Design Space and QbD Approach for Production of Drug Nanocrystals by Wet Media Milling Techniques
Abstract
1. Introduction
2. Milling Process
2.1. Stabilizers
2.2. Process Parameters in Milling
2.2.1. Frequency of Compressions and Contact Pressure
2.2.2. Energy Input
2.2.3. Milling Design
3. Quality by Design, QbD, Concept
3.1. QbD Approach for Nanosuspension Production
3.1.1. Quality Target Product Profile, QTPP
3.1.2. Determination of Critical Quality Attributes, CQAs
3.1.3. Defining CPPs
3.1.4. Establishment of Design Space
4. Discussion
Funding
Conflicts of Interest
References
- Müller, R.H.; Gohla, S.; Keck, C.M. State of the art of nanocrystals—Special features, production, nanocoxicology aspects and intracellular delivery. Eur. J. Pharm. Biopharm. 2011, 78, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zheng, Y.; Zhang, L.; Wang, Q.; Zhang, D. Stability of nanosuspensions in drug delivery. J. Control. Release 2013, 172, 1126–1141. [Google Scholar] [CrossRef] [PubMed]
- Tuomela, A.; Hirvonen, J.; Peltonen, L. Stabilizing agents for drug nanocrystals: Effect on bioavailability. Pharmaceutics 2016, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Peltonen, L.; Hirvonen, J. Drug nanocrystals—Versatile option for formulation of poorly soluble materials. Int. J. Pharm. 2018, 537, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Peltonen, L.; Hirvonen, J.; Laaksonen, T. Drug nanocrystals and nanosuspensions in medicine. In Handbook of Nanobiomedical Research, Fundamentals, Applications and Recent Developments, 1 Materials for Nanomedicine; Torchilin, V., Ed.; World Scientific: Singapore, 2014; Volume 3, pp. 169–197. [Google Scholar]
- Noyes, A.A.; Whitney, W.R. The rate of solution of solid substances in their own solutions. J. Am. Chem. Soc. 1897, 19, 930–934. [Google Scholar] [CrossRef]
- Valo, H.; Arola, S.; Laaksonen, P.; Torkkeli, M.; Peltonen, L.; Linder, M.B.; Serimaa, R.; Kuga, S.; Hirvonen, J.; Laaksonen, T. Drug release from nanoparticles embedded in four different nanofibrillar cellulose aerogels. Eur. J. Pharm. Sci. 2013, 50, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Darville, N.; Saarinen, J.; Isomäki, A.; Khriachtchev, L.; Cleeren, D.; Sterkens, P.; van Heerden, M.; Annaert, P.; Peltonen, L.; Santos, H.A.; et al. Multimodal non-linear optical imaging for the investigation of drug nano-/microcrystal-cell interactions. Eur. J. Pharm. Biopharm. 2015, 96, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Tuomela, A.; Liu, P.; Puranen, J.; Rönkkö, S.; Laaksonen, T.; Kalesnykas, G.; Oksala, O.; Ilkka, J.; Laru, J.; Järvinen, K.; et al. Brinzolamide nanocrystal formulations for ophthalmic delivery: Reduction of elevated intraocular pressure in vivo. Int. J. Pharm. 2014, 467, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Li, Y.; Wu, W. Injected nanocrystals for targeted drug delivery. Acta Pharm. Sin. B 2016, 6, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Gad, S.F.; Park, J.; Park, F.E.; Fetih, G.N.; Tous, S.S.; Lee, W.; Yeo, Y. Enhancing docetaxel delivery to multidrug-resistant cancer cells with albumin-coated nanocrystals. Mol. Pharm. 2018, 15, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Möschwitzer, J. Drug nanocrystals in the commercial pharmaceutical development process. Int. J. Pharm. 2013, 453, 142–156. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Yang, H.; Zhang, R.; Li, Y.; Duan, L. Preparation and in vitro-in vivo evaluation of teniposide nanosuspensions. Int. J. Pharm. 2015, 478, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Rong, X.; Laru, J.; van Veen, B.; Kiesvaara, J.; Hirvonen, J.; Laaksonen, T.; Peltonen, L. Nanosuspensions of poorly soluble drugs: Preparation and development by wet milling. Int. J. Pharm. 2011, 411, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Keck, C.M.; Müller, R.H. Drug nanocrystals of poorly soluble drugs produced by high pressure homogenization. Eur. J. Pharm. Biopharm. 2006, 62, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Van Eerdenbrugh, B.; Van den Mooter, G.; Augustijns, P. Top-down production of drug nanocrystals: Nanosuspension stabilization, miniaturization and transformation into solid products. Int. J. Pharm. 2008, 364, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Bilgili, E.; Afolabi, A. A combined microhydrodynamics-polymer adsorption analysis for elucidation of the roles of stabilizers in wet stirred media milling. Int. J. Pharm. 2012, 439, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Valo, H.; Kovalainen, M.; Laaksonen, P.; Häkkinen, M.; Auriola, S.; Peltonen, L.; Linder, M.; Järvinen, K.; Hirvonen, J.; Laaksonen, T. Immobilization of protein-coated drug nanoparticles in nanfibrillar cellulose matrices—Enhanced stability and release. J. Control. Release 2011, 156, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Gao, B.; Wang, J.; Wang, D.; Zhu, Z.; Qiao, Z.; Yang, G.; Nie, F. A novel preparation method for drug nanocrystals and characterization by ultrasonic spray-assisted electrostatic adsorption. Int. J. Nanomed. 2013, 8, 3927–3936. [Google Scholar] [CrossRef] [PubMed]
- Sinha, B.; Müller, R.H.; Möschwitzer, J.P. Bottom-up approaches for preparing drug nanocrystals: Formulations and factors affecting particle size. Int. J. Pharm. 2013, 453, 126–141. [Google Scholar] [CrossRef] [PubMed]
- De Waard, H.; Frijlink, H.W.; Hinrichs, W.L.J. Bottom-up preparation techniques for nanocrystals of lipophilic drugs. Pharm. Res. 2011, 28, 1220–1223. [Google Scholar] [CrossRef] [PubMed]
- Salazar, J.; Müller, R.H.; Mäschwitzer, J.P. Combinative particle size reduction technologies for the production of drug nanocrystals. J. Pharm. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Chen, X.; Zhang, L.; Lin, X.; Zhang, Y.; Tang, X.; Wang, Y. A combined bottom-up/top-down approach to prepare a sterile injectable nanosuspension. Int. J. Pharm. 2014, 472, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Attari, Z.; Kalvakuntla, S.; Reddy, M.S.; Deshpande, M.; Rao, C.M.; Koteshwara, K.B. Formulation and characterisation of nanosuspensions of BCS class II and IV drugs by combinative method. J. Exp. Nanosci. 2016, 11, 276–288. [Google Scholar] [CrossRef]
- Eon-Duval, A.; Valax, P.; Solacroup, T.; Broly, H.; Gleixner, R.; Le Strat, C.; Sutter, J. Application of the quality by design approach to the drug substance manufacturing process of an Fc fusion protein: Towards a global multi-step design space. J. Pharm. Sci. 2012, 101, 3604–3618. [Google Scholar] [CrossRef] [PubMed]
- Martin-Moe, S.; Lim, F.J.; Wong, R.L.; Sreedhara, A.; Sundaram, J.; Sane, S.U. A new roadmap for biopharmaceutical drug product development: Integrating development, validation, and quality by design. J. Pharm. Sci. 2011, 100, 3031–3043. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, H.; Read, E.; Agarabi, C.; Brorson, K.; Lute, S.; Yoon, S. A design space exploration for control of critical quality attributes of mAb. Int. J. Pharm. 2016, 512, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Qiao, Y.; Wu, Z. Nanosystem trends in drug delivery using quality-by-design concept. J. Control. Release 2017, 256, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, A. Variables affecting the fine grinding of minerals using stirred mills. Miner. Eng. 2003, 16, 337–345. [Google Scholar] [CrossRef]
- Liversidge, G.G.; Cundy, K.C.; Bishop, J.F.; Czekai, D.A. Surface Modified Drug Nanoparticles. U.S. Patent 5,145,684, 8 September 1992. [Google Scholar]
- Liversidge, G.G.; Cundy, K.C. Particle size reduction for improvement of oral bioavailability of hydrophobic drugs: I. Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int. J. Pharm. 1995, 125, 91–97. [Google Scholar] [CrossRef]
- Merisko-Liversidge, E.; Liversidge, G.G.; Cooper, E.R. Nanosizing: A formulation approach for poorly-water-soluble compounds. Eur. J. Pharm. Sci. 2003, 18, 113–120. [Google Scholar] [CrossRef]
- Cerdeira, A.M.; Mazzotti, M.; Gander, B. Miconazole nanosuspensions: Influence of formulation variables on particle size reduction and physical stability. Int. J. Pharm. 2010, 396, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Peltonen, L.; Hirvonen, J. Pharmaceutical nanocrystals by nanomilling: Critical process parameters, particle fracturing and stabilization methods. J. Pharm. Pharmacol. 2010, 62, 1569–1579. [Google Scholar] [CrossRef] [PubMed]
- Kevadiya, B.D.; Barvaliya, M.; Zhang, L.; Anovadiya, A.; Brahmbhatt, H.; Paul, P.; Tripathi, C. Fenofibrate nanocrystals embedded in oral strip-films for bioavailability enhancement. Bioengineering 2018, 5, 16. [Google Scholar] [CrossRef] [PubMed]
- Juhnke, M.; Berghausenm, J.; Timpe, C. Accelerated formulation development for nanomilled active pharmaceutical ingredients using a screening approach. Chem. Eng. Technol. 2010, 33, 1412–1418. [Google Scholar] [CrossRef]
- Li, M.; Azad, M.; Davé, R.; Bilgili, E. Nanomilling of drugs for bioavailability enhancement: A holistic formulation-process perspective. Pharmaceutics 2016, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; De Wulf, O.; Laru, J.; Heikkilä, T.; van Veen, B.; Kiesvaara, J.; Hirvonen, J.; Peltonen, L.; Laaksonen, T. Dissolution studies of poorly soluble drug nanosuspensions in non-sink conditions. AAPS PharmSciTech 2013, 14, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Juhnke, M.; Märtin, D.; John, E. Generation of wear during the production of drug nanosuspensions by wet media milling. Eur. J. Pharm. Biopharm. 2012, 81, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Yaragudi, N.; Li, M.; Afolabi, A.; Dave, R.; Bilgili, E. Sub-100 nm drug particle suspensions prepared via wet milling with low bead contamination through novel process intensification. Chem. Eng. Sci. 2015, 130, 207–220. [Google Scholar] [CrossRef]
- Becker, M.; Schwedes, J. Comminution of ceramics in stirred media mills and wear of grinding beads. Powder Technol. 1999, 105, 374–381. [Google Scholar] [CrossRef]
- Knieke, C.; Sommer, M.; Peukert, W. Identifying the apparent and true grinding limit. Powder Technol. 2009, 195, 25–30. [Google Scholar] [CrossRef]
- Breitung-Faes, S.; Kwade, A. Prediction of energy effective grinding conditions. Miner. Eng. 2013, 43–44, 36–43. [Google Scholar] [CrossRef]
- Kwade, A. Wet comminution in stirred media mills—Research and its practical application. Powder Technol. 1999, 105, 14–20. [Google Scholar] [CrossRef]
- Morales, J.O.; Watts, A.B.; McConville, J.T. Mechanical particle-size reduction techniques. In Formulating Poorly Water Soluble Drugs, 2nd ed.; Williams, R.O., III, Watts, A.B., Miller, D.A., Eds.; Springer: New York, NY, USA, 2016; pp. 165–213. [Google Scholar]
- Brosh, T.; Kalman, H.; Levy, A.; Peyron, I.; Ricard, F. DEM-CFD simulation of particle comminution in jet-mill. Powder Technol. 2014, 257, 104–112. [Google Scholar] [CrossRef]
- Colombo, M.; Orthmann, S.; Bellini, M.; Staufenbiel, S.; Bodmeier, R. Influence of drug brittleness, nanomilling time, and freeze-drying on the crystallinity of poorly water-soluble drugs and its implications for solubility enhancement. AAPS PharmSciTech 2017, 18, 2437–2445. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.S.; York, P.; Blagden, N. Preparation of hydrocortisone nanosuspension through a bottom-up nanoprecipitation technique using microfluidic reactors. Int. J. Pharm. 2009, 375, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Denny, W.A.; Garq, S. Effect of wet milling process on the solid state of indomethacin and simvastatin. Int. J. Pharm. 2009, 380, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.H.; Lamberto, D.J.; Liu, L.; Kwong, E.; Nelson, T.; Rhodes, T.; Bal, A. A new and improved method for the preparation of drug nanosuspension formulations using acoustic mixing technology. Int. J. Pharm. 2014, 473, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Siewert, C.; Moog, R.; Alex, R.; Kretzer, P.; Rothenhäusler, B. Process and scaling parameters for wet media milling in early phase drug development: A knowledge based approach. Eur. J. Pharm. Sci. 2018, 115, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Jog, R.; Shen, J.; Zolnik, B.; Sadrieh, N.; Burgess, D.J. In vitro and in vivo performance of different sized spray-dried crystalline itraconazole. J. Pharm. Sci. 2015, 104, 3018–3028. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, I.; Schenck, D.; Bose, S.; Liu, F.; Motto, M. Identification of critical process parameters and its interplay with nanosuspension formulation prepared by top down media milling technology—A QbD perspective. Pharm. Dev. Technol. 2013, 18, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Viitala, T.; Kartal-Hodzic, A.; Liang, H.; Laaksonen, T.; Hirvonen, J.; Peltonen, L. Interaction studies between indomethacin nanocrystals and PEO/PPO copolymer stabilizers. Pharm. Res. 2015, 32, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Krull, S.M.; Patel, H.V.; Li, M.; Bilgili, E.; Davé, R.N. Critical material attributes (CMAs) of strip films loaded with poorly water-soluble drug nanoparticles: I. Impact of plasticizer on film properties and dissolution. Eur. J. Pharm. Sci. 2016, 92, 146–155. [Google Scholar] [CrossRef] [PubMed]
- Muller, R.-H.; Moschwitzer, J.; Bushrab, F.-N. Manufacturing of nanoparticles by milling and homogenization techniques. In Nanoparticle Technology for Drug Delivery; Gupta, R.B., Kompella, U.B., Eds.; CRC Press: Boca Raton, FL, USA, 2006; pp. 21–51. [Google Scholar]
- Yadav, D.; Kumar, N. Nanonization of curcumin by antisolvent precipitation: Process development, characterization, freeze drying and stability performance. Int. J. Pharm. 2014, 477, 564–577. [Google Scholar] [CrossRef] [PubMed]
- Geng, T.; Banerjee, P.; Lu, Z.; Zoghbi, A.; Li, T.; Wang, B. Comparative study on stabilizing ability of food protein, non-ionic surfactant and anionic surfactant on BCS type II drug carvedilol loaded nanosuspension: Physicochemical and pharmacokinetic investigation. Eur. J. Pharm. Sci. 2017, 109, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Van Eerdenbrugh, B.; Froven, S.B.; Van Humbeeck, J.; Martens, J.A.; Augustijns, P.; Van den Mooter, G. Downscaling drug nanosuspension production: Processing aspects and physicochemical characterization. AAPS PharmSciTech 2009, 10, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Lestari, M.L.A.D.; Müller, R.H.; Möschwitzer, J.P. Systematic screening of different surface modifiers for the production of physically stable nanosuspensions. J. Pharm. Sci. 2015, 104, 1128–1140. [Google Scholar] [CrossRef] [PubMed]
- Van Eerdenbrugh, B.; Vermant, J.; Martens, J.A.; Froyen, L.; Van, H.J.; Augustijns, P.; Van den Mooter, G. A screening study of surface stabilization during the production of drug nanocrystals. J. Pharm. Sci. 2009, 98, 2091–2103. [Google Scholar] [CrossRef] [PubMed]
- Vidlarova, L.; Romero, G.B.; Hanus, J.; Stepanek, F.; Müller, R.H. Nanocrystals for dermal penetration enhancement—Effect of concentration and underlying mechanisms using curcumin as model. Eur. J. Pharm. Biopharm. 2016, 104, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Staufenbiel, S.; Rühl, E.; Bodmeier, R. In situ determination of the saturation solubility of nanocrystals of poorly soluble drugs for dermal application. Int. J. Pharm. 2017, 521, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, I.; Bose, S.; Vippagunta, R.; Harmon, F. Nanosuspension for improving the bioavailability of a poorly soluble drug and screening of stabilizing agents to inhibit crystal growth. Int. J. Pharm. 2011, 409, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, I.; Schenck, D.; Bose, S.; Ruegger, C. Optimization of formulation and process parameters for the production of nanosuspension be wet media milling technique: Effect of vitamin E TPGS and nanocrystal particle size on oral absorption. Eur. J. Pharm. Sci. 2012, 47, 718–728. [Google Scholar] [CrossRef] [PubMed]
- Molls, H.H.; Hornle, R. Third European Symposium on Size Reduction. Dechema Monogr. 1972, 69, 631–661. [Google Scholar]
- Sarnes, A.; Østergaard, J.; Jensen, S.S.; Aaltonen, J.; Rantanen, J.; Hirvonen, J.; Peltonen, L. Dissolution study of nanocrystal powders of a poorly soluble drug by UV imaging and channel flow methods. Eur. J. Pharm. Sci. 2013, 50, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Romeis, S.; Schmidt, J.; Peukert, W. Mechanochemical aspects in wet stirred media milling. Int. J. Miner. Process. 2016, 156, 24–31. [Google Scholar] [CrossRef]
- Afolabi, A.; Akinlabi, O.; Bilgili, E. Impact of process parameters on the breakage kinetics of poorly water-soluble drugs during wet stirred media milling: A microhydrodynamic view. Eur. J. Pharm. Sci. 2014, 51, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Hennart, S.L.A.; Domingues, M.C.; Wildeboer, W.J.; van Heen, P.; Meesters, G.M.H. Study of the process of stirred ball milling of poorly water soluble organic products using factorial design. Powder Technol. 2010, 198, 56–60. [Google Scholar] [CrossRef]
- Inkyo, M.; Tahara, T.; Iwaki, T.; Iskandar, F.; Hogan, C.J., Jr.; Okuyama, K. Experimental investigation of nanoparticle dispersion by beads milling with centrifugal bead separation. J. Colloid Interface Sci. 2006, 304, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Medarevic, D.; Djuris, J.; Ibric, S.; Mitric, M.; Kachrimanis, K. Optimization of formulation and process parameters for the production of carvedilol nanosuspension by wet media milling. Int. J. Pharm. 2018, 540, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Narayan, R.; Pednekar, A.; Bhuyan, D.; Gowda, C.; Koteshwara, K.B.; Nayak, U.Y. A top-down technique to improve the solubility and bioavailability of aceclofenac: In vitro and in vivo studies. Int. J. Nanomed. 2017, 12, 4921–4935. [Google Scholar] [CrossRef] [PubMed]
- Nakach, M.; Authelin, J.-R.; Perrin, M.-A.; Lakkireddy, H.R. Comparison of high pressure homogenization and stirred bead milling for the production of nano-crystalline suspensions. Int. J. Pharm. 2018, 547, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Toziopoulou, F.; Malamatari, M.; Nikolakakis, I.; Kachrimanis, K. Production of aprepitant nanocrystals by wet media milling and subsequent solidification. Int. J. Pharm. 2017, 533, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Singare, D.S.; Marella, S.; Gowthamrajan, K.; Kulkarni, G.T.; Vooturi, R.; Rao, P.S. Optimization of formulation and process variable of nanosuspension: An industrial perspective. Int. J. Pharm. 2010, 402, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Patel, G.V.; Patel, V.B.; Pathak, A.; Rajput, S.J. Nanosuspension of efavirenz for improved oral bioavailability: Formulation optimization, in vitro, in situ and in vivo evaluation. Drug Dev. Ind. Pharm. 2014, 40, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Srivalli, K.M.R.; Mishra, B. Drug nanocrystals: A way toward scale-up. Saudi Pharm. J. 2016, 24, 386–404. [Google Scholar] [CrossRef] [PubMed]
- Kwade, A. A stressing model for the description and optimization of grinding processes. Chem. Eng. Technol. 2003, 26, 199–205. [Google Scholar] [CrossRef]
- Takatsuka, T.; Endo, T.; Jianguo, Y.; Yuminoki, K.; Hashimoto, N. Nanosizing of poorly water soluble compounds using rotation/revolution mixer. Chem. Pharm. Bull. 2009, 57, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Yuminoki, K.; Tachibana, S.; Nishimura, Y.; Mori, H.; Takatsuka, T.; Hashimoto, N. Scaling up nano-milling of poorly water soluble compounds using a rotation/revolution pulverizer. Pharmazie 2016, 71, 56–64. [Google Scholar] [PubMed]
- Finkler, C.; Krummen, L. Introduction to the application of QbD principles for the development of monoclonal antibodies. Biologicals 2016, 44, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Hakemeyer, C.; McKnight, N.; St. John, R.; Meier, S.; Trexler-Schmidt, M.; Kelley, B.; Zetti, F.; Puskeiler, R.; Kleinjans, A.; Lim, F.; et al. Process characterization and design space definition. Biologicals 2016, 44, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Djuris, J.; Djuric, Z. Modeling in the quality by design environment: Regulatory requirements and recommendations for design space and control strategy appointment. Int. J. Pharm. 2017, 533, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Peltonen, L.; Strachan, C. Understanding critical quality attributes for nanocrystals from preparation to delivery. Molecules 2015, 20, 22286–22300. [Google Scholar] [CrossRef] [PubMed]
- Gora, S.; Mustafa, G.; Sahni, J.K.; Ali, J.; Baboota, S. Nanosizing of valsartan by high pressure homogenization to produce dissolution enhanced nanosuspension: Pharmacokinetics and pharmacodynamic study. Drug Deliv. 2016, 23, 930–940. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.R.; Ferreira, N.R.; Fangueiro, J.F.; Santos, A.C.; Veiga, F.J.; Cabral, C.; Silva, A.M.; Souto, E.B. Ibuprofen nanocrystals developed by 22 factorial design experiment: A new approach for poorly water-soluble drugs. Saudi Pharm. J. 2017, 25, 1117–1124. [Google Scholar] [CrossRef]
- Mishra, B.; Sahoo, J.; Dixit, P.K. Enhanced bioavailability of cinnarizine nanosuspensions by particle size engineering: Optimization and physicochemical investigations. Mater. Sci. Eng. C 2016, 63, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.; Sahoo, J.; Dixit, P.K. Formulation and process optimization of naproxen nanosuspensions stabilized by hydroxyl propyl methyl cellulose. Carbohydr. Polym. 2015, 127, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Ilevbare, G.A.; Liu, H.; Edgar, K.J.; Taylor, L.S. Inhibition of solution crystal growth of ritonavir by cellulose polymersdfactors influencing polymer effectiveness. CrystEngComm 2012, 14, 6503–6514. [Google Scholar] [CrossRef]
- Frank, K.J.; Boeck, G. Development of a nanosuspension for iv administration: From miniscale screening to a freeze dried formulation. Eur. J. Pharm. Sci. 2016, 87, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Iurian, S.; Bogdan, C.; Tomuta, I.; Szabo-Revesz, P.; Chvatal, A.; Leucuta, S.E.; Moldovan, M.; Ambrus, R. Development of oral lyophilisates containing meloxicam nanocrystals using QbD approach. Eur. J. Pharm. Sci. 2017, 104, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Shen, J.; Zolnik, B.; Sadrieh, N.; Burgess, D.J. Optimization and dissolution performance of spray-dried naproxen nano-crystals. Int. J. Pharm. 2015, 486, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Karakucuk, A.; Celebi, N.; Teksin, Z.S. Preparation of ritonavir nanosuspensions by microfluidization using polymeric stabilizers: I. A design of experiment approach. Eur. J. Pharm. Sci. 2016, 95, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Chogale, M.M.; Ghodake, V.N.; Patravale, V.B. Performance parameters and characterizations of nanocrystals: A brief review. Pharmaceutics 2016, 8, 26. [Google Scholar] [CrossRef] [PubMed]

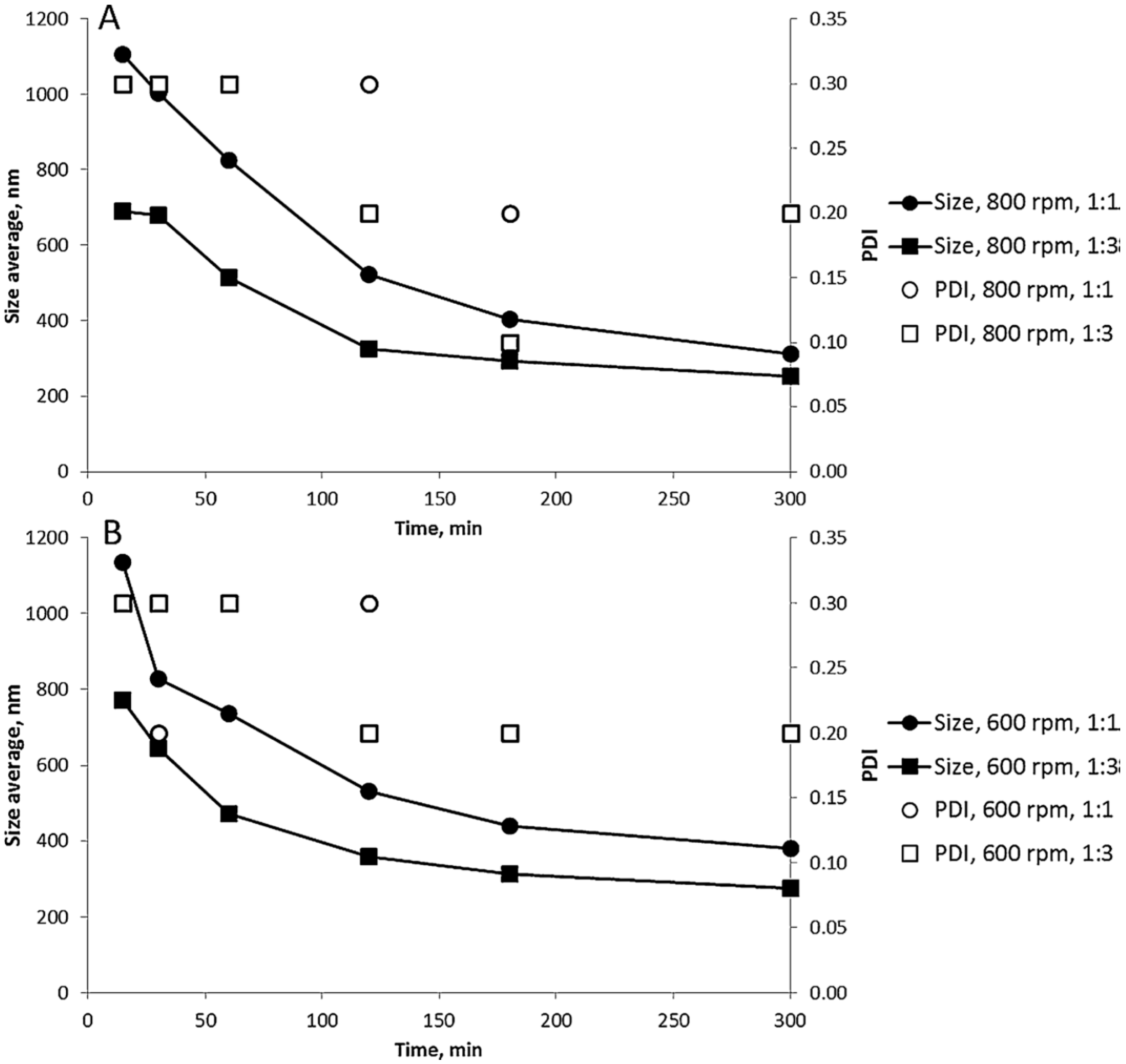
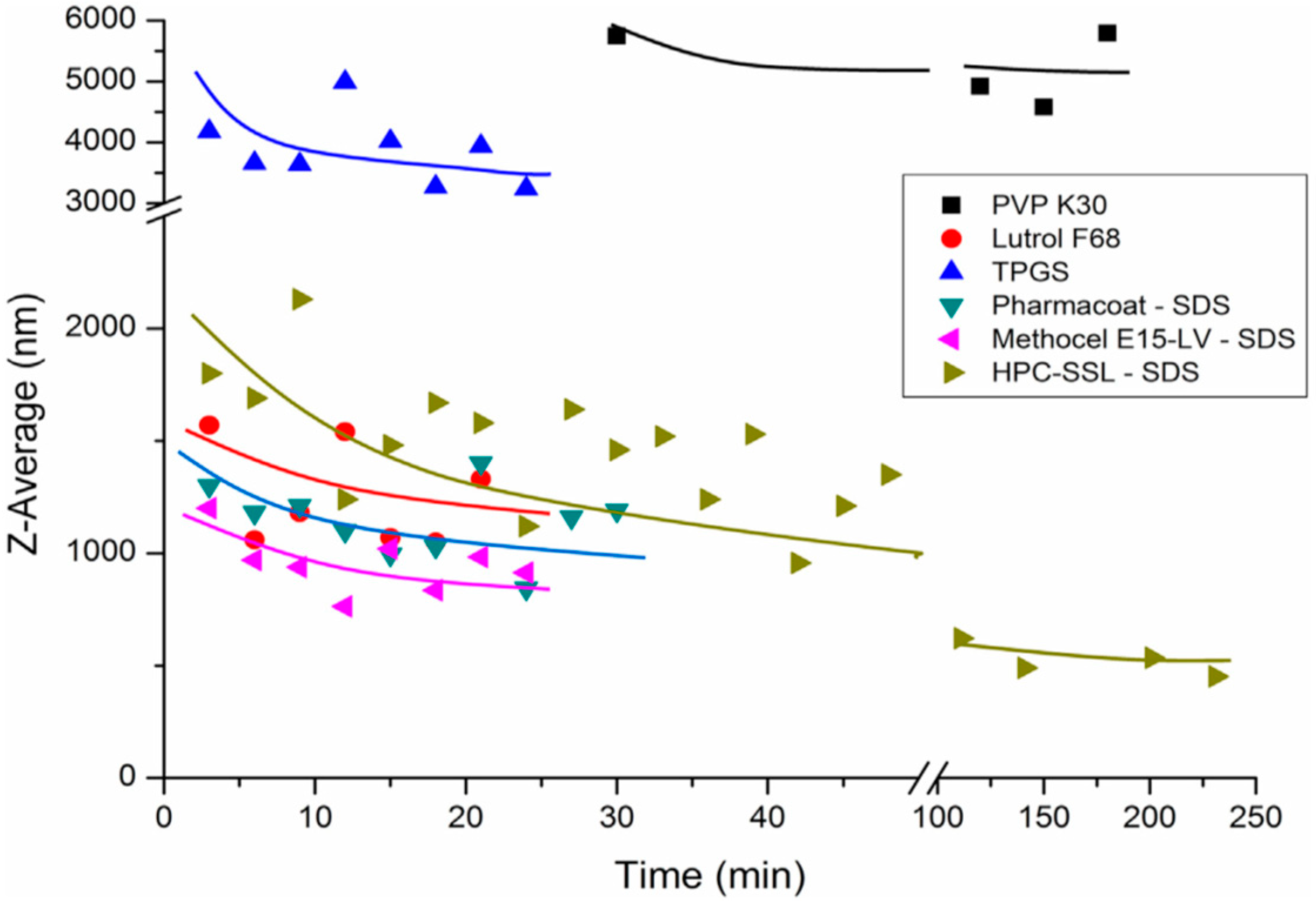
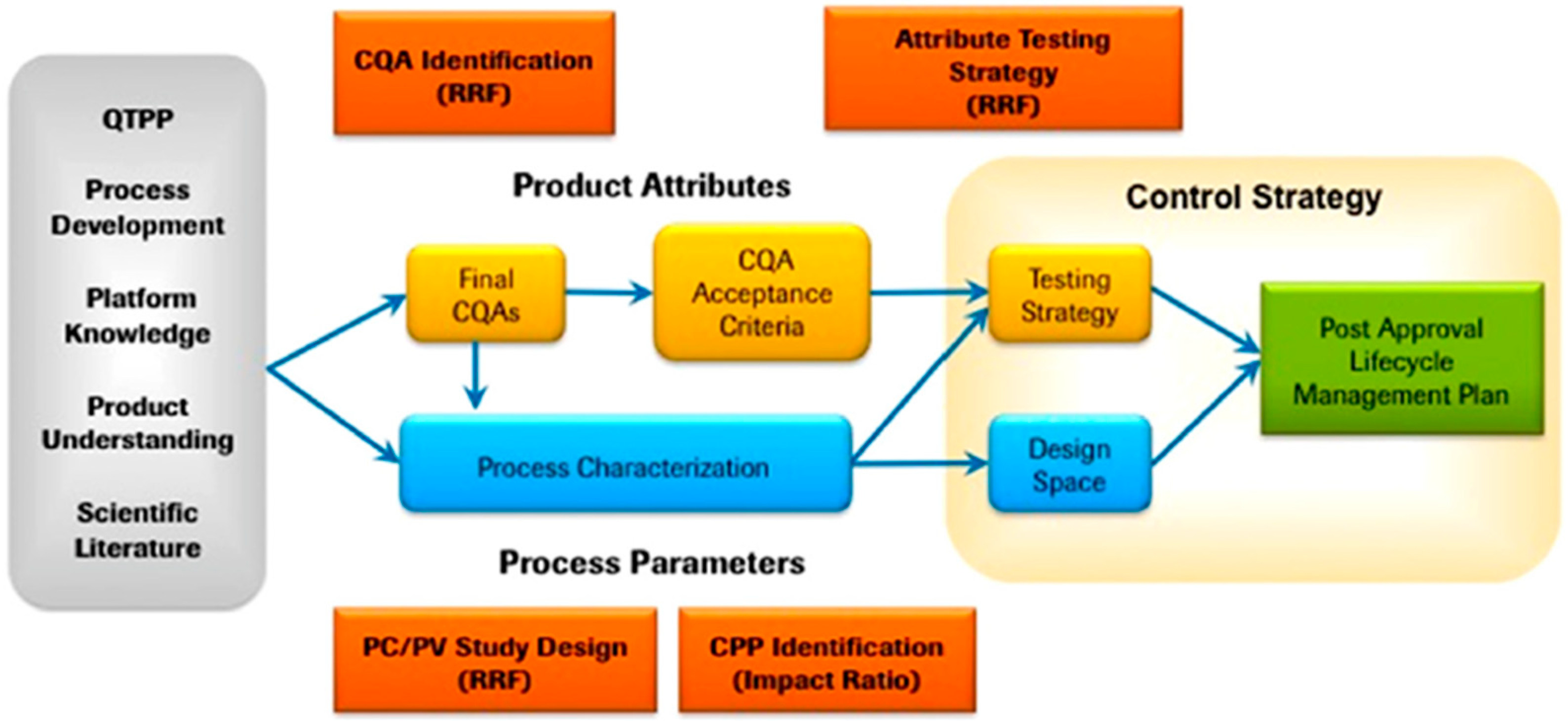
| Stabilizer | Drug | Reference |
|---|---|---|
| Methyl cellulose (MC), hydroxypropyl methylcellulose (HPMC), gelatin, sodium alginate, polyvinyl alcohol (PVA), polyvinyl pyrrolidone (PVP), polyethylene glycol (PEG), Tween 80, sodium deoxycholate, Pluronic F68, sodium dodecyl sulphate (SDS), sodium carboxymethyl cellulose (NaCMC), bovine serum albumin | Curcumin | [57] |
| Whey protein isolate, Poloxamer 188, SDS | Carvedilol | [58] |
| D-α-tocopherol polyethylene glycol 1000 succinate (TPGS) | Loviride, itraconazole, cinnarizine, indomethacin, mebendazole, naproxen, phenytoin | [59] |
| SDS, sodium docusate, sodium cholate, Tween 80, Dehyquart® A-CA, Poloxamer 188, TPGS, PVA, HPMC | Amphotericin B, curcumin, hesperetin, ibuprofen, resveratrol, rutoside trihydrate | [60] |
| PVP, PVA-PEG, Poloxamer 188, TPGS, Tween 80, HPMC, hydroxyethyl cellulose (HEC), hydroxypropyl cellulose (HPC), MC, NaCMC, sodium alginate | Loviride, itraconazole, cinnarizine, griseofulvin, indomethacin, mebendazole, naproxen, phenylbutazone, phenytoin | [61] |
| Pluronic® F68, Pluronic® 17R4, Tetronic® 908, Tetronic® 1107, Pluronic® L64 | Indomethacin | [54] |
| Plantacare 810 UP, Plantacare 1200 UP, Plantacare 2000 UP | Curcumin | [62] |
| Poloxamer 407, Poloxamer 188, vitamin E-TPGS, lecithin, Poloxamer 188 + lecithin, Plantacare 2000 UP | Dexamethasone, ibuprofen, tacrolimus | [63] |
| Parameter | Comments |
|---|---|
| Milling time | Commonly longer milling times lead to more monodisperse particles and decreased particle sizes; however, too long milling times may cause aggregation |
| Milling speed | Higher milling speed typically increase the milling rate and lead smaller particle sizes |
| Milling medium (bead size and the amount of beads) | Typically smaller beads lead to smaller nanocrystals; higher amount of beads improve the milling efficiency, though too high bead amount increase contamination |
| Drug amount | Very small drug amount increases contamination and lowers milling efficiency; very high drug amount increases viscosity and decreases milling efficiency |
| Milling design | Parameters can change a lot between different milling design: important especially in scaling-up/scaling-down |
| TPPs | Target | CQAs |
|---|---|---|
| Dosage form | Oral capsule | Identity, assay, content uniformity |
| Microbial quality | No contamination | Test for microbial growth |
| Product performance | Robust manufacturing | Drug content, particle size, polydispersity, morphology, solid state form, in vitro drug release |
| Pharmacokinetic target efficacy and safety | Better pharmacokinetic profile than pure drug | Pharmacokinetic parameters (Tmax, Cmax, AUC, MRT) |
| Stability | Stable at least 3 months at accelerated storage conditions | Drug content, particle size and polydispersity of redispersed sample |
| CQAs | Risk Assessment Matrix | |||||
|---|---|---|---|---|---|---|
| CMAs | CPPs | |||||
| Surfactant Type | Surfactant Concentration | Drug Amount | Milling Time | Milling Speed | Bead Size | |
| Particle size | Medium | Medium | Medium | High | High | High |
| PDI | Medium | High | Medium | High | High | High |
| Zeta potential | High | High | Medium | Medium | Medium | Medium |
| Drug content | Low | Low | Low | low | Low | Low |
| Drug release | Medium | Medium | Medium | High | High | High |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peltonen, L. Design Space and QbD Approach for Production of Drug Nanocrystals by Wet Media Milling Techniques. Pharmaceutics 2018, 10, 104. https://doi.org/10.3390/pharmaceutics10030104
Peltonen L. Design Space and QbD Approach for Production of Drug Nanocrystals by Wet Media Milling Techniques. Pharmaceutics. 2018; 10(3):104. https://doi.org/10.3390/pharmaceutics10030104
Chicago/Turabian StylePeltonen, Leena. 2018. "Design Space and QbD Approach for Production of Drug Nanocrystals by Wet Media Milling Techniques" Pharmaceutics 10, no. 3: 104. https://doi.org/10.3390/pharmaceutics10030104
APA StylePeltonen, L. (2018). Design Space and QbD Approach for Production of Drug Nanocrystals by Wet Media Milling Techniques. Pharmaceutics, 10(3), 104. https://doi.org/10.3390/pharmaceutics10030104





