Chitosan Composites Synthesized Using Acetic Acid and Tetraethylorthosilicate Respond Differently to Methylene Blue Adsorption
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Synthesis of Chitosan Beads Using Acetic Acid
2.2.2. Synthesis of Chitosan Beads Using TEOS
2.2.3. Synthesis of Chitosan Beads Using Double Cross-Linking (Acetic Acid Followed by TEOS)
2.2.4. Synthesis of Chitosan Beads Using One-Step Method (Equimolar Concentrations of Acetic Acid and TEOS)
2.2.5. Sample Characterization
2.3. Analysis
2.3.1. Point of Zero Charge
2.3.2. Swelling Ratio
2.3.3. Methylene Blue Adsorption and Release Properties of the Composites
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Algul, D.; Gokce, A.; Onal, A.; Servet, E.; Dogan Ekici, A.I.; Yener, F.G. In vitro release and In vivo biocompatibility studies of biomimetic multilayered alginate-chitosan/beta-TCP scaffold for osteochondral tissue. J. Biomater. Sci. Polym. Ed. 2016, 27, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Koc, A.; Finkenzeller, G.; Elcin, A.E.; Stark, G.B.; Elcin, Y.M. Evaluation of adenoviral vascular endothelial growth factor-activated chitosan/hydroxyapatite scaffold for engineering vascularized bone tissue using human osteoblasts: In vitro and in vivo studies. J. Biomater. Appl. 2014, 29, 748–760. [Google Scholar] [CrossRef] [PubMed]
- Bernkop-Schnurch, A.; Dunnhaupt, S. Chitosan-based drug delivery systems. Eur. J. Pharm. Biopharm. 2012, 81, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Cabral, J.D.; McConnell, M.A.; Fitzpatrick, C.; Mros, S.; Williams, G.; Wormald, P.J.; Moratti, S.C.; Hanton, L.R. Characterization of the in vivo host response to a bi-labeled chitosan-dextran based hydrogel for postsurgical adhesion prevention. J. Biomed. Mater. Res. A 2015, 103, 2611–2620. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Chatterjee, T.; Lim, S.R.; Woo, S.H. Adsorption of a cationic dye, methylene blue, on to chitosan hydrogel beads generated by anionic surfactant gelation. Environ. Technol. 2011, 32, 1503–1514. [Google Scholar] [CrossRef] [PubMed]
- Park, S.B.; You, J.O.; Park, H.Y.; Haam, S.J.; Kim, W.S. A novel pH-sensitive membrane from chitosan-TEOS IPN; preparation and its drug permeation characteristics. Biomaterials 2001, 22, 323–330. [Google Scholar] [CrossRef]
- Jothimani, B.; Sureshkumar, S.; Venkatachalapathy, B. Hydrophobic structural modification of chitosan and its impact on nanoparticle synthesis—A physicochemical study. Carbohydr. Polym. 2017, 173, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.; Han, J.; Li, Q.; He, B.; Zha, B.; Wu, J.; Zhou, R.; Ye, T.; Wang, W.; Xu, H.; et al. Synthesis of galactosylated chitosan/5-fluorouracil nanoparticles and its characteristics, in vitro and in vivo release studies. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 2035–2043. [Google Scholar] [CrossRef] [PubMed]
- Delval, F.; Crini, G.; Vebrel, J. Removal of organic pollutants from aqueous solutions by adsorbents prepared from an agroalimentary by-product. Bioresour. Technol. 2006, 97, 2173–2181. [Google Scholar] [CrossRef] [PubMed]
- Xin, C.; Zhao, N.; Zhan, H.; Xiao, F.; Wei, W.; Sun, Y. Phase transition of silica in the TMB-P123-H2O-TEOS quadru-component system: A feasible route to different mesostructured materials. J. Colloid Interface Sci. 2014, 433, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Budnyak, T.M.; Pylypchuk, I.V.; Tertykh, V.A.; Yanovska, E.S.; Kolodynska, D. Synthesis and adsorption properties of chitosan-silica nanocomposite prepared by sol-gel method. Nanoscale Res. Lett. 2015, 10, 87. [Google Scholar] [CrossRef] [PubMed]
- Toskas, G.; Cherif, C.; Hund, R.D.; Laourine, E.; Mahltig, B.; Fahmi, A.; Heinemann, C.; Hanke, T. Chitosan(PEO)/silica hybrid nanofibers as a potential biomaterial for bone regeneration. Carbohydr. Polym. 2013, 94, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Pipattanawarothai, A.; Suksai, C.; Srisook, K.; Trakulsujaritchok, T. Non-cytotoxic hybrid bioscaffolds of chitosan-silica: Sol-gel synthesis, characterization and proposed application. Carbohydr. Polym. 2017, 178, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Liu, W.; Feng, Q.; Dong, C.; Liu, Q.; Duan, L.; Huang, J.; Zhu, W.; Li, Z.; Xiong, J.; et al. Effect of inorganic/organic ratio and chemical coupling on the performance of porous silica/chitosan hybrid scaffolds. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 70, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Matsuhisa, H.; Tsuchiya, M.; Hasebe, Y. Protein and polysaccharide-composite sol-gel silicate film for an interference-free amperometric glucose biosensor. Colloids Surf. B Biointerfaces 2013, 111, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, J.; Zhang, S. Molecularly imprinted polymer grafted on polysaccharide microsphere surface by the sol-gel process for protein recognition. Talanta 2008, 74, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Elsagh, A. Synthesis of Silica Nanostructures and Optimization of their Size and Morphology by Use of Changing in Synthesis Conditions. J. Chem. 2011, 9, 659–668. [Google Scholar] [CrossRef]
- Gurses, A.; Dogar, C.; Yalcin, M.; Acikyildiz, M.; Bayrak, R.; Karaca, S. The adsorption kinetics of the cationic dye, methylene blue, onto clay. J. Hazard. Mater. 2006, 131, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, C.C.; Mouta, R.; Junior, M.C.C.; Santana, S.A.A.; Silva, H.; Bezerra, C.W.B. Chitosan-edible oil based materials as upgraded adsorbents for textile dyes. Carbohydr. Polym. 2018, 180, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Ju, H.; Zhou, J.; Cal, C.; Chen, H. The Electrochemical Behaviour of Methylene Blue at a Microcylinder Carbon Fiber Electrode. Electroanalysis 1994, 7, 1165–1169. [Google Scholar] [CrossRef]
- Kamari, A.; Ngah, W.S.W.; Liew, L.K. Chitosan and Chemically Modified Beads for acid dyes sorption. J. Environ. Sci. 2009, 21, 296–302. [Google Scholar]
- Tiburu, E.K.; Fleischer, H.N.A.; Aidoo, E.O.; Salifu, A.A.; Asimeng, B.O.; Zhou, H. Crystallization of Linde Type A nanomaterials at two Temperatures Exhibit Differential Inhibition of HeLa Cervical Cancer Cells In vitro. J. Biomim. Biomater. Biomed. Eng. 2016, 28, 66–77. [Google Scholar] [CrossRef]
- Copello, J.G.; Villanueva, M.E.; Gonzalez, J.A.; Eguez, L.S.; Diaz, L.E. TEOS as an Improved Alternative for Chitosan Beads Cross-Linking: A Comparative Adsorption Study. Appl. Polym. 2014, 131, 41005–41012. [Google Scholar] [CrossRef]
- Lopez-Ramon, M.V.; Stoeckli, F.; Moreno-Castilla, C.; Carrasco-Marin, F. On the characterization of acidic and basic surface sites on carbons by various technoques. Carbon 1999, 37, 1215. [Google Scholar] [CrossRef]
- Gouda, R.; Baishya, H.; Qing, Z. Application of Mathematical Models in Drug Release Kinetics of Carbidopa and Levodopa ER Tablets. J. Dev. Drugs 2017, 6, 171. [Google Scholar]
- Tiburu, E.K.; Salifu, A.A.; Aidoo, E.O.; Fleischer, H.N.A.; Manu, G.; Yaya, A.; Zhou, H.; Efavi, J.K. Formation of Chitosan Nanoparticles Using Deacetylated Chitin Isolated from Freshwater Algae and Locally Synthesized Zeolite A and their Influence on Cancer Cell Growth. J. Nanopart. Res. 2017, 48, 156. [Google Scholar] [CrossRef]
- Argüelles-Monal, W.M.; Lizardi-Mendoza, J.; Fernández-Quiroz, D.; Recillas-Mota, M.T. Montiel-Herrera, M. Chitosan Derivatives: Introducing New Functionalities with a Controlled Molecular Architecture for Innovative Materials. Polymers 2018, 10, 342. [Google Scholar] [CrossRef]
- Gohi, B.F.C.A.; Zeng, H.-Y.; Pan, A.D.; Han, J.; Yuan, J. pH Dependence of Chitosan Enzymolysis. Polymers 2017, 9, 174. [Google Scholar] [CrossRef]
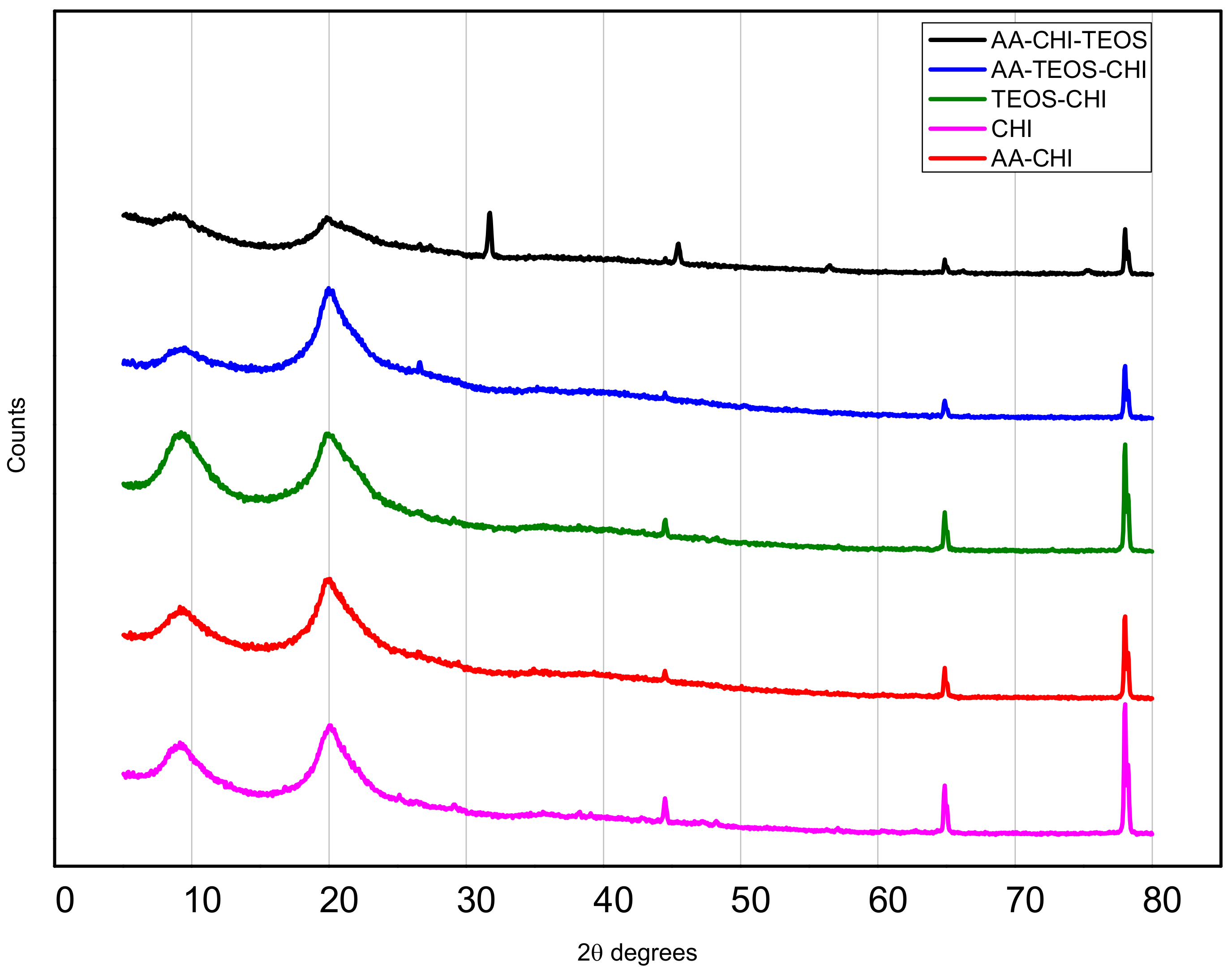

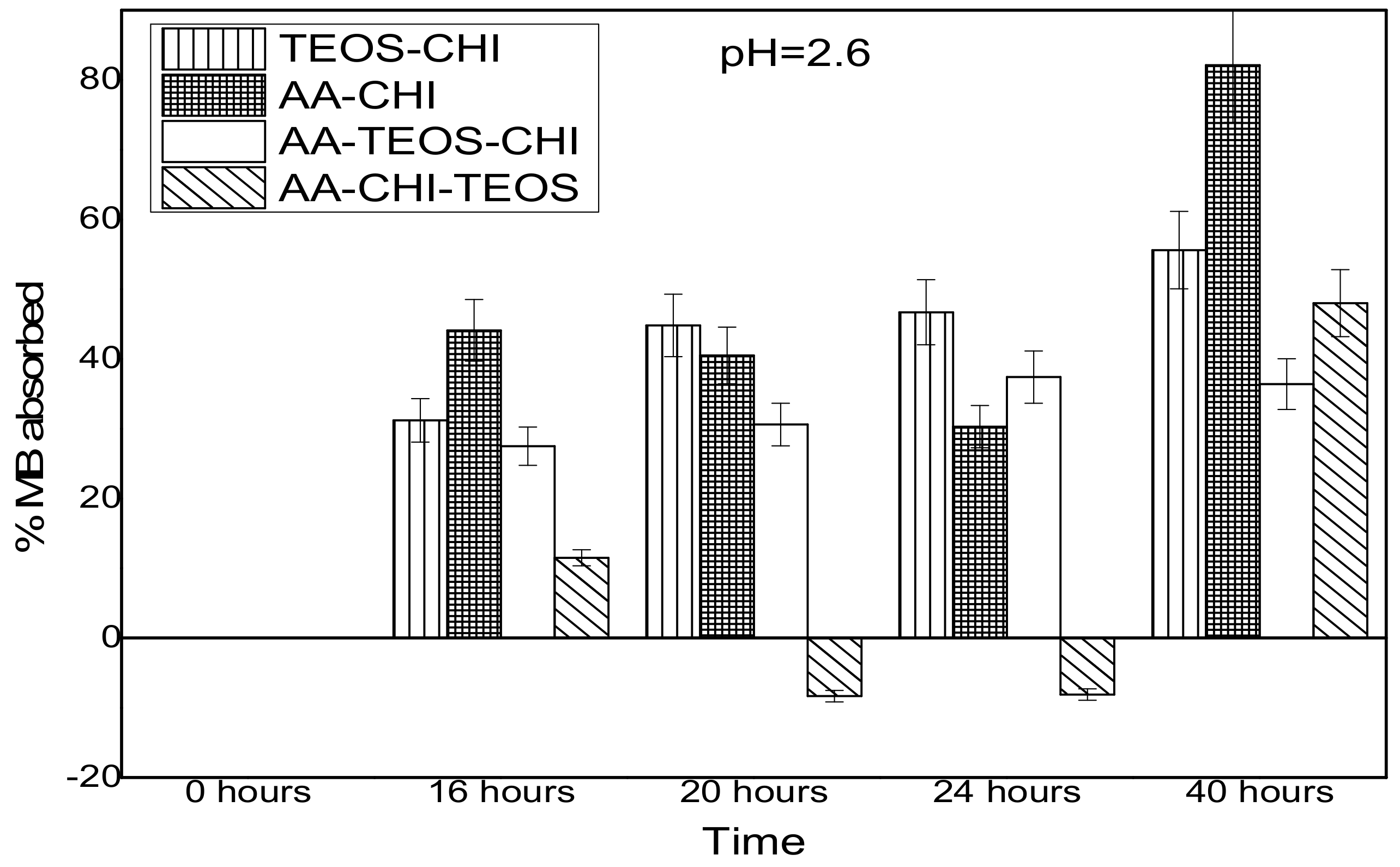
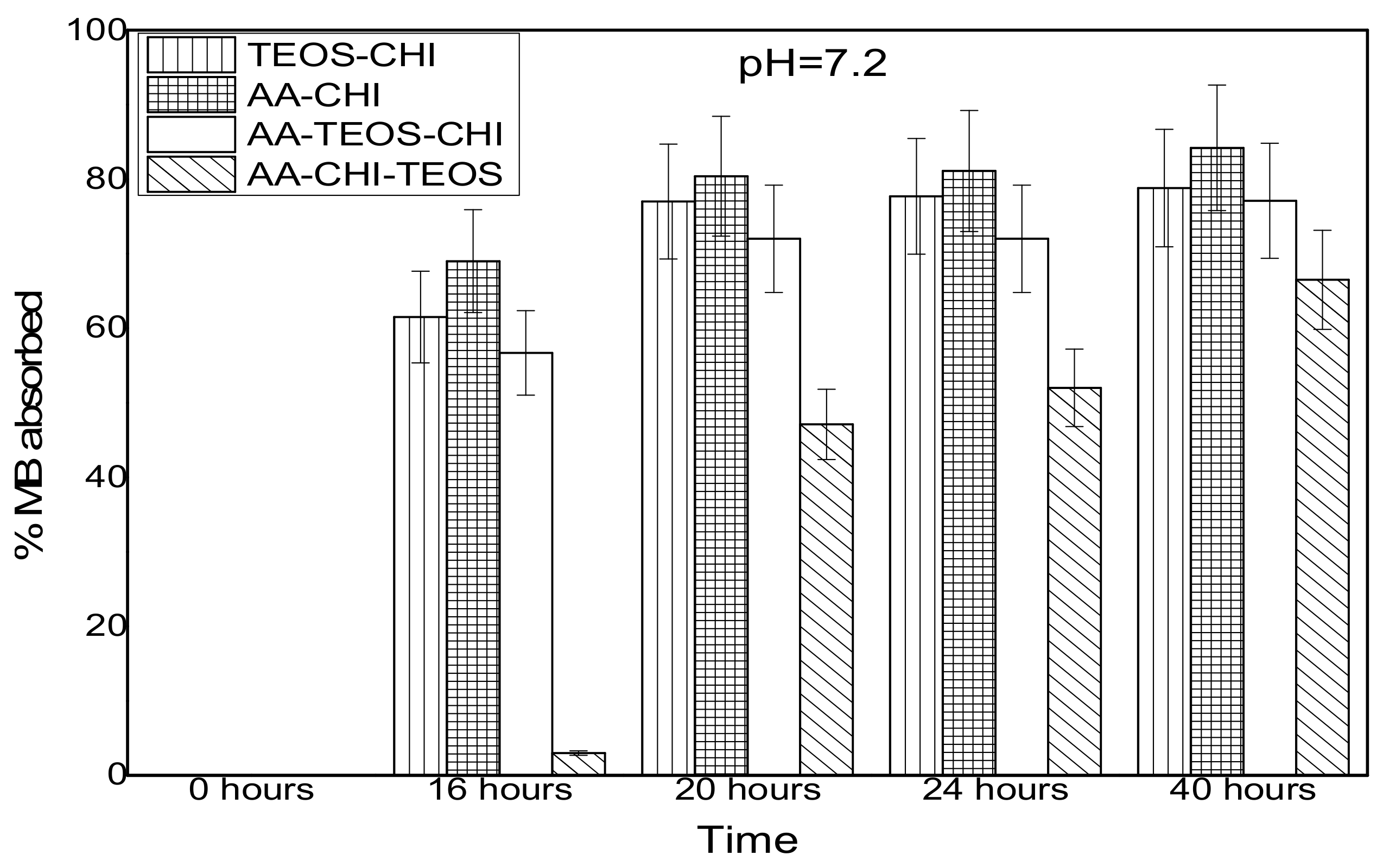

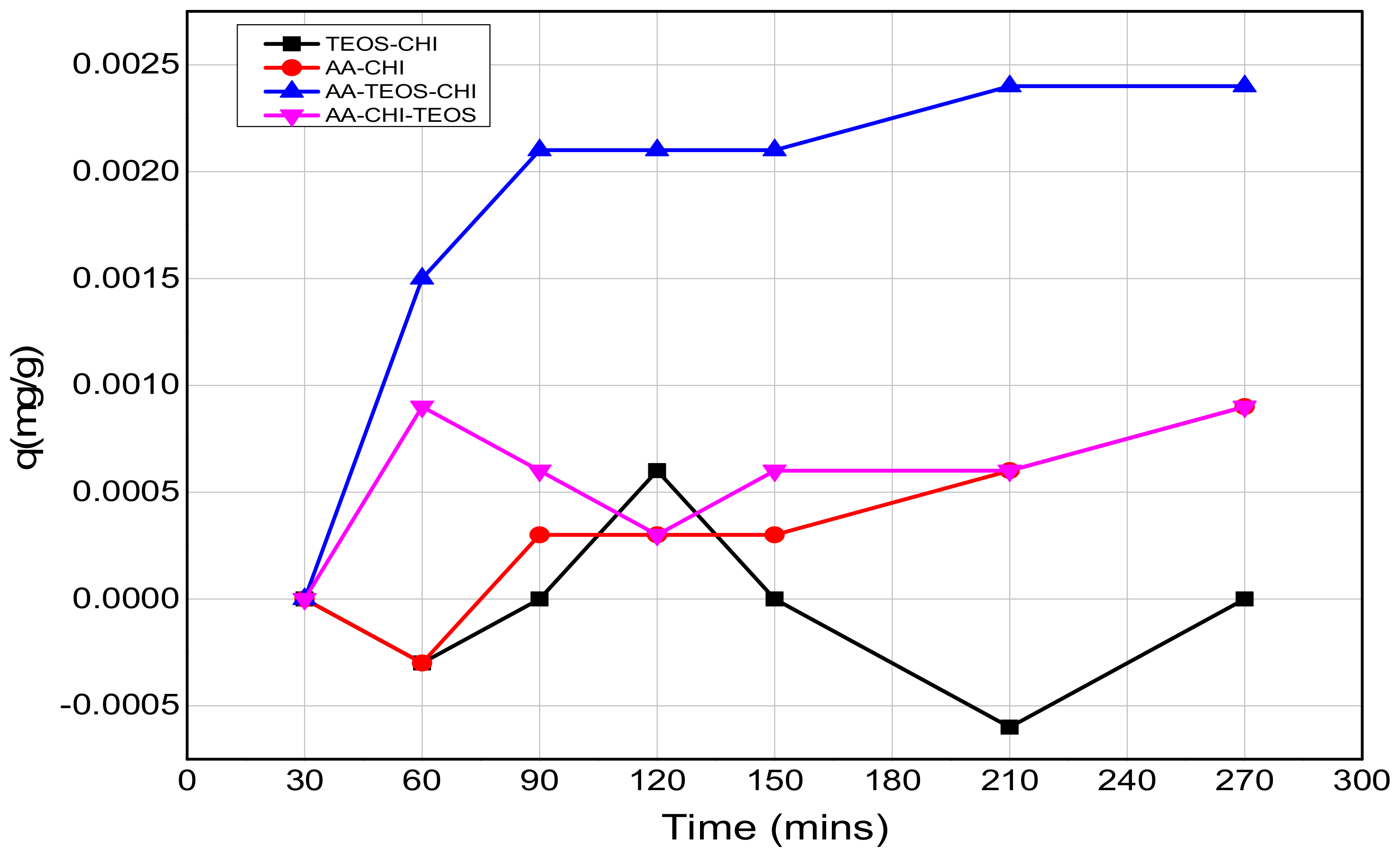
| Chitosan (CHI) | Acetic acid (AA) | TEOS | |
|---|---|---|---|
| CHI | √ | ||
| AA-CHI | √ | √ | |
| TEOS-CHI | √ | √ | |
| a AA-CHI-TEOS | √ | √ | √ |
| b AA-TEOS-CHI | √ | √ | √ |
| Sample | Swelling ratio | % Porosity (%ε) | pHpzc | ||
|---|---|---|---|---|---|
| pH = 2.6 | pH = 7.2 | pH = 2.6 | pH = 7.2 | ||
| AA-CHI | 3.0 | 2.4 | 86 | 83 | 7.6 |
| TEOS-CHI | 4.0 | 2.6 | 92 | 89 | 3.5 |
| a AA-CHI-TEOS | 4.0 | 2.0 | 87 | 77 | 7.1 |
| b AA-TEOS-CHI | 3.0 | 1.0 | 88 | 70 | 7.5 |
| Samples | R2 Values | |
|---|---|---|
| pH = 2.6 | pH = 7.2 | |
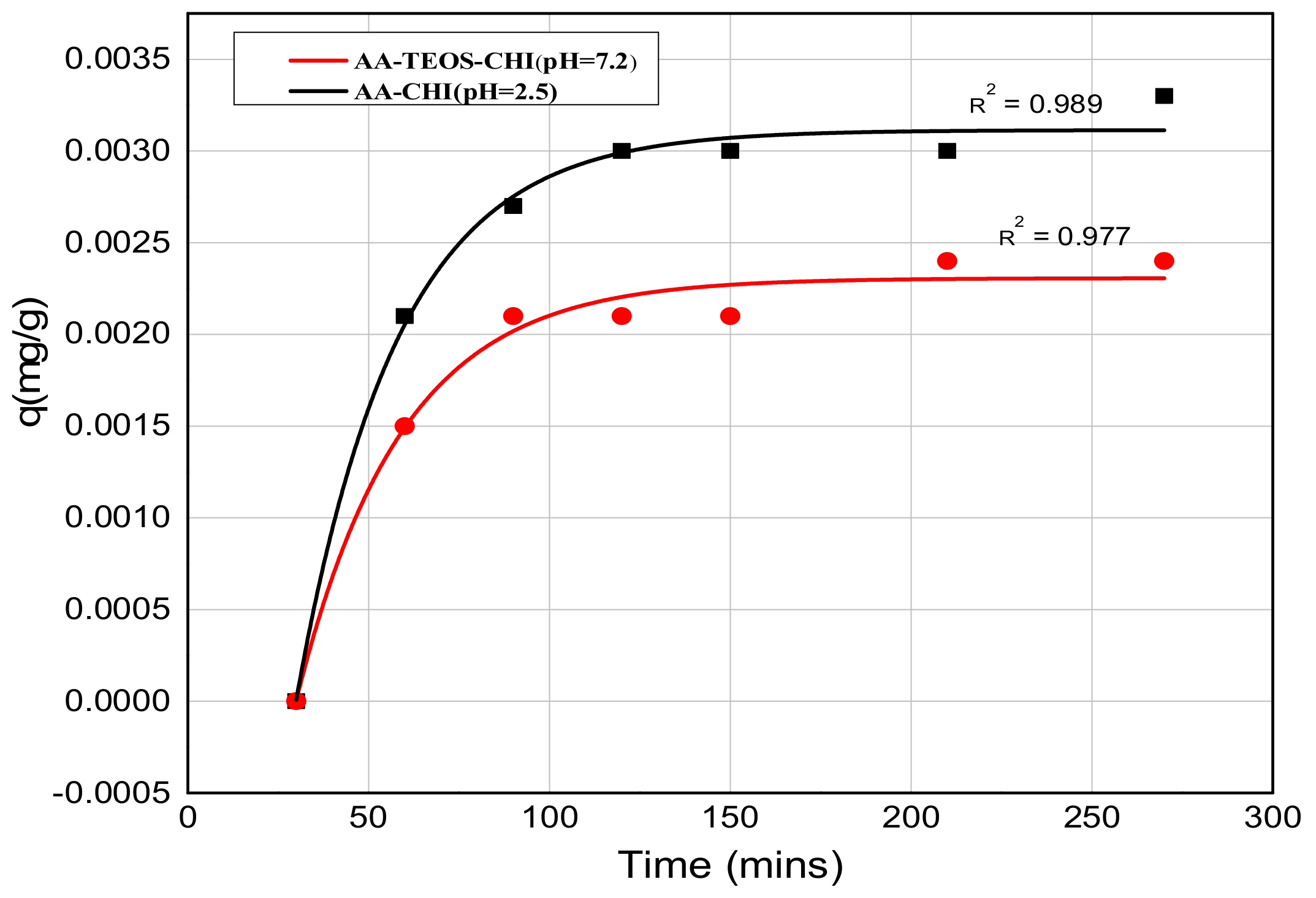
| AA-CHI | 0.989 | 0.757 |
| TEOS-CHI | 0.539 | −0.5 |
| a AA-CHI-TEOS | 0.156 | 0.380 |
| b AA-TEOS-CHI | 0.658 | 0.977 |
| Sample | TEOS-CHI | AA-CHI | AA-TEOS-CHI | AA-CHI-TEOS | |
|---|---|---|---|---|---|
| Pearson’s R | pH = 2.6 | −0.18 | 0.92 | 0.76 | 0.52 |
| pH = 7.2 | 0.77 | 0.745 | 0.748 | 0.38 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Essel, T.Y.A.; Koomson, A.; Seniagya, M.-P.O.; Cobbold, G.P.; Kwofie, S.K.; Asimeng, B.O.; Arthur, P.K.; Awandare, G.; Tiburu, E.K. Chitosan Composites Synthesized Using Acetic Acid and Tetraethylorthosilicate Respond Differently to Methylene Blue Adsorption. Polymers 2018, 10, 466. https://doi.org/10.3390/polym10050466
Essel TYA, Koomson A, Seniagya M-PO, Cobbold GP, Kwofie SK, Asimeng BO, Arthur PK, Awandare G, Tiburu EK. Chitosan Composites Synthesized Using Acetic Acid and Tetraethylorthosilicate Respond Differently to Methylene Blue Adsorption. Polymers. 2018; 10(5):466. https://doi.org/10.3390/polym10050466
Chicago/Turabian StyleEssel, Thomas Y. A., Albert Koomson, Marie-Pearl O. Seniagya, Grace P. Cobbold, Samuel K. Kwofie, Bernard O. Asimeng, Patrick K. Arthur, Gordon Awandare, and Elvis K. Tiburu. 2018. "Chitosan Composites Synthesized Using Acetic Acid and Tetraethylorthosilicate Respond Differently to Methylene Blue Adsorption" Polymers 10, no. 5: 466. https://doi.org/10.3390/polym10050466
APA StyleEssel, T. Y. A., Koomson, A., Seniagya, M.-P. O., Cobbold, G. P., Kwofie, S. K., Asimeng, B. O., Arthur, P. K., Awandare, G., & Tiburu, E. K. (2018). Chitosan Composites Synthesized Using Acetic Acid and Tetraethylorthosilicate Respond Differently to Methylene Blue Adsorption. Polymers, 10(5), 466. https://doi.org/10.3390/polym10050466






