Perspective on Global Measles Epidemiology and Control and the Role of Novel Vaccination Strategies
Abstract
:1. Introduction
2. MeV Infection and Pathogenesis
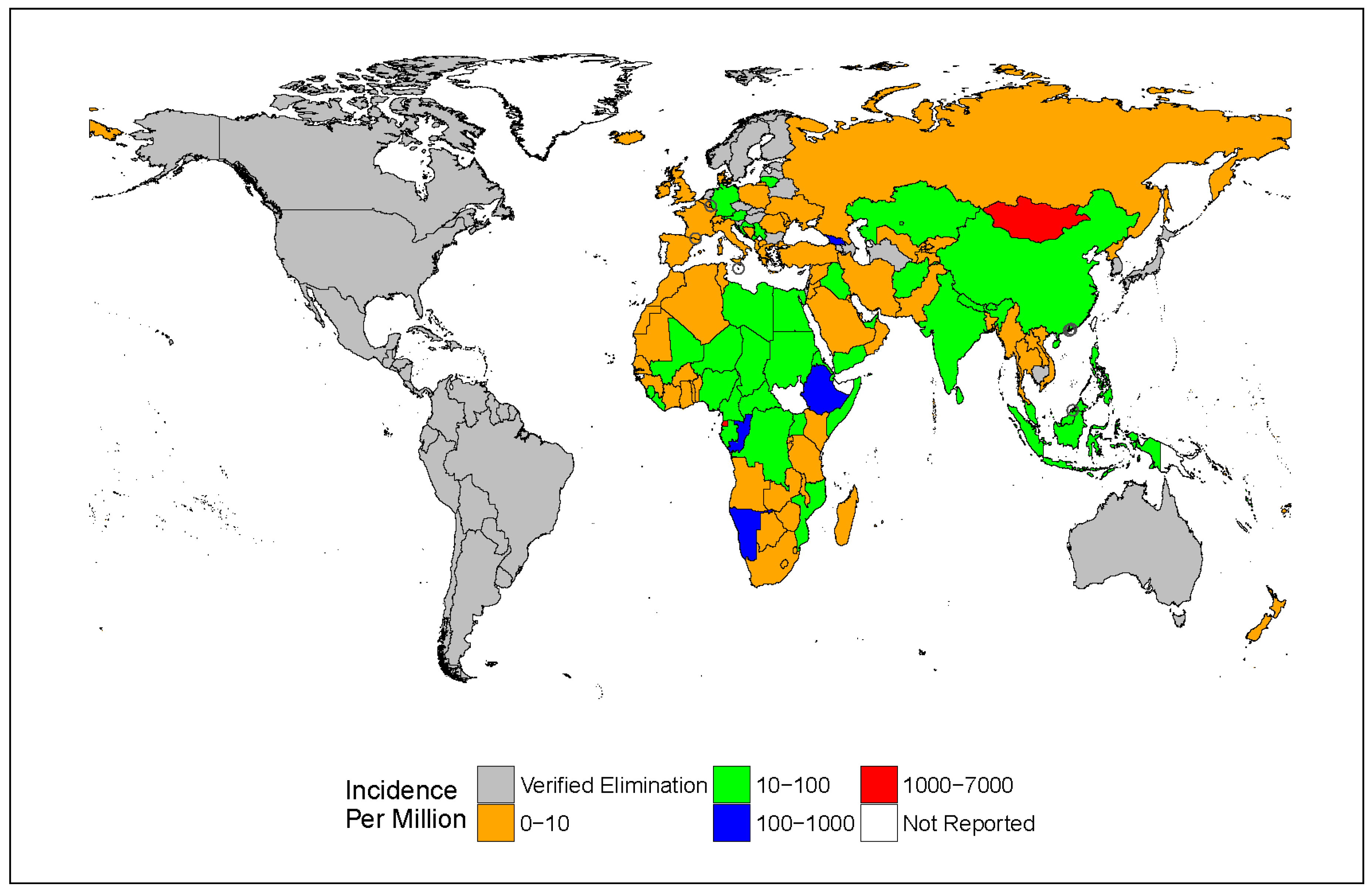
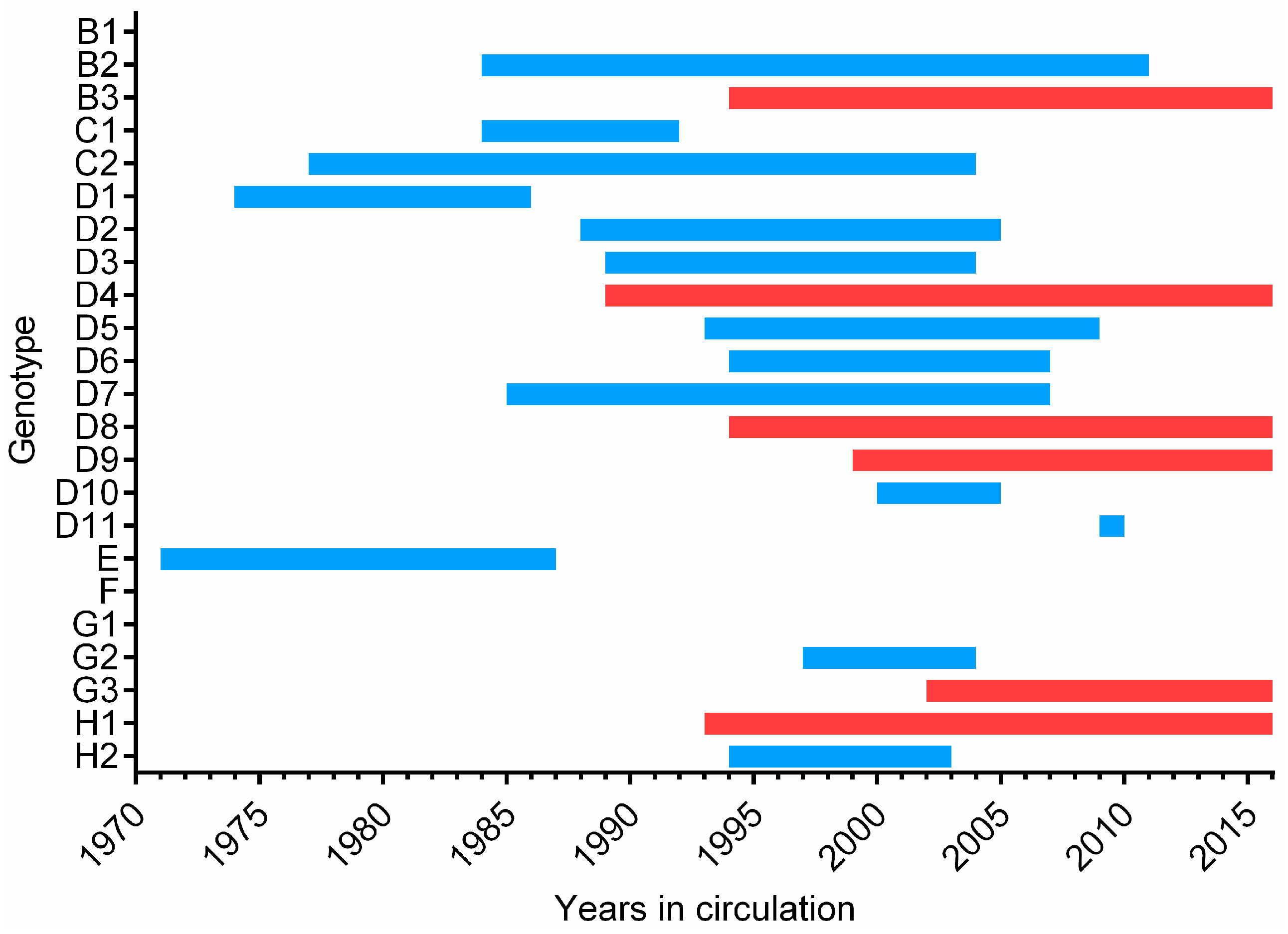
3. Epidemiology
4. Surveillance
5. Measles Vaccine
6. Novel Vaccination Strategies
7. Global Control and Prospects of Eradication
Acknowledgments
Data Statement
Conflicts of Interest
References
- Griffin, D.E. Paramyxoviridae: Measles Virus, 6th ed.; Lippincott Williams& Wilkins: Philadelphia, PA, USA, 2007; Volume 1, pp. 1043–1069. [Google Scholar]
- Liljeroos, L.; Huiskonen, J.T.; Ora, A.; Susi, P.; Butcher, S.J. Electron cryotomography of measles virus reveals how matrix protein coats the ribonucleocapsid within intact virions. Proc. Natl. Acad. Sci. USA 2011, 108, 18085–18090. [Google Scholar] [CrossRef] [PubMed]
- Tatsuo, H.; Ono, N.; Tanaka, K.; Yanagi, Y. Slam (CDw150) is a cellular receptor for measles virus. Nature 2000, 406, 893–897. [Google Scholar] [PubMed]
- Frenzke, M.; Sawatsky, B.; Wong, X.X.; Delpeut, S.; Mateo, M.; Cattaneo, R.; von Messling, V. Nectin-4-dependent measles virus spread to the cynomolgus monkey tracheal epithelium: Role of infected immune cells infiltrating the lamina propria. J. Virol. 2013, 87, 2526–2534. [Google Scholar] [CrossRef] [PubMed]
- Muhlebach, M.D.; Mateo, M.; Sinn, P.L.; Prufer, S.; Uhlig, K.M.; Leonard, V.H.; Navaratnarajah, C.K.; Frenzke, M.; Wong, X.X.; Sawatsky, B.; et al. Adherens junction protein nectin-4 is the epithelial receptor for measles virus. Nature 2011, 480, 530–533. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.K.; Li, N.; Mark, A.C.; Mateo, M.; Cattaneo, R.; Sinn, P.L. Cell-to-cell contact and nectin-4 govern spread of measles virus from primary human myeloid cells to primary human airway epithelial cells. J. Virol. 2016, 90, 6808–6817. [Google Scholar] [CrossRef] [PubMed]
- Hsu, E.C.; Sarangi, F.; Iorio, C.; Sidhu, M.S.; Udem, S.A.; Dillehay, D.L.; Xu, W.; Rota, P.A.; Bellini, W.J.; Richardson, C.D. A single amino acid change in the hemagglutinin protein of measles virus determines its ability to bind CD46 and reveals another receptor on marmoset B cells. J. Virol. 1998, 72, 2905–2916. [Google Scholar] [PubMed]
- Lecouturier, V.; Fayolle, J.; Caballero, M.; Carabana, J.; Celma, M.L.; Fernandez-Munoz, R.; Wild, T.F.; Buckland, R. Identification of two amino acids in the hemagglutinin glycoprotein of measles virus (MV) that govern hemadsorption, HeLa cell fusion, and CD46 downregulation: Phenotypic markers that differentiate vaccine and wild-type MV strains. J. Virol. 1996, 70, 4200–4204. [Google Scholar] [PubMed]
- Tahara, M.; Takeda, M.; Seki, F.; Hashiguchi, T.; Yanagi, Y. Multiple amino acid substitutions in hemagglutinin are necessary for wild-type measles virus to acquire the ability to use receptor CD46 efficiently. J. Virol. 2007, 81, 2564–2572. [Google Scholar] [CrossRef] [PubMed]
- Tahara, M.; Burckert, J.P.; Kanou, K.; Maenaka, K.; Muller, C.P.; Takeda, M. Measles virus hemagglutinin protein epitopes: The basis of antigenic stability. Viruses 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Rota, P.A.; Featherstone, D.A.; Bellini, W.J. Molecular epidemiology of measles virus. Curr. Top. Microbiol. Immunol. 2009, 330, 129–150. [Google Scholar] [PubMed]
- Tahara, M.; Ohno, S.; Sakai, K.; Ito, Y.; Fukuhara, H.; Komase, K.; Brindley, M.A.; Rota, P.A.; Plemper, R.K.; Maenaka, K.; et al. The receptor-binding site of the measles virus hemagglutinin protein itself constitutes a conserved neutralizing epitope. J. Virol. 2013, 87, 3583–3586. [Google Scholar] [CrossRef] [PubMed]
- Beaty, S.M.; Lee, B. Constraints on the genetic and antigenic variability of measles virus. Viruses 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Fulton, B.O.; Sachs, D.; Beaty, S.M.; Won, S.T.; Lee, B.; Palese, P.; Heaton, N.S. Mutational analysis of measles virus suggests constraints on antigenic variation of the glycoproteins. Cell Rep. 2015, 11, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Tahara, M.; Ito, Y.; Brindley, M.A.; Ma, X.; He, J.; Xu, S.; Fukuhara, H.; Sakai, K.; Komase, K.; Rota, P.A.; et al. Functional and structural characterization of neutralizing epitopes of measles virus hemagglutinin protein. J. Virol. 2013, 87, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.S.; Frenzke, M.; Leonard, V.H.; Welstead, G.G.; Richardson, C.D.; Cattaneo, R. Measles virus infection of alveolar macrophages and dendritic cells precedes spread to lymphatic organs in transgenic mice expressing human signaling lymphocytic activation molecule (SLAM, CD150). J. Virol. 2010, 84, 3033–3042. [Google Scholar] [CrossRef] [PubMed]
- Lemon, K.; de Vries, R.D.; Mesman, A.W.; McQuaid, S.; van Amerongen, G.; Yuksel, S.; Ludlow, M.; Rennick, L.J.; Kuiken, T.; Rima, B.K.; et al. Early target cells of measles virus after aerosol infection of non-human primates. PLoS Pathog. 2011, 7, e1001263. [Google Scholar] [CrossRef] [PubMed]
- Laksono, B.M.; de Vries, R.D.; McQuaid, S.; Duprex, W.P.; de Swart, R.L. Measles virus host invasion and pathogenesis. Viruses 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Kutty, P.; Rota, J.; Bellini, W.; Redd, S.; Barskey, A.; Wallace, G. Chapter 7: Measles. In VPD Surveillance Manual; Roush, S.W.A.L.M.B., Ed.; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2013. [Google Scholar]
- De Vries, R.D.; McQuaid, S.; van Amerongen, G.; Yuksel, S.; Verburgh, R.J.; Osterhaus, A.D.; Duprex, W.P.; de Swart, R.L. Measles immune suppression: Lessons from the macaque model. PLoS Pathog. 2012, 8, e1002885. [Google Scholar] [CrossRef] [PubMed]
- Tamashiro, V.G.; Perez, H.H.; Griffin, D.E. Prospective study of the magnitude and duration of changes in tuberculin reactivity during uncomplicated and complicated measles. Pediatr. Infect. Dis. J. 1987, 6, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Gremillion, D.H.; Crawford, G.E. Measles pneumonia in young adults. An analysis of 106 cases. Am. J. Med. 1981, 71, 539–542. [Google Scholar] [CrossRef]
- Mina, M.J.; Metcalf, C.J.; de Swart, R.L.; Osterhaus, A.D.; Grenfell, B.T. Long-term measles-induced immunomodulation increases overall childhood infectious disease mortality. Science 2015, 348, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Imdad, A.; Yakoob, M.Y.; Sudfeld, C.; Haider, B.A.; Black, R.E.; Bhutta, Z.A. Impact of vitamin a supplementation on infant and childhood mortality. BMC Public Health 2011, 11 (Suppl. 3). [Google Scholar] [CrossRef] [PubMed]
- Mahamud, A.; Burton, A.; Hassan, M.; Ahmed, J.A.; Wagacha, J.B.; Spiegel, P.; Haskew, C.; Eidex, R.B.; Shetty, S.; Cookson, S.; et al. Risk factors for measles mortality among hospitalized Somali refugees displaced by famine, Kenya, 2011. Clin. Infect. Dis. 2013, 57, e160–e166. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, R.; Bonthius, D.J. Measles virus and associated central nervous system sequelae. Semin. Pediatr. Neurol. 2012, 19, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Sugerman, D.E.; Barskey, A.E.; Delea, M.G.; Ortega-Sanchez, I.R.; Bi, D.; Ralston, K.J.; Rota, P.A.; Waters-Montijo, K.; LeBaron, C.W. Measles outbreak in a highly vaccinated population, San Diego, 2008: Role of the intentionally undervaccinated. Pediatrics 2010, 125, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.E. Measles virus and the nervous system. Handb. Clin. Neurol. 2014, 123, 577–590. [Google Scholar] [PubMed]
- Freeman, A.F.; Jacobsohn, D.A.; Shulman, S.T.; Bellini, W.J.; Jaggi, P.; de Leon, G.; Keating, G.F.; Kim, F.; Pachman, L.M.; Kletzel, M.; et al. A new complication of stem cell transplantation: Measles inclusion body encephalitis. Pediatrics 2004, 114, e657–e660. [Google Scholar] [CrossRef] [PubMed]
- Bellini, W.J.; Rota, J.S.; Lowe, L.E.; Katz, R.S.; Dyken, P.R.; Zaki, S.R.; Shieh, W.J.; Rota, P.A. Subacute sclerosing panencephalitis: More cases of this fatal disease are prevented by measles immunization than was previously recognized. J. Infect. Dis. 2005, 192, 1686–1693. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Beard, S.; Hunjan, R.; Brown, D.W.; Miller, E. Characterization of measles virus strains causing sspe: A study of 11 cases. J. Neurovirol. 2002, 8, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Rota, P.A.; Moss, W.J.; Takeda, M.; de Swart, R.L.; Thompson, K.M.; Goodson, J.L. Measles. Nat. Rev. Dis. Primers 2016, 2. [Google Scholar] [CrossRef] [PubMed]
- Bharti, N.; Tatem, A.J.; Ferrari, M.J.; Grais, R.F.; Djibo, A.; Grenfell, B.T. Explaining seasonal fluctuations of measles in niger using nighttime lights imagery. Science 2011, 334, 1424–1427. [Google Scholar] [CrossRef] [PubMed]
- Fine, P.E.; Clarkson, J.A. Measles in ·England and Wales—I: An analysis of factors underlying seasonal patterns. Int. J. Epidemiol. 1982, 11, 5–14. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The Number of Global Measles Cases Reported to Who. Available online: http://www.who.int/en/ (accessed on 8 November 2016).
- Waaijenborg, S.; Hahne, S.J.; Mollema, L.; Smits, G.P.; Berbers, G.A.; van der Klis, F.R.; de Melker, H.E.; Wallinga, J. Waning of maternal antibodies against measles, mumps, rubella, and varicella in communities with contrasting vaccination coverage. J. Infect. Dis. 2013, 208, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Simons, E.; Ferrari, M.; Fricks, J.; Wannemuehler, K.; Anand, A.; Burton, A.; Strebel, P. Assessment of the 2010 global measles mortality reduction goal: Results from a model of surveillance data. Lancet 2012, 379, 2173–2178. [Google Scholar] [CrossRef]
- Rentsen, T.; Enkhtuya, B.; Nymadawa, P.; Kobune, F.; Suzuki, K.; Yoshida, H.; Hachiya, M. Measles outbreak after a post-honeymoon period in Mongolia, 2001. Jpn. J. Infect. Dis. 2007, 60, 198–199. [Google Scholar] [PubMed]
- Wang, X.; Boulton, M.L.; Montgomery, J.P.; Carlson, B.; Zhang, Y.; Gillespie, B.; Wagner, A.L.; Ding, Y.; Luo, X.; Hong, T. The epidemiology of measles in Tianjin, China, 2005–2014. Vaccine 2015, 33, 6186–6191. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, H.; Xu, S.; Mao, N.; Zhu, Z.; Shi, J.; Huang, G.; Liu, C.; Bo, F.; Feng, D.; et al. Monitoring progress toward measles elimination by genetic diversity analysis of measles viruses in China 2009–2010. Clin. Microbiol. Infect. 2014, 20, O566–O577. [Google Scholar] [CrossRef] [PubMed]
- Rota, P.A.; Brown, K.; Mankertz, A.; Santibanez, S.; Shulga, S.; Muller, C.P.; Hubschen, J.M.; Siqueira, M.; Beirnes, J.; Ahmed, H.; et al. Global distribution of measles genotypes and measles molecular epidemiology. J. Infect. Dis. 2011, 204 (Suppl. 1), S514–S523. [Google Scholar] [CrossRef] [PubMed]
- Thompson, K.M. Evolution and use of dynamic transmission models for measles and rubella risk and policy analysis. Risk Anal. 2016, 36, 1383–1403. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, S.; Metcalf, C.J.E.; Ferrari, M.J.; Moss, W.J.; Truelove, S.A.; Tatem, A.J.; Grenfell, B.T.; Lessler, J. Reduced vaccination and the risk of measles and other childhood infections post-ebola. Science 2015, 347, 1240–1242. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.K.; Gacic-Dobo, M.; Strebel, P.M.; Dabbagh, A.; Mulders, M.N.; Okwo-Bele, J.; Dumolard, L.; Rota, P.A.; Kretsinger, K.; Goodson, J.L. Progress toward regional measles elimination—Worldwide, 2000–2015. Morb. Mortal. Wkly. Rep. 2016, 65, 1228–1233. [Google Scholar] [CrossRef] [PubMed]
- Six, C.; Blanes de Canecaude, J.; Duponchel, J.; Lafont, E.; Decoppet, A.; Travanut, M.; Pingeon, J.; Coulon, L.; Peloux-Petiot, F.; Grenier-Tisserant, P.; et al. Spotlight on measles 2010: Measles outbreak in the Provence-Alpes-Cote d’Azur region, France, January to November 2010—Substantial underreporting of cases: Measles outbreak in the Provence-Alpes-Cote d’ Azur region, France, January to November 2010—Substantial underreporting of cases. Euro Surveill. 2010, 15, 19754. [Google Scholar] [PubMed]
- Takla, A.; Wichmann, O.; Rieck, T.; Matysiak-Klose, D. Measles incidence and reporting trends in Germany, 2007–2011. Bull. World Health Organ. 2014, 92, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Simons, E.; Mort, M.; Dabbagh, A.; Strebel, P.; Wolfson, L. Strategic planning for measles control: Using data to inform optimal vaccination strategies. J. Infect. Dis. 2011, 204 (Suppl. 1), S28–S34. [Google Scholar] [CrossRef] [PubMed]
- Wolfson, L.J.; Grais, R.F.; Luquero, F.J.; Birmingham, M.E.; Strebel, P.M. Estimates of measles case fatality ratios: A comprehensive review of community-based studies. Int. J. Epidemiol. 2009, 38, 192–205. [Google Scholar] [CrossRef] [PubMed]
- Global control and regional elimination of measles, 2000–2012. Wkly. Epidemiol. Rec. 2014, 89, 45–52.
- Perry, R.T.; Murray, J.S.; Gacic-Dobo, M.; Dabbagh, A.; Mulders, M.N.; Strebel, P.M.; Okwo-Bele, J.M.; Rota, P.A.; Goodson, J.L. Progress toward regional measles elimination—Worldwide, 2000–2014. Morb. Mortal. Wkly. Rep. 2015, 64, 1246–1251. [Google Scholar] [CrossRef] [PubMed]
- Strebel, P.; World Health Organization, Geneva, Switzerland. Personal Communication, 2016.
- World Health Organization. Region of the Americas is Declared Free of Measles; World Health Organization: Washington, DC, USA, 2016. [Google Scholar]
- Pan Americas World Health Organization. 55th Directing Council 68th Session of the Region Committee of Who for the Americas; World Health Organization: Washington, DC, USA, 2016. [Google Scholar]
- World Health Organization Regianl Official for Europe. 4th Meeting of the European Regional Verification Commission for Measles and Rubella Elimination (RVC); World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization Western Pacific Region. Fourth Annual Meeting of the Regional Verification Commission for Measles Eminimation in the Western Pacific; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- World Health Organization Measles Surveillance Data. Available online: http://www.who.int/immunization/monitoring_surveillance/burden/vpd/surveillance_type/active/measles_monthlydata/en/index1.html (accessed on 8 November 2015).
- McCarthy, M. US sees first measles death in 12 years. BMJ 2015, 351. [Google Scholar] [CrossRef] [PubMed]
- Wyplosz, B.; Lafarge, M.; Escaut, L.; Stern, J.B. Fatal measles pneumonitis during Hodgkin’s lymphoma. BMJ Case Rep. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sun, L.Y.; Zhu, Z.J.; Lin, W.; Qu, W.; Zeng, Z.G. Measles virus infection in pediatric liver transplantation recipients. Transpl. Proc. 2015, 47, 2715–2718. [Google Scholar] [CrossRef] [PubMed]
- Mulders, M.N.; Rota, P.; Icenogle, J.P.; Brown, K.E.; Takeda, M.; Rey, G.J.; Ben Mamou, M.C.; Dosseh, A.; Byabamazima, C.R.; Ahmed, H.; et al. Global measles and rubella laboratory network support for elimination goals, 2010–2015. Morb. Mortal. Wkly. Rep. 2016, 65, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Update of the nomenclature for describing the genetic characteristics of wild-type measles viruses: New genotypes and reference strains. Wkly. Epidemiol. Rec. 2003, 78, 229–232.
- Genetic diversity of wild-type measles viruses and the global measles nucleotide surveillance database (means). Wkly. Epidemiol. Rec. 2015, 90, 373–380.
- Kessler, J.R.; Kremer, J.R.; Shulga, S.V.; Tikhonova, N.T.; Santibanez, S.; Mankertz, A.; Semeiko, G.V.; Samoilovich, E.O.; Tamfum, J.-J.M.; Pukuta, E.; et al. Revealing new measles virus transmission routes by use of sequence analysis of phosphoprotein and hemagglutinin genes. J. Clin. Microbiol. 2011, 49, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Gardy, J.L.; Naus, M.; Amlani, A.; Chung, W.; Kim, H.; Tan, M.; Severini, A.; Krajden, M.; Puddicombe, D.; Sahni, V.; et al. Whole-genome sequencing of measles virus genotypes H1 and D8 during outbreaks of infection following the 2010 Olympic winter games reveals viral transmission routes. J. Infect. Dis. 2015, 212, 1574–1578. [Google Scholar] [CrossRef] [PubMed]
- Penedos, A.R.; Myers, R.; Hadef, B.; Aladin, F.; Brown, K.E. Assessment of the utility of whole genome sequencing of measles virus in the characterisation of outbreaks. PLoS ONE 2015, 10, e0143081. [Google Scholar] [CrossRef] [PubMed]
- Rota, P.A.; Bankamp, B. Whole-genome sequencing during measles outbreaks. J. Infect. Dis. 2015, 212, 1529–1530. [Google Scholar] [CrossRef] [PubMed]
- Smits, G.P.; van Gageldonk, P.G.; Schouls, L.M.; van der Klis, F.R.; Berbers, G.A. Development of a bead-based multiplex immunoassay for simultaneous quantitative detection of IgG serum antibodies against measles, mumps, rubella, and varicella-zoster virus. Clin. Vaccine Immunol. 2012, 19, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.E.; Pan, C.H. Measles: Old vaccines, new vaccines. Curr. Top. Microbiol. Immunol. 2009, 330, 191–212. [Google Scholar] [PubMed]
- Rota, J.S.; Wang, Z.D.; Rota, P.A.; Bellini, W.J. Comparison of sequences of the H, F, and N coding genes of measles virus vaccine strains. Virus Res. 1994, 31, 317–330. [Google Scholar] [CrossRef]
- Bankamp, B.; Takeda, M.; Zhang, Y.; Xu, W.; Rota, P.A. Genetic characterization of measles vaccine strains. J. Infect. Dis. 2011, 204 (Suppl. 1), S533–S548. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Bankamp, B.; Xu, W.; Bellini, W.J.; Rota, P.A. The genomic termini of wild-type and vaccine strains of measles virus. Virus Res. 2006, 122, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Auwaerter, P.G.; Lee, S.H.; Nousari, H.C.; Valsamakis, A.; Leiferman, K.M.; Diwan, A.; Adams, R.J.; Griffin, D.E. Production of atypical measles in rhesus macaques: Evidence for disease mediated by immune complex formation and eosinophils in the presence of fusion-inhibiting antibody. Nat. Med. 1999, 5, 629–634. [Google Scholar] [PubMed]
- Aaby, P.; Jensen, T.G.; Hansen, H.L.; Kristiansen, H.; Tharup, J.; Poulsen, A.; Sodemann, M.; Jakobsen, M.; Knudsen, K.; Clotilde da Silva, M.; et al. Trial of high-dose Edmonston-Zagreb measles vaccine in Guinea-Bissau: Protective efficacy. Lancet 1988, 2, 809–811. [Google Scholar] [CrossRef]
- Whittle, H.; Hanlon, P.; O’Neill, K.; Hanlon, L.; Marsh, V.; Jupp, E.; Aaby, P. Trial of high-dose Edmonston-Zagreb measles vaccine in the Gambia: Antibody response and side-effects. Lancet 1988, 2, 811–814. [Google Scholar] [CrossRef]
- Aaby, P.; Jensen, H.; Samb, B.; Cisse, B.; Sodemann, M.; Jakobsen, M.; Poulsen, A.; Rodrigues, A.; Lisse, I.M.; Simondon, F.; et al. Differences in female-male mortality after high-titre measles vaccine and association with subsequent vaccination with diphtheria-tetanus-pertussis and inactivated poliovirus: Reanalysis of West African studies. Lancet 2003, 361, 2183–2188. [Google Scholar] [CrossRef]
- Aaby, P.; Ravn, H.; Benn, C.S.; Rodrigues, A.; Samb, B.; Ibrahim, S.A.; Libman, M.D.; Whittle, H.C. Randomized trials comparing inactivated vaccine after medium or high titer measles vaccine with standard titer measles vaccine after inactivated vaccine: A meta-analysis. Pediatr. Infect. Dis. J. 2016, 35, 1232–1241. [Google Scholar] [CrossRef] [PubMed]
- Aaby, P.; Kollmann, T.R.; Benn, C.S. Nonspecific effects of neonatal and infant vaccination: Public-health, immunological and conceptual challenges. Nat. Immunol. 2014, 15, 895–899. [Google Scholar] [CrossRef] [PubMed]
- Hales, C.M.; Johnson, E.; Helgenberger, L.; Papania, M.J.; Larzelere, M.; Gopalani, S.V.; Lebo, E.; Wallace, G.; Moturi, E.; Hickman, C.J.; et al. Measles outbreak associated with low vaccine effectiveness among adults in Pohnpei state, federated states of Micronesia, 2014. Open Forum Infect. Dis. 2016, 3. [Google Scholar] [CrossRef] [PubMed]
- Coughlin, M.M.; Collins, M.; Saxon, G.; Jarrahian, C.; Zehrung, D.; Cappello, C.; Dhere, R.; Royals, M.; Papania, M.; Rota, P.A. Effect of jet injection on infectivity of measles, mumps, and rubella vaccine in a bench model. Vaccine 2015, 33, 4540–4547. [Google Scholar] [CrossRef] [PubMed]
- Mitragotri, S. Current status and future prospects of needle-free liquid jet injectors. Nat. Rev. Drug Discov. 2006, 5, 543–548. [Google Scholar] [CrossRef] [PubMed]
- De Menezes Martins, R.; Curran, B.; Maia, M.d.S.; Ribeiro, M.d.G.T.; Camacho, L.A.B.; Freire, M.d.; Yoshida, A.M.; Siqueira, M.M.; Lemos, M.C.F.; de Albuquerque, E.M.; von Doellinger, V.d.R.; Homma, A.; Saganic, L.; Jarrahian, C.; Royalsi, M.; Zehrung, D. Immunogenicity and safety of measles-mumps-rubella vaccine delivered by disposable-syringe jet injector in healthy Brazilian infants: A randomized non-inferiority study. Contemp. Clin. Trials 2015, 41, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sarno, M.J.; Blase, E.; Galindo, N.; Ramirez, R.; Schirmer, C.L.; Trujillo-Juarez, D.F. Clinical immunogenicity of measles, mumps and rubella vaccine delivered by the injex jet injector: Comparison with standard syringe injection. Pediatr. Infect. Dis. J. 2000, 19, 839–842. [Google Scholar] [CrossRef] [PubMed]
- Griffin, D.E. Current progress in pulmonary delivery of measles vaccine. Expert Rev. Vaccines 2014, 13, 751–759. [Google Scholar] [CrossRef] [PubMed]
- MacLoughlin, R.J.; van Amerongen, G.; Fink, J.B.; Janssens, H.M.; Duprex, W.P.; de Swart, R.L. Optimization and dose estimation of aerosol delivery to non-human primates. J. Aerosol. Med. Pulm. Drug Deliv. 2016, 29, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Wong-Chew, R.M.; Garcia-Leon, M.L.; Espinosa-Torres Torrija, B.; Hernandez-Perez, B.; Cardiel-Marmolejo, L.E.; Beeler, J.A.; Audet, S.; Santos-Preciado, J.I. Increasing the time of exposure to aerosol measles vaccine elicits an immune response equivalent to that seen in 9-month-old Mexican children given the same dose subcutaneously. J. Infect. Dis. 2011, 204, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Ortega, J.L.; Bennett, J.V.; Castaneda-Desales, D.; Quintanilla, D.M.; Martinez, D.; de Castro, J.F. Booster immune response in children 6–7 years of age, randomly assigned to four groups with two MMR vaccines applied by aerosol or by injection. Vaccine 2014, 32, 3680–3686. [Google Scholar] [CrossRef] [PubMed]
- Low, N.; Bavdekar, A.; Jeyaseelan, L.; Hirve, S.; Ramanathan, K.; Andrews, N.J.; Shaikh, N.; Jadi, R.S.; Rajagopal, A.; Brown, K.E.; et al. A randomized, controlled trial of an aerosolized vaccine against measles. N. Engl. J. Med. 2015, 372, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Dilraj, A.; Cutts, F.T.; de Castro, J.F.; Wheeler, J.G.; Brown, D.; Roth, C.; Coovadia, H.M.; Bennett, J.V. Response to different measles vaccine strains given by aerosol and subcutaneous routes to schoolchildren: A randomised trial. Lancet 2000, 355, 798–803. [Google Scholar] [CrossRef]
- Dilraj, A.; Sukhoo, R.; Cutts, F.T.; Bennett, J.V. Aerosol and subcutaneous measles vaccine: Measles antibody responses 6 years after re-vaccination. Vaccine 2007, 25, 4170–4174. [Google Scholar] [CrossRef] [PubMed]
- Whittle, H.C.; Rowland, M.G.; Mann, G.F.; Lamb, W.H.; Lewis, R.A. Immunisation of 4–6 month old Gambian infants with Edmonston-Zagreb measles vaccine. Lancet 1984, 2, 834–837. [Google Scholar] [CrossRef]
- Bennett, J.V.; Fernandez de Castro, J.; Valdespino-Gomez, J.L.; Garcia-Garcia Mde, L.; Islas-Romero, R.; Echaniz-Aviles, G.; Jimenez-Corona, A.; Sepulveda-Amor, J. Aerosolized measles and measles-rubella vaccines induce better measles antibody booster responses than injected vaccines: Randomized trials in Mexican schoolchildren. Bull. World Health Organ. 2002, 80, 806–812. [Google Scholar] [PubMed]
- Sepulveda-Amor, J.; Valdespino-Gomez, J.L.; Garcia-Garcia Mde, L.; Bennett, J.; Islas-Romero, R.; Echaniz-Aviles, G.; de Castro, J.F. A randomized trial demonstrating successful boosting responses following simultaneous aerosols of measles and rubella (MR) vaccines in school age children. Vaccine 2002, 20, 2790–2795. [Google Scholar] [CrossRef]
- De Swart, R.L.; LiCalsi, C.; Quirk, A.V.; van Amerongen, G.; Nodelman, V.; Alcock, R.; Yuksel, S.; Ward, G.H.; Hardy, J.G.; Vos, H.; et al. Measles vaccination of macaques by dry powder inhalation. Vaccine 2007, 25, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.H.; Griffin, D.E.; Rota, P.A.; Papania, M.; Cape, S.P.; Bennett, D.; Quinn, B.; Sievers, R.E.; Shermer, C.; Powell, K.; et al. Successful respiratory immunization with dry powder live-attenuated measles virus vaccine in rhesus macaques. Proc. Natl. Acad. Sci. USA 2011, 108, 2987–2992. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, A.; Vaidya, V.; Mulay, R.; Agarkhedkar, S.; Shermer, C.; Collins, M.; Anderson, R.; Agarkhedkar, S.; Kulkarni, P.S.; Winston, S.; et al. Safety and immunogenicity of dry powder measles vaccine administered by inhalation: A randomized controlled phase I clinical trial. Vaccine 2014, 32, 6791–6797. [Google Scholar]
- Khanum, S.; Uddin, N.; Garelick, H.; Mann, G.; Tomkins, A. Comparison of Edmonston-Zagreb and Schwarz strains of measles vaccine given by aerosol or subcutaneous injection. Lancet 1987, 1, 150–153. [Google Scholar] [CrossRef]
- Wong-Chew, R.M.; Islas-Romero, R.; Garcia-Garcia Mde, L.; Beeler, J.A.; Audet, S.; Santos-Preciado, J.I.; Gans, H.; Lew-Yasukawa, L.; Maldonado, Y.A.; Arvin, A.M.; et al. Immunogenicity of aerosol measles vaccine given as the primary measles immunization to nine-month-old Mexican children. Vaccine 2006, 24, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Wong-Chew, R.M.; Islas-Romero, R.; Garcia-Garcia Mde, L.; Beeler, J.A.; Audet, S.; Santos-Preciado, J.I.; Gans, H.; Lew-Yasukawa, L.; Maldonado, Y.A.; Arvin, A.M.; et al. Induction of cellular and humoral immunity after aerosol or subcutaneous administration of Edmonston-Zagreb measles vaccine as a primary dose to 12-month-old children. J. Infect. Dis. 2004, 189, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Dilraj, A.; Cutts, F.T.; Bennett, J.V.; Fernandez de Castro, J.; Cohen, B.; Coovadia, H.M. Persistence of measles antibody two years after revaccination by aerosol or subcutaneous routes. Pediatr. Infect. Dis. J. 2000, 19, 1211–1213. [Google Scholar] [CrossRef] [PubMed]
- Edens, C.; Collins, M.L.; Goodson, J.L.; Rota, P.A.; Prausnitz, M.R. A microneedle patch containing measles vaccine is immunogenic in non-human primates. Vaccine 2015, 33, 4712–4718. [Google Scholar] [CrossRef] [PubMed]
- Mistilis, M.J.; Joyce, J.C.; Esser, E.S.; Skountzou, I.; Compans, R.W.; Bommarius, A.S.; Prausnitz, M.R. Long-term stability of influenza vaccine in a dissolving microneedle patch. Drug Deliv. Transl. Res. 2016, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Park, J.H.; Prausnitz, M.R. Microneedles for drug and vaccine delivery. Adv. Drug Deliv. Rev. 2012, 64, 1547–1568. [Google Scholar] [CrossRef] [PubMed]
- Hampton, L.M.; Farrell, M.; Ramirez-Gonzalez, A.; Menning, L.; Shendale, S.; Lewis, I.; Rubin, J.; Garon, J.; Harris, J.; Hyde, T.; et al. Cessation of trivalent oral poliovirus vaccine and introduction of inactivated poliovirus vaccine—Worldwide, 2016. Morb. Mortal. Wkly. Rep. 2016, 65, 934–938. [Google Scholar] [CrossRef] [PubMed]
- Mariner, J.C.; House, J.A.; Mebus, C.A.; Sollod, A.E.; Chibeu, D.; Jones, B.A.; Roeder, P.L.; Admassu, B.; van’t Klooster, G.G. Rinderpest eradication: Appropriate technology and social innovations. Science 2012, 337, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Proceedings of the global technical consultation to assess the feasibility of measles eradication, 28–30 July 2010. J. Infect. Dis. 2011, 204 (Suppl. 1), S4–S13. [Google Scholar]
- Strebel, P.M.; Cochi, S.L.; Hoekstra, E.; Rota, P.A.; Featherstone, D.; Bellini, W.J.; Katz, S.L. A world without measles. J. Infect. Dis. 2011, 204, S1–S3. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.B.; Goodson, J.L.; Chu, S.Y.; Rota, P.A.; Meltzer, M.I. Assessing the potential cost-effectiveness of microneedle patches in childhood measles vaccination programs: The case for further research and development. Drugs R D 2016, 16, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Garrison, L.P., Jr.; Bauch, C.T.; Bresnahan, B.W.; Hazlet, T.K.; Kadiyala, S.; Veenstra, D.L. Using cost-effectiveness analysis to support research and development portfolio prioritization for product innovations in measles vaccination. J. Infect. Dis. 2011, 204 (Suppl. 1), S124–S132. [Google Scholar] [CrossRef] [PubMed]
- Phadke, V.K.; Bednarczyk, R.A.; Salmon, D.A.; Omer, S.B. Association between vaccine refusal and vaccine-preventable diseases in the United States: A review of measles and pertussis. JAMA 2016, 315, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Notes from the field: Measles outbreak among members of a religious community—Brooklyn, New York, March–June 2013. Morb. Mortal. Wkly. Rep. 2013, 62, 752–753. [Google Scholar]
- Zipprich, J.; Winter, K.; Hacker, J.; Xia, D.; Watt, J.; Harriman, K. Measles outbreak—California, December 2014–February 2015. Morb. Mortal. Wkly. Rep. 2015, 64, 153–154. [Google Scholar]
- Gastanaduy, P.A.; Budd, J.; Fisher, N.; Redd, S.B.; Fletcher, J.; Miller, J.; McFadden, D.J., 3rd; Rota, J.; Rota, P.A.; Hickman, C.; et al. A measles outbreak in an underimmunized amish community in Ohio. N. Engl. J. Med. 2016, 375, 1343–1354. [Google Scholar] [CrossRef] [PubMed]
| Vaccine Formulation | Study Population | Immune Response b | Vaccine Virus Strain | Comments | Study Year c | Ref. |
|---|---|---|---|---|---|---|
| Aerosol | 4–6 month old infants First dose | Seroconversion d (↓) | Edmonston-Zagreb (Institute of Immunology, Zagreb) Schwarz (Smith-Kline-RIT) | Administered aerosol exposure for 10 s. Aerosol dose given at 10× SQ f dose with an assumed 10% delivery. Less than 50% seroconversion in SQ. | 1987 | [96] |
| Aerosol | 4–6 month old infants First dose | Seroconversion (~) | Edmonston-Zagreb (Institute of Immunology, Zagreb) | Longer exposure time higher Ab g. PRN titers lower than older infants in all groups. | 1984 | [90] |
| Aerosol | 9 month old infants First dose | Seroconversion seroprotection e and T cell response (↓) | Edmonston-Zagreb (SII) | Lower dose administered in aerosol group. | 2006 | [97] |
| Aerosol | 9 month old infants First dose | Seroconversion, seroprotection and T cell response (~) | Edmonston-Zagreb (SII) | Exposure time increased to 2.5 min. IFN-γ production equivalent. | 2011 | [85] |
| Aerosol | 9–11.9 month old infants First dose | Seroprotection and seroconversion (↓) | Edmonston-Zagreb (SII) | Administered aerosol exposure 30 s. Difference of 9.4% did not meet non-inferiority (5%) criteria. | 2015 | [87] |
| Aerosol | 12 month old infants First dose | Seroconversion, seroprotection and T cell response (↓) | Edmonston-Zagreb (Mexican National Institute of Virology) | Lower dose administered in aerosol group. All children boosted at 15 months SQ. IFN-γ production equivalent. | 2004 | [98] |
| Aerosol | 5–6 year old children | Seroconversion and Seroprotection (↑) | Edmonston-Zagreb (Swiss Serum and Vaccine Institute) | Booster dose. MR used. | 2002 | [92] |
| Aerosol | 6–7 year old children | Seroconversion and Seroprotection (↑) | Edmonston-Zagreb (SII) Attenuvax (Merk MMRII) | Aerosol delivery for 30 s sufficient for boosting response in school age children. MMR vaccine used. | 2014 | [86] |
| Aerosol | 6–8 year old children | Seroconversion (↑) | Edmonston-Zagreb (Swiss Serum and Vaccine Institute) | Aerosol delivery for 30 s M and MR vaccines used. Better boosting of Ab response in aerosol groups at all titers even “low dose” 1000 pfu. | 2002 | [91] |
| Aerosol | 5–14 year old children | Seroconversion (↑) with Edmonston-Zagreb Seroconversion (↓) with Schwarz | Schwarz Edmonston-Zagreb (SmithKline Beecham) | Administered aerosol exposure 30 s. Schwarz vaccine shown to lose potency following 2 min nebulization. Aerosol delivery boosted response in school aged children. | 2000 | [88] |
| Aerosol | 2 year follow up previous study | Seroconversion Maintenance (↑) with Edmonston-Zagreb | 86% of children from initial study included; 6% titers below seropositive cutoff compared to 13%–19% SQ. | 2000 | [99] | |
| Aerosol | 6 year follow up previous study | Ab titers and proportion seroprotected (↑) | 70% of children from initial study included. | 2007 | [89] | |
| Dry powder | Cynomolgus macaques | Ab response (↓) Upon challenge secondary response peaked earlier | Edmonston-Zagreb (Birmex or Berna) | Iron tracer study demonstrated most of dose did not reach deep lung. | 2007 | [93] |
| Dry powder | Rhesus macaques | Ab response (~) T cell response (↑) Protection from challenge Memory response | Edmonston-Zagreb (SII) | Puffhaler® and BD Solovent® administration comparable Smaller particle size dry powder. | 2011 | [94] |
| Dry powder | 18–45 year old seropositive males | Ab response (~) | Edmonston-Zagreb (SII) | Administration by Puffhaler® and BD Solovent® devices was well-tolerated, no adverse events reported. Baseline titer high, seroconversion only detectable in 20%–30% individuals. | 2014 | [95] |
| Microneedle | Rhesus macaques | Ab titer (~) | Edmonston-Zagreb (SII) | Increased stabilization. No site specific reactions noted. | 2015 | [100] |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coughlin, M.M.; Beck, A.S.; Bankamp, B.; Rota, P.A. Perspective on Global Measles Epidemiology and Control and the Role of Novel Vaccination Strategies. Viruses 2017, 9, 11. https://doi.org/10.3390/v9010011
Coughlin MM, Beck AS, Bankamp B, Rota PA. Perspective on Global Measles Epidemiology and Control and the Role of Novel Vaccination Strategies. Viruses. 2017; 9(1):11. https://doi.org/10.3390/v9010011
Chicago/Turabian StyleCoughlin, Melissa M., Andrew S. Beck, Bettina Bankamp, and Paul A. Rota. 2017. "Perspective on Global Measles Epidemiology and Control and the Role of Novel Vaccination Strategies" Viruses 9, no. 1: 11. https://doi.org/10.3390/v9010011
APA StyleCoughlin, M. M., Beck, A. S., Bankamp, B., & Rota, P. A. (2017). Perspective on Global Measles Epidemiology and Control and the Role of Novel Vaccination Strategies. Viruses, 9(1), 11. https://doi.org/10.3390/v9010011






