Electrochemical Performance of Micropillar Array Electrodes in Microflows
Abstract
1. Introduction
2. Methods
2.1. Materials and Instrumentations
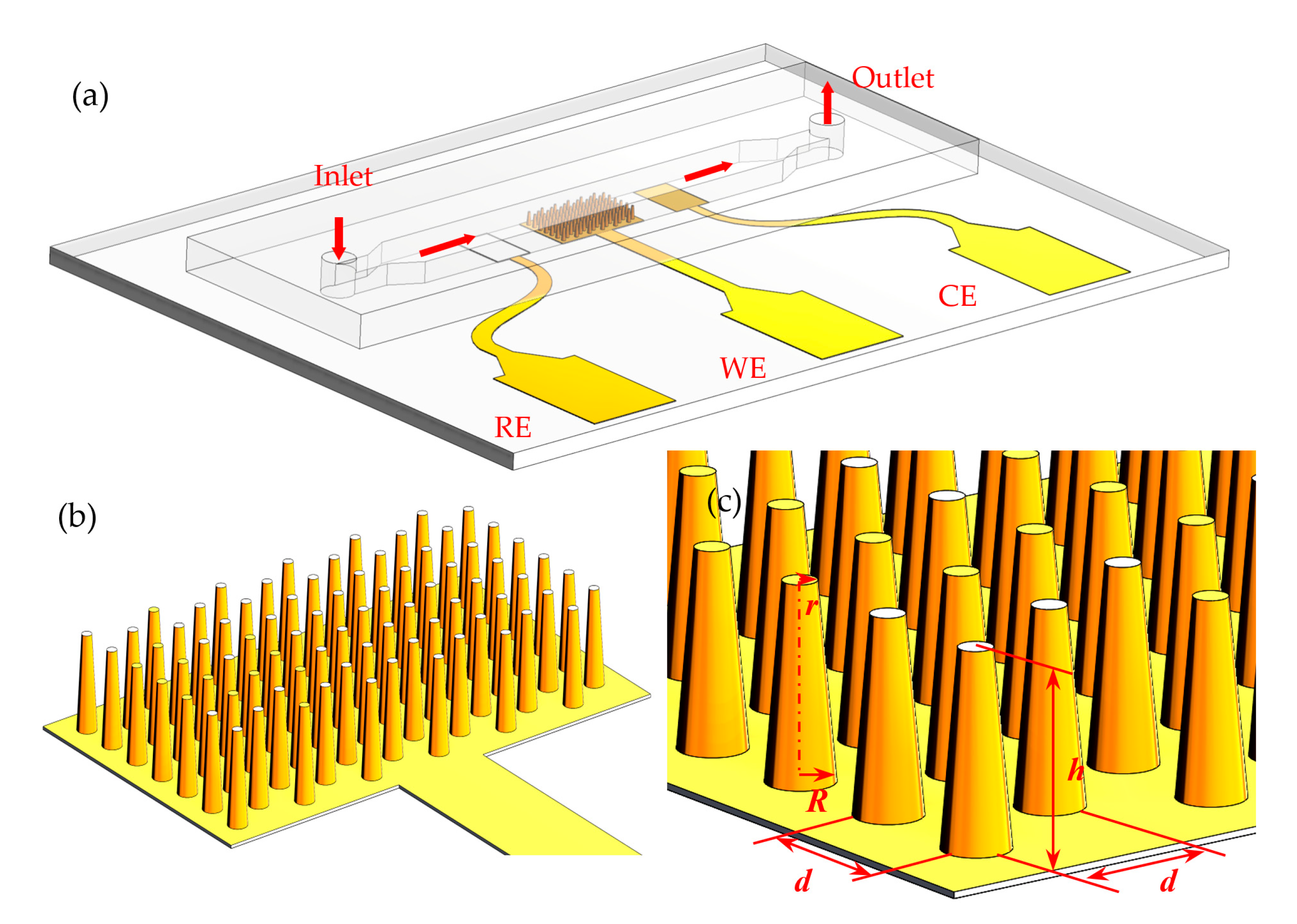
2.2. Configuration of Microchip-Based Electrochemical Detection System (μEDS)
2.3. Numerical Simulation Method of the μEDS
2.3.1. Theory
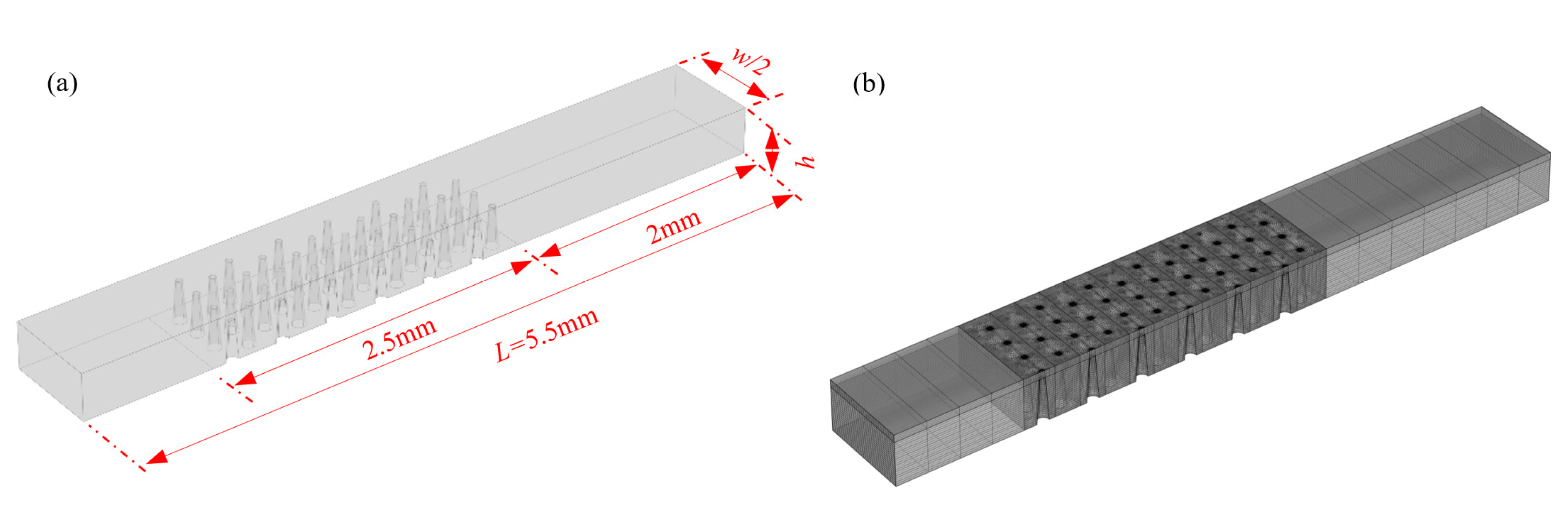
2.3.2. Numerical Model
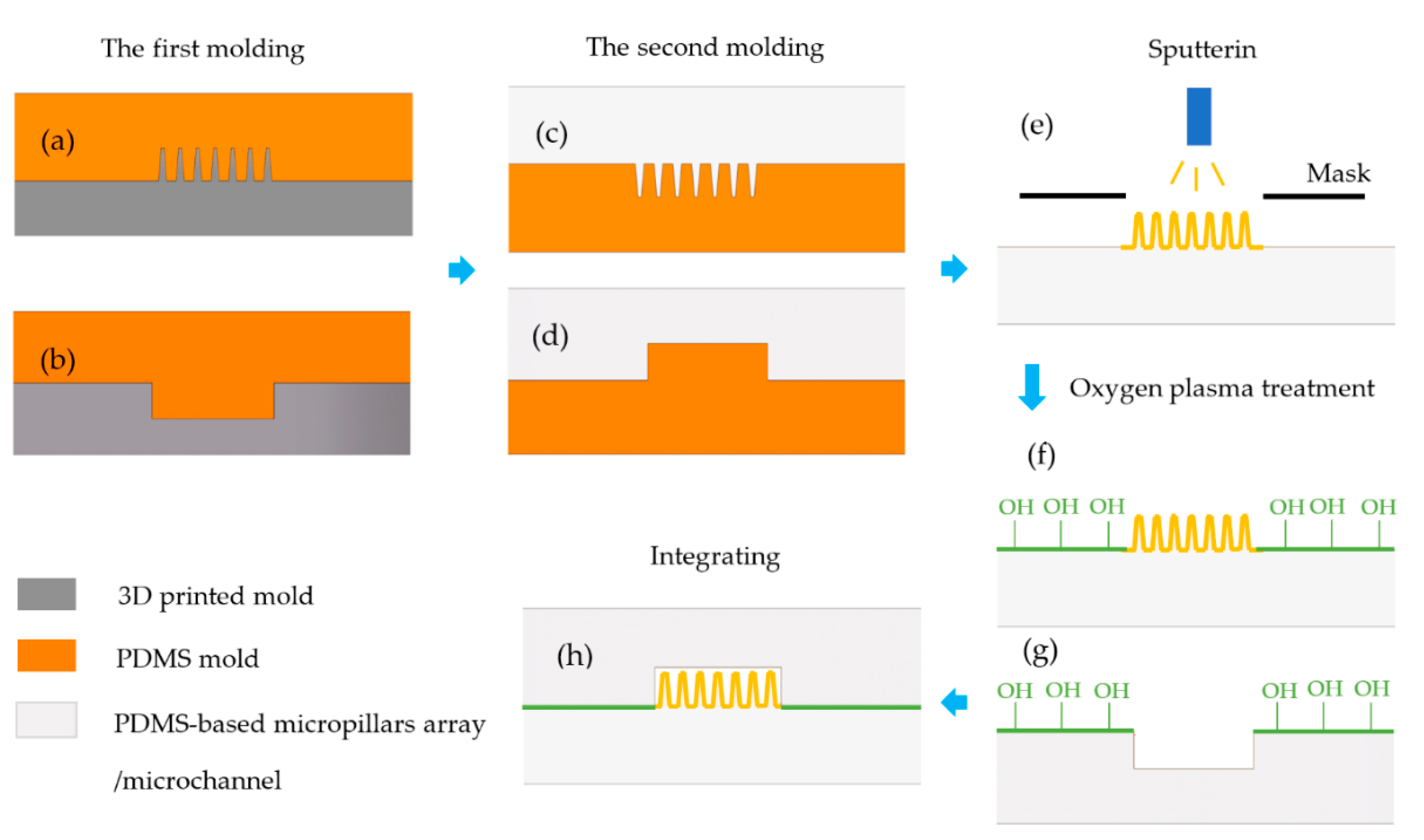
2.4. Fabrication of μEDS
2.5. Experiments of the Electrochemical Detection
3. Results and Discussion
3.1. Effect of Flow Rate and Spacing
3.2. Effect of Micropillar Height
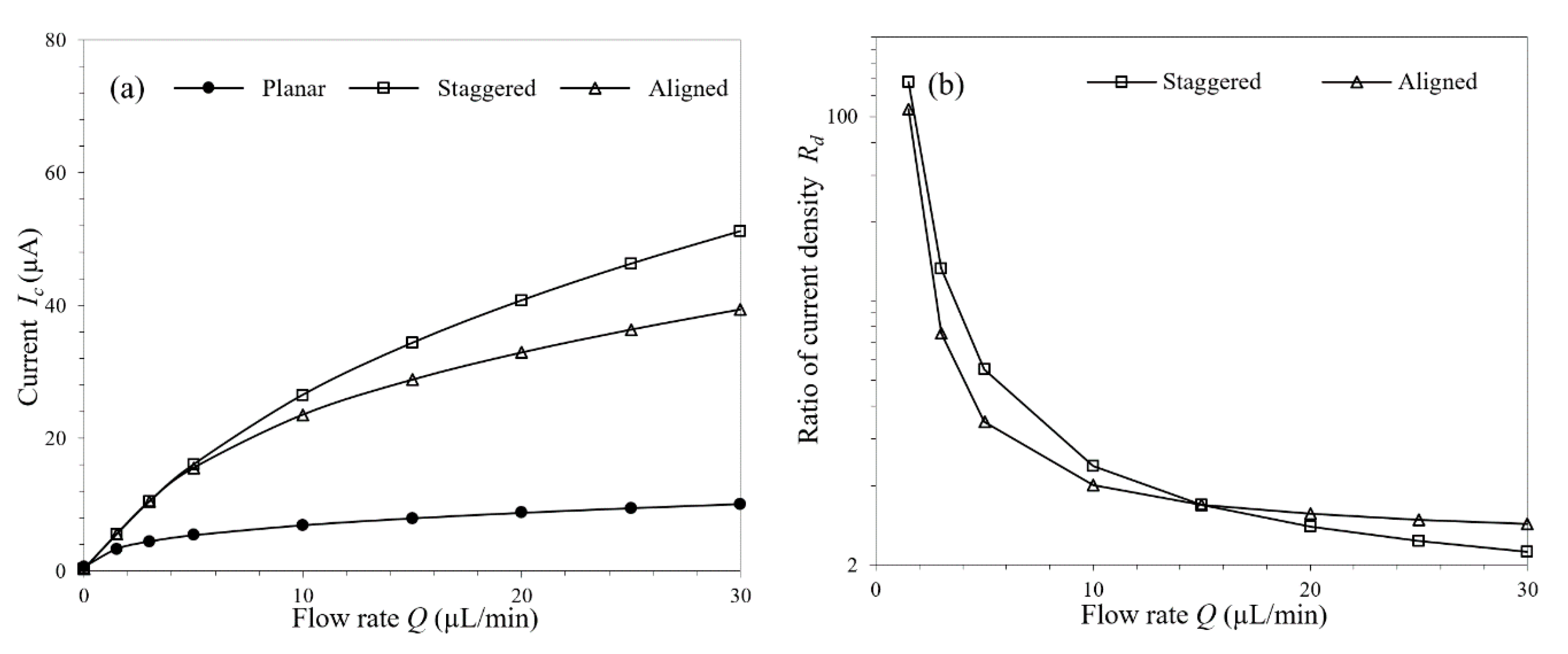
3.3. Effect of Micropillar Layout
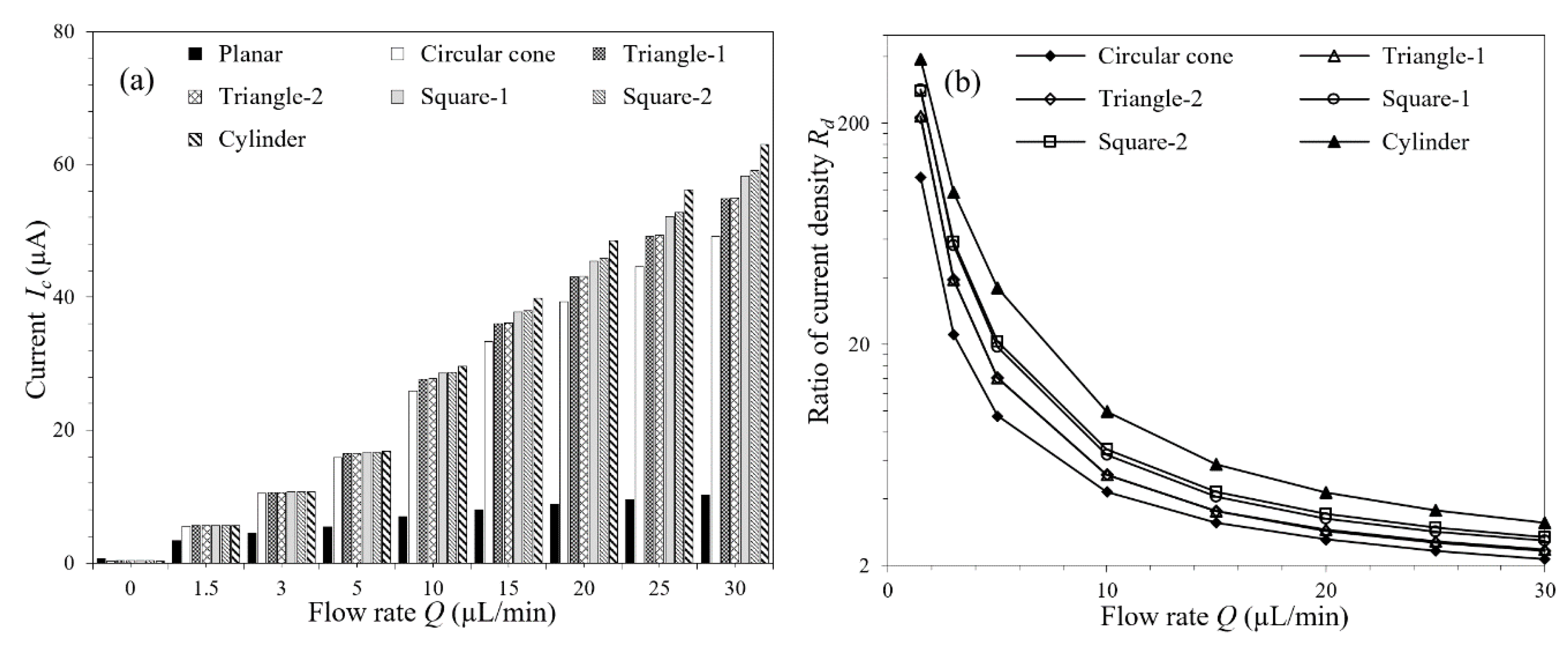
3.4. Effect of Micropillar Shape
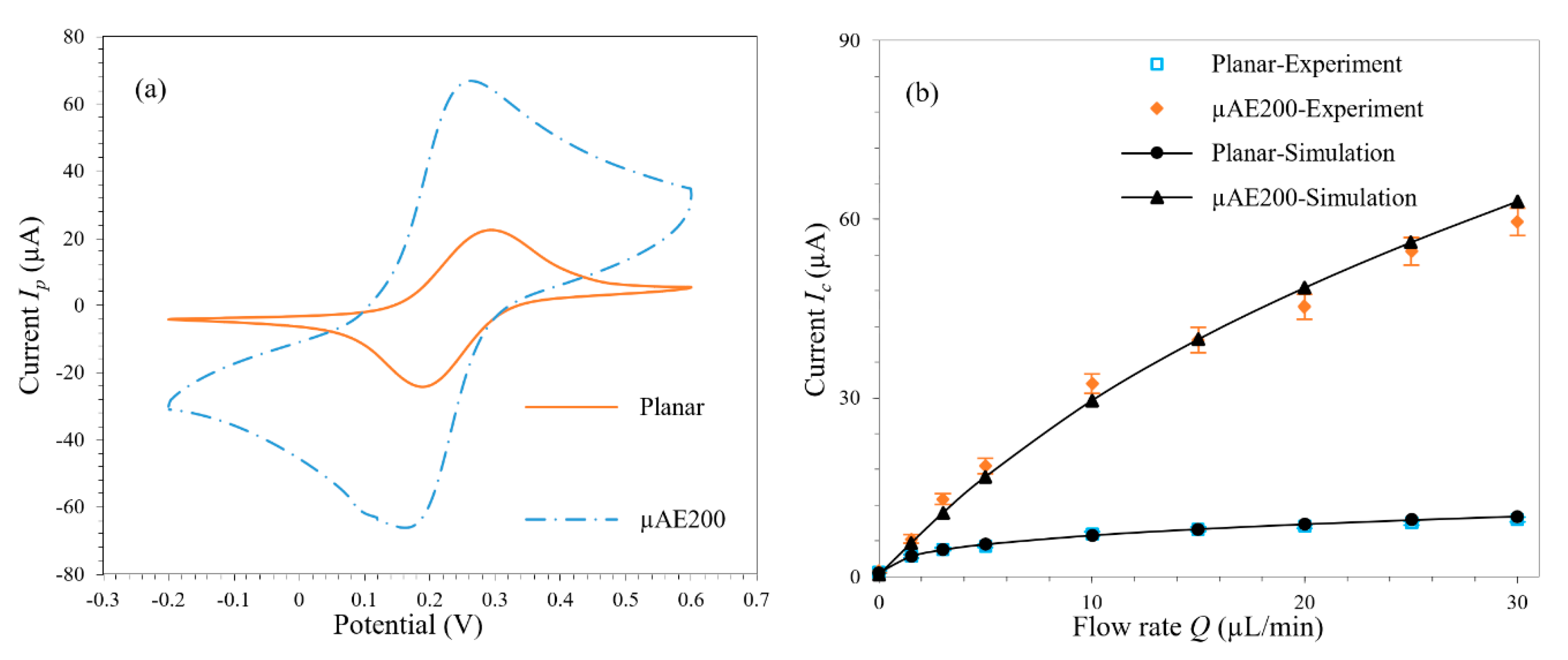
3.5. Experimental Verification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wang, J.; Chen, G.; Chatrathi, M.P.; Musameh, M. Capillary Electrophoresis Microchip with a Carbon Nanotube-Modified Electrochemical Detector. Anal. Chem. 2004, 76, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Beuer, M.; Bartoli, J.; Martin-Chapa, S.O.; Madou, M. Wireless Electrochemical Detection on a Microfluidic Compact Disc (CD) and Evaluation of Redox-Amplification during Flow. Micromachines 2019, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Pumera, M.; Chatrathi, M.P.; Escarpa, A.; Konrad, R.; Griebel, A.; Dorner, W.; Lowe, H. Towards disposable lab-on-a-chip: Poly(methylmethacrylate) Microchip Electrophoresis Device with Electrochemical Detection. Electrophoresis 2015, 23, 596–601. [Google Scholar] [CrossRef]
- Martin, R.S.; Ratzlaff, K.L.; Huynh, B.H.; Lunte, S.M. In-Channel Electrochemical Detection for Microchip Capillary Electrophoresis Using an Electrically Isolated Potentiostat. Anal. Chem. 2002, 74, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Song, Y.; Bo, X.; Min, J.; Pak, O.S.; Zhu, L.; Wang, M.; Tu, J.; Kogan, A.; Zhang, H.; et al. A Laser-engraved Wearable Sensor for Sensitive Detection of Uric Acid and Tyrosine in Sweat. Nat. Biotechnol. 2020, 38, 217–224. [Google Scholar] [CrossRef]
- Zhang, H.; Chuai, R.; Li, X.; Zhang, B. Design, Preparation and Performance Study of On-chip Flow-through Amperometric Sensors with an Integrated Ag/AgCl Reference Electrode. Micromachines 2018, 9, 114. [Google Scholar] [CrossRef]
- Azmana, M.; Mahmood, S.; Hilles, A.R.; Mandal, U.K.; Raman, S. Transdermal Drug Delivery System through Polymeric Microneedle: A Recent Update. J. Drag Deliv. Sci. Technol. 2020, 60, 101877. [Google Scholar] [CrossRef]
- Pires, L.R.; Vinayakumar, K.B.; Turos, M.; Miguel, V.; Gaspar, J. A Perspective on Microneedle-Based Drag Delivery and Diagnostics in Paediatrics. J. Pers. Med. 2019, 9, 49. [Google Scholar] [CrossRef]
- Leif, N. Electrochemical Techniques for Lab-on-a-chip Applications. Analyst 2005, 130, 599–605. [Google Scholar]
- Nair, P.R.; Alam, M.A. A Compact Analytical Formalism for Current Transients in Electrochemical System. Analyst 2013, 138, 525–538. [Google Scholar] [CrossRef]
- Davies, T.J.; Compton, R.G. The Cyclic and Linear Sweep Voltammetry of Regular and Random Arrays of Microdisc Electrodes: Theory. J. Electroanal. Chem. 2005, 585, 63–82. [Google Scholar] [CrossRef]
- Amatore, C.; Oleinick, A.; Svir, I. Numerical Simulation of Diffusion Processes at Recessed Disk Microelectrode Arrays Using the Quasi-Conformal Mapping Approach. Anal. Chem. 2009, 81, 4397–4405. [Google Scholar] [CrossRef] [PubMed]
- Jin, B.K.; Gui, X.; Shi, H.; Zhang, Z. Application of the Finite Analytical Numerical Method. Part 3. Digital Simulation of Charge Transfer to a Micro-ring Electrode Interface. J. Electroanal. Chem. 1996, 417, 45–51. [Google Scholar] [CrossRef]
- Eswari, A.; Rajendran, L. Analytical Expressions of Concentration and Current in Homogeneous Catalytic Reactions at Spherical Microelectrodes: Homotopy Perturbation Approach. J. Electroanal. Chem. 2011, 651, 173–184. [Google Scholar] [CrossRef]
- Yin, H.; Chao, T.; Shabnam, S.; Arumugam, P.U. Electrochemical Behaviour of a Gold Nanoring Electrode Microfabricated on a Silicon Micropillar. Sens. Actuators Chem. 2019, 281, 392–398. [Google Scholar] [CrossRef]
- David, S.; Juan, P.E.; Neus, S.; Xavier, M.; Campo, F.J. High Aspect-Ratio, Fully Conducting Gold Micropillar Array Electrodes: Silicon Micromachining and Electrochemical Characterization. J. Phys. Chem. C 2012, 116, 18831–18846. [Google Scholar]
- Numthuam, S.; Kakegawa, T.; Anada, T.; Khademhosseini, A. Synergistic Effects of Micro/Nano Modifications on Electrodes for Microfluidic Electrochemical ELISA. Sens. Actuators B Chem. 2011, 156, 637–644. [Google Scholar] [CrossRef]
- Prehn, R.; Abad, L.; David, S.; Duch, M.; Neus, S.; Campo, F.J.; Munoz, F.X.; Compton, R.G. Microfabrication and Characterization of Cylinder Micropillar Array Electrodes. J. Electroanal. Chem. 2011, 662, 361–370. [Google Scholar] [CrossRef]
- Liu, Q.; Li, J.; Yang, W.; Zhang, X.; Zhang, C.; Labbe, C.; Portier, X.; Liu, F.; Yao, J.; Liu, B. Simultaneous Detection of Trace Ag(I) and Cu(II) Ions Using Homoepitaxially Grown GaN Micropillar Electrode. Anal. Chim. Acta 2019, 1100, 22–30. [Google Scholar] [CrossRef]
- Pai, R.S.; Walsh, K.M.; Crain, M.M.; Roussel, T.J.; Jackson, D.J.; Baldwin, R.P.; Keynton, R.S.; Naber, J.F. Fully Integrated Three-Dimensional Electrodes for Electrochemical Detection in Microchips: Fabrication, Characterization, and Applications. Anal. Chem. 2009, 81, 4762–4769. [Google Scholar] [CrossRef]
- Edmund, J.F.D.; Streeter, I.; Compton, R.G. Chronoamperometry and Cyclic Voltammetry at Conical Electrodes, Microelectrodes, and Electrode Arrays: Theory. J. Phys. Chem. B 2008, 112, 4059–4066. [Google Scholar]
- Amatore, C.; Oleinick, A.; Svir, I. Simulation of Diffusion-convection Processes in Microfluidic Channels Equipped with Double Band Microelectrode Assemblies: Approach through Quasi-conformal Mapping. Electrochem. Commun. 2004, 6, 1123–1130. [Google Scholar] [CrossRef]
- Amatore, C.; Mota, N.D.; Sella, C.; Thouin, L. Theory and Experiments of Transport at Channel Microband Electrodes under Laminar Flows. 1. Steady-State Regimes at a Single Electrode. Anal. Chem. 2007, 79, 8502–8510. [Google Scholar] [CrossRef]
- Amatore, C.; Mota, N.D.; Lemmer, C.; Pebay, C.; Thouin, L. Theory and Experiments of Transport at Channel Microband Electrodes under Laminar Flows. 2. Electrochemical Regimes at Double Microband Assemblies under Steady State. Anal. Chem. 2008, 80, 9483–9490. [Google Scholar] [CrossRef] [PubMed]
- Amatore, C.; Mota, N.D.; Lemmer, C.; Pebay, C.; Thouin, L. Theory and Experiments of Transport at Channel Microband Electrodes under Laminar Flow. 3. Electrochemical Detection at Electrode Arrays under Steady State. Anal. Chem. 2010, 82, 2434–2440. [Google Scholar] [CrossRef] [PubMed]
- Rees, N.V.; Dryfe, R.A.W.; Cooper, J.A.; Coles, B.A.; Compton, R.G. Voltammetry under High Mass Transport Conditions. A High Speed Channel Electrode for the Study of Ultrafast Kinetics. J. Phys. Chem. 1995, 99, 7096–7101. [Google Scholar] [CrossRef]
- Compton, R.G.; Dryfe, R.A.W.; Alden, J.A.; Rees, N.V. Hydrodynamic Voltammetry with Channel Microband Electrodes: Potential Step Transients. J. Phys. Chem. 1994, 98, 1270–1275. [Google Scholar] [CrossRef]
- Christian, A.; Klymenko, O.V.; Svir, I. In Situ and Online Monitoring of Hydrodynamic Flow Profiles in Microfluidic Channels Based upon Microelectrochemistry: Optimization of Electrode Locations. ChemPhysChem 2006, 7, 482–487. [Google Scholar]
- Amatore, C.; Klymenko, O.V.; Oleinick, A.; Svir, I. In Situ and On-line Monitoring of Hydrodynamic Flow Profiles in Microfluidic Channels Based on Microelectrochemistry: Optimization of Channel Geometrical Parameters for Best Performance of Flow Profile Reconstruction. Chemphyschem 2010, 8, 1870–1874. [Google Scholar] [CrossRef]
- Aoki, K.; Tokuda, K.; Matsuda, H. Derivation of an Approximate Equation for Chronoamperometric Curves at Microband Electrodes and its Experimental Verification. J. Electroanal. Chem. 1987, 230, 61–67. [Google Scholar] [CrossRef]
- Christian, A.; Sella, C.; Thouin, L. Diffusional Cross-Talk between Paired Microband Electrodes Operating within a Thin Film: Theory for Redox Couples with Unequal Diffusion Coefficients. J. Phys. Chem. B 2002, 106, 11565–11571. [Google Scholar]
- Ferrigno, R.; Brevet, P.F.; Girault, H.H. Finite Element Simulation of the Amperometric Response of Recessed and Protruding Microband Electrodes in Flow Channels. J. Electroanal. Chem. 1997, 430, 235–242. [Google Scholar] [CrossRef]
- Stevens, N.P.C.; Fisher, A.C. Finite Element Simulations in Electrochemistry. 2. Hydrodynamic Voltammetry. J. Phys. Chem. B 1997, 93, 225–232. [Google Scholar] [CrossRef]
- Alden, J.A.; Compton, R.G. A Comparison of Finite Difference Algorithms for the Simulation of Microband Electrode Problems with and without Convective Flow. J. Electroanal. Chem. 1996, 402, 1–10. [Google Scholar] [CrossRef]
- Stevens, N.P.C.; Fisher, A. Transient Voltammetry under Hydrodynamic Conditions. Electroanalysis 2015, 10, 16–20. [Google Scholar] [CrossRef]
- Sullivan, S.P.; Johns, M.J.; Matthews, S.W.; Fisher, A.C. Lattice Boltzmann Simulations of Electrolysis Reactions: Microfluidic Voltammetry. Electrochem. Commun. 2005, 7, 1323–1328. [Google Scholar] [CrossRef]
- Tait, R.J.; Bury, P.C.; Finnin, B.C.; Reed, B.L. An Explicit Finite Difference Simulation for Chronoamperometry at a Disk Microelectrode in a Channel Flow Solution. J. Electroanal. Chem. 2002, 356, 25–42. [Google Scholar] [CrossRef]
- Zoski, E.C. Handbook of Electrochemistry; Elsevier: Amsterdam, The Netherlands, 2007; pp. 3–30. [Google Scholar]
- Baur, J.E.; Motsegood, P.N. Diffusional Interactions at Dual Disk Microelectrodes: Comparison of Experiment with Three-dimensional Random Walk Simulation. J. Electroanal. Chem. 2004, 572, 29–40. [Google Scholar] [CrossRef]
- Maher, A.K.; Bellini, M.; Pogni, R.; Giaccherini, A.; Innocenti, M.; Vizza, F.; Lavacchi, A. Effect of Electrode Shape and Flow Conditions on the Electrochemical Detection with Band Microelectrodes. Sensors 2018, 18, 3196. [Google Scholar]
- Cui, H.; Ye, J.; Zhang, W.; Li, C.; Luong, J.H.T.; Sheu, F. Selective and Sensitive Electrochemical Detection of Glucose in Neutral Solution Using Platinum-lead Alloy Nanoparticle/Carbon Nanotube Nanocomposites. Anal. Chim. Acta 2007, 594, 175–183. [Google Scholar] [CrossRef]
- Chen, Z.; Zhu, Y.; Xu, D.; Alam, M.M.; Shui, L.; Chen, H. Cell Elasticity Measurement Using a Microfluidic Device with Real-time Pressure Feedback. Lab Chip 2020, 20, 2343–2353. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xu, D.; Bai, S.; Yu, Z.; Zhu, Y.; Xing, X.; Chen, H. Dynamic Screening and Printing of Single Cells Using a Microfluidic Chip with Dual Microvalves. Lab Chip 2020, 20, 1227–1237. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Piao, Y.; Choi, J.S.; Seo, T.S. Three-dimensional Graphene Micropillar Based Electrochemical Sensor for Phenol Detection. Biosens. Bioelectron 2013, 50, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Mawatari, K.; Tsukahara, T.; Sugii, Y.; Kitamori, T. Extended-nano Fluidic Systems for Analytical and Chemical Technologies. Nanoscale 2010, 2, 1588–1595. [Google Scholar] [CrossRef] [PubMed]
- Sanghavi, B.J.; Varhue, W.; Rohani, A.; Liao, K.T.; Bazydlo, L.A.; Chou, C.F.; Swami, N.S. Ultrafast Immunoassays by Coupling Dielectrophoretic Biomarker Enrichment in Nanoslit Channel with Electrochemical Detection on Graphene. Lab Chip 2015, 15, 4563–4570. [Google Scholar] [CrossRef]
- Shaw, K.; Contento, N.M.; Xu, W.; Bohn, P.W. Nanofluidic Structures for Coupled Sensing and Remediation of Toxins. Proc. SPIE 2014, 9107, 1–12. [Google Scholar]
- Sparreboom, W.; Eijkel, J.C.T.; Bomer, J.; Berg, A. Rapid Sacrificial Layer Etching for the Fabrication of Nanochannels with Integreated Metal Electrodes. Lab Chip 2007, 8, 402–407. [Google Scholar] [CrossRef]
- Xu, W.; Foster, E.; Ma, C.; Bohn, P. On-demand in Situ Generation of Oxygen in a Nanofluidic Embedded Planar Microband Electrochemical Reactor. Microfluid. Nanofluidics 2015, 19, 1181–1189. [Google Scholar] [CrossRef]
- Steentjes, T.; Sarkar, S.; Jonkheijm, P.; Lemay, S.G.; Huskens, J. Electron Transfer Mediated by Surface-tethered Redox Groups in Nanofluidic Devices. Small 2017, 13, 1–7. [Google Scholar] [CrossRef]



| Parameters | Planar | Conical Micropillar | ||
|---|---|---|---|---|
| Projection area l × w (mm2) | 1.5 × 2.5 | |||
| Top radius rt (μm) | - | 25 | ||
| Base radius (μm) | - | 50 | ||
| Height h (μm) | - | 100/200/300 | ||
| Spacing d (μm) 1 | - | 150 | 200 | 250 |
| Number of pillars n | - | 136 | 78 | 55 |
| Surface area S (mm2) | 3.75 | 7.33 | 8.82 | 12.60 |
| Area ratio 2 Sg | 1.0 | 1.95 | 2.35 | 3.36 |
| Parameters | Unit | Value |
|---|---|---|
| Diffusion coefficient D | m2/s | 6.5 × 10−5 |
| Faraday’s constant F | C/mol | 96,485.33 |
| Standard heterogeneous rate constant k0 | m/s | 1 × 10−4 |
| Transfer coefficient α | - | 0.6 |
| Gas constant R | J/(mol·K) | 8.314 |
| Absolute temperature T | K | 298.15 |
| Applied potential E | V | 0.25 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, B.; Lv, C.; Chen, C.; Ran, B.; Lan, M.; Chen, H.; Zhu, Y. Electrochemical Performance of Micropillar Array Electrodes in Microflows. Micromachines 2020, 11, 858. https://doi.org/10.3390/mi11090858
Liu B, Lv C, Chen C, Ran B, Lan M, Chen H, Zhu Y. Electrochemical Performance of Micropillar Array Electrodes in Microflows. Micromachines. 2020; 11(9):858. https://doi.org/10.3390/mi11090858
Chicago/Turabian StyleLiu, Bo, Chuanwen Lv, Chaozhan Chen, Bin Ran, Minbo Lan, Huaying Chen, and Yonggang Zhu. 2020. "Electrochemical Performance of Micropillar Array Electrodes in Microflows" Micromachines 11, no. 9: 858. https://doi.org/10.3390/mi11090858
APA StyleLiu, B., Lv, C., Chen, C., Ran, B., Lan, M., Chen, H., & Zhu, Y. (2020). Electrochemical Performance of Micropillar Array Electrodes in Microflows. Micromachines, 11(9), 858. https://doi.org/10.3390/mi11090858








