TP53 and the Ultimate Biological Optimization Steps of Curative Radiation Oncology
Abstract
Simple Summary
Abstract
1. Introduction
2. TP53 and Cell Survival and Apoptosis at Low and High Doses and LETs
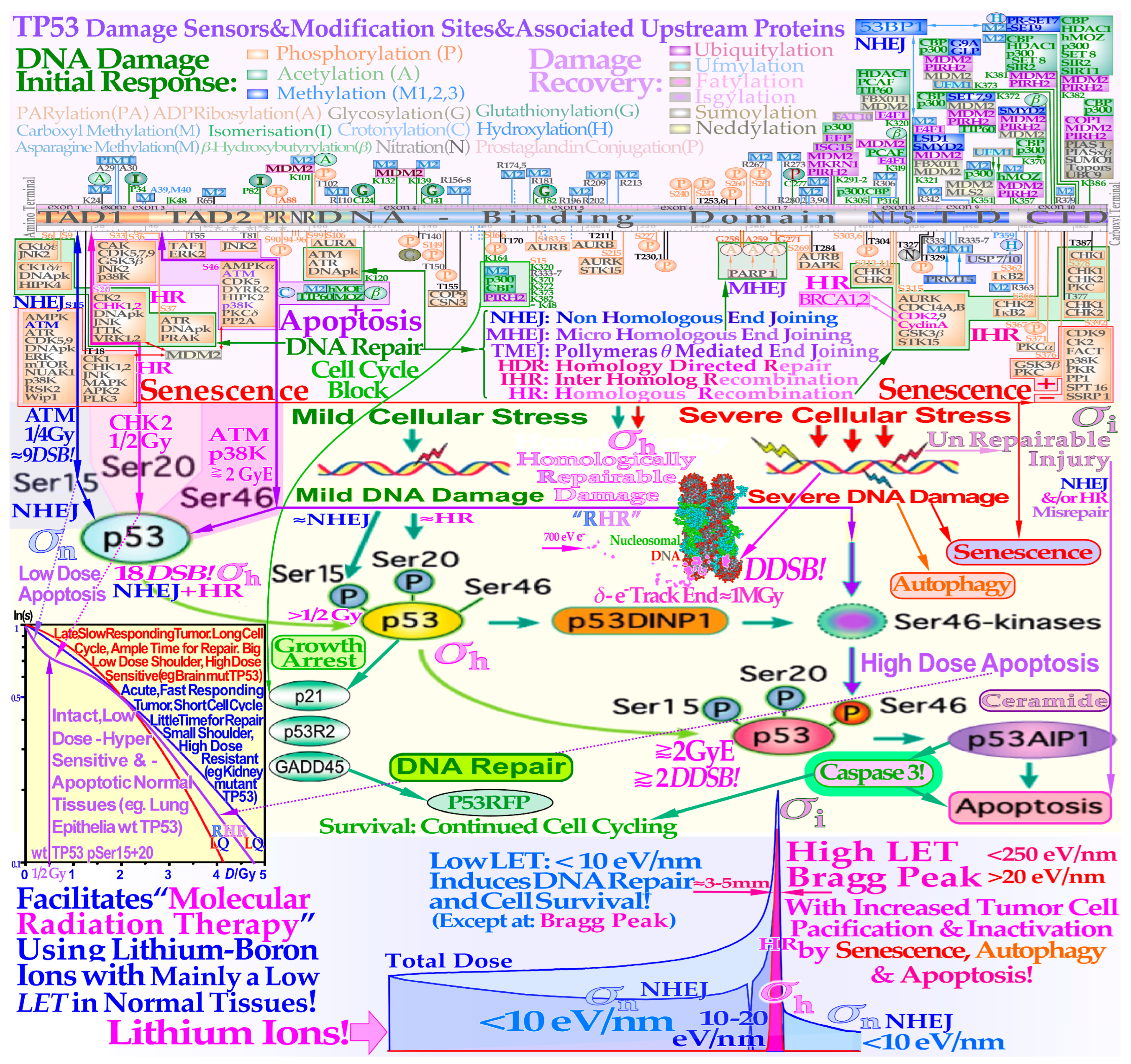
2.1. TP53 Damage Response
2.2. Cell Survival
2.3. Apoptosis Induction
2.4. Reactivation of Mutant TP53
2.5. The Fractionation Window
2.6. Secondary Cancer Induction
2.7. Simplistic Clinical Example
3. Influence of Microdosimetric Beam Characteristics on the Dose–Response Relation of Tumors and Normal Tissues
3.1. The Dose–Response Relation
3.2. The Dose–Response Steepness
3.3. Microdosimetric Heterogeneity Effects on the Dose–Response
3.4. Treatment Optimization
3.5. Optimal Use of Low-LET Beams
4. Consideration of Low-Dose Hypersensitivity and Apoptosis and Photons, Electrons, and Light Ions in Radiation Therapy Optimization
- The peak absorbed dose to critical normal tissues with adverse reactions, when quasi-uniformly irradiated (organs at risk), should preferably be in the range of 1.8–2.3 Gy/Fraction and of the lowest possible LET and biological effectiveness (Figure 5). Interestingly, this is the dose and LET range that maximizes the LDHS-related normal tissue tolerance with wt TP53, as seen in Figure 1, Figure 2, Figure 4 and Figure 6 [1,2,29,46,54]. A full minimization of the total risk for complications would naturally be preferred or preferably a full so-called P++ optimization strategy approach combining 1. here with 2. and 4. below [60].
- In order to make the treatment as curative as possible, it is desirable that the mean dose to the tumor (internal target volume [36]) is as high as possible to ensure a true complication-free cure (P+) and perfect clonogenic tumor cell eradication. Interestingly, this can be achieved quite accurately today via advanced biologically optimized intensity-modulated radiation therapy from a few inversely planned beam directions [38,50,54,60]. This will work well even for intact TP53 and ATM pathway tumors (Figure 6 and [2] (Figure 7)) since a simple LQ-type calculation may be far from optimal.
- To further minimize normal tissue damage as far as possible, it is desirable to introduce an optimal weekly dose fractionation schedule where the DNA repair of normal tissues is really taken into account to minimize their injury. Up to about 50% higher tumor doses should optimally be delivered Monday morning, Wednesday midday, Friday evening, and the last evening of treatment, to use the weekend and end of therapy for maximal normal tissue recovery (see the dashed line in the Graphical Abstract, [1] (Figure 21) and [61]) and preferably still staying below the 2.3 Gy/Fr to organs at risk. This will especially optimize the weekly HR recovery towards ≈72+ h since NHEJ achieves it quite well in the 24+ h from day to day, as shown in the lower right part of the Graphical Abstract. This fractionation advantage works well for low-LET radiations but also for the lightest ions with mainly a low LET in normal tissues.
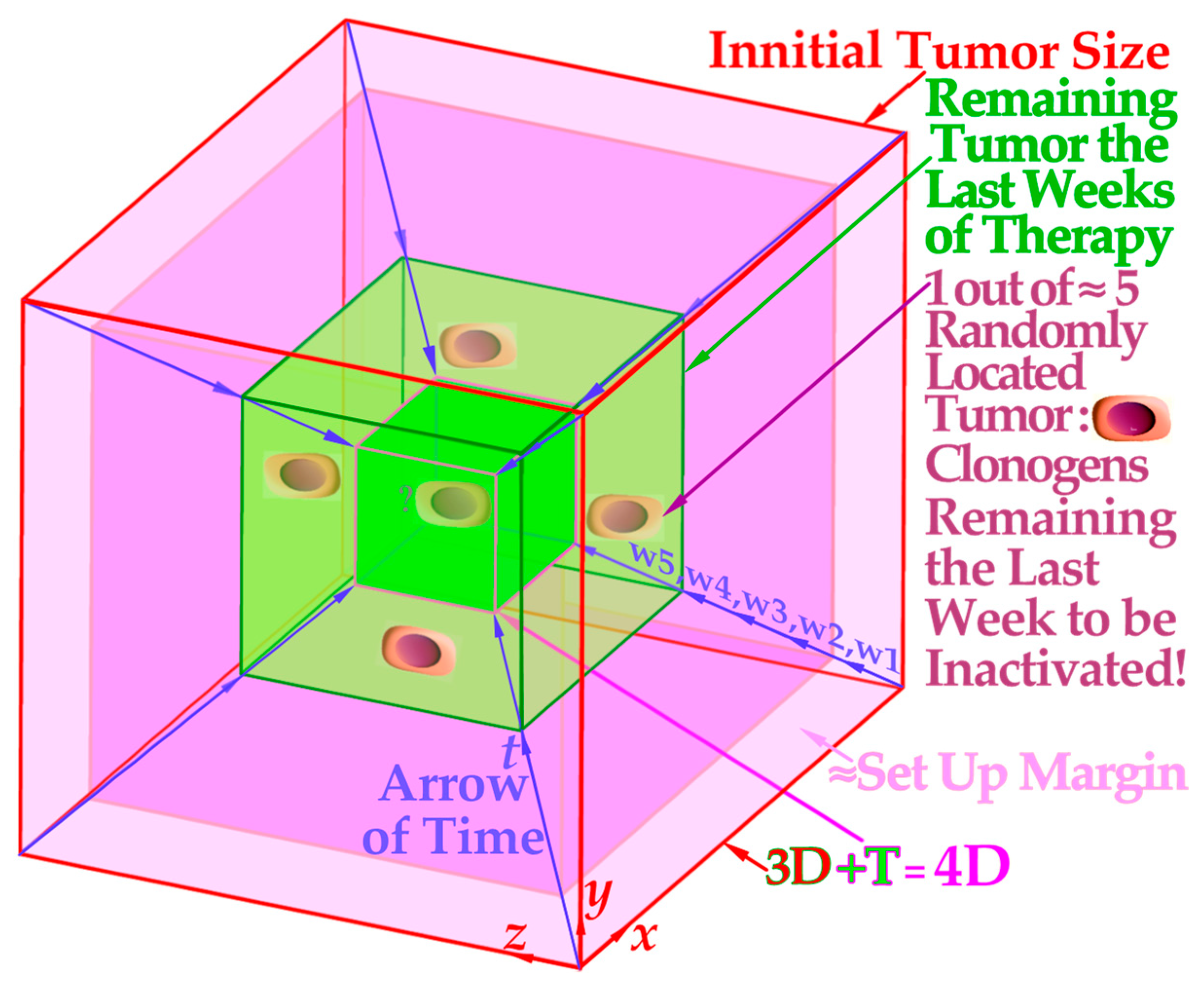
- For elderly patients, a larger number of optimized beam portals may be ideal, whereas younger patients may benefit from fewer beams (<5) and low-to-medium LET ions (see [5]) to reduce the risk for secondary cancers in extended low-dose regions (1–6 Gy total dose; see Figure 6 and [39,40]). These volumes should therefore be reduced as far as possible using sharp penumbras simultaneously as the complication-free cure (P+) or preferably the P++ optimization strategy (P+ followed by a constrained injury relaxation) are the key objectives of the treatment [60] (Figure 22).
- To further increase the biologically effective tumor dose delivery, a few light ion beam portals should be used preferably in the range from helium to boron ions only with their Bragg peaks located in the gross tumor volume, to keep the LET low (<10 eV/nm) and the dose within 1.8–2.3 Gy/fraction in organs at risk [1] (Figure 22). Organs at risk have to be passed through with beams to reach the target volume, and with the lightest ions (He-B), this can be carried out using a fairly low LET (<10 eV/nm). To maximize the complication-free cure, it is best to switch to electrons or photons in the last 10–15 GyE, and for bulky tumors, possibly a light ion concomitant gross tumor boost should be used in the last 5 GyE before the final plain 10 GyE low-LET round-up (Figure 8, Figure 9 and Figure 10; [47,54]).
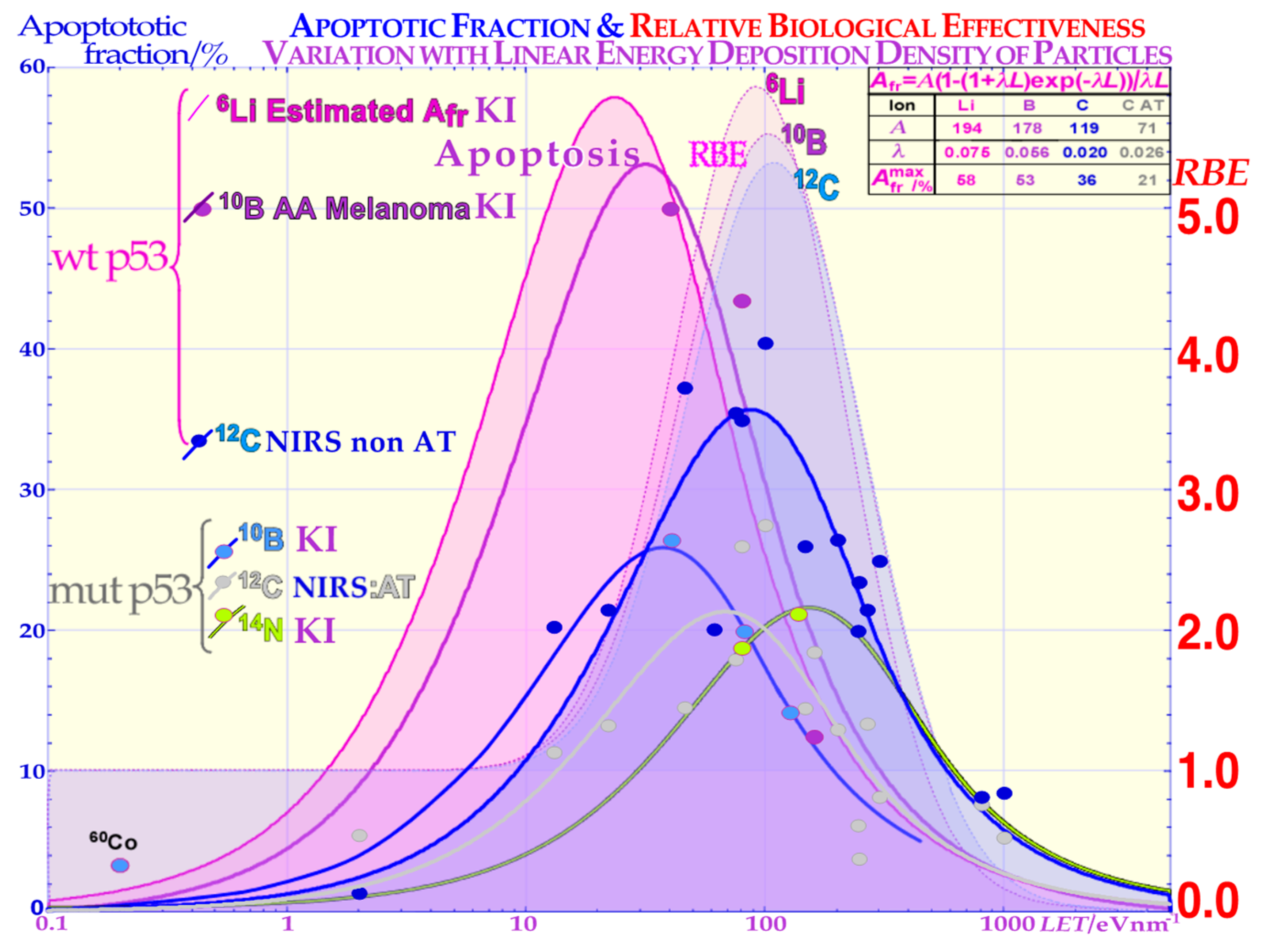
- The influence of tumor vasculature heterogeneity on the distribution of hypoxia was carefully calculated for key tumor types and showed good agreement with clinically measured Eppendorf distributions of hypoxia [62,63,64,65]. This clinically very useful dataset for treating common hypoxic tumors with low LET later showed that the optimal LET for treating them is only as low as 25 eV/nm [46,47,54,65]. This is in good agreement with the optimal LET window of 15–55 eV/nm [1,31,54], so it also can cover other types of tumor heterogeneity and radiation resistance using helium to boron ions.
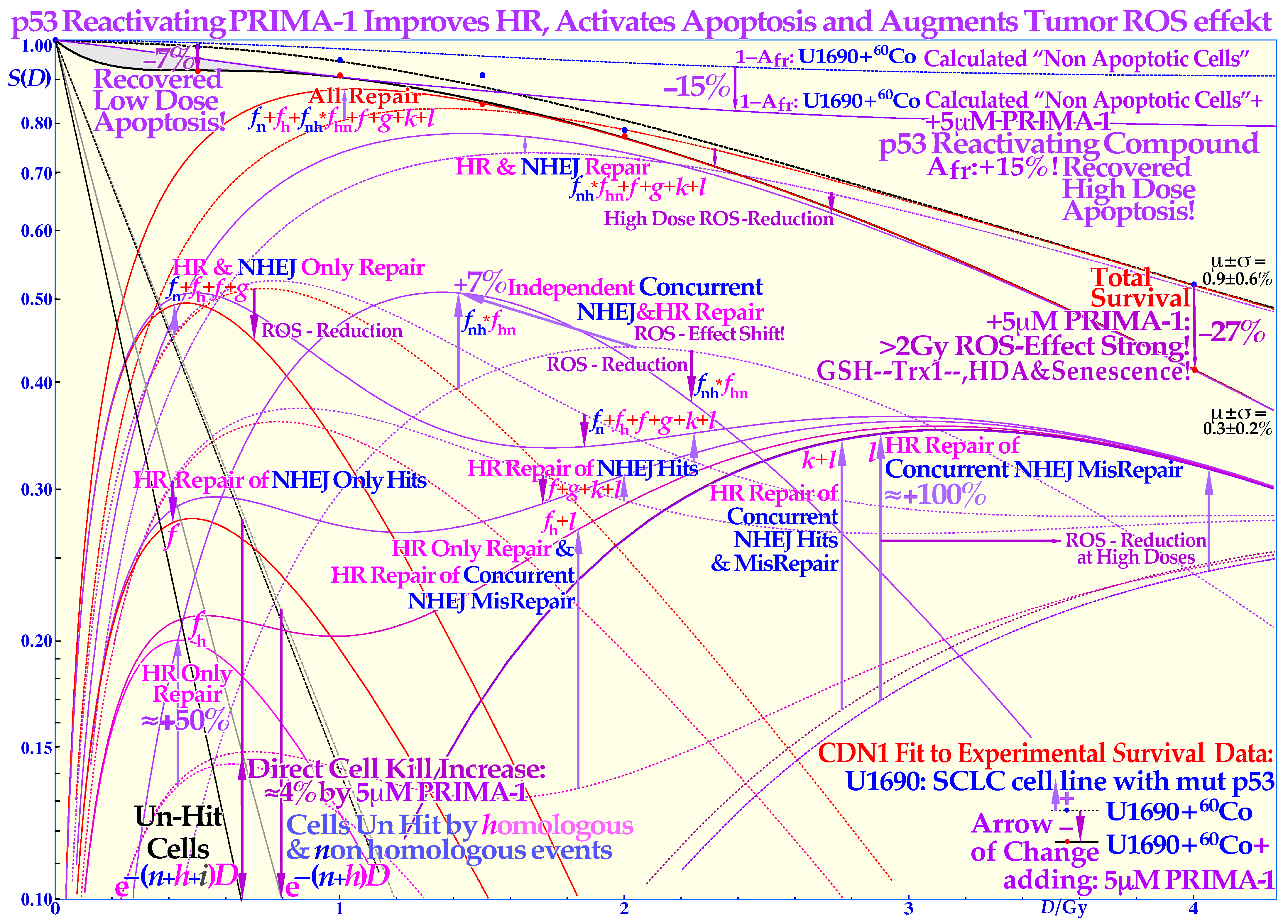
- For the multitude of radiation-resistant TP53 and/or ATM-mutated tumors that are often a severe clinical problem, the interesting p53 reactivating PRIMA-1 and APR-246 pharmaca may be useful to increase tumor cell apoptosis and further augment the radiation-induced reactive oxygen species effects in the high-dose tumor volume. Interestingly, PRIMA-1 and APR-246 promote the normal function of a missense mutant p53 protein-increasing LDA and HDA apoptosis in the tumor as well as senescence (Figure 4; [2,7,26,27,28,35]). Among other effects, as shown in Figure 4, it inhibits the enzyme thioredoxin reductase 1 and thioredoxin and decreases cellular glutathione levels, which is especially valuable with low-LET radiations, when the lightest ions are not available [2] (Figure 17).
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AT | Ataxia telangiectasia cell line |
| ATM | Ataxia telangiectasia-mutated |
| CDN1 | One-dimensional closest distance norm |
| CHK2 | Checkpoint kinase 2 |
| DDSB | Dual double-strand break |
| DSB | Double-strand break |
| DYRK2 | Dual-specificity tyrosine-regulated kinase 2 |
| GSH | Glutathione |
| HDA | High-dose apoptosis |
| HR | Homologous recombination |
| LDA | Low-dose apoptosis |
| LDHS | Low-dose hypersensitivity |
| LET | Linear energy transfer |
| LQ | Linear quadratic |
| Mut | Mutant type |
| NHEJ | Nonhomologous end-joining |
| p | Phosphorylated |
| RBE | Relative biological effectiveness |
| RCR | Repairable–conditionally repairable model |
| RHR | Repairable–homologically repairable formulation |
| Trx1 | Thioredoxin reductase 1 |
| wt | Wild type |
References
- Brahme, A. A DNA Repair-based model of cell survival with important clinical consequences. Radiat. Res. 2020, 194, 202–235. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Quantifying Cellular Repair, Misrepair and Apoptosis Induced by Boron Ions, Gamma Rays and PRIMA-1 Using the RHR Formulation. Radiat. Res. 2022, 198, 271–296. [Google Scholar] [CrossRef] [PubMed]
- Buscemi, G.; Perego, P.; Carenini, N.; Nakanishi, M.; Chessa, L.; Chen, J.; Khanna, K.; Delia, D. Activation of ATM and Chk2 kinases in relation to the amount of DNA strand breaks. Oncogene 2004, 23, 7691–7700. [Google Scholar] [CrossRef] [PubMed]
- Enns, L.; Bogen, K.T.; Wizniak, J.; Murtha, A.D.; Weinfeld, M. Low-dose radiation hypersensitivity is associated with p53-dependent apoptosis. Mol. Cancer Res. 2004, 2, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Short, S.C.; Bourne, S.; Martindale, C.; Woodcock, M.; Jackson, S.P. DNA Damage Responses at Low Radiation Doses. Radiat. Res. 2005, 164, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Wang, Y.; Zhao, Y.; He, S.; Zhao, R.; Song, Y.; Cheng, J.; Gong, Y.; Xie, J.; Wang, Y.; et al. Caspase-3 knockout attenuates radiation-induced tumor repopulation via impairing the ATM/p53/Cox-2/PGE2 pathway in non-small cell lung cancer. Aging 2020, 12, 21758–21776. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. High resolution molecular radiation therapy and tumor imaging for the 21st century. J. Nucl. Med. Radiat. Ther. 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Brahme, A. Development of Highly Specific Molecular Cancer Therapy with the Lightest Ions. In Proceedings of the 5th Takahashi Memorial International Symposium, Sendai, Japan, 7–9 September 2007; p. 57, Book of Abstract Japan. [Google Scholar]
- Brahme, A. Optimal use of Light Ions for Radiation Therapy. Radiol. Sci. 2010, 53, 35–61. Available online: http://www.nirs.go.jp/publication/rs-sci/pdf/201008.pdf (accessed on 7 May 2023).
- Wang, Y.H.; Ho, T.L.F.; Hariharan, A.; Goh, H.C.; Wong, Y.L.; Verkaik, N.S.; Lee, M.Y.; Tam, W.L.; van Gent, D.C.; Venkitaraman, A.R.; et al. Rapid recruitment of p53 to DNA damage sites directs DNA repair choice and integrity. Proc. Natl. Acad. Sci. USA 2022, 119, e2113233119. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y. Isolation of p53-target genes and their functional analysis. Cancer Sci. 2004, 95, 7–11. [Google Scholar] [CrossRef]
- Williams, A.B.; Schumacher, B. p53 in the DNA-damage-repair process. Cold Spring Harb. Perspect. Med. 2016, 6, a026070. [Google Scholar] [CrossRef]
- Brahme, A.; Rydberg, B.; Blomqvist, P. Dual spatially correlated nucleosomal double strand breaks in cell inactivation. In Microdosimetry: An Interdisciplinary Approach; Goodhead, D.T., O’Neill, P., Menzel, H.G., Eds.; The Royal Society of Chemistry: Cambridge, UK, 1997; pp. 125–128. [Google Scholar]
- Brahme, A.; Lorat, Y. Dual Nucleosomal Double Strand Breaks are the Key Effectors of Curative Radiation Therapy. Int. J. Mol. Sci. 2023. In press. [Google Scholar]
- Kastenhuber, E.R. Lowe SW. Putting p53 in Context. Cell 2017, 170, 1062–1078. [Google Scholar] [CrossRef]
- Myler, L.R.; Gallardo, I.F.; Soniat, M.M.; Deshpande, R.A.; Gonzalez, X.B.; Kim, Y.; Paull, T.T.; Finkelstein, I.J. Single-Molecule Imaging Reveals How Mre11-Rad50-Nbs1 Initiates DNA Break Repair. Mol. Cell 2017, 67, 891–898. [Google Scholar] [CrossRef]
- Gerelchuluun, A.; Manabe, E.; Ishikawa, T.; Sun, L.; Itoh, K.; Sakae, T.; Suzuki, K.; Hirayama, R.; Asaithamby, A.; Chen, D.J.; et al. The major DNA repair pathway after both proton and carbon ion radiation is NHEJ, but the HR pathway is more relevant in carbon ions. Radiat. Res. 2015, 183, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Kubo, M.; Ma, H.; Nakagawa, A.; Yoshida, Y.; Isono, M.; Kanai, T.; Ohno, T.; Furusawa, Y.; Funayama, T.; et al. Nonhomologous End-Joining Repair. Plays a More Important Role than Homologous Recombination Repair. in Defining Radiosensitivity after Exposure to High-LET Radiation. Radiat. Res. 2014, 182, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Scully, R.; Panday, A.; Elango, R.; Willis, N.A. DNA double-strand break repair-pathway choice in somatic mammalian cells. Nat. Rev. Mol. Cell Biol. 2019, 20, 698–714. [Google Scholar] [CrossRef]
- Singh, B.; Arrand, J.E.; Joiner, M.C. Hypersensitive response of normal human lung epithelial cells at low radiation doses. Int. J. Radiat. Biol. 1994, 65, 457–464. [Google Scholar] [CrossRef]
- Krueger, S.A.; Collis, S.J.; Joiner, M.C.; Wilson, G.D.; Marples, B. Transition in Survival from low-dose hyper-radiosensitivity to increased radioresistance is independent of activation of ATM Ser1981 activity. Int. J. Radiat. Oncol. Biol. Phys. 2007, 69, 1262–1271. [Google Scholar] [CrossRef] [PubMed]
- Turesson, I.; Nyman, J.; Qvarnström, F.; Simonsson, M.; Book, M.; Hermansson, I.; Sigurdardottir, S.; Johansson, K.-A. A low-dose hypersensitive keratinocyte loss in response to fractionated radiotherapy is associated with growth arrest and apoptosis. Radiot. Oncol. 2010, 94, 90–101. [Google Scholar] [CrossRef]
- Riballo, E.; Kuhne, M.; Rief, N.; Doherty, A.; Smith, G.C.M.; Recio, M.J.; Reis, C.; Dahm, K.; Fricke, A.; Krempler, A.; et al. A pathway of double-strand break rejoining dependent upon ATM, artemis, and proteins locating to γ-H2AX foci. Mol. Cell 2004, 16, 715–724. [Google Scholar] [CrossRef]
- Xue, L.; Yu, D.; Furusawa, Y.; Cao, J.; Okayasu, R.; Fan, S. ATM-dependent hyper-radiosensitivity in mammalian cells irradiated by heavy ions. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Vreede, P.; Brahme, A. Development of Biologically Optimized Radiation Therapy: Maximizing the Apoptotic Cell Kill. Radiol. Sci. 2009, 52, 31–52. Available online: http://www.nirs.go.jp/info/report/rs-sci/pdf/200907.pdf (accessed on 8 September 2014).
- Schmitt, C.A. Senescence, apoptosis and therapy—Cutting the lifelines of cancer. Nat. Rev. Cancer 2003, 3, 283–295. [Google Scholar] [CrossRef] [PubMed]
- d’Adda di Fagagna, F. Living on a break: Cellular senescence as a DNA-damage response. Nat. Rev. Cancer 2008, 8, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Nardella, C.; Clohessy, J.G.; Alimonti, A.; Pandolfi, P.P. Pro-senescence therapy for cancer treatment. Nat. Rev. Cancer 2011, 11, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Lind, B.K.; Persson, L.M.; Edgren, M.R.; Hedlöf, I.; Brahme, A. Repairable-conditionally repairable damage model based on dual Poisson processes. Radiat. Res. 2003, 160, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Joiner, M.C.; Johns, H. Renal damage in the mouse: The response to very small doses per fraction. Radiat. Res. 1988, 114, 385–398. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Physical, Biological and Clinical Merits of High Energy Boron Ions for Radiation Therapy. In Boron, Boron Compounds and Boron-Based Materials and Structures; Aydin, M., Ed.; IntechOpen: London, UK, 2023. [Google Scholar] [CrossRef]
- Bykov, V.J.N.; Eriksson, S.E.; Bianchi, J.; Wiman, K.G. Targeting mutant p53 for efficient cancer therapy. Nat. Rev. Cancer 2018, 18, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Perdrix, A.; Najem, A.; Saussez, S.; Awada AJourne, F.; Ghanem, G.; Krayem, M. PRIMA-1 and PRIMA-1MET (APR-246): From Mutant/Wild Type p53 Reactivation to Unexpected Mechanisms Underlying Their Potent Anti-Tumor Effect in Combinatorial Therapies. Cancers 2017, 9, 172. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Makino, T.; Yamasaki, M.; Tanaka, K.; Miyazaki, Y.; Takahashi, T.; Kurokawa, Y.; Nakajima, K.; Takiguchi, S.; Mori, M.; et al. PRIMA-1 induces p53-mediated apoptosis by upregulating Noxa in esophageal squamous cell carcinoma with TP53 missense mutation. Cancer Sci. 2018, 109, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Fan, C.; Luo, B.; Zhang, J.; Jensen, L.D.; Burman, J.; Jönsson, C.; Ljusberg, A.; Larsson, P.; Zhao, Z.; et al. APR-246 Enhances Colorectal Cancer Sensitivity to Radiotherapy. Available online: http://aacrjournals.org/mct/article-pdf/doi/10.1158/1535-7163.MCT-22-0275/3333376/mct-22-0275.pdf (accessed on 27 July 2013).
- Aaltonen, P.; Brahme, A.; Lax, I.; Levernes, S.; Näslund, I.; Reitan, J.V.; Turesson, I. Specification of dose delivery in radiation therapy. Recommendations by the NACP. Acta Oncol. 1997, 36 (Suppl. S10), 1–32. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Biologically based treatment planning. Acta Oncol. 1999, 38, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Individualizing cancer treatment: Biological optimization models in treatment planning and delivery. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Optimization of radiation therapy and the development of multileaf collimation. Editorial. Int. J. Rad. Oncol. Biol. Phys. 1993, 25, 373–375. [Google Scholar] [CrossRef] [PubMed]
- Hall, E.J.; Wuu, C.S. Radiation-Induced Second Cancers: The Impact of 3D-CRT and IMRT. Int. J. Radiat. Oncol. Biol. Phys. 2003, 56, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Brand, D.H.; Kirby, A.M.; Yarnold, J.R.; Somaiah, N. How Low. Can. You Go? The Radiobiology of Hypofractionation. Clin. Oncol. 2022, 34, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Dosimetric precision requirements in radiation therapy. Acta Radiol. Oncol. 1984, 23, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Tilikidis, A.; Brahme, A. Microdosimetric description of beam quality and biological effectiveness in radiation therapy. Acta Oncol. 1994, 33, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Hiroshima, Y.; Kanematsu, N.; Inaniwa, T.; Shirai, T.; Imai, R.; Suzuki, H.; Akakura, K.; Wakatsuki, M.; Ichikawa, T.; et al. Carbon-ion radiotherapy for urological cancers. Int. J. Urol. 2022, 29, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Lorat, Y.; Reindl, J.; Isermann, A.; Rübe, C.; Friedl, A.A.; Rübe, C.E. Focused Ion Microbeam Irradiation Induces Clustering of DNA Double-Strand Breaks in Heterochromatin Visualized by Nanoscale-Resolution Electron Microscopy. Int. J. Mol. Sci. 2021, 22, 7638. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Accurate Description of the Cell Survival and Biological Effect at Low and High Doses and LETs. J. Rad. Res. 2011, 52, 389–407. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brahme, A. Biologically Optimized Lightion Therapy. In Chief: Comprehensive BioMedical Physics; Major Reference Work; Brahme, A., Ed.; Elsevier: Oxford, UK, 2014; Volume 9, pp. 529–554. [Google Scholar]
- Cohen, L. The statistical prognosis in radiation therapy. Am. J. Roentgenol. 1960, 84, 741–753. [Google Scholar]
- Ågren-Cronqvist, A.; Källman, P.; Turesson, I.; Brahme, A. Volume and heterogeneity dependence of the dose-response relationship for head and neck tumours. Acta Oncol. 1995, 34, 851–860. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Physical and biologic aspects on the optimum choice of radiation modality. Acta Radiol. Oncol. 1982, 21, 469–479. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lindborg, L.; Brahme, A. Influence of microdosimetric quantities on observed dose-response relationships in radiation therapy. Rad. Res. 1990, 124 (Suppl. S1), S23–S28. [Google Scholar] [CrossRef]
- Ågren, A.; Brahme, A.; Turesson, I. Optimization of uncomplicated control for head and neck tumors. Int. J. Rad. Onc. Biol. Phys. 1990, 19, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A. Optimized radiation therapy based on radiobiological objectives. Sem. Radiat. Oncol. 1999, 9, 35–47. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A.; Svensson, H. Physical, biological and clinical background for the development of biologically optimized light ion therapy. In Biologically Optimized Radiation Therapy; Brahme, A., Ed.; World Scientific Publishing: Singapore, 2014; pp. 499–648. [Google Scholar]
- Eichorhn, J.; Lessel, A. Four years experience with combined neutron-telecobalt therapy. Int. J. Radiat. Oncol. Biol. Phys. 1977, 3, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Peters, L.J.; Hussey, D.H.; Fletcher, G.H.; Baumann, P.A.; Olson, M.H. Preliminary report of the M.D. Anderson-Texas A.&M. variable energy cyclotron fast neutron therapy pilot study. Amer. J. Roentgenol. 1979, 132, 637. [Google Scholar]
- Griffit, N.; Blasko, J.; Laramor, G.E. Results of fast neutron beam radiotherapy pilot studies at the University of Washington. In High-LET Radiations in Clinical Radiotherapy; Barendsen, G.W., Broerse, J.J., Breuer, K., Eds.; Pergamon Press: Oxford, UK, 1979; pp. 23–29. [Google Scholar]
- Tsunemoto, H.; Umegaki, Y.; Kutsutani, Y.; Arai, T.; Morita, S.; Kurisu, A.; Kawashima, K.; Maruyama, T. Results of clinical applications of fast neutrons in Japan. In High-LET Radiations in Clinical Radiotherapy; Barendsen, G.W., Broerse, J.J., Breuer, K., Eds.; Pergamon Press: Oxford, UK, 1979; pp. 75–78. [Google Scholar]
- Maor, M.H.; Hussey, D.H.; Fletcher, G.H.; Jesse, R.H. Fast neutron therapy for locally advanced head and neck tumors. Int. J. Radiat. Oncol. Biol. Phys. 1981, 7, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Brahme, A.; Löf, J. Fundamentals of Physically and Biologically Based Radiation Therapy Optimization. In Comprehensive BioMedical Physics; Major Reference Work; Brahme, A., Ed.; Elsevier: Oxford, UK, 2014; Volume 9, pp. 271–313. [Google Scholar]
- Siddiqi, M.; Lind, B.K.; Brahme, A. Optimal dose fractionation of lung cancer using biologically optimized IMRT. 525 Poster. Radiother. Oncol. 2004, 73, S235. Available online: https://bit.ly/2LCN1m7 (accessed on 4 May 2006).
- Brahme, A.; Ågren, A.K. On the optimal dose distribution for eradication of heterogeneous tumors. Acta Oncol. 1987, 26, 377–385. [Google Scholar] [CrossRef]
- Nilsson, J.; Lind, B.K.; Brahme, A. Radiation response of hypoxic and generally heterogeneous tissues. Int. J. Radiat. Biol. 2002, 78, 389–405. [Google Scholar] [CrossRef]
- Lind, B.K.; Brahme, A. The radiation response of heterogeneous tumors. Phys Med. 2008, 23, 91–99. [Google Scholar] [CrossRef]
- Brahme, A. Accurate description of heterogeneous tumors by their effective radiation-sensitive and resistant cell compartments. In Comprehensive BioMedical Physics; Major Reference Work; Brahme, A., Ed.; Elsevier: Oxford, UK, 2014; Volume 9, pp. 191–203. [Google Scholar]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brahme, A. TP53 and the Ultimate Biological Optimization Steps of Curative Radiation Oncology. Cancers 2023, 15, 4286. https://doi.org/10.3390/cancers15174286
Brahme A. TP53 and the Ultimate Biological Optimization Steps of Curative Radiation Oncology. Cancers. 2023; 15(17):4286. https://doi.org/10.3390/cancers15174286
Chicago/Turabian StyleBrahme, Anders. 2023. "TP53 and the Ultimate Biological Optimization Steps of Curative Radiation Oncology" Cancers 15, no. 17: 4286. https://doi.org/10.3390/cancers15174286
APA StyleBrahme, A. (2023). TP53 and the Ultimate Biological Optimization Steps of Curative Radiation Oncology. Cancers, 15(17), 4286. https://doi.org/10.3390/cancers15174286







