Chemo-Radio-Immunotherapy for NSCLC III: ESR/ATS Thresholds for DLCO Correlate with Radiation Dosimetry and Pneumonitis Rate
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Patients
2.2. Sequential Chemo-Radio(-Immuno)Therapy
2.3. Clinical Endpoints
2.3.1. Pneumonitis
2.3.2. DLCO Thresholds
Pre-Treatment DLCO Threshold of 60%
Post-Treatment DLCO Decline ≥10%
2.4. Modeling DLCO as a Function of V65–45%
2.5. Statistics
3. Results
3.1. Patients
3.2. Sequential Chemo-Radio(-Immuno)-Therapy
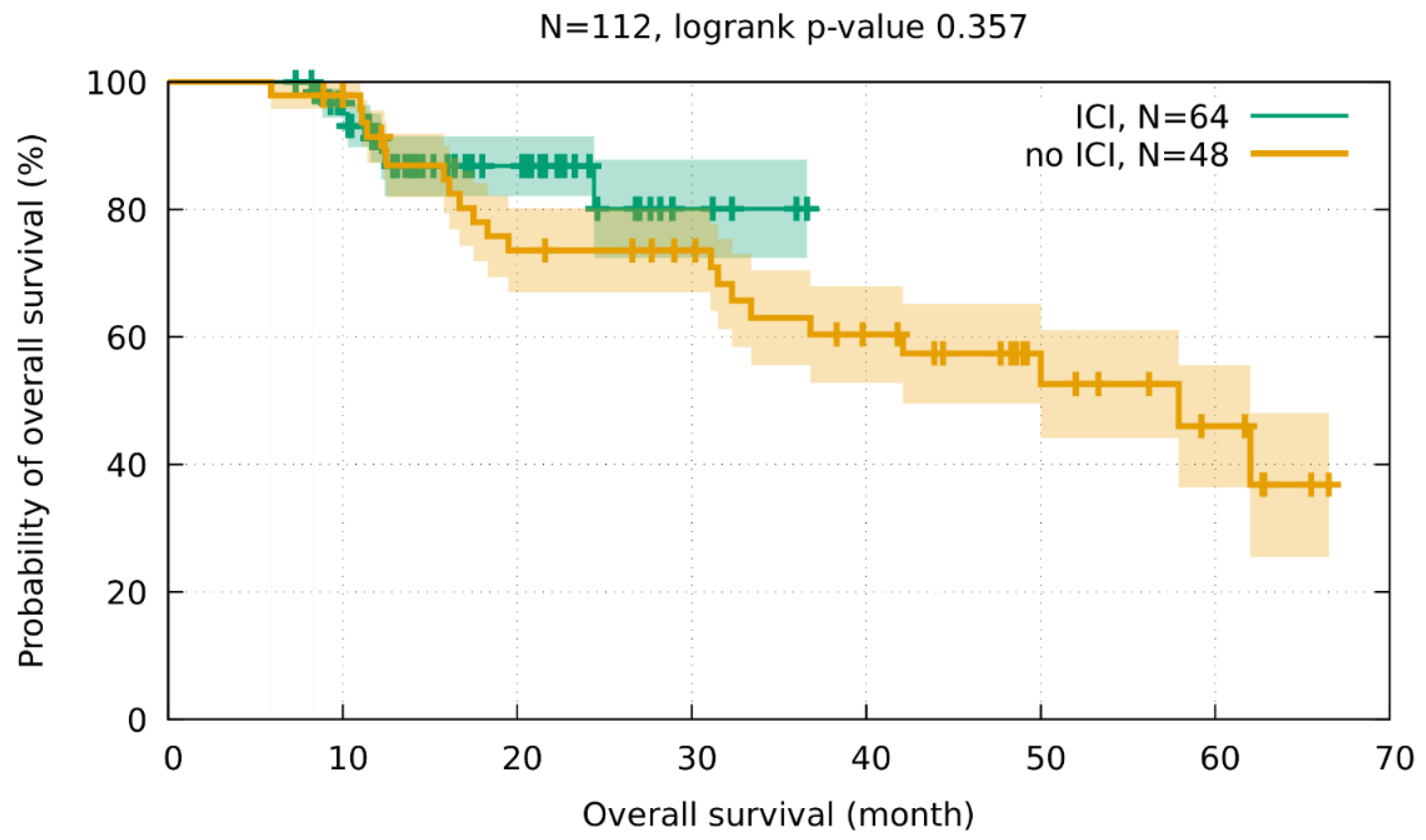
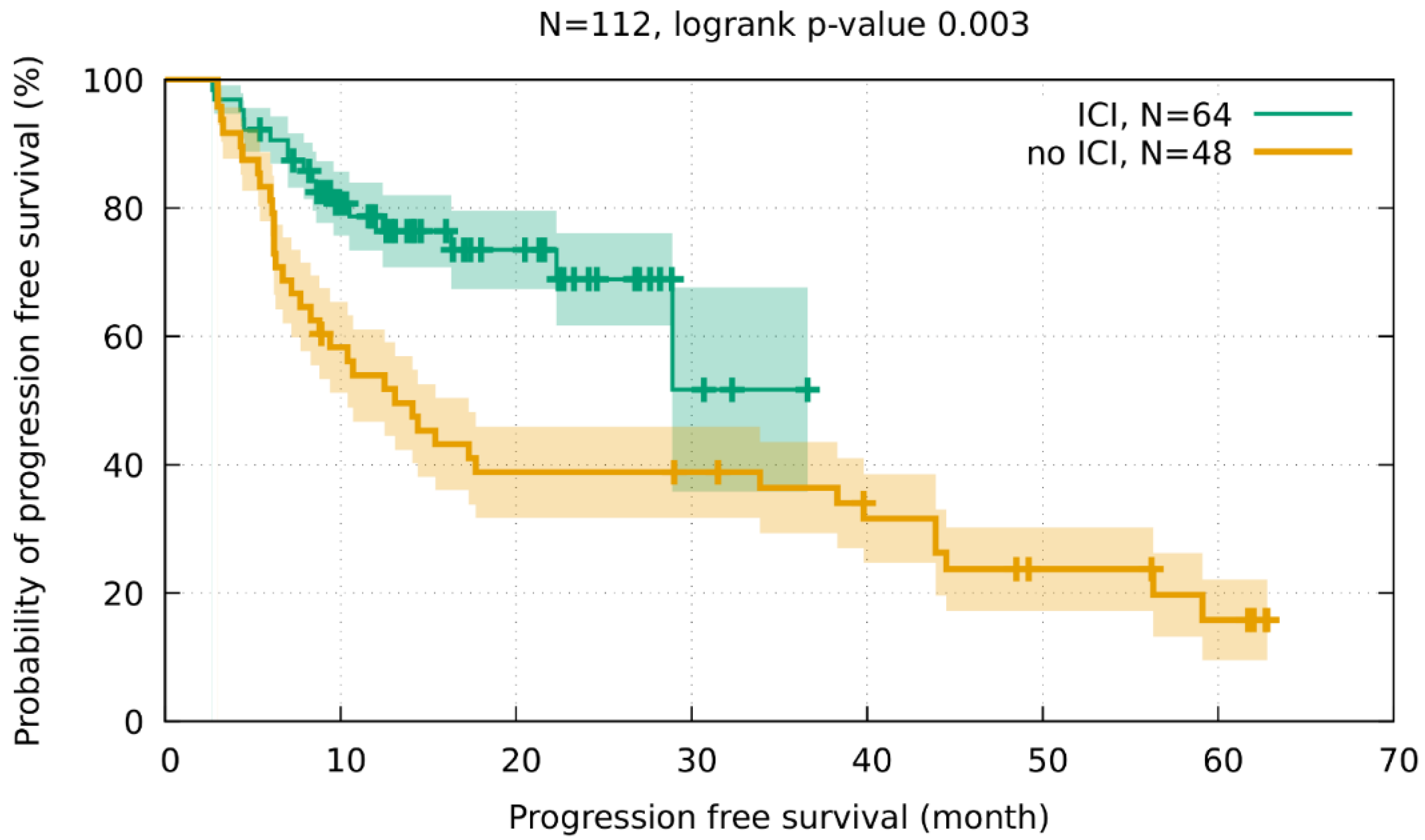
3.3. Clinical Outcome and Pulmonary Toxicity
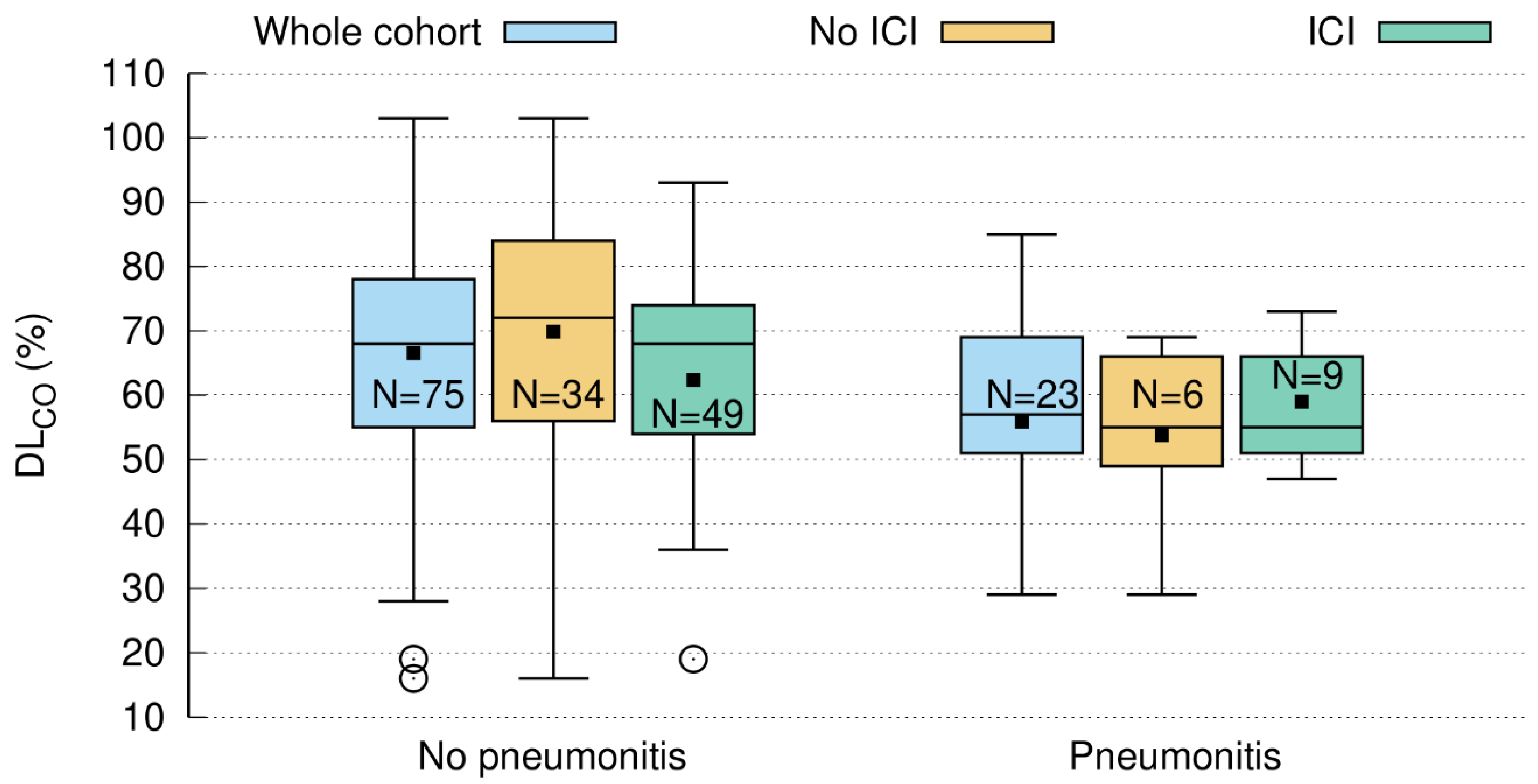
3.4. Baseline DLCO < 60% Predicts Pneumonitis
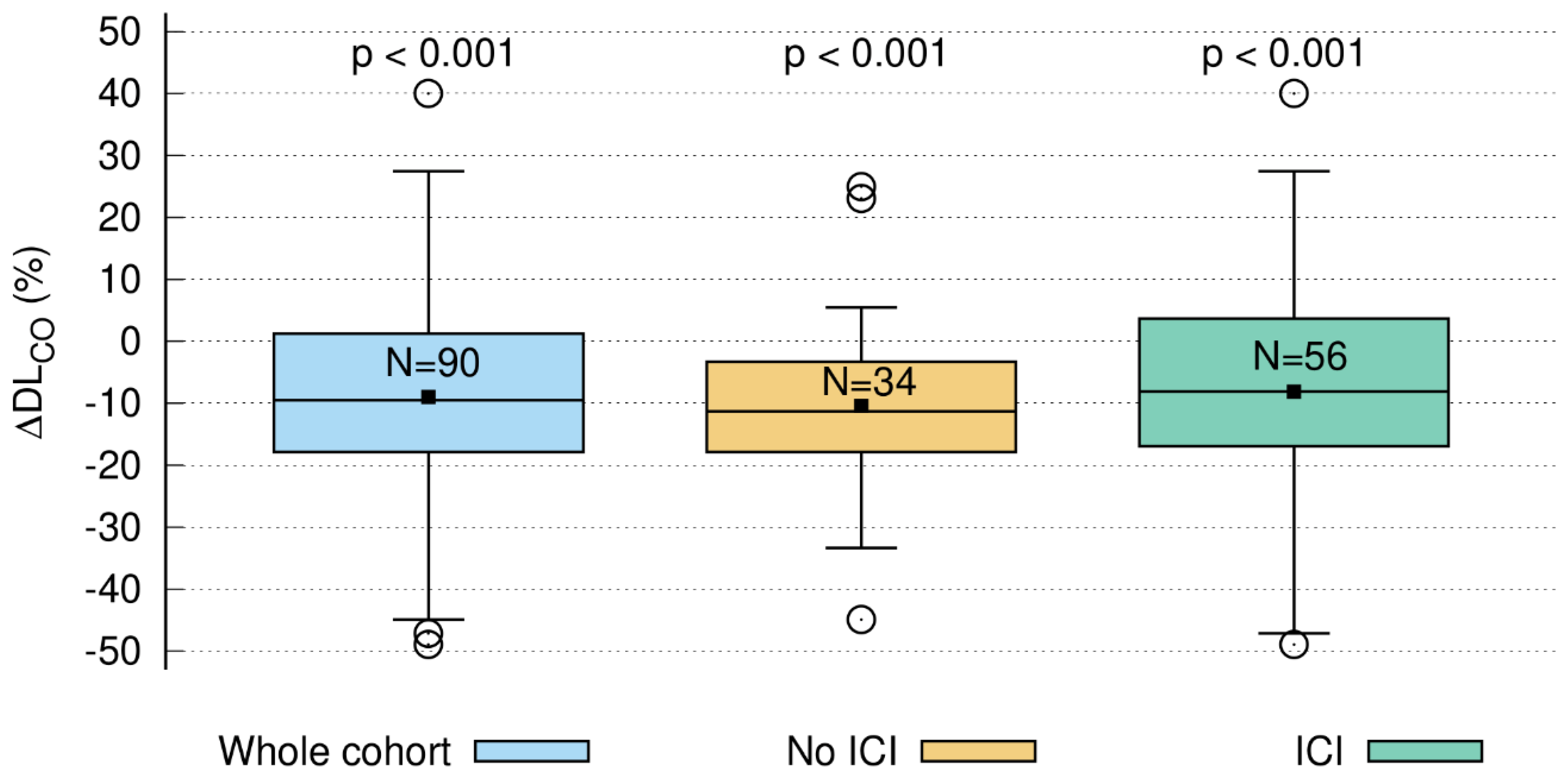
3.5. Short-Term DLCO Decline > 10%
3.5.1. Time Course of DLCO Decline
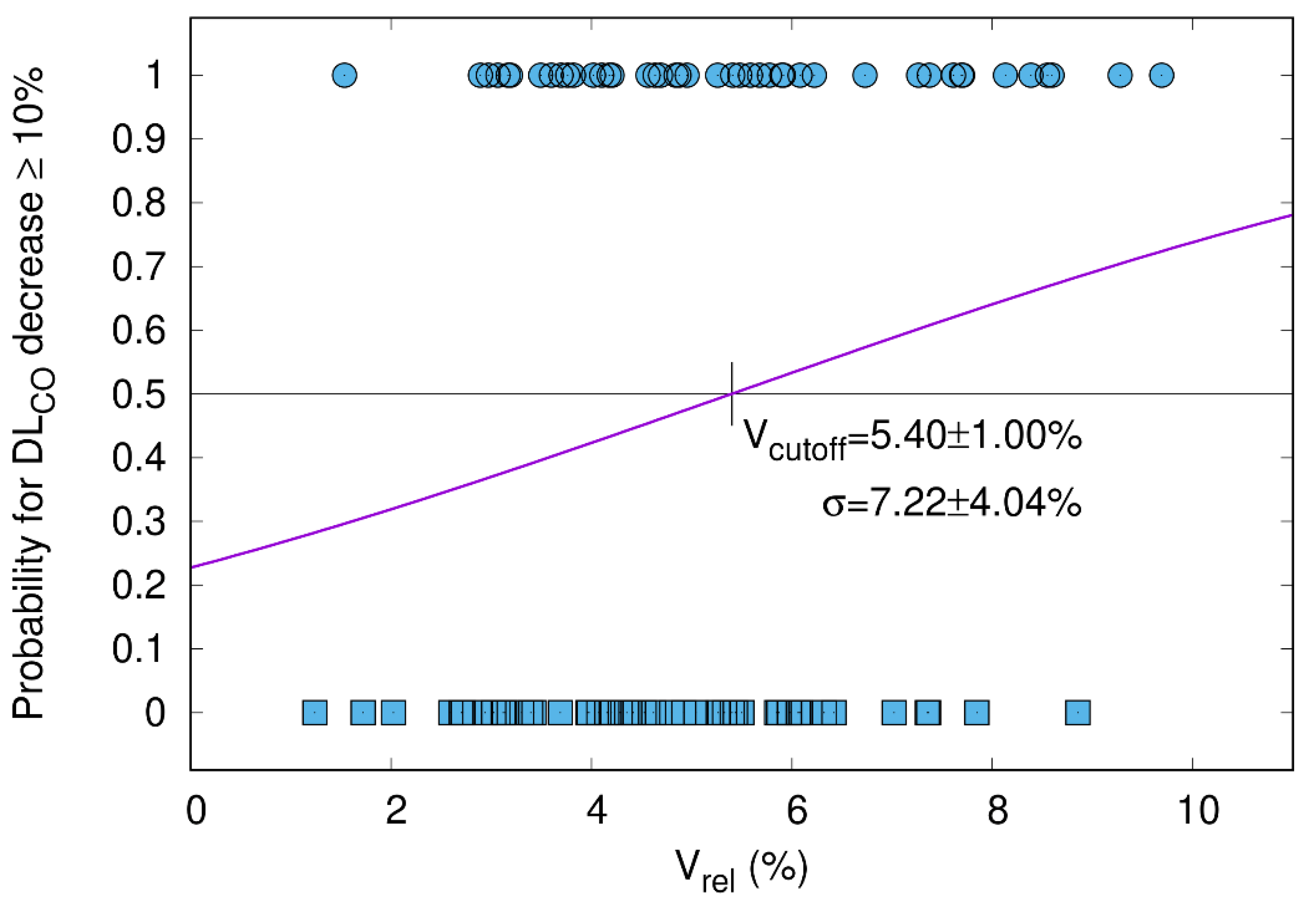
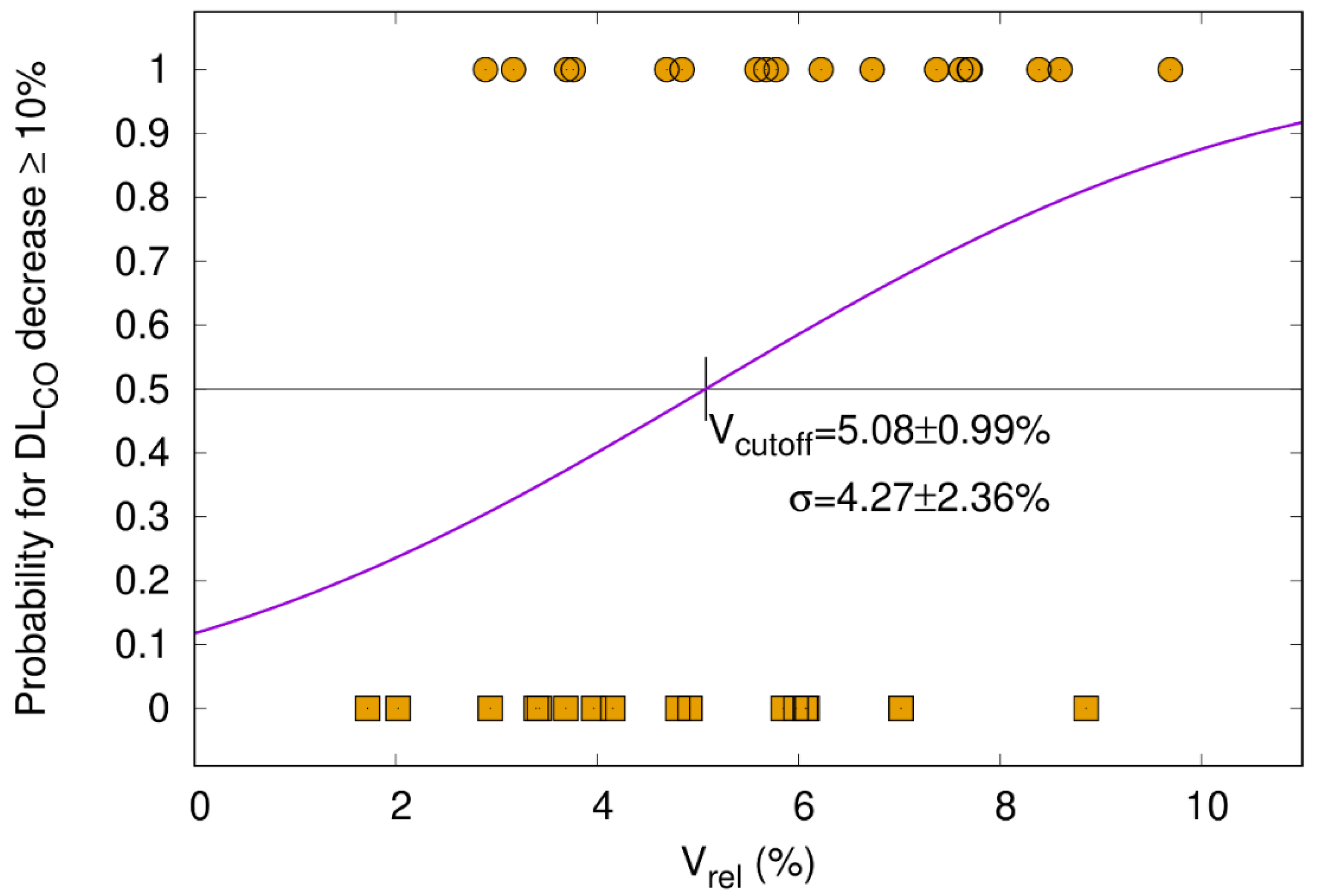
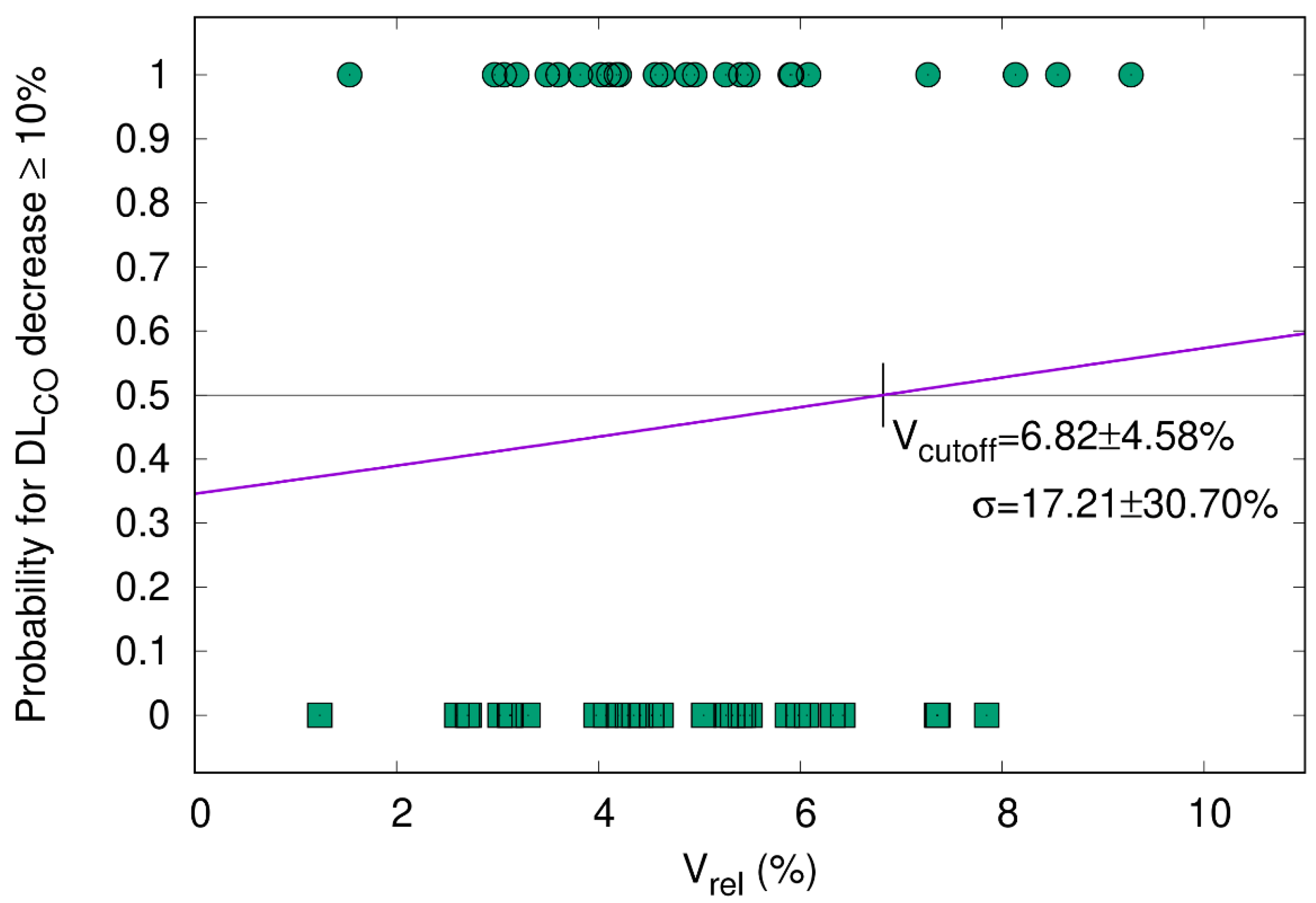
3.5.2. DLCO Decline ≥ 10% Correlates with V65–45% > 5.1%
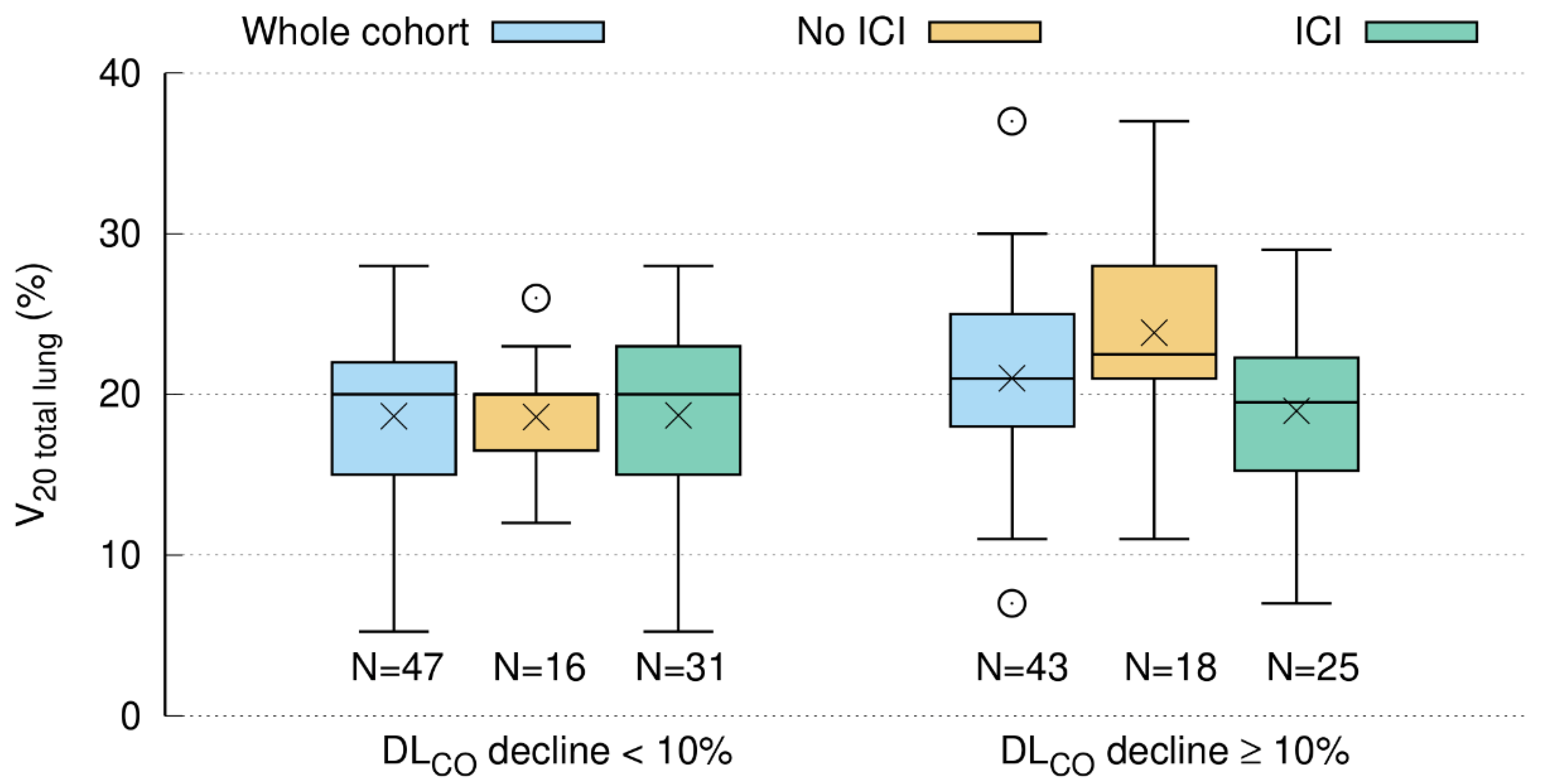
3.5.3. DLCO Decline Is Related to V20 Total Lung
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATS | American Thoracic Society |
| CDF | cumulative distribution function |
| CRT | chemoradiotherapy |
| cCRT | concomitant chemoradiotherapy |
| DART | dose differentiated accelerated radiotherapy |
| DLCO | carbon monoxide diffusing capacity |
| Dmax | maximum dose |
| EQD2 | biologically equivalent dose in 2 Gy fractions |
| ERS | European Respiratory Society |
| FEV1 | forced expiratory volume in one second |
| GTV | gross tumor volume |
| ICI | immune checkpoint inhibitor |
| IMRT | intensity-modulated radiotherapy |
| LA-NSCLC | locally advanced non-small cell lung cancer |
| MED | mean esophageal dose |
| MLD | mean lung dose |
| OS | overall survival |
| PF | pulmonary function |
| PFS | progression-free survival |
| RT | radiotherapy |
| SoC | standard of care |
| sCRT | sequential chemoradiotherapy |
| V25 Heart | heart volume receiving 25 Gy or more |
| V20 Total Lung | lung volume receiving 20 Gy or more |
| V65–45% | volume receiving between 45% and 65% of the prescribed total radiation dose |
| VMAT | volumetric arc therapy |
Appendix A
| Patients n = 112 | ||||
|---|---|---|---|---|
| sCRT (n = 48) | sCRT + ICI (n = 64) | p-Value | ||
| Age | Median | 65.5 | 68.0 | 0.147 |
| Range | 46–82 | 29–88.5 | ||
| Sex | Male | 34 (71%) | 41 (64%) | 0.453 |
| Female | 14 (29%) | 23 (36%) | ||
| Weight loss | <5% | 42 (88%) | 60 (94%) | 0.253 |
| >5% | 6 (13%) | 4 (6%) | ||
| ECOG | 0–1 | 46 (96%) | 62 (97%) | 0.474 |
| 2 | 2 (4%) | 2 (3%) | ||
| Smoking status | Ex | 28 (58%) | 35 (55%) | 0.888 |
| Current | 15 (31%) | 21 (33%) | ||
| Never | 5 (10%) | 6 (9%) | ||
| Unknown | 0 (0%) | 2 (3%) | ||
| Histology | NSCLC | 48 (100%) | 64 (100%) | n.a. |
| Unknown | 0 (0%) | 0 (0%) | ||
| UICC stage | III | 48 (100%) | 64 (100%) | n.a. |
| Unknown | 0 (0%) | 0 (0%) | ||
| COPD grade | 0 | 27 (56%) | 31 (49%) | 0.709 |
| 1 | 4 (8%) | 6 (9%) | ||
| 2 | 6 (13%) | 18 (28%) | ||
| 3 | 9 (19%) | 6 (9%) | ||
| 4 | 2 (4%) | 3 (5%) | ||
| Treatment n = 112 | ||||
|---|---|---|---|---|
| sCRT (n = 48) | sCRT + ICI (n = 64) | p-Value | ||
| GTV (mL) | Median | 15.1 | 43.0 | 0.023 |
| Range | 1.0–132.0 | 0.2–408.5 | ||
| Tumor location (n) | Peripheral | 28 (58%) | 25 (39%) | 0.044 |
| Central | 20 (42%) | 39 (61%) | ||
| RT technique (n) | IMRT | 43 (90%) | 9 (14%) | <0.001 |
| VMAT | 5 (10%) | 55 (86%) | ||
| ICI | 0 (0%) | 64 (100%) | n.a. | |
| MLD (Gy) | Median | 12.1 | 12.0 | 0.130 |
| Range | 7–17.9 | 4.0–14.8 | ||
| V20 total lung (%) | Median | 21 | 20 | 0.052 |
| Range | 11.0-37.0 | 5.5–29.0 | ||
| EQD2 tumor (Gy) | Median | 77.6 | 71.5 | <0.001 |
| Range | 58.3–88.2 | 39–77.6 | ||
| EQD2 lymph nodes (Gy) | Median | 60 | 57.3 | 0.003 |
| Range | 0–60.0 | 0–60.0 | ||
| Pearson Correlation Coefficient | Raw p-Value | Corrected p-Value | |
|---|---|---|---|
| Age | 0.099 | 0.290 | n.s. |
| Sex | 0.022 | 0.450 | n.s. |
| Weight loss | −0.274 | 0.058 | n.s. |
| ECOG | −0.204 | 0.123 | n.s. |
| Smoking status | 0.038 | 0.416 | n.s. |
| Histology | n.a. | ||
| UICC stage | n.a. | ||
| COPD | 0.038 | 0.415 | n.s. |
| GTV | 0.060 | 0.367 | n.s. |
| Tumor location | −0.174 | 0.163 | n.s. |
| Radiation technique | −0.122 | 0.246 | n.s. |
| ICI | n.a. | ||
| MLD | 0.394 | 0.010 | n.s. |
| V20 total lung | 0.466 | 0.003 | 0.042 |
| EQD2 tumor | 0.224 | 0.102 | n.s. |
| EQD2 lymph nodes | 0.257 | 0.071 | n.s. |
| V65–45% | 0.354 | 0.020 | n.s. |
References
- Malvezzi, M.; Carioli, G.; Bertuccio, P.; Boffetta, P.; Levi, F.; La Vecchia, C.; Negri, E. European cancer mortality predictions for the year 2017, with focus on lung cancer. Ann. Oncol. 2017, 28, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T.; Chiappori, A.; Lee, K.H.; De Wit, M.; et al. Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N. Engl. J. Med. 2018, 379, 2342–2350. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; De Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef]
- Spigel, D.R.; Faivre-Finn, C.; Gray, J.E.; Vicente, D.; Planchard, D.; Paz-Ares, L.; Vansteenkiste, J.F.; Garassino, M.C.; Hui, R.; Quantin, X.; et al. Five-Year Survival Outcomes from the PACIFIC Trial: Durvalumab After Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2022, 40, 1301–1311. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, D.S.; Wood, D.E.; Aisner, D.L.; Akerley, W.; Bauman, J.R.; Bharat, A.; Bruno, D.S.; Chang, J.Y.; Chirieac, L.R.; D'Amico, T.A.; et al. Non-Small Cell Lung Cancer, Version 2.2021 Featured Updates to the NCCN Guidelines. J. Natl. Compr. Cancer Netw. 2021, 19, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Furuse, K.; Fukuoka, M.; Kawahara, M.; Nishikawa, H.; Takada, Y.; Kudoh, S.; Katagami, N.; Ariyoshi, Y. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with mitomycin, vindesine, and cisplatin in unresectable stage III non-small-cell lung cancer. J. Clin. Oncol. 1999, 17, 2692–2699. [Google Scholar] [CrossRef]
- Curran, W.J.; Paulus, R.; Langer, C.J.; Komaki, R.; Lee, J.S.; Hauser, S.; Movsas, B.; Wasserman, T.; Rosenthal, S.A.; Gore, E.; et al. Sequential vs. concurrent chemoradiation for stage III non-small cell lung cancer: Randomized phase III trial RTOG 9410. J. Natl. Cancer Inst. 2011, 103, 1452–1460. [Google Scholar] [CrossRef]
- Nishino, M.; Giobbie-Hurder, A.; Hatabu, H.; Ramaiya, N.H.; Hodi, S. Incidence of Programmed Cell Death 1 Inhibitor-Related Pneumonitis in Patients with Advanced Cancer: A Systematic Review and Meta-analysis. JAMA Oncol. 2016, 2, 1607–1616. [Google Scholar] [CrossRef]
- Suresh, K.; Voong, K.R.; Shankar, B.; Forde, P.M.; Ettinger, D.S.; Marrone, K.A.; Kelly, R.J.; Hann, C.L.; Levy, B.; Feliciano, J.L.; et al. Pneumonitis in Non-Small Cell Lung Cancer Patients Receiving Immune Checkpoint Immunotherapy: Incidence and Risk Factors. J. Thorac. Oncol. 2018, 13, 1930–1939. [Google Scholar] [CrossRef]
- Fukihara, J.; Sakamoto, K.; Koyama, J.; Ito, T.; Iwano, S.; Morise, M.; Ogawa, M.; Kondoh, Y.; Kimura, T.; Hashimoto, N.; et al. Prognostic Impact and Risk Factors of Immune-Related Pneumonitis in Patients with Non-Small-Cell Lung Cancer Who Received Programmed Death 1 Inhibitors. Clin. Lung Cancer 2019, 20, 442–450.e4. [Google Scholar] [CrossRef]
- Landman, Y.; Jacobi, O.; Kurman, N.; Yariv, O.; Peretz, I.; Rotem, O.; Dudnik, E.; Zer, A.; Allen, A.M. Durvalumab after concurrent chemotherapy and high-dose radiotherapy for locally advanced non-small cell lung cancer. Oncoimmunology 2021, 10, 1959979. [Google Scholar] [CrossRef]
- Wass, R.; Hochmair, M.; Kaiser, B.; Grambozov, B.; Feurstein, P.; Weiß, G.; Moosbrugger, R.; Sedlmayer, F.; Lamprecht, B.; Studnicka, M.; et al. Durvalumab after Sequential High Dose Chemoradiotherapy versus Standard of Care (SoC) for Stage III NSCLC: A Bi-Centric Trospective Comparison Focusing on Pulmonary Toxicity. Cancers 2022, 14, 3226. [Google Scholar] [CrossRef]
- Liptay, M.J.; Basu, S.; Hoaglin, M.C.; Freedman, N.; Faber, L.P.; Warren, W.H.; Hammoud, Z.T.; Kim, A.W. Diffusion Lung Capacity for Carbon Monoxide (DLCO) Is an Independent Prognostic Factor for Long-Term Survival After Curative Lung Resection for Cancer. J. Surg. Oncol. 2009, 100, 703–707. [Google Scholar] [CrossRef]
- Ferguson, M.K.; Reeder, L.B.; Mick, R. Optimizing Selection of Patients for Major Lung Resection. J. Thorac. Cardiov. Sur. 1995, 109, 275–283. [Google Scholar] [CrossRef]
- Pellegrino, R.; Viegi, G.; Brusasco, V.; Crapo, R.O.; Burgos, F.; Casaburi, R.; Coates, A.; Van Der Grinten, C.P.M.; Gustafsson, P.; Hankinson, J.; et al. Interpretative strategies for lung function tests. Eur. Respir. J. 2005, 26, 948–968. [Google Scholar] [CrossRef]
- Stana, M.; Grambozov, B.; Gaisberger, C.; Karner, J.; Ruznic, E.; Berchtold, J.; Zellinger, B.; Moosbrugger, R.; Studnicka, M.; Fastner, G.; et al. Carbon Monoxide Diffusing Capacity (DLCO) Correlates with CT Morphology after Chemo-Radio-Immunotherapy for Non-Small Cell Lung Cancer Stage III. Diagnostics 2022, 12, 1027. [Google Scholar] [CrossRef] [PubMed]
- Grambozov, B.; Wolf, F.; Kaiser, J.; Wass, R.; Fastner, G.; Gaisberger, C.; Rettenbacher, L.; Studnicka, M.; Pirich, C.; Sedlmayer, F.; et al. Pulmonary function decreases moderately after accelerated high-dose irradiation for stage III non-small cell lung cancer. Thorac. Cancer 2020, 11, 369–378. [Google Scholar] [CrossRef]
- Wurstbauer, K.; Deutschmann, H.; Dagn, K.; Kopp, P.; Zehentmayr, F.; Lamprecht, B.; Porsch, P.; Wegleitner, B.; Studnicka, M.; Sedlmayer, F. DART-bid (Dose-differentiated accelerated radiation therapy, 1.8 Gy twice daily)—A novel approach for non-resected NSCLC: Final results of a prospective study, correlating radiation dose to tumor volume. Radiat. Oncol. 2013, 8, 49. [Google Scholar] [CrossRef]
- Marks, L.B.; Bentzen, S.M.; Deasy, J.O.; Kong, F.-M.; Bradley, J.D.; Vogelius, I.S.; El Naqa, I.; Hubbs, J.L.; Lebesque, J.V.; Timmerman, R.D.; et al. Radiation dose-volume effects in the lung. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S70–S76. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.; Arish, N.; Rokach, A.; Zaltzman, Y.; Marcus, E.L. The effect of body position on pulmonary function: A systematic review. BMC Pulm. Med. 2018, 18, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.L.; Brusasco, V.; Burgos, F.; Cooper, B.G.; Jensen, R.; Kendrick, A.; MacIntyre, N.R.; Thompson, B.R.; Wanger, J. Executive Summary: 2017 ERS/ATS standards for single-breath carbon monoxide uptake in the lung. Eur. Respir. J. 2017, 49, 16E0016. [Google Scholar] [CrossRef] [PubMed]
- Hathaway, E.H.; Tashkin, D.P.; Simmons, M.S. Intraindividual Variability in Serial Measurements of Dlco and Alveolar Volume over One Year in 8 Healthy-Subjects Using 3 Independent Measuring Systems. Am. Rev. Respir. Dis. 1989, 140, 1818–1822. [Google Scholar] [CrossRef]
- Fournel, P.; Robinet, G.; Thomas, P.; Souquet, P.J.; Léna, H.; Vergnenégre, A.; Delhoume, J.Y.; Le Treut, J.; Silvani, J.A.; Dansin, E.; et al. Randomized phase III trial of sequential chemoradiotherapy compared with concurrent chemoradiotherapy in locally advanced non-small-cell lung cancer: Groupe Lyon-Saint-Etienne d'Oncologie Thoracique-Groupe Francais de Pneumo-Cancerologie NPC 95-01 Study. J. Clin. Oncol. 2005, 23, 5910–5917. [Google Scholar] [CrossRef]
- Zatloukal, P.; Petruzelka, L.; Zemanova, M.; Havel, L.; Janku, F.; Judas, L.; Kubik, A.; Krepela, E.; Fiala, P.; Pecen, L. Concurrent versus sequential chemoradiotherapy with cisplatin and vinorelbine in locally advanced non-small cell lung cancer: A randomized study. Lung Cancer 2004, 46, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Bradley, J.D.; Paulus, R.; Komaki, R.; Masters, G.; Blumenschein, G.; Schild, S.; Bogart, J.; Hu, C.; Forster, K.; Magliocco, A.; et al. Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): A randomised, two-by-two factorial phase 3 study. Lancet Oncol. 2015, 16, 187–199. [Google Scholar] [PubMed]
- Garassino, M.; Mazieres, J.; Reck, M.; Chouaid, C.; Bischoff, H.; Reinmuth, N.; Cove-Smith, L.; Mansy, T.; Cortinovis, D.; Migliorino, M.; et al. Safety and efficacy outcomes with durvalumab after sequential chemoradiotherapy (sCRT) in stage III, unresectable NSCLC (PACIFIC-6). Ann. Oncol. 2022, 33, S81–S82. [Google Scholar] [CrossRef]
- Aupérin, A.; Le Péchoux, C.; Rolland, E.; Curran, W.J.; Furuse, K.; Fournel, P.; Belderbos, J.; Clamon, G.; Ulutin, H.C.; Paulus, R.; et al. Meta-analysis of concomitant versus sequential radiochemotherapy in locally advanced non-small-cell lung cancer. J. Clin. Oncol. 2010, 28, 2181–2190. [Google Scholar] [CrossRef]
- Kuang, Y.; Pierce, C.M.; Chang, H.C.; Sosinsky, A.Z.; Deitz, A.C.; Keller, S.M.; Samkari, A.; Uyei, J. Chemoradiation-induced pneumonitis in patients with unresectable stage III non-small cell lung cancer: A systematic literature review and meta-analysis. Lung Cancer 2022, 174, 174–185. [Google Scholar] [CrossRef]
- Hughes, J.M.B.; Bates, D.V. Historical review: The carbon monoxide diffusing capacity (DLCO) and its membrane (D-M) and red cell (Theta center dot Vc) components. Resp. Physiol. Neurobi. 2003, 138, 115–142. [Google Scholar] [CrossRef]
- Wang, J.; Olak, J.; Ferguson, M.K. Diffusing capacity predicts operative mortality but not long-term survival after resection for lung cancer. J. Thorac. Cardiov. Sur. 1999, 117, 581–587. [Google Scholar] [CrossRef]
- Celli, B.; MacNee, W.; Agusti, A.; Anzueto, A.; Berg, B.; Buist, A.; Calverley, P.; Chavannes, N.; Dillard, T.; Fahy, B.; et al. Standards for the diagnosis and treatment of patients with COPD: A summary of the ATS/ERS position paper. Eur. Respir. J. 2004, 23, 932–946. [Google Scholar] [CrossRef]
- Brennan, D.; Schubert, L.; Diot, Q.; Castillo, R.; Castillo, E.; Guerrero, T.; Martel, M.K.; Linderman, D.; Gaspar, L.E.; Miften, M.; et al. Clinical Validation of 4-Dimensional Computed Tomography Ventilation with Pulmonary Function Test Data. Int. J. Radiat. Oncol. 2015, 92, 423–429. [Google Scholar] [CrossRef]
- Videtic, G.M.; Stitt, L.W.; Ash, R.B.; Truong, P.T.; Dar, A.; Yu, E.W.; Whiston, F. Impaired diffusion capacity predicts for decreased treatment tolerance and survival in limited stage small cell lung cancer patients treated with concurrent chemoradiation. Lung Cancer 2004, 43, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Zhang, J.; Zhou, S.; Hubbs, J.L.; Foltz, R.J.; Hollis, D.R.; Light, K.L.; Wong, T.Z.; Kelsey, C.R.; Marks, L.B. Association between RT-induced changes in lung tissue density and global lung function. Int. J. Radiat. Oncol. Biol. Phys. 2009, 74, 781–789. [Google Scholar] [CrossRef]
- Borst, G.R.; De Jaeger, K.; Belderbos, J.S.; Burgers, S.A.; Lebesque, J.V. Pulmonary function changes after radiotherapy in non-small-cell lung cancer patients with long-term disease-free survival. Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 639–644. [Google Scholar] [CrossRef] [PubMed]
- Enache, I.; Noel, G.; Jeung, M.Y.; Meyer, N.; Oswald-Mammosser, M.; Pistea, C.; Jung, G.-M.; Mennecier, B.; Quoix, E.; Charloux, A. Impact of 3D conformal radiotherapy on lung function of patients with lung cancer: A prospective study. Respiration 2013, 86, 100–108. [Google Scholar] [CrossRef]
- Verbanck, S.; Van Parijs, H.; Schuermans, D.; Vinh-Hung, V.; Storme, G.; Fontaine, C.; De Ridder, M.; Verellen, D.; Vanderhelst, E.; Hanon, S. Lung Restriction in Patients with Breast Cancer After Hypofractionated and Conventional Radiation Therapy: A 10-Year Follow-up. Int. J. Radiat. Oncol. 2022, 113, 561–569. [Google Scholar] [CrossRef] [PubMed]
| Patients n = 112 | ||
|---|---|---|
| Age (years) | Median | 66 |
| Range | 29–85 | |
| Sex | Male | 75 (67%) |
| Female | 37 (33%) | |
| Weight loss (%) | >5% | 9 (8%) |
| <5% | 103 (92%) | |
| ECOG | 0–1 | 108 (96%) |
| 2 | 4 (4%) | |
| Smoking status | Ex | 63 (56%) |
| Current | 36 (32%) | |
| Never | 11 (10%) | |
| Unknown | 2 (2%) | |
| Histology | NSCLC | 112 (100%) |
| Unknown | 0 (0%) | |
| UICC | III | 112 (100%) |
| COPD grade | 0 | 58 (52%) |
| 1 | 10 (9%) | |
| 2 | 24 (21%) | |
| 3 | 15 (13%) | |
| 4 | 5 (5%) | |
| Unknown | 0 (0%) | |
| Treatment n = 112 | ||
|---|---|---|
| GTV (mL) | Median | 29.4 |
| Range | 0.3–408 | |
| Tumor location (n) | Peripheral | 53 (47%) |
| Central | 59 (53%) | |
| RT technique (n) | IMRT | 52 (46%) |
| VMAT | 60 (54%) | |
| ICI | Yes | 64 (57%) |
| No | 48 (43%) | |
| MLD (Gy) | Median | 12.0 |
| Range | 4.0–18.0 | |
| V20 Total lung (%) | Median | 20.0 |
| Range | 5.3–37.0 | |
| EQD2 Tumor (Gy) | Median | 72.3 |
| Range | 39.0–88.2 | |
| EQD2 Lymphnodes (Gy) | Median | 57.3 |
| Range | 0–60.0 | |
| Pneumonitis | |||||
|---|---|---|---|---|---|
| Grade 2 | Grade 3 | Grade 4 | Grade 5 | p-Value | |
| Whole cohort (n = 112) | 13 (12%) | 3 (3%) | 0 (0%) | 0 (0%) | n.a. |
| No ICI (n = 48) | 4 (8%) | 2 (4%) | 0 (0%) | 0 (0%) | 0.691 |
| ICI (n = 64) | 9 (14%) | 1 (2%) | 0 (0%) | 0 (0%) | |
| Pneumonitis in Dependence of Baseline DLCO | |||
|---|---|---|---|
| Baseline DLCO | |||
| <60% | >60% | n.a. | |
| Pneumonitis cases | 9 (22%) | 6 (11%) | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stana, M.; Grambozov, B.; Karner, J.; Gollner, I.; Gaisberger, C.; Ruznic, E.; Zellinger, B.; Moosbrugger, R.; Studnicka, M.; Fastner, G.; et al. Chemo-Radio-Immunotherapy for NSCLC III: ESR/ATS Thresholds for DLCO Correlate with Radiation Dosimetry and Pneumonitis Rate. Cancers 2023, 15, 1966. https://doi.org/10.3390/cancers15071966
Stana M, Grambozov B, Karner J, Gollner I, Gaisberger C, Ruznic E, Zellinger B, Moosbrugger R, Studnicka M, Fastner G, et al. Chemo-Radio-Immunotherapy for NSCLC III: ESR/ATS Thresholds for DLCO Correlate with Radiation Dosimetry and Pneumonitis Rate. Cancers. 2023; 15(7):1966. https://doi.org/10.3390/cancers15071966
Chicago/Turabian StyleStana, Markus, Brane Grambozov, Josef Karner, Isabella Gollner, Christoph Gaisberger, Elvis Ruznic, Barbara Zellinger, Raphaela Moosbrugger, Michael Studnicka, Gerd Fastner, and et al. 2023. "Chemo-Radio-Immunotherapy for NSCLC III: ESR/ATS Thresholds for DLCO Correlate with Radiation Dosimetry and Pneumonitis Rate" Cancers 15, no. 7: 1966. https://doi.org/10.3390/cancers15071966
APA StyleStana, M., Grambozov, B., Karner, J., Gollner, I., Gaisberger, C., Ruznic, E., Zellinger, B., Moosbrugger, R., Studnicka, M., Fastner, G., Sedlmayer, F., & Zehentmayr, F. (2023). Chemo-Radio-Immunotherapy for NSCLC III: ESR/ATS Thresholds for DLCO Correlate with Radiation Dosimetry and Pneumonitis Rate. Cancers, 15(7), 1966. https://doi.org/10.3390/cancers15071966








