Abstract
Medium and highly sulfidic tailings are high-volume wastes that can lead to severe environmental damage if not properly managed. Due to the high content of sulfide minerals, these tailings can undergo weathering if put in contact with oxygen and water, generating acid mine drainage (AMD). The moderate-to-high sulfide content is also an important technical limitation for their implementation in the production of construction materials. This paper reviews the use of sulfidic tailings as raw material in construction products, with a focus on cement, concrete, and ceramics. When used as aggregates in concrete, this can lead to concrete degradation by internal sulfate attack. In building ceramics, their implementation without prior treatment is undesirable due to the formation of black reduction core, efflorescence, SOx emissions, and their associated costs. Moreover, their intrinsic low reactivity represents a barrier for their use as supplementary cementitious materials (SCMs) and as precursors for alkali-activated materials (AAMs). Nevertheless, the production of calcium sulfoaluminate (CSA) cement can be a suitable path for the valorization of medium and highly sulfidic tailings. Otherwise difficult to upcycle, sulfidic tailings could be used in the clinker raw meal as an alternative raw material. Not only the SO3 and SiO2-rich bulk material is incorporated into reactive clinker phases, but also some minor constituents in the tailings may contribute to the production of such low-CO2 cements at lower temperatures. Nevertheless, this valorization route remains poorly explored and demands further research.
1. Introduction
The extraction of metals from their natural occurrences involves several steps, where each step is accompanied by the production of considerable amounts of waste [1]. At the global level, annual waste production is estimated to vary from 5–7 billion tonnes [2] to up to 10 billion tonnes [3]. Estimates suggest that the EU has a stock of 4700 Mt mining wastes, of which 1200 Mt is mine tailings [4]. Mine tailings are the wastes generated during the several mineral processing steps—e.g., crushing; grinding; gravity, magnetic or electrostatic separation; and flotation—used to physically separate the ore concentrate from the mined material [5]. The properties and quantities of those tailings can vary significantly based on the properties of the mined materials and the extraction methods utilized [6]. Among them, the tailings rich in sulfidic minerals are of particular concern because of their severe environmental impacts including acid mine drainage (AMD) accompanied by environmental leaching of pollutants with potentially severe impacts on groundwater quality, local biodiversity, etc., [7,8,9].
AMD is generated by aqueous oxidation of sulfidic minerals leading to the formation of sulfuric acid. In abiotic conditions, oxidation occurs either in a single step by direct oxidation with oxygen or in multiple steps by indirect oxidation through Fe3+; indirect oxidation is usually found to be the dominant route [5]. Therefore, while Fe-rich sulfides like pyrite and pyrrhotite exhibit very high reactivities, Fe-poor sulfides like sphalerite, covellite, etc., have lower reactivities [10]. Due to the high importance of Fe3+ in the oxidation of sulfidic minerals, the rate of oxidation is known to decrease as the solution pH is increased to >3, since Fe3+ concentrations in the solutions decrease with an increase in solution pH [11]. In addition to the Fe3+ concentration and mineral composition, the rate of AMD generation of sulfidic tailings is dependent on a range of other variables like particle size and surface area; crystallinity/amorphicity; crystal defects due to the presence of trace elements; the co-presence of multiple sulfidic minerals; temperature; bed porosity; availability of water; microbial activities; metal to sulfur ratios; and precipitation of secondary minerals [10].
Present tailings management techniques try to prevent the oxidation of sulfidic mining wastes mainly by managing their accessibility to oxygen. In this context, tailings dams and impoundment have been among the most popular tailing management methods due to their ease of application and low economic costs [12]. The popularity of the tailing dams can be judged from the presence of more than 3500 tailing dams globally, some of which can even exceed 100 m in height with millions of m3 water-saturated tailings [5]. Due to their huge size, tailing dams are susceptible to catastrophic failures, threatening human lives, the environment, and economic activities in the short term [13], in addition to prolonged environmental contamination. There have been more than 120 tailing dam failures since 1960, leading to the release of billions of tonnes of tailings [14]. Among the most recent major failures, in 2020, nearly 2.53 million m3 of Mo tailings from the Tieli mine in China traveled 200 km down the Yijimi River contaminating drinking water of at least 70,000 residents [15]. In one of the biggest tailing dam failures of 2019, nearly 12 million m3 tailings were released from the Córrego de Feijão mine in Brazil, causing the death of more than 250 people, and severe contamination of the Rio Paraopeba River [16]. In 2017, the failure of a dam in the Mishor Rotem phosphate mine in Israel led to the release of ~100,000 m3 acidic water, causing severe destruction to the local ecology and environment [17]. Clearly, the risks of failure of tailing dams and impoundments is not too infrequent and poses a severe risk to the local population and biodiversity.
To mitigate some of the problems related to tailing dam construction and their failures, surface paste disposal (SPD) techniques involving dewatering of the tailings to prepare stacks of dried tailings has also been pursued [18,19]. While they offer several advantages including water recovery, they introduce additional engineering challenges and costs, such as dust generation, waste liquefaction, control of rheological parameters for paste transportation [2]. Moreover, long-term environmental concerns related to leaching and AMD are still applicable [20]. Another emerging alternative is to utilize/dispose thickened tailings in cemented paste backfills (CPB). This involves the preparation of composites of binders and mine tailings to fill back the void created by mining [21,22]. These composites are usually composed of <10–20% binders and have compressive strengths in the range of 150–300 KPa [23,24]. In addition to the safe disposal of the tailings, CPB offers added advantages of allowing underground storage leading to lower failure risk compared to tailing dams; reduced area requirement due to higher density; reclamation of water, reagents, and energy; lower environmental leaching; and stabilization of the adjacent mines by backfilling, enabling safer mining, etc., [22,25]. While CPB is considered relatively safer compared to tailing dams, instability of the composites, especially due to the reaction of the binders with sulfur, acids, or minor elements like arsenic, and eventual oxidation of the sulfidic tailings in the failed CPB are major concerns [26]. Another alternative is to utilize low-permeability dry covers with materials like geotextiles, low sulfide waste rocks, oxide wastes, organic wastes, clay, soils, clay-rich subsoils, etc., [5,12,27]. The alkaline covers especially offer the dual advantage of low oxygen permeability and its role as a neutralizing agent.
In the above-mentioned popular tailing-management approaches, the sulfidic tailings remain in their relatively unstable states, ready to oxidize when oxygen is available. Moreover, while Fe-poor sulfides are not prime candidates for AMD, they are also known to cause environmental leaching [28]. Valorization strategies are therefore welcome to minimize the economic and environmental cost of tailings disposal. Recycling of such tailings is aligned with the United Nations’ Sustainable Development Goals (SDGs), particularly to SDG11 “Sustainable Cities and Communities” and SDG12 “Responsible Consumption and Production” by minimizing the output of waste [29], and offers an exciting, and rather unexplored, opportunity to transition from waste-management to waste-valorization. In this paper, we will focus on the potential ways to valorize these wastes. We will first assess in detail various compositions from the literature. This will allow us to draw perspective on valorization potentials depending on their physical, mineralogical and chemical properties. We will consider the following valorization routes: (a) raw material in clinker production, (b) supplementary cementitious material (SCM), (c) precursor in alkali activated materials (AAM), (d) aggregates in concrete, and (e) building ceramics. CPBs will not be considered here.
2. Sulfidic Tailings
The sulfidic wastes are mainly produced during the extraction of certain metals that occur mainly as sulfidic minerals. The major sulfidic minerals are listed in Table 1.

Table 1.
Major sulfidic ores and respective metals of interest [5,30].
A summary showing the scale of metal production mainly from sulfidic minerals is presented in Table 2. The low average percentage of concentrate with respect to the gross ore, associated with the large yearly production of individual metals, give a picture of the scale of global sulfidic tailings production. Those massive amounts are of great concern in the world as well as in Europe. Some examples of huge volumes of sulfidic mine tailings produced by individual mines are (a) the Neves Corvo Cu-Zn mine (Portugal) produces 2.9 million tonnes of tailings per year [31], (b) the Mamut Cu-Au-Ag mine (Indonesia) produced 250 Mt of overburden and 150 Mt tailings during its operation (1975–1999) [32], (c) the Lavrion Ag-Pb mine (Greece) has produced nearly 800,000 m3 of sulfidic flotation tailings, occupying a 94,000 m2 area [33], (d) the Navodari mine (Romania) has generated nearly 1,000,000 m3 of pyritic cinders [34], (e) the Laver Cu-Au-Ag mine and Stekenjokk Cu-Zn-Cu mines (Sweden) respectively stored 1.2 million tonnes and 4.4 million tonnes of tailings in containments [35], (f) it is estimated that Alberta, Canada needed at least 130 km2 for tailings slurry impoundment [36], (g) the O’Kiep Cu-Pb-Zn ores in South Africa has nearly 5.8 Mt metalliferous tailings [37].

Table 2.
Metals production mainly from sulfidic sources (values reported for 2016), and their composition in the gross ores.
2.1. Physical Properties
Tailing particle size distributions (PSD) are highly variable and difficult to generalize, as they are delineated by specific process requirements [9]. Froth flotation, for instance, has a requirement for particles in the range of approximately 20–150 μm [40], while the gravitational settling method utilizes particles of around 1 mm [9]. Physical properties such as particle size, shape, and density are of critical importance for tailings management and valorization. Not only for applications that demand a higher level of rheological control, i.e., CPB, but also for other uses of sulfidic tailings in construction materials. The tailing fineness affects the packing, water demand, and workability of mortar, concrete, and CPBs [41], and their reactivity if used as a partial replacement for cement.
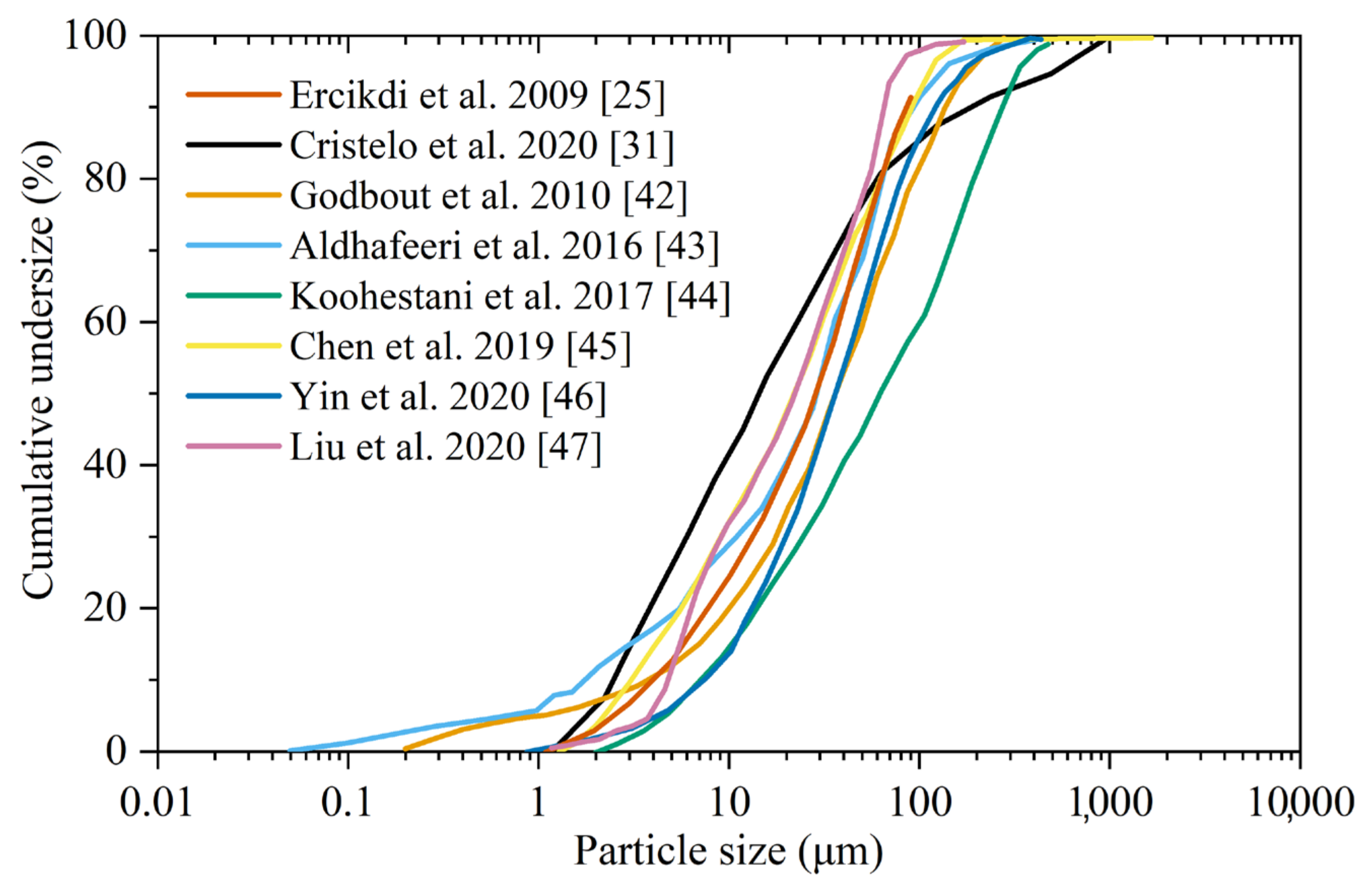
Examples of the PSD of medium and highly sulfidic tailings obtained utilizing laser diffraction granulometry [25,31,42,43,44,45,46,47] were redrawn from the literature and are presented in Figure 1. Table 3 shows the diameter ranges observed for d10, d50, and d90, specific surface area (SSA), and specific gravity (SG).

Figure 1.
Particle size distributions of medium and highly sulfidic tailings obtained from the literature [25,31,42,43,44,45,46,47].

Table 3.
Physical properties of medium and highly sulfidic tailings obtained from the literature. D-values obtained from PDS curves in [25,31,42,43,44,45,46,47]; SG values from [31,42,43,44,47,48,49,50,51,52,53,54,55], and SSA values from [48,51,52,53,55,56].
The comparison of particle size distributions shows some divergences between tailings. Some of them are very fine, with d90 < 65 μm, values that are similar to Portland cement (d90 < 60 μm). Other tailings contain large amounts of coarser particles (d90 between 100 and 277 μm). Nevertheless, overall, sulfidic tailings present a large fraction of fine particles. The values of specific surface area (SSA) are between 0.366 and 1.895 m2/g. The specific gravity of the tailings varies between 2.85, in the low-sulfide tailings reported by [47], and 4.13 g/cm3, in the highly sulfidic tailings used by [53]. The high variability in specific gravity is linked to the variable chemical and mineralogical composition of the tailings. However, the values found correspond to intermediates between typical values of siliceous matrices (SGquartz = 2.65 g/cm3) and pyrite (SGpyrite = 5.00 g/cm3).
The particle size distribution of tailings can be modified by several processes i.e., grinding, desliming, and granulation. The modifications caused by such processes may affect not only physical properties like particle size and shape but also the chemical composition and mineralogy of tailings. One example of such changes is described by [51]. The authors reported that after removal of fine particles and clay minerals of highly sulfidic tailings by desliming, the SiO2 + Al2O3 content and specific surface area (cm2/g) of the tailings were observed to decrease by 28% and 47%, respectively. Additionally, the amount of sulfidic minerals was seen to increase, thereby increasing the specific gravity of the deslimed tailings.
2.2. Chemical Composition
Sulfidic tailings can be classified according to their sulfur content as low (S < 5%), medium (5% < S < 20%), and high sulfur-containing tailings (S > 20%) [57]. In order to discuss general aspects of the chemical composition of sulfidic tailings, examples of the chemical compositions of medium and highly sulfidic metal tailings were collected from 18 publications, most of which address solidification/stabilization and CPB of sulfidic tailings [25,31,42,44,45,46,47,48,50,52,53,54,55,56,58,59,60,61]. Table 4 shows a summary of the compositions collected. The trace elements, including a complete list of the heavy metals reported, are presented in Table 5.

Table 4.
Summary of chemical compositions (elements > 1%) of medium and highly sulfidic tailings obtained from the literature. Since a few studies have not reported the contents of all major oxides, only the higher limit was included in the % range of some oxides.

Table 5.
Trace elements of medium and highly sulfidic tailings obtained from the literature.
The chemical composition of tailings is variable since it depends on many factors: the mineralogy of the ore body, the nature and efficiency of the metal extraction process, and the degree of weathering during storage in the dammed impoundment [9]. Nonetheless, medium and highly sulfidic tailings typically contain high Fe2O3, SiO2, and SO3 contents, and low to moderate Al2O3 and CaO contents. In the collected papers, summed primary oxides (SiO2, Al2O3, and Fe2O3) range from 45 to 89%, which is generally lower in highly sulfidic tailings, due to higher SO3 content. Most of the tailings (16) present CaO content below 6%, except for a few examples, which have calcite as one of the major minerals, as is indicated in the XRD patterns [45,56]. On the other hand, the Al2O3 content tends to be slightly higher than CaO due to the usual presence of Al-bearing minerals. Because metal extraction processes are never 100% efficient, metals and metalloids other than Fe are present in the tailings [9]. Mg, Mn, K, Ti, and Na oxides are generally present in minor amounts, and a number of residual heavy metals can be systematically observed in concentrations below 1.5% (Zn, As, Cu, Pb, Cr).
In many cases, the tailings present low loss-on-ignition (LOI) values, as in [32,56], which indicates the absence of significant organic and residual carbon contents. In other cases, however, LOI values range from 14–29%. It is known that sulfide or iron oxidation, dehydration of gypsum, metallic oxyhydroxides, and clays, volatilization of components, and loss of inorganic carbon affect LOI values [62]. Those reported high values are most probably caused by mass losses due to the decarbonation of carbonate minerals and sulfur volatilization in the tailings.
2.3. Mineral Phase Composition
The chemical composition of tailings is very much linked to their mineralogy. Although amorphous or poorly crystalline ferric hydroxides may precipitate as a result of AMD [5], overall, tailings are rather crystalline and low in organic matter content. Although many publications present only qualitative X-ray diffraction (XRD) analyses of the tailings and unreported amorphous content, a collection of quantitative mineralogical results was obtained from the literature. Table 6 presents a summary of the example mineral phase compositions collected; results are expressed as percentages of crystalline phases.

Table 6.
Summary of mineral phase compositions of medium and highly sulfidic tailings obtained from the literature [12,43,44,49,50,53,54,59,61], expressed as percentages of crystalline phases.
Sulfidic tailings have highly heterogeneous and deposit-specific mineralogy, which may include sulfides, silicates, oxides, hydroxides, phosphates, halides, and carbonates [5]. The major sulfide phase is most often the gangue mineral pyrite (FeS2). However, most of the tailings are polysulfide, including other sulfides such as pyrrhotite (Fe(1−x)S), sphalerite ((Zn, Fe)S), chalcopyrite (CuFeS2), galena (PbS), covellite (CuS), and arsenopyrite (FeAsS). Pyrite content ranges from 15–56% in the data collection, while the contents of other sulfides fall below 4%, and most often below 1%. As a result of the wide variability, significantly different mineralogical compositions are also found, as the 80% pyrrhotite-tailings reported by [61].
Quartz is a typical major mineral in the tailings, followed by other gangue minerals such as chlorites, feldspars, and carbonates. Calcite (CaCO3) and dolomite (CaMg(CO3)2) are common carbonates, but other examples are magnesite (MgCO3), siderite (FeCO3), huntite (Mg3Ca(CO3)4), ankerite (Ca(Fe,Mg,Mn)(CO3)2) and magnesian kutnohorite (Ca(Mg,Mn)(CO3)2). Their total amount is below 16% in all tailings from the reviewed papers.
Clay minerals such as kaolinite and montmorillonite are of special interest in the context of calcined tailings. However, aluminosilicate clay minerals, when present, are reported in minor amounts in the literature. Some examples are 2.1% and 0.7% kaolinite (Al2Si2O5(OH)4), as reported in [51,55]. Non-quantified illite was also reported [49], in addition to 16.4% of a magnesium silicate, talc (Mg3Si4O10(OH)2) [43]. Along with the groups of minerals mentioned in Table 6, amphiboles, some of which are known for their severe effects on human health [63], spinels, and pyroxene have also been mentioned in some of the publications collected.
Sulfidic tailings are known to be unstable even in ambient conditions [10]. Depending on the reaction conditions and exposure durations, several secondary, tertiary, and quaternary minerals are known to co-exist with the primary minerals [64]. Oxidation of sulfides can lead the sulfidic tailings to be rich in sulfates, hydrous sulfates, oxides, hydroxides, etc., [5]. Many of these secondary minerals can still be highly water-soluble, leading to severe environmental consequences [65]. In order to discuss the possibilities of reuse and valorization of sulfidic mine tailings, their chemical composition, mineralogy, and physical properties need to be known.
3. Applications of Sulfidic Tailings in Construction Materials
As the utilization of industrial by-products and wastes as binder components and aggregates constitute a valuable segment of cement and concrete technology [66], the construction sector offers unique opportunities for tailings valorization. Besides, this sector has been intensively driven towards diversification of resources. The huge world annual production of cement and concrete (several billions of tonnes per annum) requires that alternative resources have a long-term secure supply of at least tens of thousands of tonnes per annum to feed into local production facilities processing similar volumes of cement or concrete [67]. At a local level, a significant environmental benefit can beachieved through the exploitation of local opportunities for raw materials. This allows for the development of products and technologies optimized for a specific context or application, rather than the one-type-fits-all approach [68]. The sustainable use of construction materials produced from wastes must be guaranteed throughout their entire life cycle. In order to guarantee their environmental performance during their production, service life, recycling and disposal, trace elements must be permanently immobilized onto those products.
3.1. Clinker Production
Alternative raw materials and alternative fuels are among the decarbonization strategies used by the cement industry to reduce the environmental impact of clinker production [68]. Those strategies consist of utilizing industrial, municipal, and agricultural wastes and by-products to integrate the clinker raw meal or as a source of energy in the cement kiln. By doing so, the cement industry lowers the consumption of natural resources, reduces the CO2 emissions in clinker production, and provides appropriate disposal for wastes that would otherwise be incinerated or landfilled. While wastes consisting mainly of combustible organics are usually regarded as alternative fuels, wastes containing CaO, SiO2, Al2O3, and Fe2O3 are usually used as alternative raw materials [69].
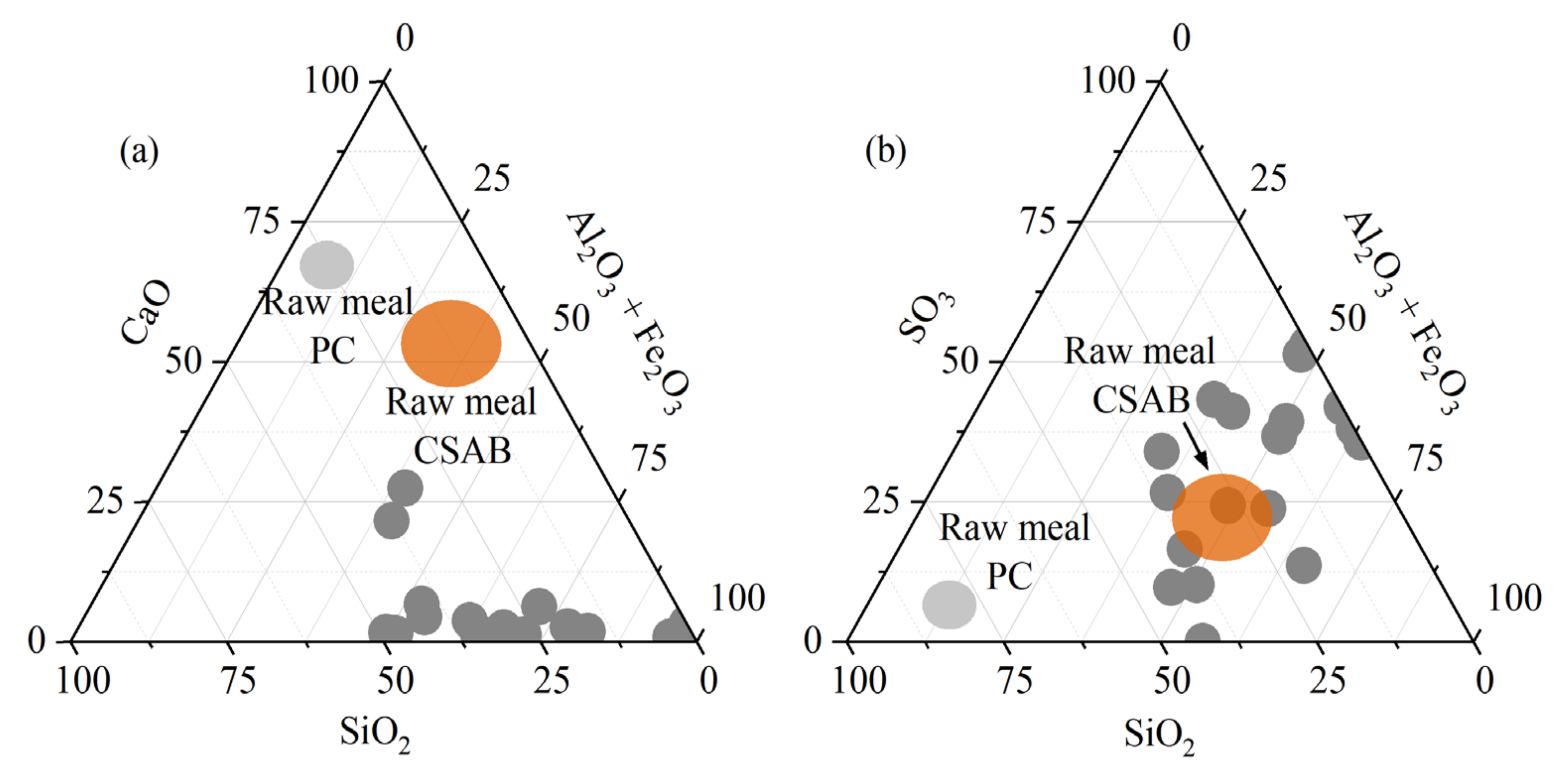
Tailings of different origins have been proposed as alternative sources of CaO, SiO2, Al2O3, and Fe2O3 to produce PC and special clinkers, as reviewed by [70]. The authors suggested that the high variability in tailings composition can challenge their effective implementation as clinker raw materials. Nevertheless, even limited utilization ratios as corrective materials were found to offer environmental and technical benefits to tailings-based clinkers. Figure 2 presents the positions of medium and highly sulfidic tailings in ternary diagrams (using data from Section 2.2). In Figure 2a, chemical compositions arerepresented in terms of CaO, SiO2, and Al2O3 + Fe2O3 to enable comparisons of the selected materials to typical clinker raw meal compositions. Figure 2b includes an important component of the tailings, SO3.

Figure 2.
Chemical composition of the medium and highly sulfidic tailings expressed in terms of (a) CaO, SiO2, and Al2O3 + Fe2O3, and (b) SO3, SiO2, and Al2O3 + Fe2O3. Zones in light gray and orange correspond to typical compositions of clinker raw meals of PC and CSAB, respectively.
It can be seen in Table 4 and Figure 2a that, apart from a few calcite-containing tailings, the CaO content in medium and highly sulfidic tailings is low, which makes them unsuitable to be used as a calcium source in clinker raw meal. The displacement of composition points towards the Al2O3 + Fe2O3 corner is caused mainly by the high iron content in the tailings, rather than high alumina concentrations. Figure 2b shows that the tailings contain considerable amounts of SiO2, Fe2O3, and SO3, besides modest Al2O3, in proportions compatible with typical raw meals of low-CO2 clinkers, such as calcium sulfoaluminate (CSA).
CSA cements belong to a group of ye’elimite-containing cements, which also include Calcium sulfoaluminate-belite (CSAB) and Belite-Ye’elimite-Ferrite (BYF) cements. They have been developed on a commercial basis and used in real applications, primarily in China, since the 1970s [71], and are produced by mixing sources of calcium, silicon/aluminum, and sulfur, usually limestone, clay, and/or bauxite and gypsum, respectively. Ranges of individual phase contents vary according to clinker type, as well as the predominant phase. Commercial CSA cement contains 50–80% of ye’elimite (Ca4(AlO2)6SO3, or C4A3$ in cement notation), followed by 30–10 wt.% of belite (Ca2SiO4, or C2S), and are used mainly in applications that require rapid setting, early strength, or shrinkage compensation [72]. This class of cement can be produced in existing Portland cement plant installations, at temperatures around 1250 °C, using a conventional cement kiln system. Other advantages include lower CO2 release during the production of CSA clinker main phase (C4A3$), when compared to PC clinker main phase (alite, Ca3SiO5, or C3S), easier grindability, larger clinker dilutions with calcium sulfate, and the possibility of using various industrial by-products in the kiln feed [73,74].
In terms of chemical composition, the utilization of tailings as alternative raw materials in the CSA clinker kilns allows for higher utilization rates than in Portland cement raw meals. While the SO3 levels must be kept low in the PC raw meals, moderate SO3 content is required in order to produce C4A3$ in CSA clinkers. Candidates for incorporation of Fe2O3 in such clinkers are ferrite (Ca2AlxFe2−xO5, or C4AF) and iron containing-ye’elimite (Ca4(AlxFe1−xO2)6SO3, or C4A3−XFX$). In addition, tailings can be a secondary silica resource for the production of C2S in such clinkers.
Limited literature exists on the use of medium and highly sulfidic tailings as raw materials for clinker production. The existing studies [75,76,77] focus on their upcycling in CSA clinker raw meals and utilize tailings containing 9.12–48.4 wt.% SO3. In one of the studies [75], CSAB was produced from tailings containing calcite, dolomite, quartz, kaolinite, gypsum, pyrite, galena, and sphalerite, in addition to high concentrations of heavy metals (Pb, Zn, Sr, Ba). Clinkers containing belite, ye’elimite, and ferrite were obtained utilizing 45–50% replacement ratios in the raw meal. The authors showed that partial immobilization of Pb, Zn, and Cu was obtained after clinkerization and improved after hydration of the cements. Another study also showed efficient immobilization of heavy metals [78].
Most of the sulfide minerals have been found to be highly volatile, and their decomposition is usually accompanied by the release of mainly SOx during clinkerization. Thus, although S is partially incorporated into solid phases, a fraction of it escapes to the gas phase mainly as SO2 [79] and may require gas cleaning installations—e.g., SO2 scrubbers—to reduce SOx emissions to the environment. If standard calcium oxide based scrubbing is used, the products—anhydrite or gypsum—can be reincorporated in the clinker raw meal feed. As envisioned by Hanein et al. in their study utilizing sulfur as fuels for CSAB production [80], the recovered sulfur product could find an immediate use in the final cement product. It has been shown that, at optimum conditions, 4–5% of sulfur decomposes during the production of CSA cement using highly pyritic tailings [76]. Volatilization of heavy metals and alkalis during the clinkerization of sulfidic tailings has not yet been addressed in publications.
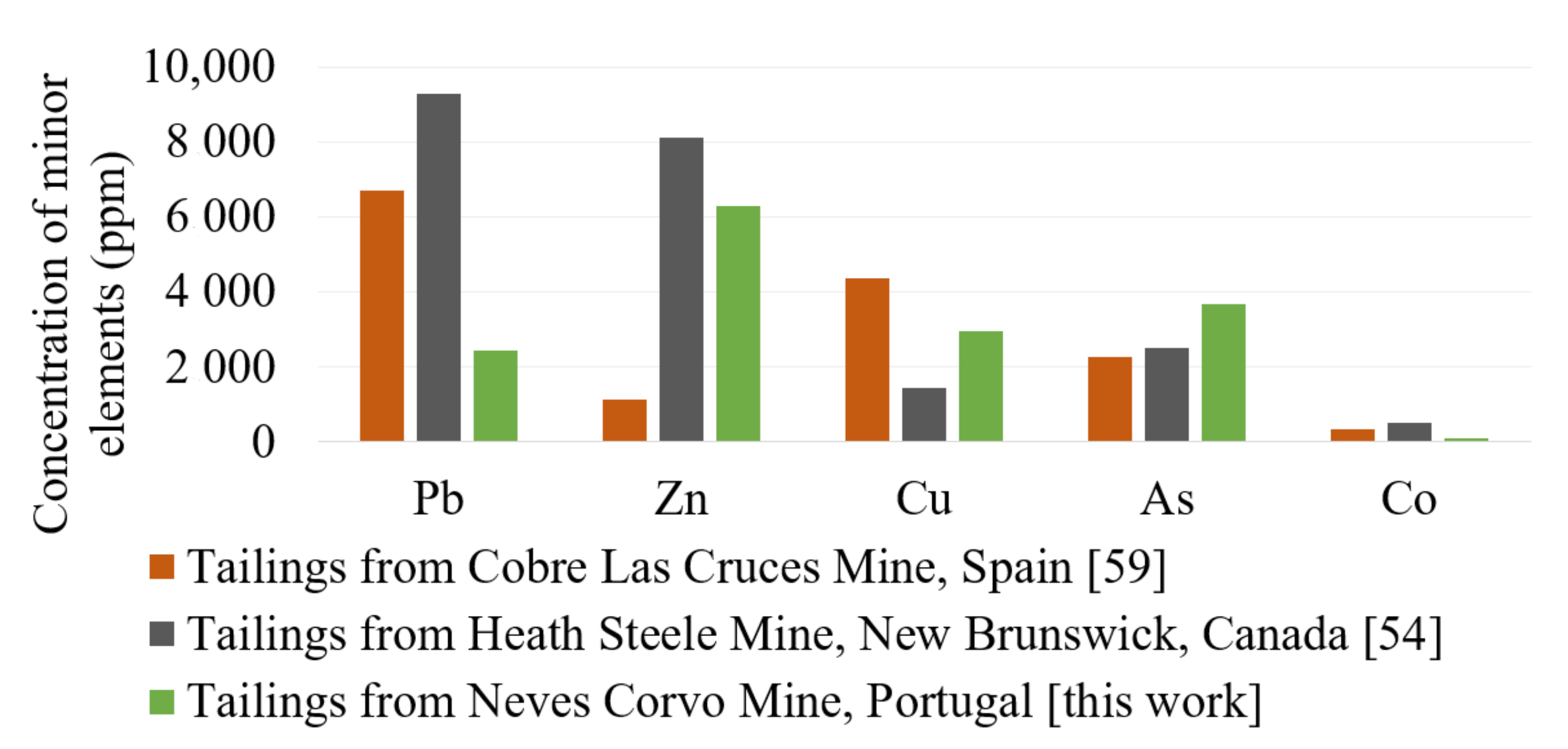
Minor constituents contained in tailings, reported in Table 5, may have a mineralizing/fluxing effect on the clinkering process, lowering processing temperatures and enabling the coexistence of phases that would otherwise be unstable at a given temperature range. Examples of minor elements which can have a mineralizing effect in CSA clinkers are CuO [81,82,83] and ZnO [84]. Concentrations of minor elements in three examples of highly sulfidic tailings are shown in Figure 3. ZnO and CuO are minor oxides present in amounts that can potentially play a role in mineralization. Nevertheless, the effect may not always be beneficial, and the burnability of the mix can be reduced if refractory minerals are contained in the raw materials—e.g., coarse quartz [85].

Figure 3.
Minor elements in sulfidic tailings [54,59].
The effect of minor elements is not limited to the clinkerization process. For ye’elimite-containing cements, in particular, several studies have demonstrated that trace elements can affect cement hydration, performance, morphology of hydrates, and microstructure of pastes and mortars. Interested readers are referred to Supplementary Table S1 (refer to electronic Supplementary Materials), which presents the highlights of publications on the effect of minor elements in ye’elimite-containing cements. In order to promote the upcycling of sulfidic tailings as a raw material in CSAB clinker production, the mineralizing effects of minor elements, as well as their influence on early hydration and environmental performance of CSAB cement must be further investigated.
The matrix of ye’elimite-containing cements has shown to be suitable for immobilization of hazardous elements in insoluble hydration products. Those cements provide a different cementitious environment, i.e., the pH of CSA lies at 10.5–11 whereas PC has a pH of 13, and, according to clinker mineralogy and the amount of extra sulfate added (in the form of gypsum or anhydrite), the hydrate phase assemblage includes ettringite (3CaO·Al2O3·3CaSO4·32H2O), AFm (monosulfate) phases, aluminum hydroxide (Al(OH)3 or AH3) and C-S-H. Fixation of heavy metals may take place not only in ettringite crystal lattice, which can accommodate substitutions of bivalent and trivalent cations and oxyanions [86,87] but also in AFm phases and C-S-H, as reviewed by [88]. The leaching of heavy metals incorporated in CSAB hydrates was investigated by [89]. Zn has been added by the dissolution of 0.5 mol/L of ZnCl2 in mixing water during the preparation of cement pastes. Zinc was not detected in the leachate, and the mechanisms of immobilization proposed were the precipitation of an unknown phase (possibly LDH) at an early age (<1 day), and probable sorption of Zn2+ onto aluminum hydroxide (AH3) at a later age. Excellent retention values of Pb, Cd, Zn, and Cr3+ were obtained utilizing a CSA clinker (53.5% C4A3$ and 21.2% C2S) and 20–30% gypsum addition [90], as well as in a synthetic system containing C4A3$, gypsum, and lime in stoichiometric ratios for ettringite formation [86]. In the latter, high concentrations (10 wt.% of binder) were used to simulate extreme conditions. A waste stream containing even higher concentrations of hazardous elements (>60%) was partially stabilized by chemical and mechanical encapsulation utilizing CSAB [91]. Nevertheless, the immobilization of oxyanions, i.e., (CrO4)2−, (Cr2O7)2− [90,92], SeO2− [91] can be more challenging and dependent on the gypsum addition. In CSAB cementitious systems containing up to 50% cement replacement with low sulfidic tailings, a study also shows an effective reduction in the release of heavy metals and sulfate in leaching tests [93]. Even for challenging elements (As, V) immobilization was achieved already after 7 days of hydration, and improved after longer curing times.
3.2. Supplementary Cementitious Materials (SCMs)
Widely used to partially replace cement, supplementary cementitious materials (SCMs) are finely ground materials that contribute to the properties of Portland cement-based materials through hydraulic or pozzolanic activity or both. Globally, it is estimated that 650 million tonnes of SCMs are used each year [94]. The use of those materials leads to a significant reduction in CO2 emissions per tonne of cementitious materials mainly due to a reduction in the clinker content in cement production. Besides, it also enables the utilization of the by-products and wastes of industrial manufacturing processes as binder components. Materials conventionally used as SCMs are blast furnace slag, fly ash from coal combustion, natural pozzolans, silica fume, calcined clays, and limestone.
Considering the limited supply of high-quality SCMs, significant effort has beenspent to explore the potential of several waste materials over the last decades. Some waste materials, however, present physical and chemical properties that are undesirable or unacceptable for their use in blended cements [94]. Very limited data have been published on the use of mine tailings as SCMs. As far as sulfidic tailings are concerned, the publications are particularly scarce. Reasons for that are relative to several incompatibilities observed on their properties. It was verified, for instance, that sulfidic tailings do not comply with common cement standards in terms of composition, in particular SO3 contents. Based on the data from Section 2.3, mineralogical composition of sulfidic tailings can also limit their direct utilization as SCMs. Most of the tailings contain major minerals that do not actively participate in pozzolanic or hydration reactions, such as quartz and sulfides. Besides, it has to be pointed out that the tailings undergo chemical transformation in the long term due to oxidation of sulfide minerals, which can lead to sulfide-induced degradation in the cementitious matrix [95,96].
In the study performed by [93], mortars were prepared from blends of CSAB clinker (48% ye’elimite, 29% belite), gypsum, and untreated tailings (7.4% SO3). The authors targeted the use of tailings as an additional source of sulfate to contribute to CSAB cement hydration and strength development. The CSAB–gypsum ratio was kept constant and equal to 9:1; w/c = 0.5 (water–cement ratio) and sand content (75 wt.%) were also kept constant. The increasing replacement of CSAB by untreated tailings reduced the strength and workability of the mortars, but all mortars showed strength gain with increasing age (strength was reported up to 90 days). However, when normalizing the strength to the CSAB content in each mortar (MPa/g of CSAB cement), mortars containing 25% and 50% wt. tailings seem to show a slight improvement in strength. The improvements are slight, therefore not enough to compensate for the dilution effect due to partial replacement of CSAB with the sulfidic tailings, resulting in the observed reduction on measured strength values. Reduction on measured strength but not on normalized strength was also observed in 7 day pastes containing 40% replacement with copper tailings (0.7% SO3) activated by calcination and grinding [97]. Along with chemical activity, finely ground materials can provide a simultaneous physical contribution to cement hydration known as the filler effect [98]. The filler effect of fine tailings can be pointed out as a reason for the apparent improvement in performance considering normalized values. At the high replacement ratios investigated by [92], dilution effects due to cement replacement arefully compensated only in the case of reactive SCMs. In the case of largely inert fine materials, contributions to performance due to filler effects are more evident at low (up to 20%) replacement ratios [99]. For that reason, in the case of fine sulfidic tailings-cement mortars, low replacement ratios can lead to improvement in performance even if the tailings have low intrinsic pozzolanic or hydraulic reactivity. This aspect has not yet been the subject of a comprehensive investigation.
Aiming at increasing the reactivity of tailings, several authors have reported the utilization of pre-treatments before their use in combination with Portland cement. Those studies have been performed in tailings other than sulfidic, but could be adapted for the treatment of sulfidic tailings. Highlights are presented individually in Supplementary Table S2. Some studies have successfully demonstrated that calcination of tailings containing clay minerals [97,100,101] and grinding [97,102,103] are effective activation methods for mine tailings. Through calcination, kaolinite decomposes and produces a highly reactive amorphous material, i.e., metakaolinite. Other clay minerals like palygorskite, montmorillonite, and illite can also be artificially transformed into pozzolans after calcination [104]. Nevertheless, a minimum clay mineral content, especially kaolinite, appears to be required to significantly enhance the mechanical properties of mortars produced from calcined tailings. It must be noted that those minerals are not typically present in major amounts in sulfidic tailings (Table 6). Besides, the decarbonation of carbonate minerals such as calcite, dolomite, and siderite takes place when calcination temperatures are beyond the decarbonation threshold of such minerals, as observed by [97]. A recent study has shown that co-calcination with carbonate minerals can lead to an increase in pozzolanic activity of low-grade aluminosilicates—e.g., mica and illite-rich materials [105]. In addition to reacting with the decomposed aluminosilicates, the reactive lime and periclase reacts with some feldspar and silica phases forming poorly crystalline phases that contribute to an increase in pozzolanic activity. Incorporation of Ca released from the decarbonation of calcite into the amorphous fraction was also observed in another study [106]. The calcination of sulfidic tailings rich in low-grade aluminosilicates and carbonate minerals may increase their pozzolanic activity. It must be noted that thermal activation leads to a reduction of the environmental benefits of the use of tailings as SCMs, especially in low-strength concretes. This is due to the relatively low amount of cement that is necessary to obtain the same mechanical performance at low strength levels [107].
The oxidation of sulfide minerals can also take place as a result of thermal treatments, as observed in tailings subjected to roasting [59,108]. Unlike calcination, roasting is carried out at lower temperatures, typically half of those used in calcination, and mostly in sulfidic ores. Particularly for highly pyritic tailings, calcination and roasting can dramatically modify the mineralogy of the tailings materials, as well as their behavior in cementitious systems. Pyrite oxidation during roasting takes place according to different reactions, resulting in a number of intermediate and final products, such as hematite (Fe2O3), magnetite (Fe3O4), iron (ferric or ferrous) sulfates (Fe2(SO4)3, FeSO4), and sulfur dioxide (SO2) [109]. Roasted sulfidic tailings containing soluble sulfates could be utilized as a replacement for anhydrite in cement. However, this valorization route remains unexplored.
3.3. Alkali-Activated Materials (AAMs)
Alkali-activated materials (AAMs) are novel binder materials coined by Davidovits in 1972 [110], where the negative charge of aluminosilicate tetrahedra units (precursors) from typical calcined kaolinite (i.e., metakaolin) was balanced with positive cations from alkali activator such as sodium hydroxide and sodium silicate, forming a three-dimensional aluminosilicate framework [111]. Aluminosilicates are commonly found in many of the industrial side streams such as tailings, slags and ashes, which gives an opportunity to produce such AAMs in more sustainable way.
Prior to the recycling or reuse of mine wastes, mineralogical analysis is rather essential to determine the subsequent application in AAMs as precursors [112]. The combination with industrial side streams or metakaolin to produce co-binders is well studied during the last decades. The available studies referring to sulfidic mine tailings are detailed in Supplementary Table S3. Alongside the fabrication of co-binders, heavy metals (including Pb, Cu, Zn, Mn, Ni, and Co) are prone to be stabilized in AAMs matrix via chemical and physical immobilization [113,114]. However, some oxyanions such as Mo, As, Se, Cr and Sb can be more leachable after alkali-activation due to the high pH environment [115].
Nevertheless, the valorization of sulfidic mine tailings in AAMs as sole precursor has been little investigated. A detailed summary is provided in Supplementary Table S4. The main obstacle can be the originally low aluminosilicate content and following aluminosilicate dissolution in alkali activator. The alkali-activation of untreated mine tailings is difficult, and they usually do not harden if there is no treatment applied or co-binders introduced [116,117]. Particularly, Pacheco-Torgal et al. [118] observed that calcination of tailings (3.10% SO3) at 950 °C for 2 h can enhance the compressive strength of alkali-activated mortar above 35 MPa. These results were attributed to the activation of muscovite in the tailings by thermal treatment, which is commonly observed in several non-sulfidic tailings [119]. Yao et al. [120] conducted mechanical activation of gold mine tailings (0.16% SO3) as the precursor for alkali-activation. Decreased particle size and crystallinity along with increased specific surface area and crystal defects were observed as a result of mechanical activation [121,122]. The mechanically activated mine tailings–cement mortar reached an activity index—the compressive strength ratio with standard cement sample—of 79% as cement additives, being a greener and more effective approach to upscale the use of mine tailings. Nevertheless, the tailings used in the cited studies have sulfur contents lower than 5 wt.%, whilst the utilization of mining wastes containing medium and high sulfur content as sole precursors has not yet been reported.
In summary, mining wastes that are rich in aluminosilicate can be promising candidates for precursor for AAMs. The sulphur and metal(loid)s can be sufficiently immobilized in a geopolymer matrix [123]. Nevertheless, pre-treatments are essential for AAM preparation based on the previous research. For that, two pre-treatment methods are typically applied: thermal treatment and/or mechanical treatment. Both facilitate the formation of amorphous phase(s) through dehydroxylation and structural collapse of the aluminosilicates, thereby increasing their alkaline dissolution. To fully achieve the potential of complex minerals wastes upcycling, techniques such as flotation and sieving could be applied prior to the pre-treatment step. The application of such separation techniques is needed in a prospective view, once they could produce an aluminosilicate-rich fraction of the tailings. To illustrate, the mechanical treatment of quartz and sulfides leads to energy consumption, yet there is no crystalline disorder occurring. It is also expected that new methods can be sought out in the aspects of alternative alkali activators [124], production of wet mix [125], and novel binder formation (Fe-rich geopolymers) [126].
3.4. Concrete Aggregates
Aggregates are granular materials with particle size greater than 75 µm that constitute a major portion (60–80%) of concrete. They have significant influence on workability, concrete strength, dimensional stability, durability, and cost of production [127,128]. In addition to the silicate, carbonate, and oxide minerals, sulfide minerals may also be present in the natural aggregates [129]. However, according to technical recommendations, aggregates are required to have low S content [130], as low as 0.1% S by mass if the presence of pyrhotite is proven [131], to be recommended for concrete production. The limitation is directly related to the degradation phenomenon initiated by the oxidation of sulfide minerals when in contact with oxygen and humidity [132,133,134]. Even at very low sulfide content—e.g., 0.28% by volume [135], aggregates can cause severe cracking in concrete in the long term. The cracks originate from iron sulfide-containing regions and then extend into the cement paste. The degradation mechanism is known as internal sulfate attack and was found to cause several structural failures [132,133,135,136].
The progress of the deleterious reaction is determined by the change in surface area of the reactants and, therefore, on the initial grain size of the sulfides [136]. The finer the sulfide particles, the larger the potential damage induced by sulfidic tailings in concrete. Soluble sulfates generated by sulfide oxidation can react with hydrated PC according to a series of chemical reactions to form gypsum, monosulfoaluminate, and ettringite [136,137]. The type of degradation product formed is dependent on the pH and the availability of sulfate and calcium [136]. Expansion takes place due to the generation of larger volume products which include the mentioned sulfate-containing phases and iron hydroxides [132,133,135,136]. Together with structural damage, aesthetic issues such as staining are also known to develop [138].
Hence, the high sulfide content makes the direct use of untreated sulfidic tailings in aggregate applications impractical [31,139]. Moreover, the typically high content of fine particles harms workability, in addition to the increase in water demand and resulting in an undesirable effect on the microstructure and strength development [140]. Innovative techniques are therefore required to allow for the use of such materials as aggregates in construction. The techniques of granulation or combined granulation/carbonation have been employed to produce secondary aggregates from fly ash [141], basic oxygen furnace (BOF) steel slag [142,143], MSWI ash [144,145], and mine tailings [146]. Improvements in particle size distribution are obtained by the production of granules with adequate mechanical properties for civil engineering applications, as well as reduced leaching of contaminants and CO2 uptake. The use of co-binders in the granulation process is expected to further improve the stabilization of the deleterious heavy metals and sulfides present in the tailings.
Granular aggregates have been produced from sulfidic tailings (S = 9%) using combined hydration of a dual MgO/GGBS binder and CO2 curing [146]. The major mineral phases in the tailings were quartz, calcite, pyrite, arsenopyrite, muscovite, and dolomite. The strength of the granules develops when MgO activates GGBS to produce hydrated phases such as C–S–H and magnesium–silicate–hydrates (M–S–H), which form structural networks in the solidified materials and contribute to the granule strength. The accelerated carbonation conditions contribute to the conversion of MgO to magnesium carbonates, as well as to the carbonation of C–S–H and M–S–H. Factors found to significantly influence the strength of the granules were carbonation conditions, MgO/GGBS ratio, and the granule size i.e., smaller granules (4 mm) were found to be stronger than larger granules (up to 10 mm) due to increased CO2 uptake by the external layer [143,146]. The mine tailings used in the study contained high concentrations of heavy metals like PbO (13.9%) and ZnO (3.1%). However, the leaching behavior of toxic elements has not been assessed. Further studies should investigate the environmental safety of secondary aggregates produced from mine tailings in order to guarantee that they meet environmental acceptance criteria set by national legislations.
While the low aluminosilicate content in such tailings makes them unsuitable candidates for alkali-activation, those materials have been employed as fine aggregates—often referred to in the literature as co-binder, as discussed in Section 3.3—in inorganic polymer-based mortars [31,60]. In addition to good metal immobilization, moderate strength (around 4–5 MPa, suitable for backfilling applications) was obtained. Nevertheless, long-term investigations are required to assess the durability and environmental impact assessment of such mixes.
3.5. Building Ceramics
Non-metallic mineral deposits—e.g., sandstones, gravel, and clay mineral deposits—accounted for almost half (48%) of the global natural resource extraction [147]. Depletion of these natural resources [148] calls for more sustainable approaches within the raw materials sector. For a raw-material-intensive sector like ceramics, which uses large quantities of clay and sand, alternative materials, such as mine tailings, can present a solution to moderate natural resource scarcity and promote environmental net gain. Therefore, from the volume perspective, ceramic production presents an opportunity for the utilization of side stream materials such as sulfidic mine tailings. Nevertheless, the valorization of medium to highly sulfidic mine tailings in traditional ceramics, such as roof tiles, inner wall blocks, pavers, and facing bricks, has not been broadly studied mainly due to their high sulfur and hazardous metal(loid)s contents.
The high sulfur contents of tailing materials can lead to several environmental problems, as well as technical and aesthetical issues of ceramic products. It has been found that the liberation of SOx during the firing process not only leads to increased SOx emissions, but it can also lead to bloating effects in the product [149]. Apart from increasing the SOx emissions and associated costs during the firing process (flue gas cleaning), the high content of sulfur can result in the formation of a black reduction core in the ceramic bodies and, in extreme situations, bloating of the product. A black core is typical for bodies with high releases of SOx and COx [150], which create a reducing atmosphere where Fe2O3 is then reduced to Fe3O4 [151]. While for some ceramic products black core is beneficial as it increases their strength, for ceramic roof tiles a black core can lower their frost resistance during freeze–thaw cycles [152]. Moreover, high concentrations of sulfur in the ceramic body, present in the form of soluble sulfates, can cause efflorescence on the surface of the product. The soluble salts within the raw materials are carried to the surface of the body by capillarity as the water is being driven off during drying process. The formation of soluble sulfates is caused by the reaction between the available SOx and free Na2O, K2O, CaO, the later coming from the decomposition of lime, which originates soluble calcium sulfates, mainly in the mineral phase of gypsum [153] or anhydrite [154]. If soluble sulfates are already present in the unfired wet blend, dry efflorescence can be avoided by adding some BaCO3, as insoluble BaSO4 is formed [155].
Several studies deal with the incorporation of mine tailings, mainly in ceramic bricks, as showcased in previous review articles [156,157,158,159,160,161,162,163]. Only a few studies deal with incorporation into roof tiles [150,164], while wall and floor tiles are more often considered, as well as building glass-ceramics due to immobilization of metal(loid)s in crystalline phases [165]. Nevertheless, due to the aforementioned technical and aesthetical issues raised by high sulfur content, mainly low sulfidic mine tailings were used in detailed investigations. The collected studies and their main conclusions are presented in Supplementary Table S5.
The research studies dealing with environmental performance of sulfidic tailings-based building ceramics are limited. The use of gold (Au) and rare earth elements (REEs) tailings (up to 40 wt.%) was studied in clay-based bricks [149]. Even though open porosities and densities were acceptable for brick production, the material failed the environmental performance test, showing high mobility of As, Pb, and Zn on the fired bricks. The most probable cause is further mobilization of metal(loid)s after the decomposition of sulfides during firing. In another study, bricks incorporating up to 30 wt.% treated calamine (lead) tailings and up to 10 wt.% glass waste showed suitable physical and mechanical properties. However, the fired bricks showed exceeded leaching of Zn after the end-of-life test [166].
In order to overcome the challenges of using sulfidic mine tailings with high sulfur and metal(loid)s content in traditional building ceramics, novel and green cleaning techniques (e.g., microwave roasting, bioleaching) must be further developed and implemented. Such techniques must be applied to the tailings before incorporation in ceramics to remove undesirable sulfur [59,167,168,169] and recover valuable metals [170,171,172,173,174]. These pre-treatment techniques need to be optimized and aligned with the end-product intrinsic characteristics, as well as the technical and aesthetical properties, and environmental compliance. Nevertheless, pre-treatment may not always be required if the sulfide content in the tailings is low. A recent study dealing with low sulfidic Pb-Zn tailings showed that ceramic roof tiles and blocks containing 20 wt.% tailings can achieve satisfying technical and aesthetical properties and can be recycled as aggregate material in a 2nd life scenario, following environmental regulations, without the need of any pre-treatment [175].
4. Concluding Remarks and Further Perspectives
Sulfidic tailings usually accumulate near the mining sites forming heaps of deposits or in tailings impoundments, which occupy considerably large areas and may cause serious environmental contamination, especially for rivers, groundwater, and soils, in case of containment failure. Alternatively, they are used in applications that are capable of tolerating multiple contaminants and require low performance, such as CPBs. Due to the significantly high volumes of production, the requirements for management, and numerous environmental concerns, it is desirable to valorize sulfidic mine tailings in high-volume applications such as construction materials.
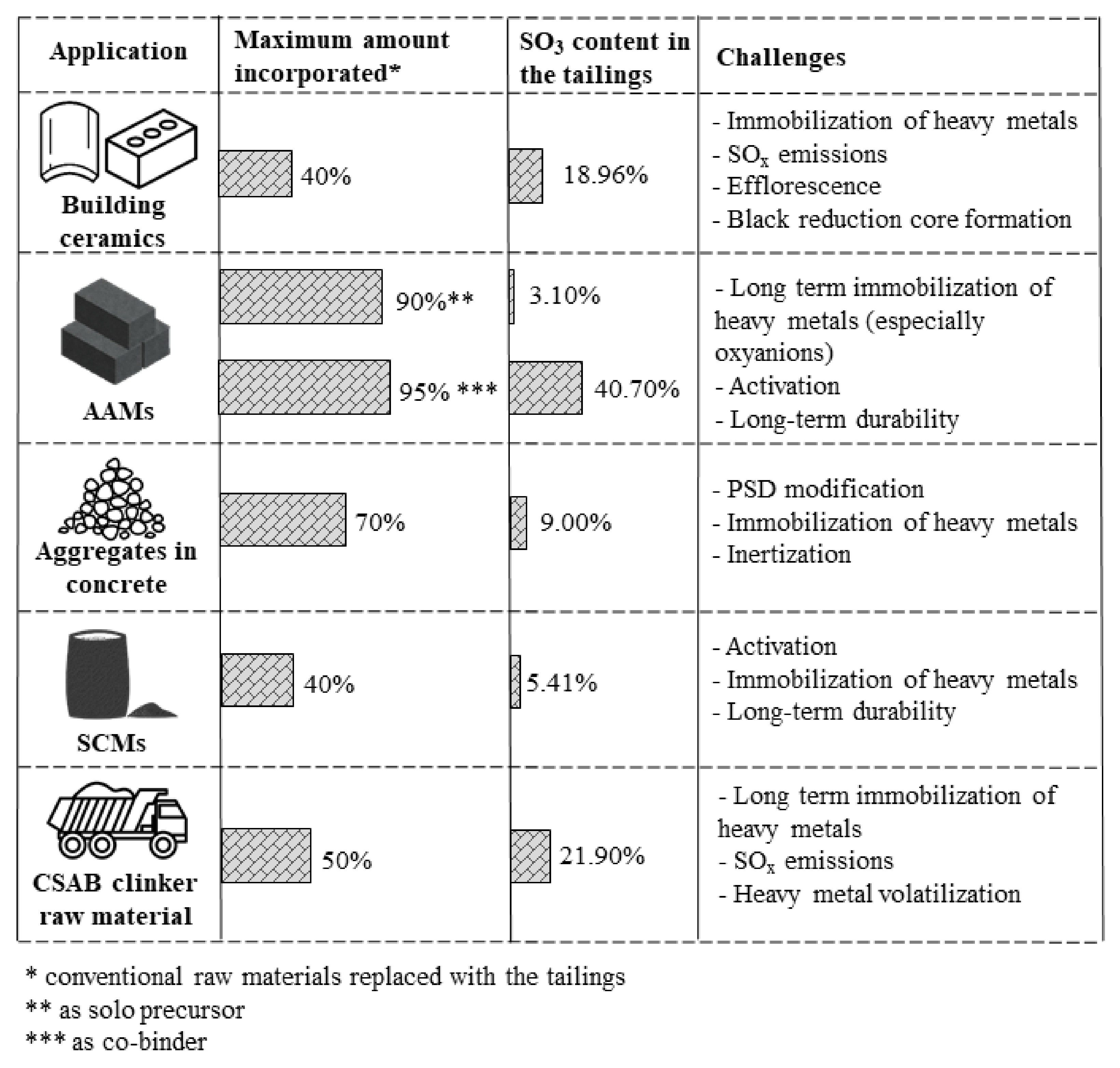
In this review, the potential for utilization of sulfide-rich mine tailings in construction materials was explored. Only a very limited number of studies have attempted to utilize sulfidic mine tailings with >25% SO3 as SCMs, alternative aggregates, building ceramics, clinker raw material, and precursors for AAMs. This is mainly due to several important technical incompatibilities, mostly related to the high sulfur content in the tailings, presence of unstable mineral phases, and high concentrations of heavy metals. Such characteristics originate the technical challenges summarized in Figure 4.

Figure 4.
Overview of challenges of the use of sulfidic tailings in construction materials. Values of maximum amount of tailings incorporated into the products and SO3 content in the tailings correspond to the ones reported in [48,75,100,146,176,177]. Higher incorporation rates tested in those studies show less promising results.
Nevertheless, the average oxide composition of medium and highly sulfidic tailings suggests a high potential for their incorporation in CSAB clinker raw meal. Since clinker making takes place at high temperatures, the mineralogical compositions of the tailings is of lower importance than for applications such as alternative aggregates, SCMs, and precursors in AAMs. Although the rare existing studies confirm this promising valorization path, the research on the topic is still emerging. Additionally, the use of tailings as a co-binder in the preparation of alkali-activated materials has also shown encouraging results especially with respect to efficient immobilization of toxic metals. For that application, as indicated in Figure 4, high incorporation rates of highly sulfidic tailings have been reported in the literature.
Ideally, the removal of heavy metals (As, Cr, Sb, B, V) and extraction of valuable metals (Zn, Fe, Cu, Ni, Pb) must take place upper in the value chain, which requires interdisciplinary collaboration and the development on new technologies. Nevertheless, lower need of pre-treatments can benefit the implementation of sulfidic tailings-based products in terms of cost and energy efficiency, given that the developed tailings-based materials are capable of providing safe disposal of hazardous metals. For most of the applications discussed in this article, the environmental performance of sulfidic tailings-based building materials remains a challenge. In order to comply with the regulatory framework, the hazardous elements present in the tailings must be permanently immobilized onto the products, which demands comprehensive knowledge of the mechanisms of immobilization under both normal and adverse conditions. In addition, in the context of the circular economy, it is important to assess in advance the environmental performance of the products’ second life application in order to avoid landfilling or incinerating. Studies of such nature employing medium or highly sulfidic tailings have not been widely reported.
In the particular case of CSAB clinker production, the existing studies show promising results regarding leaching of heavy metals. The immobilization takes place during the formation of clinker phases and remains after cement is hydrated. Future studies must address the mechanisms of immobilization during both stages, as well as assess the generation of toxic emissions due to heavy metal volatilization. With respect to lowering SOx emissions during clinker making, the raw meal formulation must aim at maximizing sulfur incorporation into clinker phases. Additionally, exploring the potential of minor elements as mineralizers would further promote the upcycling of tailings.
Although the sulfidic tailings present high content of fines, their latent hydraulic or pozzolanic properties are low. Activation processes are therefore required to enhance their suitability for application as an SCM or as a precursor in AAM. As the available literature in AAM shows, those tailings have only been employed to produce mixtures in which other materials i.e., slag, metakaolin, display the role of main precursors. Less attention is given to the active chemical role of the tailings in the mix, and they are often regarded as fillers or fine aggregates is such materials. Future work on the activation of medium and highly sulfidic tailings must take into account that conventional activation methods may not be directly applicable to such materials, given their particular mineralogy. Transforming unstable sulfides into soluble sulfates in a pre-treatment step can be cited as a potentially interesting and rather unexplored route for tailings activation—e.g., to be used as a replacement for anhydrite.
However, the effective selection of appropriate treatments can only be done when the potentially reactive components in the tailings fraction are identified. For that to happen, proper physical, chemical and mineralogical characterization of tailings must be performed. On the other hand, the development of methods for sulfidic tailings’ inertization, i.e., reducing the sulfide and sulfate content, would favor their application as a filler material, aggregate in concrete, and in the ceramics industry. The energy intensity of the pre-treatments needs to be taken into account—while sometimes prohibitive, in other cases highly energy intensive pre-treatments may be justified by the end use potential of the treated tailings.
A clear need was identified to map, identify and characterize sulfidic tailings—e.g., creating a materials passport—in order to fully understand their composition and potential use as resources for other industries. The development of economically feasible and low-impact technologies that extract potentially valuable elements, neutralize or remove incompatible compounds and activate the tailings is key to overcome the barriers for their upcycling in construction materials.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/su132112150/s1, includes highlights of publications on the effect of minor elements in ye’elimite-containing cements (Table S1), a short review of experimental studies on the use of tailings of different origins as a partial replacement for cement (Table S2), a detailed summary of studies on alkali activated co-binders produced by sulfidic mine tailings with other side streams (Table S3), and as solo precursor (Table S4), and a collection of available publications on the production of building ceramics from mine tailings (Table S5).
Author Contributions
Conceptualization, N.P.M., S.S. and P.P.; Writing—original draft, N.P.M., S.S., F.V.S. and H.N.; Writing—review & editing, N.P.M., S.S., F.V.S., H.N., P.P., R.S., M.I., H.C. and G.H. All authors have read and agreed to the published version of the manuscript.
Funding
The research leading to these results has received funding from the European Union’s Horizon 2020 Framework Programme for Research and Innovation under the Grant Agreement no. 812580 (EU H2020 MSCA -ETN SULTAN) and the Academy of Finland (Grant Agreement no. 319676). This publication reflects only the authors’ view, exempting the EU from any liability.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Adiansyah, J.S.; Rosano, M.; Vink, S.; Keir, G. A framework for a sustainable approach to mine tailings management: Disposal strategies. J. Clean. Prod. 2015, 108, 1050–1062. [Google Scholar] [CrossRef] [Green Version]
- Edraki, M.; Baumgartl, T.; Manlapig, E.; Bradshaw, D.; Franks, D.M.; Moran, C.J. Designing mine tailings for better environmental, social and economic outcomes: A review of alternative approaches. J. Clean. Prod. 2014, 84, 411–420. [Google Scholar] [CrossRef]
- Barcelos, D.A.; Pontes, F.V.; da Silva, F.A.; Castro, D.C.; dos Anjos, N.O.; Castilhos, Z.C. Gold mining tailing: Environmental availability of metals and human health risk assessment. J. Hazard. Mater. 2020, 397, 122721. [Google Scholar] [CrossRef] [PubMed]
- BRGM. Management of Mining, Quarrying and Ore-Processing Waste in the European Union 2001. Available online: https://ec.europa.eu/environment/pdf/waste/studies/mining/0204finalreportbrgm.pdf (accessed on 14 April 2021).
- Lottermoser, B.G. Mine Wastes: Characterization, Treatment and Environmental Impacts, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Vidal, O. General Information on Mineral Raw Materials and Metals. In Commodities and Energy; Elsevier: Amsterdam, The Netherlands, 2018; pp. 11–26. [Google Scholar]
- Rimstidt, D.D.; Vaughan, D.J. Pyrite oxidation: A state-of-the-art assessment of the reaction mechanism. Geochim. Cosmochim. Acta 2003, 67, 873–880. [Google Scholar] [CrossRef]
- Hansen, R. Process network modelling of the geochemical reactions responsible for acid mine drainage emanating from the Witwatersrand tailings facilities. S. Afr. J. Geol. 2020, 123, 357–365. [Google Scholar] [CrossRef]
- Kossoff, D.; Dubbin, W.E.; Alfredsson, M.; Edwards, S.J.; Macklin, M.G.; Hudson-Edwards, K.A. Mine tailings dams: Characteristics, failure, environmental impacts, and remediation. Appl. Geochem. 2014, 51, 229–245. [Google Scholar] [CrossRef] [Green Version]
- Plumlee, G.S. The environmental geology of mineral deposits. In The Environmental Geochemistry of Mineral Deposits, Part A. Processes, Techniques, and Health Issues; Society of Economic Geologists: Littleton, CO, USA, 1999; Volume 6A, pp. 71–116. [Google Scholar]
- Ficklin, W.; Mosier, E.; Plumlee, G.; Logsdon, M.; Filipek, L. Field Methods for Sampling and Analysis of Environmental Samples for Unstable and Selected Stable Constituents. In The Environmental Geochemistry of Mineral Deposits; Society of Economic Geologists: Littleton, CO, USA, 1997; pp. 249–264. [Google Scholar]
- Xenidis, A.; Mylona, E.; Paspaliaris, I. Potential use of lignite fly ash for the control of acid generation from sulphidic wastes. Waste Manag. 2002, 22, 631–641. [Google Scholar] [CrossRef]
- Fourie, A. Preventing catastrophic failures and mitigating environmental impacts of tailings storage facilities. Procedia Earth Planet. Sci. 2009, 1, 1067–1071. [Google Scholar] [CrossRef] [Green Version]
- WISE-Uranium-Project. Chronology of Major Tailings Dam Failures. Available online: http://www.wise-uranium.org/mdaf.html (accessed on 10 April 2021).
- Petley, D. The Luming Mine Tailings Spill: So What Happened Next? 2020. Available online: https://blogs.agu.org/landslideblog/2020/04/24/luming-mine-nex/ (accessed on 14 April 2021).
- Thompson, F.; de Oliveira, B.C.; Cordeiro, M.C.; Masi, B.P.; Rangel, T.P.; Paz, P.; Freitas, T.; Lopes, G.; Silva, B.S.; Cabral, A.S.; et al. Severe impacts of the Brumadinho dam failure (Minas Gerais, Brazil) on the water quality of the Paraopeba River. Sci. Total Environ. 2020, 705, 135914. [Google Scholar] [CrossRef]
- Petley, D. Mishor Rotem—Another Tailings Dam Failure, This Time in Israel. 2017. Available online: https://blogs.agu.org/landslideblog/2017/07/07/mishor-rotem-1/ (accessed on 15 April 2021).
- Yilmaz, E.; Benzaazoua, M.; Bussière, B.; Pouliot, S. Influence of disposal configurations on hydrogeological behaviour of sulphidic paste tailings: A field experimental study. Int. J. Miner. Process. 2014, 131, 12–25. [Google Scholar] [CrossRef]
- Bascetin, A.; Tuylu, S. Application of Pb-Zn tailings for surface paste disposal: Geotechnical and geochemical observations. Int. J. Min. Reclam. Environ. 2018, 32, 312–326. [Google Scholar] [CrossRef]
- Moreno, L.; Neretnieks, I. Long-term environmental impact of tailings deposits. Hydrometallurgy 2006, 83, 176–183. [Google Scholar] [CrossRef]
- Nehdi, M.; Tariq, A. Stabilization of sulphidic mine tailings for prevention of metal release and acid drainage using cementitious materials: A review. J. Environ. Eng. Sci. 2007, 6, 423–436. [Google Scholar] [CrossRef]
- Qi, C.; Fourie, A. Cemented paste backfill for mineral tailings management: Review and future perspectives. Miner. Eng. 2019, 144, 106025. [Google Scholar] [CrossRef]
- Jewell, J.; Fourie, A.B. Paste and Thickened Tailings—A Guide, 3rd ed.; Australian Centre for Geomechanics: Perth, Australia, 2015. [Google Scholar]
- Aldhafeeri, Z.; Fall, M. Coupled effect of sulphate and temperature on the reactivity of cemented tailings backfill. Int. J. Min. Reclam. Environ. 2021, 35, 80–94. [Google Scholar] [CrossRef]
- Ercikdi, B.; Kesimal, A.; Cihangir, F.; Deveci, H.; Alp, I. Cemented paste backfill of sulphide-rich tailings: Importance of binder type and dosage. Cem. Concr. Compos. 2009, 31, 268–274. [Google Scholar] [CrossRef]
- Ghirian, A.; Fall, M. Long-term coupled behaviour of cemented paste backfill in load cell experiments. Geomech. Geoengin. Int. J. 2016, 11, 237–251. [Google Scholar] [CrossRef]
- Romano, C.G.; Mayer, K.U.; Jones, D.R.; Ellerbroek, D.A.; Blowes, D.W. Effectiveness of various cover scenarios on the rate of sulfide oxidation of mine tailings. J. Hydrol. 2003, 271, 171–187. [Google Scholar] [CrossRef]
- Hofmann, T.; Schuwirth, N. Zn and Pb release of sphalerite (ZnS)-bearing mine waste tailings. J. Soils Sediments 2008, 8, 433–441. [Google Scholar] [CrossRef]
- Kinnunen, P.H.M.; Kaksonen, A.H. Towards circular economy in mining: Opportunities and bottlenecks for tailings valorization. J. Clean. Prod. 2019, 228, 153–160. [Google Scholar] [CrossRef]
- Dutrow, B.; Klein, C. Manual of Mineral Science, 22nd ed.; John Wiley & Sons: New York, NY, USA, 2002. [Google Scholar]
- Cristelo, N.; Coelho, J.; Oliveira, M.; Consoli, N.C.; Palomo, Á.; Fernández-Jiménez, A. Recycling and application of mine tailings in alkali-activated cements and mortars-strength development and environmental assessment. Appl. Sci. 2020, 10, 2084. [Google Scholar] [CrossRef] [Green Version]
- Van der Ent, A.; Edraki, M. Environmental geochemistry of the abandoned Mamut Copper Mine (Sabah) Malaysia. Environ. Geochem. Health 2018, 40, 189–207. [Google Scholar] [CrossRef] [Green Version]
- Kontopoulos, A.; Komnitsas, K.; Xenidis, A.; Papassiopi, N. Environmental characterisation of the sulphidic tailings in Lavrion. Miner. Eng. 1995, 8, 1209–1219. [Google Scholar] [CrossRef]
- Komnitsas, K.; Kontopoulos, A.; Lazar, I.; Cambridge, M. Risk assessment and proposed remedial actions in coastal tailings disposal sites in Romania. Miner. Eng. 1998, 11, 1179–1190. [Google Scholar] [CrossRef]
- Holmström, H.; Ljungberg, J.; Öhlander, B. Role of carbonates in mitigation of metal release from mining waste. Evidence from humidity cells tests. Environ. Geol. 1999, 37, 267–280. [Google Scholar] [CrossRef]
- Qiu, G.; Luo, Y.; Chen, C.; Lv, Q.; Tan, W.; Liu, F.; Liu, C. Influence factors for the oxidation of pyrite by oxygen and birnessite in aqueous systems. J. Environ. Sci. 2016, 45, 164–176. [Google Scholar] [CrossRef]
- Erdogan, I.G.; Fosso-Kankeu, E.; Ntwampe, S.K.O.; Waanders, F.B.; Hoth, N. Management of Metalliferous Solid Waste and its Potential to Contaminate Groundwater. In Recovery of Byproducts from Acid Mine Drainage Treatment; Wiley: Hoboken, NJ, USA, 2020; pp. 1–21. [Google Scholar]
- USGS. Minerals Yearbook: Volume I. In Metals and Minerals; 2016. Available online: https://www.usgs.gov/centers/nmic/minerals-yearbook-metals-and-minerals (accessed on 4 April 2021).
- Eurostat. Economy-Wide Material Flow Accounts Handbook; Publications Office of the European Union: Luxembourg, 2018. [Google Scholar]
- Norori-McCormac, A.; Brito-Parada, P.R.; Hadler, K.; Cole, K.; Cilliers, J.J. The effect of particle size distribution on froth stability in flotation. Sep. Purif. Technol. 2017, 184, 240–247. [Google Scholar] [CrossRef]
- Westerholm, M.; Lagerblad, B.; Silfwerbrand, J.; Forssberg, E. Influence of fine aggregate characteristics on the rheological properties of mortars. Cem. Concr. Compos. 2008, 30, 274–282. [Google Scholar] [CrossRef]
- Godbout, J.; Bussière, B.; Benzaazoua, M.; Aubertin, M. Influence of pyrrhotite content on the mechanical and chemical behaviour of cemented paste backfill. In Proceedings of the 13th International Seminar on Paste and Thickened Tailings, Toronto, ON, Canada, 3–6 May 2010; pp. 163–174. [Google Scholar]
- Aldhafeeri, Z.; Fall, M.; Pokharel, M.; Pouramini, Z. Temperature dependence of the reactivity of cemented paste backfill. Appl. Geochem. 2016, 72, 10–19. [Google Scholar] [CrossRef]
- Koohestani, B.; Bussière, B.; Belem, T.; Koubaa, A. Influence of polymer powder on properties of cemented paste backfill. Int. J. Miner. Process. 2017, 167, 1–8. [Google Scholar] [CrossRef]
- Chen, X.; Shi, X.; Zhou, J.; Du, X.; Chen, Q.; Qiu, X. Effect of overflow tailings properties on cemented paste backfill. J. Environ. Manag. 2019, 235, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Shao, Y.; Wu, A.; Wang, Z.; Yang, L. Assessment of expansion and strength properties of sulfidic cemented paste backfill cored from deep underground stopes. Constr. Build. Mater. 2020, 230, 116983. [Google Scholar] [CrossRef]
- Liu, L.; Xin, J.; Huan, C.; Qi, C.; Zhou, W.; Song, K.I.I.L. Pore and strength characteristics of cemented paste backfill using sulphide tailings: Effect of sulphur content. Constr. Build. Mater. 2020, 237, 117452. [Google Scholar] [CrossRef]
- Cihangir, F.; Ercikdi, B.; Kesimal, A.; Turan, A.; Deveci, H. Utilisation of alkali-activated blast furnace slag in paste backfill of high-sulphide mill tailings: Effect of binder type and dosage. Miner. Eng. 2012, 30, 33–43. [Google Scholar] [CrossRef]
- Deschamps, T.; Benzaazoua, M.; Bussière, B.; Aubertin, M.; Belem, T. Microstructural and geochemical evolution of paste tailings in surface disposal conditions. Miner. Eng. 2008, 21, 341–353. [Google Scholar] [CrossRef]
- Nehdi, M.; Tariq, A. Evaluation of sulfidic mine tailings solidified/stabilized with cement kiln dust and fly ash to control acid mine drainage. Miner. Metall. Process. 2008, 25, 185–198. [Google Scholar] [CrossRef]
- Cihangir, F.; Akyol, Y. Effect of Desliming of Tailings on the Fresh and Hardened Properties of Paste Backfill Made from Alkali-Activated Slag. Adv. Mater. Sci. Eng. 2020, 2020, 4536257. [Google Scholar] [CrossRef]
- Ercikdi, B.; Külekci, G.; Yilmaz, T. Utilization of granulated marble wastes and waste bricks as mineral admixture in cemented paste backfill of sulphide-rich tailings. Constr. Build. Mater. 2015, 93, 573–583. [Google Scholar] [CrossRef]
- Ouellet, S.; Bussière, B.; Mbonimpa, M.; Benzaazoua, M.; Aubertin, M. Reactivity and mineralogical evolution of an underground mine sulphidic cemented paste backfill. Miner. Eng. 2006, 19, 407–419. [Google Scholar] [CrossRef]
- Tariq, A.; Nehdi, M. Developing durable paste backfill from sulphidic tailings. Proc. Inst. Civ. Eng. Waste Resour. Manag. 2008, 160, 155–166. [Google Scholar] [CrossRef]
- Yılmaz, T.; Ercikdi, B.; Deveci, H. Evaluation of geochemical behaviour of flooded cemented paste backfill of sulphide-rich tailings by dynamic-tank leaching test. Int. J. Min. Reclam. Environ. 2021, 35, 336–355. [Google Scholar] [CrossRef]
- Zheng, J.; Guo, L.; Zhao, Z. Effect of calcined hard kaolin dosage on the strength development of CPB of fine tailings with sulphide. Adv. Mater. Sci. Eng. 2017, 2017, 8631074. [Google Scholar] [CrossRef] [Green Version]
- Benzaazoua, M.; Quellet, J.; Servant, S.; Newman, P.; Verburg, R. Cementitious backfill with high sulfur content physical, chemical, and mineralogical characterization. Cem. Concr. Res. 1999, 29, 719–725. [Google Scholar] [CrossRef]
- Deng, D.Q.; Liang, Y.H.; Huangfu, F.C. Properties of gobi aggregate and sulfide-rich tailings cemented paste backfill and its application in a high-stress metal mine. Adv. Civ. Eng. 2021, 2021, 6624915. [Google Scholar]
- Everaert, M.; Lemmens, V.; Atia, T.A.; Spooren, J. Sulfidic mine tailings and marl waste rock as compatible resources in a microwave-assisted roasting process. J. Clean. Prod. 2020, 274, 122628. [Google Scholar] [CrossRef]
- Paiva, H.; Yliniemi, J.; Illikainen, M.; Rocha, F.; Ferreira, V.M. Mine tailings geopolymers as awaste management solution for a more sustainable habitat. Sustainability 2019, 11, 995. [Google Scholar] [CrossRef] [Green Version]
- Amaratunga, L.M. Cold-bond agglomeration of reactive pyrrhotite tailings for backfill using low cost binders: Gypsum β-hemihydrate and cement. Miner. Eng. 1995, 8, 1455–1465. [Google Scholar] [CrossRef]
- Santisteban, J.I.; Mediavilla, R.; Lopez-Pamo, E.; Dabrio, C.J.; Zapata, M.B.R.; García, M.J.G.; Castano, S.; Martínez-Alfaro, P.E. Loss on ignition: A qualitative or quantitative method for organic matter and carbonate mineral content in sediments? J. Paleolimnol. 2004, 32, 287–299. [Google Scholar] [CrossRef] [Green Version]
- Luus, K. Asbestos: Mining exposure, health effects and policy implications. McGill J. Med. MJM Int. Forum Adv. Med. Sci. Stud. 2007, 10, 121–126. [Google Scholar]
- Cravotta, C.A. Secondary Iron-Sulfate Minerals as Sources of Sulfate and Acidity. In Environmental Geochemistry of Sulfide Oxidation; American Chemical Society: Washington, DC, USA, 1993; Volume 550, pp. 23–345. [Google Scholar]
- Jambor, J.L.; Nordstrom, D.K.; Alpers, C.N. Metal-sulfate salts from sulfide mineral oxidation. Rev. Mineral. Geochem. 2000, 40, 303–350. [Google Scholar] [CrossRef]
- Sobolev, K.; Kozhukhova, M.; Sideris, K.; Menéndez, E.; Santhanam, M. Alternative supplementary cementitious materials. In Properties of Fresh and Hardened Concrete Containing Supplementary Cementitious Materials: State-of-the-Art Report of the RILEM Technical Committee 238-SCM, Working Group 4; De Belie, N., Soutsos, M., Gruyaert, E., Eds.; Springer: Cham, Switzerland, 2018; pp. 233–282. [Google Scholar]
- Bernal, S.A.; Rodríguez, E.D.; Kirchheim, A.P.; Provis, J.L. Management and valorisation of wastes through use in producing alkali-activated cement materials. J. Chem. Technol. Biotechnol. 2016, 91, 2365–2388. [Google Scholar] [CrossRef]
- Habert, G.; Miller, S.A.; John, V.M.; Provis, J.L.; Favier, A.; Horvath, A.; Scrivener, K.L. Environmental impacts and decarbonization strategies in the cement and concrete industries. Nat. Rev. Earth Environ. 2020, 1, 559–573. [Google Scholar] [CrossRef]
- Ludwig, H.M.; Zhang, W. Research review of cement clinker chemistry. Cem. Concr. Res. 2015, 78, 24–37. [Google Scholar] [CrossRef]
- Gou, M.; Zhou, L.; Then, N.W.Y. Utilization of tailings in cement and concrete: A. review. Sci. Eng. Compos. Mater. 2019, 26, 449–464. [Google Scholar] [CrossRef]
- Zhang, L.; Su, M.; Wang, Y. Development of the use of sulfo- and ferroaluminate cements in China. Adv. Cem. Res. 1999, 11, 15–21. [Google Scholar] [CrossRef]
- Aranda, M.A.G.; De la Torre, A.G. Sulfoaluminate cement. In Eco-Efficient Concrete; Woodhead Publishing: Cambridge, UK, 2013; pp. 488–522. [Google Scholar]
- Glasser, F.P.; Zhang, L. High-performance cement matrices based on calcium sulfoaluminate-belite compositions. Cem. Concr. Res. 2001, 31, 1881–1886. [Google Scholar] [CrossRef]
- Gartner, E. Industrially interesting approaches to ‘low-CO2′ cements. Cem. Concr. Res. 2004, 34, 1489–1498. [Google Scholar] [CrossRef]
- Nouairi, J.; Hajjaji, W.; Costa, C.S.; Senff, L.; Patinha, C.; da Silva, E.F.; Labrincha, J.A.; Rocha, F.; Medhioub, M. Study of Zn-Pb ore tailings and their potential in cement technology. J. African Earth Sci. 2018, 139, 165–172. [Google Scholar] [CrossRef]
- Dong, K.; Xie, F.; Wang, W.; Chang, Y.; Chen, C.; Gu, X. Calcination of calcium sulphoaluminate cement using pyrite-rich cyanide tailings. Crystals 2020, 10, 971. [Google Scholar] [CrossRef]
- Martins, N.P.; Snellings, R.; Habert, G. Sulfidic mine tailings as raw material for CSA clinker. In Proceedings of the 74th RILEM Annual Week & The 40th Cement and Concrete Science Conference, Sheffield, UK, 31 August–4 September 2020. [Google Scholar]
- Helser, J.; Cappuyns, V. Environmental assessment of sulfidic mine waste and its integration into green construction materials. In Proceedings of the 23rd EGU General Assembly, Online, 19–30 April 2021. [Google Scholar] [CrossRef]
- Kolovos, K.; Loutsi, P.; Tsivilis, S.; Kakali, G. The effect of foreign ions on the reactivity of the CaO-SiO2-Al2O3-Fe2O3 system: Part I. Anions. Cem. Concr. Res. 2001, 31, 425–429. [Google Scholar] [CrossRef]
- Hanein, T.; Galan, I.; Elhoweris, A.; Khare, S.; Skalamprinos, S.; Jen, G.; Whittaker, M.; Imbabi, M.S.; Glasser, F.P.; Bannerman, M.N. Production of belite calcium sulfoaluminate cement using sulfur as a fuel and as a source of clinker sulfur trioxide: Pilot kiln trial. Adv. Cem. Res. 2016, 28, 643–653. [Google Scholar] [CrossRef] [Green Version]
- Tiantong, P.; Suriwong, T.; Julphunthong, P. Effects of CaF2-CuO additives and various firing temperatures on characteristics of alite calcium sulfoaluminate clinkers. Case Stud. Constr. Mater. 2021, 14, e00493. [Google Scholar]
- Ma, S.; Shen, X.; Gong, X.; Zhong, B. Influence of CuO on the formation and coexistence of 3CaO·SiO2 and 3CaO·3Al2O3·CaSO4 minerals. Cem. Concr. Res. 2006, 36, 1784–1787. [Google Scholar] [CrossRef]
- Ben Haha, M.; Bullerjahn, F. Mineralizer for Calcium Sulfoaluminate Ternesite Cements. WO2016/206785A1, 16 June 2016. Available online: https://patents.google.com/patent/CA2990086A1/en (accessed on 10 April 2021).
- Zea-Garcia, J.D.; Santacruz, I.; Aranda, M.A.G.; De la Torre, A.G. Alite-belite-ye’elimite cements: Effect of dopants on the clinker phase composition and properties. Cem. Concr. Res. 2019, 115, 192–202. [Google Scholar] [CrossRef]
- Luo, L.; Zhang, Y.; Bao, S.; Chen, T. Utilization of Iron Ore Tailings as Raw Material for Portland Cement Clinker Production. Adv. Mater. Sci. Eng. 2016. [Google Scholar] [CrossRef] [Green Version]
- Berardi, R.; Cioffi, R.; Santoro, L. Matrix stability and leaching behaviour in ettringite-based stabilization systems doped with heavy metals. Waste Manag. 1998, 17, 535–540. [Google Scholar] [CrossRef]
- Chrysochooum, M.; Dermatas, D. Evaluation of ettringite and hydrocalumite formation for heavy metal immobilization: Literature review and experimental study. J. Hazard. Mater. 2006, 136, 20–33. [Google Scholar] [CrossRef]
- Fan, C.; Wang, B.; Zhang, T. Review on Cement Stabilization/Solidification of Municipal Solid Waste Incineration Fly Ash. Adv. Mater. Sci. Eng. 2018, 2018, 5120649. [Google Scholar] [CrossRef] [Green Version]
- Berger, S.; Coumes, C.C.D.; Champenois, J.B.; Douillard, T.; Le Bescop, P.; Aouad, G.; Damidot, D. Stabilization of ZnCl2-containing wastes using calcium sulfoaluminate cement: Leaching behaviour of the solidified waste form, mechanisms of zinc retention. J. Hazard. Mater. 2011, 194, 268–276. [Google Scholar] [CrossRef]
- Peysson, S.; Péra, J.; Chabannet, M. Immobilization of heavy metals by calcium sulfoaluminate cement. Cem. Concr. Res. 2005, 35, 2261–2270. [Google Scholar] [CrossRef]
- Piekkari, K.; Ohenoja, K.; Isteri, V.; Tanskanen, P.; Illikainen, M. Immobilization of heavy metals, selenate, and sulfate from a hazardous industrial side stream by using calcium sulfoaluminate-belite cement. J. Clean. Prod. 2020, 258, 120560. [Google Scholar] [CrossRef]
- Luz, C.A.; Pera, J.; Cheriaf, M.; Rocha, J.C. Behaviour of calcium sulfoaluminate cement in presence of high concentrations of chromium salts. Cem. Concr. Res. 2007, 37, 624–629. [Google Scholar] [CrossRef]
- Kiventerä, J.; Piekkari, K.; Isteri, V.; Ohenoja, K.; Tanskanen, P.; Illikainen, M. Solidification/stabilization of gold mine tailings using calcium sulfoaluminate-belite cement. J. Clean. Prod. 2019, 239, 118008. [Google Scholar] [CrossRef]
- Snellings, R. Assessing, Understanding and Unlocking Supplementary Cementitious Materials. RILEM Tech. Lett. 2016, 1, 50. [Google Scholar] [CrossRef] [Green Version]
- Dong, Q.; Liang, B.; Jia, L.; Jiang, L. Effect of sulfide on the long-term strength of lead-zinc tailings cemented paste backfill. Constr. Build. Mater. 2019, 200, 436–446. [Google Scholar] [CrossRef]
- Benzaazoua, M.; Belem, T.; Bussière, B. Chemical factors that influence the performance of mine sulphidic paste backfill. Cem. Concr. Res. 2002, 32, 1133–1144. [Google Scholar] [CrossRef]
- Vargas, F.; Lopez, M. Development of a new supplementary cementitious material from the activation of copper tailings: Mechanical performance and analysis of factors. J. Clean. Prod. 2018, 182, 427–436. [Google Scholar] [CrossRef]
- Zunino, F.; Lopez, M. Decoupling the physical and chemical effects of supplementary cementitious materials on strength and permeability: A multi-level approach. Cem. Concr. Compos. 2016, 65, 19–28. [Google Scholar] [CrossRef]
- Gutteridge, W.A.; Dalziel, J.A. Filler cement: The effect of the secondary component on the hydration of Portland cement: Part, I. A fine non-hydraulic filler. Cem. Concr. Res. 1990, 20, 778–782. [Google Scholar] [CrossRef]
- Guo, Z.; Feng, Q.; Wang, W.; Huang, Y.; Deng, Z.; Xu, Z. Study on Flotation Tailings of Kaolinite-type Pyrite when Used as Cement Admixture and Concrete Admixture. Procedia Environ. Sci. 2016, 31, 644–652. [Google Scholar] [CrossRef] [Green Version]
- De Magalhaes, L.F.; França, S.; dos Santos Oliveira, M.; Peixoto, R.A.F.; Bessa, S.A.L.; da Silva Bezerra, A.C. Iron ore tailings as a supplementary cementitious material in the production of pigmented cements. J. Clean. Prod. 2020, 274, 123260. [Google Scholar] [CrossRef]
- Peng, K.; Yang, H.; Ouyang, J. Tungsten tailing powders activated for use as cementitious material. Powder Technol. 2015, 286, 678–683. [Google Scholar] [CrossRef]
- Ramanathan, S.; Perumal, P.; Illikainen, M.; Suraneni, P. Mechanically activated mine tailings for use as supplementary cementitious materials. RILEM Tech. Lett. 2021, 6, 61–69. [Google Scholar] [CrossRef]
- Habert, G. Assessing the environmental impact of conventional and ‘green’ cement production. In Eco-Efficient Construction and Building Materials: Life Cycle Assessment (LCA), Eco-Labelling and Case Studies; Elsevier Inc.: Amsterdam, The Netherlands, 2013; pp. 199–238. [Google Scholar]
- Bullerjahn, F.; Zajac, M.; Pekarkova, J.; Nied, D. Novel SCM produced by the co-calcination of aluminosilicates with dolomite. Cem. Concr. Res. 2020, 134, 106083. [Google Scholar] [CrossRef]
- Snellings, R.; Cizer, Ö.; Horckmans, L.; Durdziński, P.T.; Dierckx, P.; Nielsen, P.; Van Balen, K.; Vandewalle, L. Properties and pozzolanic reactivity of flash calcined dredging sediments. Appl. Clay Sci. 2016, 129, 35–39. [Google Scholar] [CrossRef]
- Vargas, F.; Lopez, M.; Rigamonti, L. Environmental impacts evaluation of treated copper tailings as supplementary cementitious materials. Resour. Conserv. Recycl. 2020, 160, 104890. [Google Scholar] [CrossRef]
- Xanthopoulos, P.; Kalebić, D.; Kamariah, N.; Busse, J.; Dehaen, W.; Spooren, J.; Binnemans, K. Recovery of Copper from Ammoniacal Leachates by Ion Flotation. J. Sustain. Metall. 2021, 1–13. [Google Scholar] [CrossRef]
- Hu, G.; Dam-Johansen, K.; Wedel, S.; Hansen, J.P. Decomposition and oxidation of pyrite. Prog. Energy Combust. Sci. 2006, 32, 295–314. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymers Inorganic polymeric new materials. J. Therm. Anal. 1991, 37, 1633–1656. [Google Scholar] [CrossRef]
- Provis, J.L.; Van Deventer, J.S.J. Introduction to geopolymers. In Geopolymers; Elsevier: Amsterdam, The Netherlands, 2009; pp. 1–11. [Google Scholar]
- Kiventerä, J.; Perumal, P.; Yliniemi, J.; Illikainen, M. Mine tailings as a raw material in alkali-activation—A review. Int. J. Miner. Metall. Mater. 2020, 27, 1009. [Google Scholar] [CrossRef]
- Van Jaarsveld, J.G.S.; Van Deventer, J.S.J.; Schwartzman, A. The potential use of geopolymeric materials to immobilise toxic metals: Part II. Material and leaching characteristics. Miner. Eng. 1999, 12, 75–91. [Google Scholar] [CrossRef]
- Van Jaarsveld, J.G.S.; Van Deventer, J.S.J.; Lorenzen, L. The potential use of geopolymeric materials to immobilise toxic metals: Part I. Theory and applications. Miner. Eng. 1997, 10, 659–669. [Google Scholar] [CrossRef]
- Álvarez-Ayuso, E.; Querol, X.; Plana, F.; Alastuey, A.; Moreno, N.; Izquierdo, M.; Font, O.; Moreno, T.; Diez, S.; Vázquez, E.; et al. Environmental, physical and structural characterisation of geopolymer matrixes synthesised from coal (co-)combustion fly ashes. J. Hazard. Mater. 2008, 154, 175–183. [Google Scholar] [CrossRef]
- Kiventerä, J.; Golek, L.; Yliniemi, J.; Ferreira, V.; Deja, J.; Illikainen, M. Utilization of sulphidic tailings from gold mine as a raw material in geopolymerization. Int. J. Miner. Process. 2016, 149, 104–110. [Google Scholar] [CrossRef]
- Kiventerä, J.; Lancellotti, I.; Catauro, M.; Poggetto, F.D.; Leonelli, C.; Illikainen, M. Alkali activation as new option for gold mine tailings inertization. J. Clean. Prod. 2018, 187, 76–84. [Google Scholar] [CrossRef]
- Pacheco-Torgal, F.; Castro-Gomes, J.; Jalali, S. Geopolymeric binder using tungsten mine waste: Preliminary investigation. In Proceedings of the Geopolymer 2005 World Congress, Saint Quentin, France, 29 June–1 July 2005; pp. 93–98. [Google Scholar]
- Perumal, P.; Piekkari, K.; Sreenivasan, H.; Kinnunen, P.; Illikainen, M. One-part geopolymers from mining residues—Effect of thermal treatment on three different tailings. Miner. Eng. 2019, 144, 106026. [Google Scholar] [CrossRef]
- Yao, G.; Liu, Q.; Wang, J.; Wu, P.; Lyu, X. Effect of mechanical grinding on pozzolanic activity and hydration properties of siliceous gold ore tailings. J. Clean. Prod. 2019, 217, 12–21. [Google Scholar] [CrossRef]
- Perumal, P.; Niu, H.; Kiventerä, J.; Kinnunen, P.; Illikainen, M. Upcycling of mechanically treated silicate mine tailings as alkali activated binders. Miner. Eng. 2020, 158, 106587. [Google Scholar] [CrossRef]
- Niu, H.; Kinnunen, P.; Sreenivasan, H.; Adesanya, E.; Illikainen, M. Structural collapse in phlogopite mica-rich mine tailings induced by mechanochemical treatment and implications to alkali activation potential. Miner. Eng. 2020, 151, 106331. [Google Scholar] [CrossRef]
- Ahmari, S.; Zhang, L. Durability and leaching behavior of mine tailings-based geopolymer bricks. Constr. Build. Mater. 2013, 44, 743–750. [Google Scholar] [CrossRef]
- Adesanya, E.; Ohenoja, K.; Di Maria, A.; Kinnunen, P.; Illikainen, M. Alternative alkali-activator from steel-making waste for one-part alkali-activated slag. J. Clean. Prod. 2020, 274, 123020. [Google Scholar] [CrossRef]
- Khalifa, A.Z.; Cizer, Ö.; Pontikes, Y.; Heath, A.; Patureau, P.; Bernal, S.A.; Marsh, A.T. Advances in alkali-activation of clay minerals. Cem. Concr. Res. 2020, 132, 106050. [Google Scholar] [CrossRef]
- Bell, J.L.; Kriven, W.M. Formation of an iron-based inorganic polymer (geopolymer). In Proceedings of the Mechanical Properties and Performance of Engineering Ceramics and Composites IV—A Collection of Papers Presented at the 33rd International Conference on Advanced Ceramics and Composites, Daytona Beach, FL, USA, 18–23 January 2009; pp. 301–312. [Google Scholar]
- Mehta, P.K.; Monteiro, P.J.M. Concrete: Microstructure, Properties, and Materials, 4th ed.; McGraw-Hill: New York, NY, USA, 2014. [Google Scholar]
- Pacheco-Torgal, F.; Labrincha, J.A.; Leonelli, C.; Palomo, A.; Chindaprasirt, P. Handbook of Alkali-Activated Cements, Mortars and Concretes, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Alexander, M.; Mindess, S. Aggregates in Concrete Modern Concrete Technology, 1st ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Capraro, A.P.B.; Braga, V.; de Medeiros, M.H.F.; Hoppe Filho, J.; Bragança, M.O.; Portella, K.F.; de Oliveira, I.C. Internal attack by sulphates in cement pastes and mortars dosed with different levels of pyrite. J. Build. Pathol. Rehabil. 2017, 2, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Duchesne, J.; Rodrigues, A.; Fournier, B. Concrete damage due to oxidation of sulfide-bearing aggregate: A review of recent advances and limitations. RILEM Tech. Lett. 2021, 6, 82–92. [Google Scholar] [CrossRef]
- Rodrigues, A.; Duchesne, J.; Fournier, B.; Durand, B.; Rivard, P.; Shehata, M. Mineralogical and chemical assessment of concrete damaged by the oxidation of sulfide-bearing aggregates: Importance of thaumasite formation on reaction mechanisms. Cem. Concr. Res. 2012, 42, 1336–1347. [Google Scholar] [CrossRef]
- Tagnit-Hamou, A.; Saric-Coric, M.; Rivard, P. Internal deterioration of concrete by the oxidation of pyrrhotitic aggregates. Cem. Concr. Res. 2005, 35, 99–107. [Google Scholar] [CrossRef]
- Rodrigues, A.; Duchesne, J.; Fournier, B. Quantitative assessment of the oxidation potential of sulfide-bearing aggregates in concrete using an oxygen consumption test. Cem. Concr. Compos. 2016, 67, 93–100. [Google Scholar] [CrossRef]
- Schmidt, T.; Leemann, A.; Gallucci, E.; Scrivener, K. Physical and microstructural aspects of iron sulfide degradation in concrete. Cem. Concr. Res. 2011, 41, 263–269. [Google Scholar] [CrossRef]
- Casanova, I.; Aguado, A.; Agulló, L. Aggregate expansivity due to sulfide oxidation—II. Physico-chemical modeling of sulfate attack. Cem. Concr. Res. 1997, 27, 1627–1632. [Google Scholar] [CrossRef]
- Shayan, A. Deterioration of a concrete surface due to the oxidation of pyrite contained in pyritic aggregates. Cem. Concr. Res. 1988, 18, 723–730. [Google Scholar] [CrossRef]
- Midgley, H.G. The staining of concrete by pyrite. Mag. Concr. Res. 1958, 10, 75–78. [Google Scholar] [CrossRef]
- Ndlovu, S.; Simate, G.S.; Matinde, E. Mining and Beneficiation Waste Production and Utilization. In Waste Production and Utilization in the Metal Extraction Industry; CRC Press: Boca Raton, FL, USA, 2017; pp. 65–112. [Google Scholar] [CrossRef]
- Cihangir, F.; Ercikdi, B.; Kesimal, A.; Ocak, S.; Akyol, Y. Effect of sodium-silicate activated slag at different silicate modulus on the strength and microstructural properties of full and coarse sulphidic tailings paste backfill. Constr. Build. Mater. 2018, 185, 555–566. [Google Scholar] [CrossRef]
- Arsla, H.; Baykal, G. Utilization of fly ash as engineering pellet aggregates. Environ. Geol. 2006, 50, 761–770. [Google Scholar] [CrossRef]
- Morone, M.; Costa, G.; Georgakopoulos, E.; Manovic, V.; Stendardo, S.; Baciocchi, R. Granulation–Carbonation Treatment of Alkali Activated Steel Slag for Secondary Aggregates Production. Waste Biomass Valorization 2017, 8, 1381–1391. [Google Scholar] [CrossRef] [Green Version]
- Morone, M.; Costa, G.; Polettini, A.; Pomi, R.; Baciocchi, R. Valorization of steel slag by a combined carbonation and granulation treatment. Miner. Eng. 2014, 59, 82–90. [Google Scholar] [CrossRef] [Green Version]
- Cioffi, R.; Colangelo, F.; Montagnaro, F.; Santoro, L. Manufacture of artificial aggregate using MSWI bottom ash. Waste Manag. 2011, 31, 281–288. [Google Scholar] [CrossRef]
- Medici, F.; Piga, L.; Rinaldi, G. Behaviour of polyaminophenolic additives in the granulation of lime and fly-ash. Waste Manag. 2000, 20, 491–498. [Google Scholar] [CrossRef]
- Kim, T.Y.; Ahn, J.Y.; Kim, C.; Choi, S.J.; Ho, T.T.; Moon, D.H.; Hwang, I. Carbonation/granulation of mine tailings using a MgO/ground-granule blast-furnace-slag binder. J. Hazard. Mater. 2019, 378, 120760. [Google Scholar] [CrossRef]
- UN Environment Programme. Global Resources Outlook 2019: Natural Resources for the Future We Want. 2019. Available online: https://www.resourcepanel.org/reports/global-resources-outlook (accessed on 5 April 2021).
- Bendixen, M.; Best, J.; Hackney, C.; Iversen, L.L. Time is running out for sand. Nature 2019, 571, 29–31. [Google Scholar] [CrossRef]
- Belmonte, L.J.; Ottosen, L.M.; Kirkelund, G.M.; Jensen, P.E.; Vestbø, A.P. Screening of heavy metal containing waste types for use as raw material in Arctic clay-based bricks. Environ. Sci. Pollut. Res. 2018, 25, 32831–32843. [Google Scholar] [CrossRef] [Green Version]
- Veiga Simão, F.; Chambart, H.; Vandemeulebroeke, L.; Cappuyns, V. Incorporation of sulphidic mining waste material in ceramic roof tiles and blocks. J. Geochemical. Explor. 2021, 225, 106741. [Google Scholar] [CrossRef]
- Gredmaier, L.; Banks, C.J.; Pearce, R.B. Calcium and sulphur distribution in fired clay brick in the presence of a black reduction core using micro X-ray fluorescence mapping. Constr. Build. Mater. 2011, 25, 4477–4486. [Google Scholar] [CrossRef] [Green Version]
- Pavlovets, A.Y.; Pavlova, I.A.; Farafontova, E.P. Bochkarihinskoe clay as raw material to building ceramics production. IOP Conf. Ser. Mater. Sci. Eng. 2020, 972, 12045. [Google Scholar] [CrossRef]
- Chwast, J.; Todorović, J.; Janssen, H.; Elsen, J. Gypsum efflorescence on clay brick masonry: Field survey and literature study. Constr. Build. Mater. 2015, 85, 57–64. [Google Scholar] [CrossRef] [Green Version]
- Guzulla, M.F.; Gomez-Tena, M.P.; Orduna, M.; Vicente, S.; Amoros, J.L. Determination of soluble salts and efflorescence in ceramic roofing tiles. Bol. Soc. Esp. Ceram. Vidr. 2010, 49, 189–196. [Google Scholar]
- Ersoy, B.; Kavas, T.; Evcin, A.; Başpinar, S.; Sariişik, A.; Önce, G. The effect of BaCO3 addition on the sintering behavior of lignite coal fly ash. Fuel 2008, 87, 2563–2571. [Google Scholar] [CrossRef]
- Zhang, L. Production of bricks from waste materials—A review. Constr. Build. Mater. 2013, 47, 643–655. [Google Scholar] [CrossRef]
- Monteiro, S.N.; Vieira, C.M.F. On the production of fired clay bricks from waste materials: A critical update. Constr. Build. Mater. 2014, 68, 599–610. [Google Scholar] [CrossRef]
- Da Silva, F.L.; Araújo, F.G.S.; Teixeira, M.P.; Gomes, R.C.; Von Krüger, F.L. Study of the recovery and recycling of tailings from the concentration of iron ore for the production of ceramic. In Ceramics International; Elsevier Ltd.: Amsterdam, The Netherlands, 2014; Volume 40, pp. 16085–16089. [Google Scholar]
- Bories, C.; Borredon, M.E.; Vedrenne, E.; Vilarem, G. Development of eco-friendly porous fired clay bricks using pore-forming agents: A review. J. Environ. Manag. 2014, 143, 186–196. [Google Scholar] [CrossRef]
- Boltakova, N.V.; Faseeva, G.R.; Kabirov, R.R.; Nafikov, R.M.; Zakharov, Y.A. Utilization of inorganic industrial wastes in producing construction ceramics. Review of Russian experience for the years 2000–2015. Waste Manag. 2017, 60, 230–246. [Google Scholar] [CrossRef]
- Murmu, A.L.; Patel, A. Towards sustainable bricks production: An overview. Constr. Build. Mater. 2018, 165, 112–125. [Google Scholar] [CrossRef]
- Muñoz, P.; Morales, M.P.; Letelier, V.; Mendivil, M.A. Fired clay bricks made by adding wastes: Assessment of the impact on physical, mechanical and thermal properties. Constr. Build. Mater. 2016, 125, 241–252. [Google Scholar] [CrossRef]
- Shreekant, R.L.; Aruna, M.; Vardhan, H. Utilisation of mine waste in the construction industry—A Critical Review. Int. J. Earth Sci. Eng. 2016, 9, 182–195. [Google Scholar]
- Flores-Badillo, J.; Ávila, J.H.; Cardona, F.P.; Pineda, N.Y.T.; Santos, J.A.O. Developing Alternative Industrial Materials from Mining Waste. In Engineering Solutions for Sustainability: Materials and Resources II; Fergus, J.W., Mishra, B., Anderson, D., Sarver, E.A., Neelameggham, N.R., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 119–125. [Google Scholar]
- Marabini, A.M.; Plescia, P.; Maccari, D.; Burragato, F.; Pelino, M. New materials from industrial and mining wastes: Glass-ceramics and glass-and rock-wool fibre. Int. J. Miner. Process. 1998, 53, 121–134. [Google Scholar] [CrossRef]
- Taha, Y.; Benzaazoua, M.; Mansori, M.; Hakkou, R. Recycling feasibility of glass wastes and calamine processing tailings in fired bricks making. Waste Biomass Valorization 2017, 8, 1479–1489. [Google Scholar] [CrossRef]
- Amankwah, R.K.; Ofori-Sarpong, G. Microwave roasting of flash flotation concentrate containing pyrite, arsenopyrite and carbonaceous matter. Miner. Eng. 2020, 151, 106312. [Google Scholar] [CrossRef]
- Hu, N.; Chen, W.; Ding, D.X.; Li, F.; Dai, Z.R.; Li, G.Y.; Wang, Y.D.; Zhang, H.; Lang, T. Role of water contents on microwave roasting of gold bearing high arsenic sulphide concentrate. Int. J. Miner. Process. 2017, 161, 72–77. [Google Scholar] [CrossRef]
- Ma, S.J.; Luo, W.J.; Mo, W.; Su, X.J.; Liu, P.; Yang, J.L. Removal of arsenic and sulfur from a refractory gold concentrate by microwave heating. Miner. Eng. 2010, 23, 61–63. [Google Scholar] [CrossRef]
- Martin, M.; Janneck, E.; Kermer, R.; Patzig, A.; Reichel, S. Recovery of indium from sphalerite ore and flotation tailings by bioleaching and subsequent precipitation processes. Miner. Eng. 2015, 75, 94–99. [Google Scholar] [CrossRef]
- Sadeghieh, S.M.; Ahmadi, A.; Hosseini, M.R. Effect of water salinity on the bioleaching of copper, nickel and cobalt from the sulphidic tailing of Golgohar Iron Mine, Iran. Hydrometallurgy 2020, 198, 105503. [Google Scholar] [CrossRef]
- Akbari, S.; Ahmadi, A. Recovery of copper from a mixture of printed circuit boards (PCBs) and sulphidic tailings using bioleaching and solvent extraction processes. Chem. Eng. Process. Process Intensif. 2019, 142, 107584. [Google Scholar] [CrossRef]
- Mäkinen, J.; Salo, M.; Khoshkhoo, M.; Sundkvist, J.-E.; Kinnunen, P. Bioleaching of cobalt from sulfide mining tailings; a mini-pilot study. Hydrometallurgy 2020, 196, 105418. [Google Scholar] [CrossRef]
- Zhang, R.; Hedrich, S.; Römer, F.; Goldmann, D.; Schippers, A. Bioleaching of cobalt from Cu/Co-rich sulfidic mine tailings from the polymetallic Rammelsberg mine, Germany. Hydrometallurgy 2020, 197, 105443. [Google Scholar] [CrossRef]
- Simão, F.V.; Chambart, H.; Vandemeulebroeke, L.; Nielsen, P.; Cappuyns, V. Turning Mine Waste into a Ceramic Resource: Plombières Tailing Case. J. Sustain. Metall. 2021. [Google Scholar] [CrossRef]
- Liu, T.; Tang, Y.; Han, L.; Song, J.; Luo, Z.; Lu, A. Recycling of harmful waste lead-zinc mine tailings and fly ash for preparation of inorganic porous ceramics. Ceram. Int. 2017, 43, 4910–4918. [Google Scholar] [CrossRef]
- Pacheco-Torgal, F.; Castro-Gomes, J.P.; Jalali, S. Investigations on mix design of tungsten mine waste geopolymeric binder. Constr. Build. Mater. 2008, 22, 1939–1949. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).