Abstract
In recent decades, the microcapsules of lipids, compound lipids, and essential oils, have found numerous potential practical applications in food, textiles, agricultural products, as well as pharmaceuticals. This article discusses the encapsulation of fat-soluble vitamins, essential oils, polyunsaturated fatty acids, and structured lipids. Consequently, the compiled information establishes the criteria to better select encapsulating agents as well as combinations of encapsulating agents best suited to the types of active ingredient to be encapsulated. This review shows a trend towards applications in food and pharmacology as well as the increase in research related to microencapsulation by the spray drying of vitamins A and E, as well as fish oil, thanks to its contribution of omega 3 and omega 6. There is also an increase in articles in which spray drying is combined with other encapsulation techniques, or modifications to the conventional spray drying system.
1. Introduction
Bioactive ingredients have become an important component in the diet of humans, particularly because consumers are more aware of the direct impact of food on human health [1]. Some statistics show that consumers are more keen to consume functional foods, as reported by Market Research Transparency in 2016. It is expected that the functional food global market could exceed more than 309 billion on sale by 2027 [2]. According to technological advances, functional food products have gained special attention on the design and manufacture of both functional and nutraceuticals food types through the incorporation of bioactive ingredients [3]. Functional ingredients are of paramount importance for obtaining stable microparticles that maintain bioactive ingredients aimed at providing protection against different physical or chemical reactions that could potentially conduce to degradation [4]. The production of functional foods implies some challenges, both in the incorporation of the bioactive ingredients in the food matrix, demonstrating their stability during the processing period, but also their stability during storage. Additionally, interactions should be certainly minimized. It is also essential to guarantee the continued functionality of the bioactive ingredients until they reach the gastro-intestinal tract, to be released at the targeted site [5]. The above microencapsulation requirements seek to ensure both protection and release under target conditions; however, these are not the only parameters that must be considered during the encapsulation of bioactive ingredients [6]. In addition, it is important to understand the characteristics of bioactive compounds such as solubility, chemical structure, and physiological functions, which allows to determine protection requirements against various environmental factors. Knowing that lipids are fats and fat-like substances, which differ in chemical composition, structure, and by physiological and biochemical function [7], it could be possible to predict the optimum encapsulation process, as well as encapsulation agents.
Lipids, compound lipids and essential oils are particularly sensitive to heat and susceptible to factors such as oxidation, humidity, light, and air [6,8], as such conditions limit their application. Microencapsulation is widely used to extend the stability of bioactive lipidic compounds through processing, storage, and handling [5]. In this sense, the structured lipids have gained commercial relevance as it is possible to optimize the bioavailability of individual or the totality of the component fatty acids within the triacylglyceride or phospholipid in question. A structured lipid, e.g., a structured triacylglyceride, is a “tailor made” molecule formulated for a specific nutritional and/or technological function. Structured lipids have gained relevance as it is possible to optimize the bioavailability of the individual or the totality of the component fatty acids within the triacylglyceride or phospholipid in question. This would allow to determine the type of fatty acids, as well as their order for the desired structure [9].
Microencapsulation technology is as a process for coating bioactive ingredients in the form of droplets. Such bioactive ingredients could be incorporated evenly in a homogeneous or heterogeneous matrix, obtaining as a final result small capsules that allow the protection of the active ingredient within the core, while allowing their controlled release [10]. The microencapsulation process encompasses a set of steps or conditions required to protect the bioactive ingredients of solid, liquid, and gaseous materials observed as micro or nano capsules, with the characteristic of releasing their contents under conditions and quantities specifics through the time [11]. Among the areas in which microencapsulation technology has been used are biomedical, textiles, and medicine, and it has also been widely used in pharmaceutical applications, as well as food industries [12]. The main reasons why microencapsulation is of such relevance in food and pharma are associated with: (i) reducing the interaction of the bioactive ingredients in different environments (heat, humidity, air and light); (ii) reducing the probability of losing bioactivity; (iii) facilitating handling; (iv) releasing under specific conditions the encapsulated, active ingredient; (v) preventing the occurrence of undesired flavors and odors; and (vi) the dosing of the bioactive ingredient when used in small quantities [13].
As previously mentioned, one of the most common uses of microencapsulation technology is the production of high value-added food with the aim of obtaining stable functional foods that possess desirable, specific properties, such as mentioned before. The cost is one of the most prevalent variables that will determine the applicability of the microencapsulation process for a certain product. Low microcapsule production cost will allow for maintaining a competitive price without compromising product quality, especially when it comes to health-related products [11]. At the industrial/commercial level, there are three aspects that must be evaluated during the micro encapsulation process, these are: (a) the production cost of the microcapsules; (b) the possibility that said microcapsules can be used in various products, or in a wide range of products; and (c) the possibility of scaling-up from proof of concept to large-scale production.
Despite the widespread use of the spray drying technique for the production of microcapsules, it is important to highlight other encapsulation techniques that have gained strength in recent years. Among these are coacervation and molecular inclusion, as well as fluidized bed, and coacervation. All microencapsulation techniques are governed by the same basic principle, which is the protection of a specific compound. However, the technique of choice is governed by aspects such as the properties of the bioactive ingredient (physical and chemical), desired particle size, the physical-chemical properties of the encapsulating material or substrate, production costs associated with microencapsulation, and controlled release mechanisms [14,15]. In light of the aforementioned factors, spray drying has widely demonstrated attractive advantages over other encapsulation techniques, such as: (1) low microcapsule manufacturing costs; (2) the wide variety of available encapsulating agents; (3) the high retention and stability of volatile compounds in the final product; and (4) the possibility of scaling at an industrial level allowing for large-scale production [16,17,18,19].
This review aims to ask the question: are there significant advances in the encapsulation of essential oils, lipids, or compound lipids, by spray drying? This review considers the main encapsulating agents and bioactive compounds used in the spray drying of essential oils and polyunsaturated fatty acids. It includes a theoretical framework of the encapsulation process, the most representative encapsulation techniques, and also describes the spray drying encapsulation methodology. Additionally, the spray drying microencapsulation method will be discussed, followed by an overview of relevant literature published in the space of the microencapsulation of different lipids, and its comparison with other microencapsulation techniques.
2. Materials and Methods
2.1. Methods
The period between the years 2000 and 2021 was selected in order to know the progress of the subject of the encapsulation of essential oils, lipids, and lipid components, by spray drying during the course of the 21st century. Additionally, it seeks to identify the main encapsulating materials and the appearance of new encapsulating materials over time.
Phase 1: Information sources and keywords were identified in Scopus (“encapsulation”, “spray dry”, “essential oil”, “lipids”, and “compound lipid”). The search criteria was selected, focusing on the years 2000–2021. The search equation was TITLE-ABS-KEY ((“essential oil” OR “lipids” OR “lipid compound”) AND encapsulation AND “spray drying”), which led to a total of 382 articles (Figure 1).

Figure 1.
Methodology outline.
Phase 2: A table format was created that contains the following criteria: article name, author, year, country, keywords, abstract, journal, Scimago quartile, and explicit mention of spray drying and encapsulation of essential oil or lipids or compound lipids.
Phase 3: The analysis was carried out in two thematic axes found in 120 papers: axis 1 related to encapsulation by spray drying and axis 2 on the encapsulation of essential oils, or lipids, or compound lipids.
2.2. Theortical Framework of the Microencapsulation
In microencapsulation, small particles of an active ingredient are coated or trapped within a film composed of one or more materials [20,21,22]. The active ingredient to be protected is known as the core material, bioactive ingredient, or internal phase, and can be encapsulated pure or in combination with other materials [12,17,23]. On the other hand, the coating material is called encapsulating agent or wall material [14,17]. The coating can be composed by a mixture of materials that have different physical and chemical properties, thus allowing to obtain a reinforced coating adapted to specific conditions [10,24].
The coacervation technique for microencapsulation has been recorded to be applied for the first time at an industrial level in 1950, when the National Cash Register Company developed “carbonless carbon paper” [25], and it has been used ever since then. Currently, microencapsulation is used in various productive sectors such as textiles [26,27,28], agricultural [10,20,29], cosmetics [30,31,32,33], and pharmaceuticals [13,16,28,29,34]. In the food industry, microencapsulation is utilized to avoid the loss of flavor during processing or storage, because they are usually volatile compounds, highly sensitive to environmental conditions such as light, heat, humidity, oxygen, and radiation, among others, thus reducing unwanted interactions of the bioactive ingredients with the exterior environment [31,35]. Other advantages associated with microencapsulation include the conservation of nutritional value, controlling the release of the encapsulation, the ability to mask some undesired flavors of certain compounds, and facilitating the handling and transportation of materials by changing their original shape and volume [11,20,36].
The size, shape, and structure of these microcapsules is a function of the selected type of encapsulating material, the active ingredient to be encapsulated, and microencapsulation technique [14,31,37]. On the other hand, encapsulation efficiency, the stability of the microparticles, and the characteristics of the final product, are directly related to the encapsulating material of choice. The final particles may present different structures and morphologies, such as single-core, multinuclear, nonspherical multinuclear, multilayer, or multicore (Figure 2).
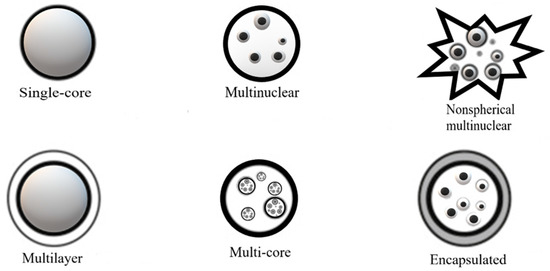
Figure 2.
Morphologies of particles obtained using encapsulation processes (adapted from [17]).
In order to predict the physical and chemical interactions that could occur between the bioactive ingredient and wall material, it is necessary to know the encapsulating material and its properties. Ideally, the encapsulating agent will present the following characteristics: (1) must not react with the encapsulated active ingredient; (2) must protect the encapsulated ingredient from the outside environment; (3) it must not be toxic; and (4) should not be cost prohibitive, among others [38,39]. The techniques used for the encapsulation of compounds are varied. The selection of the encapsulation technique to be used is carried out considering the physical and chemical properties of both the encapsulating agent and the encapsulated agent. Additionally, by knowing the targeted particle size as well as its shape, release rate, process, and its possible scaling, it is of paramount importance for the selection of the correct microencapsulation technique [40].
2.2.1. Representative Encapsulation Techniques
Currently, there are various encapsulation methods used at both laboratory and industrial level. Over time, and with increasing technological developments, new encapsulation techniques will continue to emerge and adapt to the needs for various bioactive compounds to be protected, encapsulating substrate materials and market needs. Microencapsulation was originally used to protect the active ingredients against environmental and/or external factors [41,42]; however, over time, controlled release has become a fundamental factor in the development of encapsulation systems [43,44]. Encapsulation techniques can be classified into chemical, physical-chemical, or mechanical [41,45] techniques. Among the most important techniques used for lipids protection are: coacervation; spray drying; liposomes; fluid bed spray coating; and emulsification solvent evaporation/extraction (Figure 3). These encapsulation techniques are explained below.
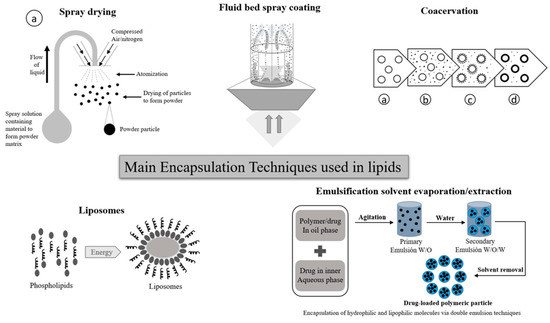
Figure 3.
Encapsulation techniques used for lipid protection.
- -
- Spray drying is a widely used manufacturing technique used in food, agri-food, and the pharmaceutical industry, among others. This technology allows to obtain a powder product, starting from a concentrated liquid solution or suspension. Its operating principle is based on the atomization of the solution, thus generating small micro droplets upon contact with a sprayed stream of hot air (between 150 °C to 300 °C) [45].
- -
- In the case of coacervation, a homogeneous solution of charged macromolecules separates into two liquid phases in equilibrium, a colloidal suspension in which the more concentrated phase is known as the “coacervated phase”, and the other is known as the “equilibrium phase”. During the simple coacervation process of a polyelectrolyte, the addition of salt or alcohol normally promotes phase segregation, through the self-neutralization of the loads [45,46]. Additionally, the coacervation complex involves the interaction of at least two biopolymers fillers. Parameters such as concentration, biopolymers ratio, temperature, pH, ionic strength, and charge density, must be balanced for an efficient phase separation [47,48].
- -
- Liposomes are vesicular structures made up of lipids organized in bilayers or multilayers. These structures are similar to the structure of the membrane lipid. In the medical field, model studies have been developed of the physical behavior and chemistry of the cell membrane, cell compartments, and the cells themselves vesicular transport structures in and out of the cell. Liposomes are considered vesicles with a unilamellar spherical structure or multilamellar; that is, its vesicular lipid conformation may have a bilayer lipid or several concentric lipid bilayers. By their conformation, unilamellar liposomes are classified into small (SUV), medium (MUV), large (LUV), or giant (GUV) [49,50].
- -
- Spray coating in a fluidized bed system allows for obtaining a suitable surface coating by uniformly applying the coating material. In this method, the coating material, which is in liquid state, is sprayed onto all the particles as they move in the fluidized bed [51,52], at the time that the aqueous or organic solution is evaporated, forming the coating layer. With this technique, it is possible to obtain particle size of 100 μm up to 3 mm [52,53]. On the other hand, emulsification solvent evaporation/extraction consists in forming an emulsion by combining a polymer and a volatile organic solvent. By heating the emulsion, the solvent is evaporated [54,55]. The solution is formed by the dispersion of the active component and the encapsulating agent, this dispersion is emulsified in an external aqueous phase, in which the polymer is insoluble. In this technique, the use of stabilizing agents that favor the formation of the particles and the retention of the bioactive compound inside each capsule is common [55]. This technique is normally used to microencapsulate hydrophobic bioactive compounds; it can additionally perform both a single emulsion and a double emulsion, also known as water-in-oil-in-water (w/o/w) [56].
2.2.2. Spray Drying Encapsulation
The spray drying technique has been used to encapsulate bioactive compounds in food ingredients. This technique consists of evaporating the solvent (generally water) rapidly, generating a capsule in which the compound of interest is almost instantly trapped [57,58]. The material used for encapsulation by this method, as well as the operating conditions, are of vital importance to achieve a correct microencapsulation and the adequate protection of the active principle. In spray drying, a liquid mixture is sprayed into a stream of a gas; normally air, although an inert gas, such as nitrogen, can also be used (Figure 4).
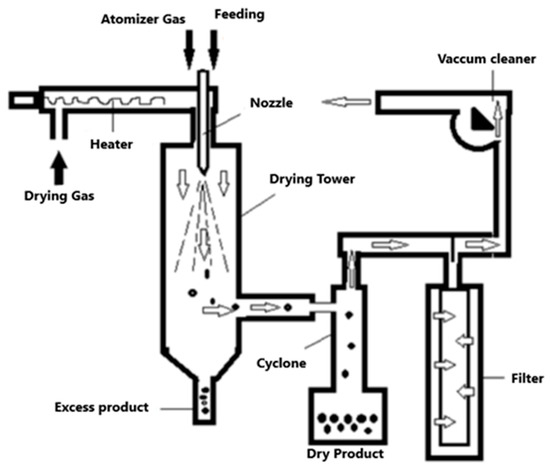
Figure 4.
Spray drying encapsulation process.
In this method, the liquid to be dried (encapsulating an active ingredient to be encapsulated) is flowed to a nozzle where it is atomized into the drying chamber, in such a way that the liquid increases the surface area in contact with hot air, which allows its complete drying in a short period of time. In this way, the sensible heat of the supplied hot air is used for the evaporation of the solvent, turning the droplets into a fine powder in less than a second [57,59]. In general, the spray drying process reduces the water content and the aqueous activity in the dehydrated material, helping the microbiological stability of the food, preventing chemical or biological degradation, reducing storage costs, and also obtaining a product with specific and desired properties [60,61].
This process is considered the most common and economical technique for producing microencapsulated food materials, for example, when compared with lyophilization. Spray drying is between 30 and 50 times cheaper [45,62], even knowing that this technique is considered low energy efficiency, since it impossible to use all the energy available as heat in the drying chamber [63,64]. The main encapsulating materials that have been reported include acacia gum, maltodextrins, modified starch, and their mixtures [65]. To a lesser extent, polysaccharides (alginate, carboxymethylcellulose, guar gum) and proteins (serum milk, soybeans, sodium caseinate) have also been used. In these cases, costs are higher due to their low solubility and the need to evaporate a greater quantity of liquid to produce the same quantity of dry matter [19,65]. The main advantages of spray drying include high level of homogeneity in production, continuous and controlled process, as well as ease to operate and high performance.
3. Results
After the de-analysis of the selected articles, considering the general aspects of each paper and carrying out a classification related to aspects such as country, year, and number of publications, some graphics were built. Additionally, the types of wall materials employed in spray drying microencapsulation, applications, and journals, were separated aiming to visualize all the information in an easier way. Finally, patent analysis related to spray drying encapsulation of essential oils, lipids, or compound lipids, were elaborated aiming to show some important industrial and academy discoveries, or novelties.
3.1. Relevance of Technology per Geographical Regions and Market Push
Figure 5 shows countries’ publications with respect to the most common encapsulation techniques used to protect essential oils, lipids, or compound lipids. The highest number of publications corresponds to Brazil (16 publications). The Latin American food encapsulation market is driven by factors such as the growing popularity of exotic flavors, cuisines and gourmet varieties, changing dietary habits, rising importance of the preservation of food products, and the growing speed and agility in new product innovation [66].
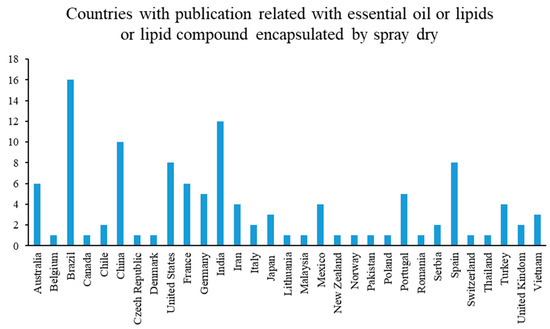
Figure 5.
Countries and publications.
Additionally, the Latin American food encapsulation market was worth USD 4.46 billion in 2021 and estimated to reach USD 6.09 billion by the end of 2026, with a growing potential of 6.43%, where Brazil, Argentina, and Chile are the main contributors [66]. Worldwide, 10 essential oils represent 85% of the market: orange, lemon, mint, java-citronella, cedar, eucalyptus, spice with citral such as may chang and lemongrass, lavender, lavandins, and pines. Brazil is located as the fourth largest exporter of oils essential [67].
In the year 2020, Brazil has been reported as the country with the greatest export potential in Latin America of a great variety of products, among which are: orange essential oils; lemon essential oils; citrus essential oils; and essential oils concentrated in fats and fixed oils [68]. The food encapsulation market is estimated to be valued at USD 9.9 billion in 2020 and is projected to grow at a CAGR (Compound Annual Growth Rate) of 7.5%, recording a value of USD 14.1 billion by 2025 [69].
India and China are the next countries with the most active research in this area. In the case of both countries the highest number of publication (12 and 10 publications, respectively,) could be due to the fact that they are the most populated countries in the world with 18% and 19%, respectively [70]. Additionally, in both India and China there has been a millenary tradition in the production of essential oils. These two countries are among the main exporters of essential oils worldwide. At 24.20 million tons (mt) in 2016–17, and an estimated 23.95 mt in 2017–2018, India’s edible oil consumption ranks number two globally, behind China (35 mt). Seventy percent (14 mt) of this demand is satisfied by imports, composed mainly of palm oil (9.5 mt), soybean (2.99 mt), and sunflower oil (1.54 mt) [71].
Additionally, initiatives to develop collaborations between universities from different countries and institutions have been developed, such as Centro de Educação Superior da Região Sul (Brazil), Centro Tecnológico de la Carne de Galicia (Spain), Nutrition and Food Science Area, Preventive Medicine and Public Health, Food Sciences, Toxicology and Forensic Medicine Department (Spain), Universidade Federal de Santa Maria (Brazil), and the Department of Animal Source Food Technology (Serbia); as well as the increasing partnership between universities or private sector from the European Union with Latin America countries and United States.
3.2. Years and Publications
Figure 6 shows the trends of spray drying encapsulations for essential oils, lipids, or compound lipid publications, according to a year in the last 20 years. The figure is evidence of a growing behavior of spray drying encapsulations for essential oils, lipids, or compound lipids along time between 2000 and 2021. The number of publications related to “microencapsulation” (February 2021), reaches 13,979 and, of these, 382 are simultaneously related to “encapsulation”, “spray drying”, “essential oil”, “lipids”, or “compound lipid”. From 2000 to 2010 there were only 49 papers published, while between 2011 and 2020, 333 papers were generated (source: www.scopus.com, 18 February 2021). The rise of publication numbers from 2010 could be due to the application of new wall materials, as well as new ingredients with possible health benefits [72].
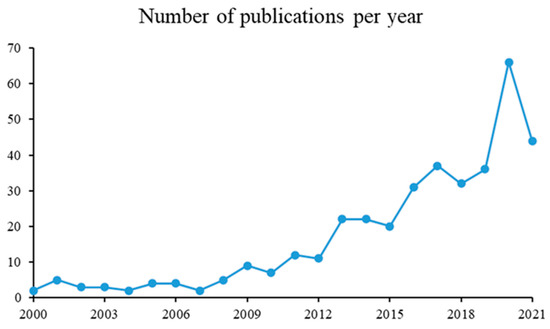
Figure 6.
Number of publications per year.
The years 2015 and 2018 showed the greatest growth in the number of patents, with China leading the domains of origin, inventors, and owners of microencapsulation technology. The largest number of applications of microcapsules were observed in the food industry, and the foods containing microencapsulated oils were powdered seasonings, dairy products, rice flour, nutritional formulae, pasta, nutritional supplements, and bread. The increase in the oxidative stabilities of oils was the most cited objective to microencapsulate oils [73].
Furthermore, during the year of the COVID-19 pandemic (2020), the number of publications doubled, compared to 2019.
3.3. Journals
Microencapsulation has been utilized to enhance the discharge properties of medications, to cover unpleasant taste, and to give specific tranquilize conveyance in pharmaceutical enterprises [74]. Additionally, it has been used in various industries, including the make-up, pharmaceutical, agrochemical, and food business, among others.
The increasing interest in spray drying encapsulations for essential oils, lipids, or compound lipids, and therefore creating a large number of indexed journals, is shown in Figure 7 and Figure 8, and are related to economic benefits and increasing research in these fields [75]. Figure 7 and Figure 8 evidence some journals related to the food science of pharmacology, which have published papers associated with spray drying encapsulation and essential oils, lipids, or compound lipids. The total number of journals was divided into two groups, the journals with a Q1 classification (Figure 7) and the journals with a Q2, Q3, and Q4 classification (Figure 8).
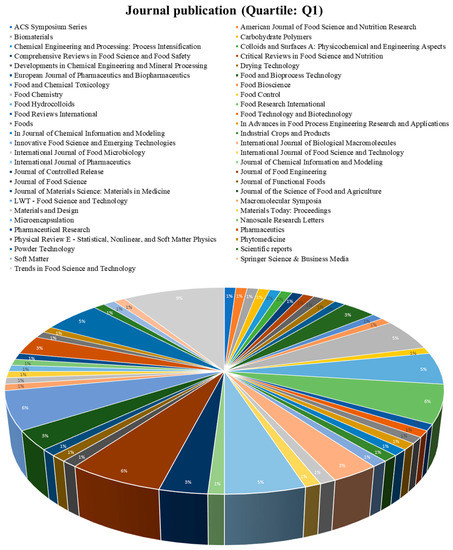
Figure 7.
Journal publication (Quartile: Q1).
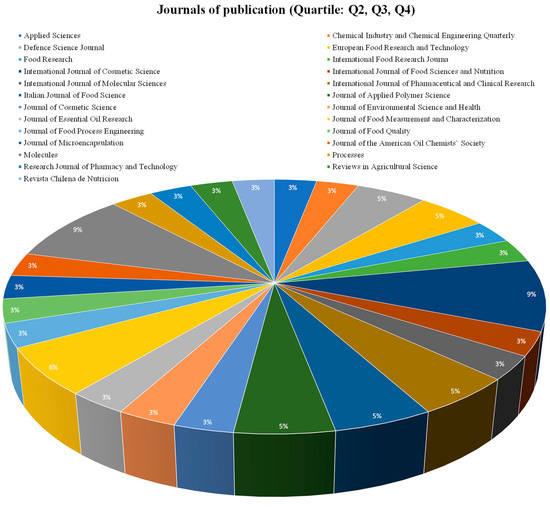
Figure 8.
Journal publication (Quartile: Q2, Q3, Q4).
3.3.1. Q1 Articles
From this group, the journal “Trends in Food Science and Technology “is the one with the most publications related to this review topic (9%) from the United Kingdom, associated with food science and biotechnology with a Q1 quartile [76].
Additionally, the second journal with the most publications is “Food Research International” (6%) from the United Kingdom, with the food science topic a Q1 quartile [77].
Other journals, such as “Macromolecular Symposia” (5%), “Journal of Food Engineering” (5%), and “Food Chemistry” (5%), also evidence that the topic of the present paper is the subject of wide research.
3.3.2. Q2, Q3, and Q4 Articles
Among this second group, the percentage of participation from each journal has more uniformity. The first journal with more publications around this topic is “International Journal of Cosmetic Science” (9.9%) from the United Kingdom, with chemistry, dermatology, and pharmaceutical science, with a Q2 quartile and aging, colloid and surface chemistry, and drug discovery, with a Q3 quartile [78].
The trend of these papers are related to characteristics such as spray drying as the main encapsulation technique, main wall materials employed and industrial applications. Hence, the largest distribution of publications is not grouped into a single journal, but 65% of the journals are in the Q1 quartile, and 35% of the journal of this research are Q2, Q3, and Q4, indicating the quality and validity of research found during this review and the numerous countries, research groups, and industrial interest focusing on new wall material and the improved protection of essential oils, lipids, and compound lipids [79].
4. Discussion
4.1. Substrates Used in Microencapsulation
There are a huge variety of materials used for microencapsulation; however, all of them must meet certain basic characteristics such as: promote release in accordance with the objectives set for the bioactive compound; protect the bioactive compound from environmental factors such as light, heat, and oxygen, among others; do not react with the bioactive compound; decrease the diffusion of the bioactive compound to the external environment; be chemically compatible; be easy to handle; and be waterproof, stable, and flexible [11,22,28,80,81]. Encapsulated materials can be natural, semi-synthetic, or synthetic polymers [19,82]. Among the most used encapsulating agents for lipids, lipophilic compounds, or essential oils are gum arabic, barley protein, carrageenan, chitosan, maltodextrin, methylcellulose, skim milk powder, and whey protein (Table 1). Additionally, Table 1 shows some common encapsulating agents used in microencapsulation processes, as well as some of the common combinations among the different encapsulating agents used for protecting lipid components.

Table 1.
Common encapsulating agents used in lipids (years 2000–2015).
4.1.1. Natural Polymers
Natural polymers are biodegradable, abundant, and generally non-toxic; they are plant extracts, as well as n-OSA starch and barley protein (Table 1). Natural materials: this category is made up of polysaccharides, proteins, and lipids. Among the most widely used polysaccharides are carrageenan and gum Arabic; examples of proteins are collagen and gelatin; examples of lipids are lecithin and heparin. The most widely used natural materials are polysaccharides, including chitosan and alginate. Chitosan is a polysaccharide with β (1–4) bonds, which can be obtained by alkalinization, and the deacetylation of chitin, naturally found in the exoskeleton of crustaceous. Some advantages of chitosan are its low cost, biodegradability, non-toxicity, and biocompatibility [107].
Alginate is derived from brown algae and is a polyanionic copolymer of D-mannuronic acid and L-guluronic acid linked by b (1–4) bonds. Alginate is widely used as an encapsulating agent for the food industry and the pharmaceutical industry due to its low immunogenicity and biocompatibility [82].
4.1.2. Synthetic Polymers
Synthetic polymers are available in various compositions and various molecules. One of the main advantages of these polymers is that their properties, optimization, and modulation are easy, since they are available in a wide range of compositions and molecular configurations. A disadvantage of these materials is that synthetic polymers show a lack of biocompatibility [82] and are generally biodegradability. Some examples of synthetic polymers are aliphatic polyesters, such as poly (lactic acid) and copolymers of lactic and glycolic acids (e.g., PLGA) [55].
As of 2015, two trends are observed regarding encapsulating materials: the first trend focuses on improving the encapsulation efficiency of conventional materials [33,108,109], and the second trend focuses on the use of novel encapsulating materials [42,59,110]. Both trends can be observed in Table 2, Table 3 and Table 4.

Table 2.
Vitamin A encapsulation by spray drying: recent articles.

Table 3.
Vitamin D encapsulation by spray drying: recent articles.

Table 4.
Vitamin E Encapsulation by spray drying: recent articles.
4.2. Active Ingredients
4.2.1. Liposoluble Vitamins
Fat-soluble vitamins (A, D, E, and K) have many health benefits. These vitamins are found mainly in food. In pharmacology, these vitamins have been used to treat various diseases, among which are mainly skin diseases, various types of cancer, and the reduction of oxidative stress. These molecules are sensitive to oxidation, so encapsulation could constitute an appropriate means to preserve their properties during storage and improve their physiological potencies. It is interesting that, even when the encapsulation of this kind of vitamin is an important topic, there in not enough. Considering the importance of these types of vitamins and their sensitivity to various factors, there is not abundant information regarding the use of the encapsulation technique by dry spray for the protection of fat-soluble proteins [110].
In 2010, the paper “New trends in encapsulation of liposoluble vitamins” mentioned the spray drying technique as a new trend, at that time. This study also highlighted the need for future research and optimization in the field of encapsulation. Although this article covers all fat-soluble vitamins, including carotenoids, only two articles are cited evidencing the use of spray drying for the protection of fat-soluble vitamins: the first article “Modified polyvinyl alcohol for encapsulation of all-trans-retinoic acid in polymeric micelles” in 2005; the second article, “Stability of spray-dried encapsulated carrot” in 1995 [139].
Vitamin A
Vitamin A is a hydrophobic compound that can aid in the formation and maintenance of healthy teeth, soft and bone tissues, mucous membranes, and skin [139]. Additionally, it is well known that vitamin A is poorly soluble in water due to its low polarity [140]. Vitamin A is also highly sensitive to environmental factors, such as humidity, temperature, light, traces of metals, and heat, among others [141]. It is possible to increase the stability of vitamin A as well as its dispersibility encapsulation using agents with specific physical and chemical properties. Additionally, it is necessary to use the appropriate encapsulation method. This can also benefit vitamin A in determining controlled release kinetics, allowing this vitamin to reach certain sites where absorption is optimized. In this way, both bioavailability and bioaccessibility in the human body are improved [25].
Microencapsulation of carotenoids (vitamin A) has been carried out by various techniques such as coacervation [25,142], and emulsion systems [143]; however, spray drying is the most widely used technique [112,144]. Additionally, biopolymers have been reported as encapsulating agents [97]. One of the carotenoid encapsulating agents that has shown advantages in terms of emulsion stability, low viscosity, and good solubility, is Arabic gum [145].
Table 2 summarizes the main advances in the use of encapsulation by spray drying for the protection of vitamin A. During the review of encapsulating agents, no new materials have been found, however, investigations are evidenced that seek to improve the efficiency of encapsulation [111,114], as well as mixtures of various encapsulating agents [111].
Vitamin D
Vitamin D participates in the calcium-phosphorus homeostasis of the body. Its sustained deficiency causes rickets in children and osteomalacia in adults. However, in addition to this, in recent years it has been observed that vitamin D influences a significant number of physiological processes, especially in relation to the immune system. Thus, various diseases such as cancer, multiple sclerosis, inflammatory bowel disease, arterial hypertension, and cardiovascular disease, have been associated with low levels of vitamin D [146]. Two relevant forms of vitamin D are D2 (ergochole-calciferol) and D3 [147].
The sensitivity of vitamin D towards heat, pH, light, oxidation, and hydrolysis, is a big concern during any formulation development [121]. Additionally, the photosensitivity of vitamin D3 in fortified milk during storage resulting in off flavor development. The enrichment of foods with vitamin D has the main role to produce healthy foods in terms of public health [50].
Microencapsulation by dry spray generates particles that exceed five microns, however, there is a trend marked by nanotechnology leading to the modification of the spray drying system towards the generation of nanoparticles obtained by this technique [148].
The article “Vitamin D microencapsulation and fortification: Trends and technologies” written in 2019 shows the little progress in the encapsulation of vitamin D using the spray drying technique. Even though spray drying uses high temperatures, something undesirable for heat-sensitive compounds, this technique offers great flexibility both in wall materials as well as in the proper handling of equipment variables that would allow the preservation of bioactive compounds, sensitive to heat, and that you want to protect. It is evident that the full potential of spray drying is still fully unexplored for vitamin D encapsulation (Table 3).
Recently, articles on this topic have attracted attention, showing advances in the encapsulation of vitamin D by spray drying. Among the most recent news are: saccharomyces cerevisiae yeast used as a vitamin D capsulant [125]; Casein micelles loaded with vitamin D2 [123]; functional yogurt powder fortified with nanoliposomal vitamin D through spray drying [50]; and the co-encapsulation of vitamins B12 and D3 using spray drying [121].
Vitamin E
The term, vitamin E, describes a family of eight related fat-soluble molecules. Of these, alpha-tocopherol has the highest biological activity and is the most abundant in the human body. The name tocopherol derives from the Greek word tocos, which means birth, and pherein, which means to carry [149]. A sufficient intake of vitamin E (alpha-tocopherol) is important since it works as an antioxidant, protecting cells, tissues, and organs against the harmful effects of free radicals, which are responsible for the aging process. These can cause a series of conditions, such as: coronary heart disease; cancer or inflammations; inhibits harmful blood clotting, which can block blood flow; regulates the opening of blood vessels, important for blood to flow smoothly [150].
Due to its antioxidant action, vitamin E could protect against clouding of the lenses of the eyes (cataracts) and a progressive deterioration of the retina at the back of the eye (age-related macular degeneration). These two conditions tend to occur with aging, causing a loss of vision [151]. Clinical studies have shown that people with cancer often have low levels of vitamin E in their blood. In addition, population trials suggest that diets rich in antioxidants, including vitamin E, may be associated with a reduced risk of certain types of cancer [150].
Since researchers believe that oxidative stress contributes to the development of Alzheimer’s disease, antioxidants such as vitamin E may help prevent this condition. The fat-soluble vitamin can easily reach the brain and apply its antioxidant properties [152].
Many bioactive substances are lipophilic, such as vitamin E, showing a poor dispersibility in water; high susceptibility to light, heat, and oxygen; and relatively low bioavailability after ingestion. Encapsulating these substances in hydrophilic matrix can convert them to a water-dispersible form, protect them from external factors, mask their flavors, and enhance their bioavailability as well as applicability in food products (Fangmeier et al. 2019) (Table 4).
For vitamin E, it is possible to observe a high numbers or papers in which the spray drying technique is applied not only for general vitamin E, but for specific vitamin E compounds, too. It is interesting that the production of articles related to the use of dry spray to encapsulate vitamin E coincides with the general statistics (Figure 5) where, as of 2015, a high number of publications is evidenced.
There is a trend towards applied research, especially in pharmacology and nutraceutical. Among the most recent applications are: preparation and evaluation of orally disintegrating tablets [138]; the enhancement of oral bioavailability of vitamin E [136]; microcapsules packed in amber colored glass bottle with stability in vitamin E content [135]; the co-encapsulation of coenzyme Q10 and vitamin E [129,132]; and the controlled release of vitamin E from chitosan/sodium lauryl ether sulfate microcapsules [128,131].
4.2.2. Essential Oils
Essential oils (EO) can be extracted from different types and parts of plants (as medicinal, edible, and aromatic), including nutmeg, cloves, oregano, basil, celery seed, coriander, cinnamon, lemongrass, tarragon, thyme, marjoram, garlic, sage, and rosemary. Essential oils are generally recognized as safe (GRAS), thus allowing their use in food [153,154]. Essential oils are characterized by their pleasant and strong aromas, with antioxidant, antibacterial, and antifungal properties, which are conferred by their content of phenolic and aliphatic compounds [155]. Essential oils contain main components which are found in relatively high concentrations (20–70%), some of these main compounds are carvacrol and thymol (oregano), linalol (coriander), α- and β-thujone and camphor (Artemisia herba-alba), α-phelandrene (dill), menthol, and mentone (mint). Low molecular weight essential oils have become a very important group that includes aromatic components, terpenoids and terpenes, as well as aliphatic components [156].
Recently, the article “Green biopolymers from by-products as wall materials for spray drying microencapsulation of phytochemicals” summarize the growing trend towards the use of new encapsulating materials as well as orange waste fiber, carrot fiber, inulin, and pectin, among others. This article is focused in the encapsulation of phytochemicals by spray drying, however, these green biopolymers have been used for the encapsulation of essential oil, lipids or compound lipids, with growing acceptance [45,110].
On the other hand, several review articles were analyzed, with particular attention to two recent review articles focused on the encapsulation of essential oils by spray drying: “Essential oils microencapsulated obtained by spray drying: a review” [155], and “Microencapsulation of Essential Oils by Spray-Drying and Influencing Factors” [28]. Both documents show the use and importance of spray drying, agreeing that the main fields of application of spray drying microencapsulation are food and pharmacology with growth in some fields such as textiles, cosmetics, and packaging. However, these review articles do not include compound lipids and fat-soluble vitamins that are of great interest today.
In food applications, they are used due to their antimicrobial, insecticidal, and antifungal characteristics. On the other hand, in the pharmaceutical industry, health benefits are sought in various diseases such as cancer [157,158,159], stress, antisicosis [160], obesity [161,162], diabetes, antidiabetic, hypolipidemic, anti-inflammatory [163], tuberculosis [164], the polyneuropathy of hereditary transthyretin [165], and acne [166].
Consumers in some industrial sectors have become increasingly demanding regarding the type and quality of the products offered. Among the industry sectors that have implemented innovative technologies are the personal care, perfumery, and cosmetic industries. In order to meet the demands of consumers, effective, natural, and safe products have been created, focused on improving health, contributing to well-being, and highlighting beauty [167,168]. It is possible to find a large number of products that combine new delivery or delivery systems as well as natural and environmentally friendly ingredients [169]. Essential oils have gained great strength in the cosmetic industry and in perfumery because they are natural ingredients proven health benefits and therefore can be used in creams, soaps, skin creams, and shampoos [170]. Since the impact of microencapsulation in the late 1980s, and thanks to the creation of fabrics with aromas developed by Kanebo Ltd., various microencapsulation techniques have been proposed that allow controlling the release of the active compounds of various formulations, whether they are food, cosmetic, or perfumery [171,172].
Some microencapsulation techniques are more used for certain bioactive compounds, for example, the microencapsulation of flavors is mainly carried out using spray drying or extrusion; however, microencapsulation techniques such as coacervation or lyophilization are also used. An essential aspect of the spray drying technique for industry is its easy scalability. The success of spray drying lies in the knowledge of the different variables of the process and the final properties of the microparticles. Some advantages of spray drying are the relative ease of obtaining microparticles in a relatively simple, continuous, and rapid manner. Additionally, it has low production and processing costs and the equipment is available. This technique has been especially used to microencapsulate volatile materials as essential oils, since they require a short contact time in the dryer [173,174].
4.2.3. Polyunsaturated Fatty Acids (PUFA)
The benefits of polyunsaturated fatty acids have been widely reported in relation to health benefits. PUFAs must be supplied through dietary supplements because the human body does not synthesize them; these are also called essential fatty acids (EFAs). Among the most recognized PUFAs are omega-3 (ω-3) and omega-6 (ω-6), reporting great benefits for health and their contribution in terms of energy and well-being. Among the benefits of PUFAs are their ability to minimize the risk of cardiovascular and neurodegenerative diseases as well as help with diabetes, arthritis, and even some types of cancer [175]. The main sources of PUFA are found on the seabed in algae and fish, while on land they can be found in some plant seeds. PUFAs can be short chain like α-linolenic acid and linoleic acid, or they can be long chain such as eicosapentaenoic acid, docosapentaenoic acid, docosahexaenoic acid, and arachidonic acid [176,177].
The spray drying of emulsions is one of the most used microencapsulation and drying technologies in the food and pharmaceutical industries because the process is flexible, economical, efficient, easy to scale-up, uses easily available equipment, and produces good quality powder. It has been extensively used in the encapsulation of fats, oils, flavors, and oil soluble ingredients (Table 5). The general process of spray drying involves the dispersion of a core material into a polymer solution, forming an emulsion or dispersion, the pumping of the feed solution/emulsion, and the atomization of the mixture and the dehydration of the atomized droplets to produce microcapsules [178].

Table 5.
Essential oils and PUFA Encapsulation by spray drying: recent articles.
4.2.4. Structured Lipids
Structured lipids are technological innovations based on the chemical composition of fats, with the aim of generating a great nutritional and technological impact in the future. Most lipids of natural origin have limited applications in their original state due to specific fatty acid and triacylglycerol composition [198]. Although human milk fat and cocoa butter are among the few exceptions with inherently excellent functionality, neither of them can meet the substantial demand in the food industry for infant nutrition or chocolate products because of certain social and/or environmental factors [198]. There are increasing health concerns and/or regulatory restrictions on the dietary ingestion of saturated fatty acids (SFAs) and trans fatty acids (TFAs). Trans fatty acids (TFAs) are unsaturated fatty acids that contain at least one nonconjugated double bond in the trans configuration, resulting in a straighter shape. Most commercially prepared foods contain TFAs. Recent data regarding TFA has implicated this lipid as being particularly deleterious to human health, contributing to problems such as cardiovascular diseases, systemic inflammation, dyslipidemia, endothelial dysfunction, and more recently hepatic and neurodegenerative diseases [199].
Lipid modification has been proved to be a powerful tool for addressing the above challenges, where structured lipids with improved function and/or nutrition can be obtained by changing the fatty acid profiles (for example, chain length, unsaturation level, and positional distribution) of natural fats and oils (Kadhum & Shamma, 2017; Osborn and Akoh, 2002).
Structured lipids (SLs) can be produced enzymatically or chemically via interesterification, acidolysis, and/or esterification processes from the conventional fats/oils in order to improve their nutritional and functional properties (Lu, Jin, Wang, and Wang, 2017; Zhao et al., 2014). Recently, the synthesis of SLs via enzymatic reactions is becoming a popular topic of lipid modification, which uses lipases as reaction catalysts. Lipid modifications enable to combine advantages of both medium chain FAs at sn-1,3 positions and long chain FAs at sn-2 position of one triacyglycerol molecule (MLM-type SLs) that makes lipids functional as they can be designed for the special health requirements of patients and meet the demands of consumers who desire functional food products with health-promoting and disease preventing properties (Akoh, Sellappan, Fomuso, and Yankah, 2002).
The chemical structures and molecular architectures of SLs define mainly their physicochemical properties and nutritional values, which are also affected by the processing conditions. The article “Synthesis, physicochemical properties, and health aspects of structured lipids: A review” explains in detail the obtaining, properties, uses and future trends of structured lipids [198]. However, even though these compounds are sensitive to various factors, the various existing or employed methods for the protection of this type of lipids are not shown.
Examples of commercially available structured lipids include the low- or zero-calorie food products such as olestra (Procter and Gambler, Cincinnati, OH, USA), salatrim (Nabisco, East Hanover, NJ, USA) and diacylglycerol (Kao Cooperation, Sumida city, Tokio, Japan) [200].
Structured lipids have applications mainly in health, aiming to treat or prevent diseases such as diabetes, cancer, and obesity, among others. In the last 20 years, there are few investigations focused on the protection of structured lipids using the spray drying technique; this may be due to the great difficulty that occurs during the spraying process because, normally, emulsions have high value viscosity causing plugging in the spraying process [201].
Although structured lipids have important value on their own, they have also been used as the transporters and protectors of compounds sensitive to various environmental factors, in combination with other encapsulating agents [201].
Some of the most relevant articles where spray drying is used for the protection of structured lipids are shown in Table 6.

Table 6.
Structured lipids Encapsulation by spray drying: recent articles.
The production of structured lipids is a growing topic of research. Due to the importance of structured lipids in health and the modifications that these can undergo due to external factors, it is necessary to protect them using an encapsulation technique. The table shows the main investigations carried out with the purpose of protecting structured lipids using the spray drying technique. In this sense, only seven papers were found. The encapsulating agent most used as wall material was isolated whey protein followed by maltodextrin.
The commonly used inlet temperature was 180 °C (Korma et al., 2019) or higher [202], however, lower temperatures also showed good results [201].
5. Conclusions and Future Perspectives
Microencapsulation is one of the most popular technologies in recent years due to its wide application in multiple industries, such as pharmaceuticals, food, health, nutrition, and agri-food, among others. Microencapsulation consists of the protection and isolation of a material of interest or biocomposite through the use of a coating material that isolates the biocomposite from environmental factors. Depending on various factors, such as the encapsulating agent, the agent to be encapsulated, and the microencapsulation technique, it is possible to obtain varied morphologies as well as to modify the performance of the microparticles. In pharmacology, microencapsulation is widely used in processes such as controlled release and prolonged release. In the case of the food industry, microencapsulation is used for the elaboration of functional foods protecting bioactive compounds, such as enzymes or vitamins. In medicine, it has been used for cell immobilization mainly for cell encapsulation for use in regenerative medicine and tissue engineering.
Each microencapsulation technique has particular characteristics that allow them to be applied more efficiently and effectively in certain areas and certain processes. The foregoing implies that each microencapsulation technique has both advantages and disadvantages since microencapsulation techniques differ in parameters such as particle size, encapsulating material used, particle morphology, release kinetics, stability and useful lifetime of the bioactive component, as well as ease of handling and production costs. Regarding the associated costs, it is of great importance to consider each microencapsulation technique used since the scaling processes or their simulations are vital before deciding to implement a certain technique.
Microencapsulation is widely used for the protection of compound lipids; however, despite the fact that many microencapsulation techniques have been used, spray drying is the most widely used encapsulation technique for the protection of this type of compound. Spray drying has been used to encapsulate essential oils, fat-soluble vitamins, and polyunsaturated fatty acids. Spray drying is a technique that is easy to implement, has good encapsulation performance, is inexpensive, and is easy to scale. Additionally, this technique is compatible with the use of a large number of encapsulating materials, including starches, maltodextrins, gums, and sugars.
In short, microencapsulation is a growing technique. Within encapsulation techniques, spray drying stands out as it offers wide advantages for various applications in different industries. However, there is the challenge of optimization in terms of the percentage of performance of this technique, as well as the great challenge of responding to the demands of consumers regarding the protection of bioactive compounds of lipid origin that respond to nutritional needs, food, and health.
Future trends in the field of microencapsulation will likely focused on: (i) the implications for the food industry, medicine, cosmetic, and textile industries based on different lipidic bioactive compounds aiming to create a synergistic effect; (ii) it is important to deepen the encapsulation of structured lipids due to their contributions to health, and (iii) the stability and bioavailability into the gastrointestinal system.
Author Contributions
D.M.S.-O.: formal analysis, writing—original draft, writing—review and editing. A.V.C.P.: review and editing. M.C.L.-J.: conceptualization, review and editing. A.L.V.: writing—review and editing. M.S.P.: conceptualization, review and editing, supervision. J.P.M.-G.: conceptualization, writing—review and editing, supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
We would like to acknowledge MinCiencias Colombia (Ministerio de Ciencia, Tecnología e Innovación) (convocatoria 848).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Goldberg, L. Functional Foods: Designer Foods, Pharmafoods, Nutraceuticals; Springer Science & Business Media: Cham, Switzerland, 2012. [Google Scholar]
- GlobeNewswire. Functional Food Market Size Worth Around USD 309 Bn by 2027 2020. Available online: https://www.globenewswire.com/news-release/2020/11/20/2130656/0/en/Functional-Food-Market-Size-Worth-Around-USD-309-Bn-by-2027.html?faodatalab=2020-11-20-1 (accessed on 25 March 2021).
- Vicentini, A.; Liberatore, L.; Mastrocola, D. Functional Foods: Trends and Development. Ital. J. Food Sci. 2016, 28, 338–352. [Google Scholar]
- Murúa-Pagola, B.; Beristain-Guevara, C.I.; Martínez-Bustos, F. Preparation of starch derivatives using reactive extrusion and evaluation of modified starches as shell materials for encapsulation of flavoring agents by spray drying. J. Food Eng. 2009, 91, 380. [Google Scholar] [CrossRef]
- McClements, D.J.; Rao, J. Food-Grade nanoemulsions: Formulation, fabrication, properties, performance, biological fate, and Potential Toxicity. Crit. Rev. Food Sci. Nutr. 2011, 51, 285–330. [Google Scholar] [CrossRef] [PubMed]
- Okuro, P.K.; Eustáquio de Matos, F.; Favaro-Trindade, C.S. Technological challenges for spray chilling encapsulation of functional food ingredients. Food Technol. Biotechnol. 2013, 51, 171–182. [Google Scholar]
- Azimova, S.S.; Glushenkova, A.I.; Vinogradova, V.I. Lipids, Lipophilic Components and Essential Oils from Plant Sources; Springer: London, UK, 2011. [Google Scholar]
- Müller, R.H.; Radtke, M.; Wissing, S.A. Nanostructured lipid matrices for improved microencapsulation of drugs. Int. J. Pharm. 2002, 242, 121–128. [Google Scholar] [CrossRef]
- Valenzuela, B.A.; Sanhueza, C.J. Estructuracion de lipidos y sustitutos de grasas, ¿lipidos del futuro? Rev. Chil. Nutr. 2008, 35, 394–405. [Google Scholar] [CrossRef]
- Mishra, M. Handbook of Encapsulation and Controlled Release. Available online: https://books.google.com/books?id=pY7wCgAAQBAJ&pgis=1 (accessed on 23 March 2021).
- Madene, A.; Jacquot, M.; Scher, J.; Desobry, S. Flavour encapsulation and controlled release—A review. Int. J. Food Sci. Technol. 2006, 41, 1–21. [Google Scholar] [CrossRef]
- Bah, M.G.; Bilal, H.M.; Wang, J. Fabrication and application of complex microcapsules: A review. Soft Matter. R. Soc. Chem. 2020, 16, 570–590. [Google Scholar]
- Vehring, R. Pharmaceutical particle engineering via spray drying. Pharm. Res. 2008, 25, 999–1022. [Google Scholar] [CrossRef]
- Hoyos-Leyva, J.D.; Bello-Pérez, L.A.; Alvarez-Ramirez, J.; Garcia, H.S. Microencapsulation using starch as wall material: A review. Food Rev. Int. 2018, 34, 148–161. [Google Scholar] [CrossRef]
- Ranjha, N.M.; Khan, H.; Naseem, S. Encapsulation and characterization of controlled release flurbiprofen loaded microspheres using beeswax as an encapsulating agent. J. Mater. Sci. Mater. Med. 2010, 21, 1621–1630. [Google Scholar] [CrossRef] [PubMed]
- Risch, S. Encapsulation: Overview of uses and techniques. ACS Symp. Ser. 1995, 2–7. [Google Scholar] [CrossRef]
- Gibbs, B.F.; Kermasha, S.; Alli, I.; Mulligan, C.N. Encapsulation in the food industry: A review. Int. J. Food Sci. Nutr. 1999, 50, 213–224. [Google Scholar] [PubMed]
- Arici, M.; Topbas, O.; Karavana, S.Y.; Ertan, G.; Sariisik, M.; Ozturk, C. Preparation of naproxen–ethyl cellulose microparticles by spray-drying technique and their application to textile materials. J. Microencapsul. 2014, 31, 654. [Google Scholar] [CrossRef] [PubMed]
- Samborska, K.; Boostani, S.; Geranpour, M.; Hosseini, H.; Dima, C.; Khoshnoudi-Nia, S.; Rostamabadi, H.; Falsafi, S.R.; Shaddel, R.; Akbari-Alavijeh, S.; et al. Green biopolymers from by-products as wall materials for spray drying microencapsulation of phytochemicals. Trends Food Sci. Technol. 2021, 108, 297–325. [Google Scholar] [CrossRef]
- Gimeno, M. An overview of the latest development of microencapsulation for agricultural products. J. Environ. Sci. Heal. Part B Pestic Food Contam. Agric. Wastes 1996, 31, 407–420. [Google Scholar] [CrossRef]
- Gbassi, G.K.; Vandamme, T. Probiotic encapsulation technology: From microencapsulation to release into the gut. Pharmaceutics 2012, 4, 149–163. [Google Scholar] [CrossRef]
- Dubey, R.; Shami, T.C.; Bhasker Rao, K.U. Microencapsulation Technology and Applications. Def. Sci. J. 2009, 59, 82–95. [Google Scholar]
- Krasaekoopt, W.; Bhandari, B.; Deeth, H. Evaluation of encapsulation techniques of probiotics for yoghurt. Int. Dairy J. 2003, 13, 3–13. [Google Scholar] [CrossRef]
- Chávarri, M.; Marañón, I.; Ares, R.; Ibáñez, F.C.; Marzo, F.; Villarán, C. Microencapsulation of a probiotic and prebiotic in alginate-chitosan capsules improves survival in simulated gastro-intestinal conditions. Int. J. Food Microbiol. 2010, 142, 185–189. [Google Scholar] [CrossRef]
- Gonçalves, A.; Estevinho, B.N.; Rocha, F. Microencapsulation of vitamin A: A review. Trends Food Sci. Technol. 2016, 51, 76–87. [Google Scholar] [CrossRef]
- Nelson, G. Application of microencapsulation in textiles. Int. J. Pharm. 2002, 242, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Marinković, S.Š.; Bezbradica, D.; Škundrić, P. Microencapsulation in the textile industry. Chem. Ind. Chem. Eng. Q. 2006, 12, 58–62. [Google Scholar] [CrossRef]
- Nguyen, T.T.T.; Le, T.V.A.; Dang, N.N.; Nguyen, D.C.; Nguyen, P.T.N.; Tran, T.T.; Nguyen, Q.V.; Bach, L.G.; Thuy Nguyen Pham, T.D. Microencapsulation of Essential Oils by Spray-Drying and Influencing Factors. J. Food Qual. 2021, 2021, 5525879. [Google Scholar] [CrossRef]
- Peanparkdee, M.; Iwamoto, S.; Yamauchi, R. Microencapsulation: A Review of Applications in the Food and Pharmaceutical Industries. Rev. Agric. Sci. 2016, 4, 56–65. [Google Scholar] [CrossRef]
- Miyazawa, K.; Yajima, I.; Kaneda, I.; Yanaki, T. Preparation of a new soft capsule for cosmetics. J. Cosmet. Sci. 2000, 51, 239–252. [Google Scholar]
- Suganya, V.; Anuradha, V. Microencapsulation and Nanoencapsulation: A Review. Int. J. Pharm. Clin. Res. 2017, 9, 233–239. [Google Scholar] [CrossRef]
- Tekin, R.; Bac, N.; Erdogmus, H. Microencapsulation of fragrance and natural volatile oils for application in cosmetics, and household cleaning products. Macromol. Symp. 2013, 333, 35–40. [Google Scholar] [CrossRef]
- Nguyen, P.; Vo, T.; Tran, T.; Le, T.; Mai, H.; Long, G. Encapsulation efficiency and thermal stability of lemongrass (Cymbopogon citratus) essential oil microencapsulated by the spray-drying process. Food Res. 2021, 5, 195–202. [Google Scholar] [CrossRef]
- Comunian, T.A.; Favaro-Trindade, C.S. Microencapsulation using biopolymers as an alternative to produce food enhanced with phytosterols and omega-3 fatty acids: A review. Food Hydrocoll. 2016, 61, 442. [Google Scholar] [CrossRef]
- Eratte, D.; Dowling, K.; Barrow, C.J.; Adhikari, B. Recent advances in the microencapsulation of omega-3 oil and probiotic bacteria through complex coacervation: A review. Trends Food Sci. Technol. 2018, 71, 121. [Google Scholar] [CrossRef]
- Kudryavtseva, V.; Boi, S.; Read, J.; Gould, D.; Szewczyk, P.K.; Stachewicz, U.; Kiryukhin, M.V.; Pastorino, L.; Sukhorukov, G.B. Micro-sized “pelmeni”—A universal microencapsulation approach overview. Mater. Des. 2021, 202, 109527. [Google Scholar] [CrossRef]
- Arenas-Jal, M.; Suñé-Negre, J.M.; García-Montoya, E. An overview of microencapsulation in the food industry: Opportunities, challenges, and innovations. Eur. Food Res. Technol. 2020, 246, 1371. [Google Scholar] [CrossRef]
- Antigo, J.L.D.; Stafussa, A.P.; de Cassia Bergamasco, R.; Madrona, G.S. Chia seed mucilage as a potential encapsulating agent of a natural food dye. J. Food Eng. 2020, 285, 110101. [Google Scholar] [CrossRef]
- Chiou, D.; Langrish, T.A.G. Development and characterisation of novel nutraceuticals with spray drying technology. J. Food Eng. 2007, 82, 84–91. [Google Scholar] [CrossRef]
- Jyothi, N.V.N.; Prasanna, P.M.; Sakarkar, S.N.; Prabha, K.S.; Ramaiah, P.S.; Srawan, G.Y. Microencapsulation techniques, factors influencing encapsulation efficiency. J. Microencapsul. 2010, 27, 187–197. [Google Scholar] [CrossRef]
- Fanger, G.O. Microencapsulation: A brief history and introduction. Microencapsulation 1974, 1–20. [Google Scholar] [CrossRef]
- Korma, S.A.; Wei, W.; Ali, A.; Abed, S.M.; Zheng, L.; Jin, Q.; Wang, X. Spray-dried novel structured lipids enriched with medium-and long-chain triacylglycerols encapsulated with different wall materials: Characterization and stability. Food Res. Int. 2019, 116, 538. [Google Scholar] [CrossRef]
- Kanakdande, D.; Bhosale, R.; Singhal, R.S. Stability of cumin oleoresin microencapsulated in different combination of gum arabic, maltodextrin and modified starch. Carbohydr. Polym. 2007, 67, 536–541. [Google Scholar] [CrossRef]
- Linke, A.; Weiss, J.; Kohlus, R. Oxidation rate of the non-encapsulated- and encapsulated oil and their contribution to the overall oxidation of microencapsulated fish oil particles. Food Res. Int. 2020, 127, 108705. [Google Scholar] [CrossRef]
- Santos, M.B.; Garcia-Rojas, E.E. Recent advances in the encapsulation of bioactive ingredients using galactomannans-based as delivery systems. Food Hydrocoll. 2021, 18, 106815. [Google Scholar] [CrossRef]
- Gupta, A.; Bohidar, H.B. Kinetics of phase separation in systems exhibiting simple coacervation. Phys. Rev. E Stat. Nonlinear Soft Matter. Phys. 2005, 72, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nori, M.P.; Favaro-Trindade, C.S.; Matias de Alencar, S.; Thomazini, M.; de Camargo Balieiro, J.C.; Contreras Castillo, C.J. Microencapsulation of propolis extract by complex coacervation. LWT Food. Sci. Technol. 2011, 44, 429. [Google Scholar] [CrossRef]
- Glomm, W.R.; Molesworth, P.P.; Sandru, E.M.; Truong, L.T.; Brunsvik, A.; Johnsen, H. Microencapsulation of peppermint oil by complex coacervation and subsequent spray drying using bovine serum albumin/gum acacia and an oxidized starch crosslinker. Appl. Sci. 2021, 11, 3956. [Google Scholar] [CrossRef]
- Akbarzadeh, A.; Rezaei-sadabady, R.; Davaran, S.; Joo, S.W.; Zarghami, N. Liposome: Classification, preparation, and applications. Nanoscale Res. Lett. 2013, 8, 1–9. [Google Scholar] [CrossRef]
- Jafari, S.M.; Vakili, S.; Dehnad, D. Production of a Functional Yogurt Powder Fortified with Nanoliposomal Vitamin D Through Spray Drying. Food Bioprocess Technol. Food Bioprocess Technol. 2019, 12, 1220–1231. [Google Scholar] [CrossRef]
- Link, K.C.; Schlünder, E.U. Fluidized bed spray granulation Investigation of the coating process on a single sphere. Chem. Eng. Process Process Intensif. 1997, 36, 443–457. [Google Scholar] [CrossRef]
- Bakry, A.M.; Abbas, S.; Ali, B.; Majeed, H.; Abouelwafa, M.Y.; Mousa, A.; Liang, L. Microencapsulation of Oils: A Comprehensive Review of Benefits, Techniques, and Applications. Compr. Rev. Food Sci. Food Saf. 2016, 15, 143–182. [Google Scholar] [CrossRef]
- Dewettinck, K.; Huyghebaert, A. Fluidized bed coating in food technology. Trends Food Sci. Technol. 1999, 10, 163–168. [Google Scholar] [CrossRef]
- Boury, F.; Marchais, H.; Benoit, J.P.; Proust, J.E. Surface characterization of poly(α-hydroxy acid) microspheres prepared by a solvent evaporation/extraction process. Biomaterials 1997, 18, 125–136. [Google Scholar] [CrossRef]
- Freitas, S.; Merkle, H.P.; Gander, B. Microencapsulation by solvent extraction/evaporation: Reviewing the state of the art of microsphere preparation process technology. J. Control Release 2005, 102, 313–332. [Google Scholar] [CrossRef] [PubMed]
- Ratner, B.D.; Hoffman, A.S.; Schoen, F.J.; Lemons, J.E. Biomaterials Science: An Introduction to Materials in Medicine; Academic Press: Cambridge, MA, USA, 2004; pp. 162–164. [Google Scholar]
- Gharsallaoui, A.; Roudaut, G.; Chambin, O.; Voilley, A.; Saurel, R. Applications of spray-drying in microencapsulation of food ingredients: An overview. Food Res. Int. 2007, 40, 1107–1121. [Google Scholar] [CrossRef]
- Lucas, J.; Ralaivao, M.; Estevinho, B.N.; Rocha, F. A new approach for the microencapsulation of curcumin by a spray drying method, in order to value food products. Powder Technol. 2020, 362, 428. [Google Scholar] [CrossRef]
- Methaapanon, R.; Kornbongkotmas, S.; Ataboonwongse, C.; Soottitantawat, A. Microencapsulation of n-octadecane and methyl palmitate phase change materials in silica by spray drying process. Powder Technol. 2020, 361, 910. [Google Scholar] [CrossRef]
- Bhandari, B.; Bansal, N.; Zhang, M.; Schuck, P. Handbook of Food Powders, Processes and Properties; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Ozdemir, N.; Bayrak, A.; Tat, T.; Altay, F.; Kiralan, M.; Kurt, A. Microencapsulation of basil essential oil: Utilization of gum arabic/whey protein isolate/maltodextrin combinations for encapsulation efficiency and in vitro release. J. Food Meas. Charact. 2021, 15, 1865–1876. [Google Scholar] [CrossRef]
- Desobry Sa Netto, F.M.; Labuza, T.P. Comparison of Spray-drying, Drum-drying and Freeze-drying for b-Carotene Encapsulation and Preservation. J. Food Sci. 1997, 62, 1158–1162. [Google Scholar] [CrossRef]
- Nedović, V.; Kalušević, A.; Manojlović, V.; Petrović, T.; Bugarski, B. Encapsulation Systems in the Food Industry. In Advances in Food Process Engineering Research and Applications; Springer: Boston, MA, USA, 2013; Volume 229, Available online: https://books.google.com/books?id=rxO4BAAAQBAJ&pgis=1 (accessed on 12 June 2021).
- Zhang, C.; Ada Khoo, S.L.; Chen, X.D.; Quek, S.Y. Microencapsulation of fermented noni juice via micro-fluidic-jet spray drying: Evaluation of powder properties and functionalities. Powder Technol. 2020, 361, 995. [Google Scholar] [CrossRef]
- Gouin, S. Microencapsulation: Industrial appraisal of existing technologies and trends. Trends Food Sci. Technol. 2004, 15, 330–347. [Google Scholar] [CrossRef]
- Market Data Forecast. Latin America Food Encapsulation Market | 2021–2026 | Mexico, Brazil, Argentina, Chile. Available online: https://www.marketdataforecast.com/market-reports/latin-america-food-encapsulation-market/request-sample (accessed on 21 August 2021).
- Forero-Peñuela, L.Y.; Forero-Peñuela, Z.L.; Deschamps, C.; Alves-Porsse, A. Análisis exploratorio de las exportaciones de aceites esenciales en Brasil: Evidencia desde 2000 hasta. Idesia 2017. [CrossRef]
- Naciones Únicas—CEPAL. Perspectivas del Comercio Internacional de América Latina y el Caribe. 2020. Available online: https://www.cepal.org/es/publicaciones/46613-perspectivas-comercio-internacional-america-latina-caribe-2020-la-integracion (accessed on 24 July 2021).
- Bussineswire. Global Food Encapsulation Market (2020 to 2025)—Players Profiled Include Friesl and Campina, DSM & Ingredion Incorporated Among Others—ResearchAndMarkets.com. 2021. Available online: https://www.businesswire.com/news/home/20200407005388/en (accessed on 24 July 2021).
- Naciones Unidas. DESAFÍOS GLOBALES Población. Nac. Unidas. 2021. Available online: https://www.un.org/es/global-issues/population (accessed on 24 August 2021).
- Abhishek Parameswaran. Olive Oil Times. 2018. Available online: https://www.oliveoiltimes.com/es/business/is-india-poised-for-an-edible-oils-revolution/62877 (accessed on 24 August 2021).
- Poshadri, A.; Aparna, K. Microencapsulation technology: A review. J. Res. Angrau. 2010, 38, 86–102. [Google Scholar]
- De Jesus Freitas, T.; Assunção, L.S.; de Lima Silva, V.; Oliveira, T.S.; Conceição, I.S.; Machado, B.A.S.; Nunes, I.L.; Otero, D.M.; Ribeiro, C.D.F. Prospective study on microencapsulation of oils and its application in foodstuffs. Recent Pat. Nanotechnol. 2021, 16, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, A.; Sood, M.; Singh, J.; Bandral, J.D.; Gupta, N.; Bhat, A. Microencapsulation and its applications in food industry. J. Pharmacogn. Phytochem. 2019, 8, 32. [Google Scholar]
- Mohammed, N.K.; Tan, C.P.; Manap, Y.A.; Muhialdin, B.J.; Hussin, A.S.M. Spray Drying for the Encapsulation of Oils—A Review. Molecules 2020, 25, 3873. [Google Scholar] [CrossRef] [PubMed]
- Scimago. Trends in Food Science and Technology Scimago. 2021. Available online: https://www.scimagojr.com/journalsearch.php?q=22475&tip=sid&clean=0 (accessed on 29 August 2021).
- Scimago. Food Research International. Scimago. 2021. Available online: https://www.scimagojr.com/journalsearch.php?q=23180&tip=sid&clean=0 (accessed on 29 August 2021).
- Scimago. International Journal of Cosmetic Science. Scimago. 2021. Available online: https://www.scimagojr.com/journalsearch.php?q=26568&tip=sid&clean=0 (accessed on 29 August 2021).
- Vijeth, S.; Heggannavar, G.B.; Kariduraganavar, M.Y. Encapsulating Wall Materials for Micro-/Nanocapsules; IntechOpen: London, UK, 2019. [Google Scholar]
- Xiao, Z.; Liu, W.; Zhu, G.; Zhou, R.; Niu, Y. A review of the preparation and application of flavour and essential oils microcapsules based on complex coacervation technology. J. Sci. Food Agric. 2014, 94, 1482–1494. [Google Scholar] [CrossRef] [PubMed]
- Mali Snehal, D.; Khochage Swapna, R.; Nitalikar Manoj, M.; Magdum Chandrakant, S. Microencapsulation: A review. Res. J. Pharm. Technol. 2013, 6, 954–961. [Google Scholar]
- Huang, H.J.; Chen, X.D.; Yuan, W.K. Microencapsulation based on emulsification for producing pharmaceutical products: A literature review. Dev. Chem. Eng. Miner. Process. 2006, 14, 515–544. [Google Scholar] [CrossRef]
- Wang, R.; Tian, Z.; Chen, L. A novel process for microencapsulation of fish oil with barley protein. Food Res. Int. 2011, 44, 2735–2741. [Google Scholar] [CrossRef]
- Scremin, F.R.; Veiga, R.S.; Silva-Buzanello, R.A.; Becker-Algeri, T.A.; Corso, M.P.; Torquato, A.S.; Bittencourt, P.R.S.; Flores, E.L.M.; Canan, C. Synthesis and characterization of protein microcapsules for eugenol storage. J. Therm. Anal. 2017, 131, 653–660. [Google Scholar] [CrossRef]
- Baik, M.-Y.; Suhendro, E.L.; Nawar, W.W.; McClements, D.; Decker, E.; Chinachoti, P. Effects of antioxidants and humidity on the oxidative stability of microencapsulated fish oil. J. Am. Oil Chem. Soc. 2004, 81, 355–360. [Google Scholar] [CrossRef]
- Morales-Medina, R.; Tamm, F.; Guadix, A.; Drusch, S. Functional and antioxidant properties of hydrolysates of sardine (S. pilchardus) and horse mackerel (T. mediterraneus) for the microencapsulation of fish oil by spray-drying. Food Chem. 2015, 194, 1208–1216. [Google Scholar] [CrossRef]
- Shaikh, J.; Bhosale, R.; Singhal, R. Microencapsulation of black pepper oleoresin. Food Chem. 2006, 94, 105–110. [Google Scholar] [CrossRef]
- Soottitantawat, A.; Bigeard, F.; Yoshii, H.; Furuta, T.; Ohkawara, M.; Linko, P. Influence of emulsion and powder size on the stability of encapsulated d-limonene by spray drying. Innov. Food Sci. Emerg. Technol. 2005, 6, 107–114. [Google Scholar] [CrossRef]
- Faria, A.F.; Mignone, R.A.; Montenegro, M.A.; Mercadante, A.Z.; Borsarelli, C.D. Characterization and Singlet Oxygen Quenching Capacity of Spray-Dried Microcapsules of Edible Biopolymers Containing Antioxidant Molecules. J. Agric. Food Chem. 2010, 58, 8004–8011. [Google Scholar] [CrossRef] [PubMed]
- de Barros Fernandes, R.V.; Borges, S.V.; Botrel, D.A. Gum arabic/starch/maltodextrin/inulin as wall materials on the microencapsulation of rosemary essential oil. Carbohydr. Polym. 2014, 101, 524–532. [Google Scholar] [CrossRef]
- Asensio, C.M.; Paredes, A.J.; Martin, M.P.; Allemandi, D.A.; Nepote, V.; Grosso, N.R. Antioxidant Stability Study of Oregano Essential Oil Microcapsules Prepared by Spray-Drying. J. Food Sci. 2017, 82, 2864–2872. [Google Scholar] [CrossRef]
- Luna-Guevara, J.; Ochoa-Velasco, C.; Hernández-Carranza, P.; Guerrero-Beltrán, J. Microencapsulation of walnut, peanut and pecan oils by spray drying. Food Struct. 2017, 12, 26–32. [Google Scholar] [CrossRef]
- Mehrad, B.; Shabanpour, B.; Jafari, S.M.; Pourashouri, P. Characterization of dried fish oil from menhaden encapsulated by spray drying. AACL Bioflux. 2015, 8, 57–69. [Google Scholar]
- Jafari, S.M.; Assadpoor, E.; Bhandari, B.; He, Y. Nano-particle encapsulation of fish oil by spray drying. Food Res. Int. 2008, 41, 172–183. [Google Scholar] [CrossRef]
- Davidov-Pardo, G.; Roccia, P.; Salgado, D.; Leon, A.; Pedroza-Islas, R. Utilization of Different Wall Materials to Microencapsulate Fish Oil Evaluation of its Behavior in Bread Products. Am. J. Food Technol. 2008, 3, 384–393. [Google Scholar] [CrossRef]
- Kolanowski, W.; Ziolkowski, M.; Weißbrodt, J.; Kunz, B.; Laufenberg, G. Microencapsulation of fish oil by spray drying--impact on oxidative stability. Part 1. Eur. Food Res. Technol. 2005, 222, 336–342. [Google Scholar] [CrossRef]
- Rascón, M.P.; Bonilla, E.; García, H.S.; Salgado, M.A.; González-Arnao, M.T.; Beristain, C.I. Tg and aw as criteria for the oxidative stability of spray-dried encapsulated paprika oleoresin. Eur. Food Res. Technol. 2015, 241, 217–225. [Google Scholar] [CrossRef]
- Serfert, Y.; Drusch, S.; Schmidt-Hansberg, B.; Kind, M.; Schwarz, K. Process engineering parameters and type of n-octenylsuccinate-derivatised starch affect oxidative stability of microencapsulated long chain polyunsaturated fatty acids. J. Food Eng. 2009, 95, 386–392. [Google Scholar] [CrossRef]
- Baranauskienė, R.; Venskutonis, P.R.; Dewettinck, K.; Verhé, R. Properties of oregano (Origanum vulgare L.), citronella (Cymbopogon nardus G.) and marjoram (Majorana hortensis L.) flavors encapsulated into milk protein-based matrices. Food Res. Int. 2006, 39, 413–425. [Google Scholar] [CrossRef]
- Aghbashlo, M.; Mobli, H.; Madadlou, A.; Rafiee, S. The correlation of wall material composition with flow characteristics and encapsulation behavior of fish oil emulsion. Food Res. Int. 2012, 49, 379–388. [Google Scholar] [CrossRef]
- Anwar, S.H.; Kunz, B. The influence of drying methods on the stabilization of fish oil microcapsules: Comparison of spray granulation, spray drying, and freeze drying. J. Food Eng. 2011, 105, 367–378. [Google Scholar] [CrossRef]
- Drusch, S.; Serfert, Y.; Scampicchio, M.; Schmidt-Hansberg, B.; Schwarz, K. Impact of Physicochemical Characteristics on the Oxidative Stability of Fish Oil Microencapsulated by Spray-Drying. J. Agric. Food Chem. 2007, 55, 11044–11051. [Google Scholar] [CrossRef]
- Polavarapu, S.; Oliver, C.M.; Ajlouni, S.; Augustin, M.A. Physicochemical characterisation and oxidative stability of fish oil and fish oil–extra virgin olive oil microencapsulated by sugar beet pectin. Food Chem. 2011, 127, 1694–1705. [Google Scholar] [CrossRef]
- Bae, E.K.; Lee, S.J. Microencapsulation of avocado oil by spray drying using whey protein and maltodextrin. J. Microencapsul. 2008, 25, 549–560. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, W.; Chen, X.D.; Selomulya, C. Micro-encapsulation and stabilization of DHA containing fish oil in protein-based emulsion through mono-disperse droplet spray dryer. J. Food Eng. 2016, 175, 74–84. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, Y.; Zhong, Q. Physical and antimicrobial properties of spray-dried zein–casein nanocapsules with co-encapsulated eugenol and thymol. J. Food Eng. 2015, 144, 93–102. [Google Scholar] [CrossRef]
- Estevinho, B.N.; Rocha, F.; Santos, L.; Alves, A. Microencapsulation with chitosan by spray drying for industry applications—A review. Trends Food. Sci. Technol. 2013, 31, 138–155. [Google Scholar] [CrossRef]
- de Melo Ramos, F.; Júnior, V.S.; Prata, A.S. Impact of vacuum spray drying on encapsulation of fish oil: Oxidative stability and encapsulation efficiency. Food Res. Int. 2021, 143, 110283. [Google Scholar] [CrossRef] [PubMed]
- Szabo, K.; Teleky, B.E.; Ranga, F.; Simon, E.; Pop, O.L.; Babalau-Fuss, V.; Kapsalis, N.; Vodnar, D.C. Bioaccessibility of microencapsulated carotenoids, recovered from tomato processing industrial by-products, using in vitro digestion model. LWT 2021, 152, 112285. [Google Scholar] [CrossRef]
- Aguiar, M.C.S.; das Graças Fernandes da Silva, M.F.; Fernandes, J.B.; Forim, M.R. Evaluation of the microencapsulation of orange essential oil in biopolymers by using a spray-drying process. Sci. Rep. 2020, 10, 11799. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.M.; Shahgol, M.; Estevinho, B.N.; Rocha, F. Microencapsulation of Vitamin A by spray-drying, using binary and ternary blends of gum arabic, starch and maltodextrin. Food Hydrocoll. 2020, 108, 106029. [Google Scholar] [CrossRef]
- Mujica-Álvarez, J.; Gil-Castell, O.; Barra, P.A.; Ribes-Greus, A.; Bustos, R.; Faccini, M.; Matiacevich, S. Encapsulation of Vitamins A and E as spray-dried additives for the feed industry. Molecules 2020, 25, 1357. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.-F.; Liu, X.; Zhou, M.-L.; Zhang, Q.-L. Preparation of vitamin A acetate microcapsules and process optimization. J. Chem. Eng. Chin. Univ. 2019, 33, 400–409. [Google Scholar]
- Li, X.; Feng, Y.; Ting, S.; Jiang, J.; Liu, Y. Effect of processing conditions on the physiochemical properties and nutrients retention of spray-dried microcapsules using mixed protein system. CYTA J. Food 2019, 17, 25–35. [Google Scholar] [CrossRef]
- Correâ-Filho, L.C.; Lourenço, M.M.; Moldaõ-Martins, M.; Alves, V.D. Microencapsulation of β-Carotene by Spray Drying: Effect of Wall Material Concentration and Drying Inlet Temperature. Int. J. Food Sci. 2019, 2019, 8914852. [Google Scholar] [CrossRef]
- O’Connor, G.; Krishnan, N.; Fagan-Murphy, A.; Cassidy, J.; O’Leary, S.; Robertson, B.D.; Keane, J.; O’Sullivan, M.P.; Cryan, S.A. Inhalable poly (lactic-co-glycolic acid)(PLGA) microparticles encapsulating all-trans-Retinoic acid (ATRA) as a host-directed, adjunctive treatment for Mycobacterium tuberculosis infection. Eur. J. Pharm. Biopharm. 2019, 134, 153–165. [Google Scholar] [CrossRef]
- Garcia, C.M.; Fernandez, M.; Lopez, O.D.; Castiñeira, M.; Martinez, B.; Nogueira, A.; Turiño, L. Microencapsulation of shark liver oil pool by spray drying. Lat. Am. Appl. Res. 2018, 48, 89–93. [Google Scholar] [CrossRef]
- Kosasih, E.A.; Warjito Imansyah, I.H.; Ruhyat, N. Use of a Double Condenser in a Dehumidifier with a Spray Dryer for Vitamin A Extraction in Tomato as a Heat-Sensitive Material. In AIP Conference Proceedings; AIP Publishing LLC: Anchorage, AK, USA, 2017. [Google Scholar]
- Gonçalves, A.; Estevinho, B.N.; Rocha, F. Design and characterization of controlled-release vitamin A microparticles prepared by a spray-drying process. Powder Technol. 2017, 305, 411–417. [Google Scholar] [CrossRef]
- Xie, Y.L.; Zhou, H.M.; Liang, X.H.; He, B.S.; Han, X.X. Study on the morphology, particle size and thermal properties of Vitamin A microencapsulated by starch octenylsucciniate. Agric. Sci. China 2010, 9, 1058–1064. [Google Scholar] [CrossRef]
- Bajaj, S.R.; Marathe, S.J.; Singhal, R.S. Co-encapsulation of vitamins B12 and D3 using spray drying: Wall material optimization, product characterization, and release kinetics. Food Chem. 2021, 335, 127642. [Google Scholar] [CrossRef]
- Shi, X.; Tan, T. Preparation of chitosan/ethylcellulose complex microcapsule and its application in controlled release of Vitamin D2. Biomaterials 2002, 23, 4469–4473. [Google Scholar] [CrossRef]
- Moeller, H.; Martin, D.; Schrader, K.; Hoffmann, W.; Chr, P. Spray- or freeze-drying of casein micelles loaded with vitamin D2: Studies on storage stability and in vitro digestibility. LWT Food Sci. Technol. 2018, 97, 87–93. [Google Scholar] [CrossRef]
- Mahdi Jafari, S.; Masoudi, S.; Bahrami, A. A Taguchi approach production of spray-dried whey powder enriched with nanoencapsulated vitamin D3. Dry Technol. 2019, 37, 2059. [Google Scholar] [CrossRef]
- Dadkhodazade, E.; Mohammadi, A.; Shojaee-Aliabadi, S.; Mortazavian, A.M.; Mirmoghtadaie, L.; Hosseini, S.M. Yeast Cell Microcapsules as a Novel Carrier for Cholecalciferol Encapsulation: Development, Characterization and Release Properties. Food Biophys. 2018, 13, 404–411. [Google Scholar] [CrossRef]
- Wei-hong, T.; Min-chang, G.; Zhen, X.; Jie, S. Pharmacological and pharmacokinetic studies with vitamin D-loaded nanoemulsions in asthma mode. Inflammation 2014, 37, 723–728. [Google Scholar] [CrossRef]
- Górska, A.; Szulc, K.; Ostrowska-Ligęza, E.; Wirkowska, M.; Bryś, J. The influence of trehalose–maltodextrin and lactose–maltodextrin matrices on thermal and sorption properties of spray-dried β-lactoglobulin–vitamin D 3 complexes. J. Therm. Anal. Calorim. 2013, 112, 429–436. [Google Scholar] [CrossRef]
- Budinčić, J.M.; Petrović, L.; Đekić, L.; Fraj, J.; Bučko, S.; Katona, J.; Spasojević, L. Study of vitamin E microencapsulation and controlled release from chitosan/sodium lauryl ether sulfate microcapsules. Carbohydr. Polym. 2021, 251, 116988. [Google Scholar] [CrossRef] [PubMed]
- Fu, N.; You, Y.J.; Quek, S.Y.; Wu, W.D.; Chen, X.D. Interplaying Effects of Wall and Core Materials on the Property and Functionality of Microparticles for Co-Encapsulation of Vitamin E with Coenzyme Q Food Bioprocess Technol. Food Bioprocess. Technol. 2020, 13, 705–721. [Google Scholar] [CrossRef]
- Surini, S.; Khotima, N.H. Stability study of ethylcellulose coated-tocotrienol microcapsules prepared by solvent evaporation and spray drying techniques. Int. J. Appl. Pharm. 2020, 12, 197–201. [Google Scholar] [CrossRef]
- Jiang, M.; Hong, Y.; Gu, Z.; Cheng, L.; Li, Z.; Li, C. Preparation of a starch-based carrier for oral delivery of Vitamin E to the small intestine. Food Hydrocoll. 2019, 91, 26–33. [Google Scholar] [CrossRef]
- Huang, E.; Quek, S.Y.; Fu, N.; Wu, W.D.; Chen, X.D. Co-encapsulation of coenzyme Q10 and vitamin E: A study of microcapsule formation and its relation to structure and functionalities using single droplet drying and micro-fluidic-jet spray drying. J. Food Eng. 2019, 247, 45–55. [Google Scholar] [CrossRef]
- Selamat, S.N.; Mohamad, S.N.H.; Muhamad, I.I.; Khairuddin, N.; Md Lazim, N.A. Characterization of Spray-Dried Palm Oil Vitamin E Concentrate. Arab. J. Sci. Eng. 2018, 43, 6165–6169. [Google Scholar] [CrossRef]
- Tarigan, J.B.; Kaban, J.; Zulmi, R. Microencapsulation of Vitamin E from palm fatty acid distillate with galactomannan and gum acacia using spray drying method. IOP Conf. Ser. Mater. Sci. Eng. 2018, 309, 012095. [Google Scholar] [CrossRef]
- Gangurde, A.B.; Ali, M.T.; Pawar, J.N.; Amin, P.D. Encapsulation of vitamin E acetate to convert oil to powder microcapsule using different starch derivatives. J. Pharm. Investig. 2017, 47, 559–574. [Google Scholar] [CrossRef]
- Parthasarathi, S.; Anandharamakrishnan, C. Enhancement of oral bioavailability of vitamin E by spray-freeze drying of whey protein microcapsules. Food Bioprod. Process. 2016, 100, 469–476. [Google Scholar]
- Hategekimana, J.; Masamba, K.G.; Ma, J.; Zhong, F. Encapsulation of vitamin E: Effect of physicochemical properties of wall material on retention and stability. Carbohydr. Polym. 2015, 124, 172–179. [Google Scholar] [CrossRef]
- Ikematsu, Y.; Uchida, S.; Namiki, N. Preparation and evaluation of orally disintegrating tablets containing vitamin e as a model fat-soluble drug. Chem. Pharm. Bull. 2015, 63, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Gonnet, M.; Lethuaut, L.; Boury, F. New trends in encapsulation of liposoluble vitamins. J. Control Release 2010, 146, 276. [Google Scholar] [CrossRef] [PubMed]
- National Coordinating Committee on Food and Nutrition. Vitamin A. Recommended Nutrition Intakes Malaysia. 2005; pp. 111–120. Available online: https://www.moh.gov.my/moh/images/gallery/rni/insert.pdf (accessed on 6 August 2021).
- Vilanova, N.; Solans, C. Vitamin A, Palmitate-B-cyclodextrin inclusion complexes: Characterization, protection and emulsification properties. Food Chem. 2015, 175, 529. [Google Scholar] [CrossRef] [PubMed]
- Wilson, N.; Shah, N.P. Microencapsulation of vitamins. Int. Food Res J. 2007, 14, 1–14. [Google Scholar]
- Yoshida, K.; Sekine, T.; Matsuzaki, F.; Yanaki, T.; Yamaguchi, M. Stability of vitamin A in oil-in-water-in-oil-type multiple emulsions. J. Am. Oil Chem. Soc. 1999, 76, 1–6. [Google Scholar] [CrossRef]
- Bartilucci, A.; Foss, N.E. Cyanocobalamin (Vitamin B12). J. Am. Pharm. Assoc. 1954, 43, 159. [Google Scholar] [CrossRef]
- Romo-Hualde, A.; Yetano-Cunchillos, A.I.; González-Ferrero, C.; Sáiz-Abajo, M.J.; González-Navarro, C.J. Supercritical fluid extraction and microencapsulation of bioactive compounds from red pepper (Capsicum annum L.) by-products. Food Chem. 2012, 133, 1045. [Google Scholar] [CrossRef]
- Zanuy, M.V.; Carranza, F.H. Metabolismo, fuentes endógenas y exógenas de vitamina D. Rev. Española Enfermedades Metabólicas Óseas 2007, 16, 63–70. [Google Scholar] [CrossRef]
- Forchielli, M.L.; Conti, M.; Patrono, D.; Mancini, R.; Pession, A.; Puggioli, C.; Bersani, G. Potential intake of vitamins “A” and “D” through branded intravenous lipid emulsions: Liquid Chromatography-Tandem Mass Spectrometry Analysis. Clin. Nutr. 2017, 36, 530. [Google Scholar] [CrossRef]
- Maurya, V.K.; Bashir, K.; Aggarwal, M. Vitamin D microencapsulation and fortification: Trends and technologies. J. Steroid Biochem. Mol. Biol. 2019, 196, 105489. [Google Scholar] [CrossRef] [PubMed]
- Meydani, S.N.; Blumberg, J.B. Vitamin E and the Immune Response. In Nutrient Modulation of the Immune Response; CRC Press: Boca Raton, FL, USA, 2020; pp. 223–238. [Google Scholar]
- Constantinou, C.; Charalambous, C.; Kanakis, D. Vitamin E and cancer: An update on the emerging role of γ and δ tocotrienols. Eur. J. Nutr. 2020, 59, 845–857. [Google Scholar] [CrossRef] [PubMed]
- Marino, P.F.; Rossi, G.C.M.; Campagna, G.; Capobianco, D.; Costagliola, C.; Qualicos Study Group. Effects of Citicoline, Homotaurine, and Vitamin E on Contrast Sensitivity and Visual-Related Quality of Life in Patients with Primary Open-Angle Glaucoma—A Preliminary Study. Molecules 2020, 25, 5614. [Google Scholar] [CrossRef] [PubMed]
- Casati, M.; Boccardi, V.; Ferri, E.; Bertagnoli, L.; Bastiani, P.; Ciccone, S.; Mansi, M.; Scamosci, M.; Rossi, P.D.; Mecocci, P.; et al. Vitamin E and Alzheimer’s disease: The mediating role of cellular aging. Aging Clin. Exp. Res. 2020, 32, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Palou, L.; Ali, A.; Fallik, E.; Romanazzi, G. GRAS, plant- and animal-derived compounds as alternatives to conventional fungicides for the >control of postharvest diseases of fresh horticultural produce. Postharvest. Biol. Technol. 2016, 122, 41. [Google Scholar] [CrossRef]
- Smith, R.L.; Cohen, S.M.; Doull, J.; Feron, V.J.; Goodman, J.I.; Marnett, L.J.; Portoghese, P.S.; Waddell, W.J.; Wagner, B.M.; Hall, R.; et al. A procedure for the safety evaluation of natural flavor complexes used as ingredients in food: Essential oils. Food Chem. Toxicol. 2005, 43, 345–363. [Google Scholar] [CrossRef]
- Veiga RDSDa Aparecida Da Silva-Buzanello, R.; Corso, M.P.; Canan, C. Essential oils microencapsulated obtained by spray drying: A review. J. Essent. Oil Res. 2019, 31, 457. [Google Scholar] [CrossRef]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial activity of terpenes and terpenoids present in essential oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Chraibi, S.; Rosière, R.; Larbanoix, L.; Gérard, P.; Hennia, I.; Laurent, S.; Vermeersch, M.; Amighi, K.; Wauthoz, N. The combination of an innovative dry powder for inhalation and a standard cisplatin-based chemotherapy in view of therapeutic intensification against lung tumours. Eur. J. Pharm. Biopharm. 2021, 164, 93–104. [Google Scholar] [CrossRef]
- Nafee, N.; Gaber, D.M.; Elzoghby, A.O.; Helmy, M.W.; Abdallah, O.Y. Promoted Antitumor Activity of Myricetin against Lung Carcinoma Via Nanoencapsulated Phospholipid Complex in Respirable Microparticles. Pharm. Res. 2020, 37, 1–24. [Google Scholar] [CrossRef]
- Khan, I.; Lau, K.; Bnyan, R.; Houacine, C.; Roberts, M. A Facile and Novel Approach to Manufacture Paclitaxel-Loaded Proliposome Tablet Formulations of Micro or Nano Vesicles for Nebulization. Pharm. Res. 2020, 37, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Meola, T.R.; Paxton, K.; Joyce, P.; Schultz, H.B.; Prestidge, C.A. The effect of drug ionization on lipid-based formulations for the oral delivery of anti-psychotics. ADMET DMPK 2020, 8, 437–451. [Google Scholar] [PubMed]
- Dening, T.J.; Joyce, P.; Kovalainen, M.; Gustafsson, H.; Prestidge, C.A. Spray Dried Smectite Clay Particles as a Novel Treatment against obesity. Pharm. Res. 2018, 36, 21. [Google Scholar] [CrossRef] [PubMed]
- Meola, T.R.; Abuhelwa, A.Y.; Joyce, P.; Clifton, P.; Prestidge, C.A. A safety, tolerability, and pharmacokinetic study of a novel simvastatin silica-lipid hybrid formulation in healthy male participants. Drug Deliv. Transl. Res. 2021, 11, 1261–1272. [Google Scholar] [CrossRef] [PubMed]
- Momordica, D. Antidiabetic, hypolipidemic, antioxidant and anti-inflammatory effects of Momordica charantia L. foliage extract. J. Pharm. Pharmacogn. Res. 2021, 9, 537–548. [Google Scholar]
- Rodrigues, S.; Cunha, L.; Kollan, J.; Neumann, P.R.; da Costa, A.M.R.; Dailey, L.A.; Grenha, A. Cytocompatibility and cellular interactions of chondroitin sulfate microparticles designed for inhaled tuberculosis treatment. Eur. J. Pharm. Biopharm. 2021, 163, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Dormenval, C.; Lokras, A.; Cano-Garcia, G.; Wadhwa, A.; Thanki, K.; Rose, F.; Thakur, A.; Franzyk, H.; Foged, C. Identification of factors of importance for spray drying of small interfering RNA-loaded lipidoid-polymer hybrid nanoparticles for inhalation. Pharm. Res. 2019, 36, 1–15. [Google Scholar] [CrossRef]
- Wijayadi, L.J.; Rusli, T.R. Encapsulated Lime Peel Essential Oil (Citrus hystrix) into Chitosan Nanoparticle: New entity to enhanced effectivity against Propionil bacterium acne in vitro. IOP Conf. Ser. Mater. Sci. Eng. 2020, 852, 012016. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64. [Google Scholar] [CrossRef]
- Vargas-Ramella, M.; Pateiro, M.; Barba, F.J.; Franco, D.; Campagnol, P.C.B.; Munekata, P.E.S.; Tomasevic, I.; Domínguez, R.; Lorenzo, J.M. Microencapsulation of healthier oils to enhance the physicochemical and nutritional properties of deer pâté. LWT 2020, 125, 109223. [Google Scholar] [CrossRef]
- Patravale, V.B.; Mandawgade, S.D. Novel cosmetic delivery systems: An application update. Int. J. Cosmet. Sci. 2008, 30, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Karuppayil, S.M. A status review on the medicinal properties of essential oils. Ind. Crops Prod. 2014, 62, 250. [Google Scholar] [CrossRef]
- Carvalho, I.T.; Estevinho, B.N.; Santos, L. Application of microencapsulated essential oils in cosmetic and personal healthcare products—A review. Int. J. Cosmet Sci. 2016, 38, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Shahidi Noghabi, M.; Molaveisi, M. Microencapsulation optimization of cinnamon essential oil in the matrices of gum Arabic, maltodextrin, and inulin by spray-drying using mixture design. J. Food Process Eng. 2020, 43, 1–13. [Google Scholar] [CrossRef]
- Adamiec, J.; Kalemba, D. Analysis of microencapsulation ability of essential oils during spray drying. Dry Technol. 2006, 24, 1127–1132. [Google Scholar] [CrossRef]
- Thuong Nhan, N.P.; Tan Thanh, V.; Huynh Cang, M.; Lam, T.D.; Cam Huong, N.; Hong Nhan, L.T.; Thanh Truc, T.; Tran, Q.T.; Bach, L.G. Microencapsulation of lemongrass (Cymbopogon citratus) essential oil via spray drying: Effects of feed emulsion parameters. Processes 2020, 8, 40. [Google Scholar] [CrossRef]
- Orsavova, J.; Misurcova, L.; Vavra Ambrozova, J.; Vicha, R.; Mlcek, J. Fatty acids composition of vegetable oils and its contribution to dietary energy intake and dependence of cardiovascular mortality on dietary intake of fatty acids. Int. J. Mol. Sci. 2015, 16, 12871–12890. [Google Scholar] [CrossRef]
- Rubio-Rodríguez, N.; Beltrán, S.; Jaime, I.; de Diego, S.M.; Sanz, M.T.; Carballido, J.R. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innov. Food Sci. Emerg. Technol. 2010, 11, 1. [Google Scholar] [CrossRef]
- Hashim, N.A.; Abdul Mudalip, S.K.; Sulaiman, S.Z.; Md Shaarani, S. Nutritional values and microencapsulation techniques of fish oil from different sources: A mini review. Mater. Today Proc. 2021, 42, 222–228. [Google Scholar] [CrossRef]
- Kaushik, P.; Dowling, K.; Barrow, C.J.; Adhikari, B. Microencapsulation of omega-3 fatty acids: A review of microencapsulation and characterization methods. J. Funct. Foods 2015, 19, 868. [Google Scholar] [CrossRef]
- Damerau, A.; Ogrodowska, D.; Banaszczyk, P.; Dajnowiec, F.; Tańska, M.; Linderborg, K.M. Baltic herring (Clupea harengus membras) oil encapsulation by spray drying using a rice and whey protein blend as a coating material. J. Food Eng. 2022, 314, 110769. [Google Scholar] [CrossRef]
- Copado, C.N.; Julio, L.M.; Diehl, B.W.K.; Ixtaina, V.Y.; Tomás, M.C. Multilayer microencapsulation of chia seed oil by spray-drying using electrostatic deposition technology. LWT 2021, 152, 112206. [Google Scholar] [CrossRef]
- Jia, C.; Huang, S.; Liu, R.; You, J.; Xiong, S.; Zhang, B.; Rong, J. Storage stability and in-vitro release behavior of microcapsules incorporating fish oil by spray drying. Colloids Surf. Physicochem. Eng. Asp. 2021, 628, 127234. [Google Scholar] [CrossRef]
- Yang, W.; Shan, Z. Application of wool keratin: An anti-ultraviolet wall material in spray drying. J. Food Sci. Technol. 2021, 58, 4235–4244. [Google Scholar] [CrossRef]
- Castejón, N.; Luna, P.; Señoráns, F.J. Microencapsulation by spray drying of omega-3 lipids extracted from oilseeds and microalgae: Effect on polyunsaturated fatty acid composition. LWT 2021, 148, 111789. [Google Scholar] [CrossRef]
- Cui, T.; Chen, C.; Jia, A.; Li, D.; Shi, Y.; Zhang, M.; Bai, X.; Liu, X.; Liu, C. Characterization and human microfold cell assay of fish oil microcapsules: Effect of spray drying and freeze-drying using konjac glucomannan (KGM)-soybean protein isolate (SPI) as wall materials. J. Funct. Foods 2021, 83, 104542. [Google Scholar] [CrossRef]
- Zhu, Y.; Peng, Y.; Wen, J.; Quek, S.Y. A Comparison of Microfluidic-Jet Spray Drying, Two-Fluid Nozzle Spray Drying, and Freeze-Drying for Co-Encapsulating β-Carotene, Lutein, Zeaxanthin, and Fish Oil. Foods 2021, 10, 1522. [Google Scholar] [CrossRef]
- Yu, F.; Xue, C.; Zhang, Z. Mechanical characterization of fish oil microcapsules by a micromanipulation technique. LWT 2021, 144, 111194. [Google Scholar] [CrossRef]
- Paulo, B.B.; Alvim, I.D.; Reineccius, G.; Prata, A.S. Barrier properties of spray-dried emulsions containing flavorings or unsaturated triglycerides. LWT 2021, 142, 111040. [Google Scholar] [CrossRef]
- Encina, C.; Giménez, B.; Márquez-Ruiz, G.; Holgado, F.; Vergara, C.; Romero-Hasler, P.; Soto-Bustamante, E.; Robert, P. Hydroxypropyl-inulin as a novel encapsulating agent of fish oil by conventional and water-free spray drying. Food Hydrocoll. 2021, 113, 106518. [Google Scholar] [CrossRef]
- Linke, A.; Weiss, J.; Kohlus, R. Impact of the oil load on the oxidation of microencapsulated oil powders. Food Chem. 2021, 341, 128153. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Yang, L.; McClements, D.J.; Wang, X.; Ye, J.; Liu, C. Spray drying and rehydration of macadamia oil-in-water emulsions: Impact of macadamia protein isolate to chitosan hydrochloride ratio. Food Chem. 2021, 342, 128380. [Google Scholar] [CrossRef] [PubMed]
- Karrar, E.; Mahdi, A.A.; Sheth, S.; Ahmed, I.A.M.; Manzoor, M.F.; Wei, W.; Wang, X. Effect of maltodextrin combination with gum arabic and whey protein isolate on the microencapsulation of gurum seed oil using a spray-drying method. Int. J. Biol. Macromol. 2021, 171, 208–216. [Google Scholar] [CrossRef] [PubMed]
- López-Miranda, S.; Berdejo, D.; Pagán, E.; García-Gonzalo, D.; Pagán, R. Modified cyclodextrin type and dehydration methods exert a significant effect on the antimicrobial activity of encapsulated carvacrol and thymol. J. Sci. Food Agric. 2021, 101, 3827–3835. [Google Scholar] [CrossRef]
- Garcia, L.G.S.; da Rocha, M.G.; Lima, L.R.; Cunha, A.P.; de Oliveira, J.S.; de Andrade, A.R.C.; Ricardo, N.M.P.S.; Pereira-Neto, W.A.; Sidrim, J.J.C.; Rocha, M.F.G.; et al. Essential oils encapsulated in chitosan microparticles against Candida albicans biofilms. Int. J. Biol. Macromol. 2021, 166, 621–632. [Google Scholar] [CrossRef]
- Umaña, M.; Wawrzyniak, P.; Rosselló, C.; Llavata, B.; Simal, S. Evaluation of the addition of artichoke by-products to O/W emulsions for oil microencapsulation by spray drying. LWT 2021, 151, 112146. [Google Scholar] [CrossRef]
- Fernandes, S.S.; Greque, L.; Santos, M.D.F.C.; Novais, L.M.R.; D’Oca, C.D.R.M.; Prentice, C.; Salas-Mellado, M.D.L.M. Effect of the spray drying conditions on the physicochemical and structural characteristics and the stability of chia oil microparticles. J. Appl. Polym. Sci. 2021, 138, 1–14. [Google Scholar] [CrossRef]
- Porras-Saavedra, J.; Pérez-Pérez, N.C.; Villalobos-Castillejos, F.; Alamilla-Beltrán, L.; Tovar-Benítez, T. Influence of Sechium edule starch on the physical and chemical properties of multicomponent microcapsules obtained by spray-drying. Food Biosci. 2021, 43, 101275. [Google Scholar] [CrossRef]
- Partheniadis, I.; Vergkizi, S.; Lazari, D.; Reppas, C.; Nikolakakis, I. Formulation, characterization and antimicrobial activity of tablets of essential oil prepared by compression of spray-dried powder. J. Drug Deliv. Sci. Technol. 2019, 50, 226. [Google Scholar] [CrossRef]
- Guo, Y.; Cai, Z.; Xie, Y.; Ma, A.; Zhang, H.; Rao, P.; Wang, Q. Synthesis, physicochemical properties, and health aspects of structured lipids: A review. Compr. Rev. Food Sci. Food Saf. 2020, 19, 759–800. [Google Scholar] [CrossRef]
- Longhi, R. Trans Fatty Acid in the Liver and Central Nervous System. In Dietary Interventions in Liver Disease; Ronald, R.W., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 275–286. [Google Scholar]
- Lee, Y.-Y.; Tang, T.-K.; Tan, C.-P.; Alitheen, N.B.M.; Phuah, E.-T.; Ab Karim, N.A.; Lai, O.-M. Entrapment of Palm-Based Medium- and Long-Chain Triacylglycerol via Maillard Reaction Products. Food Bioprocess Technol. 2015, 8, 1571–1582. [Google Scholar] [CrossRef]
- Baldim, I.; Souza, C.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.; Oliveira, W. Spray-dried structured lipid carriers for the loading of rosmarinus officinalis: New nutraceutical and food preservative. Foods 2020, 9, 1110. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Zhang, G.; Li, C.; Liu, L.; Sun, L.; Liu, N.; Li, X. Characteristic of microencapsulated 1,3-dioleoyl-2-palmitoylglycerol and its application in infant formula powder. Int. J. Food Prop. 2018, 21, 2355–2365. [Google Scholar] [CrossRef]
- Wei, W.; Li, C.; Zhang, W.; Liu, N. Strengthening oxidative stability of the infant liquid milk by structured lipid microcapsules. J. Chin. Inst. Food Sci. Technol. 2014, 14, 180–189. [Google Scholar]
- Nagachinta, S.; Akoh, C.C. Spray-dried structured lipid containing long-chain polyunsaturated fatty acids for use in infant formulas. J. Food Sci. 2013, 78, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Nagachinta, S.; Akoh, C.C. Synthesis of structured lipid enriched with omega fatty acids and sn -2 palmitic acid by enzymatic esterification and its incorporation in powdered infant formula. J. Agric. Food Chem. 2013, 61, 4455–4463. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).