1. Introduction
In the modern era, sensors may enhance the world through the development of the healthcare system. A new mechanism in universal healthcare is known as wireless body area networks, which allow sensors to be injected or worn on the body to gather and transmit actual patient health data, including hypertension, pulse rate, and respiration [
1]. The value of human life can be further enhanced by using wearable or flexible electronics for a variety of purposes, such as motion tracking, regeneration, communication modules, illness diagnostics, etc. Among these, several wearable sensors are carried on the person or connected directly to the skin to collect sensory data and show promising results in identifying diverse body behaviors. Typically, smooth data interchange for human motion recognition combined with wearable sensors will support healthcare monitoring applications [
2,
3,
4]. Diseases that interfere with the human heart’s ability to function are referred to as heart diseases. It covers flaws in the heart’s anatomy, irregular heartbeat or rhythm, and diseases of the bloodstream arteries connected to the heart. Cardiologists frequently use an electrocardiogram (ECG) sensor to promptly and noninvasively evaluate for indicators of probable heart illness and irregular cardiac rhythm. The World Health Organization (WHO) [
5] estimates that 18 million individuals globally die from heart disease every year. To prevent unexpected mortality from a heart attack or cardiac arrest, early identification and treatment of heart disease are crucial.
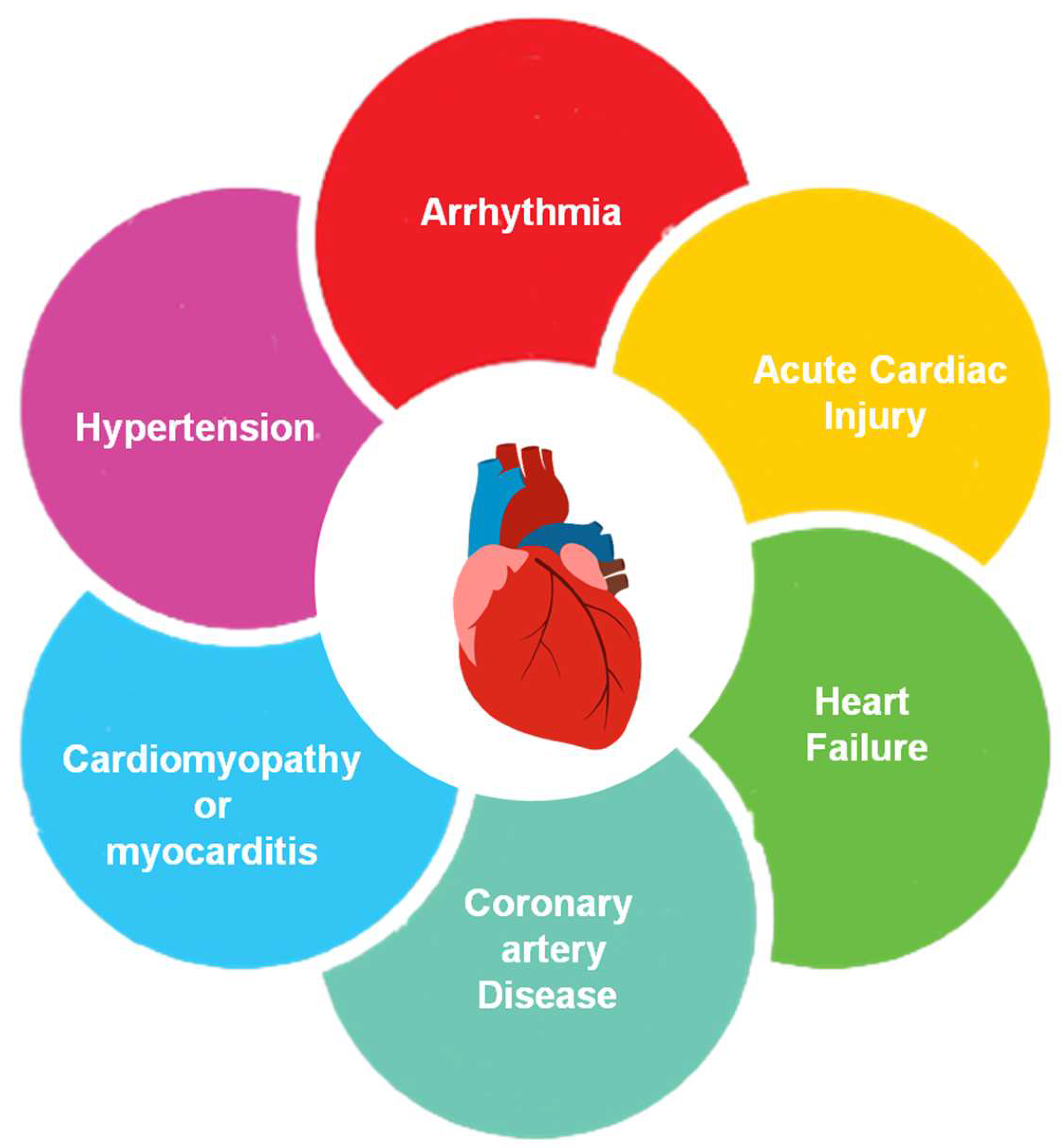
Figure 1 depicts the causes of heart disease: cardiovascular arrests, coronary artery disease (CAD), vascular disease, circulatory diseases, etc. To prevent tragic deaths and preserve the average lifespan, a condition must be diagnosed [
6]. The Internet of Things (IoT), computer networking, and 5G are examples of automated networks that are used in healthcare. Applying emerging technology to behavioral systems and protective policies can help in the advanced identification of potential health issues and enable the timing of pertinent actions, such as tracking treatments and creating new assessments. It is made up of a dynamic setting with many different aspects, including decision-making, administration of the healthcare system, illness diagnosis and prevention, analysis, and rating. Recently, Internet of Medical Things (IoMT) technology has been used in healthcare systems to gather sensor data for the evaluation and prognosis of cardiac disease. Cardiac illness weakens the body, because it impairs blood flow in the body and leads to artery infections, especially in adults and the elderly. An IoT approach uses the end-to-end detection of cardiac conditions using a single-channel ECG. A digitized stethoscope may be used to monitor patients’ heart sounds instantaneously and identify any problems. By improving these advanced techniques in the healthcare system, we proposed an IoT platform that offers more accurate evaluations of cardiac illness using the Modified Self-Adaptive Bayesian algorithm (MSABA).
Our heart disease prediction project’s objective is to determine whether a patient should receive a heart disease diagnosis or not, which is a binary result, so: positive result = 1, the patient will receive a heart disease diagnosis. If the test yields a negative result of 0, the patient will not have heart disease. The “smart heart disease prediction system” developed collects patient data from the IoT or smart gadgets. These items, which are placed on a patient’s body and include activity sensors, medical sensors, and environmental sensors, are also referred to as hardware components.
The main contribution of this study includes an IoT platform that uses a Modified Self-Adaptive Bayesian algorithm (MSABA) to provide more precise assessments of heart disease. An ECG monitors the heart’s electrical signals to detect various cardiac problems. This is the most effective method for constantly monitoring and forecasting patient ECG signals and accomplished with a predictive performance that was sufficient.
The remainder of this article is addressed as follows. In
Section 2, the related works and problem statement are provided. The proposed methodology is offered in
Section 3.
Section 4 contains the results and discussion.
Section 5 contains the conclusion.
2. Related Works
The work discussed in [
7] proposed a hybrid sparrow clustered (HSC) system. Its usefulness and efficiency are shown using the movie lens dataset. A sparrow is a tiny, intelligent, and memory-rich bird. Sparrows frequently alternate between creating and foraging food. In their immediate vicinity, sparrows observe one other’s behavior. The scalability of algorithms using real-world datasets is one of the main problems. Under the top N-recommendation systems, a significant amount of changing data is produced by user interactions in the form of ratings and reviews; as a result, scalability is a major challenge for these datasets. This work provides the viability analysis and the development of data mining and signal processing approaches for heart disease predictions. By using a special improvement in distance- and density-based clustering, the suggested methodology adopts the optimum clustering strategy. K-means clustering is employed as the density-based clustering in this instance, while DBSCAN, which uses the density-based spatial clustering of applications with noise, is used as the distance-based clustering. The new weighted feature extraction is implemented in the regular clinical repository, with the prediction initially concentrating on the data taken from there. After the informative characteristics have been retrieved, they are treated to a hybrid clustering approach, where the results of both clusters are taken into account when determining the final result. Here, the improved DBCAN and K-means clustering are combined to create the hybrid clustering. K-means clustering (KMC) optimizes the centroid, while DBSCAN optimizes the value of using a new metaheuristic technique called Modified Updating-based Chicken Swarm optimization [
8].
For the accurate and quick delivery of results, the suggested paradigm integrates Edge–Fog–Cloud computing. Data from several patients are collected by the hardware components. To obtain important features, cardiac feature extraction from signals is performed. The feature extraction of additional attributes is also gathered. Utilizing an Optimized Cascaded Convolution Neural Network, all of these features are collected and submitted to the diagnostic system. Galactic Swarm Optimization is used in this instance to optimize the CCNN hyperparameters [
9]. The authors of [
10] suggested the system gathers data from IoT devices, and patient history-related electronic clinical data that are saved in the cloud are subjected to predictive analytics. The Bi-LSTM (bidirectional long short-term memory)-based smart healthcare system for tracking and accurately predicting heart disease risk exhibits an accuracy of 98.86%, precision of 98.9%, sensitivity of 98.8%, specificity of 98.89%, and an F-measure of 98.86%, which are significantly better than the current smart heart disease prediction systems.
This study used four machine learning models to identify cardiac illness, including random forest (RF), decision tree (DT), AdaBoost (AB), and K-nearest neighbor (KNN). The strength of the pertinent parameters that influence the prediction of heart disease was examined using a generalized algorithm. The Cleveland, Hungary, Switzerland, and Long Beach (CHSLB) datasets, which were all obtained from Kaggle, were used to assess the models. The accuracy of the RF, DT, AB, and KNN model predictions on the CHSLB dataset was 99.03%, 96.10%, 100%, and 100%, respectively. Two models only—RF and KNN—show good accuracy in the case of a single (Cleveland) dataset at 93.437% and 97.83%, respectively. Finally, the study created a computer-aided smart system for disease prediction using Streamlit, a cloud hosting platform based on the internet [
11]. In order to address this issue, we introduced BioLearner, a machine learning-based intelligent heart disease prediction system for the identification of critical biomedical markers. By identifying the most crucial biological indicators, this study hopes to increase the precision of heart disease prediction. The goal is to create a set of markers that most strongly influences the onset of heart disease. The likelihood of developing heart disease depends on a variety of factors. These variables are believed to include age, smoking, heart rate, past history of chest pain (of various kinds), past fasting blood sugar, and other crucial variables. Analyzing the dataset and contrasting its many components, the accuracy of our forecast for the onset of heart disease in the future is evaluated using a variety of machine learning models, including K-nearest neighbors, neural networks, and support vector machine (SVM). With a 95% accuracy rate, BioLearner outperforms the standard approaches in predicting the likelihood of developing heart disease [
12]. The goal-oriented requirements extraction technique is presented in this work. It is an elicitation method that leverages particular healthcare business objectives to determine the needs of the upcoming e-health system [
13]. The mixed kernel-based extreme learning machine (BMDA-MKELM) methodology, the biogeography optimization algorithm, and the Mexican hat wavelet are used in this paper to enhance the optimization of the Dragonfly algorithm for the prediction of heart disease. Here, information is compiled from two sources, including electronic medical records and sensor nodes. In order to collect patient data, an android-based design is used, and a trustworthy cloud-based storage system is used. Cloud computing services are used to collect data for subsequent analysis on the prediction of heart disease. Finally, cardiovascular illnesses can be categorized using the BMDA-MKELM-based prediction system. Furthermore, the proposed prediction system is contrasted with another approach in terms of metrics such as accuracy, precision, specificity, and sensitivity. The experimental findings show that, when compared to other approaches, the proposed strategy performs better in heart disease prediction [
14]. The authors of [
15] proposed the use of data mining classification techniques to predict the likelihood of coronary heart disease, including Naive Bayes (NB), support vector machine (SVM), k-nearest neighbor (k-NN), decision tree (DT), neural network (NN), logistic regression (LR), random forest (RF), and gradient boosting. Researchers are working tirelessly in the modern world to improve the smart healthcare system. An automated system that can forecast the likelihood of developing heart disease might be considered a major accomplishment. The dataset from the UCI machine learning repository is used to assess this work on heart disease prediction to accurately forecast cardiac disease using a feature selection and classification approach. This study has therefore suggested a unique Multi-Layer Perceptron for Enhanced Brownian Motion-based on Dragonfly Algorithm (MLP-EBMDA) and an optimized unsupervised technique for feature selection. Preprocessing is done after obtaining the dataset for heart disease. Through the use of the improved unsupervised technique, features are chosen. The unique hybrid MLP-EBMDA technique is used to classify heart disease based on the chosen features [
16]. In the proposed study, we developed a data fusion strategy employing data from the BSNs and fog computing. A variety of sensors are used to collect data on daily activities, which are then combined to create high-quality data. In order to forecast heart-related disorders early on, the data thus gathered is subsequently used as the input to an ensemble classifier. The data collector is set up, and the calculation is carried out using a decentralized system using a fog computing environment. After integrating the nodes’ results using the fog computing database, a final output is generated [
17].
On this article, a sine cosine weighted K-nearest neighbor (SCA WKNN) method based on machine learning is suggested for the prediction of heart disease. This algorithm learns from the data being kept in a blockchain. Since the data saved on the blockchain cannot be altered, it serves as a secure setting for storing patient information, as well as a genuine source for learning data. The effectiveness of the suggested SCA WKNN is evaluated in terms of accuracy, precision, recall, F-score, and root mean square error in comparison to other methods. According to our investigation, SCA WKNN outperforms W K-NN and K-NN in terms of maximum accuracy by 4.59% and 15.61%, respectively. In terms of latency and throughput, peer-to-peer storage is contrasted with blockchain-based storage. Decentralized storage powered by blockchains outperforms peer-to-peer storage in maximum throughput by 25.03% [
18]. The purpose of the proposed study is to use machine learning techniques to discover critical indicators of heart disease prediction [
19]. Heart disease detection has been the focus of numerous research studies; however the accuracy of the results is subpar. Therefore, an IoMT architecture utilizing Recurrent CONVoluted neural networks (Rec-CONVnet) is proposed for the diagnosis of heart disease in order to increase prediction precision. Gradient-based learning is a key component of Rec-CONVnet’s learning strategy, although it can easily get caught in local minima [
20]. To categorize cardiac illness, the fuzzy proportional integral and derivative (Fuzzy PID) controller was utilized in the study together with a support vector machine. The goal of particle swarm optimization is to eliminate noise from the electrocardiogram signal. For the purpose of diagnosing and predicting diseases, fuzzy PID controllers were used. The outputs from a fuzzy PID controller are the most accurate and consistent [
21]. This paper’s goal is to boost the prediction values and accuracy. The research can use a variety of heart disease databases. Deep learning (DL) algorithms are crucial for heart disease prediction. To lower the danger of human fatality, prediction can be made early on. An Ensemble Deep Dynamic Algorithm (EDDA) is presented in this research to improve the prediction value accuracy. The EDDA processes heart disease predictions in accordance with a set of procedures. Following are the steps: On the chosen dataset, linear regression and Deep Boltzmann Machine (DBM) are applied. With the compared results, performance is calculated in terms of sensitivity, specificity, and accuracy [
22]. The aim of the paper’s contents is to present a thorough analysis of ML methods for heart disease projection. Using a machine learning approach for the University of California, Irvin (UCI) database, we review representative research efforts that have been conducted on this subject. Only if there is a shared benchmark on the dataset can comparisons be made. As a result, we selected studies that have used the same dataset, the UCI database, and machine learning techniques. The future prospects for heart disease prediction are highlighted, and suggestions for further research are given. A few unresolved concerns and challenges that, in comparison, received very little attention are examined [
23].
The amount of patient-related data that is prepared each month is enormous. The gathered data can be used as a source for identifying potential future weaknesses. Heart disease has been detected using unusual data mining and machine learning techniques. In this study, multiple modified recurrent neural network (mRNN) deep learning algorithms were developed for the prediction of heart disease. With the aid of specially created IoT settings, numerous feature extraction and selection techniques have been employed to obtain crucial attributes and collect data. In a runtime context, the system efficiently and accurately provides heart risk scores [
24]. To monitor easily and effectively predict traffic characteristics, intelligent transportation systems (ITS) must be able to recognize the kind of observed items. Moreover, major depressive disorder (MDD) is demonstrated in [
25]. HAM-D is used in individuals with MDD that were receiving treatment with two distinct treatments.
The authors of [
26] introduced a number of machine learning techniques for heart disease prediction that make use of patient data on key health indicators. In order to construct the prediction models, the study exhibited four classification techniques: multilayer perceptron (MLP), support vector machine (SVM), random forest (RF), and Nave Bayes (NB). Before creating the models, processes for data pretreatment and feature selection were taken. On the basis of accuracy, precision, recall, and F1-score, the models were assessed. The SVM model had the highest accuracy, 91.67%. The major goal of the suggested research is to categorize data and forecast cardiac disease utilizing medical information and imaging data. The suggested model functions in two stages and classifies and predicts medical data. Stage two is not necessary if the results from stage one are capable of accurately predicting cardiac disease. First, information from medical sensors attached to the patient’s body was categorized, and in stage two, an echocardiogram’s image was categorized in order to provide a heart disease prediction [
27]. Renal disease can be detected and managed utilizing medical knowledge from the past in those who are at high risk for cardiovascular disease. However, CKD (Chronic Kidney Disease) is a disorder that has no analytical symptoms; is difficult to locate, identify, and prevent; and it can permanently harm the immune system. For these reasons, AI is being used for treatment forecast and analysis. This study’s main goal is to develop a vision model for CKD and heart disease data using free and open-source Python libraries. Predictions can be made using machine learning algorithms, and the accuracy of those predictions is assessed by contrasting different techniques, such as K-nearest neighbors (KNN) and Fast-Recurrent Neural Networks. Using this technique, a dataset collected from a patient’s medical history is forecasted. Renal disease can be detected and managed utilizing medical knowledge from the past in those who are at high risk for cardiovascular disease. However, CKD (Chronic Kidney Disease) is a disorder that has no analytical symptoms; is difficult to locate, identify, and prevent; and it can permanently harm the immune system. For these reasons, AI is being used for treatment forecast and analysis. This study’s main goal is to develop a vision model for CKD and heart disease data using free and open-source Python libraries. Predictions can be made using machine learning algorithms, and the accuracy of those predictions is assessed by contrasting different techniques, such as K-nearest neighbor (KNN) and Fast-Recurrent Neural Networks. Using this technique, a dataset collected from a patient’s medical history is forecasted [
28]. The development of a wearable biomedical prototype to detect the existence of heart disease is the main goal of this study. The study’s conclusions will be particularly useful in nations with shockingly low doctor-to-patient ratios, because wearable technology may be used to track patients’ vital signs anywhere, not only in hospitals. Using machine learning algorithms, the goal is to foresee the possibility of heart disease. The wearable biomedical prototype’s ECG sensor, which is integrated within the device, provides electrocardiogram (ECG) patterns. Monitoring is done for changes in the ECG patterns. The R-to-R approach is applied to ECG patterns to determine the heart rate [
29]. Artificial intelligence has also drawn greater interest recently, since it enables a deeper understanding of healthcare data and produces precise prediction outcomes. This accurate forecast will address complex questions about cardiac problems and help clinical professionals make wise treatment choices. Consequently, this work aims to improve the feature selection and classification methods to accurately forecast cardiac disease. The Grey-wolf with Firefly algorithm is used in the study to choose features efficiently, and the Differential Evolution Algorithm is used to adjust the hyperparameters of the artificial neural network. In order to better classify the chosen features, it is called the Grey-wolf with Firefly algorithm with Differential Evolution (GF-DE). This suggested classification model optimizes a large number of hyperparameters and trains the neural network to acquire optimal weights [
30]. In [
31], they suggested a unique approach to improve the precision of cardiovascular disease prediction by identifying key features using machine learning techniques. Different feature combinations and many well-known categorization strategies are used to introduce the prediction model. In [
32], they indicated an efficient heart disease prediction model (HDPM) for a CDSS, which consists of XGBoost to predict heart disease, a hybrid Synthetic Minority Oversampling Technique-Edited Nearest Neighbor (SMOTE-ENN) to balance the training data distribution, and Density-Based Spatial Clustering of Applications with Noise (DBSCAN) to detect and eliminate outliers. The provider may suffer consequences from CDS, despite the fact that it has been shown to increase clinical quality results. Due to the potential legal ramifications of CDS, alert fatigue, loss of autonomy, workflow modifications, increased EHR usage, and anxiety are all associated with CDS alerts.
Numerous heart disease-related characteristics are presented in [
33], along with a model built using supervised learning techniques such as the naïve Bayes, decision trees, K-nearest neighbor, and random forest algorithms. It makes use of the current dataset from the UCI heart disease patient repository’s Cleveland database. In [
34], they determined the most accurate machine learning classifiers for these diagnostic uses. For the purpose of predicting cardiac disease, several supervised machine learning algorithms were used, and their effectiveness was evaluated. In [
35], utilizing the UCI repository dataset for training and testing, we measured the accuracy of machine learning methods for predicting cardiac disease. These algorithms included k-nearest neighbor, decision tree, linear regression, and support vector machine (SVM). In [
36], data science was used to predict cardiac problems in the medical industry. There have been numerous studies conducted in relation to that issue, but more work has to be done to increase the forecast accuracy. In [
37], they examined the data that are now accessible regarding cardiovascular diseases to predict heart problems at an early stage and to prevent them from happening. This study sought to find the most accurate machine learning classifiers for these diagnostic uses. For the purpose of predicting cardiac disease, a number of supervised machine learning algorithms were used, and their effectiveness was evaluated. With the exception of MLP and KNN, all applied algorithms were estimated to have feature relevance ratings for each feature. To determine which features were most important for making accurate forecasts about heart disease, all features were prioritized [
34]. In [
38], they combined ensemble learning techniques to try and construct an IOT-based algorithm for predicting cardiac disease. The implementation that gathers data from patients and stores it in the cloud environment incorporates sensors for the prediction of heart disease.
In [
39], they proposed a smart healthcare system for feature fusion and ensemble deep learning for the prediction of heart disease. In [
40], they provided a comprehensive analysis of the current methods for monitoring heart function and predicting cardiovascular disease. These systems transmit detected heart data to a doctor via the Internet of Things, Bluetooth, a GSM module, and a cloud-based server. Using machine learning methods, this work offered the preliminary architecture of a cloud-based cardiac disease prediction system [
41]. Using weighted associative rule mining, the study aims to forecast heart disease based on the scores of relevant aspects [
42]. This work provides the viability analysis and the development of data mining and signal processing approaches for heart disease predictions. By using a special improvement in distance- and density-based clustering, the suggested methodology adopts the optimum clustering strategy. K-means clustering is employed as the density-based clustering in this instance, while DBSCAN, which uses density-based spatial clustering of applications with noise, is used as the distance-based clustering [
8]. The UCI Repository dataset and healthcare sensors are both used in this study [
36] to establish an effective framework for predicting cardiac disease in the general population. In [
43], they evaluated the research conducted by several researchers on the reliability of heart disease prediction using various methods.
As a result, the focus of this work is to provide a novel method for predicting the development of heart disease by taking into account specific procedures, including feature extraction, record keeping, attribute minimization, and classification. Both the higher-order and statistical features are initially extracted in feature extraction. Then, attribute and record minimization are carried out; PCA, a component analysis technique, plays a crucial role in overcoming the dimensionality curse. Finally, a prediction process employing the neural network (NN) model is carried out, consuming the dimensionally reduced features [
44]. For the prediction of cardiovascular illness, numerous intelligent healthcare frameworks have been developed recently using various machine learning and swarm optimization techniques. However, because to a lack of data-recognized approaches and appropriate prediction methodology, the majority of the current strategies for cardiovascular disease prediction failed to reach a higher accuracy. In this paper, we offer an intelligent healthcare framework based on the Swarm-Artificial Neural Network (Swarm-ANN) technique for predicting cardiovascular heart disease, motivated by the current issues [
45]. Using the heart disease dataset accessible in the UCI Machine Repository, this study aims to provide an accurate diagnosis of heart disease. An optimization technique can be useful for improving the sensitivity and accuracy of a heart disease diagnosis. Optimization is the process of identifying the best possible solution among all feasible solutions to a particular problem [
46]. In [
47], they demonstrated the usefulness of local Arabica coffee, focusing on the Influence of the attention level of local Arabica coffee on specific human brainwaves. In [
48], the mental model in human–computer interactions was the main topic of the study. This review study uses a variety of tactics, one of which is to highlight the current methods, findings, and trends in human–computer interactions.
Problem Statement
The work discussed in [
49] suggested that significant Health Tracking Device adoption is prone to failure if the patient is unwilling to take an active role in their treatment. Another factor preventing its broad usage is the expense. The complication of wired sensors for heart disease is higher [
50]. The work presented in [
51] indicated that inadequate detection results from the smart sensor’s design requirement to employ preset embedded functionalities. An outside microprocessor must be used to control sensor calibration. Hence, we proposed the Modified Self-Adaptive Bayesian algorithm (MSABA) to provide more precise assessments of heart disease [
52].
The patient’s surroundings act as a significant source of germs. Gram-negative bacilli and Gram-positive cocci that are multidrug-resistant germs present in the environment and are significant causes of healthcare-associated illnesses (HAI). The primary objective of this study is to improve emergency medical treatment from the initial response to hospital admissions, especially in developing nations, by leveraging IoT to autonomously and automatically deliver emergency healthcare providers and emergency care in real time.
4. Performance Evaluation
In this section, we evaluate the efficacy of our suggested approach using the performance indicator indicated before. Our proposed technique is performed and also matched with other standard techniques (MANAGE-HF [
50], GA [
51], ML [
52], BSNs [
17], MSABA). Heart failure patients experience a range of physical and psychological symptoms, including dyspnea, fatigue, edema, trouble sleeping, sadness, and chest discomfort. Patients’ everyday social and physical activities are limited by these symptoms, which lowers their quality of life. High hospitalization and mortality rates are associated with low QOL. The limitation of genetic algorithms (GA) includes identifying the fitness function as a challenge. Definition of the problem’s representation. It is a case of premature convergence. Defining the values to use for things such as population size, mutation rate, crossover rate, selection method, strength, and so on can be difficult. Problem-specific knowledge cannot be easily included. Training datasets for machine learning systems need to be large, diverse, and accurate. In other cases, they may even have to hold off until fresh information is generated. Machine learning requires a significant length of time for the algorithms to learn and mature so that they can accomplish their goal with a high degree of accuracy and relevance. Likewise, it requires a lot of energy and money to run. Since there is no data protection system in place, there are concerns regarding the privacy and security of patient monitoring data. Standards of BAN implementation are nonexistent. These are the limitations of the existing methods. Therefore, we can use the proposed method of the modified self-adaptive Bayesian algorithm to overcome these issues.
Figure 4 depicts the comparison of accuracy with proposed and existing techniques regarding the given financial collections. In this graph, the x-axis denotes financial datasets, and the y-axis denotes accuracy. In
Figure 4, the existing methods of management of heart failure have 56%, the genetic algorithm has 64%, machine learning have 70%, body sensor networks have 82%, and the proposed method of modified, self-adaptive Bayesian algorithm have 90%, so the proposed technique has a high degree of accuracy when compared to other methods.
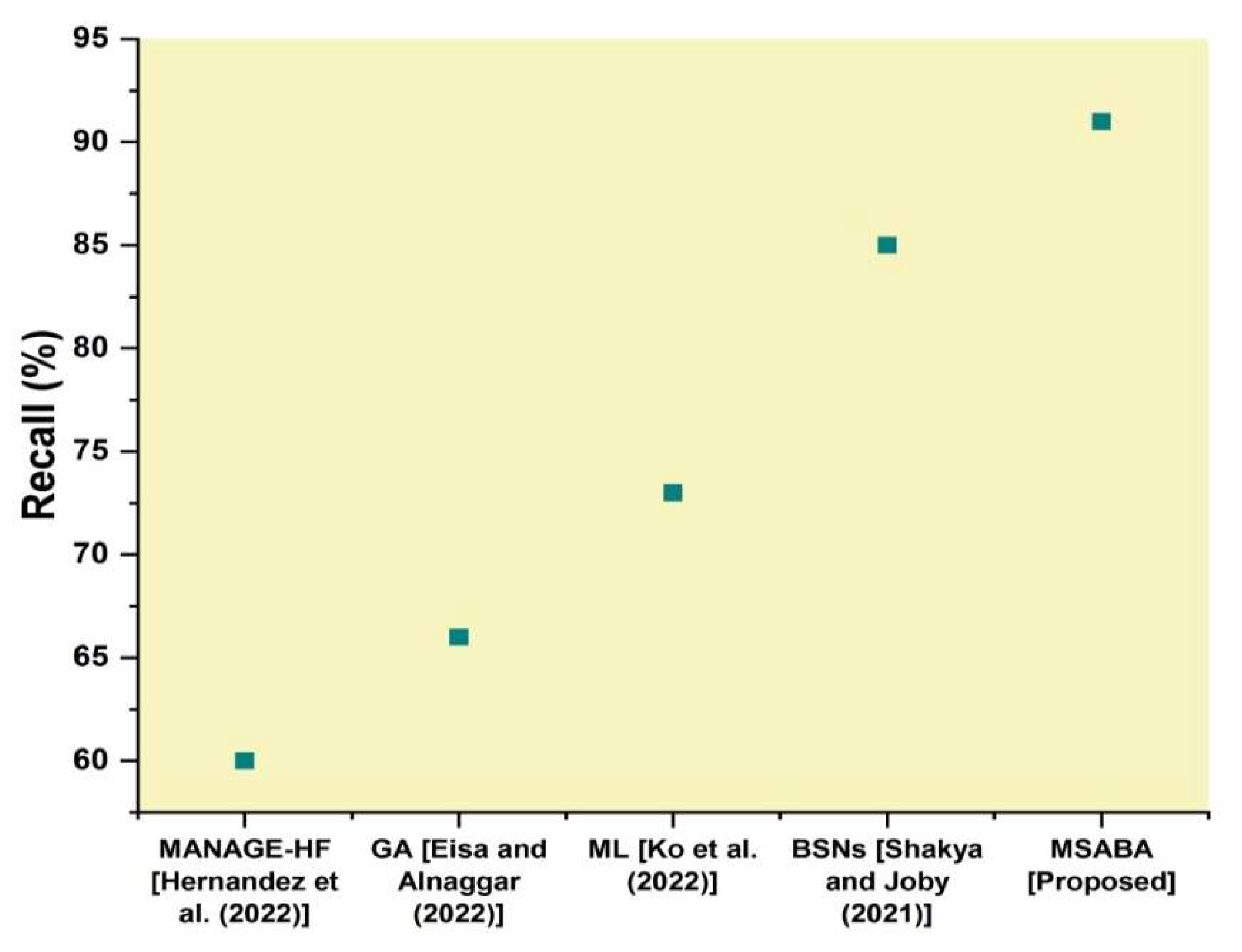
Figure 5 represented the comparison of recall with the proposed and existing techniques regarding the given financial collections. In
Figure 5, the existing methods of management of heart failure have 60%, the genetic algorithm have 66%, machine learning have 73%, body sensor networks have 85%, and the proposed method of the modified self-adaptive Bayesian algorithm have 91%, so the proposed technique has a high degree of recall when compared to other methods.
The comparison of precision with proposed and existing techniques is represented in
Figure 6. In
Figure 6, the existing methods of management of heart failure have 58%, the genetic algorithm have 65%, machine learning have 72%, body sensor networks have 84%, and the proposed method of modified self-adaptive Bayesian algorithm have 92%, so the existing methods have a low degree of precision when compared to the proposed method.
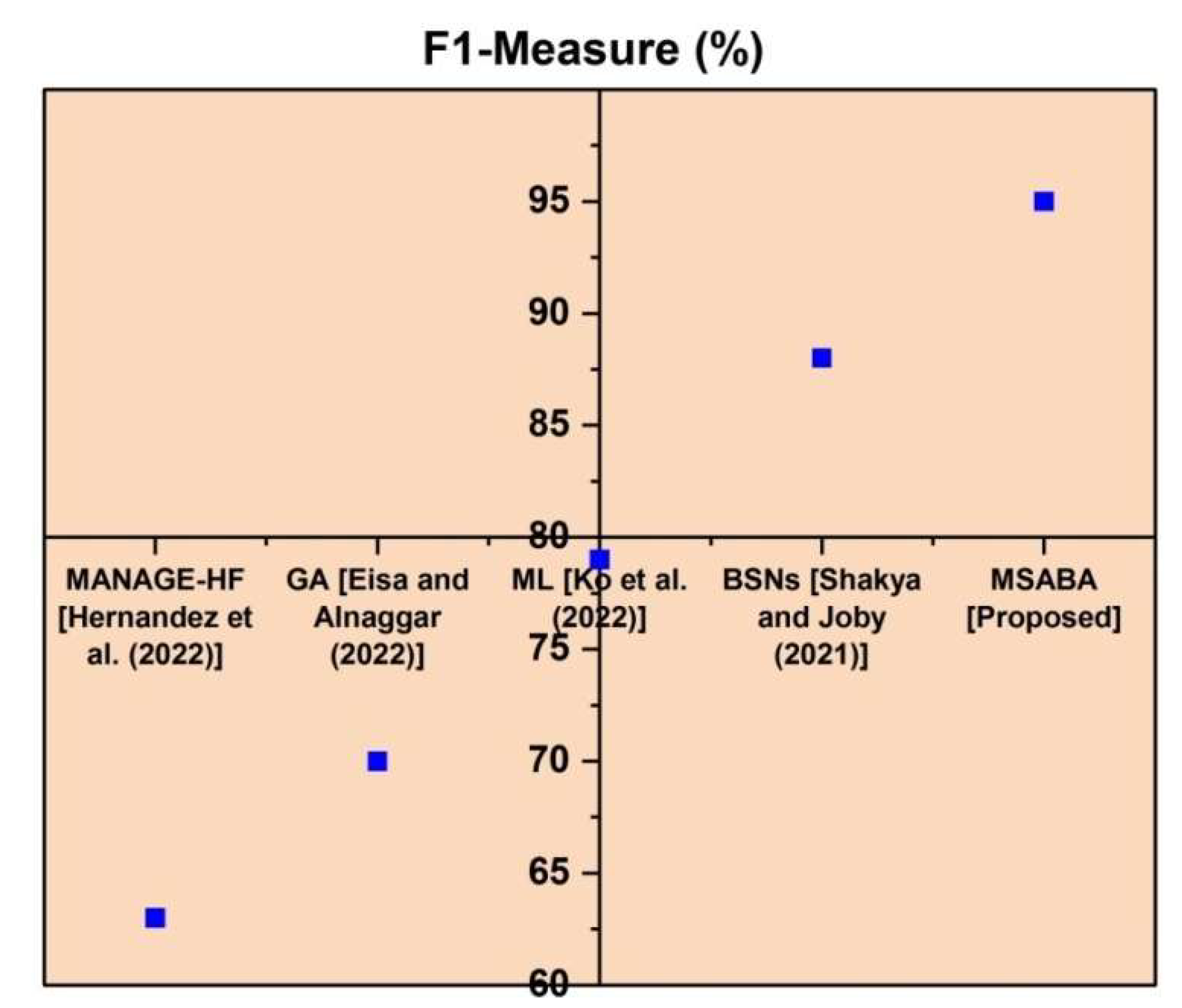
Figure 7 depicts the comparison of the F1 measure with proposed and existing techniques regarding the given financial collections. In this graph, the x-axis denotes financial datasets, and the y-axis denotes accuracy. In
Figure 7 the existing methods of management of heart failure have 63%, the genetic algorithm have 70%, machine learning have 79%, body sensor networks have 88% and the proposed method of modified self-adaptive Bayesian algorithm have 95%, so the proposed method has a high degree of F1-measures when compared to other existing methods.
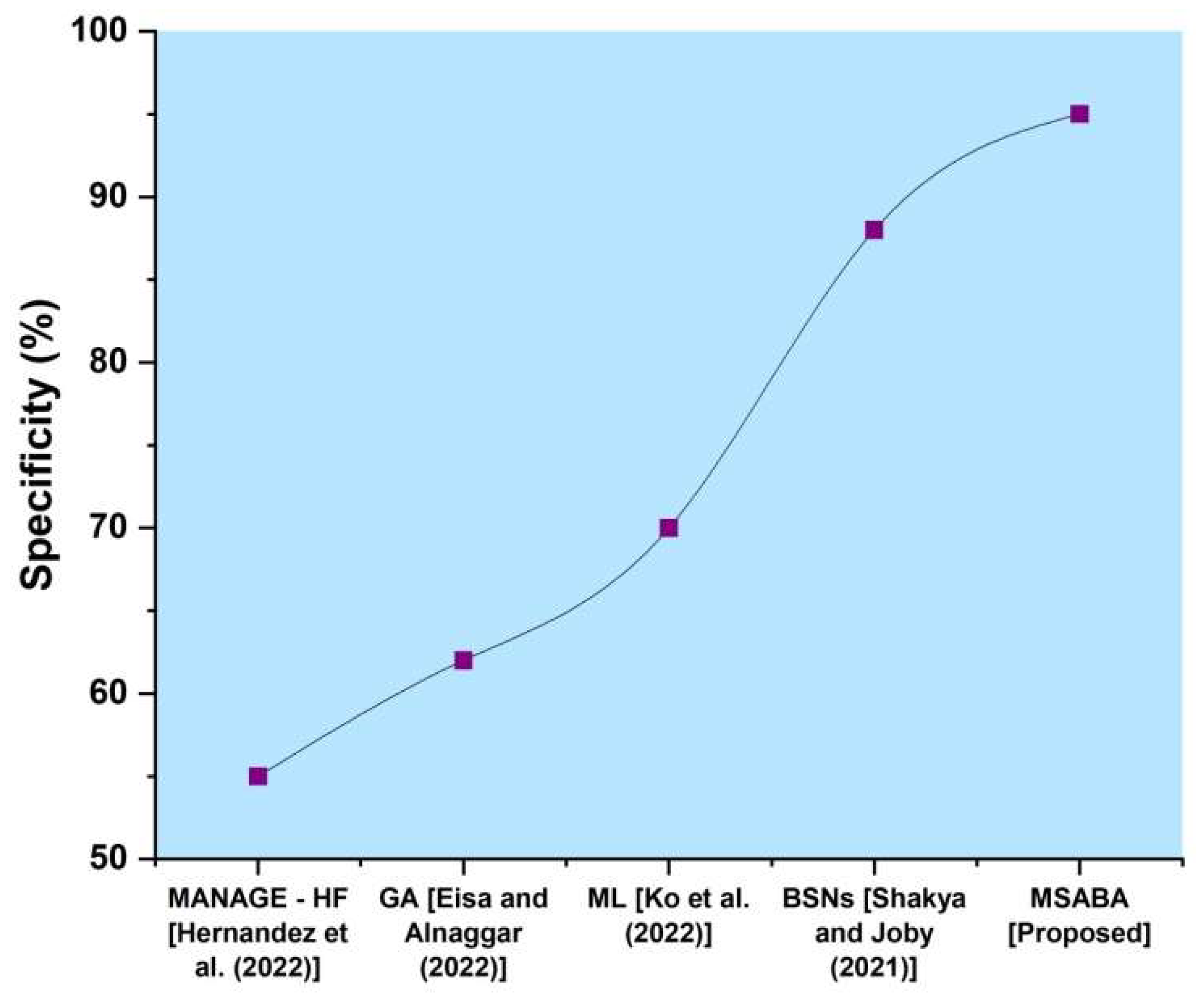
Figure 8 depicts the comparison of specificity with proposed and existing techniques regarding the given financial collections. In this graph, the x-axis denotes financial datasets, and the y-axis denotes specificity. In
Figure 8, the existing methods of management of heart failure have 55%, the genetic algorithm have 62%, machine learning have 70%, body sensor networks have 88%, and the proposed method of modified self-adaptive Bayesian algorithm have 95%, so the proposed method has a high degree of specificity when compared to other existing methods.
Figure 9 represents the comparison of root mean square error with proposed and existing techniques regarding the given financial collections. In this graph, the x-axis denotes financial datasets, and the y-axis denotes the root mean square error. In
Figure 9, the existing methods of management of heart failure have 96%, genetic algorithm have 87%, machine learning have 71%, body sensor networks have 63%, and the proposed method of modified self-adaptive Bayesian algorithm have 56%, so the proposed method has a low degree of root mean square error when compared to other existing methods.
Future research will use data mining techniques to create a more precise dataset for heart disease diagnostics, improving the performance of feature fusion. Additionally, unique feature reduction techniques will be developed to manage massive feature counts and volumes of medical records. To attain effective results, a more advanced strategy will be investigated for eliminating unimportant characteristics and controlling missing data and noise.
The reliability of the registries and the linking determine the validity. As a result, we checked the predicted incidence rates of coronary heart disease (CHD), acute myocardial infarction, unstable angina pectoris, and heart failure against the cardiovascular registry Maastricht cohort study’s disease registry. The cohort comprises 21,148 people who were randomly selected from Maastricht and its neighboring districts between 1987 and 1997 and were born between 1927 and 1977.
In this study, a modified self-adaptive Bayesian algorithm (MSABA) is suggested to offer more accurate evaluations of heart disease. In this section, the patient’s health status, heart rate, and course of therapy are analyzed. The important metrics are accuracy, recall, precision, and F1 measure. The suggested algorithm’s efficacy is evaluated using these metrics. The outcomes were compared to those obtained using conventional methods such as Multiple Cardiac Sensors for the management of heart failure (MANAGE-HF) [
50], genetic algorithms (GA) [
51], machine learning approaches (ML) [
52], and body sensor networks (BSNs) [
17]. As per data collection, the datasets are gathered out of public health datasets. The Jupiter notebook simulation tool system is used in this work. Jupiter notebook is a convenient tool for python programming projects and is used as a simulation tool. The Jupytor notebook includes code, as well as rich text features, including equations, links, and many other types of data. These documents are the ideal place to combine an analysis description and its results, since they include rich text elements with code, and they also enable real-time data analysis. A web-based interactive tool for creating images, maps, charts, visualizations, and narrative prose is called Jupyter notebook.
In the management of the heart failure (MANAGE-HF) existing method, the heart logic index datasets are used. Cleveland heart disease datasets are used in the genetic algorithms existing method. Heart rate data is implemented using machine learning (ML) approaches. Data are collected using wearable sensors fitted onto the human body in the body sensor networks existing method.
4.1. Accuracy
The proximity among measurements and their “real” values are referred to as accuracy. A measurement’s accuracy decreases with distance from the expected or true value. The degree to which evaluation results of a proportion are much closer to the actual value of that number indicates more accuracy of the device. The accuracy of the suggested approach and existing approaches are displayed in
Figure 4. The proposed approach is demonstrated to be more accurate when contrasted with the existing method. The accuracy of the proposed algorithm and the existing systems’ estimations of heart disease are highlighted. While MANAGE-HF has a 56% accuracy rate, GA has a 64% accuracy rate, ML has a 70% accuracy rate, and BSNs have an 82% accuracy rate, the suggested method (MSABA) has a 90% accuracy rate. It demonstrates that the suggested strategy is more successful than the existing approaches.
4.2. Recall
The capacity of a model to locate all pertinent instances in a data source. Recall is calculated mathematically as the product of the number of true positives divided by a sum of the true positives and false negatives. The percentage of pertinent instances that were found constitutes a recall. The true positive rate sometimes referred to as sensitivity is also referred to as recall.
Figure 5 illustrates how the current and suggested methods are remembered. When compared to the commonly employed methods, the proposed procedure had the highest recall for identifying heart disease. The recall of the recommended algorithm and the estimates of heart disease made by the existing systems are established. In comparison to MANAG-memory HF rates of 60% from GA, 66% from ML, and 85% from BSNs, the suggested technique MSABA has a 91% recall rate. It illustrates that the proposed methodology is more effective than the traditionally used methods.
4.3. Precision
A classification model’s capacity to isolate only the pertinent data points, precision is calculated by dividing the total number of true positives by the total number of true positives + false positives. Precision, or the positive predictive value, is the percentage of pertinent ideas among the recovered occurrences. It may indicate that the standard for quality is precise. Precision is the probability of pertaining recovery on average. The precision of suggested and existing methodologies is compared in
Figure 6. In comparison to the existing approaches, the suggested study has substantially higher precision in the healthcare monitoring system of heart disease. The precision of the existing systems is as follows: MANAG- HF has a 58% precision level, GA has a 65% precision level, ML has a 72% level, and BSNs have an 84% level. The suggested system MSABA has a 92% precision level. As a result, the suggested system has the highest level of functionality
4.4. F1-Measure
The accuracy of a model on a dataset is gauged by the F-score, also known as the F1-score. It’s employed to assess binary categorization schemes that label examples as “positive” or “negative”. By calculating the harmonic means, the clarity and memory of a system are merged into a single statistic known as the F1 measure.
Figure 7 demonstrates the F1 measure for both existing and newly proposed methodologies. Improved system performance is indicated by a higher F1 metric for the prediction of heart disease. According to
Figure 6, the suggested system achieves 95% of the F1 measure, compared to MANAGE-HF 63%, GA 70%, ML 79%, and BSNs 88%. It indicates that the suggested system is more valuable.
The accuracy of a test at identifying people who do not have a condition or feature is known as specificity. It is the percentage of people who are accurately diagnosed as actually not at risk or without a condition (such as a trait, disease, categorization, or label) by a diagnostic tool.
Figure 8 demonstrates the specificity of both existing and new proposed methodologies. According to
Figure 8, the suggested system of MSABA achieves 95% specificity, compared to MANAGE-HF 55%, GA 62%, ML 70%, and BSNs 88%. It indicates that the suggested system is more valuable than the existing approaches.
The square root of the mean of the square of all the errors is known as the root mean squared error (RMSE). The RMSE is frequently employed and is regarded as a superior all-purpose error metric for numerical forecasts. The precision of the suggested and existing methodologies is compared in
Figure 9. In comparison to existing approaches, the suggested study has a substantially lower root mean square error in the healthcare monitoring system of heart disease. The root mean square error of the existing systems is as follows: MANAG- HF has a 96% root mean square level, GA has an 87% root mean square level, ML has a 71% level, and BSNs have a 63% level. The suggested system MSABA has a 56% precision level. As a result, the suggested system has the lowest level of functionality.