Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution
Abstract
1. Introduction
1.1. Introduction to Family Poxviridae
1.2. History of Monkeypox
1.3. Monkeypox Virus: Morphology, Genome Organization, and Morphogenesis
2. Monkeypox Virus Infection Biology, Diagnosis and Treatment
2.1. Animal Models
2.2. Transmission
2.3. Diagnosis
2.3.1. Genetic Methods
2.3.2. Phenotypic Methods
2.3.3. Immunological Methods
2.3.4. Electron Microscopy
2.4. Virus–Host Interaction
2.4.1. Host and Tissue Tropism
2.4.2. Signaling in Orthopoxvirus Infection
2.4.3. Host Immune Responses to MPXV
2.5. Treatment
2.5.1. Vaccination
2.5.2. Antivirals
3. Ecology and Epidemiology of Monkeypox Virus in Nigeria
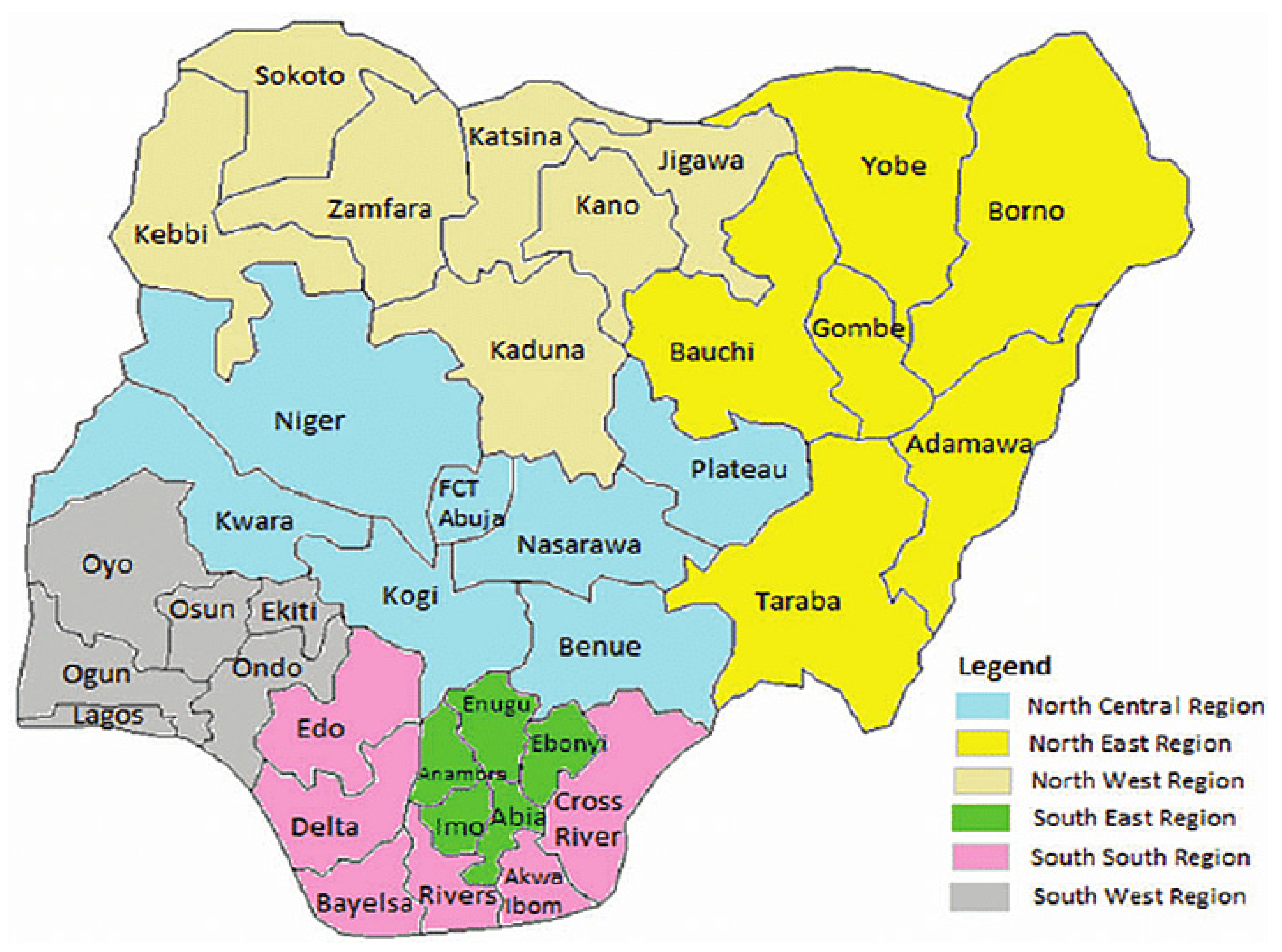
3.1. Geographic Distribution of Human Monkeypox in Nigeria
3.2. Reservoir Host Species of Monkeypox Virus in Nigeria
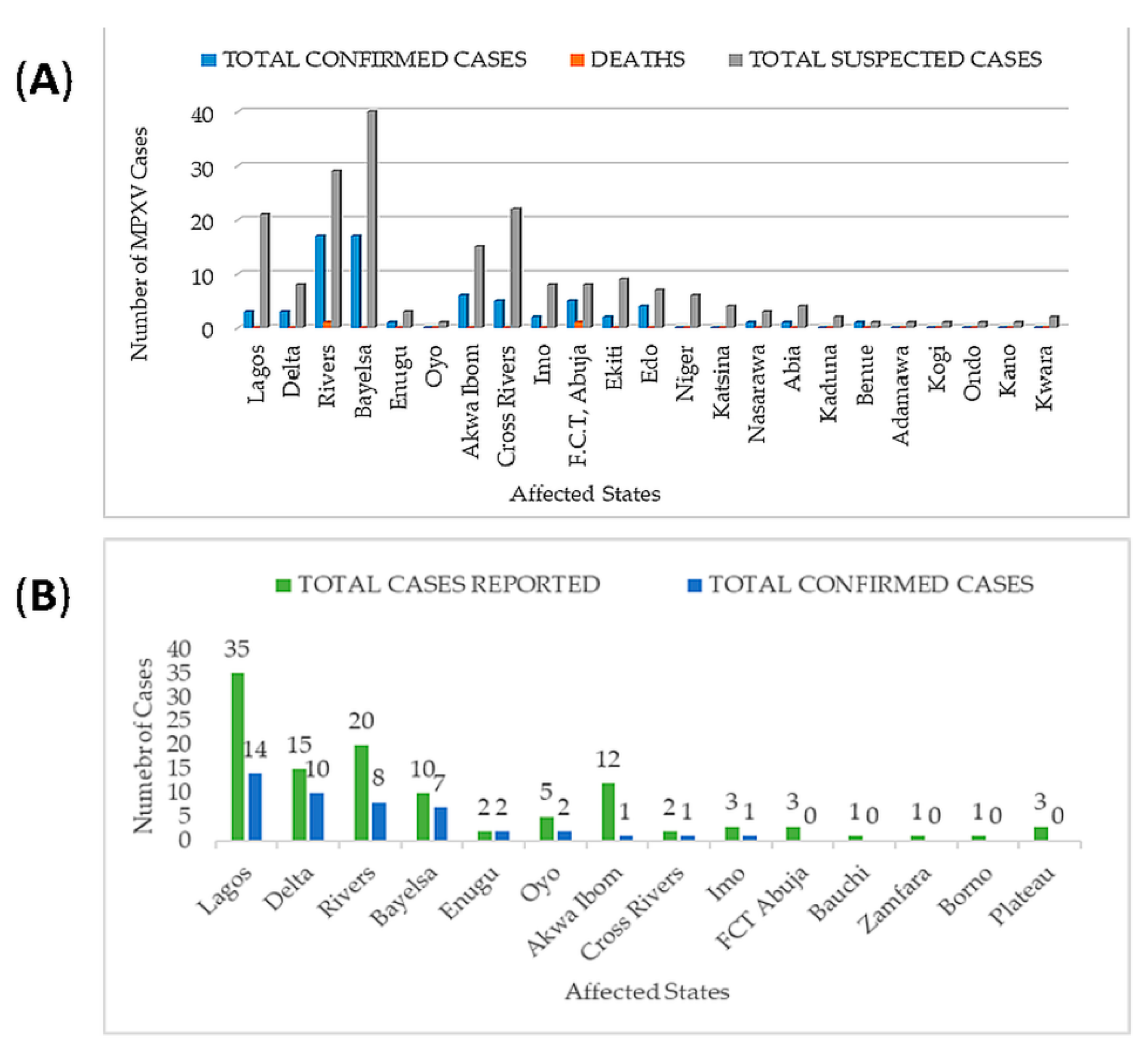
3.3. Epidemiology of Monkeypox Virus in Nigeria
4. Phylogeny and Evolution of Monkeypox Virus in Nigeria
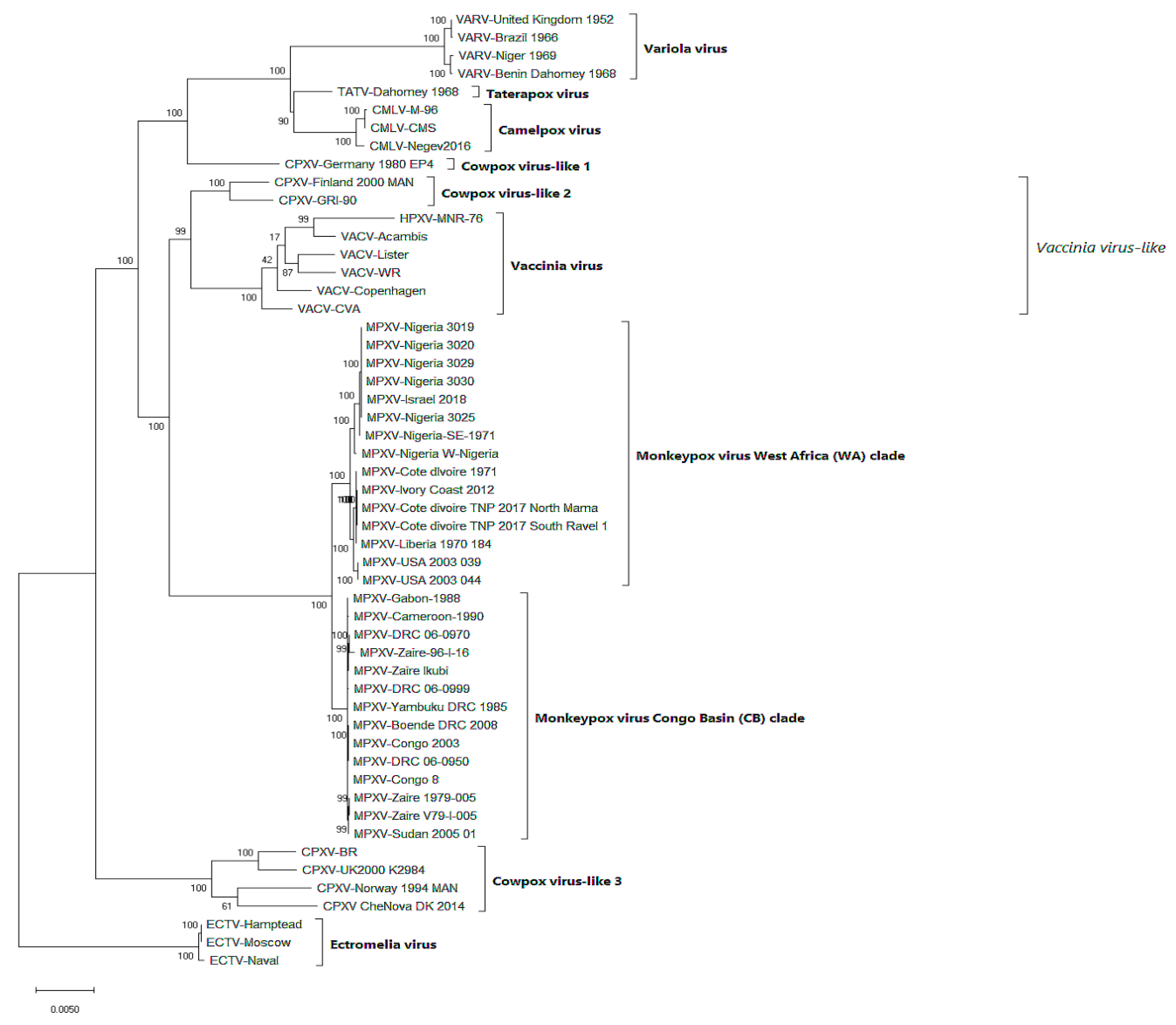
4.1. Phylogeny
4.2. Recombination
4.3. Gene Loss, Gene Gain, and SNPs
5. Gaps in Knowledge, Omitted Research, and Conclusion
5.1. Reservoir Host Species and Tissue Tropism
5.2. Co-Infection and Recombination
5.3. Monkeypox Virus Infectome
5.4. Antibody-Dependent Enhancement (ADE) of Infection
5.5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barrett, J.W.; McFadden, G. Origin and Evolution of Poxviruses. In Origin and Evolution of Viruses; Elsevier Ltd.: Singapore, 2008; pp. 431–446. ISBN 9780123741530. [Google Scholar]
- Hughes, A.L.; Irausquin, S.; Friedman, R. The evolutionary biology of poxviruses. Infect. Genet. Evol. 2010, 10, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Diven, D.G. An overview of poxviruses. J. Am. Acad. Dermatol. 2001, 44, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Odom, M.R.; Curtis Hendrickson, R.; Lefkowitz, E.J. Poxvirus protein evolution: Family wide assessment of possible horizontal gene transfer events. Virus Res. 2009, 144, 233–249. [Google Scholar] [CrossRef] [PubMed]
- Lefkowitz, E.J.; Wang, C.; Upton, C. Poxviruses: Past, present and future. Virus Res. 2006, 117, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Essbauer, S.; Pfeffer, M.; Meyer, H. Zoonotic poxviruses. Vet. Microbiol. 2010, 140, 229–236. [Google Scholar] [CrossRef]
- International Committee on Taxonomy of Viruses (ICTV). Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 17 October 2020).
- Von Magnus, P.; Andersen, E.K.; Petersen, K.B.; Birch-Andersen, A. A Pox-like Disease in Cynomolgus Monkeys. Acta Pathol. Microbiol. Scand. 2009, 46, 156–176. [Google Scholar] [CrossRef]
- Breman, J.G.; Ruti, K.; Steniowski, M.V. Human monkeypox, 1970–1979. Bull. World Health Organ. 1980, 58, 165–182. [Google Scholar] [PubMed]
- Jezek, Z.; Gromyko, A.I.; Szczeniowski, M.V. Human monkeypox. J. Hyg. Epidemiol. Microbiol. Immunol. 1983, 27, 13–28. [Google Scholar]
- Ladnyj, I.D.; Ziegler, P.; Kima, E. A human infection caused by monkeypox virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593–597. [Google Scholar]
- Foster, S.O.; Brink, E.W.; Hutchins, D.L.; Pifer, J.M.; Lourie, B.; Moser, C.R.; Cummings, E.C.; Kuteyi, O.E.K.; Eke, R.E.A.; Titus, J.B.; et al. Human monkeypox. Bull. World Health Organ. 1972, 46, 569–576. [Google Scholar] [CrossRef]
- Monkeypox. Available online: https://www.who.int/health-topics/monkeypox/#tab=tab_1 (accessed on 19 October 2020).
- Monkeypox Virus|MSDSonline. Available online: https://www.msdsonline.com/resources/sds-resources/free-safety-data-sheet-index/monkeypox-virus/ (accessed on 17 October 2020).
- Cho, C.T.; Wenner, H.A. Monkeypox virus. Bacteriol. Rev. 1973, 37, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Fenner, F.; Wittek, R.; Dumbell, K.R. Orthopoxviruses. Biodefense Res. Methodol. Anim. Model. Second Ed. 2012, 255–270. [Google Scholar] [CrossRef]
- Jahrling, P.B.; Huggins, J.W.; Ibrahim, M.S.; Lawler, J.V.; Martin, J.W. Smallpox and Related Orthopoxviruses. In Medical Aspects of Biological Warfare; Office of the Surgeon General: Washington, DC, USA, 2007; pp. 215–240. [Google Scholar]
- Kugelman, J.R.; Johnston, S.C.; Mulembakani, P.M.; Kisalu, N.; Lee, M.S.; Koroleva, G.; Mccarthy, S.E.; Gestole, M.C.; Wolfe, N.D.; Fair, J.N.; et al. Genomic Variability of Monkeypox Virus among Humans, Democratic Republic of the Congo. Emerg. Infect. Dis. 2014, 20, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Esposito, J.J.; Knight, J.C. Orthopoxvirus DNA: A comparison of restriction profiles and maps. Virology 1985, 143, 230–251. [Google Scholar] [CrossRef]
- Boyle, K. Paula Traktman Poxviruses. In Viral Genome Replication; Cameron, C.E., Raney, K.D., Götte, M., Eds.; Springer: New York, NY, USA, 2009; pp. 1–636. ISBN 9780387894560. [Google Scholar]
- Takemura, M. Poxviruses and the origin of the eukaryotic nucleus. J. Mol. Evol. 2001, 52, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Remichkova, M. Poxviruses: Smallpox vaccine, its complications and chemotherapy. Virus Adapt. Treat. 2010, 2, 41–46. [Google Scholar] [CrossRef]
- Moss, B. The Molecular Biology of Poxviruses. In The Molecular Basis of Viral Replication; Springer: New York, NY, USA, 1987; Volume 37, pp. 99–102. [Google Scholar]
- Smith, G.L.; Murphy, B.J.; Law, M. Vaccinia Virus Motility. Annu. Rev. Microbiol. 2003, 57, 323–342. [Google Scholar] [CrossRef]
- Schmelz, M.; Sodeik, B.; Ericsson, M.; Wolffe, E.J.; Shida, H.; Hiller, G.; Griffiths, G. Assembly of vaccinia virus: The second wrapping cisterna is derived from the trans Golgi network. J. Virol. 1994, 68, 130–147. [Google Scholar] [CrossRef]
- Meiser, A.; Sancho, C.; Krijnse Locker, J. Plasma Membrane Budding as an Alternative Release Mechanism of the Extracellular Enveloped Form of Vaccinia Virus from HeLa Cells. J. Virol. 2003, 77, 9931–9942. [Google Scholar] [CrossRef]
- Okeke, M.I.; Nilssen, Ø.; Traavik, T. Modified vaccinia virus Ankara multiplies in the rat IEC-6 cells and limited production of mature virions occurs in other mammalian cell lines. J. Gen. Virol. 2006, 87, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Shida, H.; Tanabe, K.; Matsumoto, S. Mechanism of Virus Occlusion into A-Type Infection during Poxvirus. Virology 1977, 233, 217–233. [Google Scholar] [CrossRef]
- Okeke, M.I.; Adekoya, O.A.; Moens, U.; Tryland, M.; Traavik, T.; Nilssen, O. Comparative sequence analysis of A-type inclusion (ATI) and P4c proteins of orthopoxviruses that produce typical and atypical ATI phenotypes. Virus Genes 2009, 39, 200–209. [Google Scholar] [CrossRef]
- Howard, A.R.; Weisberg, A.S.; Moss, B. Congregation of Orthopoxvirus Virions in Cytoplasmic A-Type Inclusions Is Mediated by Interactions of a Bridging Protein (A26p) with a Matrix Protein (ATIp) and a Virion Membrane-Associated Protein (A27p). J. Virol. 2010, 84, 7592–7602. [Google Scholar] [CrossRef]
- Hutson, C.L.; Damon, I.K. Monkeypox virus infections in small animal models for evaluation of anti-poxvirus agents. Viruses 2010, 2, 2763–2776. [Google Scholar] [CrossRef]
- Susceptibility of Some Rodent Species to Monkeypox Virus, and Course of the Infection. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2366409/ (accessed on 27 June 2020).
- Xiao, S.Y.; Sbrana, E.; Watts, D.M.; Siirin, M.; Travassos Da Rosa, A.P.A.; Tesh, R.B. Experimental infection of prairie dogs with monkeypox virus. Emerg. Infect. Dis. 2005, 11, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Tesh, R.B.; Watts, D.M.; Sbrana, E.; Siirin, M.; Popov, V.L. Experimental Infection of Ground Squirrels with Monkeypox Virus. Emerg. Infect. Dis. 2004, 10, 1563–1567. [Google Scholar] [CrossRef]
- Hatch, G.J.; Graham, V.A.; Bewley, K.R.; Tree, J.A.; Dennis, M.; Taylor, I.; Funnell, S.G.P.; Bate, S.R.; Steeds, K.; Tipton, T.; et al. Assessment of the Protective Effect of Imvamune and Acam2000 Vaccines against Aerosolized Monkeypox Virus in Cynomolgus Macaques. J. Virol. 2013, 87, 7805–7815. [Google Scholar] [CrossRef] [PubMed]
- Schultz, D.A.; Sagartz, J.E.; Huso, D.L.; Buller, R.M.L. Experimental infection of an African dormouse (Graphiurus kelleni) with monkeypox virus. Virology 2009, 383, 86–92. [Google Scholar] [CrossRef]
- Earl, P.L.; Americo, J.L.; Moss, B. Lethal Monkeypox Virus Infection of CAST/EiJ Mice Is Associated with a Deficient Gamma Interferon Response. J. Virol. 2012, 86, 9105–9112. [Google Scholar] [CrossRef] [PubMed]
- Americo, J.L.; Sood, C.L.; Cotter, C.A.; Vogel, J.L.; Kristie, T.M.; Moss, B.; Earl, P.L. Susceptibility of the wild-derived inbred CAST/Ei mouse to infection by orthopoxviruses analyzed by live bioluminescence imaging. Virology 2015, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Americo, J.L.; Moss, B.; Earl, P.L. Identification of Wild-Derived Inbred Mouse Strains Highly Susceptible to Monkeypox Virus Infection for Use as Small Animal Models. J. Virol. 2010, 84, 8172–8180. [Google Scholar] [CrossRef]
- Earl, P.L.; Americo, J.L.; Moss, B. Natural killer cells expanded in vivo or ex vivo with IL-15 overcomes the inherent susceptibility of CAST mice to lethal infection with orthopoxviruses. PLoS Pathog. 2020, 16, 1–25. [Google Scholar] [CrossRef]
- Jenewari, F.O. Monkey Pox in Nigeria: Epidemiology and Prevention. In Proceedings of the Problems and Prospects for the Development of Modern Medicine, GomSMU, Kharkiv, Ukrainian, 2–3 May 2019; pp. 194–195. [Google Scholar]
- Kabuga, A.I.; El Zowalaty, M.E. A review of the monkeypox virus and a recent outbreak of skin rash disease in Nigeria. J. Med. Virol. 2019, 91, 533–540. [Google Scholar] [CrossRef]
- Nasir, I.A.; Dangana, A.; Ojeamiren, I.; Emeribe, A.U. Reminiscing the recent incidence of monkeypox in Nigeria: Its ecologic-epidemiology and literature review. Port Harcourt Med. J. 2018, 11, 1–9. [Google Scholar] [CrossRef]
- Sadeuh-Mba, S.A.; Yonga, M.G.; Els, M.; Batejat, C.; Eyangoh, S.; Caro, V.; Etoundi, A.; Carniel, E.; Njouom, R. Monkeypox virus phylogenetic similarities between a human case detected in Cameroon in 2018 and the 2017-2018 outbreak in Nigeria. Infect. Genet. Evol. 2019, 69, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.; Kantele, A.; Koopmans, M.; Asogun, D.; Ogunlete, A.; Ihekweazu, C.; Zumla, A. Human Monkeypox–Epidemiological and Clinical characteristics, Diagnosis and Prevention. Infect. Dis. Clin. 2019, 33, 1027–1043. [Google Scholar] [CrossRef]
- Brown, K.; Leggat, P.A. Human monkeypox: Current state of knowledge and implications for the future. Trop. Med. Infect. Dis. 2016, 1, 8. [Google Scholar] [CrossRef]
- Ugorji, C.V.; Nworuh; Ibe, S.N.O.; Iwuoha, G.; Abanobi, O.C. Factors associated with the practice of monkey pox preventive behaviours among health workers in Yenagoa LGA, Bayelsa state, Nigeria. IOSR J. Nurs. Health Sci. 2019, 8, 75–85. [Google Scholar] [CrossRef]
- Okoli, S.C.; Kikelomo Amao, L.; Akinrogbe, J.T.; Iniobong, J.-I.; Burga, J. Outbreak Investigation of Monkeypox in Akwa Ibom State: A Matched Case Control Study 14th-24th October 2019. East Afr. J. Health Sci. 2019, 4, 37–44. [Google Scholar]
- Ellis, C.K.; Carroll, D.S.; Lash, R.R.; Townsend Peterson, A.; Damon, I.K.; Malekani, J.; Formenty, P. Ecology and geography of human monkeypox case occurrences across Africa. J. Wildl. Dis. 2012, 48, 335–347. [Google Scholar] [CrossRef]
- Ihekweazu, C.; Yinka-Ogunleye, A.; Lule, S.; Ibrahim, A. Importance of epidemiological research of monkeypox: Is incidence increasing? Expert Rev. Anti. Infect. Ther. 2020, 18, 389–392. [Google Scholar] [CrossRef]
- Vaughan, A.; Aarons, E.; Astbury, J.; Balasegaram, S.; Beadsworth, M.; Beck, C.R.; Chand, M.; O’connor, C.; Dunning, J.; Ghebrehewet, S.; et al. Two cases of monkeypox imported to the United Kingdom, september 2018. Eurosurveillance 2018, 23, 1800509. [Google Scholar] [CrossRef]
- Yinka-Ogunleye, A.; Aruna, O.; Ogoina, D.; Aworabhi, N.; Eteng, W.; Badaru, S.; Mohammed, A.; Agenyi, J.; Etebu, E.N.; Numbere, T.W.; et al. Reemergence of human monkeypox in Nigeria, 2017. Emerg. Infect. Dis. 2018, 24, 1149–1151. [Google Scholar] [CrossRef]
- Nolen, L.D.; Osadebe, L.; Katomba, J.; Likofata, J.; Mukadi, D.; Monroe, B.; Doty, J.; Hughes, C.M.; Kabamba, J.; Malekani, J.; et al. Extended human-to-human transmission during a monkeypox outbreak in the Democratic Republic of the Congo. Emerg. Infect. Dis. 2016, 22, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Ogoina, D.; Izibewule, J.H.; Ogunleye, A.; Ederiane, E.; Anebonam, U.; Neni, A.; Oyeyemi, A.; Etebu, E.N.; Ihekweazu, C. The 2017 human monkeypox outbreak in Nigeria—Report of outbreak experience and response in the Niger Delta University Teaching Hospital, Bayelsa State, Nigeria. PLoS ONE 2019, 14, 1–12. [Google Scholar] [CrossRef]
- Beer, E.M.; Bhargavi Rao, V. A systematic review of the epidemiology of human monkeypox outbreaks and implications for outbreak strategy. PLoS Negl. Trop. Dis. 2019, 13, 1–20. [Google Scholar] [CrossRef] [PubMed]
- McMullen, C.L.; Mulembekani, P.; Hoff, N.A.; Doshi, R.H.; Mukadi, P.; Shongo, R.; Kebela-Illunga, B.; Okitolonda, E.; Muyembe, J.-J.; Rimoin, A.W. Human monkeypox transmission dynamics thirty years after smallpox eradication in the Sankuru district, democratic republic of Congo. Am. J. Trop. Med. Hyg. 2015, 93, 341. [Google Scholar]
- Fine, P.E.M.; Jezek, Z.; Grab, B.; Dixon, H. The transmission potential of monkeypox virus in human populations. Int. J. Epidemiol. 1988, 17, 643–650. [Google Scholar] [CrossRef]
- Fowotade, A.; Fasuyi, T.O.; Bakare, R.A. Re-emergence of monkeypox in Nigeria: A cause for concern and public enlightenment. Afr. J. Clin. Exp. Microbiol. 2018, 19, 307. [Google Scholar] [CrossRef]
- Li, Y.; Olson, V.A.; Laue, T.; Laker, M.T.; Damon, I.K. Detection of monkeypox virus with real-time PCR assays. J. Clin. Virol. 2006, 36, 194–203. [Google Scholar] [CrossRef]
- Yinka-Ogunleye, A.; Aruna, O.; Dalhat, M.; Ogoina, D.; McCollum, A.; Disu, Y.; Mamadu, I.; Akinpelu, A.; Ahmad, A.; Burga, J.; et al. Outbreak of human monkeypox in Nigeria in 2017–18: A clinical and epidemiological report. Lancet Infect. Dis. 2019, 19, 872–879. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Carroll, D.S.; Olson, V.A.; Hughes, C.; Galley, J.; Likos, A.; Montgomery, J.M.; Suu-ire, R.; Kwasi, M.O.; Root, J.J.; et al. A Silent Enzootic of an Orthopoxvirus in Ghana, West Africa: Evidence for Multi-Species Involvement in the Absence of Widespread Human Disease. Am. J. Trop. Med. Hyg. 2010, 82, 746–754. [Google Scholar] [CrossRef]
- Orba, Y.; Sasaki, M.; Yamaguchi, H.; Ishii, A.; Thomas, Y.; Ogawa, H.; Hang, B.M.; Mweene, S.; Morikawa, S.; Saijo, M.; et al. Orthopoxvirus infection among wildlife in Zambia. Gen. Virol. 2015, 96, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Kulesh, D.A.; Loveless, B.M.; Norwood, D.; Garrison, J.; Whitehouse, C.A.; Hartmann, C.; Mucker, E.; Miller, D.; Wasieloski, L.P.; Huggins, J.; et al. Monkeypox virus detection in rodents using real-time 3′-minor groove binder TaqMan®® assays on the Roche LightCycler. Lab. Investig. 2004, 84, 1200–1208. [Google Scholar] [CrossRef]
- Meyer, H.; Pfeffer, M.; Rziha, H.-J. Sequence alterations within and downstream of the A-type inclusion protein genes allow differentiation of Orthopoxvirus species by polymerase chain reaction. Gen. Virol. 1994, 75, 1975–1981. [Google Scholar] [CrossRef]
- Ropp, S.L.; Jin, Q.I.; Knight, J.C.; Massung, R.F.; Esposito, J.J. PCR Strategy for Identification and Differentiation of Smallpox and Other Orthopoxviruses. Clin. Microbiol. 1995, 33, 2069–2076. [Google Scholar] [CrossRef]
- Radonić, A.; Metzger, S.; Dabrowski, P.W.; Couacy-hymann, E.; Schuenadel, L.; Kurth, A.; Mätz-rensing, K.; Boesch, C.; Leendertz, F.H.; Nitsche, A. Fatal Monkeypox in Wild-Living Sooty Mangabey, Côte Ivoire, 2012. Emerg. Infect. Dis. 2014, 20, 1009–1011. [Google Scholar] [CrossRef]
- Farlow, J.; Ichou, M.A.; Huggins, J.; Ibrahim, S. Comparative whole genome sequence analysis of wild-type and cidofovir-resistant Monkeypoxvirus. Virol. J. 2010, 7, 1–15. [Google Scholar] [CrossRef]
- Cohen-gihon, I.; Israeli, O.; Shifman, O.; Erez, N.; Melamed, S.; Paran, N.; Beth-din, A.; Zvi, A. Identification and Whole-Genome Sequencing of a Monkeypox. Microbiol. Resour. Announc. 2020, 9, 5–6. [Google Scholar] [CrossRef]
- Meyer, H.; Damon, I.K.; Esposito, J.J. Orthopoxvirus Diagnostics. Methods Mol. Biol. 2004, 269, 119–133. [Google Scholar] [PubMed]
- Quick, J.; Loman, N.J.; Duraffour, S.; Simpson, J.T.; Severi, E.; Cowley, L.; Bore, J.A.; Koundouno, R.; Dudas, G.; Mikhail, A.; et al. Real-time, portable genome sequencing for Ebola surveillance. Nature 2016, 530, 228–232. [Google Scholar] [CrossRef]
- Wilson, M.E.; Hughes, J.M.; McCollum, A.M.; Damon, I.K. Human monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef]
- Sklenovská, N.; Van Ranst, M. Emergence of Monkeypox as the Most Important Orthopoxvirus Infection in Humans. Front. Public Health 2018, 6, 1–12. [Google Scholar] [CrossRef]
- Osadebe, L.; Hughes, C.M.; Shongo Lushima, R.; Kabamba, J.; Nguete, B.; Malekani, J.; Pukuta, E.; Karhemere, S.; Muyembe Tamfum, J.J.; Wemakoy Okitolonda, E.; et al. Enhancing case definitions for surveillance of human monkeypox in the Democratic Republic of Congo. PLoS Negl. Trop. Dis. 2017, 11, 1–13. [Google Scholar] [CrossRef]
- Petersen, E.; Abubakar, I.; Ihekweazu, C.; Heymann, D.; Ntoumi, F.; Blumberg, L.; Asogun, D.; Mukonka, V.; Lule, S.A.; Bates, M.; et al. Monkeypox—Enhancing public health preparedness for an emerging lethal human zoonotic epidemic threat in the wake of the smallpox post-eradication era. Int. J. Infect. Dis. 2019, 78, 78–84. [Google Scholar] [CrossRef]
- Weaver, J.R.; Isaacs, S.N. Monkeypox virus and insights into its immunomodulatory proteins. Immunol. Revolut. 2008, 225, 96–113. [Google Scholar] [CrossRef]
- MacNeil, A.; Abel, J.; Reynolds, M.G.; Lash, R.; Fonnie, R.; Kanneh, L.D.; Robert, W.; Lungay, V.K.; Goba, A.; Moses, L.M.; et al. Serologic evidence of human orthopoxvirus infections in Sierra Leone. BMC Res. Notes 2011, 4, 1–5. [Google Scholar] [CrossRef]
- Karem, K.L.; Reynolds, M.; Braden, Z.; Lou, G.; Bernard, N.; Patton, J.; Damon, I.K. Characterization of acute-phase humoral immunity to monkeypox: Use of immunoglobulin m enzyme-linked immunosorbent assay for detection of monkeypox infection during the 2003 north American outbreak. Clin. Diagn. Lab. Immunol. 2005, 12, 867–872. [Google Scholar] [CrossRef]
- Risi, G.F. Orthopoxviruses. In Nebraska Isolation and Quarantine Manual; Theodore, J., Mark, C., Kortepeter, G., Christopher, J., Kratochvil, J.V.L., Eds.; University of Nebraska Medical Center: Omaha, NE, USA, 2019; pp. 125–138. [Google Scholar]
- McFadden, G. Poxvirus tropism. Nat. Rev. Microbiol. 2005, 3, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Hutson, C.L.; Olson, V.A.; Carroll, D.D.; Abel, J.A.; Hughes, C.M.; Braden, Z.H.; Weiss, S.; Self, J.; Osorio, J.E.; Hudson, P.N.; et al. A prairie dog animal model of systemic orthopoxvirus disease using west African and Congo Basin strains of Monkeypox virus. J. Gen. Virol. 2009, 90, 323–333. [Google Scholar] [CrossRef]
- Bösl, K.; Ianevski, A.; Than, T.T.; Andersen, P.I.; Kuivanen, S.; Teppor, M.; Zusinaite, E.; Dumpis, U.; Vitkauskiene, A.; Cox, R.J.; et al. Common Nodes of Virus–Host Interaction Revealed Through an Integrated Network Analysis. Front. Immunol. 2019, 10, 1–12. [Google Scholar] [CrossRef]
- Shisler, J.L. Immune evasion strategies of molluscum contagiosum virus. In Advances in Virus Research; Academic Press: Cape Town, South Africa, 2015; Volume 92, pp. 201–252. [Google Scholar]
- Mandl, J.N.; Ahmed, R.; Barreiro, L.B.; Daszak, P.; Epstein, J.H.; Virgin, H.W.; Feinberg, M.B. Reservoir host immune responses to emerging zoonotic viruses. Cell 2015, 160, 20–35. [Google Scholar] [CrossRef]
- Nolen, L.D.; Osadebe, L.; Katomba, J.; Likofata, J.; Mukadi, D.; Monroe, B.; Doty, J.; Kalemba, L.; Malekani, J.; Kabamba, J.; et al. Introduction of monkeypox into a community and household: Risk factors and zoonotic reservoirs in the democratic republic of the congo. Am. J. Trop. Med. Hyg. 2015, 93, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Hutson, C.L.; Nakazawa, Y.J.; Self, J.; Olson, V.A.; Regnery, L.; Braden, Z.; Weiss, S.; Malekani, J.; Jackson, E.; Tate, M.; et al. Laboratory Investigations of African Pouched Rats (Cricetomys gambianus) as a Potential Reservoir Host Species for Monkeypox Virus. PLoS Negl. Trop. Dis. 2015, 9, 1–20. [Google Scholar] [CrossRef]
- Sale, T.A.; Melski, J.W.; Stratman, E.J. Monkeypox: An epidemiologic and clinical comparison of African and US disease. J. Am. Acad. Dermatol. 2006, 55, 478–481. [Google Scholar] [CrossRef] [PubMed]
- Giulio, D.B.D.; Eckburg, P.B. Human monkeypox: An emerging zoonosis. Lancet Infect. Dis. 2004, 4, 15–25. [Google Scholar] [CrossRef]
- Formenty, P.; Muntasir, M.O.; Damon, I.; Chowdhary, V.; Opoka, M.L.; Monimart, C.; Mutasim, E.M.; Manuguerra, J.; Davidson, W.B.; Karem, K.L.; et al. Human Monkeypox Outbreak Caused by Novel Virus Belonging to Congo Basin clade, Sudan, 2005. Emerg. Infect. Dis. 2010, 16, 1539–1545. [Google Scholar] [CrossRef]
- Fuller, T.; Thomassen, H.A.; Mulembakani, P.M.; Johnston, S.C.; Lloyd-smith, J.O.; Kisalu, N.K.; Lutete, T.K.; Blumberg, S.; Fair, J.N.; Wolfe, N.D.; et al. Using Remote Sensing to Map the Risk of Human Monkeypox Virus in the Congo Basin. Ecohealth 2011, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.L.; Nichols, D.K.; Martinez, M.J.; Raymond, J.W. Animal models of orthopoxvirus infection. Vet. Pathol. 2010, 47, 852–870. [Google Scholar] [CrossRef]
- Lopera, J.G.; Falendysz, E.A.; Rocke, T.E.; Osorio, J.E. Attenuation of monkeypox virus by deletion of genomic regions. Virology 2015, 475, 129–138. [Google Scholar] [CrossRef]
- Arita, I.; Henderson, D.A. Smallpox and monkeypox in non-human primates. Bull. World Health Organ. 1968, 39, 277–283. [Google Scholar]
- Cleri, D.J.; Ricketti, A.J.; Vernaleo, J.R. Fever of Unknown Origin Due to Zoonoses. Infect. Dis. Clin. N. Am. 2007, 21, 963–996. [Google Scholar] [CrossRef]
- Parker, S.; Buller, R.M. A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Future Virol. 2013, 8, 129–157. [Google Scholar] [CrossRef]
- Heymann, D.L.; Szczeniowski, M.; Esteves, K. Re-emergence of monkeypox in Africa: A review of the past six years. Br. Med. Bull. 1998, 54, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Kindrachuk, J.; Arsenault, R.; Kusalik, A.; Kindrachuk, K.N.; Trost, B.; Napper, S.; Jahrling, P.B.; Blaney, J.E. Systems kinomics demonstrates congo basin monkeypox virus infection selectively modulates host cell signaling responses as compared to West African monkeypox virus. Mol. Cell. Proteom. 2012, 11, 1–12. [Google Scholar] [CrossRef]
- Arita, I.; Henderson, D.A. Smallpox and monkeypox in primates. Primates Med. 1969, 3, 122–123. [Google Scholar] [PubMed]
- Hutin, Y.J.F.; Williams, R.J.; Malfait, P.; Pebody, R.; Loparev, V.N.; Ropp, S.L.; Rodriguez, M.; Knight, J.C.; Tshioko, F.K.; Khan, A.S.; et al. Outbreak of Human Monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg. Infect. Dis. 2001, 7, 434–438. [Google Scholar] [CrossRef]
- Hutson, C.L.; Lee, K.N.; Abel, J.; Carroll, D.S.; Montgomery, J.M.; Olson, V.A.; Li, Y.; Davidson, W.; Hughes, C.; Dillon, M.; et al. Monkeypox zoonotic associations: Insights from laboratory evaluation of animals associated with the multi-state US outbreak. Am. J. Trop. Med. Hyg. 2007, 76, 757–767. [Google Scholar] [CrossRef]
- Ježek, Z.; Fenner, F. Human Monkeypox; S. Karger AG: Basel, Switzerland, 1988. [Google Scholar]
- Parker, S.; Nuara, A.; Buller, R.M.L.; Schultz, D.A. Human monkeypox: An emerging zoonotic disease. Future Microbiol. 2007, 2, 17–34. [Google Scholar] [CrossRef]
- Adebayo, O.; Owoeye, D. Monkeypox. Ann. Ibadan Postgrad. Med. 2017, 15, 145–148. [Google Scholar]
- Gispen, R.; Verlinde, J.D.; Zwart, P. Histopathological and virological studies on monkeypox. Arch. Gesamte Virusforsch. 1967, 21, 205–216. [Google Scholar] [CrossRef]
- Guarner, J.; Johnson, B.J.; Paddock, C.D.; Shieh, W.J.; Goldsmith, C.S.; Reynolds, M.G.; Damon, I.K.; Regnery, R.L.; Zaki, S.R.; Greer, P.; et al. Monkeypox Transmission and Pathogenesis in Prairie Dogs. Emerg. Infect. Dis. 2004, 10, 426–431. [Google Scholar] [CrossRef]
- Smith, S.K.; Self, J.; Weiss, S.; Carroll, D.; Braden, Z.; Regnery, R.L.; Davidson, W.; Jordan, R.; Hruby, D.E.; Damon, I.K. Effective Antiviral Treatment of Systemic Orthopoxvirus Disease: ST-246 Treatment of Prairie Dogs Infected with Monkeypox Virus. J. Virol. 2011, 85, 9176–9187. [Google Scholar] [CrossRef]
- Falendysz, E.A.; Londoño-Navas, A.M.; Meteyer, C.U.; Pussini, N.; Lopera, J.G.; Osorio, J.E.; Rocke, T.E. Evaluation of monkeypox virus infection of black-tailed prairie dogs (Cynomys Ludovicianus) using in vivo bioluminescent imaging. J. Wildl. Dis. 2014, 50, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Rimoin, A.W.; Alfonso, V.H.; Hoff, N.A.; Doshi, R.H.; Mulembakani, P.; Kisalu, N.K.; Muyembe, J.J.; Okitolonda, E.W.; Wright, L.L. Human Exposure to Wild Animals in the Sankuru Province of the Democratic Republic of the Congo. Ecohealth 2017, 14, 552–563. [Google Scholar] [CrossRef]
- Khodakevich, L.; Jezek, Z.; Kinzanzka, K. Isolation of Monkeypox Virus From Wild Squirrel Infected in Nature. Lancet 1986, 327, 98–99. [Google Scholar] [CrossRef]
- Thomassen, H.A.; Fuller, T.; Asefi-Najafabady, S.; Shiplacoff, J.A.G.; Mulembakani, P.M.; Blumberg, S.; Johnston, S.C.; Kisalu, N.K.; Kinkela, T.L.; Fair, J.N.; et al. Pathogen-Host Associations and Predicted Range Shifts of Human Monkeypox in Response to Climate Change in Central Africa. PLoS ONE 2013, 8, e66071. [Google Scholar] [CrossRef] [PubMed]
- Osorio, J.E.; Iams, K.P.; Meteyer, C.U.; Rocke, T.E. Comparison of monkeypox viruses pathogenesis in mice by in vivo imaging. PLoS ONE 2009, 4, 1–10. [Google Scholar] [CrossRef]
- Zaucha, G.M.; Jahrling, P.B.; Geisbert, T.W.; Swearengen, J.R.; Hensley, L. The pathology of experimental aerosolized monkeypox virus infection in cynomolgus monkeys (Macaca fascicularis). Lab. Investig. 2001, 81, 1581–1600. [Google Scholar] [CrossRef]
- Reece, S.; Simmons, C.P.; Fitzhenry, R.J.; Matthews, S.; Phillips, A.D.; Dougan, G.; Frankel, G. Site-directed mutagenesis of intimin α modulates intimin-mediated tissue tropism and host specificity. Mol. Microbiol. 2001, 40, 86–98. [Google Scholar] [CrossRef]
- DuShane, J.K.; Maginnis, M.S. Human DNA virus exploitation of the MAPK-ERK cascade. Int. J. Mol. Sci. 2019, 20, 3427. [Google Scholar] [CrossRef] [PubMed]
- Greber, U.F. Signalling in viral entry. Cell. Mol. Life Sci. 2002, 59, 608–626. [Google Scholar] [CrossRef]
- Krajcsi, P.; Wold, W.S.M. Viral proteins that regulate cellular signalling. J. Gen. Virol. 1998, 79, 1323–1335. [Google Scholar] [CrossRef]
- Bourquain, D.; Dabrowski, P.W.; Nitsche, A. Comparison of host cell gene expression in cowpox, monkeypox or vaccinia virus-infected cells reveals virus-specific regulation of immune response genes. Virology 2013, 10, 1–13. [Google Scholar] [CrossRef]
- Kyriakis, J.M.; Avruch, J. MAP Kinase Pathways. Compend. Inflamm. Dis. 2016, 892–908. [Google Scholar] [CrossRef]
- Lu, Y.; Stuart, J.H.; Talbot-Cooper, C.; Agrawal-Singh, S.; Huntly, B.; Smid, A.I.; Snowden, J.S.; Dupont, L.; Smith, G.L. Histone deacetylase 4 promotes type I interferon signaling, restricts DNA viruses, and is degraded via vaccinia virus protein C6. Proc. Natl. Acad. Sci. USA 2019, 116, 11997–12006. [Google Scholar] [CrossRef]
- Richter, K.; Haslbeck, M.; Buchner, J. The Heat Shock Response: Life on the Verge of Death. Mol. Cell 2010, 40, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Liem, J.; Liu, J. Stress Beyond Translation: Poxviruses and More. Viruses 2016, 8, 169. [Google Scholar] [CrossRef]
- Filone, C.M.; Caballero, I.S.; Dower, K.; Mendillo, M.L.; Cowley, G.S.; Santagata, S.; Rozelle, D.K.; Yen, J.; Rubins, K.H.; Hacohen, N.; et al. The Master Regulator of the Cellular Stress Response (HSF1) Is Critical for Orthopoxvirus Infection. PLoS Pathog. 2014, 10, 1–14. [Google Scholar] [CrossRef]
- Amaya, M.; Keck, F.; Bailey, C.; Narayanan, A. The role of the IKK complex in viral infections. Pathog. Dis. 2014, 72, 32–44. [Google Scholar] [CrossRef]
- Shchelkunov, S.N. Orthopoxvirus genes that mediate disease virulence and host tropism. Adv. Virol. 2012, 2012, 1–17. [Google Scholar] [CrossRef]
- Izmailyan, R.; Hsao, J.-C.; Chung, C.-S.; Chen, C.-H.; Hsu, P.W.-C.; Liao, C.-L.; Chang, W. Integrin β1 Mediates Vaccinia Virus Entry through Activation of PI3K/Akt Signaling. Virology 2012, 86, 6677–6687. [Google Scholar] [CrossRef]
- Benedict, C.A.; Ware, C.F. Poxviruses aren’t StuPYD. Immunity 2005, 23, 553–555. [Google Scholar] [CrossRef][Green Version]
- Townsend, M.B.; Keckler, M.S.; Patel, N.; Davies, D.H.; Felgner, P.; Damon, I.K.; Karem, K.L. Humoral Immunity to Smallpox Vaccines and Monkeypox Virus Challenge: Proteomic Assessment and Clinical Correlations. J. Virol. 2013, 87, 900–911. [Google Scholar] [CrossRef]
- Song, H.; Josleyn, N.; Janosko, K.; Skinner, J.; Reeves, R.K.; Cohen, M.; Jett, C.; Johnson, R.; Blaney, J.E.; Bollinger, L.; et al. Monkeypox Virus Infection of Rhesus Macaques Induces Massive Expansion of Natural Killer Cells but Suppresses Natural Killer Cell Functions. PLoS ONE 2013, 8, e77804. [Google Scholar] [CrossRef]
- Hammarlund, E.; Dasgupta, A.; Pinilla, C.; Norori, P.; Früh, K.; Slifka, M.K. Monkeypox virus evades antiviral CD4+ and CD8+ T cell responses by suppressing cognate T cell activation. Proc. Natl. Acad. Sci. USA 2008, 105, 14567–14572. [Google Scholar] [CrossRef]
- Wenner, H.A.; Bolano, C.; Cho, C.T.; Kamitsuka, P.S. Monkey pox. iv. modification of disease pattern by antilymphocytic sera. J. Infect. Dis. 1969, 120, 318–331. [Google Scholar] [CrossRef]
- Jahrling, P.B.; Hensley, L.E.; Martinez, M.J.; LeDuc, J.W.; Rubins, K.H.; Relman, D.A.; Huggins, J.W. Exploring the potential of variola virus infection of cynomolgus macaques as a model for human smallpox. Proc. Natl. Acad. Sci. USA 2004, 101, 15196–15200. [Google Scholar] [CrossRef]
- Downie, A.W.; Mccarthy, K.; Macdonald, A. Virus and virus antigen in the blood of smallpox patients. Their significance in early diagnosis. Lancet 1953, 164–166. [Google Scholar] [CrossRef]
- Akinyemi, A.I.; Isiugo-Abanihe, U.C. Demographic dynamics and development in Nigeria: Issues and perspectives. Etude Popul. Afr. 2013, 27, 239–248. [Google Scholar] [CrossRef]
- Nigeria Population (2020)—Worldometer. Available online: https://www.worldometers.info/world-population/nigeria-population/ (accessed on 21 October 2020).
- Nigeria: Age Distribution of Population, by Gender 2019|Statista. Available online: https://www.statista.com/statistics/1121317/age-distribution-of-population-in-nigeria-by-gender/ (accessed on 21 October 2020).
- Petersen, B.W.; Damon, I.K.; Pertowski, C.A.; Meaney, D.; Guarnizo, J.T.; Beigi, R.H.; Edwards, K.M.; Fisher, M.C.; Frey, S.E.; Lynfield, R.; et al. Clinical Guidance for Smallpox Vaccine Use in a Postevent Vaccination Program. Morb. Mortal. Wkly. Rep. Recomm. Rep. 2015, 64, 1–26. [Google Scholar]
- Petersen, B.W.; Kabamba, J.; Mccollum, A.M.; Lushima, R.S.; Wemakoy, E.O.; Jacques, J.; Tamfum, M.; Nguete, B.; Reynolds, M.G. Vaccinating against monkeypox in the Democratic Republic of the Congo. Antivir. Res. 2018, 11, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Treatment|Monkeypox|Poxvirus|CDC. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/treatment.html (accessed on 22 June 2020).
- Vaccines|Smallpox|CDC. Available online: https://www.cdc.gov/smallpox/clinicians/vaccines.html (accessed on 22 June 2020).
- Wogu, J.O.; Chukwu, C.O.; Orekyeh, E.S.S.; Nwankiti, C.O.; Okoye-Ugwu, S. Assessment of media reportage of monkeypox in southern Nigeria. Medicine (Baltim.) 2020, 99, 1–4. [Google Scholar] [CrossRef]
- Delaune, D.; Iseni, F. Drug development against smallpox: Present and future. Antimicrob. Agents Chemother. 2020, 64, 1–22. [Google Scholar] [CrossRef]
- Merchlinsky, M.; Albright, A.; Olson, V.; Schiltz, H.; Merkeley, T.; Hughes, C.; Petersen, B.; Challberg, M. The development and approval of tecoviromat (TPOXX®), the first antiviral against smallpox. Antivir. Res. 2019, 168, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.O.; Bray, M.; Huggins, J.W. Potential antiviral therapeutics for smallpox, monkeypox and other orthopoxvirus infections. Antivir. Res. 2003, 57, 13–23. [Google Scholar] [CrossRef]
- Durski, K.N.; McCollum, A.M.; Nakazawa, Y.; Petersen, B.W.; Reynolds, M.G.; Briand, S.; Harouna Djingarey, M.; Olson, V.; Damon, I.K.; Khalakdina, A. Emergence of Monkeypox—West and Central Africa, 1970–2017. Morb. Mortal. Wkly. Rep. 2018, 67, 306. [Google Scholar] [CrossRef]
- OT, O.; OM, M. Monkeypox in Nigeria: A case report of re-emerged disease outbreak. J. Microbiol. Exp. 2018, 6, 89–91. [Google Scholar] [CrossRef][Green Version]
- Nigeria Population (2020)—Population Stat. Available online: https://populationstat.com/nigeria/ (accessed on 27 September 2020).
- Bourke, A.T.; Dumbell, K.R. An unusual poxvirus from Nigeria. Bull. World Health Organ. 1972, 46, 621–623. [Google Scholar]
- Silenou, B.C.; Tom-Aba, D.; Adeoye, O.; Arinze, C.C.; Oyiri, F.; Suleman, A.K.; Yinka-Ogunleye, A.; Dörrbecker, J.; Ihekweazu, C.; Krause, G. Use of surveillance outbreak response management and analysis system for human monkeypox outbreak, Nigeria, 2017–2019. Emerg. Infect. Dis. 2020, 26, 345–349. [Google Scholar] [CrossRef]
- Gayawan, E.; Arogundade, E.; Adebayo, S.B. Possible determinants and spatial patterns of anaemia among young children in Nigeria: A Bayesian semi-parametric modelling. Int. Health 2014, 6, 35–45. [Google Scholar] [CrossRef]
- Okanume, O. Ben Perception of Warri Metropolitan Residents on Online Newspaper Reportage of the Monkey Pox Virus Vaccination Hoax. Int. J. Multidiscip. Res. Publ. 2018, 1, 1–8. [Google Scholar]
- Joshua, D.S.; Ajah, I. Knowledge and Perception of Monekeypox Disease in Yenagoa, Bayelsa State. FUDMA J. Sci. 2019, 3, 418–426. [Google Scholar] [CrossRef]
- Nigeria Centre for Disease Control (NCDC). Nigeria Monkeypox Monthly Situation Report; Nigeria Centre for Disease Control (NCDC): Abuja, Nigeria, 2019.
- Khodakevich, L.; Jezek, Z.; Messinger, D. Monkeypox virus: Ecology and public health significance. Bull. World Health Organ. 1988, 66, 747–752. [Google Scholar]
- Olayemi, A.; Akinpelu, A. Diversity and distribution of murid rodent populations between forest and derived savanna sites within south western Nigeria. Biodivers. Conserv. 2008, 17, 2411–2425. [Google Scholar] [CrossRef]
- Angelici, F.M.; Akani, G.C. Some new mammal records from the rainforests of south-eastern Nigeria. Hystrix, Ital. J. Mammal. 2001, 12, 37–43. [Google Scholar] [CrossRef]
- Petrozzi, F.; Akani, G.C.; Amadi, N.; Eniang, E.A.; Gippoliti, S.; Luiselli, L. Surveys of mammal communities in a system of five forest reserves suggest an ongoing biotic homogenization process for the Niger Delta (Nigeria). Trop. Zool. 2015, 28, 95–113. [Google Scholar] [CrossRef]
- Reynolds, M.G.; Damon, I.K. Outbreaks of human monkeypox after cessation of smallpox vaccination. Trends Microbiol. 2012, 20, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Faye, O.; Pratt, C.B.; Faye, M.; Fall, G.; Chitty, J.A.; Diagne, M.M.; Wiley, M.R.; Yinka-Ogunleye, A.F.; Aruna, S.; Etebu, E.N.; et al. Genomic characterisation of human monkeypox virus in Nigeria. Lancet Infect. Dis. 2018, 18, 246. [Google Scholar] [CrossRef]
- Nigeria Centre for Disease Control (NCDC). Monkeypox Outbreak in Nigeria December 2017 Report; Nigeria Centre for Disease Control (NCDC): Abuja, Nigeria, 2017.
- NCDC. Nigeria Monkeypox Outbreak Report; NCDC: Abuja, Nigeria, 2018.
- Nigeria Centre for Disease Control (NCDC). Monkeypox Outbreak in Nigeria; Nigeria Centre for Disease Control (NCDC): Abuja, Nigeria, 2018.
- Nigeria Centre for Disease Control (NCDC). Monkeypox Outbreak in Nigeria; Nigeria Centre for Disease Control (NCDC): Abuja, Nigeria, 2017.
- Nigeria Centre for Disease Control (NCDC). First-Case-of-Corona-Virus-Disease-Confirmed-in-Nigeria. Available online: https://ncdc.gov.ng/news/227/first-case-of-corona-virus-disease-confirmed-in-nigeria (accessed on 25 July 2020).
- Wogu, J.O.; Chukwu, C.O.; Nwafor, K.A.; Anikpe, E.A.; Ugwuoke, J.C.; Ugwulor-Onyinyechi, C.C.; Eseadi, C. Mass media reportage of Lassa fever in Nigeria: A viewpoint. J. Int. Med. Res. 2019, 48, 0300060518821552. [Google Scholar] [CrossRef]
- Sivapalasingam, S.; Kennedy, J.S.; Borkowsky, W.; Valentine, F.; Zhan, M.X.; Pazoles, P.; Paolino, A.; Ennis, F.A.; Steigbigel, N.H. Immunological memory after exposure to variola virus, monkeypox virus, and vaccinia virus. J. Infect. Dis. 2007, 195, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Hammarlund, E.; Lewis, M.W.; Carter, S.V.; Amanna, I.; Hansen, S.G.; Strelow, L.I.; Wong, S.W.; Yoshihara, P.; Hanifin, J.M.; Slifka, M.K. Multiple diagnostic techniques identify previously vaccinated individuals with protective immunity against monkeypox. Nat. Med. 2005, 11, 1005–1011. [Google Scholar] [CrossRef]
- Simpson, K.; Heymann, D.; Brown, C.S.; Edmunds, W.J.; Elsgaard, J.; Fine, P.; Hochrein, H.; Hoff, N.A.; Green, A.; Ihekweazu, C.; et al. Human monkeypox—After 40 years, an unintended consequence of smallpox eradication. Vaccine 2020, 38, 5077–5081. [Google Scholar] [CrossRef]
- Sejvar, J.J.; Chowdary, Y.; Schomogyi, M.; Stevens, J.; Patel, J.; Karem, K.; Fischer, M.; Kuehnert, M.J.; Zaki, S.R.; Paddock, C.D.; et al. Human monkeypox infection: A family cluster in the Midwestern United States. J. Infect. Dis. 2004, 190, 1833–1840. [Google Scholar] [CrossRef]
- Centre for Disease Control. Multistate Outbreak of Monkeypox—Illinois, Indiana, and Wisconsin, 2003; American Medical Association (AMA): Chicago, IL, USA, 2003; Volume 52. [Google Scholar]
- Erez, N.; Achdout, H.; Milrot, E.; Schwartz, Y.; Wiener-Well, Y.; Paran, N.; Politi, B.; Tamir, H.; Israely, T.; Weiss, S.; et al. Diagnosis of imported monkeypox, Israel, 2018. Emerg. Infect. Dis. 2019, 25, 980–983. [Google Scholar] [CrossRef]
- Vaughan, A.; Aarons, E.; Astbury, J.; Brooks, T.; Chand, M.; Flegg, P.; Hardman, A.; Harper, N.; Jarvis, R.; Mawdsley, S.; et al. Human-to-human transmission of monkeypox virus, United Kingdom, October 2018. Emerg. Infect. Dis. 2020, 26, 782–785. [Google Scholar] [CrossRef]
- Fang, E.S.Y.; Ng, O.T.; Marc, J.Z.H.; Mak, T.M.; Marimuthu, K.; Vasoo, S.; Yeo, T.W.; Ng, Y.K.; Cui, L.; Ferdous, Z.; et al. Imported Monkeypox, Singapore. Emerg. Infect. Dis. 2020, 26, 1826–1830. [Google Scholar]
- Pickett, B.E.; Sadat, E.L.; Zhang, Y.; Noronha, J.M.; Squires, R.B.; Hunt, V.; Liu, M.; Kumar, S.; Zaremba, S.; Gu, Z.; et al. ViPR: An open bioinformatics database and analysis resource for virology research. Nucleic Acids Res. 2012, 40, 593–598. [Google Scholar] [CrossRef]
- Nakazawa, Y.; Mauldin, M.R.; Emerson, G.L.; Reynolds, M.G.; Lash, R.R.; Gao, J.; Zhao, H.; Li, Y.; Muyembe, J.J.; Kingebeni, P.M.; et al. A phylogeographic investigation of African monkeypox. Viruses 2015, 7, 2168–2184. [Google Scholar] [CrossRef]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2018, 20, 1160–1166. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Mauldin, M.R.; McCollum, A.M.; Nakazawa, Y.J.; Mandra, A.; Whitehouse, E.R.; Davidson, W.; Zhao, H.; Gao, J.; Li, Y.; Doty, J.; et al. Exportation of Monkeypox virus from the African continent. Infect. Dis. Soc. Am. 2020, 1–10. [Google Scholar]
- Likos, A.M.; Sammons, S.A.; Olson, V.A.; Frace, A.M.; Li, Y.; Olsen-Rasmussen, M.; Davidson, W.; Galloway, R.; Khristova, M.L.; Reynolds, M.G.; et al. A tale of two clades: Monkeypox viruses. J. Gen. Virol. 2005, 86, 2661–2672. [Google Scholar] [CrossRef]
- Carroll, D.S.; Emerson, G.L.; Li, Y.; Sammons, S.; Olson, V.; Frace, M.; Czerny, C.P.; Tryland, M.; Kolodziejek, J.; Nowotny, N.; et al. Chasing Jenner’s Vaccine: Revisiting Cowpox Virus Classification. PLoS ONE 2011, 6, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Franke, A.; Pfaff, F.; Jenckel, M.; Hoffmann, B.; Höper, D.; Antwerpen, M.; Meyer, H.; Beer, M.; Hoffmann, D. Classification of cowpox viruses into several distinct clades and identification of a novel lineage. Viruses 2017, 9, 142. [Google Scholar] [CrossRef] [PubMed]
- Smithson, C.; Kampman, S.; Hetman, B.M.; Upton, C. Incongruencies in vaccinia virus phylogenetic trees. Computation 2014, 2, 182–198. [Google Scholar] [CrossRef]
- Posada, D. jModelTest: Phylogenetic model averaging. Mol. Biol. Evol. 2008, 25, 1253–1256. [Google Scholar] [CrossRef]
- Gubser, C.; Hué, S.; Kellam, P.; Smith, G.L. Poxvirus genomes: A phylogenetic analysis. J. Gen. Virol. 2004, 85, 105–117. [Google Scholar] [CrossRef]
- Pérez-Losada, M.; Arenas, M.; Galán, J.C.; Palero, F.; González-Candelas, F. Recombination in viruses: Mechanisms, methods of study, and evolutionary consequences. Infect. Genet. Evol. 2015, 30, 296–307. [Google Scholar] [CrossRef]
- Okeke, M.I.; Hansen, H.; Traavik, T. A naturally occurring cowpox virus with an ectromelia virus A-type inclusion protein gene displays atypical A-type inclusions. Infect. Genet. Evol. 2012, 12, 160–168. [Google Scholar] [CrossRef]
- Esposito, J.J.; Sammons, S.A.; Frace, A.M.; Osborne, J.D.; Glsen-Rasmussen, M.; Zhang, M.; Govil, D.; Damon, I.K.; Kline, R.; Laker, M.; et al. Genome sequence diversity and clues to the evolution of variola (smallpox) virus. Science 2006, 313, 807–812. [Google Scholar] [CrossRef]
- Coulson, D.; Upton, C. Characterization of indels in poxvirus genomes. Virus Genes 2011, 42, 171–177. [Google Scholar] [CrossRef]
- Babkin, I.V.; Babkina, I.N. A retrospective study of the orthopoxvirus molecular evolution. Infect. Genet. Evol. 2012, 12, 1597–1604. [Google Scholar] [CrossRef]
- Sasani, T.A.; Cone, K.R.; Quinlan, A.R.; Elde, N.C. Long read sequencing reveals poxvirus evolution through rapid homogenization of gene arrays. eLife 2018, 7, 1–24. [Google Scholar] [CrossRef]
- Cone, K.R.; Kronenberg, Z.N.; Yandell, M.; Elde, N.C. Emergence of a Viral RNA Polymerase Variant during Gene Copy Number Amplification Promotes Rapid Evolution of Vaccinia Virus. J. Virol. 2017, 91, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Estep, R.D.; Messaoudi, I.; O’Connor, M.A.; Li, H.; Sprague, J.; Barron, A.; Engelmann, F.; Yen, B.; Powers, M.F.; Jones, J.M.; et al. Deletion of the Monkeypox Virus Inhibitor of Complement Enzymes Locus Impacts the Adaptive Immune Response to Monkeypox Virus in a Nonhuman Primate Model of Infection. J. Virol. 2011, 85, 9527–9542. [Google Scholar] [CrossRef] [PubMed]
- Goff, A.; Mucker, E.; Raymond, J.; Fisher, R.; Bray, M.; Hensley, L.; Paragas, J. Infection of cynomolgus macaques with a recombinant monkeypox virus encoding green fluorescent protein. Arch. Virol. 2011, 156, 1877–1881. [Google Scholar] [CrossRef]
- Hughes, A.L.; Friedman, R. Poxvirus genome evolution by gene gain and loss. Mol. Phylogenet. Evol. 2005, 35, 186–195. [Google Scholar] [CrossRef]
- Hendrickson, R.C.; Wang, C.; Hatcher, E.L.; Lefkowitz, E.J. Orthopoxvirus genome evolution: The role of gene loss. Viruses 2010, 2, 1933–1967. [Google Scholar] [CrossRef]
- Meyer, H.; Sutter, G.; Mayr, A. Mapping of deletions in the genome of the highly attenuated vaccinia virus MVA and their influence on virulence. J. Gen. Virol. 1991, 72, 1031–1038. [Google Scholar] [CrossRef]
- Hatcher, E.L.; Hendrickson, R.C.; Lefkowitz, E.J. Identification of Nucleotide-Level Changes Impacting Gene Content and Genome Evolution in Orthopoxviruses. J. Virol. 2014, 88, 13651–13668. [Google Scholar] [CrossRef]
- Elde, N.C.; Child, S.J.; Eickbush, M.T.; Kitzman, J.O.; Rogers, K.S.; Shendure, J.; Geballe, A.P.; Malik, H.S. Poxviruses deploy genomic accordions to adapt rapidly against host antiviral defenses. Cell 2012, 150, 831–841. [Google Scholar] [CrossRef]
- Falendysz, E.A.; Lopera, J.G.; Doty, J.B.; Nakazawa, Y.; Crill, C.; Lorenzsonn, F.; Kalemba, L.N.; Ronderos, M.D.; Mejia, A.; Malekani, J.M.; et al. Characterization of Monkeypox virus infection in African rope squirrels (Funisciurus sp.). PLoS Negl. Trop. Dis. 2017, 11, 1–23. [Google Scholar] [CrossRef]
- Keasey, S.; Pugh, C.; Tikhonov, A.; Chen, G.; Schweitzer, B.; Nalca, A.; Ulrich, R.G. Proteomic basis of the antibody response to monkeypox virus infection examined in cynomolgus macaques and a comparison to human smallpox vaccination. PLoS ONE 2010, 5, e15547. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Doty, J.B.; McCollum, A.M.; Olson, V.A.; Nakazawa, Y. Monkeypox re-emergence in Africa: A call to expand the concept and practice of One Health. Expert Rev. Anti. Infect. Ther. 2019, 17, 129–139. [Google Scholar] [CrossRef]
- Gruber, C.E.M.; Giombini, E.; Selleri, M.; Tausch, S.H.; Andrusch, A.; Tyshaieva, A.; Cardeti, G.; Lorenzetti, R.; De Marco, L.; Carletti, F.; et al. Whole genome characterization of orthopoxvirus (Opv) abatino, a zoonotic virus representing a putative novel clade of old world orthopoxviruses. Viruses 2018, 10, 546. [Google Scholar] [CrossRef]
- Noyce, R.S.; Evans, D.H. Synthetic horsepox viruses and the continuing debate about dual use research. PLoS Pathog. 2018, 14, 1–5. [Google Scholar] [CrossRef]
- Pajer, P.; Dresler, J.; Kab, H.; Libor, P.; Aganov, P.; Fucik, K.; Elleder, D.; Hron, T.; Kuzelka, V.; Velem, P.; et al. Characterization of Two Historic Smallpox Specimens from a Czech Museum. Viruses 2017, 9, 200. [Google Scholar] [CrossRef] [PubMed]
- Townsend, M.B.; Gallardo-romero, N.F.; Khmaladze, E.; Vora, N.M.; Maghlakelidze, G.; Geleishvili, M.; Carroll, D.S.; Emerson, G.L.; Reynolds, M.G.; Satheshkumar, P.S. Retrospective Proteomic Analysis of Serum After Akhmeta Virus Infection: New Suspect Case Identification and Insights Into Poxvirus Humoral Immunity. J. Infect. Dis. 2017, 216, 1505–1512. [Google Scholar] [CrossRef]
- Rimoin, A.W.; Kisalu, N.; Kebela-Ilunga, B.; Mukaba, T.; Wright, L.L.; Formenty, P.; Wolfe, N.D.; Shongo, R.L.; Tshioko, F.; Okitolonda, E.; et al. Endemic Human Monkeypox, Republic of Congo, Democratic 2001–2004. J. Phys. A Math. Theor. 2011, 44, 1689–1699. [Google Scholar] [CrossRef]
- Hoff, N.A.; Morier, D.S.; Kisalu, N.K.; Johnston, S.C.; Doshi, R.H.; Hensley, L.E.; Okitolonda-Wemakoy, E.; Muyembe-Tamfum, J.J.; Lloyd-Smith, J.O.; Rimoin, A.W. Varicella Coinfection in Patients with Active Monkeypox in the Democratic Republic of the Congo. Ecohealth 2017, 14, 564–574. [Google Scholar] [CrossRef]
- Meyer, H.; Perrichot, M.; Stemmler, M.; Emmerich, P.; Schmitz, H.; Varaine, F.; Shungu, R.; Tshioko, F.; Formenty, P. Outbreaks of disease suspected of being due to human monkeypox virus infection in the Democratic Republic of Congo in 2001. J. Clin. Microbiol. 2002, 40, 2919–2921. [Google Scholar] [CrossRef]
- Ogoina, D.; Iroezindu, M.; James, H.I.; Oladokun, R.; Yinka-Ogunleye, A.; Wakama, P.; Otike-odibi, B.; Usman, L.M.; Obazee, E.; Aruna, O.; et al. Clinical Course and Outcome of Human Monkeypox in Nigeria. Clin. Infect. Dis. 2020, 16, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Bhunu, C.P.; Mushayabasa, S.; Hyman, J.M. Modelling HIV/AIDS and monkeypox co-infection. Appl. Math. Comput. 2012, 218, 9504–9518. [Google Scholar] [CrossRef]
- Rubins, K.H.; Hensley, L.E.; Bell, G.W.; Wang, C.; Lefkowitz, E.J.; Patrick, O.; Relman, D.A. Comparative Analysis of Viral Gene Expression Programs during Poxvirus Infection: A Transcriptional Map of the Vaccinia and Monkeypox Genomes. PLoS ONE 2008, 3, 1–12. [Google Scholar] [CrossRef]
- Alkhalil, A.; Hammamieh, R.; Hardick, J.; Ichou, M.A.; Jett, M.; Ibrahim, S. Gene expression profiling of monkeypox virus-infected cells reveals novel interfaces for host-virus interactions. Virology 2010, 7, 1–19. [Google Scholar] [CrossRef]
- Brown, J.N.; Estep, R.D.; Lopez-ferrer, D.; Brewer, H.M.; Clauss, T.R.; Manes, N.P.; Connor, M.O.; Li, H.; Adkins, J.N.; Wong, S.W.; et al. Characterization of Macaque Pulmonary Fluid Proteome during Monkeypox Infection. Mol. Cell. Proteom. 2010, 9, 2760–2771. [Google Scholar] [CrossRef] [PubMed]
- Arndt, W.D.; Cotsmire, S.; Trainor, K.; Harrington, H.; Hauns, K.; Kibler, K.V.; Huynh, T.P.; Jacobs, B.L. Evasion of the Innate Immune Type I Interferon System by Monkeypox Virus. J. Virol. 2015, 89, 10489–10499. [Google Scholar] [CrossRef]
- Rubins, K.H.; Hensley, L.E.; Relman, D.A.; Brown, P.O. Stunned silence: Gene expression programs in human cells infected with monkeypox or vaccinia virus. PLoS ONE 2011, 6, e15615. [Google Scholar] [CrossRef]
- Kulkarni, R. Antibody-Dependent Enhancement of Viral Infections. In Dynamics of Immune Activation in Viral Diseases; Bramhachari, P.V., Ed.; Springer: New York, NY, USA, 2020; pp. 1–283. ISBN 9789811510458. [Google Scholar]
- Morens, D.M. Antibody-dependent enhancement of infection and the pathogenesis of viral disease. Clin. Infect. Dis. 1994, 19, 500–512. [Google Scholar] [CrossRef]
- Arvin, A.M.; Fink, K.; Schmid, M.A.; Cathcart, A.; Spreafico, R.; Havenar-Daughton, C.; Lanzavecchia, A.; Corti, D.; Virgin, H.W. A perspective on potential antibody-dependent enhancement of SARS-CoV-2. Nature 2020, 584, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Eroshenko, N.; Gill, T.; Keaveney, M.K.; Church, G.M.; Trevejo, J.M.; Rajaniemi, H. Implications of antibody-dependent enhancement of infection for SARS-CoV-2 countermeasures. Nat. Biotechnol. 2020, 38, 788–789. [Google Scholar] [CrossRef]
- Tryland, M.; Nilsen, H.; Olsvik, O. Antibodies to orthopoxvirus in domestic cats in Norway. Vet. Rec. 1998, 143, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Kinnunen, P.M.; Henttonen, H.; Hoffmann, B.; Kallio, E.R. Orthopox Virus Infections in Eurasian Wild Rodents. Vector-Bone Zoonotic Dseases 2011, 11. [Google Scholar] [CrossRef]
- Heuser, E.; Fischer, S.; Ryll, R.; Mayer-Scholl, A.; Hoffmann, D.; Spahr, C.; Imholt, C.; Alfa, D.M.; Fröhlich, A.; Lüschow, D.; et al. Survey for zoonotic pathogens in Norway rat populations from Europe. Pest Manag. Sci. 2017, 73, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Stagegaard, J.; Kurth, A.; Stern, D.; Dabrowski, P.W.; Pocknell, A.; Nitsche, A.; Schrick, L. Seasonal recurrence of cowpox virus outbreaks in captive cheetahs (Acinonyx jubatus). PLoS ONE 2017, 12, 1–15. [Google Scholar] [CrossRef]
- Sandvik, T.; Tryland, M.; Hansen, H.; Mehl, R.; Moens, U.; Olsvik, Ø.; Traavik, T. Naturally occurring orthopoxviruses: Potential for recombination with vaccine vectors. J. Clin. Microbiol. 1998, 36, 2542–2547. [Google Scholar] [CrossRef]
- Tryland, M.; Okeke, M.I.; Traavik, T. Orthopoxvirus DNA in Eurasian. Emerg. Infect. Dis. 2011, 17, 1–7. [Google Scholar] [CrossRef]
- Prkno, A.; Hoffmann, D.; Goerigk, D.; Kaiser, M.; Catherine, A.; Van Maanen, F.; Jeske, K.; Jenckel, M.; Pfaff, F.; Vahlenkamp, T.W.; et al. Epidemiological Investigations of Four Cowpox Virus Outbreaks in Alpaca Herds, Germany. Viruses 2017, 9, 344. [Google Scholar] [CrossRef]
- Kaysser, P.; Von Bomhard, W.; Dobrzykowski, L.; Meyer, H. Genetic diversity of feline cowpox virus, Germany 2000–2008. Vet. Microbiol. 2011, 141, 282–288. [Google Scholar] [CrossRef]
- Fischer, S.; Franke, A.; Imholt, C.; Spierling, N.G.; Jacob, J.; Beer, M.; Hoffmann, D.; Ulrich, R.G. Patchy Occurrence of Cowpox Virus in Voles from Germany. Vector-Bone Zoonotic Dseases 2020, 20, 471–475. [Google Scholar] [CrossRef] [PubMed]

| Geographic Location/Countries | Method of Detection | References | |
|---|---|---|---|
| Sooty mangabey monkey (Cercocebus atys) | Côte d’Ivoire | PCR | [66,84] |
| Gambian-pouched rat (Cricetomys gambianus) | Africa | PCR and viral isolation | [85,86,87,88,89] |
| Rhesus macaques (Macaca mulatta) | Copenhagen | Serological test | [15,90,91,92,93,94] |
| Cynomolgus Macaque (Macaca fascicularis) | Singapore/Copenhagen | Viral isolation | [49,92,95,96] |
| Asian Monkeys (M. fascicularis) | Copenhagen | Viral isolation | [97,98,99,100] |
| Southern opossum (Didelphis marsupialis) | South America | PCR and viral isolation | [88,89,98,99] |
| Sun squirrel (Heliosciurus sp.) | Zaire | Antibody detection test | [62,88,89,98,101,102] |
| African hedgehogs (Atelerix sp.) | Africa | PCR, antibody detection test, and viral isolation | [81] |
| Jerboas (Jaculus sp.) | Illinois, USA | PCR, antibody detection test, and viral isolation | [49,81] |
| Woodchucks (Marmota monax) | USA | PCR and viral isolation | [98,100] |
| Shot-tailed opossum (Monodelphis domestica) | USA | PCR and viral isolation | [98,100] |
| Porcupines (Atherurus africanus) | Zaire | PCR and viral isolation | [58,61,78,89] |
| Giant anteaters (Myrmecophaga tridactyla) | Rotterdam | Viral isolation | [103] |
| Prairie dogs (Cynomys spp.) | USA | PCR and viral isolation | [33,81,99,104,105,106] |
| Elephant shrew (Petrodromus tetradactylus) | DR Congo | Serological test | [99,107] |
| Domestic pig (Sus scrofa) | DR Congo | Serological test | [99,108] |
| Rope squirrel (Funisciurus sp.) | Zaire | PCR and viral isolation | [55,87,88,104,107,108,109] |
| African dormice (Graphiurus spp.) | USA | PCR and viral isolation | [55,104,109] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. https://doi.org/10.3390/v12111257
Alakunle E, Moens U, Nchinda G, Okeke MI. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses. 2020; 12(11):1257. https://doi.org/10.3390/v12111257
Chicago/Turabian StyleAlakunle, Emmanuel, Ugo Moens, Godwin Nchinda, and Malachy Ifeanyi Okeke. 2020. "Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution" Viruses 12, no. 11: 1257. https://doi.org/10.3390/v12111257
APA StyleAlakunle, E., Moens, U., Nchinda, G., & Okeke, M. I. (2020). Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses, 12(11), 1257. https://doi.org/10.3390/v12111257





