Multi-Functional Ethylene-vinyl Acetate Copolymer Flexible Composite Film Embedded with Indium Acetate-Passivated Perovskite Quantum Dots
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
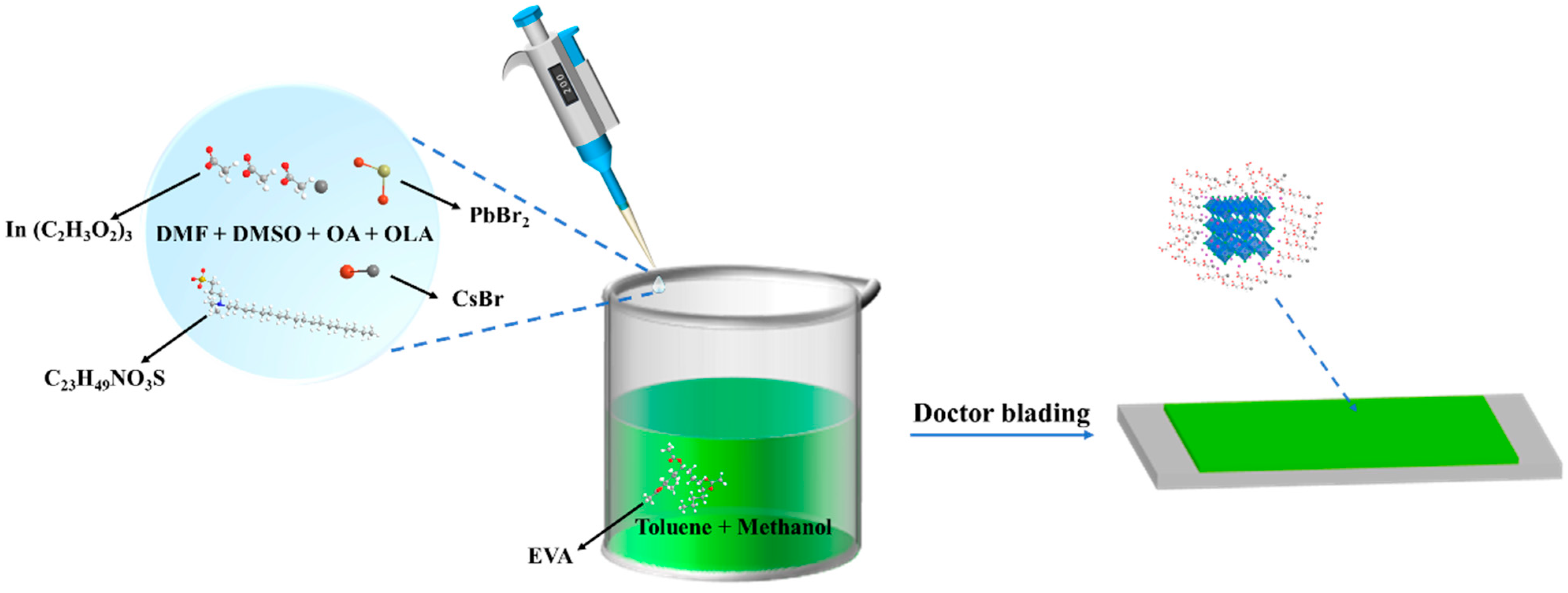
2.2. Preparation of Precursor and EVA Solution
2.3. Synthesis of CsPbBr3-In/EVA Film
2.4. Material Characterization
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guo, H.; Pei, Y.; Zhang, J.; Cai, C.; Zhou, K.; Zhu, Y. Doping with SnBr2 in CsPbBr3 to enhance the efficiency of all-inorganic perovskite solar cells. J. Mater. Chem. C 2019, 7, 11234–11243. [Google Scholar] [CrossRef]
- Yang, L.; Fu, B.; Li, X.; Chen, H.; Li, L. Poly (vinylidene fluoride)-passivated CsPbBr3 perovskite quantum dots with near-unity photoluminescence quantum yield and superior stability. J. Mater. Chem. C 2021, 9, 1983–1991. [Google Scholar] [CrossRef]
- Wang, S.; Feng, S.; Li, R.; Jin, J.; Wu, J.; Zheng, W.; Xia, Z.; Chen, X.; Ling, Q.; Lin, Z. Multiexciton Generation from a 2D Organic–Inorganic Hybrid Perovskite with Nearly 200% Quantum Yield of Red Phosphorescence. Adv. Mater. 2023, 35, 2211992. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Wang, S.; Chen, G.; Chen, R.; Chen, S.; Jiang, N.; Huang, F.; Zheng, Y.; Zheng, Y.; Chen, D. Ultra-stable narrowband green-emitting CsPbBr3 quantum dot-embedded glass ceramics for wide color gamut backlit displays. J. Mater. Chem. C 2022, 10, 7263–7272. [Google Scholar] [CrossRef]
- Zhumekenov, A.A.; Saidaminov, M.I.; Haque, M.A.; Alarousu, E.; Sarmah, S.P.; Murali, B.; Dursun, I.; Miao, X.; Abdelhady, A.L.; Wu, T.; et al. Formamidinium Lead Halide Perovskite Crystals with Unprecedented Long Carrier Dynamics and Diffusion Length. ACS Energy Lett. 2016, 1, 32–37. [Google Scholar] [CrossRef]
- Chen, Z.; Zhao, J.; Zeng, R.; Liu, X.; Zou, B.; Xiang, W. High efficiency fluorescent perovskite quantum dots encapsulated in superhydrophobic silica aerogel for wide color gamut backlight displays. Chem. Eng. J. 2022, 433, 133195. [Google Scholar] [CrossRef]
- Tao, C.; Wei, Y.; Zhang, J.; Cao, Y.; Wang, S.; Xu, L.; Wen, K.; Wang, J.; Kuang, Z.; Wang, X.; et al. Indirect Bandgap Emission of the Metal Halide Perovskite FAPbI3 at Low Temperatures. J. Phys. Chem. Lett. 2023, 14, 3805–3810. [Google Scholar] [CrossRef]
- Xuan, T.; Guo, S.; Bai, W.; Zhou, T.; Wang, L.; Xie, R. Ultrastable and highly efficient green-emitting perovskite quantum dot composites for Mini-LED displays or backlights. Nano Energy 2022, 95, 107003. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, T.; Ma, Z.; Yuan, F.; Zhou, X.; Wang, H.; Liu, Z.; Qing, J.; Chen, H.; Li, X.; et al. A Multifunctional “Halide-Equivalent” Anion Enabling Efficient CsPb(Br/I)3 Nanocrystals Pure-Red Light-Emitting Diodes with External Quantum Efficiency Exceeding 23%. Adv. Mater. 2023, 35, 2209002. [Google Scholar] [CrossRef]
- Yu, M.; Ji, Y.; Yan, H.; Hu, J.; Li, S.; Xu, H.; Chen, P.; Zhao, L.; Guan, S.; Bi, X.; et al. High-Radiance Near-Infrared Perovskite Light-Emitting Diodes with Improved Roll-Off Degradation. Adv. Opt. Mater. 2023, 11, 2202043. [Google Scholar] [CrossRef]
- Su, B.; Jin, J.; Han, K.; Xia, Z. Ceramic Wafer Scintillation Screen by Utilizing Near-Unity Blue-Emitting Lead-Free Metal Halide (C8H20N)2Cu2Br4. Adv. Funct. Mater. 2023, 33, 2210735. [Google Scholar] [CrossRef]
- Zou, L.; Li, X.; Yang, M.; Yan, J.; Wang, J.; Cheng, J.; Xing, J.; Liu, H.; Hao, H.; Dong, J. ZnPc/CsPbBr3 QDs collaborative interface modification to improve the performance of CsPbBr3 perovskite solar cells. Sol. Energy Mater. Sol. Cells 2023, 251, 112157. [Google Scholar] [CrossRef]
- Song, J.; Ren, Y.; Gong, S.; Zhao, L.; Xuan, W.; Zhu, L.; Zhao, Y.; Qiang, H.; Gao, L.; Huang, S. Performance Enhancement of Crystal Silicon Solar Cell by a CsPbBr3-Cs4PbBr6 Perovskite Quantum Dot@ZnO/Ethylene Vinyl Acetate Copolymer Downshifting Composite Film. Sol. RRL 2022, 6, 2200336. [Google Scholar] [CrossRef]
- Wang, Q.; Fu, R.; Sun, T.; Liu, M.; Sun, S.; Jiang, H.; Li, Z.; Zhang, Y.; Liu, D.; Chen, Y.; et al. Continuously in-situ manufacture of perovskite quantum dots/POE encapsulation adhesive film for silicon solar cell enhancement application. Sol. Energy Mater. Sol. Cells 2023, 259, 112450. [Google Scholar] [CrossRef]
- Xuan, W.; Shan, H.; Hu, D.; Zhu, L.; Guan, T.; Zhao, Y.; Qiang, Y.; Song, J.; Zhang, J.; Sui, M.; et al. In-situ synthesis of stable ZnO-coated CsPbBr3 nanocrystals for room-temperature heptanal sensors. Mater. Today Chem. 2022, 26, 101155. [Google Scholar] [CrossRef]
- Xuan, W.; Zheng, L.; Cao, L.; Miao, S.; Hu, D.; Zhu, L.; Zhao, Y.; Qiang, Y.; Gu, X.; Huang, S. Machine Learning-Assisted Sensor Based on CsPbBr3@ZnO Nanocrystals for Identifying Methanol in Mixed Environments. ACS Sens. 2023, 8, 1252–1260. [Google Scholar] [CrossRef]
- Shan, H.; Xuan, W.; Li, Z.; Hu, D.; Gu, X.; Huang, S. Room-Temperature Hydrogen Sulfide Sensor Based on Tributyltin Oxide Functionalized Perovskite CsPbBr3 Quantum Dots. ACS Appl. Nano Mater. 2022, 5, 6801–6809. [Google Scholar] [CrossRef]
- Zhang, J.; Jiao, B.; Dai, J.; Wu, D.; Wu, Z.; Bian, L.; Zhao, Y.; Yang, W.; Jiang, M.; Lu, S. Enhance the responsivity and response speed of self-powered ultraviolet photodetector by GaN/CsPbBr3 core-shell nanowire heterojunction and hydrogel. Nano Energy 2022, 100, 107437. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Y.; Xu, Z.; Ye, H.; Yang, Z.; You, J.; Liu, M.; He, Y.; Kanatzidis, M.G.; Liu, S. Nucleation-controlled growth of superior lead-free perovskite Cs3Bi2I9 single-crystals for high-performance X-ray detection. Nat. Commun. 2020, 11, 2304. [Google Scholar] [CrossRef]
- Xue, J.; Zhu, Z.; Xu, X.; Gu, Y.; Wang, S.; Xu, L.; Zou, Y.; Song, J.; Zeng, H.; Chen, Q. Narrowband perovskite photodetector-based image array for potential application in artificial vision. Nano Lett. 2018, 18, 7628–7634. [Google Scholar] [CrossRef]
- Yin, G.; Qi, X.; Chen, Y.; Peng, Q.; Jiang, X.; Wang, Q.; Zhang, W.; Gong, X. Constructing an all zero-dimensional CsPbBr3/CdSe heterojunction for highly efficient photocatalytic CO2 reduction. J. Mater. Chem. A 2022, 10, 22468–22476. [Google Scholar] [CrossRef]
- Zhang, Q.; He, M.; Wan, Q.; Zheng, W.; Liu, M.; Zhang, C.; Liao, X.; Zhan, W.; Kong, L.; Guo, X.; et al. Suppressing thermal quenching of lead halide perovskite nanocrystals by constructing a wide-bandgap surface layer for achieving thermally stable white light-emitting diodes. Chem. Sci. 2022, 13, 3719–3727. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, X.; Xue, W.; Wang, W.; Zhu, W.; Zhao, L. Highly luminescent and stable CsPbBr3 perovskite quantum dots modified by phosphine ligands. Nano Res. 2019, 12, 785–789. [Google Scholar] [CrossRef]
- Ye, S.; Huang, M.; Han, Q.; Song, J.; Qu, J. Significantly enhanced thermal and water stability of heterostructured CsPbBr3/Cs4PbBr6@PbS nanocrystals. J. Alloys Compd. 2023, 965, 171442. [Google Scholar] [CrossRef]
- Xie, Q.; Wu, D.; Wang, X.; Li, Y.; Fang, F.; Wang, Z.; Ma, Y.; Su, M.; Peng, S.; Liu, H.; et al. Branched capping ligands improve the stability of cesium lead halide (CsPbBr3) perovskite quantum dots. J. Mater. Chem. C 2019, 7, 11251–11257. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, H.; Fang, Z.; Yi, J.; Song, P.; Yu, X.; Zhou, D.; Qiu, J.; Xu, X. One-step precipitated all-inorganic perovskite QDs from amorphous media for backlighting display and reproducible laser-driven white lighting. Chem. Eng. J. 2022, 427, 131379. [Google Scholar] [CrossRef]
- Chen, D.; Chen, X. Luminescent perovskite quantum dots: Synthesis, microstructures, optical properties and applications. J. Mater. Chem. C 2019, 7, 1413–1446. [Google Scholar] [CrossRef]
- Feng, X.; Liu, J.; Zhao, X.; Xu, P.; Liu, J. Surface regulation by bifunctional BODIPY to fabricate stable CsPbBr3 for multi-layered optical anti-counterfeiting. J. Colloid Interface Sci. 2023, 629, 63–72. [Google Scholar] [CrossRef]
- Li, Y.; Cai, M.; Shen, M.; Cai, Y.; Xie, R. Bidentate aliphatic quaternary ammonium ligand-stabilized CsPbBr3 perovskite nanocrystals with high PLQY (92.3%) and superior stability. J. Mater. Chem. C 2022, 10, 8356–8363. [Google Scholar] [CrossRef]
- Grisorio, R.; Fasulo, F.; Muñoz-García, A.B.; Pavone, M.; Conelli, D.; Fanizza, E.; Striccoli, M.; Allegretta, I.; Terzano, R.; Margiotta, N.; et al. In situ formation of zwitterionic ligands: Changing the passivation paradigms of CsPbBr3 nanocrystals. Nano Lett. 2022, 22, 4437–4444. [Google Scholar] [CrossRef]
- Lu, C.H.; Biesold-McGee, G.V.; Liu, Y.; Kang, Z.; Lin, Z. Doping and ion substitution in colloidal metal halide perovskite nanocrystals. Chem. Soc. Rev. 2020, 49, 4953–5007. [Google Scholar] [CrossRef]
- Yang, D.; Li, X.; Wu, Y.; Wei, C.; Qin, Z.; Zhang, C.; Sun, Z.; Li, Y.; Wang, Y.; Zeng, H. Surface halogen compensation for robust performance enhancements of CsPbX3 perovskite quantum dots. Adv. Opt. Mater. 2019, 7, 1900276. [Google Scholar] [CrossRef]
- Wang, C.; Huang, H.; Weng, B.; Verhaeghe, D.; Keshavarz, M.; Jin, H.; Liu, B.; Xie, H.; Ding, Y.; Gao, Y.; et al. Planar heterojunction boosts solar-driven photocatalytic performance and stability of halide perovskite solar photocatalyst cell. Appl. Catal. B 2022, 301, 120760. [Google Scholar] [CrossRef]
- Liu, G.; Li, Z.; Lai, Z.; Qiu, Z.; Yang, M.; Wu, X.; Zeng, Z.; Deng, S.; Yang, B. Flexible, Stretchable, and Luminescent Hydrogels Based on a Polydimethylsiloxane-Coated CsPbBr3 Nanostructure for Elastomers. ACS Appl. Nano Mater. 2023, 11, 9588–9597. [Google Scholar] [CrossRef]
- Shi, J.; Wang, M.; Wang, H.; Zhang, C.; Ji, Y.; Wang, J.; Zhou, Y.; Bhatti, A.S. Preparation of ultra-stable and environmentally friendly CsPbBr3@ZrO2/PS composite films for white light-emitting diodes. Nanoscale 2022, 14, 16548–16559. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Wang, L.; Tong, P.; Zhang, Y.; Jiang, X.; Mao, H.; Xu, T.; Fan, B.; Shao, G.; Liu, W.; et al. Stability study of all-inorganic perovskite CsPbBr3 QDs@SiO2/EVA film composites prepared by one-step. J. Cryst. Growth 2021, 560, 126045. [Google Scholar] [CrossRef]
- Shi, J.; Ge, W.; Gao, W.; Xu, M.; Zhu, J.; Li, Y. Enhanced thermal stability of halide perovskite CsPbX3 nanocrystals by a facile TPU encapsulation. Adv. Opt. Mater. 2020, 8, 1901516. [Google Scholar] [CrossRef]
- Lai, W.; Wu, C.; Han, X. Facile synthesis of hyperbranched Eu-MOF structures for the construction of a CsPbBr3/Eu-MOF composite and its application as a ratiometric fluorescent probe. J. Mater. Chem. C 2023, 11, 2995–3002. [Google Scholar] [CrossRef]
- Pan, J.; Shang, Y.; Yin, J.; Bastiani, M.D.; Peng, W.; Dursun, I.; Sinatra, L.; EI-Zohry, A.M.; Hedhili, M.N.; Emwas, A.; et al. Bidentate ligand-passivated CsPbI3 perovskite nanocrystals for stable near-unity photoluminescence quantum yield and efficient red light-emitting diodes. J. Am. Chem. Soc. 2017, 140, 562–565. [Google Scholar] [CrossRef]
- Wang, H.; Lu, W.; Xu, P.; Luo, J.; Yao, K.; Zhang, J.; Wei, X.; Peng, S.; Cheng, H.; Hu, H.; et al. Highly Stable CsPbBr3 Nanocrystals for Photocatalytic Reduction of CO2: Ionic Liquid as a Surface Passivation Ligand and Reaction Precursor. ACS Sustain. Chem. Eng. 2023, 11, 5963–5972. [Google Scholar] [CrossRef]
- Li, L.; Zhang, Z.; Chen, Y.; Xu, Q.; Zhang, J.; Chen, Z.; Chen, Y.; Zhu, J. Sustainable and Self-enhanced Electrochemiluminescent Ternary Suprastructures Derived from CsPbBr3 Perovskite Quantum Dots. Adv. Funct. Mater. 2019, 29, 1902533. [Google Scholar] [CrossRef]
- Yuan, X.; Hou, X.; Li, J.; Qu, C.; Zhang, W.; Zhao, J.; Li, H. Thermal degradation of luminescence in inorganic perovskite CsPbBr3 nanocrystals. Phys. Chem. Chem. Phys. 2017, 19, 8934–8940. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Wang, M.; Yang, Z.; Qiu, H.; Ji, S.; Dou, J.; Gaponenko, N.V. Highly stable Na: CsPb(Br, I)3@Al2O3 nanocomposites prepared by a pre-protection strategy. Nanoscale 2020, 12, 6403–6410. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Li, J.; Lu, Z.; Zhang, H.; Li, W.; Zhuang, J.; Hu, C.; Liu, Y.; Lei, B.; Zhang, X. Modulating the local structure of glass to promote in situ precipitation of perovskite CsPbBr3 quantum dots by introducing a network modifier. J. Mater. Chem. C 2022, 10, 8634–8641. [Google Scholar] [CrossRef]
- Woo, J.Y.; Kim, Y.; Bae, J.; Kim, T.G.; Kim, J.W.; Lee, D.C.; Jeong, S. Highly Stable Cesium Lead Halide Perovskite Nanocrystals through in Situ Lead Halide Inorganic Passivation. Chem. Mater. 2017, 29, 7088–7092. [Google Scholar] [CrossRef]
- Xie, G.; Lu, X.; Duan, J.; Dong, Y.; Jiang, X.; Tu, F.; Duan, Y.; Tang, Q. Alkali chloride doped SnO2 electron-transporting layers for boosting charge transfer and passivating defects in all-inorganic CsPbBr3 perovskite solar cells. J. Mater. Chem. A 2021, 9, 15003–15011. [Google Scholar] [CrossRef]
- Liu, X.; Tan, X.; Liu, Z.; Zhang, X.; Shi, T.; Liao, G. Enhancing the performance of all vapor-deposited electron-conductor-free CsPbBr3 photodetectors via interface engineering for their applications in image sensing. J. Mater. Chem. C 2020, 8, 14409–14422. [Google Scholar] [CrossRef]
- Li, Y.; Lv, Y.; Guo, Z.; Dong, L.; Zheng, J.; Chai, C.; Chen, N.; Lu, Y.; Chen, C. One-Step Preparation of Long-Term Stable and Flexible CsPbBr3 Perovskite Quantum Dots/Ethylene Vinyl Acetate Copolymer Composite Films for White Light-Emitting Diodes. ACS Appl. Mater. Interfaces 2018, 10, 15888–15894. [Google Scholar] [CrossRef]
- Law, A.M.; Bukhari, F.; Jones, L.O.; Isherwood, P.J.M.; Walls, J.M. Multilayer antireflection coatings for cover glass on silicon solar modules. IEEE J. Photovolt. 2022, 12, 1205–1210. [Google Scholar] [CrossRef]







| Sample | τ1 (ns) | A1 | τ2 (ns) | A2 | τA (ns) |
|---|---|---|---|---|---|
| 0 mmol In | 9.37 | 0.71 | 36.41 | 0.29 | 25.96 |
| 0.01 mmol In | 6.85 | 0.62 | 27.07 | 0.38 | 21.16 |
| 0.02 mmol In | 6.58 | 0.67 | 24.57 | 0.33 | 18.23 |
| 0.03 mmol In | 7.30 | 0.70 | 24.11 | 0.30 | 17.15 |
| 0.04 mmol In | 6.03 | 0.69 | 20.13 | 0.31 | 14.49 |
| 0.05 mmol In | 5.98 | 0.71 | 19.43 | 0.29 | 13.65 |
| Figure | Sample | Voc (V) | JSC (mA/cm2) | Factor | Efficiency | ΔE |
|---|---|---|---|---|---|---|
| a | Solar cell | 0.61296384 | 32.15262214 | 66.9669 | 13.1981 | 0.0975 |
| EVA | 0.61512954 | 32.21299025 | 67.0983 | 13.2956 | ||
| b | Solar cell | 0.61570937 | 32.69598568 | 68.3807 | 13.7659 | 0.1352 |
| CsPbBr3/EVA | 0.61768150 | 32.71814702 | 68.7855 | 13.9011 | ||
| c | Solar cell | 0.61018635 | 31.92773015 | 68.1391 | 13.2748 | 0.3111 |
| CsPbBr3-In/EVA | 0.61358988 | 32.21761684 | 68.7256 | 13.5859 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, S.; Gao, S.; Zhang, H.; Bian, C.; Zhao, Y.; Gu, X.; Xu, W. Multi-Functional Ethylene-vinyl Acetate Copolymer Flexible Composite Film Embedded with Indium Acetate-Passivated Perovskite Quantum Dots. Polymers 2023, 15, 3986. https://doi.org/10.3390/polym15193986
Huang S, Gao S, Zhang H, Bian C, Zhao Y, Gu X, Xu W. Multi-Functional Ethylene-vinyl Acetate Copolymer Flexible Composite Film Embedded with Indium Acetate-Passivated Perovskite Quantum Dots. Polymers. 2023; 15(19):3986. https://doi.org/10.3390/polym15193986
Chicago/Turabian StyleHuang, Sheng, Shasha Gao, Hui Zhang, Ce Bian, Yulong Zhao, Xiuquan Gu, and Wenjie Xu. 2023. "Multi-Functional Ethylene-vinyl Acetate Copolymer Flexible Composite Film Embedded with Indium Acetate-Passivated Perovskite Quantum Dots" Polymers 15, no. 19: 3986. https://doi.org/10.3390/polym15193986
APA StyleHuang, S., Gao, S., Zhang, H., Bian, C., Zhao, Y., Gu, X., & Xu, W. (2023). Multi-Functional Ethylene-vinyl Acetate Copolymer Flexible Composite Film Embedded with Indium Acetate-Passivated Perovskite Quantum Dots. Polymers, 15(19), 3986. https://doi.org/10.3390/polym15193986






