FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis
Abstract
1. Introduction
2. FDA-Approved Drugs in 2022
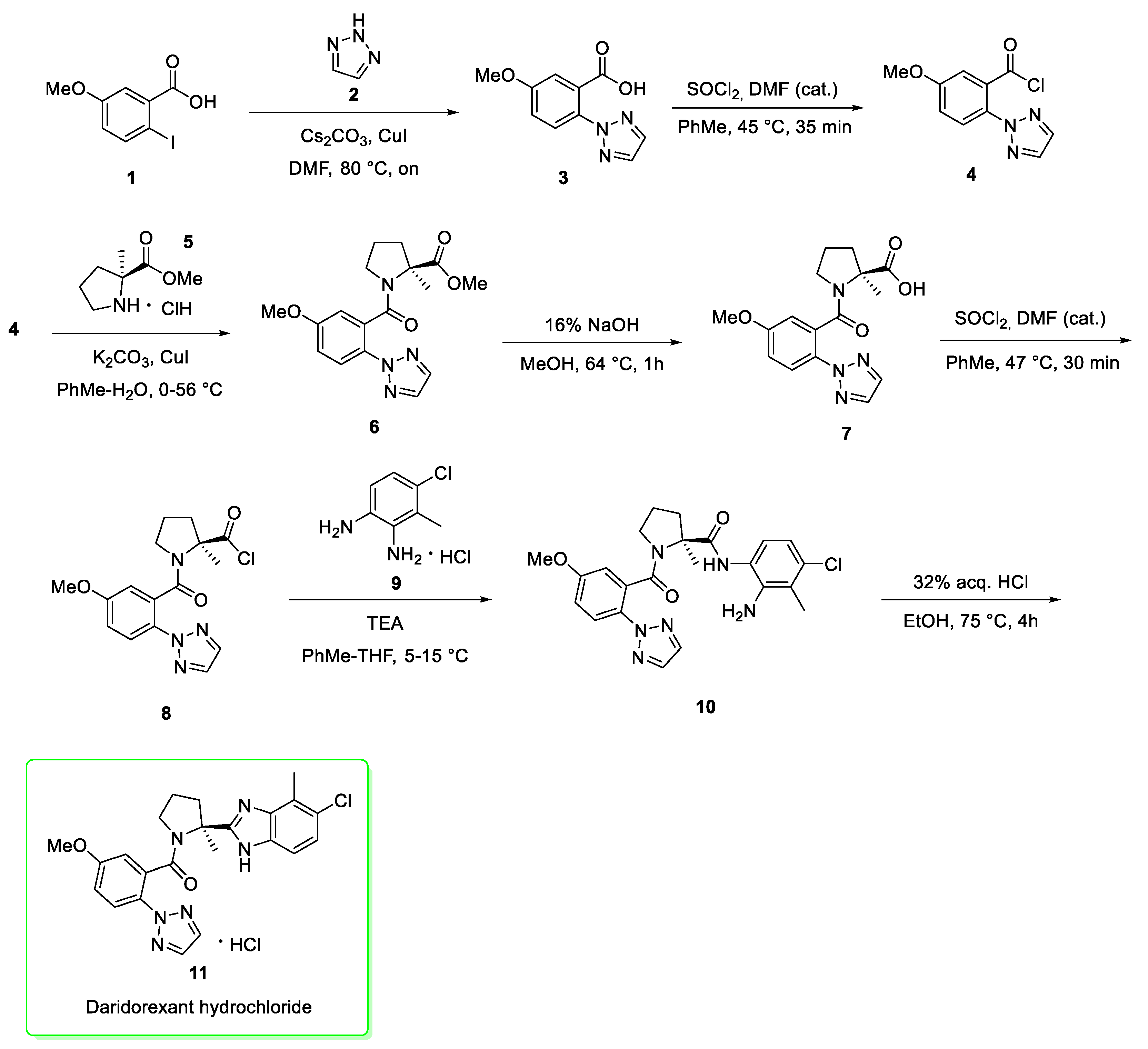
2.1. Daridorexant
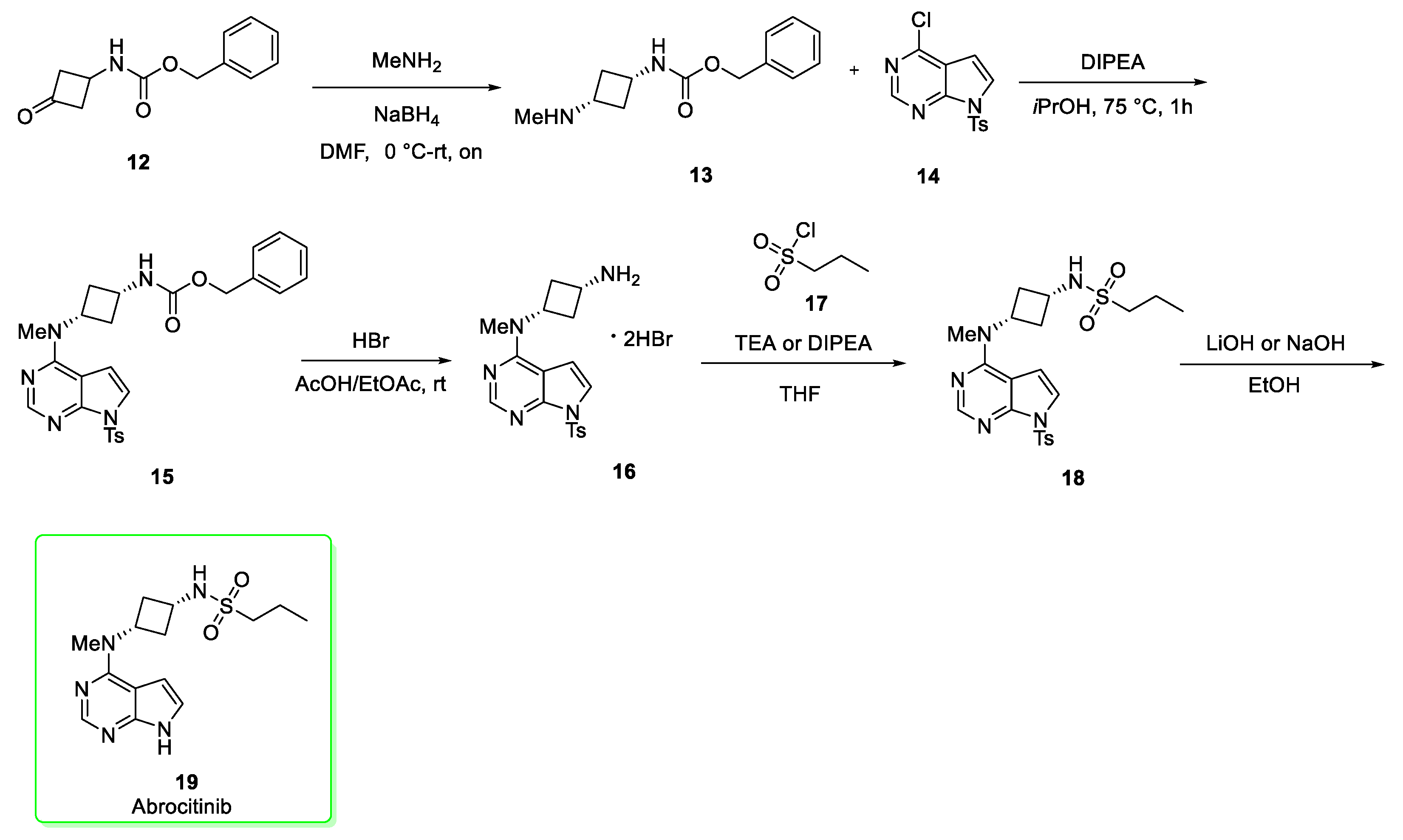
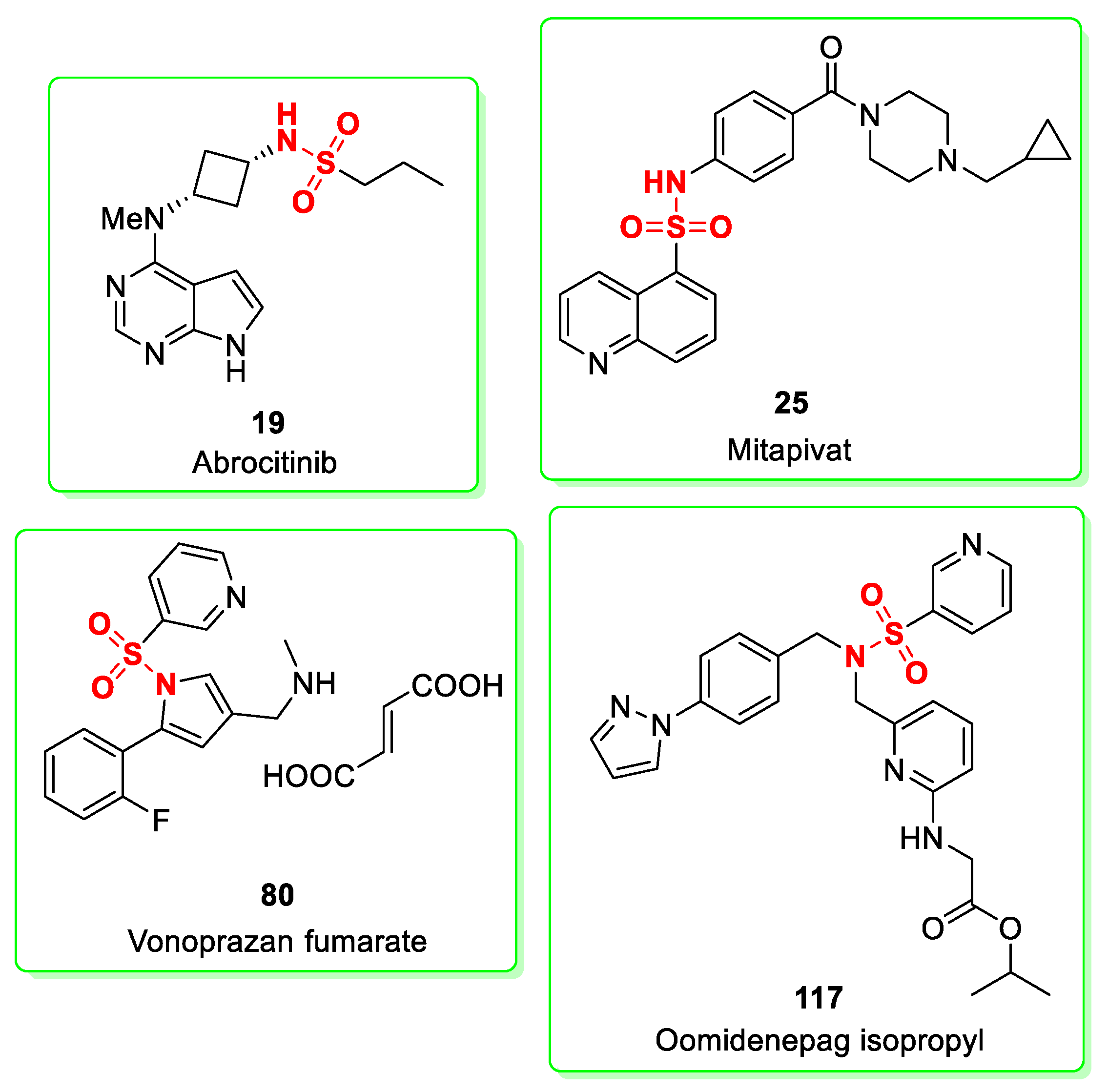
2.2. Abrocitinib
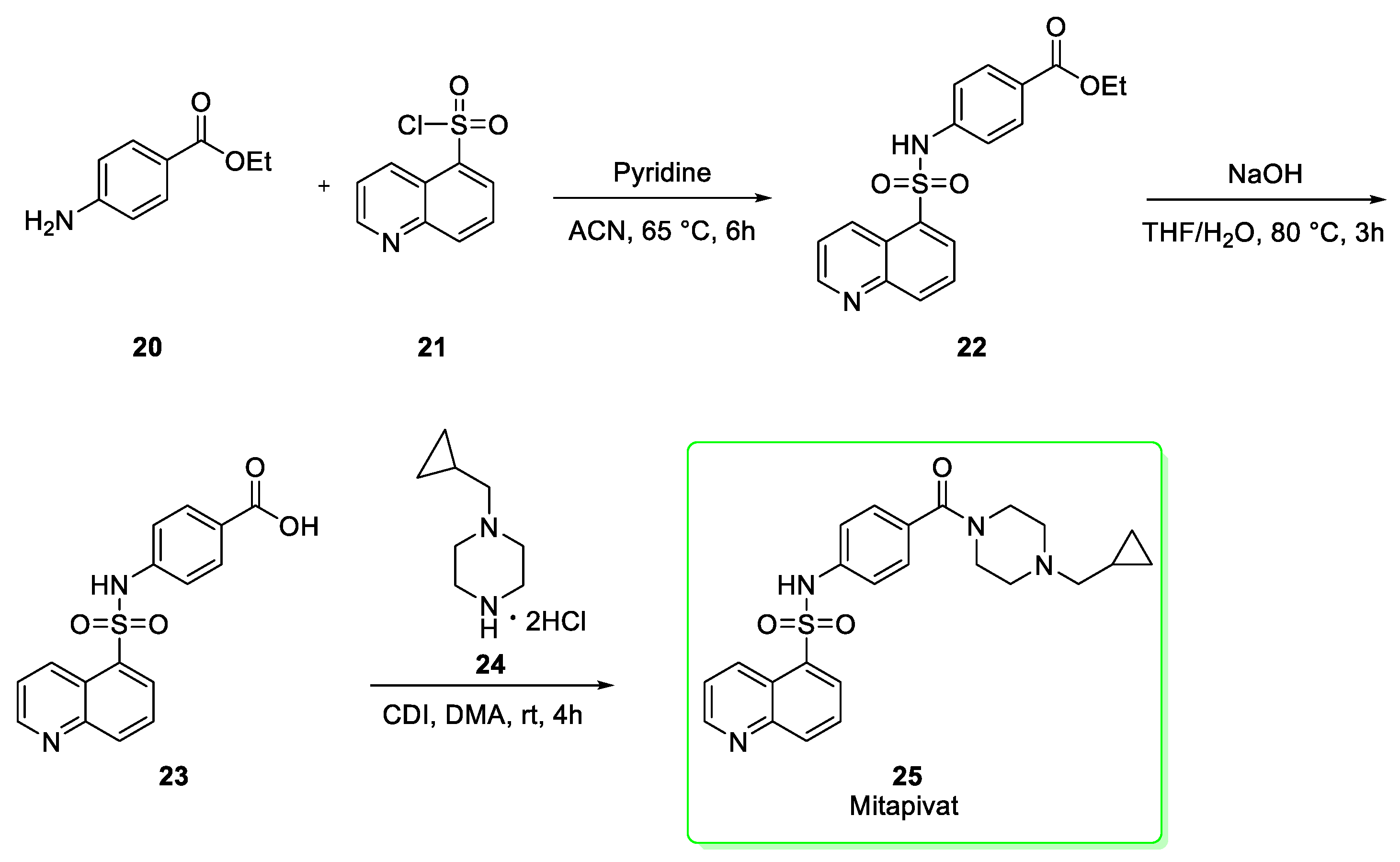
2.3. Mitapivat
2.4. Pacritinib
2.5. Ganaxolone
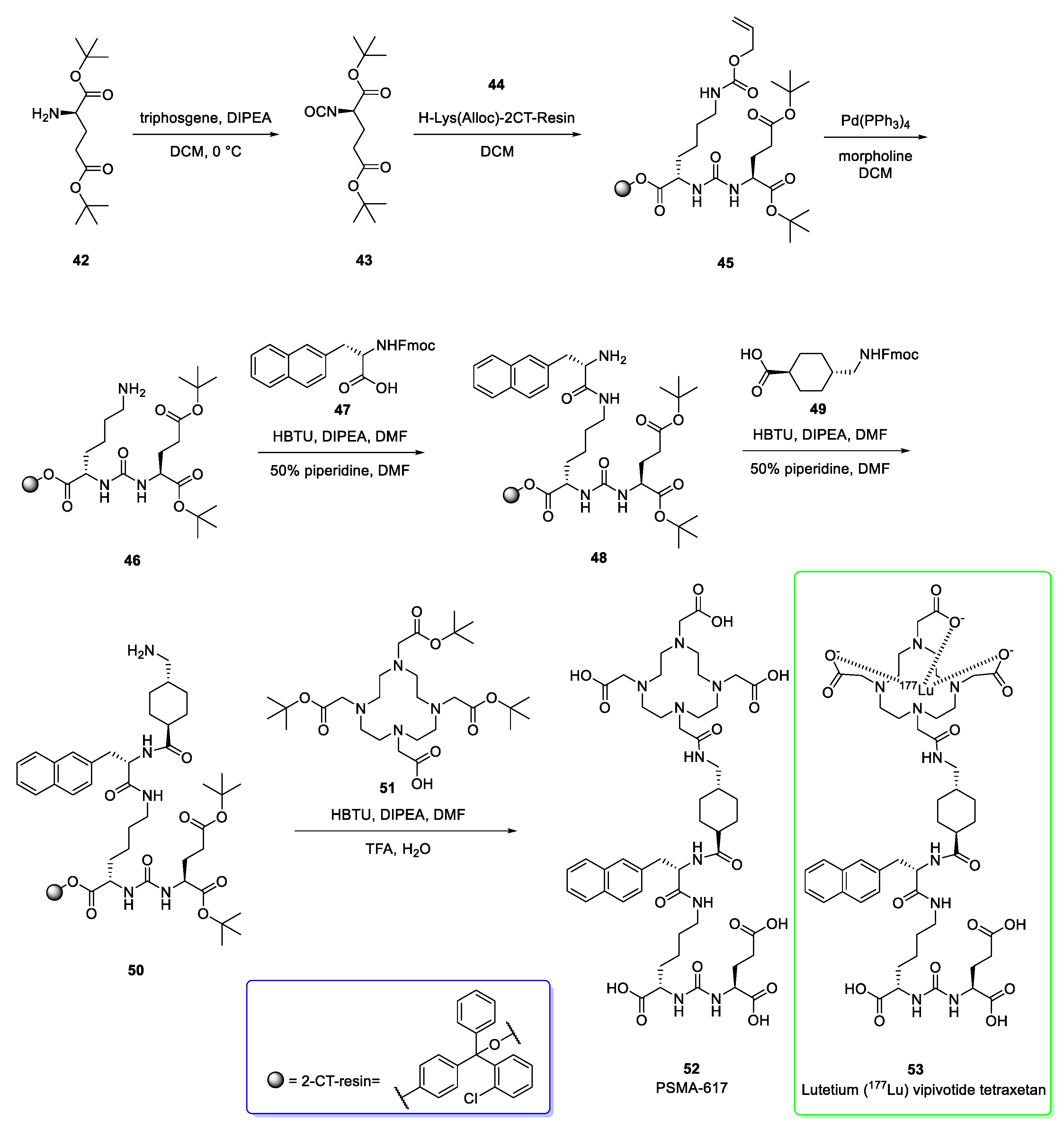
2.6. Lutetium (177Lu) Vipivotide Tetraxetan
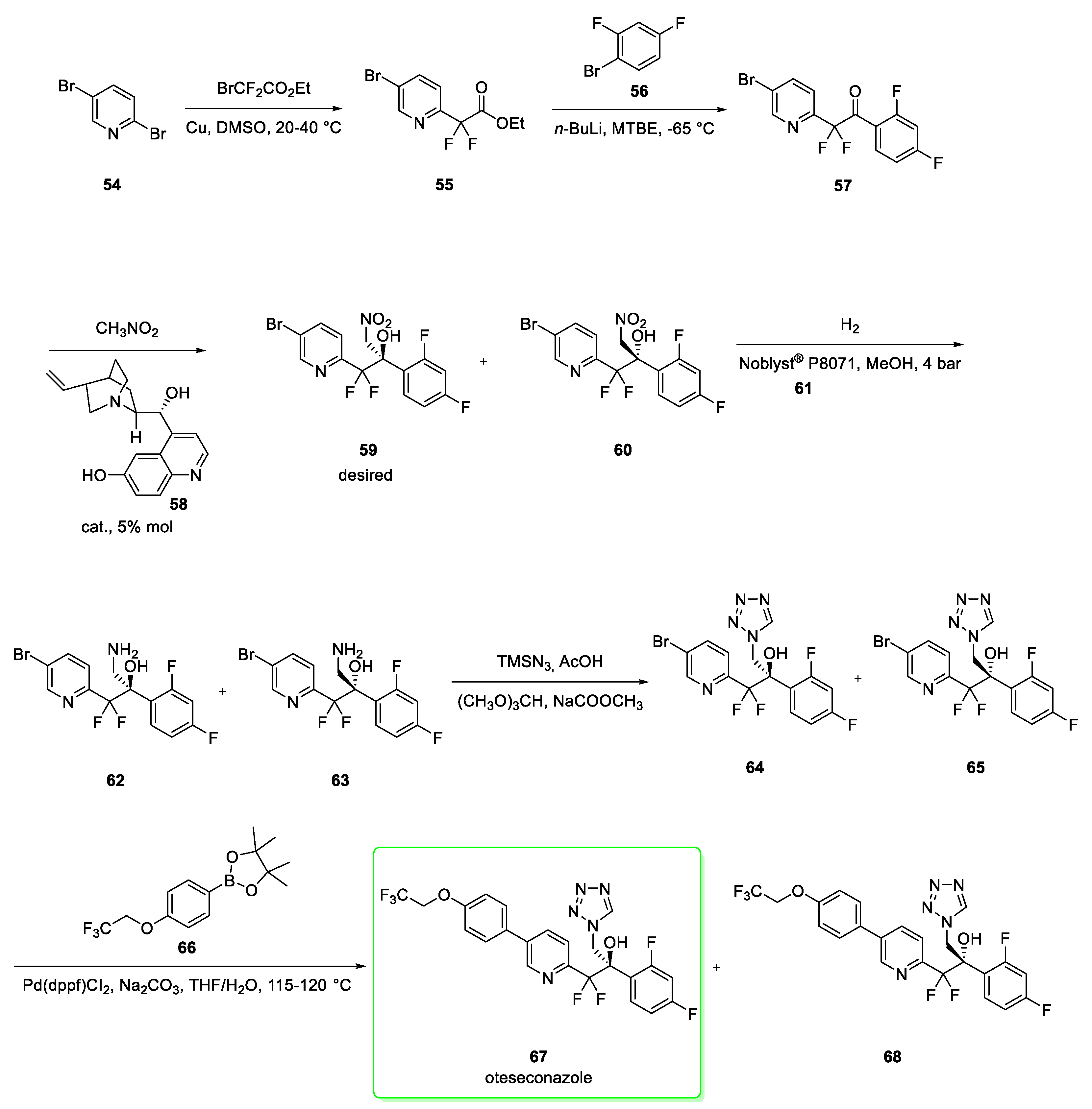
2.7. Oteseconazole
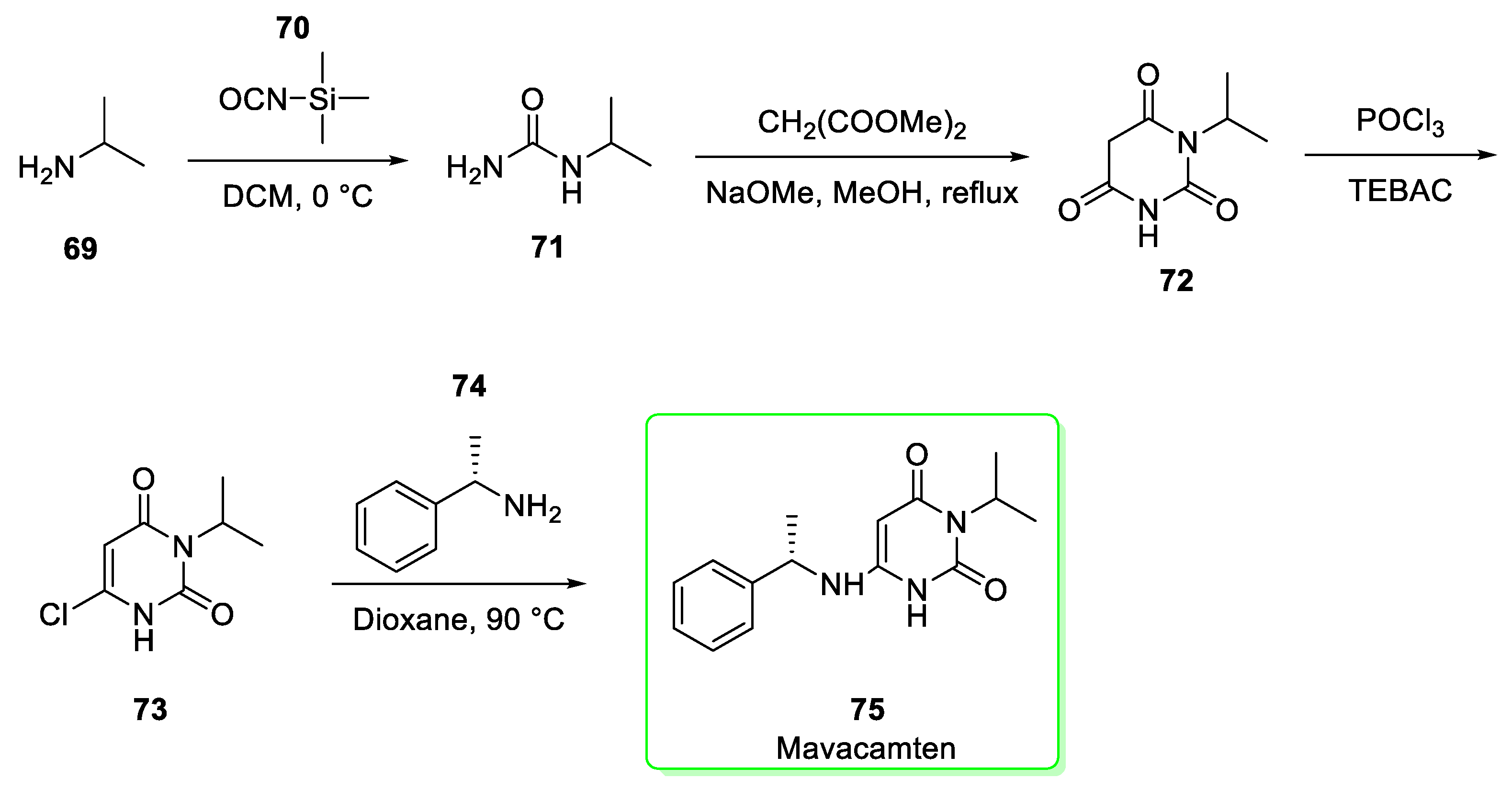
2.8. Mavacamten
2.9. Vonoprazan (in Combination with Amoxicillin, and Clarithromycin)
2.10. Tapinarof
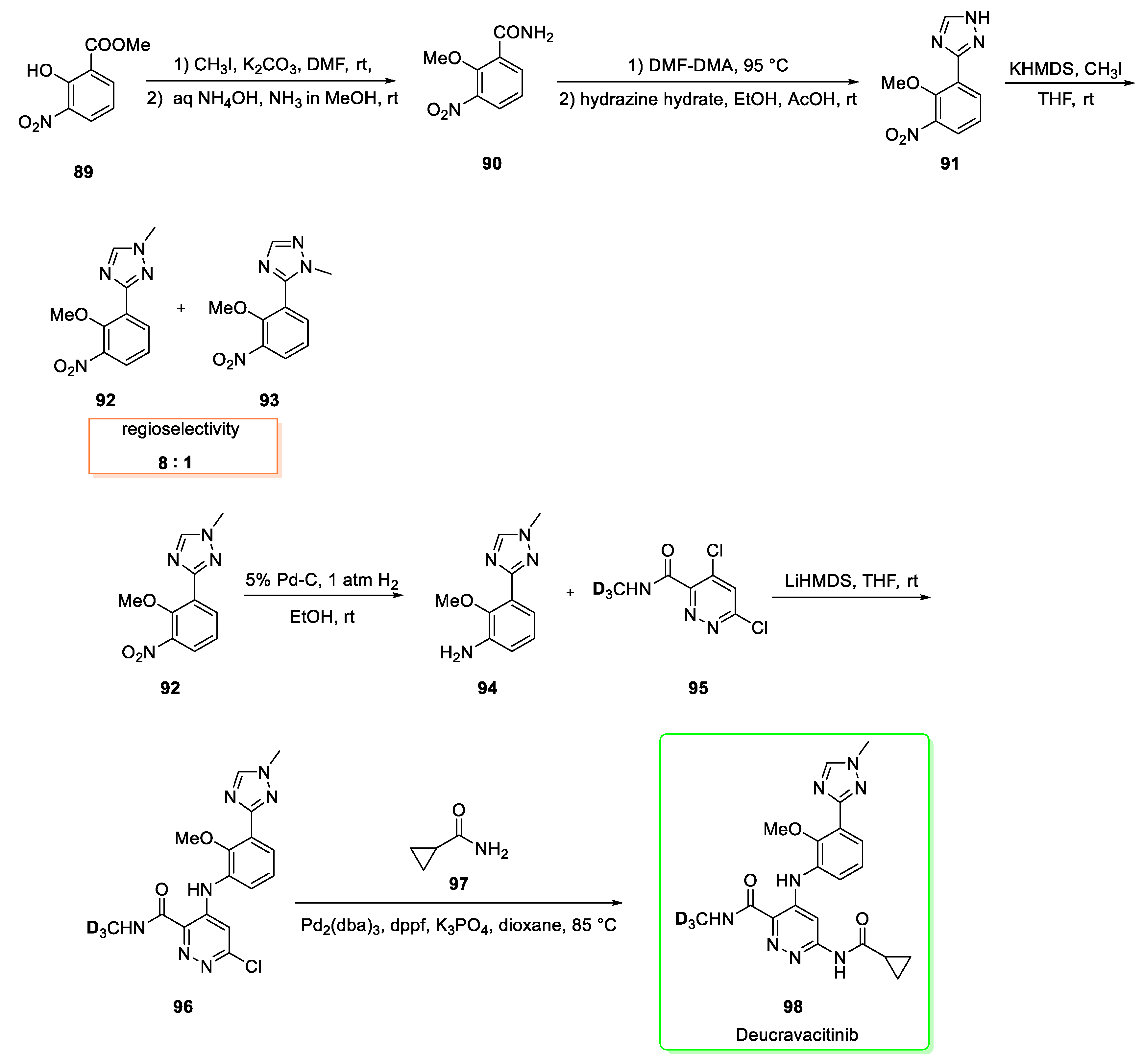
2.11. Deucravacitinib
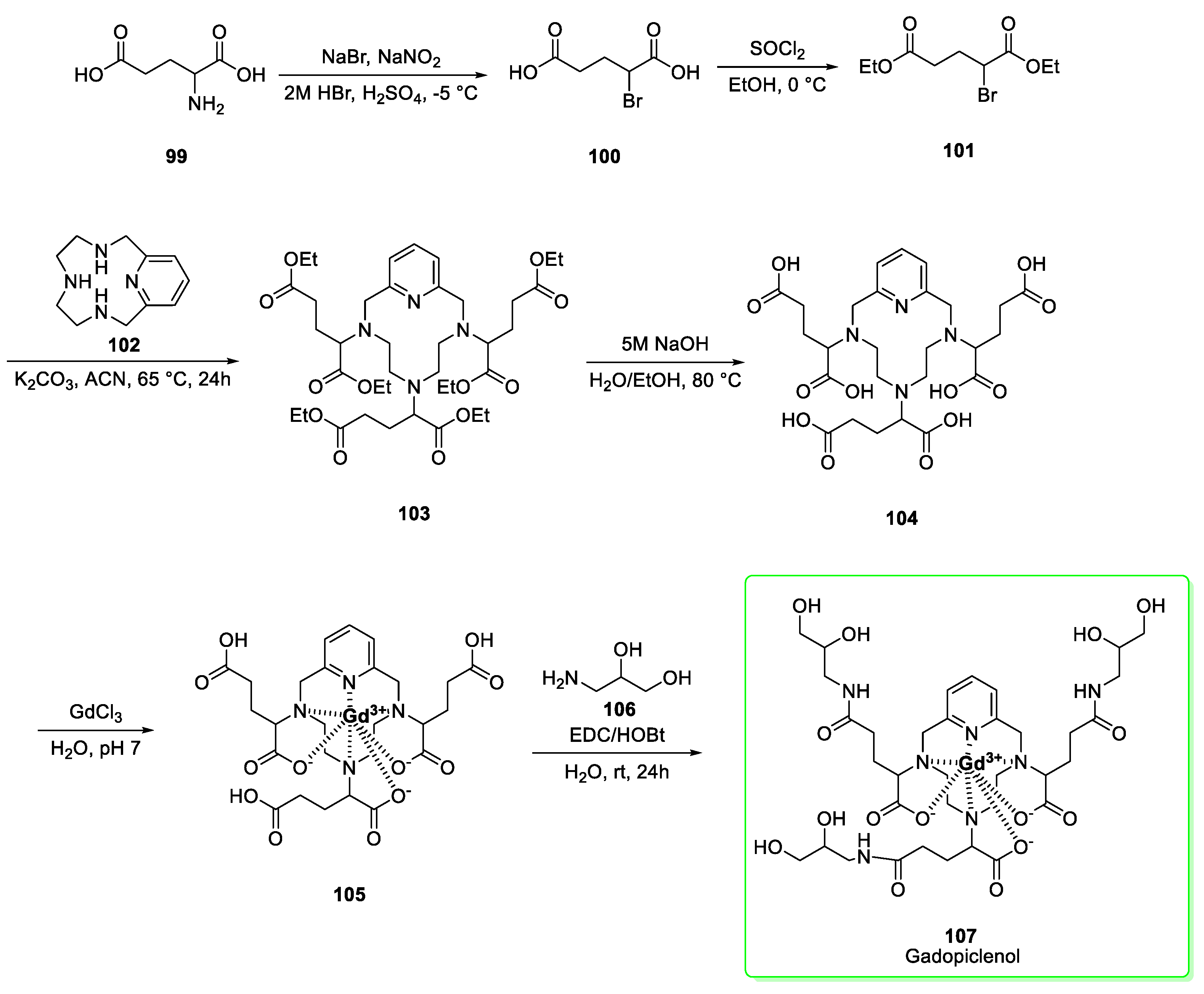
2.12. Gadopiclenol
2.13. Oomidenepag Isopropyl
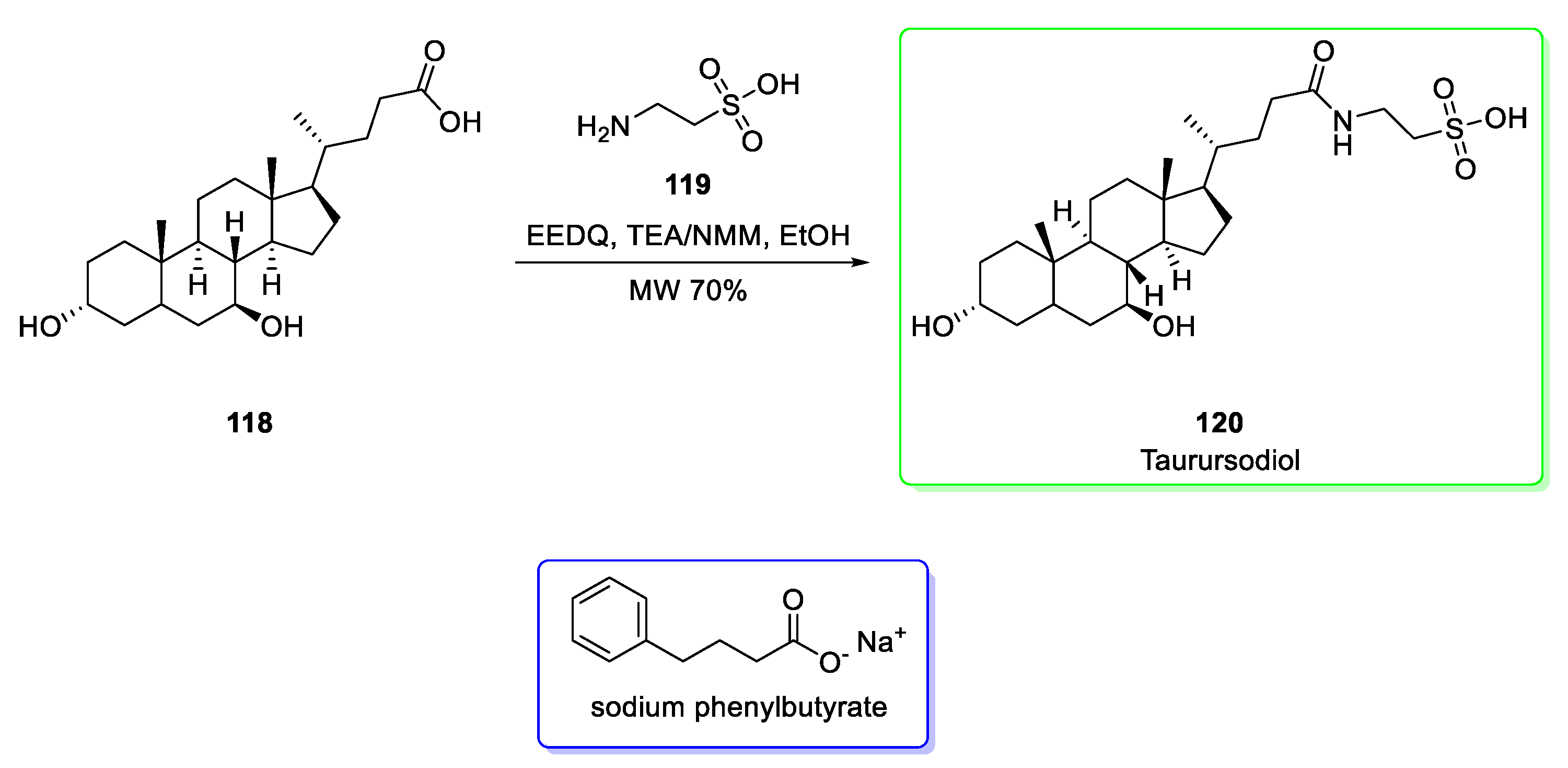
2.14. Taurursodiol (in Combination with Sodium Phenylbutyrate)
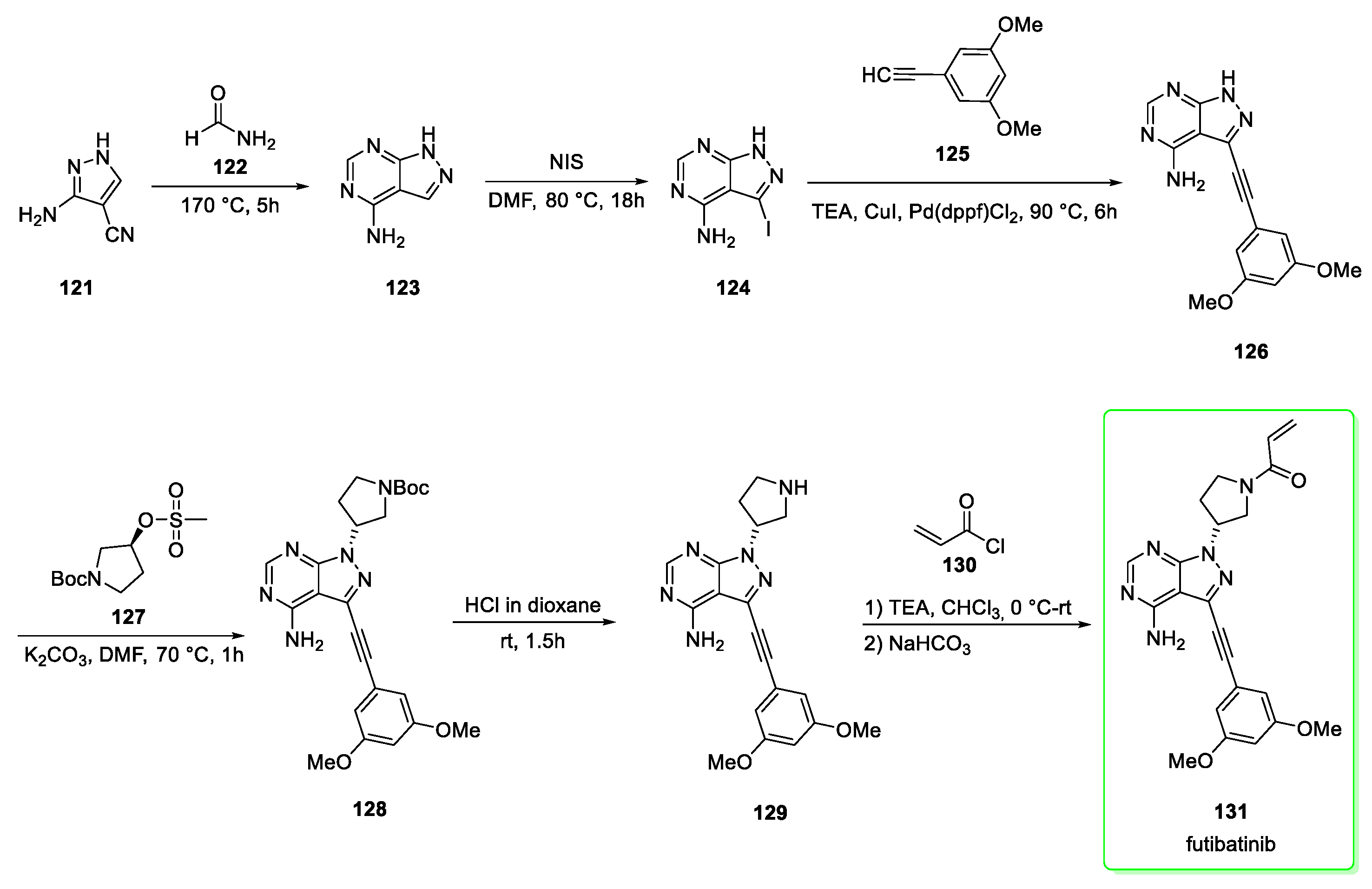
2.15. Futibatinib
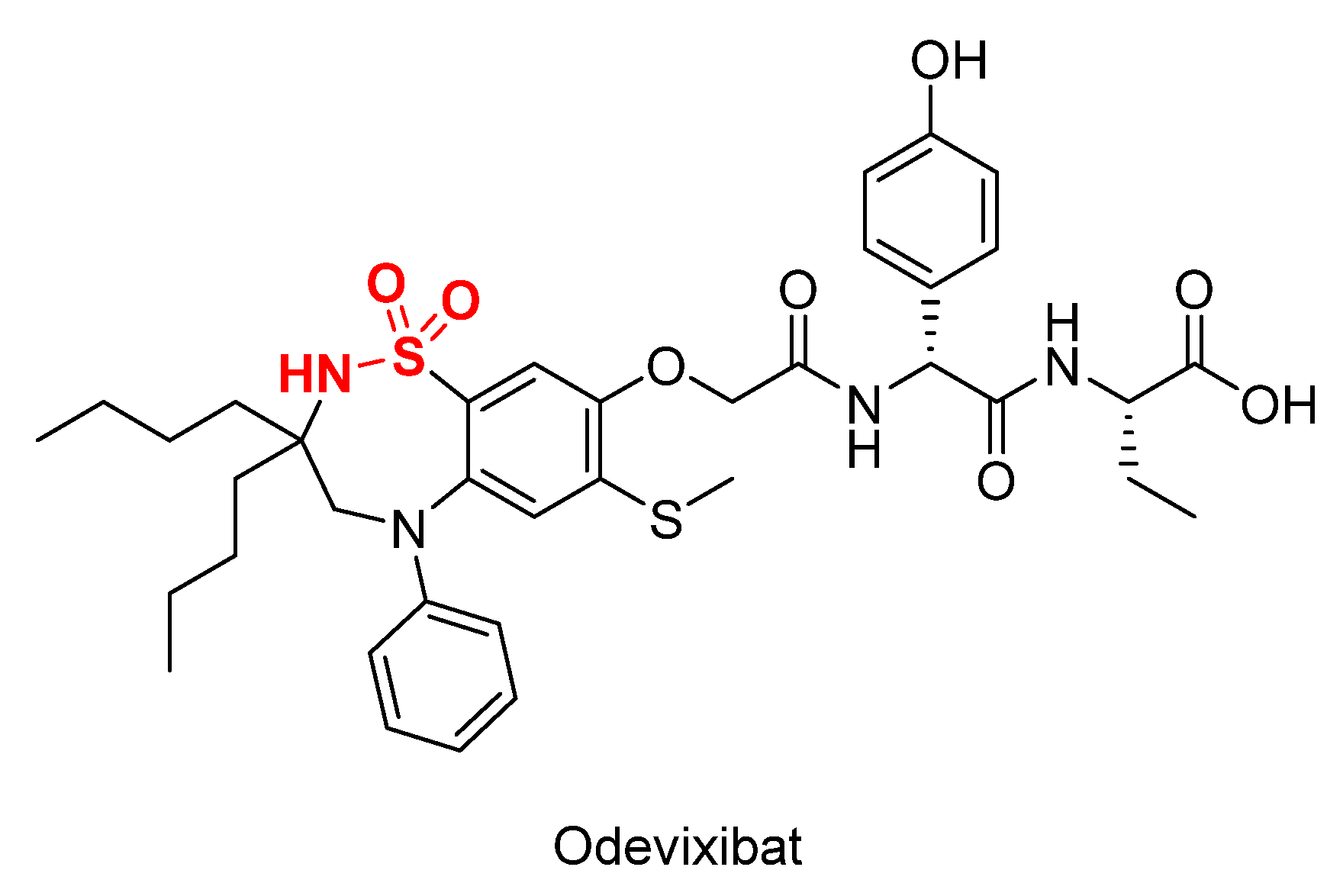
3. Sulfonamide: An Historical Re-Occurring Moiety in the FDA-Approved Drug List
4. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hunting for Drugs in Chemical Space. Available online: https://cen.acs.org/pharmaceuticals/drug-discovery/Hunting-drugs-chemical-space/100/i23 (accessed on 24 October 2022).
- Small Molecules. Available online: https://www.astrazeneca.com/r-d/next-generation-therapeutics/small-molecule.html (accessed on 27 October 2022).
- Novel Drug Approvals for 2022|FDA. Available online: https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2022 (accessed on 24 October 2022).
- Benedetto Tiz, D.; Bagnoli, L.; Rosati, O.; Marini, F.; Sancineto, L.; Santi, C. New Halogen-Containing Drugs Approved by FDA in 2021: An Overview on Their Syntheses and Pharmaceutical Use. Molecules 2022, 27, 1643. [Google Scholar] [CrossRef] [PubMed]
- Mignot, E.; Mayleben, D.; Fietze, I.; Leger, D.; Zammit, G.; Bassetti, C.L.A.; Pain, S.; Kinter, D.S.; Roth, T. Safety and Efficacy of Daridorexant in Patients with Insomnia Disorder: Results from Two Multicentre, Randomised, Double-Blind, Placebo-Controlled, Phase 3 Trials. Lancet Neurol. 2022, 21, 125–139. [Google Scholar] [CrossRef]
- Urquhart, L. FDA New Drug Approvals in Q1 2022. Nat. Rev. Drug Discov. 2022, 21, 329. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, T. The Role of Orexin in Motivated Behaviours. Nat. Rev. Neurosci. 2014, 15, 719–731. [Google Scholar] [CrossRef] [PubMed]
- Kocienski, P. Synthesis of Daridorexant. Synfacts 2021, 17, 0244. [Google Scholar] [CrossRef]
- Napolitano, M.; Fabbrocini, G.; Ruggiero, A.; Marino, V.; Nocerino, M.; Patruno, C. The Efficacy and Safety of Abrocitinib as a Treatment Option for Atopic Dermatitis: A Short Report of the Clinical Data. Drug Des. Dev. Ther. 2021, 15, 1135–1147. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, M.L.; Kaila, N.; Strohbach, J.W.; Trzupek, J.D.; Brown, M.F.; Flanagan, M.E.; Mitton-Fry, M.J.; Johnson, T.A.; TenBrink, R.E.; Arnold, E.P.; et al. Identification of N-{Cis-3-[Methyl(7H-Pyrrolo[2,3-d]Pyrimidin-4-Yl)Amino]Cyclobutyl}propane-1-Sulfonamide (PF-04965842): A Selective JAK1 Clinical Candidate for the Treatment of Autoimmune Diseases. J. Med. Chem. 2018, 61, 1130–1152. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Karmilowicz, M.J.; Burke, D.; Burns, M.P.; Clark, L.A.; Connor, C.G.; Cordi, E.; Do, N.M.; Doyle, K.M.; Hoagland, S.; et al. Biocatalytic Reductive Amination from Discovery to Commercial Manufacturing Applied to Abrocitinib JAK1 Inhibitor. Nat. Catal. 2021, 4, 775–782. [Google Scholar] [CrossRef]
- Al-Samkari, H.; van Beers, E.J. Mitapivat, a Novel Pyruvate Kinase Activator, for the Treatment of Hereditary Hemolytic Anemias. Ther. Adv. Hematol. 2021, 12, 204062072110660. [Google Scholar] [CrossRef] [PubMed]
- Sizemore, J.; Guo, L.; Mirmehrabi, M.; Su, Y. Crystalline Forms of N-(4-(4-(Cyclopropylmethyl) Piperazine-1-Carbonyl)Phenyl)Quinoline-8-Sulfonamide. WO2019104134A1, 31 May 2019. [Google Scholar]
- Verstovsek, S.; Komrokji, R.S. A Comprehensive Review of Pacritinib in Myelofibrosis. Future Oncol. 2015, 11, 2819–2830. [Google Scholar] [CrossRef]
- Hart, S.; Goh, K.C.; Novotny-Diermayr, V.; Tan, Y.C.; Madan, B.; Amalini, C.; Ong, L.C.; Kheng, B.; Cheong, A.; Zhou, J.; et al. Pacritinib (SB1518), a JAK2/FLT3 Inhibitor for the Treatment of Acute Myeloid Leukemia. Blood Cancer J. 2011, 1, e44. [Google Scholar] [CrossRef] [PubMed]
- William, A.D.; Lee, A.C.-H.; Blanchard, S.; Poulsen, A.; Teo, E.L.; Nagaraj, H.; Tan, E.; Chen, D.; Williams, M.; Sun, E.T.; et al. Discovery of the Macrocycle 11-(2-Pyrrolidin-1-Yl-Ethoxy)-14,19-Dioxa-5,7,26-Triaza-Tetracyclo[19.3.1.1(2,6).1(8,12)]Heptacosa-1(25),2(26),3,5,8,10,12(27),16,21,23-Decaene (SB1518), a Potent Janus Kinase 2/Fms-Like Tyrosine Kinase-3 (JAK2/FLT3) Inhibitor for the Treatment of Myelofibrosis and Lymphoma. J. Med. Chem. 2011, 54, 4638–4658. [Google Scholar] [CrossRef] [PubMed]
- Carter, R.B.; Wood, P.L.; Wieland, S.; Hawkinson, J.E.; Belelli, D.; Lambert, J.J.; White, H.S.; Wolf, H.H.; Mirsadeghi, S.; Tahir, S.H.; et al. Characterization of the Anticonvulsant Properties of Ganaxolone (CCD 1042; 3alpha-Hydroxy-3beta-Methyl-5alpha-Pregnan-20-One), a Selective, High-Affinity, Steroid Modulator of the Gamma-Aminobutyric Acid(A) Receptor. J. Pharmacol. Exp. Ther. 1997, 280, 1284–1295. [Google Scholar] [PubMed]
- Ganaxolone. Available online: https://www.drugs.com/monograph/ganaxolone.html (accessed on 15 November 2022).
- Reddy, D. Neurosteroid Compounds and Methods for Their Preparation and Use in Treating Central Nervous System Disorders. WO2019209850A1, 31 October 2019. [Google Scholar]
- Highlights of Prescribing Information. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215833s000lbl.pdf (accessed on 24 October 2022).
- Keam, S.J. Lutetium Lu 177 Vipivotide Tetraxetan: First Approval. Mol. Diagn. Ther. 2022, 26, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Benešová, M.; Schäfer, M.; Bauder-Wüst, U.; Afshar-Oromieh, A.; Kratochwil, C.; Mier, W.; Haberkorn, U.; Kopka, K.; Eder, M. Preclinical Evaluation of a Tailor-Made DOTA-Conjugated PSMA Inhibitor with Optimized Linker Moiety for Imaging and Endoradiotherapy of Prostate Cancer. J. Nucl. Med. 2015, 56, 914–920. [Google Scholar] [CrossRef]
- Eder, M.; Schäfer, M.; Bauder-Wüst, U.; Hull, W.-E.; Wängler, C.; Mier, W.; Haberkorn, U.; Eisenhut, M. 68 Ga-Complex Lipophilicity and the Targeting Property of a Urea-Based PSMA Inhibitor for PET Imaging. Bioconjugate Chem. 2012, 23, 688–697. [Google Scholar] [CrossRef]
- Hoy, S.M. Oteseconazole: First Approval. Drugs 2022, 82, 1017–1023. [Google Scholar] [CrossRef]
- Wirth, D.D.; Yates, C.M.; Hoekstra, W.J. Antifungal Compound Process. WO2017049096A1, 23 March 2017. [Google Scholar]
- Wirth, D.D.; Yates, C.M.; Hoekstra, W.J.; Bindl, M.F.; Hartmann, E. Antifungal Compound Process. WO2017049080A1, 23 March 2017. [Google Scholar]
- Keam, S.J. Mavacamten: First Approval. Drugs 2022, 82, 1127–1135. [Google Scholar] [CrossRef]
- Urquhart, L. FDA New Drug Approvals in Q2 2022. Nat. Rev. Drug Discov. 2022, 21, 550. [Google Scholar] [CrossRef]
- Oslob, J.; Anderson, R.; Aubele, D.; Evanchik, M.; Fox, J.C.; Kane, B.; Lu, P.; McDowell, R.; Rodriguez, H.; Song, Y.; et al. Pyrimidinedione Compounds. USOO9585883B2, 7 March 2017. [Google Scholar]
- Garnock-Jones, K.P. Vonoprazan: First Global Approval. Drugs 2015, 75, 439–443. [Google Scholar] [CrossRef]
- Sachs, G.; Shin, J.M.; Hunt, R. Novel Approaches to Inhibition of Gastric Acid Secretion. Curr. Gastroenterol. Rep. 2010, 12, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.-Y.; Zeng, H.; Yao, K.; Li, J.-Q.; Liu, Y. Novel and Practical Synthesis of Vonoprazan Fumarate. Synth. Commun. 2017, 47, 1169–1174. [Google Scholar] [CrossRef]
- Keam, S.J. Tapinarof Cream 1%: First Approval. Drugs 2022, 82, 1221–1228. [Google Scholar] [CrossRef] [PubMed]
- Richardson, W.H.; Schmidt, T.M.; Nealson, K.H. Identification of an Anthraquinone Pigment and a Hydroxystilbene Antibiotic from Xenorhabdus Luminescens. Appl. Environ. Microbiol. 1988, 54, 1602–1605. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Webster, J.; Li, J.; Hu, K.; Zhu, J. Anti-Inflammatory and Psoriasis Treatment and Protein Kinase Inhibition by Hydroxyltilbenes and Novel Stilbene Derivatives and Analogues. US20030171429A, 11 September 2003. [Google Scholar]
- Brown, A. FDA New Drug Approvals in Q3 2022. Nat. Rev. Drug Discov. 2022, 21, 788. [Google Scholar] [CrossRef]
- Mullard, A. First de Novo Deuterated Drug Poised for Approval. Nat. Rev. Drug Discov. 2022, 21, 623–625. [Google Scholar] [CrossRef]
- Wrobleski, S.T.; Moslin, R.; Lin, S.; Zhang, Y.; Spergel, S.; Kempson, J.; Tokarski, J.S.; Strnad, J.; Zupa-Fernandez, A.; Cheng, L.; et al. Highly Selective Inhibition of Tyrosine Kinase 2 (TYK2) for the Treatment of Autoimmune Diseases: Discovery of the Allosteric Inhibitor BMS-986165. J. Med. Chem. 2019, 62, 8973–8995. [Google Scholar] [CrossRef]
- Moslin, R.; Zhang, Y.; Wrobleski, S.T.; Lin, S.; Mertzman, M.; Spergel, S.; Tokarski, J.S.; Strnad, J.; Gillooly, K.; McIntyre, K.W.; et al. Identification of N -Methyl Nicotinamide and N -Methyl Pyridazine-3-Carboxamide Pseudokinase Domain Ligands as Highly Selective Allosteric Inhibitors of Tyrosine Kinase 2 (TYK2). J. Med. Chem. 2019, 62, 8953–8972. [Google Scholar] [CrossRef]
- Robic, C.; Port, M.; Rousseaux, O.; Louguet, S.; Fretellier, N.; Catoen, S.; Factor, C.; Le Greneur, S.; Medina, C.; Bourrinet, P.; et al. Physicochemical and Pharmacokinetic Profiles of Gadopiclenol: A New Macrocyclic Gadolinium Chelate With High T1 Relaxivity. Investig. Radiol. 2019, 54, 475–484. [Google Scholar] [CrossRef]
- Gadopiclenol: Another Milestone Achieved|Bracco Corporate. Available online: https://www.bracco.com/en/news/gadopiclenol-another-milestone-achieved (accessed on 24 October 2022).
- Napolitano, R.; Lattuada, L.; Baranyai, Z.; Guidolin, N.; Marazzi, G. Gadolinium Bearing Pcta-Based Contrast Agents. WO2020030618A1, 13 February 2020. [Google Scholar]
- Duggan, S. Omidenepag Isopropyl Ophthalmic Solution 0.002%: First Global Approval. Drugs 2018, 78, 1925–1929. [Google Scholar] [CrossRef]
- Aihara, M.; Ropo, A.; Lu, F.; Kawata, H.; Iwata, A.; Odani-Kawabata, N.; Shams, N. Intraocular Pressure-Lowering Effect of Omidenepag Isopropyl in Latanoprost Non-/Low-Responder Patients with Primary Open-Angle Glaucoma or Ocular Hypertension: The FUJI Study. Jpn. J. Ophthalmol. 2020, 64, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Hagihara, M.; Yoneda, K.; Okanari, E.; Shigetomi, M. Pharmaceutical Composition for Treating or Preventing Glaucoma. WO2010113957A1, 7 October 2010. [Google Scholar]
- Iwamura, A.; Tanaka, M.; Katsube, T.; Shigetomi, M.; Okasei, E.; Tokunaga, H. Medicine Containing Pyridylaminoacetic Acid Compound. JP2011057633A, 24 March 2011. [Google Scholar]
- Iwamura, R.; Tanaka, M.; Katsube, T.; Shigetomi, M.; Okanari, E.; Tokunaga, Y.; Fujiwara, H. Pyridylaminoacetic Acid Compound. WO2009113600A1, 17 September 2009. [Google Scholar]
- Relyvrio (Sodium Phenylbutyrate and Taurursodiol) FDA Approval History. Available online: https://www.drugs.com/history/relyvrio.html (accessed on 25 October 2022).
- Hagey, L.; Crombie, D.; Espinosa, E.; Carey, M.; Igimi, H.; Hofmann, A. Ursodeoxycholic Acid in the Ursidae: Biliary Bile Acids of Bears, Pandas, and Related Carnivores. J. Lipid Res. 1993, 34, 1911–1917. [Google Scholar] [CrossRef]
- Tonin, F.; Arends, I.W.C.E. Latest Development in the Synthesis of Ursodeoxycholic Acid (UDCA): A Critical Review. Beilstein J. Org. Chem. 2018, 14, 470–483. [Google Scholar] [CrossRef] [PubMed]
- Dayal, B.; Bhojawala, J.; Rapole, K.R.; Pramanik, B.N.; Ertel, N.H.; Shefer, S.; Salen, G. Chemical Synthesis, Structural Analysis, and Decomposition of N-Nitroso Bile Acid Conjugates. Bioorganic Med. Chem. 1996, 4, 885–890. [Google Scholar] [CrossRef]
- Meric-Bernstam, F.; Bahleda, R.; Hierro, C.; Sanson, M.; Bridgewater, J.; Arkenau, H.-T.; Tran, B.; Kelley, R.K.; Park, J.O.; Javle, M.; et al. Futibatinib, an Irreversible FGFR1–4 Inhibitor, in Patients with Advanced Solid Tumors Harboring FGF / FGFR Aberrations: A Phase I Dose-Expansion Study. Cancer Discov. 2022, 12, 402–415. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Geng, M.; Wang, Y.; Ai, J.; Fan, J.; Dai, Y.; Ding, J. New Compound Having Fgfr Inhibitory Activity and Preparation and Application Thereof. WO2017215485A1, 21 December 2017. [Google Scholar]
- Sootome, H. Therapeutic Agent for Fgfr Inhibitor-Resistant Cancer. WO2015008844A1, 22 January 2015. [Google Scholar]
- Ballatore, C.; Huryn, D.M.; Smith, A.B. Carboxylic Acid (Bio)Isosteres in Drug Design. ChemMedChem 2013, 8, 385–395. [Google Scholar] [CrossRef]
- Novel Drug Approvals for 2021|FDA. Available online: https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2021 (accessed on 25 October 2022).
- Novel Drug Approvals for 2020|FDA. Available online: https://www.fda.gov/drugs/new-drugs-fda-cders-new-molecular-entities-and-new-therapeutic-biological-products/novel-drug-approvals-2020 (accessed on 24 October 2022).
- Zhong, D.; Wu, D.; Zhang, Y.; Lu, Z.; Usman, M.; Liu, W.; Lu, X.; Liu, W.-B. Synthesis of Sultams and Cyclic N -Sulfonyl Ketimines via Iron-Catalyzed Intramolecular Aliphatic C–H Amidation. Org. Lett. 2019, 21, 5808–5812. [Google Scholar] [CrossRef]
- Mondal, S.; Malakar, S. Synthesis of Sulfonamide and Their Synthetic and Therapeutic Applications: Recent Advances. Tetrahedron 2020, 76, 131662. [Google Scholar] [CrossRef]
- Ovung, A.; Bhattacharyya, J. Sulfonamide Drugs: Structure, Antibacterial Property, Toxicity, and Biophysical Interactions. Biophys. Rev. 2021, 13, 259–272. [Google Scholar] [CrossRef]
- Carta, F.; Supuran, C.T.; Scozzafava, A. Sulfonamides and Their Isosters as Carbonic Anhydrase Inhibitors. Future Med. Chem. 2014, 6, 1149–1165. [Google Scholar] [CrossRef]
- Białk-Bielińska, A.; Stolte, S.; Matzke, M.; Fabiańska, A.; Maszkowska, J.; Kołodziejska, M.; Liberek, B.; Stepnowski, P.; Kumirska, J. Hydrolysis of Sulphonamides in Aqueous Solutions. J. Hazard. Mater. 2012, 221–222, 264–274. [Google Scholar] [CrossRef] [PubMed]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benedetto Tiz, D.; Bagnoli, L.; Rosati, O.; Marini, F.; Santi, C.; Sancineto, L. FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis. Pharmaceutics 2022, 14, 2538. https://doi.org/10.3390/pharmaceutics14112538
Benedetto Tiz D, Bagnoli L, Rosati O, Marini F, Santi C, Sancineto L. FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis. Pharmaceutics. 2022; 14(11):2538. https://doi.org/10.3390/pharmaceutics14112538
Chicago/Turabian StyleBenedetto Tiz, Davide, Luana Bagnoli, Ornelio Rosati, Francesca Marini, Claudio Santi, and Luca Sancineto. 2022. "FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis" Pharmaceutics 14, no. 11: 2538. https://doi.org/10.3390/pharmaceutics14112538
APA StyleBenedetto Tiz, D., Bagnoli, L., Rosati, O., Marini, F., Santi, C., & Sancineto, L. (2022). FDA-Approved Small Molecules in 2022: Clinical Uses and Their Synthesis. Pharmaceutics, 14(11), 2538. https://doi.org/10.3390/pharmaceutics14112538