Increased Reflux Esophagitis after Helicobacter pylori Eradication Therapy in Cases Undergoing Endoscopic Submucosal Dissection for Early Gastric Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
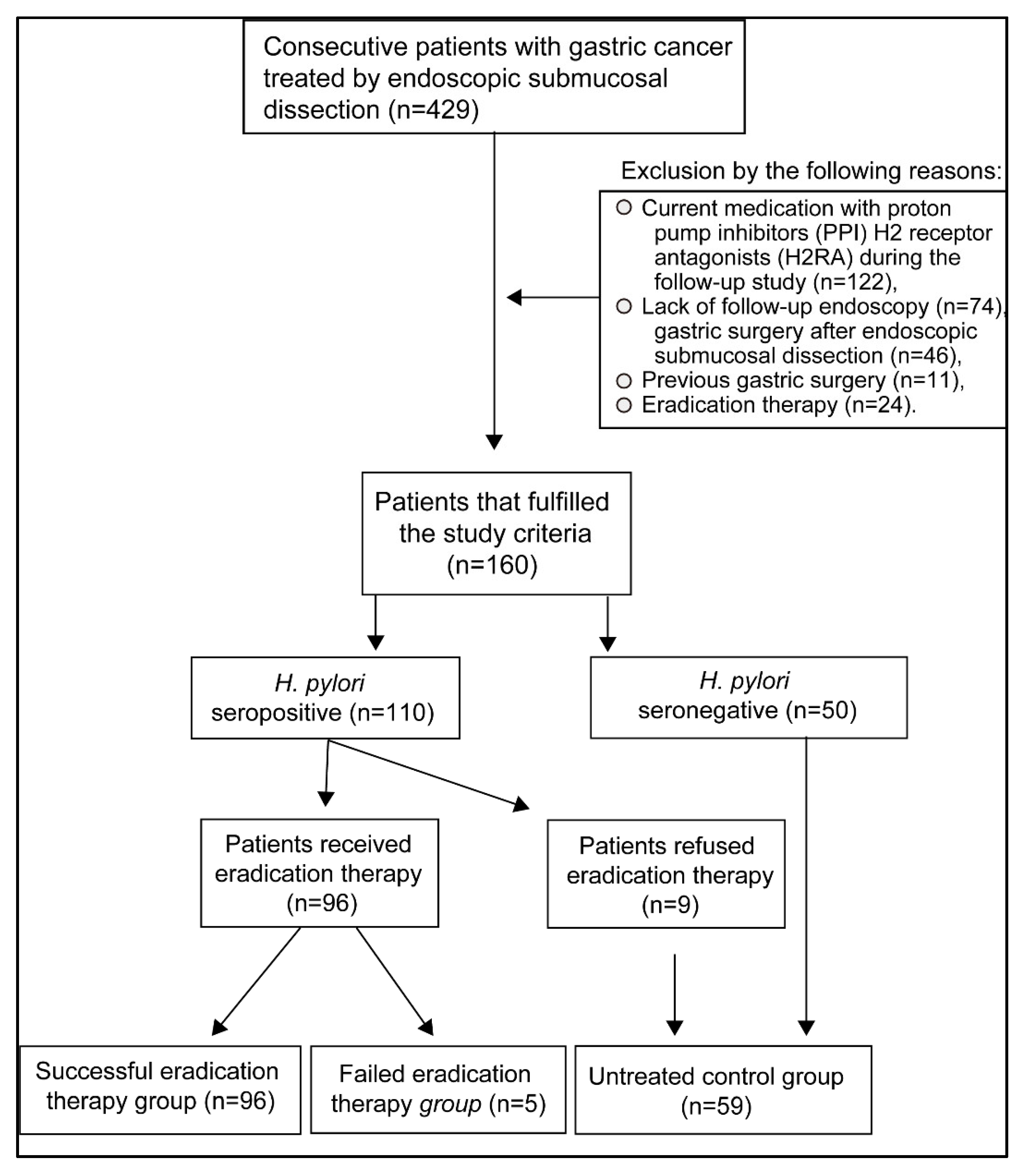
2.1. Patients
2.2. Study Design
2.3. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Frequency and Severity of Reflux Esophagitis before and after Therapy
3.3. Univariate and Multivariate Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fukase, K.; Kato, M.; Kikuchi, S.; Inoue, K.; Uemura, N.; Okamoto, S.; Terao, S.; Amagai, K.; Hayashi, S.; Asaka, M.; et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: An open-label, randomised controlled trial. Lancet 2008, 372, 392–397. [Google Scholar] [CrossRef]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Gisbert, J.P.; Kuipers, E.J.; Axon, A.T.; Bazzoli, F.; Gasbarrini, A.; Atherton, J.; Graham, D.Y.; et al. Management of Helicobacter pylori infection—The Maastricht V/Florence consensus report. Gut 2017, 66, 6–30. [Google Scholar] [CrossRef]
- Usai Satta, P.; Oppia, F.; Cabras, F. Overview of pathophysiological features of GERD. Minerva Gastroenterol. Dietol. 2017, 63, 184–197. [Google Scholar]
- Schutze, K.; Hentschel, E.; Dragosics, B.; Hirschl, A.M. Helicobacter pylori reinfection with identical organisms: Transmission by the patients’ spouses. Gut 1995, 36, 831–833. [Google Scholar] [CrossRef] [PubMed]
- Labenz, J.; Blum, A.L.; Bayerdorffer, E.; Meining, A.; Stolte, M.; Borsch, G. Curing Helicobacter pylori infection in patients with duodenal ulcer may provoke reflux esophagitis. Gastroenterology 1997, 112, 1442–1447. [Google Scholar] [CrossRef]
- Befrits, R.; Sjostedt, S.; Odman, B.; Sorngard, H.; Lindberg, G. Curing Helicobacter pylori infection in patients with duodenal ulcer does not provoke gastroesophageal reflux disease. Helicobacter 2000, 5, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Atherton, J.; Axon, A.T.; Bazzoli, F.; Gensini, G.F.; Gisbert, J.P.; Graham, D.Y.; Rokkas, T.; et al. Management of Helicobacter pylori infection—The Maastricht IV/Florence Consensus Report. Gut 2012, 61, 646–664. [Google Scholar] [CrossRef] [PubMed]
- McColl, K.E.; Dickson, A.; El-Nujumi, A.; El-Omar, E.; Kelman, A. Symptomatic benefit 1–3 years after H. pylori eradication in ulcer patients: Impact of gastroesophageal reflux disease. Am. J. Gastroenterol. 2000, 95, 101–105. [Google Scholar]
- Hamada, H.; Haruma, K.; Mihara, M.; Kamada, T.; Yoshihara, M.; Sumii, K.; Kajiyama, G.; Kawanishi, M. High incidence of reflux oesophagitis after eradication therapy for Helicobacter pylori: Impacts of hiatal hernia and corpus gastritis. Aliment. Pharmacol. Ther. 2000, 14, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Inoue, H.; Imoto, I.; Taguchi, Y.; Kuroda, M.; Nakamura, M.; Horiki, N.; Oka, S.; Gabazza, E.C.; Adachi, Y. Reflux esophagitis after eradication of Helicobacter pylori is associated with the degree of hiatal hernia. Scand. J. Gastroenterol. 2004, 39, 1061–1065. [Google Scholar] [CrossRef]
- Take, S.; Mizuno, M.; Ishiki, K.; Nagahara, Y.; Yoshida, T.; Yokota, K.; Oguma, K.; Okada, H.; Yamamoto, K. Helicobacter pylori eradication may induce de novo, but transient and mild, reflux esophagitis: Prospective endoscopic evaluation. J. Gastroenterol. Hepatol. 2009, 24, 107–113. [Google Scholar] [CrossRef]
- Li, Y.; Xia, R.; Zhang, B.; Li, C. Chronic atrophic gastritis: A review. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 241–259. [Google Scholar] [CrossRef]
- Na, H.K.; Lee, J.H.; Park, S.J.; Park, H.J.; Kim, S.O.; Ahn, J.Y.; Kim, D.H.; Jung, K.W.; Choi, K.D.; Song, H.J.; et al. Effect of Helicobacter pylori eradication on reflux esophagitis and GERD symptoms after endoscopic resection of gastric neoplasm: A single-center prospective study. BMC Gastroenterol. 2020, 20, 123. [Google Scholar] [CrossRef]
- Hill, L.D.; Kozarek, R.A.; Kraemer, S.J.; Aye, R.W.; Mercer, C.D.; Low, D.E.; Pope, C.E., II. The gastroesophageal flap valve: In vitro and in vivo observations. Gastrointest. Endosc. 1996, 44, 541–547. [Google Scholar] [CrossRef]
- Kimura, K.; Satoh, K.; Ido, K.; Taniguchi, Y.; Takimoto, T.; Takemoto, T. Gastritis in the Japanese stomach. Scand. J. Gastroenterol. Suppl. 1996, 214, 17–20. [Google Scholar] [CrossRef]
- Hirayama, Y.; Kawai, T.; Otaki, J.; Kawakami, K.; Harada, Y. Prevalence of Helicobacter pylori infection with healthy subjects in Japan. J. Gastroenterol. Hepatol. 2014, 29, 16–19. [Google Scholar] [CrossRef]
- Kinoshita, Y.; Kawanami, C.; Kishi, K.; Nakata, H.; Seino, Y.; Chiba, T. Helicobacter pylori independent chronological change in gastric acid secretion in the Japanese. Gut 1997, 41, 452–458. [Google Scholar] [CrossRef]
- Furukawa, N.; Iwakiri, R.; Koyama, T.; Okamoto, K.; Yoshida, T.; Kashiwagi, Y.; Ohyama, T.; Noda, T.; Sakata, H.; Fujimoto, K. Proportion of reflux esophagitis in 6010 Japanese adults: Prospective evaluation by endoscopy. J. Gastroenterol. 1999, 34, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Arakawa, T. Epidemiology and clinical characteristics of GERD in the Japanese population. J. Gastroenterol. 2009, 44, 518–534. [Google Scholar] [CrossRef]
- Correa, P.; Piazuelo, M.B. The gastric precancerous cascade. J. Dig. Dis. 2012, 13, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Laine, L.; Sugg, J. Effect of Helicobacter pylori eradication on development of erosive esophagitis and gastroesophageal reflux disease symptoms: A post hoc analysis of eight double blind prospective studies. Am. J. Gastroenterol. 2002, 97, 2992–2997. [Google Scholar] [CrossRef] [PubMed]
- Qian, B.; Ma, S.; Shang, L.; Qian, J.; Zhang, G. Effects of Helicobacter pylori eradication on gastroesophageal reflux disease. Helicobacter 2011, 16, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Saad, A.M.; Choudhary, A.; Bechtold, M.L. Effect of Helicobacter pylori treatment on gastroesophageal reflux disease (GERD): Meta-analysis of randomized controlled trials. Scand. J. Gastroenterol. 2012, 47, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Cui, X.; Zheng, H.; Chen, D.; He, L.; Jiang, B. Meta-analysis: Eradication of Helicobacter pylori infection is associated with the development of endoscopic gastroesophageal reflux disease. Eur. J. Gastroenterol. Hepatol. 2013, 25, 1195–1205. [Google Scholar] [CrossRef]
- Sugimoto, M.; Murata, M.; Mizuno, H.; Iwata, E.; Nagata, N.; Itoi, T.; Kawai, T. Endoscopic reflux esophagitis and reflux-related symptoms after Helicobacter pylori eradication therapy: Meta-analysis. J. Clin. Med. 2020, 9, 3007. [Google Scholar] [CrossRef] [PubMed]
- El-Omar, E.; Penman, I.; Dorrian, C.A.; Ardill, J.E.; McColl, K.E. Eradicating Helicobacter pylori infection lowers gastrin mediated acid secretion by two thirds in patients with duodenal ulcer. Gut 1993, 34, 1060–1065. [Google Scholar] [CrossRef]
- Furuta, T.; El-Omar, E.M.; Xiao, F.; Shirai, N.; Takashima, M.; Sugimura, H. Interleukin 1beta polymorphisms increase risk of hypochlorhydria and atrophic gastritis and reduce risk of duodenal ulcer recurrence in Japan. Gastroenterology 2002, 123, 92–105. [Google Scholar] [CrossRef]
- Yamaoka, Y.; Orito, E.; Mizokami, M.; Gutierrez, O.; Saitou, N.; Kodama, T.; Osato, M.S.; Kim, J.G.; Ramirez, F.C.; Mahachai, V.; et al. Helicobacter pylori in North and South America before Columbus. FEBS Lett. 2002, 517, 180–184. [Google Scholar] [CrossRef]
| Successful Eradication Group | % | |
|---|---|---|
| First-line therapy | 78/101 | 77.2% |
| Second-line therapy | 15/21 | 71.4% |
| Third-line therapy | 2/3 | 66.7% |
| Fourth-line therapy | 1/1 | 100% |
| Total | 96/101 | 95.0% |
| Variables | Successful Eradication Group (n = 96) | Failed Eradication Group (n = 5) | p Values * | Untreated Control Group (n = 59) | p Values * |
|---|---|---|---|---|---|
| Age (years) | 69.4 ± 9.1 | 71.4 ± 5.1 | 0.610 | 69.9 ± 7.4 | 0.848 |
| Sex (male/female) | 73/23 | 5/0 | 0.213 | 44/15 | 0.837 |
| Body mass index (kg/m2) | 22.6 ± 3.1 | 22.2 ± 2.6 | 0.803 | 23.0 ± 3.3 | 0.381 |
| Hiatus hernia (presence/absence) | 56/40 | 3/2 | 0.941 | 38/21 | 0.452 |
| Gastritis (close/open) | 18/78 | 1/4 | 0.944 | 21/38 | 0.019 |
| Diabetes (yes/no/unknown) | 10/83/3 | 1/4 | 0.523 | 12/47 | 0.102 |
| Hypertension (yes/no/unknown) | 37/56/3 | 2/3 | 0.992 | 31/28 | 0.123 |
| Hyperlipidemia (yes/no/unknown) | 16/73/3 | 0/5 | 0.311 | 15/44 | 0.220 |
| Alcohol (yes/no/unknown) | 33/58/5 | 3/2 | 0.286 | 20/38/1 | 0.662 |
| Smoking (yes/no/unknown) | 10/81/5 | 0/5 | 0.434 | 2/57/0 | 0.094 |
| NSAID use (yes/no/unknown) | 5/86/5 | 0/5 | 0.590 | 7/52/0 | 0.160 |
| Univariate Analysis | Odds Ratio | 95% Confidence Interval | p Values |
|---|---|---|---|
| Age (<70) | 2.781 | 0.929–8.324 | 0.067 |
| Sex | 1.041 | 0.318–3.413 | 0.947 |
| Body mass index (≧25) | 0.436 | 0.094–2.014 | 0.287 |
| Hiatus hernia | 0.741 | 0.269–2.039 | 0.561 |
| Gastritis | 0.738 | 0.242–2.254 | 0.594 |
| Diabetes | 0.359 | 0.0425–2.863 | 0.333 |
| Hypertension | 1.041 | 0.240–2.026 | 0.507 |
| Hyperlipidemia | 0.436 | 0.604–5.907 | 0.275 |
| Alcohol | 0.741 | 0180–1.974 | 0.397 |
| Smoking | 0.738 | 0.095–6.603 | 0.83 |
| NSAID use | 0.359 | 0.374–9.583 | 0.441 |
| Successful eradication | 5.577 | 1.227–25.347 | 0.026 |
| Multivariate analyses | |||
| Successful eradication | 5.887 | 1.238–28.001 | 0.026 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katsurahara, M.; Imoto, I.; Umeda, Y.; Miura, H.; Tsuboi, J.; Yamada, R.; Yasuma, T.; Nakamura, M.; Hamada, Y.; Inoue, H.; et al. Increased Reflux Esophagitis after Helicobacter pylori Eradication Therapy in Cases Undergoing Endoscopic Submucosal Dissection for Early Gastric Cancer. Cancers 2021, 13, 1779. https://doi.org/10.3390/cancers13081779
Katsurahara M, Imoto I, Umeda Y, Miura H, Tsuboi J, Yamada R, Yasuma T, Nakamura M, Hamada Y, Inoue H, et al. Increased Reflux Esophagitis after Helicobacter pylori Eradication Therapy in Cases Undergoing Endoscopic Submucosal Dissection for Early Gastric Cancer. Cancers. 2021; 13(8):1779. https://doi.org/10.3390/cancers13081779
Chicago/Turabian StyleKatsurahara, Masaki, Ichiro Imoto, Yuhei Umeda, Hiroshi Miura, Junya Tsuboi, Reiko Yamada, Taro Yasuma, Misaki Nakamura, Yasuhiko Hamada, Hiroyuki Inoue, and et al. 2021. "Increased Reflux Esophagitis after Helicobacter pylori Eradication Therapy in Cases Undergoing Endoscopic Submucosal Dissection for Early Gastric Cancer" Cancers 13, no. 8: 1779. https://doi.org/10.3390/cancers13081779
APA StyleKatsurahara, M., Imoto, I., Umeda, Y., Miura, H., Tsuboi, J., Yamada, R., Yasuma, T., Nakamura, M., Hamada, Y., Inoue, H., Tanaka, K., Horiki, N., Gabazza, E. C., & Takei, Y. (2021). Increased Reflux Esophagitis after Helicobacter pylori Eradication Therapy in Cases Undergoing Endoscopic Submucosal Dissection for Early Gastric Cancer. Cancers, 13(8), 1779. https://doi.org/10.3390/cancers13081779







