Abstract
We reviewed the literature to evaluate potential associations between vitamins, nutrients, nutritional status or nutritional interventions and presence or healing of foot ulceration in diabetes. Embase, Medline, PubMed, and the Cochrane Library were searched for studies published prior to September 2020. We assessed eligible studies for the association between nutritional status or interventions and foot ulcers. Fifteen studies met the inclusion criteria and were included in this review. Overall, there is a correlation between poor nutritional status and the presence of foot ulceration or a delay in healing. However, there is not enough data to reach conclusions about whether the relationships are causal or only association. Further research is required to test whether any forms of nutritional supplementation improve foot ulcer healing.
1. Introduction
Diabetic foot ulcers (DFU) are a severe, yet unfortunately common, complication of diabetes which cause substantial morbidity. They are also associated with high mortality risk, which is similar to overall five-year cancer mortality [1]. It is estimated that 19–34% of people with diabetes will develop a foot ulcer in their lifetime, and many of these will result in a digit or lower limb amputation [2]. Every year in Australia, there are 10,000 hospital admissions for DFU, with approximately 4400 of these resulting in an amputation [3,4]. This has large costs to the health care system. Research suggests that investing in evidence-based practice for DFU could save the Australian government and health-care system around $2.7 billion over five years [5].
It is estimated that 85% of these diabetes related amputations could be prevented if wounds were detected early enough and managed appropriately [6]. Prevention of lower limb amputation would have significant positive effect on quality of life (QoL) of the individual and would be cost-effective.
The International Working Group on the Diabetic Foot (IWGDF) 2019 guidelines state on page 164, “do not use interventions aimed at correcting the nutritional status (including supplementation of protein, vitamin and trace elements, pharmacotherapy with agents promoting angiogenesis) of patients with a diabetic foot ulcer, with the aim of improving healing in preference to best standard of care”. However, it is known that certain vitamins and minerals play important roles in wound healing [7]] and if people are deficient in those factors, their wound healing would be impaired.
Deficiency of vitamin C in its severest form causes scurvy. Symptoms can include bleeding gums, petechiae, arthralgia, tiredness, delayed wound healing, and an ability to bruise easily [8]. Vitamin C deficiency results in defective formation of connective tissues and collagen [8]. Vitamin C is a co-factor in lysine and proline hydroxylation on collagen molecules and hydroxylation of these amino acids is essential in stabilising the triple-helix structure of mature collagen [7]. This is especially important in the proliferation phase of wound healing when fibroblasts produce collagen which forms the scaffolding structure to support healing. The hydroxylation also provides essential tensile strength to the newly formed collagen, which would otherwise not stretch without tearing [9]. Tensile strength is important in healed wounds; if it is not sufficient, wounds may break down, leading to re-ulceration [2].
Vitamin C also plays a critical role in immune function which needs to be taken into consideration in foot ulcers because of the break in the epithelial barrier [9]. It is required for innate immune function, including the neutrophil ‘oxidative burst’ which is important for early bacterial killing. Anderson [2005] advocates that vitamin C is the most essential micronutrient in wound healing due to its role in collagen synthesis and angiogenesis [9].
Vitamin A is an essential fat soluble vitamin which has numerous bodily functions. Dietary vitamin A is absorbed as retinol from either preformed retinoids or provitamin A carotenoids [10]. Retinoids play an important role in wound healing as it is involved in cell mitosis, angiogenesis, increasing epithelial thickness, and collagen synthesis [11].
Zubair et al. reported that lower concentrations of vitamin D played a role in the pathogenesis of DFU and a 2019 meta-analysis reported that severe vitamin D deficiency is associated with the pathogenesis of foot ulceration [12,13]. Vitamin D is important for normal skin function; mice with deletion of the vitamin D receptor having a number of skin problems and poor wound healing [14].
Minerals such as zinc, copper, and selenium have roles in protein and collagen synthesis and cell replication, as well as antioxidant properties [15]. Zinc deficiency affects all phases of wound healing and causes a reduction in mature B-cells. Copper assists in the formation of red blood cells and aids vitamin C to form elastin [15]. Selenium is involved in antioxidant function and may protect the body from infection [16].
This paper will review published data about nutritional deficiencies or supplementations and healing of DFU.
2. Materials and Methods
The following databases were searched to identify relevant studies for inclusion:
- (i)
- Cochrane Library (searched 17 September 2020)
- (ii)
- Ovid Embase (searched 17 September 2020)
- (iii)
- Ovid MEDLINE (searched 17 September 2020)
- (iv)
- PubMed (searched 17 September 2020)
The combination of search terms used was as follows: (vitamin OR nutrient OR supplement) AND (foot OR feet) AND (wound OR ulcer OR ulceration). For PubMed, the search terms used were MeSH (Foot ulcer) and nutri*.
No limitations were placed on date of publication or language. Titles and abstracts were reviewed, and full copies were obtained for those potentially eligible for inclusion.
One author (N.B.) screened the search results. After duplicates were removed, titles and abstracts of the remaining papers were screened. The full manuscripts for those considered potentially relevant were read. The reference lists for these papers were also examined for potential studies to be included in this review resulting in the retrieval of one further study for full text reading. The search process is outlined in Figure 1.
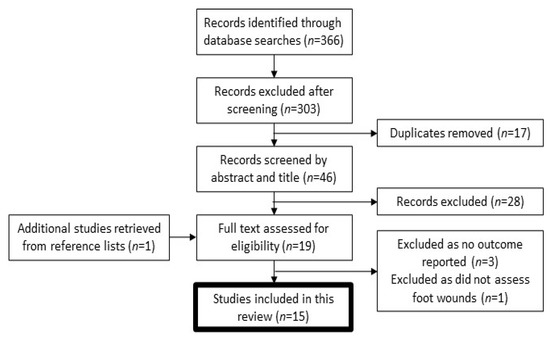
Figure 1.
Flow diagram of study selection for the systematic review.
Inclusion and Exclusion Criteria
Studies had to meet the following criteria for inclusion: (a) randomised controlled trials, cohort studies or case-control studies; (b) analysis of the relationship between a nutrient or mineral, and foot ulceration; (c) report on serum concentrations or deficiency rates of the nutrient or mineral being analysed; and (d) human studies.
Systematic review articles and case reports were excluded. For the study to be included, there had to be at least one foot outcome reported.
3. Results
3.1. Study Selection
Following the database searches, a total of 366 articles were found. Of these, 303 papers were excluded after screening and 17 duplicates were removed, leaving 46 articles for abstract review. Eighteen full text articles were assessed along with their reference lists. One study was retrieved for full text reading from a reference list. Of these 19 articles, 15 were suitable for inclusion in the study. The other four were excluded because no outcome was reported (n = 3) or because they did not assess foot wounds (n = 1).
3.2. Included Studies
Nine studies were randomised controlled trials [17,18,19,20,21,22,23,24,25]. There were two case control studies [26,27]. Four were cohort studies; one was a retrospective study [28], and three were prospective [12,29,30]. The studies are described in Table 1.

Table 1.
Characteristics of the study groups.
All studies were published in English. Six studies were reported from Iran [17,19,20,21,24,25] all by the same group. Two studies were carried out in India [12,18], two in the USA [22,28], and one in each of the following; Australia [29], Bahrain [30], Nigeria [26], Saudi Arabia [27], and Sweden [23].
3.3. Demographic Data
The studies had a total of 1737 participants in the pooled sample; 929 were in the intervention or case groups, and 808 people were controls. Of the 808 in the control groups, 381 had diabetes and foot ulcers, 377 had diabetes without foot ulcers, and 50 did not have diabetes or foot ulceration. Thirteen studies included both males and females, one study included males only [28], and the remaining study did not report gender [26].
The characteristics of both groups of participants, those with and without foot disease, are summarised in Table 1.
Populations evaluated in the included studies were as follows:
- (i)
- Six RCTs, all from the same senior author in Iran, included only participants with grade 3 DFU according to the Wagner classification system [17,19,20,21,24,25];
- (ii)
- One RCT included participants with grade 2–3 DFU [18,23];
- (iii)
- One RCT included participants grade 1–2 DFU [23];
- (iv)
- The remaining RCT included participants with diabetes who had at least one University of Texas grade 1A foot ulcer [22];
- (v)
- One retrospective [28] and one prospective study [29] included those with diabetes and foot ulceration;
- (vi)
- One case-control [27] and one prospective study [12] included participants with diabetes and foot ulceration, and a control group with diabetes but no foot ulceration;
- (vii)
- The other case-control study included participants with diabetes and foot ulceration, and a control group without diabetes or foot ulceration [26];
- (viii)
- The remaining prospective cohort study included any participants with foot ulceration; 90% of whom had diabetes [30].

Table 2.
Wagner-Meggitt classification.

Table 3.
University of Texas foot ulcer classification.
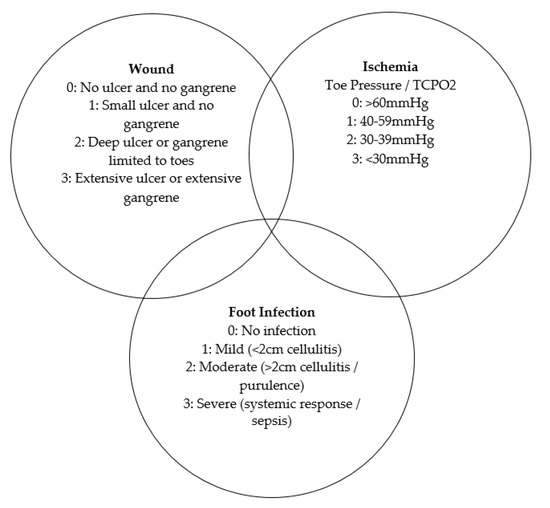
Figure 2.
The three intersection rings of the WIfI scoring system to help identify which risk is dominant at a given time [31].
3.3.1. Vitamin C
Two articles met the inclusion criteria and reported vitamin C concentrations in individuals with foot ulceration [26,29] (Table 4).

Table 4.
Overview of study design and results of included articles other than the Aseni group.
A case-control study by Bolajoko et al. (2017) assessed vitamin C, vitamin E, and selected minerals in people with diabetes and foot ulceration. There were 70 individuals with DFU and 50 participants free of diabetes and foot ulceration. The researchers measured vitamin C, vitamin E, copper, selenium, zinc and oxidative stress markers including lipid peroxide, total antioxidant status, superoxide dismutase and glutathione peroxidase. Significantly lower vitamin C (3.8 vs. 5.6 μmol/L, p = 0.003), selenium (0.48 vs. 0.81 μmol/L, p < 0.001), vitamin E (19.6 vs. 25.6 μmol/L, p < 0.001) and total antioxidant status (0.67 vs. 1.42 mmol/L, p < 0.001) concentrations were found in the DFU group compared to controls. Unexpectedly, people with DFU had higher glutathione peroxidase. Vitamin C was significantly positively correlated with glutathione peroxidase. The vitamin C assay was ‘old school’ and it is noteworthy that everyone tested had severe deficiency by usual-assay-standards, including the people without diabetes or foot ulcers [32]. People with DFU had concentrations which were clearly lower at 3.76 vs. 5.57 μmol/L.
In the prospective cohort study by Pena et al. (2020) serum concentrations of vitamins A, C, and D, and copper and zinc were measured in patients with DFU [29]. Vitamin C deficiency (<11.4 μmol/L) affected 50.8% of patients, with suboptimal concentrations (between 11.4 and 22.7 μmol/L) in another 22.2%. Over half of the 131 participants had low or undetectable plasma vitamin C. It was concluded that increased severity of DFU, assessed using the WIfI score (Figure 2), was associated with lower concentrations of vitamin C (p = 0.02).
3.3.2. Vitamin D
Four studies report on vitamin D as the primary measure [12,18,19,30]. Another study by Pena et al. (2020) also reported on vitamin D deficiency in people with DFU.
Kamble et al. (2020) conducted a RCT with 60 participants. Group A was prescribed a weekly 60,000 IU sachet of cholecalciferol, and group B received a placebo. The trial ran for a period of 12 weeks and vitamin D concentrations significantly increased (p = 0.0001). They reported significant improvements in HbA1c (p = 0.008) and cholesterol concentrations (p = 0.0001). The study reported beneficial effects on ulcer healing when compared to the control group (p = 0.0001). This is one of the 6 studies from the Aseni group in Iran. We note the six studies have improbable homogeneity of recruited subjects and benefits from the six treatments (Table 5).

Table 5.
Reports from the Aseni group.
Razzaghi et al. (2017), also from the Aseni group completed a study of very similar design with 60 participants. The intervention group received 50,000 IU of vitamin D every 2 weeks over 12 weeks, and controls received placebo. The patients had diabetes with 100% using insulin therapy, and 100% of patients had Wagner-Meggitt’s grade 3 lesions (deep ulcer with abscess or osteomyelitis). Randomisation was by computer generated random numbers. “The randomised allocation sequence, enrolling participants and allocating them to interventions were conducted by a trained staff at the clinic”. While practical for a busy clinic, this would not usually be considered a reliable means of maintaining study blinding.
They report significant decreases in ulcer length (p = 0.001), width (p = 0.02) and depth (p < 0.001) between the groups, however, the study inappropriately reports size to the nearest cm for each dimension (see Figure 1 and Figure 2 in the paper). Data is not provided for success with complete healing. Baseline vitamin D was lower in the vitamin D group, probably non-significantly. As the mean level was deficient, a large proportion of that group were presumably deficient at baseline. A significant reduction in HbA1c compared to the placebo was also reported (p = 0.004) as were reductions in LDL and total HDL to cholesterol ratio and C-reactive protein (CRP).
Two prospective cohort studies assessed vitamin D and wound healing [12,30]. Smart et al. (2019) included 80 participants with current foot ulceration who had blood values for vitamin D and HbA1c measured. A three-dimensional camera was used to measure each ulcer. None of the participants included in the study had sufficient vitamin D concentrations, 15% were insufficient and 85% were deficient. The researchers concluded that poor wound healing is linked to vitamin D deficiency and poor glycaemic control using general linear modelling.
The other prospective study included a total of 324 participants, all with diabetes [12]. Of these, 162 had foot ulcers and 162 were ulcer free. Greater vitamin D inadequacy (97.1%) was reported in DFU patients when compared to controls. Low serum vitamin D correlated with foot ulceration by linear regression (p < 0.001).
In the study by Pena et al. (2020), it was also reported that 55.7% of patients attending the foot wound clinics in Adelaide were deficient in vitamin D.
These studies indicate potential for vitamin D in the treatment of foot ulcers, particularly in people with baseline deficiency. Replication of the studies in larger cohorts is needed.
3.3.3. Vitamins B12 and Folic Acid
Two studies assessed impact of vitamin B on foot ulceration [27,28]. Badedi et al. (2019) included 323 participants; 108 with DFU and 215 control participants with diabetes. Vitamin B12 concentrations were measured and those with concentrations <148 pmol/L were defined as deficient. Researchers reported a significant correlation between DFU and vitamin B12 deficiency. When covariates were adjusted, there was a significant association between vitamin B12 deficiency and the presence of a DFU, with a 95% CI. Those with diabetes and vitamin B12 deficiency had odds 3.1 times higher of a DFU than those with diabetes without vitamin B12 deficiency. However it has been reported that long term metformin use increases the risk of vitamin B12 deficiency [33]. When compared with placebo, individuals taking metformin had lower vitamin B12 concentrations by 19% [33]. Therefore, this should be taken into consideration for those who have been on metformin for a long period of time.
A non-randomised cohort study by Boykin et al. (2020) recruited participants who were given a daily supplement of 5 mg folic acid (B9), 4 mg cyanocobalamin (B12), and 50 mg pyridoxine (B6) for one month. This study reported significant improvements in ulcer dimensions when comparing the four-week periods before and after supplements (p < 0.05).
While further studies are indicated, vitamin B12 deficiency is relatively common in people taking metformin and in people who have a vegan diet. We recommend measuring concentrations in those groups, and treating deficiency whenever it is present.
3.3.4. Vitamin E
In a third study from the Aseni group, Afzali et al. (2019) completed a RCT with 57 participants who were divided into two groups for a 12-week study. Those in the intervention group received 250 mg magnesium oxide plus 400 IU vitamin E daily and the controls received placebo daily. The study reports that magnesium plus vitamin E, compared to placebo, reduced ulcer length (p = 0.003), width (p = 0.02), and depth (p = 0.02). They also reported a significant reduction in HbA1c (p < 0.003) and a decrease in triglycerides, LDL-cholesterol, erythrocyte sedimentation rate (ESR) and increase in HDL-cholesterol. It is not clear which part of the co-supplementation led to these positive effects. The study has similar potential issues with measurement of ulcers, and randomization and blinding to the other study discussed above.
3.3.5. Omega-3 Polyunsaturated Fatty Acids
Soleimani et al. (2017) in a 4th study from the Aseni group reported a RCT with 60 subjects; participants were allocated to receive either 1000 mg omega-3 polyunsaturated fatty acids (PUFA) or a placebo twice daily for 12 weeks. By 12 weeks, they reported significant decreases in ulcer length (p = 0.03), width (p = 0.02) and depth (p = 0.01). A significant reduction in HbA1c (p = 0.01) and rise in insulin sensitivity (p = 0.002) was also reported when compared with the placebo group. Omega-3 PUFA supplementation also showed to significantly decrease CRP levels (p = 0.01). The study has similar potential issues with measurement of ulcers, and randomization and blinding.
We note lack of effect of omega-3 PUFA on HbA1c or markers of insulin resistance in people with type 2 diabetes reported in a Cochrane review [34] and in a more recent meta-analysis in 2015 [35].
3.3.6. Minerals (Magnesium, Selenium, Zinc, Copper)
As described above, Afzali et al. (2019) reported testing co-supplementation of magnesium and vitamin E. Magnesium (mg/dL) in the control group was 1.51 at baseline compared to 1.50 at end of study, compared to 1.55 vs. 1.83 (p < 0.001) in the intervention group. They reported this had beneficial effects on ulcer size, HbA1c, and lipid concentrations.
In the study by Bolajoko et al. (2017) significant differences in selenium (0.48 vs. 0.81 μmol/L) were noted between the DFU group and the healthy controls (p < 0.001). Only non-significant decreases in super-oxide dismutase activity and copper and zinc concentrations were observed (p > 0.05).
Pena et al. (2020) measured serum concentrations of copper and zinc in the 131 patients included in the study. Zinc deficiency was present in 26.9% of DFU patients. This study did not perform any statistical analyses for copper as all participants were in the normal range.
Razzaghi et al. (2018) in a fifth study from the Aseni group reported a RCT of 70 participants with DFU. The subjects were randomised to receive either 250 mg of magnesium oxide supplements daily, or a placebo, over a period of 12 weeks. Following the 12-week treatment period, they reported that magnesium supplementation resulted in decreased ulcer length (p = 0.01), width (p = 0.02), and depth (p = 0.003), compared with placebo. The researchers also reported significant reductions in HbA1c (p = 0.03) and fasting glucose (p = 0.04). Additionally, when compared with the placebo group, there was also a marked reduction in CRP levels (p = 0.01). We note that this trial is under investigation by the publishing journal as concerns have been raised about the report.
Momen-Heravi et al. (2017), again from the Aseni group completed a RCT of another cohort of 60 people with DFU who were all also 100% insulin-requiring people who all had 100% Wagner-Meggitt’s Grade 3 ulcers. The participants were randomised to take either 220 mg zinc sulfate, containing 50 mg elemental zinc, or a placebo daily over a period of 12 weeks. The researchers reported that zinc supplementation resulted in significant decreases in ulcer length (p = 0.02), width (p = 0.02), and depth (p = 0.05), when compared to the placebo group. They also reported significant reductions in HbA1c (p = 0.01) from baseline to the end of study in the intervention group.
3.3.7. Probiotics
Mohseni et al. (2018) in a sixth study the Aseni group reported evaluating the effects of probiotic supplementation on healing of DFU in 60 participants. Participants were randomly divided into two groups, to receive either a probiotic capsule or placebo daily for 12 weeks. The probiotic contained Lactobacillus acidophilus, Lactobacillus casei, Lactobacillus fermentum, and Bifidobacterium bifidum. Once again, they reported significant improvements in ulcer length (p = 0.01), width (p = 0.02), and depth (p = 0.02) in the intervention group, when compared with placebo. Additionally, when compared with the placebo, decreases in total cholesterol (p = 0.02) and CRP (p = 0.02) levels were also reported. As with all of the other studies from this group, the treatment was reported to cause a significant decrease in HbA1c.
3.3.8. Amino Acids
A RCT by Armstrong et al. (2014) recruited 270 participants with at least one University of Texas grade 1A foot ulcer. Participants were randomised to receive either arginine, glutamine, and beta-hydroxy-beta-methylbutyrate or a placebo, twice daily, for 16 weeks. There were no significant differences in wound healing in non-ischemic patients or those with sufficient baseline albumin concentrations. However, a significantly greater number of those with low baseline albumin concentrations, healed at 16 weeks in the intervention group when compared with controls (p = 0.03). Those with an ankle-brachial index of <1.0 also had significant healing rates when compared to the placebo (p = 0.008). These analyses are not stated in the report to be pre-specified, so are likely to be post-hoc.
3.3.9. Protein-Energy Supplementation
Eneroth et al. (2004) completed a 6-month RCT of 53 participants with DFU. Participants received either a 400 mL liquid nutritional supplement (20 g protein per 200 mL bottle, 1 kcal/mL, with added vitamins, minerals and trace elements) or a 400 mL placebo daily. Forty patients completed the six-month study and there was no significant effect of supplementation. Baseline protein malnutrition was associated with a non-significantly lower rate of healing at 6 months (24% versus 50% in those without).
4. Discussion
Foot ulceration is a challenging complication seen in patients with diabetes, peripheral neuropathy, abnormal foot architecture and/or peripheral arterial disease. Normal wounds should re-epithelialize within 72 h. Foot ulceration markedly increases the risk of lower limb amputation and can result in prolonged hospitalisations with an increased burden and cost to the healthcare system and the patient [6].
Unfortunately, 6 of the 15 identified studies from the same group (reviewed in Table 5) raise concerns due to homogeneity of the recruited patients and the results from the different treatments, including the report that each of the treatments improved HbA1c and ulcer outcomes significantly. It has also been noted that some of these studies are currently under investigation.
Eight of the other nine studies concluded that concentrations of their studied nutrient correlated with healing or that supplements improved foot ulcer healing. Overall, we conclude that better understanding the mechanism of action of these nutritional supplements, may add to future protocols and guidelines for the treatment of foot ulceration. Most of the sample sizes were small, but the associations are significant enough to suggest that a number of micronutrients may be important in wound healing.
To date, there has only been one similar systematic review completed in 2020 by Moore et al. The authors only included RCTs in their review and reported that it was unclear whether there was a difference in healing or amputations rates between intervention and control groups. Therefore they concluded that there is insufficient evidence to support or refute treating foot ulceration with nutritional interventions and that future studies should be larger and designed to a high standard [36].
Based on the findings of this review, vitamin D and B-complex vitamins may be effective in healing of foot ulceration. Both of these are relatively safe but should be done so under dietetic and/or medical supervision. Supplementation with vitamin C is also considered safe in usual supplemental doses (up to 1000 mg daily) as it is water soluble and excess intake is excreted in the urine [7]. A pilot study available after the date of this systematic review suggests that there is benefit from vitamin C supplements in people with foot ulcers [37].
Depending on the local clinic population, we recommend consideration of testing of appropriate nutritional factors and correction of any deficiencies identified. It is important to note that supplementation of some nutrients can be harmful when the correct dosage is exceeded. Dietary supplementation in the setting of foot ulcer should only be commenced following individual advice, and preferably after measurement of the nutrient under consideration.
Strengths and Limitations
This is a systematic review, a method which limits bias in determining study inclusion. We deliberately used relatively open search terms, as described in the methods. However, while this review concludes that there are correlations between nutrient deficiencies and foot ulceration, the studies themselves have limitations. As discussed above, six of the articles, which were by the same team of investigators (reviewed in Table 5), raise concerns due to the close similarities between recruited patients and highly similar outcomes across the set of studies. We are unsure whether this data is reliable.
This study did not require HREC/IRB approval as it was a systematic review.
Many of the other studies had small sample sizes and completing further studies using large RCTs will be important. Unfortunately, most of the studies currently published do not report on the two outcomes of greatest clinical interest; complete healing of the ulcer(s) and rate of amputation, and obviously, future studies providing this data is desirable.
Future trials should be of moderate to large scale, preferably multi-centre and look at specific supplementation and the effects on healing of foot ulceration over a period of time. This future research may have the ability to strengthen the evidence that is currently available, and in turn improving outcomes for those with foot ulcers and reducing amputation rates.
5. Conclusions
Most published reports find a correlation between nutritional status/supplementation and foot ulcers. However, many of the sample sizes are small, or the studies cannot conclude causation between a specific deficiency and the delay in healing and thus provide associative data. Therefore, further studies should be completed to investigate macronutrient or micronutrient supplementation and foot ulcer healing. Regardless, in clinically appropriate populations, we recommend consideration of testing for nutritional status and replacement in all cases where deficiency is identified.
Author Contributions
Conceptualisation, N.B. and J.E.G.; Methodology, N.B. and J.E.G.; Validation, N.B., J.E.G. and V.F.; Formal analysis, N.B. and J.E.G.; Investigation, N.B.; Writing—original draft preparation, N.B.; Writing—review and editing, J.E.G., V.F., T.-M.H. and C.M.; Visualisation, N.B. and J.E.G.; Supervision, J.E.G., V.F. and T.-M.H.; Project administration, N.B. All authors have read and agreed to the published version of the manuscript.
Funding
N.B. is supported by a Kickstarter Grant from the Research and Education Network, Western Sydney Local Health District. J.E.G. receives research support from NHMRC Program Grant Complexity in Nutrition.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
CRP, C-reactive protein; Cu, Copper; DFU, Diabetic foot ulcers; ESR, Erythrocyte sedimentation rate; HDL, High-density lipoprotein; IWGDF, International Working Group on the Diabetic Foot; LDL, Low-density lipoprotein; Mg, Magnesium; PUFA, Polyunsaturated fatty acids; QoL, Quality of Life; RCT, Randomised controlled trial; Se, Selenium; Vit, Vitamin; Zn, Zinc.
References
- Boulton, A. The diabetic foot: A global view. Diabetes Metab. Res. Rev. 2000, 16, S2–S5. [Google Scholar] [CrossRef]
- Reardon, R.; Simring, D.; Kim, B.; Mortensen, J.; Williams, D.; Leslie, A. The diabetic foot ulcer. Aust. J. Gen. Pract. 2020, 49, 250–255. [Google Scholar] [CrossRef] [PubMed]
- ACSQHC. Australian Atlas of Healthcare Variation Series: Australian Government. 2016. Available online: http://www.safetyandquality.gov.au/atlas/ (accessed on 24 July 2020).
- Davis, W.; Norman, P.E.; Bruce, D.; Davis, T. Predictors, consequences and costs of diabetes-related lower extremity amputation complicating type 2 diabetes: The Fremantle Diabetes Study. Diabetology 2006, 49, 2634–2641. [Google Scholar] [CrossRef]
- Cheng, Q.; A Lazzarini, P.; Gibb, M.; Derhy, P.H.; Kinnear, E.M.; Burn, E.; Graves, N.; E Norman, R. A cost-effectiveness analysis of optimal care for diabetic foot ulcers in Australia. Int. Wound J. 2016, 14, 616–628. [Google Scholar] [CrossRef]
- Bakker, K.; Apelqvist, J.; Schaper, N.; International Working Group on the Diabetic Foot Editorial Board. Practical guidelines on the management and prevention of the diabetic foot 2011. Diabetes Metab. Res. Rev. 2012, 28, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Collins, N. Nutrition 411: Revisiting vitamin C and wound healing. Ostomy Wound Manag. 2013, 59, 12. [Google Scholar]
- Christie-David, D.J.; Gunton, J.E. Vitamin C deficiency and diabetes mellitus—Easily missed? Diabet. Med. 2017, 34, 294–296. [Google Scholar] [CrossRef] [Green Version]
- Anderson, B. Nutrition and wound healing: The necessity of assessment. Br. J. Nurs. 2005, 14, S30–S38. [Google Scholar] [CrossRef]
- Polcz, M.E.; Barbul, A. The Role of Vitamin A in Wound Healing. Nutr. Clin. Pract. 2019, 34, 695–700. [Google Scholar] [CrossRef]
- Zinder, R.; Cooley, R.; Vlad, L.G.; Molnar, J.A. Vitamin A and Wound Healing. Nutr. Clin. Pract. 2019, 34, 839–849. [Google Scholar] [CrossRef]
- Zubair, M.; Malik, A.; Meerza, D.; Ahmad, J. 25-Hydroxyvitamin D [25(OH)D] levels and diabetic foot ulcer: Is there any relationship? Diabetes Metab. Syndr. Clin. Res. Rev. 2013, 7, 148–153. [Google Scholar] [CrossRef]
- Dai, J.; Jiang, C.; Chen, H.; Chai, Y. Vitamin D and diabetic foot ulcer: A systematic review and meta-analysis. Nutr. Diabetes 2019, 9, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oda, Y.; Tu, C.-L.; Menendez, A.; Nguyen, T.; Bikle, D.D. Vitamin D and calcium regulation of epidermal wound healing. J. Steroid Biochem. Mol. Biol. 2016, 164, 379–385. [Google Scholar] [CrossRef] [Green Version]
- Quain, A.M.; Khardori, N.M. Nutrition in Wound Care Management: A Comprehensive Overview. Wounds 2015, 27, 327–335. [Google Scholar] [PubMed]
- ODS. Selenium USA: USA Government. 2020; [updated 11 March 2020]. Available online: https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/ (accessed on 24 July 2020).
- Afzali, H.; Kashi, A.H.J.; Momen-Heravi, M.; Razzaghi, R.; Amirani, E.; Bahmani, F.; Gilasi, H.R.; Asemi, Z. The effects of magnesium and vitamin E co-supplementation on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. Wound Repair Regen. 2019, 27, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Kamble, A.; Ambad, R.; Padamwar, M.; Kakade, A.; Yeola, M. To study the effect of oral vitamin D supplements on wound healing in patient with diabetic foot ulcer and its effect on lipid metabolism. Int. J. Res. Pharm. Sci. 2020, 11, 2701–2706. [Google Scholar] [CrossRef]
- Razzaghi, R.; Pourbagheri, H.; Momen-Heravi, M.; Bahmani, F.; Shadi, J.; Soleimani, Z.; Asemi, Z. The effects of vitamin D supplementation on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J. Diabetes Its Complicat. 2017, 31, 766–772. [Google Scholar] [CrossRef]
- Soleimani, Z.; Hashemdokht, F.; Bahmani, F.; Taghizadeh, M.; Memarzadeh, M.R.; Asemi, Z. Clinical and metabolic response to flaxseed oil omega-3 fatty acids supplementation in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. J. Diabetes Its Complicat. 2017, 31, 1394–1400. [Google Scholar] [CrossRef]
- Razzaghi, R.; Pidar, F.; Momen-Heravi, M.; Bahmani, F.; Akbari, H.; Asemi, Z. Magnesium Supplementation and the Effects on Wound Healing and Metabolic Status in Patients with Diabetic Foot Ulcer: A Randomized, Double-Blind, Placebo-Controlled Trial. Biol. Trace Element Res. 2017, 181, 207–215. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Hanft, J.R.; Driver, V.R.; Smith, A.P.S.; Martinez, J.L.L.; Reyzelman, A.M.; Furst, G.J.; Vayser, D.J.; Cervantes, H.L.; Snyder, R.J.; et al. Effect of oral nutritional supplementation on wound healing in diabetic foot ulcers: A prospective randomized controlled trial. Diabet. Med. 2014, 31, 1069–1077. [Google Scholar] [CrossRef]
- Eneroth, M.; Larsson, J.; Oscarsson, C.; Apelqvist, J. Nutritional supplementation for diabetic foot ulcers: The first RCT. J. Wound Care 2004, 13, 230–234. [Google Scholar] [CrossRef]
- Mohseni, S.; Bayani, M.; Bahmani, F.; Tajabadi-Ebrahimi, M.; Bayani, M.A.; Jafari, P.; Asemi, Z. The beneficial effects of probiotic administration on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. Diabetes/Metab. Res. Rev. 2018, 34, e2970. [Google Scholar] [CrossRef]
- Momen-Heravi, M.; Barahimi, E.; Razzaghi, R.; Bahmani, F.; Gilasi, H.R.; Asemi, Z. The effects of zinc supplementation on wound healing and metabolic status in patients with diabetic foot ulcer: A randomized, double-blind, placebo-controlled trial. Wound Repair Regen. 2017, 25, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Bolajoko, E.B.; Akinosun, O.M.; Anetor, J.; Mossanda, K.S. Relationship between selected micronutrient deficiencies and oxidative stress biomarkers in diabetes mellitus patients with foot ulcers in Ibadan, Nigeria. Turk. J. Med Sci. 2017, 47, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Badedi, M.; Darraj, H.; Hummadi, A.; Solan, Y.; Zakri, I.; Khawaji, A.; Daghreeri, M.; Budaydi, A. Vitamin B12 Deficiency and Foot Ulcers in Type 2 Diabetes Mellitus: A Case–Control Study. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 2589–2596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boykin, J.V.; Hoke, G.D.; Driscoll, C.R.; Dharmaraj, B.S. High-dose folic acid and its effect on early stage diabetic foot ulcer wound healing. Wound Repair Regen. 2020, 28, 517–525. [Google Scholar] [CrossRef]
- Pena, G.; Kuang, B.; Cowled, P.; Howell, S.; Dawson, J.; Philpot, R.; Fitridge, R. Micronutrient Status in Diabetic Patients with Foot Ulcers. Adv. Wound Care 2020, 9, 9–15. [Google Scholar] [CrossRef]
- Smart, H.; AlGhareeb, A.M.; Smart, S.-A. 25-Hydroxyvitamin D Deficiency: Impacting Deep-Wound Infection and Poor Healing Outcomes in Patients With Diabetes. Adv. Ski. Wound Care 2019, 32, 321–328. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Mills, J.L. Juggling risk to reduce amputations: The three-ring circus of infection, ischemia and tissue loss-dominant conditions. Wound Med. 2013, 1, 13–14. [Google Scholar] [CrossRef]
- Kyaw, A. A simple colorimetric method for ascorbic acid determination in blood plasma. Clin. Chim. Acta 1978, 86, 153–157. [Google Scholar] [CrossRef]
- de Jager, J.; Kooy, A.; Lehert, P.; Wulffelé, M.; van der Kolk, J.; Bets, D.; Verburg, J.; Donker, J.M.; Stehouwer, D.A. Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin B-12 deficiency: Randomised placebo controlled trial. BMJ 2010, 340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartweg, J.; Perera, R.; Montori, V.; Dinneen, S.; Neil, A.H.; Farmer, A.J. Omega-3 polyunsaturated fatty acids (PUFA) for type 2 diabetes mellitus. Cochrane Database Syst. Rev. 2008, CD003205. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Yu, X.; Shao, S. Effects of Omega-3 Fatty Acid Supplementation on Glucose Control and Lipid Levels in Type 2 Diabetes: A Meta-Analysis. PLoS ONE 2015, 10, e0139565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moore, Z.; Corcoran, M.; Patton, D. Nutritional interventions for treating foot ulcers in people with diabetes (review). Cochrane Database Syst. Rev. 2020, 7. [Google Scholar] [CrossRef]
- Gunton, J.E.; Girgis, C.M.; Lau, T.; Vicaretti, M.; Begg, L.; Flood, V. Vitamin C Improves Healing of Foot Ulcers; A Randomised, Double-Blind, Placebo-Controlled Trial. Br. J. Nutr. 2020, 2020, 1–21. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).