Abstract
Cadmium (Cd) contamination harms plant growth and human health. The application of hemin (ferroprotoporphyrin IX) can effectively relieve abiotic stresses in plants. This work investigates the effects of hemin on alleviating Cd toxicity and enhancing Cd tolerance in maize seedlings. In this study, maize seedlings were cultivated in nutrient solutions, with a combination of CdCl2 (464 μmol L−1) and hemin (100 μmol L−1). We measured plant growth status, water status, Cd concentration, and Cd distribution in maize seedlings. The results indicated that Cd stress increased Cd accumulation in plants and inhibited plant growth. However, hemin alleviated the growth inhibition and improved water balance, root morphology, and root vitality under Cd stress. Additionally, hemin increased 2,4-dihydroxy-7-methoxy-1,4-benzoxazin-3-one (DIMBOA), total phenolic content, and phenylalanine ammonia lyase (PAL) activity to enhance tolerance to Cd stress. Hemin reduced Cd concentration, the translocation factor (TF), and the bioconcentration factor (BCF) in maize seedlings under Cd stress. Furthermore, hemin increased Cd concentrations in the cell wall and the soluble fraction of seedling roots, which helped reduce Cd transport from root to shoot. In summary, exogenous hemin could be used for alleviating adverse impacts on maize seedling induced by Cd stress.
1. Introduction
Cadmium (Cd) is one of the most hazardous and widespread environmental contaminants [1,2]. Cd is increasingly seriously exposed to the ecological system due to the influence of human activities like industrial waste emissions, sewage irrigation, and unreasonable fertilizer application in agricultural production [3,4]. Plants grown on Cd-contaminated soils can easily absorb Cd through their roots, and Cd can accumulate in other parts of the plant due to Cd’s high mobility and availability in soil [5]. High Cd concentrations in plants inhibit plant growth and lead to a potential detriment to human health through the food chain cycle [6]. Humans ingesting Cd-contaminated food over a long time will easily accumulate Cd content above the safety margin. Therefore, it is essential for food security and human health to alleviate the negative impacts of Cd on plant growth and restrict Cd absorption and accumulation in plants under Cd stress.
High Cd content in crops produces a series of toxic effects, thus reducing agricultural yield and quality [7]. As is well known, plant roots are in direct contact with Cd-contaminated soil, and their growth will be severely inhibited; accordingly, this impairs the absorption of water and nutrients by plant roots [8]. High Cd concentrations in plants cause a reduction of photosynthetic pigment content and enzyme activities and stomatal conductance, resulting in an inhibition of photosynthesis and a decrease in plant stature and biomass [9,10]. Moreover, Cd stress causes severe damage to the cell membrane structure and antioxidant system, thereby destroying plant growth [11]. Much effort has been devoted to exploring approaches to alleviate excess Cd toxicity in plants. Exogenous application is one of the most effective approaches to alleviating the negative effects of Cd toxicity. For example, exogenous melatonin improves Cd tolerance by regulating the antioxidant systems in wheat seedlings [12]. Chitosan alleviates the growth inhibition of the root system, improving antioxidant enzyme activities and enhancing the photosynthesis of plants under Cd stress [13].
Hemin (ferroprotoporphyrin IX) is a metal porphyrin compound that is formed by the complexation of a molecule of ferrous iron and protoporphyrin [14]. Over the past few years, hemin has been widely used in multiple industries in safe artificial pigments [15], iron supplements [16], and pharmaceutical materials [17]. Recently, many studies have shown that exogenously applied hemin effectively relieves the adverse effects of abiotic stresses in various plants. For example, hemin treatment alleviates the inhibition of root elongation caused by salt stress, which enhances the tolerance to salt toxicity in wheat [18]. Exogenous applications of hemin significantly alleviated the oxidative damage triggered by Cd stress in Medicago sativa [19]. The role of hemin in increasing photosynthetic activity, decreasing Cd accumulation, and improving plant growth has also been reported in rice seedlings [20].
Maize is one of the major cereal crops in the world; it greatly contributes to food crop production [21]. In recent years, soil Cd contamination has become increasingly severe. High levels of Cd in maize have destroyed the growth and development of maize and decreased the grain’s nutritional quality, causing a further threat to human health [22,23]. Therefore, efforts to impede Cd absorption and transport from soil to plants and reduce Cd accumulation in the aboveground fraction are crucial for alleviating Cd toxicity in maize and safeguarding human health. One of the most effective means to alleviating Cd toxicity in plants is applying exogenous additives. The beneficial role of hemin against Cd toxicity has been reported in previous studies [24,25]. However, they have mostly focused on the role of improving shoot and root growth and enhancing antioxidative capacity [25,26]. There are few studies on the effect of exogenously applied hemin on growth, root characteristics, Cd accumulation, translocation, and subcellular distribution in maize seedlings under Cd stress. This study aims to reveal the response and mitigation mechanism of hemin on maize seedlings under Cd stress, providing an experimental basis for improving plant growth and enhancing the tolerance of maize to Cd stress.
2. Materials and Methods
2.1. Plant Growth Conditions and Treatments
Maize seeds (Zea may L., cultivar Tiannong 9) were sterilized with 10% sodium hypochlorite for 10 min and then rinsed with distilled water. The seeds were soaked in distilled water for 7 h and then germinated on wet filter paper in the dark at 25 °C for 48 h. The germinated seeds were transferred to a growth chamber where the temperature was set to 28 /25 °C, corresponding to a 12 h/12 h (day/night) photoperiod. Light intensity was kept at 400 μmol m−2 s−1, and relative humidity was maintained at 60–70%. After the first two leaves were fully expanded, uniform-germinated seedlings were selected and sown in plastic pots containing 1/2-strength Hoagland nutrient solution (40 L, pH 6.8). The nutrient solution was renewed every 3 days. When the seedlings fully expanded their third leaf, 4 treatments were established as follows: (1) 1/2-strength Hoagland nutrient solution (CK); (2) 1/2-strength Hoagland nutrient solution + 100 μmol L−1 hemin (Hemin); (3) 1/2-strength Hoagland nutrient solution + 464 μmol L−1 CdCl2 (Cd); (4) 1/2-strength Hoagland nutrient solution + 464 μmol L−1 CdCl2 + 100 μmol L−1 hemin (Hemin + Cd). Each treatment contained 3 replicates with 30 plants (90 plants in total). The seedlings were cultured for 4 days after the treatments and collected for measurement.
2.2. Determination of Plant Growth and Root Characteristic Parameters
The plants were separated into roots and shoots after 4 days of treatment. The leaf area was recorded by leaf meter (SHY-150, Harbin, China). The root/shoot ratio was calculated as dry root weight/dry shoot weight. The root characteristic parameters, including root length, surface area, volume, mean diameter, and the number of root tips, were scanned using a root scanner (MRS-9600TFU2L, Shanghai, China). To determine dry weight, the roots and shoots were oven-dried at 105 °C for 0.5 h and dried to constant weight at 80 °C.
2.3. Determination of Leaf Relative Water Content and Leaf Water Potential
To measure relative water content (RWC), the second fully expanded leaf per replicate was collected and weighed to estimate fresh weight. After that, leaf samples were immediately immersed in distilled water for 24 h in the dark to determine turgid weight. Finally, the samples were dried at 80 °C for 48 h to determine dry weight. The following formula was used to calculate RWC: RWC (%) = (fresh weight − dry weight)/(turgid weight − dry weight) × 100.
The second fully expanded leaf’s water potential (Ψw) was measured using a 3115 pressure chamber (Beijing, China) system. Pressure was applied to the leaf surface in the chamber; when water began to appear at the cut end of the leaf’s midrib, the pressure value was recorded as leaf Ψw.
2.4. Determination of Root Hydraulic Conductance
Each shoot was cut off at the base of the root system, leaving 4 cm of stem, and sealed with silicone seals. Thereafter, the whole root was placed in a pressure chamber. The pressure was raised in steps of 0.1 MPa up to 0.4 MPa. The sap was collected with absorbent cotton at each pressure gradient (for 1 min) and then weighed, generating a range of sap flows that represent the whole-plant. Root hydraulic conductance (Lp) was determined according to the following equation: Lp = Jv/P, where Jv (m s−1) and P (MPa) indicated the flow rate and external pressure, respectively [27].
2.5. Determination of Root Vitality and the Root Length Stress Tolerance Index
Root vitality was determined using triphenyltetrazolium chloride (TTC) [28]. The roots were cut into small pieces and incubated in the solution containing 5 mL 0.4% TTC and 0.1 M PBS (pH 7.0) for 1 h at 37 °C. Then, we added 20 mL methanol, followed by incubation for 6 h at 37 °C. The total extracted solution was determined at the absorbance of 485 nm. The Root Length Stress Tolerance Index (RLSI) was calculated following the method in [29]: root length of treatment plant/root length of control plant.
2.6. Determination of Cd Concentration, the Translocation Factor, and the Bioconcentration Factor
After 4 d of treatments, the powdery dried plant parts (shoots and roots) were used to determine Cd concentrations. The powders (0.2 g) were digested with mixed acid (HNO3/HClO4, v/v = 4/1) at 220 °C for 4 h and then diluted to 25 mL. The Cd concentration was then determined by an Optima3000 ICP-AES instrument (Perkin-Elmer, Waltham, MA, USA).
The translocation factor (TF) was calculated following the method in [30]: Cd concentration in shoots/Cd concentration in roots. The bioconcentration factor (BCF) was calculated following the method in [31]: Cd concentration in the aerial part of the plant/Cd concentration in the nutrient solution.
2.7. Determination of Nonprotein Thiol and Phytochelatin Contents
Fresh leaf samples (1 g) were ground in liquid nitrogen and homogenized in 8 mL 5% sulfosalicylic acid. The homogenate was centrifuged at 10,000× g at 4 °C for 15 min, and the supernatant was collected for determination. Nonprotein thiol (NPT) and GSH were determined according to the method of Rama Devi [32] and Li [33], respectively. The amount of phytochelatins (PCs) was calculated using the following formula: PCs = NPT − GSH.
2.8. Determination of Subcellular Cd Distribution
Fresh samples were homogenized with precooled extraction buffer, and then the homogenate was filtered using nylon cloth, and the filtrate was centrifuged at 5000× g for 20 min. The cell wall fraction was the deposit of the two operations. Next, the supernatant was centrifuged at 15,000× g for 45 min—the deposit was the cell organelle fraction, and the supernatant was the soluble fraction. Finally, these fractions were dried and digested with HNO3/HClO4 (v/v = 4/1). The Cd concentration in each fraction was determined by ICP-MS [34].
2.9. Determination of Total Phenolic and 2,4-Dihydroxy-7-Methoxy-1,4-Benzoxazin-3-One (DIMBOA) Contents and Phenylalanine Ammonia Lyase (PAL) Activity
The samples were ground to a fine powder under liquid nitrogen and then 5 mL water was added to incubate for 1 h at room temperature. After centrifugation, the supernatant was collected and extracted four times with an equal volume of ethyl acetate (EtOAc). Pooled extracts were evaporated, followed by dissolving the solid residue in 1.0 mL of MeOH for HPLC analysis. Analyses were performed using an Agilent 1100 HPLC. The mobile phase was 25% methanol and 75% acetic acid solution (pH2.6). The mobile phase flow rate was 1 mL min−1, and the total analysis time was 20 min. The sample injection volume was 20 µL, and the detection wavelength was monitored at 262 nm. The DIMBOA content was calculated by using the absorption peak areas of different concentrations of DIMBOA standard samples as a standard curve [35].
The samples were extracted with 10 mL of 70% (v/v) ethanol containing 1% hydrochloric acid and then centrifuged, and the extract was obtained after filtering. Extracts were thoroughly mixed with 1 mL Folin–Ciocalteu reagent. After standing for 10 min, 0.5 mL 7.5% Na2CO3 was added to the mixture. Spectrophotometric measurements were done at 765 nm. Content of total phenolics was calculated using gallic acid as standard [36].
The samples were homogenized in liquid nitrogen and extracted with 6 mL of sodium borate buffer (0.05 mol L−1, pH 8.8, containing 5 mmol L−1 2-mercaptoethanol, 1 mmol L−1 EDTA, 0.2 g PVP). The homogenate was centrifuged at 10,000× g for 15 min at 4 °C to obtain the supernatant. PAL activity was determined at the absorbance of 290 nm [37].
2.10. Statistical Analysis
The data were summarized to calculate mean value and standard error (SE). The mean value was compared by analysis of variance (ANOVA) to analyze the significant differences between samples with different treatments (p < 0.05). All statistical analyses were performed using SPSS 19.0 procedures (SPSS Inc., Chicago, IL, USA). Microsoft Excel 2010 was used to draw the tables.
3. Results
3.1. Seedling Growth
The results of the impact of different treatments on the growth traits of maize seedlings are presented in Table 1. In the absence of Cd stress, the application of hemin increased maize seedling leaf area, dry shoot weight, dry root weight, and root/shoot ratio, but the difference was not significant except for dry shoot weight. Compared with CK, Cd stress significantly inhibited seedling growth and decreased leaf area, dry shoot weight, and dry root weight by 20.99%, 23.68%, and 13.27%, respectively. Applying hemin significantly alleviated the growth inhibition caused by Cd and increased leaf area, dry shoot weight, and dry root weight. Hemin treatment had different effects on the root/shoot ratio under Cd stress. Cd stress significantly increased the root/shoot ratio, while hemin application reduced the root/shoot ratio by 5.49% compared with Cd stress.

Table 1.
Effects of hemin on leaf area, dry shoot weight, dry root weight, and the root/shoot ratio under Cd stress.
3.2. RWC and Ψw and Lp
From 1 to 4 d after Cd treatment, leaf Ψw and RWC values of maize seedlings showed an overall downward trend. In the absence of Cd stress, the application of hemin had no significant effect on Ψw and RWC. Compared with CK, leaf Ψw and RWC decreased by 128.14% and 28.63%, respectively, after 4 days of Cd stress. Hemin reduced Cd’s adverse effects on leaf Ψw and RWC and increased leaf Ψw and RWC by 15.94% and 11.52%, respectively. Hemin treatment had a similar impact on Lp under Cd stress. Cd stress significantly reduced Lp, while hemin application increased Lp by 29.74% compared with Cd stress (Table 2).

Table 2.
Effects of hemin on leaf relative water content (RWC), leaf water potential (Ψw), and root hydraulic conductance (Lp) under Cd stress.
3.3. Root Traits
In the absence of Cd stress, the application of hemin significantly increased root surface area, length, and diameter. Cd stress inhibited root growth, leading to 19.7%, 23.4%, 26.1%, 23.8%, and 32.8% decreases in root length, surface area, volume, mean diameter, and number of root tips, respectively. Hemin significantly relieved the inhibition of root growth under Cd stress and increased root length, surface area, volume, mean diameter, and number of root tips by 12.48%, 15.05%, 18.49%, 15.15%, and 27.03%, respectively. In the absence of Cd stress, the application of hemin significantly improved root vitality. Cd stress reduced root vitality by 47.3% compared with CK. Hemin alleviated the Cd-induced constraint of root vitality and increased root vitality by 55.9%. The values of RLSI in Cd treatment and Hemin + Cd treatment were 0.8 and 0.9, respectively. Therefore, the application of hemin significantly increased RLSI under Cd stress (Table 3).

Table 3.
Effects of hemin on root morphological traits (root length, surface area, volume, mean diameter, and number of root tips), root vitality and the root length stress tolerance index (RLSI) under Cd stress.
3.4. Cd Concentrations and Translocation
We measured Cd concentrations in root, shoot, and whole seedling in each treatment. Under Cd stress, the Cd concentration in roots was higher than shoots. The application of hemin impeded the accumulation of Cd in the plants. Compared to Cd treatment, the Cd concentrations in root, shoot, and seedling in the Hemin + Cd treatment were decreased by 13.12%, 33.8%, and 19.97%, respectively. The application of hemin inhibited Cd translocation from underground to shoot. The Hemin + Cd treatment decreased TF and BCF by 23.73% and 33.83%, respectively (Table 4).

Table 4.
Effects of hemin on Cd concentrations of shoot and root, total Cd concentration, the translocation factor (TF), and the bioconcentration factor (BCF) under Cd stress.
3.5. NPT and PC Contents
Hemin treatment observably increased NPT and PC contents of leaf and root compared with CK. Cd treatment also increased NPT and PC contents compared with CK. The application of Hemin further increased the contents of NPT and PCs in seedlings. Compared with Cd treatment, the Hemin + Cd treatment increased NPT and PC contents by 39.1% and 69.7% in leaf and 58.1% and 63.1% in root, respectively (Table 5).

Table 5.
Effects of hemin on nonprotein thiol (NPT) and phytochelatin (PC) contents of leaf and root under Cd stress.
3.6. Subcellular Distribution of Cd
Hemin has a significant influence on the distribution of Cd in subcellular fractions of maize leaf and root under Cd stress. In leaf, the Hemin + Cd treatment reduced Cd concentration in the soluble fraction and the organelle by 68.72% and 63.05% compared with Cd treatment, while Cd concentration in the cell wall increased, although not significantly. In root, the Hemin + Cd treatment increased Cd concentration in the soluble fraction and the cell wall by 92.4% and 118.7% compared with Cd treatment, while Cd concentration in the organelle decreased, although not significantly (Table 6).

Table 6.
Effects of hemin on subcellular distribution (soluble fraction, cell organelle, and cell wall) of Cd in roots and shoots under Cd stress.
3.7. Total Phenolics, DIMBOA, and PAL Activity
The contents of DIMBOA and total phenolics and PAL activity in roots were higher than those in leaves. In the absence of Cd stress, the application of hemin increased DIMBOA and total phenolic contents and PAL activity in roots and leaves on the second and fourth days after treatment. Cd stress significantly reduced the contents of DIMBOA and total phenolics and PAL activity compared with CK in the seedlings. However, the application of hemin counteracted the Cd-induced reduction of secondary metabolite contents and PAL activity. In root, the Hemin + Cd treatment increased DIMBOA content, total phenolics content, and PAL activity by 34.93%, 33.68%, and 23.31% on the second day after treatment compared with Cd treatment and increased them by 51.47%, 49.50%, and 33.33% on the fourth day after treatment, respectively. In leaf, the Hemin + Cd treatment increased DIMBOA content, total phenolic content, and PAL activity by 36.27%, 34.22%, and 19.58% on the second day after treatment compared with Cd treatment and increased them by 74.91%, 70.28%, and 39.90% on the fourth day after treatment, respectively (Table 7).

Table 7.
Effects of hemin on 2,4-dihydroxy-7-methoxy-1,4-benzoxazin-3-one (DIMBOA) and total phenolic contents and phenylalanine ammonia lyase (PAL) activity in roots and shoots under Cd stress.
4. Discussion
Under Cd stress, plants often show growth inhibition and biomass reduction [38,39]. It has been reported that the suppression of plant growth is one of the most crucial characteristics in plants under Cd stress [40]. This study shows that Cd treatment significantly decreases leaf area, dry shoot weight, and dry root weight in maize seedlings. Hemin alleviated the Cd-induced repression of maize growth in the presence of Cd stress. It has been reported that root morphology is an important indicator for evaluating Cd toxicity in plants. Cd exposure decreases total root length, number of root tips, and root radial expansion, which inhibit root growth and decrease plant root absorption capacity [41,42,43]. In this study, Cd stress reduced root length, root surface area, root volume, root average diameter, and the number of root tips. Exogenous hemin improved the characteristic parameters of the maize root system and increased root vitality and RLSI under Cd stress, which are beneficial for promoting the absorption of water and nutrients. Cd affects the permeability of the plasma membrane by inducing lipid peroxidation, leading to an imbalance between Cd and water and causing a water deficit [44]. Previous studies have shown that hemin reduces the active oxygen contents in plants under heavy metal stress and reduces lipid peroxidation induced by oxidative stress [26,45]. In this study, Cd stress decreased leaf RWC, leaf Ψω, and root Lp. However, hemin enhanced water flow efficiency through maize roots and improved the water status in shoots, accordingly increasing the tolerance of maize seedlings under Cd stress.
Plants growing in Cd-contaminated soil can easily absorb and accumulate Cd. Cd is harmful to plant growth, and crops that contain Cd threaten human food security through the food chain [46,47]. Therefore, reducing Cd accumulation is the key to alleviating Cd toxicity in plants. In this study, we found that exogenous hemin reduced the Cd concentration in the shoots and roots of maize seedlings under Cd stress. This result is consistent with another study that reduced Cd accumulation in Chinese cabbage seedlings by exogenous hemin [24]. At present, the mechanism of hemin reducing Cd accumulation in plants is not clear. Hemin is an inducer of HO1, which can induce the expression of HO1 [48]. The expression of HO1 has been shown to be beneficial in inhibiting the absorption and accumulation of heavy metals in Indian mustard [49]. HO1 can also specifically degrade heme to produce biliverdin, Fe, and CO. HY1(AtHO-1) might improve Fe homeostasis under Cd stress, thereby relieving Cd accumulation in Arabidopsis [50]. Han et al. [51] considered that CO could induce alfalfa tolerance against Cd toxicity. The mechanism of hemin reducing Cd absorption and accumulation in plants needs to be further explored.
Cd’s chemical forms and subcellular distribution directly determine Cd activity in the cell and further reflect the plants’ toxicity level [52,53]. NPTs and PCs are important heavy metal chelating agents in plants, which can chelate with Cd ions in the cytoplasm and then be transported to the vacuole to synthesize high molecular complexes with stable properties, thereby alleviating Cd toxicity [54,55]. In this study, NPT and PC contents increased under Cd stress. However, the NPT and PC contents in maize seedlings further increased after applying hemin under Cd stress. This indicates that hemin plays an important role in alleviating Cd toxicity in maize seedlings by increasing Cd ion chelation under Cd stress. Plant root cell walls are directly contacted with heavy metals dissolved in the soil solution, which is the outermost barrier to protect protoplasts from Cd toxicity [56,57]. The constituent of the root cell wall (such as pectin, hemicellulose, and protein) can combine with Cd and facilitate Cd deposit on the cell wall, thereby restraining Cd transport into the protoplast and reducing Cd toxicity [58,59]. In this study, exogenous hemin increased Cd concentration in the root cell wall, which enhanced the detoxification ability of maize seedlings under Cd exposure. Moreover, hemin improved Cd transport from protoplasts to vacuoles by increasing Cd concentration in the soluble fraction of maize roots. Cd concentration in the soluble fraction (vacuole) and the organelle in leaves was significantly reduced by hemin, which further reduced Cd toxicity. Furthermore, we found that hemin reduced TF and BCF in maize seedlings under Cd stress. This indicated that hemin could reduce Cd absorption and transportation in maize seedlings. The results showed that hemin reduced Cd toxicity by altering Cd’s chemical form and increased Cd retention in the root system by promoting the deposit of Cd on the root cell wall and the compartmentalization of the vacuole, which ultimately resulted in the reduction of Cd transport.
Secondary metabolites play an important role in regulating plant growth and responding to various biotic and abiotic stresses [60]. Cyclic hydroxamates are the main secondary metabolite in cereal plants, and their main substances include DIMBOA [61]. When destroyed by mechanical damage, insect pests, and pathogen invasion, plants will release activated DIMBOA [62,63,64]. Polyphenol is an important secondary metabolite, with antioxidant activity in plants [65]. As part of the plant defense system, polyphenols can effectively relieve the damage caused by free radical accumulation in plants [66]. As a key enzyme in phenylpropane metabolism, PAL has an important impact on plant secondary metabolism [67]. In this study, Cd treatment significantly reduced DIMBOA and total phenol contents and PAL activity in root and leaf, while hemin treatment increased DIMBOA and total phenol contents and PAL activity under Cd stress. This shows that exogenous hemin can improve the chemical defense response to Cd stress and improve the tolerance of maize seedlings.
5. Conclusions
Hemin promotes water status by improving root traits, root Lp, leaf RWC, and Ψw of maize. Hemin increases Cd deposition in maize root by improving chelation and subcellular distribution, and hemin reduces Cd absorption and transport, which contribute to reducing Cd concentration in maize seedlings. Moreover, hemin increases secondary metabolites in maize seedlings, which improves the chemical defense response to Cd stress (Figure 1). Therefore, exogenous hemin reduces the growth inhibition caused by Cd stress and enhances Cd tolerance of maize seedlings.
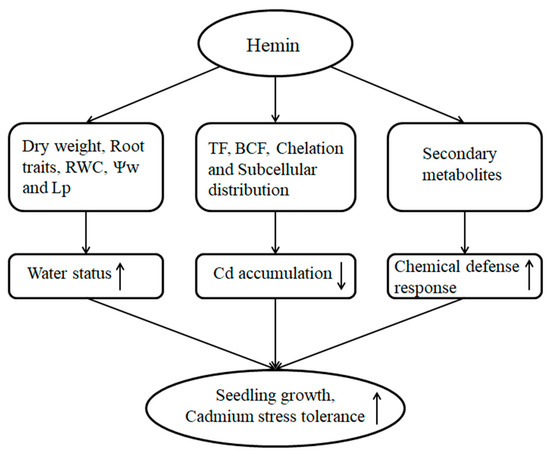
Figure 1.
Schematic representation of the positive role of hemin on tolerance of maize to Cd stress. A model was developed to show that water status, Cd concentration, and chemical defense response were regulated by hemin in maize under Cd stress. The arrows (↑) and (↓) represent the positive and passive roles of hemin, respectively.
Author Contributions
X.L. and Y.M. performed the entire experiment, analyzed the results, and drafted the manuscript. W.G. designed the entire experiment and was in charge of manuscript revisions. S.W. and W.G. interpreted the results and prepared the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Key R&D Program of China (Grant No. 2016YFD0300103 and 2017YFD0300506).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data sets analysed during the current study are available from the current author on reasonable request.
Acknowledgments
We thank Li Congfeng, Institute of Crop Sciences, Chinese Academy of Agricultural Sciences, for providing the experimental design and laboratory of this study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Larson, C. China Gets Serious About Its Pollutant-Laden Soil. Science 2014, 343, 1415–1416. [Google Scholar] [CrossRef]
- Luo, J.; Huang, J.; Zeng, D.; Peng, J.; Zhang, G.; Ma, H.; Guan, Y.; Yi, H.; Fu, Y.; Han, B.; et al. A Defensin-like Protein Drives Cadmium Efflux and Allocation in Rice. Nat. Commun. 2018, 9, 645. [Google Scholar] [CrossRef]
- Wu, J.; Shi, Y.; Zhu, Y.; Wang, Y.; Gong, H. Mechanisms of Enhanced Heavy Metal Tolerance in Plants by Silicon: A Review. Pedosphere 2013, 23, 815–825. [Google Scholar] [CrossRef]
- Kikuchi, T.; Okazaki, M.; Toyota, K.; Motobayashi, T.; Kato, M. The Input-output Balance of Cadmium in a Paddy Field of Tokyo. Chemosphere 2007, 67, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Zia-ur-Rehman, M.; Khalid, H.; Akmal, F.; Ali, S.; Rizwan, M.; Qayyum, M.F.; Iqbal, M.; Khalid, M.U.; Azhar, M. Effect of Limestone, Lignite and Biochar Applied Alone and Combined on Cadmium Uptake in Wheat and Rice under Rotation in an Effluent Irrigated Field. Environ. Pollut. 2017, 227, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Benavides, M.; Gallego, M.S.; Tomaro, M.L. Cadmium Toxicity in Plants. Braz. J. Plant Physiol. 2005, 17, 21–34. [Google Scholar] [CrossRef]
- Chen, J.; Duan, B.; Xu, G.; Korpelainen, H.; Niinemets, U.; Li, C. Sexual Competition Affects Biomass Partitioning, Carbon-nutrient Balance, Cd Allocation and Ultrastructure of Populus cathayana Females and Males Exposed to Cd Stress. Tree Physiol. 2016, 36, 1353–1368. [Google Scholar] [CrossRef]
- Lux, A.; Martinka, M.; Vaculik, M.; White, P.J. Root Responses to Cadmium in the Rhizosphere: A Review. J. Exp. Bot. 2010, 62, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Parmar, P.; Kumari, N.; Sharma, V. Structural and Functional Alterations in Photosynthetic Apparatus of Plants under Cadmium Stress. Bot. Stud. 2013, 54, 45. [Google Scholar] [CrossRef]
- Mobin, M.; Khan, N.A. Photosynthetic Activity, Pigment Composition and Antioxidative Response of Two Mustard (Brassica juncea) Cultivars Differing in Photosynthetic Capacity Subjected to Cadmium Stress. J. Plant Physiol. 2007, 164, 601–610. [Google Scholar] [CrossRef]
- Semane, B.; Dupae, J.; Cuypers, A.; Noben, J.P.; Tuomainen, M.; Tervahauta, A.; Karenlampi, S.; Van Belleghem, F.; Smeets, K.; Vangronsveld, J. Leaf Proteome Responses of Arabidopsis thaliana Exposed to Mild Cadmium Stress. J. Plant Physiol. 2010, 167, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Wang, Q.; Shah, F.A.; Liu, W.; Wang, D.; Huang, S.; Fu, S.; Wu, L. Exogenous Melatonin Confers Cadmium Tolerance by Counterbalancing the Hydrogen Peroxide Homeostasis in Wheat Seedlings. Molecules 2018, 23, 799. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.; Gu, W.; Zhang, L.; Li, C.; Chen, X.; Li, J.; Li, L.; Xie, T.; Wei, S. Role of Chitosan in the Regulation of the Growth, Antioxidant System and Photosynthetic Characteristics of Maize Seedlings under Cadmium stress. Russ. J. Plant Physiol. 2019, 66, 140–151. [Google Scholar] [CrossRef]
- Wagener, F.A.; Volk, H.D.; Willis, D.; Abraham, N.G.; Soares, M.P.; Adema, G.J.; Figdor, C.G. Different Faces of the Heme-heme Oxygenase System in Inflammation. Pharmacol. Rev. 2003, 55, 551–571. [Google Scholar] [CrossRef] [PubMed]
- Becker, E.M.; Cardoso, D.R.; Skibsted, L.H. Quenching of Excited States of Red-pigment Zinc Protoporphyrin IX by Hemin and Natural Reductors in Dry-cured Hams. Eur. Food. Res. Technol. 2011, 232, 343–349. [Google Scholar] [CrossRef]
- Sander, A.; Kretzer, S.; Bredt, W.; Oberle, K.; Bereswill, S. Hemin-dependent Growth and Hemin Binding of Bartonella henselae. FEMS Microbiol. Lett. 2000, 189, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Katoh, Y.; Nemoto, N.; Tanaka, M.; Takayama, S. Inhibition of Benzo[a] Pyrene-induced Mutagenesis in Chinese Hamster V79 Cells by Hemin and Related Compounds. Mutat. Res. 1983, 121, 153–157. [Google Scholar] [CrossRef]
- Xie, Y.; Cui, W.; Yuan, X.; Shen, W.; Yang, Q. Heme Oxygenase-1 is Associated with Wheat Salinity Acclimation by Modulating Reactive Oxygen Species Homeostasis. J. Integr. Plant Biol. 2011, 53, 653–670. [Google Scholar] [CrossRef]
- Han, Y.; Xuan, W.; Yu, T.; Fang, W.; Lou, T.; Gao, Y.; Chen, X.; Xiao, X.; Shen, W. Exogenous Hematin Alleviates Mercury-induced Oxidative Damage in the Roots of Medicago sativa. J. Integr. Plant Biol. 2007, 49, 1703–1713. [Google Scholar] [CrossRef]
- Chen, Q.; Gong, C.; Ju, X.; Zhu, Z.; Shen, W.; Shen, Z.; Cui, J. Hemin Through the Heme Oxygenase 1/Ferrous Iron, Carbon Monoxide System Involved in Zinc Tolerance in Oryza Sativa, L. J. Plant Growth Regul. 2018, 37, 947–957. [Google Scholar] [CrossRef]
- Palacios-Rojas, N.; McCulley, L.; Kaeppler, M.; Titcomb, T.J.; Gunaratna, N.S.; Lopez-Ridaura, S.; Tanumihardjo, S.A. Mining Maize Diversity and Improving Its Nutritional Aspects within Agro-food Systems. Compr. Rev. Food Sci. F. 2020, 19, 1809–1834. [Google Scholar] [CrossRef] [PubMed]
- Malcovska, S.M.; Ducaiova, Z.; Maslanakova, I.; Backor, M. Effect of Silicon on Growth, Photosynthesis, Oxidative Status and Phenolic Compounds of Maize (Zea mays L.) Grown in Cadmium Excess. Water Air Soil Poll. 2014, 225, 2056. [Google Scholar] [CrossRef]
- Sun, K.; Yue, Y.; Wen, D.; Li, X.; Yang, Y.; Yang, N.; Zhang, H.; Chen, N.; Wang, K. Effects of Exogenous Sulfur on Maize (Zea mays L.) Growth and Cd Accumulation in Cd-contaminated Plastic Shed Soil. Environ. Monit. Assess. 2020, 192, 651. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Huang, Y.; Wu, X.; Liu, Z.; Zou, J.; Chen, Y.; Su, N.; Cui, J. Increased Antioxidative Capacity and Decreased Cadmium Uptake Contribute to Hemin-induced Alleviation of Cadmium Toxicity in Chinese Cabbage Seedlings. Ecotox. Environ. Safety 2019, 177, 47–57. [Google Scholar] [CrossRef]
- Fu, G.; Zhang, L.; Cui, W.; Wang, Y.; Shen, W.; Ren, Y.; Zheng, T. Induction of Heme Oxygenase-1 with β-CD-hemin Complex Mitigates Cadmium-induced Oxidative Damage in the Roots of Medicago sativa. Plant Soil 2011, 345, 271–285. [Google Scholar] [CrossRef]
- Cui, W.; Zhang, J.; Xuan, W.; Xie, Y. Up-regulation of Heme Oxygenase-1 Contributes to the Amelioration of Aluminum-induced Oxidative Stress in Medicago sativa. J. Plant Physiol. 2013, 170, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Gu, W.; Zhang, L.; Li, C.; Chen, X.; Qian, C.; Wang, Z.; Li, W.; Zuo, S.; Wei, S. Exogenous 2-(3,4-Dichlorophenoxy) Triethylamine Alleviates Salinity Stress in Maize by Enhancing Photosynthetic Capacity, Improving Water Status and Maintaining K+ /Na+ Homeostasis. BMC Plant Biol. 2020, 20, 348. [Google Scholar] [CrossRef]
- Jiang, H.; Zhang, J.; Han, Z.; Yang, J.; Ge, C.; Wu, Q. Revealing New Insights into Different Phosphorus-starving Responses between Two Maize (Zea mays) Inbred Lines by Transcriptomic and Proteomic Studies. Sci. Rep. 2017, 7, 44294. [Google Scholar] [CrossRef] [PubMed]
- Salahuddin, M.; Nawaz, F.; Shahbaz, M.; Naeem, M.; Zulfiqar, B.; Shabbir, R.; Hussain, R. Effect of Exogenous Nitric Oxide (NO) Supply on Germination and Seedling Growth of Mungbean (cv. Nm-54) under Salinity Stress. Legume Res. 2017, 40, 846–852. [Google Scholar] [CrossRef]
- Song, T.; Su, X.; He, J.; Liang, Y.; Zhou, T.; Liu, C. Selenium (Se) Uptake and Dynamic Changes of Se Content in Soil-plant Systems. Environ. Sci. Pollut. Res. 2018, 25, 34343–34350. [Google Scholar] [CrossRef]
- Dumbrava, A.; Birghila, S.; Munteanu, M. Contributions on Enhancing the Copper Uptake by Using Natural Chelators, with Applications in Soil Phytoremediation. Int. J. Environ. Sci. Technol. 2015, 12, 929–938. [Google Scholar] [CrossRef]
- Rama Devi, S.; Prasad, M.N.V. Copper Toxicity in Ceratophyllum demersum L. (Coontail), a Free Floating Macrophyte: Response of Antioxidant Enzymes and Antioxidants. Plant Sci. 1998, 138, 157–165. [Google Scholar] [CrossRef]
- Li, D.; Chen, G.; Lu, Q.; Li, Y.; Wang, J.; Li, H. Responses of Two Kidney Bean (Phaseolus vulgaris) Cultivars to the Combined Stress of Sulfur Deficiency and Cadmium Toxicity. Biometals 2018, 31, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Bao, Y.; Chen, X.; Yao, Q.; Wang, C.; Chai, S.; Zeng, J.; Fan, X.; Kang, H.; Sha, L.; et al. Different Nitrogen Forms Differentially Affect Cd Uptake and Accumulation in Dwarf Polish Wheat (Triticum polonicum L.) Seedlings. J. Hazard. Mater. 2020, 400, 123209. [Google Scholar] [CrossRef] [PubMed]
- Mao, J.; Burt, A.J.; Ramputh, A.I.; Simmonds, J.; Cass, L.; Hubbard, K.; Miller, S.; Altosaar, I.; Arnason, J.T. Diverted Secondary Metabolism and Improved Resistance to European Corn Borer (Ostrinia nubilalis) in Maize (Zea mays L.) Transformed with Wheat Oxalate Oxidase. J. Agric. Food Chem. 2007, 55, 2582–2589. [Google Scholar] [CrossRef] [PubMed]
- Sheng, K.; Shui, S.; Yan, L.; Liu, C.; Zheng, L. Effect of Postharvest UV-B or UV-C Irradiation on Phenolic Compounds and Their Transcription of Phenolic Biosynthetic Genes of Table Grapes. J. Food Sci. Technol. 2018, 55, 3292–3302. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Hu, D.; Liu, X.; She, H.; Ruan, R.; Yang, H.; Yi, Z.; Wu, D. Effects of Uniconazole on the Lignin Metabolism and Lodging Resistance of Culm in Common Buckwheat (Fagopyrum esculentum M.). Field Crop. Res. 2015, 180, 46–53. [Google Scholar] [CrossRef]
- Shah, A.A.; Khan, W.U.; Yasin, N.A.; Akram, W.; Ahmad, A.; Abbas, M.; Ali, A.; Safdar, M.N. Butanolide Alleviated Cadmium Stress by Improving Plant Growth, Photosynthetic Parameters and Antioxidant Defense System of Brassica oleracea. Chemosphere 2020, 261, 127728. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Shang, Y.; Li, L.; Chen, Y.; Qin, Z.; Zhou, L.; Yuan, M.; Ding, C.; Liu, J.; Huang, Y.; et al. Cadmium Stress in Dongying Wild Soybean Seedlings: Growth, Cd Accumulation, and Photosynthesis. Photosynthetica 2018, 56, 1346–1352. [Google Scholar] [CrossRef]
- Zadeh, R.; Arvin, S.M.J.; Jamei, R.; Mozaffari, H.; Nejhad, F.R. Response of Tomato Plants to Interaction Effects of Magnetic (Fe3O4) Nanoparticles and Cadmium Stress. J. Plant Interact. 2019, 14, 474–481. [Google Scholar] [CrossRef]
- Valentovicova, K.; Mistrik, I.; Zelinova, V.; Tamas, L. How Cobalt Facilitates Cadmium- and Ethylene Precursor-induced Growth Inhibition and Radial Cell Expansion in Barley Root Tips. Cent. Eur. J. Biol. 2012, 7, 551–558. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, C.; Wang, X.; Shi, G. Cadmium-induced Alterations in Morpho-physiology of Two Peanut Cultivars Differing in Cadmium Accumulation. Acta Physiol. Plant. 2013, 35, 2105–2112. [Google Scholar] [CrossRef]
- Xu, J.; Wang, W.; Yin, H.; Liu, X.; Sun, H.; Mi, Q. Exogenous Nitric Oxide Improves Antioxidative Capacity and Reduces Auxin Degradation in Roots of Medicago truncatula Seedlings under Cadmium Stress. Plant Soil 2010, 326, 321–330. [Google Scholar] [CrossRef]
- Barcelo, J.; Cabot, C.; Poschenrieder, C. Cadmium-Induced Decrease of Water Stress Resistance in Bush Bean Plants (Phaseolus vulgaris L. cv. Contender) II. Effects of Cd on Endogenous Abscisic Acid Levels. J. Plant Physiol. 1986, 125, 27–34. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, X.; Liu, Y.; Wei, J.; Shen, W.; Shen, Z.; Cui, J. Hemin-mediated Alleviation of Zinc, Lead and Chromium Toxicity is Associated with Elevated Photosynthesis, Antioxidative Capacity; Suppressed Metal Uptake and Oxidative Stress in Rice Seedlings. Plant Growth Regul. 2017, 81, 253–264. [Google Scholar] [CrossRef]
- Farooq, H.; Asghar, H.; Khan, M.; Saleem, M.; Zahir, Z. Auxin-mediated Growth of Rice in Cadmium-contaminated Soil. Turk. J. Agric. For. 2015, 39, 272–276. [Google Scholar] [CrossRef]
- Zheng, S.; Wang, Q.; Yuan, Y.; Sun, W. Human Health Risk Assessment of Heavy Metals in Soil and Food Crops in the Pearl River Delta Urban Agglomeration of China. Food Chem. 2020, 316, 126213. [Google Scholar] [CrossRef]
- Jin, Q.; Feng, J.; Sun, Y.; Cui, W.; Han, B.; Xu, S.; Shen, W.; Cui, J. Characterization of a Heme Oxygenase–1 from Tobacco That Is Involved in Some Abiotic Stress Responses. Int. J. Plant Sci. 2012, 173, 113–123. [Google Scholar] [CrossRef]
- Li, H.; Jiang, M.; Che, L.; Nie, L.; Yang, Z. BjHO-1 is Involved in the Detoxification of Heavy Metal in India Mustard (Brassica juncea). Biometals 2012, 25, 1269–1279. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Yang, Z.; Xie, Y.; Nie, L.; Cui, J.; Shen, W. Arabidopsis HY1 Confers Cadmium Tolerance by Decreasing Nitric Oxide Production and Improving Iron Homeostasis. Mol. Plant 2014, 7, 388–403. [Google Scholar] [CrossRef]
- Han, Y.; Zhang, J.; Chen, X.; Gao, Z.; Xuan, W.; Xu, S.; Ding, X.; Shen, W. Carbon Monoxide Alleviates Cadmium-induced Oxidative Damage by Modulating Glutathione Metabolism in the Roots of Medicago sativa. New Phytol. 2008, 177, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Fang, Z.; Hu, Z.; Yin, X.; Song, G.; Cai, Q. Exogenous Glutathione Alleviation of Cd Toxicity in Italian Ryegrass (Lolium multiflorum) by Modulation of the Cd Absorption, Subcellular Distribution, and Chemical Form. Int. J. Environ. Res. Public Health. 2020, 17, 8143. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Zhou, Y.; Yang, Z.; Lin, B.; Yuan, J.; Wu, S. Comparisons in Subcellular and Biochemical Behaviors of Cadmium between Low-Cd and High-Cd Accumulation Cultivars of Pakchoi (Brassica chinensis L.). Front. Environ. Sci. Eng. 2014, 8, 226–238. [Google Scholar] [CrossRef]
- Mendoza-Cozatl, D.G.; Moreno-Sanchez, R. Control of Glutathione and Phytochelatin Synthesis under Cadmium Stress. Pathway Modeling for Plants. J. Theor. Biol. 2006, 238, 919–936. [Google Scholar] [CrossRef] [PubMed]
- Hou, M.; Li, M.; Yang, X.; Pan, R. Responses of Nonprotein Thiols to Stress of Vanadium and Mercury in Maize (Zea mays L.) Seedlings. Bull. Environ. Contam. Toxicol. 2019, 102, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.L. Cellular Mechanisms for Heavy Metal Detoxification and Tolerance. J. Exp. Bot. 2002, 53, 1–11. [Google Scholar] [CrossRef]
- Fu, X.; Dou, C.; Chen, Y.; Chen, X.; Shi, J.; Yu, M.; Xu, J. Subcellular Distribution and Chemical Forms of Cadmium in Phytolacca americana L. J. Hazard. Mater. 2011, 186, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Vatehova-Vivodova, Z.; Kollarova, K.; Malovikova, A.; Liskova, D. Maize Shoot Cell Walls under Cadmium Stress. Environ. Sci. Pollut. Res. 2018, 25, 22318–22322. [Google Scholar] [CrossRef]
- Wu, X.; Song, H.; Guan, C.; Zhang, Z. Boron Alleviates Cadmium Toxicity in Brassica napus by Promoting the Chelation of Cadmium onto the Root Cell Wall Components. Sci. Total Environ. 2020, 728, 138833. [Google Scholar] [CrossRef]
- Singh, A.K.; Dhanapal, S.; Yadav, B.S. The Dynamic Responses of Plant Physiology and Metabolism during Environmental Stress Progression. Mol. Biol. Rep. 2020, 47, 1459–1470. [Google Scholar] [CrossRef]
- Niemeyer, H.M. Hydroxamic Acids (4-Hydroxy-1,4-benzoxazin-3- ones), Defence Chemicals in the Gramineae. Phytochemistry 1988, 27, 3349–3358. [Google Scholar] [CrossRef]
- Rollo, D.; Borden, J.H.; Casey, I.B. Endogenously Produced Repellent from American Cockroach (Blattaria: Blattidae): Function in Death Recognition. Environ. Entomol. 1995, 24, 116–124. [Google Scholar] [CrossRef]
- Cuevas, L.; Niemeyer, H.M.; Jonsson, L.M.V. Partial Purification and Characterization of a Hydroxamic Acid Glucoside β-d-glucosidase from Maize. Phytochemistry 1992, 31, 2609–2612. [Google Scholar] [CrossRef]
- Hofman, J.; Hofmanova, O. 1, 4-Benzoxazine Derivatives in Plants. Eur. J. Biochem. 1969, 8, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Tufarelli, V.; Casalino, E.; D’Alessandro, A.G.; Laudadio, V. Dietary Phenolic Compounds: Biochemistry, Metabolism and Significance in Animal and Human Health. Curr. Drug Metab. 2017, 18, 905–913. [Google Scholar] [CrossRef] [PubMed]
- Tusevski, O.; Stanoeva, J.P.; Stefova, M.; Spasenoski, M.; Simic, S. State of Antioxidant Systems and Phenolic Compounds’ Production in Hypericum perforatum L. Hairy Roots. Acta Physiol. Plant. 2019, 41, 132. [Google Scholar] [CrossRef]
- Wang, C.; Lu, J.; Zhang, S.; Wang, P.; Hou, J.; Qian, J. Effects of Pb Stress on Nutrient Uptake and Secondary Metabolism in Submerged Macrophyte Vallisneria natans. Ecotox. Environ. Safety 2011, 74, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).