with manure > soil texture. In the case of N-NH4 and N-NO3, mineral fertilization was the most critical variable. The carbon and nitrogen governance in agroecosystems should consider the ranks of factors controlling their contents.
1. Introduction
Soil organic carbon (SOC) and mineral nitrogen (Nmin), especially nitrates (NO3−) in agroecosystems, have attracted much attention over the past few decades due to their crucial roles in soil fertility, crop productivity, environmental quality, and/or climate change mitigation and adaptation [1,2,3,4,5,6].
The soil carbon (C) pool, estimated at 2293 Pg, including 1425 Pg of SOC to 1 m depth, is the third principal C stock, which accumulates 5–15% of the annual global C emissions [2,7,8]. In merely the topsoil of EU farmland, 51 billion tons of CO2eq are stored, which is equivalent to over ten times the European Union’s annual emissions. SOC content is crucial to diverse soil functions (physical, chemical, and biological) and ecosystem services [1,2,3,9,10,11], e.g., (i) soil structure and aggregation, (ii) water use efficiency and retention, (iii) plant nutrient availability, (iv) the diversity and biological activity of soil organisms, and (v) gaseous emissions/sequestration. The critical level of SOC in the root zone, essential to maintaining these functions, is 1.5–2.0% [12], whereas ca. 45% of the mineral soils in Europe have low or very low SOC content, i.e., below 2% [13].
Nitrogen (N) is both an essential element strongly related to food security and resource sustainability that often limits crop yields and a major pollutant in modern agroecosystems [5,14,15,16]. It is the most unstable plant nutrient, especially its mineral forms, subjected to many transformations sometimes in a short time, such as uptake by plants, immobilization by microorganisms, nitrification, denitrification, volatilization, leaching, and adsorption [17]. Approximately 25% of the N inputs are emitted into the air, and nearly 20% are leached as nitrate, hampering productivity and profitability of crop production as well as environmental quality [15]. Anthropogenic activities have doubled the global rates of reactive N applications in the last century and substantially affected C and N cycles in agricultural ecosystems [7]. Unfortunately, there are lack or contradictory reports regarding the effect of anthropogenic (fertilization with manure, crop, and pre-crop fertilization with mineral nitrogen) and environmental (soil pH and texture) factors on C and N pools [6,18]. Mineral fertilizers, affecting the increase in crop yields and the amount of post-harvest residues, have a positive effect on the SOC content. Some authors [19,20], however, believe that one-sided mineral fertilization, especially in light soils, causes intensification of mineralization. Exogenous organic matter is another possible significant source of SOC and Nmin in agroecosystems. On the other hand, the direction of such impact is determined by its chemical composition, mainly the content of N, polyphenols, and lignins [19]. Adjusting the pH by liming allows the retention of C and N in the soil. It is also a factor that potentially generates gaseous losses in the form of carbon dioxide, nitrogen oxides, dinitrogen oxide, ammonia, and/or dissolved forms of C and N [21,22,23]. Hence, there is a need to investigate these issues in various climatic and soil conditions and to update research allowing the implementation of effective mitigation and adaptation measures in the context of the currently introduced carbon farming initiative as a part of the European Green Deal and the evaluation of action programs related to the Nitrates Directive (91/676/EEC).
The aim of the study was to (i) evaluate the contents of organic carbon, ammonium, and nitrate in soils under differentiated pH, texture, and fertilization rates and (ii) identify the variables that were most important in contributing to the accumulation of carbon and mineral nitrogen in Polish soils using a novel analytical approach in large-scale research.
2. Materials and Methods
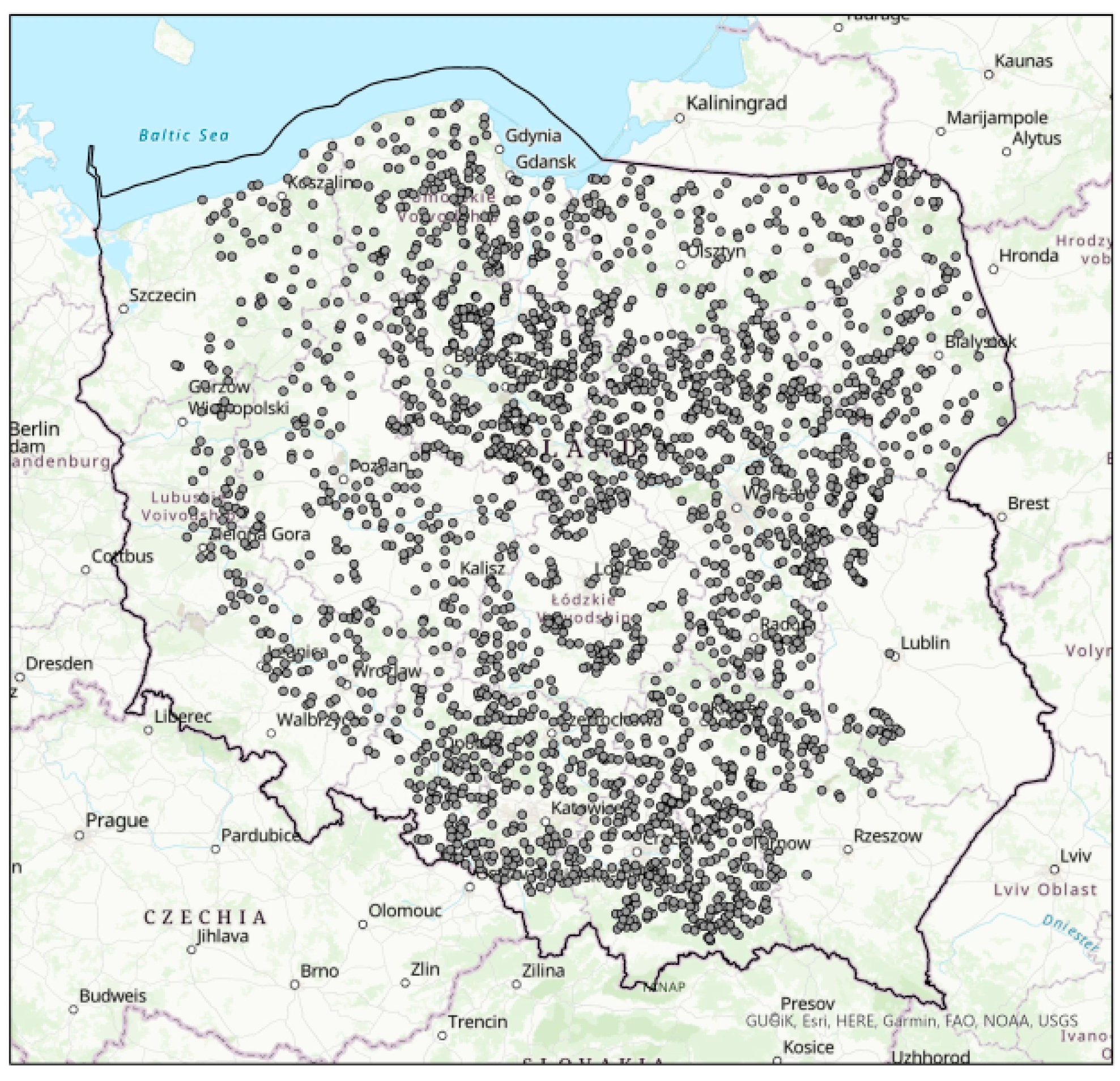
Soil samples were taken in the autumn after harvesting crops at 3172 permanent monitoring points evenly covering Polish arable lands (Figure 1), from 0–30 cm depth and an area of 100 m2. The spatial distribution of the sampling points reflected agricultural production conditions, variability of soil properties, and representativeness of textures that are characteristic of Poland. The consumption of mineral nitrogen fertilizers (urea and ammonium nitrate), as well as farmyard manure (FYM) in the studied agroecosystems established following the procedure of soil monitoring in Poland (a direct interview with farmers) varied from <50 kg N ha−1 to >150 kg N ha−1.
Figure 1.
Map of the study and sampling area.
Poland belongs to the same climate zone as the Czech Republic, Slovakia, partly Germany and Austria, Hungary, Romania, Ukraine, and Belarus [24]. The annual rainfall during the investigation was 573.3 mm. Mean monthly temperatures in January and June were: −1.5 and 21.2 °C, respectively.
Each sample taken from the fields for analysis consisted of 20 primary soil samples collected from the surface layer from an area of 100 m2. Soil samples were air-dried, homogenized, sieved (2 mm sieve-mesh), and kept in the dark. The soil samples for Nmin analysis were placed in polyethylene bags and stored in the refrigerator.
In air-dried and sieved soil samples, the following parameters were determined: pH in 1 mol KCl dm−3 by the potentiometric method in a soil:solution ratio of 1:2.5 (w/v), total organic carbon by sulfochromic oxidation with titration of excess K2Cr2O7 with FeSO4(NH4)2SO4·6H2O, and particle size distribution using the laser diffraction method (LDM) based on the light intensity distribution pattern of the scattered light emitted from that particle group. Mineral nitrogen was determined colorimetrically (N-NO3 with diphenylamine sulfonic acid and N-NH4 with Nessler’s reagent) using a San Plus Segmented Flow Analyzer after extraction with 1% of K2SO4 [25,26].
In order to compare the average value of parameters in individual classes, one-way analysis of variance (ANOVA) and Tukey’s honest significant difference test with significance level α = 0.05 were performed. The importance of variables was assessed by CART, which is a multivariate regression tool appropriate for identifying the most important factors controlling SOC and Nmin contents in soils. Statistical analyses were performed using Statistica 13.3.
3. Results and Discussion
Carbon and mineral nitrogen accumulation in soils depended on soil pH, texture as well as mineral and organic fertilization (Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7 and Figure 8).
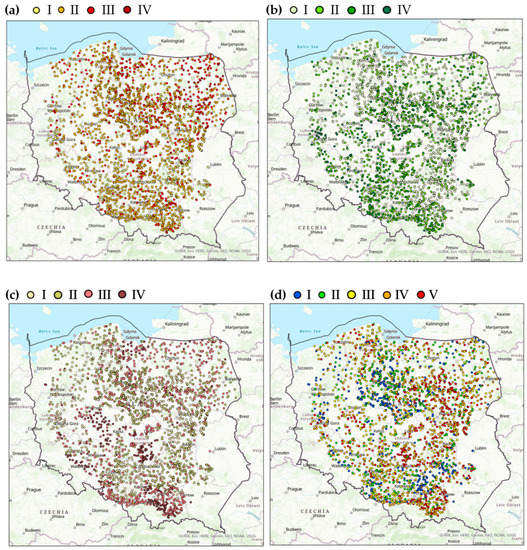
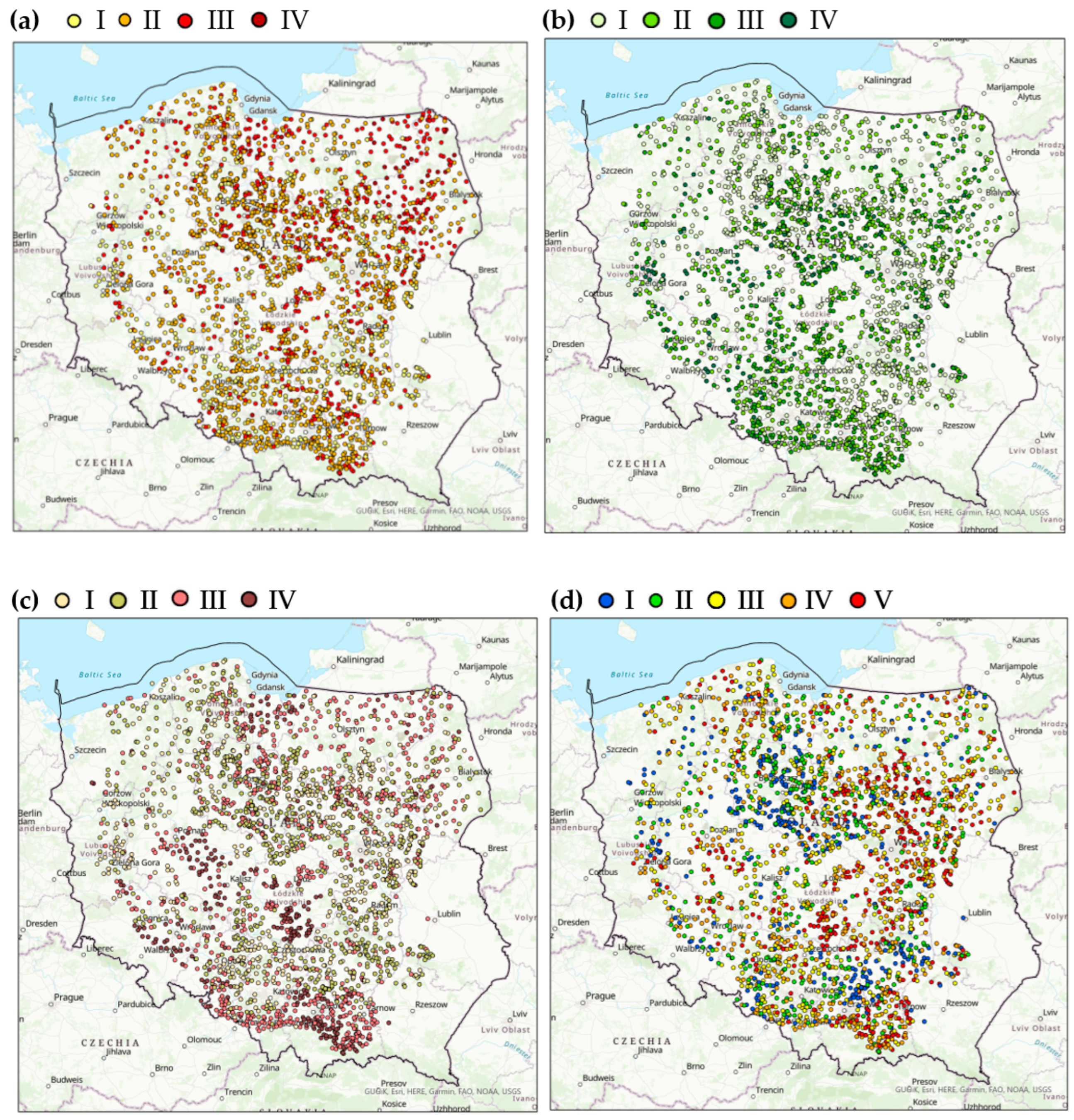
Figure 2.
The soil characteristic distribution among the sites (a) organic carbon: I—<1% C (very low), II—1–2% C (low), III—3–6% C (medium), IV—>6% C (high), (b) mineral nitrogen: I—<10 mg kg−1, II—10.01–20.0 mg kg−1 III—20.01–40.0 mg kg−1, I—>40.0 mg kg−1, (c) fractions smaller than 0.02 mm: I—<10%, II—10–20%, III—20–35%, IV—>35%, (d) soil pH: I—alkaline soils, pH > 7.2, II—neutral soils, pH = 6.6–7.2, III—slightly acidic soils, pH = 5.6–6.5, IV—acidic soils, pH = 4.6–5.5, V–highly acidic soils, pH < 4.5.
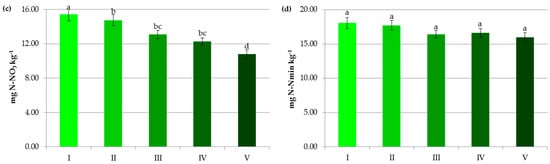
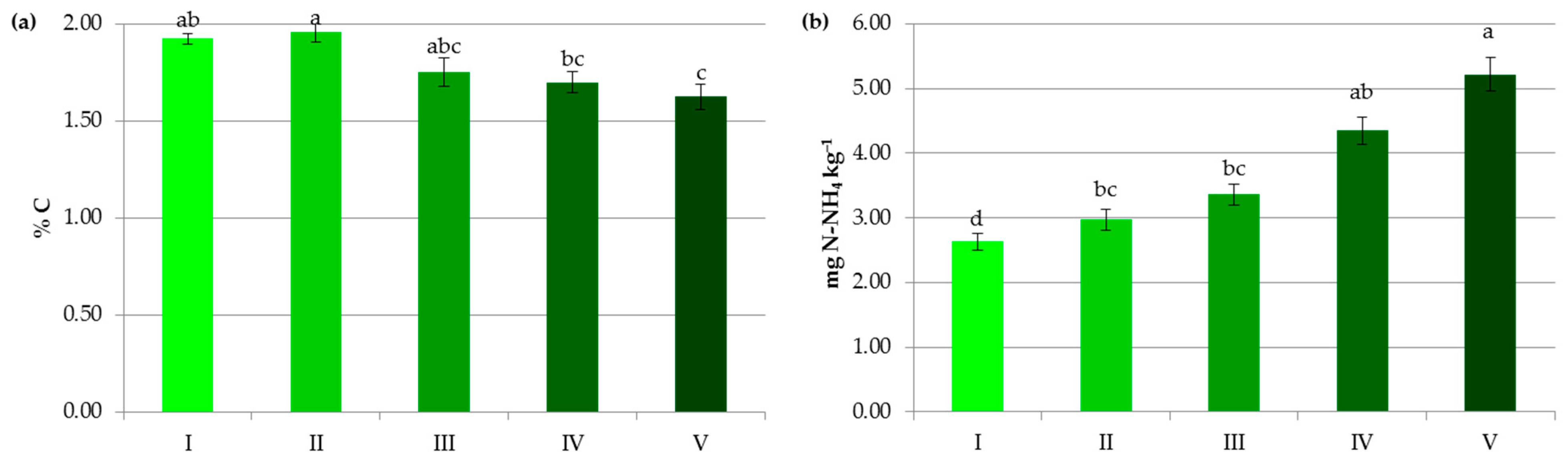
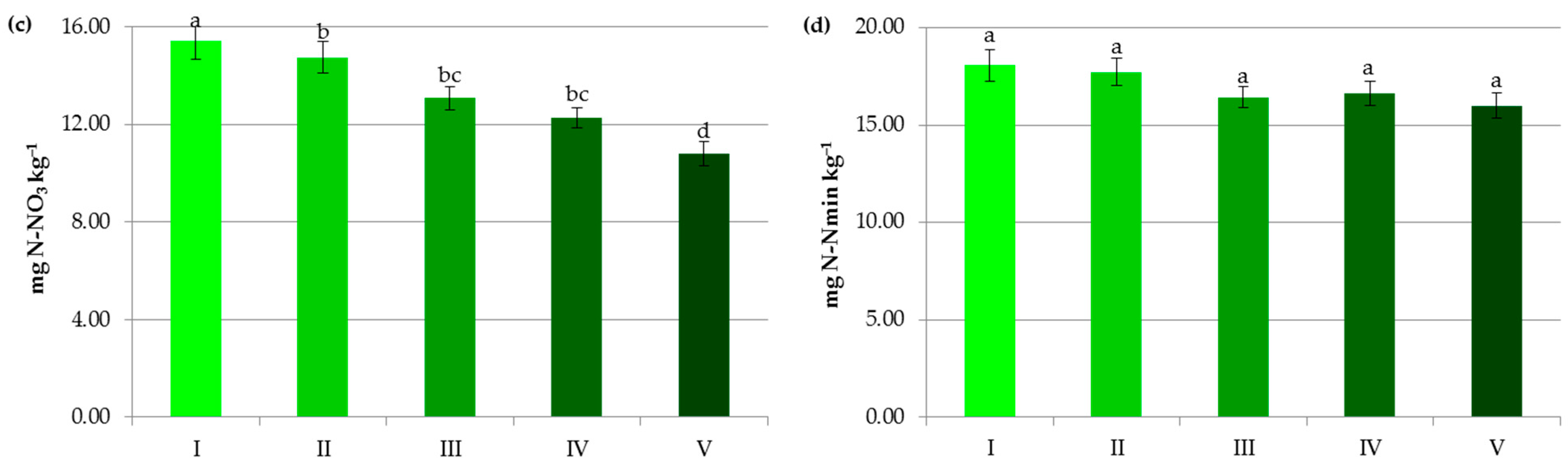
Figure 3.
Impact of soil pH on contents of organic carbon (a), ammonium nitrogen (b), nitrate nitrogen (c) and mineral nitrogen (d). I—alkaline soils, II—neutral soils, III—slightly acidic soils, IV—acidic soils, V—highly acidic soils. The same letter means not significantly different.
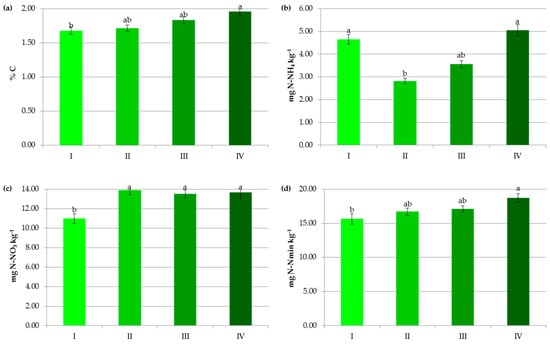
Figure 4.
Impact of soil texture on contents of organic carbon (a), ammonium nitrogen (b), nitrate nitrogen (c) and mineral nitrogen (d). Fractions smaller than 0.02 mm: I—<10%, II—10–20%, III—20–35%, IV—>35%. The same letter means not significantly different.
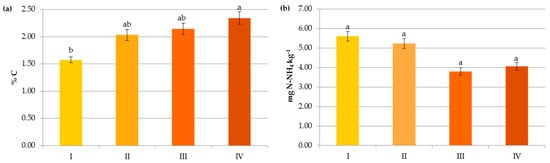
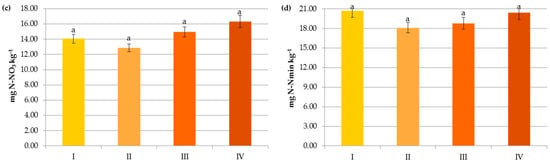
Figure 5.
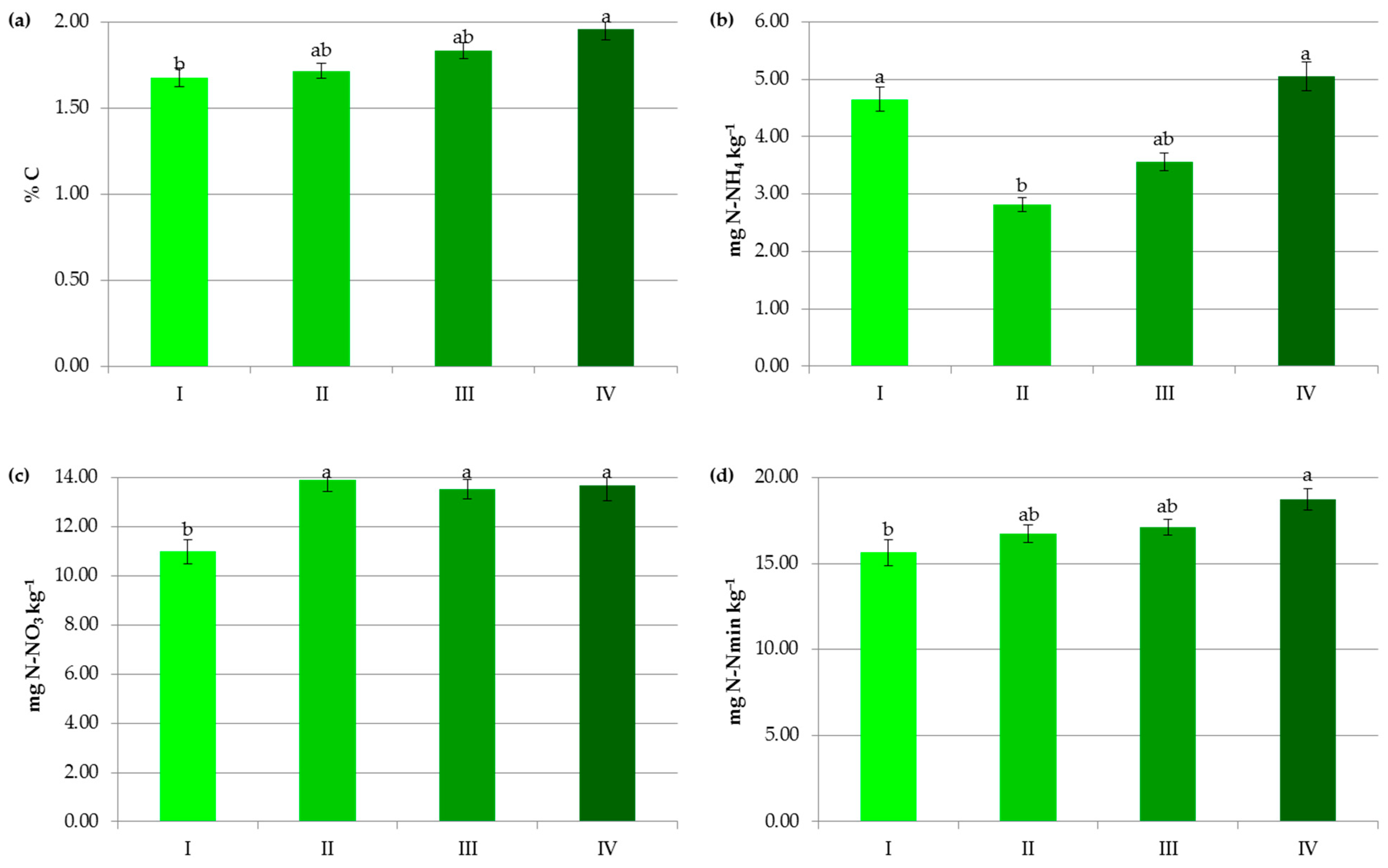
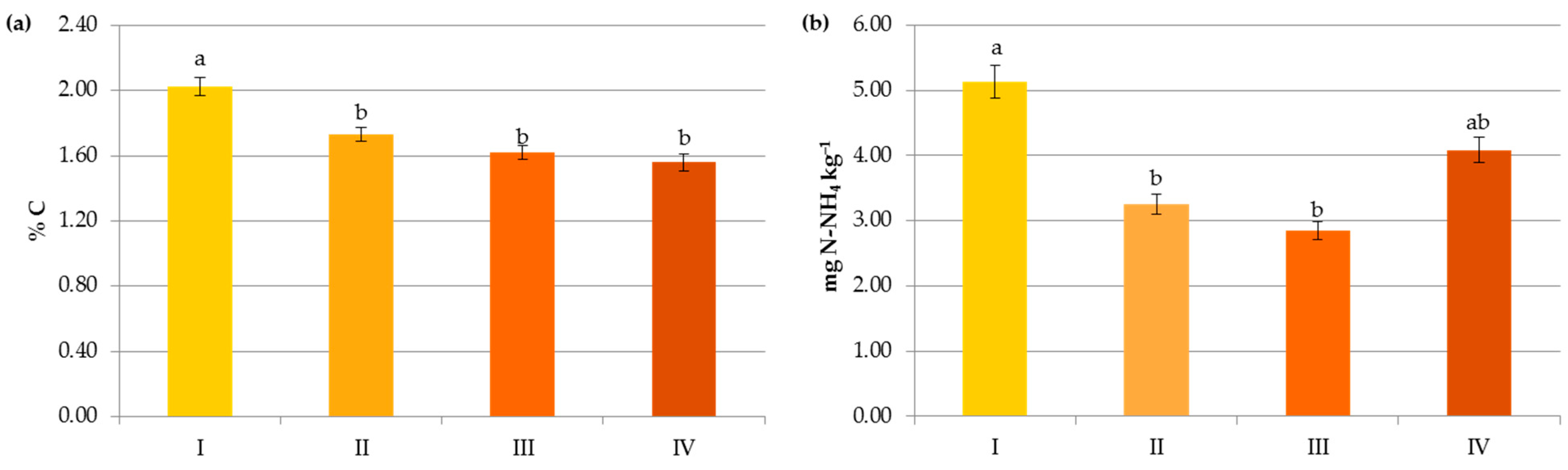
Impact of N manure application on contents of organic carbon (a), ammonium nitrogen (b), nitrate nitrogen (c) and mineral nitrogen (d): I—<50 kg N ha−1, II—50–100 kg N ha−1, III—100–150 kg N ha−1, IV—>150 kg N ha−1. The same letter means not significantly different.
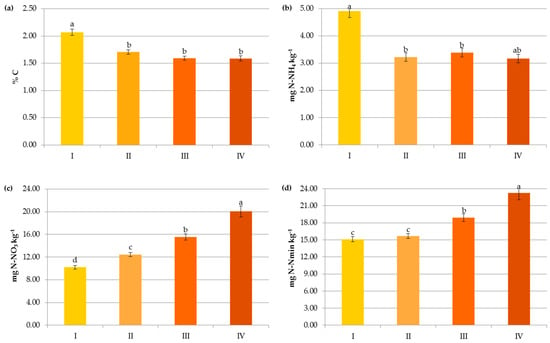
Figure 6.
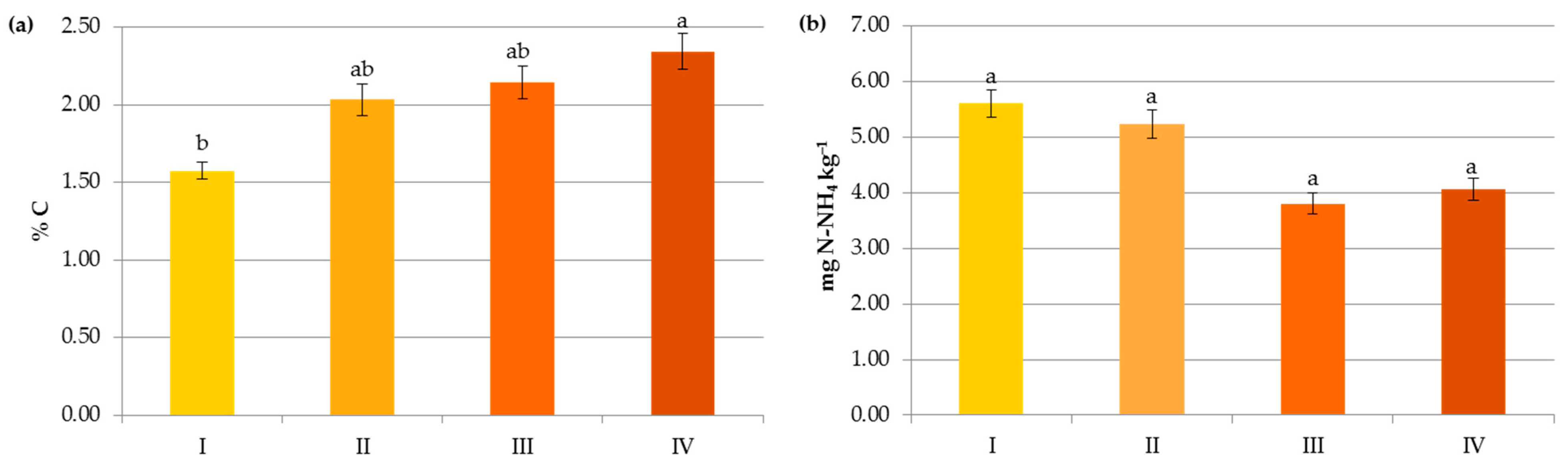
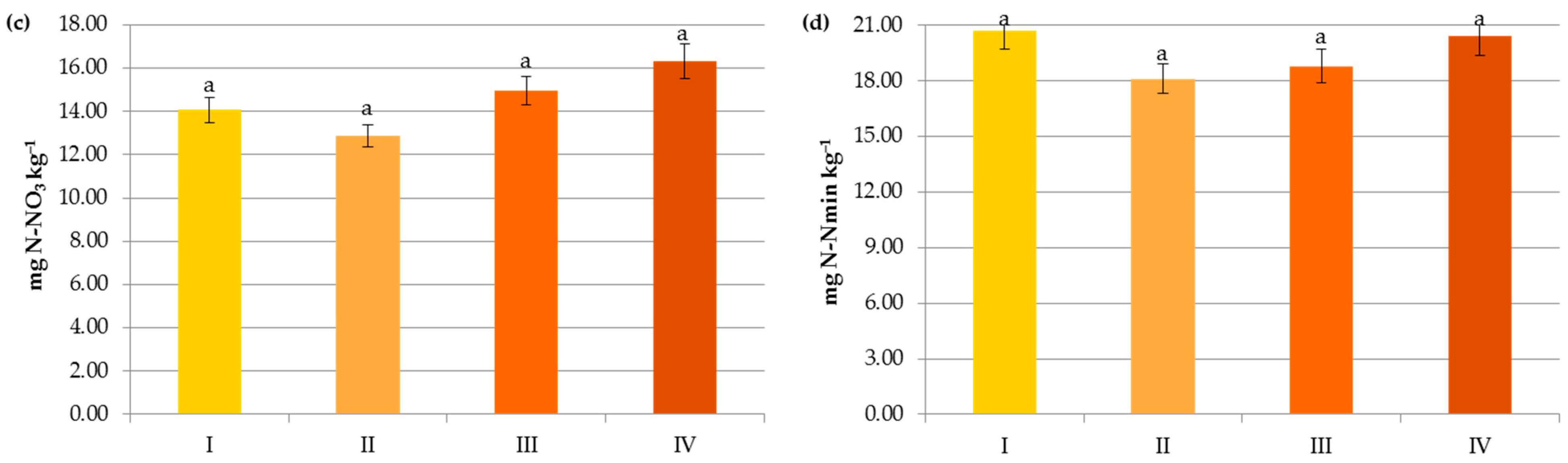
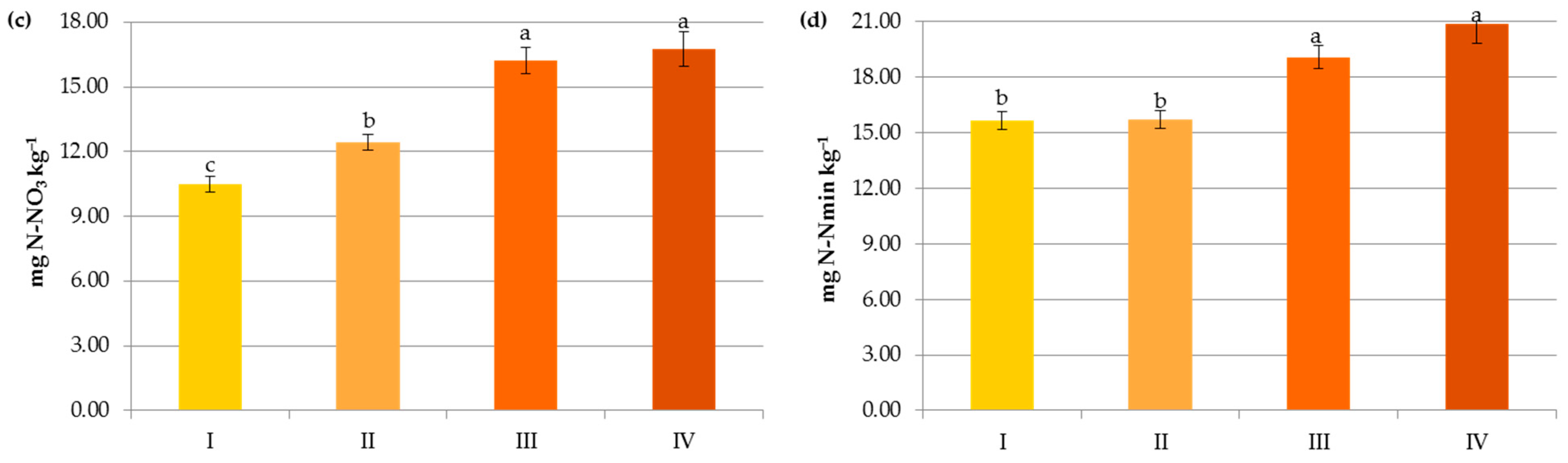
Impact of crop N fertilization on contents of organic carbon (a), ammonium nitrogen (b), nitrate nitrogen (c) and mineral nitrogen (d): I—<50 kg N ha−1, II—50–100 kg N ha−1, III—100–150 kg N ha−1, IV—>150 kg N ha−1. The same letter means not significantly different.
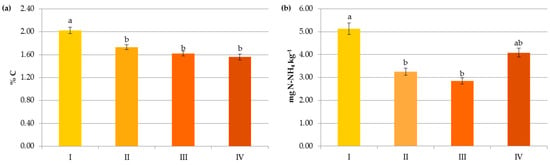
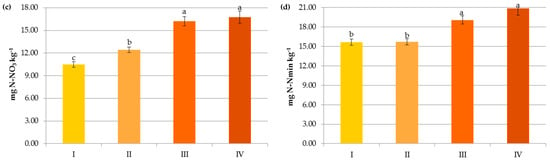
Figure 7.
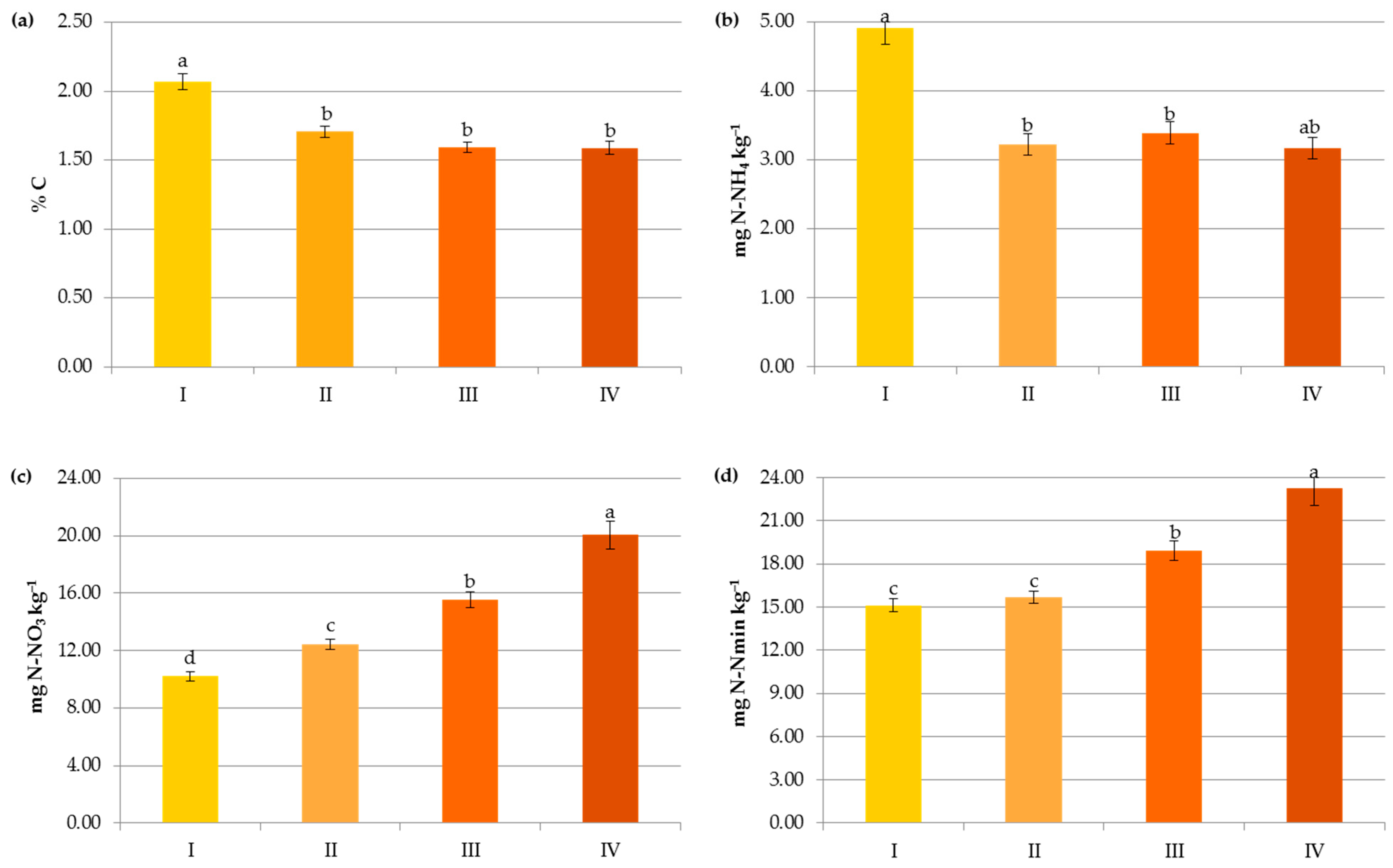
Impact of pre-crop N fertilization on contents of organic carbon (a), ammonium nitrogen (b), nitrate nitrogen (c) and mineral nitrogen (d): I—<50 kg N ha−1, II—50–100 kg N ha−1, III—100–150 kg N ha−1, IV—>150 kg N ha−1. The same letter means not significantly different.
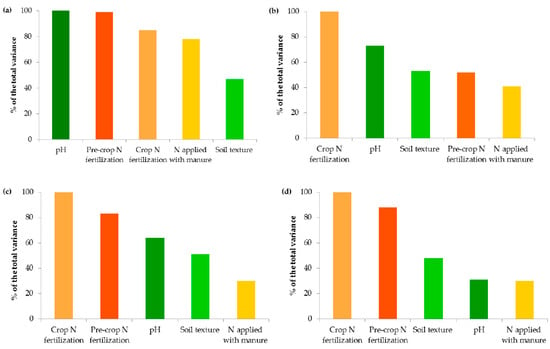
Figure 8.
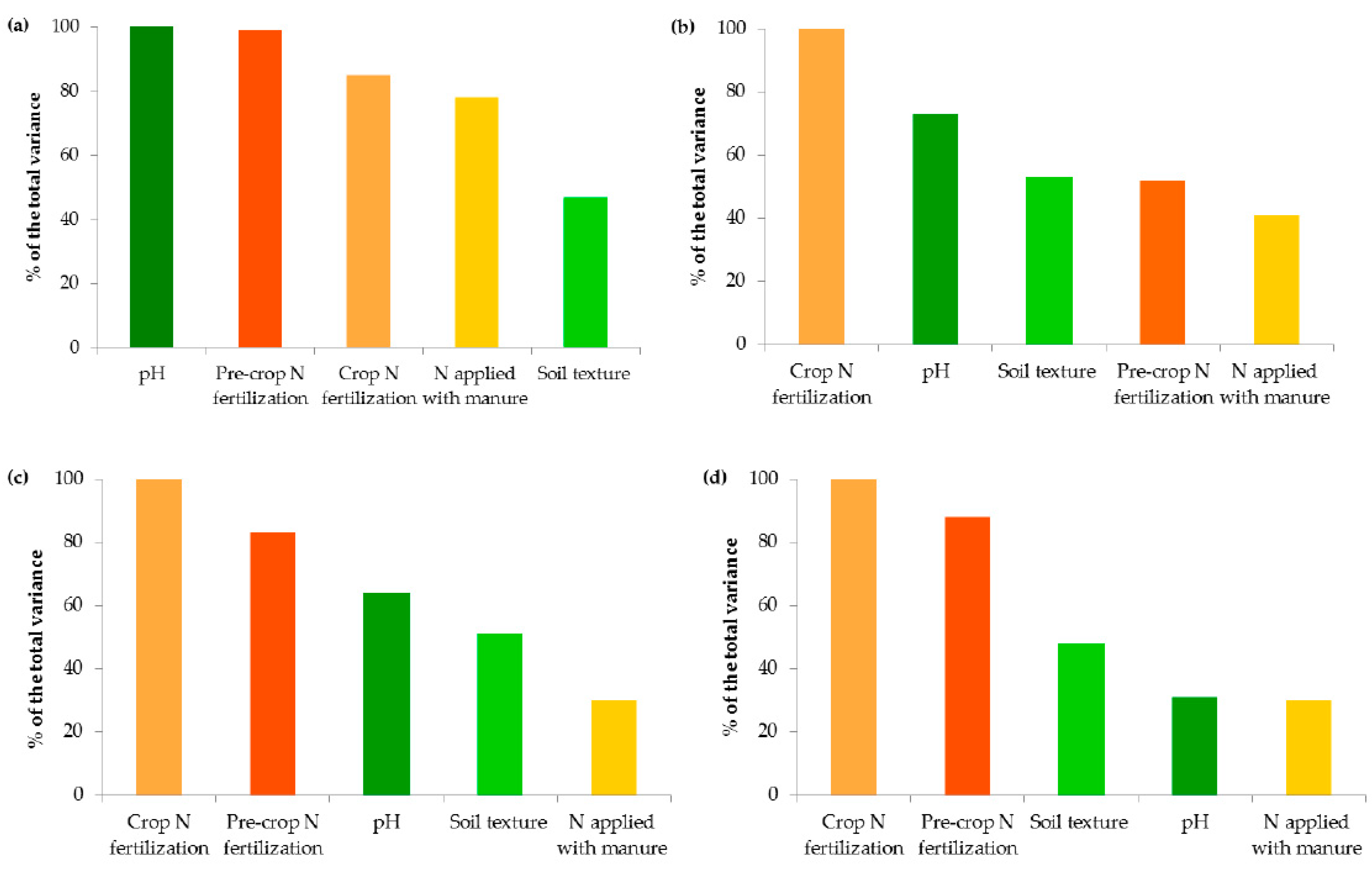
Variables in terms of their importance on the contents of organic carbon (a), ammonium nitrogen (b), nitrate nitrogen (c) and mineral nitrogen (d).
3.1. Soil Organic Carbon Contents
The average content of organic carbon in the studied soils was 1.78%. Soils with pH of 6.5–7.2 and above 7.2 accumulated the highest amounts of organic carbon, respectively 1.96% and 1.92%. This was in contrast to some research [27], which has demonstrated that alkaline soils are more prone to SOC losses than acidic ones due to the reduction in SOM complexation on mineral surfaces via ligand exchange. In the present study, a statistically significant decrease in the level of this parameter was observed in acidic and very acidic soils (Figure 3a). Solly et al. [28] also found that SOC content was lower in soils with pH < 5.5. Leifeld et al. [29] stated that the processes of carbon transformations in soils resemble strong pH dependencies, and their midpoints are reached at pH values between 4.3 and 5.3. A lot of authors [21,30,31] reported that low pH had a negative impact on SOM. In the present study, the identification of soil pH as the most important variable affecting SOC (Figure 8a) and a positive correlation between these parameters (r = 0.947) seem to confirm this thesis. Undoubtedly, these results were due to the influence of pH on the quantity and quality of the humus substances in the soil and the possibility of their stabilization. In the current research, the correlations of crop yields with pH and SOC were, respectively, r = 0.886 and r = 0.759. Apparently, higher pH optimized the plant growth environment, the use of macro- and microelements from soil reserves, and fertilizers, and stimulated biomass production [21,22]. The more significant the difference between the current pH value and the pH requirements of crops, the more liming contributes to increasing plant yields. It should be underlined that soil acidification limits biomass production and threatens the environment in Poland to a much greater extent than in many European and non-European countries. This is mainly due to the postglacial acidified parent material of Polish soils and the leaching of calcium and magnesium anions under the predominance of precipitation over evaporation, and a large share of light soils with high permeability in Poland [20,21]. According to some authors [22], greater inputs of post-harvest residues in limed soils compensate for initial carbon losses, caused by increased mineralization. Moreover, non-acid soils favor the transformation of humus substances into relatively stable organo-mineral complexes that are more difficult to decompose [21,32,33,34]. Lime, by releasing Ca2+ and Mg2+ ions and increasing the ionic strength of the soil solution, may enhance the flocculation of clay minerals and stimulate the activity of microorganisms secreting polysaccharides (binding agents). Thus, it contributes to the formation of stable soil aggregates, which improves the effectiveness of physical protection of organic matter. Calcium and magnesium ions and a higher pH also favor the transformation of humic substances to form calcium humates and other more complex organo-mineral compounds that are less prone to mineralization [21,35].
Soil texture was another factor significantly influencing the accumulation of soil organic carbon in the studied soils. However, its importance was lower in comparison to the soil pH (Figure 4a and Figure 8a). The amount of SOC in heavy (fine) soils rich with clay and silt fractions was significantly higher than in other categories (Figure 4a). According to some authors [9,27,32,36], clay-rich soils have longer turnover times and slower decomposition of SOM than sandy ones. There are relationships, not only between clay contents but also their types and SOC level. Singh et al. [37] stated that the rate of SOM decomposition was greater in the kaolinitic-illitic clay minerals, followed by smectitic and allophanic. The greater SOC stabilization in the latter resulted from their higher specific surface area and/or proportion of micropores as well as a lower microbial activity. Our results were supported by Matus [38], who, based on a study of 1.5 million arable French soil samples, stated that 85% of the soil organic carbon content was sequestered in the <20 µm mineral fraction. Interestingly, the differences in the carbon content in soils containing <10% and 20–35% of fractions with a diameter below 0.02 mm did not exceed 10% (Figure 4a). This could be associated with the limited capacity of carbon stabilization. Frasier et al. [39] have demonstrated that if carbon inputs cannot be stabilized, as in the predominant number of analyzed soils, SOC remains in particulate forms, which are susceptible to mineralization. It should be noted that organic matter is stabilized in soils in two major ways: (i) by interactions with fine soil particles <20 μm, i.e., mineral components, via weaker (e.g., Van der Waals forces) or stronger bonds (e.g., ligand exchange), (ii) and by SOC incorporation within soil aggregates and physical protection of SOM inside them [8,11,27,39,40,41]. The first one is considered dominant [38].
In the present study, SOC content was significantly affected by organic and mineral fertilization (Figure 5, Figure 6 and Figure 7). Its significant accumulation (2.34%) occurred when at least 150 kg N ha−1 in manure was applied. Undoubtedly, this result was due to the direct effect of the manure composition and the indirect impact of the increased crop growth and its residue in response to the additional nutrient supply [33,42]. Moreover, FYM, which was used in the analyzed agroecosystems, contains stabilized material that has already undergone the decomposition process [9], as well as a substantial amount of lignin and polyphenols and a high C:N ratio, which leads to the formation of stable complexes more resistant to oxidation [15,41].
In contrast to the manure inputs, a significant reduction, by 14.7–23.2%, in the content of SOC was observed with the increasing rate of crop and pre-crop N fertilization above 50 kg N ha−1 (Figure 6 and Figure 7). Apparently, the acceleration of SOM mineralization through a lower C-to-N ratio [32,42,43], soil acidification [34], as well as negative changes in the stability of soil aggregates and/or in the microbial community [44] caused by the current and/or recent mineral fertilization prevailed on the amount of C accumulated in agricultural soils as a result of greater root exudates and increased biomass production, later returned to the soil as crop residues [33,44,45]. Some authors reported that N fertilizers enlarge carbon stocks only when crop straws were returned to soils [7,42,46] and their decline was observed under intensive cultivation and imbalanced fertilization [15]. Others [47] have stated that only manure inputs resulted in a significant enhancement of SOC in comparison to NPK treatments, and the crop residues had nothing to do with the increase in soil organic carbon after several years.
Interestingly, the variables analyzed in the study in terms of their importance on the organic carbon content can be ranked as follows: soil pH > pre-crop N fertilization > crop N fertilization > N applied with manure > soil texture (Figure 8a). This indicates that the observed levels of organic carbon in the studied soils were mainly determined by the factors limiting SOC accumulation.
3.2. Mineral Nitrogen Contents
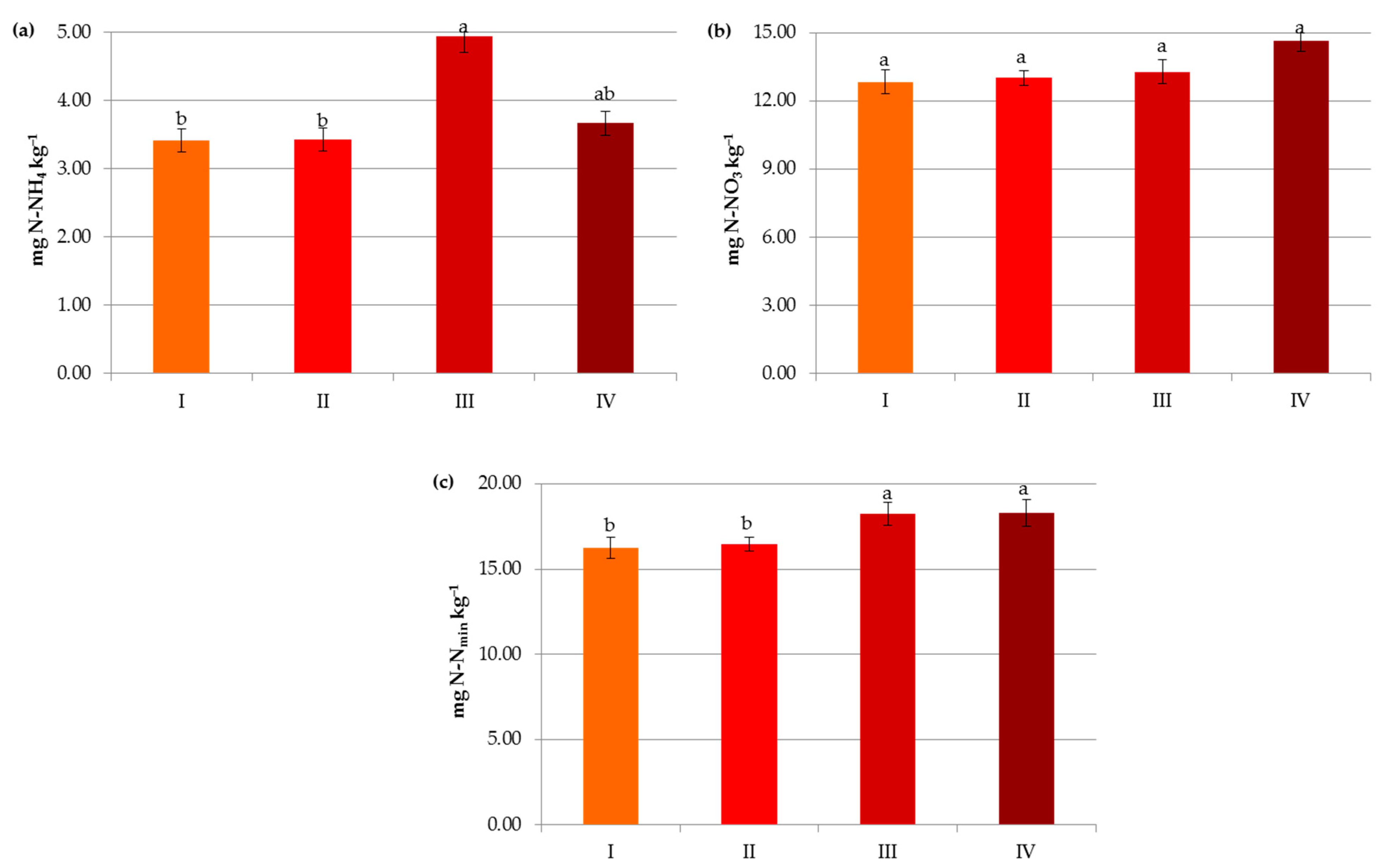
In the present study, organic matter contributed significantly to the availability of mineral nitrogen (N-NO3 and N-NH4) in the soils (Figure 9). The maximum content of Nmin (18.24–18.31 mg kg−1) was noted in soils containing above 2% of C (Figure 9c). These results are consistent with the findings of Ladha et al. [43] and Ren et al. [44] in which soil organic matter strongly influences soil N turnover and provides more than 50% of the nitrogen requirements of the crops. According to Soinne et al. [48], the amount of inorganic N available for plants is determined by net nitrogen mineralization, i.e., the difference between gross N mineralization and N immobilization in the microbial biomass. It should be noted that the contents of ammonium nitrogen were lower and more differentiated in carbon classes compared to nitrates (Figure 9a,b). These results may be explained by the fact that N-NH4 and N-NO3 have different biochemical characteristics (e.g., biological preference and ionic charge), and the former is adsorbed in exchangeable and non-exchangeable forms and nitrified as well as preferred by microorganisms due to the low energy cost [17,49,50,51,52].
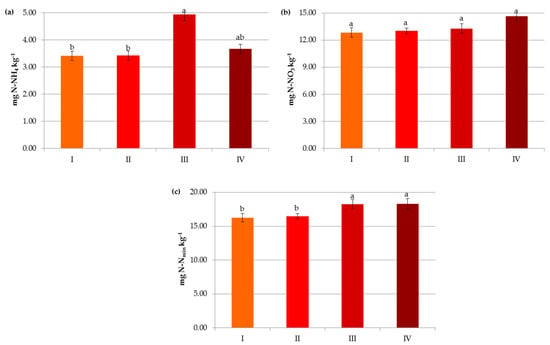
Figure 9.
Impact of carbon level on contents of ammonium nitrogen (a), nitrate nitrogen (b) and mineral nitrogen (c): I—<1% C (very low), II—1–2% C (low), III—3–6% C (medium), IV—>6% C (high). The same letter means not significantly different.
The presence of nitrogen in the plant available pool was influenced by the same factors that govern SOM, but their importance was slightly different, e.g., both in the case of N-NH4 and N-NO3, mineral fertilization was the most important factor affecting their contents (Figure 8b–d). Interestingly, the pH and texture had less effect on the latter form of nitrogen (Figure 3 and Figure 4).
In our research, the decline in soil pH was accompanied by a decrease in nitrate concentration (from 15.43 mg kg−1 at pH > 7.2 to 10.79 mg kg−1 at pH < 4.5) and an increase in NH4+ content (from 2.63 mg kg−1 at pH > 7.2 to 5.22 mg kg−1 at pH < 4.5) (Figure 3b,c). The lower soil pH undoubtedly reduced the provision of substrate for nitrification and the rate of this process itself [50,53]. In some experiments, at pH < 5.5 ammonium cations produced from the SOM mineralization were accumulated in the soil profile [53] and alkaline conditions were beneficial for nitrification and elevation of the nitrate concentration in the soil solution [5]. It should be noted that higher soil pH greatly affects NO3− uptake by plants but is also likely to drive nitrate leaching and/or N loss by volatilization [5,51,52,54], which should be considered in N management decisions. Kemmit [55] even suggested that a combination of natural or human-induced soil acidification may be a useful mechanism to reduce nitrate leaching from some agroecosystems.
Our results were consistent with other findings [56,57] in which soil texture significantly influenced N-NH4 and N-NO3 pools (Figure 4b–d). The content of ammonium increased both in coarse- and fine-textured soils, indicating different processes that control it (Figure 4b). Soils rich in fine clay particles have a higher adsorption capacity for N-NH4 [58] and usually contain more soil organic matter—a reservoir of ammonium cations. However, sandy soils are characterized by a faster mineralization rate and usually acidic conditions [56], limiting the biological oxidation of N-NH4. The latter process also had an apparently predominant role in regulating the nitrate concentration, since its significantly lowest value (10.98 mg kg−1) was reported in soils containing the smallest level of fine particle fractions (Figure 4c). Additionally, poorly structured sandy soils are prone to nitrate leaching losses due to faster water movement. Cameron et al. [59] cited data that gave an approximate ratio of leaching losses of 1:5 for a clay loam soil compared to a silt loam soil.
Changes in the Nmin pool observed in the studied agricultural ecosystems, similarly to other studies [16,59], were mainly a result of the higher mineral N application rates in these systems (Figure 6). Interestingly, significant accumulation of nitrates occurred at a rate above 50 kg N ha−1 (12.43 mg kg−1) and their concentration almost doubled after exceeding 150 kg N ha−1 (20.07 mg kg−1), which might increase the risk of N losses due to leaching. In contrast to N-NO3, the availability of ammonium declined by 34.35% when more than 50 kg N ha−1 was applied and remained constant with increasing doses of N (Figure 6). This was undoubtedly due to the N fluxes, i.e., mineralization, nitrification, denitrification, and volatilization, which are highly variable in response to mineral N input [7]. Similar dynamics of mineral nitrogen were observed in the case of pre-crop N fertilization (Figure 7). According to Cameron et al. [59], 15–25% of the N in applied mineral fertilizers remains in organic forms after harvest. Furthermore, the residual mineral N may be leached over the subsequent winter if excessive rates of nitrogen inputs are used.
Organic amendments did not significantly support mineral nitrogen contents in the present study (Figure 5). Most of the nitrogen in manured soils may be subjected to immobilization, and its transformation into mineral forms available to plants occurs in the long term, as reported by Christodoulou et al. [14].
4. Conclusions
In the environmental study, soil organic carbon levels were determined by the factors limiting its accumulation and contributed significantly to the availability of mineral nitrogen. The variables analyzed in the study in terms of their importance on the organic carbon content were ranked as follows: soil pH > pre-crop N fertilization > crop N fertilization > N applied with manure > soil texture. The presence of nitrogen in the plant available pool was influenced by the same factors that governed SOM, but their importance was slightly different, i.e., both in the case of N-NH4 and N-NO3, mineral fertilization was the most important variable affecting their contents. Carbon and nitrogen management strategy in agroecosystems should take into account the ranks of factors controlling their contents.
Author Contributions
Conceptualization, S.K. and M.S.; methodology, S.K. and M.S.; formal analysis, S.K., M.S., P.T., W.L. and J.M.; writing–original draft preparation, review and editing, M.S. and S.K. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the Ministry of Science and Higher Education, and the Ministry of Agriculture and Rural Development.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Lal, R. Digging deeper: A holistic perspective of factors affecting soil organic carbon sequestration in agroecosystems. Glob. Change Biol. 2018, 24, 3285–3301. [Google Scholar] [CrossRef]
- Lal, R. Managing soils for negative feedback to climate change and positive impact on food and nutritional security. J. Soil Sci. Plant Nutr. 2020, 66, 1–9. [Google Scholar] [CrossRef]
- Lal, R. Soil health and carbon management. Food Energy Secur. 2016, 5, 212–222. [Google Scholar] [CrossRef]
- Parfitt, R.L.; Salt, G.J. Carbon and nitrogen mineralisation in sand, silt, and clay fractions of soils under maize and pasture. Soil Res. 2001, 39, 361–371. [Google Scholar] [CrossRef]
- Wang, Y.; Ying, H.; Yin, Y.; Zheng, H.; Cui, Z. Estimating soil nitrate leaching of nitrogen fertilizer from global meta-analysis. Sci. Total Environ. 2019, 657, 96–102. [Google Scholar] [CrossRef]
- Xu, H.; Liu, K.; Zhang, W.; Rui, Y.; Zhang, J.; Wu, L.; Colinet, G.; Huang, Q.; Chen, X.; Xu, M. Long-term fertilization and intensive cropping enhance carbon and nitrogen accumulated in soil clay-sized particles of red soil in South China. J. Soils Sediments 2020, 20, 1824–1833. [Google Scholar] [CrossRef]
- Lu, X.; Hou, E.; Guo, J.; Gilliam, F.S.; Li, J.; Tang, S.; Kuang, Y. Nitrogen addition stimulates soil aggregation and enhances carbon storage in terrestrial ecosystems of China: A meta-analysis. Glob. Change Biol. 2021, 27, 2780–2792. [Google Scholar] [CrossRef]
- Cai, A.; Feng, W.; Zhang, W.; Xu, M. Climate, soil texture, and soil types affect the contributions of finefraction-stabilized carbon to total soil organic carbon in different land uses across China. J. Environ. Manag. 2016, 172, 2–9. [Google Scholar] [CrossRef]
- Rodrigues, L.; Hardy, B.; Huyghebeart, B.; Fohrafellner, J.; Fornara, D.; Barančíková, G.; Bárcena, T.G.; De Boever, M.; Di Bene, C.; Feizienė, D.; et al. Achievable agricultural soil carbon sequestration across Europe from countryspecific estimates. Glob. Chang. Biol. 2021, 27, 6363–6380. [Google Scholar] [CrossRef]
- COWI; Ecologic Institute; IEEP. Technical Guidance Handbook—Setting up and Implementing Result-Based Carbon Farming Mechanisms in the EU.; Report to the European Commission, DG Climate Action, under Contract No. CLIMA/C.3/ETU/2018/007; COWI: Kongens Lyngby, Denmark, 2021. [Google Scholar]
- Geng, J.; Cheng, S.; Fang, H.; Pei, J.; Xu, M.; Lu, M.; Yang, Y.; Cao, Z.; Li, Y. Different molecular characterization of soil particulate fractions under N deposition in a subtropical forest. Forests 2019, 10, 914. [Google Scholar] [CrossRef]
- Lal, R.; Negassa, W.; Lorenz, K. Carbon sequestration in soil. Curr. Opin. Environ. Sustain. 2015, 15, 79–86. [Google Scholar] [CrossRef]
- Navarro-Pedreño, J.; Almendro-Candel, M.B.; Zorpas, A.A. The increase of soil organic matter reduces global warming, myth or reality? Science 2021, 3, 18. [Google Scholar] [CrossRef]
- Christodoulou, E.; Agapiou, A.; Anastopoulos, I.; Omirou, M.; Ioannides, I.M. The effects of different soil nutrient management schemes in nitrogen cycling. J. Environ. Manag. 2019, 243, 168–176. [Google Scholar] [CrossRef]
- Majumder, S.P.; Krishna Chaitanya, A.; Datta, A.; Padhan, D.; Badole, S.; Mandal, B. Dynamics of Carbon and Nitrogen in Agricultural Soils: Role of Organic and Inorganic Sources. In Soil Management and Climate Change: Effects on Organic Carbon, Nitrogen Dynamics, and Greenhouse Gas Emissions; Academic Press: Cambridge, MA, USA, 2018; pp. 151–169. [Google Scholar]
- Templer, P.H.; Mack, M.C.; Chapin, F.S.; Christenson, L.M.; Compton, J.E.; Crook, H.D.; Currie, W.S.; Curtis, C.J.; Dail, D.B.; D’Antonio, C.M.; et al. Sinks for nitrogen inputs in terrestrial ecosystems: A meta-analysis of 15N tracer field studies. Ecology 2012, 93, 1816–1829. [Google Scholar] [CrossRef]
- Dal Molin, S.J.; Ernani, P.R.; Gerber, J.M. Soil acidification and nitrogen release following application of nitrogen fertilizers. Commun. Soil Sci. Plant Anal. 2020, 51, 2551–2558. [Google Scholar] [CrossRef]
- O’Brien, S.L.; Jastrow, J.D.; Grimley, D.A.; Gonzalez-Meler, M.A. Edaphic controls on soil organic carbon stocks in restored grasslands. Geoderma 2015, 251, 117–123. [Google Scholar] [CrossRef]
- Skowrońska, M. Impact of wastes application on selected soil quality parameters. Proc. ECOpole 2007, 1, 227–232. [Google Scholar]
- Šimanský, V.; Juriga, M.; Jonczak, J.; Uzarowicz, Ł.; Stępień, W. How relationships between soil organic matter parameters and soil structure characteristics are affected by the long-term fertilization of a sandy soil. Geoderma 2019, 342, 75–84. [Google Scholar] [CrossRef]
- Filipek, T.; Badora, A.; Lipiński, W.; Brodowska, M.S.; Domańska, J.; Harasim, P.; Kozłowska-Strawska, J.; Skowron, P.; Skowrońska, M.; Tkaczyk, P. Acidification and Liming of Soils; FAPA: Warsaw, Poland, 2015; p. 236. (In Polish) [Google Scholar]
- Paradelo, R.; Virto, I.; Chenu, C. Net effect of liming on soil organic carbon stocks: A review. Agric. Ecosyst. Environ. 2015, 202, 98–107. [Google Scholar] [CrossRef]
- Zhuang, S.; Li, Q.; Lv, P.; Gui, R. Effect of liming on soil nitrogen loss in runoff from a cultivated bamboo stand. Soil Water Conserv. 2016, 71, 356–363. [Google Scholar] [CrossRef]
- Skowrońska, M.; Bielińska, E.J.; Szymański, K.; Futa, B.; Antonkiewicz, J.; Kolodziej, B. An integrated assessment of the long-term impact of municipal sewage sludge on the chemical and biological properties of soil. Catena 2020, 189, 104484. [Google Scholar] [CrossRef]
- Lityński, T.; Jurkowska, H.; Gorlach, E. Chemical and Agricultural Analysis; PWN: Warsaw, Poland, 1976. (In Polish) [Google Scholar]
- Jones, J.B., Jr.; Case, V.V. Sampling, handling, and analyzing plant tissue samples. In Soil Testing and Plant Analysis, 3rd ed.; Westerman, R.L., Ed.; SSSA Book Series, 3; Soil Science Society of America: Madison, WI, USA, 1990. [Google Scholar]
- Kögel-Knabner, I.; Amelung, W. Soil organic matter in major pedogenic soil groups. Geoderma 2021, 384, 114785. [Google Scholar] [CrossRef]
- Solly, E.F.; Weber, V.; Zimmermann, S.; Walthert, L.; Hagedorn, F.; Schmidt, M.W.I. Is the content and potential preservation of soil organic carbon reflected by cation exchange capacity? A case study in Swiss forest soils. Biogeosciences Discuss. 2019; preprint. [Google Scholar] [CrossRef]
- Leifeld, J.; Zimmermann, M.; Fuhrer, J. Simulating decomposition of labile soil organic carbon: Effects of pH. Soil Biol. Biochem. 2008, 40, 2948–2951. [Google Scholar] [CrossRef]
- Ghimire, R.; Machado, S.; Bista, P. Soil pH, soil organic matter, and crop yields in winter wheat-summer fallow systems. J. Agron. 2017, 109, 706–717. [Google Scholar] [CrossRef]
- Neina, D. The role of soil pH in plant nutrition and soil remediation. Appl. Environ. Soil Sci. 2019, 2019, 5794869. [Google Scholar] [CrossRef]
- Chowdhury, S.; Bolan, N.; Farrell, M.; Sarkar, B.; Sarkar, J.R.; Kirkham, M.B.; Hossain, M.Z.; Kim, G.-H. Role of cultural and nutrient management practices in carbon sequestration in agricultural soil. Adv. Agron. 2021, 166, 131–196. [Google Scholar]
- Gross, A.; Glaser, B. Meta-analysis on how manure application changes soil organic carbon storage. Sci. Rep. 2021, 11, 5516. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Kuzyakov, I.; Raza, S.; Zhou, J.; Zamanian, K. Letter-to-the-Editor: Does acidification really increase soil carbon in croplands? How statistical analyses of large datasets might mislead the conclusions. Geoderma 2021, 384, 114806. [Google Scholar] [CrossRef]
- Anderssona, S.; Nilsson, I.; Saetre, P. Leaching of dissolved organic carbon (DOC) and dissolved organic nitrogen (DON) in mor humus as affected by temperature and pH. Soil Biol. Biochem. 2000, 32, 1–10. [Google Scholar] [CrossRef]
- Wiesmeier, M.; Urbanski, L.; Hobley, E.; Lang, B.; von Lützow, M.; Marin-Spiotta, E.; van Wesemael, B.; Rabot, E.; Ließ, M.; Garcia-Franco, N.; et al. Soil organic carbon storage as a key function of soils—A review of drivers and indicators at various scales. Geoderma 2019, 333, 149–162. [Google Scholar] [CrossRef]
- Singh, M.; Sarkar, B.; Bolan, N.S.; Ok, Y.S.; Churchman, G.J. Decomposition of soil organic matter as affected by clay types, pedogenic oxides and plant residue addition rates. J. Hazard. Mater. 2019, 374, 11–19. [Google Scholar] [CrossRef]
- Matus, F.J. Fine silt and clay content is the main factor defining maximal C and N accumulations in soils: A meta-analysis. Sci. Rep. 2021, 11, 6438. [Google Scholar] [CrossRef]
- Frasier, I.; Quiroga, A.; Fernández, R.; Álvarez, C.; Gómez, F.; Scherger, E.; Gili, A.; Noellemeyer, E. Soil type, land-use and -management as drivers of root-C inputs and soil C storage in the semiarid pampa region, Argentina. Soil Tillage Res. 2019, 192, 134–143. [Google Scholar] [CrossRef]
- Angst, G.; Mueller, K.E.; Nierop, K.G.; Simpson, M.J. Plant-or microbial-derived? A review on the molecular composition of stabilized soil organic matter. Soil Biol. Biochem. 2021, 156, 108189. [Google Scholar] [CrossRef]
- Mustafa, A.; Hu, X.; Abrar, M.M.; Shah, S.A.A.; Nan, S.; Saeed, Q.; Kamran, M.; Naveed, M.; Conde-Cid, M.; Hongjun, G.; et al. Long-term fertilization enhanced carbon mineralization and maize biomass through physical protection of organic carbon in fractions under continuous maize cropping. Appl. Soil Ecol. 2021, 165, 103971. [Google Scholar] [CrossRef]
- Plaza-Bonilla, D.; Álvaro-Fuentes, J.; Cantero-Martínez, C. Soil aggregate stability as affected by fertilization type under semiarid no-tillage conditions. Soil Sci. Soc. Am. J. 2013, 77, 284. [Google Scholar] [CrossRef]
- Ladha, J.K.; Reddy, C.K.; Padre, A.T.; van Kessel, C. Role of nitrogen fertilization in sustaining organic matter in cultivated soils. J. Environ. Qual. 2011, 40, 1756–1766. [Google Scholar] [CrossRef]
- Ren, T.; Wang, J.; Chen, Q.; Zhang, F.; Lu, S. The effects of manure and nitrogen fertilizer applications on soil organic carbon and nitrogen in a high-input cropping system. PLoS ONE 2014, 9, e97732. [Google Scholar] [CrossRef]
- Vasbieva, M. Effect of long-term application of organic and mineral fertilizers on the organic carbon content and nitrogen regime of Soddy-Podzolic soil. Eurasian Soil Sci. 2019, 52, 1422–1428. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Z.-L.; Zhang, Q.; Hu, N.; Li, Z.; Lou, Y.; Li, Y.; Xue, D.; Chen, Y.; Wu, C.; et al. Long-term effects of nitrogen fertilization on aggregation and localization of carbon, nitrogen and microbial activities in soil. Sci. Total Environ. 2018, 624, 1131–1139. [Google Scholar] [CrossRef]
- Ding, X.; Yuan, Y.; Liang, Y.; Li, L.; Han, X. Impact of long-term application of manure, crop residue, and mineral fertilizer on organic carbon pools and crop yields in a mollisol. J. Soils Sediments 2014, 14, 854–859. [Google Scholar] [CrossRef]
- Soinne, H.; Keskinen, R.; Räty, M.; Kanerva, S.; Kaseva, J.; Nuutinen, V.; Simojoki, A.; Salo, T. Soil organic carbon and clay content as deciding factors for net nitrogen mineralization and cereal yields in boreal mineral soils. Eur. J. Soil Sci. 2021, 72, 1497–1512. [Google Scholar] [CrossRef]
- He, X.; Chi, Q.; Cai, Z.; Cheng, Y.; Zhang, J.; Müller, C. 15N tracing studies including plant N uptake processes provide new insights on gross N transformations in soil-plant systems. Soil Biol. Biochem. 2019, 141, 107666. [Google Scholar] [CrossRef]
- Li, J.; Nie, M.; Powell, J.R.; Bissett, A.; Pendall, E. Soil physico-chemical properties are critical for predicting carbon storage and nutrient availability across Australia. Environ. Res. Lett. 2020, 15, 094088. [Google Scholar] [CrossRef]
- Marchezan, C.; Ferreira, P.A.A.; Silva, L.S.; Bacca, A.; Krug, A.V.; Nicoloso, F.T.; Tarouco, C.P.; Tiecher, T.L.; Brunetto, G.; Ceretta, C.A. Nitrogen availability and physiological response of corn after 12 years with organic and mineral fertilization. J. Plant. Nutr. Soil Sci. 2020, 20, 979–989. [Google Scholar] [CrossRef]
- Zhu, G.; Song, X.; Ju, X.; Zhang, J.; Müller, C.; Sylvester-Bradley, R.; Thorman, R.E.; Bingham, I.; Rees, R.M. Gross N transformation rates and related N2O emissions in Chinese and UK agricultural soils. Sci. Total Environ. 2019, 666, 176–186. [Google Scholar] [CrossRef]
- Aciego Pietri, J.C.; Brookes, P.C. Nitrogen mineralisation along a pH gradient of a silty loam UK soil. Soil Biol. Biochem. 2008, 40, 797–802. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, J.; Li, B. Determining the influence factors of soil organic carbon stock in opencast coal-mine dumps based on complex network theory. Catena 2019, 173, 433–444. [Google Scholar] [CrossRef]
- Kemmitt, S.J.; Wright, D.; Jones, D.L. Soil acidification used as a management strategy to reduce nitrate losses from agricultural land. Soil Biol. Biochem. 2005, 37, 867–875. [Google Scholar] [CrossRef]
- Fioreze, C.; Ceretta, C.A.; Giacomini, S.J.; Trentin, G.; Lorensini, F. N release in different textures soils with or without organic fertilizers|[Liberação do N em solos de diferentes texturas com ou sem adubos orgânicos]. Cienc. Rural 2012, 42, 1187–1192. [Google Scholar] [CrossRef]
- Xia, L.; Lam, S.K.; Wolf, B.; Kiese, R.; Chen, D.; Butterbach-Bahl, K. Trade-offs between soil carbon sequestration and reactive nitrogen losses under straw return in global agroecosystems. Glob. Change Biol. 2018, 24, 5919–5932. [Google Scholar] [CrossRef] [PubMed]
- Cai, A.; Xu, H.; Duan, Y.; Zhang, X.; Ashraf, M.N.; Zhang, W.; Xu, M. Changes in mineral-associated carbon and nitrogen by long-term fertilization and sequestration potential with various cropping across China. Soil Tillage Res. 2021, 205, 104725. [Google Scholar] [CrossRef]
- Cameron, K.C.; Di, H.J.; Moir, J.L. Nitrogen losses from the soil/plant system: A review. Ann. Appl. Biol. 2013, 162, 145–173. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).