A Pilot Study Comparing the Efficacy of Lactate Dehydrogenase Levels Versus Circulating Cell-Free microRNAs in Monitoring Responses to Checkpoint Inhibitor Immunotherapy in Metastatic Melanoma Patients
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
2.1. LDH Levels at Treatment Baseline as a Predictive Factor for CII Response in Metastatic Melanoma Patients
2.2. Identification of cfmiRs in Pre- and Post-Treatment Samples from Patients Non-Responsive to CII
2.3. MiR-615-3p Correlates with Melanoma Response to CII
2.4. A cfmiR Signature to Assess CII Responses in Stage III Melanoma Patients
3. Discussion
4. Materials and Methods
4.1. Consent to Participate and Patient Specimen Accrual
4.2. Blood Sample Collection
4.3. CII Response
4.4. Sample Processing for HTG WTA
4.5. Biostatistical Analysis
4.6. Data Deposit
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AJCC | American Joint Committee on Cancer |
| CfmiR | Cell-free miR |
| CfDNA | Cell-free DNA |
| CfNA | Cell-free Nucleic Acids |
| CII | Checkpoint Inhibitor Immunotherapy |
| CR | Complete Response |
| CTLA-4 | Cytotoxic T Lymphocyte-Associated Antigen 4 |
| IRAE | Immune-Related Adverse Events |
| DE | Differentially Expressed |
| FC | Fold-Change |
| FDR | False Discovery Rate |
| GEO | Gene Expression Omnibus |
| MBM | Melanoma Brain Metastasis |
| miR | MicroRNA |
| NED | No Evidence of Disease |
| NGS | Next-Generation Sequencing |
| PD-1 | Programmed Cell Death Protein-1 |
| PD-L1 | Programmed Cell Death Protein-1 Ligand |
| PD | Progressive Disease |
| PFS | Progressive-Free Survival |
| PR | Partial Response |
| NS | Non-Significant |
| RECIST | Response Evaluation Criteria In Solid Tumors 1.1 |
| REMARK | Reporting Recommendations For Tumour Marker |
| SD | Stable Disease |
| LDH | Lactate Dehydrogenase |
| ULN | Upper Limit Normal |
| WTA | Whole Transcriptome Assay |
References
- Berk-Krauss, J.; Stein, J.A.; Weber, J.; Polsky, D.; Geller, A.C. New Systematic Therapies and Trends in Cutaneous Melanoma Deaths among US Whites, 1986–2016. Am. J. Public Health 2020, 110, 731–733. [Google Scholar] [CrossRef]
- Robert, C.; Thomas, L.; Bondarenko, I.; O’Day, S.; Weber, J.; Garbe, C.; Lebbe, C.; Baurain, J.-F.; Testori, A.; Grob, J.-J.; et al. Ipilimumab plus Dacarbazine for Previously Untreated Metastatic Melanoma. N. Engl. J. Med. 2011, 364, 2517–2526. [Google Scholar] [CrossRef] [PubMed]
- Schachter, J.; Ribas, A.; Long, G.V.; Arance, A.; Grob, J.J.; Mortier, L.; Daud, A.; Carlino, M.S.; McNeil, C.; Lotem, M.; et al. Pembrolizumab versus ipilimumab for advanced melanoma: Final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet 2017, 390, 1853–1862. [Google Scholar] [CrossRef]
- Ribas, A.; Lawrence, D.; Atkinson, V.; Agarwal, S.; Miller, W.H., Jr.; Carlino, M.S.; Fisher, R.; Long, G.V.; Hodi, F.S.; Tsoi, J.; et al. Combined BRAF and MEK inhibition with PD-1 blockade immunotherapy in BRAF-mutant melanoma. Nat. Med. 2019, 25, 936–940. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Ribas, A.; Hamid, O.; Daud, A.; Wolchok, J.; Joshua, A.M.; Hwu, W.-J.; Weber, J.S.; Gangadhar, T.C.; Joseph, R.W.; et al. Durable Complete Response After Discontinuation of Pembrolizumab in Patients With Metastatic Melanoma. J. Clin. Oncol. 2018, 36, 1668–1674. [Google Scholar] [CrossRef]
- Haslam, A.; Prasad, V. Estimation of the Percentage of US Patients With Cancer Who Are Eligible for and Respond to Checkpoint Inhibitor Immunotherapy Drugs. JAMA Netw. Open 2019, 2, e192535. [Google Scholar] [CrossRef]
- Mckean, W.B.; Moser, J.C.; Rimm, D.; Hu-Lieskovan, S. Biomarkers in Precision Cancer Immunotherapy: Promise and Challenges. Am. Soc. Clin. Oncol. Educ. Book 2020, 40, e275–e291. [Google Scholar] [CrossRef]
- Gide, T.N.; Wilmott, J.S.; Scolyer, R.A.; Long, G.V. Primary and Acquired Resistance to Immune Checkpoint Inhibitors in Metastatic Melanoma. Clin. Cancer Res. 2017, 24, 1260–1270. [Google Scholar] [CrossRef]
- Van Wilpe, S.; Koornstra, R.; Brok, M.D.; De Groot, J.W.; Blank, C.; De Vries, J.; Gerritsen, W.; Mehra, N. Lactate dehydrogenase: A marker of diminished antitumor immunity. OncoImmunology 2020, 9, 1731942. [Google Scholar] [CrossRef]
- Mishra, D.; Banerjee, D. Lactate Dehydrogenases as Metabolic Links between Tumor and Stroma in the Tumor Microenvironment. Cancers 2019, 11, 750. [Google Scholar] [CrossRef]
- Amin, M.B.; Greene, F.L.; Edge, S.B.; Compton, C.C.; Gershenwald, J.E.; Brookland, R.K.; Meyer, L.; Gress, D.M.; Byrd, D.R.; Winchester, D.P. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J. Clin. 2017, 67, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Egberts, F.; Hitschler, W.N.; Weichenthal, M.; Hauschild, A. Prospective monitoring of adjuvant treatment in high-risk melanoma patients: Lactate dehydrogenase and protein S-100B as indicators of relapse. Melanoma Res. 2009, 19, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Kelderman, S.; Heemskerk, B.; Van Tinteren, H.; Brom, R.R.H.V.D.; Hospers, G.A.P.; Eertwegh, A.J.M.V.D.; Kapiteijn, E.W.; De Groot, J.W.B.; Soetekouw, P.; Jansen, R.L.; et al. Lactate dehydrogenase as a selection criterion for ipilimumab treatment in metastatic melanoma. Cancer Immunol. Immunother. 2014, 63, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Wagner, N.B.; Forschner, A.; Leiter, U.; Garbe, C.; Eigentler, T. S100B and LDH as early prognostic markers for response and overall survival in melanoma patients treated with anti-PD-1 or combined anti-PD-1 plus anti-CTLA-4 antibodies. Br. J. Cancer 2018, 119, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Diem, S.; Kasenda, B.; Spain, L.; Martin-Liberal, J.; Marconcini, R.; Gore, M.; Larkin, J. Serum lactate dehydrogenase as an early marker for outcome in patients treated with anti-PD-1 therapy in metastatic melanoma. Br. J. Cancer 2016, 114, 256–261. [Google Scholar] [CrossRef]
- Lin, S.Y.; Huang, S.K.; Huynh, K.T.; Salomon, M.P.; Chang, S.-C.; Marzese, D.M.; Lanman, R.B.; Talasaz, A.; Hoon, D.S. Multiplex Gene Profiling of Cell-Free DNA in Patients With Metastatic Melanoma for Monitoring Disease. JCO Precis. Oncol. 2018, 2, 1–30. [Google Scholar] [CrossRef]
- Goh, J.Y.; Feng, M.; Wang, W.; Oguz, G.; Yatim, S.M.J.M.; Lee, P.L.; Bao, Y.; Lim, T.H.; Wang, P.; Tam, W.L.; et al. Chromosome 1q21.3 amplification is a trackable biomarker and actionable target for breast cancer recurrence. Nat. Med. 2017, 23, 1319–1330. [Google Scholar] [CrossRef]
- Leung, F.; Kulasingam, V.; Diamandis, E.P.; Hoon, D.S.; Kinzler, K.; Pantel, K.; Alix-Panabières, C. Circulating Tumor DNA as a Cancer Biomarker: Fact or Fiction? Clin. Chem. 2016, 62, 1054–1060. [Google Scholar] [CrossRef]
- Huynh, K.; Hoon, D.S. Liquid Biopsies for Assessing Metastatic Melanoma Progression. Crit. Rev. Oncog. 2016, 21, 141–154. [Google Scholar] [CrossRef]
- Huang, S.K.; Hoon, D.S. Liquid biopsy utility for the surveillance of cutaneous malignant melanoma patients. Mol. Oncol. 2016, 10, 450–463. [Google Scholar] [CrossRef]
- Hoshimoto, S.; Faries, M.B.; Morton, D.L.; Shingai, T.; Kuo, C.; Wang, H.-J.; Elashoff, R.; Mozzillo, N.; Kelley, M.C.; Thompson, J.F.; et al. Assessment of Prognostic Circulating Tumor Cells in a Phase III Trial of Adjuvant Immunotherapy After Complete Resection of Stage IV Melanoma. Ann. Surg. 2012, 255, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Hoshimoto, S.; Shingai, T.; Morton, D.L.; Kuo, C.; Faries, M.B.; Chong, K.; Elashoff, D.; Wang, H.-J.; Elashoff, R.M.; Hoon, D.S. Association Between Circulating Tumor Cells and Prognosis in Patients with Stage III Melanoma with Sentinel Lymph Node Metastasis in a Phase III International Multicenter Trial. J. Clin. Oncol. 2012, 30, 3819–3826. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.Y.; Chang, S.-C.; Lam, S.; Ramos, R.I.; Tran, K.; Ohe, S.; Salomon, M.P.; Bhagat, A.A.S.; Lim, C.T.; Fischer, T.D.; et al. Prospective Molecular Profiling of Circulating Tumor Cells from Patients with Melanoma Receiving Combinatorial Immunotherapy. Clin. Chem. 2019, 66, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Fleischhacker, M.; Schmidt, B. Circulating nucleic acids (CNAs) and cancer—A survey. Biochim. Biophys. Acta (BBA)-Bioenerg. 2007, 1775, 181–232. [Google Scholar] [CrossRef]
- Schwarzenbach, H.; Hoon, D.S.B.; Pantel, K. Cell-free nucleic acids as biomarkers in cancer patients. Nat. Rev. Cancer 2011, 11, 426–437. [Google Scholar] [CrossRef]
- Ono, S.; Oyama, T.; Lam, S.; Chong, K.; Foshag, L.J.; Hoon, D.S. A direct plasma assay of circulating microRNA-210 of hypoxia can identify early systemic metastasis recurrence in melanoma patients. Oncotarget 2015, 6, 7053–7064. [Google Scholar] [CrossRef]
- Iida, Y.; Ciechanover, A.; Marzese, D.M.; Hata, K.; Bustos, M.; Ono, S.; Wang, J.; Salomon, M.P.; Tran, K.; Lam, S.; et al. Epigenetic Regulation of KPC1 Ubiquitin Ligase Affects the NF-κB Pathway in Melanoma. Clin. Cancer Res. 2017, 23, 4831–4842. [Google Scholar] [CrossRef]
- Bustos, M.A.; Ono, S.; Marzese, D.M.; Oyama, T.; Iida, Y.; Cheung, G.; Nelson, N.; Hsu, S.C.; Yu, Q.; Hoon, D.S.B. MiR-200a Regulates CDK4/6 Inhibitor Effect by Targeting CDK6 in Metastatic Melanoma. J. Investig. Dermatol. 2017, 137, 1955–1964. [Google Scholar] [CrossRef]
- Gajos-Michniewicz, A.; Czyz, M. Role of miRNAs in Melanoma Metastasis. Cancers 2019, 11, 326. [Google Scholar] [CrossRef]
- Mumford, S.L.; Towler, B.P.; Pashler, A.L.; Gilleard, O.; Martin, Y.; Newbury, S.F. Circulating MicroRNA Biomarkers in Melanoma: Tools and Challenges in Personalised Medicine. Biomolecules 2018, 8, 21. [Google Scholar] [CrossRef]
- Thyagarajan, A.; Tsai, K.Y.; Sahu, R.P. MicroRNA heterogeneity in melanoma progression. Semin. Cancer Biol. 2019, 59, 208–220. [Google Scholar] [CrossRef] [PubMed]
- Bustos, M.A.; Tran, K.D.; Rahimzadeh, N.; Gross, R.; Lin, S.Y.; Shoji, Y.; Murakami, T.; Boley, C.L.; Tran, L.T.; Cole, H.; et al. Integrated Assessment of Circulating Cell-Free MicroRNA Signatures in Plasma of Patients with Melanoma Brain Metastasis. Cancers 2020, 12, 1692. [Google Scholar] [CrossRef] [PubMed]
- Deichmann, M.; Benner, A.; Bock, M.; Jäckel, A.; Uhl, K.; Waldmann, V.; Näher, H. S100-Beta, Melanoma-Inhibiting Activity, and Lactate Dehydrogenase Discriminate Progressive From Nonprogressive American Joint Committee on Cancer Stage IV Melanoma. J. Clin. Oncol. 1999, 17, 1891. [Google Scholar] [CrossRef] [PubMed]
- Yokota, K.; Uchi, H.; Uhara, H.; Yoshikawa, S.; Takenouchi, T.; Inozume, T.; Ozawa, K.; Ihn, H.; Fujisawa, Y.; Qureshi, A.; et al. Adjuvant therapy with nivolumab versus ipilimumab after complete resection of stage III / IV melanoma: Japanese subgroup analysis from the phase 3 CheckMate 238 study. J. Dermatol. 2019, 46, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Hanniford, D.; Zhong, J.; Koetz, L.; Gaziel-Sovran, A.; Lackaye, D.J.; Shang, S.; Pavlick, A.C.; Shapiro, R.; Berman, R.S.; Darvishian, F.; et al. A miRNA-Based Signature Detected in Primary Melanoma Tissue Predicts Development of Brain Metastasis. Clin. Cancer Res. 2015, 21, 4903–4912. [Google Scholar] [CrossRef] [PubMed]
- Ba, N.H.F.; Zhong, J.; Silva, I.C.; De Miera, E.V.-S.; Brady, B.; Han, S.W.; Hanniford, D.; Wang, J.; Shapiro, R.L.; Hernando, E.; et al. Serum-based miRNAs in the prediction and detection of recurrence in melanoma patients. Cancer 2014, 121, 51–59. [Google Scholar] [CrossRef]
- Gaziel-Sovran, A.; Segura, M.F.; Di Micco, R.; Collins, M.K.; Hanniford, D.; De Miera, E.V.-S.; Rakus, J.F.; Dankert, J.F.; Shang, S.; Kerbel, R.S.; et al. miR-30b/30d Regulation of GalNAc Transferases Enhances Invasion and Immunosuppression during Metastasis. Cancer Cell 2011, 20, 104–118. [Google Scholar] [CrossRef]
- Noman, M.Z.; Buart, S.; Romero, P.; Ketari, S.; Janji, B.; Mari, B.; Mami-Chouaib, F.; Chouaib, S. Hypoxia-Inducible miR-210 Regulates the Susceptibility of Tumor Cells to Lysis by Cytotoxic T Cells. Cancer Res. 2012, 72, 4629–4641. [Google Scholar] [CrossRef]
- Huber, V.; Vallacchi, V.; Fleming, V.; Hu, X.; Cova, A.; Dugo, M.; Shahaj, E.; Sulsenti, R.; Vergani, E.; Filipazzi, P.; et al. Tumor-derived microRNAs induce myeloid suppressor cells and predict immunotherapy resistance in melanoma. J. Clin. Investig. 2018, 128, 5505–5516. [Google Scholar] [CrossRef]
- Carpi, S.; Polini, B.; Fogli, S.; Nieri, P.; Romanini, A. Circulating MicroRNAs in Cutaneous Melanoma Diagnosis and Prognosis. Management Malignant Melanoma. 2016. Available online: http://www.smgebooks.com/management-of-malignant-melanoma/chapters/MMM-16-02.pdf (accessed on 8 September 2020).
- Godínez-Rubí, M.; Ortuño-Sahagún, D. miR-615 Fine-Tunes Growth and Development and Has a Role in Cancer and in Neural Repair. Cells 2020, 9, 1566. [Google Scholar] [CrossRef]
- Pan, X.; Peng, G.; Liu, S.; Sun, Z.; Zou, Z.-W.; Wu, G. MicroRNA-4649-3p inhibits cell proliferation by targeting protein tyrosine phosphatase SHP-1 in nasopharyngeal carcinoma cells. Int. J. Mol. Med. 2015, 36, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Walbrecq, G.; Lecha, O.; Gaigneaux, A.; Fougeras, M.R.; Philippidou, D.; Margue, C.; Nomigni, M.T.; Bernardin, F.; Dittmar, G.; Behrmann, I.; et al. Hypoxia-Induced Adaptations of miRNomes and Proteomes in Melanoma Cells and Their Secreted Extracellular Vesicles. Cancers 2020, 12, 692. [Google Scholar] [CrossRef] [PubMed]
- Da-Via, M.; Solimando, A.G.; Garitano-Trojaola, A.; Barrio, S.; Munawar, U.; Strifler, S.; Haertle, L.; Rhodes, N.; Teufel, E.; Vogt, C.; et al. CIC Mutation as a Molecular Mechanism of Acquired Resistance to Combined BRAF-MEK Inhibition in Extramedullary Multiple Myeloma with Central Nervous System Involvement. Oncology 2019, 25, 112–118. [Google Scholar] [CrossRef]
- Motti, M.L.; Minopoli, M.; Di Carluccio, G.; Ascierto, P.A.; Carriero, M.V. MicroRNAs as Key Players in Melanoma Cell Resistance to MAPK and Immune Checkpoint Inhibitors. Int. J. Mol. Sci. 2020, 21, 4544. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Krall, E.B.; Aguirre, A.J.; Kim, M.; Widlund, H.R.; Doshi, M.B.; Sicinska, E.; Sulahian, R.; Goodale, A.; Cowley, G.S.; et al. ATXN1L, CIC, and ETS Transcription Factors Modulate Sensitivity to MAPK Pathway Inhibition. Cell Rep. 2017, 18, 1543–1557. [Google Scholar] [CrossRef]
- Vergani, E.; Di Guardo, L.; Dugo, M.; Rigoletto, S.; Tragni, G.; Ruggeri, R.; Perrone, F.; Tamborini, E.; Gloghini, A.; Arienti, F.; et al. Overcoming melanoma resistance to vemurafenib by targeting CCL2-induced miR-34a, miR-100 and miR-125b. Oncotarget 2015, 7, 4428–4441. [Google Scholar] [CrossRef]
- Varrone, F.; Caputo, E. The miRNAs Role in Melanoma and in Its Resistance to Therapy. Int. J. Mol. Sci. 2020, 21, 878. [Google Scholar] [CrossRef]
- McShane, L.M.; for the Statistics Subcommittee of the NCI-EORTC Working Group on Cancer Diagnostics; Altman, D.G.; Sauerbrei, W.; Taube, S.E.; Gion, M.; Clark, G.M. REporting recommendations for tumour MARKer prognostic studies (REMARK). Br. J. Cancer 2005, 93, 387–391. [Google Scholar] [CrossRef]
- Sauerbrei, W.; Taube, S.E.; McShane, L.M.; Cavenagh, M.M.; Altman, D.G. Reporting Recommendations for Tumor Marker Prognostic Studies (REMARK): An Abridged Explanation and Elaboration. J. Natl. Cancer Inst. 2018, 110, 803–811. [Google Scholar] [CrossRef]
- Chia, P.L.; Gedye, C.; Boutros, P.C.; Wheatley-Price, P.; John, T. Current and Evolving Methods to Visualize Biological Data in Cancer Research. J. Natl. Cancer Inst. 2016, 108, djw031. [Google Scholar] [CrossRef]
- R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. Available online: https://www.gbif.org/tool/81287/r-a-language-and-environment-for-statistical-computing (accessed on 18 August 2020).
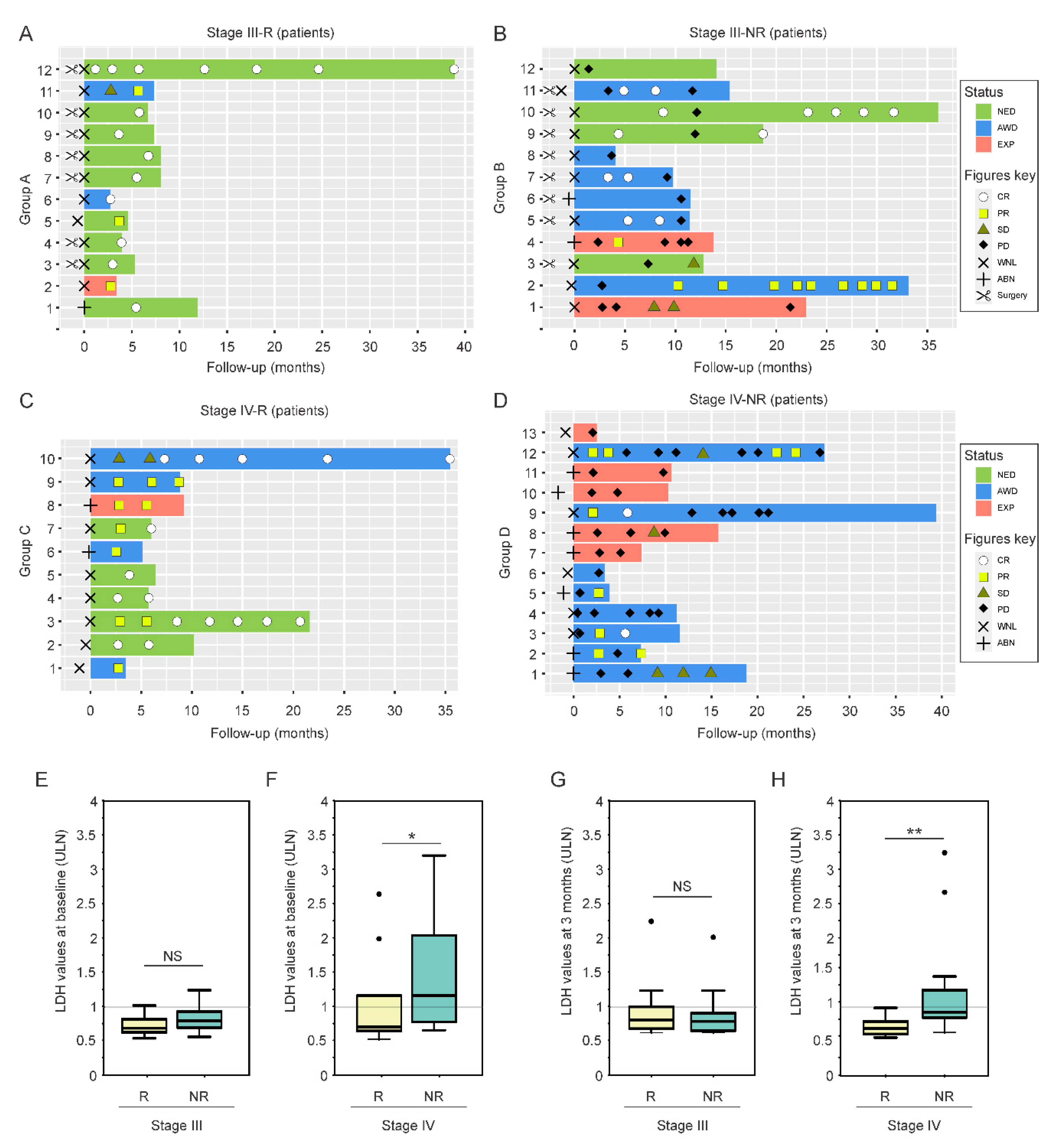
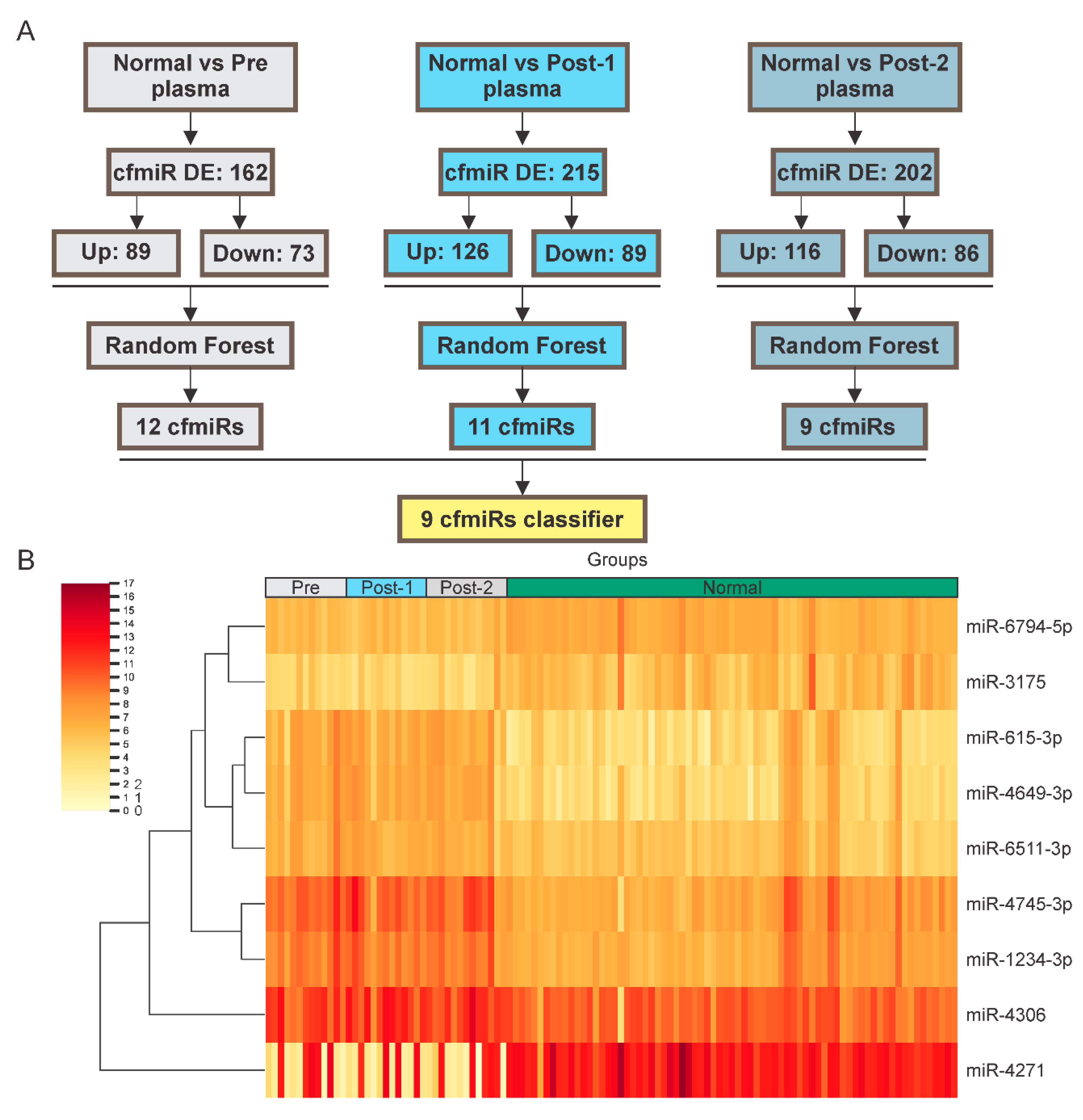
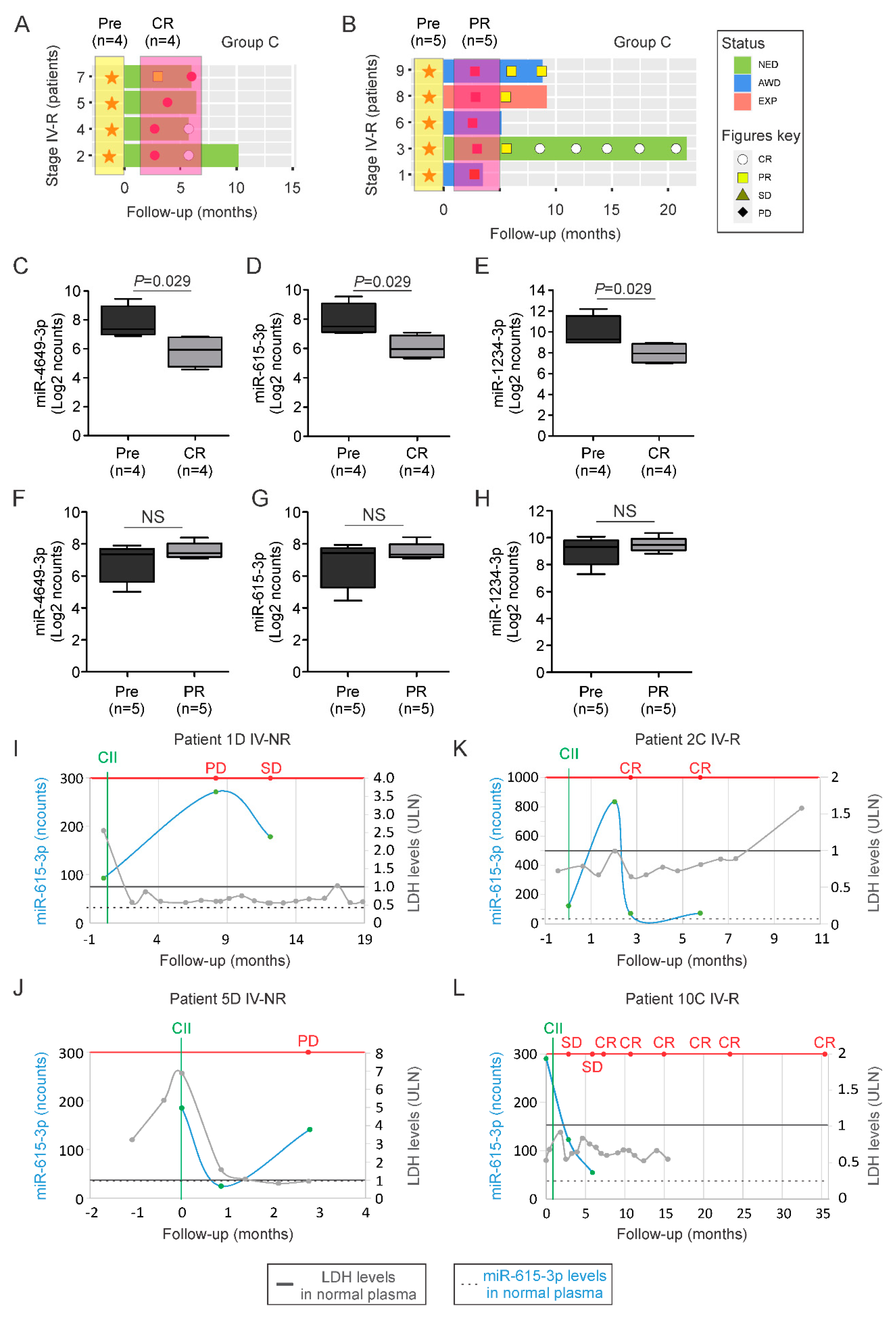
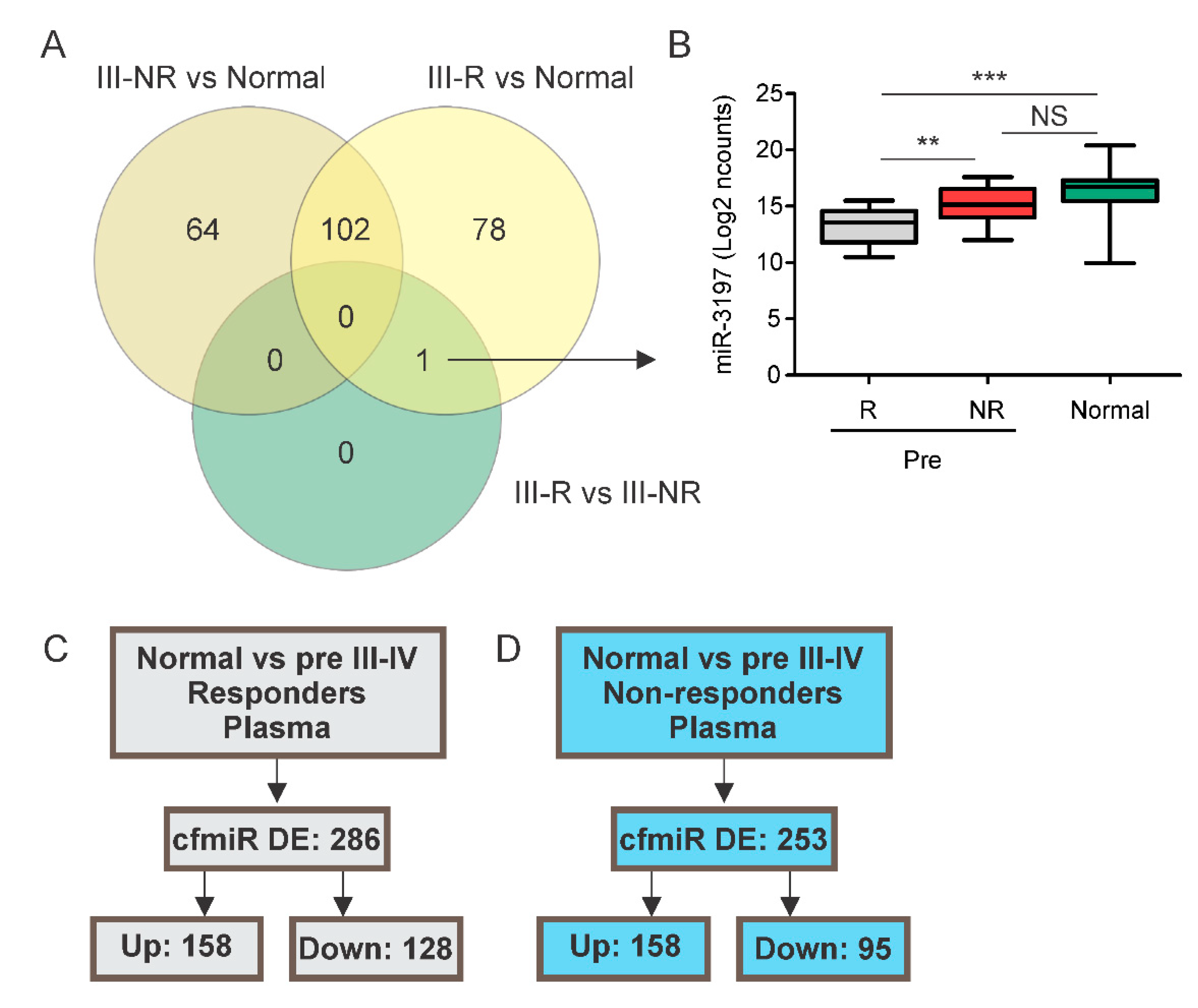
| Melanoma Patients (n = 47) | |
|---|---|
| Variables | n (%) |
| Age at diagnosis, mean (SD 4) | 62.0 (13.9) |
| Age at treatment, mean (SD 4) | 65.9 (13.5) |
| <60 | 14 (29.8) |
| ≥60 | 33 (70.2) |
| Gender | |
| Male | 30 (63.8) |
| Female | 17 (36.2) |
| Treatment regimen | |
| Anti-PD-1 | 31 (65.95) |
| Anti-PD-1/anti-CTLA-4 | 16 (34.05) |
| AJCC 8th ed. Stages 3 | |
| III b/c | 24 (51.1) |
| IV a/b/c/d | 23 (48.9) |
| BRAF mutation | |
| Positive | 25 (53.2) |
| Negative | 22 (46.8) |
| CII-response based on RECIST 5 1.1 | |
| Responders | 22 (46.8) |
| Non-responders | 25 (53.2) |
| Number of metastasis | |
| 1 | 24 (51.06) |
| ≥2 | 16 (34.04) |
| unknown | 7 (14.90) |
| LDH 6 level at baseline | |
| ≤1X 7 ULN | 35 (74.5) |
| >1X ULN | 12 (23.5) |
| Probe | Pre-Treatment FIS 3 | Post-Treatment-1 FIS 3 | Post-Treatment-2 FIS 3 |
|---|---|---|---|
| miR-4271 | 0.06 | 0.05 | 0.07 |
| miR-3175 | 0.05 | 0.05 | 0.04 |
| miR-4745-3p | 0.04 | 0.01 | 0.02 |
| miR-6862-3p | 0.03 | N/A | N/A |
| miR-4649-3p | 0.03 | 0.04 | 0.02 |
| miR-6510-3p | 0.02 | 0.01 | N/A |
| miR-4306 | 0.01 | 0.02 | 0.02 |
| miR-1234-3p | 0.01 | 0.05 | 0.01 |
| miR-6511a-3p | 0.01 | 0.04 | 0.01 |
| miR-615-3p | 0.01 | 0.02 | 0.02 |
| miR-6794-5p | 0.01 | 0.02 | 0.03 |
| miR-1301-5p | 0.01 | 0.02 | N/A |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bustos, M.A.; Gross, R.; Rahimzadeh, N.; Cole, H.; Tran, L.T.; Tran, K.D.; Takeshima, L.; Stern, S.L.; O’Day, S.; Hoon, D.S.B. A Pilot Study Comparing the Efficacy of Lactate Dehydrogenase Levels Versus Circulating Cell-Free microRNAs in Monitoring Responses to Checkpoint Inhibitor Immunotherapy in Metastatic Melanoma Patients. Cancers 2020, 12, 3361. https://doi.org/10.3390/cancers12113361
Bustos MA, Gross R, Rahimzadeh N, Cole H, Tran LT, Tran KD, Takeshima L, Stern SL, O’Day S, Hoon DSB. A Pilot Study Comparing the Efficacy of Lactate Dehydrogenase Levels Versus Circulating Cell-Free microRNAs in Monitoring Responses to Checkpoint Inhibitor Immunotherapy in Metastatic Melanoma Patients. Cancers. 2020; 12(11):3361. https://doi.org/10.3390/cancers12113361
Chicago/Turabian StyleBustos, Matias A., Rebecca Gross, Negin Rahimzadeh, Hunter Cole, Linh T. Tran, Kevin D. Tran, Ling Takeshima, Stacey L. Stern, Steven O’Day, and Dave S. B. Hoon. 2020. "A Pilot Study Comparing the Efficacy of Lactate Dehydrogenase Levels Versus Circulating Cell-Free microRNAs in Monitoring Responses to Checkpoint Inhibitor Immunotherapy in Metastatic Melanoma Patients" Cancers 12, no. 11: 3361. https://doi.org/10.3390/cancers12113361
APA StyleBustos, M. A., Gross, R., Rahimzadeh, N., Cole, H., Tran, L. T., Tran, K. D., Takeshima, L., Stern, S. L., O’Day, S., & Hoon, D. S. B. (2020). A Pilot Study Comparing the Efficacy of Lactate Dehydrogenase Levels Versus Circulating Cell-Free microRNAs in Monitoring Responses to Checkpoint Inhibitor Immunotherapy in Metastatic Melanoma Patients. Cancers, 12(11), 3361. https://doi.org/10.3390/cancers12113361






