Use of Antibiotics following Snakebite in the Era of Antimicrobial Stewardship
Abstract
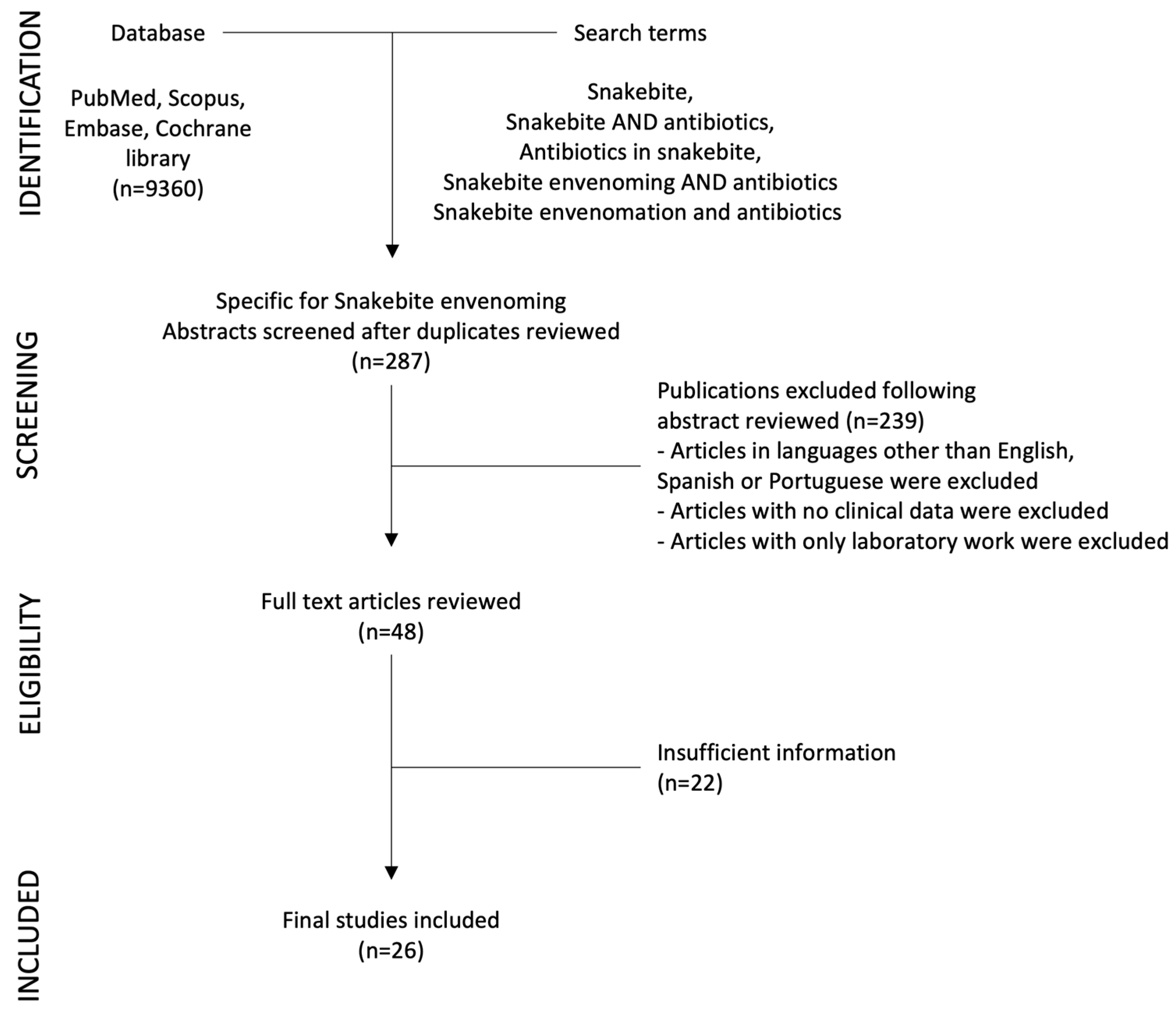
1. Introduction
2. Results
3. Discussion
4. Conclusions
5. Materials and Methods
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Minghui, R.; Malecela, M.N.; Cooke, E.; Abela-Ridder, B. WHO’s Snakebite Envenoming Strategy for prevention and control. Lancet Glob. Health 2019, 7, e837–e838. [Google Scholar] [CrossRef] [PubMed]
- Bhaumik, S.; Menon, G.R.; Habib, A.G. Prioritising snakebite in the child and adolescent health agenda. Lancet Child. Adolesc. Health 2023, 7, 753–755. [Google Scholar] [CrossRef] [PubMed]
- Iliyasu, G.; Dayyab, F.M.; Michael, G.C.; Hamza, M.; Habib, M.A.; Gutierrez, J.M.; Habib, A.G. Case fatality rate and burden of snakebite envenoming in children—A systematic review and meta-analysis. Toxicon 2023, 234, 107299. [Google Scholar] [CrossRef] [PubMed]
- Le Geyt, J.; Pach, S.; Gutierrez, J.M.; Habib, A.G.; Maduwage, K.P.; Hardcastle, T.C.; Hernandez Diaz, R.; Avila-Aguero, M.L.; Ya, K.T.; Williams, D.; et al. Paediatric snakebite envenoming: Recognition and management of cases. Arch. Dis. Child. 2021, 106, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Kshirsagar, V.Y.; Ahmed, M.; Colaco, S.M. Clinical profile of snake bite in children in rural India. Iran. J. Pediatr. 2013, 23, 632–636. [Google Scholar] [PubMed]
- Brenes-Chacon, H.; Ulloa-Gutierrez, R.; Soriano-Fallas, A.; Camacho-Badilla, K.; Valverde-Munoz, K.; Avila-Aguero, M.L. Bacterial Infections Associated with Viperidae Snakebites in Children: A 14-Year Experience at the Hospital Nacional de Ninos de Costa Rica(dagger). Am. J. Trop. Med. Hyg. 2019, 100, 1227–1229. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.H.; Hsueh, J.H.; Liu, W.C.; Yang, K.C.; Hsu, K.C.; Lin, C.T.; Ho, Y.Y.; Chen, L.W. Contributing Factors for Complications and Outcomes in Patients with Snakebite: Experience in a Medical Center in Southern Taiwan. Ann. Plast. Surg. 2017, 78, S32–S36. [Google Scholar] [CrossRef]
- Jorge, M.T.; de Mendonca, J.S.; Ribeiro, L.A.; da Silva, M.L.; Kusano, E.J.; Cordeiro, C.L. [Bacterial flora of the oral cavity, fangs and venom of Bothrops jararaca: Possible source of infection at the site of bite]. Rev. Inst. Med. Trop. Sao Paulo 1990, 32, 6–10. [Google Scholar] [CrossRef]
- Chen, C.M.; Wu, K.G.; Chen, C.J.; Wang, C.M. Bacterial infection in association with snakebite: A 10-year experience in a northern Taiwan medical center. J. Microbiol. Immunol. Infect. 2011, 44, 456–460. [Google Scholar] [CrossRef]
- Otero-Patino, R. Epidemiological, clinical and therapeutic aspects of Bothrops asper bites. Toxicon 2009, 54, 998–1011. [Google Scholar] [CrossRef]
- Resiere, D.; Gutierrez, J.M.; Neviere, R.; Cabie, A.; Hossein, M.; Kallel, H. Antibiotic therapy for snakebite envenoming. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020, 26, e20190098. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.K.; Vardanega, J.; Smith, S.; White, J.; Little, M.; Hanson, J. The Incidence of Infection Complicating Snakebites in Tropical Australia: Implications for Clinical Management and Antimicrobial Prophylaxis. J. Trop. Med. 2023, 2023, 5812766. [Google Scholar] [CrossRef] [PubMed]
- Parker-Cote, J.; Meggs, W.J. First Aid and Pre-Hospital Management of Venomous Snakebites. Trop. Med. Infect. Dis. 2018, 3, 45. [Google Scholar] [CrossRef] [PubMed]
- Gil-Alarcon, G.; Sanchez-Villegas Mdel, C.; Hugo Reynoso, V. Pre-hospital treatment of ophidian accidents: Review, update, and current problems. Gac. Med. Mex. 2011, 147, 195–208. [Google Scholar] [PubMed]
- Hardcastle, T.C.; Kajee, M.; Lachenicht, K.; Van der Walt, N. Approach to the diagnosis and management of snakebite envenomation in South Africa in humans. S. Afr. Med. J. 2023, 113, 10–18. [Google Scholar] [CrossRef]
- Kerrigan, K.R. Bacteriology of snakebite abscess. Trop. Doct. 1992, 22, 158–160. [Google Scholar] [CrossRef]
- Wagener, M.; Naidoo, M.; Aldous, C. Wound infection secondary to snakebite. S. Afr. Med. J. 2017, 107, 315–319. [Google Scholar] [CrossRef]
- Lin, C.C.; Chen, Y.C.; Goh, Z.N.L.; Seak, C.K.; Seak, J.C.; Shi-Ying, G.; Seak, C.J.; Spot, I. Wound Infections of Snakebites from the Venomous Protobothrops mucrosquamatus and Viridovipera stejnegeri in Taiwan: Bacteriology, Antibiotic Susceptibility, and Predicting the Need for Antibiotics-A BITE Study. Toxins 2020, 12, 575. [Google Scholar] [CrossRef]
- Yeh, H.; Gao, S.Y.; Lin, C.C. Wound Infections from Taiwan Cobra (Naja atra) Bites: Determining Bacteriology, Antibiotic Susceptibility, and the Use of Antibiotics-A Cobra BITE Study. Toxins 2021, 13, 183. [Google Scholar] [CrossRef]
- Mendes, V.; Pereira, H.D.S.; Elias, I.C.; Soares, G.S.; Santos, M.; Talhari, C.; Cordeiro-Santos, M.; Monteiro, W.M.; Sachett, J.A.G. Secondary infection profile after snakebite treated at a tertiary referral center in the Brazilian Amazon. Rev. Soc. Bras. Med. Trop. 2022, 55, e0244. [Google Scholar] [CrossRef]
- Kerrigan, K.R.; Mertz, B.L.; Nelson, S.J.; Dye, J.D. Antibiotic prophylaxis for pit viper envenomation: Prospective, controlled trial. World J. Surg. 1997, 21, 369–372, discussion 372–363. [Google Scholar] [CrossRef]
- Resiere, D.; Mehdaoui, H.; Neviere, R.; Olive, C.; Severyns, M.; Beaudoin, A.; Florentin, J.; Brouste, Y.; Banydeen, R.; Cabie, A.; et al. Infectious Complications Following Snakebite by Bothrops lanceolatus in Martinique: A Case Series. Am. J. Trop. Med. Hyg. 2020, 102, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Senthilkumaran, S.; Salim, A.; Almeida, J.R.; Williams, J.; Vijayakumar, P.; Thirunavukarasu, A.; Christopoulos, M.A.; Williams, H.F.; Thirumalaikolundusubramanian, P.; Patel, K.; et al. The Effectiveness of Antibiotics in Managing Bacterial Infections on Bite Sites following Snakebite Envenomation. Toxins 2023, 15, 190. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.C.; Liu, P.Y.; Hung, D.Z.; Lai, W.C.; Huang, S.T.; Hung, Y.M.; Yang, C.C. Bacteriology of Naja atra Snakebite Wound and Its Implications for Antibiotic Therapy. Am. J. Trop. Med. Hyg. 2016, 94, 1129–1135. [Google Scholar] [CrossRef]
- Clark, R.F.; Selden, B.S.; Furbee, B. The incidence of wound infection following crotalid envenomation. J. Emerg. Med. 1993, 11, 583–586. [Google Scholar] [CrossRef] [PubMed]
- LoVecchio, F.; Klemens, J.; Welch, S.; Rodriguez, R. Antibiotics after rattlesnake envenomation. J. Emerg. Med. 2002, 23, 327–328. [Google Scholar] [CrossRef] [PubMed]
- Campbell, B.T.; Corsi, J.M.; Boneti, C.; Jackson, R.J.; Smith, S.D.; Kokoska, E.R. Pediatric snakebites: Lessons learned from 114 cases. J. Pediatr. Surg. 2008, 43, 1338–1341. [Google Scholar] [CrossRef]
- Jorge, M.T.; Ribeiro, L.A.; da Silva, M.L.; Kusano, E.J.; de Mendonca, J.S. Microbiological studies of abscesses complicating Bothrops snakebite in humans: A prospective study. Toxicon 1994, 32, 743–748. [Google Scholar] [CrossRef]
- Jorge, M.T.; Malaque, C.; Ribeiro, L.A.; Fan, H.W.; Cardoso, J.L.; Nishioka, S.A.; Sano-Martins, I.S.; Franca, F.O.; Kamiguti, A.S.; Theakston, R.D.; et al. Failure of chloramphenicol prophylaxis to reduce the frequency of abscess formation as a complication of envenoming by Bothrops snakes in Brazil: A double-blind randomized controlled trial. Trans. R. Soc. Trop. Med. Hyg. 2004, 98, 529–534. [Google Scholar] [CrossRef]
- Garg, A.; Sujatha, S.; Garg, J.; Acharya, N.S.; Chandra Parija, S. Wound infections secondary to snakebite. J. Infect. Dev. Ctries. 2009, 3, 221–223. [Google Scholar] [CrossRef]
- Huang, L.W.; Wang, J.D.; Huang, J.A.; Hu, S.Y.; Wang, L.M.; Tsan, Y.T. Wound infections secondary to snakebite in central Taiwan. J. Venom. Anim. Toxins incl Trop. Dis. 2012, 18, 272–276. [Google Scholar] [CrossRef]
- Palappallil, D.S. Pattern of Use of Antibiotics Following Snake Bite in a Tertiary Care Hospital. J. Clin. Diagn. Res. 2015, 9, OC05–OC09. [Google Scholar] [CrossRef] [PubMed]
- Sachett, J.A.G.; da Silva, I.M.; Alves, E.C.; Oliveira, S.S.; Sampaio, V.S.; do Vale, F.F.; Romero, G.A.S.; Dos Santos, M.C.; Marques, H.O.; Colombini, M.; et al. Poor efficacy of preemptive amoxicillin clavulanate for preventing secondary infection from Bothrops snakebites in the Brazilian Amazon: A randomized controlled clinical trial. PLoS Negl. Trop. Dis. 2017, 11, e0005745. [Google Scholar] [CrossRef] [PubMed]
- August, J.A.; Boesen, K.J.; Hurst, N.B.; Shirazi, F.M.; Klotz, S.A. Prophylactic Antibiotics Are Not Needed Following Rattlesnake Bites. Am. J. Med. 2018, 131, 1367–1371. [Google Scholar] [CrossRef] [PubMed]
- Ngo, N.D.; Le, Q.X.; Pham, A.Q.; Nguyen, N.T.; Ha, H.T.; Dinh, M.M.Q.; Le, T.Q. Clinical Features, Bacteriology, and Antibiotic Treatment Among Patients with Presumed Naja Bites in Vietnam. Wilderness Environ. Med. 2020, 31, 151–156. [Google Scholar] [CrossRef]
- Kriengkrairut, S.; Othong, R. Bacterial infection secondary to Trimeresurus species bites: A retrospective cohort study in a university hospital in Bangkok. Emerg. Med. Australas. 2021, 33, 1006–1012. [Google Scholar] [CrossRef]
- Chiang, L.C.; Chaou, C.H.; Li, Y.Y.; Seak, C.J.; Yu, S.R.; Lin, C.C. Management and Prognosis of Snake Envenomation Among Pediatric Patients: A National Database Study. J. Acute Med. 2022, 12, 13–22. [Google Scholar] [CrossRef]
- Houcke, S.; Resiere, D.; Lontsingoula, G.R.; Cook, F.; Lafouasse, P.; Pujo, J.M.; Demar, M.; Matheus, S.; Hommel, D.; Kallel, H. Characteristics of Snakebite-Related Infection in French Guiana. Toxins 2022, 14, 89. [Google Scholar] [CrossRef]
- Hu, S.; Lou, Z.; Shen, Y.; Tu, M. Bacteriological Studies of Venomous Snakebite Wounds in Hangzhou, Southeast China. Am. J. Trop. Med. Hyg. 2022, 107, 925–929. [Google Scholar] [CrossRef]
- Soares Coriolano Coutinho, J.V.; Fraga Guimaraes, T.; Borges Valente, B.; Gomes Martins de Moura Tomich, L. Epidemiology of secondary infection after snakebites in center-west Brazil. PLoS Negl. Trop. Dis. 2023, 17, e0011167. [Google Scholar] [CrossRef]
- Michael, G.C.; Thacher, T.D.; Shehu, M.I. The effect of pre-hospital care for venomous snake bite on outcome in Nigeria. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Singaravelu, K.P.; Pandit, V.R.; Chinnakali, P.; Bammigatti, C. Pre-hospital care and its association with clinical outcome of snakebite victims presenting at a tertiary care referral hospital in South India. Trop. Doct. 2021, 51, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, M.C.; Traynor, M.; Bruce, J.L.; Bekker, W.; Laing, G.L.; Aho, J.M.; Kong, V.Y.; Klinkner, D.B.; Zielinski, M.D.; Clarke, D.L. Surgical Considerations for Pediatric Snake Bites in Low- and Middle-Income Countries. World J. Surg. 2019, 43, 1636–1643. [Google Scholar] [CrossRef] [PubMed]
- Tekin, R.; Sula, B.; Cakirca, G.; Aktar, F.; Deveci, O.; Yolbas, I.; Celen, M.K.; Bekcibasi, M.; Palanci, Y.; Dogan, E. Comparison of snakebite cases in children and adults. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 2711–2716. [Google Scholar] [PubMed]
- Correa, J.A.; Fallon, S.C.; Cruz, A.T.; Grawe, G.H.; Vu, P.V.; Rubalcava, D.M.; Kaziny, B.; Naik-Mathuria, B.J.; Brandt, M.L. Management of pediatric snake bites: Are we doing too much? J. Pediatr. Surg. 2014, 49, 1009–1015. [Google Scholar] [CrossRef] [PubMed]
- Resiere, D.; Hossein, M.; Megarbane, B. Snake Bites by Bothrops lanceolatus in Martinique. Med. Sante Trop. 2018, 28, 37–43. [Google Scholar] [CrossRef]
- Oliveira, I.S.; Pucca, M.B.; Cerni, F.A.; Vieira, S.; Sachett, J.; Seabra de Farias, A.; Lacerda, M.; Murta, F.; Baia-da-Silva, D.; Rocha, T.A.H.; et al. Snakebite envenoming in Brazilian children: Clinical aspects, management and outcomes. J. Trop. Pediatr. 2023, 69, fmad010. [Google Scholar] [CrossRef]
- Saravia-Otten, P.; Gutierrez, J.M.; Arvidson, S.; Thelestam, M.; Flock, J.I. Increased infectivity of Staphylococcus aureus in an experimental model of snake venom-induced tissue damage. J. Infect. Dis. 2007, 196, 748–754. [Google Scholar] [CrossRef]
- Saravu, K.; Somavarapu, V.; Shastry, A.B.; Kumar, R. Clinical profile, species-specific severity grading, and outcome determinants of snake envenomation: An Indian tertiary care hospital-based prospective study. Indian. J. Crit. Care Med. 2012, 16, 187–192. [Google Scholar] [CrossRef]
- Nelson, B.K. Snake envenomation. Incidence, clinical presentation and management. Med. Toxicol. Adverse Drug Exp. 1989, 4, 17–31. [Google Scholar] [CrossRef]
- Padhi, L.; Panda, S.K.; Mohapatra, P.P.; Sahoo, G. Antibiotic susceptibility of cultivable aerobic microbiota from the oral cavity of Echis carinatus from Odisha (India). Microb. Pathog. 2020, 143, 104121. [Google Scholar] [CrossRef] [PubMed]
- Chugh, K.S.; Pal, Y.; Chakravarty, R.N.; Datta, B.N.; Mehta, R.; Sakhuja, V.; Mandal, A.K.; Sommers, S.C. Acute renal failure following poisonous snakebite. Am. J. Kidney Dis. 1984, 4, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Morales-Alvarez, M.C. Nephrotoxicity of Antimicrobials and Antibiotics. Adv. Chronic Kidney Dis. 2020, 27, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Lindbaek, M.; Berild, D.; Straand, J.; Hjortdahl, P. Influence of prescription patterns in general practice on anti-microbial resistance in Norway. Br. J. Gen. Pract. 1999, 49, 436–440. [Google Scholar]
- Tagwireyi, D.D.; Ball, D.E.; Nhachi, C.F. Routine prophylactic antibiotic use in the management of snakebite. BMC Clin. Pharmacol. 2001, 1, 4. [Google Scholar] [CrossRef]
| Title | Author(s) | Year | Objective | Population | Snake Species | Isolated Bacteria | Antibiotic Use Recommendation | Findings and Recommendations |
|---|---|---|---|---|---|---|---|---|
| Bacteriology of snakebite abscess | Kerrigan, K [16] | 1992 | Efficacy of prophylactic antibiotic in reducing incidence of snakebite injuries has never been documented and needs to be studied. | 312 patients | Viperidae family | S. aureus Streptococcus sp. | Gentamicin and chloramphenicol | Abscesses in 9% of patients. Broad spectrum antibiotic prophylaxis should be used based on local flora of snakes in every setting. |
| The incidence of wound infection following crotalid envenomation | Clark, R et al. [25] | 1993 | Assess the incidence of wound infection and evaluate the need for prophylactic antibiotics. | 54 patients | Crotalid (rattlesnake) | P. aeruginosa Proteus sp. Coagulase negative Staphylococcus, Clostridium sp. B. fragilis | Amoxicillin/clavulanate, nafcillin Cephalexin, Cefazolin, ceftriaxone | 22% used prophylactic antibiotics. 1.8% developed wound infection. Routine use of prophylactic antibiotics may not be warranted. |
| Microbiological studies of abscesses complicating Bothrops snakebite in humans: A prospective Study | Jorge, M et al. [28] | 1994 | Identify microorganisms responsible for abscess formation at the bite site and antimicrobial sensitivity. | 40 patients | Bothrops spp. | M. morganii P. rettgeri Bacterioides sp. Enterobacter sp. Streptococcus sp. | Chloramphenicol Benzyl penicillin + gentamycin | 75% presented with abscesses. Chloramphenicol is recommended in cases of infection. No specific recommendation about use of prophylactic antimicrobial treatment is stated. |
| Antibiotic prophylaxis for pit viper envenomation: Prospective, Controlled Trial | Kerrigan, K [21] | 1997 | Determine whether prophylactic antibiotics can decrease the incidence of infectious complications at the anatomical site of pit viper envenomation. | 114 patients | Viperidae family | E. coli Klebsiella sp. Enterobacter sp. Proteus sp. S. aureus | Gentamicin and chloramphenicol | 7.9% developed abscesses. 67% of patients who developed abscesses received antibiotic prophylaxis. Antibiotics are not indicated as prophylactic therapy for pit viper envenomation. Antibiotics do not prevent infectious complications, are not cost-effective, and may select resistant organisms. |
| Antibiotics after Rattlesnake envenomation | LoVecchio, F et al. [26] | 2002 | Describe the incidence of infection following rattlesnake bite. | 56 patients | Crotalid (rattlesnake) | No bacteria isolated | Antibiotics used are not specified | 5% received antibiotics. No cases of documented infection. Prophylactic antibiotics are not indicated in patients with rattlesnake bites. |
| Failure of chloramphenicol prophylaxis to reduce the frequency of abscess formation as a complication of envenoming by Bothrops snakes in Brazil: A double-blind randomized controlled trial | Jorge, M et al. [29] | 2004 | Comparison between chloramphenicol and placebo prophylaxis to reduce abscess formation as a complication of Bothrops spp. Envenoming. | 251 patients | Bothrops spp. | M. morganii E. coli | Chloramphenicol vs. placebo prophylaxis | Abscesses developed in 6 (4.9%) patients with chloramphenicol and 6 (4.7%) in the placebo group. Use of chloramphenicol for snakebite victims with local signs of envenoming is not effective for the prevention of local infection. |
| Wound infections secondary to snakebite | Garg, A et al. [30] | 2009 | Evaluate the aerobic bacteria responsible for snakebite-associated wound infection and antibiogram of these isolates. | 43 patients | Snake species are not specified | S. aureus E. coli Coagulase negative Staphylococcus | Gentamicin, amikacin, ciprofloxacin, ceftriaxone, meropenem | Antibiotic use should be prescribed according to local susceptibilities. |
| Bacterial infection in association with snakebite: A 10-year experience in a northern Taiwan medical center | Chen, CM et al. [9] | 2011 | Survey of patients admitted for snakebites at a medical center in northern Taiwan. | 231 patients | Trimeresurus spp. Naja, B. multicinctus | M. morganii Enterococcus sp. B. fragilis P. aeruginosa | Amoxicillin–clavulanate, ciprofloxacin, piperacillin–tazobactam | 25% developed cellulitis or wound infection. Cobra bite-related injuries were more severe than those inflicted by other species. Prophylactic use of antibiotics is controversial. |
| Wound infections secondary to snakebite in central Taiwan | Huang, LW et al. [31] | 2012 | Investigate the treatment of secondary infection following snakebites in Taiwan. | 121 patients | Naja atra, T. mucrosquamatus, T. stejnegeri, B. multicinctus | M. morganii A. hydrophila Enterococcus sp. | piperacillin-tazobactam, quinolonee, cephalosporins | 28% developed wound infection. |
| Pattern of use of Antibiotics following snakebite in a tertiary care hospital | Palappallil, D et al. [32] | 2015 | Pattern of antibiotics used following snakebite envenomation in a tertiary care hospital of Kerala. | 313 patients | Snake species are not specified | Bacteria identified are not specified | Ampicillin, cloxacillin, metronidazole, cefotaxime, piperacillin–tazobactam, ciprofloxacin | There is a high prescription of antibiotics in snake bitten patients (95%). Clinical outcomes of patients with or without antibiotics were not different. |
| Bacteriology of Naja atra snakebite wound and its implications for antibiotic therapy | Mao, YC et al. [24] | 2016 | Understand the bacteriology of N. atra bite wound. | 112 patients | Naja atra | Gram-negative rod M. morganii Enterococcus sp. Proteus sp. A. hydrophila Bacterioides sp. | Ureidopenicillin Aminopenicillin + third-generation cephalosporin or fluoroquinolone | 77% developed wound infection, including cellulitis, tissue necrosis, gangrene, and necrotizing fasciitis. Patients may have received antibiotics in the prehospital setting. |
| Poor efficacy of preemptive amoxicillin clavulanate for preventing secondary infection from Bothrops snakebites in the Brazilian Amazon: A randomized controlled clinical trial | Sachett, J et al. [33] | 2017 | Assess the efficacy of amoxicillin clavulanate for reducing secondary infection. Identify associated factors for secondary infections from snakebites. | 186 patients | Bothrops spp. | M. morganii S. aureus | Amoxicillin-clavulanate use is not recommended | Antibiotic schemes suggested for the treatment of secondary infection are not based on good evidence. No evidence that antibiotics decreased risk of associated secondary infection. Higher risk of infection in patients with elevated fibrinogen, ALT, CRP. |
| Wound infection secondary to snakebite | Wagener, M et al. [17] | 2017 | Determine the bacterial causation of wound infection secondary to snakebite. | 164 patients | Snake species are not specified | M. morganii Proteus sp. E. faecalis | Ceftriaxone, ciprofloxacin, gentamicin, amikacin | 26% patients developed infection. Recommendation advises against prophylactic use of antibiotics to treat all snakebites. Use of antibiotics in snakebite is widespread and not directed. Good antibiotic policy is strongly advocated. |
| Prophylactic antibiotics are not needed following Rattlesnake Bite | August, J et al. [34] | 2018 | Determine efficacy of prophylactic antibiotics for venomous snakebites in the US. | 2748 patients | Crotalid (rattlesnake) | S. aureus E. coli Enterococcus sp. B. fragilis | Antibiotics used are not specified | Self-manipulation of wounds is associated with post bite infection. No recommendation in prophylaxis after rattlesnake bites. |
| Bacterial infections associated with Viperidae snakebites in children: a 14 year experience at the Hospital Nacional de Niños de Costa Rica | Brenes-Chacon, H et al. [6] | 2019 | Infectious complications associated with Viperidae snakebites in children. | 75 patients | Viperidae family | M. morganii A. hydrophila P. rettgeri | Combination of penicillin or clindamycin with an aminoglycoside | 19% developed wound infection. Infection complications are more frequent in patients with severe envenomation or patients with delayed medical care. Use of antibiotic prophylaxis is controversial. |
| Infectious complications following snakebite by Bothrops Ianceolatus in Martinique: a case series | Resiere, D et al. [22] | 2020 | Investigate the infectious complications related to B. lanceolatus bite. | 170 patients | Bothrops lanceolatus | A. hydrophila M. morganii K. pneumoniae Bacillus sp. Enterococcus sp. | Cephalosporins, aminoglycoside, ciprofloxacin, and metronidazole | Wound infection occurred in 12% of patients Soft tissue infection occurs in patients with severe envenoming. |
| Wound infections of snakebites from the venomous Protobothrops mucrosquamatus and Viridovipera stejnegeri in Taiwan: Bacteriology, antibiotic susceptibility, and predicting the need for antibiotics-A BITE Study | Lin, C et al. [18] | 2020 | Develop a suitable tool to predict the probability of developing a snakebite wound infection. | 726 patients | Protobothrops mucrosquamatus Viridovipera stejnegeri | E. faecalis Staphylococcus sp. Corynebacterium sp. M. morganii | Amoxicillin-clavulanate, oxacillin, cefazolin, ampicillin/sulbactam | 22.5% of patients developed wound infection. Use of antibiotics according to local susceptibility 44% of patients received antibiotic prophylaxis. BITE score considers hospitalization and laboratory findings for severity. Recommendation to only give antibiotics to patients with a BITE score = 5. |
| Clinical features, bacteriology, and antibiotic treatment among patients with presumed Naja bites in Vietnam | Ngo, N et al. [35] | 2020 | Describe the clinical and bacteriological characteristics of local wounds in patients with presumed Naja bite and their antibiotic treatment. | 46 patients | Naja spp. | E. faecalis M. morganii Enterobacter sp. Proteus sp. | Clindamycin Ciprofloxacin | 80% of cases had positive bacterial wound cultures. Early antibiotic use could be indicated to prevent wound necrosis and infection. |
| Bacterial infection secondary to Trimeresurus species bites: A retrospective cohort study in a university hospital in Bangkok | Kriengkrairut, S Othong, R [36] | 2021 | Determine the infection rate in those bitten by Trimeresurus spp. | 123 patients | Trimeresurus spp. | No bacteria were identified in those infected | Antibiotics used are not specified | 6.5% developed a bacterial wound infection presented as cellulitis, tenosynovitis, or necrotizing fasciitis. Antibiotic prescription rate was 12.2%. Hemorrhagic manifestations were found to be the only risk factor for infection. |
| Wound infection from Taiwan Cobra (Naja atra) Bites: Determining bacteriology, ATB susceptibility, and use of antibiotics—A Cobra BITE Study | Yeh, H et al. [19] | 2021 | Bacteriology of infected wounds. Compare rate of infection in wounds with and without necrosis. | 195 patients | Naja atra | M. morganii E. faecalis Coagulase negative Staphylococcus | Gentamicin, ceftriaxone, ciprofloxacin, or levofloxacin as monotherapy | 27% of patients developed wound infection. Wound infection was more prone to occur in moderate to severe cases. |
| Management and prognosis of snake envenomation among pediatric patients: A national database study | Chiang, L et al. [37] | 2022 | Investigate the epidemiology, management process, and endpoints of pediatric snakebite envenomation in Taiwan. | 106 patients | Snake species are not specified | No bacteria were identified in those infected | Antibiotics used are not specified | 65% of patients received antibiotics, but only 35% were hospitalized. Use of prophylactic antibiotics ranged from 15 to 100%. Antibiotic prophylaxis and treatment are controversial. |
| Secondary infection profile after snakebite treated at a tertiary referral center in the Brazilian Amazon | Mendes, V et al. [20] | 2022 | Characterize local secondary infections from snakebites. | 545 patients | Bothrops spp. | P. mirabilis Morganella spp. E. coli Streptococcus sp. Aeromonas spp. S. aureus Clostridium spp. | Ceftriaxone, piperacillin-tazobactam, ciprofloxacin | 49% of patients were diagnosed with moderate envenomation and 23% developed secondary bacterial infection. Secondary infection occurred in those with moderate envenoming. |
| Characteristics of snakebite-related infection in French Guiana | Houcke, S et al. [38] | 2022 | Assess the prevalence of wound infection after snakebite envenoming to identify the involved bacteria and monitor the use of antibiotics. | 172 patients | Bothrops spp. | A. hydrophila M. morganii S. aureus P. rettgeri | Amoxicillin/clavulanate Cefotaxime | 36% received antibiotics at admission. 32% developed wound infection: 19% had grade 1 envenoming, 35% grade 2, and 53% grade 3. 83% of isolates were resistant to amox/clav, so administration is not advised. Favor the promotion of proper use of antibiotics. |
| Bacteriological Studies of Venomous Snakebite Wounds in Hangzhou, Southeast China | Hu, S et al. [39] | 2022 | Define the pattern of wound bacterial flora of venomous snakebites and their susceptibility to common antibiotics | 331 patients | D. acutus, G. brevicaudus, T. stejnegeri, N. atra | M. morganii S. aureus A. hydrophila | Aminoglycosides Cephalosporins Quinolones | 25% developed wound infection. 23% developed localized tissue necrosis. 13% had positive bacterial cultures. |
| The effectiveness of antibiotics in managing bacterial infections on bite sites following snakebite envenomation | Senthilkumaran, S et al. [23] | 2023 | Document the bacterial profiles in local bite sites and provide guidance for the effective management of infections. | 266 patients | Daboia russelii | S. aureus Klebsiella sp. E. coli P. aeruginosa | Linezolid Amikacin Clindamycin Piperacillin-tazobactam Colistin | 82% of patients had a bacterial growth identified. Use of antibiotics as preventive measure is controversial; they should only be used when there is confirmation of a wound infection. Overuse of antibiotics can contribute to antimicrobial resistance. |
| Epidemiology of secondary infection after snakebites in center-west Brazil | Soares Coriolano Coutinho, J et al. [40] | 2023 | Evaluate incidence of secondary infections, characterize microbiological profile and empirical therapy failure rates. | 326 patients | Bothrops spp. | A. hydrophila | Amoxicillin/clavulanate TMP-SMX | 47.5% developed secondary infection. Only 7 had microbiological cultures. Association between infection and severity of envenomation. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brenes-Chacon, H.; Gutiérrez, J.M.; Avila-Aguero, M.L. Use of Antibiotics following Snakebite in the Era of Antimicrobial Stewardship. Toxins 2024, 16, 37. https://doi.org/10.3390/toxins16010037
Brenes-Chacon H, Gutiérrez JM, Avila-Aguero ML. Use of Antibiotics following Snakebite in the Era of Antimicrobial Stewardship. Toxins. 2024; 16(1):37. https://doi.org/10.3390/toxins16010037
Chicago/Turabian StyleBrenes-Chacon, Helena, José María Gutiérrez, and María L. Avila-Aguero. 2024. "Use of Antibiotics following Snakebite in the Era of Antimicrobial Stewardship" Toxins 16, no. 1: 37. https://doi.org/10.3390/toxins16010037
APA StyleBrenes-Chacon, H., Gutiérrez, J. M., & Avila-Aguero, M. L. (2024). Use of Antibiotics following Snakebite in the Era of Antimicrobial Stewardship. Toxins, 16(1), 37. https://doi.org/10.3390/toxins16010037







