Nano-Based Drug Delivery Systems of Potent MmpL3 Inhibitors for Tuberculosis Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
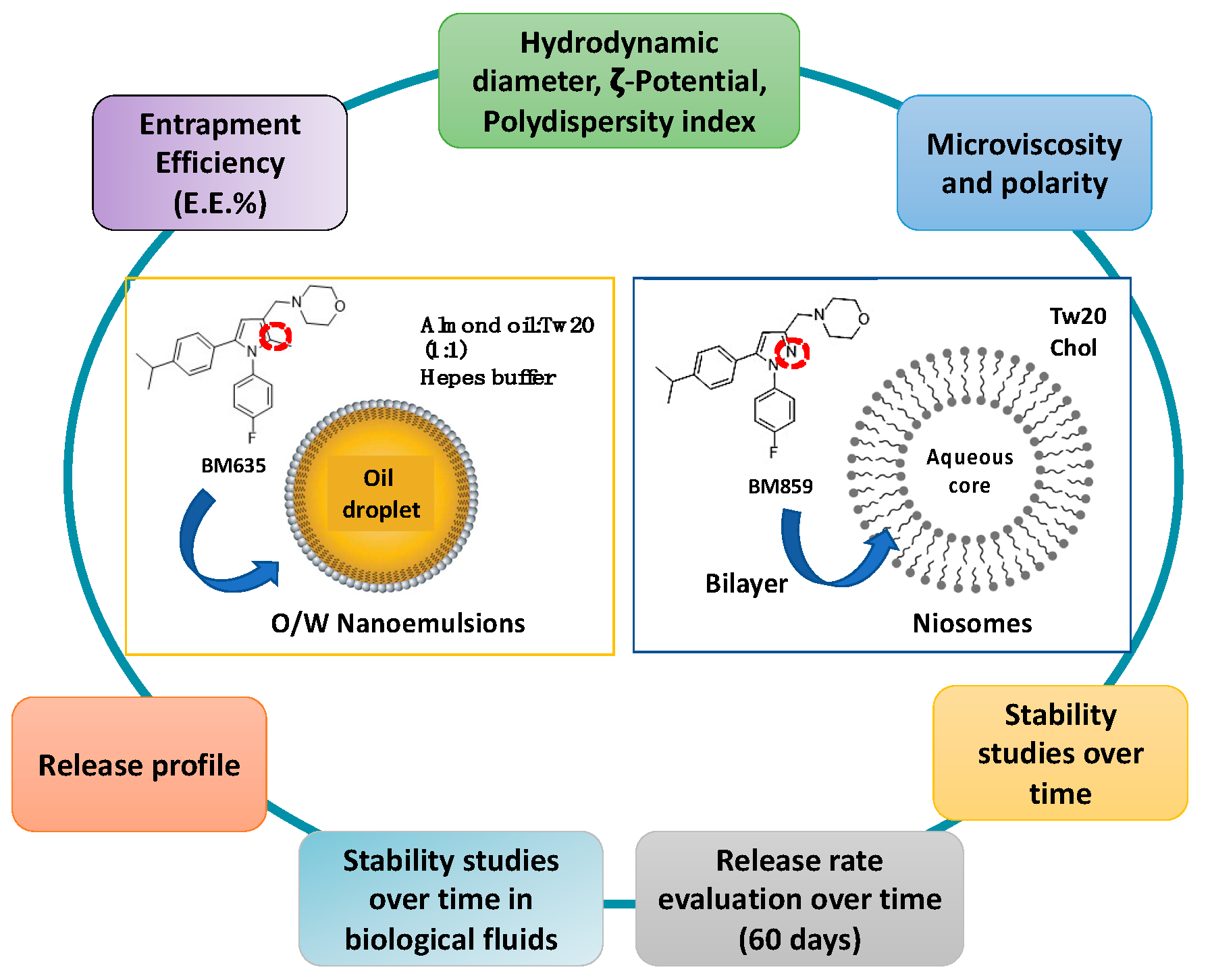
2.2. Ternary Diagram Construction and Empty and Loaded NEs Preparation
2.3. Preparation of NIs and Drug-Loaded NIs
2.4. Dynamic Light Scattering and ζ-Potential Measurements
2.5. Fluorometric Measurements
2.6. Drug-Entrapment Efficiency (E.E.%)
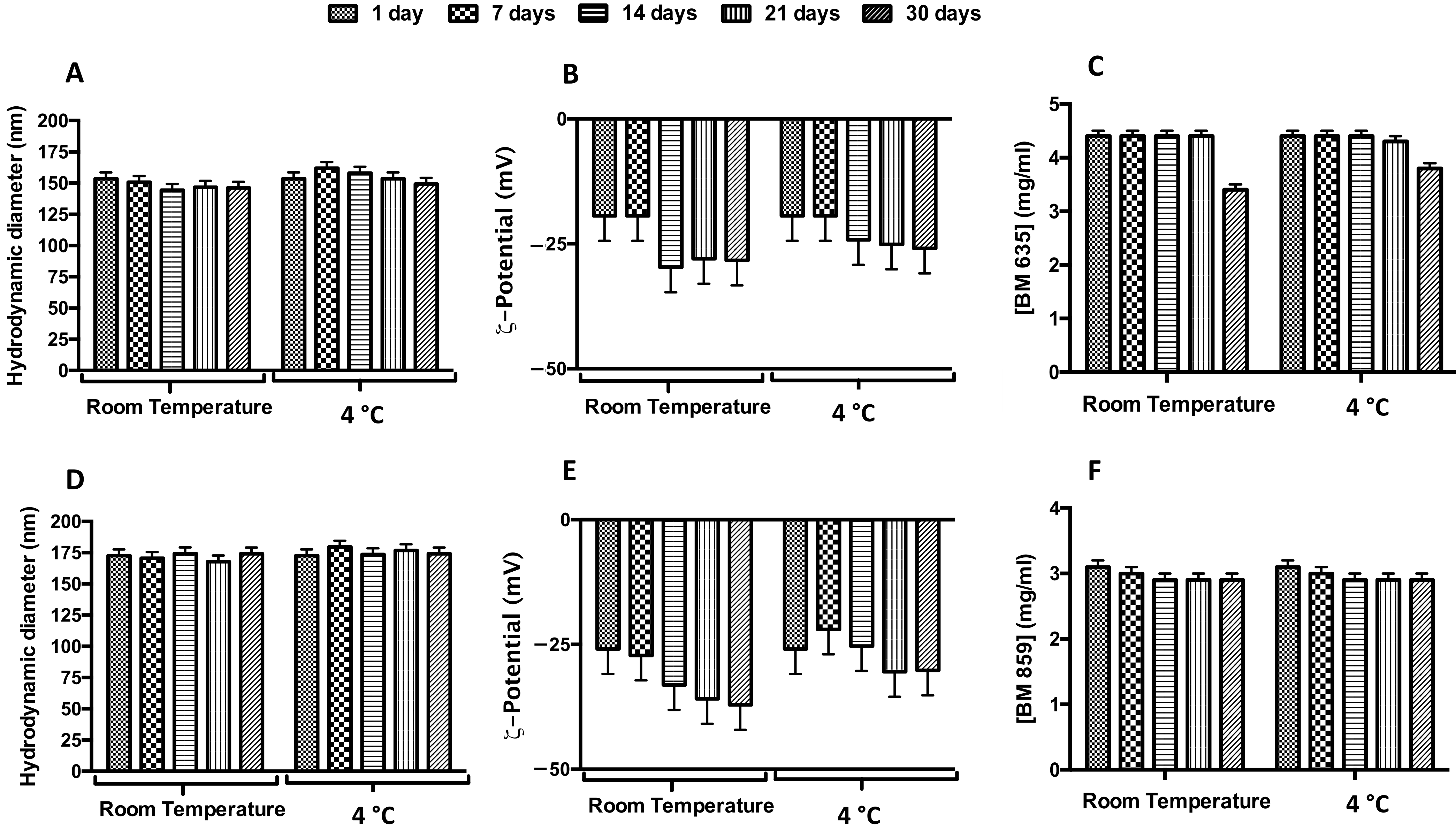
2.7. Physicochemical Stability
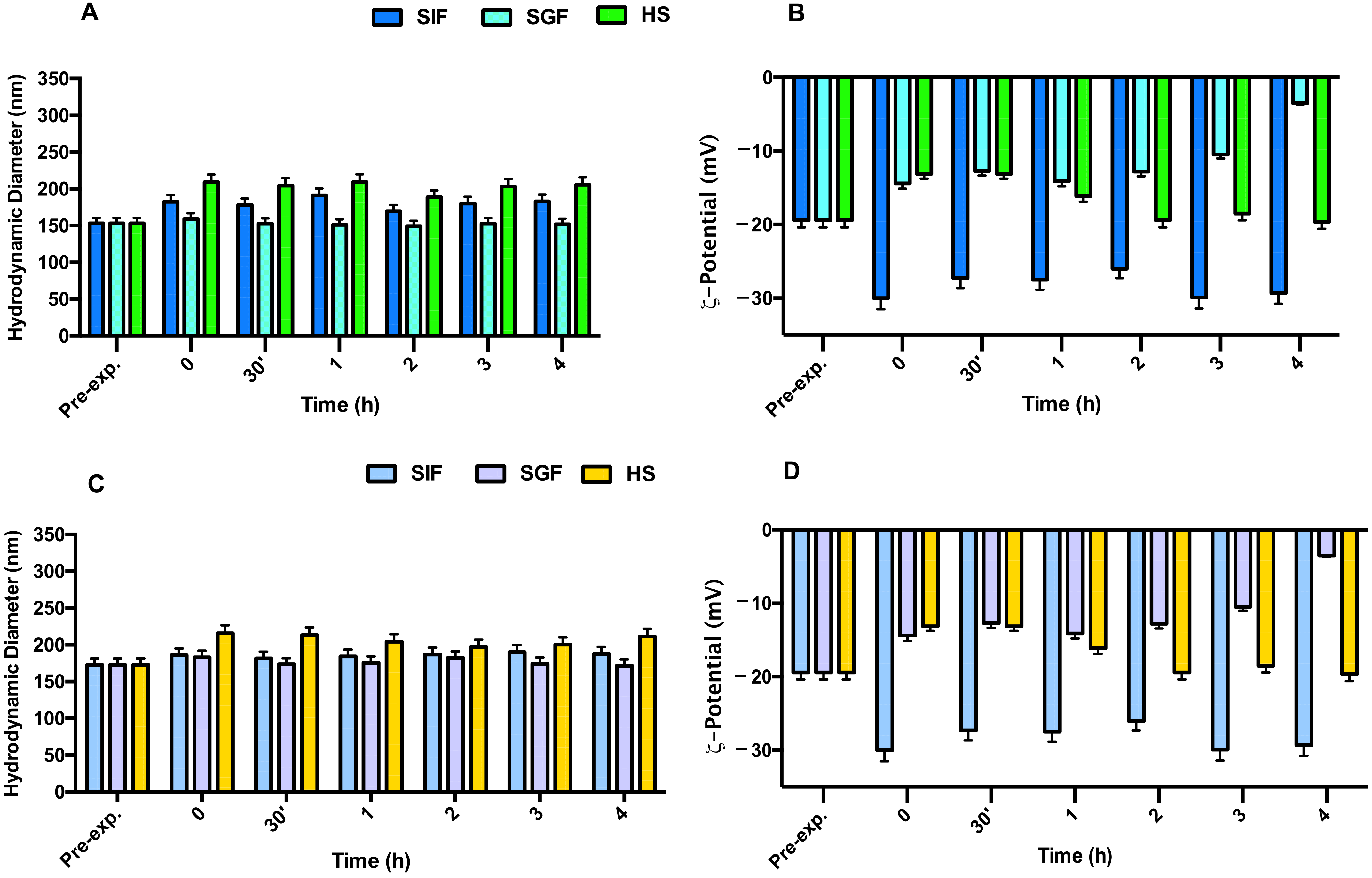
2.8. NEs and NIs Biological Stability
2.9. NEs and NIs Stability in Culture Medium
2.10. In Vitro Release Studies
2.11. MIC Determination
2.12. Statistical Analysis
3. Results and Discussion
3.1. NEs and NIs Characterization
Physicochemical Stability
3.2. Nes and Nis Biological Stability
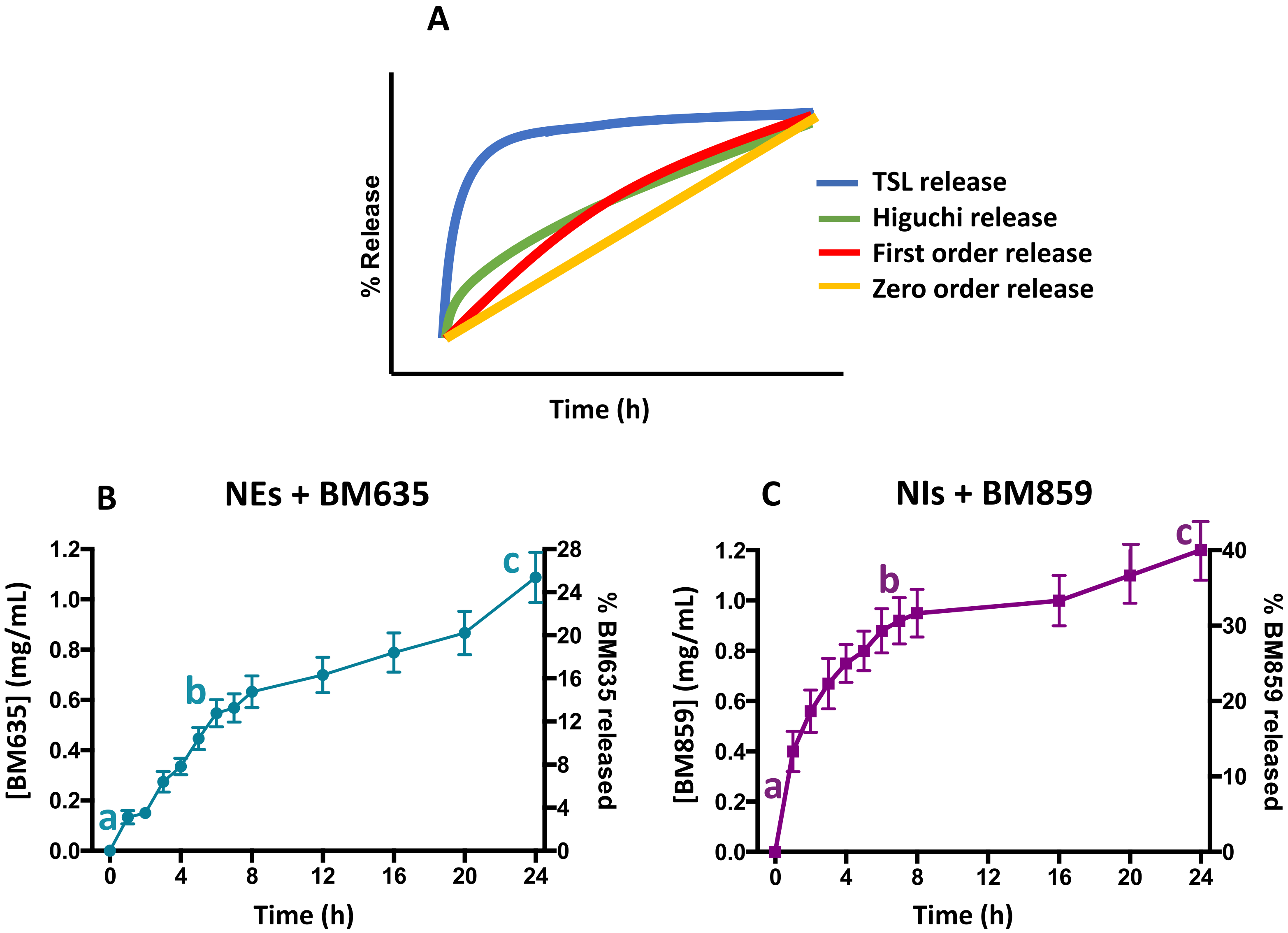
3.3. Release Studies
3.4. Microbiological Activity of Formulated Compounds
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Christie, S.; Potter, J. P40 Is Routine Liver Function Testing Necessary for Patients Receiving Latent Tuberculosis Treatment? Thorax 2021, 76, A87. [Google Scholar] [CrossRef]
- Poce, G.; Bates, R.H.; Alfonso, S.; Cocozza, M.; Porretta, G.C.; Ballell, L.; Rullas, J.; Ortega, F.; Logu, A.D.; Agus, E.; et al. Improved BM212 MmpL3 Inhibitor Analogue Shows Efficacy in Acute Murine Model of Tuberculosis Infection. PLoS ONE 2013, 8, e56980. [Google Scholar] [CrossRef] [PubMed]
- Poce, G.; Consalvi, S.; Venditti, G.; Alfonso, S.; Desideri, N.; Fernandez-Menendez, R.; Bates, R.H.; Ballell, L.; Barros Aguirre, D.; Rullas, J.; et al. Novel Pyrazole-Containing Compounds Active against Mycobacterium Tuberculosis. ACS Med. Chem. Lett. 2019, 10, 1423–1429. [Google Scholar] [CrossRef] [PubMed]
- Marianecci, C.; Di Marzio, L.; Rinaldi, F.; Celia, C.; Paolino, D.; Alhaique, F.; Esposito, S.; Carafa, M. Niosomes from 80s to Present: The State of the Art. Adv. Colloid Interface Sci. 2014, 205, 187–206. [Google Scholar] [CrossRef] [PubMed]
- Information for the Package Leaflet Regarding Polysorbates Used as Excipients in Medicinal Products for Human Use. Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/draft-information-package-leaflet-regarding-polysorbates-used-excipients-medicinal-products-human_en.pdf (accessed on 28 January 2022).
- Baranyai, Z.; Soria-Carrera, H.; Alleva, M.; Millán-Placer, A.C.; Lucía, A.; Martín-Rapún, R.; Aínsa, J.A.; la Fuente, J.M. Nanotechnology-Based Targeted Drug Delivery: An Emerging Tool to Overcome Tuberculosis. Adv. Therap. 2021, 4, 2000113. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers as Novel Drug Delivery Systems: Applications, Advantages and Disadvantages. Res. Pharm. Sci. 2018, 13, 288–303. [Google Scholar] [CrossRef] [PubMed]
- Di Marzio, L.; Esposito, S.; Rinaldi, F.; Marianecci, C.; Carafa, M. Polysorbate 20 Vesicles as Oral Delivery System: In Vitro Characterization. Colloids Surf. B Biointerfaces 2013, 104, 200–206. [Google Scholar] [CrossRef]
- Li, Y.; Li, M.; Qi, Y.; Zheng, L.; Wu, C.; Wang, Z.; Teng, F. Preparation and Digestibility of Fish Oil Nanoemulsions Stabilized by Soybean Protein Isolate-Phosphatidylcholine. Food Hydrocoll. 2020, 100, 105310. [Google Scholar] [CrossRef]
- Naderinezhad, S.; Amoabediny, G.; Haghiralsadat, F. Co-Delivery of Hydrophilic and Hydrophobic Anticancer Drugs Using Biocompatible PH-Sensitive Lipid-Based Nano-Carriers for Multidrug-Resistant Cancers. RSC Adv. 2017, 7, 30008–30019. [Google Scholar] [CrossRef] [Green Version]
- Sharma, V.; Anandhakumar, S.; Sasidharan, M. Self-Degrading Niosomes for Encapsulation of Hydrophilic and Hydrophobic Drugs: An Efficient Carrier for Cancer Multi-Drug Delivery. Mater. Sci. Eng. C 2015, 56, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Abdelbary, A.A.; AbouGhaly, M.H.H. Design and Optimization of Topical Methotrexate Loaded Niosomes for Enhanced Management of Psoriasis: Application of Box–Behnken Design, in-Vitro Evaluation and in-Vivo Skin Deposition Study. Int. J. Pharm. 2015, 485, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, F.; Seguella, L.; Gigli, S.; Hanieh, P.N.; Del Favero, E.; Cantù, L.; Pesce, M.; Sarnelli, G.; Marianecci, C.; Esposito, G.; et al. InPentasomes: An Innovative Nose-to-Brain Pentamidine Delivery Blunts MPTP Parkinsonism in Mice. J. Control. Release 2019, 294, 17–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koppel, D.E. Analysis of Macromolecular Polydispersity in Intensity Correlation Spectroscopy: The Method of Cumulants. J. Chem. Phys. 1972, 57, 4814–4820. [Google Scholar] [CrossRef]
- De Vos, C.; Deriemaeker, L.; Finsy, R. Quantitative Assessment of the Conditioning of the Inversion of Quasi-Elastic and Static Light Scattering Data for Particle Size Distributions. Langmuir 1996, 12, 2630–2636. [Google Scholar] [CrossRef]
- Sennato, S.; Bordi, F.; Cametti, C.; Marianecci, C.; Carafa, M.; Cametti, M. Hybrid Niosome Complexation in the Presence of Oppositely Charged Polyions. J. Phys. Chem. B 2008, 112, 3720–3727. [Google Scholar] [CrossRef] [PubMed]
- Ingallina, C.; Rinaldi, F.; Bogni, A.; Ponti, J.; Passeri, D.; Reggente, M.; Rossi, M.; Kinsner-Ovaskainen, A.; Mehn, D.; Rossi, F.; et al. Niosomal Approach to Brain Delivery: Development, Characterization and in Vitro Toxicological Studies. Int. J. Pharm. 2016, 511, 969–982. [Google Scholar] [CrossRef]
- Zachariasse, K.A. Intramolecular Excimer Formation with Diarylalkanes as a Microfluidity Probe for Sodium Dodecyl Sulphate Micelles. Chem. Phys. Lett. 1978, 57, 429–432. [Google Scholar] [CrossRef]
- Naseema, A.; Kovooru, L.; Behera, A.K.; Kumar, K.P.P.; Srivastava, P. A Critical Review of Synthesis Procedures, Applications and Future Potential of Nanoemulsions. Adv. Colloid Interface Sci. 2021, 287, 102318. [Google Scholar] [CrossRef]
- Stepanovic, S.; Cirkovic, I.; Ranin, L.; Svabic-Vlahovic, M. Biofilm Formation by Salmonella Spp. and Listeria Monocytogenes on Plastic Surface. Lett. Appl. Microbiol. 2004, 38, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Mahdi, Z.H.; Maraie, N.K. Overview on Nanoemulsion as a Recently Developed Approach in Drug Nanoformulation. Res. J. Pharm. Technol. 2019, 12, 5554. [Google Scholar] [CrossRef]
- Rinaldi, F.; Maurizi, L.; Conte, A.L.; Marazzato, M.; Maccelli, A.; Crestoni, M.E.; Hanieh, P.N.; Forte, J.; Conte, M.P.; Zagaglia, C.; et al. Nanoemulsions of Satureja Montana Essential Oil: Antimicrobial and Antibiofilm Activity against Avian Escherichia Coli Strains. Pharmaceutics 2021, 13, 134. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, R.C.d.A.; Barreto, S.M.A.G.; Ostrosky, E.A.; Rocha-Filho, P.A.d.; Veríssimo, L.M.; Ferrari, M. Production and Characterization of Cosmetic Nanoemulsions Containing Opuntia Ficus-Indica (L.) Mill Extract as Moisturizing Agent. Molecules 2015, 20, 2492–2509. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hillaireau, H.; Couvreur, P. Nanocarriers’ Entry into the Cell: Relevance to Drug Delivery. Cell. Mol. Life Sci. 2009, 66, 2873–2896. [Google Scholar] [CrossRef] [PubMed]
- Di Marzio, L.; Marianecci, C.; Petrone, M.; Rinaldi, F.; Carafa, M. Novel PH-Sensitive Non-Ionic Surfactant Vesicles: Comparison between Tween 21 and Tween 20. Colloids Surf. B Biointerfaces 2011, 82, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; ten Hagen, T.L.M. A Novel Kinetic Model to Describe the Ultra-Fast Triggered Release of Thermosensitive Liposomal Drug Delivery Systems. J. Control. Release 2020, 324, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Akbari, V.; Abedi, D.; Pardakhty, A.; Sadeghi-Aliabadi, H. Release Studies on Ciprofloxacin Loaded Non-Ionic Surfactant Vesicles. Avicenna J. Med. Biotechnol. 2015, 7, 69–75. [Google Scholar] [PubMed]
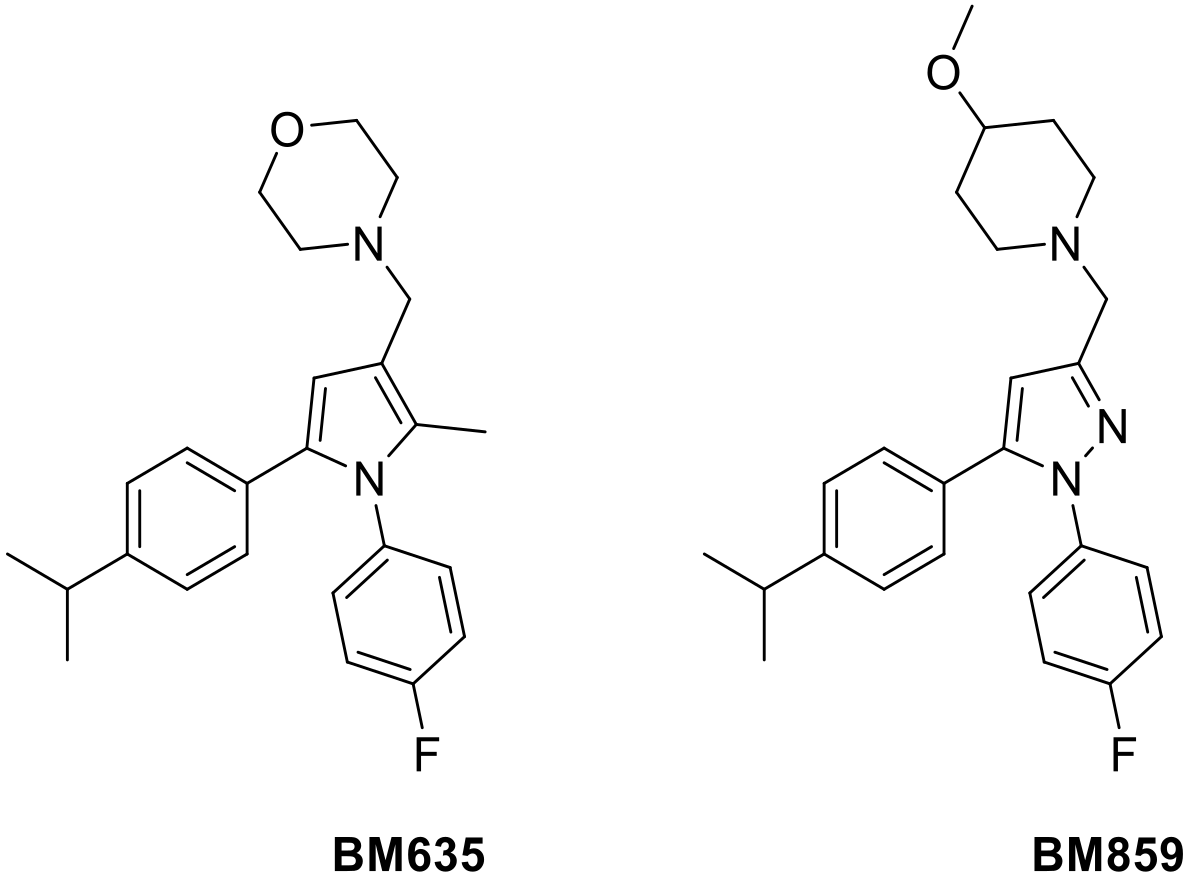

| Sample | Tween 20 (mg/mL) | Almond Oil (mg/mL) | BM635 Loaded (mg/mL) | Cholesterol (mg/mL) | BM859 Loaded (mg/mL) |
|---|---|---|---|---|---|
| NEs | 18.4 | 18.4 | - | - | - |
| Nes + BM635 | 4.6 | ||||
| NIs | - | - | 5.8 | - | |
| Nis + BM859 | 3.8 |
| Sample | Hydrodynamic Diameter (nm) ± SD | ζ-Potential (mV) ± SD | PDI ± SD | Drug-Entrapment Efficiency (E.E.%) | I1/I3 (Polarity) | IE/I3 (Microviscosity) |
|---|---|---|---|---|---|---|
| NEs | 132.6 ± 3.4 159.5 ± 4.6 | −21.9 ± 1.2 −25.9 ± 2.3 | 0.1 ± 0.01 | - | 0.96 | 1.87 |
| NEs + BM635 | 0.1 ± 0.01 | 95.6 | 0.97 | 2.15 | ||
| NIs | 161.0 ± 3.3 172.5 ± 4.2 | −28.5 ± 1.4 −19.4 ± 3.3 | 0.1 ± 0.01 | - | 1.26 | 0.67 |
| NIs + BM859 | 0.1 ± 0.01 | 81.6 | 1.32 | 0.82 |
| Compound | MIC (µM) |
|---|---|
| Nes + BM635 | 40 |
| BM635 | 0.12 |
| NEs | ND a |
| Nis + BM859 | 0.6 |
| BM859 | 0.3 |
| NIs | ND a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanieh, P.N.; Consalvi, S.; Forte, J.; Cabiddu, G.; De Logu, A.; Poce, G.; Rinaldi, F.; Biava, M.; Carafa, M.; Marianecci, C. Nano-Based Drug Delivery Systems of Potent MmpL3 Inhibitors for Tuberculosis Treatment. Pharmaceutics 2022, 14, 610. https://doi.org/10.3390/pharmaceutics14030610
Hanieh PN, Consalvi S, Forte J, Cabiddu G, De Logu A, Poce G, Rinaldi F, Biava M, Carafa M, Marianecci C. Nano-Based Drug Delivery Systems of Potent MmpL3 Inhibitors for Tuberculosis Treatment. Pharmaceutics. 2022; 14(3):610. https://doi.org/10.3390/pharmaceutics14030610
Chicago/Turabian StyleHanieh, Patrizia Nadia, Sara Consalvi, Jacopo Forte, Gianluigi Cabiddu, Alessandro De Logu, Giovanna Poce, Federica Rinaldi, Mariangela Biava, Maria Carafa, and Carlotta Marianecci. 2022. "Nano-Based Drug Delivery Systems of Potent MmpL3 Inhibitors for Tuberculosis Treatment" Pharmaceutics 14, no. 3: 610. https://doi.org/10.3390/pharmaceutics14030610
APA StyleHanieh, P. N., Consalvi, S., Forte, J., Cabiddu, G., De Logu, A., Poce, G., Rinaldi, F., Biava, M., Carafa, M., & Marianecci, C. (2022). Nano-Based Drug Delivery Systems of Potent MmpL3 Inhibitors for Tuberculosis Treatment. Pharmaceutics, 14(3), 610. https://doi.org/10.3390/pharmaceutics14030610











