De novo Phasing Xenons Observed in the Frog Ependymin-Related Protein
Abstract
1. Introduction
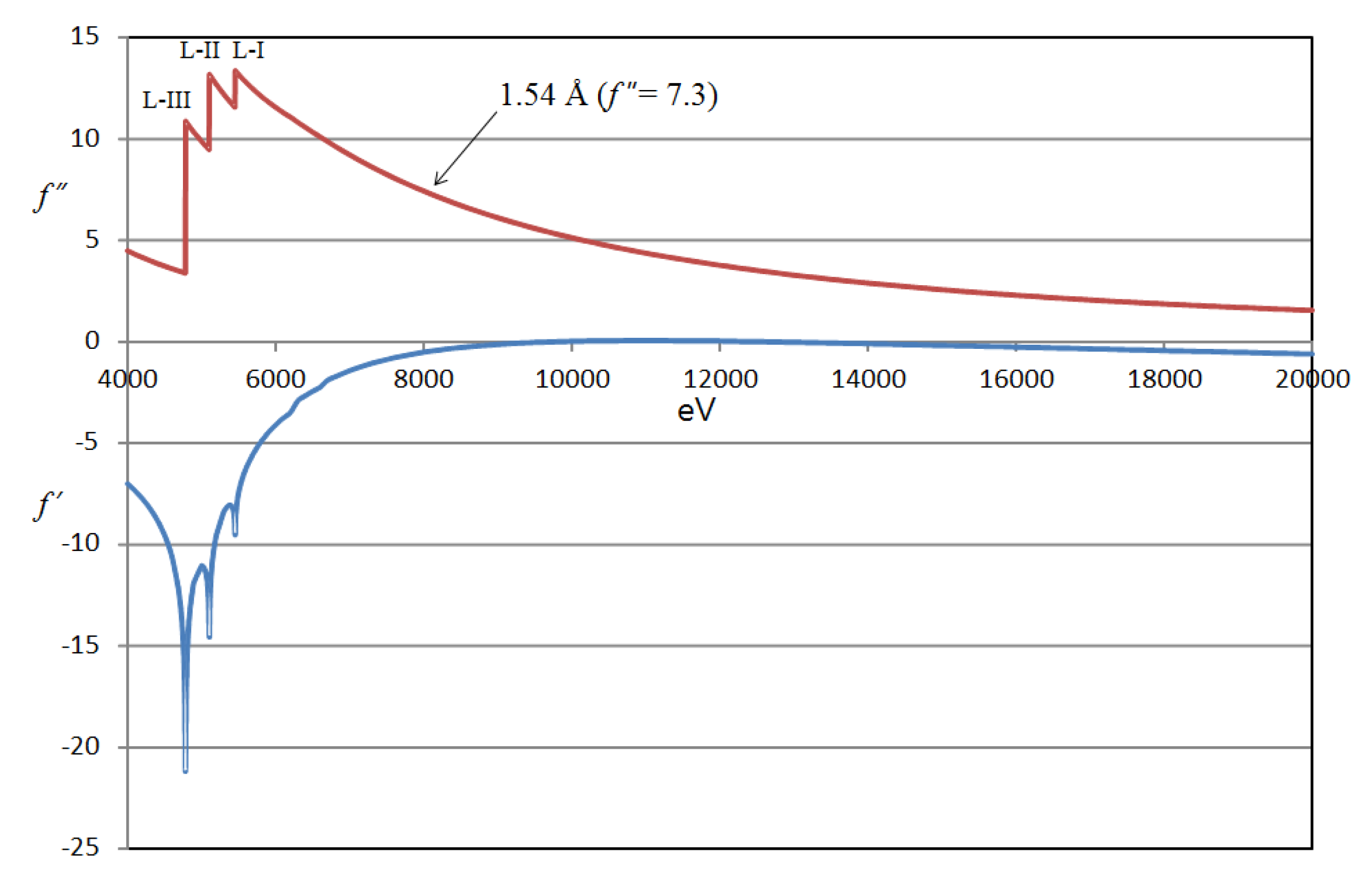
2. Materials and Methods
3. Results and Discussion
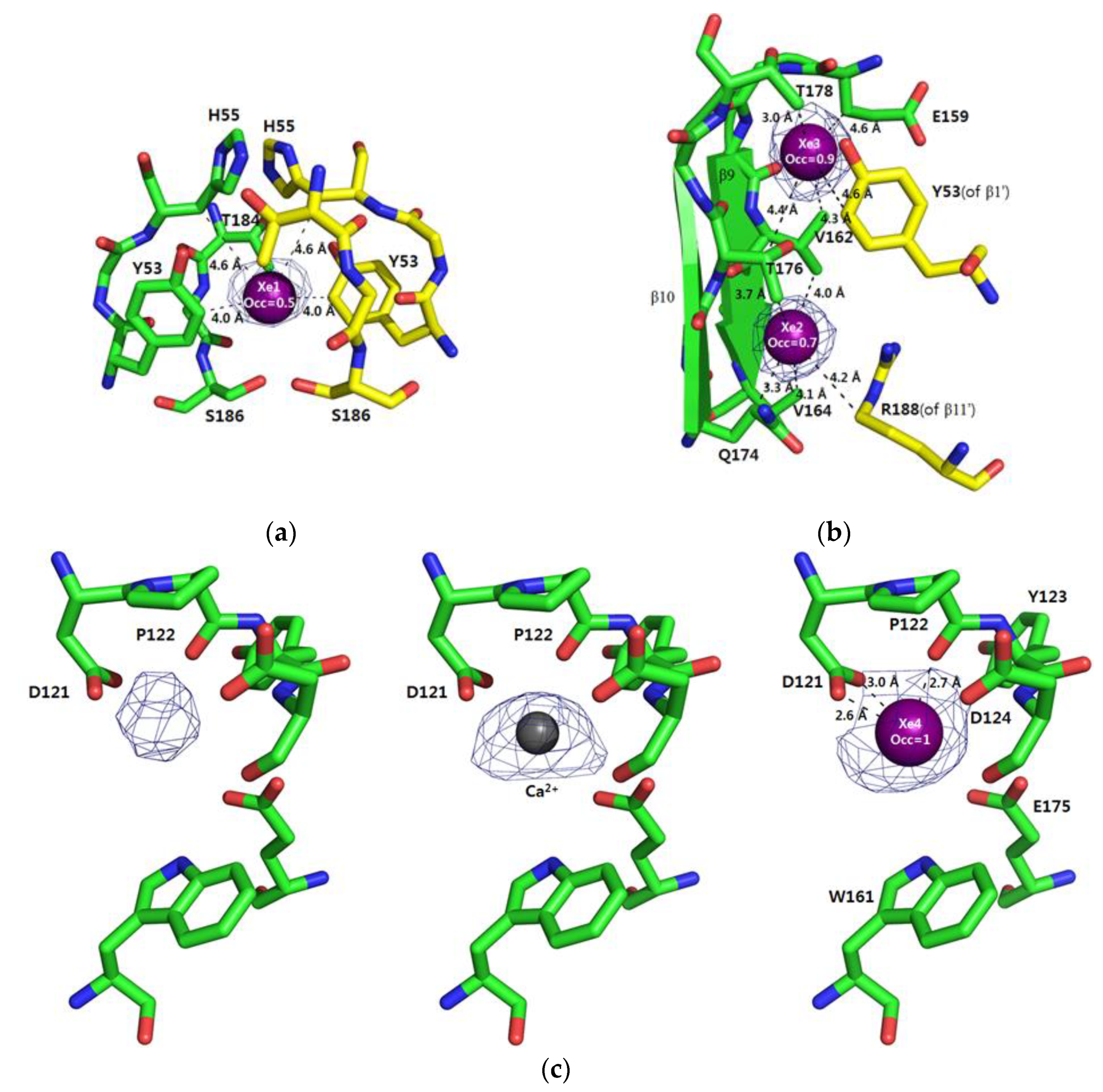
3.1. Xe1–3 in the Dimeric Interface
3.2. Xe4 in the Ca2+-Binding Site
Funding
Accession Number
Acknowledgments
Conflicts of Interest
References
- Prangé, T.; Schiltz, M.; Pernot, L.; Colloc’h, N.; Longhi, S.; Bourguet, W.; Fourme, R. Exploring hydrophobic sites in proteins with xenon or krypton. Proteins 1998, 30, 61–73. [Google Scholar] [CrossRef]
- Cohen, A.; Ellis, P.; Kresge, N.; Soltis, S.M. MAD phasing with krypton. Acta. Cryst. D 2001, 57, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Miyatake, H.; Park, S.-Y.; Kawamoto, M.; Kamiya, N.; Mikia, K. Multi-wavelength anomalous diffraction method for I and Xe atoms using ultra-high-energy X-rays from SPring-8. J. Appl. Cryst. 2004, 37, 925–933. [Google Scholar] [CrossRef]
- Cianci, M.; Rizkallah, P.J.; Olczak, A.; Raftery, J.; Chayen, N.E.; Zagalsky, P.F.; Helliwell, J.R. Structure of lobster apocrustacyanin A1 using softer X-rays. Acta Cryst. D 2001, 57, 1219–1229. [Google Scholar] [CrossRef] [PubMed]
- Olczak, A.; Cianci, M.; Hao, Q.; Rizkallah, P.J.; Raftery, J.; Helliwell, J.R. S-SWAT (softer single-wavelength anomalous technique): Potential in high-throughput protein crystallography. Acta Cryst. A 2003, 59, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Xiong, Z.J.; Li, J.; Zou, C.; Cairo, C.W.; Klassen, J.S.; Privé, G.G. Crystal structures of human lysosomal EPDR1 reveal homology with the superfamily of bacterial lipoprotein transporters. Commun. Biol. 2019, 2, 52. [Google Scholar] [CrossRef] [PubMed]
- Park, J.K.; Kim, K.Y.; Sim, Y.W.; Kim, Y.I.; Kim, J.K.; Lee, C.; Han, J.; Kim, C.U.; Lee, J.E.; Park, S. Structures of three ependymin-related proteins suggest their function as a hydrophobic molecule binder. IUCrJ. 2019, 6, 729–739. [Google Scholar] [CrossRef] [PubMed]
- Shashoua, V.E. Identification of specific changes in the pattern of brain protein synthesis after training. Science 1976, 193, 1264–1266. [Google Scholar] [CrossRef] [PubMed]
- McDougall, C.; Hammond, M.J.; Dailey, S.C.; Somorjai, I.M.L.; Cummins, S.F.; Degnan, B.M. The evolution of ependymin-related proteins. BMC Evol. Biol. 2018, 18, 182. [Google Scholar] [CrossRef] [PubMed]
- Sleat, D.E.; Sohar, I.; Lackland, H.; Majercak, J.; Lobel, P. Rat brain contains high levels of mannose-6-phosphorylated glycoproteins including lysosomal enzymes and palmitoyl-protein thioesterase, an enzyme implicated in infantile neuronal lipofuscinosis. J. Biol. Chem. 1996, 271, 19191–19198. [Google Scholar] [CrossRef] [PubMed]
- Della Valle, M.C.; Sleat, D.E.; Sohar, I.; Wen, T.; Pintar, J.E.; Jadot, M.; Lobel, P. Demonstration of lysosomal localization for the mammalian ependymin-related protein using classical approaches combined with a novel density shift method. J. Biol. Chem. 2006, 281, 35436–35445. [Google Scholar] [CrossRef] [PubMed]
- Hirano, S.; Asamizu, S.; Onaka, H.; Shiro, Y.; Nagano, S. Crystal structure of VioE, a key player in the construction of the molecular skeleton of violacein. J. Biol. Chem. 2008, 283, 6459–6466. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.S.; Balibar, C.J.; Turo, K.E.; Walsh, C.T.; Drennan, C.L. The violacein biosynthetic enzyme VioE shares a fold with lipoprotein transporter proteins. J. Biol. Chem. 2008, 283, 6467–6475. [Google Scholar] [CrossRef]
- Takeda, K.; Miyatake, H.; Yokota, N.; Matsuyama, S.; Tokuda, H.; Miki, K. Crystal structures of bacterial lipoprotein localization factors, LolA and LolB. EMBO J. 2003, 22, 3199–3209. [Google Scholar] [CrossRef]
- Park, J.K.; Sim, Y.W.; Park, S.Y. Over-expression, secondary structure characterization, and preliminary X-ray crystallographic analysis of Xenopus tropicalis ependymin. Crystals 2018, 8, 284. [Google Scholar] [CrossRef]
- Otwinowski, Z.; Minor, W. Processing of X-ray Diffraction Data Collected in Oscillation Mode. Methods Enzymol. 1997, 276, 307–326. [Google Scholar]
- Adams, P.D.; Afonine, P.V.; Bunkoczi, G.; Chen, V.B.; Davis, I.W.; Echols, N.; Headd, J.J.; Hung, L.-W.; Kapral, G.J.; Grosse-Kunstleve, R.W.; et al. PHENIX: A comprehensive Python-based system for macromolecular structure solution. Acta. Cryst. D Biol. Cryst. 2001, 66, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Emsley, P.; Cowtan, K. Coot: Model-building tools for molecular graphics. Acta. Cryst. D Biol. Cryst. 2004, 60, 2126–2132. [Google Scholar] [CrossRef] [PubMed]
- Vagin, A.A.; Steiner, R.A.; Lebedev, A.A.; Potterton, L.; McNicholas, S.; Long, F.; Murshudov, G.N. REFMAC5 dictionary: Organisation of prior chemical knowledge and guidelines for its use. Acta. Cryst. D 2004, 60, 2284–2295. [Google Scholar] [CrossRef] [PubMed]

| Data Collection | |
| Diffraction source | Pohang Light Source (PLS 7A, Pohang, Korea) |
| Space group | P6522 |
| Detector | ADSC Q270 |
| Wavelength (Å) | 1.54 |
| Oscillation(°)/Frame | 1/360 |
| Cell dimensions | 61.46 Å, 61.45 Å, 233.84 Å, 90°, 90°, 120° |
| Resolution (Å) | 50−2.90 (2.95−2.90)1 |
| Rsym (%) | 9.7 (58.2) |
| Rp.i.m. (%) | 2.1 (12.5) |
| CC1/2 | (0.976) |
| I/σ(I) | 71.0 (10.5) |
| Completeness (%) | 99.9 (100.0) |
| Redundancy | 21.9 (22.5) |
| Unique reflections | 6,390 |
| Overall B factor from Wilson plot (Å2) | 63.6 |
| Refinement | |
| Resolution (Å) | 2.90 |
| NCS molecules in AU | 1 |
| Rwork/Rfree (%) | 0.219/0.308 |
| No. atoms | |
| Protein | 1,514 |
| Ligand/ion | 4 Xe |
| B-factors | |
| Protein (main/side chain) | 69.9/76.6 |
| Xe | 135.6 |
| R.m.s. deviations | |
| Bond lengths (Å) | 0.011 |
| Bond angles (°) | 1.500 |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S. De novo Phasing Xenons Observed in the Frog Ependymin-Related Protein. Crystals 2020, 10, 32. https://doi.org/10.3390/cryst10010032
Park S. De novo Phasing Xenons Observed in the Frog Ependymin-Related Protein. Crystals. 2020; 10(1):32. https://doi.org/10.3390/cryst10010032
Chicago/Turabian StylePark, SangYoun. 2020. "De novo Phasing Xenons Observed in the Frog Ependymin-Related Protein" Crystals 10, no. 1: 32. https://doi.org/10.3390/cryst10010032
APA StylePark, S. (2020). De novo Phasing Xenons Observed in the Frog Ependymin-Related Protein. Crystals, 10(1), 32. https://doi.org/10.3390/cryst10010032




