Abstract
Magnolia wufengensis, a rare ornamental tree species, is now in a huge gap between market demand and actual supply of seedlings. As cutting propagation is one of the most important means to solve the shortage of seedling supply, this study developed an efficient cutting propagation procedure of M. wufengensis, revealed the morphological and histological changes of adventitious root formation, and explored the rhythm correlation between rooting process and physiological and biochemical changes. Cuttings pre-treated with NAA:IBA (2:1) exhibited the best rooting performance. Anatomical analysis demonstrated that adventitious root primordia of M. wufengensis were initiated from cambial and parenchyma cells of xylem, with no relationship to the callus formed on the epidermis. The rooting process of M. wufengenis can be divided into four periods: induction phase (0–8 dap) (dap means days after planting), initiation phase (8–13 dap), expression phase (13–18 dap), and extension phase (18–28 dap). NAA:IBA (2:1) induced the accumulations of 3-indoleacetic-acid and increased the contents of peroxidase and polyphenol-oxidase near the wounding at induction phase. The initiation phase, with the first histological modifications to the formation of meristemoids, correspond to the increase of peroxidase, polyphenol-oxidase, and soluble protein contents. The synergistic reaction of low 3-indoleacetic-acid and high levels of gibberellins and zeatin also stimulates the initiation phase. In the expression and extension phase, high activities of polyphenol-oxidase, IAA-oxidase, and increased contents of soluble protein co-stimulate the emergence and outgrowth of adventitious roots. The present study not only provides optimized protocol by application of auxin combination but also presents insights in the histological, morpho-physiological, and biochemical changes in stem cuttings of M. wufengensis.
1. Introduction
Magnolia wufengensis L.Y.Ma et L.R.Wang, a new Magnolia species (Magnoliaceae), was discovered in the southwest of Hubei Province, China [1,2] (Figure 1). It is a type of hysteranthous deciduous tree with different colorful flowers (pure, dark, or pale red) and varied petal numbers (9–25, 32, or 46) [3,4]. M. wufengensis can be used in urban landscaping and mountain forestation with broad application and popularization prospects, and it has been widely introduced to 23 provinces such as Yunnan, Guangxi, Guizhou, Henan, Jiangsu, Shandong, and Beijing in China [5]. Moreover, potentially suitable areas in the world for the introduction of this species have been identified [5].

Figure 1.
Magnolia wufengensis flower in Jiufeng, Beijing, China.
However, the slow breeding process of M. wufengensis cannot meet the increasing demands in local and international markets. Currently, M. wufengensis is propagated mainly by seed and grafting [6]. These labor-intensive and time-consuming conventional methods are costly processes such that they are not always suitable for commercial purposes [7]. These low efficiency methods spurred the development of alternative propagation methods. Taking into consideration the disadvantages of propagation through seeds and grafting, cutting propagation can improve production within a short period of time [8]. Ulmus villosa, Magnolia Biondii, Diospyros virginiana have achieved 90%, 83.11% and close to 100% of rooting percentage through cutting propagation, respectively [9,10,11], which greatly accelerated the process of seedling breeding and improved the efficiencies in horticultural industries. Thus, cutting propagation is thought to be an ideal alternative way to achieve conservation and meet the increasing demand of M. wufengensis.
Modern methods of cutting propagation have utilized sophisticated technologies such as plant growth regulators, misting and fogging systems, and advanced hygiene protocols to optimize for adventitious root formation and thereby maximize propagation efficiency [8,12]. In many difficult-to-root species, root stimulation is usually accomplished using various pre-planting hormonal treatments, such as application of the auxins indole-3-butyric acid (IBA) and 1-naphthaleneacetic acid (NAA). Recent studies have established that M. wufengensis propagation by cutting is viable, and applications of NAA and IBA are effective in stimulating the rooting capacity of M. wufengensis [13]. However, the rooting percentages were in a low range variable from 1–53.3% with the application of single auxin [13,14]. Several studies indicate that combined application of auxins enhances the rooting performance over single auxin application, and proper application of the combined auxin treatment reduces the dose usage of auxin and achieves ideal root formation and development [15,16,17,18,19]. Thus, the potential motivated us to work on the optimization of the existing protocols on exogenous hormones. Adventitious root formation from stem cuttings usually involves a series of development phase according to physiological and biochemical changes [20]. The most widely recognized development phases of adventitious rooting are the induction, the initiation, and the expression phase [21]. Some also contain extension or elongation phases [22,23,24]. Endogenous hormones, such as auxin, cytokinin, gibberellin, and abscisic acid, are the major endogenous regulators of root growth that regulate root growth through their cross-talk [25]. Oxidative enzymes such as peroxidase (POD), polyphenol oxidase (PPO), and IAA oxidase (IAAO) play an important role in the adventitious rooting process by controlling synthesis and degradation of some substances during root development [26]. Soluble protein also plays crucial role in adventitious root formation, as its content is relevant for both structural and metabolic processes of plant growth [27]. However, to our knowledge, no published reports are available on histological, morpho-physiological, and biochemical changes during the rooting process of M. wufengensis through stem cuttings. Precise knowledge about these changes during rooting provides insights into the fascinating process of adventitious root formation and open up new perspectives for sustainable and efficient utilization of M. wufengensis.
Therefore, in this study, we analyzed the histological, morpho-physiological, and biochemical changes during the adventitious root formation in M. wufengensis cuttings. The purpose of our study is to: (1) Explore the effect of auxin combinations on the comprehensive rooting performance of M. wufengensis; (2) Document the histological origin and develop a spatially explicit timeline of M. wufengensis adventitious root formation; and (3) Explore the rhythm correlation between rooting process and physiological and biochemical changes in stem cutting of M. wufengensis.
2. Materials and Methods
2.1. Plant Materials, Growth Conditions, and Planting
The investigations were carried out in Jiufeng Experiment Station of Beijing Forestry University, Beijing, located at 40°3′ N, 116°05′ E. A total of 14 cutting seedbeds were prepared, with a dimension of 1.5 × 6 × 1 m, covered with transparent plastic canvas (Figure 2).

Figure 2.
Cutting seedbeds and seedlings.
The stem cuttings were obtained from 5–6-year-old M. wufengensis plants grown at the study site, the Nursey of Jiufeng Experiment Station of Beijing Forestry University. The semi-lignified stem cuttings were collected in the early morning of cloudy days, the stem cuttings were sectioned into 12–15 cm in length, each containing 2–3 buds with two half leaves. The cuttings were then disinfected with carbendazim solution for 3 min to prevent pest and disease. Afterward, the cuttings were immersed in the specific solutions for 2 h (different combination of auxins (shown in Table 1) formulated as per each treatment. First, we dissolved IBA and NAA in 95% ethanol and 1 mol/L NaOH solutions, respectively, and then added distilled water to 1 L, stirred and heated in the water bath). The treated explants were planted vertically in the seedbeds filled with thoroughly washed river sand, previously disinfected by 0.2% potassium permanganate solution (KMnO4). Each cutting had 1/3 of its length submerged into the substrates. During the whole experiment, plastic sheets were kept on the seedbeds with one flap left open to allow air flow and were misted for an appropriate time interval by foggers that were placed at top of tents on each 1 m distance. Intermittent misting was automatically controlled between 08:00 and 21:00 at 15 min for 15 s duration to maintain relative humidity at 85–100%. On cool and cloudy days, the interval time increased to 20 min, and on hot days, the interval time reduced to 10 min. Exhaust fans and reflective aluminized shade fabric were used on extra hot and sunny days to maintain ambient air temperature between 19–29 °C. The air temperature and relative humidity data were recorded by RC-4HC temperature and humidity recorder (Figure 3).

Table 1.
Implementation methods of different auxin combinations.
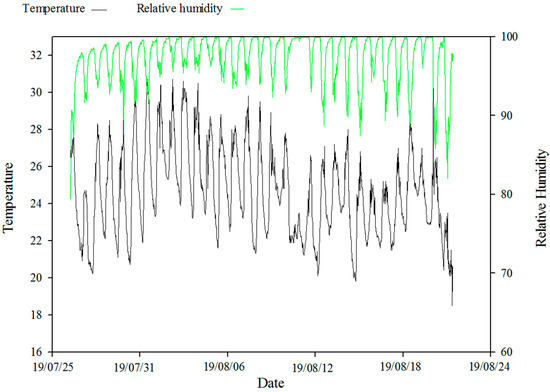
Figure 3.
Dynamic changes of ambient air temperature and relative humidity of the cutting seedbeds. Temperature and humidity data were recorded each half hour.
2.2. Rooting Assessments
This experiment was conducted to check the effect of different combinations of NAA and IBA on the semi-lignified stem cuttings of M. wufengensis. The experiment consisted of 6 treatments (Table 1), each repeated 3 times in a complete randomized design with 30 cuttings per replicates.
Data were recorded on the 80th day after planting; cuttings were carefully removed from the medium and rooting percent, number of roots per cutting, root length (cm), root diameter (mm), and root quality score rate were recorded.
Root quality score rate was measured by following the method of Wang Y et al. [13].First, we scored each rooting cutting, then calculated the root quality score rate of each treatment with Equation (1), scoring criteria of the roots are presented in Table 2.

Table 2.
Scoring criteria of roots.
Root quality score rate of each treatment =
N1, N2, N3 in Equation (1) indicates the number of cutting seedlings score 1, 2, 3.
In the next year, the best treatment according to the rooting results was selected for further experiments and 1000 cuttings were implanted for histological, morpho-physiological, and biochemical analysis.
2.3. Morphological and Histological Analysis of the Cuttings during Rooting
On the basis of continuous observation of the morphological changes in the cuttings, destructive sampling was carried out on day 0, 3, 8, 13, 18, and 28 after planting with the help of a sharp, sterilized shear and the samples were 2–3 cm long from the base. The collected samples were fixed immediately in the FAA solution (38% formaldehyde: glacial acetic: 70% ethanol 5:5:90, by vol.). The air was pulled with the help of a vacuum pump for about 2 h until the samples were sunk, eluted in alcohol, transparented in xylene, and embedded in paraffin block. Sections (5 µm) were made by Leica RM2016, cross-dyed with safranine and fast green, Canadian gum mounting, and made into permanent section. NikonECLIPSE CI Microscope and software CaseViewer 2.3 were used for observation of sections.
2.4. Measurement of Antioxidant Enzymes Activities and Soluble Protein Contents
The material for antioxidant enzymes and soluble protein were collected on day 0, 3, 8, 13, 18, 23, 28, and 38 after planting. Samples were selected randomly (20 cuttings, 3 biological replicates per treatment) and rinsed with water. The bark of the cuttings at about 0–3 cm long from the base was peeled and preserved in the liquid nitrogen and then stored at −80 °C until analyses.
To measure the activity of antioxidant enzymes and content of soluble proteins, samples were ground and 0.5 g of each sample was taken for enzyme extraction. Peroxidase (POD) activity was determined using the guaiacol chromogenic method [28], polyphenol (PPO) activity was determined using the catechol colorimetry method [29], IAA-oxidase (IAAO) activity was determined using the FeCl3 and perchloric chromogenic method [28], and the soluble protein was determined by the Coomassie brilliant blue G-250 staining method [29].
2.5. Endogenous Hormones Quantification by HPLC
The collecting time and storage method of samples were same as aforementioned in the measurement of antioxidant enzymes activities and soluble protein contents. Endogenous hormones extraction and quantification from cuttings were performed according to Xiao [30]. The measurement indicators included IAA, ABA, GA3, and ZT.
2.6. Statistical Analysis
The data recorded in the percentage were firstly transformed to arcsine value [31]. All statistical analyses were conducted using Microsoft Excel 2010 and Statistical Package for Social Sciences 20.0 (SPSS 20.0). One-way ANOVA analysis was used to test significant effect of treatments, and Duncan’s Significant Difference tests were performed for significant differences. Means were presented with standard errors. Graphs were prepared by using Sigmaplot 12.5 software.
3. Results
3.1. Effect of Auxin Combinations on Stem Cutting of M. wufengensis
Cuttings immersed in different proportions of NAA and IBA exhibited a wide range of rooting percentages (Table 3), ranging from 35.6% to 71.1%. The highest rooting percentage was noticed in NAA:IBA (2:1), it was significantly higher than other proportions (p < 0.01), compared to NAA:IBA (1:0) (average 51.1%) increased by 20%, which was about 16.16 fold higher compared to control (average 4.4%).

Table 3.
Effects of different auxin combinations on rooting percentage of M. wufengensis.
The application of auxin combinations significantly enhanced the number of roots compared to CK (Table 4). The highest number of roots (11.34) was observed in NAA:IBA (1:0), closely followed by the cuttings immersed in NAA:IBA (2:1) solutions, which measured 10.10 in the number of roots. There was no statistically significant difference between these two treatments, and they produced 6.79- and 6.05-fold more adventitious roots than CK, respectively.

Table 4.
Effect of different auxin combinations on rooting morphology indicators of M. wufengensis.
All 5 combinations of auxin showed a significant increase in average root length compared to CK (5.50 cm, p < 0.05) (Table 4). Cuttings immersed in NAA:IBA (1:0) solutions demonstrated the highest root length, 12.09 cm, had a significant difference from CK (p < 0.01). NAA:IBA (2:1) showed the lowest root length (8.19 cm), it was decreased 3.9 cm compared to NAA:IBA (1:0), and increased by about 48.9% compared to CK.
Lowest root diameter was found in CK (1.75 mm) (Table 4). It was found that NAA:IBA (0:1) treatment was ineffective to enhance the root diameter, while the other four treatments showed significantly different results compared to NAA:IBA (0:1). When the cuttings were immersed in NAA:IBA (2:1) solutions, it showed the highest root diameter (2.47 mm), which was approximately 1.41 times that of CK.
The application of NAA:IBA (0:1), NAA:IBA (1:2), and NAA:IBA (2:1) significantly enhanced the rate of root quality score compared to CK (33.33%) (Table 4). Cuttings treated with NAA:IBA (2:1) solution demonstrated the highest rate of root quality score (74.21%), which was about 2.23-fold that of CK.
In order to evaluate the comprehensive rooting effect of different treatments more intuitively, we performed a principal component analysis (Table 5) on the five morphological parameters of all treatments (all data had been standardized by SPSS for z-score). SPSS extracted 1 principal component with a feature value greater than 1. The cumulative contribution rate of the extracted principal component was 72.39%.

Table 5.
Variance and variance cumulative contribution rate of each composition.
Principal component expression and the formula of the comprehensive score are as follows:
C = 0.975X1 + 0.871X2 + 0.686X3 + 0.841X4 + 0.856X5
C indicates the comprehensive principal component score; X1 to X5 indicate rooting percentage, average number of roots, average root length, average root diameter, and rate of root quality score, respectively.
Substituting the five morphological parameters of each treatment into Equation (2), the score and ranking of each treatment were listed in Table 6. It can be seen from the comprehensive rank that among the 5 combinations of auxin, cuttings immersed in NAA:IBA (2:1) solution was found with the highest comprehensive score.

Table 6.
Principal component scores and comprehensive scores of each process.
3.2. Morphological and Histological Observations in Stem Cuttings of M. wufengensis
The morphological changes of cuttings treated with NAA:IBA (2:1) were shown in Figure 4. When compared with day 0, there were no changes in the external morphological structure in the basal cuttings on the 3rd day. The basal part of cuttings near the incision was thickened and swollen slightly on the 8th day. On the 13th day, the epidermis near the lower incision of the cuttings showed minor cracks and bursts, and then formed many white callus tissues on the epidermis. On the 18th day, the green color at the basal epidermis of cuttings gradually faded, the cracking and burst further expanded and increased, presented in longitudinal split shape. After 28 days of planting, the root primordia were developed, the transparent and white young adventitious roots penetrated the stem cortex, erupted successively in the epidermis callus, and were projected well beyond the stem surface. On the 38th day, most of the cuttings formed adventitious roots at the base of shoot stems. In successive days, the number of adventitious roots increased. In the stem explants, comparably advanced roots were observed after 58 days. On the 78th day, we observed profuse adventitious roots concentrated in the range of 3 cm from the base of cuttings, the uniformly distributed root system was developed, with a lot of lateral roots. As for CK (control), formations of the adventitious roots were less observed, and there was no obvious change at base of the cuttings from day 0 to day 8 after planting. On the 13th day, base of the cuttings formed many white callus tissues on the epidermis. In the successive 20 days, the callus continued to expand, then cuttings gradually rotted from the 38th day, and almost completely rotted on the 58th day after planting (Figure A1, Figure A2 and Figure A3 are shown in Appendix A).

Figure 4.
Morphological changes of cuttings treated with NAA:IBA (2:1) on day 0, 3, 8, 13, 18, 28, 38, 58, and 78 after planting. dap means days after planting. Bars: 1 cm.
Figure 5 shows the well-outlined configuration of M. wufengensis stem from the outside to the inside. During the rooting process, a uniseriate epidermis covered the stem surface, and a few cortical sclerenchyma and parenchyma cells which consisted of the cortex were found (Figure 5b,c). A continuous ring of sclerenchyma which contained 2–7 layers of small, oval, and compactly arranged cells were present adjacent to the cortex (Figure 5b,c). The phloem, vascular cambium, xylem, pith, and pith ray formed the vascular tissue. There was no latent root primordium observed in the sections.
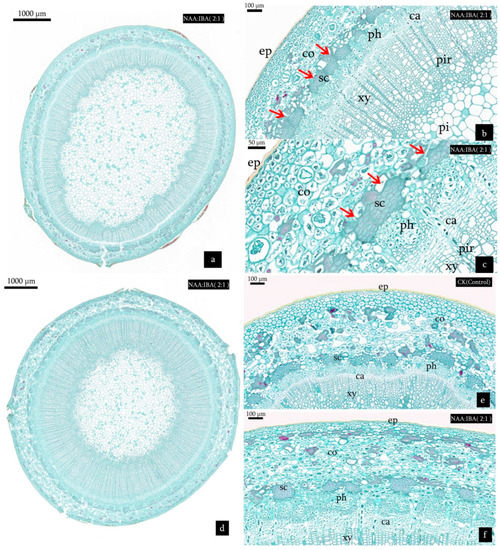
Figure 5.
Transverse sections derived from cuttings on day 0 and 8 after planting. (a) Histological structure of cutting treated with NAA:IBA (2:1) on day 0 after planting. (b,c) Close-up images of histological structures of cuttings treated with NAA:IBA (2:1). Arrow showing the ring of sclerenchyma. (d) Histological structure of the cutting treated with NAA:IBA (2:1) on the 8th day after planting. (e) Transverse sections derived from cuttings in CK (Control) on the 8th day after planting. (f) Transverse sections derived from cuttings treated with NAA:IBA (2:1) on the 8th day after planting, showing the thickening of cortex, phloem, and cambium areas. Abbreviations: ep = epidermis; co = cortex; sc = sclerenchyma; ph = phloem; ca= cambium; xy = xylem; pi = pith; pir = pith ray.
The basal part of cuttings near the incision thickened and swelled slightly on the 8th day after planting (Figure 4). Stem segments of M. wufengensis experienced an obvious thickening of cortex, phloem, and cambium areas (Figure 5e,f). The number of cortical cells of cuttings treated with NAA:IBA (2:1) increased significantly, and the cell division of phloem and cambium was vigorous, the nucleus was more prominent than that of CK (Control). Combined action of these three factors led to the thickening of cuttings.
On the 13th day after planting, the epidermis showed minor cracks and bursts, cell proliferations initiated in the cortex developed in the opening of the epidermis a callus tissue for protection against microorganism attack and drought (Figure 6a). At the same time, certain cells produced by periclinal and anticlinal mitotic divisions were occasionally detected in the cambium and xylem zone (Figure 6d,e), they were more densely stained than the cells neighbor and their nuclei became more pronounced (Figure 6f). The specific cellular mass formed by small clumps of cells with meristematic characteristics showed no overall polarity at this stage, and it might be the precursor of adventitious root primordia (Figure 6d–f). The xylem was squeezed and became scattered with the proliferation of meristematic cells (Figure 6g).
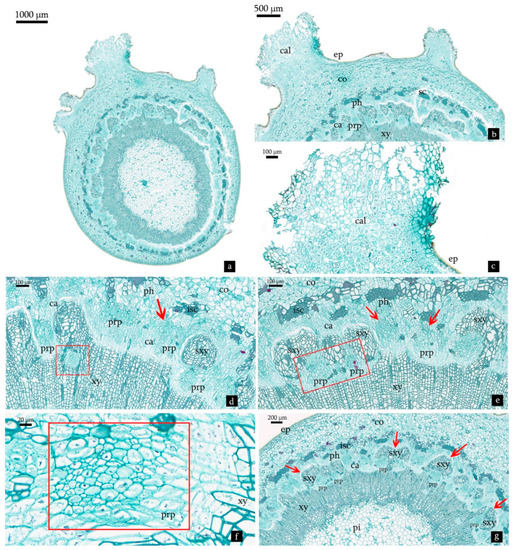
Figure 6.
Transverse sections derived from cuttings treated with NAA:IBA (2:1) on the 13th day after planting. (a) Histological structure of cutting treated with NAA:IB (2:1). (b,c) Callus tissue on the epidermis, showing cortical cell proliferation. (d) Two sources of adventitious root primordia in M. wufengensis cuttings. Red frame shows the root meristem cells initiated from xylem. Red arrow shows the root primordia precursors initiated from cambium zone. (e) Two types of root primordia precursors in M. wufengensis cuttings. Red frame shows the root primordia precursors initiated from xylem. Red arrow shows the root primordia precursors initiated from cambium zone. (f) Cells of root primordia precursors were more densely stained than the cells neighbor and their nuclei became more pronounced. (g) The scattered xylem squeezed by the proliferation of meristematic cells. Abbreviations: co = cortex; sc = sclerenchyma; ph = phloem; ca = cambium; xy = xylem; pi= pith; pir = pith ray; isc = intermittent sclerenchyma; cal = callus tissue; prp = precursor of adventitious root primordia; sxy = scattered xylem.
At 18 days after planting, the cracking and burst further expanded and increased, the number of callus tissues on epidermis increased rapidly (Figure 7a,b), accompanied with the increased amounts of the precursor root primordia in the phloem and cambium zone (Figure 7c). With cell proliferation and division, some precursors of root primordia became true root primordia (Figure 7d). At the same time, cells in some root primordia became polarized, with the apex cells of root primordia extending outward, the polarization of divisions of the root primordium gave birth to the typical pointed shape, the cells which were smaller than their neighbor were presumed as procambium strand of root primordia (Figure 7e). Other cells in the base of root primordium arranged radially were presumed to be the precursors of the future cortical and vascular tissues (Figure 7e).
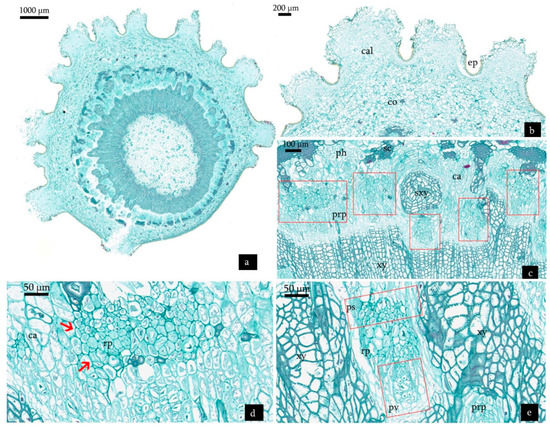
Figure 7.
Transverse sections derived from cuttings treated with NAA:IBA (2:1) on the 18th day after planting. (a) Histological structure of cuttings treated with NAA:IBA (2:1) on the 18th day after planting. (b) Increased number of callus tissue on epidermis. (c) Increased number of precursors of adventitious root primordia. Red frame shows precursors of root primordia. (d) With cell proliferation and division, some precursors of root primordia became true root primordia. Arrows showing root primordia. (e) Root primordia (dome-like structures) in differentiation. Red frame shows the procambium strand and precursors of vascular tissues. Abbreviations: ep = epidermis; co = cortex; sc = sclerenchyma; ph = phloem; ca = cambium; xy = xylem; prp = precursor of adventitious root primordia; sxy = scattered xylem; cal = callus tissue; pv = precursors of vascular tissue; rp = root primordium; ps = procambium strand.
As shown in Figure 8a, different stages of root development were observed in the same section as the adventitious root formation was an asynchronous process. On the 28th day, the differentiation of root primordia was completed (Figure 8b). The achievement of the differentiation signaled the initiation of adventitious roots. On the tip of the adventitious root, a group of cell was distinguishable arranged in a semicircle and forming a root cap (Figure 8b,c). The precursors of adventitious root vascular grew through the surrounding layers of phloem and cortical tissues and developed a true vascular system connected to that of the stem (Figure 8d,e). At the same time, further elongation of adventitious root proceeded in the cortex. When the internally growing root reached the surface of stem, it ruptured the cortex and epidermis, passed through the callus tissue, and projected well beyond the stem surface (Figure 8f). The callus tissue on epidermis was just functioning as the root cap of adventitious roots (Figure 8f,g).
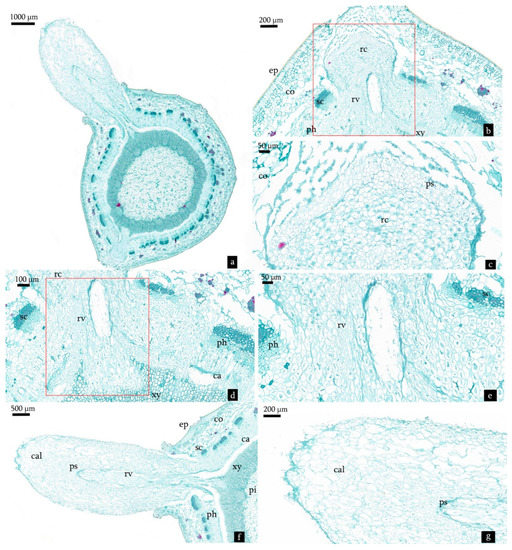
Figure 8.
Transverse sections derived from cuttings treated with NAA:IBA (2:1) on the 28th day after planting. (a) Histological structure of cuttings treated with NAA:IBA (2:1) on the 28th day after planting. (b) Root primordia which achieved differentiation pushing into the cortex through the sclerenchyma of the cuttings. Red frame shows the adventitious root. (c) Root cap of the adventitious roots. (d,e) Root vascular bundles connected with the stem. Red frame shows the root vascular. (f) The adventitious root penetrated through cortex and callus tissue. (g) The callus tissue on epidermis functionally formed the root cap of adventitious roots. Abbreviations: ep = epidermis; co = cortex; sc = sclerenchyma; ph = phloem; ca = cambium; xy = xylem; prp = precursor of adventitious root primordia; cal = callus tissue; ps = procambium strand; pi = pith; rv = root vascular cylinder; rc = root cap.
Through the histological and external morphology observations, it is clear that the adventitious roots of M. wufengensis originated from cambial and parenchyma cells of xylem, extended outwards from the cells of the phloem, sclerenchyma, cortex, epidermis, and callus tissue on epidermis to the outside of the cuttings. There was no callus rooting observed in this study. The rooting process for M. wufengenis stem cuttings can be divided into four phases: induction phase (0–8 dap), initiation phase (8–13 dap), expression phase (13–18 dap), and extension phase (18–28 dap).
3.3. Metabolic Changes during the Adventitious Root Formation of M. wufengensis
The presence of NAA and IBA in the rooting agent significantly improved POD activity (Figure 9a). POD activities in NAA:IBA (2:1)-treated cuttings were higher than CK (Control) during the whole rooting process. It followed a double-A-shaped trend, it initially increased and reached a low peak at 13 days, then dropped slightly up to the 18th day after planting. Afterward, the level of POD continuously increased, and reached the peak value on the 28th day. The time to reach the two peak values synchronized well with the initiation and extension phase of adventitious root formation.
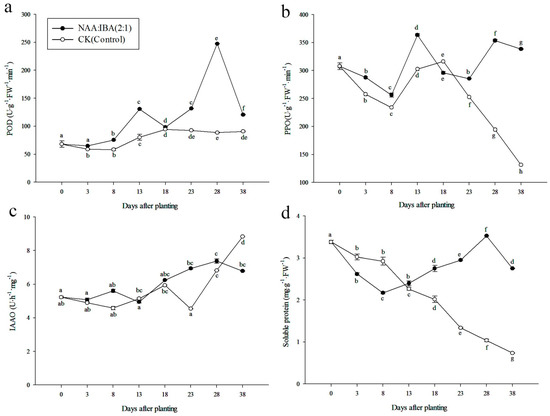
Figure 9.
Changes in the enzymatic activities and soluble protein contents during rooting in stem cuttings of M. wufengensis. (a) POD activities. (b) PPO activities. (c) IAAO activities. (d) Soluble protein contents. Data are the means of three replicates ± SE, for 20 cuttings per treatment. Letters on the top of each point show p < 0.05, same letters show the non-significant difference while different letters show a significant difference between different days after planting.
Significant differences were observed in PPO activities in NAA:IBA (2:1) (Figure 9b). PPO activity in NAA:IBA (2:1) treatment followed an A-shaped trend, it initially increased, then reached a peak value on the 28th day after planting (363.578 U·(g·FW·min)−1), which synchronized well with extension phase, whereas the activities of PPO were found lower in CK than NAA:IBA (2:1)-treated cuttings during rooting process, which might have induced the low rooting percentage.
Supplementation of NAA with IBA significantly affected the activity of IAAO (Figure 9c). The IAAO activity trend showed less remarkable changes than POD and PPO. Enzymatic activities of IAAO in NAA:IBA (2:1) followed a double-A-shaped trend: it continuously increased and showed the first peak on the 8th day after planting (5.60 U·h−1·mg−1), then showed stabilization and modest increase of IAAO activities in expression and extension phase, which had no significant differences in days 13–38. At initiation phase, the IAAO activity was low which might be related to high IAA content. In CK, IAAO activities showed a remarkable increase at initiation, expression, and extension phase, reached the peak value at 38 days (8.8267 U·h−1·mg−1), and was 1.3-fold that of the NAA:IBA (2:1) treatment.
The soluble protein contents of CK unceasingly decreased and reached the minimum value on the 38th day after planting (Figure 9d). However, in NAA:IBA (2:1) treatment, the contents of soluble protein first decreased to the minimum on the 8th day (2.171 mg·(g·FW)−1), then showed stabilization and modest increase during the initiation, expression, and extension phase. On the 28th day, contents of soluble protein in NAA:IBA (2:1) treatment reached the peak and then showed a downward trend. The increase in soluble protein contents observed in NAA:IBA (2:1) treatment during the extension phase might serve as a good marker for rooting ability in cuttings [32,33].
3.4. Endogenous Hormones Changes during the Adventitious Root Formation of M. wufengensis
The IAA contents in NAA:IBA (2:1)-treated cuttings were found 2.52 times higher on the 8th day than the initial day (Figure 10a), which synchronized well with the induction phase. In the initiation and expression phase, IAA contents continuously decreased and dropped to 6.849 μg·g−1 on the 18th day. In the extension phase, IAA contents increased and showed the peak on the 28th day, about 2.41 times higher than the initial value. In addition, the level of endogenous IAA in CK was lower than NAA:IBA (2:1) treatment during the entire rooting process. On the last day of measurements, the endogenous IAA contents in cuttings treated with NAA:IBA (2:1) was two times higher than CK.
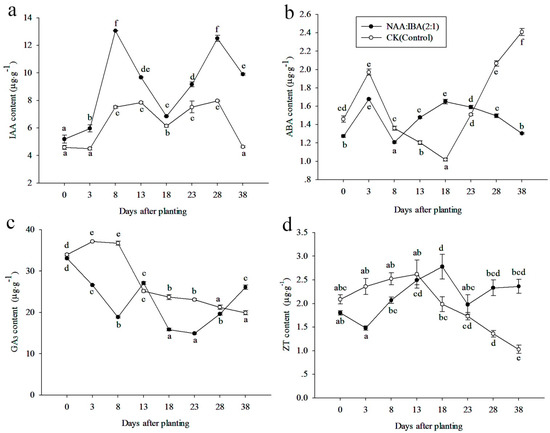
Figure 10.
Changes in IAA, ABA, GA3, and ZT quantifications during rooting in cuttings of M. wufengensis. (a) IAA contents. (b) ABA contents. (c) GA3 contents. (d) ZT contents. Data are the means of three replicates ± SE, for 20 cuttings per treatment. Letters on the top of each point show p < 0.05, same letters show the non-significant difference while different letters show significant difference between different days after planting.
ABA contents remained in a low range of 1.20–1.68μg·g−1 during the rooting process in NAA:IBA (2:1)-treated cuttings (Figure 10b). It followed a double-A-shaped trend, peaked at the 3rd and 18th days, valued 1.6781 μg·g−1 and 1.6514 μg·g−1, respectively. In CK, contents of ABA first increased to 1.9707 μg·g−1 on the 3rd day; in the successive 15 days, it decreased to the lowest value, 1.0186 μg·g−1. During the last 20 days, it increased again, reaching the highest value on the 38th day, 2.4082 μg·g−1, which was about 1.85 times higher than that of NAA:IBA (2:1) treatment.
The exogenous application of NAA and IBA significantly affected the contents of GA3 (Figure 10c). Endogenous GA3 contents in NAA:IBA (2:1) treatment showed a W-shaped trend, it rapidly decreased in the induction phase and then increased to a peak value on the 13th day, 27.0932 μg·g−1. During the successive 10 days, GA3 contents dropped significantly and reached the minimum value on the 23rd day (14.9176 μg·g−1), which was 45.12% of the initial day. Endogenous GA3 contents in CK were higher than NAA:IBA (2:1) treatment in the early 8 days of rooting. For example, on the 8th day after planting, endogenous GA3 content increased 1.95 times higher than in NAA:IBA (2:1) treatment. After 8 days, endogenous GA3 contents in CK kept falling to the end of experiment.
Endogenous ZT contents showed a slightly decrease in the first 3 days (Figure 10d), and then increased to the peak value on the 18th day (2.7763 μg·g−1), which was 1.54 times higher than the initial day. In the successive 10 days, ZT contents dropped rapidly to 1.9791 μg·g−1 and then increased to 2.3630 μg·g−1 on the 38th day. In CK, ZT contents showed an increase-decrease trend, the peak value of CK was found on the 13th day, 2.6216 μg·g−1. During the successive 25 days, ZT contents dropped significantly and reached the minimum value on the 38th day (1.0296 μg·g−1), which was 49.33% of the day initial.
4. Discussion
4.1. Auxin Combinations Positively Affect Rooting Performance of M. wufengensis
Adventitious root (AR) formation is a complex process influenced by a large set of external and internal factors [34,35,36]. It is generally accepted that the application of exogenous hormones has a certain role in adventitious root initiation. Auxins are a response to the induction of the meristems of root primordia, redistribution of nutrients, and biosynthesis of endogenous hormones in the cuttings, thereby stimulating adventitious root formation [15,37]. Synthetic auxins such as IBA and NAA have been used to promote rooting in plant cuttings [38,39,40]. Unfortunately, for some hard-to-root plants, application of single auxins usually failed to stimulate meristematic activity and root initiation, or had only a slight rooting effect [39,41]. In the case of M. wufengensis stem cuttings, a study on the role of auxins showed NAA and IBA exhibited better rooting results compared with IAA and GGR6 (a synthetic plant growth regulator, active ingredient: amino acids ≥ 20%, trace elements ≥ 2%; Beijing Erbitux Biological Technology Co., Ltd., Peking, China), as in the different sensitivity of M. wufengensis rhizomes to these two plant growth regulators, NAA was more stable and effective than IBA [13]. In the present study, combined applications of NAA and IBA were found to be more effective than using NAA separately to increase the rooting ability of M. wufengensis, and when treated with the median level (1000 mg/L NAA:IBA (2:1) solutions), we achieved the best rooting performance (Table 6), which had significant differences to single auxin application and CK. The rooting percentage was about 16.16 times higher than CK and 1.39 times higher than NAA:IBA (1:0) treatment. Complementary application of NAA and IBA is found to be synergetic in terms of cells division and root formation [15,42]. These findings are also supported by B. Kaviani, who studied the effects of combinations of NAA and IBA on softwood cuttings of Buxus hyrcana and found that 1000–2000 mg/L NAA and IBA treatments provided greater consistencies in rooting and shooting [17]. Shahram [43] investigated the effect of auxin combinations on stem cuttings of Mugo pine; when compared with CK, the rooting percentage of cuttings under NAA and IBA application increased from 14% to 55%, which confirms the positive effect of auxin combination.
In addition, the promoting effect of auxin combination on stem cuttings was closely related with the proportions of auxins. With the continuous increase of IBA proportion, the higher concentration of IBA may have a negative impact on stem cuttings and even suppress the activity of NAA [17,44,45]. In this study, when cuttings were immersed in NAA:IBA (1:1), NAA:IBA (1:2), or NAA:IBA (0:1) (the proportion of IBA was 50%, 66.7%, or 100%, respectively), the rooting percentage was reduced by 45.3%, 35.9%, or 49.9%, compared to NAA:IBA (2:1) (the proportion of IBA was 33.3%). Similar results were found in the findings of Shahram [43] who studied different concentrations of IBA and NAA on stem cutting of Mugo pine; the results indicated NAA:IBA (4:1)(the proportion of IBA was 20%) treatment exhibited the highest rooting percentage and dry weight. When cuttings were immersed in NAA:IBA (1:2) and NAA:IBA (1:4) (the proportion of IBA was 66.7% and 80%), the rooting percentage of these two treatments were decreased [43]. Taken together, combined application of exogenous hormones is very important for the formation and improvement of root system in M. wufengensis.
4.2. Anatomical Evaluation and Root Primordia Development during Adventitious Root Formation
The origin of adventitious roots in stem cuttings has been reported in various tissues and varies among species to species [11,46,47]. There are two cell sources from which adventitious root differentiate: (a) various existing cells, for example the fascicular cambium; (b) callus tissue formed at the base of cuttings in response to wounding occurring during cutting collection [48]. In this study, we firstly reported the formation of adventitious roots in M. wufengensis. As shown in Figure 4 and Figure 6, little callus tissue was formed on the epidermis of M. wufengensis cuttings, as a natural reaction of stems to wounding caused by cutting both in woody [49,50,51] and herbaceous plants [52]. At first glance, the roots of M. wufengensis seemed to originate from the callus developed on the epidermis. However, the cells of callus generated from M. wufengensis cuttings did not acquire the rooting competence (Figure 6c and Figure 7b), as they expressed different genetic routes with root-forming meristemoids which can initiate the establishment of a root development program [53]. Furthermore, it was observed from the sections that the adventitious roots of M. wufengensis arose directly from the stem vascular cambium and xylem parenchyma (Figure 6d,e), which have been reported to have an enormous potential for the formation of root primordia [54]. The histological structure was not sequentially performed, the function of callus on epidermis was only to heal the wound and emergent root, and it had no physical association with the adventitious root primordia in this species (Figure 8d). Similar results were obtained in other difficult-to-root species such as Diospyros virginiana and chestnut [11,53,55]. Some studies indicated that the excessive growth of callus may inhibit the production of adventitious roots in cuttings, as a high level of cytokinin and a low level of auxin in callus would lead to a continuous formation of callus and to a large consumption of nutrients and energy, thus inhibiting the formation of adventitious roots [51,56,57]. Fortunately, in NAA:IBA (2:1) treatment, callus on the epidermis did not present the phenomenon of overgrowth, and the small amount of callus could also protect against losses of water and nutrients of cuttings.
4.3. Changes of Metabolic Process in Response to Application of Auxin Combination during Adventitious Root Formation
The initiation, induction, and growth of adventitious root is an intensive metabolic process. Promoted by auxins and other plant growth regulators, these processes will lead to the increase of enzyme activities and the synthesis of RNA and proteins [22,54,58,59].
Plant peroxidases (POD) influence rooting by stimulating the production of iso-2-tyrosin in hydroxyproline-rich glycoproteins (HRGP, extensin) and catalyzing the biosynthesis of lignin and phellem layer of specific cells which will then be differentiated as root primordial cells [24,60,61]. In cuttings, total POD activity has been considered as a biochemical marker of AR development [27,62,63]. In this study, we observed two peak values of the POD activity synchronized well with the critical rooting points in NAA:IBA (2:1) treatment: one at the beginning of initiation phase, while the other was at the beginning of extension phase (Figure 9a). The increase in POD activity at these two stages indicated that the pre-treatment with combination of NAA and IBA facilitated a lot in scavenging for H2O2 molecules. It can also increase cell wall strength, and subsequently increase resistance to stress. Similar results were reported in Persian walnut and Malus hupehensis [24,64].
Polyphenol oxidases (PPO), which contained four atoms of copper per molecule and bound sites for two aromatic compounds and oxygen, are a group of copper enzymes localized in plastid tylacoids; they were able to catalyze the oxidation of aromatic compounds by oxygen [26]. In this study, the trends that PPO activities exhibited were totally different in NAA:IBA (2:1) and CK. When cuttings were immersed in NAA:IBA (2:1) solutions, PPO activity increased continuously during induction, initiation, and expression phase but declined slowly after extension phase (Figure 9b). As a previous study reported, PPO played an important role during root differentiation and root primordia formation and development. This might be due to its ability to participate in regulating synthesis of phenolic precursors during lignin biosynthesis [64,65,66,67,68]. The continuous increase of PPO activities in NAA:IBA (2:1) treatment might have promoted adventitious root formation in the entire process of induction to development. The same results were obtained by Zhang [24].
IAA oxidase (IAAO) affects rooting due to the IAA oxidation and regulates levels of IAA [26,69,70]. In this study, the activity trend of IAAO in NAA:IBA (2:1) treatment showed less remarkable changes than that of CK. At 0–8 days after planting, IAAO activities of NAA:IBA (2:1) treatment was higher than CK (Figure 9c). As previous study, an increase in the IAAO activity and the high contents of endogenous IAA and their derivatives could stimulate the root primordia induction and development on the auxin-supplemented medium [26], this was in accord with our results. High activity of IAAO enzyme will inhibit rooting, as was found in previous studies; high IAAO activity reduced contents of endogenous IAA, which inhibited root development [32,70,71]. In NAA:IBA (2:1) treatment, the low amount of IAAO activity 8–13 days after planting may be conducive to the initiation of root primordia. In expression and extension phase (13–28 dap), although IAAO activities of NAA:IBA (2:1) treatment continued to rise, the inhibitory effect on rooting was not exerted as there was no significant difference with the value on the 8th day. In Control, the activities of IAAO showed a rapid increase in the initiation, expression, and extension phases, which might induce low rooting percentage. A similar relationship was found during rooting of poplar, Populus tomentosa, and smoke trees [26,71,72].
Soluble protein content is relevant for both structural and regulatory/metabolic processes [27]. Studies have shown that nucleic acid and protein synthesis are necessary for adventitious root formation [64]. Presence of the auxin increases protein content in the base of a stem [34]. In this study, the soluble protein contents showed stabilization and modest increase during 8–28 days in NAA:IBA (2:1) treatment. Nutrient substance was adequate throughout the initiation, expression, and extension phases. It suggested that the pre-treatment with auxin combination was beneficial to the sufficient nutrient supply during the rooting process (Figure 9d). While in control, the soluble protein exhibited a continuously decrease trend; it was speculated that cuttings of control might suffer a defect of nutrient substance, and this finally led to the decline of rooting percentage (Figure 9d). Taking into account the obtained results, we can speculate that the positive effect of auxin combination on stem cuttings might be linked to the progressive transition from heterotrophic metabolism to the autotrophic metabolism and the attaining of functionality of the water and nutrient balance system. These factors jointly promoted the development of the roots [11].
4.4. Changes of Endogenous Hormones in Response to Application of Auxin Combination during Adventitious Root Formation
Plant hormones play an important role in the control of AR formation as they respond to the changing environment, provide a signaling network within the plant, and are decisive for cell fate determination and specification [35]. 3-indoleacetic acid (IAA) is common in plants as a main hormone of the auxin group. In this study, endogenous IAA showed a modest increase in NAA:IBA (2:1) treatment in the first 3 days (Figure 10a). It was speculated that the isolation of stem cuttings may induce the polar auxin transport, the basipetally transported auxin from shoot tips and young leaves may accumulate at the cutting base, and then create a local concentration gradient that drives the induction of adventitious root formation [27,34,56]. The early peak of endogenous IAA appeared at 8 days after planting, which synchronized well with the induction phase. It was reported that IAA could affect the root formation by acting directly in the cambium cells that initiate root primordia or indirectly through its engagement in the overall metabolism [73,74]. Our results confirmed the promotion role of IAA in the induction phase (0–8 days), the results were in accord with the findings obtained by Ahkami [74]. In the initiation and expression phases (8–18 days), cuttings showed a decrease of IAA (Figure 10a). This might be reduced by catabolism and the physiologically active conjugate of jasmonic acid after the induction phase [35,75,76].
Abscisic acid (ABA), which is a stress-related hormone, has been shown to inhibit adventitious root emergence and lateral root development in deepwater rice, Arachis hypogaea, and tomato, possibly by interfering with ethylene (ET) and gibberellic acids (GAs) signaling pathways and blocking cell cycle progression [23,34,77,78]. In this study, ABA contents of cuttings immersed in NAA:IBA (2:1) treatment increased slightly in the first 3 days after planting (Figure 10b). This was speculated to be related to the isolation of the cuttings. It was reported that isolation of the cuttings from donor trees broke the soil–plant–atmosphere continuum and led to numerous stresses caused by the interruption of water and nutrient uptake, and then altered transport of endogenous phytohormones and the activation of wound responses [47,79]. At 3–8 days, ABA contents showed a decreased trend both in NAA:IBA (2:1) and CK, while after 8 days, ABA contents of cuttings immersed in NAA:IBA (2:1) solutions increased slightly (Figure 10b). This coincided with the stage when root primordia are determined but their vascular connection with the stem has not been established yet. The same results were observed in the stem cutting of carnation [47]. In these conditions, the incipient root primordia might suffer some stress such as water shortage, and that leads to a transitory increase in their ABA contents [47]. When the root development was gradually completed, the contents of ABA decreased.
Gibberellins (GAs) are a complex family of tetracyclic diterpenoid plant growth regulators, some of which, such as GA1, GA3, GA4, and GA7, are thought to function as bioactive hormones [25]. These bioactive hormones control diverse aspects of plant growth and development, including seed germination, flower initiation, fruit development, stem elongation, leaf expansion, and trichome differentiation [80,81,82,83]. In cutting propagation, GA3 is generally considered an inhibitor of adventitious root formation [34]. In poplar plants, in which defects in GA production or perception cause the growth of lateral root to be promoted, higher root mass and highly branched roots are produced [82], while according to some findings, GAs are assumed to have an adventitious root phase-dependent effect [34]. In our study, the results indicated that GA3 contents in cuttings immersed in NAA:IBA (2:1) solutions exhibited an increase trend in the initiation and extension phases (Figure 10c), which confirmed the phase-dependent effect of GAs. Similar results were observed in deep water rice, Malus hupehensis, and chestnut [23,24,55]. The effect of GAs being inhibitory to root induction and stimulatory to root formation might contribute to their synthesis and transportation characteristics. Previous studies indicated that GAs synthesized in the meristem and cortical and epidermal cell layers [25,84,85], then specifically accumulated in the endodermis of the root elongation zone [86].
Cytokinins play key roles in regulation of root development [87]. The major cytokinins in leaves, stems, and roots of plants have been rigorously identified by several physicochemical methods [88]. Zeatin (ZT) and zeatin riboside have been identified as two major cytokinins in root exudate [89]. Zeatin is considered more active than its riboside [47]. In this study, contents of ZT showed a W-shaped trend with the presence of NAA and IBA. In the first 3 days, the contents of ZT showed a slight decrease, this was speculated to be related to the isolation from the stock plants (Figure 10d). As the previous study reported, the root primordia cells required a dedifferentiation process before adventitious root induction [35]. Early wounds in cuttings induced accumulation of ethylene and jasmonic acid, these two hormones together with cytokinins which exhibited a transient decrease in levels contributed to the dedifferentiation process by enhancing auxin responsiveness [35]. During days 3–8, ZT contents of cuttings immersed in NAA:IBA (2:1) increased, while there were no significant differences with the initial value (Figure 10d). According to the previous reports, cytokinins regulated adventitious root formation in a stage-specific manner, the low level of cytokinins combined with high auxin levels was thought to be involved in the contribution of adventitious root induction [34,47,90,91,92,93]. In the present study, cuttings immersed in NAA:IBA (2:1) solutions revealed a high level of IAA and low level of ZT at induction phase (0–8 days), which confirmed the theory that a rise in both auxin and cytokinin depletion controls induction of adventitious roots in cuttings [47,91]. At 8–18 days, contents of ZT continuously increased in NAA:IBA (2:1) treatment (Figure 10d), the increasing cytokinin could regulate the longitudinal zonation and radial patterning of root vasculature by controlling cell differentiation, promoting protophloem cell identity, and spatially inhibiting protoxylem formation in root meristem [93]. Owing to the increasing ZT contents, massive root primordia emerge, the vascular reconnection between newly formed roots and shoot was fully established, allowing root nutrition, hydration, and growth [47,94]. At the same time, the requirement for ZT further expanded with the cell differentiation and proliferation in the phloem, xylem, and cambium zones, which could explain the increase of ZT contents in the extension phase of NAA:IBA (2:1) treatment.
5. Conclusions
The effect of auxin combination on stem cuttings of M. wufengensis was studied for the first time to provide an optimized protocol that can be used to develop a healthier and more dense root system for its propagation. The stem cuttings pretreated with NAA:IBA (2:1) solution significantly exhibited the overall rooting performance. Furthermore, this study scientifically revealed the origin and spatially explicit timeline of adventitious root formation and explored the rhythm correlation between the rooting process and the physiological and biochemical changes in stem cuttings of M. wufengensis.
Author Contributions
Conceptualization, Y.W. and M.A.K.; data curation, Y.W. and T.H.; formal analysis, Y.W.; funding acquisition, Z.J. and L.M.; investigation, Y.W., M.A.K., Z.Z. and T.H.; methodology, Y.W. and M.A.K.; project administration, L.M.; resources, Z.Z. and Z.S.; software, Y.W.; supervision, L.M.; visualization, Y.W.; writing—original draft, Y.W.; writing—review and editing, Y.W. and Z.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was financially supported by the Special Fund for Forest Fund for Scientific Research in the Public Welfare under grant no. 201504704.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Acknowledgments
We are thankful to Wufeng Bo Ling Magnolia wufengensis Technology Development Co., Ltd. for providing Magnolia wufengensis seedlings. We are thankful to Special Fund for Forest Scientific Research in the Public Welfare under grant no. 201504704 for providing funds.
Conflicts of Interest
The authors declare that they have no conflict of interest.
Appendix A

Figure A1.
At 13 days after planting, cuttings in Control formed some white callus tissues on the epidermis.
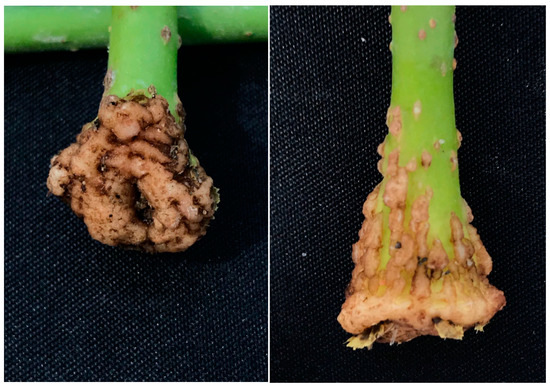
Figure A2.
At 28 days after planting, cuttings in Control showing the overgrowth of callus tissue, which finally will inhibit rooting.

Figure A3.
Cuttings in Control gradually rotted and almost completely rotted at 58 days after planting.
References
- Ma, L.Y.; Wang, L.R.; He, S.C.; Liu, X.; Wang, X.Q. A new species of Magnolia (Magnoliaceae) from Hubei, China. Bull. Bot. Res. 2006, 26, 4–7. [Google Scholar]
- Ma, L.Y.; Wang, L.R.; He, S.C.; Liu, X.; Wang, X.Q. A new variety of Magnolia (Magnoliaceae) from Hubei, China. Bull. Bot. Res. 2006, 26, 517–519. [Google Scholar]
- Sang, Z.Y.; Ma, L.Y.; Chen, F.J.; Zhang, P.; Zhu, Y.C. Protection status and utilization countermeasure of germplasm resources of Magnolia wufengensis in Wufeng County. Hubei Agric. Sci. 2011, 50, 1564–1567. [Google Scholar] [CrossRef]
- Yang, Y.; Jia, Z.K.; Chen, F.J.; Sang, Z.Y.; Ma, L.Y. Natural cold acclimatisation and de-acclimatisation of Magnolia wufengensis in response to alternative methods of application of abscisic acid. J. Hortic. Sci. Biotech. 2016, 90, 704–710. [Google Scholar] [CrossRef]
- Shi, X.D.; Yin, Q.; Sang, Z.Y.; Zhu, Z.L.; Jia, Z.K.; Ma, L.Y. Prediction of potentially suitable areas for the introduction of Magnolia wufengensis under climate change. Ecol. Indic. 2021, 127, 107762. [Google Scholar] [CrossRef]
- Hao, Y.; Peng, Z.D.; Liang, D.W. Sowing, seedling raising and tending management technology of Magnolia wufengensis. For. Sci. Technol. 2010, 3, 47–48. [Google Scholar] [CrossRef]
- Justamante, M.S.; Ibanez, S.; Villanova, J.; Perez-Perez, J.M. Vegetative propagation of argan tree (Argania spinosa (L.) Skeels) using in vitro germinated seeds and stem cuttings. Sci. Hortic. 2017, 225, 81–87. [Google Scholar] [CrossRef]
- Kimber, W.; Harsharn, G.; Jamie, S. Willow bark extract and the biostimulant complex Root Nectar® increase propagation efficiency in chrysanthemum and lavender cuttings. Sci. Hortic. 2020, 263, 109108. [Google Scholar] [CrossRef]
- Bhardwaj, D.R.; Mishra, V.K. Vegetative propagation of Ulmus villosa: Effects of plant growth regulations, collection time, type of donor and position of shoot on adventitious root formation in stem cuttings. New For. 2005, 29, 105–116. [Google Scholar] [CrossRef]
- Khan, M.A.; Wang, Y.; Uddin, S.; Muhammad, B.; Badshan, M.T.; Khan, D. Propagation of Magnolia Biondii Pamp through stem cuttings using exogenous hormones. Appl. Ecol. Environ. Res. 2020, 182, 2213–2229. [Google Scholar] [CrossRef]
- Anat, I.; Yiftah, Y.; Tsuf, B.; Israel, D.; Arie, R.; Joseph, R. Rooting of cuttings of selected Diosphyros virginiana clonal rootstocks and bud growth in rooted cuttings. Sci. Hortic. 2018, 232, 13–21. [Google Scholar] [CrossRef]
- Preece, J.E. A century of progress with vegetative plant propagation. Hortic. Sci. 2003, 38, 1015–1025. [Google Scholar] [CrossRef]
- Wang, Y.; Jia, Z.K.; Ma, L.Y.; Deng, S.X.; Zhu, Z.L.; Sang, Z.Y. Effects of four plant growth regulators on rooting of softwood cutting of Magnolia wufengensis. Sci. Slivae Sin. 2019, 55, 35–45. [Google Scholar] [CrossRef]
- Liang, Y.T.; Deng, Y.X.; Xia, W.S. Study of indole-3-acetic acid and 1-naphthaleneacetic acid treatment on the rooting of softwood-cutting of Magnolia wufengensis L.Y.Ma et L.R.Wang. Anhui Agric. Sci. Bull. 2018, 24, 87–88. [Google Scholar]
- Zheng, L.; Xiao, Z.B.; Song, W.T. Effects of substrate and exogenous auxin on the adventitious rooting of Dianthus caryophyllus L. Hortic. Sci. 2020, 55, 170–173. [Google Scholar] [CrossRef]
- Blythe, E.K.; Sibley, J.L.; Ruter, J.M.; Tilt, K.M. Cutting propagation of foliage crops using a foliar application of auxin. Sci. Hortic. 2004, 103, 31–37. [Google Scholar] [CrossRef]
- Kaviani, B.; Negahdar, N. Propagation, micropropagation and cryopreservation of Buxus hyrcana Pojark., an endangered ornamental shrub. S. Afr. J. Bot. 2017, 111, 326–335. [Google Scholar] [CrossRef]
- Su, J.S.; Chung, H.K.; Un, S.S.; Hye, J.O. Successful stem cutting propagation of Patrinia rupestris for horticulture. Rhizosphere 2019, 9, 90–92. [Google Scholar] [CrossRef]
- Zhou, L.L.; Lin, S.B.; Huang, P.; Lin, S.J. Effects of leaf age and exogenous hormones on callus initiation, rooting formation, bud germination and plantlet formation in Chinese Fir leaf cuttings. Forests 2018, 9, 478. [Google Scholar] [CrossRef]
- Bellini, C.; Pacurar, D.I.; Perrone, I. Adventitious roots and lateral roots: Similarities and differences. Annu. Rev. Plant Biol. 2014, 65, 639–666. [Google Scholar] [CrossRef]
- Kevers, C.; Hausman, J.F.; Faivre Rampant, O.; Evers, D.; Gaspar, T. Hormonal control of adventitious rooting: Progress and questions. Angew. Bot. 1997, 71, 71–79. [Google Scholar]
- De Klerk, G.J.; van der Krieken, W.; DeJong, J.C. The formation of adventitious roots: New concepts, new possibilities. In Vitro Cell. Dev. Biol. Plant 1999, 35, 189–199. [Google Scholar] [CrossRef]
- Steffens, B.; Wang, J.; Sauter, M. Interactions between ethylene, gibberellin and abscisic acid regulate emergence and growth rate of adventitious roots in deepwater rice. Planta 2006, 223, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.X.; Fan, J.J.; Tian, Q.Q.; Zhao, M.M.; Zhou, T.; Cao, F.L. The effects of exogenous hormones on rooting process and the activities of key enzymes of Malus hupehensis stem cuttings. PLoS ONE 2017, 12, e0172320. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Elena, P.; Laura, P.; Sabrina, S. Plant hormone cross-talk: The pivot of root growth. J. Exp. B 2015, 66, 1113–1121. [Google Scholar] [CrossRef]
- Ilczuk, A.; Jacygrad, E. The effect of IBA on anatomical changes and antioxidant enzyme activity during the in vitro rooting of smoke tree (Cotinus coggygria Scop.). Sci. Hortic. 2016, 210, 268–276. [Google Scholar] [CrossRef]
- Aumond, M.L., Jr.; de Araujo, A.T., Jr.; Junkes, C.F.d.; de Almeida, M.R.; Matsuura, H.N.; de Costa, F.; Fett-Neto, A.G. Events associated with early age-related decline in adventitious rooting competence of Eucalyptus globulus Labill. Front. Plant Sci. 2017, 8, 1734. [Google Scholar] [CrossRef]
- Zhang, Z.L.; Qu, W.J. Experimental Course of Plant Physiology; China Forestry Press: Beijing, China, 2003; pp. 88–104. [Google Scholar]
- Lu, W.J.; Li, Y.S. Experimental Course of Plant Physiology; China Forestry Press: Beijing, China, 2012; pp. 43–45. [Google Scholar]
- Xiao, A.H.; Chen, F.J.; Jia, Z.K.; Sang, Z.; Zhu, Z.L.; Ma, L.Y. Determination of 4 plant hormones in Magnolia wufengensis by gradient elution high performance liquid chromatography. Chin. J. Anal. Lab. 2020, 39, 249–254. [Google Scholar] [CrossRef]
- Anderson, V.L.; McLean, R.A. Design of Experiments; Marcel Dekker Inc.: New York, NY, USA, 1974. [Google Scholar]
- Moncousin, C.H.; Gaspar, T.H. Peroxidase as a marker for rooting improvement of cynara scolymus L. cultured in vitro. Biochem. Physiol. Pflanz. 1983, 178, 263–271. [Google Scholar] [CrossRef]
- Nordstrom, A.C.; Eliasson, J. Levels of endogenous indole-3-acetic acid and indole-3-acetylaspartic acid during adventitious root formation in pea cuttings. Physiol. Plant. 1991, 82, 599–605. [Google Scholar] [CrossRef]
- Da Costa, C.T.; De Almeida, M.R.; Ruedell, C.M.; Schwambach, J.; Maraschin, F.D.S.; Fett-Neto, A.G. When stress and development go hand in hand: Main hormonal controls of adventitious rooting in cuttings. Front. Plant. Sci. 2013, 4, 133. [Google Scholar] [CrossRef]
- Druege, U.; Hilo, A.; Perez-Perez, J.M.; Klopotek, Y.; Acosta, M.; Shahinnia, F. Molecular and physiological control of adventitious rooting in cuttings: Phytohormone action meets resource allocation. Ann. Bot. 2019, 123, 929–949. [Google Scholar] [CrossRef] [PubMed]
- Sergio, I.; Helena, R.C.; María, A.F.; Ana, B.S.G.; Joan, V.; José, L.M.; José, M.P.P. A network-guided genetic approach to identify novel regulators of adventitious root formation in Arabidopsis thaliana. Front. Plant Sci. 2019, 10, 461. [Google Scholar] [CrossRef]
- Garrido, G.; Guerrero, J.R.; Cano, E.A.; Acosta, M.; Sanchez-Bravo, J. Origin and basipetal transport of the IAA responsible for rooting of carnation cuttings. Physiol. Plant 2002, 114, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, H.T.; Kester, D.E.; Davies, F.T.; Geneve, R.L. Plant Propagation Principles and Practices, 5th ed.; Prentice Hall: Hoboken, NJ, USA, 2001. [Google Scholar]
- Denaxa, N.K.; Vemmos, S.N.; Roussos, P.A.; Kostelenos, G. The effect of IBA, NAA and carbohydrates on rooting capacity of leafy cuttings in three olive cultivars (Olea europaea, L.). Acta Hortic. 2010, 924, 101–109. [Google Scholar] [CrossRef]
- Ioannis, D.; Katerina, B.; Despoina, B.; Maritina, S. The effect that indole-3-butyric acid (IBA) and position of cane segment have on the rooting of cuttings from grapevine rootstocks and from Cabernet franc (Vitis vinifera, L.) under conditions of a hydroponic culture system. Sci. Hortic. 2018, 227, 79–84. [Google Scholar] [CrossRef]
- Wiesman, Z.; Lavee, S. Enhancement of stimulatory effects on rooting of olive cultivar stem cuttings. Sci. Hortic. 1995, 62, 189–198. [Google Scholar] [CrossRef]
- Ragonezi, C.; Klimaszewska, K.; Castro, M.R.; Lim, M.; Oliveira, P.; Zavattieri, M.A. Adventitious rooting of conifers: Influence of physical and chemical factors. Trees 2010, 24, 975–992. [Google Scholar] [CrossRef]
- Sedaghathoor, S.; Kayghobadi, S.; Tajvar, Y. Rooting of Mugo pine (Pinus mugo) cuttings as affected by IBA, NAA and planting substrate. For. Syst. 2016, 25, eSC08. [Google Scholar] [CrossRef]
- Henselova, M. Synergistic effect of benzolinone with IBA and fungicides on the vegetative propagation of ornamental plants, park and fruit woody species. Hortic. Sci. 2002, 29, 41–50. [Google Scholar] [CrossRef]
- Kesari, V.; Krishnamachari, A.; Rangan, L. Effect of auxins on adventitious rooting from stem cuttings of candidate plus tree Pongamia pinnata (L.), a potential biodiesel plant. Trees 2009, 23, 597–604. [Google Scholar] [CrossRef]
- Naija, S.; Elloumi, N.; Jbir, N.; Ammar, S.; Kevers, C. Anatomical and biochemical changes during adventitious rooting of apple rootstocks MM 106 cultured in vitro. Comptes Rendus Biol. 2008, 331, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Agulló-Antón, M.Á.; Ferrández-Ayela, A.; Fernández-Garcia, N.; Nicolás, C.; Albacete, A.; Pérez-Alfocea, F. Early steps of adventitious rooting: Morphology, hormonal profiling and carbohydrate turnover in carnation stem cuttings. Physiol. Plant 2014, 150, 446–462. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, H.T.; Kester, D.E.; Davies, F.T.; Geneve, R.L. Hartmann and Kester’s Plant Propagation: Principles and Practices, 8th ed.; Prentice Hall: Englewood Cliffs, NJ, USA, 2014. [Google Scholar]
- Strzelecka, K. Anatomical structure and adventitious root formation in Rhododendron ponticum, L. cuttings. Acta Sci. Pol. Hortorum Cultus 2007, 6, 15–22. [Google Scholar] [CrossRef]
- Trofmuk, L.P.; Kirillov, P.S.; Egorov, A.A. Application of biostimulants for vegetative propagation of endangered Abies gracilis. J. For. Res. 2020, 31, 1195–1199. [Google Scholar] [CrossRef]
- Chen, W.Z.; He, L.B.; Tian, S.Y.; Joseph, M.; Xiong, H.; Zou, F.; Yuan, D.Y. Factors involved in the success of Castanea henryi stem cuttings in different cutting mediums and cutting selection periods. J. For. Res. 2021, 32, 627–1639. [Google Scholar] [CrossRef]
- Wróblewska, K. The infuence of benzyladenine and naphthalene-1-acetic acid on rooting and growth of Fuchsia hybrida cuttings. Acta Sci. Pol. Hortorum Cultus 2013, 12, 101–113. [Google Scholar] [CrossRef]
- Tetsumura, T.; Ishimura, S.; Honsho, C.; Chijiwa, H. Improvement in rooting of cuttings of FDR-1, a dwarfing rootstock for kaki©. Acta Hortic. 2016, 1140, 113–116. [Google Scholar] [CrossRef]
- Legué, V.; Rigal, A.; Bhalerao, R.P. Adventitious root formation in tree species: Involvement of transcription factors. Physiol. Plant 2014, 151, 192–198. [Google Scholar] [CrossRef]
- Jesús, M.V.; Nieves, V.; Jose, M.C.S.; Saleta, R.; Conchi, S. Recent advances in adventitious root formation in Chestnut. Plants 2020, 9, 1543. [Google Scholar] [CrossRef]
- Druege, U.; Franken, P.; Hajirezaei, M.R. Plant hormone homeostasis, signaling, and function during adventitious root formation in cuttings. Front. Plant Sci. 2016, 7, 381. [Google Scholar] [CrossRef] [PubMed]
- Du, C.J.; Sun, J.C.; Chen, W.; Ji, J.; Jang, Z.P.; Shi, S.Q. Comparison of physiological and anatomical characteristics between seedlings and graftings derived from old Platycladus orientalis. Sci. Silvae Sin. 2019, 55, 41–49. [Google Scholar] [CrossRef]
- Hartmann, H.T.; Kester, D.E. Plant Propagation Principles and Practices, 4th ed.; Upper Saddle River: Prentice Hall, NJ, USA, 1983. [Google Scholar]
- Sergio, T.; Alberto, P.; Stefano, P.; Daniela, F. Influence of light and shoot development stage on leaf photosynthesis and carbohydrate status during the adventitious root formation in cuttings of Corylus avellana L. Front. Plant Sci. 2015, 6, 973. [Google Scholar] [CrossRef]
- Sato, Y.; Sugiyama, M.; Gorecki, R.J.; Fukuda, H.; Komamine, A. Interrelationship between lignin deposition and the activities of peroxidase isoenzymes in differentiating tracheary elements of Zinnia. Planta 1993, 189, 584–589. [Google Scholar] [CrossRef]
- Aeschbacher, R.A.; Schiefelbein, J.W.; Benfey, P.N. The genetic and molecular basis of root development. Ann. Rev. Plant Biol. 1994, 45, 25–45. [Google Scholar] [CrossRef]
- Fett-Neto, A.G.; Teixeira, S.L.; Da Silva, E.A.M.; Sant’ Anna, R. Biochemical and morphological changes during in vitro rhizogenesis in cuttings of Sequoia sempervirens (D. Don) Endl. J. Plant Physiol. 1992, 140, 720–728. [Google Scholar] [CrossRef]
- Schwambach, J.; Ruedell, C.M.; de Almeida, M.R.; Penchel, R.M.; de Araújo, E.F.; Fett-Neto, A.G. Adventitious rooting of Eucalyptus globulus × maidennii mini-cuttings derived from mini-stumps grown in sand bed and intermittent flooding trays: A comparative study. New For. 2008, 36, 261–271. [Google Scholar] [CrossRef]
- Haissig, B.E. Metabolic processes in adventitious rooting of cuttings. In New Root Formation in Plants and Cuttings; Jackson, M.B., Ed.; Springer: Dordrecht, The Netherlands, 1986; pp. 141–189. [Google Scholar]
- González, A.; Tamés, R.S.; Rodríguez, R. Ethylene in relation to protein, peroxidase and polyphenol oxidase activities during rooting in hazelnut cotyledons. Physiol. Plant 1991, 83, 611–620. [Google Scholar] [CrossRef]
- Yilmaz, H.; Taşkin, T.; Otludil, B. Polyphenol oxidase activity during rooting in cuttings of grape (Vitis vinifera, L.) varieties. Turk. J. Bot. 2003, 27, 495. [Google Scholar] [CrossRef]
- Pizzeghello, D.; Francioso, O.; Ertani, A.; Muscolo, A.; Nardi, S. Isopentenyladenosine and cytokinin-like activity of different humic substances. J. Geochem. Explor. 2013, 129, 70–75. [Google Scholar] [CrossRef]
- Mohamed, S.E.; Hong, Z.; Yan, C.; Bing, L.; Xia, Y.P. The effect of humic acid on endogenous hormone levels and antioxidant enzyme activity during in vitro rooting of evergreen azalea. Sci. Hortic. 2018, 227, 234–243. [Google Scholar] [CrossRef]
- Fu, Z.; Xu, P.; He, S.; Teixeira da Silva, J.A.; Tanaka, M. Dynamic changes in enzyme activities and phenolic content during in vitro rooting of tree peony (Paeonia suffruticosa Andr.) plantlets. Maejo Int. J. Sci. Technol. 2011, 5, 252–265. [Google Scholar]
- Meng, X.Y.; Wang, Z.; He, S.L.; Shi, L.Y.; Song, Y.L. Endogenous hormone levels and avtivities of IAA-modifying enzymes during adventitious rooting of tree peony cuttings and grafted scions. Hortic. Environ. Biotechnol. 2019, 60, 187–197. [Google Scholar] [CrossRef]
- Song, J.Y.; He, W.L.; Li, S.B.; Liu, Y.J.; Sun, Y.J. Analysis of physiological and biochemical characteristics related to cutting and rooting of chimera in populous tomentosa carr. Sci. Silvae Sin. 2001, 37, 64–67. [Google Scholar] [CrossRef]
- Günes, T. Peroxidase and IAA-oxidase activities during rooting in cuttings of three poplar species. Turk. J. Bot. 2000, 24, 97–101. [Google Scholar] [CrossRef]
- Altman, A.; Wareing, P.F. Effect of IAA on sugar accumulation and basipetal transport of C14-labeled assimilates in relation to root formation in Phaseolus vulgaris cuttings. Physiol. Plant 1975, 33, 32–38. [Google Scholar] [CrossRef]
- Ahkami, A.H.; Melzer, M.; Ghaffari, M.R.; Pollmann, S.; Majid, G.J.; Shahinnia, F.; Hajirezaei, M.R.; Druege, U. Distribution of indole-3-acetic acid in Petunia hybrida shoot tip cuttings and relationship between auxin transport, carbohydrate metabolism and adventitious root formation. Planta 2013, 238, 499–517. [Google Scholar] [CrossRef]
- Lakehal, A.; Bellini, C. Control of adventitious root formation: Insights into synergistic and antagonistic hormonal interactions. Physiol. Plant 2019, 165, 90–100. [Google Scholar] [CrossRef]
- Druege, U. Overcoming physiological bottlenecks of leaf vitality and root development in cuttings: A systemic perspective. Front. Plant Sci. 2020, 11, 907. [Google Scholar] [CrossRef]
- Guo, D.; Liang, J.; Qiao, Y.; Yan, Y.; Li, L.; Dai, Y. Involvement of G1-to-S transition and AhAUX-dependent auxin transport in abscisic acid-induced inhibition of lateral root primodia initiation in Arachis hypogaea L. J. Plant Physiol. 2012, 169, 1102–1111. [Google Scholar] [CrossRef]
- McAdam, S.A.M.; Brodribb, T.J.; Ross, J.J. Shoot-derived abscisic acid promotes root growth. Plant Cell Environ. 2016, 39, 652–659. [Google Scholar] [CrossRef] [PubMed]
- Leon, J.; Rojo, E.; S’anchez-Serrano, J.J. Wound signalling in plants. J. Exp. Bot. 2001, 52, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hooley, R. Gibberellins: Perception, transduction and responses. Plant Mol. Biol. 1994, 26, 1529–1555. [Google Scholar] [CrossRef]
- Davies, P.J. Plant Hormones: Physiology, Biochemistry, and Molecular Biology; Kluwer: London, UK, 1995; pp. 6–7. [Google Scholar]
- Busov, V.; Meilan, R.; Pearce, D.W.; Rood, S.B.; Ma, C.; Tschaplinski, T.J.; Strauss, S.H. Transgenic modification of gai or rgl1 causes dwarfing and alters gibberellins, root growth, and metabolite profiles in Populus. Planta 2006, 224, 288–299. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, S. Gibberellin metabolism and its regulation. Annu. Rev. Plant Biol. 2008, 59, 225–251. [Google Scholar] [CrossRef]
- Silverstone, A.L.; Chang, C.; Krol, E.; Sun, T.P. Developmental regulation of the gibberellin biosynthetic gene GA1 in Arabidopsis thaliana. Plant J. 1997, 12, 9–19. [Google Scholar] [CrossRef]
- Birnbaum, K.; Shasha, D.E.; Wang, J.Y.; Jung, J.W.; Lambert, G.M.; Galbraith, D.W.; Benfey, P.N. A gene expression map of the Arabidopsis root. Science 2003, 302, 1956–1960. [Google Scholar] [CrossRef]
- Shani, E.; Weinstain, R.; Zhang, Y.; Castillejo, C.; Kaiserli, E.; Chory, J.; Tsien, R.Y.; Estelle, M. Gibberellins accumulate in the elongating endodermal cells of Arabidopsis root. Proc. Natl. Acad. Sci. USA 2013, 110, 4834–4839. [Google Scholar] [CrossRef]
- Dello, I.R.; Nakamura, K.; Moubayidin, L.; Perilli, S.; Taniguchi, M.; Morita, M.T. A genetic framework for the control of cell division and differentiation in the root meristem. Science 2008, 322, 1380–1384. [Google Scholar] [CrossRef]
- Martin, V.P.; Wong, O.C. Identification of cytokinins from xylem exudate of Phaseolus vulgaris L. Plant Physiol. 1985, 79, 296–298. [Google Scholar] [CrossRef]
- Davey, J.E.; van Staden, J. Cytokinin translocation: Changes in zeatin and zeatin-riboside levels in the root exudate of tomato plants during their development. Planta 1976, 13, 69–72. [Google Scholar] [CrossRef]
- Blakesley, D. Auxin metabolism and adventitious root initiation. In Biology of Adventitious Root Formation; Davis, T.D., Haissig, B.E., Eds.; Plenum Press: New York, NY, USA, 1994; pp. 143–154. [Google Scholar]
- Rasmussen, A.; Hosseini, S.A.; Hajirezaei, M.R.; Druege, U.; Geelen, D. Adventitious rooting declines with the vegetative to reproductive switch and involves a changed auxin homeostasis. J. Exp. Bot. 2015, 66, 1437–1452. [Google Scholar] [CrossRef] [PubMed]
- Villacorta-Martín, C.; Sanchez-Garcia, A.B.; Villanova, J. Gene expression profiling during adventitious root formation in carnation stem cuttings. BMC Genom. 2015, 16, 789. [Google Scholar] [CrossRef] [PubMed]
- Marta, D.B.; Leonardo, G.; Sabrina, S. Spatiotemporal changes in the role of cytokinin during root development. New Phytol. 2013, 199, 324–338. [Google Scholar] [CrossRef]
- Hatzilazarou, S.P.; Syrosa, T.D.; Yupsanis, T.A.; Bosabalidis, A.M.; Economou, A.S. Peroxidases, lignin, and anatomy during in vitro and ex vitro rooting of gardenia (Gardenia jasminoides Ellis) microshoots. J. Plant. Physiol. 2006, 163, 827–836. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).