Abstract
Using pseudoboehmite and cerium nitrate as raw materials, a cerium dioxide-doped alumina support was prepared by the hot oil column method. Subsequently, with platinum nitrate and palladium nitrate solutions as precursor salts, the active components were loaded onto the supports via the incipient wetness impregnation, followed by an activation treatment, thus obtaining platinum-palladium bimetallic catalysts for hydrocarbon elimination in zero-air generators. The catalyst was characterized by XRD, BET, SEM, TEM, XPS, and Raman spectroscopy. The results showed that the as-prepared supports possess a large specific surface area, and the noble metals Pt and Pd are uniformly distributed on the support surface. After activation treatment, the structural stability and catalytic reaction activity of the catalysts are significantly enhanced. Performance tests simulating the actual operating conditions of zero-air generators show that the catalysts exhibit excellent hydrocarbon elimination capability: when the inlet methane concentration is 50 ppm, the outlet methane content can be reduced to below 10 ppb. Moreover, no obvious attenuation of catalyst activity is observed after a 1000-h long-term stability test, which meets the practical application requirements of zero-air generators.
1. Introduction
High-purity zero gas is an indispensable core carrier gas and background gas for fields such as environmental monitoring and industrial production, and its purity level directly determines the accuracy of analytical test results and the stability and reliability of industrial processes [1,2,3]. As a key piece of equipment for producing high-purity zero gas, the zero gas generator can efficiently remove interfering impurities such as moisture, sulfur dioxide (SO2), nitrogen oxides, and various hydrocarbons from compressed air, and continuously supply clean and stable gas sources for environmental monitoring systems, serving as a core supporting component to ensure the accuracy and reliability of monitoring data. According to current international and Chinese technical standards, the total hydrocarbon content of hydrocarbon-free air must meet the strict limits of less than 0.1 ppm [4] or not more than 0.40 mg/m3 (calculated as methane) [5,6]. For some high-precision monitoring scenarios, an even more stringent requirement of total hydrocarbon content not exceeding 10 ppb has been proposed [7]. Against this backdrop of rigorous demands, the performance parameters of catalysts have become a core factor restricting the operational efficiency of zero gas generators and their capacity to achieve deep impurity removal compliance. Therefore, the development of high-efficiency purification catalysts with low noble metal loading and high catalytic activity holds significant theoretical guiding value and practical application significance for promoting the technological iteration and upgrading of zero gas generators and meeting the needs of high-end, high-precision applications.
Among various hydrocarbons, methane (CH4) is the most difficult component to be catalytically oxidized due to its stable molecular structure and high C-H bond energy [8,9,10], which poses severe challenges to the low-temperature activity and long-term stability of catalysts for zero air generators. In addition, especially for portable devices, the catalyst module is required to handle a relatively large gas flow rate under the condition of limited volume. This demands that the catalyst carrier not only has a high specific surface area, but also possesses extremely high mechanical strength (high crush strength and low wear rate) and regular sphericity, so as to avoid powdering and pipeline blockage and ensure the long-term stable operation of the equipment [11,12].
Alumina is widely used as a catalyst support owing to its high specific surface area, excellent thermal stability, and tunable surface properties [13,14]. The main industrial methods for preparing millimeter-scale alumina spheres include the rolling ball method, hot oil column method, oil-ammonia column method, and spray drying method. Among these, alumina spheres fabricated via the hot oil column method exhibit outstanding performance in crush strength, pore volume, sphericity, and surface smoothness [15]. Furthermore, this method features flexible process conditions, allowing precise regulation of particle characteristics by adjusting the oil-phase composition and dispersion parameters. Consequently, such alumina spheres are particularly suitable as catalyst supports for high-performance zero air generators.
Currently, most research on the catalytic combustion of low-concentration methane focuses on monolithic catalysts or alumina supports prepared via the powder or rolling ball method, which suffer from drawbacks such as poor mechanical strength, easy pulverization, and elevated costs caused by high precious metal loading [16,17]. To address these challenges, the mainstream research directions currently focus on two strategies: first, introducing rare earth elements (e.g., cerium) to enhance support stability and promote the dispersion of precious metals, thereby reducing the dosage of platinum, palladium, and other noble metals while improving catalytic performance [18,19,20,21]; second, constructing Pt-Pd bimetallic active sites to exploit synergistic effects, which serves as an effective approach to boost catalytic activity and sintering resistance [22,23,24].
Based on the above considerations, this study aims to develop a high-performance, high-strength, and low-cost hydrocarbon removal catalyst suitable for zero air generators. Using pseudoboehmite and cerium nitrate as raw materials, ceria-doped alumina spherical supports were prepared via the hot oil column method. Subsequently, low-content Pt-Pd active components were loaded onto the supports by the incipient wetness impregnation method using a coating machine, with platinum nitrate and palladium nitrate as precursors. The resulting materials were then activated under specific conditions to obtain Pt-Pd bimetallic catalysts. A variety of characterization techniques including XRD, BET, SEM, TEM, XPS, and Raman spectroscopy were employed to systematically investigate the effects of support structure and activation conditions on the physicochemical properties and catalytic performance of the catalysts. This work is expected to provide new insights for the development of industrially feasible, high-performance catalysts for zero air generators, and promote the advancement of China’s zero air preparation technology toward high-end and low-cost directions.
2. Results and Discussion
2.1. Catalytic Performance
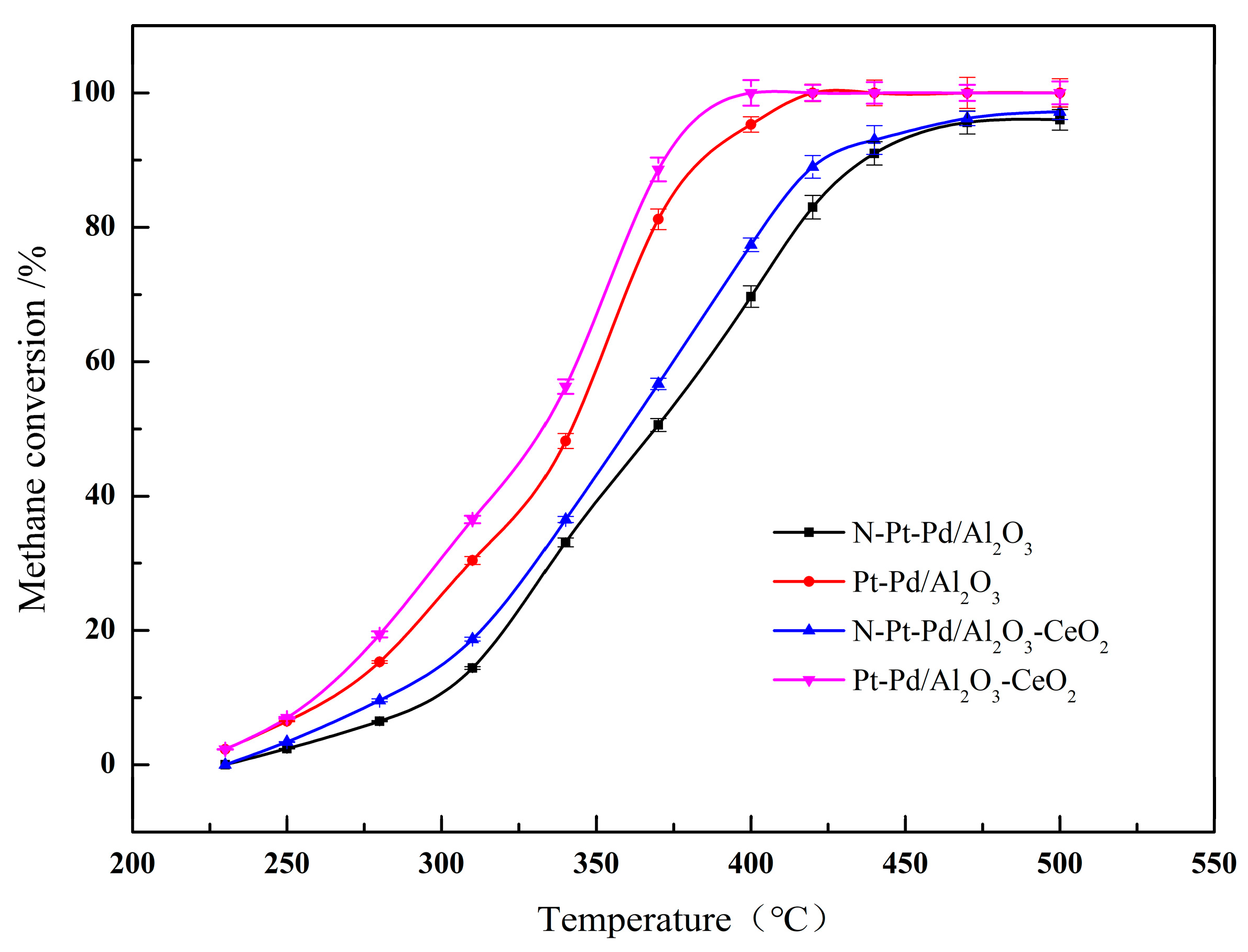
Two metal oxides, namely Al2O3 and Al2O3-CeO2, were selected as supports for platinum-palladium (Pt-Pd) bimetallic catalysts. The labeling of each sample is specified in the Experimental Section. Figure 1 presents the methane conversion curves of different catalysts as a function of reaction temperature, while Table 1 lists the corresponding methane conversion characteristic temperatures (T10, T50, T90) for each catalyst. Specifically, T10, T50, and T90 denote the reaction temperatures at which the methane conversion reaches 10%, 50%, and 90%, respectively. These characteristic temperatures are key indicators for evaluating the methane abatement activity of the catalysts—lower characteristic temperatures correspond to superior catalytic activity. All data reported herein are the average results of three parallel measurements, with the standard deviation of methane conversion for all samples in the range of ±1.2~±2.5%, and error bars representing standard deviations are added in Figure 1 to ensure the reliability and repeatability of the experimental data.
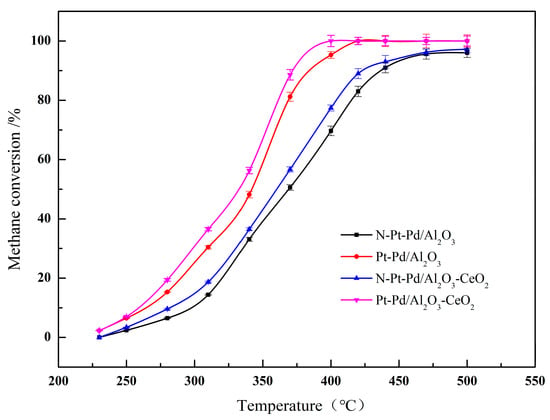
Figure 1.
Methane conversion rates of catalysts prepared with different supports.

Table 1.
Catalytic conversion of catalysts with different supports.
A Pt/Pd mass ratio of 1:2 (with a nominal total noble metal loading of 0.5 wt%) was determined as the optimal proportion, based on systematic evaluation of catalysts with various ratios (1:1, 1:2, 2:1, 1:3) under identical reaction conditions. Among the series, the catalyst with a 1:2 ratio exhibited the lowest light-off temperatures (T10/T50/T90), demonstrating superior low-temperature activity for lean methane combustion (see Table S1). This optimal performance is attributed to the synergistic interplay between Pt and Pd, which exploits their complementary functions under lean conditions: Pd serves as the primary site for activating the C-H bond of methane (the rate-determining step); Pt modulates the electronic structure of Pd, inducing a downshift of the Pd d-band center. According to the d-band theory, this electronic modification optimizes the adsorption/desorption behavior of reaction intermediates. Specifically, it weakens the over-strong binding of oxygen-containing species (e.g., surface carbonates or hydroxyl groups) that would otherwise lead to surface passivation (over-oxidation), thereby shifting the reaction pathway toward the more efficient Mars-van Krevelen (MvK) cycle involving lattice oxygen. Furthermore, Pt acts as a structural promoter that stabilizes the active PdO phase and inhibits its sintering and thermal deactivation at high temperatures [25,26,27].
By correlating the data presented in Figure 1 and Table 1, it can be observed that the catalytic activity of all samples increases progressively with rising temperature. However, pronounced disparities exist in the methane abatement performance among the various catalysts, with the activity ranking from the lowest to highest as follows: N-Pt-Pd/Al2O3 < N-Pt-Pd/Al2O3-CeO2 < Pt-Pd/Al2O3 < Pt-Pd/Al2O3-CeO2. Even when the standard deviation is taken into account, the methane conversion curves of CeO2-doped catalysts and undoped catalysts show no overlap in the entire test temperature range (230~500 °C), which clearly confirms the significant promotion effect of CeO2 doping on the catalytic activity of Pt-Pd bimetallic catalysts. These findings demonstrate that the non-activated catalysts exhibit relatively inferior catalytic activity, whereas the activated Pt-Pd bimetallic catalysts supported on CeO2-doped Al2O3 deliver optimal methane abatement performance and achieve higher conversion rates within the low-temperature range. Evidently, the incorporation of CeO2 into the support facilitates the uniform dispersion of noble metal active components on the support surface, thereby markedly enhancing the catalytic efficiency of the resultant catalysts.
Table 2 systematically presents the key physical property parameters of catalysts prepared with different supports. Experimental data indicate that despite the differences in support compositions, the values of compressive strength and attrition rate of all catalyst types show relatively small fluctuations. This result fully demonstrates that the modification strategy of doping CeO2 into the Al2O3 support does not exert a significant negative effect on the mechanical stability of the catalysts, which can meet the basic operational requirements for impact resistance and wear resistance of catalysts during the practical operation of zero-air generators. Meanwhile, the introduction of CeO2 can effectively promote the uniform dispersion of Pt-Pd active components on the support surface through its unique oxygen storage and release performance as well as the support-active component interaction, thereby avoiding the agglomeration of active sites and further significantly enhancing the catalytic reaction activity of the catalysts.

Table 2.
Physical parameters of catalysts with different supports.
Table 3 shows the test results of methane elimination performance of the series of catalysts under simulated practical operating conditions of zero-air generators. During the test, the inlet methane concentration was strictly controlled at 50 ppm, and other reaction conditions (such as space velocity, reaction temperature, humidity, etc.) were consistent with the actual operating parameters of zero-air generators. The test results clearly reveal that compared with the catalyst prepared with pure Al2O3 support, the catalyst based on the Pt-Pd/Al2O3-CeO2 composite support exhibits the most excellent methane removal efficiency. The concentration of methane was below the limit of detection (LOD) of the Gas Chromatograph, which is far below the limit requirements for hydrocarbon impurities in zero air specified in relevant technical standards. In addition, after a 1000-h continuous stability test, the methane removal efficiency of this catalyst does not show obvious attenuation and still maintains stable and high-efficiency catalytic performance, which fully highlights its great potential and value in the industrial application scenarios of zero-air generators.

Table 3.
Performance test results of catalysts under the simulated application scenario of zero-air generators.
In addition to methane concentration detection, the gaseous products (CO and CO2) from methane catalytic oxidation were synchronously quantified. Under simulated practical operating conditions (with an inlet CH4 concentration of 50 ppm), the Pt-Pd/Al2O3-CeO2 catalyst achieved the complete oxidation of methane to CO2. At nearly 100% conversion, no CO was detected in the reaction products (below the instrument detection limit), and the CO2 concentration was measured as 49.8 ppm, with a carbon balance above 99.5%. In contrast, for the other catalysts (N-Pt-Pd/Al2O3, Pt-Pd/Al2O3, and N-Pt-Pd/Al2O3-CeO2), trace levels of CO (9.8–17.2 ppb) were detected in the effluent gas, consistent with their incomplete methane conversion. The CO2 yield was positively correlated with methane conversion, consistent with the catalytic activity order. These results demonstrate that the Pt-Pd/Al2O3-CeO2 catalyst exhibits superior selectivity towards complete oxidation, effectively suppressing the formation of toxic CO byproducts. This performance meets the stringent purity requirements for zero-air generators where strict control of CO at extremely low levels is required.
2.2. Characterization of Catalysts
2.2.1. XRD Analysis
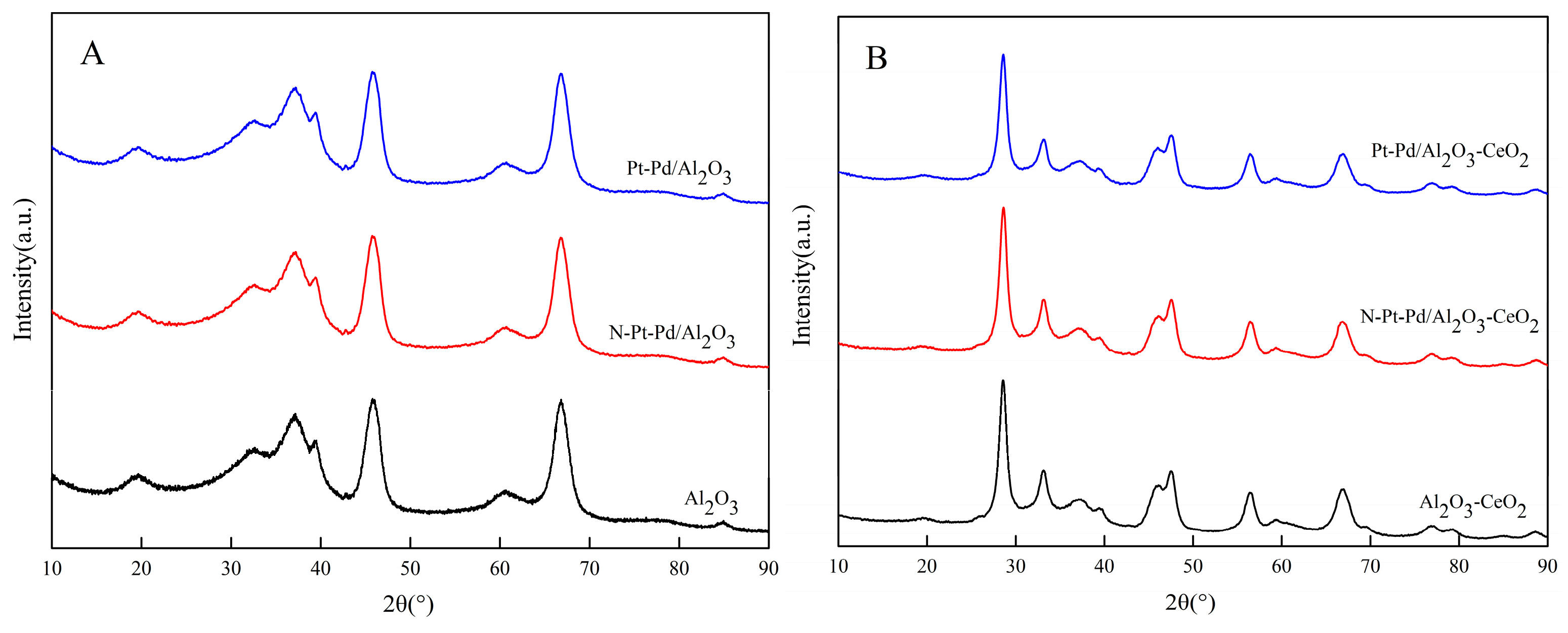
To investigate the effect of support types on the crystal structure of catalysts, X-ray diffraction (XRD) characterization was conducted on the as-prepared supports and the corresponding catalysts loaded with active components.
Figure 2A shows the XRD patterns of the pure Al2O3-based support and its supported catalyst in both the non-activated state and the high-temperature activated state. By comparing the positions and intensities of the diffraction peaks with the standard Al2O3 diffraction card (PDF#29-0063), the Al2O3 support was confirmed to have a typical cubic crystal system and be characterized as γ-Al2O3. After high-temperature activation, no obvious shift or attenuation was observed in the position and intensity of the catalyst’s diffraction peaks, indicating that the high-temperature activation treatment did not induce crystal phase transformation of the Al2O3 support and the corresponding catalyst, and their crystal structures maintained good stability during the activation process.
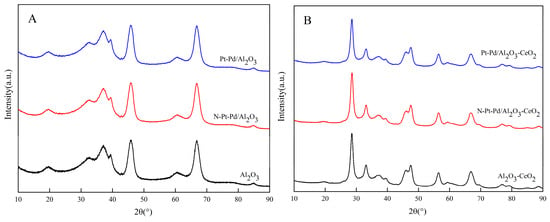
Figure 2.
XRD diffraction patterns of catalysts prepared with different supports: (A) Al2O3; (B) Al2O3-CeO2.
Figure 2B presents the XRD patterns of the CeO2-doped Al2O3-based support and the corresponding catalyst under both non-activated and activated conditions. Comparison with the standard diffraction card for CeO2 (PDF#43-1002) confirms that the doped ceria also crystallizes in a cubic fluorite structure. More importantly, it is observed that the characteristic diffraction peaks corresponding to Al2O3 in the composite support exhibit a significant decrease in intensity along with a slight broadening of the full width at half maximum (FWHM). According to the Scherrer equation, which correlates peak broadening with crystallite size, these observations indicate that the introduction of CeO2 effectively inhibits the grain growth of Al2O3, resulting in refined Al2O3 crystallites. This phenomenon is attributed to a strong interfacial interaction between CeO2 and Al2O3, which hinders the agglomeration and sintering of Al2O3 crystallites during preparation and activation. The reduction in grain size is the direct cause of the attenuated and broadened diffraction peaks observed here. This phenomenon, consistent with the reported role of rare-earth oxides in refining the microstructure of alumina supports, provides clear evidence for the significant Al2O3-CeO2 interaction and lays the structural foundation for the enhanced catalytic performance [28,29].
Comparison of the XRD patterns before and after loading with Pt and Pd shows that no obvious shift is observed in the characteristic diffraction peaks of the support, indicating that the loading of Pt and Pd does not destroy the crystalline structure of the support. In addition, no characteristic diffraction peaks corresponding to Pt and Pd species are detected in the XRD patterns of all the catalysts. Two main reasons are speculated for this phenomenon: first, the Pt and Pd species are highly dispersed on the support surface without agglomeration, failing to form regular crystalline phases with sufficient crystallinity and particle size to meet the requirements of XRD detection; their excellent dispersibility renders the characteristic diffraction signals undetectable by XRD characterization. Second, the actual loading amounts of Pt and Pd in the experiment are relatively low, which are below the detection limit of the XRD instrument. Furthermore, the inherent diffraction signal intensity of Pt and Pd species is weak, and the masking effect of the strong diffraction peaks of the support further prevents their characteristic diffraction peaks from being effectively detected.
2.2.2. BET Characterization
Table 4 presents the specific surface area, pore volume and pore size data of different supports and the catalysts after noble metal loading. It can be seen from the data in the table that after loading Pt and Pd on the supports, their specific surface area and pore volume both decreased slightly, while the pore size remained basically unchanged. This phenomenon is mainly attributed to the filling and surface covering effects of precious metal particles on partial pores of the supports, without causing substantial damage to the pore structure framework of the supports.

Table 4.
Structural parameters of catalysts prepared with different supports.
Compared with pure Al2O3 spheres, the Al2O3 support doped with CeO2 exhibits a significant increase in specific surface area, pore volume, and pore size. The reasons can be attributed to two aspects. On the one hand, the doping of 10 wt% CeO2 effectively inhibits the high-temperature sintering and grain growth of Al2O3 particles during the preparation process. As a result, CeO2 mainly exists as a separate fluorite-type cubic nanocrystalline phase (Figure 2B) and is uniformly dispersed on the Al2O3 surface, forming an intimate Al2O3-CeO2 interface contact. This inhibition of sintering also reduces particle agglomeration, thereby maintaining the integrity of the support’s porous structure.
There is a close positive correlation between the specific surface area of the catalyst and its catalytic activity. A larger specific surface area is not only conducive to the uniform dispersion of Pt and Pd species on the support surface during the impregnation process and avoids the agglomeration of active components, but also provides more active sites for the methane oxidation reaction, thereby significantly improving the overall catalytic performance of the catalyst.
2.2.3. SEM and TEM Characterization
Figure 3 displays the SEM images of catalysts prepared with different supports before and after activation, while Figure 4 presents the corresponding HR-TEM images of these catalysts.

Figure 3.
SEM images of catalysts prepared with different supports: (A) N-Pt-Pd/Al2O3; (B) Pt-Pd/Al2O3; (C) N-Pt-Pd/Al2O3-CeO2; (D) Pt-Pd/Al2O3-CeO2.

Figure 4.
HR-TEM images of catalysts prepared with different supports: (A) N-Pt-Pd/Al2O3; (B) Pt-Pd/Al2O3; (C) N-Pt-Pd/Al2O3-CeO2; (D) Pt-Pd/Al2O3-CeO2; (E) STEM-EDS elemental mapping of Pt-Pd/Al2O3-CeO2.
SEM can directly reflect the surface distribution of the active components of catalysts. According to the SEM characterization results in Figure 3, the catalysts prepared with pure Al2O3 as the sole support exhibit larger particle sizes of Pt-Pd active components along with agglomeration, which leads to insufficient exposure of active sites and thus hinders the full exertion of catalytic performance. In contrast, the catalysts fabricated with CeO2-doped modified Al2O3 supports show smaller particle sizes of Pt-Pd active components, which are uniformly dispersed on the support surface without obvious agglomeration characteristics. This effectively enhances the effective exposure degree of active sites, providing a structural basis for the improvement of catalytic performance.
The HR-TEM characterization results in Figure 4 further clarify the regulatory effect of support types on the microstructure of Pt and Pd active components. In the Pt-Pd/Al2O3 catalyst prepared with pure Al2O3 as the support, the particle size of precious metal particles is approximately 8 nm, and the particles tend to aggregate, which is not conducive to the mass transfer process and the efficient utilization of active sites during the catalytic reaction. For the Pt-Pd/Al2O3-CeO2 catalyst fabricated using CeO2-doped modified Al2O3 as the support, the particle size of the active components is significantly reduced to around 5 nm, with dispersibility greatly improved, thus providing more abundant active sites and smoother mass transfer channels. The measurement of its lattice fringes (inset of D) yields a lattice spacing of d = 0.32 nm, corresponding to the (111) crystal plane of CeO2, which confirms that CeO2 has been successfully doped into the support and participates in the structural regulation of the active components [30].
To characterize the nanoscale distribution of the catalyst, STEM-EDS elemental mapping was conducted (Figure 4E). The results reveal that Ce (10 wt%) is homogeneously dispersed within the Al2O3 matrix, verifying the successful doping of CeO2. Notably, even at an ultra-low total noble metal loading of 0.5 wt%, Pt and Pd species are uniformly dispersed on the support without discernible agglomeration. Their distribution exhibits a strong overlap with that of Ce, implying that the doped CeO2 provides abundant anchoring sites. These sites strengthen the metal-support interaction and effectively suppress the agglomeration of Pt-Pd species, thus realizing their uniform nanoscale dispersion. Owing to the resolution limitation of EDS mapping, the exact particle size cannot be determined. However, the critical absence of large, bright aggregates (which would indicate severe sintering) supports the conclusion that the metal species are highly dispersed. This observation is fully consistent with the small average particle size of ~5 nm obtained from HR-TEM/chemisorption.
In summary, a smaller particle size and superior dispersibility of the catalyst’s active components translate to more extensive exposure of active sites, which in turn contributes to enhanced catalytic performance.
2.2.4. H2-Chemisorption Characterization
H2-chemisorption was employed to determine the average crystallite size and dispersion of Pt-Pd bimetals in the entire catalyst sample. The reported values are overall averages calculated based on the total amount of irreversibly chemisorbed H2 and the total number of Pt and Pd atoms. The calculation employs the spherical model and assumes, as a simplified and widely applied approach for bimetallic systems, that each surface metal atom adsorbs one hydrogen atom (i.e., an average H/M_total stoichiometry of 1). While the precise H/M adsorption stoichiometry may differ for individual Pt and Pd, this method provides a reliable and consistent metric for comparing relative trends in metal dispersion and particle size among the catalyst series [31,32].
The H2-chemisorption data for catalysts with different supports are displayed in Table 5. The results demonstrate that the Pt-Pd/Al2O3 catalyst has an average Pt-Pd crystallite size of 7.8 nm with a corresponding metal dispersion of 37.5%. In contrast, the Pt-Pd/Al2O3-CeO2 catalyst features a reduced average size of 5.2 nm alongside a markedly higher metal dispersion of 52.1%. These quantitative findings, which represent volume-averaged and statistically significant data for the entire sample, are in good agreement with the reduced particle size trend (e.g., ~5 nm) observed via HR-TEM. The excellent agreement between the H2-chemisorption-derived average crystallite size (5.2 nm) and the HR-TEM observation for the Pt-Pd/Al2O3-CeO2 catalyst cross-validates the applicability of this analytical approach. The significant enhancement in dispersion upon CeO2 doping can be attributed to the increased surface oxygen storage capacity and defect sites introduced by CeO2, which promote a strong metal-support interaction (SMSI) that effectively inhibits particle coalescence during calcination. This further confirms that CeO2 doping effectively refines the Pt-Pd bimetallic particles and improves their dispersion on the support surface.

Table 5.
H2-chemisorption test of catalysts with different supports.
2.2.5. XPS Characterization
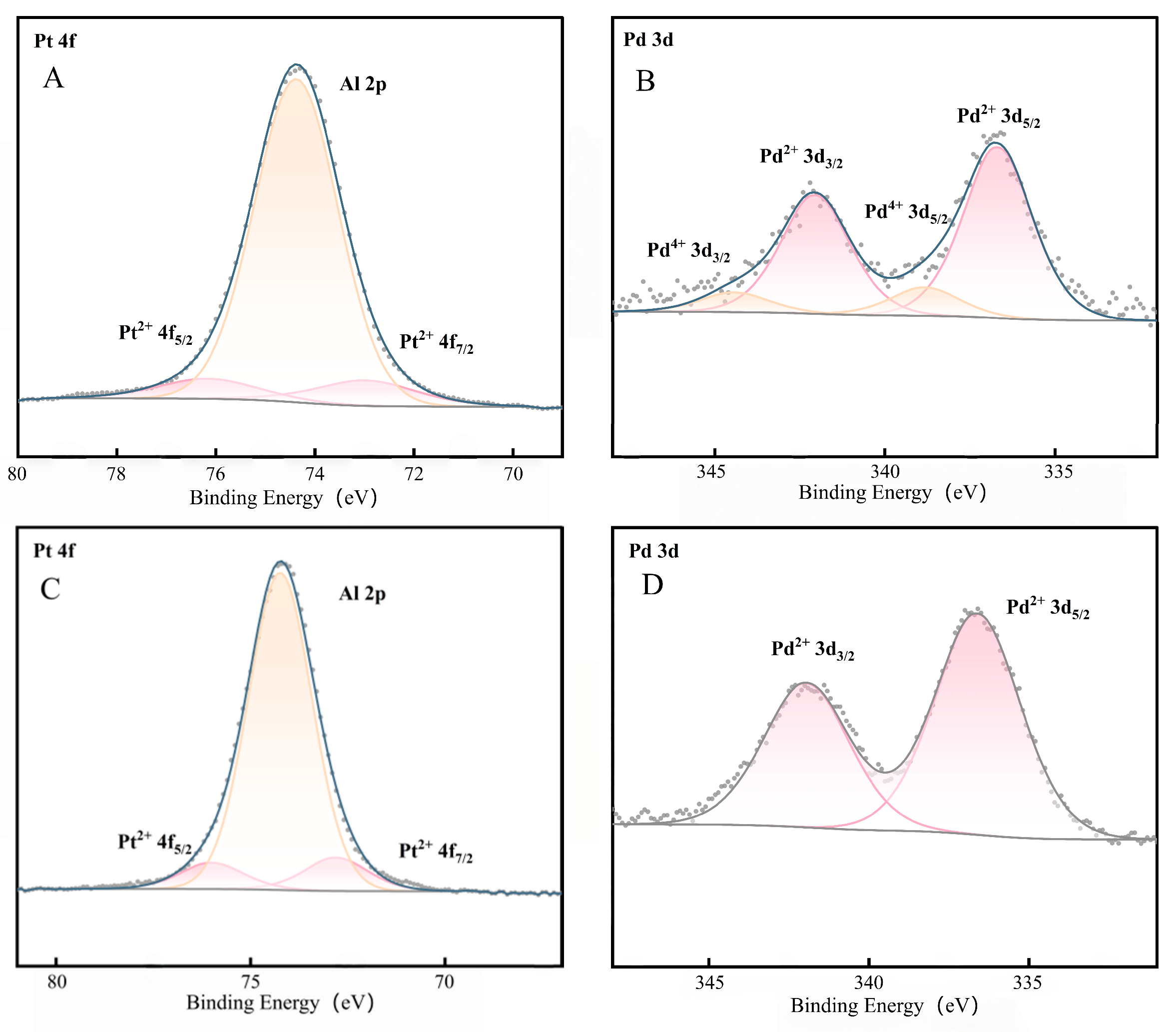
Figure 5 shows the XPS spectra of Pt and Pd in the CeO2-doped Al2O3-supported catalyst before and after activation, which are analogous to the corresponding spectra of the pure Al2O3-supported catalyst under the same activation conditions. Based on the characterization results of N-Pt-Pd/Al2O3-CeO2 (spectra A and B) and Pt-Pd/Al2O3-CeO2 (spectra C and D) in Figure 5, the evolution rules of chemical valence states of surface Pt and Pd species as well as the variation characteristics of metal-support interaction (MSI) before and after catalyst activation were analyzed via peak fitting of characteristic peaks and binding energy data analysis, and the conclusions are drawn as follows:
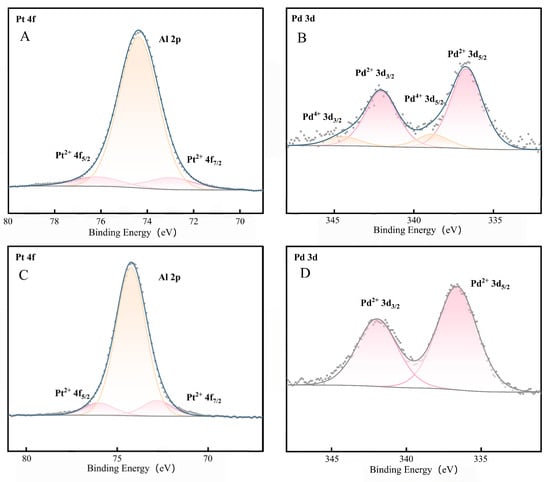
Figure 5.
XPS spectra of Pt and Pd in the catalysts with Al2O3-doped CeO2 supports before and after activation: (A,B) N-Pt-Pd/Al2O3-CeO2; (C,D) Pt-Pd/Al2O3-CeO2.
A comparison of Spectra A and C reveals that the valence state of Pt remained unchanged for both catalysts before and after activation, existing stably in the form of Pt2+. Full-spectrum and high-resolution fine scanning of Pt 4f confirmed that no characteristic peaks of metallic Pt0 (Pt 4f7/2 at ~70.9 eV, Pt 4f5/2 at ~74.2 eV) were detected in all samples. This phenomenon indicates that the Al2O3-CeO2 composite support exerts an excellent stabilizing effect on the valence state of Pt species; even after activation treatment, it can maintain the intrinsic chemical state of Pt active components, which is related to the strong metal-support interaction between Pt and the composite support and the mild activation conditions, thereby laying a foundation for the stable performance of the catalyst. Prior to activation, the binding energies of the characteristic peaks of Pt 4f7/2 and Pt 4f5/2 corresponding to Pt2+ were 72.97 eV and 76.17 eV, respectively. After activation, the binding energies of these two characteristic peaks decreased to 72.79 eV and 75.99 eV, respectively, shifting by 0.18 eV toward the lower binding energy direction compared with those before activation [33]. Meanwhile, the binding energy of the Al 2p characteristic peak in the support exhibited a similar shifting trend: the binding energy of the Al 2p peak was 74.38 eV before activation and dropped to 74.24 eV after activation, with a shift amplitude of 0.14 eV. The shift of binding energy toward the low-energy end is a direct indication of the enhanced strong metal-support interaction (SMSI). This result demonstrates that the metal-support interaction of the catalyst was significantly strengthened during the activation process. The strong interaction can effectively inhibit the migration and agglomeration of Pt active components under high-temperature activation conditions, thus maintaining the structural stability of the catalyst.
A comparison of Spectra B and D shows that the valence state of Pd species underwent a significant evolution before and after activation. Before activation, Pd species existed in the form of two coexisting valence states (Pd2+ and Pd4+), among which the proportion of Pd2+ was as high as 85.8% while that of Pd4+ was only 14.2%. Following activation in 0.5% CH4/air at 400 °C, Pd4+ was selectively reduced to a trace amount (<3%), with Pd2+ becoming the dominant species (>97%). The selective reduction of Pd4+ under net oxidizing conditions is attributed to the concerted action of CH4 and the unique properties of the CeO2-Al2O3 composite support. CH4 adsorbed on the surface acts as a mild reductant, generating active hydrogen species upon dehydrogenation that preferentially target the less stable Pd4+. More importantly, the well-dispersed CeO2 nanocrystals, intimately interfaced with Al2O3 as confirmed by XRD, create a highly active interface. The Ce3+/Ce4+ redox couple at this interface acts as an electron-transfer mediator, facilitating the reduction of proximate Pd4+. Concurrently, the oxygen vacancies and the high oxygen mobility of CeO2 help buffer the local oxygen chemical potential, which kinetically favors the stabilization of Pd2+ and prevents its re-oxidation or over-reduction, thereby explaining the high selectivity towards Pd2+. This change indicates that the activation process can facilitate the reduction reaction of high-valence Pd, which ultimately exists in the form of stable low-valence Pd2+, further enhancing the chemical stability of Pd active components and providing the main active valence state for methane catalytic oxidation. Prior to activation, the binding energies of the characteristic peaks of Pd 3d5/2 and Pd 3d3/2 corresponding to Pd2+ were 336.68 eV and 342.04 eV, respectively; the binding energies of the characteristic peaks of Pd 3d5/2 and Pd 3d3/2 corresponding to Pd4+ were 338.82 eV and 344.42 eV, respectively [34]. After activation, the binding energies of the two characteristic peaks corresponding to Pd2+ decreased to 336.60 eV and 341.95 eV, respectively, showing a slight shift toward the lower binding energy direction. This shifting trend is consistent with the changes observed in the Pt 4f and Al 2p characteristic peaks, which further corroborates the enhancement effect of metal-support interaction during the activation process.
2.2.6. Raman Characterization
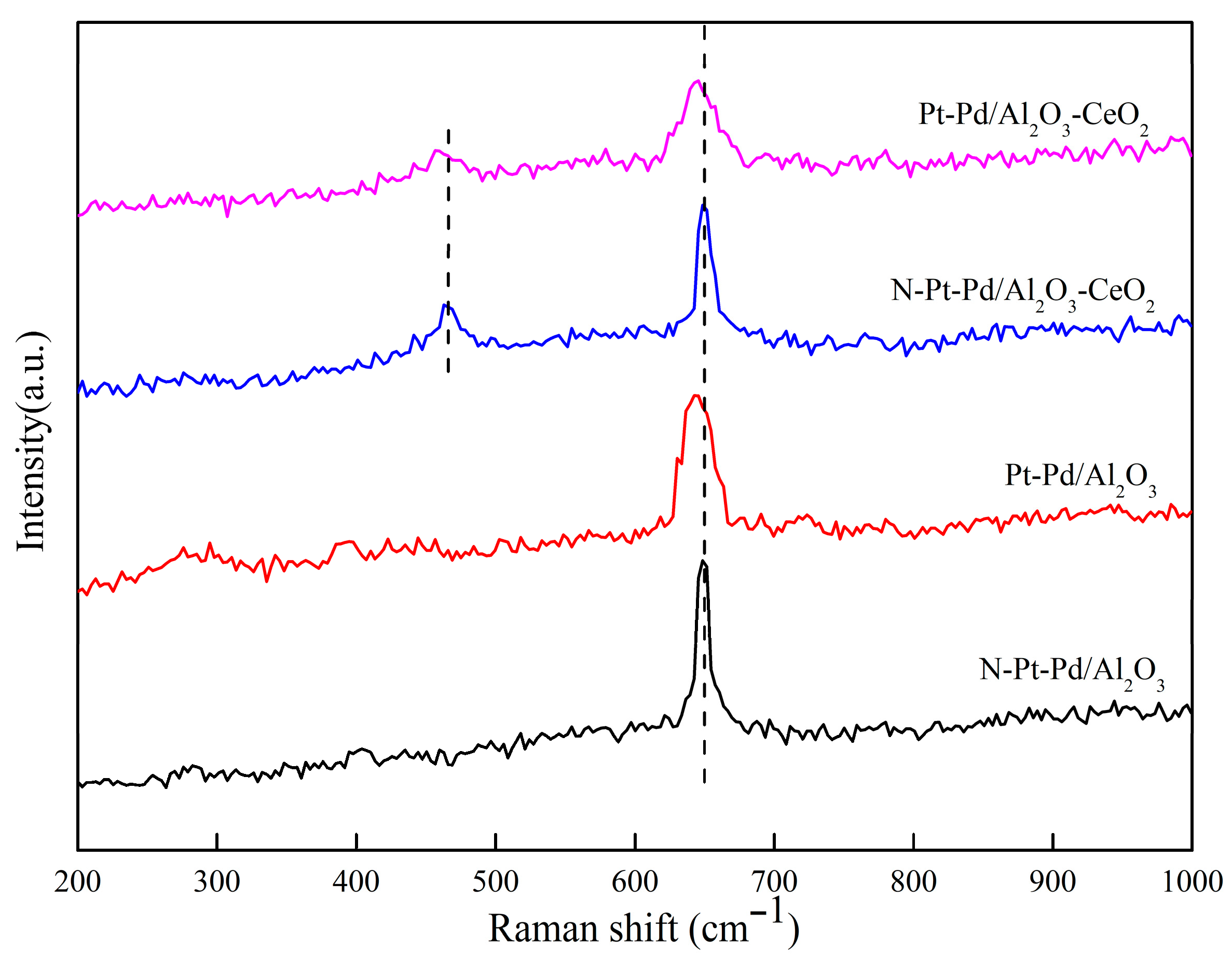
Figure 6 shows the Raman spectra of the catalysts prepared with different supports in both unactivated and activated states.
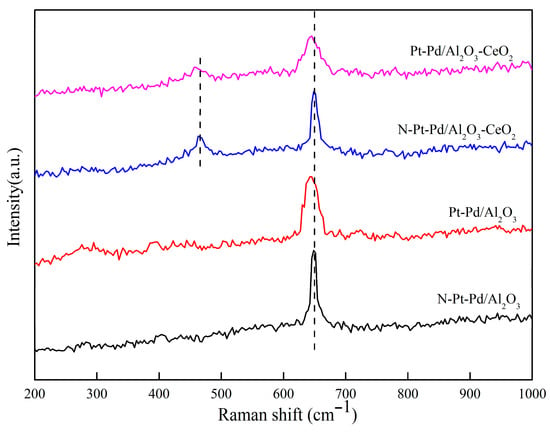
Figure 6.
Raman spectra of catalysts prepared with different supports.
Raman spectroscopy was employed to comparatively analyze the structural differences of catalysts prepared with different supports before and after activation, and the results are presented in Figure 6. The characteristic peak at 643 cm−1 corresponds to the B1g characteristic vibration peak of PdO species [35]; after catalyst activation, this peak shifts 3 cm−1 toward the lower-wavenumber direction with peak broadening observed. The characteristic peak at 465 cm−1 originates from the F2g symmetric stretching vibration of CeO2 [36,37], which also exhibits a 3 cm−1 shift to lower wavenumbers and peak broadening after activation.
A peak shift toward lower wavenumbers is generally associated with increased bond length and decreased bond energy of substances, while peak broadening can be attributed to the improved dispersion of active components and increased lattice defects. The aforementioned peak shifts and broadening phenomena directly reflect that the interaction between Pt-Pd active components and the support is significantly enhanced after catalyst activation. This interaction not only promotes the uniform dispersion of active components on the support surface, but also increases the quantity and exposure degree of active sites, thereby providing a favorable structural basis for subsequent catalytic reactions of methane elimination.
2.2.7. ICP-OES Quantitative Analysis
ICP-OES quantitative analysis was conducted to verify the nominal Pt-Pd loading of 0.5 wt% (Pt/Pd mass ratio = 1:2) in the prepared catalysts.
As shown in Table 6, ICP-OES quantitative analysis confirmed the accuracy of the metal precursor dosage. The results revealed that all four catalysts exhibited actual total Pt-Pd loadings in the range of 0.486–0.492 wt%, deviating by 1.6% to 2.8% from the nominal 0.5 wt%. The measured Pt/Pd mass ratios were consistently ~1:1.98, closely matching the target ratio of 1:2. These findings attest to the high precision and excellent reproducibility of the catalyst preparation process.

Table 6.
ICP-OES determination of catalysts with different supports.
2.2.8. H2-TPR Characterization
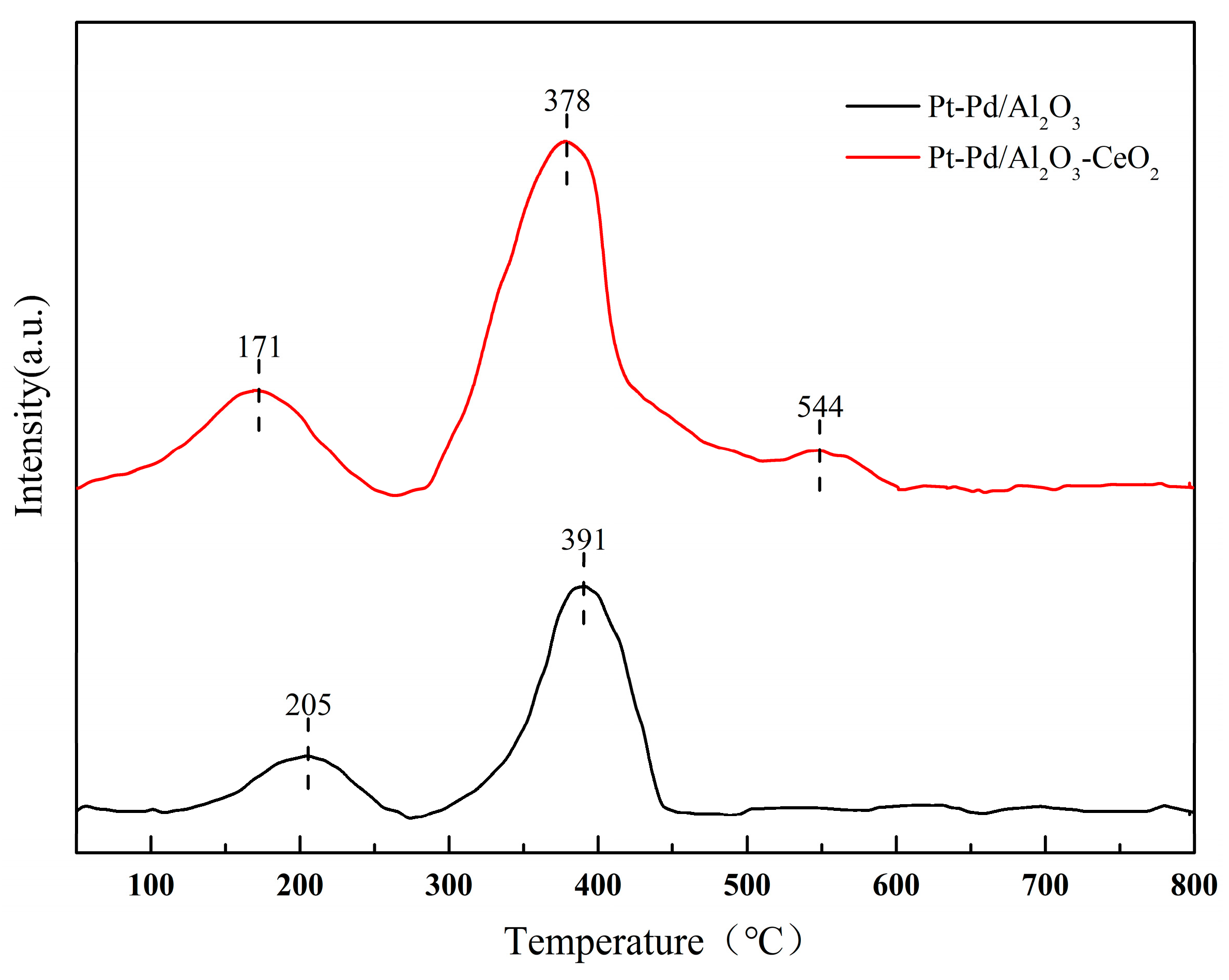
H2-TPR characterization was further used to evaluate the reducibility and reactive availability of the Pt-Pd active components. As shown in Figure 7.
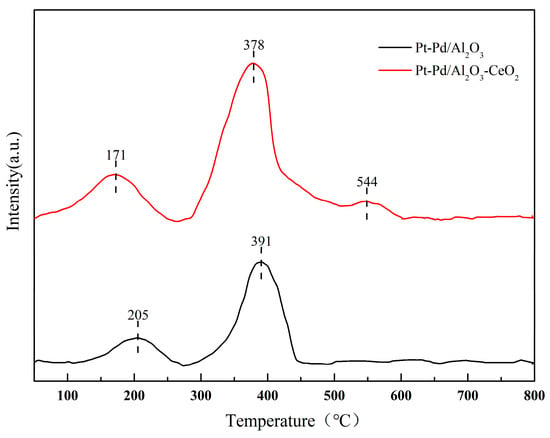
Figure 7.
H2-TPR profiles for Pt-Pd/Al2O3 and Pt-Pd/Al2O3-CeO2.
Pt-Pd/Al2O3 exhibits two main reduction peaks at 205 °C and 391 °C. The low-temperature peak (205 °C) is attributed to the reduction of PtO2 and/or surface Pd atoms, while the high-temperature peak (391 °C) corresponds to the reduction of PdO species strongly interacting with the Al2O3 support. After CeO2 modification, both peaks shift to lower temperatures (171 °C and 378 °C). This phenomenon is primarily due to the enhanced hydrogen spillover effect mediated by CeO2, which facilitates the transfer of active hydrogen to the metal oxide species, thereby accelerating the reduction kinetics. An additional peak at 544 °C is assigned to the bulk reduction of Ce4+. The decreased reduction temperature of the noble metals, coupled with the preserved Pd2+ state as confirmed by XPS, demonstrates that CeO2 not only improves the reducibility but also regulates the redox equilibrium, which is crucial for the stability of the zero-air generator [38,39].
2.2.9. Appearance Morphology
Figure 8 shows the appearance morphologies of different supports and their corresponding activated catalysts.

Figure 8.
Appearance images of different supports and their corresponding activated catalysts. (A) Al2O3; (B) Al2O3-CeO2; (C) Pt-Pd/Al2O3; (D) Pt-Pd/Al2O3-CeO2.
The alumina spherical support (A) prepared by the hot oil column method exhibits a uniform particle size distribution, providing an excellent physical basis for the subsequent loading of Pt-Pd bimetals. The support modified by ceria doping (B) presents a pale yellow color, which directly indicates that cerium species have been successfully incorporated into the support system and regulated its surface physicochemical properties. After activation, the Pt-Pd catalyst supported on pristine alumina (C) shows an uneven brown color on the surface, reflecting the inhomogeneous dispersion of noble metal particles. This phenomenon is mainly attributed to the random distribution of active sites on the surface of pristine alumina supports, which tends to cause agglomeration and segregation of noble metal particles. In contrast, the catalyst supported on ceria-doped alumina (D) displays a homogeneous light tan color after activation. This result demonstrates that ceria doping can construct abundant anchoring sites for noble metals on the support surface; by regulating the distribution of active sites, it significantly improves the dispersion of noble metal particles, thereby enhancing the atomic utilization efficiency of noble metals and boosting the potential catalytic performance of the catalyst. In summary, ceria doping modification is an effective technical strategy for optimizing the performance of alumina-based Pt-Pd catalysts.
2.3. Performance Comparison with Commercial Catalysts for Zero-Air Generators
To further validate the practical application value of the as-prepared Pt-Pd/Al2O3-CeO2 catalyst, a systematic performance comparison was conducted with four mainstream commercial Pt/Pd-based hydrocarbon removal catalysts (Parker Balston, Domnick Hunter, Claind ZeroAir HC Series, and PRUESI S30/S35) under simulated zero-air generator conditions, consistent with the test parameters described in Section 3.4. The results, summarized in Table S2, clearly demonstrate the superiority of our catalyst.
Despite its ultra-low total noble metal loading, which is significantly lower than that of the commercial catalysts, the Pt-Pd/Al2O3-CeO2 catalyst exhibits the best low-temperature activity, with a T90 of 374.43 °C. This outperforms the best commercial sample by ~10 °C. Most critically, while commercial catalysts left residual CH4 in the hundreds of ppb range and produced detectable CO (7.6–28.7 ppb), our catalyst achieved ultra-low outlet concentrations below the detection limit for both CH4 and CO simultaneously.
Accordingly, the Pt-Pd/Al2O3-CeO2 catalyst not only exhibits superior catalytic activity and deep purification performance but also enables a substantial reduction in precious metal usage, highlighting its significant technical and economic advantages for practical zero-air generation applications.
3. Materials and Methods
3.1. Reagents
Platinum nitrate and palladium nitrate were prepared by Purification Equipment Research Institute of CSSC (Handan, China) with purity ≥ 99.9%. Pt(NO3)2 (CAS 18496-40-7): Pt oxidation state +2, solvent deionized water, Pt mass fraction 15 wt%; Pd(NO3)2·2H2O (CAS 10102-05-3): Pd oxidation state +2, solvent deionized water, Pd mass fraction 10 wt%. Urea, hexamethylenetetramine, cerium nitrate hexahydrate and nitric acid were purchased from Sinopharm Chemical Reagents Co., Ltd. (Shanghai, China) with an analytical purity of ≥99%, along with pseudoboehmite powder (specific surface area was 254 m2/g, Al2O3 dry basis content was 69%).
3.2. Catalyst Preparation
3.2.1. Support Preparation
The alumina sol was prepared via the sol-gel method: 37.3 g of urea and 17.4 g of cerium nitrate hexahydrate were added to 160 g of distilled water, followed by stirring on a magnetic stirrer until homogeneous. Subsequently, 90 g of pseudoboehmite powder was slowly incorporated, and the mixture was stirred at 100 r/min for 30 min. Thereafter, 23 mL of nitric acid solution with a mass fraction of 20% was added, and stirring was continued for 2 h to form a stable cerium-promoted alumina sol (the Al2O3 content in the sol slurry was approximately 20 wt%). To the aforementioned alumina sol, 24.8 g of hexamethylenetetramine solution with a mass fraction of 20% was added, and continuous stirring was performed for 10 min to obtain a mixed sol.
The temperature of the hot oil column bath was set to 98 °C, and the oil phase height was controlled at 2 m. The mixed sol was added dropwise into the hot oil column for spheronization reaction. The spherical products were directly collected at the bottom of the apparatus, with a particle diameter ranging from 1.6 to 2 mm. The spherical products were washed with distilled water three times, then dried at 120 °C for 2 h, and subsequently calcined at 600 °C for 4 h to obtain CeO2-doped Al2O3 spheres (CeO2 weight fraction: 10%).
Pure Al2O3 spheres without CeO2 doping were prepared using the identical procedure described above, with the sole exception of omitting the addition of cerium nitrate hexahydrate.
3.2.2. Active Metal Loading
3.3 g of palladium nitrate solution (10 wt%) and 1.1 g of platinum nitrate solution (15 wt%) were added to 90 g of deionized water. The mixture was stirred to homogeneity to yield a precious metal precursor solution. This precursor solution was preheated to 80 °C in a water bath, then poured into a coating machine preheated to 70 °C. Next, 100 g of the support prepared in step (1) was added to the precursor solution in the coating machine, and rotary evaporation drying was conducted at a rotation speed of 15 r/min.
The dried catalyst was placed in an oven and dried at 120 °C for 4 h. After drying, the temperature was ramped up to 500 °C at a heating rate of 3 °C/min, followed by calcination at this temperature for 2 h. Finally, a Pt-Pd bimetallic catalyst was obtained (total precious metal content: 0.5 wt%, Pt/Pd mass ratio: 1:2).
Catalysts were prepared using the undoped Al2O3 support and the CeO2-doped Al2O3 support (Al2O3-CeO2), which were designated as N-Pt-Pd/Al2O3 and N-Pt-Pd/Al2O3-CeO2, respectively.
3.2.3. Catalyst Activation
The catalyst sample was placed in the reaction tube of a tube furnace. The temperature was programmed to increase to 400 °C, after which a 0.5% CH4 mixed gas (balanced with air) was introduced for continuous activation for 2 h. Upon completion of activation, the catalyst was naturally cooled to room temperature, then taken out and labeled as Pt-Pd/Al2O3 and Pt-Pd/Al2O3-CeO2, respectively.
3.3. Catalyst Characterization
3.3.1. X-Ray Powder Diffraction (XRD)
The phase compositions of different catalysts were analyzed using a D8 ADVANCE X-ray diffractometer manufactured by Bruker AXS GmbH, Karlsruhe, Germany, and the XRD data were processed and analyzed using Jade 6.5 software. The test conditions were set as follows: Cu target (Kα radiation, wavelength = 1.5406 Å), scanning range (2θ) = 10–90°, and scanning rate = 6°/min.
3.3.2. Surface Property Analysis (BET)
The specific surface area, pore volume, and pore size of different supports and catalysts were measured using an ASAP 3020 high-performance specific surface area and micropore analyzer produced by Micromeritics Instrument Corporation, Norcross, GA, USA. Data were analyzed using MicroActive Version 3.02 software. Prior to testing, the samples were degassed at 200 °C for 6 h to remove impurity gases. The instrument recorded the physical adsorption isotherms at 77 K. The specific surface area was calculated using the Brunauer-Emmett-Teller (BET) method, while the pore volume and average pore size were derived via the Barrett-Joyner-Halenda (BJH) method. All calculations were carried out using MicroActive software (Micromeritics).
3.3.3. Laser Raman Spectroscopy (Raman)
Raman characterization was performed on a LabRAM HR Evolution laser Raman spectrometer from Horiba Scientific, Palaiseau, France. Data acquisition and processing were carried out using LabSpec 5 software. The test conditions were configured as: laser wavelength = 532 nm, wavenumber range = 100–3500 cm−1, and parallel measurements were conducted twice for each sample.
3.3.4. X-Ray Photoelectron Spectroscopy (XPS)
XPS measurements were carried out using a Nexsa G2 X-ray photoelectron spectrometer manufactured by Thermo Fisher Scientific Inc., Waltham, MA, USA. Data acquisition and analysis were performed using Avantage software (version 59931). The test conditions were: Al Kα X-ray source, and the binding energy scale was calibrated against the C 1s peak at 284.6 eV.
3.3.5. Scanning Electron Microscopy (SEM)
The morphology of the catalysts was observed using an Apreo C scanning electron microscope from Thermo Fisher Scientific Inc.,Waltham, MA, USA. Image analysis was performed using Avizo 2023.2 and ImageJ 1.54. The samples were subjected to rotary sputter coating with gold at 10 kV for 60 s, and morphological observations were conducted under the microscope after uniform coating.
3.3.6. High-Resolution Transmission Electron Microscopy (HR-TEM) and Energy-Dispersive X-Ray Spectroscopy (EDS) Elemental Mapping
The morphological features and lattice fringes of the catalyst samples were characterized using a JEM-2100F field emission transmission electron microscope (JEOL Ltd., Akishima, Japan). Images were analyzed using ImageJ 1.53 and DigitalMicrograph 3.4. For sample preparation, the catalysts were dispersed in anhydrous ethanol and subjected to ultrasonic treatment for 10–15 min to achieve uniform dispersion. A small aliquot of the resulting suspension was drop-cast onto a carbon-coated copper grid, which was then air-dried naturally at room temperature prior to microscopic measurements. EDS elemental mapping was simultaneously conducted on the same instrument to analyze the spatial distribution of Pt and Pd active components on the support surface.
3.3.7. H2-Chemisorption
H2-chemisorption measurements were performed on a Micromeritics AutoChem II 2920 instrument (Micromeritics, USA). Data were analyzed using ChemiSorb Software Version 2.00 software. Approximately 100 mg of catalyst was loaded into a quartz reactor and pretreated under high-purity Ar flow (50 mL min−1) at 300 °C for 1 h to eliminate surface-adsorbed water and impurities. After cooling to 50 °C, pulse chemisorption was carried out using high-purity H2 (99.999%) as the adsorbate and Ar as the carrier gas.
3.3.8. H2-Temperature Programmed Reduction (H2-TPR)
The test was carried out on a Micromeritics AutoChem II 2920 instrument (Micromeritics, USA). Data were analyzed using ChemiSorb Software Version 2.00 software. Approximately 50 mg of the catalyst sample was used for temperature-programmed reduction (TPR) under a 5% H2-Ar gas mixture. The temperature was ramped from 50 to 800 °C at a heating rate of 10 °C·min−1.
3.3.9. Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES)
The quantitative determination was performed using an ICP-OES 730 spectrometer (Agilent Technologies, Santa Clara, CA, USA). Data acquisition and analysis were carried out using ICP Expert II V2.0 software. Catalyst samples were completely dissolved via high-temperature acid digestion with a HNO3-HCl-HF mixed solution, and measurements were conducted at the optimal spectral lines (Pt: 265.945 nm, Pd: 340.458 nm).
3.4. Catalyst Evaluation
The test gas was configured in a simulated air atmosphere (O2: 21 vol%, balance N2) with methane (the representative hydrocarbon) at a concentration of 50 ppm, and The gas hourly space velocity (GHSV) was set to 10,000 h−1, which was calculated as the ratio of the actual gas flow rate to the bulk volume of the catalyst packed bed (the standard calculation basis for GHSV). The catalyst module device of a commercially available zero air generator was simulated. The reaction tube was cylindrical with an inner diameter of 12 mm and a height of 300 mm. A stainless-steel mesh with a diameter of 12 mm and a pore size of 1 mm was placed at both the upper and lower ends inside the reaction tube, respectively. The inner diameter of the stainless-steel gas inlet and outlet pipes at both ends was 3 mm. 20 g of the catalyst was loaded into the reaction tube, with the measured bulk volume of the catalyst packed bed being 34 mL and the bed porosity of 0.485, the actual volumetric gas flow rate was controlled at 340 L/h by a mass flow controller to achieve the set GHSV. The temperature of the reaction tube was controlled by a tube furnace.
The concentration of methane after elimination by the above catalyst was measured using a 7890A gas chromatograph manufactured by Agilent Technologies. The oxidation products CO and CO2 were synchronously detected with the same gas chromatograph coupled with a methanizer, and the carbon balance of the catalytic reaction was calculated based on the inlet CH4 concentration and outlet CO/CO2 concentrations. The LOD and LOQ were calculated according to the IUPAC recommendation using the equations: LOD = 3.3 × σ/S and LOQ = 10 × σ/S, where σ is the standard deviation of the baseline response at the methane retention time from seven replicate blank measurements, and S is the slope of the calibration curve. The blank was a methane-free simulated air matrix (21 vol% O2, N2 balance). The calibration curve was established with five standard methane samples: 10, 20, 30, 40, and 50 ppb. Based on the parameters σ = 0.0226 and S = 0.0524 (R2 = 0.9998), the LOD and LOQ were calculated to be 1.42 ppb and 4.31 ppb, respectively.
Methane conversion rate was calculated based on the inlet and outlet concentration difference with the formula: , where is the methane conversion rate (%), is the inlet methane concentration (ppm), and is the outlet methane concentration (ppm/ppb).
4. Conclusions
In this study, the cerium dioxide-doped alumina composite support was prepared via the hot oil column method, followed by the successful synthesis of the supported Pt-Pd bimetallic catalyst using the impregnation method. Systematic characterization results demonstrate that the support type and catalyst activation mode can significantly affect the methane oxidation activity of the catalytic material by regulating the interaction strength between the active components and the support, as well as optimizing the number of oxygen vacancies on the surface of both the support and the catalyst.
The results of methane catalytic oxidation performance tests indicate that the Pt-Pd/Al2O3-CeO2 composite-supported catalyst exhibits superior methane elimination performance: at a reaction temperature of 400 °C and an inlet methane concentration of 50 ppm, the outlet methane concentration can be reduced to below 10 ppb. Furthermore, this catalyst possesses excellent long-term operational stability; even after 1000 h of continuous operation, it can still stably maintain high-efficiency methane elimination efficiency.
In conclusion, all performance indicators of this catalyst meet the application standards and service requirements for zero-air generators. Meanwhile, its low noble metal content can significantly cut down the preparation cost, endowing the catalyst with important practical application value and promising promotion potential.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal16030265/s1. Table S1. Methane catalytic oxidation performance of Pt-Pd bimetallic catalysts with different mass ratios. Table S2. Performance comparison with commercial catalysts for zero-air generators (50 ppm CH4)
Author Contributions
Conceptualization, L.Z.; methodology, L.Z.; validation, Z.C. and Q.W.; formal analysis, Z.P. and W.T.; data curation, J.H.; writing—original draft preparation, L.Z.; writing—review and editing, L.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All data have been provided in the manuscript.
Acknowledgments
The authors acknowledge the financial support and research equipment of Purification Equipment Research Institute of CSSC.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Hill-Pearce, R.E.; Resner, K.V.; Worton, D.R.; Brewer, P.J. A synthetic zero air reference material forhigh accuracy greenhouse gas measurements. Anal. Chem. 2019, 91, 1974–1979. [Google Scholar] [CrossRef] [PubMed]
- Englert, J.; Claude, A.; Demichelis, A.; Persijn, S.; Baldan, A.; Li, J.R.; Plass-Duelmer1, C.; Michl, K.; Tensing, E.; Wortman, R.; et al. Preparation and analysis of zero gases for the measurement of trace VOCs in air monitoring. Atmos. Meas. Tech. 2018, 11, 3197–3203. [Google Scholar] [CrossRef]
- Miñarro, M.D.; Brewer, P.J.; Brown, R.J.C.; Persijn, S.; Wijk, J.; Nieuwenkamp, G.; Baldan, A.; Kaiser, C.; Sutour, C.; Macé, T.; et al. Zero gas reference standards. Anal. Methods 2016, 8, 3014–3022. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Determination of Total Gaseous Organic Concentration Using a Flame Ionization Analyzer; Method 25A; U.S. Environmental Protection Agency: Washington, DC, USA, 2017; pp. 1–7. [Google Scholar]
- HJ 38-2017; Stationary Source Emission-Determination of Total Hydrocarbons, Methane and Nonmethane Hydrocarbons-Gas Chromatography. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2017.
- HJ 604-2017; Ambient Air-Determination of Total Hydrocarbons, Methane and Nonmethane Hydrocarbons-Direct Injection/Gas Chromatography. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2017.
- China National Environmental Monitoring Centre. Technical Specifications for Continuous Automatic Monitoring of Non-Methane Total Hydrocarbons in Ambient Air (Trial Implementation); China National Environmental Monitoring Centre: Beijing, China, 2021; pp. 1–27, Zongzhan Qi Zi No. 61. [Google Scholar]
- Gélin, P.; Primet, M. Complete oxidation of methane at low temperature over noble metal based catalysts: A review. Appl. Catal. B Environ. 2002, 39, 1–37. [Google Scholar] [CrossRef]
- Ciuparu, D.; Lyubovsky, M.R.; Altman, E.; Pfefferle, L.D.; Datye, A. Catalytic combustion of methane over palladium-based catalysts. Catal. Rev. 2002, 44, 593–649. [Google Scholar] [CrossRef]
- Tabakova, T.; Grahovski, B.; Karakirova, Y.; Petrova, P.; Venezia, A.M.; Liotta, L.F.; Todorova, S. Effect of support on complete hydrocarbon oxidation over Pd-based catalysts. Catalysts 2025, 15, 110. [Google Scholar] [CrossRef]
- Zhang, Y.; Lv, Y.M.; Mo, Y.F.; Li, H.Y.; Tang, P.G.; Li, D.Q.; Feng, Y.J. Facile preparation and promising hydrothermal stability of spherical γ-alumina support with high specific surface area. Catalysts 2022, 12, 1416. [Google Scholar] [CrossRef]
- Wang, S.J.; Tian, Z.W.; Liu, Q.; Qiao, Y.Y.; Tian, Y.Y. Facile preparation of a Ni/MgAl2O4 catalyst with high surface area: Enhancement in activity and stability for CO methanation. Main Group Met. Chem. 2018, 41, 73–89. [Google Scholar] [CrossRef]
- Oh, D.H.; Jo, Y.R.; Chang, J.; An, H.; Kim, H.J.; Vohs, J.M.; Jung, W.C.; Lee, S. Impact of CaO-modified γ-Al2O3 support on CO oxidation activity of Pt/LaFeO3 catalyst. ACS Appl. Mater. Interfaces 2024, 16, 64714–64724. [Google Scholar] [CrossRef]
- AbdelDayem, H.M.; Salib, B.G.; El-Hosiny, F.I. Facile synthesis of hydrothermal stable hierarchically macro-mesoporous hollow microspheres γ-Al2O3-graphene oxide composite: As a new efficient acid-base catalyst for transesterification reaction for biodiesel production. Fuel 2020, 277, 118106. [Google Scholar] [CrossRef]
- Islam, A.; Taufiq-Yap, Y.H.; Chu, C.M.; Chan, E.S.; Ravindra, P. Synthesis and characterization of millimetric gamma alumina spherical particles by oil drop granulation method. J. Porous Mater. 2012, 19, 807–817. [Google Scholar] [CrossRef]
- Yang, S.W.; Maroto-Valiente, A.; Benito-Gonzalez, M.; Rodriguez-Ramos, I.; Guerrero-Ruiz, A. Methane combustion over supported palladium catalysts: I. Reactivity and active phase. Appl. Catal. B Environ. 2000, 28, 223–233. [Google Scholar] [CrossRef]
- Auvray, X.; Lindholm, A.; Milh, M.; Olsson, L. The addition of alkali and alkaline earth metals to Pd/Al2O3 to promote methane combustion. Effect of Pd and Ca loading. Catal. Today 2018, 299, 212–218. [Google Scholar] [CrossRef]
- Fan, X.; Wang, F.; Zhu, T.L.; He, H. Effects of Ce on catalytic combustion of methane over Pd-Pt/Al2O3 catalyst. J. Environ. Sci. 2012, 24, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.H.; Zhou, R.X.; Wang, Y.J.; Han, X.X.; Zheng, X.M. Study on the combustion behavior of methane over Ce-Zr-modified Pd/Al2O3 catalysts. Appl. Surf. Sci. 2005, 246, 36–43. [Google Scholar] [CrossRef]
- Arosio, F.; Colussi, S.; Trovarelli, A.; Groppi, G. Effect of alternate CH4-reducing/lean combustion treatments on the reactivity of fresh and S-poisoned Pd/CeO2/Al2O3 catalysts. Appl. Catal. B Environ. 2008, 80, 335–342. [Google Scholar] [CrossRef]
- Lee, Y.; He, G.; Akey, A.J.; Si, R.; Flytzani-Stephanopoulos, M.; Herman, I.P. Raman analysis of mode softening in nanoparticle CeO2-δ and Au-CeO2-δ during CO Oxidation. J. Am. Chem. Soc. 2011, 133, 12952–12955. [Google Scholar] [CrossRef]
- Qi, W.J.; Ran, J.Y.; Wang, R.R.; Du, X.S.; Shi, J.; Niu, J.T.; Zhang, P.; Ran, M.C. Kinetic consequences of methane combustion on Pd, Pt and Pd-Pt catalysts. RSC Adv. 2016, 6, 109834–109845. [Google Scholar] [CrossRef]
- Enache, D.I.; Edwards, J.K.; Landon, P.; Solsona-Espriu, B.; Carley, A.F.; Herzing, A.A.; Watanabe, M.; Kiely, C.; Knight, D.W.; Hutchings, G.J. Solvent-free oxidation of primary alcohols to aldehydes using Au-Pd/TiO2 catalysts. Science 2011, 311, 362–365. [Google Scholar] [CrossRef]
- Geng, H.J.; Zhang, L.; Yang, Z.Q.; Yan, Y.F.; Ran, J.Y. Effect of Pd/Pt ratio on the reactivity of methane catalytic combustion in bimetallic Pd-Pt catalyst. Int. J. Hydrogen Energy 2018, 43, 11069–11078. [Google Scholar] [CrossRef]
- Yang, Y.F.; Lee, J.; Dorakhan, R.; Nie, H.Y.; Fu, G.S.; Quarantotto, A.; Howe, J.Y.; Chin, Y. Active site structure and methane oxidation reactivity of bimetallic Pd and Pt nanoparticles. Appl. Catal. A Gen. 2022, 629, 118290. [Google Scholar] [CrossRef]
- Stotz, H.; Maier, L.; Boubnov, A.; Gremminger, A.T.; Grunwaldt, J.D.; Deutschmann, O. Surface reaction kinetics of methane oxidation over PdO. J. Catal. 2019, 370, 152–175. [Google Scholar] [CrossRef]
- Xu, P.; Zhang, X.; Zhao, X.T.; Yang, J.; Hou, Z.Q.; Bai, L.; Chang, H.Q.; Liu, Y.X.; Deng, J.G.; Guo, G.S.; et al. Preparation, characterization, and catalytic performance of PdPt/3DOM LaMnAl11O19 for the combustion of methane. Appl. Catal. A Gen. 2018, 562, 284–293. [Google Scholar] [CrossRef]
- Darda, S.; Pachatouridou, E.; Lappas, A.; Iliopoulou, E. Effect of preparation method of Co-Ce catalysts on CH4 combustion. Catalysts 2019, 9, 219. [Google Scholar] [CrossRef]
- AlMohamadi, H.; Smith, K.J. The impact of CeO2 loading on the activity and stability of PdO/γ-AlOOH/γ-Al2O3 monolith catalysts for CH4 oxidation. Catalysts 2019, 9, 557. [Google Scholar] [CrossRef]
- Pawelec, B.; Mariscal, R.; Navarro, R.M.; Bokhorst, S.; Rojas, S.; Fierro, J.L.G. Hydrogenation of aromatics over supported Pt-Pd catalysts. Appl. Catal. A 2002, 225, 223–237. [Google Scholar] [CrossRef]
- Pawelec, B.; Murcia-Mascarós, S.; Fierro, J.L.G. Surface and structural features of Pt/Pd-loaded mesoporous silica-delaminated zirconium phosphate systems. Langmuir 2002, 18, 7953–7963. [Google Scholar] [CrossRef]
- Liyanage, A.D.; Perera, S.D.; Tan, K.; Chabal, Y.; Balkus, K.J. Synthesis, characterization, and photocatalytic activity of Y-doped CeO2 nanorods. ACS Catal. 2014, 4, 577–584. [Google Scholar] [CrossRef]
- Chen, J.J.; Wu, Y.; Hu, W.; Qu, P.F.; Liu, X.; Yuan, R.; Zhong, L.; Chen, Y.Q. Insights into the role of Pt on Pd catalyst stabilized by magnesia-alumina spinel on gama-alumina for lean methane combustion: Enhancement of hydrothermal stability. Mol. Catal. 2020, 496, 111185. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Cai, Y.F.; Guo, Y.; Wang, H.F.; Wang, L.; Lou, Y.; Guo, Y.L.; Lu, G.Z.; Wang, Y.Q. The effects of the Pd chemical state on the activity of Pd/Al2O3 catalysts in CO oxidation. Catal. Sci. Technol. 2014, 4, 3973–3980. [Google Scholar] [CrossRef]
- Shi, C.K.; Yang, L.F.; Cai, J.X. Cerium promoted Pd/HZSM-5 catalyst for methane combustion. Fuel 2007, 86, 106–112. [Google Scholar] [CrossRef]
- Wang, N.; Wang, S.; Yang, J.; Xiao, P.; Zhu, J.J. Promotion effect of Ce doping on catalytic performance of LaMnO3 for CO Oxidation. Catalysts 2022, 12, 1409. [Google Scholar] [CrossRef]
- Chen, S.Y.; Li, S.D.; You, R.Y.; Guo, Z.Y.; Wang, F.; Li, G.X.; Yuan, W.T.; Zhu, B.E.; Gao, Y.; Zhang, Z.; et al. Elucidation of active sites for CH4 catalytic oxidation over Pd/CeO2 via tailoring metal-support interactions. ACS Catal. 2021, 11, 5666–5677. [Google Scholar] [CrossRef]
- Fan, R.R.; Li, Z.Q.; Wang, Y.; Wang, Y.; Ding, Z.Y.; Zhang, C.; Kang, N.; Guo, X.; Wang, R. Promotional effect of ZrO2 and WO3 on bimetallic Pt-Pd dieseloxidation catalyst. Environ. Sci. Pollut. Res. 2022, 29, 5282–5294. [Google Scholar] [CrossRef] [PubMed]
- Pitkäaho, S.; Matejova, L.; Jiratova, K.; Ojala, S.; Keiski, R.L. Oxidation of perchloroethylene-Activity and selectivity of Pt, Pd, Rh, and V2O5 catalysts supported on Al2O3, Al2O3-TiO2 and Al2O3-CeO2. Part 2. Appl. Catal. B Environ. 2012, 126, 215–224. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.