Microplastics Determination in Gastrointestinal Tracts of European Sea Bass (Dicentrarchus labrax) and Gilt-Head Sea Bream (Sparus aurata) from Tenerife (Canary Islands, Spain)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Contamination Control
2.2. Sample Processing and Observation
2.3. MicroFourier-Transform Infrared Spectroscopy Analysis
2.4. Statistical Analysis
3. Results and Discussion
3.1. Sampling and Sample Treatment
3.2. Microplastics Occurrence
3.3. Composition of the Microfibers
3.4. Comparison with Previous Studies Published in the Literature
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ugwu, K.; Herrera, A.; Gómez, M. Microplastics in marine biota: A review. Mar. Pollut. Bull. 2021, 169, 112540. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.-L.; Liu, L.-Y.; Hu, Y.-B.; Zeng, E.Y.; Guo, Y. Microplastics: A review of analytical methods, occurrence and characteristics in food, and potential toxicities to biota. Sci. Total Environ. 2022, 809, 150263. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-Q. Ocurrence of microplastics and its pollution in the environment: A review. Sustain. Prod. Consum. 2018, 13, 16–23. [Google Scholar] [CrossRef]
- Vighi, M.; Bayo, J.; Fernández-Piñas, F.; Gago, J.; Gómez, M.; Hernández-Borges, J.; Herrera, A.; Landaburu, J.; Muniategui-Lorenzo, S.; Muñoz, A.-R.; et al. Micro and Nano-Plastics in the Environment: Research Priorities for the Near Future. Springer Cham. 2021, 257, 163–218. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Chen, P. Microplastics in freshwater systems: A review on occurrence, environmental effects, and methods for microplastics detection. Water Res. 2018, 137, 362–374. [Google Scholar] [CrossRef]
- Cheung, L.T.O.; Lui, C.Y.; Fok, L. Microplastic Contamination of Wild and Captive Flathead Grey Mullet (Mugil cephalus). Int. J. Environ. Res. Public Health 2018, 15, 597. [Google Scholar] [CrossRef] [Green Version]
- Reinold, S.; Herrera, A.; Saliu, F.; Hernández-González, C.; Martínez, I.; Lasagni, M.; Gómez, M. Evidence of microplastics ingestion by cultured European sea bass (Dicentrachus labrax). Mar. Pollut. Bull. 2021, 168, 112450. [Google Scholar] [CrossRef]
- Garcia, A.G.; Suárez, D.C.; Li, J.; Rotchell, J.M. A comparison of microplastic contamination in freshwater fish from natural and farmed sources. Environ. Sci. Pollut. Res. 2020, 12, 14488–14497. [Google Scholar] [CrossRef]
- Wu, F.; Wang, Y.; Leung, J.Y.S.; Huang, W.; Zeng, J.; Tang, Y.; Chen, J.; Shi, A.; Yu, X.; Xu, X.; et al. Accumulation of microplastics in typical commercial aquatic species: A case study at a productive aquaculture site in China. Sci. Total Environ. 2019, 708, 135432. [Google Scholar] [CrossRef]
- Corami, F.; Rosso, B.; Sfristo, A.A.; Gambaro, A.; Mistri, M.; Munari, C.; Barbante, C. Additives, plasticizers, small microplastics (<100 μm), and other microlitter components in the gastrointestinal tract of commercial teleost fish: Method of extraction, purification, quantification, and characterization using Micro-FTIR. Mar. Pollut. Bull. 2022, 177, 113477. [Google Scholar] [CrossRef]
- Froese, R.; Pauly, D. (Eds.) Fish Database. Available online: http://www.fishbase.org (accessed on 2 February 2022).
- Toledo-Guedes, K.; Sánchez-Jerez, P.; González-Lorenzo, G.; Brito-Hernández, A. Detecting the degree of establishment of a non-indigenous species in coastal ecosystems: Sea bass Dicentrarchus labrax escapes from sea cages in Canary Islands (Northeastern Central Atlantic). Hydrobiologia 2009, 623, 203–212. [Google Scholar] [CrossRef]
- Leitão, F.; Santos, M.N.; Erzini, K.; Monteiro, C.C. The effect of predation on artificial reef juvenile demersal fish species. Mar. Biol. 2008, 153, 1233–1244. [Google Scholar] [CrossRef]
- Asociación Empresarial de Acuicultura de España. Available online: https://www.apromar.es (accessed on 2 February 2022).
- Bayo, J.; Rojo, D.; Martínez-Baños, P.; López-Castellanos, J.; Olmos, S. Commercial Gilthead Seabream (Sparus aurata L.) from the Mar Menor Coastal Lagoon as Hotspots of Microplastic Accumulation in the Digestive System. Int. J. Environ. Res. Public Health 2021, 18, 6844. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Lopes, C.; Oliveira, P.; Bessa, F.; Otero, V.; Henriques, B.; Raimundo, J.; Caetano, M.; Vale, C.; Vale, C.; et al. Microplastics in wild fish from North East Atlantic Ocean and its potential for causing neurotoxic effects, lipid oxidative damage, and human health risks associated with ingestion exposure. Sci. Total Environ. 2020, 717, 134625. [Google Scholar] [CrossRef]
- Akoueson, F.; Sheldon, L.M.; Danopoulos, E.; Morris, S.; Hotten, J.; Chapman, E.; Li, J.; Rotchell, J.M. A preliminary analysis of microplastics in edible versus non-edible tissues from seafood samples. Environ. Pollut. 2020, 263, 114452. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the Marine Environment: A review of the methods used for identification and quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef] [PubMed]
- Barrows, A.P.W.; Neumann, C.A.; Pieper, C.; Berger, M.L.; Shaw, S.D. Guide to Microplastic Identification, a Comprehensive Method Guide for Microplastics Identification and Quantification in the Laboratory; Marine & Environmental Research Institute: Blue Hill, ME, USA, 2017. [Google Scholar]
- Galgani, F.; Hanke, G.; Werner, S.; Oosterbaan, L.; Nilsson, P.; Fleet, D.; Kinsey, S.; Thompson, R.C.; van Franeker, J.; Vlachogianni, T.; et al. Guidance on Monitoring of Marine Litter in European Seas; Publications Office of the European Union: Luxembourg, 2013. [CrossRef]
- Edo, C.; González-Pleiter, M.; Tamayo-Belda, M.; Ortega-Ojeda, F.E.; Leganés, F.; Fernández-Piñas, F.; Rosal, R. Microplastics in sediments of artificially recharged lagoons: Case study in a biosphere reserve. Sci. Total Environ. 2020, 729, 138824. [Google Scholar] [CrossRef]
- Villanova-Solano, C.; Díaz-Peña, F.J.; Hernández-Sánchez, C.; González-Sálamo, J.; González-Pleiter, M.; Vega-Moreno, D.; Fernández-Piñas, F.; Fraile-Nuez, E.; Machín, F.; Hernández-Borges, J. Microplastic pollution in sublittoral coastal sediments of a North Atlantic island: The case of La Palma (Canary Islands, Spain). Chemosphere 2022, 288, 132530. [Google Scholar] [CrossRef]
- Sevillano-González, M.; González-Sálamo, J.; Díaz-Peña, F.; Hernández-Sánchez, C.; Catalán-Torralbo, S.; Ródenas-Seguí, A.; Hernández-Borges, J. Assessment of microplastic content in Diadema africanum sea urchin from Tenerife (Canary Islands, Spain). Mar. Pollut. Bull. 2022, 175, 112174. [Google Scholar] [CrossRef]
- Pérez-Reverón, R.; González-Sálamo, J.; Hernández-Sánchez, C.; González-Pleiter, M.; Hernández-Borges, J.; Díaz-Peña, F.J. Recycled wastewater as a potential source of microplastics in irrigated soils from an arid-insular territory (Fuerteventura, Spain). Sci. Total Environ. 2022, 817, 152830. [Google Scholar] [CrossRef]
- Herrera, A.; Ŝtindlová, A.; Martínez, I.; Rapp, J.; Romero-Kutzner, V.; Samper, M.D.; Montoto, T.; Aguiar-González, B.; Packard, T.; Gómez, M. Microplastic ingestion by Atlantic chub mackerel (Scomber colias) in the Canary Islands coast. Mar. Pollut. Bull. 2019, 139, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Adikaa, S.A.; Mahua, E.; Craneb, R.; Marchantc, R.; Montfordd, J.; Folorunshoe, R.; Gordon, C. Microplastic ingestion by pelagic and demersal fish species from the Eastern Central Atlantic Ocean, off the Coast of Ghana. Mar. Pollut. Bull. 2020, 153, 110998. [Google Scholar] [CrossRef] [PubMed]
- da Silva, J.M.; Alves, L.M.F.; Laranjeiro, M.I.; Bessa, F.; Silva, A.V.; Norte, A.C.; Lemos, M.F.L.; Ramos, J.A.; Novais, S.C.; Ceia, F.R. Accumulation of chemical elements and occurrence of microplastics in small pelagic fish from a neritic environment. Environ. Pollut. 2022, 292, 118451. [Google Scholar] [CrossRef] [PubMed]
- Pequeño, J.; Antunes, J.; Dhimmer, V.; Bessa, F.; Sobral, P. Microplastics in Marine and Estuarine Species from the Coast of Portugal. Front. Environ. Sci. 2021, 9, 579127. [Google Scholar] [CrossRef]
- Maaghloud, H.; Houssa, R.; Bellali, F.; El Bouqdaoui, K.; Ouansafi, S.; Loulad, S.; Fahde, A. Microplastic ingestion by Atlantic horse mackerel (Trachurus trachurus) in the North and central Moroccan Atlantic coast between Larache (35°30′ N) and Boujdour (26°30′ N). Environ. Pollut. 2021, 288, 117781. [Google Scholar] [CrossRef]

| Species (n) | Fork Length (cm) | Standard Length (cm) | Specimen Weight (g) | Gastrointestinal Tracts Weight (g) |
|---|---|---|---|---|
| Europen sea-bass (Dicentrarchus labrax) n = 45 | 37.5 ± 1.4 cm Min = 33.8 cm Max = 41.6 cm | 33.7 ± 2.0 cm Min = 30.1 cm Max = 40.6 cm | 660.5 ± 46.3 g Min = 536.4 g Max = 787.3 g | 11.0 ± 1.6 g Min = 8.1 g Max = 16.4 g |
| Gilt-head sea bream (Sparus aurata) n = 41 | 31.4 ± 2.7 cm Min = 26.0 cm Max = 36.5 cm | 27.1 ± 2.3 cm Min = 22.5 cm Max = 34.0 cm | 609.7 ± 125.8 g Min = 322.2 g Max = 804.6 g | 12.7 ± 2.8 g Min = 6.8 g Max = 17.2 g |
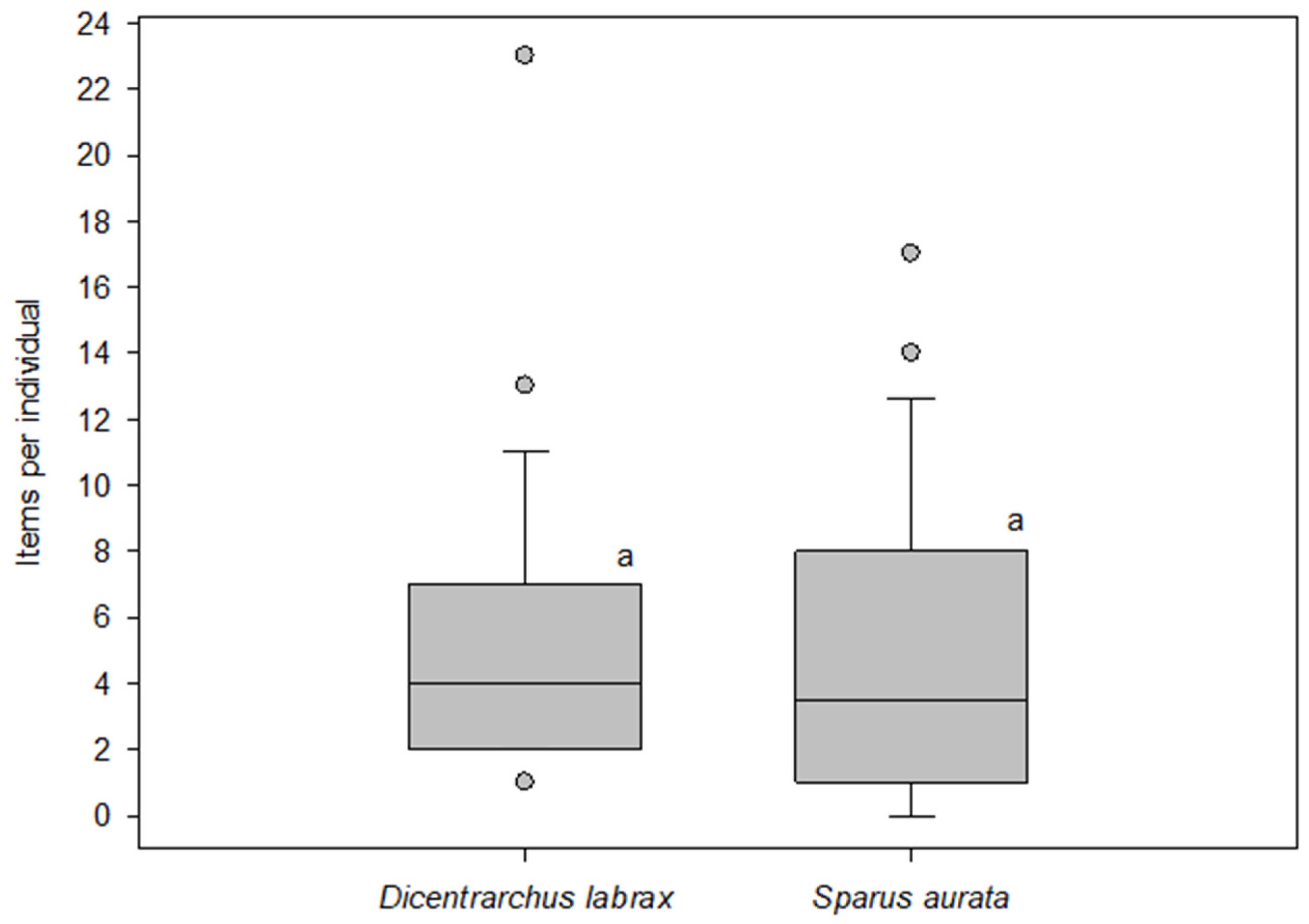
| Species (n) | Total Number of Particles Found | Average Items/Individual ± SD | Items/Individual Range | Average Items Length ± SD | Items Length Range | Shape Classification |
|---|---|---|---|---|---|---|
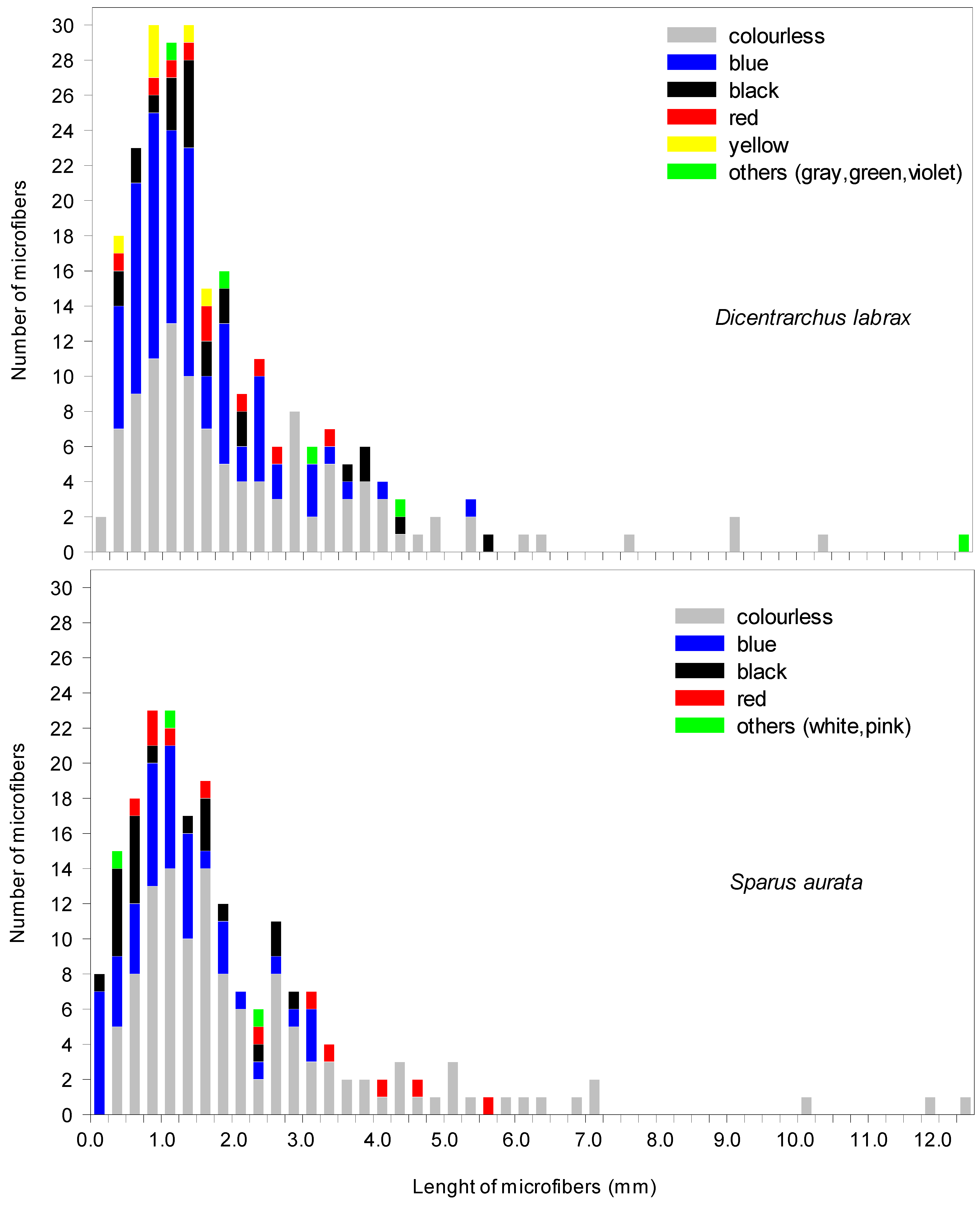
| European seabass (Dicentrarchus labrax) n = 45 | 242 | 5.4 ± 4.2 | 1–23 | 1957 ± 1699 μm | 221 μm–12.4 mm | 242 microfibers (100%) |
| Gilt-head sea bream (Sparus aurata) n = 41 | 208 * | 5.1 ± 5.1 | 0–17 | 1988 ± 1853 μm | 69 μm–12.4 mm | 200 microfibers (96.1%) 1 line (0.5%) 1 film (0.5%) 1 fragment (0.5%) 5 tangled messes (2.4%) |
| Location | Species | Number of Individuals Analysed | Digestion | Items/Individual | Shape (%) | Fibers Length | Colours (%) | Chemical Composition | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Tenerife, Canary Islands, Atlantic Ocean | Dicentrarchus labrax (farmed) | 83 | 10% KOH three times the organic material, room temperature, 2 weeks | 1.43 ± 1.75 | Fibers (81.0%) Fragments (12.0%) Films (4.0%) Lines (3.0%) | - | Blue (26.3%) Yellow (23.7%) Black (16.9%) Transparent (14.4%) Pink (5.1%) White (2.5%) Red (2.5%) Green (1.7%) Silver (1.7%) Grey (1.7%) Yellowish semitransparent (1.7%) Brown (0.8%) Purple (0.8%) | Fibers (11) Cellulose/Cellophane (55.0%) Nylon (27.0%) Rayon (9.0%) Acrylic (9.0%)] Particles (20) PE (25.0%) PP (25.0%) PS (5.0%) SAN (5.0%) PA (5.0%) EPDM (5.0%) E/p (5.0%) EVA (5.0%) Polynorbornene (5.0%) Nitrocellulose (5.0%) Epoxy resin (5.0%) Phenolic resin (5.0%) | [7] |
| Gran Canaria and Lanzarote, Canary Islands, Atlantic Ocean | Scomber colias (wild) | 120 | 10% KOH | 2.17 ± 2.04 | Fibers (74.2%) Fragments (11.9%) Paints (11.5%) Lines (1.5%) Films (0.8%) | - | Blue (55.0%) Dark/Black (23.5%) Red (10.4%) Green (5.0%) Clear/White (4.6%) Yellow/Brown (1.5%) | - | [19] |
| Eastern Central Atlantic Ocean, Coast of Ghana | Sardinella maderensis (wild) | 80 | 20 mL of 10 M KOH at 60 °C for 24 h | 32.0 ± 2.7 | Pellets (31.0%) Microbeads (29.0%) Burnt plastic films (22.0%) Clear plastic fragments (6.0%) White plastic fragments (3.0%) Green plastic fragment (5.0%) Thread plastics (2.0%) Microfibers (2.0%) Foams (<0.1%) | - | - | - | [26] |
| Sardinella aurita (wild) | 47 | 26.0 ± 1.6 | |||||||
| Dentex angolensis (wild) | 28 | 40.0 ± 3.8 | |||||||
| Portuguese coast, Atlantic Ocean | Sardina pilchardus (wild) | 20 | 10% KOH at room temperature for 2 days | 0.26 ± 0.56 | Fibers (71.0%) Fragments (24.0%) Films (6.0%) | 11.47 ± 19.05 mm | Blue (35.0%) Transparent (29.0%) Green (12.0%) Black (12.0%) Red and Purple (<6.0%) | - | [27] |
| Trachurus trachurus (wild) | 20 | 0.37 ± 0.60 | 0.54 ± 0.53 mm | ||||||
| Scomber spp. (wild) | 13 | 0.38 ± 0.51 | 1.13 ± 1.09 mm | ||||||
| Portuguese coast, Atlantic Ocean | Trachurus trachurus (wild) | 82 | 10% KOH | 2.24 ± 2.05 | Fibers (88.0%) Fragments (12.0%) | 1090 ± 1011 µm | Blue (39.0%) Black (25.0%) Red (12.0%) Green (12.0%) Transparent (12.0%) | PET (64.0%) PE (27.0%) PP (9.0%) | [28] |
| Scomber colias (wild) | 82 | 2.46 ± 4.12 | Fibers (70.0%) Fragments (30.0%) | PE (47.0%) PET (34.0%) PP (16.0%) PA (3.0%) | |||||
| North and Central Moroccan Atlantic coast | Trachurus trachurus (wild) | 147 | - | 0.46 ± 1.29 | Fibers (58.0%) Fragments (40.0%) Films (2.0%) | - | Blue (33.0%) White (31.0%) Red (16.0%) Black (11.0%) Green (7.0%) Yellow (2.0%) | Acrylic (47.0%) PS (32.0%) Others (21.0%) | [29] |
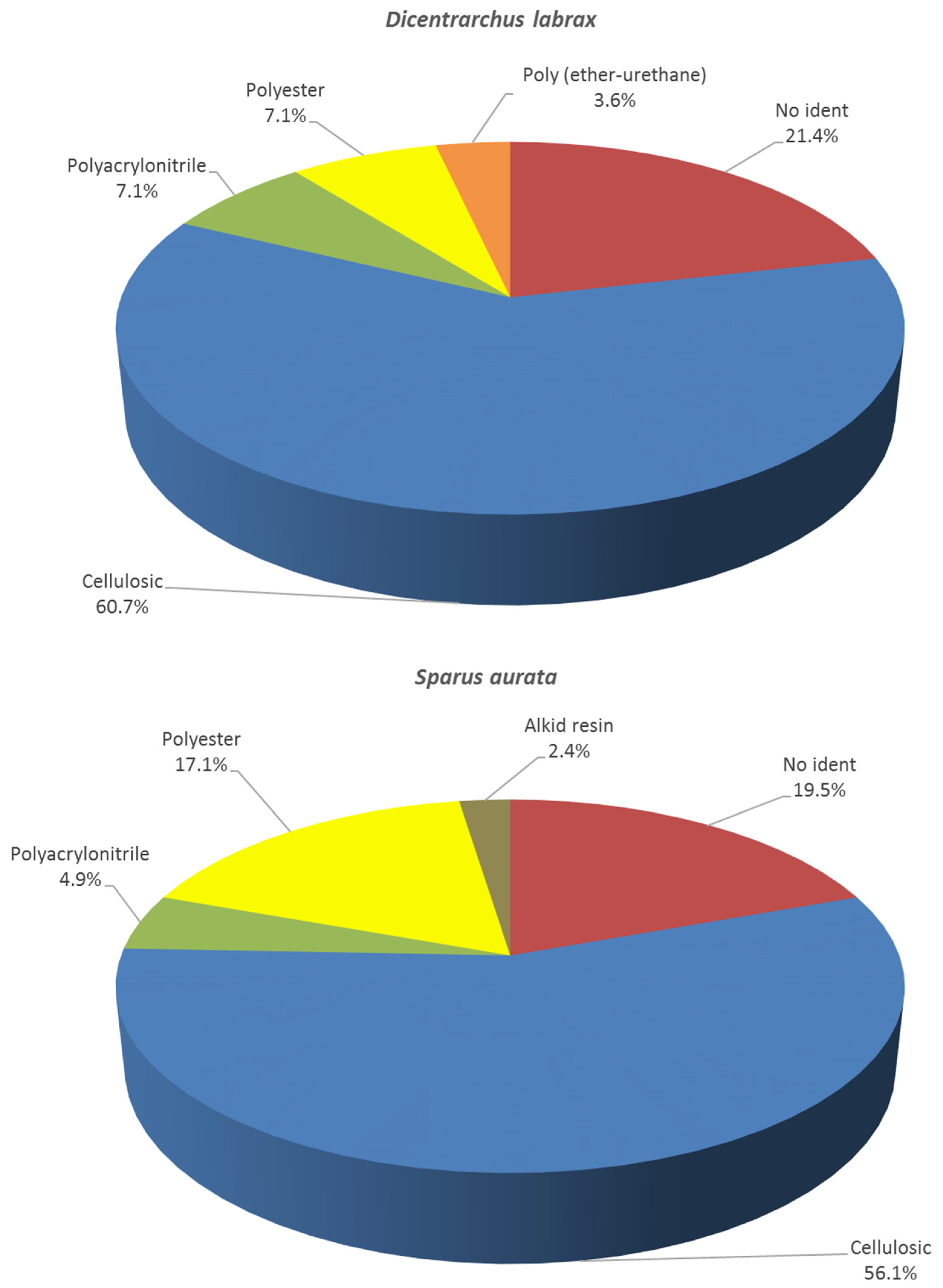
| Arona, Tenerife, Canary Islands, Atlantic Ocean | Dicentrarchus labrax (farmed) | 45 | 10% KOH 60 °C 24 h | 5.4 ± 4.2 | Fibers (100%) | 221 µm–12.4 mm | Colourless (47.7%) Blue (35.3%) Black (8.7%) Red (4.2%) Yellow (2.5%) Grey (0.8%) Green (0.4%) Violet (0.4% | Cellulosics (60.7%) Polyester (7.1%) Polyacrylonitrile (7.1%) Poly (ether-urethane) (3.6%) Non identified (21.4%) | This study |
| Sparus aurata (farmed) | 41 | 5.1 ± 5.1 | Fibers (96.1%) Line (0.5%) Tangled messes (2.4%) Films (0.5%) Fragments (0.5%) | 69 µm–12.4 mm | Colourless (60.9%) Blue (24.8%) Black (7.9%) Red (5.4%) White (0.5%) Pink (0.5%) | Cellulosics (56.1%) Alkid resin (2.4%) Polyester (17.1%) Polyacrylonitrile (4.9%) Non identified (19.5%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Almeida, R.; Hernández-Sánchez, C.; Villanova-Solano, C.; Díaz-Peña, F.J.; Clemente, S.; González-Sálamo, J.; González-Pleiter, M.; Hernández-Borges, J. Microplastics Determination in Gastrointestinal Tracts of European Sea Bass (Dicentrarchus labrax) and Gilt-Head Sea Bream (Sparus aurata) from Tenerife (Canary Islands, Spain). Polymers 2022, 14, 1931. https://doi.org/10.3390/polym14101931
Sánchez-Almeida R, Hernández-Sánchez C, Villanova-Solano C, Díaz-Peña FJ, Clemente S, González-Sálamo J, González-Pleiter M, Hernández-Borges J. Microplastics Determination in Gastrointestinal Tracts of European Sea Bass (Dicentrarchus labrax) and Gilt-Head Sea Bream (Sparus aurata) from Tenerife (Canary Islands, Spain). Polymers. 2022; 14(10):1931. https://doi.org/10.3390/polym14101931
Chicago/Turabian StyleSánchez-Almeida, Raquel, Cintia Hernández-Sánchez, Cristina Villanova-Solano, Francisco Javier Díaz-Peña, Sabrina Clemente, Javier González-Sálamo, Miguel González-Pleiter, and Javier Hernández-Borges. 2022. "Microplastics Determination in Gastrointestinal Tracts of European Sea Bass (Dicentrarchus labrax) and Gilt-Head Sea Bream (Sparus aurata) from Tenerife (Canary Islands, Spain)" Polymers 14, no. 10: 1931. https://doi.org/10.3390/polym14101931
APA StyleSánchez-Almeida, R., Hernández-Sánchez, C., Villanova-Solano, C., Díaz-Peña, F. J., Clemente, S., González-Sálamo, J., González-Pleiter, M., & Hernández-Borges, J. (2022). Microplastics Determination in Gastrointestinal Tracts of European Sea Bass (Dicentrarchus labrax) and Gilt-Head Sea Bream (Sparus aurata) from Tenerife (Canary Islands, Spain). Polymers, 14(10), 1931. https://doi.org/10.3390/polym14101931










