Abstract
Intense sports activities affect cognitive, perceptual, and physiological domains, possibly concurring in reducing pain perception and anxiety. Within a framework for a new socio-ecological narrative that considers opposition (such as fighting) and collaboration (cooperative learning) as carriers of self-control and pain perception inhibition, we investigated the acute effects of a judo-specific session on cognitive (inhibitory control and simple reaction time), perceptual (pain and fatigue), affective (anxiety) and physiological (hormone profile) domains. Eleven male black belt judokas (age 39.0 ± 13.9 years; training experience: 23.2 ± 14.4 years) were recruited and tested before and after judo randori and resting sessions. Inhibitory control and simple reaction time were assessed via the Flanker task and clinical reaction time test, pain and fatigue were assessed using Borg’s category-ratio scale (CR-10), and the state–trait anxiety inventory Y-1 test assessed anxiety. Glucose, insulin, cortisol, creatinine, and irisin levels were measured. Cognitive performance, perception of fatigue and physiological variables increased after randori bouts, while pain decreased. Inhibitory control, perception of pain, perception of fatigue and glucose, cortisol, and creatinine significantly differed (p < 0.05) between randori and resting sessions. A high-intensity randori may induce acute beneficial effects on cognitive, perceptual, and physiological domains. Further studies should compare the results with the outcomes from a collaborative and non-agonist environment and confirm the socio-ecologic framework.
1. Introduction
A multilevel socio-ecological model [1,2,3] is a current scientific form to interpret the complexity of the educative reality. Indeed, it considers the multifaced interaction between individuals and environmental features characterizing any sociocultural context [1,2].
A framework has been planned to investigate an environment based on an “opposites’ narrative” addressing two contrasting categorical conditions: sharing/integration/cooperation, and education to competition/fighting (Figure 1).
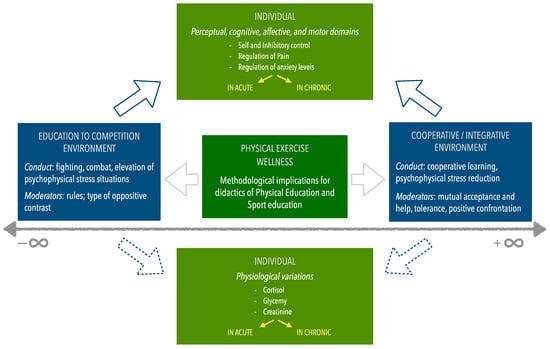
Figure 1.
The promotion of physical activity and wellness according to a socio-ecological multilevel framework based on environmental contrasts.
Both cooperative and agonistic practices could constitute coping mechanisms strategically used by people to manage stress and/or trauma, helping them to control pain and emotions influenced by physiological (hormone profile) variations. In particular, in pain management education, a contrasting approach can be characterized by both competitive activities such as martial arts (where the agonistic approach implies self and inhibitory control, and pain tolerance) and cooperative activities (which often imply the relief of a different kind of pain to the social pain) [4,5].
In this framework, the present study on combat sports is the first investigation step paralleling the effects of education between competitive and cooperative environments (firstly in acute and then chronic conditions) for methodological implications in physical and sports education.
Considering the education to competition, randori is a judo free-style practice between two opponents involving throwing, choking, holding down, and locking elbows. It emphasizes the development of perception, decision-making skills, and emotional control during competitive “free exercise” [6]. Randori simulates real judo fights with explosive movements lasting 10 to 63 s, separated by low-intensity hopping (1 to 22 s) and short referee interventions (about 9 s). It is characterized by a temporal structure of 3:1 or 2:1 effort/pause ratios with an intensity above maximal lactate steady state and a total duration between 1 and 5 min for each set [7]. The randori phase can last even 40 min in a typical training session with many fighting bouts [8]. Due to such characteristics, the free-style practice of randori is supposed to drive numerous effects within the cognitive, perceptual, and physiological domains, which are worth investigating.
In open-skill sports such as judo, successful randori requires inhibitory control and strategic selective attention. Athletes must adapt to unpredictable situations and choose appropriate responses [9]. The high intensity and perceptual-cognitive demands imposed by the randori may induce an acute positive effect on cognitive functions. In fact, the literature has extensively demonstrated that a bout of exercise can acutely improve cognitive performance when assessed immediately after the cessation of the exercise, albeit with various moderators [10,11,12].
Moreover, recent evidence supports the notion that physical activity with some forms of cognitive engagement (such as randori) may have more beneficial effects on cognitive functions as compared to physical activity with limited cognitive engagement (such as running or cycling involving automated repetitive movements) [13,14]. Consequently, this may translate into an enhanced involvement in a selective attention task with congruent and incongruent stimuli not directly related to sport-specific performance, such as in a cognitive task assessing inhibitory control [15,16]. In addition, simple reaction time implies executive functions (such as inhibitory control) to respond adequately and quickly to motor stimuli [17]. Thanks to practice, the improved efficiency in processing nervous system information considerably reduces the average reaction time in rapid hand movements [18]. A quicker motor action depends on attentiveness and better control of emotions [19,20]. Even though not directly related to judo-specific performance, reaction time combining mental and physical effort can also be utilized to indicate fatigue [21].
Pain is an impairing condition that makes actions such as thinking, acting and conducting satisfying daily interactions more difficult [22]. Pain modulation involves nociceptors detecting and transmitting pain signals to the central nervous system, which is altered during physical exercise, resulting in decreased pain perception during and after exercise [23]. The central nervous system plays a crucial role in decreasing pain in response to an acute physical exercise, in particular, reducing muscle pain [24]. The distraction determined with selective and specific attention requested in maximal competitive randori can reduce the perception of pain [25,26].
As for pain, physical exercise has also been shown to have an acute positive effect on reducing anxiety symptoms [27]. Given the features of the randori session, it might induce immediate positive effects on pain and anxiety, contributing to reduced pain perception and anxiety status during daily interactions. According to Rossi [28], anxiety or motivation could impact judokas’ performance differently from one to the other; anxiety may increase when an athlete is facing defeat. On the contrary, a successful athlete may control their emotions better and experience less anxiety. From a practical point of view, retrieving information on trait anxiety might help athletes alleviate its adverse effects immediately after exercise.
Hormonal processes are the base of the organism’s adaptative capacity. Therefore, these processes’ analysis can highlight the resulting physiological effects [29].
Although hormonal alterations can impact metabolic outcomes, studies on martial arts have scarcely investigated glucose metabolism. Nevertheless, cortisol raises plasma glucose [30] and is detected after stressful situations. However, even before competition, this response to competition occurrence has been found [31]. Similarly, single-bout exercises have been proven to increase insulin sensitivity for hours. This would reflect a better capability of the recruited muscles to take up glucose and use it for energy, irrespective of insulin level. Although, in general, physical exercise increases body sensitivity to insulin [32], little is known about how glucose and insulin would change after an acute randori session of judo-specific practice.
Irisin is a hormone-like myokine that mice and human skeletal muscle produce in response to exercise [33]. It induces white adipose tissue’s browning, increases energy expenditure, and protects against insulin resistance and obesity [34]. However, how physical activity promotes human irisin secretion has not been fully explained and controversial literature adduces exercise has great effects in increasing irisin [35] or reports unchanged irisin levels following acute and chronic exercise [36].
Since judo is a very engaging activity requiring a solid will during training and competitions to tolerate fatigue and pain, overcome fear, and manage aggressiveness and rage [37], the consequences of a judo randori on different psychophysiological variables (cognitive, perceptive, physiological, and affective domains) must be clarified.
Therefore, this study aimed to investigate the acute effects of a randori session on inhibitory control, simple reaction time, pain, perception of fatigue, anxiety and hormone profile, which are crucial variables for an individual’s general well-being. We hypothesized that such activity, which corresponds to one of the extremes of the opposite-based approach of the framework for a new socio-ecological narrative, is adequate to promote individual self-organization and cognitive, perceptive, psychological, and physiological adaptations.
2. Materials and Methods
Eleven male judo amateurs aged 18 to 58 years old (age 39.0 ± 13.9 years, height 1.74 ± 0.7 m, body mass 82.7 ± 21.6 kg, body mass index 26.8 ± 5.0 kg/m2, mean ± standard deviation, SD) participated in this study. To be included, athletes had to meet the following criteria: (i) have high skills in judo (black belt, i.e., ten years of experience at least); (ii) be healthy (no acute inflammations/illnesses); (iii) be adults (over 18 years); (iv) be active (practicing at least four hours per week in the preceding two months). The participants of this study were black belt judokas with a mean training (90 min < session practice time < 150 min) volume of 6–8 h per week and age of practice of 23.2 ± 14.4 years. In addition, they were healthy and on a regular diet, with standard glucose tolerance (<126 mg/dL according to American Diabetes Association) and were requested not to modify their diet during the data collection period.
According to the Declaration of Helsinki, all athletes signed a written informed consent before participating. Furthermore, all the procedures used complied with the Good Clinical Practice (GCP) principles approved by the ethics committee of Ospedale San Raffaele, reference number: 88/int/2016.
2.1. Study Protocol
The study was performed in a martial arts Dojo in two randomized experimental sessions (i.e., randori and resting sessions; Figure 2).
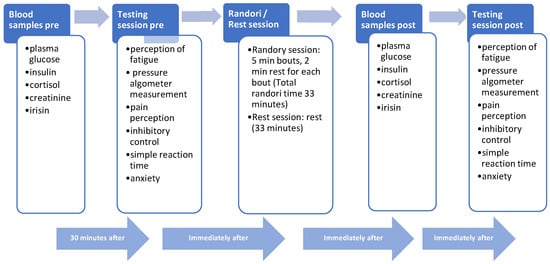
Figure 2.
Study protocol of the intervention. Each participant performed both randori and rest sessions one week apart.
In the randori session, participants performed a randori contact fighting lasting 5 min for bout, with 33 min total and 2 min rest between every bout. A federal judge supervised the randori according to the international rules of the International Judo Federation (IJF, https://www.ijf.org/ijf/documents/6, accessed on 2 March 2023).
Two blood samples were drawn from each participant to assess plasma glucose, insulin, cortisol, creatinine, and irisin pre- and post-randori. The pre-sample was collected 30 min before the randori fighting, while the post-sample was collected immediately after the end. In both the pre- and post-randori stages, perception of fatigue, pressure measurement using a pressure algometer, pain, inhibitory control were assessed after the blood sample withdrawal via the Flanker task, simple reaction time using the clinical reaction time and anxiety using the STAI-Y scale, respectively. In the experimental resting session, the participants remained seated for all 5 min of the bout for 33 total minutes instead of performing a randori session. Each participant randomly performed the randori and the rest sessions one week apart.
2.2. Procedures
2.2.1. Cognitive Domains
Cognitive performance was assessed following previous studies [38,39]. A computerized modified version of the Flanker task was used to assess inhibitory control [40]. Participants had to detect the direction of a target flanking arrow (the central of five flanking arrows) and press as quickly as possible, on a keyboard, button A when pointing to the left or button L when pointing to the right. One hundred trials were performed, equally distributed among congruent (n = 50 trials) and incongruent (n = 50) conditions. In the left and right congruent conditions, arrows pointed in the same direction as the target arrow (left: <<<<<; right: >>>>>), while in the incongruent condition, they pointed to the opposite direction (left: >><>>; right: <<><<) and incongruent (e.g., left: >><>>). Within two seconds of the stimulus presentation, participants had to decide and press the button corresponding to the target. The mean response time and number of correct answers were computed for congruent and incongruent conditions.
The clinical reaction time test as an indicator of fatigue assessed simple reaction time. The test was administered using a 1.3 m long stick ending in a weighted rubber disk and covered with high-friction tape marked every 0.5 cm. Participants were asked to sit with the dominant forearm on horizontal support to keep the upper limb relaxed. After 2 to 5 s random waits, the operator released the stick vertically suspended with the weight at the level of the open hand of the participant, who had to catch the stick as quickly as possible when it started falling. Participants performed two practice trials followed by eight testing trials. Thanks to marks on the stick, the distance between the weighted disk and the catch of each trial was measured, and the mean result was considered and converted into the reaction time.
2.2.2. Perceptual Domains
Pressure time, pressure, and pain were recorded using a pressure algometer (Salter Abbey Weighing Machines Ltd., Bury St. Edmunds, UK) pre- and post-condition [41]. The pressure algometer had a flat circular metal probe dressed in several layers of lint and measured 1 cm in diameter. The force was displayed digitally in increments of 0.1 N·cm−2. The operator set the algometer’s pointer on the mandibular branch of the facial nerve and performed an incremental pressure of 20 N·s−1. The pressure ended when the participant was no longer able to bear the pressure. The peak pressure reached (in N·cm−2), and pressure time (s) were recorded. Moreover, the operator asked for the perceived pain value using the category-ratio scale (CR-10) anchored at 10 [42]. To assess fatigue perception, participants were asked to rate their perception of effort using the CR-10 Borg’s scale [42] immediately pre-and post-each condition.
2.2.3. Affective Domain
We used the state–trait anxiety inventory (STAI-Y) to assess the affective domain. The STAI-Y is a questionnaire divided into two scales (Y1 and Y2) that evaluate state anxiety (the anxiety level at the moment) and trait anxiety (the usual anxiety level), respectively. The questionnaire is composed of 40 items (20 for trait anxiety and 20 for state anxiety). Both Y1 and Y2 scales have items addressed to determine whether anxiety is present or missing. Specifically, statements such as “I feel secure” denote a lack of anxiety, while statements such as “I feel worried” suggest anxiety is occurring. The self-evaluation is performed using a Likert scale ranging from 1 to 4. For the state anxiety, answers are scored as follows: 1 = not at all, 2 = somewhat, 3 = moderately so, 4 = very much so. For the trait anxiety: 1 = almost never, 2 = sometimes, 3 = often, 4 = almost always. The final score was calculated based on previous literature [43].
2.2.4. Physiological Domain
Blood samples (22.5) mL were taken in a sitting position via standard antecubital venipuncture and preserved on ice until plasma or serum centrifugation at 4 °C (within 1.5 h from sampling). Plasma and serum were frozen at −60 °C for later analysis in duplicate. A glucose analyzer was used to measure plasma glucose (Beckman Instruments, Fullerton, CA, USA). Free insulin was dosed via a highly specific two-site monoclonal antibodybased immunosorbent assay (ELISA; Dako Diagnostics, Cambridgeshire, UK). A commercial ELISA kit served to measure plasma cortisol. Creatinine was detected using a Beckman Coulter analyzer.
Quantifying circulating irisin levels on serum samples was executed with a specific enzyme immunoassay kit (Cat. EK-067-29, Phoenix EuropeGmbH, Karlsruhe, Germany). The minimum detectable concentration was 1.43 ng/mL. The intra- and inter-assay variations were less than 10% and 15%, respectively. Previous validation of the assays for irisin detection was performed.
2.3. Statistical Analysis
Data were expressed as mean ± standard deviation (SD). The normality of data distributions was checked using graphical methods and the Shapiro–Wilk’s test. The reliability of the Flanker task, pressure algometer and clinical reaction time test was assessed by comparing the data collected before the randori and rest sessions using the interclass correlation coefficient (ICC).
Student’s paired t-test or Wilcoxon test (in accordance with the normality test) was used to compare the difference of post and pre-conditions (delta) between the randori and the resting session for each variable. Moreover, effect sizes (ESs) were calculated for the magnitude of the difference between the delta of each condition (Cohen’s d effect size for variables normally distributed, and r effect size for variables not normally distributed). The corresponding ES thresholds for small, moderate, and large effects were classified as 0.2, 0.5, and 0.8, respectively (for Cohen’s d) and as 0.1, 0.3, 0.5, respectively (for r). Statistical analysis was performed using IBM SPSS Statistics (version 21, New York, NY, USA).
A p-value (p) lower than 0.05 was considered statistically significant, and further specifics lower than 0.01 and 0.001 were provided.
3. Results
All variables presented a moderate-to-excellent reliability for physical (pressure ICC = 0.989), cognitive (clinical reaction time ICC = 0.726, incongruent (ms) ICC = 0.808, incongruent (n° of corrects) ICC = 0.877, congruent (ms) = 0.878, congruent (n° of corrects) = 0.736), perceptual (pain ICC = 0.828, RPE ICC = 0.539) and affective (anxiety ICC = 0.905) variables.
Table 1 shows each variable’s pre, post, and delta values for randori and resting sessions, together with statistical comparisons.

Table 1.
Pre, post, and delta of each variable for the randori and resting sessions (Mean ± SD).
In the cognitive domain (Figure 3), the delta of the randori session was significantly different from the delta of the resting session in the number of correct responses of congruent (p < 0.014; d = 0.88, large) and incongruent condition (p < 0.009; r = 0.79, large), in inhibitory control of the Flanker task. The delta of the randori session was significantly different from the delta of the resting session for the response time of congruent (p = 0.014; d = 1.4, large) and incongruent condition (p = 0.009, r = 0.71, large) in inhibitory control of the Flanker task. For simple reaction time, the delta of the randori session was not significantly different from the delta of the resting session (p = 0.88; d = 0.06, small).
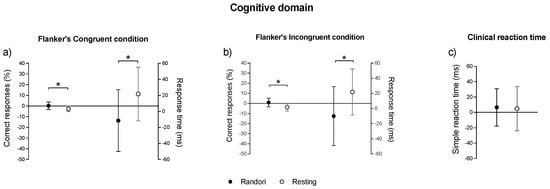
Figure 3.
Comparison between the delta values (post–pre) of cognitive domain between randori and resting sessions results, measured using the Flanker task (panels (a,b), congruent and incongruent conditions, respectively) and the clinical reaction time test (panel (c)). Significant values: * p < 0.01.
In the perceptual domain, the delta of randori session was not significantly different from delta of resting session in pressure time (p = 0.21; r = 0.25, small) and pressure (p = 0.77; d = 0.131, small). Conversely, pain (p = 0.015; d = 1.64, large) and RPE (p < 0.001; d = 0.63, moderate) evaluated using the CR-10 report were significantly different between the delta of the randori session and the delta of the resting session (Figure 4).
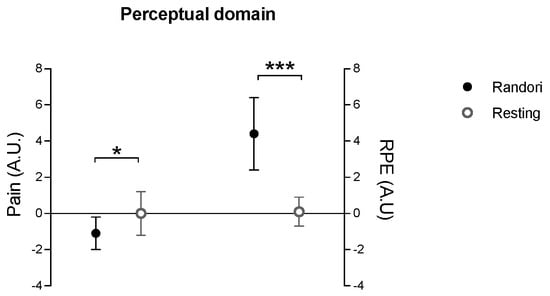
Figure 4.
Comparison between the delta values (post–pre) of perceptual domain between randori and resting sessions results. Significant values: * p < 0.01; *** p < 0.001.
In the affective domain, no significant difference was found between the delta of the randori session and the delta of the resting session in state anxiety (p = 0.44; r = 0.2, small).
In the physiological domain, significant differences were found between the delta of randori session and the delta of resting session in glucose (p = 0.005; d = 1.4, large), creatinine (p = 0.012; r = 0.7, large) and cortisol (p = 0.026; r = 0.66, large), whereas no differences were observed in irisin (p = 0.82; d = 0.07, small) and insulin (p = 0.66; r = 0.14, small).
4. Discussion
The main finding of the present study was that the cognitive, perceptual and physiological domains were differently affected by the randori session compared to the resting session. Specifically, the cognitive domain measured using the Flanker task significantly changed (number of correct responses and time, in both congruent and incongruent conditions) in response to the randori session.
The high perceptual-cognitive demands imposed by the randori session might be responsible for the positive effect on inhibitory control. Accordingly, it has been established that neuromotor exercise (as randori is) can improve cognitive performance immediately after its cessation [11,44]. In fact, besides the cardiovascular fitness hypothesis, the cognitive engagements required by physical tasks that involve non-automated repetitive movements (e.g., those targeting gross and fine body coordination) were proposed as a potential mediator for the acute beneficial effect of exercise on cognition [13,45]. The externally paced situation of randori involves selective and flexible attention to what the opponent does to suppress inappropriate actions and select correct behaviors. This strategic feature might support the exercise-induced changes of inhibitory control in response to randori.
Nevertheless, the cognitive domain evaluated using the clinical reaction time did not change after a randori session. A plausible explanation may be linked to the fact that this test assesses simple reaction time. Indeed, the increasing neuromotor benefits caused by the raising of selective attention evidenced in the Flanker task should have improved the simple reaction time; conversely, however, the incoming local fatigue of the hand caused by the randori session may have hidden this effect [46,47].
As regards physical and perceptual domains, the reduction in pain was more significant in response to the randori session than to the resting session, regardless of the applied pressure. In fact, no significant differences between the two conditions were found in the pre-post changes in pressure and pressure time (Table 1). The reduction in pain in response to the randori session was not surprising due to the well-known antalgic role of exercise [48]. Numerous studies demonstrated that an acute exercise session reduced perceived pain [48,49]. Furthermore, regular exercise reduces pain perception and affects mental health [49]. Specifically, positive effects on mood with consequent stress reduction and depression were found in response to an acute bout of exercise [50]. Heavy combat such as the randori session might mirror a condition of danger (as explained by the cortisol increase). However, in a fight-or-flight situation, the present study observed no change in state anxiety. In a situation of high emotional involvement, the evident increase in the capacity for self-control inhibition moderated by sporting rules that guarantee the safety of contenders probably prevented negative changes in anxiety levels and reduced pain perception.
Among the numerous factors proposed as potential modulators for reducing pain in response to exercise [50], particular attention has been recently devoted to the modulation of the serotonergic system in the brainstem. In addition, the mesocorticolimbic system, comprising the prefrontal cortex, amygdala nucleus accumbens, and hippocampus, represents a group of brain regions involved in pain’s cognitive and emotional aspects [50,51].
In the present study, the delta of glucose, creatinine, and cortisol levels in response to the randori session was significantly higher than those in response to the resting session; in addition, the randori session elicited higher glucose and cortisol levels from pre to post. Accordingly, cortisol is recognized as having direct effects on human increases in glucose production by soliciting gluconeogenesis [52]. Of note, it should be considered that the cortisol secretion could be induced by a natural fight-or-flight reaction to randori.
Regarding irisin, it did not significantly change after randori, although it tended to increase. As shown in a recent meta-analysis [53], plasma irisin seems to be increased in mouse and human muscles following exercise, and positive metabolic adaptations accompanied this rise. A recent meta-analysis reported that acute exercise elicited a substantial rise in circulating irisin concentration related to participants’ fitness levels [54]. The different experience levels of the participants could thus have hidden a substantial difference in the irisin level. Similarly, the creatinine values increased after the randori session. Overall, it has already been reported that endurance exercise [55] stimulates creatinine serum concentration because of an augmented creatinine release from muscle cells [56]. It may be plausible that the acute rise in creatinine depends on the potential traumatic activity of judo-specific actions. No variation in insulin was detected after the randori condition.
In synthesis, the feature of judo randori as an open-skill combat sport requiring the involvement of high-level psychophysical stress situations under rules dictating respect for the opponent might explain the improvements in participants’ inhibitory control and their reduction in pain perception.
A main limitation of this study is represented by the low number of participants and the discrepancy in their age and years of practice. As the present study was primarily addressed to investigate activities corresponding to opposite-based didactics as commonly occurring in usual education to training to competition of judo amateur (i.e., in ecologic environmental training conditions such as mixed participants, different ages and years of practice), the results have been considered without recurring to analyses using other independent variables except for technical level (that was the inclusion criterium: being a black belt). However, a broader range of participants would allow the collection of more information on the variables selected in the present investigation, permitting extensive analyses according to age, weight, and gender categories (as commonly used in competitions), possibly further strengthening generalizations of randori effects on an individual’s general well-being.
Moreover, the study’s design did not compare judo to other types of physical exercise; it used only a resting condition instead. Hence, this limited the strength of conclusions to generalize that combat sports might be of the utmost importance to induce benefits in cognitive, perceptual, and physiological domains compared to other activities.
Finally, it was impossible to control the participants’ diet. Even if they were asked not to change their diet during the study period, a standard diet would be preferable in future investigations.
Contextualization into the Framework for a New Socio-Ecological Narrative
The motor conduct that has been addressed in the present study is located at one of the two extremes assumed in the framework for a new socioecological narrative (competitive, opposed to cooperative).
The competition returns a result, with a winner and a loser. On the other hand, sports results require individual performance, respect for rules and full psychophysical involvement, implying physical effort, individual self and inhibitory control, and free selection of behaviors to achieve the goals. However, this also promotes ethical behaviors aimed at the good of oneself and others.
Upcoming steps of the framework will address acute cooperative learning effects [57] and investigate whether the related psychophysical dynamics under such an approach impact cognitive, perceptual, psychological, and physiological adaptation differently. Further intervention studies based on “in chronic” competition and cooperation implications will complete the analysis of the framework, also considering age, body mass index, gender, and practice specifics as moderators.
5. Conclusions
This study showed that a randori session probably affected inhibitory control, pain, perception of fatigue and hormone profile differently from a resting session in an amateur agonistics judoka. Fatiguing randori bouts definitely improve inhibitory control and reduce pain perception, generating acute beneficial effects on individuals practicing judo.
Competitive combat sports, lying at the competitive/fighting opposite in the conceived framework for a new socio-ecological narrative, seem to contribute to general well-being promotion and constitute the starting point of the in-progress framework’s rationale analysis.
6. Practical Applications
In the specifics of education in competitive sports for well-being with experienced black belt adult amateur judokas (10 years of experience at least), a randori practice can be used to favor acute psycho-physical pain management due to the induced activation of the inhibitory and self-control abilities. Despite some limitations, the sustainability of the present pilot study lies in a research–action approach [58], which is addressed to combine academia and practice to elucidate the importance of the coaches’ and teachers’ educational processes and make them conscious of psycho-social values of methods and didactics with respect to environmental context and specificity.
Author Contributions
Conceptualization, P.L.I.; methodology, A.T. and P.L.I.; formal analysis, A.B., D.F. and G.S.; investigation, A.T., D.F., G.S. and M.R.; resources, M.R. and R.S.; data curation, A.B., D.F., G.S. and R.S.; writing—original draft preparation, A.T., D.F., M.R., P.L.I. and R.S.; writing—review and editing, A.T., D.F., M.R., P.L.I., R.S. and S.B.; visualization, R.S. and S.B.; supervision, P.L.I. and S.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and the procedures used complied with the Good Clinical Practice (GCP) principles approved by the ethics committee of the Ospedale San Raffaele (reference number: 88/int/2016).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the first author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Bronfenbrenner, U. Toward an Experimental Ecology of Human Development. Am. Psychol. 1977, 32, 513–531. [Google Scholar] [CrossRef]
- Faigenbaum, A.D.; Rebullido, T.R.; Chulvi-Medrano, I. Youth Physical Activity Is All About the “F-Words”. Strength Cond. J. 2020, 42, 2–6. [Google Scholar] [CrossRef]
- Schölmerich, V.L.; Kawachi, I. Translating the Socio-Ecological Perspective Into Multilevel Interventions: Gaps Between Theory and Practice. Health Educ. Behav. 2016, 43, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Batelaan, P.; Gundare, I. Intercultural education, co-operative learning and the changing society. Intercult. Educ. 2000, 11, 31–34. [Google Scholar] [CrossRef]
- Chen, M.A.; Cheesman, D.J. Mental toughness of mixed martial arts athletes at different levels of competition. Percept. Mot. Skills 2013, 116, 905–917. [Google Scholar] [CrossRef]
- Torres-Luque, G.; Hernández-García, R.; Escobar-Molina, R.; Garatachea, N.; Nikolaidis, P.T. Physical and Physiological Characteristics of Judo Athletes: An Update. Sports 2016, 4, 20. [Google Scholar] [CrossRef]
- Morales, J.; Franchini, E.; Garcia-Massó, X.; Solana-Tramunt, M.; Buscà, B.; González, L.-M. The Work Endurance Recovery Method for Quantifying Training Loads in Judo. Int. J. Sports Physiol. Perform. 2016, 11, 913–919. [Google Scholar] [CrossRef]
- Carmo, K.E.O.; Pérez, D.I.V.; Valido, C.N.; dos Santos, J.L.; Miarka, B.; Mendes-Netto, R.S.; Leite, M.M.R.; Antoniêtto, N.R.; Aedo-Muñoz, E.A.; Brito, C.J. Caffeine improves biochemical and specific performance after judo training: A double-blind crossover study in a real judo training situation. Nutr. Metab. 2021, 18, 15. [Google Scholar] [CrossRef]
- Johnstone, A.; Marí-Beffa, P. The Effects of Martial Arts Training on Attentional Networks in Typical Adults. Front. Psychol. 2018, 9, 80. [Google Scholar] [CrossRef]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain. Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef]
- Formenti, D.; Cavaggioni, L.; Duca, M.; Trecroci, A.; Rapelli, M.; Alberti, G.; Komar, J.; Iodice, P. Acute Effect of Exercise on Cognitive Performance in Middle-Aged Adults: Aerobic Versus Balance. J. Phys. Act. Health 2020, 17, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; McGowan, A.L.; Chandler, M.C.; Gwizdala, K.L.; Parks, A.C.; Fenn, K.; Kamijo, K. A primer on investigating the after effects of acute bouts of physical activity on cognition. Psychol. Sport Exerc. 2019, 40, 1–22. [Google Scholar] [CrossRef]
- Pesce, C. Shifting the focus from quantitative to qualitative exercise characteristics in exercise and cognition research. J. Sport Exerc. Psychol. 2012, 34, 766–786. [Google Scholar] [CrossRef] [PubMed]
- Pesce, C.; Crova, C.; Marchetti, R.; Struzzolino, I.; Masci, I.; Vannozzi, G.; Forte, R. Searching for cognitively optimal challenge point in physical activity for children with typical and atypical motor development. Ment. Health Phys. Act. 2013, 6, 172–180. [Google Scholar] [CrossRef]
- Kao, S.C.; Drollette, E.S.; Ritondale, J.P.; Khan, N.; Hillman, C.H. The acute effects of high-intensity interval training and moderate-intensity continuous exercise on declarative memory and inhibitory control. Psychol. Sport Exerc. 2018, 38, 90–99. [Google Scholar] [CrossRef]
- Moreau, D.; Chou, E. The Acute Effect of High-Intensity Exercise on Executive Function: A Meta-Analysis. Perspect. Psychol. Sci. 2019, 14, 734–764. [Google Scholar] [CrossRef]
- Mann, D.T.; Williams, A.M.; Ward, P.; Janelle, C.M. Perceptual-cognitive expertise in sport: A meta-analysis. J. Sport Exerc. Psychol. 2007, 29, 457–478. [Google Scholar] [CrossRef] [PubMed]
- Vickers, J.N. Mind over muscle: The role of gaze control, spatial cognition, and the quiet eye in motor expertise. Cogn. Process. 2011, 12, 219–222. [Google Scholar] [CrossRef] [PubMed]
- Logan, G.D.; Cowan, W.B.; Davis, K.A. On the ability to inhibit simple and choice reaction time responses: A model and a method. J. Exp. Psychol. Hum. Percept. Perform. 1984, 10, 276–291. [Google Scholar] [CrossRef]
- Posner, M.I. Chronometric Explorations of Mind; Lawrence Erlbaum: Oxford, UK, 1978. [Google Scholar]
- Cojocariu, A.; Abalasei, B. Does the reaction time to visual stimuli contribute to performance in judo? Arch. Budo 2014, 10, 73–78. [Google Scholar]
- Katz, N. The impact of pain management on quality of life. J. Pain Symptom. Manag. 2002, 24 (Suppl. S1), S38–S47. [Google Scholar] [CrossRef] [PubMed]
- Geneen, L.J.; Moore, R.A.; Clarke, C.; Martin, D.; Colvin, L.A.; Smith, B.H. Physical activity and exercise for chronic pain in adults: An overview of Cochrane Reviews. Cochrane Database Syst. Rev. 2017, 152, 440–446. [Google Scholar] [CrossRef]
- Ray, C.A.; Carter, J.R. Central modulation of exercise-induced muscle pain in humans. J. Physiol. 2007, 585, 287–294. [Google Scholar] [CrossRef]
- Chayadi, E.; McConnell, B.L. Gaining insights on the influence of attention, anxiety, and anticipation on pain perception. J. Pain Res. 2019, 12, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Van Ryckeghem, D.M.; Van Damme, S.; Eccleston, C.; Crombez, G. The efficacy of attentional distraction and sensory monitoring in chronic pain patients: A meta-analysis. Clin. Psychol. Rev. 2018, 59, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Ströhle, A.; Feller, C.; Strasburger, C.J.; Heinz, A.; Dimeo, F. Anxiety modulation by the heart? Aerobic exercise and atrial natriuretic peptide. Psychoneuroendocrinology 2006, 31, 1127–1130. [Google Scholar] [CrossRef]
- Rossi, C.; Roklicer, R.; Tubic, T.; Bianco, A.; Gentile, A.; Manojlovic, M.; Maksimovic, N.; Trivic, T.; Drid, P. The Role of Psychological Factors in Judo: A Systematic Review. Int. J. Environ. Res. Public Health 2022, 19, 2093. [Google Scholar] [CrossRef] [PubMed]
- Urhausen, A.; Gabriel, H.; Kindermann, W. Blood Hormones as Markers of Training Stress and Overtraining. Sports Med. 1995, 20, 251–276. [Google Scholar] [CrossRef]
- Battezzati, A.; Benedini, S.; Fattorini, A.; Piceni Sereni, L.; Luzi, L. Effect of hypoglycemia on amino acid and protein metabolism in healthy humans. Diabetes 2000, 49, 1543–1551. [Google Scholar] [CrossRef]
- Schulkin, J.; McEwen, B.S.; Gold, P.W. Allostasis, amygdala, and anticipatory angst. Neurosci. Biobehav. Rev. 1994, 18, 385–396. [Google Scholar] [CrossRef]
- Nonogaki, K.; Iguchi, A. Role of central neural mechanisms in the regulation of hepatic glucose metabolism. Life Sci. 1997, 60, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Li, Q.; Liu, J.; Jia, S. Irisin, an exercise-induced myokine as a metabolic regulator: An updated narrative review. Diabetes Metab. Res. Rev. 2016, 32, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Benedini, S.; Dozio, E.; Invernizzi, P.L.; Vianello, E.; Banfi, G.; Terruzzi, I.; Luzi, L.; Corsi Romanelli, M.M. Irisin: A Potential Link between Physical Exercise and Metabolism—An Observational Study in Differently Trained Subjects, from Elite Athletes to Sedentary People. J. Diabetes Res. 2017, 2017, e1039161. [Google Scholar] [CrossRef] [PubMed]
- Joo, C.H. The effects of short term detraining and retraining on physical fitness in elite soccer players. PLoS ONE 2018, 13, e0196212. [Google Scholar] [CrossRef] [PubMed]
- Tomik, R.; Olex-Zarychta, D.; Mynarski, W. Social values of sport participation and their significance for youth attitudes towards physical education and sport. Stud. Phys. Cult. Tour. 2012, 19, 99–141. [Google Scholar]
- Formenti, D.; Trecroci, A.; Duca, M.; Cavaggioni, L.; D’Angelo, F.; Passi, A.; Longo, S.; Alberti, G. Differences in inhibitory control and motor fitness in children practicing open and closed skill sports. Sci. Rep. 2021, 11, 4033. [Google Scholar] [CrossRef]
- Formenti, D.; Trecroci, A.; Duca, M.; Vanoni, M.; Ciovati, M.; Rossi, A.; Alberti, G. Volleyball-Specific Skills and Cognitive Functions Can Discriminate Players of Different Competitive Levels. J. Strength. Cond. Res. 2022, 36, 813–819. [Google Scholar] [CrossRef]
- Eriksen, B.A.; Eriksen, C.W. Effects of noise letters upon the identification of a target letter in a nonsearch task. Percept. Psychophys. 1974, 16, 143–149. [Google Scholar] [CrossRef]
- Chesterton, L.S.; Sim, J.; Wright, C.C.; Foster, N.E. Interrater reliability of algometry in measuring pressure pain thresholds in healthy humans, using multiple raters. Clin. J. Pain 2007, 23, 760–766. [Google Scholar] [CrossRef]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998. [Google Scholar]
- Ilardi, C.R.; Gamboz, N.; Iavarone, A.; Chieffi, S.; Brandimonte, M.A. Psychometric properties of the STAI-Y scales and normative data in an Italian elderly population. Aging Clin. Exp. Res. 2021, 33, 2759–2766. [Google Scholar] [CrossRef] [PubMed]
- Budde, H.; Voelcker-Rehage, C.; Pietrabyk-Kendziorra, S.; Ribeiro, P.; Tidow, G. Acute coordinative exercise improves attentional performance in adolescents. Neurosci. Lett. 2008, 441, 219–223. [Google Scholar] [CrossRef]
- Schmidt, M.; Jäger, K.; Egger, F.; Roebers, C.M.; Conzelmann, A. Cognitively Engaging Chronic Physical Activity, But Not Aerobic Exercise, Affects Executive Functions in Primary School Children: A Group-Randomized Controlled Trial. J. Sport Exerc. Psychol. 2015, 37, 575–591. [Google Scholar] [CrossRef] [PubMed]
- Cowley, J.C.; Dingwell, J.B.; Gates, D.H. Effects of local and widespread muscle fatigue on movement timing. Exp. Brain. Res. 2014, 232, 3939–3948. [Google Scholar] [CrossRef] [PubMed]
- Pavelka, R.; Třebický, V.; Třebická Fialová, J.; Zdobinský, A.; Coufalová, K.; Havlíček, J.; Tufano, J.J. Acute fatigue affects reaction times and reaction consistency in Mixed Martial Arts fighters. PLoS ONE 2020, 15, e0227675. [Google Scholar] [CrossRef]
- Lima, L.V.; Abner, T.S.S.; Sluka, K.A. Does exercise increase or decrease pain? Central mechanisms underlying these two phenomena. J. Physiol. 2017, 595, 4141–4150. [Google Scholar] [CrossRef] [PubMed]
- Sluka, K.A.; Frey-Law, L.; Hoeger Bement, M. Exercise-induced pain and analgesia? Underlying mechanisms and clinical translation. Pain 2018, 159 (Suppl. S1), S91–S97. [Google Scholar] [CrossRef]
- Lesnak, J.B.; Sluka, K.A. Mechanism of exercise-induced analgesia: What we can learn from physically active animals. Pain Rep. 2020, 5, e850. [Google Scholar] [CrossRef]
- Kami, K.; Tajima, F.; Senba, E. Brain Mechanisms of Exercise-Induced Hypoalgesia: To Find a Way Out from “Fear-Avoidance Belief”. Int. J. Mol. Sci. 2022, 23, 2886. [Google Scholar] [CrossRef]
- Khani, S.; Tayek, J.A. Cortisol increases gluconeogenesis in humans: Its role in the metabolic syndrome. Clin. Sci. 2001, 101, 739–747. [Google Scholar] [CrossRef]
- Fatouros, I.G. Is irisin the new player in exercise-induced adaptations or not? A 2017 update. Clin. Chem. Lab. Med. 2018, 56, 525–548. [Google Scholar] [CrossRef] [PubMed]
- Fox, J.; Rioux, B.V.; Goulet, E.D.B.; Johanssen, N.M.; Swift, D.L.; Bouchard, D.R.; Loewen, H.; Sénéchal, M. Effect of an acute exercise bout on immediate post-exercise irisin concentration in adults: A meta-analysis. Scand. J. Med. Sci. Sports 2018, 28, 16–28. [Google Scholar] [CrossRef] [PubMed]
- Sierra, A.P.R.; Oliveira, R.A.; Silva, E.D.; Lima, G.H.O.; Benetti, M.P.; Kiss, M.A.P.; Sierra, C.A.; Ghorayeb, N.; Seto, J.T.; Pesquero, J.B.; et al. Association Between Hematological Parameters and Iron Metabolism Response After Marathon Race and ACTN3 Genotype. Front. Physiol. 2019, 10, 697. [Google Scholar] [CrossRef] [PubMed]
- Beunders, R.; Bongers, C.C.W.G.; Pickkers, P. The effects of physical exercise on the assessment of kidney function. J. Appl. Physiol. 2020, 128, 1459–1460. [Google Scholar] [CrossRef]
- Manning, M.L.; Lucking, R. The What, Why, and How of Cooperative Learning. Clear. House 1991, 64, 152–156. [Google Scholar] [CrossRef]
- Walter, M. Social Research Methods, 2nd ed.; Oxford University Press: South Melbourne, Australia, 2009. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).