Bio-Based Healable 2K PU Textile Coating for Durable Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Schiff Base
2.3. Bio-Based Healable 2K PU Coating
2.4. Characterisation
3. Results and Discussion
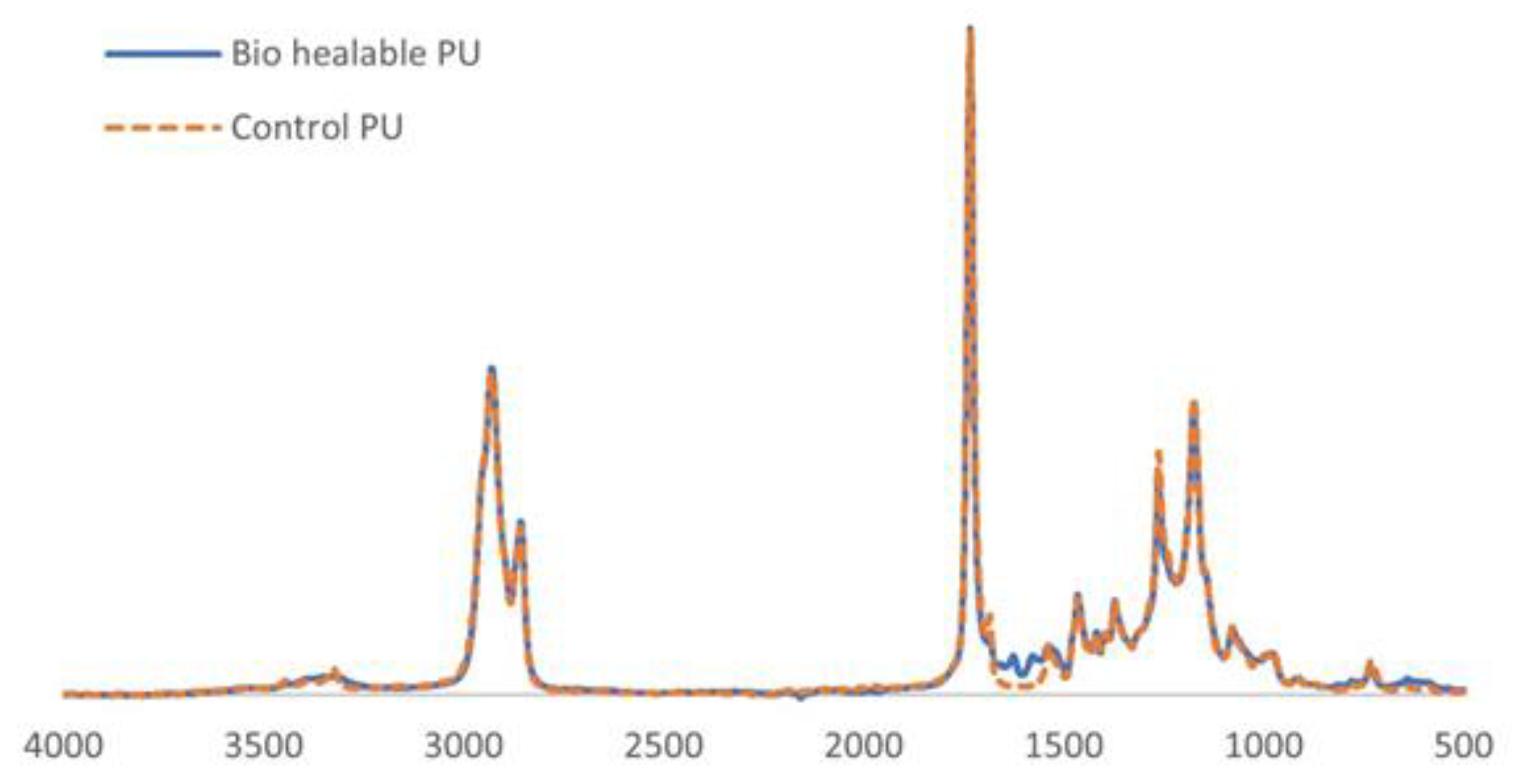
3.1. Structural Characterization of 2K PU Coatings
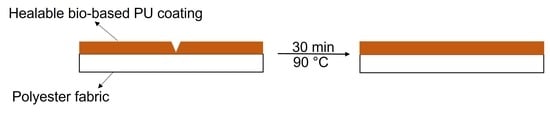
3.2. Scratch Healing
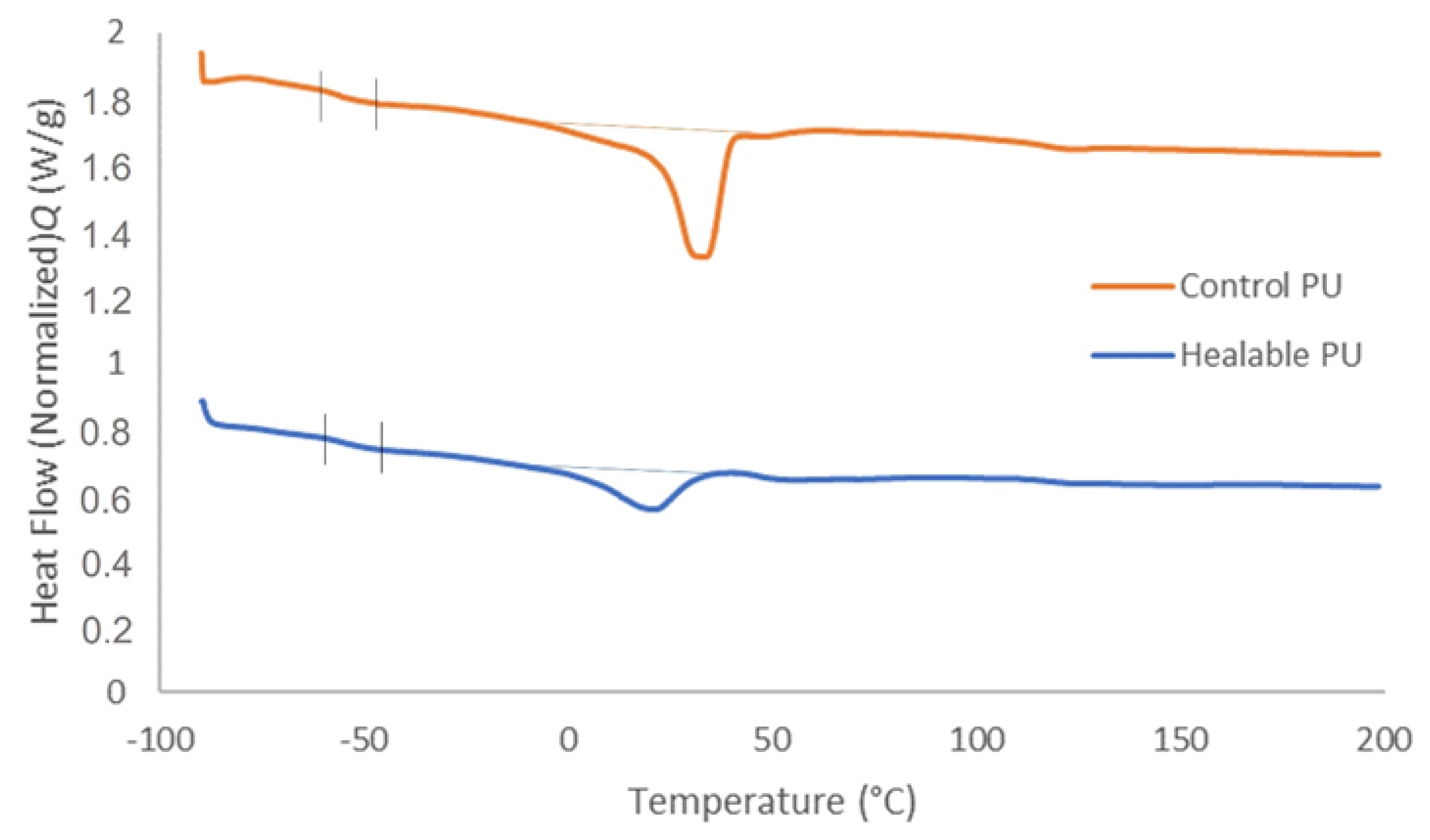
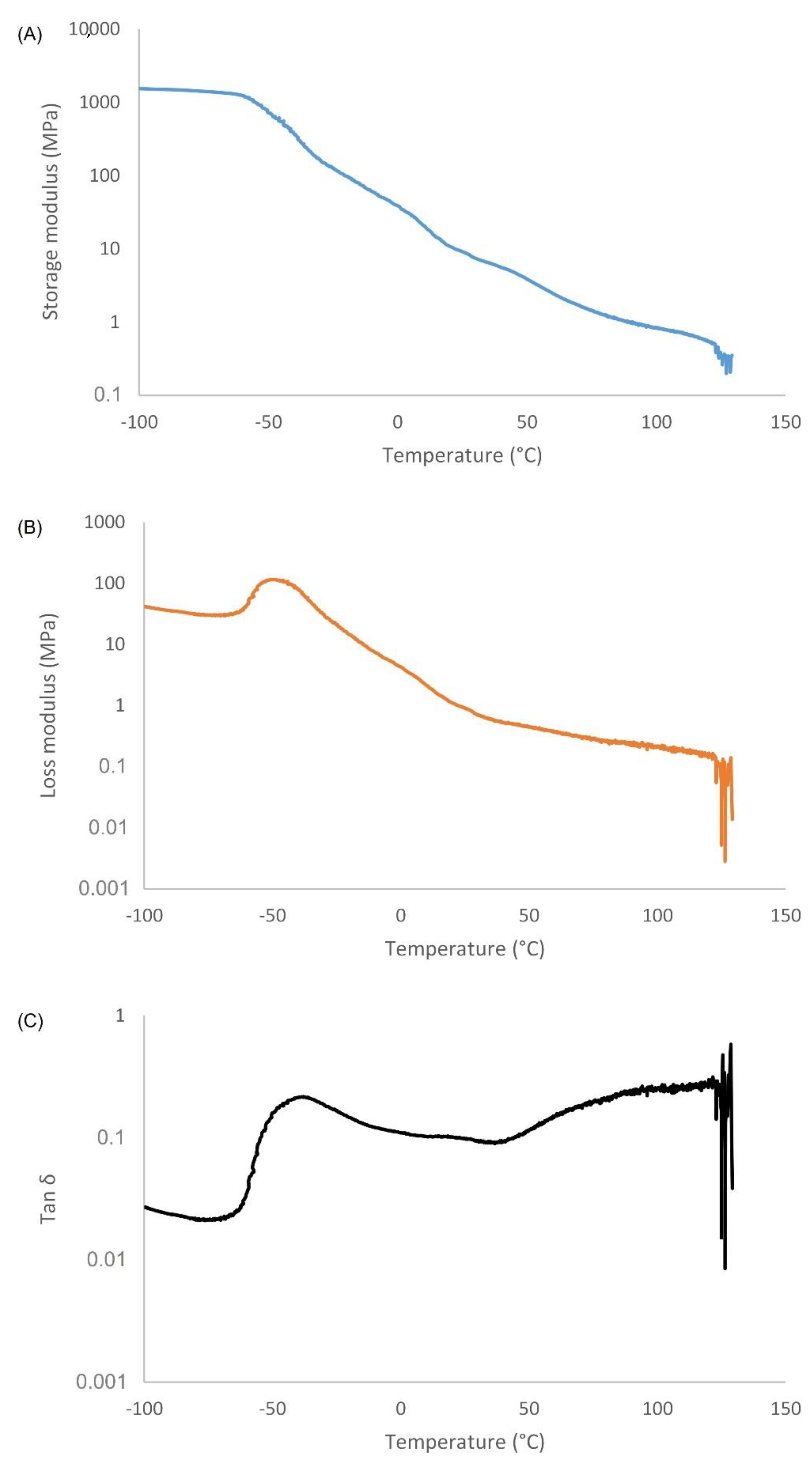
3.3. Thermal Behavior
3.4. Performance of the Bio-Based 2K PU Coating
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, X.; Li, Y.; Fang, X.; Zhang, Z.; Li, S.; Sun, J. Healable and Recyclable Polymeric Materials with High Mechanical Robustness. ACS Mater. Lett. 2022, 4, 554–571. [Google Scholar] [CrossRef]
- Lee, W.-J.; Oh, H.-G.; Cha, S.-H. A Brief Review of Self-Healing Polyurethane Based on Dynamic Chemistry. Macromol. Res. 2021, 29, 649–664. [Google Scholar] [CrossRef]
- Ghosh, S.K. Self-Healing Materials: Fundamentals, Design Strategies, and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Hillewaere, X.K.; Du Prez, F.E. Fifteen chemistries for autonomous external self-healing polymers and composites. Prog. Polym. Sci. 2015, 49–50, 121–153. [Google Scholar] [CrossRef]
- Toohey, K.S.; Sottos, N.R.; Lewis, J.A.; Moore, J.; White, S. Self-healing materials with microvascular networks. Nat. Mater. 2007, 6, 581–585. [Google Scholar] [CrossRef] [PubMed]
- Heyse, P.; De Vilder, I.; Vanneste, M. Smart durable and self-healing textile coatings. In Active Coatings for Smart Textiles, Woodhead Publishing Series in Textiles; Woodhead Publishing: Duxford, UK, 2016; pp. 55–80. [Google Scholar]
- Chen, J.; Li, F.; Luo, Y.; Shi, Y.; Ma, X.; Zhang, M.; Boukhvalov, D.W.; Luo, Z. A self-healing elastomer based on an intrinsic non-covalent cross-linking mechanism. J. Mater. Chem. A 2019, 7, 15207–15214. [Google Scholar] [CrossRef]
- Xu, S.; Sheng, D.; Zhou, Y.; Wu, H.; Xie, H.; Liu, X.; Yang, Y. A robust and healable polyurethane based on coordination bonds. Polym. Int. 2020, 69, 876–882. [Google Scholar] [CrossRef]
- Feula, A.; Pethybridge, A.; Giannakopoulos, I.; Tang, X.; Chippindale, A.; Siviour, C.R.; Buckley, C.P.; Hamley, I.W.; Hayes, W. A Thermoreversible Supramolecular Polyurethane with Excellent Healing Ability at 45 °C. Macromolecules 2015, 48, 6132–6141. [Google Scholar] [CrossRef]
- Chen, S.; Mo, F.; Yang, Y.; Stadler, F.J.; Chen, S.; Yang, H.; Ge, Z. Development of zwitterionic polyurethanes with multi-shape memory effects and self-healing properties. J. Mater. Chem. A 2014, 3, 2924–2933. [Google Scholar] [CrossRef]
- Sugane, K.; Shibata, M. Self-healing thermoset polyurethanes utilizing host–guest interaction of cyclodextrin and adamantane. Polymer 2021, 221, 123629. [Google Scholar] [CrossRef]
- Chakma, P.; Konkolewicz, D. Dynamic Covalent Bonds in Polymeric Materials. Angew. Chem. Int. Ed. 2019, 58, 9682–9695. [Google Scholar] [CrossRef]
- Hayashi, M. Implantation of Recyclability and Healability into Cross-Linked Commercial Polymers by Applying the Vitrimer Concept. Polymers 2020, 12, 1322. [Google Scholar] [CrossRef] [PubMed]
- Montarnal, D.; Capelot, M.; Tournilhac, F.; Leibler, L. Silica-Like Malleable Materials from Permanent Organic Networks. Science 2011, 334, 965–968. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Jin, B.; Wang, H.; Wu, W.; Cao, Z.; Wu, J.; Huang, G. A Degradable and Self-Healable Vitrimer Based on Non-isocyanate Polyurethane. Front. Chem. 2020, 8, 585569. [Google Scholar] [CrossRef]
- Denissen, W.; Rivero, G.; Nicolaÿ, R.; Leibler, L.; Winne, J.; Du Prez, F. Vinylogous Urethane Vitrimers. Adv. Funct. Mater. 2015, 15, 2451–2457. [Google Scholar] [CrossRef]
- Lian, W.; Han, H.; Zhang, X.; Peng, G.; Jia, Z.; Zhang, Z. Polyurethane modified epoxy vitrimer and its stress relaxation behavior. J. Polym. Eng. 2021, 41, 365–374. [Google Scholar] [CrossRef]
- Wu, P.; Cheng, H.; Wang, X.; Shi, R.; Zhang, C.; Arai, M.; Zhao, F. A self-healing and recyclable polyurethane-urea Diels–Alder adduct synthesized from carbon dioxide and furfuryl amine. Green Chem. 2020, 23, 552–560. [Google Scholar] [CrossRef]
- Farshchi, N.; Gedan-Smolka, M.; Stommel, M. Preparation and Characterization of Self-Healing Polyurethane Powder Coating Using Diels–Alder Reaction. Polymers 2021, 13, 3803. [Google Scholar] [CrossRef]
- Behera, P.K.; Raut, S.K.; Mondal, P.; Sarkar, S.; Singha, N.K. Self-Healable Polyurethane Elastomer Based on Dual Dynamic Covalent Chemistry Using Diels–Alder “Click” and Disulfide Metathesis Reactions. ACS Appl. Polym. Mater. 2021, 3, 847–856. [Google Scholar] [CrossRef]
- Lee, S.-H.; Shin, S.-R.; Lee, D.-S. Self-healing of cross-linked PU via dual-dynamic covalent bonds of a Schiff base from cystine and vanillin. Mater. Des. 2019, 172, 107774. [Google Scholar] [CrossRef]
- Chang, K.; Jia, H.; Gu, S.-Y. A transparent, highly stretchable, self-healing polyurethane based on disulfide bonds. Eur. Polym. J. 2019, 112, 822–831. [Google Scholar] [CrossRef]
- Ye, G.; Jiang, T. Preparation and Properties of Self-Healing Waterborne Polyurethane Based on Dynamic Disulfide Bond. Polymers 2021, 13, 2936. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Kim, S.; Lee, D. Synthesis and Characterization of Healable Waterborne Polyurethanes with Cystamine Chain Ex-tenders. Molecules 2019, 24, 1492. [Google Scholar] [CrossRef]
- Ji, S.; Cao, W.; Yu, Y.; Xu, H. Visible-Light-Induced Self-Healing Diselenide-Containing Polyurethane Elastomer. Adv. Mater. 2015, 27, 7740–7745. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Aguirresarobe, R.H.; Irusta, L.; Ruipérez, F.; Matxain, J.M.; Pan, X.; Aramburu, N.; Mecerreyes, D.; Sardon, H.; Zhu, J. Aromatic diselenide crosslinkers to enhance the reprocessability and self-healing of polyurethane thermosets. Polym. Chem. 2017, 8, 3641–3646. [Google Scholar] [CrossRef]
- Delpierre, S.; Willocq, B.; Manini, G.; Lemaur, V.; Goole, J.; Gerbaux, P.; Cornil, J.; Dubois, P.; Raquez, J.-M. Simple Approach for a Self-Healable and Stiff Polymer Network from Iminoboronate-Based Boroxine Chemistry. Chem. Mater. 2019, 31, 3736–3744. [Google Scholar] [CrossRef]
- De Smet, D.; Uyttendaele, W.; Vanneste, M. Bio-Based 2K PU Coating for Durable Textile Applications. Coatings 2022, 12, 169. [Google Scholar] [CrossRef]
- Wilhelm, C.; Gardette, J.-L. Infrared analysis of the photochemical behaviour of segmented polyurethanes: 1. Aliphatic poly(ester-urethane). Polymer 1997, 38, 4019–4031. [Google Scholar] [CrossRef]
- Irusta, L.; Fernanadez-Berridi, J. Aromatic poly(ester-urethanes): Effect of the polyol molecular weight on the photochemical behavior. Polymer 2000, 41, 3297–3302. [Google Scholar] [CrossRef]
- Coates, J. Interpretation of infrared spectra: A practical approach. In Encyclopedia of Analytical Chemistry; Mayers, R., Ed.; Wiley: Hoboken, NJ, USA, 2000; pp. 10815–10837. [Google Scholar]
- Kaushiva, B.; McCartney, S.; Rossmy, G.; Wilkes, G. Surfactant level influences on structure and properties of flexible slabstock polyurethane foams. Polymer 2000, 41, 285–310. [Google Scholar] [CrossRef]
- Aguirresarobe, R.H.; Nevejans, S.; Reck, B.; Irusta, L.; Sardon, H.; Asua, J.M.; Ballard, N. Healable and self-healing polyurethanes using dynamic chemistry. Prog. Polym. Sci. 2021, 114, 101362. [Google Scholar] [CrossRef]
- Anand, A.; Kulkarni, R.; Gite, V. Preparation and properties of eco-friendly two pack PU coatings based on renewable source (sorbitol) and its property improvement by nano ZnO. Prog. Org. Coat. 2012, 74, 764–767. [Google Scholar] [CrossRef]
- Ahmad, S.; Zafar, F.; Sharmin, E.; Garg, N.; Kashif, M. Synthesis and characterization of corrosion protective polyurethane fatty amide/silica hybrid coating material. Prog. Org. Coat. 2012, 201273, 112–117. [Google Scholar] [CrossRef]
- Marathe, R.; Tatiya, P.; Chaudhari, A.; Lee, J.; Mahulikar, P.; Sohn, D.; Gite, V. Neem acetylated polyester polyol—Renewable source based smart PU coatings containing quinoline (corrosion inhibitor) encapsulated polyurea microcapsules for enhance anticorrosive property. Ind. Crop. Prod. 2015, 77, 239–250. [Google Scholar] [CrossRef]
- Saleh, T.A. Polymer science and polymerization methods toward hybrid materials. In Polymer Hybrid Materials and Nano-Composites; Elsevier: Amsterdam, The Netherlands, 2021; pp. 59–103. [Google Scholar]
- Mahapatra, S.S.; Yadav, S.K.; Yoo, H.J.; Cho, J.W.; Park, J.-S. Highly branched polyurethane: Synthesis, characterization and effects of branching on dispersion of carbon nanotubes. Compos. Part B Eng. 2012, 45, 165–171. [Google Scholar] [CrossRef]
- Li, T.; Xie, Z.; Xu, J.; Weng, Y.-X.; Guo, B.-H. Design of a self-healing cross-linked polyurea with dynamic cross-links based on disulfide bonds and hydrogen bonding. Eur. Polym. J. 2018, 107, 249–257. [Google Scholar] [CrossRef]
- Available online: https://repository.tudelft.nl/islandora/object/uuid:3d93cc18-86f6-48cc-a203-910da569cfe9/datastream/OBJ/download (accessed on 14 September 2022).





| Priplast 3172 | Schiff Base | DMSO | Bismuth Catalyst | HMDI | Dynasylan 1189 | Tego Airex 900 | |
|---|---|---|---|---|---|---|---|
| Healable PU | 28 | 6 | 5 | 0.02 | 5.5 | 0.3 | 0.3 |
| Control PU | 28 | - | 5 | 0.02 | 1.69 | 0.3 | 0.3 |
| Tensile Strength (MPa) | Elongation at Break (%) | |
|---|---|---|
| Healable PU | 1.11 ± 0.58 | 384.3 ± 34.0 |
| Control PU | 0.46 ± 0.10 | 12.0 ± 1.3 |
| Tensile Strength (MPa) | Healing Efficiency (%) | |||
|---|---|---|---|---|
| Healed 1 N Scratch | Healed 2 N Scratch | Healed 1 N Scratch | Healed 2 N Scratch | |
| Healable PU | 0.89 ± 0.01 | 0.83 ± 0.08 | 81% | 75% |
| Control PU | 0.20 ± 0.05 | - * | 44% | - |
| T5% (°C) | Weight Residue (%) | |
|---|---|---|
| Healable PU | 292.35 | 10.835 |
| Control PU | 327.81 | 0.389 |
| Tg (°C) | Tm (°C) | Xc (%) | |
|---|---|---|---|
| Healable PU | −54.6 | 20.5 | 13.8 |
| Control PU | −57.5 | 32.9 | 28.2 |
| Initial | 2 N Scratch | After Healing | |
|---|---|---|---|
| Healable PU | ≥1000 | 532 | ≥1000 |
| Control PU | 702 | 252 | 276 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Smet, D.; Vanneste, M. Bio-Based Healable 2K PU Textile Coating for Durable Applications. Polymers 2022, 14, 4014. https://doi.org/10.3390/polym14194014
De Smet D, Vanneste M. Bio-Based Healable 2K PU Textile Coating for Durable Applications. Polymers. 2022; 14(19):4014. https://doi.org/10.3390/polym14194014
Chicago/Turabian StyleDe Smet, David, and Myriam Vanneste. 2022. "Bio-Based Healable 2K PU Textile Coating for Durable Applications" Polymers 14, no. 19: 4014. https://doi.org/10.3390/polym14194014
APA StyleDe Smet, D., & Vanneste, M. (2022). Bio-Based Healable 2K PU Textile Coating for Durable Applications. Polymers, 14(19), 4014. https://doi.org/10.3390/polym14194014