Abstract
We review crown ether-facilitated nucleophilic reactions using metal salts, presenting the studies using kinetic measurements and quantum chemical methods. We focus on the mechanistic features, specifically on the contact ion-pair (CIP) mechanism of metal salts for nucleophilic processes promoted by crown ethers and derivatives. Experimental verification of the CIP form of the metal salt CsF complexed with [18-Crown-6] by H-NMR spectroscopy is described. The use of chiral crown ethers and derivatives for enantioselective nucleophilic processes is also discussed.
1. Introduction
Since their discovery by Pedersen [1,2], crown ethers [3,4] have been used as prominent phase-transfer catalysts (PTCs) in various chemical reactions. This class of macrocyclic organocatalysts forms host–guest complexes with alkali metal ions and organic cations such as ammoniums and pyridiniums owing to the strongly electronegative O atoms in the ring. Crown ether-based host–guest complexes have been widely used for constructing ion sensors [5,6,7,8], artificial enzymes [9,10], and stimuli-responsive materials [11,12].
For organic reactions involving counter-cation–anion pairs, for example, in SN2 reactions, crown ether was presumed to form complexes with the counter-cation and “leave the anion bare,” thereby enhancing the reactivity of the anion. However, the detailed interactions between crown ethers and counter-cation/nucleophile pairs have not been fully examined. The most relevant question will be: Do crown ethers really “detach” the anionic nucleophile sufficiently from the counter-cation for the former to be considered “bare”?
A series of investigations on the mechanism of SN2 rate enhancement by crown ethers led researchers to theoretically propose that this conventional wisdom regarding the role of crown ethers should be revised in some circumstances. Observations of phenomenally efficient and selective SN2 reactions using metal salts activated by Lewis base promoters (bulky alcohols, oligo(ethylene glycols), ionic liquids) suggest that metal salts MX (M = K, Cs) may react as contact ion-pairs (CIPs) [13,14,15,16,17,18,19,20], in which the counter-cation and the nucleophile are in close contact (RMX < 3 Å) and that crown ethers do not really “separate” the ion pairs to large distances, but rather act as Lewis bases (through the O atoms) on the counter-cation in CIP to mitigate the latter’s harmful Coulombic influence on the nucleophile. In this novel mechanism, the counter-cation exists as a “neutralized spectator.” This new class of SN2 mechanism has not been paid a fair amount of attention except for some excellent theoretical studies [15,16,17,18,19,20], probably due to the lack of experimental techniques for directly (in situ) probing the CIPs in reaction systems in solution phase. It seems that the crown ether/metal salt system is the simplest and prototypical example for this purpose, as we propose in this review.
In this review, we describe recent developments in crown ether chemistry for SN2 technology and demonstrate the efficacy of phase-transfer catalysts. We show that, in some cases, the reactions proceed almost instantaneously at room temperature under the influence of crown ethers and derivatives. Crown ether derivatives are used for the treatment of enantioselective reactions. We specifically focus on the CIP form of the metal salt elucidated by H-NMR spectroscopy and the CIP mechanism for SN2 reactions facilitated by crown ethers. We also provide a brief review of recent work on the synergetic effects of crown ether/PTC combinations and on enantioselective SN2 reactions facilitated by chiral crown ether derivatives. Although our emphasis is on SN2 processes, we also examine other nucleophilic reactions, including SNAr and Michael addition reactions.
2. Earlier Works on Crown Ethers and Derivatives as PTCs
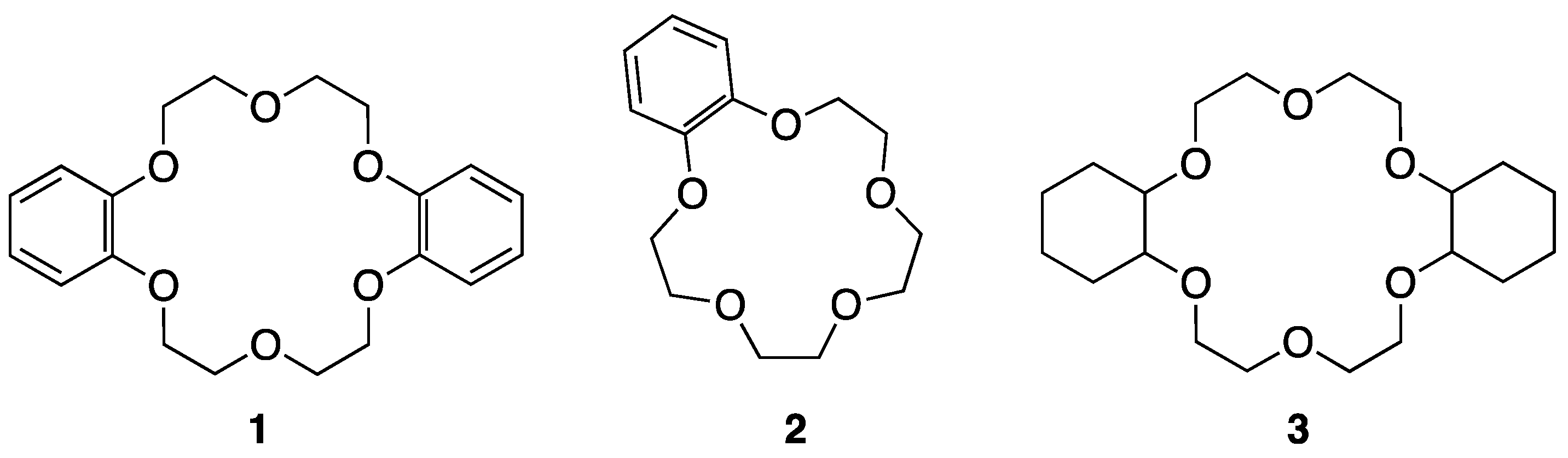
Crown ethers were first used as PTCs [21,22,23,24,25,26,27,28] after their discovery by Pedersen. Liotta and Harris in 1974 first used [18-crown-6] for the nucleophilic reactions of KF with organic substrates in aprotic organic solvents, such as acetonitrile or benzene [29]. Notably, the authors termed the anionic nucleophile interacting with the crown ether a “naked” anion, which became a conventional “standard” concept. Landini, Montanari, and coworkers [26,27] studied anion-promoted nucleophilic substitutions in aqueous–organic two-phase systems in the presence of catalytic amounts of crown ether derivatives (1–3 in Figure 1).
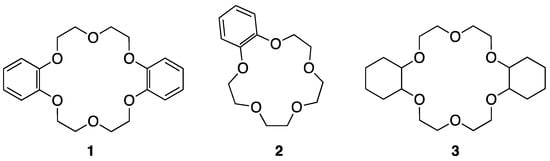
Figure 1.
Crown ether derivatives are synthesized and used for SN2 reactions. Ref. [26].
Aliphatic or aromatic side chains of these crown ethers formed stable complexes with metal cations, acting as PTCs, among which III showed the best performance (Table 1). They also observed that the removal of the water phase only led to a minimal variation in anion reactivity, balanced by a larger cation–anion interaction. However, complexed crown ethers can hardly be considered a source of “naked anions”, unlike cryptates. Thus, from the beginning, the naked nucleophile view has been suspected. Nevertheless, unambiguous evidence for the form (CIP vs. separated ion-pair (SIP)) of counter-cation–anion pairs under the influence of crown ethers in the solution phase has been lacking, and ambiguity has persisted for a long time, although some crown ether/metal salt complexes have been found to involve the CIP cation–anion pair in crystalline states [2,3]. However, this view of the separated ion-pair (SIP) mechanism for nucleophilic processes may not be correct, as illustrated below.

Table 1.
Dependence of catalytic activity on crown ethers in the conversion of n-C8H17Br into n-C8H17I under phase-transfer conditions. Ref. [26].
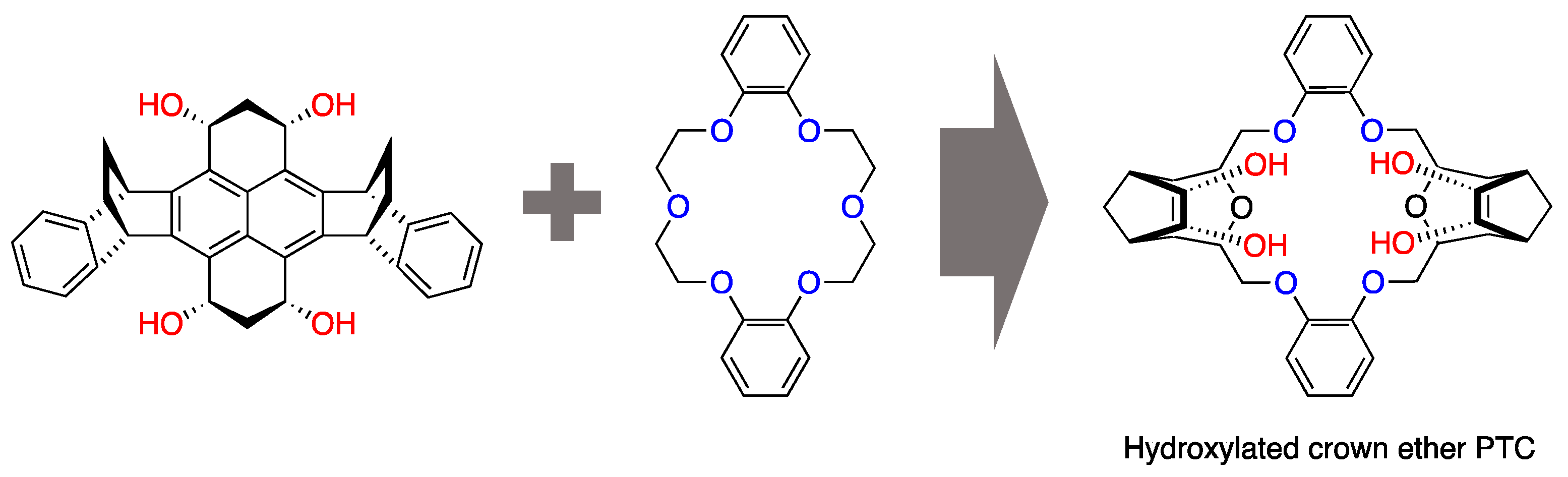
Structural modifications have also been attempted; for example, the attachment of –OHs was shown to give crown ethers the bifunctional capability of simultaneous anion solvation and cation complexation [30]. Pliego and coworkers’ design of “hydrocrowns” [31] (Figure 2) is the most recent example along this scheme line. The initial interest in crown ethers as PTCs subsided in the 1990s and was somewhat overshadowed by the development of other classes of PTCs, such as quaternary ammonium salts [20,32,33,34,35], organic phosphonium salts [36,37,38], polyethylene glycols [39], and ionic liquids [40,41,42]. However, the use of crown ether derivatives for asymmetric syntheses has revived them.
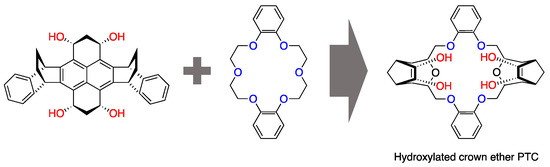
Figure 2.
Theoretical design of a crown ether scaffold for nucleophilic fluorination formed by merging two catalytic concepts. Ref. [31].
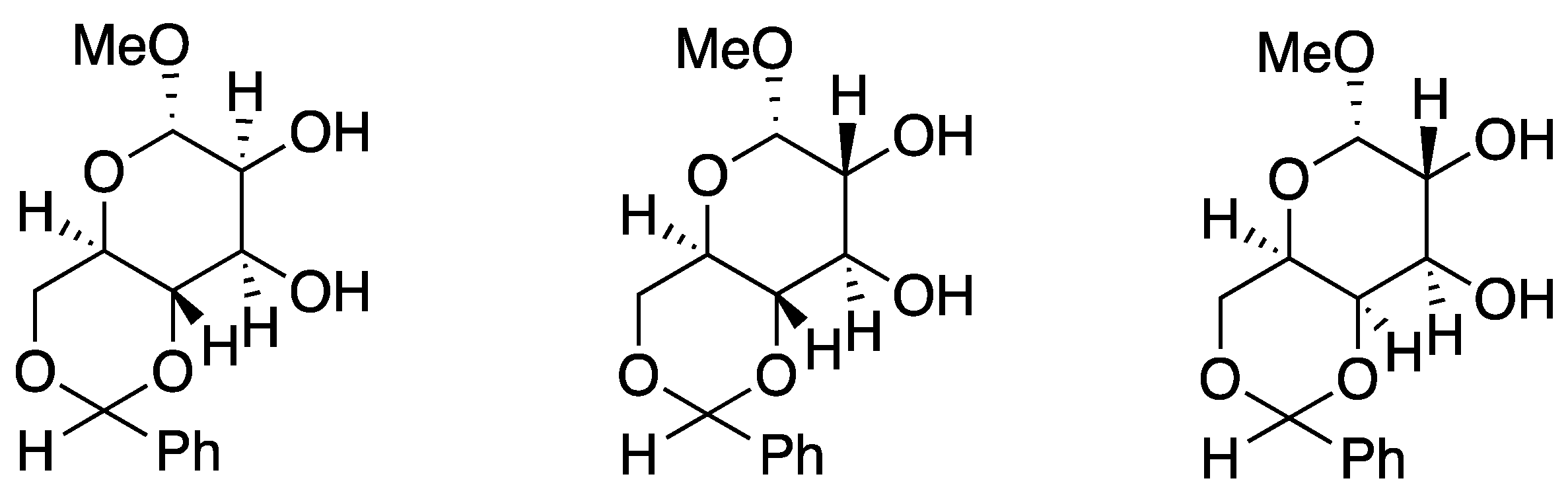
In 1984, Pietraszkiewicz and Jurczak [43] introduced carbohydrates as chiral side chains (Figure 3). Subsequently, a wide variety of chiral crown ethers have been designed and used for chiral recognition and asymmetric synthesis. Crown ether-derived peptides are one example [44,45], as discussed below.
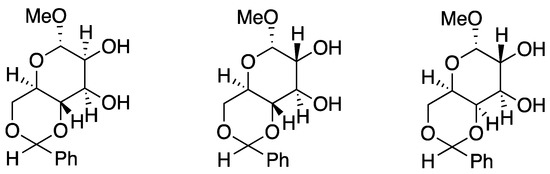
Figure 3.
Chiral diaza-crown ether incorporating a carbohydrate unit. Ref. [43].
3. Functionalized Crown Ethers
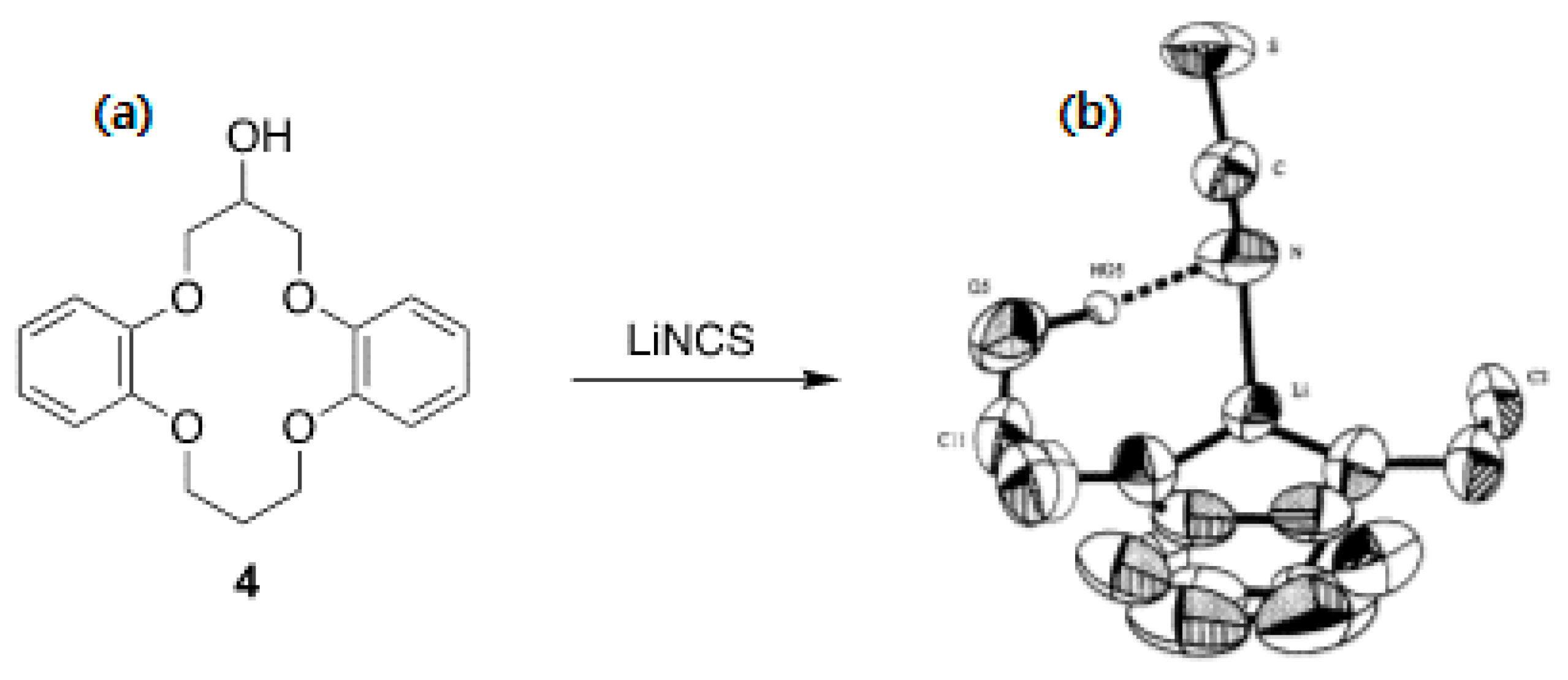
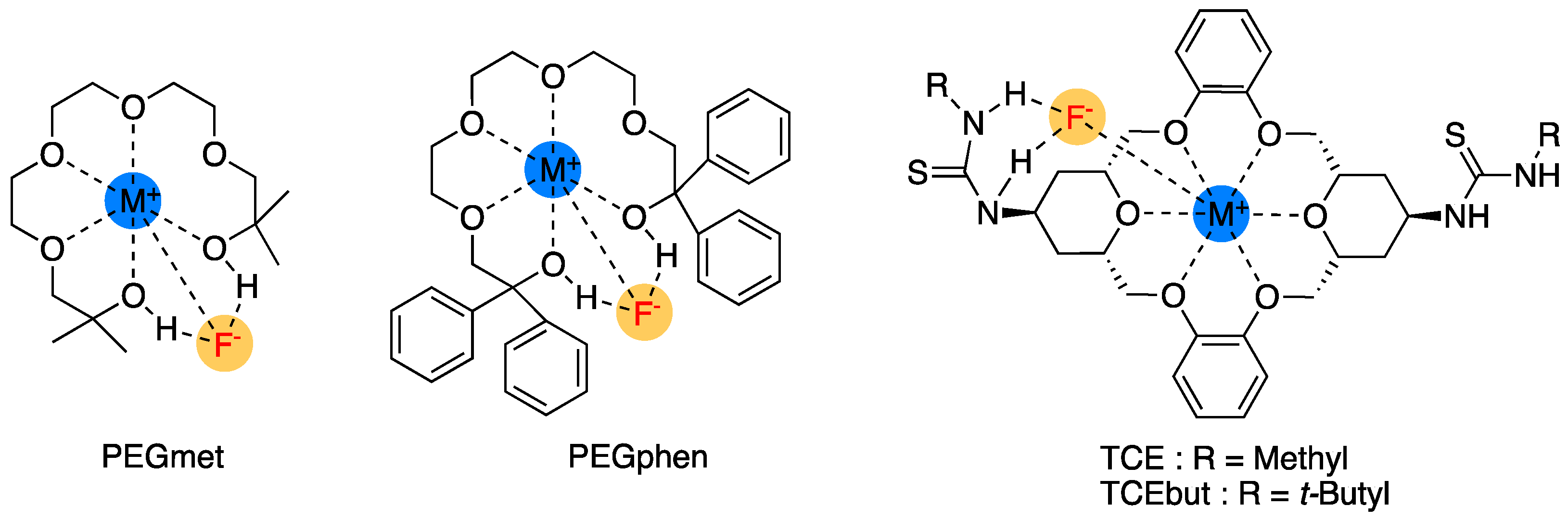
Another feature of crown ethers is the possibility of functionalizing them for specific purposes, such as ion sensing or chiral recognition. However, here, we only discuss the functionalization of crown ethers in relation to their capability as PTCs. The crown ether derivatives used for asymmetric synthesis are reviewed in Section 5. Olsher et al.’s structural modification of crown ethers [30] by attaching one or more pendant hydroxyl groups might be a typical early example. The authors found that functionalization allowed PTCs to act as bifunctional ligands for simultaneous anion solvation and cation complexation (Figure 4). They demonstrated the capability of the bifunctional crown ether by identifying the X-ray structure of its complex with LiNCS (Figure 4).
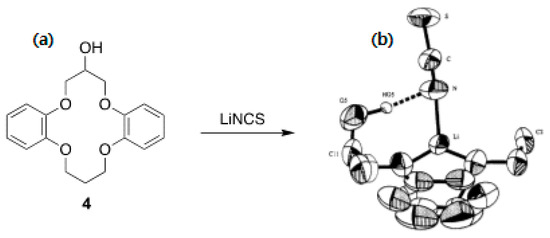
Figure 4.
(a) Crown ether derivatives with appended –OHs as bifunctional ligands and (b) crystal structure of 4•LiNCS complex. Ref. [30].
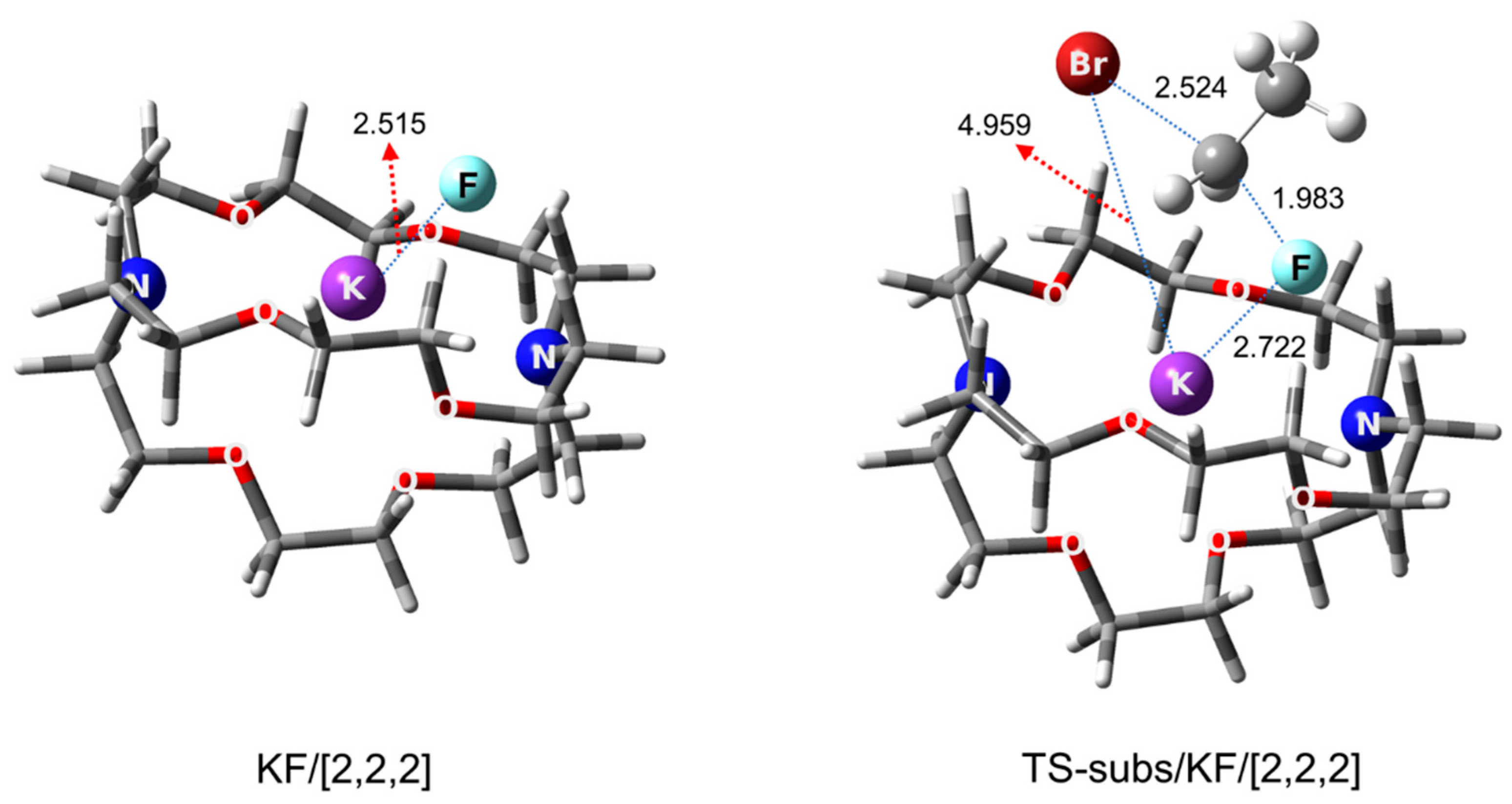
Cryptands as PTCs are very similar to crown ethers and derivatives, and they may safely be considered functionalized crown ethers. First discovered by Lehn in 1969 [46], this class of PTCs [47,48] soon found use as organocatalysts [49]. As for the mechanistic features of the best-known cryptand, [2.2.2], Pliego carried out a theoretical analysis in 2018 [50], confirming the most outstanding capability of the PTC, better than bulky alcohols or oligoEGs. KF was predicted to be in CIP form in the KF/[2.2.2] complex and in the TS for the SN2 fluorination of ethyl bromide by KF in the presence of [2.2.2] (Figure 5).
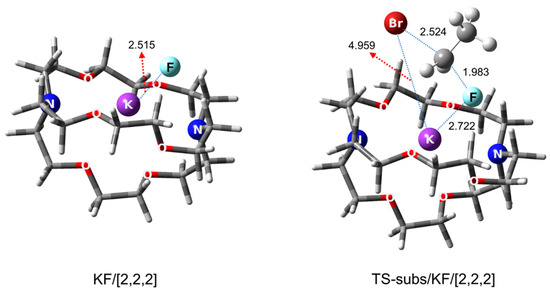
Figure 5.
Calculated structures of KF/[2.2.2] complex, and the TS for SN2 fluorination of ethyl bromide by KF in the presence of/[2.2.2]. Ref. [50].
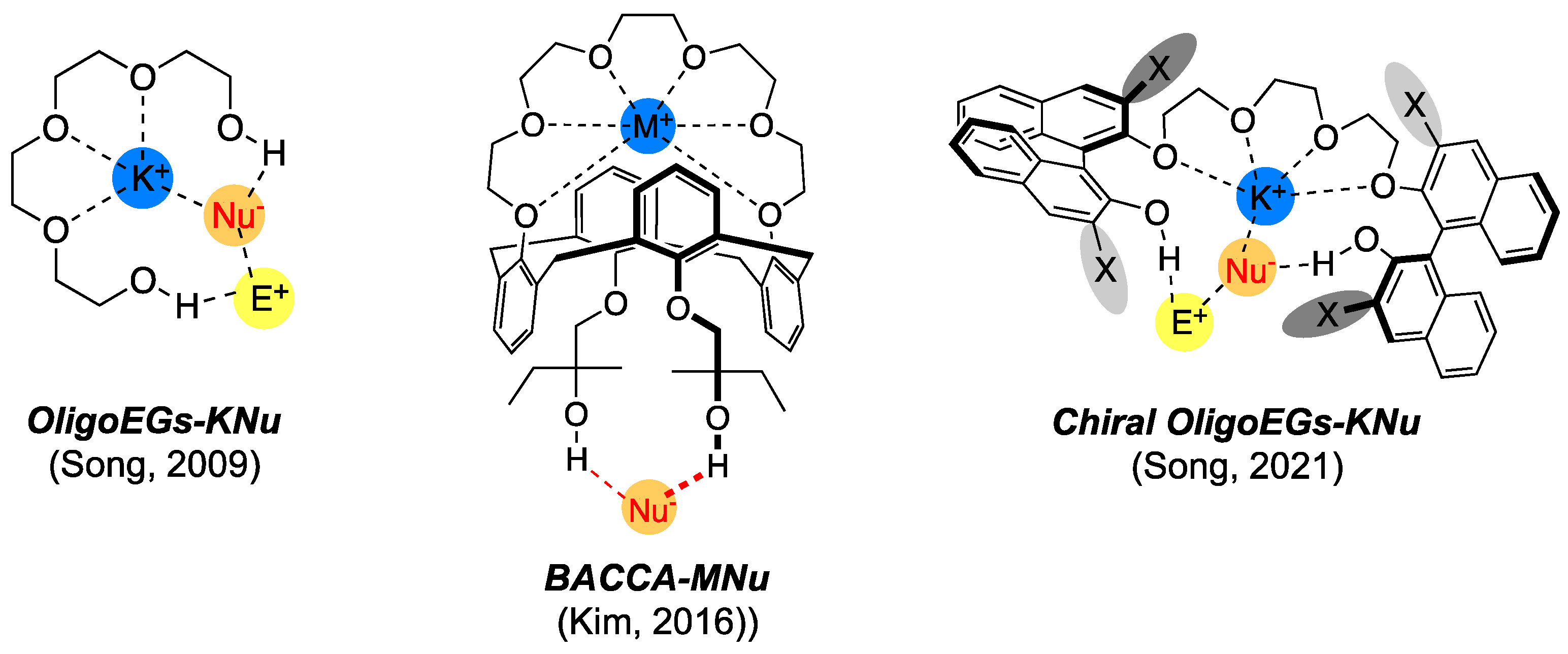
OligoEGs (Figure 6) form a major subset that can be viewed as “truncated” crown ethers. The first (achiral) oligoETs were introduced in 2009 [39] as excellent PTCs for SN2 fluorination using KF. Since then, chiral versions of OligoEGs (BINOL-based) have been developed that show excellent capability for the enantioselective induction of a fluorine atom. Because this progress has been well reviewed elsewhere [14,51], we only discuss other related functionalized crown ethers.
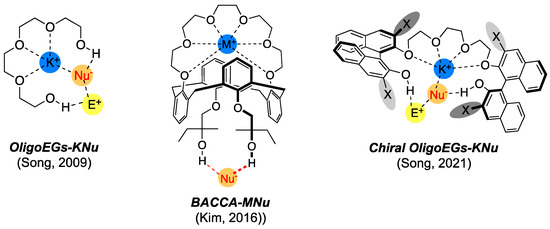
Figure 6.
Oligoethylene glycols and derivatives as PTCs for SN2 reactions. Refs. [39,51,52].
In this respect, Dalessandro and Pliego’s theoretical design of functionalized crown ethers should be noted [52] (Figure 7). The authors predicted that the newly designed catalysts would be effective in promoting SN2 fluorination reactions and that the macrocycle based on a thiourea-crown-ether structure (TCEbut in Figure 7) would be more effective than the [2.2.2]-cryptand by complexing K+ and forming four hydrogen bonds with the TS through the thiourea groups.
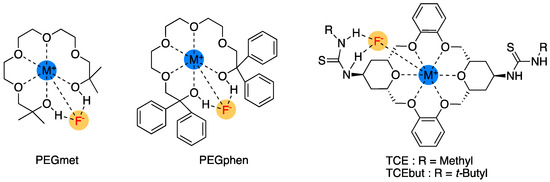
Figure 7.
Crown ether-based PTCs theoretically designed by Dalessandro and Pliego. Ref. [52].
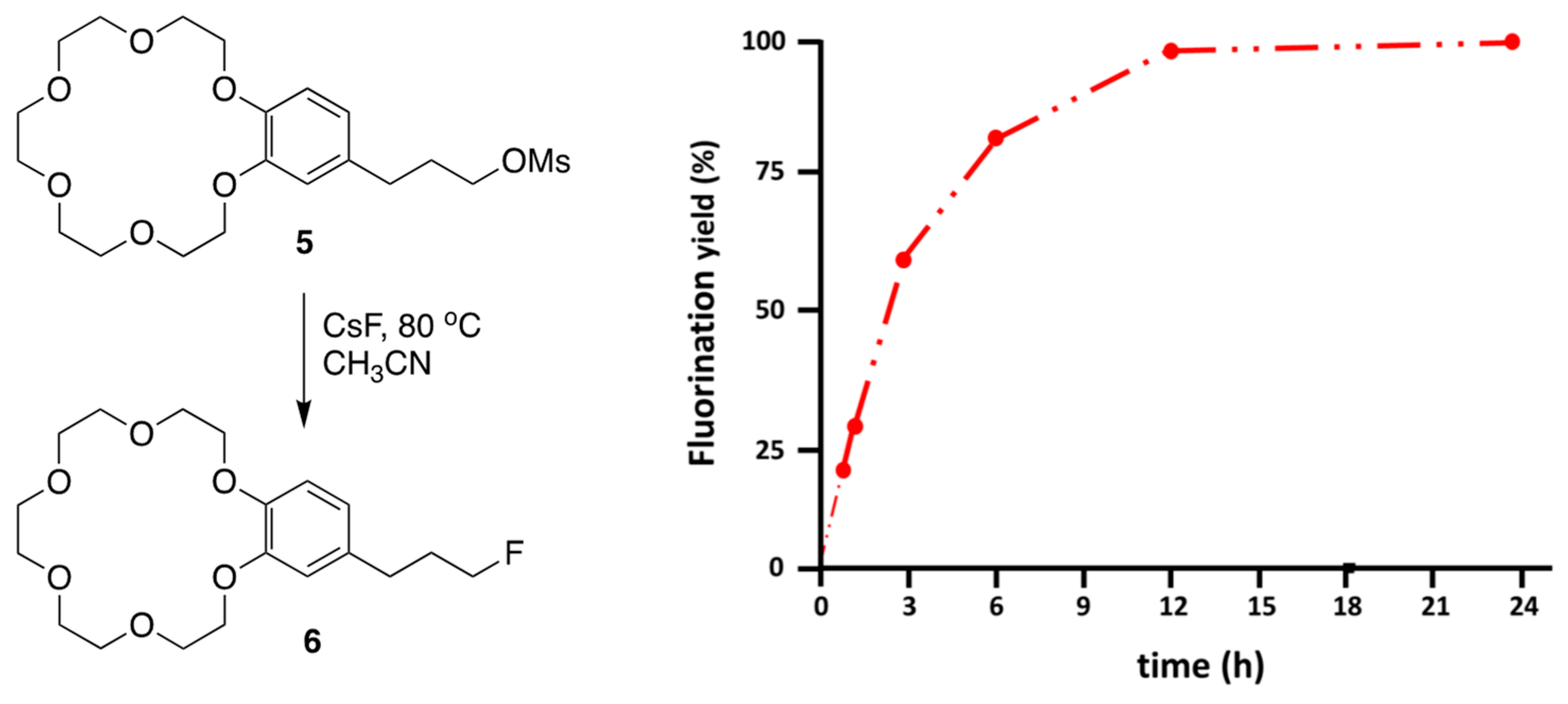
Another type of functionalized crown ether is the crown ether-integrated substrate, synthesized and used to elucidate the mechanism (CIP vs. SIP) of intramolecular SN2 reactions using metal salts activated by the [18-Crown-6] unit, recently studied by Kim and Lee. These systems were adopted to shed further light on the mechanism of crown ether-facilitated processes and to exhibit the outstanding capability of crown ethers as PTCs. Substrate 5 was designed to study the promotion of the intramolecular SN2 reaction using crown ether-integrated substrate 5. Lee and Kim [53] carried out intramolecular SN2 fluorination of 5, promoted by a fused crown ether unit.
The SN2 fluorination yields of the reaction are presented in Figure 8 as a function of time, which shows several interesting kinetic features. Intramolecular SN2 fluorination was nearly complete within 12 h, demonstrating the efficacy of the intramolecular promoter of crown ether.
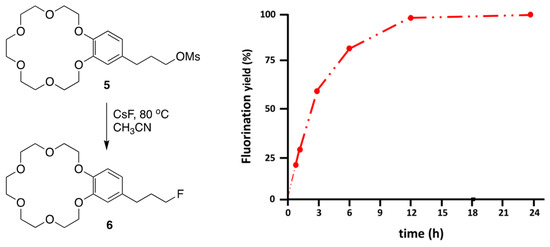
Figure 8.
Progress of SN2 fluorination reactions promoted by intramolecular (reaction A) and intermolecular (reaction B) crown ether [18-Crown-6] in CH3CN at 80 °C. Ref. [54].
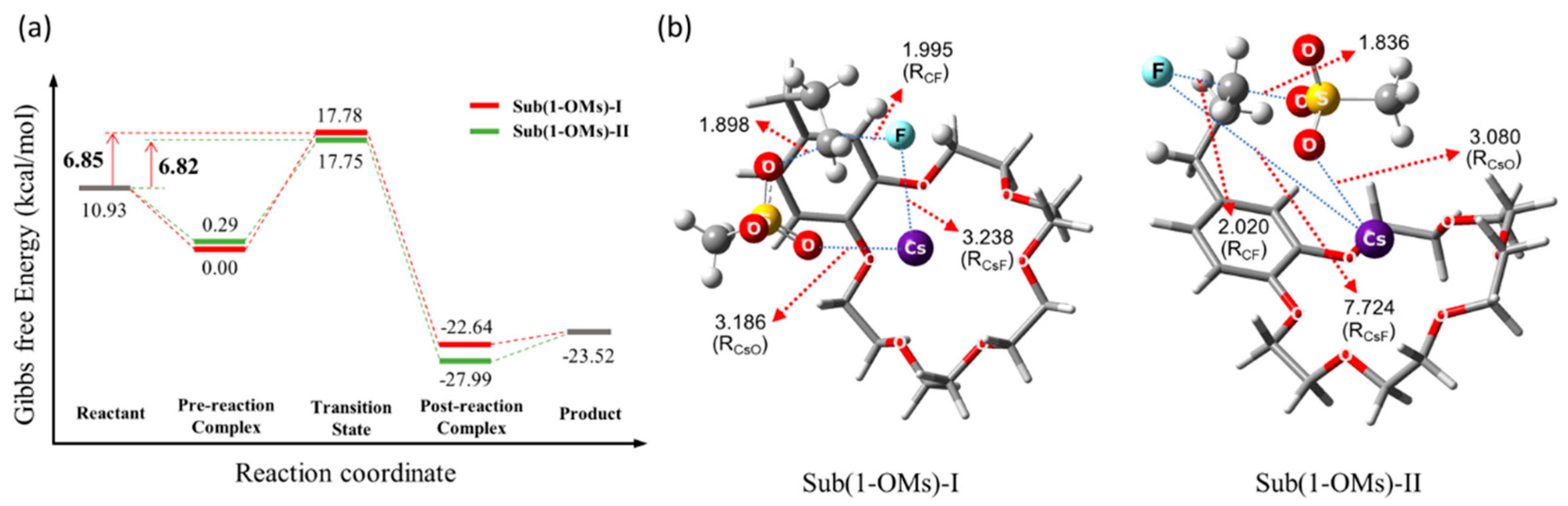
Figure 9 shows the calculated energetics for the intramolecular SN2 fluorination reaction.
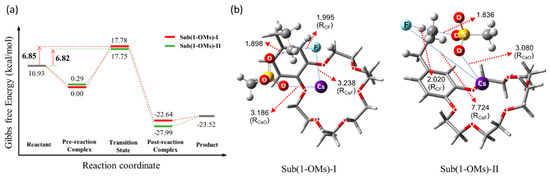
Figure 9.
(a) Energetics and (b) transition states of intramolecular SN2 fluorination of crown ether-integrated substrate using CsF in CH3CN at 80 °C. Ref. [54].
The structures of the TSs involved in the SN2 fluorination are shown in Figure 10. In SN2 fluorination of 5, the nucleophile F− seems to react with 5 either in close contact with (G‡ = 6.85 kcal/mol, RCs-F = 3.238 Å, Sub(1-OMs)-Ι; CIP mechanism) or far away from the counter-cation Cs+ (G‡ = 6.82 kcal/mol, RCs-F = 7.724 Å, Sub(1-OMs)-ΙΙ; separated ion-pair (SIP) mechanism). Because the two reaction paths have very similar energetics (via Sub(1-OMs)-Ι and Sub(1-OMs)-ΙΙ), they were predicted to contribute almost equally to the reaction.
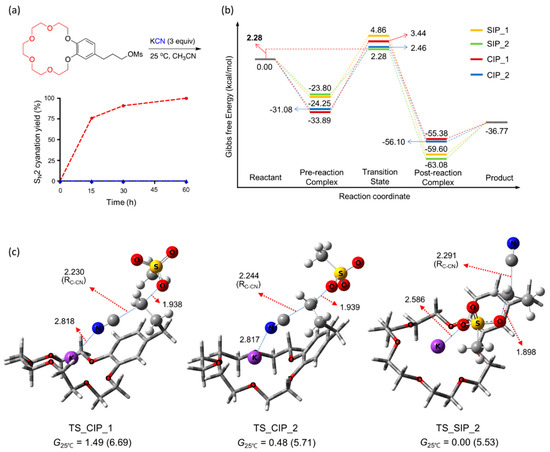
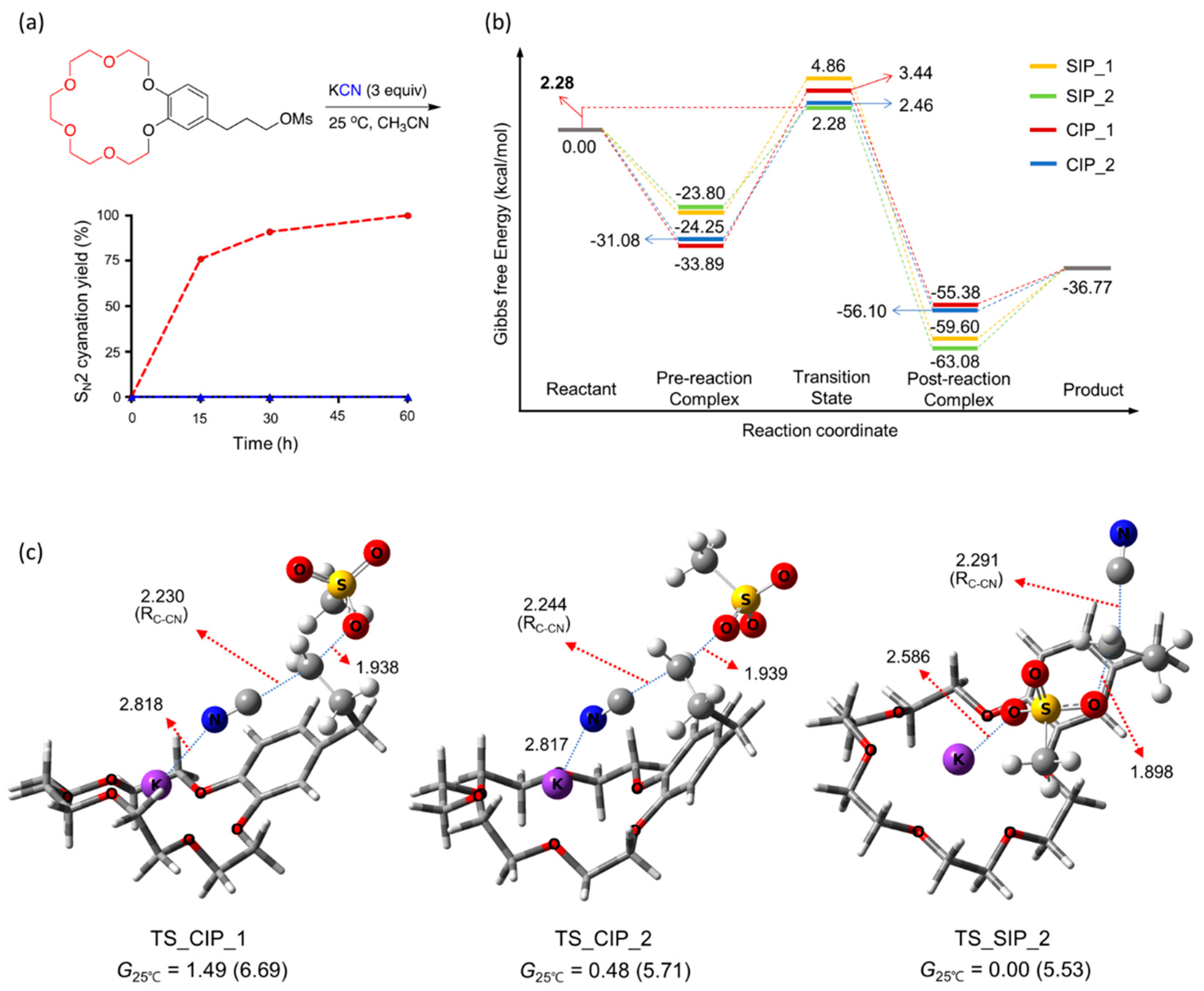
Figure 10.
(a) SN2 yields as functions of time, (b) energetics, and (c) TSs for cyanation using KCN facilitated by intramolecular crown ether [18-Crown-6] in CH3CN at 25 °C. Ref. [55].
Figure 10 presents the observed yields of SN2 cyanation of the crown ether-integrated mesylate substrate [55] as a function of time. SN2 cyanation was extremely fast, almost complete within 2 h at room temperature with no side products, yielding a cyano-product quantitatively. The relative feasibility of the CIP vs. SIP cyanation mechanism was determined by the energetics of the reactions shown in Figure 10. The Gibbs free energies of the TSs for two reaction pathways via (TS_CIP_2) and (TS_SIP_2) are very similar (the difference is only 0.2 kcal/mol), thus they may be considered to contribute almost equally to the cyanation reaction.
4. Synergetic Effects of Crown Ether/Alcohol and/Ammonium Combination
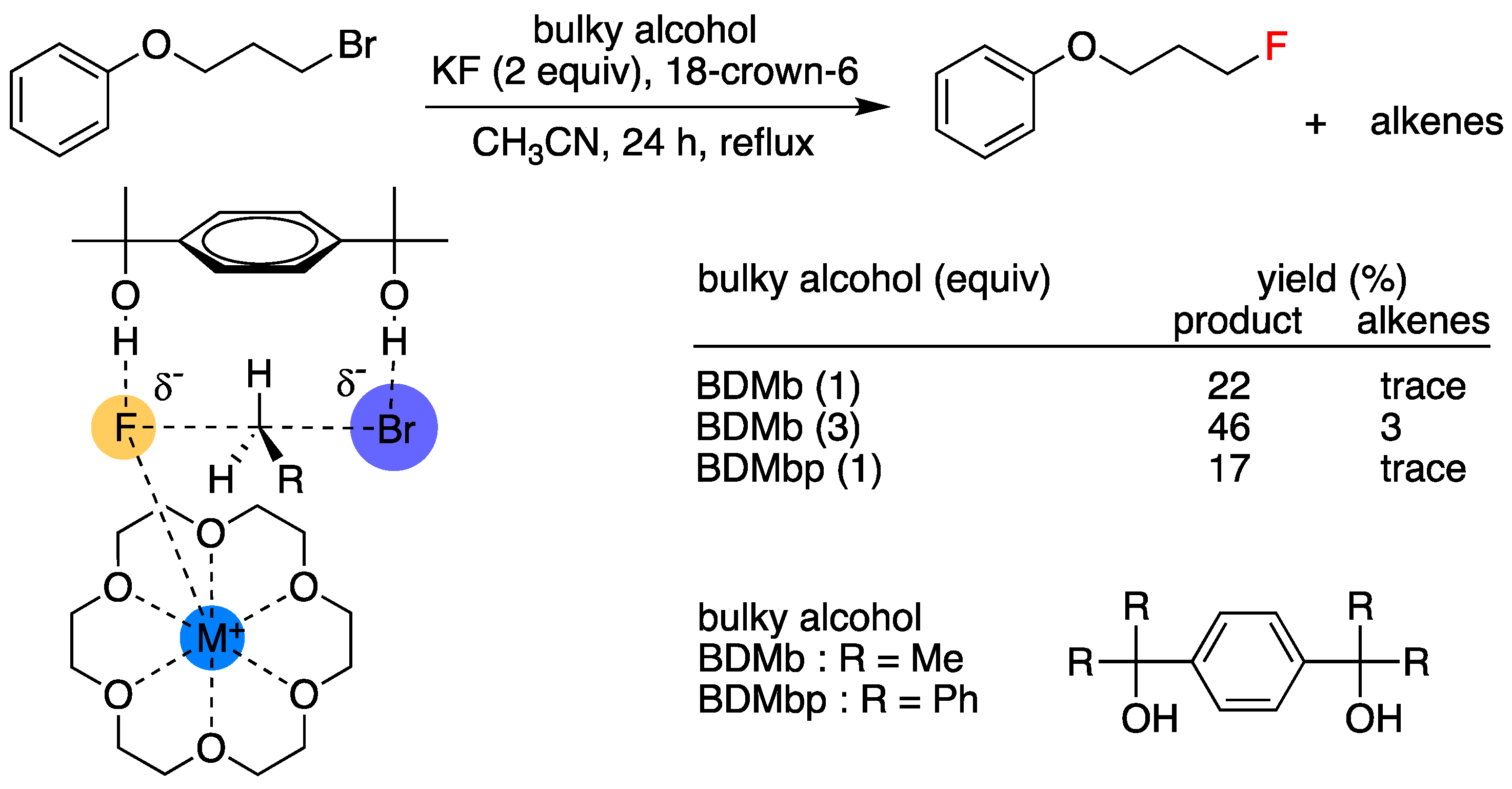
In addition to attempts to modify the structures of crown ethers, some recent studies have examined the use of crown ethers in combination with another PTC to strengthen their catalytic capability. Pliego and coworkers’ theoretical/experimental study [31,56,57,58] on the use of [18-Crown-6] in combination with diols was the most recent scheme (Figure 11).
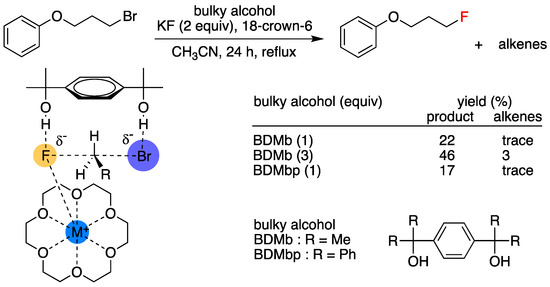
Figure 11.
SN2 fluorination is facilitated by 18-Crown-6 and diols. Ref. [56].
The authors showed that this combined PTC gave much better SN2 yields than those (4.7–13.5%) previously observed by using [18-Crown-6]/t-butanol PTC. This improvement was attributed to double hydrogen bonding between the diols. Hong et al.’s study on the cooperative action of [18-crown-6] and tetramethylammonium (TMA+) salts [59] was also noted (Figure 12). The authors’ scheme afforded fluorinated products in high yields (mostly >80%) and purities under mild conditions (i.e., did not require a rigorously anhydrous environment). A possible explanation is that [18-crown-6] and TMA+, act as cation- and anion-binding ligands, respectively, and that a quadruple CIP mechanism involving TMA+, X−, M+ (M = K, Cs), and F− is at work. Maruoka and co-workers’ [60] observation of the dramatic rate increase of alkylation by using crown ether/achiral quaternary ammonium salt combinations as PTCs is another excellent example along this line of work. This scheme looks promising, which is in line with the intramolecular combined use of BINOL and chiral OligoEGs for asymmetric SN2 reactions described below, but the mechanistic details of the reactions have not yet been systematically studied.
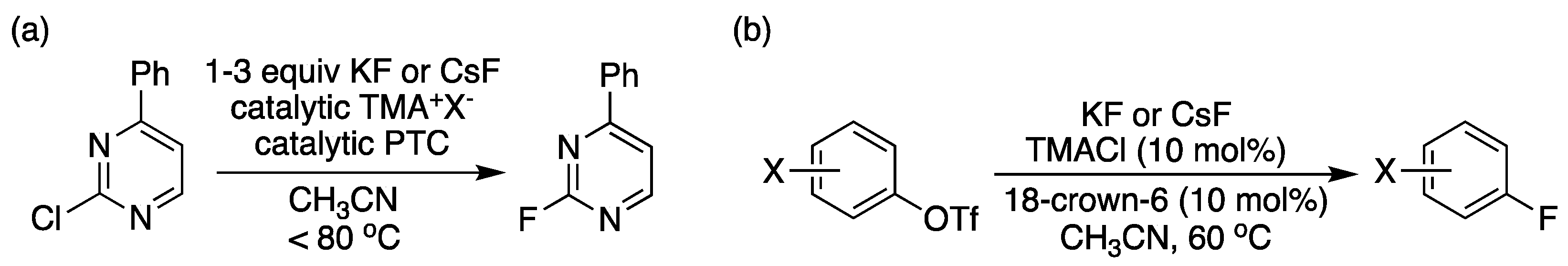
Figure 12.
Nucleophilic fluorination of (a) heteroaryl chlorides and (b) aryl triflates by the [18-crown-6]/tetramethylammonium cooperative catalysis. Ref. [59].
5. Mechanistic Features for Crown Ether-Facilitated SN2 Reactions
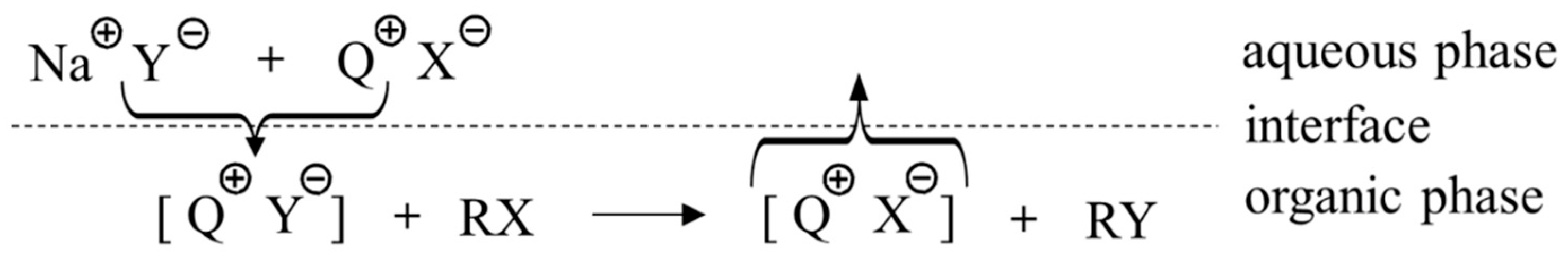
Figure 13 depicts the ion-exchange model of onium-salt-promoted reactions. In this mechanism, alkali metal counter-cations (Na+ in Figure 13), nucleophile Y−, and onium salt cations or anions undergo complicated ion exchange processes, which are not clearly understood. In contrast, crown ethers can solubilize alkali metal salts even in nonpolar organic solvents, not by ion exchange but by complexation [30]. In this sense, they may be called “solid–liquid phase-transfer catalysts” [56]. Therefore, the most relevant question for the mechanism of the crown ether-enhanced reaction is: What is the nature of the complexation?
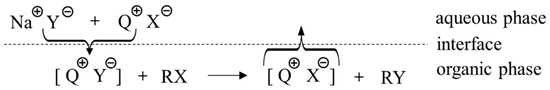
Figure 13.
Schematic mechanism of onium PTCs by ion exchange. Ref. [28].
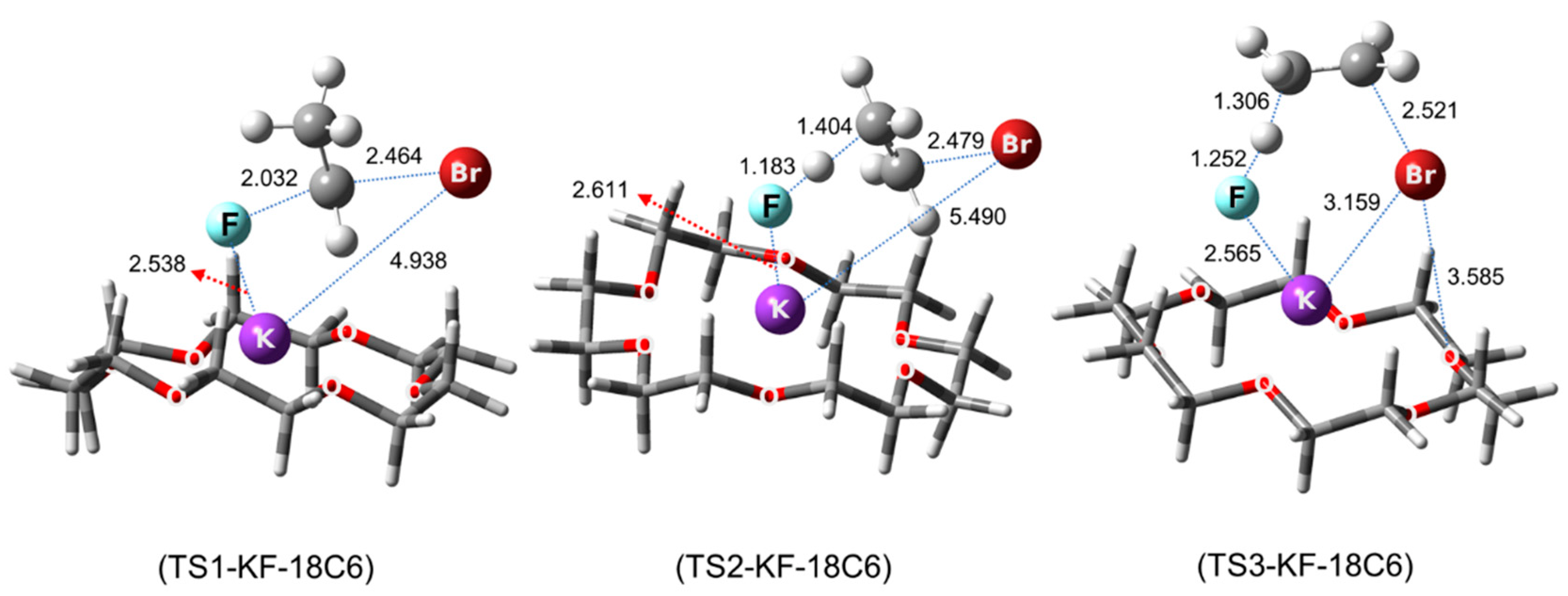
Pliego and Riveros, in 2012, were the first to systematically examine the detailed mechanism of phase-transfer catalysis of the SN2 reaction using metal salt (KF) promoted by [18-crown-6] [56]. By calculating and comparing the mechanism of SN2 fluorination of ethyl bromide by KF in toluene vs. in the presence of [18-crown-6], Pliego and Riveros found that the Gibbs free energy of activation was lowered from 24 to 14.0 kcal/mol, mostly because of the stabilization of K+ in the TSs (Figure 14). Interestingly, KF was seen to be in the CIP form (RKF = 2.54–2.61 Å) in the TSs.
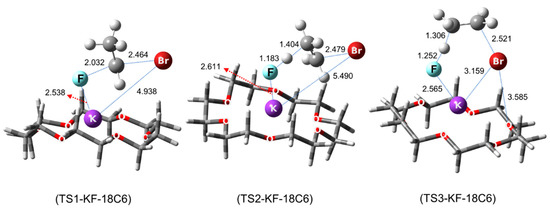
Figure 14.
Calculated transition states in SN2 fluorination of ethyl bromide in the presence of [18-crown-6]. Ref. [58].
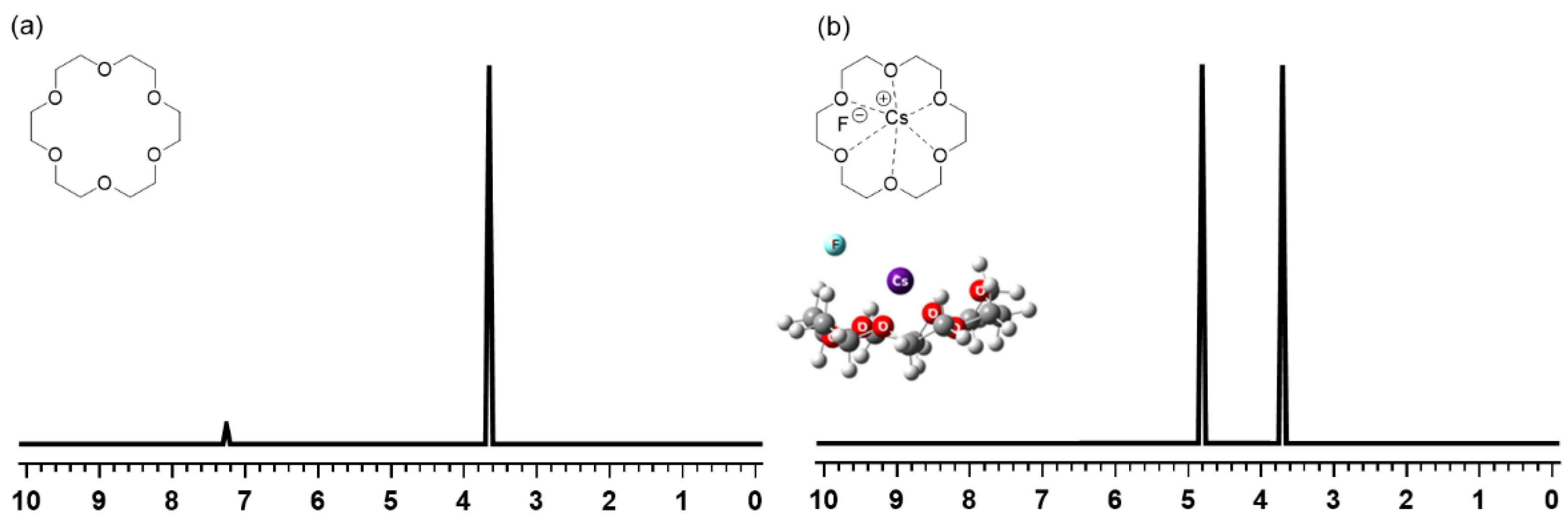
More recent interest in crown ethers has also arisen in connection with the mechanism of the SN2 reaction using metal salts activated by crown ether PTCs. Lee’s group [41,53,54,55,61] carried out systematic studies on the nature (CIP vs. SIP) of metal salts MX (M = K, Cs) in SN2 reactions in the solution phase in the presence of crown ethers. Theoretical (density functional theory) calculations preceded the experimental verification by H-NMR spectroscopic methods for SN2 reactions facilitated by [18-Crown-6]. The first clue to the CIP metal salts interacting with crown ethers was obtained from the H-NMR spectrum of [18-Crown-6]/CsF in D2O (Figure 15) [53], which exhibited two NMR peaks of very similar intensities, in contrast with the observed single peak (at 3.674 ppm) for [18-Crown-6] in CDCl3 (Figure 15). This feature of the H-NMR spectrum of the [18-Crown-6]/CsF complex in D2O was interpreted to reflect the asymmetry of the CsF/[18-Crown-6] complex; the peak at 3.703 ppm was due to the protons farther from F− (and closer to Cs+), whereas that at 4.6763 ppm corresponded to the protons closer to F−. This asymmetry of the CsF/[18-Crown-6] complex clearly manifest that in the complex CsF exists as a CIP even in D2O with a very large dielectric constant. This was indisputable evidence for the role of [18-Crown-6]: the crown ether did not “separate” the counter-cation and the nucleophile. Still, it made them react in CIP form under the minimal influence of the counter-cation Cs+, as Landini, Montanari, and coworkers suggested long ago [26,27].
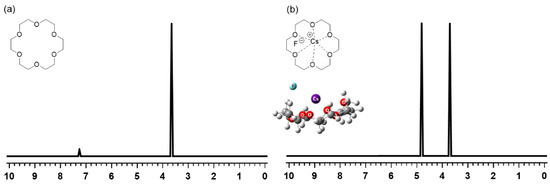
Figure 15.
H-NMR spectra of (a) [18-Crown-6] in CDCl3 and (b) [18-Crown-6]/CsF in D2O. Ref. [53].
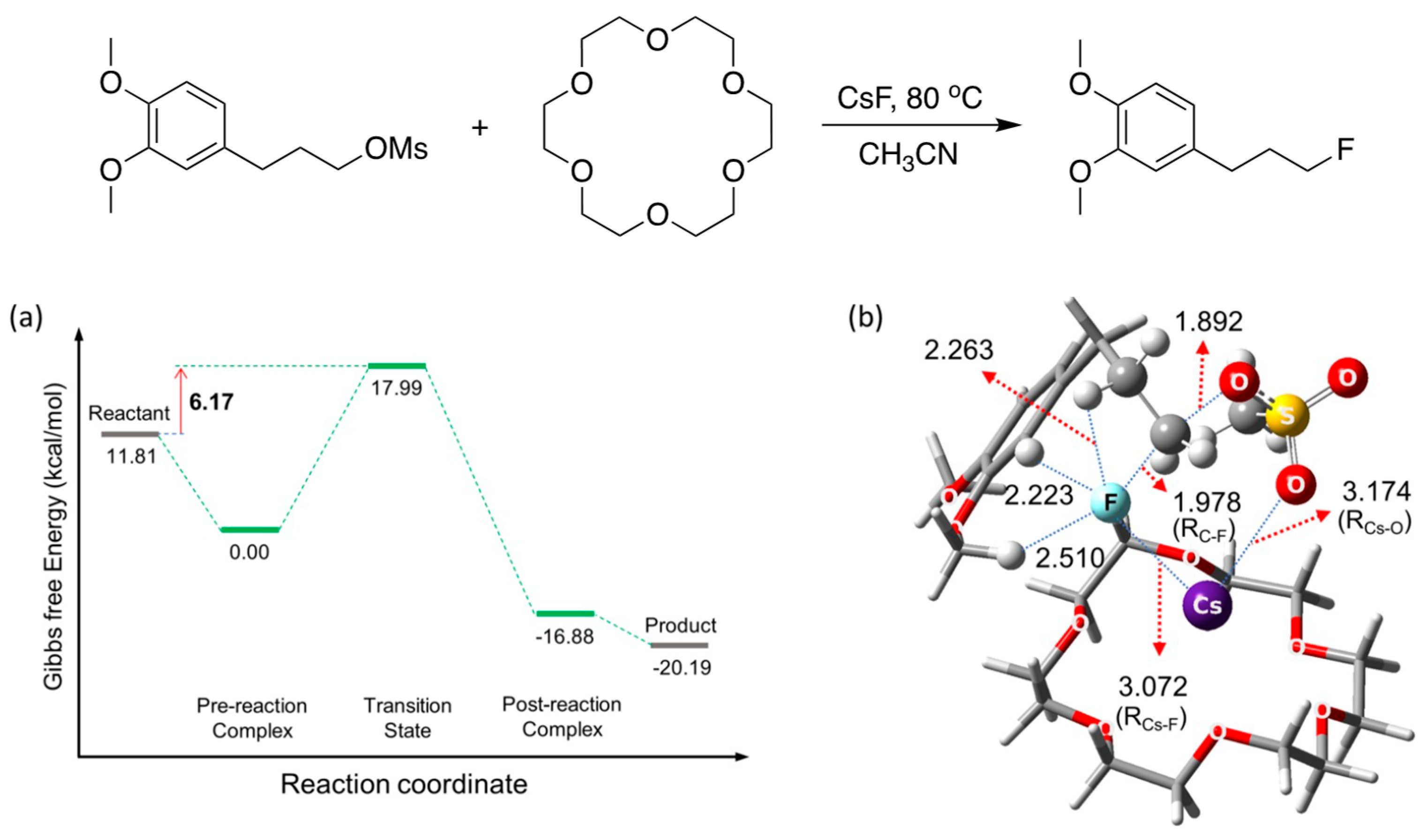
Figure 16 depicts the scheme to illustrate the efficacy of [18-Crown-6] in enhancing the rates of SN2 fluorination, and Figure 16a,b depict the calculated energetics and structure of the TS. The role of [18-crown-6] seems clear: the metal salt Cs+F− coordinates to [18-crown-6] through interactions with the O atoms that act as Lewis bases, alleviating the Coulombic influence of the counter-cation Cs+ to enhance the nucleophilicity of F−. CsF was predicted to react as CIP (RCs-F = 3.072 Å) based on the structure of the TS for the SN2 process.
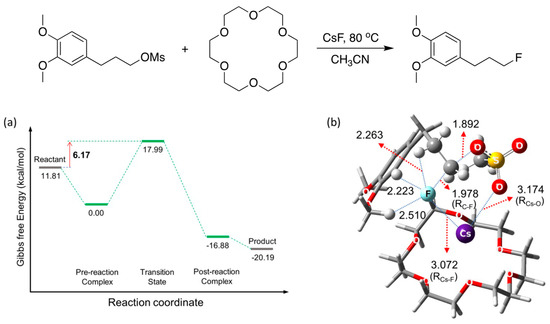
Figure 16.
(a) Energetics and (b) structure of the transition state for SN2 fluorination reactions of the veratrole substrate in the presence of [18-crown-6]. Gibbs free energy in kcal/mol. Ref. [54].
6. Chiral Crown Ethers and Derivatives for Asymmetric Synthesis
A vast number of studies on asymmetric reactions promoted by crown ethers and derivatives have been reported in recent decades, and a comprehensive review of the development of these exciting fields would be impossible. Thus, we have limited our discussion to nucleophilic processes. Crown ether-facilitated processes involving asymmetric phase-transfer [62] or supramolecular [63] catalysis have been well reviewed elsewhere; therefore, they are not included here.
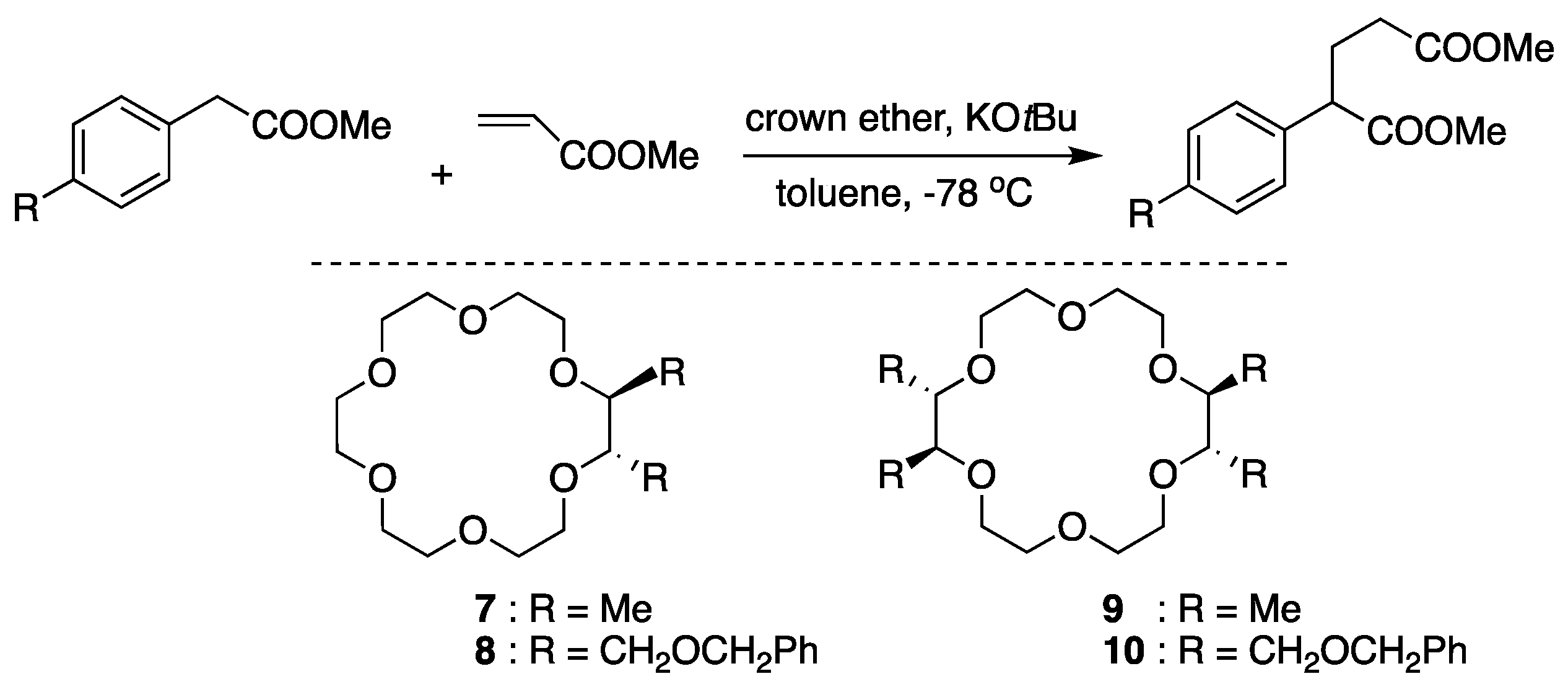
Koga and coworkers’ chiral crown ether complexed with KOtBu [64] is one of the simplest prototypes of chiral crown ethers (Figure 17). Chiral induction by the chiral catalysts in Michael addition reactions was attributed to the diaxial-like conformation of the vicinal methyl groups in 1·potassium enolate complex.
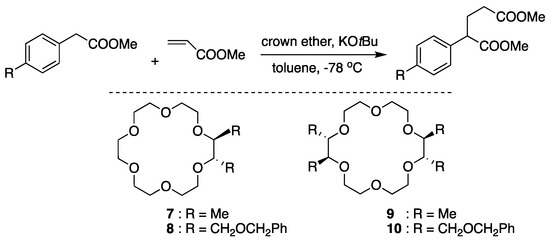
Figure 17.
Michael addition reactions by chiral crown ethers complexed with KOtBu. Ref. [64].
Dela Sala, Izzo, and co-workers’ work on the arylogous Michael addition [65] and the fluoride-promoted vinylogous Mukaiyama–Michael reaction [66] are also to be noted as primary examples in which the crown ether/CIP metal salt was proposed to work for excellent diastereoselective reactions.
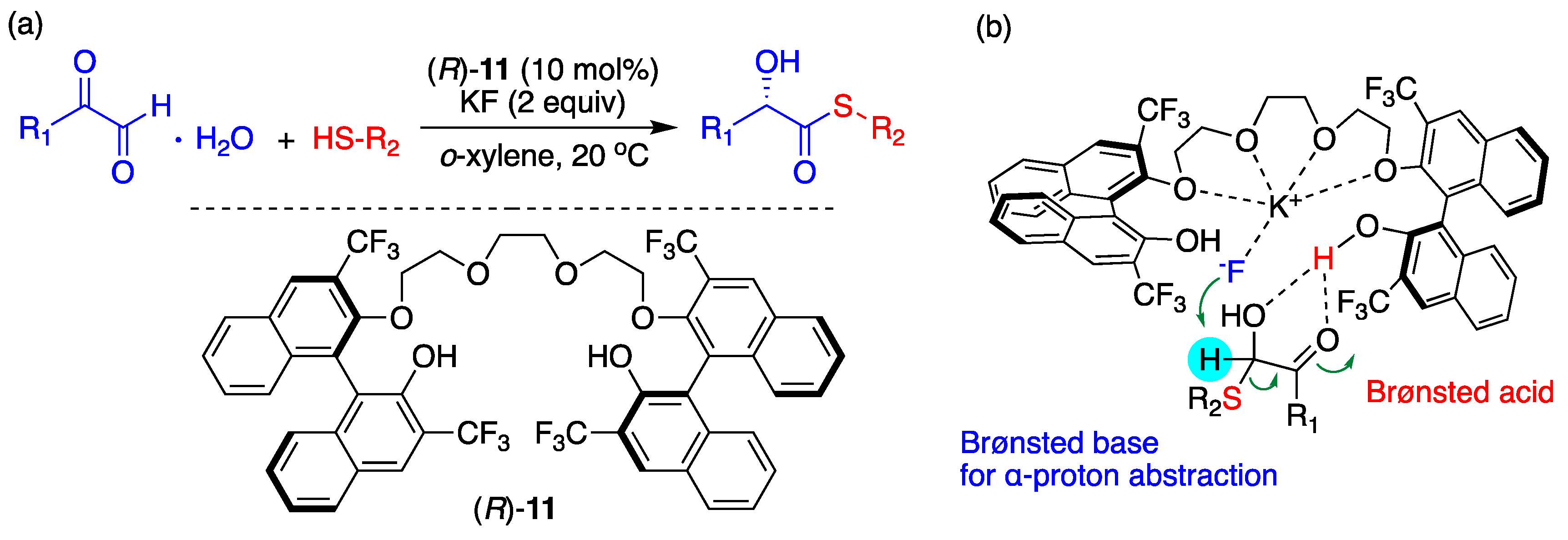
Song and coworkers’ use of BINOL-based chiral OligoEGs for various asymmetric nucleophilic reactions seems to be the most prominent recent progress. These authors utilized the cation-coordinating capability of crown ether but introduced powerful hydrogen bonds of –OHs by truncating the ring. Chirality was induced by functionalizing the oligoEGs with BINOL units. These chiral PTCs worked well with excellent yields and ee%, as discussed in a recent review [67]. Figure 18 illustrates an example (yield, ee% > 90%). Song and coworkers confirmed the formation of some BINOL-based chiral oligoEG/KF complexes by ESI-HRMS and proposed a plausible TS. From the structure of the TS depicted in Figure 18, some mechanistic features could be deduced: (1) The O atoms in the OligoEG ring coordinate K+, which is in close vicinity to F− (KF is in the form of CIP); (2) F− acts as a Brønsted base for abstracting the α-hydrogen of thiol, thus providing a partial negative charge to –SH to increase the nucleophilicity of the latter; (3) One of the two BINOL –OHs acts as an “anchor” for F−, whereas the other –OH donates acidic hydrogen to aldehyde carboxyl. Experimental elucidation of the structure of the chiral OligoEG/KF complex would unambiguously confirm the CIP mechanism.
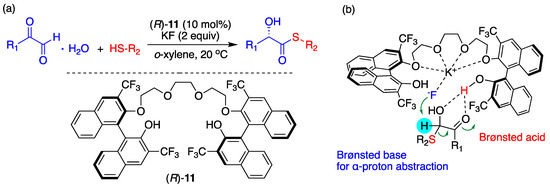
Figure 18.
(a) Asymmetric synthesis of chiral α-hydroxy thioesters from α-oxoaldehydes and (b) proposed transition state. Ref. [67].
Biomolecules are promising candidates for use as chiral side chains in crown ethers. Rapi and coworkers [68] synthesized various crown ether-D-glucoses and used them as PCTs. Figure 19 depicts the enantioselective Michael-initiated ring closure reaction. The enantioselectivity of the PTCs was excellent, with >88% ee (Table 2).
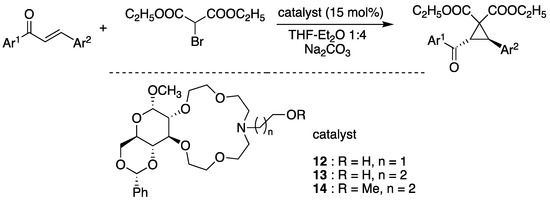
Figure 19.
Crown ether-D-glucose as an efficient phase-transfer catalyst for the enantioselective Michael-initiated ring closure reaction of chalcones and diethyl bromomalonate. Ref. [69].

Table 2.
Effect of crown ethers on the MIRC reaction of chalcones and diethyl bromomalonate. Ref. [69].
Voyer’s [44,45] use of crown ether-modified cyclic dipeptides as supramolecular chiral catalysts for enantioselective epoxidation (Table 3) is another excellent example. The authors synthesized various crown ether-modified cyclic dipeptides and examined their performance as supramolecular PTCs for enantioselective epoxidation reactions. They found that the method significantly improved the reaction yields and enantioselectivities (up to 93 and 40 ee%, respectively) from those obtained using the cyclic dipeptides alone [44] (Table 3). The mechanism or origin of the observed enantioselectivity is highly intriguing to elucidate. There also exist many examples for crown ether catalyzed asymmetric synthesis, for example, transition-metal assisted of crown ether catalyzed asymmetric synthetic methods. However, since we cannot hope to be exhaustive in this brief review, we only cite a nice recent review [63] for this topic.

Table 3.
Enantioselective epoxidation facilitated by the crown ether-modified cyclic di-leucine 21 Ref. [44].
7. Concluding Remarks
Since their discovery in 1967, crown ethers have found applications in various fields, solving some difficult chemical problems. Here, we focus mainly on the efficiency of crown ethers and the mechanistic features of PTCs. Using these materials for a wider range of asymmetric syntheses is an exciting avenue for exploration. The experimental evidence delineated in this review is expected to shed further light on the mechanism of catalysis by crown ethers, which will aid in the design, manufacture, and application of more efficient and selective crown ether PTCs because understanding the underlying mechanism is always the most effective and economical means to advance technology.
8. Materials and Methods
All calculations were carried out by M06-2X [70] density functional theory method with 6-311G** basis set for C, H, O, S, F atoms and LANL2DZ basis set for Cs, Br with its corresponding effective core potential [71] as implemented in Gaussian16 suite of programs [72]. The SMD [73] model was used to treat the effects of solvation. All frequencies for pre-/post- reaction complexes were confirmed to be real, and for TSs only one of those for reaction coordinate was ascertained to be imaginary. To determine the unique reaction pathway, the intrinsic reaction coordinate analysis was applied.
Author Contributions
D.W.K. and J.G.J. performed the reactions and H-NMR spectroscopy experiments. Y.-H.O. and S.L. performed the quantum chemical calculations. S.L. and D.W.K. devised the study’s overall concept and the system’s modeling. D.W.K. and S.L. prepared the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the Korea Research Foundation (NRF-2019R1F1A1057609, 2020R1A2C1009017), Ministry of Science and ICT (MSIT), Ministry of Trade, Industry & Energy (MOTIE), and Korea Semiconductor Research Consortium (KSRC) program (grant code:20019504), Republic of Korea, and KISTI Supercomputer Center.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pedersen, C.J. The Discovery of Crown Ethers. Science 1988, 241, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, C.J. Cyclic Polyethers and Their Complexes with Metal Salts. J. Am. Chem. Soc. 1967, 89, 7017–7036. [Google Scholar] [CrossRef]
- Gokel, G.W. Crown Ethers and Cryptands; Royal Society of Chemistry: London, UK, 1991; ISBN 0851869963. [Google Scholar]
- Inoue, Y.; Gokel, G.W. Cation Binding by Macrocycles: Complexation of Cationic Species by Crown Ethers, 1st ed.; Routledge: New York, NY, USA, 1990; ISBN 0824781872. [Google Scholar]
- Bradshaw, J.S.; Izatt, R.M. Crown Ethers: The Search for Selective Ion Ligating Agents. Acc. Chem. Res. 1997, 30, 338–345. [Google Scholar] [CrossRef]
- Gokel, G.W.; Leevy, W.M.; Weber, M.E. Crown Ethers: Sensors for Ions and Molecular Scaffolds for Materials and Biological Models. Chem. Rev. 2004, 104, 2723–2750. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Xu, C.; Ren, J.; Xu, B.; Qu, X. Sensing Metal Ions with Ion Selectivity of a Crown Ether and Fluorescence Resonance Energy Transfer between Carbon Dots and Graphene. Chem. Commun. 2012, 48, 1284–1286. [Google Scholar] [CrossRef]
- Warnock, S.J.; Sujanani, R.; Zofchak, E.S.; Zhao, S.; Dilenschneider, T.J.; Hanson, K.G.; Mukherjee, S.; Ganesan, V.; Freeman, B.D.; Abu-Omar, M.M. Engineering Li/Na Selectivity in 12-Crown-4–Functionalized Polymer Membranes. Proc. Natl. Acad. Sci. USA 2021, 118, e2022197118. [Google Scholar] [CrossRef]
- Sasaki, S.; Shionoya, M.; Koga, K. Functionalized Crown Ethers as an Approach to the Enzyme Model for the Synthesis of Peptides. J. Am. Chem. Soc. 1985, 107, 3371–3372. [Google Scholar] [CrossRef]
- Yu, L.; Li, F.Z.; Wu, J.Y.; Xie, J.Q.; Li, S. Development of the Aza-Crown Ether Metal Complexes as Artificial Hydrolase. J. Inorg. Biochem. 2016, 154, 89–102. [Google Scholar] [CrossRef]
- Zhang, M.; Yan, X.; Huang, F.; Niu, Z.; Gibson, H.W. Stimuli-Responsive Host–Guest Systems Based on the Recognition of Cryptands by Organic Guests. Acc. Chem. Res. 2014, 47, 1995–2005. [Google Scholar] [CrossRef]
- Qu, D.-H.; Wang, Q.-C.; Zhang, Q.-W.; Ma, X.; Tian, H. Photoresponsive Host–Guest Functional Systems. Chem. Rev. 2015, 115, 7543–7588. [Google Scholar] [CrossRef]
- Kim, D.W.; Ahn, D.-S.; Oh, Y.-H.; Lee, S.; Kil, H.S.; Oh, S.J.; Lee, S.J.; Kim, J.S.; Ryu, J.S.; Moon, D.H.; et al. A New Class of SN2 Reactions Catalyzed by Protic Solvents: Facile Fluorination for Isotopic Labeling of Diagnostic Molecules. J. Am. Chem. Soc. 2006, 128, 16394–16397. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-W.; Oliveira, M.T.; Jang, H.B.; Lee, S.; Chi, D.Y.; Kim, D.W.; Song, C.E. Hydrogen-Bond Promoted Nucleophilic Fluorination: Concept, Mechanism and Applications in Positron Emission Tomography. Chem. Soc. Rev. 2016, 45, 4638–4650. [Google Scholar] [CrossRef] [PubMed]
- Oh, Y.-H.; Ahn, D.-S.; Chung, S.-Y.; Jeon, G.-H.; Park, S.-W.; Oh, S.J.; Kim, D.W.; Kil, H.S.; Chi, D.Y.; Lee, S. Facile SN2 Reaction in Protic Solvent: Quantum Chemical Analysis. J. Phys. Chem. A 2007, 111, 10152–10161. [Google Scholar] [CrossRef]
- Laloo, J.Z.A.; Rhyman, L.; Larrañaga, O.; Ramasami, P.; Bickelhaupt, F.M.; de Cózar, A. Ion-Pair SN2 Reaction of OH− and CH3Cl: Activation Strain Analyses of Counterion and Solvent Effects. Chem. Asian J. 2018, 13, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
- Laloo, J.Z.A.; Rhyman, L.; Ramasami, P.; Bickelhaupt, F.M.; de Cózar, A. Ion-Pair SN2 Substitution: Activation Strain Analyses of Counter-Ion and Solvent Effects. Chem. Eur. J. 2016, 22, 4431–4439. [Google Scholar] [CrossRef] [PubMed]
- Harder, S.; Streitwieser, A.; Petty, J.T.; Ragué Schleyer, P. von Ion Pair SN2 Reactions. Theoretical Study of Inversion and Retention Mechanisms. J. Am. Chem. Soc. 1995, 117, 3253–3259. [Google Scholar] [CrossRef]
- Ren, Y.; Gai, J.-G.; Xiong, Y.; Lee, K.-H.; Chu, S.-Y. Theoretical Study on the Identity Ion Pair SN2 Reactions of LiX with CH3SX (X = Cl, Br, and I): Structure, Mechanism, and Potential Energy Surface. J. Phys. Chem. A 2007, 111, 6615–6621. [Google Scholar] [CrossRef]
- Dalessandro, E.V.; Pliego, J.R. Reactivity and Stability of Ion Pairs, Dimers and Tetramers versus Solvent Polarity: SNAr Fluorination of 2-Bromobenzonitrile with Tetramethylammonium Fluoride. Theor. Chem. Acc. 2020, 139, 27. [Google Scholar] [CrossRef]
- Stott, P.E.; Bradshaw, J.S.; Parish, W.W. Modified Crown Ether Catalysts. 3. Structural Parameters Affecting Phase Transfer Catalysis by Crown Ethers and a Comparison of the Effectiveness of Crown Ethers to That of Other Phase Transfer Catalysts. J. Am. Chem. Soc. 1980, 102, 4810–4815. [Google Scholar] [CrossRef]
- Guida, W.C.; Mathre, D.J. Phase-Transfer Alkylation of Heterocycles in the Presence of 18-Crown-6 and Potassium Tert-Butoxide. J. Org. Chem. 1980, 45, 3172–3176. [Google Scholar] [CrossRef]
- Cacciapaglia, R.; Mandolins, L. Catalysis by Metal Ions in Reactions of Crown Ether Substrates. Chem. Soc. Rev. 1993, 22, 221–231. [Google Scholar] [CrossRef]
- Di Stefano, S.; Capocasa, G.; Mandolini, L. Supramolecular Catalysts Featuring Crown Ethers as Recognition Units. Eur. J. Org. Chem. 2020, 23, 3340–3350. [Google Scholar] [CrossRef]
- Fehér, Z.; Richter, D.; Nagy, S.; Bagi, P.; Rapi, Z.; Simon, A.; Drahos, L.; Huszthy, P.; Bakó, P.; Kupai, J. Synthesis of Novel Crown Ether-Squaramides and Their Application as Phase-Transfer Catalysts. Molecules 2021, 26, 6542. [Google Scholar] [CrossRef] [PubMed]
- Landini, D.; Montanari, F.; Pirisi, F.M. Crown Ethers as Phase-Transfer Catalysts in Two-Phase Reactions. J. Chem. Soc. Chem. Commun. 1974, 21, 879–880. [Google Scholar] [CrossRef]
- Landini, D.; Maia, A.; Montanari, F.; Pirisi, F.M. Crown Ethers as Phase-Transfer Catalysts. A Comparison of Anionic Activation in Aqueous–Organic Two-Phase Systems and in Low Polarity Anhydrous Solutions by Perhydrodibenzo-18-Crown-6, Lipophilic Quaternary Salts, and Cryptands. J. Chem. Soc. Perkin Trans. 1980, 2, 46–51. [Google Scholar] [CrossRef]
- Dehmlow, E. V Advances in Phase-Transfer Catalysis [New Synthetic Methods (20)]. Angew. Chem. Int. Ed. 1977, 16, 493–505. [Google Scholar] [CrossRef]
- Liotta, C.L.; Harris, H.P. Chemistry of Naked Anions. I. Reactions of the 18-Crown-6 Complex of Potassium Fluoride with Organic Substrates in Aprotic Organic Solvents. J. Am. Chem. Soc. 1974, 96, 2250–2252. [Google Scholar] [CrossRef]
- Olsher, U.; Frolow, F.; Dalley, N.K.; Jiang, W.; Yu, Z.Y.; Knobeloch, J.M.; Bartsch, R.A. Crown Ether Alcohols as Bifunctional Ligands for Simultaneous Cation Complexation and Anion Solvation. J. Am. Chem. Soc. 1991, 113, 6570–6574. [Google Scholar] [CrossRef]
- Carvalho, N.F.; Pliego, J.R. Theoretical Design and Calculation of a Crown Ether Phase-Transfer-Catalyst Scaffold for Nucleophilic Fluorination Merging Two Catalytic Concepts. J. Org. Chem. 2016, 81, 8455–8463. [Google Scholar] [CrossRef]
- Iashin, V.; Wirtanen, T.; Perea-Buceta, J.E. Tetramethylammonium Fluoride: Fundamental Properties and Applications in CF Bond-Forming Reactions and as a Base. Catalysts 2022, 12, 233. [Google Scholar] [CrossRef]
- Lee, S.J.; Morales-Colón, M.T.; Brooks, A.F.; Wright, J.S.; Makaravage, K.J.; Scott, P.J.H.; Sanford, M.S. SNAr Radiofluorination with in Situ Generated [18F]Tetramethylammonium Fluoride. J. Org. Chem. 2021, 86, 14121–14130. [Google Scholar] [CrossRef]
- Schimler, S.D.; Froese, R.D.J.; Bland, D.C.; Sanford, M.S. Reactions of Arylsulfonate Electrophiles with NMe4F: Mechanistic Insight, Reactivity, and Scope. J. Org. Chem. 2018, 83, 11178–11190. [Google Scholar] [CrossRef] [PubMed]
- Shinde, S.S.; Bolik, K.-V.; Maschauer, S.; Prante, O. 18F-Fluorination Using Tri-Tert-Butanol Ammonium Iodide as Phase-Transfer Catalyst: An Alternative Minimalist Approach. Pharmaceuticals 2021, 14, 833. [Google Scholar] [CrossRef] [PubMed]
- Starks, C.M. Phase-Transfer Catalysis. I. Heterogeneous Reactions Involving Anion Transfer by Quaternary Ammonium and Phosphonium Salts. J. Am. Chem. Soc. 1971, 93, 195–199. [Google Scholar] [CrossRef]
- Liu, S.; Kumatabara, Y.; Shirakawa, S. Chiral Quaternary Phosphonium Salts as Phase-Transfer Catalysts for Environmentally Benign Asymmetric Transformations. Green Chem. 2016, 18, 331–341. [Google Scholar] [CrossRef]
- He, R.; Ding, C.; Maruoka, K. Phosphonium Salts as Chiral Phase-Transfer Catalysts: Asymmetric Michael and Mannich Reactions of 3-Aryloxindoles. Angew. Chem. Int. Ed. 2009, 48, 4559–4561. [Google Scholar] [CrossRef]
- Lee, J.W.; Yan, H.; Jang, H.B.; Kim, H.K.; Park, S.; Lee, S.; Chi, D.Y.; Song, C.E. Bis-Terminal Hydroxy Polyethers as All-Purpose, Multifunctional Organic Promoters: A Mechanistic Investigation and Applications. Angew. Chem. Int. Ed. 2009, 48, 7683–7686. [Google Scholar] [CrossRef]
- Kim, D.W.; Song, C.E.; Chi, D.Y. New Method of Fluorination Using Potassium Fluoride in Ionic Liquid: Significantly Enhanced Reactivity of Fluoride and Improved Selectivity. J. Am. Chem. Soc. 2002, 124, 10278–10279. [Google Scholar] [CrossRef]
- Oh, Y.-H.; Jang, H.B.; Im, S.; Song, M.J.; Kim, S.-Y.; Park, S.-W.; Chi, D.Y.; Song, C.E.; Lee, S. SN2 Fluorination Reactions in Ionic Liquids: A Mechanistic Study towards Solvent Engineering. Org. Biomol. Chem. 2011, 9, 418–422. [Google Scholar] [CrossRef]
- Oh, Y.-H.; Kim, D.W.; Lee, S. Ionic Liquids as Organocatalysts for Nucleophilic Fluorination: Concepts and Perspectives. Molecules 2022, 27, 5702. [Google Scholar] [CrossRef]
- Pietraszkiewicz, M.; Jurczak, J. Synthesis of Chiral Diaza-Crown Ethers Incorporating Carbohydrate Units. Tetrahedron 1984, 40, 2967–2970. [Google Scholar] [CrossRef]
- Bérubé, C.; Voyer, N. Crown-Ether-Modified Cyclic Dipeptides as Supramolecular Chiral Catalysts. Supramol. Chem. 2018, 30, 184–195. [Google Scholar] [CrossRef]
- Bérubé, C.; Barbeau, X.; Cardinal, S.; Boudreault, P.L.; Bouchard, C.; Delcey, N.; Lagüe, P.; Voyer, N. Interfacial Supramolecular Biomimetic Epoxidation Catalysed by Cyclic Dipeptides. Supramol. Chem. 2017, 29, 330–349. [Google Scholar] [CrossRef]
- Dietrich, B.; Lehn, J.M.; Sauvage, J.P. Les Cryptates. Tetrahedron Lett. 1969, 10, 2889–2892. [Google Scholar] [CrossRef]
- Lehn, J.M. Accounts of Chemical Research Cryptates: The Chemistry of Macropolycyclic Inclusion Complexes. Acc. Chem. Res. 1978, 11, 49–57. [Google Scholar] [CrossRef]
- Kang, S.O.; Llinares, J.M.; Day, V.W.; Bowman-James, K. Cryptand-like Anion Receptors. Chem. Soc. Rev. 2010, 39, 3980–4003. [Google Scholar] [CrossRef]
- Landini, D.; Maia, A.; Montanari, F.; Tundo, P. Lipophilic [2.2.2] Cryptands as Phase-Transfer Catalysts. Activation and Nucleophilicity of Anions in Aqueous-Organic Two-Phase Systems and in Organic Solvents of Low Polarity. J. Am. Chem. Soc. 1979, 101, 2526–2530. [Google Scholar] [CrossRef]
- Pliego, J.R. Potassium Fluoride Activation for the Nucleophilic Fluorination Reaction Using 18-Crown-6,[2.2.2]-Cryptand, Pentaethylene Glycol and Comparison with the New Hydro-Crown Scaffold: A Theoretical Analysis. Org. Biomol. Chem. 2018, 16, 3127–3137. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, A.P.; Park, S.Y.; Lee, J.-W.; Yan, H.; Song, C.E. Cooperative Asymmetric Cation-Binding Catalysis. Acc. Chem. Res. 2021, 54, 4319–4333. [Google Scholar] [CrossRef] [PubMed]
- Dalessandro, E.V.; Pliego, J.R. Theoretical Design of New Macrocycles for Nucleophilic Fluorination with KF: Thiourea-Crown-Ether Is Predicted to Overcome [2.2.2]-Cryptand. Mol. Syst. Des. Eng. 2020, 5, 1513–1523. [Google Scholar] [CrossRef]
- Jadhav, V.H.; Jeong, H.J.; Choi, W.; Kim, D.W. Crown Ether Metal Complex Fluoride Salt as a Facile and Low Hygroscopic Fluoride Source for Nucleophilic Fluorination. Chem. Eng. J. 2015, 270, 36–40. [Google Scholar] [CrossRef]
- Oh, Y.-H.; Yun, W.; Kim, C.-H.; Jang, S.-W.; Lee, S.-S.; Lee, S.; Kim, D.-W. Inter-and Intra-Molecular Organocatalysis of SN2 Fluorination by Crown Ether: Kinetics and Quantum Chemical Analysis. Molecules 2021, 26, 2947. [Google Scholar] [CrossRef]
- Oh, Y.; Yun, W.; Lee, S.; Kim, D.W. Kinetics and Quantum Chemical Analysis of Intramolecular SN2 Reactions by Using Metal Salts and Promoted by Crown Ethers: Contact Ion Pair vs. Separated Nucleophile Mechanism. ChemistrySelect 2022, 7, e202104431. [Google Scholar] [CrossRef]
- Pliego, J.R., Jr.; Riveros, J.M. New Insights on Reaction Pathway Selectivity Promoted by Crown Ether Phase-Transfer Catalysis: Model Ab Initio Calculations of Nucleophilic Fluorination. J. Mol. Catal. A Chem. 2012, 363, 489–494. [Google Scholar] [CrossRef]
- Silva, S.L.; Valle, M.S.; Pliego, J.R., Jr. Micro-Solvation and Counter Ion Effects on Ionic Reactions: Activation of Potassium Fluoride with 18-Crown-6 and Tert-Butanol in Aprotic Solvents. J. Mol. Liq. 2020, 319, 114211. [Google Scholar] [CrossRef]
- Silva, S.L.; Valle, M.S.; Pliego, J.R., Jr. Nucleophilic Fluorination with KF Catalyzed by 18-Crown-6 and Bulky Diols: A Theoretical and Experimental Study. J. Org. Chem. 2020, 85, 15457–15465. [Google Scholar] [CrossRef]
- Hong, C.M.; Whittaker, A.M.; Schultz, D.M. Nucleophilic Fluorination of Heteroaryl Chlorides and Aryl Triflates Enabled by Cooperative Catalysis. J. Org. Chem. 2021, 86, 3999–4006. [Google Scholar] [CrossRef]
- Shirakawa, S.; Yamamoto, K.; Kitamura, M.; Ooi, T.; Maruoka, K. Dramatic Rate Enhancement of Asymmetric Phase-Transfer-Catalyzed Alkylations. Angew. Chem. Int. Ed. 2005, 44, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, V.H.; Choi, W.; Lee, S.-S.; Lee, S.; Kim, D.W. Bis-tert-Alcohol-Functionalized Crown-6-Calix[4]arene: An Organic Promoter for Nucleophilic Fluorination. Chem. Eur. J. 2016, 22, 4515–4520. [Google Scholar] [CrossRef] [PubMed]
- Schettini, R.; Sicignano, M.; De Riccardis, F.; Izzo, I.; Della Sala, G. Macrocyclic Hosts in Asymmetric Phase-Transfer Catalyzed Reactions. Synthesis 2018, 50, 4777–4795. [Google Scholar] [CrossRef]
- Zhang, Z.; Shao, Y.; Tang, J.; Jiang, J.; Wang, L.; Li, S. Supramolecular Asymmetric Catalysis Mediated by Crown Ethers and Related Recognition Systems. Green Synth. Catal. 2021, 2, 156–164. [Google Scholar] [CrossRef]
- Aoki, S.; Sasaki, S.; Koga, K. Simple Chiral Crown Ethers Complexed with Potassium Tert-Butoxide as Efficient Catalysts for Asymmetric Michael Additions. Tetrahedron Lett. 1989, 30, 7229–7230. [Google Scholar] [CrossRef]
- Sicignano, M.; Schettini, R.; Sica, L.; Pierri, G.; De Riccardis, F.; Izzo, I.; Maity, B.; Minenkov, Y.; Cavallo, L.; Della Sala, G. Unprecedented Diastereoselective Arylogous Michael Addition of Unactivated Phthalides. Chem. Eur. J. 2019, 25, 7131–7141. [Google Scholar] [CrossRef]
- Della Sala, G.; Sicignano, M.; Schettini, R.; De Riccardis, F.; Cavallo, L.; Minenkov, Y.; Batisse, C.; Hanquet, G.; Leroux, F.; Izzo, I. Switchable Diastereoselectivity in the Fluoride-Promoted Vinylogous Mukaiyama–Michael Reaction of 2-[(Trimethylsilyl) Oxy] Furan Catalyzed by Crown Ethers. J. Org. Chem. 2017, 82, 6629–6637. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Hwang, I.S.; Lee, H.J.; Song, C.E. Biomimetic Catalytic Transformation of Toxic α-Oxoaldehydes to High-Value Chiral α-Hydroxythioesters Using Artificial Glyoxalase I. Nat. Commun. 2017, 8, 14877. [Google Scholar] [CrossRef]
- Rapi, Z.; Grün, A.; Nemcsok, T.; Hessz, D.; Kállay, M.; Kubinyi, M.; Keglevich, G.; Bako, P. Crown Ether Derived from D-Glucose as an Efficient Phase-Transfer Catalyst for the Enantioselective Michael Addition of Malonates to Enones. Tetrahedron Asymmetry 2016, 27, 960–972. [Google Scholar] [CrossRef]
- Orbán, I.; Bakó, P.; Rapi, Z. Carbohydrate-Based Azacrown Ethers in Asymmetric Syntheses. Chemistry 2021, 3, 550–577. [Google Scholar] [CrossRef]
- Zhao, Y.; Truhlar, D.G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other function. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar] [CrossRef]
- Hay, P.J.; Wadt, W.R. Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg. J. Chem. Phys. 1985, 82, 270–283. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16; Gaussian Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).