Conversion of Furans by Baeyer-Villiger Monooxygenases
Abstract
:1. Introduction
2. Results and Discussion
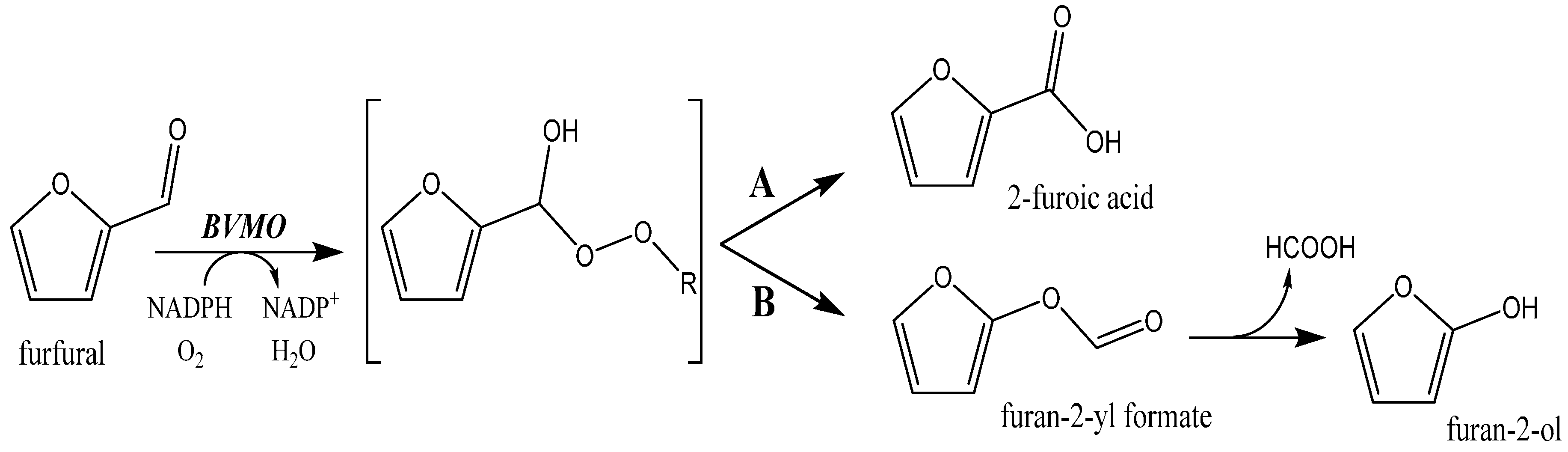
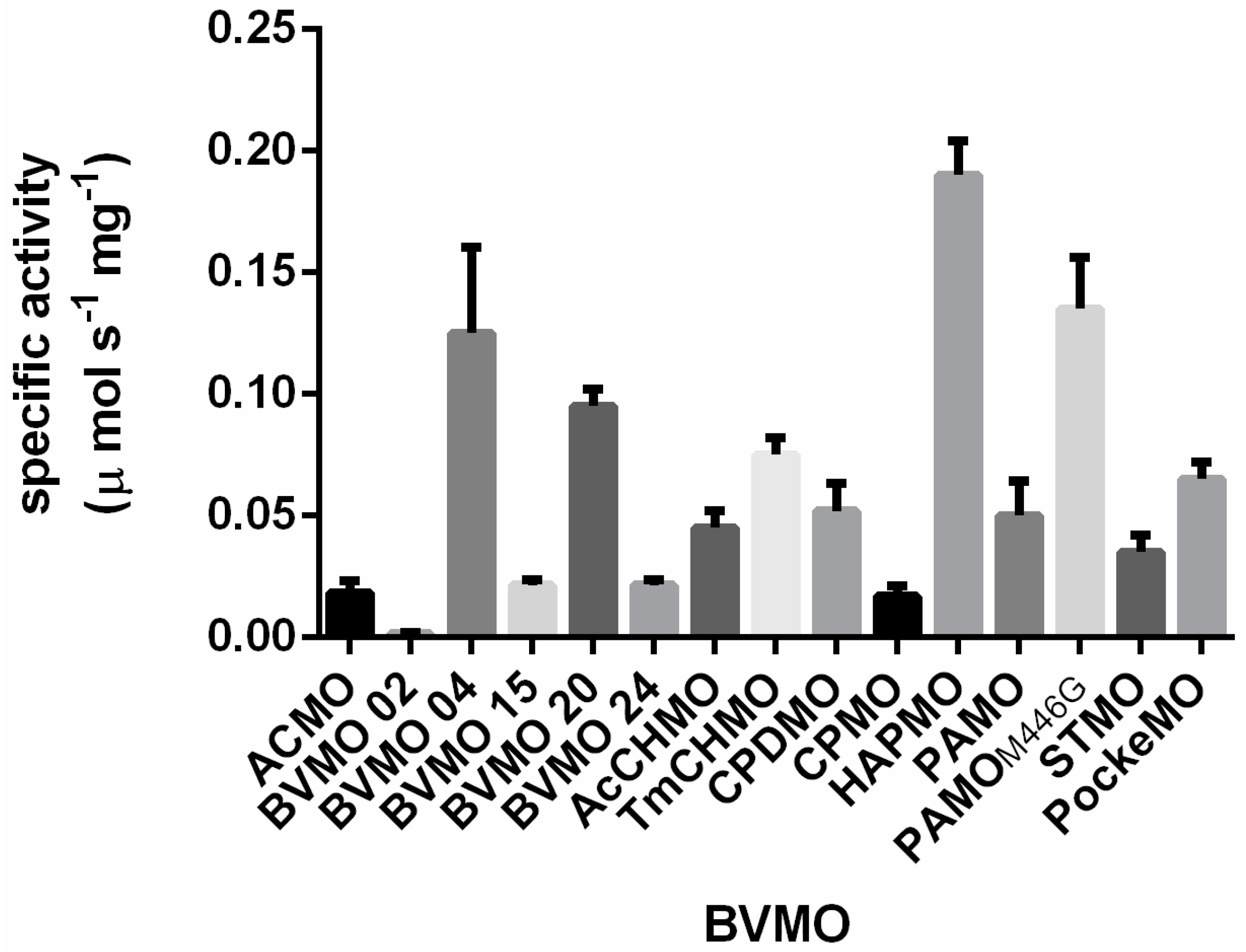
2.1. Exploring BVMOs for Activity on Furfural and HMF
2.2. Kinetic Analysis
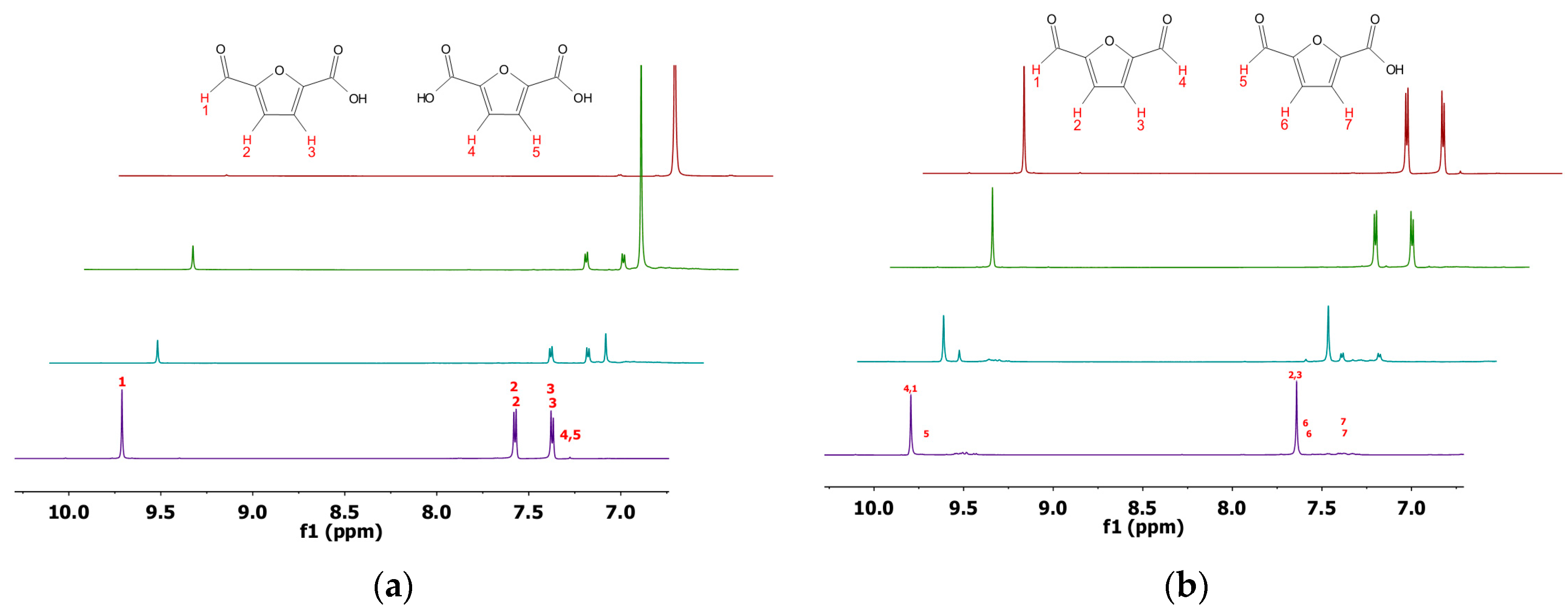
2.3. Product Analysis
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Expression and Purification
4.3. Kinetic Measurements
4.4. Product Identification Using HPLC
4.5. NMR Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhou, P.; Zhang, Z. One-pot catalytic conversion of carbohydrates into furfural and 5-hydroxymethylfurfural. Catal. Sci. Technol. 2016, 6, 3694–3712. [Google Scholar] [CrossRef]
- Sousa, A.F.; Vilela, C.; Fonseca, A.C.; Matos, M.; Freire, C.S.R.; Gruter, G.-J.M.; Coelho, J.F.J.; Silvestre, A.J.D. Biobased polyesters and other polymers from 2,5-furandicarboxylic acid: A tribute to furan excellency. Polym. Chem. 2015, 6, 5961–5983. [Google Scholar] [CrossRef]
- Mariscal, R.; Maireles-Torres, P.; Ojeda, M.; Sádaba, I.; López Granados, M. Furfural: A renewable and versatile platform molecule for the synthesis of chemicals and fuels. Energy Environ. Sci. 2016, 9, 1144–1189. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhen, J.; Liu, B.; Lv, K.; Deng, K. Selective aerobic oxidation of the biomass-derived precursor 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid under mild conditions over a magnetic palladium nanocatalyst. Green Chem. 2015, 17, 1308–1317. [Google Scholar] [CrossRef]
- Yi, G.; Teong, S.P.; Zhang, Y. Base-free conversion of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid over a Ru/C catalyst. Green Chem. 2016, 18, 979–983. [Google Scholar] [CrossRef]
- Li, S.; Su, K.; Li, Z.; Cheng, B. Selective oxidation of 5-hydroxymethylfurfural with H2O2 catalyzed by a molybdenum complex. Green Chem. 2016, 18, 2122–2128. [Google Scholar] [CrossRef]
- Mei, N.; Liu, B.; Zheng, J.; Lv, K.; Tang, D.; Zhang, Z. A novel magnetic palladium catalyst for the mild aerobic oxidation of 5-hydroxymethylfurfural into 2,5-furandicarboxylic acid in water. Catal. Sci. Technol. 2015, 5, 3194–3202. [Google Scholar] [CrossRef]
- Dijkman, W.P.; Binda, C.; Fraaije, M.W.; Mattevi, A. Structure-based enzyme tailoring of 5-hydroxymethylfurfural oxidase. ACS Catal. 2015, 5, 1833–1839. [Google Scholar] [CrossRef]
- Dijkman, W.P.; Groothuis, D.E.; Fraaije, M.W. Enzyme-catalyzed oxidation of 5-hydroxymethylfurfural to furan-2,5-dicarboxylic acid. Angew. Chem. Int. Ed. Engl. 2014, 53, 6515–6518. [Google Scholar] [CrossRef] [PubMed]
- McKenna, S.M.; Leimkühler, S.; Herter, S.; Turner, N.J.; Carnell, A.J. Enzyme cascade reactions: Synthesis of furandicarboxylic acid (FDCA) and carboxylic acids using oxidases in tandem. Green Chem. 2015, 17, 3271–3275. [Google Scholar] [CrossRef]
- Carro, J.; Ferreira, P.; Rodríguez, L.; Prieto, A.; Serrano, A.; Balcells, B.; Ardá, A.; Jiménez-Barbero, J.; Gutiérrez, A.; Ullrich, R.; et al. 5-Hydroxymethylfurfural conversion by fungal aryl-alcohol oxidase and unspecific peroxygenase. FEBS J. 2015, 282, 3218–3229. [Google Scholar] [CrossRef] [PubMed]
- Bucko, M.; Gemeiner, P.; Schenkmayerova, A.; Krajcovic, T.; Rudroff, F.; Mihovilovic, M.D. Baeyer-Villiger oxidations: Biotechnological approach. Appl. Microbiol. Biotechnol. 2016, 100, 6585–6599. [Google Scholar] [CrossRef] [PubMed]
- Leisch, H.; Morley, K.; Lau, P.C.K. Baeyer-Villiger monooxygenases: More than just green chemistry. Chem. Rev. 2011, 111, 4165–4222. [Google Scholar] [CrossRef] [PubMed]
- Torres Pazmiño, D.E.; Dudek, H.M.; Fraaije, M.W. Baeyer-Villiger monooxygenases: Recent advances and future challenges. Curr. Opin. Chem. Biol. 2010, 14, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Colonna, S.; Gaggero, N.; Carrea, G.; Ottolina, G.; Pasta, P.; Zambianchi, F. First asymmetric epoxidation catalysed by cyclohexanone monooxygenase. Tetrahedron Lett. 2002, 43, 1797–1799. [Google Scholar] [CrossRef]
- Brondani, P.B.; de Gonzalo, G.; Fraaije, M.W. Recent developments in flavin-based catalysis. In Green Biocatalysis, 1st ed.; Patel, R.N., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2013; Volume 5, pp. 403–415. [Google Scholar]
- Torres Pazmiño, D.E.; Snajdrova, R.; Baas, B.-J.; Ghobrial, M.; Mihovilovic, M.D.; Fraaije, M.W. Self-sufficient Baeyer–Villiger monooxygenases: Effective coenzyme regeneration for biooxygenation by fusion engineering. Angew. Chem. Int. Ed. 2008, 47, 2275–2278. [Google Scholar] [CrossRef] [PubMed]
- Moonen, M.J.H.; Westphal, A.H.; Rietjens, I.M.C.M.; Van Berkel, W.J.H. Enzymatic Baeyer-Villiger oxidation of benzaldehydes. Adv. Synth. Catal. 2005, 347, 1027–1034. [Google Scholar] [CrossRef]
- Bisagni, S.; Summers, B.; Kara, S.; Hatti-Kaul, R.; Grogan, G.; Mamo, G.; Hollmann, F. Exploring the substrate specificity and enantioselectivity of a Baeyer–Villiger monooxygenase from Dietzia sp. D5: Oxidation of sulfides and aldehydes. Top. Catal. 2013, 57, 366–375. [Google Scholar] [CrossRef]
- Torres Pazmiño, D.E.; Snajdrova, R.; Rial, D.V.; Mihovilovic, M.D.; Fraaije, M.W. Altering the substrate specificity and enantioselectivity of phenylacetone monooxygenase by structure-inspired enzyme redesign. Adv. Synth. Catal. 2007, 349, 1361–1368. [Google Scholar] [CrossRef]
- Branchaud, B.P.; Walsh, C.T. Functional group diversity in enzymatic oxygenation reactions catalyzed by bacterial flavin-containing cyclohexanone oxygenase. J. Am. Chem. Soc. 1985, 107, 2153–2161. [Google Scholar] [CrossRef]
- Dijkman, W.P.; Fraaije, M.W. Discovery and characterization of a 5-hydroxymethylfurfural oxidase from Methylovorus sp. strain MP688. Appl. Environ. Microbiol. 2014, 80, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, C.; de Gonzalo, G.; Fraaije, M.W.; Gotor, V. Enzymatic kinetic resolution of racemic ketones catalyzed by Baeyer–Villiger monooxygenases. Tetrahedron: Asymmetry 2007, 18, 1338–1344. [Google Scholar] [CrossRef]
- Fraaije, M.W.; Wu, J.; Heuts, D.P.H.M.; van Hellemond, E.W.; Spelberg, J.H.L.; Janssen, D.B. Discovery of a thermostable Baeyer-Villiger monooxygenase by genome mining. Appl. Microbiol. Biotechnol. 2005, 66, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Riebel, A.; Dudek, H.M.; de Gonzalo, G.; Stepniak, P.; Rychlewski, L.; Fraaije, M.W. Expanding the set of rhodococcal Baeyer-Villiger monooxygenases by high-throughput cloning, expression and substrate screening. Appl. Microbiol. Biotechnol. 2012, 95, 1479–1489. [Google Scholar] [CrossRef] [PubMed]
- Torres Pazmiño, D.E.; Riebel, A.; de Lange, J.; Rudroff, F.; Mihovilovic, M.D.; Fraaije, M.W. Efficient biooxidations catalyzed by a new generation of self-sufficient Baeyer-Villiger monooxygenases. ChemBioChem 2009, 10, 2595–2598. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, H.; Wang, S.; Grosse, S.; Bergeron, H.; Nagahashi, A.; Lertvorachon, J.; Yang, J.; Konishi, Y.; Hasegawa, Y.; Lau, P.C.K. Pseudomonad cyclopentadecanone monooxygenase displaying an uncommon spectrum of Baeyer-Villiger oxidations of cyclic ketones. Appl. Environ. Microbiol. 2006, 72, 2707–2720. [Google Scholar] [CrossRef] [PubMed]
- Kotani, T.; Yamamoto, T.; Yurimoto, H.; Sakai, Y.; Kato, N. Propane monooxygenase and NAD+-dependent secondary alcohol dehydrogenase in propane metabolism by Gordonia sp. strain TY-5. J. Bacteriol. 2003, 185, 7120–7128. [Google Scholar] [CrossRef] [PubMed]
- Romero, E.; Castellanos, J.R.G.; Mattevi, A.; Fraaije, M.W. Characterization and crystal structure of a robust cyclohexanone monooxygenase. Angew. Chem. Int. Ed. Engl. 2016, 55, 15852–15855. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, S.; van Beek, H.L.; Pennetta, A.; Martinoli, C.; Fraaije, M.W.; Mattevi, A. Exploring the structural basis of substrate preferences in Baeyer-Villiger monooxygenases: Insight from steroid monooxygenase. J. Biol. Chem. 2012, 287, 22626–22634. [Google Scholar] [CrossRef] [PubMed]
- Fürst, M.J.L.J.; Savino, S.; Dudek, H.M.; Gómez Castellanos, J.R.; Gutiérrez de Souza, C.; Rovida, S.; Fraaije, M.W.; Mattevi, A. Polycyclic ketone monooxygenase from the thermophilic fungus Thermothelomyces thermophila: A structurally distinct biocatalyst for bulky substrates. J. Am. Chem. Soc. 2017, 139, 627–630. [Google Scholar] [CrossRef] [PubMed]
| Substrate | PAMO | PAMOM446G | ||||||
|---|---|---|---|---|---|---|---|---|
| kcat (s−1) | Km (mM) | Ki (mM) | kcat/Km (s−1mM−1) | kcat (s−1) | Km (mM) | Ki (mM) | kcat/Km (s−1mM−1) | |
| Furfural | 2.8 ± 0.3 | 2.2 ± 0.5 | 36.9 ± 9.8 | 1.2 | 7 ± 0.8 | 17.3 ± 1.3 | 6.4 ± 0.5 | 0.4 |
| 5-(hydroxymethyl)furfural (HMF) | 1.4 ± 0.1 | 16.9 ± 3.3 | n.d. | 0.08 | 0.67 ± 0.1 | 20.5 ± 2.3 | n.d. | 0.03 |
| 2,5-diformylfuran (DFF) | 0.3 ± 0.1 | 4.3 ± 2.3 | n.d. | 0.07 | 1.2 ± 0.8 | 5.8 ± 2.0 | 10.3 ± 3.0 | 0.1 |
| Substrate | Concentration (mM) | Reaction Time (h) | PAMO | PAMOM446G | ||
|---|---|---|---|---|---|---|
| Concentration (µM) | Conversion (%) | Concentration (µM) | Conversion (%) | |||
| Furfural | 5 | 12 | 5 | 60 | 5 | 60 |
| HMF | 5 | 16 | 5 | 66 | 5 | 85 |
| DFF | 2 | 12 | 5 | 30 | 5 | 98 |
| 5-formyl-2-furancarboxylic acid (FFA) | 10 | 24 | 10 | 60 | 10 | 90 |
| BVMO | Construct | Source | Reference |
|---|---|---|---|
| BVMO02 | pCRE2-BVMO02 | Rhodococcus jostii RHA1 | [25] |
| BVMO04 | pBADNK-BVMO04 | Rhodococcus jostii RHA1 | [25] |
| BVMO15 | pBADNK-BVMO15 | Rhodococcus jostii RHA1 | [25] |
| BVMO20 | pBADNK-BVMO20 | Rhodococcus jostii RHA1 | [25] |
| BVMO24 | pBADNK-BVMO24 | Rhodococcus jostii RHA1 | [25] |
| HAPMO | pCRE2-HAPMO | Pseudomonas fluorescens ACB | [26] |
| CPDMO | pCRE2-CPDMO | Pseudomonas sp. strain HI-70 | [27] |
| ACMO | pCRE2-ACMO | Gordonia sp. strain TY-5 | [28] |
| AcCHMO | pCRE2-CHMO | Acinetobactor sp. | [26] |
| TmCHMO | pCRE2-TmCHMO | Thermocrispum municipale | [29] |
| CPMO | pCRE2-CPMO | Comamonas sp. | [26] |
| STMO | pCRE2-STMO | Rhodococcus rhodochrous | [30] |
| PAMO | pCRE2-PAMO | Thermobifida fusca | [24] |
| PAMOM446G | pCRE2-PAMOM446G | Thermobifida fusca | [26] |
| PockeMO | pCRE2-PoKeMO | Thermothelomyces thermophila | [31] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, H.; Fraaije, M.W. Conversion of Furans by Baeyer-Villiger Monooxygenases. Catalysts 2017, 7, 179. https://doi.org/10.3390/catal7060179
Kumar H, Fraaije MW. Conversion of Furans by Baeyer-Villiger Monooxygenases. Catalysts. 2017; 7(6):179. https://doi.org/10.3390/catal7060179
Chicago/Turabian StyleKumar, Hemant, and Marco W. Fraaije. 2017. "Conversion of Furans by Baeyer-Villiger Monooxygenases" Catalysts 7, no. 6: 179. https://doi.org/10.3390/catal7060179
APA StyleKumar, H., & Fraaije, M. W. (2017). Conversion of Furans by Baeyer-Villiger Monooxygenases. Catalysts, 7(6), 179. https://doi.org/10.3390/catal7060179







