Abstract
This study systematically investigates how organic ligand modifications—chain length adjusting and functional group incorporation—regulate the catalytic performance of UiO-66 derivatives for CO2-to-dimethyl carbonate (DMC) conversion. Through multi-technique characterization (Py-IR, TGA, FT-IR, XPS, etc.) and catalytic tests, Lewis acid/basic sites (LAS/LBS), bulk defects and electron density effects were identified as the three key factors to govern the catalytic activity. The bulk defects were believed to enhance mass transfer. Notably, MOF-801 (shortest ligand) and UiO-66-Br (electron-withdrawing-Br) achieved the highest TOFs of 0.86 h−1 and 1.10 h−1, respectively. While LAS/LBS and electron-rich Zr clusters promote methanol activation, defect-enhanced mass transfer dominated over electronic effects in boosting DMC yield. These insights highlight the tunability of MOFs for CO2 utilization via rational ligand design.
1. Introduction
As a “new block” of organic synthesis in the 21st century, dimethyl carbonate (DMC) has been widely used in industrial fields and some researchers predict that a new chemical group composed of DMC and its many derivatives will be formed in the near future [1,2]. At present, a variety of DMC process routes have been industrialized, including phosgenation, methanol oxidation carbonylation, transesterification, urea alcoholysis, and direct synthesis route of CO2 and methanol. The conventional DMC synthesis routes suffer from severe drawbacks including toxic feedstock, corrosive byproduct and explosive intermediates. These inherent risks, coupled with soaring costs of waste handling, have propelled CO2 direct esterification to DMC to be a green, promising alternative in modern chemical industry [3,4,5,6,7]. However, this pathway is severely hampered by several intertwined challenges [8,9]. Firstly, the reaction faces significant thermodynamic constraints, for instance, in CeO2-catalyzed systems, exhibiting a notably low equilibrium constant (e.g., K ~ 10−5–10−7 in the range of 80–180 °C), which inherently limits the CO2 conversion [10]. Concurrently, catalyst stability is significantly compromised by distinct deactivation mechanisms. Supported metal catalysts (e.g., Fe, Cu) are prone to carbon deposition from side reactions, leading to active site contamination [11], while inappropriate modification strategies, such as excessive La doping in LaxCeO2, can induce structural collapse and aggregation of nano-architectures, resulting in catalyst deactivation [12].
In the last decade, metal–organic frameworks (MOFs) have been intensively studied in CO2 utilization field. The adjustable architectures of MOFs, during which linker functionalization (-X) takes place, as well as linker chain length provide a promising strategy to overcome CO2-to-DMC bottlenecks [13,14,15]. Xuan et al. systematically studied MOF-808 for the CO2-to-DMC system, the results of which highlighted the pivotal role of micropore size in mass transfer, emphasizing accessibility over absolute site density; a DMC yield of 21.5% was obtained [16]. Furthermore, Xu and co-workers developed a composite strategy by dispersing CeO2 nanoparticles on the UiO-66 surface, which provided abundant oxygen vacancies for CO2 capture, leading to a synergistic effect and a high DMC formation rate of 2.84 mmol·g−1·h−1 [17]. Recent advances in the direct synthesis of dimethyl carbonate (DMC) from CO2 and methanol have demonstrated the efficiency of organic linker modification in fine-tuning the catalytic properties MOFs, particularly within the UiO-66 family. Xuan et al. utilized trifluoroacetic acid (TFA) as a modulator to engineer defects in UiO-66-X, effectively tailoring its porosity and increasing the concentration of Lewis acid–base sites and hydroxyl groups, which collectively contributed to a marked enhancement in catalytic activity [18]. Huo and colleagues prepared a series of functionalized UiO-66-X catalysts, revealing that the electron-withdrawing ability of the substituents (-F, -Cl, -Br) inversely correlated with the population of weak acid sites; consequently, the UiO-66-Br catalyst, possessing the highest density of these sites, delivered the superior DMC formation rate, underscoring the critical importance of optimal acid–base characteristics for the synergistic activation of CO2 and methanol [19]. In a complementary strategy, Zhang et al. incorporated imidazole carboxylate as a secondary ligand to construct UiO-66-Im-X materials. This approach successfully increased linker defects within the Zr6 clusters, which significantly enhanced Lewis acidity. The optimal catalyst, UiO-66-Im-0.2, achieved a DMC yield of 0.9335% at 140 °C and 3.5 MPa over 8 h and retained over 90% of its initial activity after four catalytic cycles, demonstrating excellent stability [20]. Li and co-workers have introduced 2-methylimidazole (2MI) into Cu-MOFs to replace tricarboxylic acid (BTC) as organic ligand, increasing the inherent heterogenous rate constant (k0) from 0.09 cm·s−1 to 0.13 cm·s−1. The larger surface area and faster electron transfer ability generated from the use of 2MI ligands contributed to the improved reaction rate [21]. Although works have applied the adjusting organic linker strategy, relatively fewer research studies are available on the mechanism clarification regarding how chain length and functional groups affect the properties and activities of MOFs up to now.
Herein, this work aims at revealing the effect of chain length and functional groups of organic linkers on thermocatalytic features of UiO-66 experimentally and systematically. Through experimental studies, we seek to establish the correlation between these structural modifications and the material’s defects, Lewis acidity/basicity, electron density, and ultimately, its catalytic activity. To achieve such a goal, four Zr-centered MOFs with different linker chain lengths, and another four with different electron donating/withdrawing functional groups are designed and synthesized (Figure 1). The derived MOFs were applied for the CO2 conversion, the experimental results indicate that among the first series, MOF-801 material exhibited the highest TOF of 0.86 h−1, while UiO-66-Br demonstrates the highest TOF of 1.10 h−1 among the second series, suggesting the significant ligand effects on the CO2 conversion. Lewis acid/basic sites (LAS/LBS), bulk defects (affect the mass transfer), and electron density effects are believed to be the three crucial factors that contribute to the activity of this MOF series.
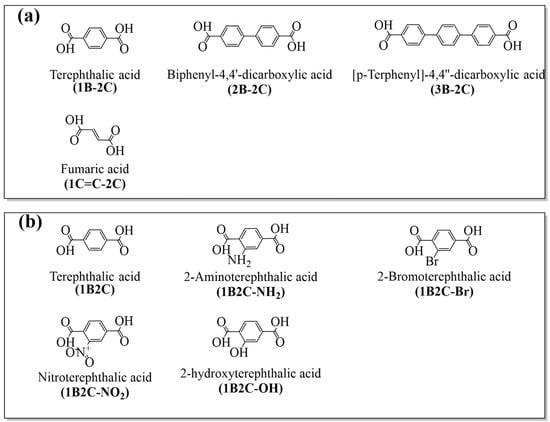
Figure 1.
(a) Organic linkers with varied chain lengths, (b) organic linker with different functional groups.
2. Results and Discussion
2.1. Effects of Chain Length of Organic Ligands on the MOFs
2.1.1. Structure and Morphology Analysis
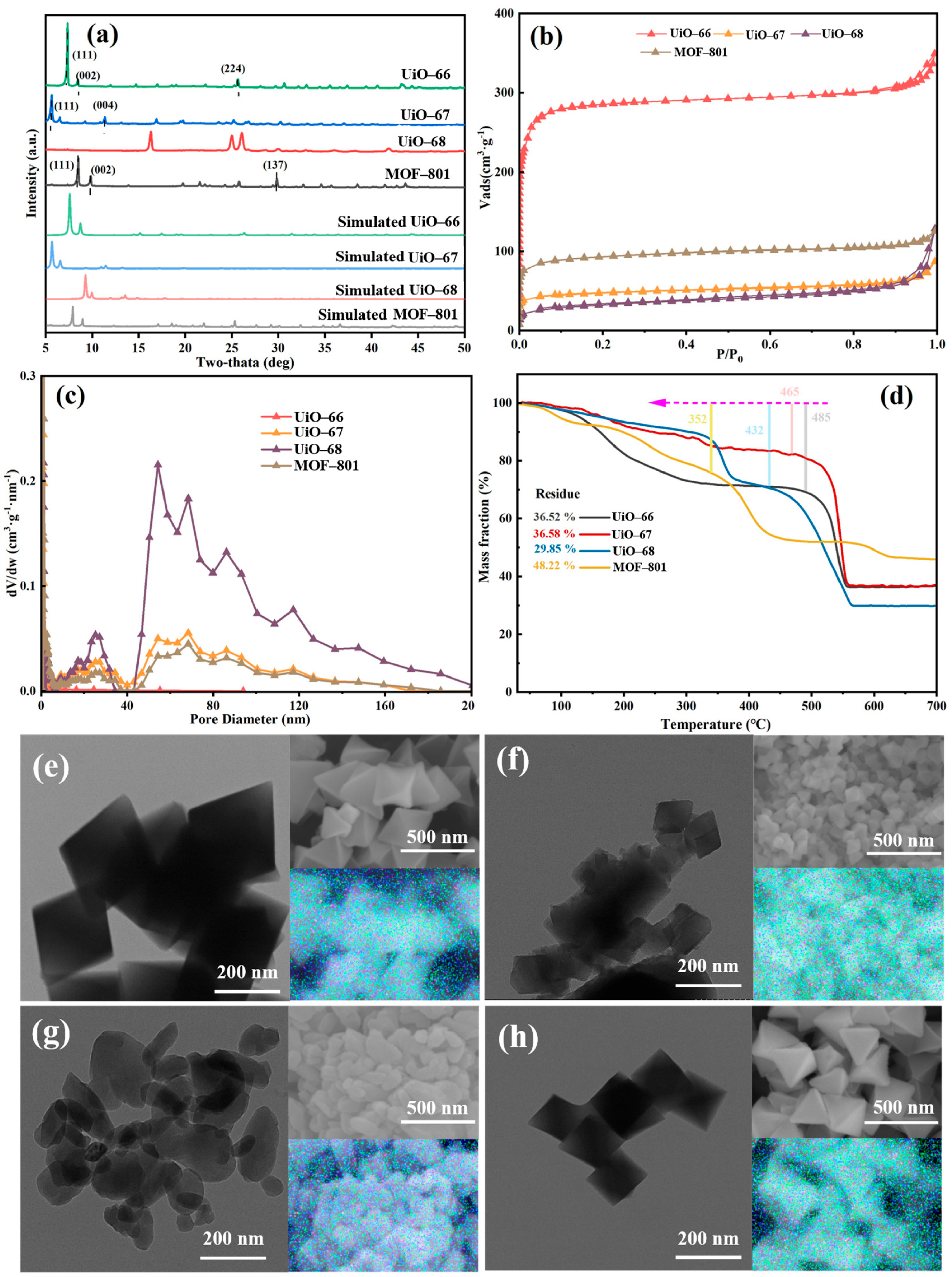
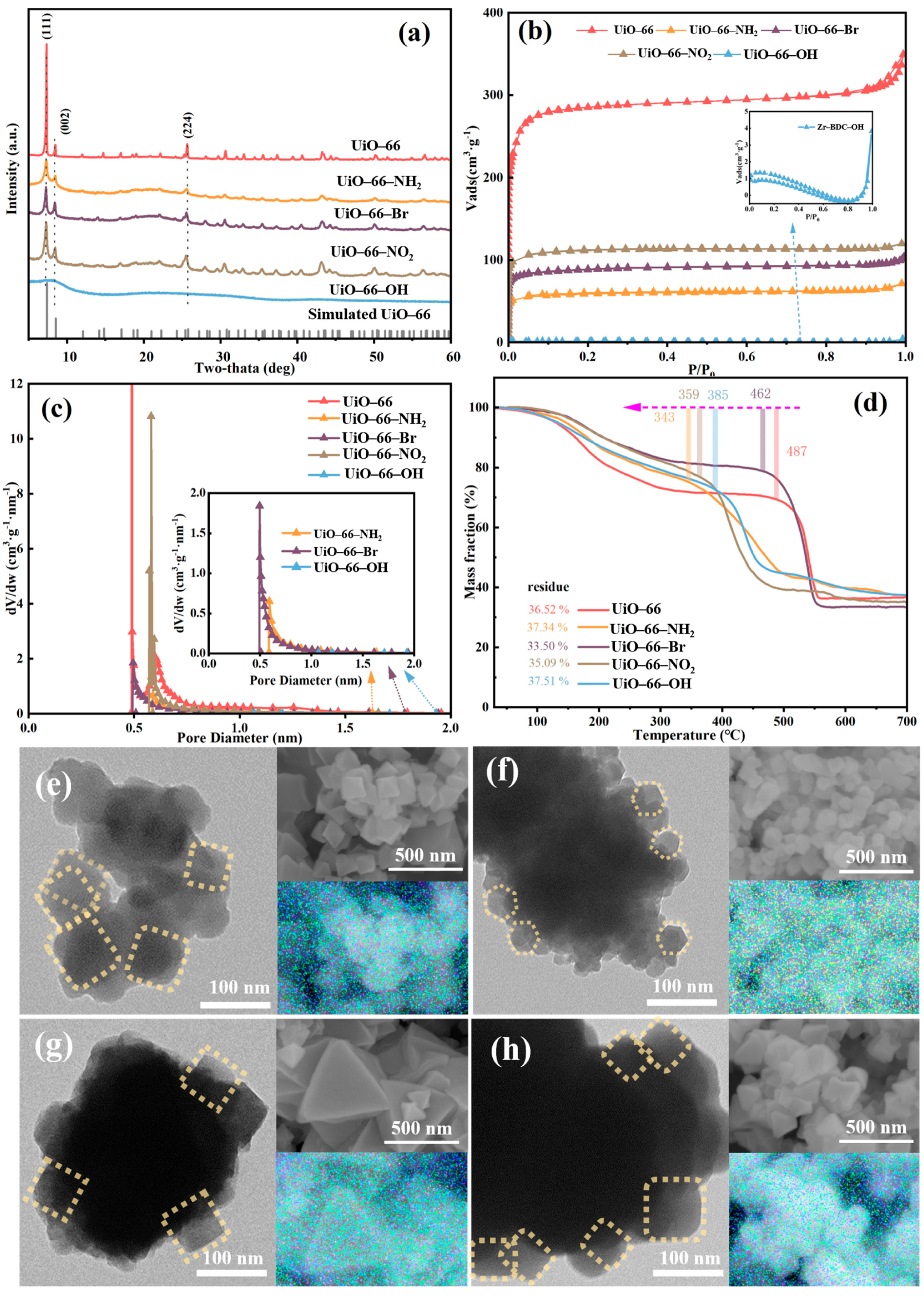
The features of four synthesized MOFs, namely UiO-66, UiO-67, UiO-68 and MOF-801, were investigated using different techniques. As shown in Figure 2a, the XRD patterns of UiO-66, UiO-67, and MOF-801 represent that of UiO-66 [22], UiO-67 [23] and MOF-801 [24] structures, suggesting the successful synthesis of the three MOFs. However, a well-defined UiO-68 crystal structure could not be formed, primarily due to the significant kinetic barriers and steric hindrance introduced by the long-chain linker during crystallization, which prevents orderly assembly with zirconium clusters, leading to high defect concentrations and even framework collapse [25]. TEM and SEM images in Figure 2 reveal morphological variations when the chain lengths changed. UiO-66 is in the form of perfect octahedral, the size of which is around 400 nm. With increasing ligand chain dimensionality, UiO-68 exhibited size reduction (decreased to around 150 nm). With further increase in chain length, partial amorphization was observed for UiO-68, which is consistent with the XRD results. Notably, MOF-801 maintains octahedral structure with smaller particle size. Additionally, all the elements were uniformly distributed in all four materials, as shown in Figure 2. To investigate the porous structure of these four materials, N2 adsorption–desorption analysis was performed. All of the composites exhibited a typical type-I isotherm, indicating the presence of microporosity [26]. The N2 adsorption–desorption isotherms of all four materials exhibited a Type-I profile (Figure 2b), indicating that the microporous nature of the UiO-type framework was maintained despite the variation in linker length. However, a quantitative analysis reveals that the porous properties were profoundly altered. As summarized in Table 1, a significant decrease in N2 uptake capacity, BET surface area, and pore volume was observed for UiO-67, UiO-68, and MOF-801 compared to the prototype UiO-66 [27,28]. To further compare the BET surface area and characteristics of porosity of materials synthesized using organic ligands with different chain lengths, their specific data are listed in Table 1. The overall trend in BET area and porosity features was consistent with that of the N2 adsorption amount of the synthesized UiO-66 analogues. From the data in Table 1, the conclusion could be drawn more intuitively that the pores of materials were mainly microporous except for UiO-68 (Figure 2d). It was worth noting that the BET surface area and pore volume of UiO-66 were up to 1173 cm2·g−1 and 0.54 cm3·g−1, respectively, which were much larger compared to other synthesized ones. The differences in BET area and porosity features of the synthesized UiO-66 analogues might be attributed to the number of missing-node defects and missing linker defects [14], which were tuned by the chain lengths of the organic ligand. Furthermore, the TGA profile was used to explore the thermal stability of the four materials, as depicted in Figure 2d. The typical behavior of the weight change upon heat exposure in TGA is represented by the sudden changes in the slope of the curve. Mainly three stages of weight loss were detected for UiO-66; the first period of loss below 150 °C corresponds to desolvation and dehydration of the structure [29]. Moreover, the most significant peak above 400 °C is associated with the overall loss of structural integrity of the MOF itself [30,31,32]. The loss between 400 °C and 500 °C can be used to evaluate the linker deficiency, all of which for the synthesized Zr-MOFs were less than the theoretical 6 of UiO-66, in line with the known linker deficiency even in well-crystallized materials [33].
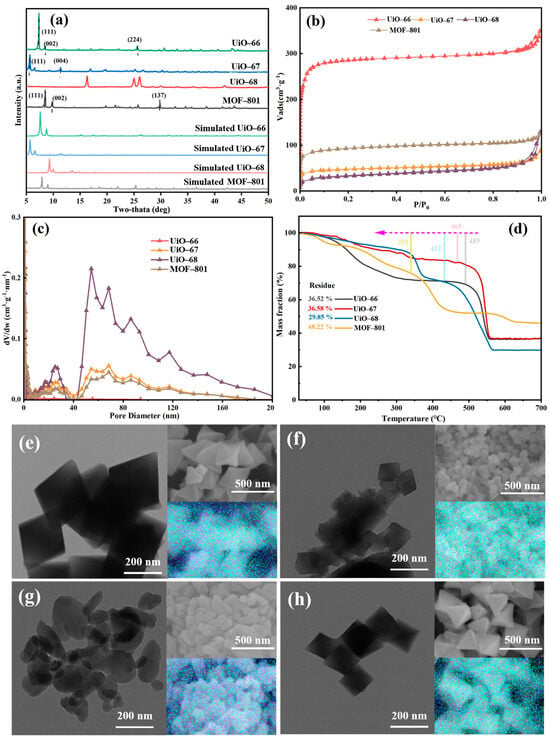
Figure 2.
(a) XRD patterns, (b) N2 adsorption–desorption isotherm, (c) pore diameter distribution, (d) TGA profile, TEM and SEM images with corresponding composite overlay mappings for (e) UiO-66, (f) UiO-67, (g) UiO-68, and (h) MOF-801.

Table 1.
Texture properties of Zr-MOFs with different size of organic linkers.
Table 1.
Texture properties of Zr-MOFs with different size of organic linkers.
| Catalyst | Specific Surface Area (m2·g−1) a | Pore Volume (cm3·g−1) a | Micropore Volume (cm3·g−1) a | Zr Content (wt %) b | Zr Content (wt %) c | Zr Content (wt %) d | Estimated Missing Linkers (per Zr6 Cluster) e |
|---|---|---|---|---|---|---|---|
| UiO-66 | 1173.26 | 0.54 | 0.52 | 26.25 | 17.61 | 13.91 | 1.52 |
| UiO-67 | 148.2 | 0.13 | 0.06 | 20.76 | 27.57 | 37.34 | 1.88 |
| UiO-68 | 108.16 | 0.20 | 0.02 | 11.97 | 24.49 | 9.11 | 1.91 |
| MOF-801 | 481.80 | 0.19 | 0.12 | 31.68 | 19.65 | 26.42 | 2.04 |
a Determined by N2-adsorption–desorption isotherms; b Estimated from XPS data; c Determined by ICP-MS; d Determined by EDX; e Calculated using the method from Shearer [26].
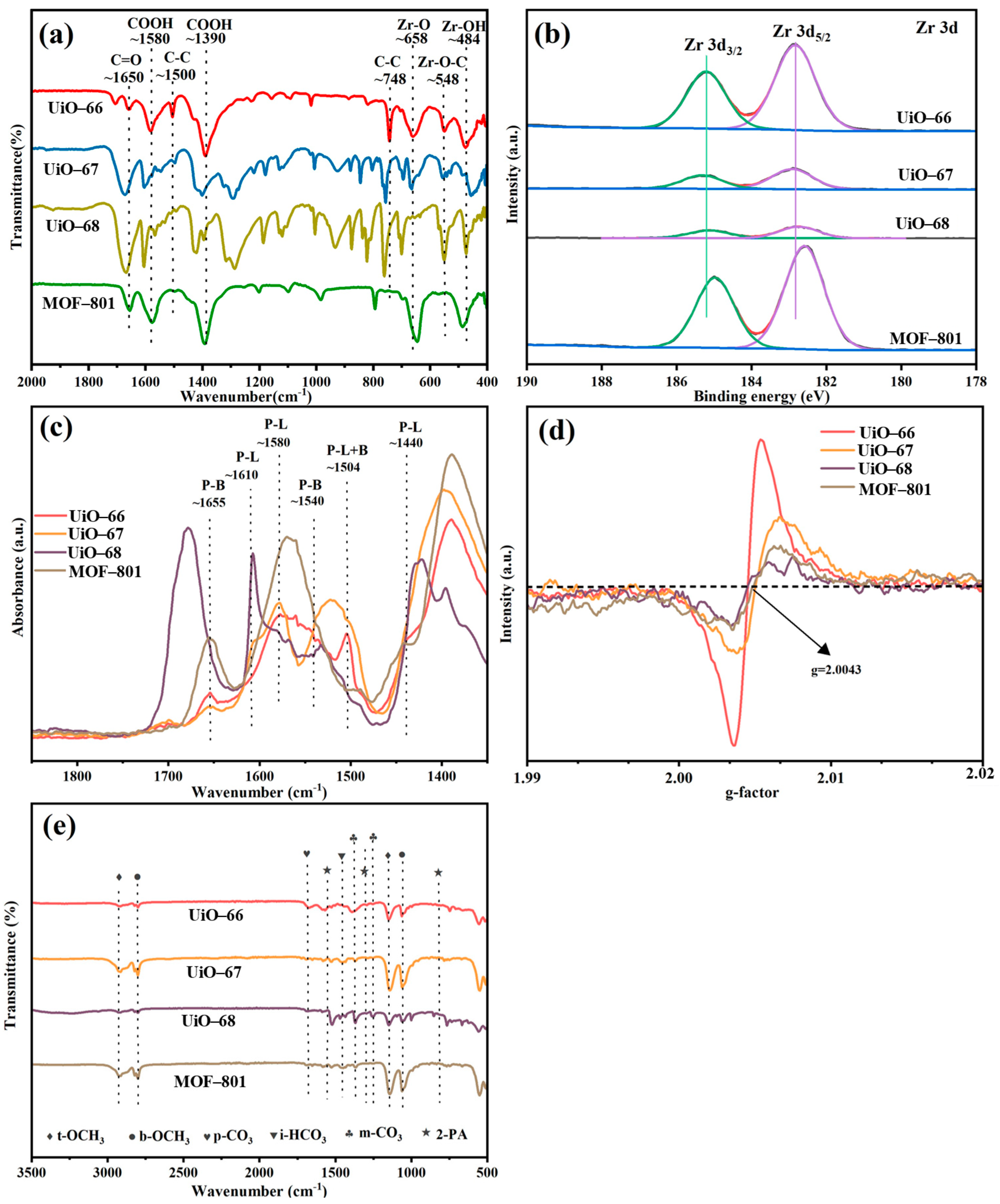
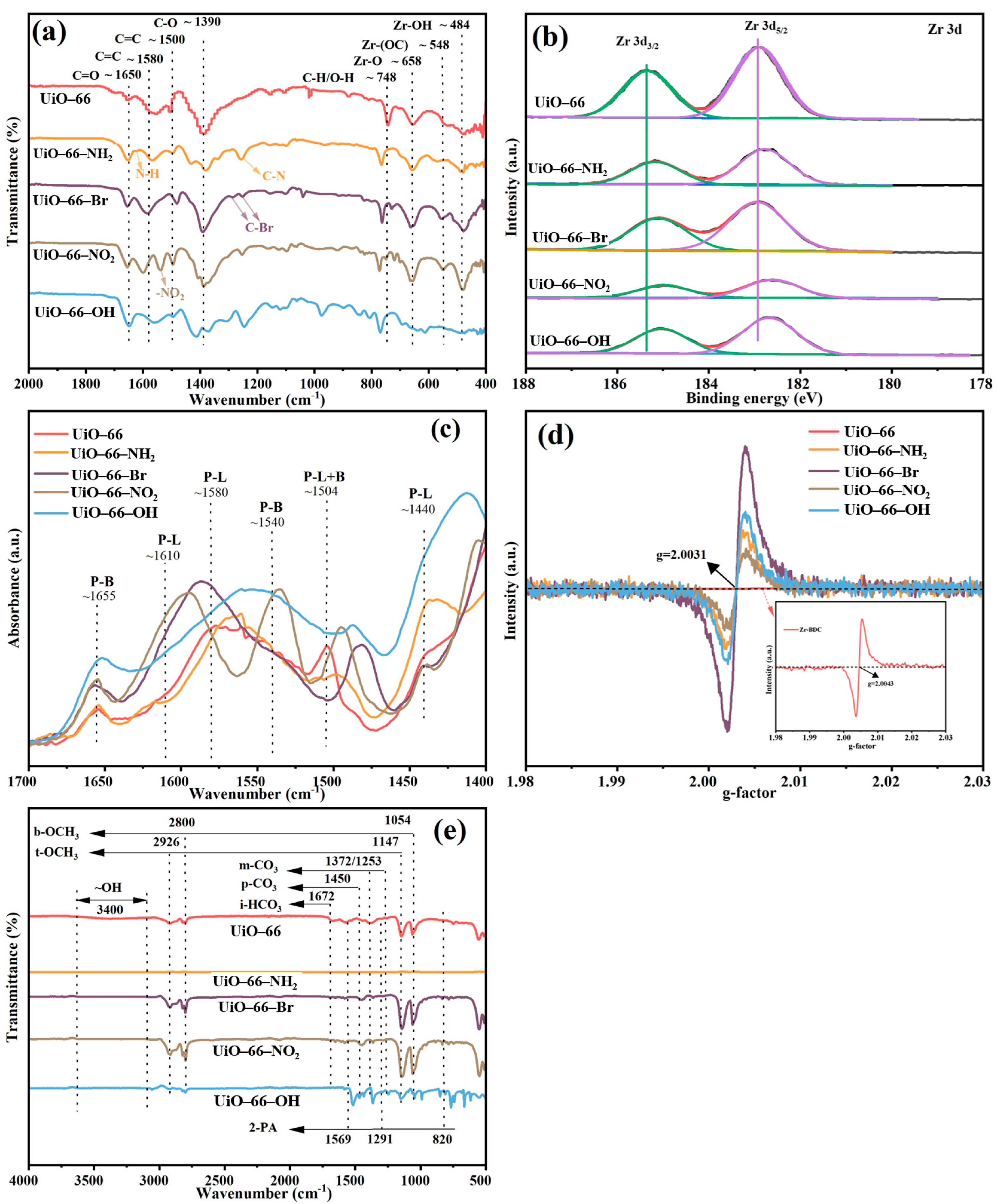
The functional groups of three MOFs were observed by means of the FT-IR spectroscopy [Figure 3a]. In the FT-IR spectra of UiO-66, peaks around 1580 and 1500 cm−1 are associated with C=C bond in the benzene rings of organic linkers [34,35]. The peak around 658 and 484 cm−1 can be attributed to the stretching vibration of Zr–Oμ3-O and Zr–Oμ3-OH from the Zr cluster, which was reported to be Lewis basic sites. Additionally, two characterized peaks at 1650 and 1390 cm−1 corresponded to the antisymmetric and symmetric stretching vibration of the -COO- group [34,36,37]. The peaks above can also be found at similar positions in the spectra of all the Zr-MOFs, indicating the analogous composition of all the MOFs. The slight peak position shift may be caused by the steric effect of different carbon chain structure as well as the different electronic effect of different carbon chains. In addition, XPS analysis revealed the electronic state modification of Zr6 clusters by the modification of chain lengths of organic ligands in UiO-66 analogues. As shown in Figure 3b, the changing of organic ligand chain into two benzene rings results in a slight oxidation of the constituting Zr6 clusters relative to that of UiO-66, whereas the introduction of three benzene rings and C=C provide a relative reduction of the Zr6 clusters. Considering that the adsorbed methanol molecules are diffusive and interact with Zr6 clusters as well as the organic ligands, the relatively electron-rich Zr6 clusters of MOF-801 may contribute to the increase in the electron density of methanol in the pores. XPS also revealed drastic Zr composition redistribution upon ligand chain modification, as summarized in Table 1, the amount of which was also detected by ICP-MS. Notably, modifying the chain length of the organic ligands of Zr-MOFs significantly altered the metal mass fraction of Zr, indicating a substantial change in active sites. The surface Zr amount of the four synthesized MOFs is in the following order: UiO-68 < UiO-67 < UiO-66 < MOF-801. In UiO-66 analogues, Zr metal clusters can generate various active sites, including terminal hydroxyl Zr-OH2 and Zr-OH, Lewis acid sites Zr4+, and Lewis basic sites Zr-O- and Zr-O-Zr [16].
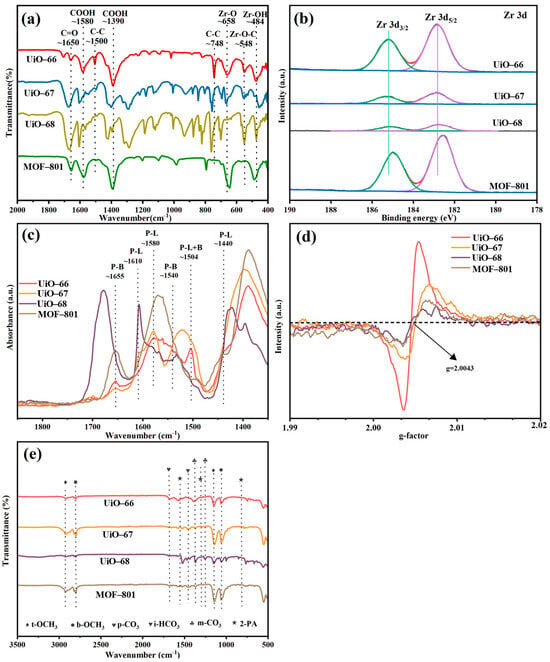
Figure 3.
(a) FT-IR spectra, (b) the XPS spectra of Zr 3d, (c) Py-IR results, (d) EPR diagram of UiO-66 analogues, (e) FT-IR of UiO-66 analogues after reaction.
2.1.2. Performance Evaluation of UiO-66 Analogues
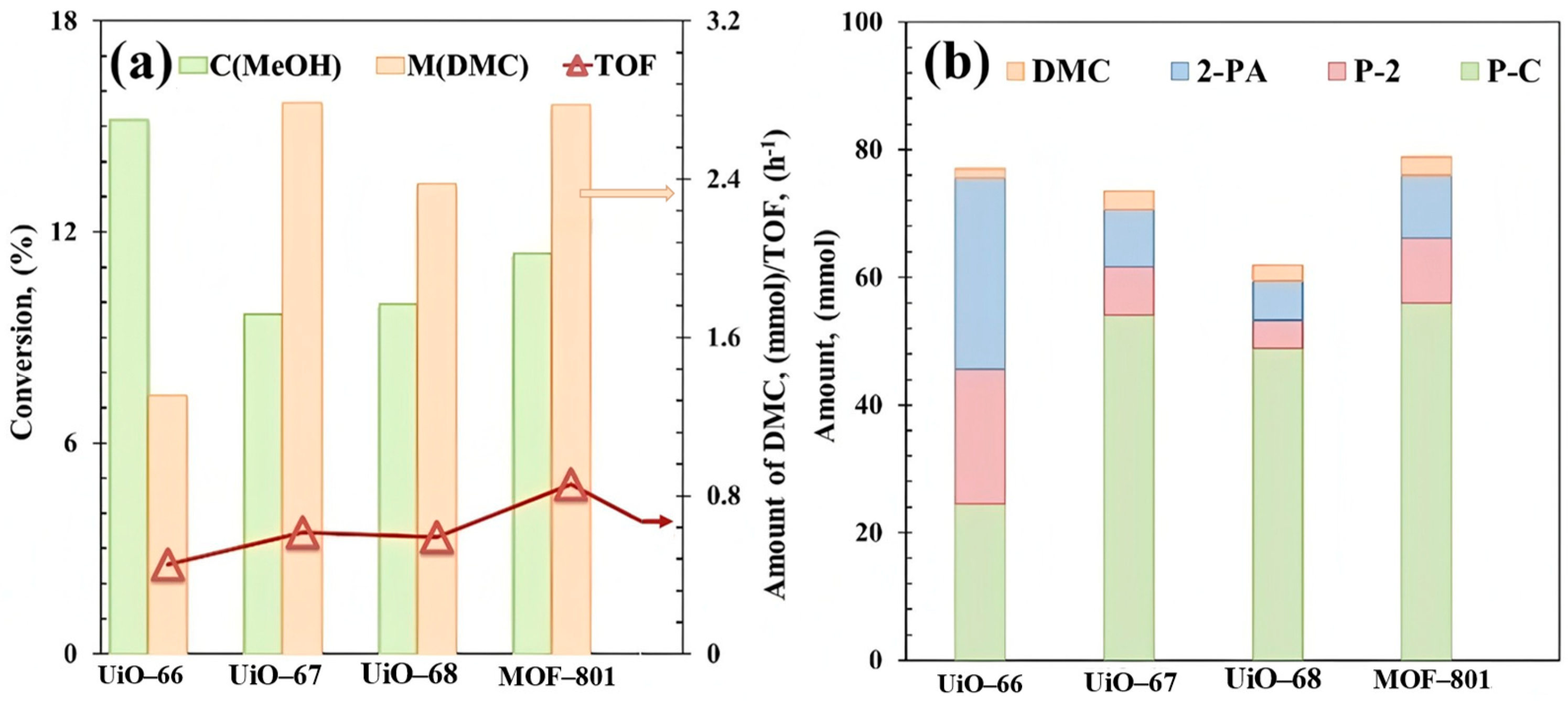
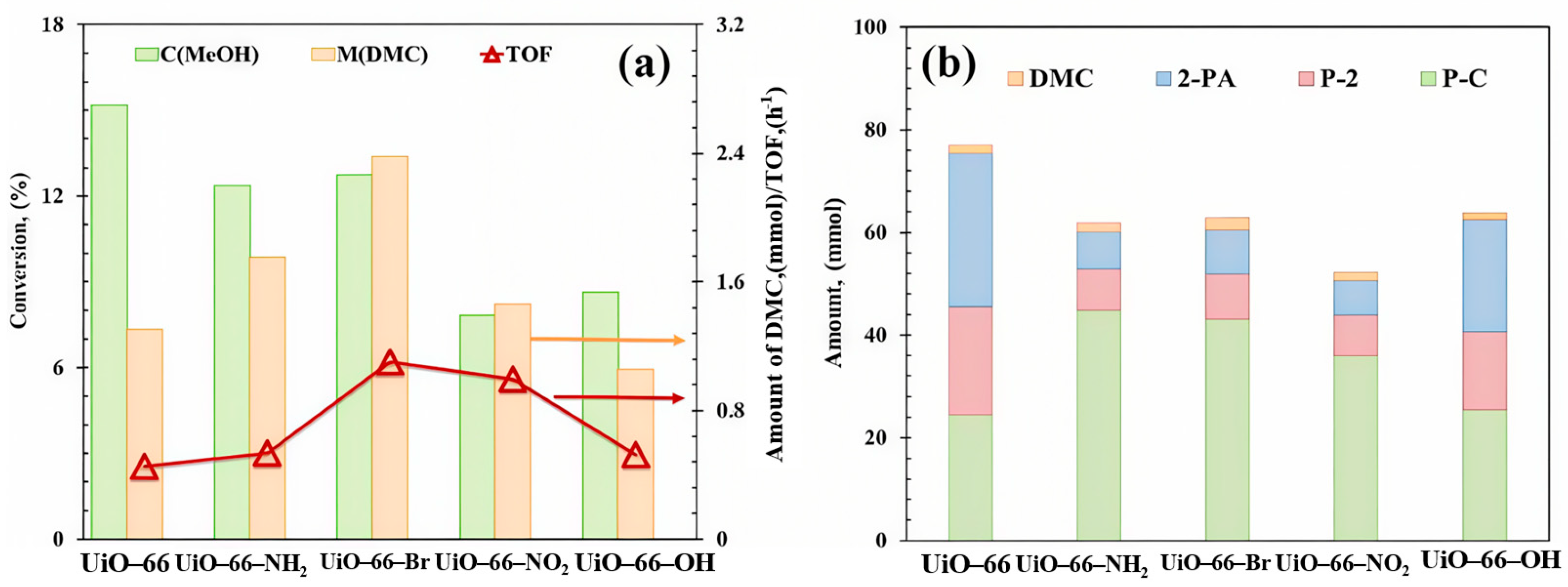
To investigate the performance of UiO-66 analogues for DMC direct synthesis from CO2, we performed the initial tests in a batch reactor using 2-cyanopyridine (2-CY) as dehydrating agent to react with co-produced H2O to form 2-picolinamide (2-PA), which is a thermodynamic driving force to push the equilibrium towards the formation of DMC [7]. Figure 4 shows the catalytic activity as well as amounts of DMC and the byproducts generated during the reaction over Zr-MOFs prepared from organic ligands with different chain lengths. A highest TOF of 0.86 h−1 was observed over MOF-801 catalyst, which has the smallest organic linker size. The activity of the MOFs is in the following order: UiO-66 < UiO-67 ≈ UiO-68 < MOF-801. Three main by-products were observed, 2-PA from the direct hydration of 2-CY, methyl picolinate (P-2) from an esterification reaction between 2-PA and methanol, and propylene carbonate (P-C) generated through reactions between 2-CY and methanol (Figure S6).
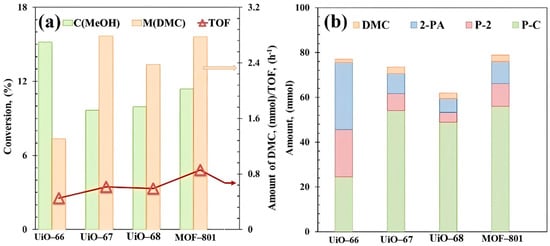
Figure 4.
(a) Catalytic evaluation and (b) product distribution of UiO-66 analogues. Reaction conditions: 0.5 mol MeOH, 0.25 mol 2-cyanopyridine, T = 150 °C, P = 2.6 MPa, t = 6 h. By-products: 2-picolinamide (2-PA), methyl picolinate (P-2), propylene carbonate (P-C).
The significant varied distribution of by-products over the series of Zr-MOFs with organic linkers of varying chain lengths (UiO-66, UiO-67, UiO-68, MOF-801) suggests an influence of ligand dimensions on reaction pathway selectivity. As shown in Figure 4b, MOF-801 exhibited a notably high yield of P-C (55.76 mmol) with highest TOF value (0.86 h−1, Figure 4a) compared to the other catalysts, which can be attributed to the highest estimated number of missing linkers of MOF-801 (2.04 per Zr6 cluster, Table 1). The missing linkers could contribute to enhanced mass transfer, potentially facilitating both reactant access and product desorption. Furthermore, the distinct micro-environment defined by its shortest ligand might preferentially favor the reaction pathway between 2-CY and methanol, leading to P-C. In contrast, UiO-66, despite its high methanol conversion, showed a more balanced product distribution. The combined yield of 2-PA and P-2 was the highest in this series (51.05 mmol, Figure 4b), which may be facilitated by its largest BET surface area (1173 m2/g) and pore volume (0.54 cm3/g, Table 1), providing ample space for the sequential hydration and esterification reactions involving 2-CY. The lower yields of all products observed for UiO-67 and UiO-68 could be related to their reduced surface areas and defect concentrations (Table 1), potentially limiting overall activity.
To gain further insight into the catalytic behavior of MOF-801, Py-IR, EPR and FT-IR of the post-reaction samples were conducted, as shown in Figure 3. As previously reported, the Lewis acid sites (LASs) and Lewis basic sites (LBSs) could promote the adsorption and activation of methanol [16], forming intermediates b-OCH3 and t-OCH3 [as evidenced by the IR spectra in Figure 3e], which could react further with CO2 to generate m-CH3OCOO−. And finally, DMC can be formed through the reaction of this CH3-OCOO− intermediate with another activated methanol [38]. We proposed that three key factors contributed to the enhanced activity of MOF-801: firstly, higher abundance of LAS and LBS in MOF-801, confirmed by the Py-IR results in Figure 3c (LAS, P-L, at 1580 cm−1, LBS, P-B, at 1655 cm−1 were prominent compared with other materials) [39], and secondly, the electron-rich Zr6 clusters of MOF-801, which increase the electron density of adsorbed methanol in the pores, promoting its dissociation into -CH3O and thus enhancing the reaction activity [24], and bulk defects in materials (benefits the mass transfer). Oxygen vacancy concentration was reported to reflect coordinatively unsaturated Zr atoms. EPR spectra [Figure 3d] revealed a symmetrical signal at g = 2.004, assigned to oxygen vacancies, with UiO-66 showing the strongest signal [40]. However, TGA-derived bulk defect concentrations (Table 1) indicated that MOF-801 had the highest defect density per Zr6 cluster due to missing linkers, which are beneficial for the mass transfer of the reactants and products. Therefore, it is speculated that the mass transfer may be more important than the improved electron density of UiO-66 analogues during the CO2-DMC process, which has also been strongly corroborated by prior research on defect engineering in UiO-66 frameworks [41].
2.2. Effects of Functional Groups of Organic Ligands on the MOFs
2.2.1. Structure and Morphology Analysis
XRD patterns of the four MOF materials synthesized with organic ligand with varied carbon chain match well with the UiO-66 except for UiO-66-OH, indicating the existence of functional groups does not alter the crystalline structure, as shown in Figure 5a [42,43]. The wide characteristic peaks of UiO-66-OH suggested poor crystallinity of the material. TEM and SEM images in Figure 5 reveal pronounced morphological variations for the one synthesized with organic ligand with -Br, while the other three materials preserve similar shapes. As the ligands are modified by different functional groups, the morphology of Zr-based MOF changes from about 200 nm cube to 80–150 nm cube or hexahedron. The nitrogen sorption isotherms obtained on all four UiO-66 analogues are shown in Figure 5b. The adsorption isotherms of all these catalysts exhibited type I behavior, suggesting formation of microporous material [26]. As can be seen, the nitrogen uptake capacity (and thus porosity) of the samples varies enormously depending on the functional groups of the organic ligand used in the MOF synthesis. Of even greater interest is the observation of two remarkable trends. (1) Samples synthesized with electron-withdrawing ligands (-NO2, -Br) exhibit significantly higher nitrogen uptake (and thus porosity) compared to those with electron-donating groups (-NH2, -OH). (2) Samples synthesized with functionalized ligands show markedly lower nitrogen uptake (and porosity) than the non-functionalized counterpart. The exceptionally low porosity of UiO-66-OH (BET surface area of only 2.96 m2·g−1) aligns with its poor crystallinity and can be attributed to the absence of modulators during the synthesis process. For instance, Katz et al. successfully synthesized UiO-66-OH with a high BET surface area of ~1000 m2·g−1 by employing concentrated hydrochloric acid as a modulator [29]. Consequently, the low surface area observed herein is characteristic of UiO-66-OH synthesized via the standard modulator-free route, which is consistent with its negligible catalytic activity. Quantitative data was extracted from the isotherms by calculating the BET surface areas of the samples (Table 2). As can be seen, the BET surface areas of the samples vary significantly, ranging from 1159 m2 g−1 (UiO-66) to 2.96 m2 g−1 (UiO-66-OH). More importantly, the trends in porosity (now quantitatively described by the BET surface area) are the same as those qualitatively observed in Figure 2. Moreover, the weight loss of all four materials was similar, as shown in the TGA profile in Figure 5d, indicating the same final solid residue. However, the presence of functional groups caused significant changes to the number of defects, as shown in Table 2. The largest amount of defects was observed for UiO-66-Br; this significant change can be ascribed to the electron transfer between the Zr6 cluster and the functional groups.
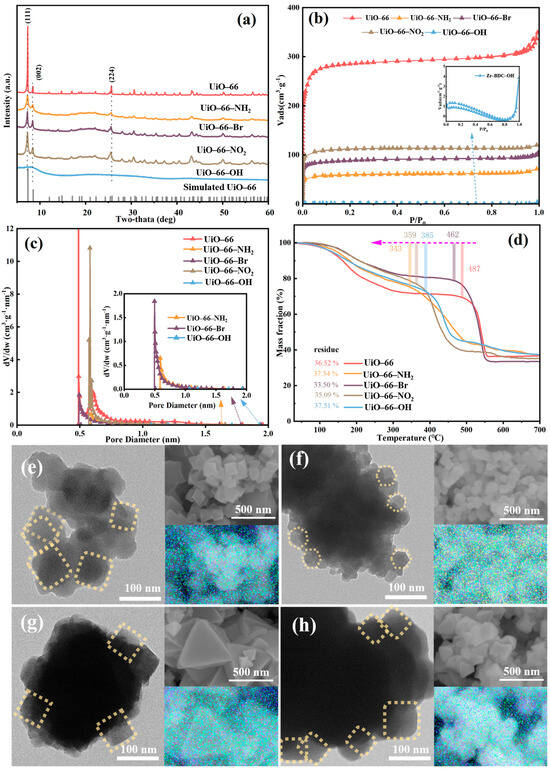
Figure 5.
(a) XRD patterns of Zr-1B2C-X materials, (b) N2 adsorption–desorption isotherm, (c) pore diameter distribution, (d) TGA graph, TEM and SEM images with corresponding composite overlay mappings for (e) UiO-66-NH2, (f) UiO-66-Br, (g) UiO-66-NO2, and (h) UiO-66-OH.

Table 2.
Texture properties of Zr-MOFs with different functional groups.
Similar to the case in the investigation of organic ligand carbon chain effect, the chemical structure of all four MOF materials with organic linkers appended with different functional groups was also characterized by FT-IR, as shown in Figure 6a. The characterized peaks corresponding to C=C bond, C-H and O-H vibrations in the benzene ring, and the stretching vibration of Zr–Oμ3-O and Zr–Oμ3-OH from the Zr cluster were detected in all four materials with slight variation [35,44,45,46]. However, the intensity of these characteristic peaks of UiO-66-OH is very weak in the FT-IR pattern, indicating that the structure of UiO-66-OH is difficult to form. Furthermore, characteristic peaks for functional groups were observed in corresponding MOF material, such as C-N tensile vibration from the aromatic amine appearing at 1256 cm−1, the bending vibration of N-H at 1622 cm−1 for UiO-66-NH2, the tensile vibration of C-Br at 1287 cm−1 and 1251 cm−1 over UiO-66-Br [44], and the asymmetric vibration characteristic peak of -NO2 at 1538 cm−1 for UiO-66-NO2 [47]. The appearance of functional group characteristic peaks once again proves the successful synthesis of UiO-66-X materials. In addition, XPS revealed the electronic state modification of Zr6 clusters by the functionalization of organic ligand in UiO-66 analogues. As shown in Figure 6b, functionalization with all four groups results in a slight reduction of the constituting Zr6 clusters relative to that of UiO-66, given the fact that functionalization with an electron-withdrawing group (the -Br group or -NO2 group) could decrease the electron density of the protons in the adsorbed methanol, compared with unfunctionalized UiO-66. On the other hand, introduction of an electron-donating group (the -NH2 group or -OH group) could increase the electron density of protons of methanol in the pores. Furthermore, characteristic peaks associated with functional groups can be observed for every functional group, as summarized in Figure S3 [48,49,50,51,52]. XPS also revealed drastic Zr composition redistribution upon ligand functionalization, as summarized in Table 2, the amount of which was also detected by ICP-MS. Notably, modifying the functional groups of the organic ligands of Zr-MOFs significantly altered the metal mass fraction of Zr, indicating substantial change in active sites. The surface Zr amount of the four synthesized MOFs is in the following order: UiO-66-NO2 < UiO-66-NH2 < UiO-66-OH < UiO-66 < UiO-66-Br.
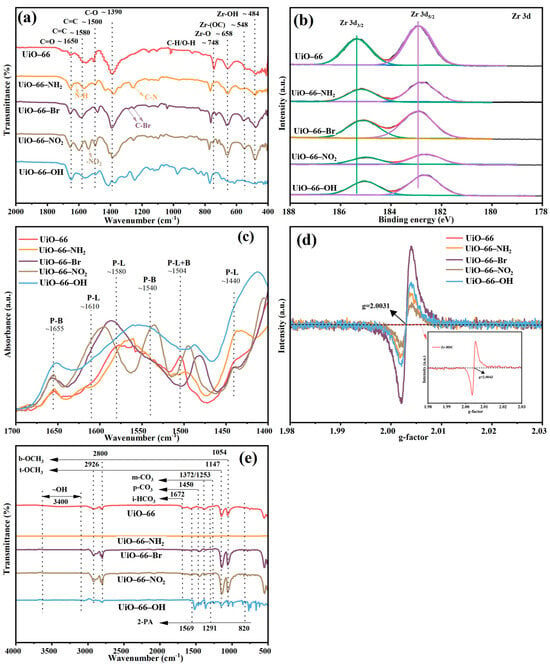
Figure 6.
(a) FT-IR spectra, (b) the XPS spectra of Zr 3d, (c) Py-IR results, (d) EPR diagram of UiO-66-X materials, (e) FT-IR of UiO-66-X materials after reaction.
2.2.2. Performance Evaluation of UiO-66-X
The activity of UiO-66-X catalyst was tested for the synthesis of DMC from CO2 conversion using 2-CY as the dehydrating agent; the methanol conversion and DMC formation amount are shown in Figure 7a. After appending functional groups, the activity of all UiO-66-X materials increased. The maximum DMC formation (2.38 mmol) was obtained over UiO-66-Br, generating from the strong interaction between the functional group with CO2 or methanol [53]. Furthermore, it is worth noting that after modifying functional groups on UiO-66, the side reactions involved in the reaction system also have significant changes, as shown in Figure 7b. UiO-66-Br showed the highest P-C yield (43.08 mmol) and the highest TOF (1.10 h−1). When functional groups were modified on the ligand, the activity of 2-CY reacting with H2O (blue) and 2-PA reacting with methanol (red) decreased significantly (the total production of 2-PA and P-2 decreased from 51.11 mmol to 14.71~37.14 mmol), while the activity of 2-CY in reaction with methanol increased (green), indicating varied catalytic requirements of these side reactions [54].
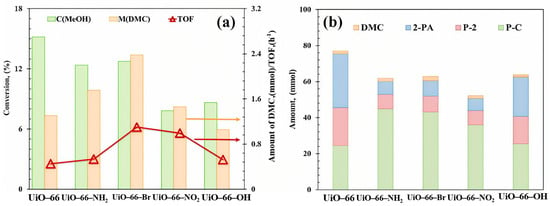
Figure 7.
(a) Catalytic evaluation and (b) product distribution of UiO-66-X materials. Reaction conditions: 0.5 mol MeOH, 0.25 mol 2-cyanopyridine, T = 150 °C, P = 2.6 MPa, t = 6 h. By-products: 2-picolinamide (2-PA), methyl picolinate (P-2), propylene carbonate (P-C).
Similar to the observation in Section 2.1.2, three factors were believed to contribute to the superior performance of UiO-66-Br, as shown in Table 2 and Figure 6. The Py-IR analysis in Figure 6c revealed that UiO-66-Br exhibited the strongest absorption band intensity belonging to LAS at 1580 cm−1 and LBS at 1655 cm−1 [55], indicating a higher concentration of active sites. EPR signals [Figure 6d] confirmed the highest OV content in UiO-66-Br, while TGA profile (Table 2) suggested it also had the most bulk defects, both of which are likely to enhance the mass transfer. Although the -Br group may reduce the electron density of adsorbed methanol (hindering its dissociation into reactive intermediate -OCH3), the TOF of UiO-66-Br remained the highest. The above results imply that the mass transfer, which is likely facilitated by the abundant bulk defects (as evidenced by the higher missing linker density in MOF-801 and UiO-66-Br), may play a more crucial role than the electronic effects in determining the overall catalytic activity. The defect-enhanced mass transfer predominates over electronic effects, which has also been elaborated previously regarding metal oxide catalysts [41,56].
3. Materials and Methods
3.1. Materials
Zirconium chloride (ZrCl4, 98%), terephthalic acid (1B-2C, 99%), biphenyl-4,4′-dicarboxylic acid (2B-2C, 97%), [p-Terphenyl]-4,4″-dicarboxylic acid (3B-2C, 98%), fumaric acid (1C=C-2C, 99%), cis, 2-aminoterephthalic acid (1B2C-NH2, 98%), 2-bromoterephthalic acid (1B2C-Br, 97%), nitroterephthalic acid (1B2C-NO2, 98%), 2-hydroxyterephthalic acid (1B2C-OH, 98%), N, N-dimethylformamide (DMF, 98%), methanol anhydrous (99.0%), mesitylene (99%), 2-cyanopyridine (2-CY, 99%) and dimethyl carbonate (DMC, 99%), were purchased from Aladdin Industrial Inc. (Shanghai, China). Carbon dioxide (99.9%) and N2 (99%) were purchased from Tianjin Vista Technology Co., Ltd. (Tianjin, China). All chemicals were used without further purification.
3.2. Catalyst Preparation
The Zr-based MOFs were prepared using the following method: 0.5 mmol ZrCl4 and 0.5 mmol terephthalic acid were fully dissolved in 100 mL of DMF, then the mixed solution was sonicated for 30 min to ensure thorough mixing. After this, the solution was transferred into a sealed Teflon-lined autoclave and heated at 120 °C for 24 h. After cooling to room temperature, the white precipitate was collected and washed with DMF and acetone to remove the uncompleted ions. UiO-66 was eventually obtained after drying at 70 °C for 5 h. UiO-67, UiO-68, MOF-801 and UiO-66-X (X = NH2, Br, NO2, OH) were prepared following the same procedure using other organic ligands of different structures as precursors (Table 3).

Table 3.
Synthesized MOFs and their corresponding organic ligands.
3.3. Catalyst Characterization
Wide angle XRD patterns of the samples were collected on an X-ray Diffraction Smart Lab using Cu Kα radiation (λ = 1.5406 Å), operated at 40 kV and 30 mA with the scanning range 5–60° at a scanning rate of 5° min−1. Transmission electron microscope (TEM) images of the catalyst samples were obtained on a HT770 instrument (HITACHI, Tokyo, Japan) at an electron acceleration voltage of 120 kV. Scanning electron microscope (SEM) images were captured on a SUPRA 55 field-emission microscope (Carl Zeiss AG, Oberkochen, Germany) operating at the beam energy of 10.0 kV. The sample powder was stacked on the conducting resin and sprayed with gold for 60 s to gain conductivity. The textural properties of the synthesized catalyst were determined by N2-adsorption-desorption isotherms using Autosorb iQ-Chemisorption and Physisorption Gas Sorption Analyzer (MicroActive for TriStar II Plus Version 1.01). The specific surface area was evaluated by the Brunauer–Emmett–Teller (BET) method and pore size distribution of the samples was determined by the Horvath–Kawazoe (HK) method.
Fourier transform infrared (FT-IR) spectra for the catalyst samples were recorded in the range of 500–4000 cm−1 on a FT-IR Nicolet iS10 spectrometer (Thermo Fisher Scientific Inc, Waltham, MA, USA). XPS spectra were recorded with X-ray photo electron spectrometer Escalab 250Xi apparatus with an Al-Kα X-ray source (hv = 1486.6 eV) and a monochromator (Thermo Fisher Scientific Inc., Waltham, MA, USA). The XPS measurement was carried out in the electron binding energy ranges corresponding to carbon 1s, oxygen 1S, zirconium 3d and bromine 3d core excitations.
Acid–base properties of the samples were studied by temperature-programmed desorption of ammonia (NH3-TPD) and temperature-programmed desorption of carbon dioxide (CO2-TPD) on a BELCAT-B instrument (MicrotracBEL Japan, Inc., Tokyo, Japan). The acidity of the samples was also detected using FT-IR with Pyridine as probe molecule. Furthermore, the acid properties were further probed by pyridine-adsorbed FT-IR (Py-IR) using a Nicolet iS10 spectrometer (OMNIC 8.2, Thermo Fisher Scientific Inc., Waltham, MA, USA). Prior to measurement, the catalyst was pretreated in pyridine vapor for 24 h, followed by vacuum dehydration at 60 °C for 4 h to remove physisorbed species. Spectra were collected to discriminate Brønsted and Lewis acid sites.
Oxygen vacancy (Ov) concentration was quantified by EPR (Bruker ELEXSYS E500, Bruker BioSpin AG, Fällanden, Switzerland) with the double integration of signals at g = 2.003 (delocalized oxygen vacancies).
3.4. Catalyst Activity Test
The direct synthesis of DMC from CO2 and CH3OH was carried out in a 100 mL stainless-steel autoclave equipped with provisions for temperature control and agitation speed control. A schematic of the experimental setup is shown in Figure S1. The standard experimental procedure was as follows. Methanol (MeOH, 0.5 mol, 32.0 g), 2-cyanopyridine (2-CY, dehydrating agent, 0.25 mol, 26.0 g), mesitylene (Mesi, internal standard, 0.02 mol, 2.7 g), and the catalyst (0.25 g) were charged into the reactor. The air in the sealed reactor was removed by purging the mixture with N2 gas three times. Subsequently, CO2 was introduced into the reactor to achieve an initial pressure of 2.6 MPa at room temperature. The reactor was then heated to the target temperature (150 °C) under low-speed stirring. Once the target temperature was reached, the stirring speed was rapidly increased to 900 rpm, which was defined as the reaction starting point (t = 0). After proceeding for a specified duration (6 h), the reactor was cooled to room temperature. Approximately 1–2 mL of the liquid mixture was then sampled for quantitative analysis.
All the liquid samples were analyzed using an Agilent gas chromatography (GC-7890A, Agilent Technologies, Inc., Santa Clara, CA, USA) equipped with a WAX capillary column and a flame ionization detector (FID). The analytical conditions are as follows: An injector temperature of 290 °C, FID detector temperature of 300 °C, and a temperature program initiating at 45 °C (hold for 4 min), ramping at 35 °C·min−1 to 250 °C (hold for 4.5 min).
MeOH conversion and the turn over frequency (i) are as follows:
4. Conclusions
This study presents a systematic investigation into the effects of organic ligand chain length and functional groups on the properties and activities of UiO-66 samples. Qualitative and quantitative analysis of multi-technique characterization data revealed that Lewis acid/basic sites (LAS/LBS), bulk defects (affect the mass transfer), and electron density effects are the three critical factors governing the catalytic activity of MOFs. Notably, it is suggested that mass transfer plays a more dominant role than electron density effect in modulating catalytic performance of MOFs.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/catal16010033/s1. Reference [57] is cited in Supplementary Materials. Figure S1: Schematic of experimental unit; Figure S2: XPS survey spectra, C 1s spectra, and O 1s spectra of the Zr-MOFs with different chain length of organic ligands; Figure S3: XPS survey spectra of the Zr-MOFs with different functional group; Figure S4: Effect of reaction temperature, CO2 pressure, molar ratio of methanol to 2-CY, and reaction time; Figure S5: Recycle experimental results including catalytic evaluation, products distribution and XRD pattern. Figure S6: The reactions involved in the synthesis of DMC from CO2 and CH3OH using 2-CY as a dehydrating agent. Figure S7: SEM and elemental mappings for (a) UiO-66, (b) UiO-67, (c) UiO-68, and (d) MOF-801. Figure S8: SEM and elemental mappings for (a) UiO-66-NH2, (b) UiO-66-Br, (c) UiO-66-NO2, and (d) UiO-66-OH. Table S1: Catalytic performance of the synthesized MOFs in the direct synthesis of DMC from CO2 and methanol. Table S2: Comparison of catalytic performance over MOF-801 and UiO-66-Br with that reported in the literature. Table S3: Elemental Composition of the Synthesized MOF Catalysts Determined by EDX Analysis.
Author Contributions
Conceptualization, Y.L. and Z.S.; methodology, Y.L. and Z.R.; validation, J.B. and K.S.; formal analysis, Z.R.; investigation, Y.L. and J.B.; resources, Z.S. and S.S.; data curation, Y.L. and K.S.; writing—original draft preparation, Y.L.; writing—review and editing, Z.S. and S.S.; visualization, Z.R.; supervision, Z.S.; project administration, Z.S.; funding acquisition, S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (22308298), Natural Science Foundation of Hebei Province (B2022203019), and Subsidy for Hebei Key Laboratory of Applied Chemistry after Operation Performance (22567616H).
Data Availability Statement
The data are available from the corresponding author upon reasonable request.
Conflicts of Interest
Author Shaotong Song was employed by the PetroChina Company Limited. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Santos, B.A.; Silva, V.M.; Loureiro, J.M.; Rodrigues, A.E. Review for the direct synthesis of dimethyl carbonate. ChemBioEng Rev. 2014, 1, 214–229. [Google Scholar] [CrossRef]
- Xue, S.; Xu, Z.; Tang, Y.; Ji, C. Polypiperazine-amide nanofiltration membrane modified by different functionalized multiwalled carbon nanotubes (MWCNTs). ACS Appl. Mater. Interfaces 2016, 8, 19135–19144. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A.; Pastore, C.; Pápai, I.; Schubert, G. Reaction mechanism of the direct carboxylation of methanol to dimethylcarbonate: Experimental and theoretical studies. Top. Catal. 2006, 40, 71–81. [Google Scholar] [CrossRef]
- Delledonne, D.; Rivetti, F.; Romano, U. Oxidative carbonylation of methanol to dimethyl carbonate (DMC): A new catalytic system. J. Organomet. Chem. 1995, 488, C15–C19. [Google Scholar] [CrossRef]
- Dahiya, S.; Srivastava, V.C.; Kumar, V. Dimethyl carbonate synthesis via transesterification of propylene carbonate using a titanium–praseodymium-based catalyst. Energy Fuels 2022, 36, 13148–13158. [Google Scholar] [CrossRef]
- Mandooie, M.; Rahimi, M.; Nikravesh, G.; Salehi, E. A comprehensive review on zinc-based mixed metal oxide catalysts for dimethyl carbonate synthesis via urea alcoholysis process. J. Ind. Eng. Chem. 2023, 117, 70–84. [Google Scholar] [CrossRef]
- Dong, L.; Zhu, S.; Yuan, Y.; Zhang, X.; Zhao, X.; Chen, Y.; Xu, L. Mechanism study on the influence of surface properties on the synthesis of dimethyl carbonate from CO2 and methanol over ceria catalysts. Chin. J. Catal. 2024, 65, 138–152. [Google Scholar] [CrossRef]
- Aresta, M.; Dibenedetto, A.; Angelini, A. Catalysis for the Valorization of Exhaust Carbon: From CO2 to Chemicals, Materials, and Fuels. Technological Use of CO2. Chem. Rev. 2014, 114, 1709–1742. [Google Scholar] [CrossRef]
- Bleken, F.L.; Barbera, K.; Bonino, F.; Olsbye, U.; Lillerud, K.P.; Bordiga, S.; Beato, P.; Janssens, T.; Svelle, S. Catalyst deactivation by coke formation in microporous and desilicated zeolite H-ZSM-5 during the conversion of methanol to hydrocarbons. J. Catal. 2013, 307, 62–73. [Google Scholar] [CrossRef]
- Xiao, Y.; Lei, B.; Jiang, H.; Xie, Y.; Du, J.; Xu, W.; Ma, D.; Zhong, M. Boosting dimethyl carbonate synthesis from CO2 and methanol through oxygen vacancy engineering on CeO2 under thermodynamically favorable conditions. J. Environ. Sci. 2025, 155, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Faria, D.J.; Moreira Dos Santos, L.; Bernard, F.L.; Pinto, I.S.; Chaban, V.V.; Romero, I.P.; Einloft, S. The direct synthesis of dimethyl carbonate out of carbon dioxide and methanol is catalyzed by D-metals supported on various matrices. J. Mol. Struct. 2023, 1292, 136110. [Google Scholar] [CrossRef]
- Hu, L.; Zhang, S.; Zhao, P.; Yang, J.; Wu, G.; Xu, W. La-doped CeO2 nanoflowers catalysts for direct synthesis of dimethyl carbonate from CO2 and methanol. J. Rare Earths 2025, 43, 2177–2185. [Google Scholar] [CrossRef]
- Yu, D.Y.; Wang, L.P.; Yang, T.Y.; Yang, G.P.; Wang, D.; Ni, H.G.; Wu, M.H. Tuning Lewis acidity of iron-based metal-organic frameworks for enhanced catalytic ozonation. Chem. Eng. J. 2021, 404. [Google Scholar] [CrossRef]
- Cui, W.; Zhang, G.; Hu, T.; Bu, X. Metal-organic framework-based heterogeneous catalysts for the conversion of C1 chemistry: CO, CO2 and CH4. Coord. Chem. Rev. 2019, 387, 79–120. [Google Scholar] [CrossRef]
- Han, B.; Chakraborty, A. Functionalization, protonation and ligand extension on MIL-53 (Al) MOFs to boost water adsorption and thermal energy storage for heat transformations. Chem. Eng. J. 2023, 472, 145137. [Google Scholar] [CrossRef]
- Xuan, K.; Pu, Y.; Li, F.; Li, A.; Luo, J.; Li, L.; Wang, F.; Zhao, N.; Xiao, F. Direct synthesis of dimethyl carbonate from CO2 and methanol over trifluoroacetic acid modulated UiO-66. J. CO2 Util. 2018, 27, 272–282. [Google Scholar] [CrossRef]
- Xu, W.; Xu, Z.; Yao, W.; Hu, L.; Ding, K.; Wu, G.; Xiao, G.; Gao, L. Directly synthesis of dimethyl carbonate from CO2 and methanol over UiO-66 @CeO2 Catalyst. Appl. Catal. A-Gen. 2023, 662, 119262. [Google Scholar] [CrossRef]
- Xuan, K.; Pu, Y.; Li, F.; Luo, J.; Zhao, N.; Xiao, F. Metal-organic frameworks MOF-808-X as highly efficient catalysts for direct synthesis of dimethyl carbonate from CO2 and methanol. Chin. J. Catal. 2019, 40, 553–566. [Google Scholar] [CrossRef]
- Huo, L.M.; Li, J.J.; Wang, L.; Zhang, S.Y.; Pu, Y.F.; Zhai, C.P.; Qiao, C.Z.; Zhou, S.S. Functionalized UiO-66 as Catalyst for Direct Synthesis of Dimethyl Carbonate from CO2 and Methanol. ChemistrySelect 2025, 10, e04044. [Google Scholar] [CrossRef]
- Zhang, Z.; Shen, Y.; Zhang, A.; Zhai, Y.; Wang, Z.; Jiang, K.; Wang, H. Preparation of imidazole ligand zirconium-based UiO-66 and its application in the direct synthesis of dimethyl carbonate from CO2 and methanol. J. CO2 Util. 2025, 98, 103148. [Google Scholar] [CrossRef]
- Li, C.L.; Shen, J.; Wu, K.B.; Yang, N.J. Metal Centers and Organic Ligands Determine Electrochemistry of Metal-Organic Frameworks. Small 2022, 18, 2106607. [Google Scholar] [CrossRef]
- Semivrazhskaya, O.O.; Salionov, D.; Clark, A.H.; Casati, N.; Nachtegaal, M.; Ranocchiari, M.; Bjelic, S.; Verel, R.; van Bokhoven, J.A.; Sushkevich, V.L. Deciphering the Mechanism of Crystallization of UiO-66 Metal-Organic Framework. Small 2023, 19, 2305771. [Google Scholar] [CrossRef]
- Skorynina, A.A.; Lazzarini, A.; Sannes, D.K.; Kozyr, E.G.; Ahoba-Sam, C.; Bordiga, S.; Olsbye, U.; Bugaev, A.L. The structure of Pd-functionalized UiO-67 during CO2 hydrogenation. J. Mater. Chem. C 2024, 12, 3564–3572. [Google Scholar] [CrossRef]
- Yan, Z.T.; Gong, Y.J.; Chen, B.H.; Wu, X.N.; Liu, Q.; Cui, L.L.; Xiong, S.S.; Peng, S.M. Methyl functionalized Zr-Fum MOF with enhanced Xenon adsorption and separation. Sep. Purif. Technol. 2020, 239, 116514. [Google Scholar] [CrossRef]
- Tan, C.X.; Han, X.; Li, Z.J.; Liu, Y.; Cui, Y. Controlled Exchange of Achiral Linkers with Chiral Linkers in Zr-Based UiO-68 Metal-Organic Framework. J. Am. Chem. Soc. 2018, 140, 16229–16236. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.C.; Chavan, S.; Bordiga, S.; Svelle, S.; Olsbye, U.; Lillerud, K.P. Defect Engineering: Tuning the Porosity and Composition of the Metal-Organic Framework UiO-66 via Modulated Synthesis. Chem. Mat. 2016, 28, 3749–3761. [Google Scholar] [CrossRef]
- Molavi, H.; Eskandari, A.; Shojaei, A.; Mousavi, S.A. Enhancing CO2/N2 adsorption selectivity via post-synthetic modification of NH2-UiO-66(Zr). Microporous Mesoporous Mat. 2018, 257, 193–201. [Google Scholar] [CrossRef]
- Shao, Z.; Wu, P.; Liu, F.; Zhang, Q.; Guo, C.; Liu, R.; Lu, J.; Ren, X.; Zheng, Y. Tailored SO2 capture in multivariate UiO-66(Zr) MOFs: Experimental insights and theoretical mechanisms. J. Environ. Chem. Eng. 2025, 13, 120101. [Google Scholar] [CrossRef]
- Motegi, H.; Yano, K.; Setoyama, N.; Matsuoka, Y.; Ohmura, T.; Usuki, A. A facile synthesis of UiO-66 systems and their hydrothermal stability. J. Porous Mater. 2017, 24, 1327–1333. [Google Scholar] [CrossRef]
- Helal, A.; Alahmari, F.; Usman, M.; Yamani, Z.H. Chalcopyrite UiO-67 metal-organic framework composite for CO2 fixation as cyclic carbonates. J. Environ. Chem. Eng. 2022, 10, 108061. [Google Scholar] [CrossRef]
- Wu, J.; Liang, D.; Song, X.; Liu, T.; Xu, T.; Wang, S.; Zou, Y. Sulfonic groups functionalized Zr-metal organic framework for highly catalytic transfer hydrogenation of furfural to furfuryl alcohol. J. Energy Chem. 2022, 71, 411–417. [Google Scholar] [CrossRef]
- Prabhu, S.M.; Chuaicham, C.; Park, C.M.; Jeon, B.; Sasaki, K. Synthesis and characterization of defective UiO-66 for efficient co-immobilization of arsenate and fluoride from single/binary solutions. Environ. Pollut. 2021, 278, 116841. [Google Scholar] [CrossRef]
- Kar, A.K.; Sarkar, R.; Manal, A.K.; Kumar, R.; Chakraborty, S.; Ahuja, R.; Srivastava, R. Unveiling and understanding the remarkable enhancement in the catalytic activity by the defect creation in UIO-66 during the catalytic transfer hydrodeoxygenation of vanillin with isopropanol. Appl. Catal. B-Environ. 2023, 325, 122385. [Google Scholar] [CrossRef]
- Chen, C.Q.; Chen, D.Z.; Xie, S.S.; Quan, H.Y.; Luo, X.B.; Guo, L. Adsorption Behaviors of Organic Micropollutants on Zirconium Metal-Organic Framework UiO-66: Analysis of Surface Interactions. ACS Appl. Mater. Interfaces 2017, 9, 41043–41054. [Google Scholar] [CrossRef]
- Wang, Y.L.; Zhang, S.; Zhao, Y.F.; Bedia, J.; Rodriguez, J.J.; Belver, C. UiO-66-based metal organic frameworks for the photodegradation of acetaminophen under simulated solar irradiation. J. Environ. Chem. Eng. 2021, 9, 106087. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, J.; Li, Y.S.; Zhuang, Q.X.; Gu, J.L. Simultaneous Degradation and Removal of Cr-VI from Aqueous Solution with Zr-Based Metal-Organic Frameworks Bearing Inherent Reductive Sites. Chem.-Eur. J. 2017, 23, 15415–15423. [Google Scholar] [CrossRef]
- Zou, C.; Vagin, S.; Kronast, A.; Rieger, B. Template mediated and solvent-free route to a variety of UiO-66 metal-organic frameworks. RSC Adv. 2016, 6, 102968–102971. [Google Scholar] [CrossRef]
- Bai, J.; Song, Z.; Liu, L.; Zhu, X.; Gao, F.; Chaudhari, R.V. Enhanced transformation of CO2 over microporous Ce-doped Zr metal–organic frameworks. RSC Adv. 2022, 12, 26307–26318. [Google Scholar] [CrossRef]
- Pyen, S.; Hong, E.; Shin, M.; Suh, Y.; Shin, C. Acidity of co-precipitated SiO2-ZrO2 mixed oxides in the acid-catalyzed dehydrations of iso-propanol and formic acid. Mol. Catal. 2018, 448, 71–77. [Google Scholar] [CrossRef]
- Chiang, C.; Lin, K.; Yu, S.; Lin, Y. Synthesis and characterization of H3PW12O40/Ce0.1Ti0.9O2 for dimethyl carbonate formation via Methanol carbonation. Int. J. Hydrog. Energy 2017, 42, 22108–22122. [Google Scholar] [CrossRef]
- Lu, D.; Cheng, Z.; Klomkliang, N.; Koo-amornpattana, W.; Verpoort, F.; Chaemchuen, S. Hydro-modulator induced defective structure and hieratical pores in UiO-66 for efficient adsorption and catalysis. Mater. Today Chem. 2024, 42, 102420. [Google Scholar] [CrossRef]
- Katz, M.J.; Brown, Z.J.; Colón, Y.J.; Siu, P.W.; Scheidt, K.A.; Snurr, R.Q.; Hupp, J.T.; Farha, O.K. A facile synthesis of UiO-66, UiO-67 and their derivatives. Chem. Commun. 2013, 49, 9449–9451. [Google Scholar] [CrossRef] [PubMed]
- Cavka, J.H.; Jakobsen, S.; Olsbye, U.; Guillou, N.; Lamberti, C.; Bordiga, S.; Lillerud, K.P. A New Zirconium Inorganic Building Brick Forming Metal Organic Frameworks with Exceptional Stability. J. Am. Chem. Soc. 2008, 130, 13850–13851. [Google Scholar] [CrossRef]
- Lammert, M.; Wharmby, M.T.; Smolders, S.; Bueken, B.; Lieb, A.; Lomachenko, K.A.; De Vos, D.; Stock, N. Cerium-based metal organic frameworks with UiO-66 architecture: Synthesis, properties and redox catalytic activity. Chem. Commun. 2015, 51, 12578–12581. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.H.; Zhang, Y.; Jiang, J.Y.; Yang, Y.B.; Liu, H. Modified UiO-66-Br Microphotocatalyst with High Electron Mobility Enhances Tetracycline Degradation. Langmuir 2023, 39, 3678–3691. [Google Scholar] [CrossRef]
- Yang, Y.Q.; Zhang, D.F.; Ji, W.Q.; Bi, F.K.; Song, L.; Zhang, X.D. Uniform platinum nanoparticles loaded on Universitetet i Oslo-66 (UiO-66): Active and stable catalysts for gas toluene combustion. J. Colloid Interface Sci. 2022, 606, 1811–1822. [Google Scholar] [CrossRef] [PubMed]
- Desai, N.C.; Dodiya, A.M. Conventional and microwave techniques for the synthesis and antimicrobial studies of novel 1-[2-(2-chloro-6-methyl(3-quinolyl))-5-(4-nitrophenyl)-( 1,3,4-oxadiazolin-3-yl)]-3-(aryl) prop-2-en-1-ones. Arab. J. Chem. 2016, 9, S379–S387. [Google Scholar] [CrossRef]
- Gowthaman, N.; Sinduja, B.; Karthikeyan, R.; Rubini, K.; John, S.A. Fabrication of nitrogen-doped carbon dots for screening the purine metabolic disorder in human fluids. Biosens. Bioelectron. 2017, 94, 30–38. [Google Scholar] [CrossRef]
- Wu, Y.F.; Xiao, Y.; Yuan, H.; Zhang, Z.Q.; Shi, S.B.; Wei, R.P.; Gao, L.J.; Xiao, G.M. Imidazolium ionic liquid functionalized UiO-66-NH2 as highly efficient catalysts for chemical fixation of CO2 into cyclic carbonates. Microporous Mesoporous Mat. 2021, 310, 110578. [Google Scholar] [CrossRef]
- Yang, W.X.; Wang, J.M.; Han, Y.; Luo, X.L.; Tang, W.Z.; Yue, T.L.; Li, Z.H. Robust MOF film of self-rearranged UiO-66-NO2 anchored on gelatin hydrogel via simple thermal-treatment for efficient Pb(II) removal in water and apple juice. Food Control 2021, 130, 108409. [Google Scholar] [CrossRef]
- Zeng, S.S.; Lyu, F.C.; Sun, L.G.; Zhan, Y.W.; Ma, F.X.; Lu, J.; Li, Y.Y. UiO-66-NO2 as an Oxygen "Pump" for Enhancing Oxygen Reduction Reaction Performance. Chem. Mat. 2019, 31, 1646–1654. [Google Scholar] [CrossRef]
- Zhao, S.J.; Huang, W.J.; Xie, J.K.; Liu, W.; Qu, Z.; Yan, N.Q. Mercury removal from flue gas using UiO-66-type metal-organic frameworks grafted with organic functionalities. Fuel 2021, 289, 119807. [Google Scholar] [CrossRef]
- Qu, Y.; Zhao, Y.F.; Li, D.Z.; Sun, J.M. Task-specific ionic liquids for carbon dioxide absorption and conversion into value-added products. Curr. Opin. Green Sustain. Chem. 2022, 34, 100599. [Google Scholar] [CrossRef]
- Giram, G.G.; Bokade, V.V.; Darbha, S. Direct synthesis of diethyl carbonate from ethanol and carbon dioxide over ceria catalysts. New J. Chem. 2018, 42, 17546–17552. [Google Scholar] [CrossRef]
- Fu, Z.W.; Zhong, Y.Y.; Yu, Y.H.; Long, L.Z.; Xiao, M.; Han, D.M.; Wang, S.J.; Meng, Y.Z. TiO2-Doped CeO2 Nanorod Catalyst for Direct Conversion of CO2 and CH3OH to Dimethyl Carbonate: Catalytic Performance and Kinetic Study. ACS Omega 2018, 3, 198–207. [Google Scholar] [CrossRef]
- Rojas-Buzo, S.; Salusso, D.; Jouve, A.; Bracciotti, E.; Signorile, M.; Bordiga, S. CO2 to dimethylcarbonate synthesis: Surface defects and oxygen vacancies engineering on MOF-derived CexZr1−xO2−y catalysts. Appl. Catal. B-Environ. 2024, 346, 123723. [Google Scholar] [CrossRef]
- Huo, L.; Wang, L.; Li, J.; Pu, Y.; Xuan, K.; Qiao, C.; Yang, H. Cerium doped Zr-based metal-organic framework as catalyst for direct synthesis of dimethyl carbonate from CO2 and methanol. J. CO2 Util. 2023, 68, 102352. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.