Abstract
Background: The loss of the Y chromosome in various malignant diseases has been described previously. There are no reliable information on the actual frequency, significance and homogeneity of Y chromosome loss (LoY) in esophageal adenocarcinoma (EAC). Methods: 400 male EAC including lymph-node metastases were analyzed with commercially available Y chromosome specific fluorescence in-situ probes. The results were correlated with molecular and immunohistochemical markers and clinicopathological aspects. Results: The entire cohort (n = 400) showed a singular LoY of one chromosome arm in 1.0% (q-arm) and 2.8% (p-arm), complete LoY in 52.5%. LoY was strongly associated with shortened overall-survival (OS). Patients with preserved Y chromosome had a median OS of 58.8 months, patients with LoY an OS of 19.4 months (p < 0.001). Multivariate analysis showed LoY as an independent prognostic marker with a hazard ratio of 1.835 (95% CI 1.233–2.725). LoY correlated with TP53 mutations (p = 0.003), KRAS amplification (p = 0.004), loss of ARID1a (p = 0.045) and presence of LAG3 (p = 0.018). Conclusions: Loss of the Y chromosome is a very common phenomenon in EAC. The LoY is heterogeneously distributed within the tumor, but corresponding lymph node metastases frequently show homogeneous LoY, indicating a selection and metastasizing advantage with poor prognosis. To date, the male predominance of EAC (7–9:1) is unclear, so genetic explanatory models are favored. The LoY in EAC may be biologically and functionally relevant and additional genomic or functional analyses are needed.
1. Introduction
The incidence of esophageal adenocarcinoma (EAC) has increased massively in recent decades, particularly in the Western world. Data from the United States show a 7-fold increase. In the past decades, there was an increase from 3.6 cases/1 million inhabitants in 1970s to 25.6/1 million inhabitants in 2006. A similar development is observed in Germany. The reason for this increase is unclear; a possible connection with rising obesity rates is discussed [1,2,3,4,5,6]. The vast majority of adenocarcinomas of the esophagus show a clear connection to the presence of a Barrett mucosa, which in turn is caused by chronic reflux of gastric acid and bile into the lower esophagus. While the majority of the studies describe a gender-neutral or up to twice as frequent occurrence of Barrett mucosa in men (maximum 2:1), the resulting adenocarcinoma shows an overwhelming male predominance of up to 9:1. The underlying reason for this gender-specific tumorigenesis is completely unclear and has not been investigated so far. A possible association with the androgen receptor signaling pathway has been discussed in the past with contradictory results [7,8,9]. In the past, the Y chromosome was presumed as limited to sex determination and spermatogenesis. Over the last years it has become evident that this disregard was false, as the Y chromosome is involved in many biological processes. An involvement is seen in coronary artery disease, including coronary artery disease, infectious diseases and, beyond hormonal influence, also autoimmune diseases [10,11,12,13,14]. Furthermore, the loss of the entire Y chromosome in various malignant diseases is a frequently described phenomenon. In bladder carcinoma, for example, loss of the Y chromosome is found in 30% of cases [15]. In older men, non tumor cells frequently show a loss of the Y chromosome - this is also discussed in the literature in connection with the overall higher propensity of the male sex for the development of a malignant disease [16]. Former studies considering only very small numbers of up to 20 cases of EAC have described the Y chromosome loss in up to 75% of the investigated cases [17,18,19,20,21].
To date, there is no reliable information on the actual frequency, significance for tumor progression including prognostic significance, the type of Y chromosome loss (isolated p-arm loss, isolated q-arm loss and complete Y chromosome loss) and the homogeneity of Y chromosome loss within the tumor in esophageal adenocarcinoma. Thus, we analyzed a very large tumor collective that includes 400 cases of EAC with different fluorescence in-situ probes and statistically analyzed them considering additional molecular tumor changes and clinical aspects.
2. Results
2.1. Patient Samples and Correlation with Clinical Data
The entire cohort considered for analysis consisted of 400 male patients with EAC. Female patients were excluded by definition. From a subset of patients, lymph node metastases corresponding to the primary tumors were included in the TMA. Within the patients’ cohort, lymph node metastases of 255 patients were available, and from these, 165 cases were analyzable for Y chromosome loss (LoY) (64.7%). Neoadjuvant treatment (either chemo-radiation or chemotherapy) was administered in 223/400 patients (55.8%). The median follow-up for the entire cohort was 57.7 months with a calculated 5-year survival rate of 26.6%. The patient collective analyzed corresponds to the normal distribution in the frequency of the individual tumor stages. In the analyzed patient collective, tumors of all stages could be analyzed (pT1–pT4) with an accumulation of pT3/pN+ tumors (Table 1).

Table 1.
Patients’ characteristics and clinicopathological data; results of immunohistochemical and molecular markers considering the Y chromosome status.
2.2. Y Chromosome Status in Cell Lines
Fluorescence in-situ hybridization (FISH) analysis of the cell lines fully agreed with the known Y chromosome status, thus the FISH probes used represent a reliable method for the analysis of the Y chromosome status (Figure 1).
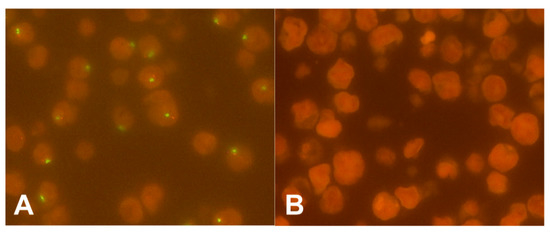
Figure 1.
FISH of esophageal adenocarcinoma cell lines: (A) Preserved Y chromosome signals of the short (red signal) and long (green signal) arm; (B) Complete loss of the Y chromosome (magnification ×630).
2.3. Y Chromosome Status in the Patients’ Cohort
Singular loss of one of the Y chromosome arms was present in 0.1% (q-arm, n = 4) and 2.8% (p-arm, n = 11), complete LoY in 52.5% (n = 210) (Figure 2). LoY in lymph nodes was found with a higher frequency (60.6%) but with no statistical difference to the LoY frequency detectable in primary tumors (p = 0.125). LoY was not correlated to administration of any kind of neoadjuvant treatment, neither in primary tumors nor in lymph node metastasis.
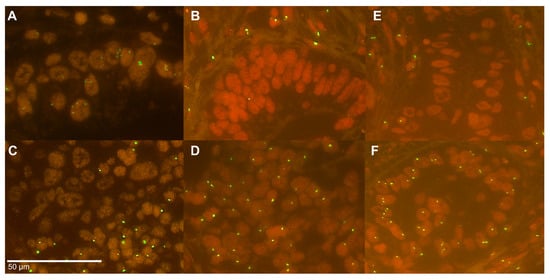
Figure 2.
FISH of the Tissue Microarray: (A) preserved Y chromosome signals of the short (red signal) and long (green signal) arm; (B,C) complete loss of the Y chromosome; (D) mosaic pattern of the Y chromosome with partial loss (only long arm with green signals); (E,F) heterogeneity of the Y chromosome with partly lost and preserved signals in the tumor cells (magnification ×630).
LoY was not associated with patients’ age (p = 0.124) or tumor stage (pT) (p = 0.314). In lymph node positive patients (pN+), LoY was seen in higher frequency 59.4%) than in nodal negative patients (p = 0.001) which is also reflected in an association with UICC stage and LoY (p = 0.015).
2.4. Heterogeneity of the Y Chromosome Status
We analyzed ten large tumor slides evaluating the intra-tumoral heterogeneity of the Y chromosome via FISH-analysis. Therefore, we screened the whole tumor for the signal distribution revealing areas with preserved and lost signals of the Y chromosome. We found a heterogeneous LoY in five cases, some with only focal heterogeneity (Figure 2E,F). Furthermore, one of the ten cases had a complete LoY, whereas two cases showed a complete loss of one arm of the Y chromosome. Two cases had a homogeneously preserved Y chromosome.
2.5. Y Chromosome Status and Correlation with Immunohistochemical and Molecular Markers
Analysis of the molecular marker profile showed a correlation between LoY and TP53 mutations and KRAS amplifications (p = 0.003 and p = 0.004, respectively), loss of chromatin remodeling protein ARID1a (SMARCA4) (p = 0.045) and presence of the immune checkpoint regulator of LAG3 (p = 0.018). A correlation with amplification of Her2/neu, PIK3CA and GATA6 and CD3 positive T-cells could not be revealed (Table 1).
2.6. LoY Correlation to Patients’ Outcome
LoY was strongly associated with shortened overall-survival (OS) in the entire patients´ cohort. Patients with presence of Y chromosome showed a median OS of 58.8 months (95% CI 33.1–83.2 months), patients with LoY an OS of 19.4 months (95% CI 14.8–24.0 months, p < 0.001) (Figure 3A). The survival difference is detectable both in patients that underwent upfront surgery without neoadjuvant treatment (median OS 117.7 months (95% CI 92.2–142.6 months) vs. 32.5 months (95% CI 13.1–51.9 months), p = 0.015) and in patients that received neoadjuvant treatment (median OS 41.3 months (95% CI 27.5–55.0 months) vs. 17.2 months (95% CI 12.6–22.0 months), p = 0.002) (Figure 3B,C).
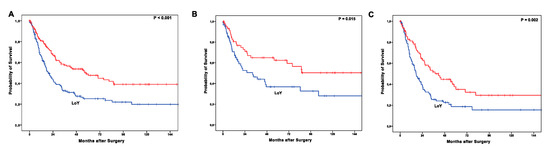
Figure 3.
Kaplan-Meier survival analysis showing shortened overall survival (OS) in case of LoY (blue line) vs. preserved Y chromosome (red line) in the entire patients’ cohort (A). The effect of impaired OS is detectable in both, patients after upfront surgery without neoadjuvant treatment (B) and in patients who received neoadjuvant treatment (C).
Interestingly, patients with preserved Y chromosome in combination with high numbers of CD3 positive tumor infiltrating T-cells (CD3+ high) showed a significantly prolonged overall- survival compared to the group with LoY and low CD3+ status. This is reflected in the Kaplan-Meier survival analysis to the effect that in the group with preserved Y chromosome and CD3+ high the median OS is not reached, whereas in the group LoY and CD3+ low a median OS of 24.6 months (95% CI 19.4–30.1 months, p < 0.001) is seen.
Next, we tested for independence of LoY as a prognostic marker using a multivariate cox regression model with age, tumor stage, lymph node metastases, and grading as covariates. LoY was seen as an independent prognostic marker with a hazard ratio of 1.835 (95%CI 1.835–2.725) (Table 2).

Table 2.
Multivariate cox regression demonstrating LoY as an independent prognostic marker for overall survival.
3. Discussion
The extent of Y-chromosome loss in adenocarcinomas of the esophagus has so far only been determined on small case numbers. With 400 adenocarcinomas analyzed, this study is by far the largest tumor cohort investigated. Our study validated that the complete loss of the Y chromosome is a common phenomenon in EAC.
It cannot be ruled out that the LoY in the tumor is a biologically-functionally irrelevant epiphenomenon that occurs as a general expression of a “complex karyotype” of esophageal carcinoma. This may be supported by the fact that the EAC is often characterized by genomic chaos, including genome duplication, chromotrypsis or pronounced telomere shortening, which contribute to complex chromosomal rearrangements [22]. TP53 mutations strongly correlate with instable genomes [23]. However, our multivariate regression analysis showed that LoY has a prognostic effect independent of TP53 status indicating that genome instability with its high diversity of selective advantages might not be the (only) cause for LoY manifestation in EAC. Further, the extremely high frequency of >50% of LoY suggests that there is a fitness advantage. To our knowledge, EAC represents the highest rate of LoY among solid tumors [15,24,25,26]. Cestari et al. found LoY in high grade dysplastic Barrett’s mucosa but not yet in low grade dysplastic or normal Barrett’s mucosa. This could indicate that LoY is biologically relevant for tumor initiation [18]. Overall, it seems well possible that LoY in EAC has an oncogenic effect. The LoY is described to be associated with shorter survival and risk of cancer in general. Some studies even suggest to use the LoY in blood cells in healthy elderly men as a predictive biomarker for carcinogenesis [16].
LoY is frequently seen in circulating leukocytes, where it can be not only linked to a higher risk of hematological malignancies but indicates also a higher risk for the development of solid cancer [16,27]. Thompson et al. recently detected a relevant overlap of LoY-risk loci with known cancer susceptibility loci and somatic drivers of tumor growth, proposing a connection between LoY and genomic instability [27]. The mechanisms and implications of the LoY are not clear, so different hypotheses are discussed. Possible mechanisms of rising LoY rates in age could be telomere shortening or that the gonosomes replicate lately in the s-phase being more vulnerable for shortening of the cell cycle [28,29]. The LoY also seems to influence signal pathways as smoking has a transient and mutagenic effect on the Y chromosome status [30]. This matches the theory of altered tissue microenvironment signaling influencing somatic evolution in an age-dependent manner [31]. The LoY might affect survival through defective immune functions of blood cells by disrupting immunosurveillance enabling tumor development and expansion [16,30]. The main risk factor for the development of EAC is the Barrett mucosa. Barrett mucosa occurs up to twice as frequently in men as in women. The resulting EAC shows a male predominance of 7 up to 9 to 1 (in our tumor collective 9 to 1). The cause of this gender shift in tumor initiation is unclear. There is currently no convincing evidence that gender-specific lifestyle aspects can explain this difference, so that genetic explanatory models are favored. Among other things, a possible role of the androgen pathways or protective aspects of female sex hormones are discussed. As explained above, LoY is already found in high-grade dysplastic Barrett mucosa and thus at an early stage of tumor initiation. LoY might render its cancer-promoting effect through tumor suppressor genes located on the X chromosome that have no homologous copy in males and that might respond to LoY by epigenetic silencing. In women, some tumor suppressor genes on the X chromosome escape X-inactivation. One study has shown that six of such genes show increased loss-of-function mutations in male tumor diseases (ATRX, CNKSR2, DDX3X, KDM5C, KDM6A, and MAGEC3) [26]. For KDM5D encoded on the X-chromosome, a direct interaction with the androgen receptor in the tumor cell nucleus could be demonstrated, and a reduction in the expression of KDM5D leads to disruption of the androgen receptor pathway [32].
Alternatively, the so-called pseudo-autosomal region (PAR) of the sex chromosomes could be relevant for tumor initiation in EAC. In this case, LoY would be advantageous for tumor development for the same reasons as mentioned above [26,33,34,35,36,37,38].
Whether a similar functional interaction is important in the development of EAC tumors should be subject of future analyses. Since sex chromosomes are commonly excluded from copy number analyses of comprehensive next generation sequencing-based analyses, it is plausible that LoY has not been recognized as a relevant factor in large genomic studies on EAC [26].
Interestingly, here we found a statistically significant association between tumors with LoY and loss of expression of ARID1a, a member of the relevant SWI-SNF chromatin remodeling complex. This trend towards mutual exclusivity between LoY and SWI-SNF inactivation might indicate that LoY has an epigenetic effect that reduces the additional selective advantage of other epigenetic alterations on cancer development.
Considering the phenomenon of LoY apparently correlating with LAG3 as a marker of the tumor microenvironment in EAC could also implicate a direct influence of Y deficient leukocytes on the tumor immune microenvironment in cancer. Former studies linked the Y chromosome to infectious and autoimmune diseases indicating direct influence of the Y chromosome on the immune system [10,13,14,16]. For example, the development of NK-T-cells is directly associated with Y chromosome-linked factors [11]. Our present study has few limitations, as we performed no analysis of the Y chromosome status of blood cells. Such analyses, however, might help enlightening the link between the Y chromosome and the immune system in cancer. LoY is known to be the most common acquired mutation in healthy males with a rising prevalence with age and is found in up to 20% of men older than 80 years [39]. For colorectal, prostate, bladder and lung cancer studies showed a significant higher frequency of LoY in blood cells compared to healthy controls [40,41]. A further limitation is that the high frequency of LoY in fully developed EAC does not explain the gender specificity of the tumor, as the Y chromosome status in Barrett´s mucosa without dysplasia has not been analyzed in large cohorts yet [20,42,43].
4. Materials and Methods
4.1. Patients and Tumor Samples
We analyzed formalin-fixed, paraffin embedded (FFPE) material from 400 male patients with EAC who underwent primary surgical resection or resection after neoadjuvant therapy at the Department of General, Visceral and Cancer Surgery, University of Cologne, Cologne, Germany. The standard surgical procedure was laparotomic or laparoscopic gastrolysis and right transthoracic en bloc esophagectomy including two-field lymphadenectomy of mediastinal and abdominal lymph nodes. Reconstruction was performed by high intrathoracic esophagogastrostomy as described previously [44,45]. Patients with advanced esophageal cancer (cT3, cNx, M0 or cN+, M0) received preoperative chemoradiation (5-FU, cisplatin, 40 Gy as treated in the area prior the CROSS trial) or chemotherapy alone. All patients were followed up according to a standardized protocol. During the first 2 years, patients were followed up clinically in the hospital every 3 months. Afterwards, annual exams were carried out. Follow-up examinations included a detailed history, clinical evaluation, abdominal ultrasound, chest X-ray and additional diagnostic procedures as required. Follow-up data were available for all patients. Patient characteristics are given in Table 1. Depending on the effect of neoadjuvant chemo- or radiochemotherapy, there is a preponderance of minor responders in the TMAs, defined as histopathological residual tumour of ≥10% [46].
All procedures followed the national and institutional ethical standards and were in accordance with the relevant version of the Helsinki Declaration. Informed and ethical approved consent from the local ethics committee (13-091) was obtained from all included patients.
4.2. Cell Culture
Commercially available FISH probes were tested and validated in different esophageal cell lines of known Y chromosome status. Immortalized human normal esophageal squamous epithelium cell lines EPC1-hTERT (XY) and EPC2-hTERT (XY) were gifts by Dr. René Thieme (Gockel laboratory, Leipzig). EPC-1 and EPC-2 cells were cultivated in Keratinocyte-SFM (KSFM) medium (Gibco, ThermoFisher Scientific, Waltham, MA, USA) supplemented with bovine pituitary extract (50 μg/mL) (Gibco, ThermoFisher Scientific, Waltham, MA, USA), and human recombinant epidermal growth factor (EGF) (1 ng/mL) (Gibco, ThermoFisher Scientific, Waltham, MA, USA). Following dissociation during subculturing, trypsin activity was blocked by soybean trypsin inhibitor (STI) (Sigma-Aldrich, Schnelldorf, Germany). The metaplastic cell line CP-A (XY) and the dysplastic cell line CP-B (XY) were purchased from ATCC and cultivated according to manufacturer’s recommendations. Esophageal adenocarcinoma cell lines OE-33 (X0), OE-19 (X0), Eso-26 (XY) and OAC-P4C (X0) were available from the German Collection of Microorganisms and Cell Cultures GmbH (DSMZ, Leibnitz Institute, Braunschweig, Germany). All EAC cell lines were cultivated in RPMI 1640 GlutaMax growth medium (Gibco, ThermoFisher Scientific, Waltham, MA, USA), supplemented with 10% fetal bovine serum (FBS). All cells were incubated at 37 °C and 5% CO2.
4.3. Formalin-Fixation and Paraffin-Embedding of Cell Line Pellets
Cells of 80% confluency were harvested by trypsinization (0.25% Trypsin + EDTA) (Gibco, ThermoFisher Scientific, Waltham, MA, USA), washed and resuspended in 4% paraformaldehyde (Walter-CMP, Ratingen, Germany). Cells were fixed overnight at 4 °C. Fixed cells were washed at room temperature (1200 rcf, 15 min) and resuspended in EtOH (97%). After adding three drops of protein glycerol (Carl Roth, Karlsruhe, Germany), cells were vortexed and subsequently spun (1200 rcf, 15 min). The cell pellet was dehydrated and embedded in paraffin.
4.4. TMA Construction
TMA construction was performed as previously described [47,48]. In brief, tissue cylinders with a diameter of 1.2 mm each were punched from selected tumor tissue blocks using a self-constructed semiautomated precision instrument and embedded in empty recipient paraffin blocks. Consecutive sections of the resulting TMA blocks were transferred to an adhesive-coated slide system (Instrumedics Inc., Hackensack, NJ) for immunohistochemistry and FISH.
4.5. Fluorescence In Situ Hybridization
FISH analysis of Y chromosome was performed with a panel containing probes for long and short arms of the Y chromosome (Abbott Molecular, Wiesbaden, Germany). Detailed procedures of FISH analysis were described elsewhere [49]. In brief, three-μm tissue sections (SuperFrost Plus) were mounted by heating at 56 °C, followed by semi-automated deparaffinization protease digestion washing steps (VP2000 processor system, Abbott Molecular, Germany) with the ready-to-use FISH pretreatment kit (Vysis IntelliFISH Universal FFPE Tissue Pretreatment Protease; Abbott Molecular, Wiesbaden, Germany). Hybridization at 37°C was done overnight with the FISH probes, followed by DAPI staining. Non tumor epithelial tissue and fibroblasts served as on-slide internal positive control except for the analysis of the cell lines, where no internal control was available by definition. Further analysis of the Y chromosome for the TMA and large tumor slides was only performed with internal positive control exhibiting clearly distinct signals of each color. The tumor spots were screened for existence or absence of green (long arm; Yq12) and orange (short arm; Yp11.3) signals. Complete loss of both was defined as loss of the Y chromosome, whereas loss in some tumor cells was defined as a mosaic pattern. Loss of one color (green or orange) was defined as partial Y chromosome loss.
Further FISH analyses were performed on TMA slides for gene amplification of KRAS, PIK3CA, Her2/neu (ERBB2) and GATA6. A detailed description of the analysis of KRAS, PIK3CA, Her2/neu and GATA6 is already published [50,51,52].
4.6. Analysis of Heterogeneity of Y Chromosome Status
To address the question of the distribution of the Y chromosome within the tumor we further performed FISH analyses on 10 large tumor slides evaluating the heterogeneity of the Y chromosome status. The analysis itself was done analogously to the TMAs. We chose cases where we observed differences of Y chromosome in the tumor and the lymph node metastasis on the TMA. We screened the whole tumor for the signal distribution of the short and long arm of the Y chromosome revealing areas with preserved and lost signals.
4.7. Immunohistochemistry
Immunohistochemistry was performed on TMA slides evaluating the expression of CD3, TP53, LAG3, ARID1a and BRG1. A detailed description of the analysis for CD3, TP53, ARID1a and BRG1 is published [45,53]. For CD3 the rabbit monoclonal antibody (SP7; Thermo Fisher Scientific, MA, USA) and for LAG3 the rabbit monoclonal antibody for LAG3 (D2G40; Cell Signaling Technology Europe) was used on the Leica BOND-MAX Stainer (Leica Biosystems, Wetzlar, Germany). For CD3, <50 positive T-lymphocytes/mm2 were defined as low positive, >50 as highly positive, and for LAG3 expression in <1% lymphocytes were defined as negative and ≥1% was assessed as positive.
4.8. Data Analysis and Statistics
The current retrospective study was performed with the approval of the Ethics Committee of the University of Cologne, utilizing clinical data that was collected prospectively according to a standardized protocol.
Univariate analysis was conducted for tables using chi-squared statistics or Fisher’s exact test if necessary. Prognosis was calculated including all types of mortality beginning on the date of surgery. Kaplan–Meier univariate analysis was used to describe survival distribution, and log-rank tests were used to evaluate survival differences. Cox proportional hazard regression with sequential backward elimination of the non-significant variables was used to analyze the effect of several risk factors on survival. Survival analysis and the multivariate cox-regression model was performed on the entire cohort (test + validation cohort).
Statistical analyses were carried out using IBM SPSS v22.0 (IBM Corporation, Armonk, NY, USA).
5. Conclusions
Taken together, here we validated the high frequency of LoY in a large cohort of EAC. EAC patients with LoY have a poor prognosis and show correlation with the immune checkpoint protein LAG3, suggesting a direct influence of Y deficiency on the tumor immune microenvironment. Further, we observe a correlation between LoY and inactivation of SWI/SNF components raising the possibility of an epigenetic effect of LoY. Our data suggest that LoY has to be seen in a larger functional context. Understanding this context may shed light on the gender bias of EAC incidence.
Author Contributions
Conceptualization, H.L., A.Q. and A.M.H.; methodology, H.L. and C.B.W.; validation, A.Q. and H.L.; formal analysis, H.L. and C.B.W.; investigation, H.L., C.B.W. and F.G.; resources, T.Z., W.S., R.B., C.J.B. and H.A.; data curation, F.G. and S.-H.C.; writing—original draft preparation, H.L., C.B.W. and A.Q.; writing—review and editing, R.B. and C.J.B.; visualization, H.L. and F.G.; supervision, A.Q. and A.M.H.; project administration, A.Q. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
We thank the Winand Dinjens laboratory, Rotterdam, for their advice and support on tissue culture experiments. Furthermore, we thank Wiebke Jeske and Elke Binot for their always prompt and excellent technical support. H.L., H.A., S.H.C., T.Z. and A.Q. are part of the Gastrointestinal Cancer Group Cologne (GCGC), which is a clinical working group.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lepage, C.; Rachet, B.; Jooste, V.; Faivre, J.; Coleman, M.P. Continuing rapid increase in esophageal adenocarcinoma in England and Wales. Am. J. Gastroenterol. 2008, 103, 2694–2699. [Google Scholar] [CrossRef] [PubMed]
- Edgren, G.; Adami, H.O.; Weiderpass, E.; Nyren, O. A global assessment of the oesophageal adenocarcinoma epidemic. Gut 2013, 62, 1406–1414. [Google Scholar] [CrossRef]
- Pohl, H.; Sirovich, B.; Welch, H.G. Esophageal adenocarcinoma incidence: Are we reaching the peak? Cancer Epidemiol. Biomarkers Prev. 2010, 19, 1468–1470. [Google Scholar] [CrossRef]
- Barrett, J.C. Mechanisms of multistep carcinogenesis and carcinogen risk assessment. Environ. Health Perspect 1993, 100, 9–20. [Google Scholar] [CrossRef]
- Koch-Institut, R. Krebs in Deutschland für 2013/2014. Available online: https://edoc.rki.de/handle/176904/3270 (accessed on 30 June 2020).
- Tustumi, F.; Kimura, C.M.; Takeda, F.R.; Uema, R.H.; Salum, R.A.; Ribeiro-Junior, U.; Cecconello, I. Prognostic Factors and Survival Analysis in Esophageal Carcinoma. Arq. Bras. Cir. Dig. 2016, 29, 138–141. [Google Scholar] [CrossRef]
- Sukocheva, O.A.; Li, B.; Due, S.L.; Hussey, D.J.; Watson, D.I. Androgens and esophageal cancer: What do we know? World J. Gastroenterol. 2015, 21, 6146–6156. [Google Scholar] [CrossRef]
- Palethorpe, H.M.; Drew, P.A.; Smith, E. Androgen Signaling in Esophageal Adenocarcinoma Cell Lines In Vitro. Dig. Dis. Sci. 2017, 62, 3402–3414. [Google Scholar] [CrossRef]
- Asanuma, K.; Iijima, K.; Shimosegawa, T. Gender difference in gastro-esophageal reflux diseases. World J. Gastroenterol. 2016, 22, 1800–1810. [Google Scholar] [CrossRef]
- Case, L.K.; Wall, E.H.; Dragon, J.A.; Saligrama, N.; Krementsov, D.N.; Moussawi, M.; Zachary, J.F.; Huber, S.A.; Blankenhorn, E.P.; Teuscher, C. The Y chromosome as a regulatory element shaping immune cell transcriptomes and susceptibility to autoimmune disease. Genome. Res. 2013, 23, 1474–1485. [Google Scholar] [CrossRef] [PubMed]
- Wesley, J.D.; Tessmer, M.S.; Paget, C.; Trottein, F.; Brossay, L. A Y chromosome-linked factor impairs NK T development. J. Immunol. 2007, 179, 3480–3487. [Google Scholar] [CrossRef]
- Charchar, F.J.; Bloomer, L.D.; Barnes, T.A.; Cowley, M.J.; Nelson, C.P.; Wang, Y.; Denniff, M.; Debiec, R.; Christofidou, P.; Nankervis, S.; et al. Inheritance of coronary artery disease in men: An analysis of the role of the Y chromosome. Lancet 2012, 379, 915–922. [Google Scholar] [CrossRef]
- Teuscher, C.; Noubade, R.; Spach, K.; McElvany, B.; Bunn, J.Y.; Fillmore, P.D.; Zachary, J.F.; Blankenhorn, E.P. Evidence that the Y chromosome influences autoimmune disease in male and female mice. Proc. Natl. Acad. Sci. USA 2006, 103, 8024–8029. [Google Scholar] [CrossRef] [PubMed]
- Case, L.K.; Toussaint, L.; Moussawi, M.; Roberts, B.; Saligrama, N.; Brossay, L.; Huber, S.A.; Teuscher, C. Chromosome y regulates survival following murine coxsackievirus b3 infection. G3 (Bethesda) 2012, 2, 115–121. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sauter, G.; Moch, H.; Wagner, U.; Novotna, H.; Gasser, T.C.; Mattarelli, G.; Mihatsch, M.J.; Waldman, F.M. Y chromosome loss detected by FISH in bladder cancer. Cancer Genet. Cytogenet 1995, 82, 163–169. [Google Scholar] [CrossRef]
- Forsberg, L.A. Loss of chromosome Y (LOY) in blood cells is associated with increased risk for disease and mortality in aging men. Hum. Genet. 2017, 136, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Persons, D.L.; Croughan, W.S.; Borelli, K.A.; Cherian, R. Interphase cytogenetics of esophageal adenocarcinoma and precursor lesions. Cancer Genet. Cytogenet 1998, 106, 11–17. [Google Scholar] [CrossRef]
- Cestari, R.; Villanacci, V.; Rossi, E.; Della Casa, D.; Missale, G.; Conio, M.; Grigolato, P.; Bassotti, G. Fluorescence in situ hybridization to evaluate dysplasia in Barrett’s esophagus: A pilot study. Cancer Lett. 2007, 251, 278–287. [Google Scholar] [CrossRef]
- Doak, S.H.; Jenkins, G.J.; Parry, E.M.; D’Souza, F.R.; Griffiths, A.P.; Toffazal, N.; Shah, V.; Baxter, J.N.; Parry, J.M. Chromosome 4 hyperploidy represents an early genetic aberration in premalignant Barrett’s oesophagus. Gut 2003, 52, 623–628. [Google Scholar] [CrossRef]
- Beuzen, F.; Dubois, S.; Flejou, J.F. Chromosomal numerical aberrations are frequent in oesophageal and gastric adenocarcinomas: A study using in-situ hybridization. Histopathology 2000, 37, 241–249. [Google Scholar] [CrossRef]
- Altaf, K.; Xiong, J.J.; la Iglesia, D.; Hickey, L.; Kaul, A. Meta-analysis of biomarkers predicting risk of malignant progression in Barrett’s oesophagus. Br. J. Surg. 2017, 104, 493–502. [Google Scholar] [CrossRef]
- Nones, K.; Waddell, N.; Wayte, N.; Patch, A.M.; Bailey, P.; Newell, F.; Holmes, O.; Fink, J.L.; Quinn, M.C.J.; Tang, Y.H.; et al. Genomic catastrophes frequently arise in esophageal adenocarcinoma and drive tumorigenesis. Nat. Commun. 2014, 5, 5224. [Google Scholar] [CrossRef] [PubMed]
- Eischen, C.M. Genome Stability Requires p53. Cold Spring Harb. Perspect. Med. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Agahozo, M.C.; Timmermans, M.A.M.; Sleddens, H.F.B.M.; Foekens, R.; Trapman-Jansen, A.M.A.C.; Schröder, C.P.; van Leeuwen-Stok, E.; Martens, J.W.M.; Dinjens, W.N.M.; van Deurzen, C.H.M. Loss of Y-Chromosome during Male Breast Carcinogenesis. Cancers 2020, 12, 631. [Google Scholar] [CrossRef]
- Hollows, R.; Wei, W.; Cazier, J.B.; Mehanna, H.; Parry, G.; Halford, G.; Murray, P. Association between loss of Y chromosome and poor prognosis in male head and neck squamous cell carcinoma. Head Neck 2019, 41, 993–1006. [Google Scholar] [CrossRef]
- Arseneault, M.; Monlong, J.; Vasudev, N.S.; Laskar, R.S.; Safisamghabadi, M.; Harnden, P.; Egevad, L.; Nourbehesht, N.; Panichnantakul, P.; Holcatova, I.; et al. Loss of chromosome Y leads to down regulation of KDM5D and KDM6C epigenetic modifiers in clear cell renal cell carcinoma. Sci. Rep. 2017, 7, 44876. [Google Scholar] [CrossRef]
- Thompson, D.J.; Genovese, G.; Halvardson, J.; Ulirsch, J.C.; Wright, D.J.; Terao, C.; Davidsson, O.B.; Day, F.R.; Sulem, P.; Jiang, Y.; et al. Genetic predisposition to mosaic Y chromosome loss in blood. Nature 2019, 575, 652–657. [Google Scholar] [CrossRef]
- Persani, L.; Bonomi, M.; Lleo, A.; Pasini, S.; Civardi, F.; Bianchi, I.; Campi, I.; Finelli, P.; Miozzo, M.; Castronovo, C.; et al. Increased loss of the Y chromosome in peripheral blood cells in male patients with autoimmune thyroiditis. J. Autoimmun. 2012, 38, J193–J196. [Google Scholar] [CrossRef]
- Stone, J.F.; Sandberg, A.A. Sex chromosome aneuploidy and aging. Mutat. Res. 1995, 338, 107–113. [Google Scholar] [CrossRef]
- Dumanski, J.P.; Rasi, C.; Lonn, M.; Davies, H.; Ingelsson, M.; Giedraitis, V.; Lannfelt, L.; Magnusson, P.K.; Lindgren, C.M.; Morris, A.P.; et al. Mutagenesis. Smoking is associated with mosaic loss of chromosome Y. Science 2015, 347, 81–83. [Google Scholar] [CrossRef]
- Rozhok, A.I.; DeGregori, J. Toward an evolutionary model of cancer: Considering the mechanisms that govern the fate of somatic mutations. Proc. Nat. Acad. Sci. USA 2015, 112, 8914–8921. [Google Scholar] [CrossRef]
- Komura, K.; Jeong, S.H.; Hinohara, K.; Qu, F.; Wang, X.; Hiraki, M.; Azuma, H.; Lee, G.S.; Kantoff, P.W.; Sweeney, C.J. Resistance to docetaxel in prostate cancer is associated with androgen receptor activation and loss of KDM5D expression. Proc. Natl. Acad. Sci. USA 2016, 113, 6259–6264. [Google Scholar] [CrossRef]
- Dunford, A.; Weinstock, D.M.; Savova, V.; Schumacher, S.E.; Cleary, J.P.; Yoda, A.; Sullivan, T.J.; Hess, J.M.; Gimelbrant, A.A.; Beroukhim, R.; et al. Tumor-suppressor genes that escape from X-inactivation contribute to cancer sex bias. Nat. Genet. 2017, 49, 10–16. [Google Scholar] [CrossRef]
- Vaz, C.V.; Correia, S.; Cardoso, H.J.; Figueira, M.I.; Marques, R.; Maia, C.J.; Socorro, S. The Emerging Role of Regucalcin as a Tumor Suppressor: Facts and Views. Curr. Mol. Med. 2016, 16, 607–619. [Google Scholar] [CrossRef]
- Weng, S.; Stoner, S.A.; Zhang, D.E. Sex chromosome loss and the pseudoautosomal region genes in hematological malignancies. Oncotarget 2016, 7, 72356–72372. [Google Scholar] [CrossRef]
- Huang, C.Y.; Chen, Y.M.; Zhao, J.J.; Chen, Y.B.; Jiang, S.S.; Yan, S.M.; Zhao, B.W.; Pan, K.; Wang, D.D.; Lv, L.; et al. Decreased expression of transcription elongation factor A-like 7 is associated with gastric adenocarcinoma prognosis. PLoS ONE 2013, 8, e54671. [Google Scholar] [CrossRef] [PubMed]
- Chien, J.; Narita, K.; Rattan, R.; Giri, S.; Shridhar, R.; Staub, J.; Beleford, D.; Lai, J.; Roberts, L.R.; Molina, J.; et al. A role for candidate tumor-suppressor gene TCEAL7 in the regulation of c-Myc activity, cyclin D1 levels and cellular transformation. Oncogene 2008, 27, 7223–7234. [Google Scholar] [CrossRef][Green Version]
- Wong, H.Y.; Wang, G.M.; Croessmann, S.; Zabransky, D.J.; Chu, D.; Garay, J.P.; Cidado, J.; Cochran, R.L.; Beaver, J.A.; Aggarwal, A.; et al. TMSB4Y is a candidate tumor suppressor on the Y chromosome and is deleted in male breast cancer. Oncotarget 2015, 6, 44927–44940. [Google Scholar] [CrossRef]
- Dumanski, J.P.; Lambert, J.C.; Rasi, C.; Giedraitis, V.; Davies, H.; Grenier-Boley, B.; Lindgren, C.M.; Campion, D.; Dufouil, C.; The European Alzheimer’s Disease Initiative Investigators; et al. Mosaic Loss of Chromosome Y in Blood Is Associated with Alzheimer Disease. Am. J. Hum. Genet. 2016, 98, 1208–1219. [Google Scholar] [CrossRef]
- Korkmaz, D.T.; Demirhan, O.; Abat, D.; Demirberk, B.; Tunc, E.; Kuleci, S. Microchimeric Cells, Sex Chromosome Aneuploidies and Cancer. Pathol. Oncol. Res. 2015, 21, 1157–1165. [Google Scholar] [CrossRef]
- Noveski, P.; Madjunkova, S.; Sukarova Stefanovska, E.; Matevska Geshkovska, N.; Kuzmanovska, M.; Dimovski, A.; Plaseska-Karanfilska, D. Loss of Y Chromosome in Peripheral Blood of Colorectal and Prostate Cancer Patients. PLoS ONE 2016, 11, e0146264. [Google Scholar] [CrossRef]
- Menke-Pluymers, M.B.; van Drunen, E.; Vissers, K.J.; Mulder, A.H.; Tilanus, H.W.; Hagemeijer, A. Cytogenetic analysis of Barrett’s mucosa and adenocarcinoma of the distal esophagus and cardia. Cancer Genet. Cytogenet. 1996, 90, 109–117. [Google Scholar] [CrossRef]
- Chaves, P.; Crespo, M.; Ribeiro, C.; Laranjeira, C.; Pereira, A.D.; Suspiro, A.; Cardoso, P.; Leitao, C.N.; Soares, J. Chromosomal analysis of Barrett’s cells: Demonstration of instability and detection of the metaplastic lineage involved. Mod. Pathol. 2007, 20, 788–796. [Google Scholar] [CrossRef] [PubMed]
- Holscher, A.H.; Schneider, P.M.; Gutschow, C.; Schroder, W. Laparoscopic ischemic conditioning of the stomach for esophageal replacement. Ann. Surg. 2007, 245, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Schallenberg, S.; Bork, J.; Essakly, A.; Alakus, H.; Buettner, R.; Hillmer, A.M.; Bruns, C.; Schroeder, W.; Zander, T.; Loeser, H.; et al. Loss of the SWI/SNF-ATPase subunit members SMARCF1 (ARID1A), SMARCA2 (BRM), SMARCA4 (BRG1) and SMARCB1 (INI1) in oesophageal adenocarcinoma. BMC Cancer 2020, 20, 12. [Google Scholar] [CrossRef]
- Schneider, P.M.; Metzger, R.; Schaefer, H.; Baumgarten, F.; Vallbohmer, D.; Brabender, J.; Wolfgarten, E.; Bollschweiler, E.; Baldus, S.E.; Dienes, H.P.; et al. Response evaluation by endoscopy, rebiopsy, and endoscopic ultrasound does not accurately predict histopathologic regression after neoadjuvant chemoradiation for esophageal cancer. Ann. Surg. 2008, 248, 902–908. [Google Scholar] [CrossRef]
- Helbig, D.; Ihle, M.A.; Putz, K.; Tantcheva-Poor, I.; Mauch, C.; Buttner, R.; Quaas, A. Oncogene and therapeutic target analyses in atypical fibroxanthomas and pleomorphic dermal sarcomas. Oncotarget 2016, 7, 21763–21774. [Google Scholar] [CrossRef]
- Simon, R.; Mirlacher, M.; Sauter, G. Tissue microarrays. Methods Mol. Med. 2005, 114, 257–268. [Google Scholar] [CrossRef]
- Loeser, H.; Waldschmidt, D.; Kuetting, F.; Heydt, C.; Zander, T.; Plum, P.; Alakus, H.; Buettner, R.; Quaas, A. Copy-number variation and protein expression of DOT1L in pancreatic adenocarcinoma as a potential drug target. Mol. Clin. Oncol. 2017, 6, 639–642. [Google Scholar] [CrossRef][Green Version]
- Essakly, A.; Loeser, H.; Kraemer, M.; Alakus, H.; Chon, S.H.; Zander, T.; Buettner, R.; Hillmer, A.M.; Bruns, C.J.; Schroeder, W.; et al. PIK3CA and KRAS Amplification in Esophageal Adenocarcinoma and their Impact on the Inflammatory Tumor Microenvironment and Prognosis. Transl. Oncol. 2019, 13, 157–164. [Google Scholar] [CrossRef]
- Plum, P.S.; Gebauer, F.; Kramer, M.; Alakus, H.; Berlth, F.; Chon, S.H.; Schiffmann, L.; Zander, T.; Buttner, R.; Holscher, A.H.; et al. HER2/neu (ERBB2) expression and gene amplification correlates with better survival in esophageal adenocarcinoma. BMC Cancer 2019, 19, 38. [Google Scholar] [CrossRef]
- Schiffmann, L.M.; Loeser, H.; Jacob, A.S.; Maus, M.; Fuchs, H.; Zhao, Y.; Tharun, L.; Essakly, A.; Iannos Damanakis, A.; Zander, T.; et al. Dickkopf-2 (DKK2) as Context Dependent Factor in Patients with Esophageal Adenocarcinoma. Cancers 2020, 12, 451. [Google Scholar] [CrossRef] [PubMed]
- Quaas, A.; Heydt, C.; Gebauer, F.; Alakus, H.; Loeser, H.; Buettner, R.; Hillmer, A.; Bruns, C.; Merkelbach-Bruse, S.; Zander, T.; et al. Genomic Characterization of TP53-Wild-Type Esophageal Carcinoma. Transl. Oncol. 2019, 12, 154–161. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).