Co-Delivery of M2e Virus-Like Particles with Influenza Split Vaccine to the Skin Using Microneedles Enhances the Efficacy of Cross Protection
Abstract
1. Introduction
2. Materials and Methods
2.1. Viruses, Influenza M2e5x VLPs, and Human Influenza Split Vaccine
2.2. Immunogold Cryogenic-Transmission Electron Microscopy of M2e5x VLPs
2.3. Preparation of MN Patches
2.4. Immunization and Challenge
2.5. Antibody Responses and Hemagglutinin Inhibition (HAI) Assay
2.6. Analysis of Antibodies in Bronchoalveolar Lavages after Challenge
2.7. Inflammatory Cytokine and Lung Viral Titer
2.8. Determination of T Cell Responses
2.9. Cross-Protective Efficacy Test of Immune Sera
2.10. Statistical Analysis
3. Results
3.1. M2e5x VLPs Present M2e Epitopes on the Particle Surfaces
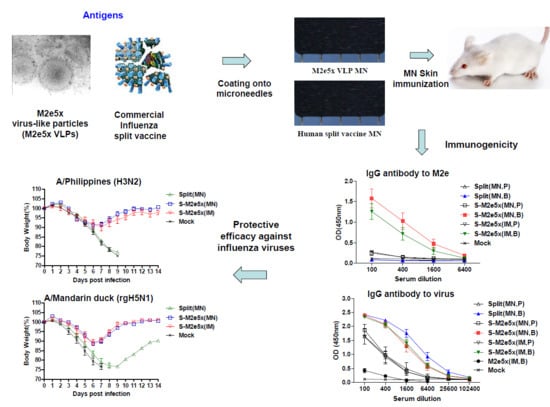
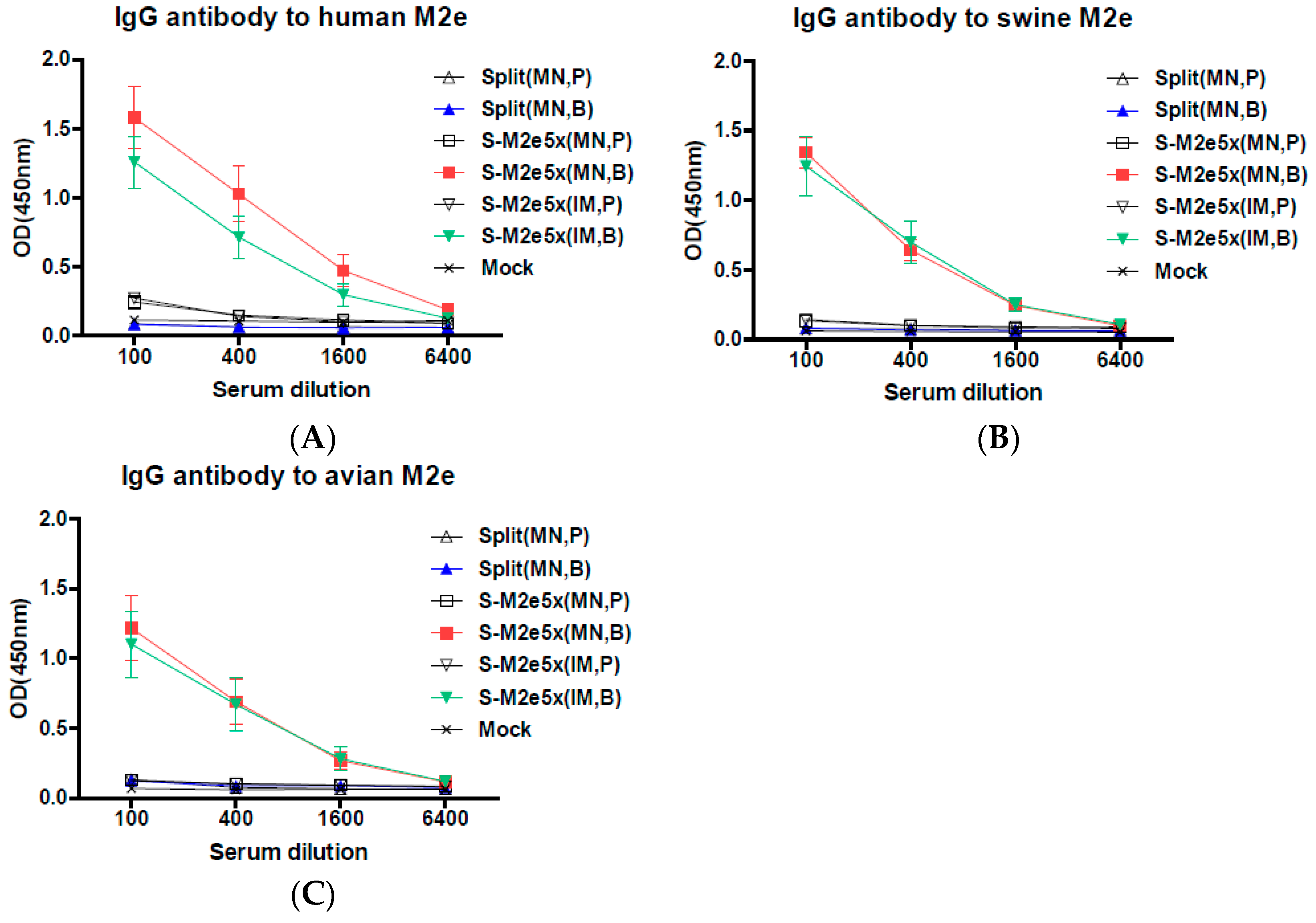
3.2. M2e5x VLP Co-Immunization with Split Vaccine in the Skin by MN Patch Enhances M2-Specific Antibody Responses
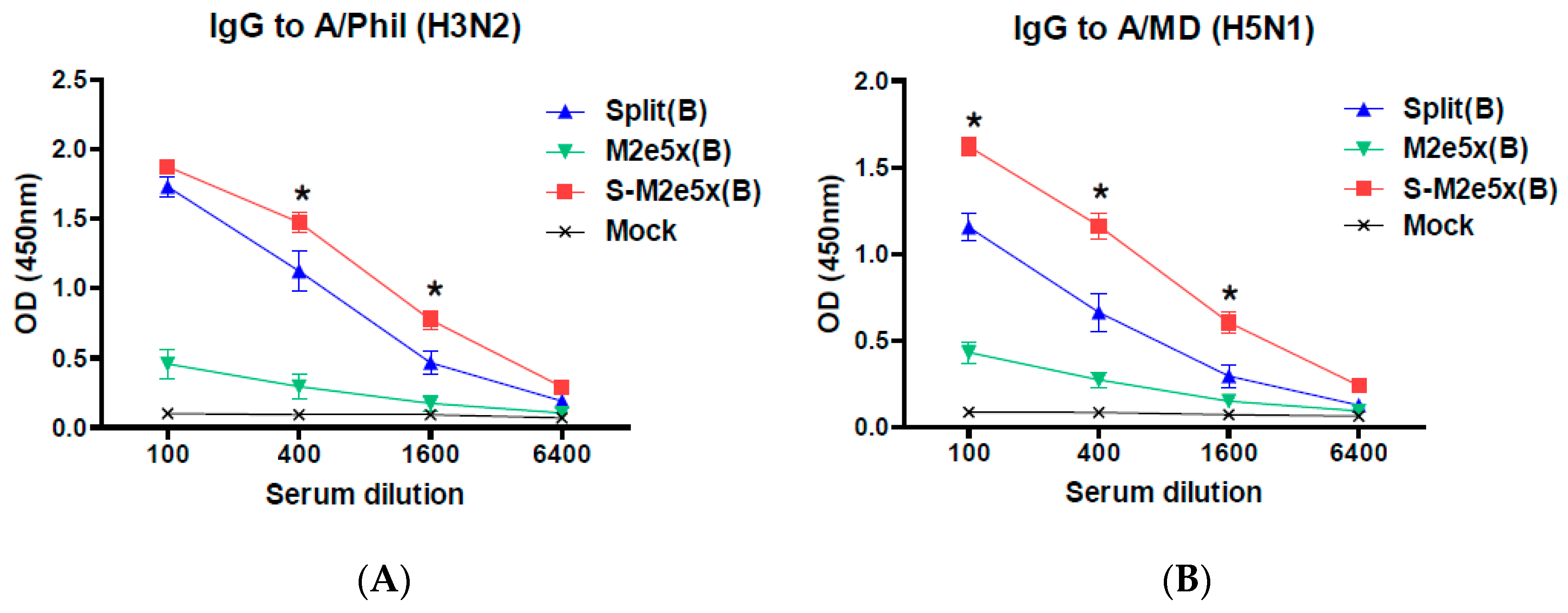
3.3. M2e5x VLP Co-Immunization with Split Vaccine Increases the Ratio of IgG2a/IgG1 Isotypes and Heterosubtypic Virus-Specific IgG Antibody Responses
3.4. M2e5x VLPs Co-Immunized with Split Vaccines Provide Enhanced Cross Protection
3.5. S-M2e5x VLP MN Co-Immunization Contributes to Inducing M2e Mucosal Antibodies
3.6. S-M2e5x VLP Co-Immunization Lowers Inflammatory Cytokine Levels and Lung Viral Titers
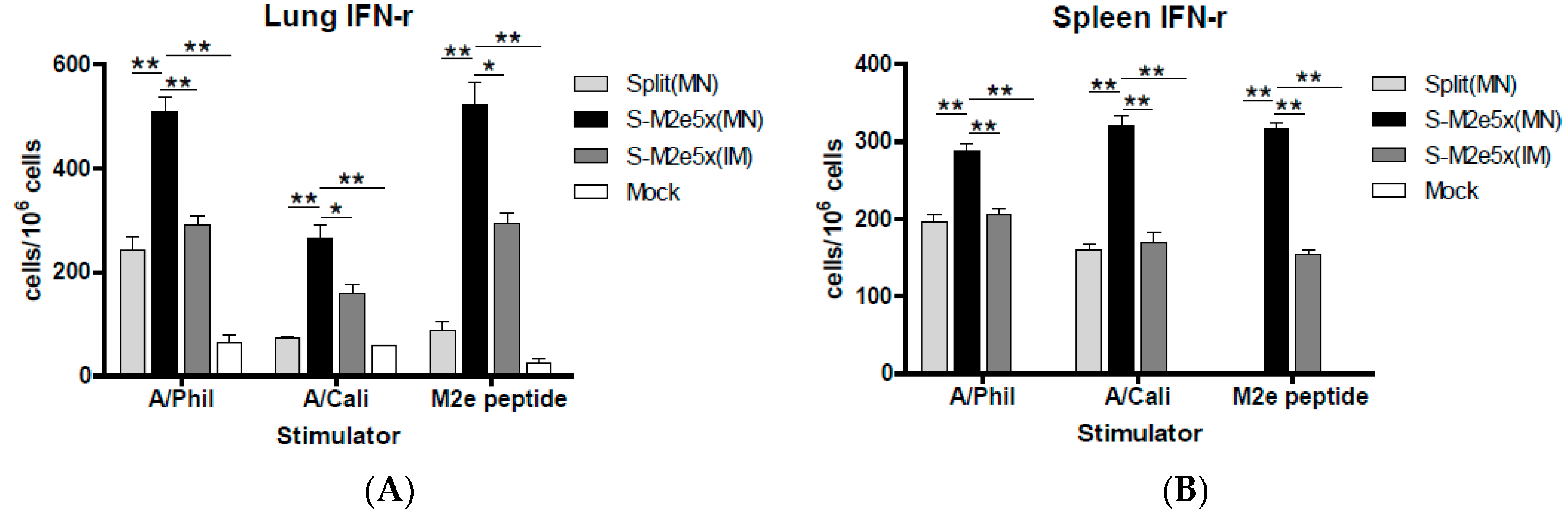
3.7. S-M2e5x VLP Co-Immunization Enhances IFN- γ Secreting T Cell Responses
3.8. S-M2e5x MN Co-Immune Sera Confer Enhanced Cross Protection.
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rabarison, J.H.; Tempia, S.; Harimanana, A.; Guillebaud, J.; Razanajatovo, N.H.; Ratsitorahina, M.; Heraud, J.M. Burden and Epidemiology of Influenza- and Respiratory Syncytial Virus-Associated Severe Acute Respiratory Illness Hospitalization in Madagascar, 2011-2016. Influenza Other Respir. Viruses 2018. [Google Scholar] [CrossRef] [PubMed]
- Viboud, C.; Miller, M.; Olson, D.; Osterholm, M.; Simonsen, L. Preliminary Estimates of Mortality and Years of Life Lost Associated with the 2009 A/H1N1 Pandemic in the US and Comparison with Past Influenza Seasons. PLoS Curr. 2010, 2, RRN1153. [Google Scholar] [CrossRef]
- Neumann, G.; Noda, T.; Kawaoka, Y. Emergence and pandemic potential of swine-origin H1N1 influenza virus. Nature 2009, 459, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.J.; Vijaykrishna, D.; Bahl, J.; Lycett, S.J.; Worobey, M.; Pybus, O.G.; Ma, S.K.; Cheung, C.L.; Raghwani, J.; Bhatt, S.; et al. Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature 2009, 459, 1122–1125. [Google Scholar] [CrossRef]
- Hancock, K.; Veguilla, V.; Lu, X.; Zhong, W.; Butler, E.N.; Sun, H.; Liu, F.; Dong, L.; DeVos, J.R.; Gargiullo, P.M.; et al. Cross-reactive antibody responses to the 2009 pandemic H1N1 influenza virus. N. Engl. J. Med. 2009, 361, 1945–1952. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, K.; Murphy, B.R.; Fauci, A.S. Development of effective vaccines against pandemic influenza. Immunity 2006, 24, 5–9. [Google Scholar] [CrossRef]
- Subbarao, K.; Matsuoka, Y. The prospects and challenges of universal vaccines for influenza. Trends Microbiol. 2013, 21, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zou, P.; Ding, J.; Lu, Y.; Chen, Y.H. Sequence comparison between the extracellular domain of M2 protein human and avian influenza A virus provides new information for bivalent influenza vaccine design. Microbes Infect. 2005, 7, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.C.; Lee, J.S.; Kwon, Y.M.; O, E.; Lee, Y.J.; Choi, J.G.; Wang, B.Z.; Compans, R.W.; Kang, S.M. Multiple heterologous M2 extracellular domains presented on virus-like particles confer broader and stronger M2 immunity than live influenza A virus infection. Antivir. Res. 2013, 99, 328–335. [Google Scholar] [CrossRef]
- Zhao, Z.; Ukidve, A.; Dasgupta, A.; Mitragotri, S. Transdermal immunomodulation: Principles, advances and perspectives. Adv. Drug Deliv. Rev. 2018, 127, 3–19. [Google Scholar] [CrossRef]
- Leone, M.; Monkare, J.; Bouwstra, J.A.; Kersten, G. Dissolving Microneedle Patches for Dermal Vaccination. Pharm. Res. 2017, 34, 2223–2240. [Google Scholar] [CrossRef] [PubMed]
- Shin, C.I.; Jeong, S.D.; Rejinold, N.S.; Kim, Y.C. Microneedles for vaccine delivery: Challenges and future perspectives. Ther. Deliv. 2017, 8, 447–460. [Google Scholar] [CrossRef] [PubMed]
- Prausnitz, M.R. Engineering Microneedle Patches for Vaccination and Drug Delivery to Skin. Annu. Rev. Chem. Biomol. Eng. 2017, 8, 177–200. [Google Scholar] [CrossRef] [PubMed]
- Rouphael, N.G.; Paine, M.; Mosley, R.; Henry, S.; McAllister, D.V.; Kalluri, H.; Pewin, W.; Frew, P.M.; Yu, T.; Thornburg, N.J.; et al. The safety, immunogenicity, and acceptability of inactivated influenza vaccine delivered by microneedle patch (TIV-MNP 2015): A randomised, partly blinded, placebo-controlled, phase 1 trial. Lancet 2017, 390, 649–658. [Google Scholar] [CrossRef]
- Fernando, G.J.P.; Hickling, J.; Jayashi Flores, C.M.; Griffin, P.; Anderson, C.D.; Skinner, S.R.; Davies, C.; Witham, K.; Pryor, M.; Bodle, J.; et al. Safety, tolerability, acceptability and immunogenicity of an influenza vaccine delivered to human skin by a novel high-density microprojection array patch (Nanopatch). Vaccine 2018, 36, 3779–3788. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Pewin, W.; Wang, C.; Luo, Y.; Gonzalez, G.X.; Mohan, T.; Prausnitz, M.R.; Wang, B.Z. A boosting skin vaccination with dissolving microneedle patch encapsulating M2e vaccine broadens the protective efficacy of conventional influenza vaccines. J. Control Release 2017, 261, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.C.; Lee, J.W.; Choi, H.J.; Lee, Y.N.; Hwang, H.S.; Lee, J.; Kim, C.; Lee, J.S.; Montemagno, C.; Prausnitz, M.R.; et al. Microneedle patch delivery to the skin of virus-like particles containing heterologous M2e extracellular domains of influenza virus induces broad heterosubtypic cross-protection. J. Control Release 2015, 210, 208–216. [Google Scholar] [CrossRef]
- Quan, F.S.; Compans, R.W.; Nguyen, H.H.; Kang, S.M. Induction of heterosubtypic immunity to influenza virus by intranasal immunization. J. Virol. 2008, 82, 1350–1359. [Google Scholar] [CrossRef]
- Kim, Y.C.; Quan, F.S.; Compans, R.W.; Kang, S.M.; Prausnitz, M.R. Formulation and coating of microneedles with inactivated influenza virus to improve vaccine stability and immunogenicity. J. Control Release 2010, 142, 187–195. [Google Scholar] [CrossRef]
- Quan, F.S.; Kim, Y.C.; Yoo, D.G.; Compans, R.W.; Prausnitz, M.R.; Kang, S.M. Stabilization of influenza vaccine enhances protection by microneedle delivery in the mouse skin. PLoS ONE 2009, 4, e7152. [Google Scholar] [CrossRef]
- Kim, M.C.; Lee, Y.N.; Ko, E.J.; Lee, J.S.; Kwon, Y.M.; Hwang, H.S.; Song, J.M.; Song, B.M.; Lee, Y.J.; Choi, J.G.; et al. Supplementation of influenza split vaccines with conserved M2 ectodomains overcomes strain specificity and provides long-term cross protection. Mol. Ther. 2014, 22, 1364–1374. [Google Scholar] [CrossRef]
- Wen, H.S.; Wang, S.P. Mouse adaptation of the Asian influenza virus. J. Infect. Dis. 1959, 105, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Song, J.M.; Hossain, J.; Yoo, D.G.; Lipatov, A.S.; Davis, C.T.; Quan, F.S.; Chen, L.M.; Hogan, R.J.; Donis, R.O.; Compans, R.W.; et al. Protective immunity against H5N1 influenza virus by a single dose vaccination with virus-like particles. Virology 2010, 405, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Song, J.M.; Van Rooijen, N.; Bozja, J.; Compans, R.W.; Kang, S.M. Vaccination inducing broad and improved cross protection against multiple subtypes of influenza A virus. Proc. Natl. Acad. Sci. USA 2011, 108, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Song, J.M.; Wang, B.Z.; Park, K.M.; Van Rooijen, N.; Quan, F.S.; Kim, M.C.; Jin, H.T.; Pekosz, A.; Compans, R.W.; Kang, S.M. Influenza virus-like particles containing M2 induce broadly cross protective immunity. PLoS ONE 2011, 6, e14538. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, D.G.; Bakkouri, K.E.; Schon, K.; Ramne, A.; Festjens, E.; Lowenadler, B.; Fiers, W.; Saelens, X.; Lycke, N. CTA1-M2e-DD: A novel mucosal adjuvant targeted influenza vaccine. Vaccine 2008, 26, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, D.G.; Omokanye, A.; Schon, K.; Wenzel, U.A.; Bernasconi, V.; Bemark, M.; Kolpe, A.; El Bakkouri, K.; Ysenbaert, T.; Deng, L.; et al. M2e-tetramer-specific memory CD4 T cells are broadly protective against influenza infection. Mucosal Immunol. 2018, 11, 273–289. [Google Scholar] [CrossRef] [PubMed]
- Gianfrani, C.; Oseroff, C.; Sidney, J.; Chesnut, R.W.; Sette, A. Human memory CTL response specific for influenza A virus is broad and multispecific. Hum. Immunol. 2000, 61, 438–452. [Google Scholar] [CrossRef]
- Deng, L.; Cho, K.J.; Fiers, W.; Saelens, X. M2e-Based Universal Influenza A Vaccines. Vaccines 2015, 3, 105–136. [Google Scholar] [CrossRef]
- Kim, M.C.; Song, J.M.; O, E.; Kwon, Y.M.; Lee, Y.J.; Compans, R.W.; Kang, S.M. Virus-like particles containing multiple M2 extracellular domains confer improved cross-protection against various subtypes of influenza virus. Mol. Ther. 2013, 21, 485–492. [Google Scholar] [CrossRef]
- Kim, M.C.; Lee, Y.N.; Hwang, H.S.; Lee, Y.T.; Ko, E.J.; Jung, Y.J.; Cho, M.K.; Kim, Y.J.; Lee, J.S.; Ha, S.H.; et al. Influenza M2 virus-like particles confer a broader range of cross protection to the strain-specific pre-existing immunity. Vaccine 2014, 32, 5824–5831. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nava, G.M.; Attene-Ramos, M.S.; Ang, J.K.; Escorcia, M. Origins of the new influenza A(H1N1) virus: Time to take action. Euro Surveill. 2009, 14. [Google Scholar] [CrossRef]
- Solovyov, A.; Palacios, G.; Briese, T.; Lipkin, W.I.; Rabadan, R. Cluster analysis of the origins of the new influenza A(H1N1) virus. Euro Surveill. 2009, 14, 19224. [Google Scholar] [CrossRef][Green Version]
- Nelson, M.I.; Vincent, A.L.; Kitikoon, P.; Holmes, E.C.; Gramer, M.R. Evolution of novel reassortant A/H3N2 influenza viruses in North American swine and humans, 2009-2011. J. Virol. 2012, 86, 8872–8878. [Google Scholar] [CrossRef] [PubMed]
- Esser, E.S.; Pulit-Penaloza, J.A.; Kalluri, H.; McAllister, D.; Vassilieva, E.V.; Littauer, E.Q.; Lelutiu, N.; Prausnitz, M.R.; Compans, R.W.; Skountzou, I. Microneedle patch delivery of influenza vaccine during pregnancy enhances maternal immune responses promoting survival and long-lasting passive immunity to offspring. Sci. Rep. 2017, 7, 5705. [Google Scholar] [CrossRef] [PubMed]
- Mistilis, M.J.; Joyce, J.C.; Esser, E.S.; Skountzou, I.; Compans, R.W.; Bommarius, A.S.; Prausnitz, M.R. Long-term stability of influenza vaccine in a dissolving microneedle patch. Drug Deliv. Transl. Res. 2017, 7, 195–205. [Google Scholar] [CrossRef]
- Nakatsukasa, A.; Kuruma, K.; Okamatsu, M.; Hiono, T.; Suzuki, M.; Matsuno, K.; Kida, H.; Oyamada, T.; Sakoda, Y. Potency of whole virus particle and split virion vaccines using dissolving microneedle against challenges of H1N1 and H5N1 influenza viruses in mice. Vaccine 2017, 35, 2855–2861. [Google Scholar] [CrossRef]
- Zhu, W.; Li, S.; Wang, C.; Yu, G.; Prausnitz, M.R.; Wang, B.Z. Enhanced Immune Responses Conferring Cross-Protection by Skin Vaccination With a Tri-Component Influenza Vaccine Using a Microneedle Patch. Front. Immunol. 2018, 9, 1705. [Google Scholar] [CrossRef]
- Wang, B.Z.; Gill, H.S.; He, C.; Ou, C.; Wang, L.; Wang, Y.C.; Feng, H.; Zhang, H.; Prausnitz, M.R.; Compans, R.W. Microneedle delivery of an M2e-TLR5 ligand fusion protein to skin confers broadly cross-protective influenza immunity. J. Control Release 2014, 178, 1–7. [Google Scholar] [CrossRef]
- Lee, Y.N.; Lee, Y.T.; Kim, M.C.; Gewirtz, A.T.; Kang, S.M. A Novel Vaccination Strategy Mediating the Induction of Lung-Resident Memory CD8 T Cells Confers Heterosubtypic Immunity against Future Pandemic Influenza Virus. J. Immunol. 2016, 196, 2637–2645. [Google Scholar] [CrossRef]
- Schotsaert, M.; Ysenbaert, T.; Neyt, K.; Ibanez, L.I.; Bogaert, P.; Schepens, B.; Lambrecht, B.N.; Fiers, W.; Saelens, X. Natural and long-lasting cellular immune responses against influenza in the M2e-immune host. Mucosal Immunol. 2013, 6, 276–287. [Google Scholar] [CrossRef]
- Zebedee, S.L.; Lamb, R.A. Influenza A virus M2 protein: Monoclonal antibody restriction of virus growth and detection of M2 in virions. J. Virol. 1988, 62, 2762–2772. [Google Scholar]
- Jegerlehner, A.; Schmitz, N.; Storni, T.; Bachmann, M.F. Influenza A vaccine based on the extracellular domain of M2: Weak protection mediated via antibody-dependent NK cell activity. J. Immunol. 2004, 172, 5598–5605. [Google Scholar] [CrossRef]
- Van den Hoecke, S.; Ehrhardt, K.; Kolpe, A.; El Bakkouri, K.; Deng, L.; Grootaert, H.; Schoonooghe, S.; Smet, A.; Bentahir, M.; Roose, K.; et al. Hierarchical and Redundant Roles of Activating FcgammaRs in Protection against Influenza Disease by M2e-Specific IgG1 and IgG2a Antibodies. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Lee, Y.N.; Lee, Y.T.; Kim, M.C.; Hwang, H.S.; Lee, J.S.; Kim, K.H.; Kang, S.M. Fc receptor is not required for inducing antibodies but plays a critical role in conferring protection after influenza M2 vaccination. Immunology 2014, 143, 300–309. [Google Scholar] [CrossRef]
- Deng, L.; Mohan, T.; Chang, T.Z.; Gonzalez, G.X.; Wang, Y.; Kwon, Y.M.; Kang, S.M.; Compans, R.W.; Champion, J.A.; Wang, B.Z. Double-layered protein nanoparticles induce broad protection against divergent influenza A viruses. Nat. Commun. 2018, 9, 359. [Google Scholar] [CrossRef]
- Mullarkey, C.E.; Bailey, M.J.; Golubeva, D.A.; Tan, G.S.; Nachbagauer, R.; He, W.; Novakowski, K.E.; Bowdish, D.M.; Miller, M.S.; Palese, P. Broadly Neutralizing Hemagglutinin Stalk-Specific Antibodies Induce Potent Phagocytosis of Immune Complexes by Neutrophils in an Fc-Dependent Manner. mBio 2016, 7. [Google Scholar] [CrossRef]
- Huber, V.C.; McKeon, R.M.; Brackin, M.N.; Miller, L.A.; Keating, R.; Brown, S.A.; Makarova, N.; Perez, D.R.; Macdonald, G.H.; McCullers, J.A. Distinct contributions of vaccine-induced immunoglobulin G1 (IgG1) and IgG2a antibodies to protective immunity against influenza. Clin. Vaccine Immunol. 2006, 13, 981–990. [Google Scholar] [CrossRef]
- Del Pilar Martin, M.; Weldon, W.C.; Zarnitsyn, V.G.; Koutsonanos, D.G.; Akbari, H.; Skountzou, I.; Jacob, J.; Prausnitz, M.R.; Compans, R.W. Local response to microneedle-based influenza immunization in the skin. mBio 2012, 3, e00012. [Google Scholar] [CrossRef]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.-C.; Kim, K.-H.; Lee, J.W.; Lee, Y.-N.; Choi, H.-J.; Jung, Y.-J.; Kim, Y.-J.; Compans, R.W.; Prausnitz, M.R.; Kang, S.-M. Co-Delivery of M2e Virus-Like Particles with Influenza Split Vaccine to the Skin Using Microneedles Enhances the Efficacy of Cross Protection. Pharmaceutics 2019, 11, 188. https://doi.org/10.3390/pharmaceutics11040188
Kim M-C, Kim K-H, Lee JW, Lee Y-N, Choi H-J, Jung Y-J, Kim Y-J, Compans RW, Prausnitz MR, Kang S-M. Co-Delivery of M2e Virus-Like Particles with Influenza Split Vaccine to the Skin Using Microneedles Enhances the Efficacy of Cross Protection. Pharmaceutics. 2019; 11(4):188. https://doi.org/10.3390/pharmaceutics11040188
Chicago/Turabian StyleKim, Min-Chul, Ki-Hye Kim, Jeong Woo Lee, Yu-Na Lee, Hyo-Jick Choi, Yu-Jin Jung, Yu-Jin Kim, Richard W. Compans, Mark R. Prausnitz, and Sang-Moo Kang. 2019. "Co-Delivery of M2e Virus-Like Particles with Influenza Split Vaccine to the Skin Using Microneedles Enhances the Efficacy of Cross Protection" Pharmaceutics 11, no. 4: 188. https://doi.org/10.3390/pharmaceutics11040188
APA StyleKim, M.-C., Kim, K.-H., Lee, J. W., Lee, Y.-N., Choi, H.-J., Jung, Y.-J., Kim, Y.-J., Compans, R. W., Prausnitz, M. R., & Kang, S.-M. (2019). Co-Delivery of M2e Virus-Like Particles with Influenza Split Vaccine to the Skin Using Microneedles Enhances the Efficacy of Cross Protection. Pharmaceutics, 11(4), 188. https://doi.org/10.3390/pharmaceutics11040188