Rapid Amplicon Nanopore Sequencing (RANS) for the Differential Diagnosis of Monkeypox Virus and Other Vesicle-Forming Pathogens
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines and Virus-Infected Samples
2.2. Clinical Samples
2.3. DNA Extraction
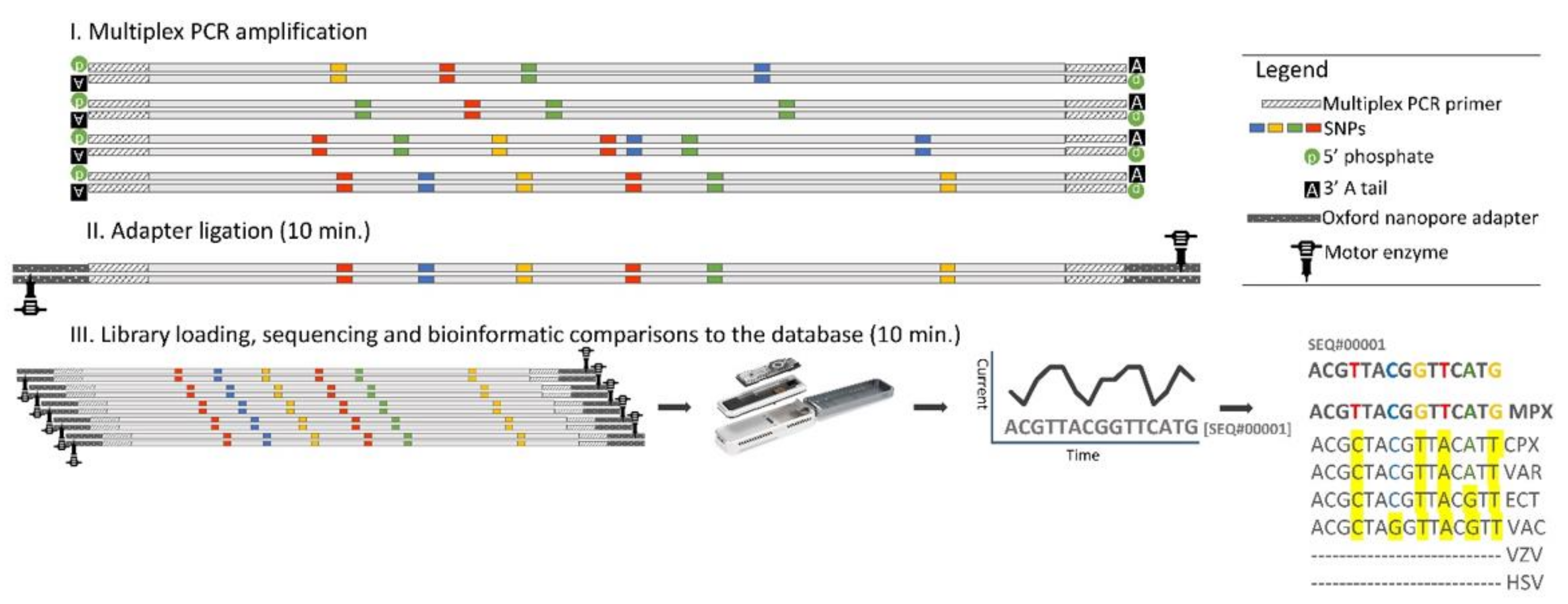
2.4. Multiplex PCR
2.5. Amplicon Library Preparation
2.6. Flongle Loading and Sequencing
2.7. Analysis
2.8. MPXV Real-Time PCR
3. Results and Discussion
3.1. Establishment of the Rapid Amplicon Nanopore Sequencing (RANS) Approach for the Differential Detection of MPXV and Other Vesicle-Forming Pathogens
3.2. Laboratory Samples
3.3. Evaluation of RANS in Clinical Samples
4. Summary and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mathieu, E.; Dattani, S.; Ritchie, S.; Spooner, F.; Roser, M. 20.10.2022. Our World in Data. Monkeypox. Available online: https://ourworldindata.org/monkeypox (accessed on 20 July 2022).
- Bunge, E.M.; Hoet, B.; Chen, L.; Lienert, F.; Weidenthaler, H.; Baer, L.R.; Steffen, R. The changing epidemiology of human monkeypox—A potential threat? A systematic review. PLoS Neglected Trop. Dis. 2022, 16, e0010141. [Google Scholar] [CrossRef] [PubMed]
- Farahat, R.A.; Abdelaal, A.; Shah, J.; Ghozy, S.; Sah, R.; Bonilla-Aldana, D.K.; Rodriguez-Morales, A.J.; McHugh, T.D.; Leblebicioglu, H. Monkeypox outbreaks during COVID-19 pandemic: Are we looking at an independent phenomenon or an overlapping pandemic? Ann. Clin. Microbiol. Antimicrob. 2022, 21, 26. [Google Scholar] [CrossRef] [PubMed]
- Babkin, I.V.; Babkina, I.N.; Tikunova, N.V. An Update of Orthopoxvirus Molecular Evolution. Viruses 2022, 14, 388. [Google Scholar] [CrossRef] [PubMed]
- Moss, B. Genetically engineered poxviruses for recombinant gene expression, vaccination, and safety. Proc. Natl. Acad. Sci. USA 1996, 93, 11341–11348. [Google Scholar] [CrossRef]
- Silva, N.; de Oliveira, J.; Kroon, E.; Trindade, G.; Drumond, B. Here, There, and Everywhere: The Wide Host Range and Geographic Distribution of Zoonotic Orthopoxviruses. Viruses 2020, 13, 43. [Google Scholar] [CrossRef]
- Bera, B.; Shanmugasundaram, K.; Barua, S.; Venkatesan, G.; Virmani, N.; Riyesh, T.; Gulati, B.; Bhanuprakash, V.; Vaid, R.; Kakker, N.; et al. Zoonotic cases of camelpox infection in India. Veter. Microbiol. 2011, 152, 29–38. [Google Scholar] [CrossRef]
- Henderson, D.A. Smallpox: Clinical and Epidemiologic Features. Emerg. Infect. Dis. 1999, 5, 537–539. [Google Scholar] [CrossRef]
- Pennington, H. Smallpox and bioterrorism. Bull. World Health Organ. 2003, 81, 762–767. [Google Scholar]
- Esposito, J.J.; Sammons, S.A.; Frace, A.M.; Osborne, J.D.; Olsen-Rasmussen, M.; Zhang, M.; Govil, D.; Damon, I.K.; Kline, R.; Laker, M.; et al. Genome Sequence Diversity and Clues to the Evolution of Variola (Smallpox) Virus. Science 2006, 313, 807–812. [Google Scholar] [CrossRef]
- Arita, M.; Tagaya, I. Structural Polypeptides of Several Strains of Orthopoxvirus. Microbiol. Immunol. 1977, 21, 343–346. [Google Scholar] [CrossRef][Green Version]
- Mercer, A.; Fleming, S.; Robinson, A.; Nettleton, P.; Reid, H. Molecular genetic analyses of parapoxviruses pathogenic for humans. Vir. Zoonoses Food Anim. Orig. 1997, 13, 25–34. [Google Scholar] [CrossRef]
- Obijeski, J.F.; Palmer, E.L.; Gafford, L.G.; Randall, C.C. Polyacrylamide gel electrophoresis of fowlpox and vaccinia virus proteins. Virology 1973, 51, 512–516. [Google Scholar] [CrossRef]
- Esposito, J.J.; Knight, J.C. Orthopoxvirus DNA: A comparison of restriction profiles and maps. Virology 1985, 143, 230–251. [Google Scholar] [CrossRef]
- Luciani, L.; Inchauste, L.; Ferraris, O.; Charrel, R.; Nougairède, A.; Piorkowski, G.; Peyrefitte, C.; Bertagnoli, S.; de Lamballerie, X.; Priet, S. A novel and sensitive real-time PCR system for universal detection of poxviruses. Sci. Rep. 2021, 11, 5961. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, H.; Wilkins, K.; Hughes, C.; Damon, I.K. Real-time PCR assays for the specific detection of monkeypox virus West African and Congo Basin strain DNA. J. Virol. Methods 2010, 169, 223–227. [Google Scholar] [CrossRef]
- Maksyutov, R.A.; Gavrilova, E.V.; Shchelkunov, S.N. Species-specific differentiation of variola, monkeypox, and varicella-zoster viruses by multiplex real-time PCR assay. J. Virol. Methods 2016, 236, 215–220. [Google Scholar] [CrossRef]
- Ryabinin, V.A.; Shundrin, L.A.; Kostina, E.B.; Laassri, M.; Chizhikov, V.; Shchelkunov, S.N.; Chumakov, K.; Sinyakov, A.N. Microarray assay for detection and discrimination ofOrthopoxvirus species. J. Med. Virol. 2006, 78, 1325–1340. [Google Scholar] [CrossRef]
- Nitsche, A.; Ellerbrok, H.; Pauli, G. Detection of Orthopoxvirus DNA by Real-Time PCR and Identification of Variola Virus DNA by Melting Analysis. J. Clin. Microbiol. 2004, 42, 1207–1213. [Google Scholar] [CrossRef]
- Laassri, M.; Chizhikov, V.; Mikheev, M.; Shchelkunov, S.; Chumakov, K. Detection and discrimination of orthopoxviruses using microarrays of immobilized oligonucleotides. J. Virol. Methods 2003, 112, 67–78. [Google Scholar] [CrossRef]
- Gelaye, E.; Mach, L.; Kolodziejek, J.; Grabherr, R.; Loitsch, A.; Achenbach, J.E.; Nowotny, N.; Diallo, A.; Lamien, C.E. A novel HRM assay for the simultaneous detection and differentiation of eight poxviruses of medical and veterinary importance. Sci. Rep. 2017, 7, srep42892. [Google Scholar] [CrossRef]
- Ropp, S.L.; Jin, Q.; Knight, J.C.; Massung, R.F.; Esposito, J.J. PCR strategy for identification and differentiation of small pox and other orthopoxviruses. J. Clin. Microbiol. 1995, 33, 2069–2076. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.; Chen, J. Application of next generation sequencing for the detection of human viral pathogens in clinical specimens. J. Clin. Virol. 2016, 86, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Beth-Din, A.; Israeli, O.; Shifman, O.; Stein, D.; Ben-Arie, E.; Paran, N.; Lustig, S.; Zvi, A.; Ariel, N.; Ordentlich, A. Genetic Detection of Vesicle Forming Pathogens by Arrayed Primer Extension (APEX). In The Challenge of Highly Pathogenic Microorganisms; Shafferman, A., Ordentlich, A., Velan, B., Eds.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar] [CrossRef]
- Erez, N.; Achdout, H.; Milrot, E.; Schwartz, Y.; Wiener-Well, Y.; Paran, N.; Politi, B.; Tamir, H.; Israely, T.; Weiss, S.; et al. Diagnosis of Imported Monkeypox, Israel, 2018. Emerg. Infect. Dis. 2019, 25, 980–983. [Google Scholar] [CrossRef] [PubMed]
- Paran, N.; Suezer, Y.; Lustig, S.; Israely, T.; Schwantes, A.; Melamed, S.; Katz, L.; Preuß, T.; Hanschmann, K.-M.; Kalinke, U.; et al. Postexposure Immunization with Modified Vaccinia Virus Ankara or Conventional Lister Vaccine Provides Solid Protection in a Murine Model of Human Smallpox. J. Infect. Dis. 2009, 199, 39–48. [Google Scholar] [CrossRef]
- Paran, N.; Lustig, S.; Zvi, A.; Erez, N.; Israely, T.; Melamed, S.; Politi, B.; Ben-Nathan, D.; Schneider, P.; Lachmi, B.; et al. Active vaccination with vaccinia virus A33 protects mice against lethal vaccinia and ectromelia viruses but not against cowpoxvirus; elucidation of the specific adaptive immune response. Virol. J. 2013, 10, 229. [Google Scholar] [CrossRef]
- Sharon, M.; Nir, P.; Lior, K.; David, B.-N.; Tomer, I.; Paula, S.; Reuven, L.; Shlomo, L. Tail scarification with Vaccinia virus Lister as a model for evaluation of smallpox vaccine potency in mice. Vaccine 2007, 25, 7743–7753. [Google Scholar] [CrossRef]
- Li, H. Minimap2: Pairwise alignment for nucleotide sequences. Bioinformatics 2018, 34, 3094–3100. [Google Scholar] [CrossRef]
- Virus Pathogen Resource (ViPR). 20.07.2022. Available online: https://www.viprbrc.org/brc/home.spg?decorator=vipr (accessed on 22 July 2022).
- Danecek, P.; Bonfield, J.K.; Liddle, J.; Marshall, J.; Ohan, V.; O Pollard, M.; Whitwham, A.; Keane, T.; A McCarthy, S.; Davies, R.M.; et al. Twelve years of SAMtools and BCFtools. GigaScience 2021, 10, giab008. [Google Scholar] [CrossRef]
- Jain, M.; Olsen, H.E.; Paten, B.; Akeson, M. The Oxford Nanopore MinION: Delivery of nanopore sequencing to the genomics community. Genome Biol. 2016, 17, 256. [Google Scholar]
- Avershina, E.; Frye, S.A.; Ali, J.; Taxt, A.M.; Ahmad, R. Ultrafast and Cost-Effective Pathogen Identification and Resistance Gene Detection in a Clinical Setting Using Nanopore Flongle Sequencing. Front. Microbiol. 2022, 13, 822402. [Google Scholar] [CrossRef]
- Israeli, O.; Cohen-Gihon, I.; Zvi, A.; Lazar, S.; Shifman, O.; Levy, H.; Tidhar, A.; Beth-Din, A. Rapid identification of unknown pathogens in environmental samples using a high-throughput sequencing-based approach. Heliyon 2019, 5, e01793. [Google Scholar] [CrossRef]
- Israeli, O.; Makdasi, E.; Cohen-Gihon, I.; Zvi, A.; Lazar, S.; Shifman, O.; Levy, H.; Gur, D.; Laskar, O.; Beth-Din, A. A rapid high-throughput sequencing-based approach for the identification of unknown bacterial pathogens in whole blood. Future Sci. OA 2020, 6, FSO476. [Google Scholar] [CrossRef]
- Isidro, J.; Borges, V.; Pinto, M.; Sobral, D.; Santos, J.D.; Nunes, A.; Mixão, V.; Ferreira, R.; Santos, D.; Duarte, S.; et al. Phylogenomic characterization and signs of microevolution in the 2022 multi-country outbreak of monkeypox virus. Nat. Med. 2022. ahead of print. [Google Scholar] [CrossRef]
| Subfamily | Chordopoxvirinae | Alphaherpesvirus | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Genus | Orthopoxvirus | Parapoxvirus | Moll. | Yata. | Simplexvirus | Varic. | |||||||||
| Area | bp | VARV | MPXV | VACV | CPXV | CMLP | ECTV | ORF | BPSV | MOLL | TANA | HSV1 | HSV2 | HBV | VZV |
| Chem | 597 | √ | √ | √ √ | √ | √ | |||||||||
| R60 | 638 | √ √ | √ √ | √ √ | √ √ | √ √ | √ √ | ||||||||
| R56 | 652 | √ √ | √ √ | √ √ | √ √ | √ | √ | ||||||||
| Y71 | 785 | √ | √ | √ | √ √ | √ | √ √ | ||||||||
| m8121L | 720 | √ √ | √ | √ | √ | √ √ | √ √ | √ √ | |||||||
| Mine | 1017 | √ √ | √ | √ √ | √ | √ √ | √ √ | √ √ | √ √ | ||||||
| B100 | 355 | √ √ | √ √ | √ √ | |||||||||||
| ORF45 | 530 | √ √ | √ √ | √ √ | √ √ | ||||||||||
| ORF27 | 460 | √ √ | √ √ | √ √ | √ √ | ||||||||||
| Mix | Primer | Sequence |
|---|---|---|
| A | Mine-sF | CGATACGACGGGCGTACTAGCGAGGTGCTCwGCGAGAAGTTCAC |
| Mine-sR1 | CGATACGACGGGCGTACTAGCGGGCAGCACCAGCATGAACTTG | |
| Mine-sR2 | CGATACGACGGGCGTACTAGCGGGCAGCATGAGCATGAACTTG | |
| Y71-F | CGATACGACGGGCGTACTAGCGCCCGTwTATGGATCwATTCAAGA | |
| Y71-R | CGATACGACGGGCGTACTAGCGCCTCTTCCyTCyGGATCCTTAGA | |
| R60-F | CGATACGACGGGCGTACTAGCGCAATATGAACAAGAAATAGAATCGTTAGAAG | |
| R60-R | CGATACGACGGGCGTACTAGCGGCGCTTCTATATCTCTCATTAGCTAGAA | |
| ORF45-F | CGATACGACGGGCGTACTAGCGTGCGAyGAyCACATGCCG | |
| ORF45-R | CGATACGACGGGCGTACTAGCGTCCTGGCTGCTrTTkCCCTC | |
| B | Chem-F | CGATACGACGGGCGTACTAGCGCAAmCATyATATGGGAATCGATGTTA |
| Chem-R | CGATACGACGGGCGTACTAGCGTCGAyATACTTrAATCCATCCTTGAC | |
| ORF27-F1 | CGATACGACGGGCGTACTAGCGCGTGGCCGACAACTGCCT | |
| ORF27-F2 | CGATACGACGGGCGTACTAGCGACAGTGGCGGATAACTGCCT | |
| ORF27-R1 | CGATACGACGGGCGTACTAGCGCGTGCTCTGCCACACGTG | |
| ORF27-R2 | CGATACGACGGGCGTACTAGCGCGTGCTCTGCCACACGTG | |
| B100-mF | CGATACGACGGGCGGACTAGCGGTACCACCCCAGCCAGTACG | |
| B100-R | CGATACGACGGGCGTACTAGCGAyGCGCACCTCGTTCAT | |
| R56-F | CGATACGACGGGCGTACTAGCGTTACCACGTCTGGATAGGAGATTGT | |
| R56-R | CGATACGACGGGCGTACTAGCGCGTGTTCTTAGTTGCTTAGCTGAAAC | |
| m8121-sF1 | CGATACGACGGGCGTACTAGCGTACTTGGAAAAGAATTTGGACCAA | |
| m8121-sF2 | CGATACGACGGGCGTACTAGCGGAGAAATTCCATTTAATATGAAAGACATG | |
| m8121-sR1 | CGATACGACGGGCGTACTAGCGCATGATAATATTAAAGATAAAGCGCTGAC | |
| m8121-sR2 | CGATACGACGGGCGTACTAGCGGGTTCGCTTAAAAATATAGACTTGTTAAATG | |
| Multiplex adapter | P* CGATACGACGGGCGTACTAGCG | |
| Sample # | Virus Strain Cultured | % Mapped Reads | Species Detected | Nearest Detected Strain (Accession Number) |
|---|---|---|---|---|
| 1 VACV | Vaccinia Lister | 94.9 | Vaccinia virus | Vaccinia Lister (KX061501) |
| 2 VACV | Vaccinia Western Reserve | 93.1 | Vaccinia virus | Vaccinia Western Reserve (AY243312) |
| 3 ECTV | Ectromelia Moscow | 91.0 | Ectromelia virus | Ectromelia Moscow (AF012825) |
| 4 CMLP | Camelpox Negev 2016 | 98.4 | Camelpox virus | Camelpox Negev 2016 (MK901851) |
| 5 CPXV | Cowpox Brighton Red | 97.0 | Cowpox virus | Cowpox Brighton Red (AF482758) |
| 6 MPXV | Monkeypox virus Israel | 99.1 | Monkeypox virus | Monkeypox virus Israel 2018 (MN648051) |
| 7 VZV | Human herpesvirus 3 | 96.8 | Human herpesvirus 3 | Human herpesvirus 3 YCO1 (KU926318) |
| 8 HSV1 | Human herpesvirus 1 | 93.8 | Human herpesvirus 1 | Mckrae (MN136524) Human herpesvirus 1 |
| Clinical Sample # | Source | MPXV Ct GE WA | % Mapped Reads | Species Detected | Nearest Strain (Accession Number) | |
|---|---|---|---|---|---|---|
| 2022 | vesicle swab | 24.7 | 25.0 | 99.6 | Monkeypox virus | MPXV/Germany/2022/RKI015 (ON694331.1) |
| 2023 | throat swab | 32.2 | 32.7 | 99.6 | Monkeypox virus | MPXV/Germany/2022/RKI020 (ON694337.1) |
| 2043 | throat swab | 38.8 | 38.5 | 99.8 | Monkeypox virus | MPXV/Germany/2022/RKI03 (ON682263.2) |
| 2072 | semen | 34.1 | 33.7 | 99.5 | Monkeypox virus | MpxV/Spain/MD-HGUGM-6532064/2022 (ON720849) |
| 2012 | vesicle swab | undet. | undet. | 95.0 | Human herpesvirus 3 | YC02 (KU926319) |
| 2068 | vesicle swab | undet. | undet. | 99.9 | Human herpesvirus 3 | YC02 (KU926319) |
| 2089 | vesicle swab | undet. | undet. | 89.6 | Human herpesvirus 2 | HG52 (Z86099) |
| 2090 | vesicle swab | undet. | undet. | 97.9 | Molluscum contagiosum virus subtype 1 | MCV1_P05S02A (MN931749) |
| 2114 | vesicle swab | undet. | undet. | 0.0 | unknown | unknown |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Israeli, O.; Guedj-Dana, Y.; Shifman, O.; Lazar, S.; Cohen-Gihon, I.; Amit, S.; Ben-Ami, R.; Paran, N.; Schuster, O.; Weiss, S.; et al. Rapid Amplicon Nanopore Sequencing (RANS) for the Differential Diagnosis of Monkeypox Virus and Other Vesicle-Forming Pathogens. Viruses 2022, 14, 1817. https://doi.org/10.3390/v14081817
Israeli O, Guedj-Dana Y, Shifman O, Lazar S, Cohen-Gihon I, Amit S, Ben-Ami R, Paran N, Schuster O, Weiss S, et al. Rapid Amplicon Nanopore Sequencing (RANS) for the Differential Diagnosis of Monkeypox Virus and Other Vesicle-Forming Pathogens. Viruses. 2022; 14(8):1817. https://doi.org/10.3390/v14081817
Chicago/Turabian StyleIsraeli, Ofir, Yehoudit Guedj-Dana, Ohad Shifman, Shirley Lazar, Inbar Cohen-Gihon, Sharon Amit, Ronen Ben-Ami, Nir Paran, Ofir Schuster, Shay Weiss, and et al. 2022. "Rapid Amplicon Nanopore Sequencing (RANS) for the Differential Diagnosis of Monkeypox Virus and Other Vesicle-Forming Pathogens" Viruses 14, no. 8: 1817. https://doi.org/10.3390/v14081817
APA StyleIsraeli, O., Guedj-Dana, Y., Shifman, O., Lazar, S., Cohen-Gihon, I., Amit, S., Ben-Ami, R., Paran, N., Schuster, O., Weiss, S., Zvi, A., & Beth-Din, A. (2022). Rapid Amplicon Nanopore Sequencing (RANS) for the Differential Diagnosis of Monkeypox Virus and Other Vesicle-Forming Pathogens. Viruses, 14(8), 1817. https://doi.org/10.3390/v14081817







