Abstract
The chemical modification of oligomers such as DNA, PNA, MORF, LNA to attach radionuclides for nuclear imaging and radiotherapy applications has become a field rich in innovation as older methods are improved and new methods are introduced. This review intends to provide a brief overview of several chelators currently in use for the labeling of oligomers with metallic radionuclides such as 99mTc, 111In and 188Re. While DNA and its analogs have been radiolabeled with important radionuclides of nonmetals such as 32P, 35S, 14C, 18F and 125I, the labeling methods for these isotopes involve covalent chemistry that is quite distinct from the coordinate-covalent chelation chemistry described herein. In this review, we provide a summary of the several chelators that have been covalently conjugated to oligomers for the purpose of radiolabeling with metallic radionuclides by chelation and including details on the conjugation, the choice of radionuclides and labeling methods.
1. Introduction
Advances in nuclear imaging require adequate methods for labeling biologicals such as DNA and its analogs (referred to herein as oligomers) with a variety of radionuclides, those that emit gamma rays for noninvasive imaging and those emit betas, alphas, Auger electrons, etc. for radiotherapy. Compared to other imaging modalities such as MRI, CT, ultrasound and optical, nuclear imaging offers extremely high sensitivity capable of detecting contrast agents at pM concentrations or lower [1]. Compared to external beam or brachytherapy, radiation therapy with internally administered radionuclides can offer superb dose distribution [2]. Oligomers are unique in their property of hybridization to their complement and this property has been exploited in the development of novel radiopharmaceuticals for nuclear imaging and radiotherapy by pretargeting, by antisense localization and by aptamer-mediated approaches [3,4,5]. While the use of unlabeled oligomers in medicine has been extensively reported over the past several decades, much less is presently in the literature regarding methods of labeling these oligomers with radionuclides, especially metallic radionuclides. Radionuclides such as 14C, 11C, 14N, 35S, 3H and 32P have been used to label biologicals including oligomers, often by isotope substitution (e.g., 11C for 12C, 3H for 1H). While labeling by isotope substitution essentially guarantees that the properties of the biological will not have been altered, the labeling process is usually nontrivial. With few exceptions, biologicals cannot be radiolabeled with metallic radionuclides in this manner but require the preliminary covalent attachment of a chelator, a chemical structure capable of binding a metal in a claw-like fashion with two or more bonds. Since chelators are usually large molecules, in contrast to nonmetals, labeling with metals is much more likely to alter biological properties. However, an important advantage of labeling by chelation is that the labeling itself can be extremely simple to the point where kit formulations are possible. This contribution is intended to provide a brief description of several chelators that have been successfully used to radiolabeled oligomers. The coverage is not intended to be comprehensive. Rather we hope merely to raise awareness of what has been done in the recent past and of some of the problems that have been successfully addressed.
2. Oligomers
The term oligomers as used herein refers to oligonucleotides such as DNAs and RNAs and includes analogs that are not polynucleotides (such as PNAs, MORFs and LNAs). To overcome the instability to nucleases of native DNA and RNAs with phosphodiester (PO) backbones, several families of synthetic analogs have become commercially available, including phosphorothioate (PS) DNAs [6], phosphorodiamidate morpholino oligomers (MORF) [7], peptide nucleic acids (PNA) [8] and locked nucleic acid (LNA) [9,10] as shown in Figure 1. The modified backbones in each of these synthetic DNA analogs have stabilized the oligomers against nuclease hydrolysis while still permitting similar, in some cases increased, affinities for their complement.
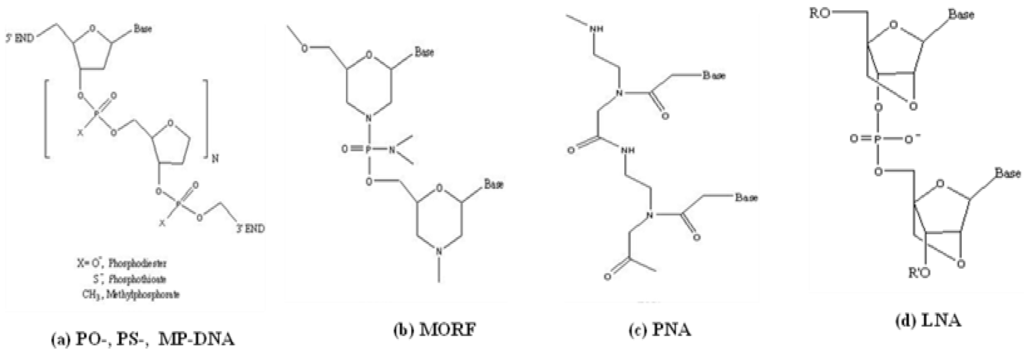
Figure 1.
Chemical structures of DNAs and several important analogs. (a) phosphodiester (PO), phosphorothioate (PS) and methylphosphonate (MP) DNAs; (b) phosphorodiamidate morpholino (MORF); (c) peptide nucleic acids (PNA); (d) locked nucleic acids (LNA).
3. Metallic Radionuclides
Theoretically, almost any radionuclide can be chemically attached to oligomers, but the choice will depend upon the application. Although justification of a particular choice is usually not now included in published reports, implicit are those factors extensively discussed decades ago [11,12,13], including (1) decay type, (2) physical half life, (3) availability and (4) ease of labeling. For example: radionuclides decaying by short-range high LET emissions such as betas, alphas and Auger electrons are highly cytotoxic and used to label biologicals designed for radiotherapeutic purposes, while diagnostic imaging agents require radionuclides that are relatively low in cytotoxicity but decay with imaginable emissions, either gammas for planar and Single Photon Computerized Emission Tomography (SPECT) imaging or annihilation photons for Positron Emission Tomography (PET). In general, radionuclides with short physical half lives are preferred for imaging to minimize the radiation exposure to subjects. However, too short a half life may not provide sufficient time for radiolabeling. Furthermore the half life must be a good match to the time between administration and imaging that is required to reach an adequate target/nontarget ratio that can vary over a wide range depending upon the application. For radiotherapy, radionuclides with longer half lives are often preferred if that results in delivering a higher radiation dose to the target. Nevertheless, an overriding prerequisite is always availability. Thus, in addition to its superior decay property, 99mTc is often a favorite choice for planar and SPECT imaging since it may be made available on demand from a 99Mo/99mTc radionuclide generator.

Table 1.
Properties of selected metallic radionuclides useful in nuclear medicine.
| Radionuclide | half-life | energy (KeV) | emitter | source |
| 64Cu | 12.7 h | 653 | β+ | cyclotron |
| 67Ga | 78.3 h | 93,185 | γ | cyclotron |
| 89Sr | 50.6 d | 1460 | β- | reactor |
| 90Y | 64.1 h | 2270 | β- | reactor |
| 99mTc | 6.02 h | 141 | γ | generator |
| 111In | 67.9 h | 171,247 | γ | cyclotron |
| 153Sm | 46.3 h | 702,810;103 | β-,γ | reactor |
| 177Lu | 6.7 d | 176,497;113,208 | β-,γ | reactor |
| 186Re | 90.6 h | 936,1070;137 | β-,γ | reactor |
| 188Re | 16.9 h | 1500;155 | β-,γ | generator |
| 201Tl | 73.1 h | 135,167 | γ | cyclotron |
4. Chelators and/or Linkers
The labeling strategy of oligomers is very similar to that of other biologicals such as peptides and antibodies. If the radionuclide is an isotope of a metal, a chelator, possibly attached via a linker to avoid steric hindrances, is required. We describe below the use of MAG3, DTPA, and DOTA as bifunctional chelators (i.e., with two functionalities, one permitting covalent attachment and another permitting chelation) because of their common use for radiolabeling oligomers with some metallic radionuclides.
4.1. MAG3 derivatives
When labeled with 99mTc, mercaptoacetyltriglycine (MAG3) is a clinical radiopharmaceutical for imaging kidney function [14]. The sulfhydryl group in the clinical MAG3 radiopharmaceutical is protected by a benzoyl group that requires high temperatures and basic pH conditions for deprotection.
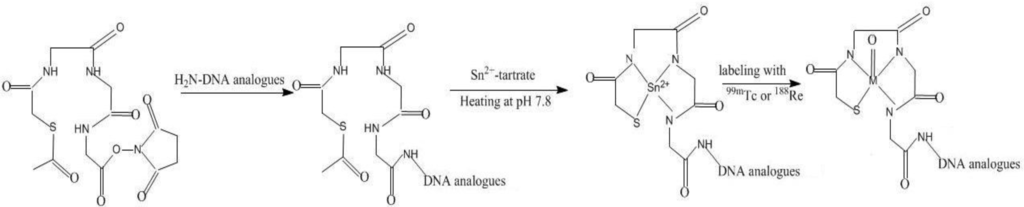
Figure 2.
Conjugation of the NHS-MAG3 to amine-derivatized oligomers, and radiolabeling.
Because of its ability to stabilize chelate 99mTc, MAG3 has also been modified into a bifunctional chelator for the labeling of biologicals. A NHS (N-Hydroxyl succinimide) activated MAG3 bifunctional chelator (S-acetyl NHS-MAG3) has been used for the labeling of amine derivatized oligomers with 99mTc (Figure 2). To avoid the harsh conditions of boiled water temperature and alkaline pH of benzoyl deprotection, S-acetyl NHS-MAG3 was synthesized in which an acetyl replaces the benzoyl group. As a better leaving group, acetyl can be more easily removed at neutral pH and room temperature [15]. However, while the labeling of DNAs in this manner with 99mTc at room temperature and neutral pH became routine [16,17,18,19], post-labeling purification was required to raise the radiochemical purity to 90% or higher [20]. Fortunately, oligomers tend to be insensitive to heat. In an investigation to examine the reasons for the lower labeling efficiency, the conjugation and labeling chemistry was found to be more complicated than expected. When the free S-acetyl NHS-MAG3 was labeled at room temperature, the product was an unidentified labeled product and not the expected 99mTcO-MAG3 [21] although the unidentified labeled products was converted into 99mTcO-MAG3 after heating. When NHS-MAG3 was first used to radiolabel an anime-derivatized MORF, low labeling efficiency was found despite boiling water temperatures [22,23]. Subsequent investigations found that the low labeling efficiency was not due to incomplete purification of the MAG3-MORF after conjugation but due to the labeling of impurities [20]. Liu et al found that these impurities could be removed by introducing a preliminary purification procedure before labeling and a labeling efficiency of over 95% is now obtained routinely [24,25].
To our knowledge, activated groups other than NHS active esters have not been used for the conjugation of MAG3 to oligomers. However, as earlier mentioned, all other conjugation approaches employed in the labeling of peptides and antibodies can be potentially translatable to the labeling of oligomers, for example MAG3 activated by an isothiocyanate group [26]. Like most chelators useful with 99mTc, MAG3 can also form stable complex with 186Re and 188Re, because of similarities in the chemistry of technetium and rhenium.
4.2. DTPA
Although the active DTPA ester formed in situ by reacting with a carbodiimide such as EDC [1-Ethyl-3(3-dimethylaminopropyl)carbodiimide Hydrochloride)] has been used for the conjugation of DTPA to biologicals [27], DTPA in the form of a bifunctional chelator has proved to be more popular. As the simplest bifunctional form of DTPA, the cyclic DTPA anhydride has been widely used [28], for example to radiolabel amine derivatized DNAs and MORFs with 99mTc [19,27]. However unlike 99mTc-MAG3, the 99mTc within the DTPA chelator is susceptible to oxidation once the excess Sn(II) used in the labeling is removed [29]. Furthermore, the labeling efficiency is usually low because DTPA is a poor chelator for 99mTc [30]. As such, DTPA has seen more use for attaching trivalent metals such as 111In as shown in Figure 3 [31] and radioactive lanthanides such as 90Y [32]. The labeling can be achieved by simply mixing the DTPA-conjugated biologicals with the acetate, the kinetics is rapid and an almost quantitative labeling efficiency is usually achieved even at neutral pH and room temperature. This method of labeling is therefore suitable for biologicals sensitive to excessive heat and acidity.
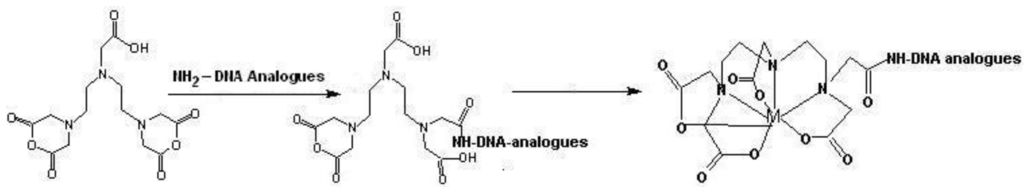
Figure 3.
Conjugation of the cyclic DTPA anhydride to amine-derivatized oligomers, and radiolabeling.
Earlier there was a concern that the use of the cyclic DTPA anhydride could cross link two biologicals because of the two anhydride groups [33]. However, to our knowledge, no direct evidence of cross linking has been reported. A plausible explanation is that the cyclic DTPA anhydride is normally added at a five-fold molar excess or higher. The excess anhydride is also often necessary to compensate for the hydrolysis of the anhydride that will occur in aqueous solution in competition with conjugation.
Another concern to the use of the cyclic anhydride is the possible compromised chelation stability because of the loss of one carboxylate to the conjugation [34,35]. Therefore other activated conjugation groups have been introduced in which the linker is attached to one of the ethylene carbons thus leaving all five carboxylates intact. For example, an isothiocyanate group has been added to the DTPA structure and used to conjugate biologicals [34] including RNA analogs [36]. The isothiocyanate benzyl DTPA is commercially available, the conjugation condition is mild, and the labeling efficiency is high for some isotopes, such as 111In, 90Y and 177Lu [37,38,39].
4.3. DOTA
Similar to DTPA, 1,4,7,10-Tetraazacyclododecane-N,N’,N’’,N’’’-tetraacetic acid (DOTA) is also a good chelator for trivalent metals, such as In3+, Y3+ and other metals of the lanthanide series [40] and can be readily modified into a bifunctional chelator, although by first activating a carboxylate with a carbodiimide as with DTPA, DOTA itself may be conjugated. Alternatively, DOTA bifunctional chelators may be prepared by derivatization of one of the carboxylates, and their NHS or SCN activated derivatives as shown in Figure 4.
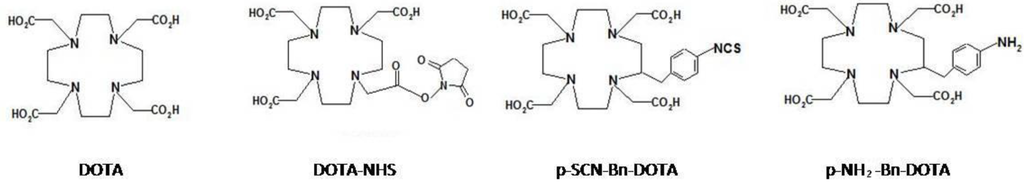
Figure 4.
DOTA and three bifunctional DOTA chelators.
Unlike DTPA, DOTA is a macrocyle with a 12-membered tetraaza macrocycle ring and due to the rigid ring structure, its radiometal complexes tend to be kinetically inert to dissociation and its complexes are also characterized by high thermodynamic stability [41]. However, because the kinetics of radiolabeling of all macrocycles tends to be slow at room temperature, heating is often applied [42]. In addition, the ring size is a sensitive parameter of radiolabeling efficiency and radiolabel stability. For example, DOTA prefers smaller ions including Y3+ and other lanthanides to In3+ and Ga3+ [43]. A MORF has been labeled with 90Y and 111In using SCN-benzyl-DOTA [44]. Figure 5 illustrates conjugation of SCN-benzyl-DOTA to an amine-derivatized oligomer and the subsequent radiolabeling.
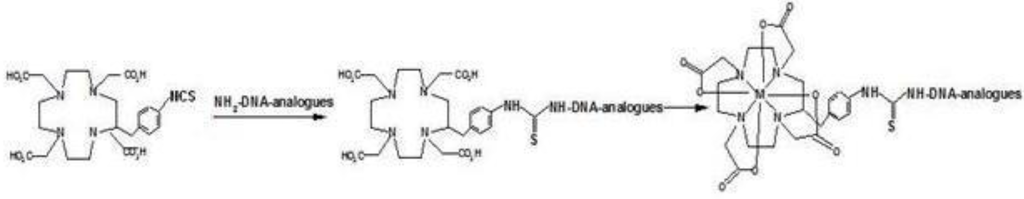
Figure 5.
Conjugation of the SCN-benzyl-DOTA to amine-derivatized oligomers, and radiolabeling.
4.4. HYNIC
The bifunctional chelator 6-Hydrazinopridine-3-carboxylic acid (Hynic) is used mainly for radiolabeling of biologicals with 99mTc [31,45,46]. Strictly speaking, Hynic is not a chelator because the strong association between Hynic and 99mTc is not due to chelation but due to the formation of a single strong diazenido double bond. The oxidation state of technetium in this structure is 5 [99mTc(V)] [47,48,49,50,51,52,53,54]. Although the 99mTc bond to Hynic is very stable towards dissociation, it is susceptible to air oxidation. While chelators for 99mTc are usually translatable to radiorhennium, Hynic may be an exception [55]. It has been reported that the labeling efficiency of a 188Re labeled Hynic conjugate dropped from 97% to 80% in 1 h upon storage [56], probably because rhenium is more easily air oxidized especially in the absence of excess tin (II).
Because Hynic occupies only one or two coordination positions (see below) of the octahedral coordination sphere of technetium, a coligand is required. The wrong choice of coligands can seriously decrease the labeling efficiency and stability of the radiolabel. If a weak coligand such as tricine [57,58,59,60,61,62,63] (Figure 6) or glucoheptonate [64,65,66,67,68,69,70] is selected, dissociation of the coligand can occur in vivo and replaced with endogenous proteins leading to high normal tissue backgrounds [71]. Stronger coligands such as EDDA and tricine/phosphine can be introduced to replace tricine after labeling to avoid this ligands exchange [72,73].
The bifunctional chelator NHS-Hynic is commercially available and has been used to label oligomers but postlabeling purification was required [19,31]. In addition to labeling with 99mTc(V), Hynic has also been used as a true bidentate chelator when used with 99mTc(I) as 99mTc(CO)3+, however the labeling chemistry remains poorly understood. One concern over this approach is high kidney accumulation that may be explained by the one remaining labile position [74].
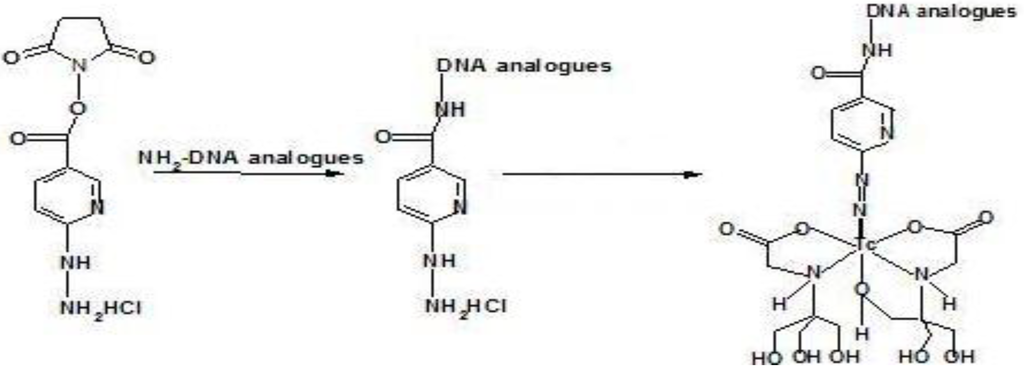
Figure 6.
Conjugation of the NHS-Hynic to amine-derivatized oligomers, and radiolabeling with 99mTc.
5. Biological Properties and Labeling
Radiolabeled biologicals used in nuclear medicine are usually required to faithfully trace the behavior of the unlabeled biological in vivo. Since determining the biodistributions accurately of unlabeled compounds is extremely difficult, the reasonable assumption is usually made that the biodistribution of a biological will be unchanged if labeled by isotope substitution in which a stable atom in the structure is replaced with its radioactive isotope as mentioned above. However, as already mentioned, there are no convenient long lived, imaginable radioisotopes of naturally occurring elements such as carbon, oxygen and nitrogen. Biologicals are therefore usually radiolabeled with elements not found in biologicals such as technetium, indium and yttrium. Because the radioisotopes of these elements are metals, the modification of the biological usually requires attaching a relatively bulky chelator. For large molecules such as IgG antibodies, the effect on the biodistribution due to the radiolabeling is normally assumed to be minimal, however for molecules of small size such as peptides, biodistribution change after radiolabeling has been observed [75,76].
A search of the literature failed to find any comparison of biodistributions of oligomers before and after radiolabeling by chelation. It has been shown that biodistribution of an oligomer can be influenced by different conjugation groups and labeling methods [19]. However, the biodistributions of a 25 mer MORF oligomer labeled with 90Y and 111In using SCN-Benzyl-DTPA and with 99mTc and 188Re using NHS-MAG3 were shown to be essentially identical [44]. This observation was partially supported by measuring the biodistributions of an 18 mer MORF oligomer labeled both with 99mTc via MAG3 and 111In via DTPA. The biodistributions were again essentially identical in normal organs except for the intestinal tract [27] where the excretion was essentially negligible in the case of 111In and was about 2% in the case of 99mTc.
6. Conclusions
Reports describing the radiolabeling of DNA and RNA and their analogs with radionuclides of diagnostic and therapeutic importance are continually appearing. However, we have focused herein only on the chelation labeling of oligomers with metallic radionuclides using common chelators popular in this and other laboratories. We hope that we have shown that a range of labeling methods are available for these biologicals to attach the more common metallic radionuclides useful in nuclear medicine but that each has its peculiar advantages and disadvantages. Compared to only a few years ago, these labeling methods may now be described as mature such that the emphasis need not be placed to the same degree on the labeling but on the use of these radiolabeled oligomers in various attractive in vivo applications.
References
- John, V.F. New technologies for human cancer imaging. J. Clin. Oncol. 2008, 26, 4012–4021. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Xie, C.; Jin, X.; Zhang, P. Simultaneous modulated accelerated radiation therapy in the treatment of nasopharyngeal cancer: A local center’s experience. Int. J. Radiat. Oncol. Biol. Phys. 2009, 66, 40–46. [Google Scholar] [CrossRef]
- Liu, G.; Dou, S.; Yin, D.; Squires, S.; Liu, X.; Wang, Y.; Rusckowski, M.; Hnatowich, D.J. A novel pretargeting method for measuring antibody internalization in tumor cells. Cancer Biother. Radio. 2007, 22, 33–39. [Google Scholar] [CrossRef]
- Wang, Y.; Nakamura, K.; Liu, X.; Kitamura, N.; Kubo, A.; Hnatowich, D.J. Simplified preparation via streptavidin of antisense oligomers/carriers nanoparticles showing improved cellular delivery in culture. Bioconjugate Chem. 2007, 18, 1338–1343. [Google Scholar] [CrossRef]
- Hicke, B.J.; Stephens, A.W.; Gould, Ty.; Chang, Y.; Lynott, C.K.; Heil, J.; Borkowski, S.; Hilger, C.; Cook, G.; Warren, S.; Schmidt, P.G. Tumor targeting by an aptamer. J. Nucl. Med. 2006, 47, 668–678. [Google Scholar] [PubMed]
- Agrawal, S.; Temsamani, J.; Tang, J.Y. Pharmacokinetics, biodistribution, and stability of oligodeoxynucleotide phosphorothioates in mice. Proc. Natl. Acad. Sci. USA 1991, 88, 7595–7599. [Google Scholar] [CrossRef]
- Summerton, J.; Stein, D.; Huang, S.B.; Matthew, P.; Weller, D.; Partridge, M. Morpholino and phosphporothioate antisense oligomers compared in cell-free and in-cell systems. Antisense Nucleic. Acid. Drug. Dev. 1997, 7, 63–70. [Google Scholar] [CrossRef]
- Nielsen, P.E. Antisense properties of peptide nucleic acid. Method. Enzymol. 1999, 313, 156–164. [Google Scholar]
- Koshkin, A.A.; Singh, S.K.; Rajwanshi, V.K.; Kumar, R.; Meldgaard, M.; Olsen, C.E.; Wengel, J. LNA(locked nucleic acids): synthesis of the adenine, cytosine, guanine, 5-methylcytosine, thymine and uracil bicyclonucleoside monomers, oligomerisation, and unprecedented nucleic acid recognition. Tetrahedron 1998, 54, 3607–3630. [Google Scholar] [CrossRef]
- Obika, S.; Nanbu, D.; Hari, Y.; Andoh, J.; Morio, K.; Doi, T.; Imanishi, T. Stability and structural features of the duplexes containing nucleoside analogues with a fixed N-type conformation, 2’-O, 4’-C-methyleneribonucleosides. Tetrahedron Lett. 1998, 39, 5401–5404. [Google Scholar] [CrossRef]
- Wolf, W.; Shani, J. Criteria for the selection of the most desirable radionuclide for radiolabeling of monoclonal antibodies. Nucl. Med. Biol. 1986, 13, 319–324. [Google Scholar]
- Fawwaz, R.A.; Wang, T.S.T.; Srivastava, C.; Hardy, M.A. The use of radionuclides for tumor therapy. Nucl. Med. Biol. 1986, 13, 429–436. [Google Scholar]
- Schubiger, P.A.; Alberto, R.; Smith, A. Vehicles, chelators, and radionuclides: chossing the “building blocks” of an effective therapeutic radioimmunocojugate. Bioconjugate Chem. 1996, 7, 165–179. [Google Scholar] [CrossRef]
- Nosco, D.L.; Manning, R.G.; Fritzberg, A. Characterization of the new 99mTc dynamic renal imaging agent, 99mTc-MAG3 (abstr). J. Nucl. Med. 1986, 27, 939. [Google Scholar]
- Duncan, R.J.S.; Weston, P.D.; Wrigglesworth, R. A new reagent which may be used to introduce sulfhydryl groups into protein, and its use in the preparation of conjugates for immunoassay. Anal. Biochem. 1983, 132, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Winnard, P.J.; Chang, F.; Rusckowski, M.; Mardirossian, G.; Hnatowich, D.J. Preparation and use of NHS-MAG3 for the technetium-99m labeling of DNA. Nucl. Med. Biol. 1997, 24, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Hnatowich, D.J.; Qu, T.; Chang, F.; Ley, A.C.; Ladner, R.C.; Rusckowski, M. Labeling peptides with technetium-99m using a bifunctional chelator of a N-hydroxysuccinimide ester of mercatoacetyltriglycine. J. Nucl. Med. 1998, 39, 56–64. [Google Scholar] [PubMed]
- Rusckowski, M.; Qu, T.; Pullman, J.; Marcel, R.; Ley, A.C.; Ladner, R.C.; Hnatowich, D.J. Imflammation and infection imaging with a 99mTc-neutrophil elastase inhibitor in monkeys. J. Nucl. Med. 2000, 41, 363–374. [Google Scholar] [PubMed]
- Zhang, Y.; Liu, N.; Zhu, Z.; Rusckowski, M.; Hnatowich, D.J. Influence of different chelators (HYNIC, MAG3 and DTPA) on tumor cell accumulation and mouse biodistribution of technetium-99m labeled to antisense DNA. Eur. J. Nucl. Med. 2000, 27, 1700–1707. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Zhang, S.; He, J.; Zhu, Z.; Rusckowski, M.; Hnatowich, D.J. Improving the labeling of S-Acetyl NHS-MAG-conjugated morpholino oligomers. Bioconjugate Chem. 2002, 13, 893–897. [Google Scholar] [CrossRef]
- Hnatowich, D.J.; Chang, F.; Lei, K.; Qu, T.; Rusckowski, M. The influence of temperature and alkaline pH on the labeling of free and conjugated MAG3 with technetium-99m. Appl. Radiat. Isot. 1997, 48, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Mang’era, K.O.; Liu, G.; Yi, W.; Liu, N.; Gupat, S.; Rusckowski, M.; Hnatowich, D.J. Initial investigation of 99mTc-labeled morpholinos for radiopharmaceutical applications. Eur. J. Nucl. Med. 2001, 28, 1682–1689. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Mang’era, K.; Liu, N.; Gupta, S.; Rusckowski, M.; Hnatowich, D.J. Tumor pretargeting in mice using 99mTc-labeled morpholino, a DNA analogue. J. Nucl. Med. 2002, 43, 384–391. [Google Scholar] [PubMed]
- Liu, G.; Dou, S.; He, J.; Yin, D.; Gupta, S.; Zhang, S.; Wang, Y.; Rusckowski, M.; Hnatowich, D.J. Radiolableing of MAG3-morpholino oligomers with 188Re at high labeling efficiency and specific radioactivity for tumor pretargeting. Appl. Radiat. Isotopes 2006, 64, 971–978. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, G.; Hnatowich, D.J. Methods for MAG3 conjugation and 99mTc radiolableing of biomolecules. Nat. Method. 2006, 1, 1477–1480. [Google Scholar]
- Ram, S.; Merriwether, W.; Buchsbaum, D.J. Synthesis and biodistribution of peptide based 99mTc/186Re-MAGIPG-D612 monoclonal antibody in nude mice bearing colon cancer xenografts. Cancer Biother. Radio. 1997, 12, 55–62. [Google Scholar] [CrossRef]
- Liu, G.; Cheng, D.; Dou, S.; Chen, X.; Liang, M.; Pretorius, H.; Rusckowski, M.; Hnatowich, D.J. Replacing 99mTc with 111In improves MORF/cMORF pretargeting by reducing intestinal accumulation. Mol. Image. Biol. 2009, 11, 303–307. [Google Scholar] [CrossRef]
- Hnatowich, D.J.; Layne, W.W.; Childs, R.L.; Lanteigne, D.; Davis, M.A. Radioactive labeling of antibody: a simple and efficient method. Science 1983, 220, 613–615. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; University of Massachusetts Medical School, Worcester, MA 01655, USA. Unpublished observations.
- Childs, R.L.; Hnatowich, D.J. Optimum conditions for labeling of DTPA-coupled antibodies with technetium-99m. J. Nucl. Med. 1985, 26, 293–299. [Google Scholar]
- Hnatowich, D.J.; Winnard, P.; Virzi, F.; Fogarasi, M.; Sano, T.; Smith, C.L.; Cantor, C.R.; Rusckowski, M. Technetium-99m labeling of DNA oligonucleotides. J. Nucl. Med. 1995, 36, 2306–2314. [Google Scholar]
- Hovinen, J. Labeling of oligonucleotides with DTPA and DOTA on solid phase. Nucleos. Nucleot. Nucl. 2007, 26, 1459–1462. [Google Scholar] [CrossRef]
- Reilly, R.M.; Scollard, D.A.; Wang, J.; Mondal, H.; Chen, P.; Henderson, L.A.; Bowen, B.M.; Vallis, K.A. A kit formulated under good manufacturing practices for labeling human epidermal growth factor with 111In for radiotherapeutic applications. J. Nucl. Med. 2004, 45, 4701–4708. [Google Scholar]
- Brechbiel, M.W.; Gansow, O.A.; Atcher, R.W.; Schlom, J.; Esteban, J.; Simpson, D.; Colcher, D. Synthesis of 1-(p-isothiocyanatobenzyl) derivatives of DTPA and EDTA. Antibody labeling and tumor-imaging studies. Inorg. Chem. 1986, 25, 2772–2781. [Google Scholar] [CrossRef]
- Brockmann, J.; Rosch, F. Determination of stability constants in Y-DTPA-peptide-systems: Evaluation of a radiochemical method using n. c. a. Yttrium-88. Radiochim. Acta 1999, 87, 79–85. [Google Scholar]
- Merkel, O.M.; Librizzi, D.; Pfestroff, A.; Schurrat, T.; Béhé, M.; Kissel, T. In vivo SPECT and Real-Time Gamma camera imaging of biodistribution and pharmacokinetics of siRNA delivery using an optimized radiolabeling and purification procedure. Bioconjugate Chem. 2009, 20, 174–182. [Google Scholar] [CrossRef]
- Tolmachev, V.; Nilsson, F.Y.; WidstrÖm, C.; Andersson, K.; Rosik, D.; Gedda, L.; Wennborg, A.; Orlova, A. 111In-Benzyl-DTPA–ZHER2:342, an affibody-based conjugate for in vivo imaging HER2 expression in malignant tumors. J. Nucl. Med. 2006, 47, 846–853. [Google Scholar] [PubMed]
- Kozak, R.W.; Raubitschek, A.; Mirzadeh, S.; Brechbiel, M.W.; Junghans, R.P.; Gansow, O.A.; Waldmann, T.A.; Junghaus, R. Nature of the bifunctional chelating agent used for radioimmunotherapy with yttrium-90 monoclonal antibodies: critical factors in determining in vivo survival and organ toxicity. Cancer Res. 1989, 49, 2639–2644. [Google Scholar] [PubMed]
- Persson, M.; Tolmachev, V.; Andersson, K.; Gedda, L.; Sandström, M.; Carlsson, J. [177Lu]pertuzumab: experimental studies on targeting of HER-2 positive tumor cells. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 1457–1462. [Google Scholar] [CrossRef]
- Schlesinger, J.; Fischer, C.; Koezle, I.; Vonhoff, S.; Klussmann, S.; Bergmann, R. Radiosynthesis of new [90Y]-DOTA-Based malemide reagents suitable for the prelabeling of thiol-bearing L-oligonucleotides and peptides. Bioconjugate Chem. 2009, 20, 1340–1348. [Google Scholar] [CrossRef]
- Leon-Rodriguez, L.M.; Kovacs, Z. The synthesis and chelation chemistry of DOTA-peptide conjugates. Bioconjugate Chem. 2008, 19, 391–402. [Google Scholar] [CrossRef]
- Liu, S. The role of coordination chemistry in the development of target specific radiopharmaceuticals. Chem. Soc. Rev. 2004, 33, 445–461. [Google Scholar] [CrossRef]
- Schlesinger, J.; Koezle, I.; Bergmann, R.; Tamburini, S.; Bolzati, C.; Tisato, F.; Noll, B.; Klussmann, S.; Vonhoff, S.; Wuest, F.; Pietzsch, H.; Steinbach, J. An 86Y-labeled mirror-image oligonucleotide: influence of Y-DOTA isomers on the biodistribution in rats. Bioconjugate Chem. 2008, 19, 928–939. [Google Scholar] [CrossRef]
- Liu, C.; Liu, G.; Liu, N.; Zhang, Y.; He, J.; Rusckowsi, M.; Hnatowich, D.J. Radiolabeling morpholino with 90Y, 111In, 188Re and 99m Tc. Nucl. Med. Bio. 2003, 30, 207–214. [Google Scholar] [CrossRef]
- Kemerink, G.J.; Liu, X.; Kieffer, D.; Ceyssens, S.; Mortelmans, L.; Verbruggen, A.M.; Steinmetz, N.; Green, A.; Verbeke, K. Safety, biodistribution, and dosimetry of 99mTc-HYNIC-annexin V, a novel human recombinant annexin V for human application. J. Nucl. Med. 2003, 44, 947–952. [Google Scholar] [PubMed]
- Babich, J.W.; Solomon, H.; Pike, M.C.; Kroon, D.; Graham, W.; Abrams, M.J.; Tompkins, R.G.; Rubin, R.H.; Fischman, A.J. Technetium-99m-labeleed hydrazine nicotinamide derivatized chemotactic peptide analogs for imaging focal sites of bacterial infection. J. Nucl. Med. 1993, 34, 1967–1974. [Google Scholar]
- Nicholson, T.; Zubieta, J. Complexes of rhenium with benzoylazo and related ligands. Crystal and molecular structures of the "green chelate" benzoylazo complex [ReCl2(PPh3)2(NNCOC6H4Cl)], of the analogous l-azophthalazine chelate complex [ReCl2(PPh3)2(NNC8H5N2)] and of the cis-dichloro organodiazenido complexes of the type [ReCl2(PPh3)2(NNR)L] (L =NCCH3, NH3,C5H5N). A comparison to the structure of the trans-dichloro dimethyl formamide derivative [ReCl2(PPh3)2(NNCO2CH3)(Me2NCHO)]. The structural characterization of the mixed hydrazido(l-) hydrazido(2-) complexes [ReCl2(PPh3)2(NNHR)(NHNHR')] (R = R' = -COC6H5; R = -COC6H5, R' = -CO2CH3). Polyhedron 1998, 7, 171–185. [Google Scholar] [CrossRef]
- Abrams, M.J.; Larsen, S.K.; Shaikh, S.N.; Zubieta, J. Investigations of technetium-organohydrazine coordination chemistry. The crystal and molecular structures of [TcCl2(C8H5N4)(PPh3)2]·0.75C7H8 and [TcNCl2(PPh3)2]·0.25CH2Cl2. Inorg. Chim. Acta. 1991, 185, 7–15. [Google Scholar] [CrossRef]
- Archer, C.M.; Dilworth, J.R.; Jobanputra, P.; Thompson, R.M.; McPartin, M.; Povey, D.C.; Smith, G.W.; Kelly, J.D. Development of new technetium cores containing technetium-nitrogen multiple bonds. Synthesis and characterization of some diazenido-, hydrazido- and imido- complexes of technetium. Polyhedron 1990, 9, 1497–1502. [Google Scholar] [CrossRef]
- Archer, C.M.; Dilworth, J.R.; Jobanputra, P.; Thompson, R.M; McPartin, M.; Hiller, W. Technetium diazenido complexes. Part 1. Synthesis and structures of [TcCl(NNC6H4Cl-4)2(PPh3)2] and TcCl(NNPh)(Ph2PCH2CH2PPh2)2][PF6]·H2O. J. Chem. Soc. Dalton Trans. 1993, 897–904. [Google Scholar]
- Nicholson, T.; Cook, J.; Davison, A.; Rose, D.J.; Maresca, K.P.; Zubieta, J.A.; Jones, A.G. The synthesis and characterization of [MCl3(N=NC5H4NH)(HN=NC5H4N)] from [MO4]-{where M = Re, Tc}, organodiazenido, organodiazene-chelate complexes. The X-ray structure of [ReCl3(N=NC5H4NH)(HN=NC5H4N)]. Inorg. Chim. Acta. 1996, 252, 421–426. [Google Scholar] [CrossRef]
- Hirsch-Kuchma, M.; Nicholson, T.; Davison, A.; Jones, A.G. Group 7 ‘organohydrazide’ chemistry: classification of ligand type based on crystal structural data. J. Chem. Soc. Dalton Trans. 1997, 3, 3189–3192. [Google Scholar] [CrossRef]
- Rose, D.J.; Maresca, K.P.; Nicholson, T.; Davison, A.; Jones, A.G.; Babich, J.; Fischman, A.; Graham, W.; DeBord, J.R.; Zubieta, J. Synthesis and characterization of organohydrazino complexes of technetium, rhenium, and molybdenum with the {M(η1-HxNNR)(η2-HyNNR)} core and their relationship to radiolabeled organohydrazine-derivatized chemotactic peptides with diagnostic applications. J. Inorg. Chem. 1998, 37, 2701–2716. [Google Scholar] [CrossRef]
- Abrams, M.J.; Juweid, M.; TenKate, C.I.; Schwartz, D.A.; Hauser, M.M.; Gaul, F.E.; Fuccello, A.J.; Rubin, R.H.; Strauss, H.W.; Fischman, A.J. Technetium-99m-human polyclonal IgG radiolabeled via the hydrazino nicotinamide derivative for imaging focal sites of infection in rats. J. Nucl. Med. 1990, 31, 2022–2208. [Google Scholar]
- Liu, G.; Hnatowich, D.J. Labeling biomolecules with radiorhenium-a review of the bifunctional chelators. Anticancer Agents Med. Chem. 2007, 7, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.M.; Jeong, H.J.; Heo, Y.J.; Moon, H.B.; Bom, H.S.; Kim, C.G. Intratumoral injection of 188Re labeled cationic polyethylenimine conjugates: a preliminary report. J. Korean Med. Sci. 2004, 19, 647–651. [Google Scholar] [CrossRef] [PubMed]
- Claessens, R.A.; Boerman, O.C.; Koenders, E.B.; Oyen, W.J.; Van der Meer, J.M.; Corstens, F.H. Technetium-99m labeled hydrazinonicotinamide human non-specific polyclonal immunoglobulin G for detection of infectious foci: a comparison with two other technetium-labelled immunoglobulin preparations. Eur. J. Nucl. Med. 1996, 23, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Claessens, R.A.; Koenders, E.B.; Oyen, W.J.G.; Corstens, F.H. Retention of technetium-99m in infectious foci in rats after release from technetium-99m labeled human non-specific polyclonal immunoglobulin G: a dual-label study with hydrazinonicotinamide and iminothiolano immunoglobulin. Eur. J. Nucl. Med. 1996, 23, 1536–1539. [Google Scholar] [CrossRef] [PubMed]
- Dams, E.T.; Oyen, W.J.; Boerman, O.C.; Claessens, R.A.; Wymenga, A.B.; Van der Meer, J.W.; Corstens, F.H. Technitium-99m labeled to human immunoglobulin G through the nicotinyl hydrazine derivative: a clinical study. J. Nucl. Med. 1998, 39, 119–124. [Google Scholar]
- Dams, E.T.; Oyen, W.J.; Boerman, O.C.; Storm, G.; Laverman, P.; Koender, E.B.; Van der Meer, J.W.; Corstens, F.H. Technetium-99m-labeled liposomes to image experimental colitis in rabbits: comparison with technetium-99m-HMPAO-granulocytes and technetium-99m-HYNIC IgG. J. Nucl. Med. 1998, 39, 2172–2178. [Google Scholar] [PubMed]
- Decristoforo, C.; Mather, S.J. 99m-Technetium labeled peptide-HYNIC conjugates. The effects of lipophilicity and stability on biodistribution. Nucl. Med. Biol. 1999, 26, 389–396. [Google Scholar] [CrossRef]
- Decristoforo, C.; Mather, S.J. Preparation, 99mTc-labeling, and in vitro characterization of HYNIC and N3S modified RC-160 and [Tyr3] octreotide. Bioconjugate Chem. 1999, 10, 431–438. [Google Scholar] [CrossRef]
- Larson, S.K.; Solomon, H.F.; Caldwell, G.; Abrams, M.J. [99mTc]tricine: a useful precursor complex for the radiolabeling of hydrazinonicotinate protein conjugates. Bioconjugate Chem. 1995, 6, 635–638. [Google Scholar] [CrossRef]
- Babich, J.W.; Solomon, H.; Pike, M.C.; Kroon, D.; Graham, W.; Abrams, M.J; Tompkins, R.G.; Rubin, R.H.; Fischman, A.J. 99mTc-labeled hydrazine nicotinamide derivatized chemotactic peptides for imaging focal sites of bacterial infection. J. Nucl. Med. 1993, 34, 1964–1974. [Google Scholar]
- Babich, J.W.; Fischman, A.J. Influence of co-ligand on the distribution of 99mTc labeled hydrazinonicotinic acid derivatized chemotactic peptides in normal rats. Nucl. Med. Biol. 1995, 22, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Babich, J.W.; Graham, W.; Barrow, S.A; Fischman, A.J. Comparison of the infection imaging properties of a 99mTc labeled chemotactic peptide with 111In IgG. Nucl. Med. Biol. 1995, 22, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Babich, J.W.; Tompkins, R.G.; Graham, W.; Barrow, S.A.; Fischman, A.J. Localization of radiolabeled chemotactic peptide at focal sites of Escherichia coli infection in rabbits: evidence for a receptor-specific mechanism. J. Nucl. Med. 1997, 38, 1316–1322. [Google Scholar] [PubMed]
- Blankenberg, F.G.; Katsikis, P.D.; Tait, J.F.; Davis, R.E.; Naumovski, L.; Ohtsuki, K.; Kopiwoda, S.; Abrams, M.J.; Strauss, H.W. Imaging of apoptosis (programmed cell death) with 99mTc annexin V. J. Nucl. Med. 1999, 40, 184–191. [Google Scholar] [PubMed]
- Callahan, R.; Barrow, S.A.; Abrams, M.J.; Rubin, R.H.; Fischman, A.J. Biodistribution and dosimetry of technetium-99m-hydrazino nicotinamide IgG: comparison with indium-111-DTPA-IgG. J. Nucl. Med. 1996, 37, 843–846. [Google Scholar] [PubMed]
- Fischman, A.J.; Rauh, D.; Soloman, O.C.; Babich, J.W.; Tompkins, R.G.; Kroon, D.; Strauss, H.W.; Rubin, R.H. In vivo bioactivity and biodistribution of chemotactic peptide analogs in nonhuman primates. J. Nucl. Med. 1993, 34, 2130–2134. [Google Scholar] [PubMed]
- Rusckowski, M.; Qu, T.; Gupta, S.; Ley, A.; Hnatowich, D.J. A comparison in monkeys of 99mTc labeled to a peptide by 4 methods. J. Nucl. Med. 2001, 42, 1870–1877. [Google Scholar] [PubMed]
- Liu, S.; Edwards, D.S.; Looby, R.J.; Harris, A.R.; Poirier, M.J.; Barrett, J.A.; Heminway, S.J.; Carroll, T.R. Labeling a hydrazine nicotinamide-modified cyclic IIb/IIIa receptor antagonist with 99mTc using aminocarboxylates as coligands. Bioconjugate Chem. 1996, 7, 63–71. [Google Scholar] [CrossRef]
- Decristoforo, C.; Mather, S.J. 99mTc-somatostatin analogues effect of labeling methods and peptide sequence. Eur. J. Nucl. Med. 1999, 26, 869–876. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Liu, C.; Vanderheyden, J.; Liu, G.; Dou, S.; Rusckowski, M.; Hnatowich, D.J. Radiolabelling morpholinos with 188Re tricarbonyl provides improved in vitro and in vivo stability to re-oxidation. Nucl. Med. Comm. 2004, 25, 731–736. [Google Scholar] [CrossRef]
- Auzeloux, P.; Papon, J.; Azim, E.M.; Bore, M.; Pasqualini, R.; Veyre, A.; Madelmont, J.C. A potential melanoma tracer: synthesis, radiolabeling, and biodistribution in mice of a new nitridotechnetium bis(aminothiol) derivative pharmacomodulated by a N-(diethylaminoethyl)benzamide. J. Med. Chem. 2000, 43, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Qu, T.; Wang, Y.; Zhu, Z.; Rusckowski, M.; Hnatowich, D.J. Different chelators and different peptides together influence the in vitro and mouse in vivo properties of 99Tcm. Nucl. Med. Comm. 2001, 22, 203–215. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).