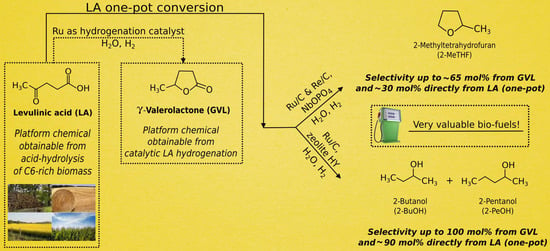
Cascade Strategy for the Tunable Catalytic Valorization of Levulinic Acid and γ-Valerolactone to 2-Methyltetrahydrofuran and Alcohols
Abstract
1. Introduction
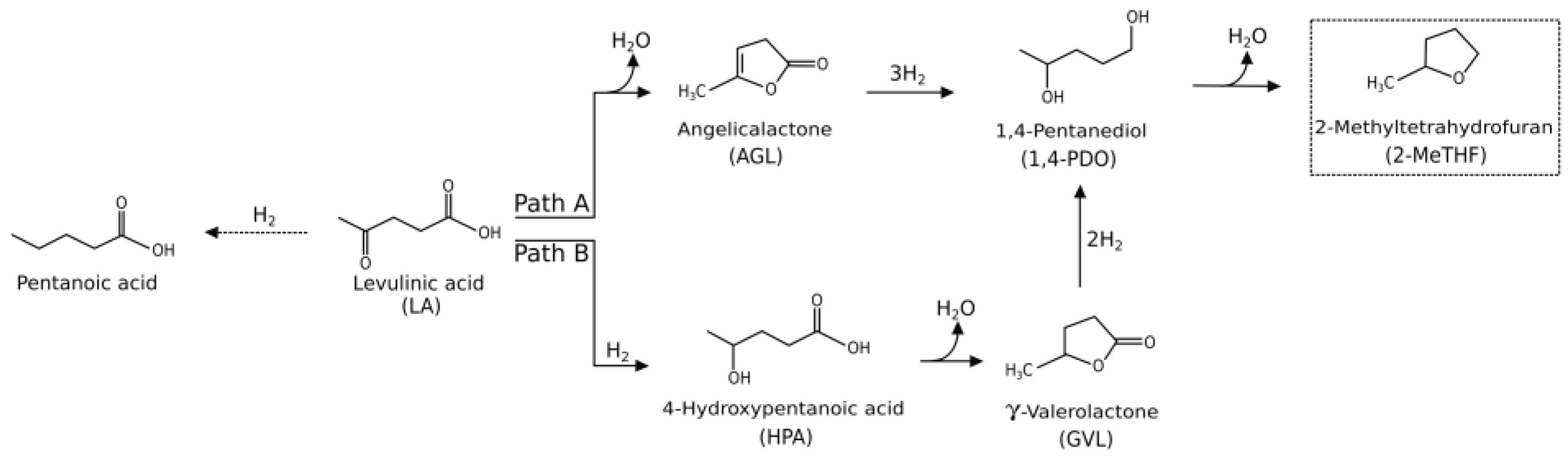
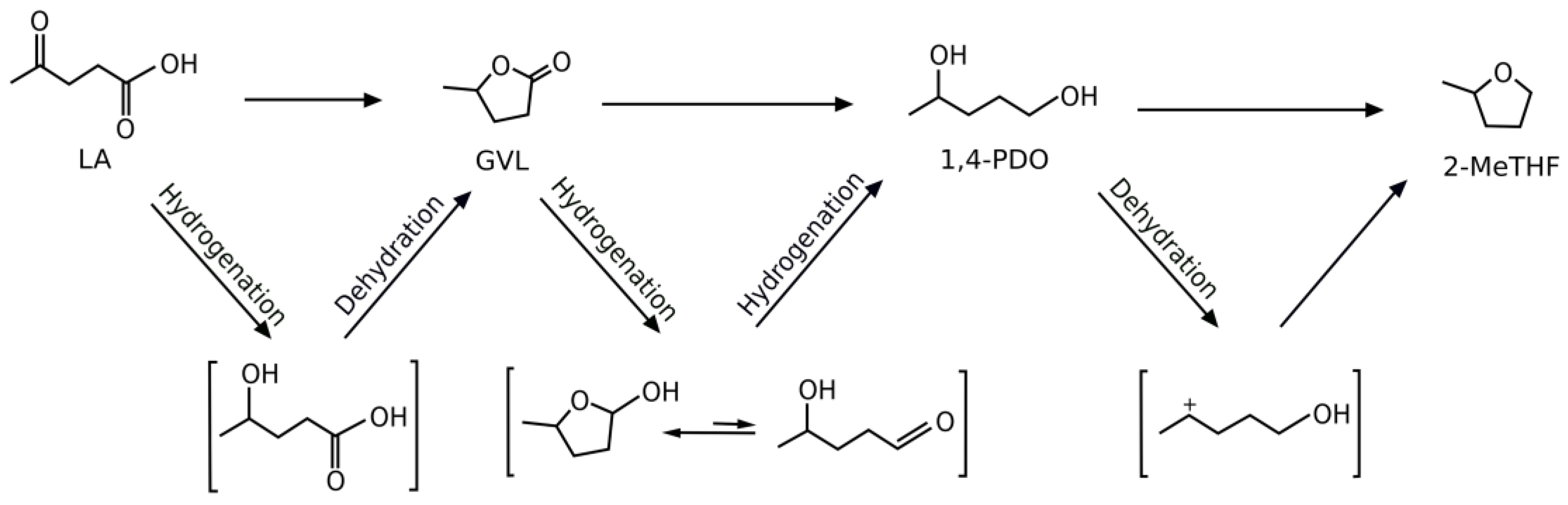
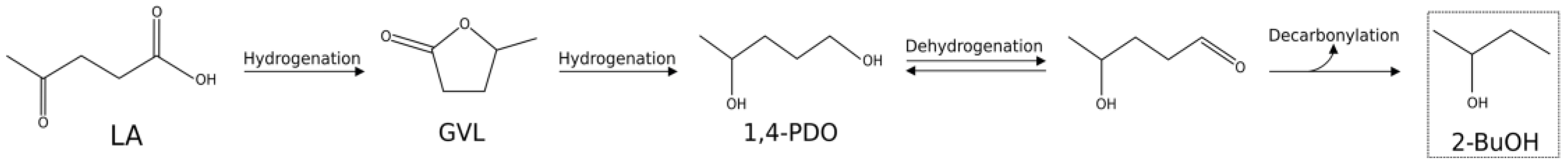
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Hydrogenation Reactions
3.3. Analysis of the Reaction Products
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Shylesh, S.; Gokhale, A.A.; Ho, C.R.; Bell, A.T. Novel strategies for the production of fuels, lubricants, and chemicals from biomass. Acc. Chem. Res. 2017, 50, 2589–2597. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, G.; Ripa, M.; Ulgiati, S. Chemicals from biomass: Technological versus environmental feasibility. A review. Biofuels Bioprod. Biorefin. 2017, 11, 195–214. [Google Scholar] [CrossRef]
- Mariscal, R.; Maireles-Torres, P.; Ojeda, M.; Sádaba, I.; López Granados, M. Furfural: A renewable and versatile platform molecule for the synthesis of chemicals and fuels. Energy Environ. Sci. 2016, 9, 1144–1189. [Google Scholar] [CrossRef]
- Li, X.; Jia, P.; Wang, T. Furfural: A promising platform compound for sustainable production of C4 and C5 chemicals. ACS Catal. 2016, 6, 7621–7640. [Google Scholar] [CrossRef]
- Yu, I.K.M.; Tsang, D.C.W. Conversion of biomass to hydroxymethylfurfural: A review of catalytic systems and underlying mechanisms. Bioresour. Technol. 2017, 238, 716–732. [Google Scholar] [CrossRef] [PubMed]
- Antonetti, C.; Raspolli Galletti, A.M.; Fulignati, S.; Licursi, D. Amberlyst A-70: A surprisingly active catalyst for the MW-assisted dehydration of fructose and inulin to HMF in water. Catal. Commun. 2017, 97, 146–150. [Google Scholar] [CrossRef]
- Antonetti, C.; Melloni, M.; Licursi, D.; Fulignati, S.; Ribechini, E.; Rivas, S.; Parajó, J.C.; Cavani, F.; Raspolli Galletti, A.M. Microwave-assisted dehydration of fructose and inulin to HMF catalyzed by Niobium and Zirconium phosphate catalysts. Appl. Catal. B 2018, 206, 364–377. [Google Scholar] [CrossRef]
- Girisuta, B.; Heeres, H.J. Levulinic acid from biomass: Synthesis and applications. In Production of Platform Chemicals from Sustainable Resources; Fang, Z., Smith, R.L., Qi, X., Eds.; Springer: Singapore, 2017; Chapter 5; pp. 143–169. ISBN 978-9811041716. [Google Scholar]
- Antonetti, C.; Licursi, D.; Fulignati, S.; Valentini, G.; Raspolli Galletti, A.M. New frontiers in the catalytic synthesis of levulinic acid: From sugars to raw and waste biomass as starting feedstock. Catalysts 2016, 6, 196. [Google Scholar] [CrossRef]
- Yan, K.; Yang, Y.; Chai, J.; Lu, Y. Catalytic reactions of gamma-valerolactone: A platform to fuels and value-added chemicals. Appl. Catal. B 2015, 179, 292–304. [Google Scholar] [CrossRef]
- Zhang, Z. Synthesis of γ-Valerolactone from carbohydrates and its applications. ChemSusChem 2016, 9, 156–171. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Yao, Q.; Fu, Y. Conversion of levulinic acid and alkyl levulinates to biofuels and high-value chemicals. Green Chem. 2017, 19, 5527–5547. [Google Scholar] [CrossRef]
- Horváth, I.T.; Mehdi, H.; Fábos, V.; Boda, L.; Mika, L.T. γ-Valerolactone-A sustainable liquid for energy and carbon-based chemicals. Green Chem. 2008, 10, 238–242. [Google Scholar] [CrossRef]
- Bond, J.Q.; Alonso, D.M.; Wang, D.; West, R.M.; Dumesic, J.A. Integrated catalytic conversion of γ-valerolactone to liquid alkenes for transportation fuels. Science 2010, 327, 1110–1114. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Zeng, X.; Li, Z.; Hu, L.; Sun, Y.; Liu, S.; Lei, T.; Lin, L. Production of γ-valerolactone from lignocellulosic biomass for sustainable fuels and chemicals supply. Renew. Sustain. Energy Rev. 2014, 40, 608–620. [Google Scholar] [CrossRef]
- Upare, P.P.; Lee, J.M.; Hwang, D.W.; Halligudi, S.B.; Hwang, Y.K.; Chang, J.S. Selective hydrogenation of levulinic acid to γ-valerolactone over carbon-supported noble metal catalysts. J. Ind. Eng. Chem. 2011, 17, 287–292. [Google Scholar] [CrossRef]
- Yan, K.; Lafleur, T.; Wu, G.; Liao, J.; Ceng, C.; Xie, X. Highly selective production of value-added γ-valerolactone from biomass-derived levulinic acid using the robust Pd nanoparticles. Appl. Catal. A 2013, 468, 52–58. [Google Scholar] [CrossRef]
- Sudholt, A.; Tripathi, R.; Mayer, D.; Glaude, P.A.; Battin-Leclerc, F.; Pitsch, H. The oxidation of the novel lignocellulosic biofuel γ-valerolactone in a low pressure flame. Proc. Combust. Inst. 2017, 36, 577–585. [Google Scholar] [CrossRef]
- Lilga, M.A.; Padmaperuma, A.B.; Auberry, D.L.; Job, H.M.; Swita, M.S. Ketonization of levulinic acid and γ-valerolactone to hydrocarbon fuel precursors. Catal. Today 2018, 302, 80–86. [Google Scholar] [CrossRef]
- Fegyverneki, D.; Orha, L.; Láng, G.; Horváth, I.T. Gamma-valerolactone-based solvents. Tetrahedron 2010, 66, 1078–1081. [Google Scholar] [CrossRef]
- Yan, K.; Lafleur, T.; Wu, X.; Chai, J.; Wu, C.; Xie, X. Cascade upgrading of γ-valerolactone to biofuels. Chem. Commun. 2015, 51, 6984–6987. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Ruiz, J.C.; Wang, D.; Dumesic, J.A. Catalytic upgrading of levulinic acid to 5-nonanone. Green Chem. 2010, 12, 574–577. [Google Scholar] [CrossRef]
- Wang, A.; Lu, Y.; Yi, Z.; Ejaz, A.; Hu, K.; Zhang, L.; Yan, K. Selective production of γ-valerolactone and valeric acid in one-pot bifunctional metal catalysts. ChemistrySelect 2018, 3, 1097–1101. [Google Scholar] [CrossRef]
- Raspolli Galletti, A.M.; Antonetti, C.; Ribechini, E.; Colombini, M.P.; Nassi, N.; Bonari, E. From giant reed to levulinic acid and gamma-valerolactone: A high yield catalytic route to valeric biofuels. Appl. Energy 2013, 102, 157–162. [Google Scholar] [CrossRef]
- Antonetti, C.; Bonari, E.; Licursi, D.; Nassi, N.; Raspolli Galletti, A.M. Hydrothermal conversion of giant reed to furfural and levulinic acid: Optimization of the process under microwave irradiation and investigation of distinctive agronomic parameters. Molecules 2015, 20, 21232–21253. [Google Scholar] [CrossRef] [PubMed]
- Rivas, S.; Raspolli Galletti, A.M.; Antonetti, C.; Licursi, D.; Santos, V.; Parajó, J.C. A biorefinery cascade conversion of hemicellulose-free Eucalyptus Globulus wood: Production of concentrated levulinic acid solutions for γ-Valerolactone sustainable preparation. Catalysts 2018, 8, 169. [Google Scholar] [CrossRef]
- Pace, V.; Hoyos, P.; Castoldi, L.; De María, P.D.; Alcántara, A.R. 2-Methyltetrahydrofuran (2-MeTHF): A biomass-derived solvent with broad application in organic chemistry. ChemSusChem 2012, 5, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Climent, M.J.; Corma, A.; Iborra, S. Conversion of biomass platform molecules into fuel additives and liquid hydrocarbon fuels. Green Chem. 2014, 16, 516–547. [Google Scholar] [CrossRef]
- Kar, Y.; Deveci, H. Importance of p-series fuels for flexible-fuel vehicles (FFVs) and alternative fuels. Energy Source A Recovery Util. Environ. Eff. 2006, 28, 909–921. [Google Scholar] [CrossRef]
- Da Silva Trindade, W.R.; Dos Santos, R.G. Review on the characteristics of butanol, its production and use as fuel in internal combustion engines. Renew. Sustain. Energy Rev. 2017, 69, 642–651. [Google Scholar] [CrossRef]
- Stevens, J.G.; Bourne, R.A.; Twigg, M.V.; Poliakoff, M. Real-time product switching using a twin catalyst system for the hydrogenation of furfural in supercritical CO2. Angew. Chem. Int. Ed. 2010, 49, 8856–8859. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Zhu, Y.; Ding, G.; Cui, J.; Li, X.; Li, Y. One-step conversion of furfural into 2-methyltetrahydrofuran under mild conditions. ChemSusChem 2015, 8, 1534–1537. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Liu, A.F.; Cai, B.; Luo, J.Y.; Pan, H.; Huang, Y.B. Catalytic transfer hydrogenation of furfural to 2-methylfuran and 2-methyltetrahydrofuran over bimetallic copper-palladium catalysts. ChemSusChem 2016, 9, 3330–3337. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Liao, J.; Wu, X.; Xie, X. A noble-metal free Cu-catalyst derived from hydrotalcite for highly efficient hydrogenation of biomass-derived furfural and levulinic acid. RSC Adv. 2013, 3, 3853–3856. [Google Scholar] [CrossRef]
- Christian, R.V.J.; Brown, H.D.; Hixon, R.M. Derivatives of γ-valerolactone, 1,4-pentanediol and 1,4-di-(β-cyanoethoxy)-pentane. J. Am. Chem. Soc. 1947, 69, 1961–1963. [Google Scholar] [CrossRef]
- Elliot, D.; Frye, J.G. Hydrogenated 5-Carbon Compound and Method of Making. U.S. Patent 5883266, 16 March 1999. [Google Scholar]
- Mehdi, H.; Fàbos, V.; Tuba, R.; Bodor, A.; Mika, L.T.; Horvàth, I.T. Integration of homogeneous and heterogeneous catalytic processes for a multi-step conversion of biomass: From sucrose to levulinic acid, γ-valerolactone, 1,4-pentanediol, 2-methyl-tetrahydrofuran, and alkanes. Top. Catal. 2008, 48, 49–54. [Google Scholar] [CrossRef]
- Geilen, F.M.A.; Engendahl, B.; Harwardt, A.; Marquardt, W.; Klankermayer, J.; Leitner, W. Selective and flexible transformation of biomass-derived platform chemicals by a multifunctional catalytic system. Angew. Chem. Int. Ed. 2010, 49, 5510–5514. [Google Scholar] [CrossRef] [PubMed]
- Upare, P.P.; Lee, J.M.; Hwang, Y.K.; Hwang, D.W.; Lee, J.H.; Halligudi, S.B.; Hwang, J.S.; Chang, J.S. Direct hydrocyclization of biomass-derived levulinic acid to 2-methyltetrahydrofuran over nanocomposite copper/silica catalysts. ChemSusChem 2011, 4, 1749–1752. [Google Scholar] [CrossRef] [PubMed]
- Du, X.L.; Bi, Q.Y.; Liu, Y.M.; Cao, Y.; He, H.Y.; Fan, K.N. Tunable copper-catalyzed chemoselective hydrogenolysis of biomass-derived γ-valerolactone into 1,4-pentanediol or 2-methyltetrahydrofuran. Green Chem. 2012, 14, 935–939. [Google Scholar] [CrossRef]
- Bermudez, J.M.; Menendez, J.A.; Romero, A.A.; Serrano, E.; Garcia-Martinez, J.; Luque, R. Continuous flow nanocatalysis: Reaction pathways in the conversion of levulinic acid to valuable chemicals. Green Chem. 2013, 15, 2786–2792. [Google Scholar] [CrossRef]
- Al-Shaal, M.G.; Dzierbinski, A.; Palkovits, R. Solvent-free γ-valerolactone hydrogenation to 2-methyltetrahydrofuran catalysed by Ru/C: A reaction network analysis. Green Chem. 2014, 16, 1358–1364. [Google Scholar] [CrossRef]
- Mizugaki, T.; Togo, K.; Maeno, Z.; Mitsudome, T.; Jitsukawa, K.; Kaneda, K. One-pot transformation of levulinic acid to 2-methyltetrahydrofuran catalyzed by Pt−Mo/H-β in water. ACS Sustain. Chem. Eng. 2016, 4, 682–685. [Google Scholar] [CrossRef]
- Upare, P.P.; Lee, M.; Lee, S.K.; Yoon, J.W.; Bae, J.; Hwang, D.W.; Lee, UH.; Chang, J.S.; Hwang, Y.K. Ru nanoparticles supported graphene oxide catalyst for hydrogenation of bio-based levulinic acid to cyclic ethers. Catal. Today 2016, 265, 174–183. [Google Scholar] [CrossRef]
- Obregón, I.; Gandarias, I.; Al-Shaal, M.G.; Mevissen, C.; Arias, P.L.; Palkovits, R. The role of the hydrogen source on the selective production of γ-valerolactone and 2-methyltetrahydrofuran from levulinic acid. ChemSusChem 2016, 9, 2488–2495. [Google Scholar] [CrossRef]
- Thomas, J.J.; Barile, R.G. Conversion of cellulose hydrolysis products to fuel and chemical feedstocks. Biomass Wastes 1985, 8, 1461–1494. [Google Scholar]
- Al-Shaal, M.G.; Hausoul, P.J.C.; Palkovits, R. Efficient, solvent-free hydrogenation of α-angelica lactone catalysed by Ru/C at atmospheric pressure and room temperature. Chem. Commun. 2014, 50, 10206–10209. [Google Scholar] [CrossRef] [PubMed]
- Al-Shaal, M.G.; Wright, W.R.H.; Palkovits, R. Exploring the ruthenium catalysed synthesis of γ-valerolactone in alcohols and utilisation of mild solvent-free reaction conditions. Green Chem. 2012, 14, 1260–1263. [Google Scholar] [CrossRef]
- Abdelrahman, O.A.; Heyden, A.; Bond, J.Q. Analysis of kinetics and reaction pathways in the aqueous-phase hydrogenation of levulinic acid to form γ-valerolactone over Ru/C. ACS Catal. 2014, 4, 1171–1181. [Google Scholar] [CrossRef]
- Zheng, J.; Zhu, J.; Xu, X.; Wang, W.; Li, J.; Zhao, Y.; Tang, K.; Song, Q.; Qi, X.; Kong, D.; et al. Continuous hydrogenation of ethyl levulinate to γ-valerolactone and 2-methyl tetrahydrofuran over alumina doped Cu/SiO2 catalyst: The potential of commercialization. Sci. Rep. 2016, 6, 28898–28907. [Google Scholar] [CrossRef] [PubMed]
- Obregón, I.; Gandarias, I.; Miletic´, N.; Ocio, A.; Arias, P.L. One-pot 2-methyltetrahydrofuran production from levulinic acid in green solvents using Ni-Cu/Al2O3 catalysts. ChemSusChem 2015, 8, 3483–3488. [Google Scholar] [CrossRef] [PubMed]
- Assima, G.P.; Zamboni, I.; Lavoie, J.M. Alcohol fuels: The thermochemical route. In Biofuels Production and Processing Technology; Riazi, R.M., Chiaramonti, D., Eds.; CRC Press: Boca Raton, FL, USA, 2018; Chapter 14; pp. 362–406. ISBN 978-1-4987-7893-0. [Google Scholar]
- Shabaker, J.W.; Weiner, A.; Rundell, D.N.; Sejour, H.; Goeden, G. Unit, Method, and Renewable Materials. U.S. Patent 20100281763A1, 11 November 2010. [Google Scholar]
- Lv, J.; Rong, Z.; Sun, L.; Liu, C.; Lu, A.H.; Wang, Y.; Qu, J. Catalytic conversion of biomass-derived levulinic acid into alcohols over nanoporous Ru catalyst. Catal. Sci. Technol. 2018, 8, 975–979. [Google Scholar] [CrossRef]
- Raspolli Galletti, A.M.; Antonetti, C.; De Luise, V.; Martinelli, M. A sustainable process for the production of γ-valerolactone by hydrogenation of biomass-derived levulinic acid. Green Chem. 2012, 12, 688–694. [Google Scholar] [CrossRef]
- Corbel-Demailly, L.; Ly, B.K.; Minh, D.P.; Tapin, B.; Especel, C.; Epron, F.; Cabiac, A.; Guillon, E.; le Besson, M.; Pinel, C. Heterogeneous catalytic hydrogenation of biobased levulinic and succinic acids in aqueous solutions. ChemSusChem 2013, 6, 2388–2395. [Google Scholar] [CrossRef] [PubMed]
- Buitrago-Sierra, R.; Serrano-Ruiz, J.C.; Rodríguez-Reinoso, F.; Sepúlveda-Escribano, A.; Dumesic, J.A. Ce promoted Pd-Nb catalysts for γ-valerolactone ring-opening and hydrogenation. Green Chem. 2012, 14, 3318–3324. [Google Scholar] [CrossRef]
- Luo, W.; Deka, U.; Beale, A.M.; Van Eck, E.R.H.; Bruijnincx, P.C.A.; Weckhuysen, B.M. Ruthenium-catalyzed hydrogenation of levulinic acid: Influence of the support and solvent on catalyst selectivity and stability. J. Catal. 2013, 301, 175–186. [Google Scholar] [CrossRef]
- Prati, L.; Jouve, A.; Villa, A. Production and upgrading of γ-valerolactone with bifunctional catalytic processes. In Production of Biofuels and Chemicals with Bifunctional Catalysts, 1st ed.; Fang, Z., Smith, R.L., Li, H., Eds.; Springer: Singapore, 2017; Volume 8, pp. 221–237. ISBN 978-981-10-5136-4. [Google Scholar]
- Freitas, F.A.; Licursi, D.; Lachter, E.R.; Raspolli Galletti, A.M.; Antonetti, C.; Brito, T.C.; Nascimento, R.S.V. Heterogeneous catalysis for the ketalisation of ethyl levulinate with 1,2-dodecanediol: Opening the way to a new class of bio-degradable surfactants. Catal. Commun. 2016, 73, 84–87. [Google Scholar] [CrossRef]
- Raspolli Galletti, A.M.; Carlini, C.; Sbrana, G.; Armaroli, T.; Busca, G. Selective saccharides dehydration to 5-hydroxymethyl-2-furaldehyde by heterogeneous niobium catalysts. Appl. Catal. A 1999, 183, 295–302. [Google Scholar] [CrossRef]
- Amaroli, T.; Busca, G.; Carlini, C.; Giuttari, M.; Raspolli Galletti, A.M.; Sbrana, G. Active acid sites characterization of niobium phosphate catalysts and their activity in fructose dehydration to 5-hydroxymethyl-2-furaldehyde. J. Mol. Catal. A: Chem. 2000, 151, 233–243. [Google Scholar] [CrossRef]
- Bond, J.Q.; Alonso, D.M.; West, R.M.; Dumesic, J.A. γ-Valerolactone ring-opening and decarboxylation over SiO2/Al2O3 in the presence of water. Langmuir 2010, 26, 16291–16298. [Google Scholar] [CrossRef] [PubMed]
- Kellicutt, A.B.; Salary, R.; Abdelrahman, O.A.; Bond, J.Q. An examination of the intrinsic activity and stability of various solid acids during the catalytic decarboxylation of γ-valerolactone. Catal. Sci. Technol. 2014, 4, 2267–2279. [Google Scholar] [CrossRef]
- Villa, A.; Schiavoni, M.; Chan-Thaw, C.E.; Fulvio, P.F.; Mayes, R.T.; Dai, S.; More, K.L.; Veith, G.M.; Prati, L. Acid-functionalized mesoporous carbon: An efficient support for ruthenium-catalyzed γ-valerolactone production. ChemSusChem 2015, 8, 2520–2528. [Google Scholar] [CrossRef] [PubMed]
- Busca, G. Acidity and basicity of zeolites: A fundamental approach. Microporous Mesoporous Mater. 2017, 254, 3–16. [Google Scholar] [CrossRef]
- Ravenelle, R.M.; Schüβler, F.; D’Amico, A.; Danilina, N.; Van Bokhoven, J.A.; Lercher, J.A.; Jones, C.W.; Sievers, C. Stability of zeolites in hot liquid water. J. Phys. Chem. C 2010, 114, 19582–19595. [Google Scholar] [CrossRef]
- Pine, L.A. Decarboxylation. U.S. Patent 3476803A, 4 November 1969. [Google Scholar]
- Di, X.; Li, C.; Zhang, B.; Qi, J.; Li, W.; Su, D.; Liang, C. Role of Re and Ru in Re. Ru/C bimetallic catalysts for the aqueous hydrogenation of succinic Acid. Ind. Eng. Chem. Res. 2017, 56, 4672–4683. [Google Scholar] [CrossRef]
- Van Borm, R.; Reyniers, M.F.; Martens, J.A.; Marin, G.B. Catalytic cracking of methylcyclohexane on FAU, MFI, and bimodal porous materials: Influence of acid properties and pore topology. Ind. Eng. Chem. Res. 2010, 49, 10486–10495. [Google Scholar] [CrossRef]
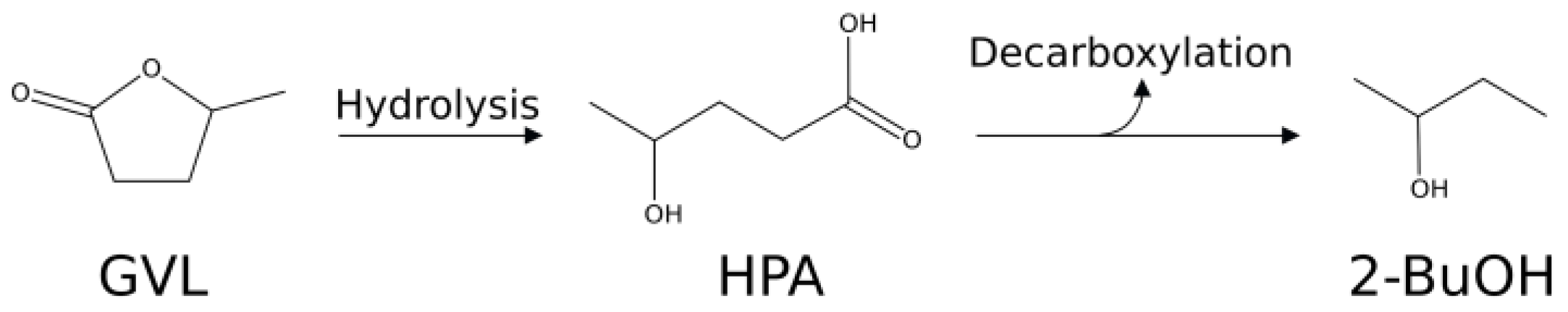
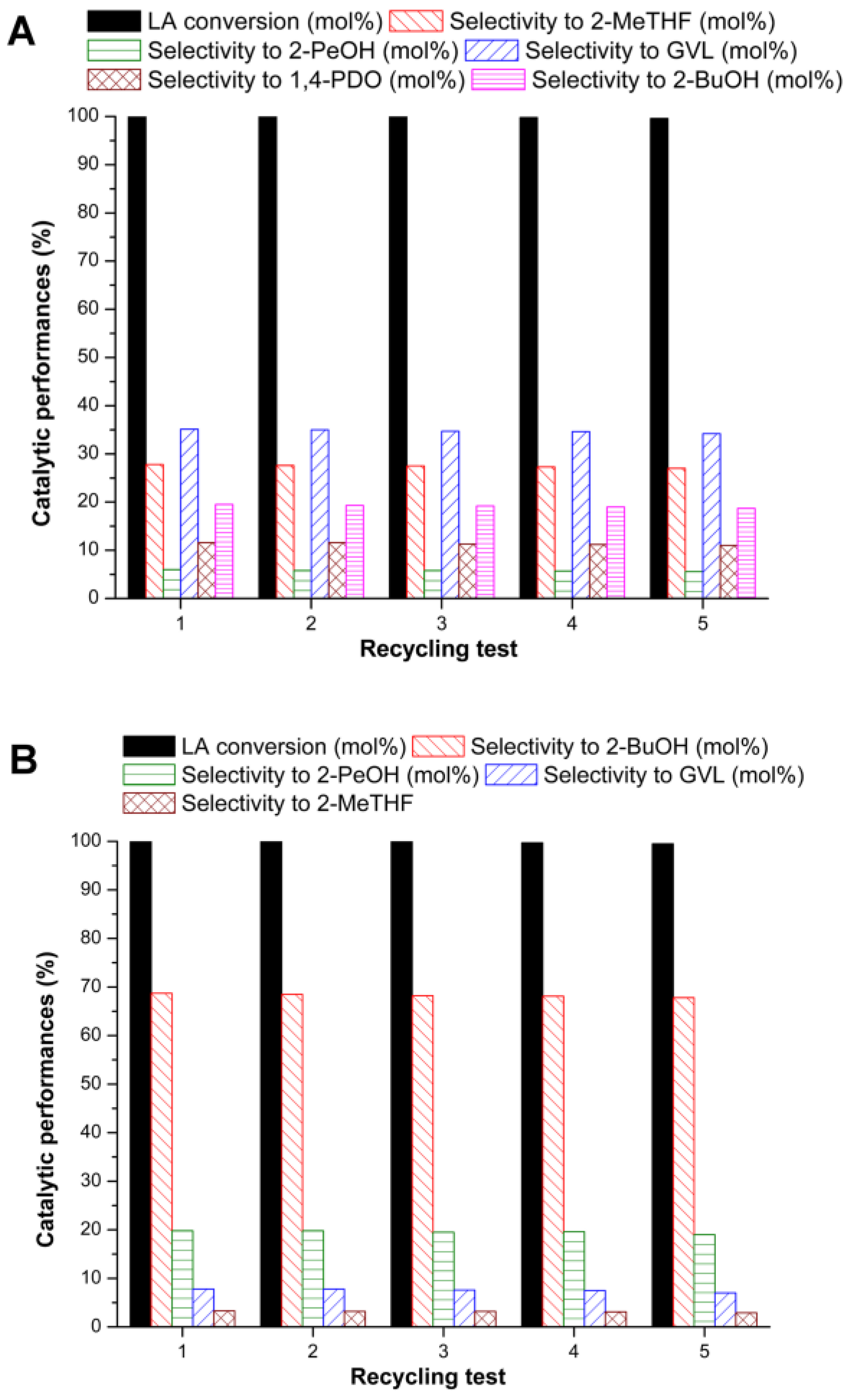
| Run | Catalytic System | GVL/Re mol/mol | T (°C) | t [h] | Conv. GVL (mol %) | Selectivity (mol %) | |||
|---|---|---|---|---|---|---|---|---|---|
| 2-MeTHF | 2-BuOH | 2-PeOH | 1,4-PDO | ||||||
| 1 | Ru/C 5% | - | 210 | 6 | traces | - | - | - | - |
| 2 | Ru/C 5% + NBP (1 g) | - | 200 | 3 | 31.7 | 35.2 | 42.2 | 5.7 | 12.2 |
| 3 | Ru/C 5% + NBP (1 g) | - | 200 | 6 | 40.5 | 38.9 | 34.0 | 6.5 | 14.0 |
| 4 | Ru/C 5% + NBP (1 g) | - | 180 | 6 | 3.6 | 49.2 | 27.0 | 0.1 | 5.4 |
| 5 | Ru/C 5% + NBP (1 g) | - | 210 | 6 | 71.1 | 21.0 | 66.8 | 7.7 | 0.4 |
| 6 | Re/C 10% (10 mg) | 312.33 | 210 | 6 | traces | - | - | - | - |
| 7 | Ru/C 5% + Re/C 10% (5 mg) + NBP (1 g) | 624.66 | 200 | 3 | 35.9 | 43.0 | 31.5 | 8.7 | 14.1 |
| 8 | Ru/C 5% + Re/C 10% (10 mg) + NBP (1 g) | 312.33 | 200 | 3 | 36.3 | 47.2 | 23.0 | 6.1 | 18.8 |
| 9 | Ru/C 5% + Re/C 10% (20 mg) + NBP (1g) | 156.16 | 200 | 3 | 36.1 | 57.5 | 11.1 | 4.6 | 21.3 |
| 10 | Ru/C 5% + Re/C 10% (20 mg), no NBP | 156.16 | 200 | 3 | 27.3 | 22.1 | 6.1 | 3.6 | 63.8 |
| 11 | Ru/C 5% + Re/C 10% (20 mg) + NBP (250 mg) | 156.16 | 200 | 3 | 30.8 | 44.6 | 7.1 | 5.1 | 30.2 |
| 12 | Ru/C 5% + Re/C 10% (20 mg) + NBP (500 mg) | 156.16 | 200 | 3 | 39.3 | 64.9 | 7.3 | 3.7 | 21.5 |
| Run | Catalyst and co-catalyst | Reaction conditions T, PH2 | Conv. GVL (mol %) | Sel. 2-MeTHF (mol %) | Sel. 2-BuOH (mol %) | Sel. 2-PeOH (mol %) | Sel. 1,4-PDO (mol %) |
|---|---|---|---|---|---|---|---|
| 13 | 5% Ru/C NBP | 200 °C, 9.0 MPa | 98.9 | 8.5 | 75.7 | 15.8 | - |
| 14 | 5% Ru/C NBP | 200 °C, 5.0 MPa | 77.9 | 5.7 | 83.3 | 10.9 | 0.1 |
| 15 | 5% Ru/C NBP | 180 °C, 5.0 MPa | 26.6 | 9.7 | 60.6 | 4.2 | 20.7 |
| 16 | 5% Ru/C 10% Re/C NBP | 180 °C, 5.0 MPa | 48.3 | 37.8 | 36.6 | 7.5 | 18.1 |
| 17 | 5% Ru/C HY | 180 °C 5.0 MPa | 66.3 | 21.3 | 63.3 | 13.8 | 1.6 |
| 18 | 5% Ru/C HY | 200 °C 3.0 MPa | 100 | - | 81.3 | 18.7 | - |
| 19 | 5% Ru/C 10% Re/C HY | 200 °C 3.0 MPa | 100 | 35.7 | 47.6 | 16.7 | - |
| Run | Catalyst and Co-Catalyst | T (°C) | Conv. LA (mol %) | Sel. GVL (mol %) | Sel. 2-MeTHF (mol %) | Sel. 2-BuOH (mol %) | Sel. 2-PeOH (mol %) | Sel. 1,4-PDO (mol %) |
|---|---|---|---|---|---|---|---|---|
| 20 | 5% Ru/C, NBP | 200 | 99.7 | 73.4 | 8.8 | 9.9 | 0.9 | 1.3 |
| 21 | 5% Ru/C, NBP | 210 | 99.9 | 70.6 | 8.7 | 13.8 | 1.5 | 1.7 |
| Run | Catalyst and Co-Catalyst | Reaction Conditions T, PH2 | Conv. LA (mol %) | Sel. GVL (mol %) | Sel. 2-MeTHF (mol %) | Sel. 2-BuOH (mol %) | Sel. 2-PeOH (mol %) | Sel. 1,4-PDO (mol %) |
|---|---|---|---|---|---|---|---|---|
| 22 | 5%Ru/C NBP | 180 °C, 5.0 MPa | 100 | 36.2 | 10.1 | 37.8 | 13.0 | 2.9 |
| 23 | 5%Ru/C HY | 180 °C, 5.0 MPa | 100 | 30.8 | 3.8 | 35.0 | 28.7 | 1.7 |
| 24 | 5%Ru/C HY | 180 °C, 3.0 MPa | 100 | 46.8 | 3.6 | 36.5 | 13.1 | - |
| 25 | 5%Ru/C HY | 180 °C, 1.0 MPa | 100 | 81.1 | 0.6 | 13.6 | 4.7 | - |
| 26 | 5%Ru/C HY | 200 °C, 3.0 MPa | 100 | 7.9 | 3.3 | 68.9 | 19.9 | - |
| 27 | 5%Ru/C NBP | 200 °C 3.0 MPa | 100 | 25.8 | 7.0 | 52.0 | 15.2 | - |
| 28 | 5%Ru/C10%R/C, NBP | 180 °C 5.0 MPa | 100 | 35.1 | 27.8 | 19.5 | 6.0 | 11.6 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Licursi, D.; Antonetti, C.; Fulignati, S.; Giannoni, M.; Raspolli Galletti, A.M. Cascade Strategy for the Tunable Catalytic Valorization of Levulinic Acid and γ-Valerolactone to 2-Methyltetrahydrofuran and Alcohols. Catalysts 2018, 8, 277. https://doi.org/10.3390/catal8070277
Licursi D, Antonetti C, Fulignati S, Giannoni M, Raspolli Galletti AM. Cascade Strategy for the Tunable Catalytic Valorization of Levulinic Acid and γ-Valerolactone to 2-Methyltetrahydrofuran and Alcohols. Catalysts. 2018; 8(7):277. https://doi.org/10.3390/catal8070277
Chicago/Turabian StyleLicursi, Domenico, Claudia Antonetti, Sara Fulignati, Michael Giannoni, and Anna Maria Raspolli Galletti. 2018. "Cascade Strategy for the Tunable Catalytic Valorization of Levulinic Acid and γ-Valerolactone to 2-Methyltetrahydrofuran and Alcohols" Catalysts 8, no. 7: 277. https://doi.org/10.3390/catal8070277
APA StyleLicursi, D., Antonetti, C., Fulignati, S., Giannoni, M., & Raspolli Galletti, A. M. (2018). Cascade Strategy for the Tunable Catalytic Valorization of Levulinic Acid and γ-Valerolactone to 2-Methyltetrahydrofuran and Alcohols. Catalysts, 8(7), 277. https://doi.org/10.3390/catal8070277