Star-Shaped Conjugated Systems
Abstract
:- Table of Content
- 1. Introduction
- 2. Molecular Architecture and Conjugation
- 3. Three-arm Systems with one Central Atom
- 3.1. [3]Star Systems with Methylium Core
- 3.2. [3]Star Systems with Boron Core
- 3.3. [3]Star Compounds with Nitrogen Core
- 4. Star Compounds with a Benzene Core
- 4.1. Three-armed Stars (C-4)
- 4.1.1. Structure and Conjugation
- 4.1.2. Synthesis
- 4.1.3. Three-arm Stars with Benzene Centers and Materials Science
- 4.2. Four-arm Systems-Tetrasubstituted Benzene (C-5)
- 4.2.1. Structure and Conjugation
- 4.2.2. Synthesis
- 4.2.3. Four-arm Stars with Benzene Centers and Materials Science
- 4.3. Six-arm Systems-Hexasubstituted Benzenes (C-6)
- 4.3.1. Structure and Conjugation of Parent Systems
- 4.3.2. Synthesis of the Parent Systems
- 4.3.3. Hexaarm Stars with Benzene Centers and Materials Science
- 5. Compounds with Heterocyclic Cores
- 5.1. Pyridine-based Stars (C-7-A-6, C-7-A-7)
- 5.2. Stars with a Pyrimidine Core
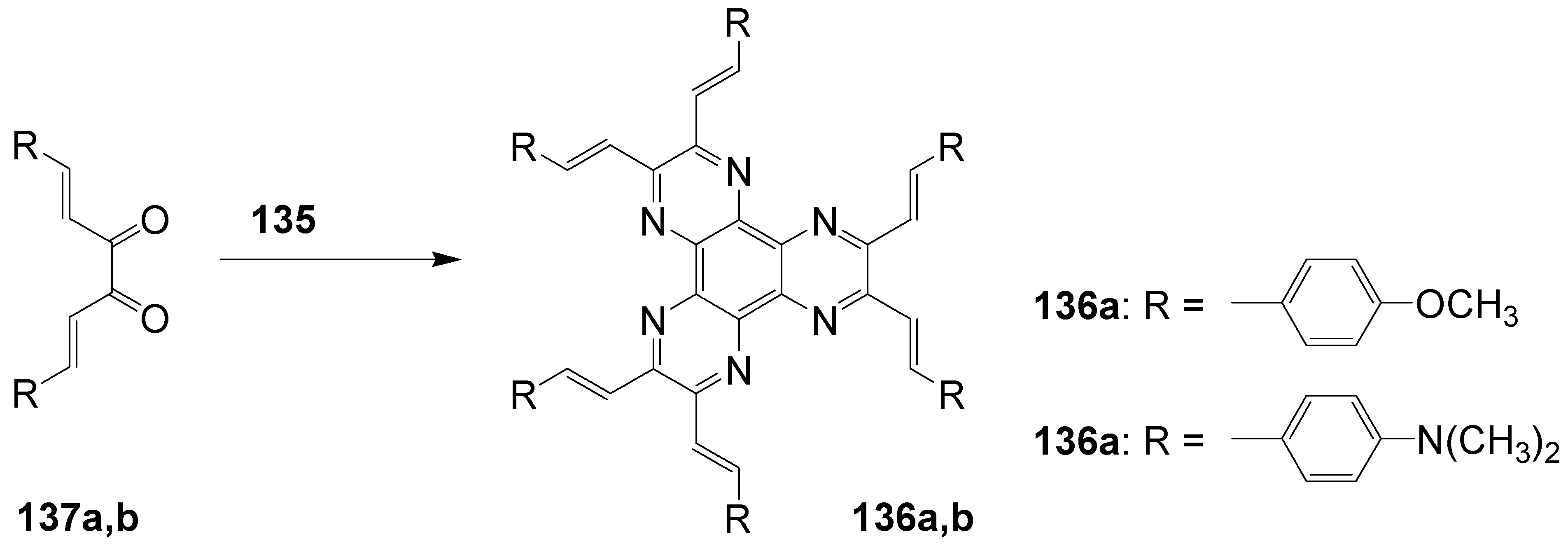
- 5.3. Pyrazine as Core (C-10-A-6)
- 5.4. 1,3,5-Triazine as Core
- 5.5. Borazine as a Core
- 6. Condensed Ring Systems as Cores
- 6.1. Triphenylene Star Compounds (C-12)
- 6.1.1. Structures and Synthesis
- 6.1.2. Triphenylene Derivatives and Materials Science
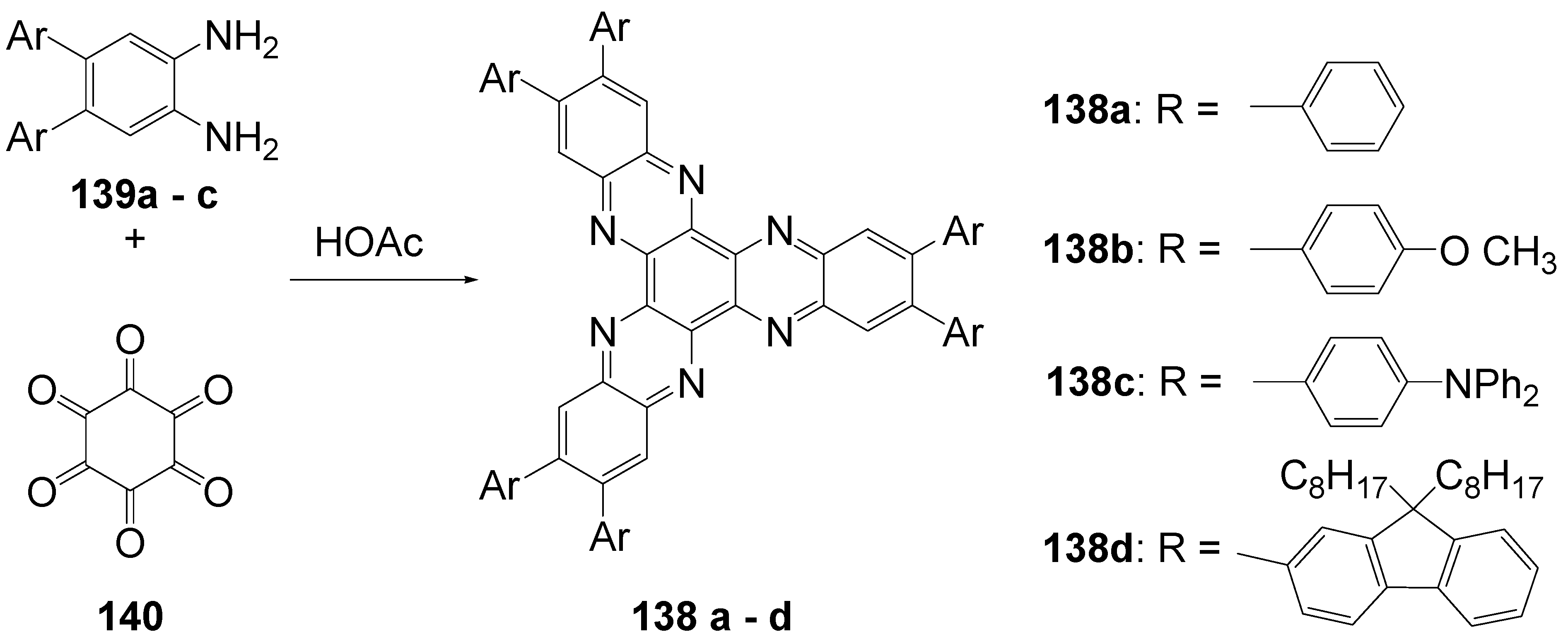
- 6.2. Hexaazatriphenylene (C-13-A-3 and C-13-A-6)
- 6.3. Triazatruxenes (C-14-A-3 and C-14-A-8)
- 6.4. Tristriazolotriazines (C-15-A-3 and C-15-A-9)
- 7. Summary and Conclusion
1. Introduction
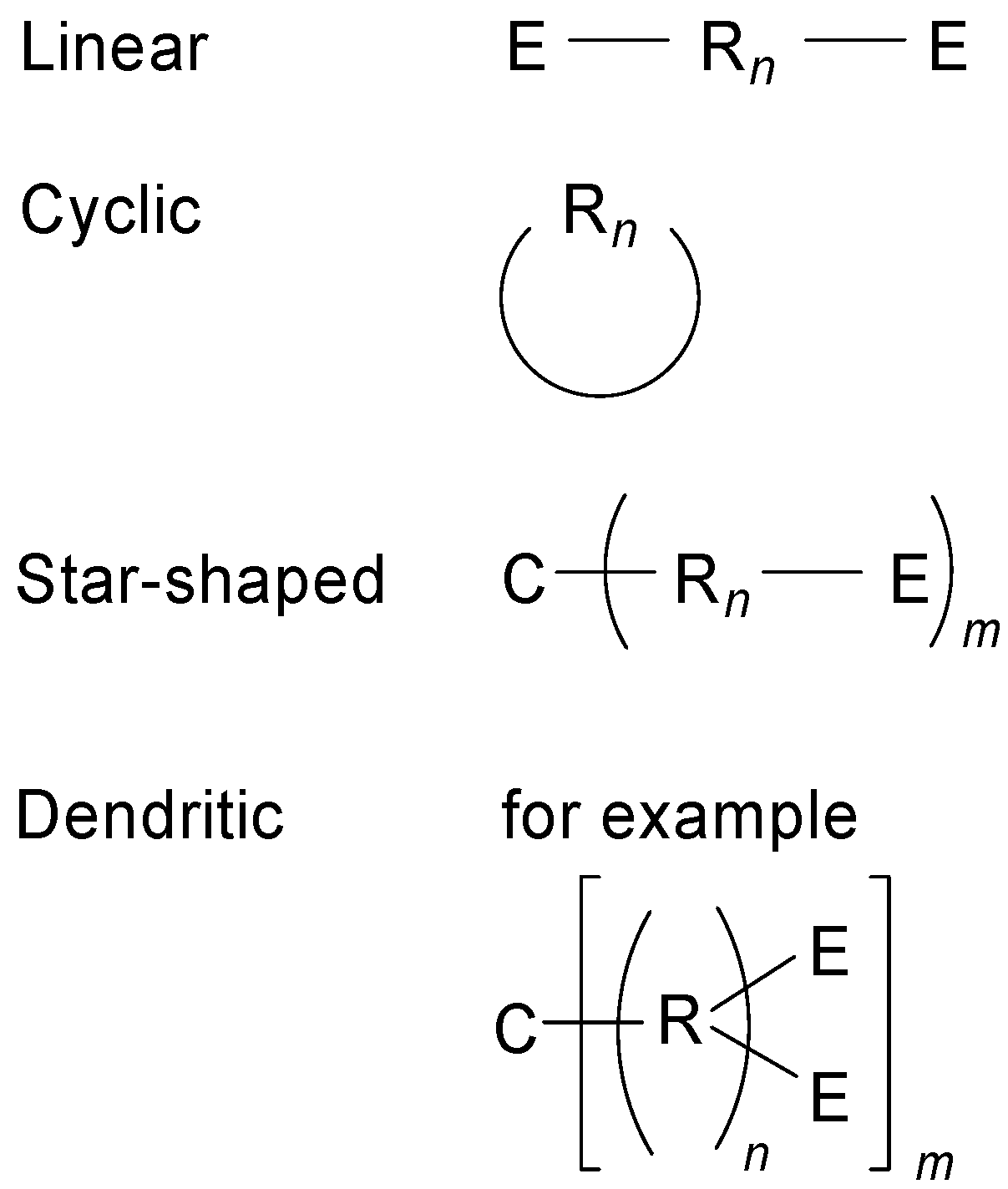
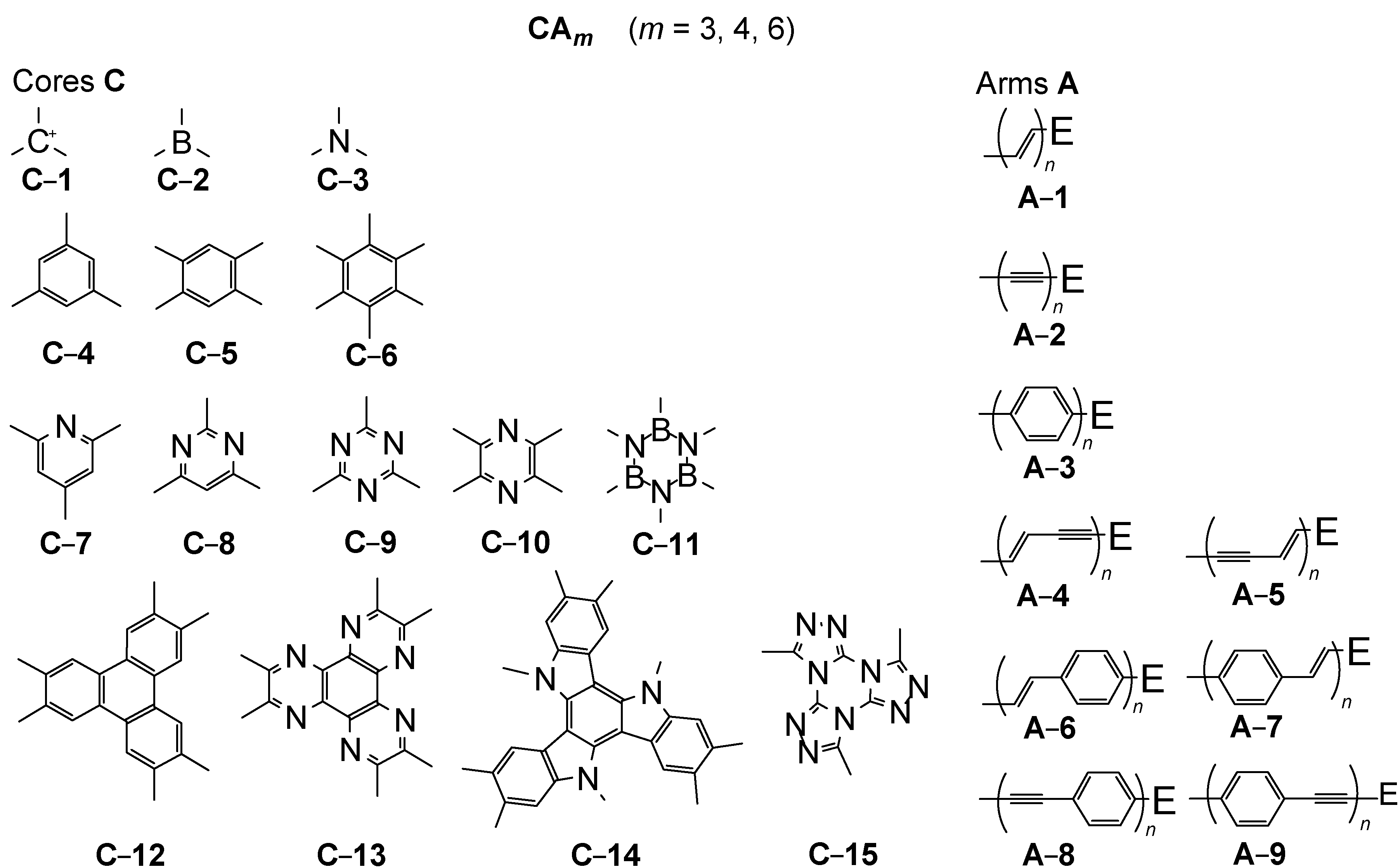
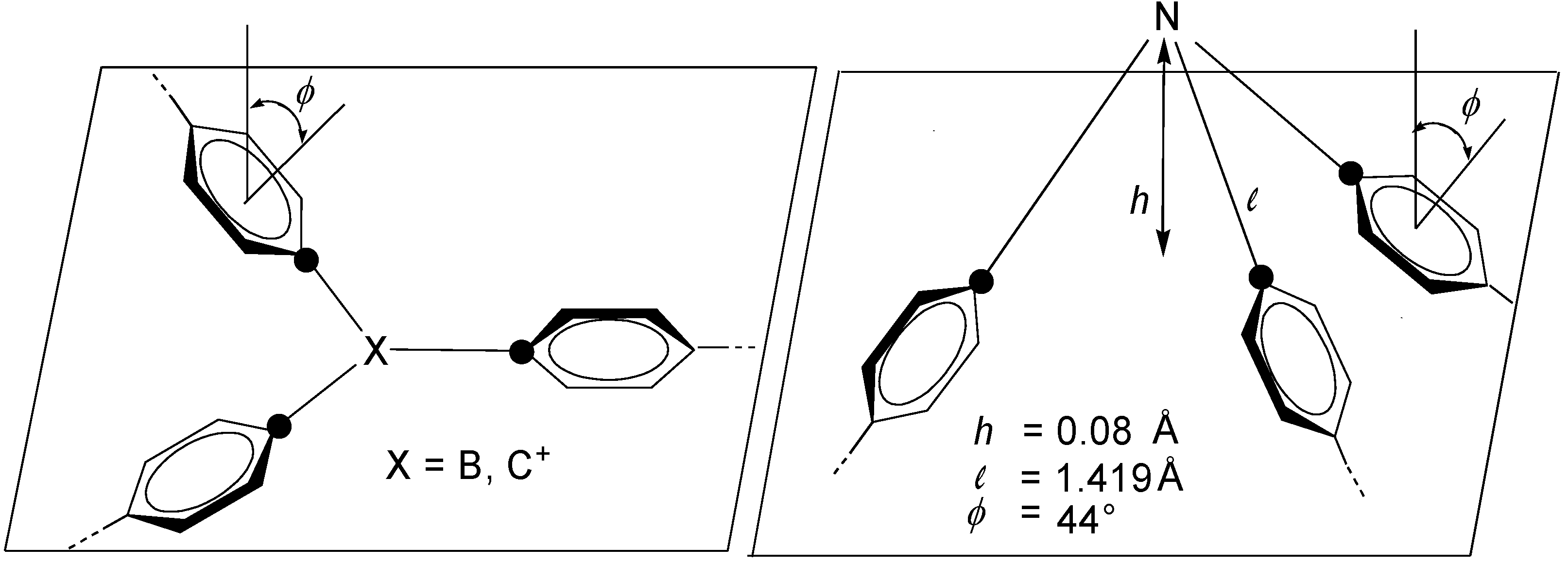
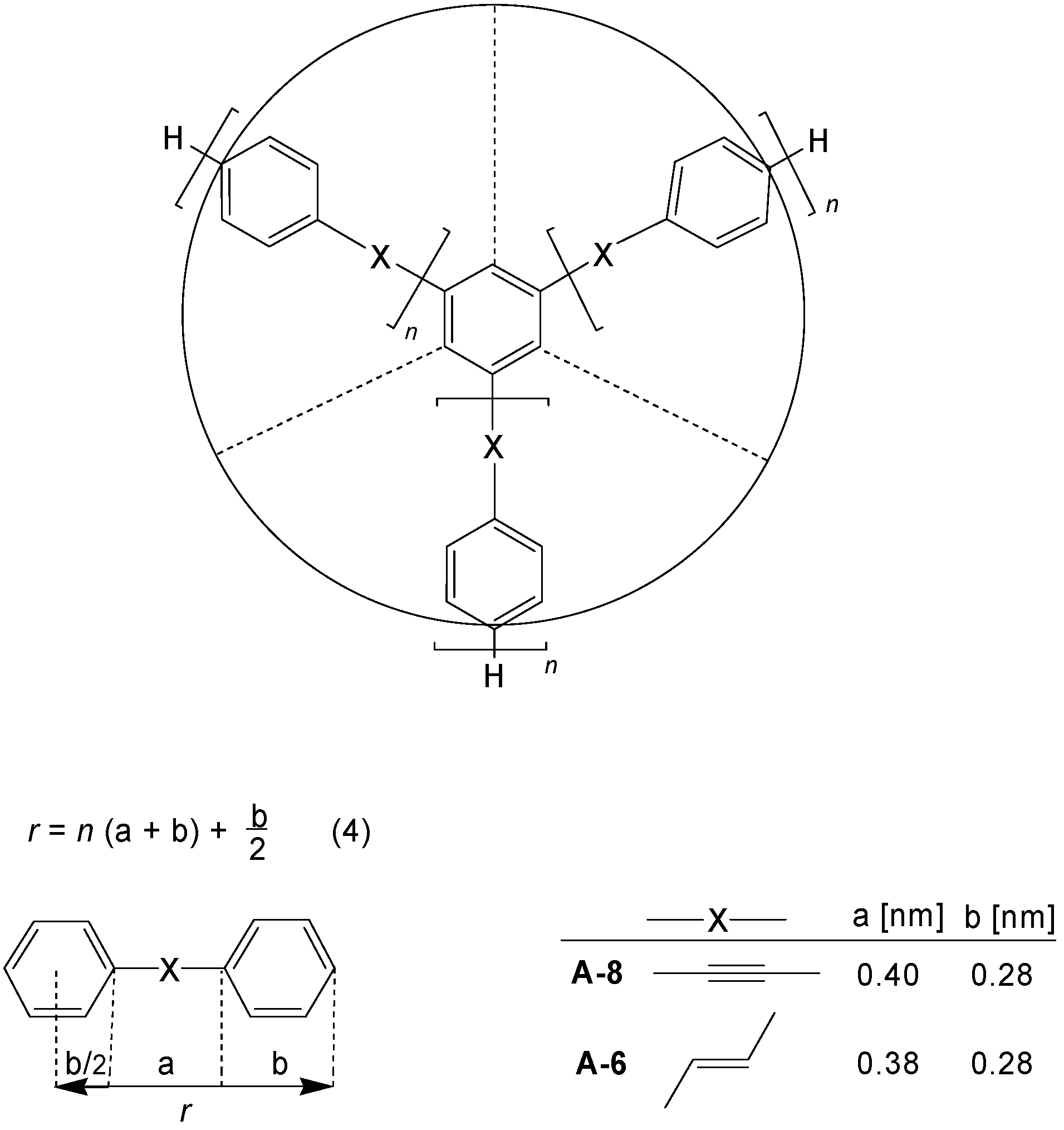
2. Molecular Architecture and Conjugation
| Generation n | Core Symmetry D3h C2v Cs |
| 1 2 3 4 | 2 4 8 12 32 64 88 256 512 696 512 1024 |
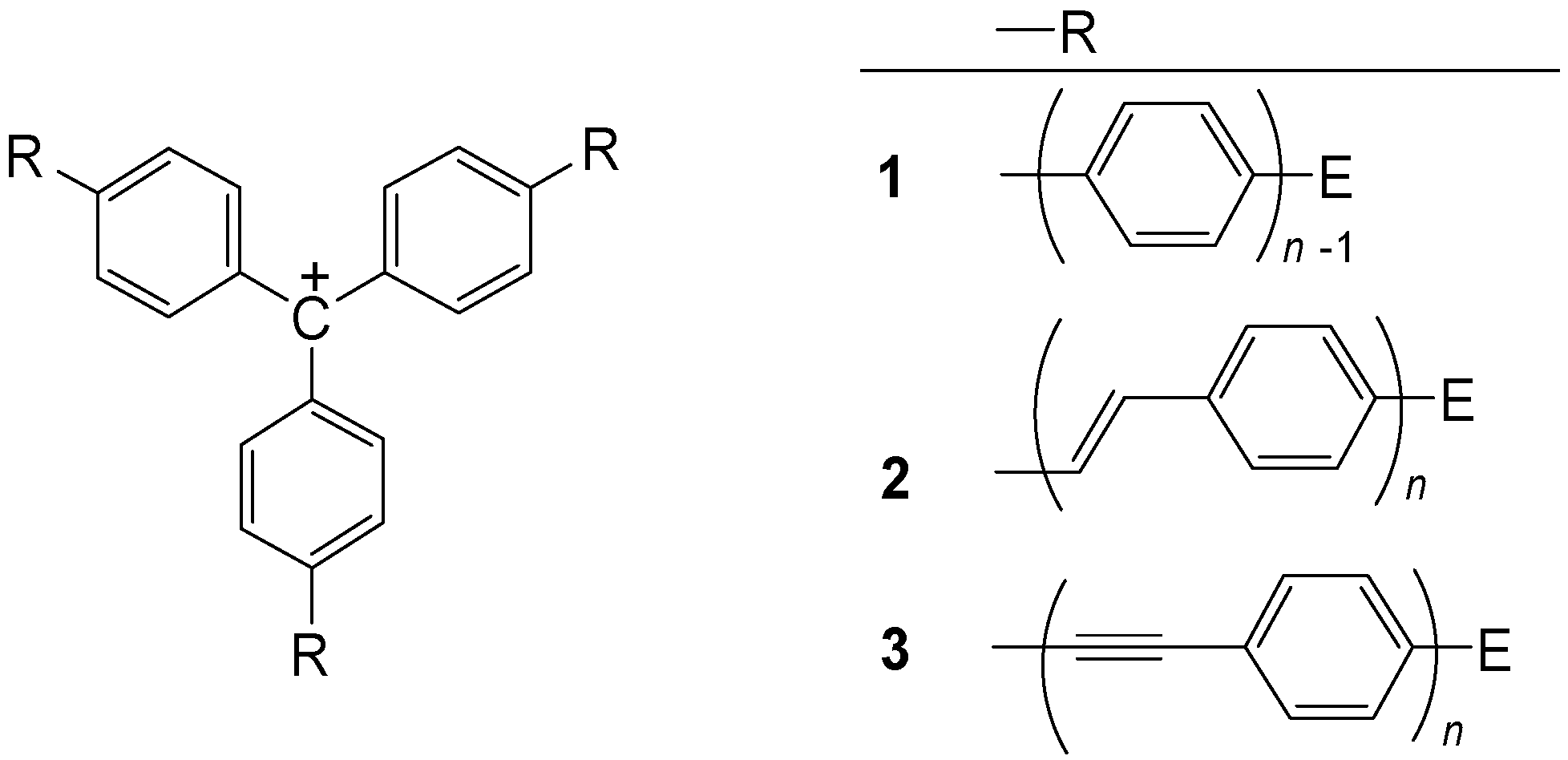
3. Three-Arm Systems with One Central Atom
3.1. [3]Star Systems with Methylium Core

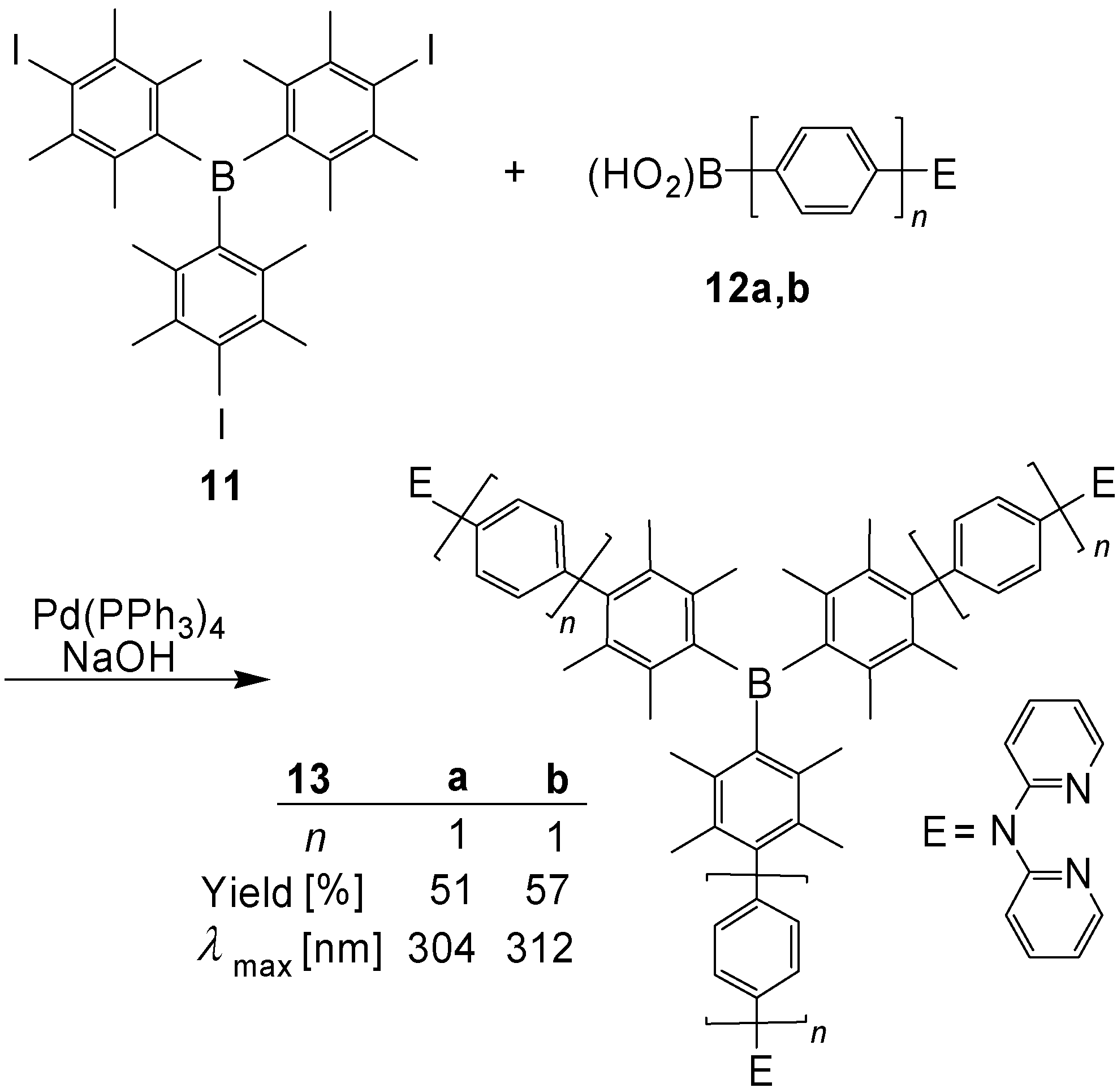
3.2. [3]Star Systems with Boron Core

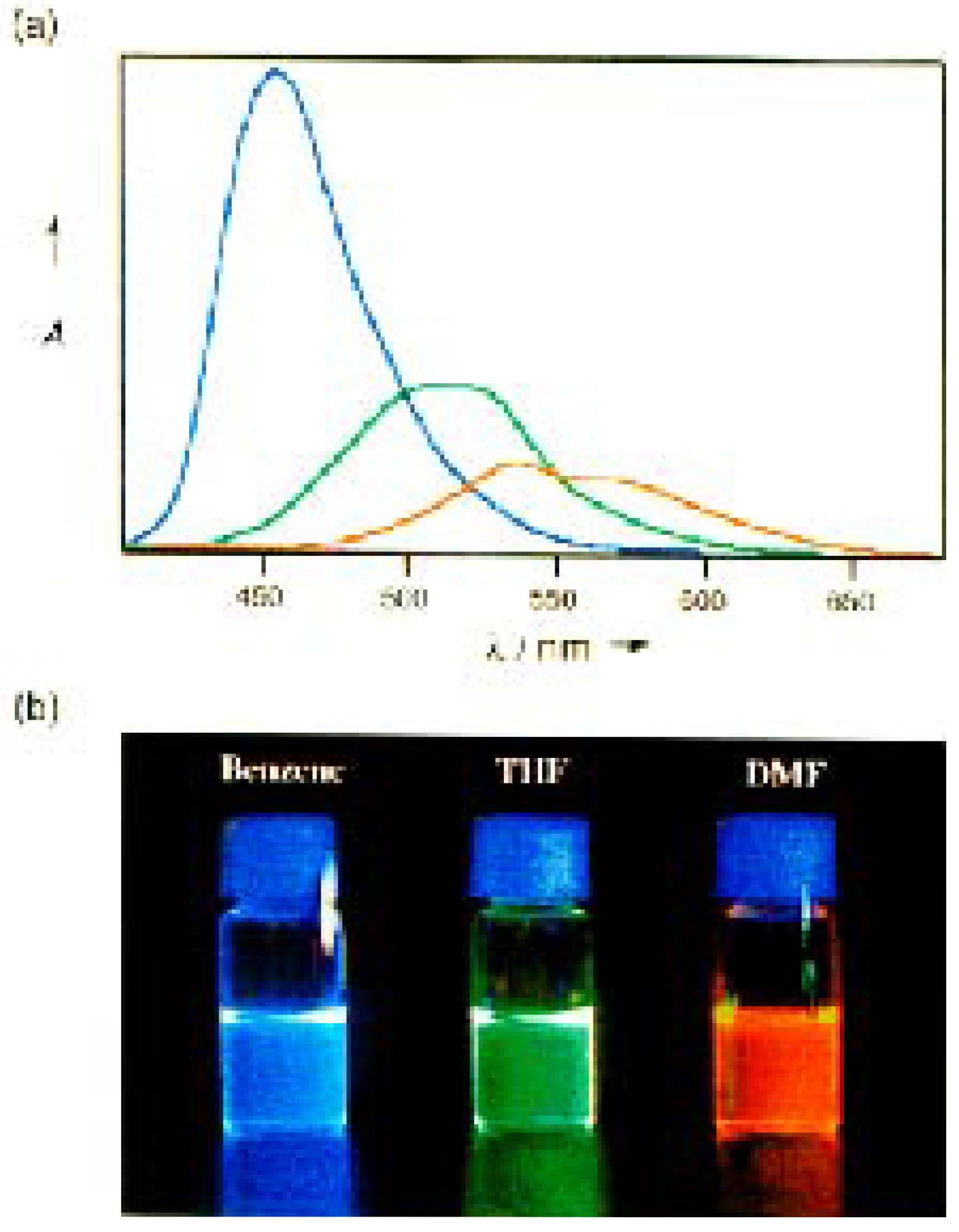
3.3. [3]Star Compounds with Nitrogen Core
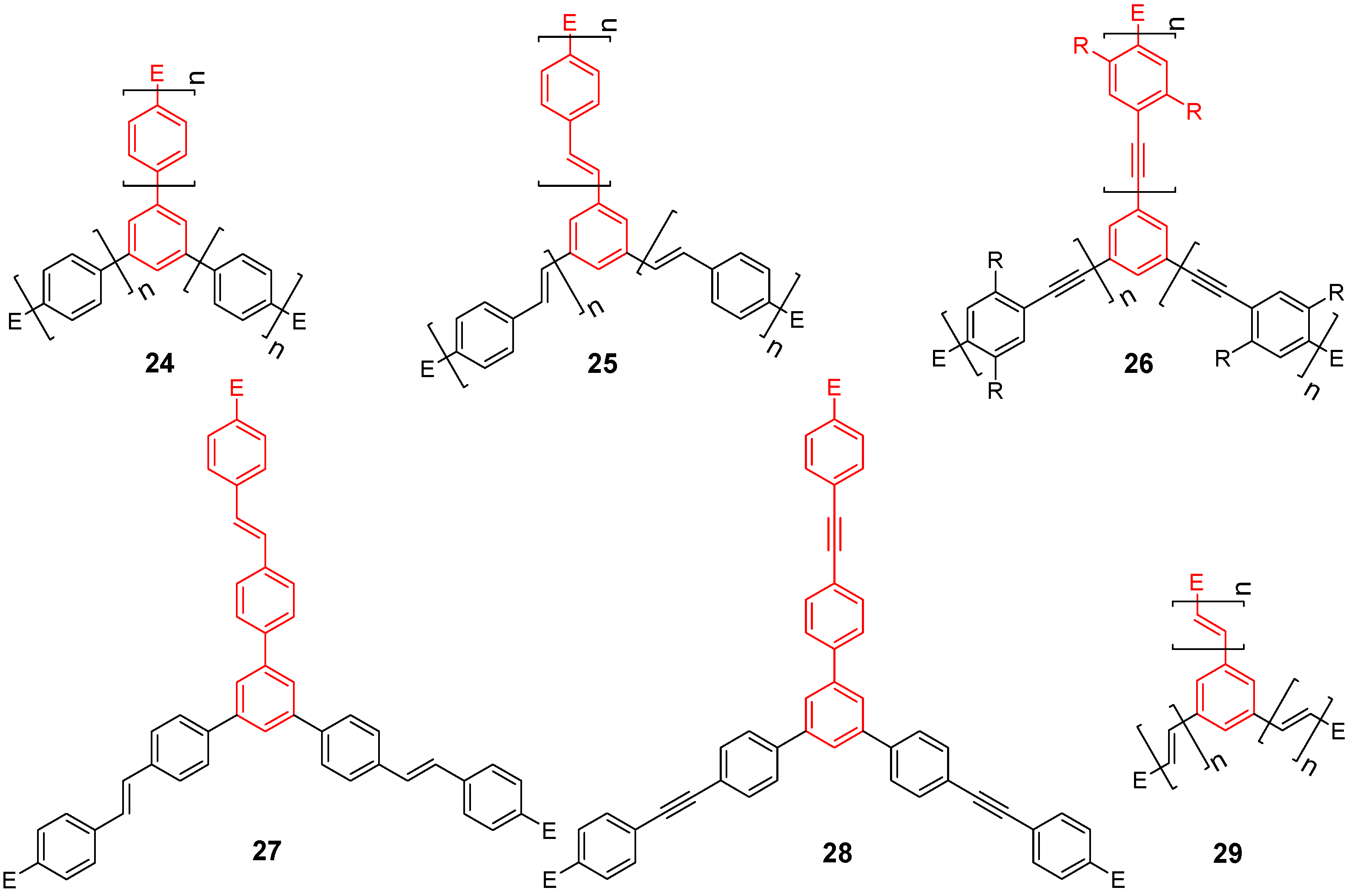
4. Star Compounds with a Benzene Core
4.1. Three-armed Stars (C-4)
4.1.1. Structure and Conjugation
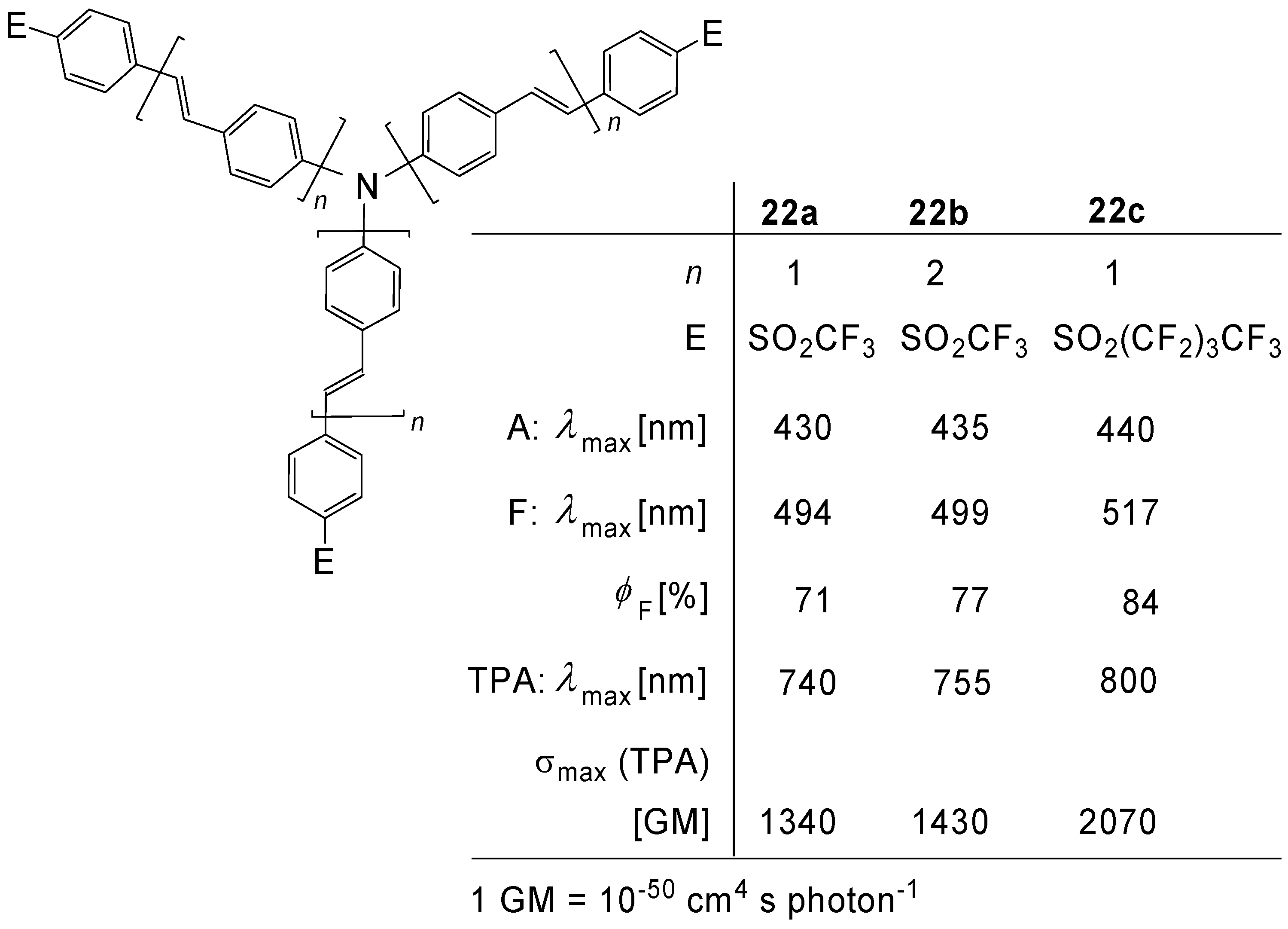
| Compounda | Substituents E, R Solvent, T (K) | Absorption λmax [nm] (ε [Lcm-1mol-1]) | Emission λmax [nm] (Quantum yield Ф) | Reference | |
| 24a | star | H (dioxane) | 250 (59,600) | 354 (0.10) | [62] |
| linear | H (dioxane) | 246 (18,600) | 316 (0.18) | [62] | |
| 25a | star | tristyryl (hexane, 293 K) | 317 (47,863) | 410 | [63] [64] |
| linear | H (methylpentanes, 295 K) EPA,c 77 K | 294 (26,000) 303 (51,500) | 347 343 | [65] [66] | |
| 26a | star | H CHCl3, 295 K | 305 (85,100) | 353 (0.15) | [67] |
| linear | H CHCl3 EPA,c 77 K | 300 (24,000) 284 (42,900) 302 (44,500) | — 326 | [66] | |
| 27a | star | H DMF | 337 (142,000) | — | [70] |
| linear | H DMF | 328 (45,200) | — | [70] | |
| 28a | star | H acetonitril | 308 (158,000) | 365 (0.92) | [68] |
| linear | H EPA,c 77 K | 307 (55,000) 325 (51,400) | 352 | [66] | |
| 24b n = 2 | star | H dioxane | 288 (103,000) | 364 (0.27) | [62] |
| linear | H dioxane | 276 (30,200) | 342 (0.55) | [62] | |
| 24c n = 3 | star | H dioxane | 307 (136,000) | 375 (0.71) | [62] |
| linear | H dioxane | 294 (48,600) | 369 (0.71) | [62] | |
| 25c n = 0 | star | Trisdodecyloxy-phenylethenyl CHCl3, rt | 331 (81,000) | 426b | [69] |
| 25d n = 1 | star | Trisdodecyloxy-phenylethenyl CHCl3, rt | 376 (170,000) | 447b | [69] |
| linear | Trisdodecyloxy-phenylethenyl,R = Methyl | 366 (64,000) | [70] | ||
| 25e n = 2 | star | Trisdodecyloxy-phenylethenyl CHCl3, rt | 397 (313,000) | - | [69] |
| linear | Trisdodecyloxy-phenylethenyl, R = CH2OH CH2Cl2, 298 K | 390 (67,000) | 468 nm | [71] | |
| 26b n = 1 | star | H, OCH3 CHCl3, 295 K | 334 (64,600) | 384 (0.46) | [67] |
| 26c n = 2 | star | H, OCH3 CHCl3, 295 K | 380 (128,800) | 409 (0.85) | [67] |
| 26d n = 3 | star | H, OCH3 CHCl3, 295 K | 426 (195,000) | 464 (0.98) | [67] |
| linear | H, OCH3 CHCl3, 295 K | 390 (56,200) | 430 (0.81) | [67] | |
| 26e n = 0 | star | 2-methoxyphenylethynyl, OCH3 CHCl3, 295 K | 314 (58,900) | 359 (0.24) | [67] |
| 26f n = 1 | star | 2-methoxyphenylethynyl, OCH3 CHCl3, 295 K | 377 (104,700) | 406 (0.83) | [67] |
| 26g n = 2 | star | 2-methoxyphenylethynyl, OCH3 CHCl3, 295 K | 405 (128,800) | 433 (0.97) | [67] |
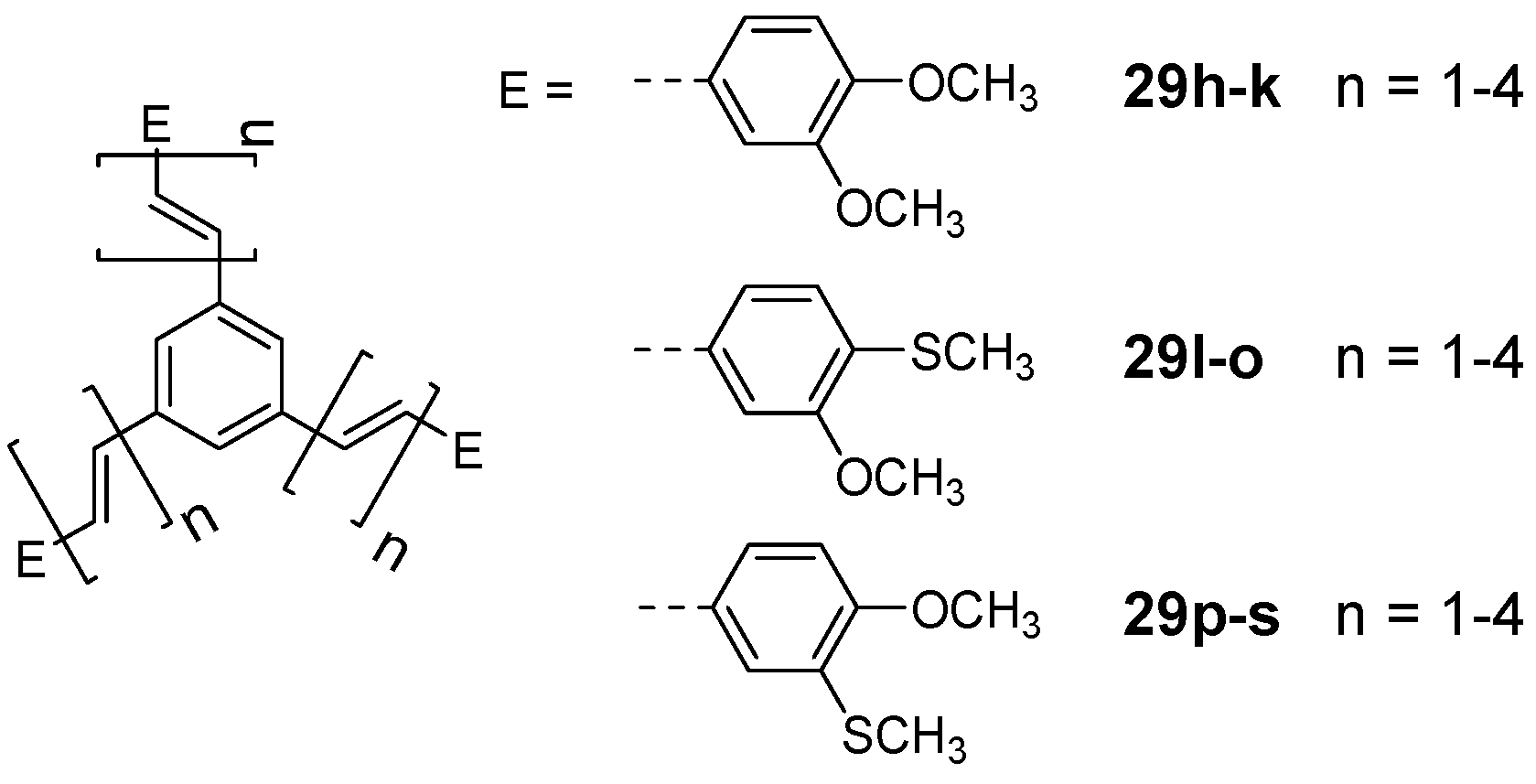
| 29a n = 1 | star | 3,4-dibutoxy-phenyl | 340 | [72] | |
| linear | 3,4-dibutoxy-phenyl | 325 | [72] | ||
| 29b n = 2 | star | 3,4-dibutoxy-phenyl | 360 | [72] | |
| linear | 3,4-dibutoxy-phenyl | 350 | [72] | ||
| 29c n = 3 | star | 3,4-dibutoxy-phenyl | 380 | [72] | |
| linear | 3,4-dibutoxy-phenyl | 375 | [72] | ||
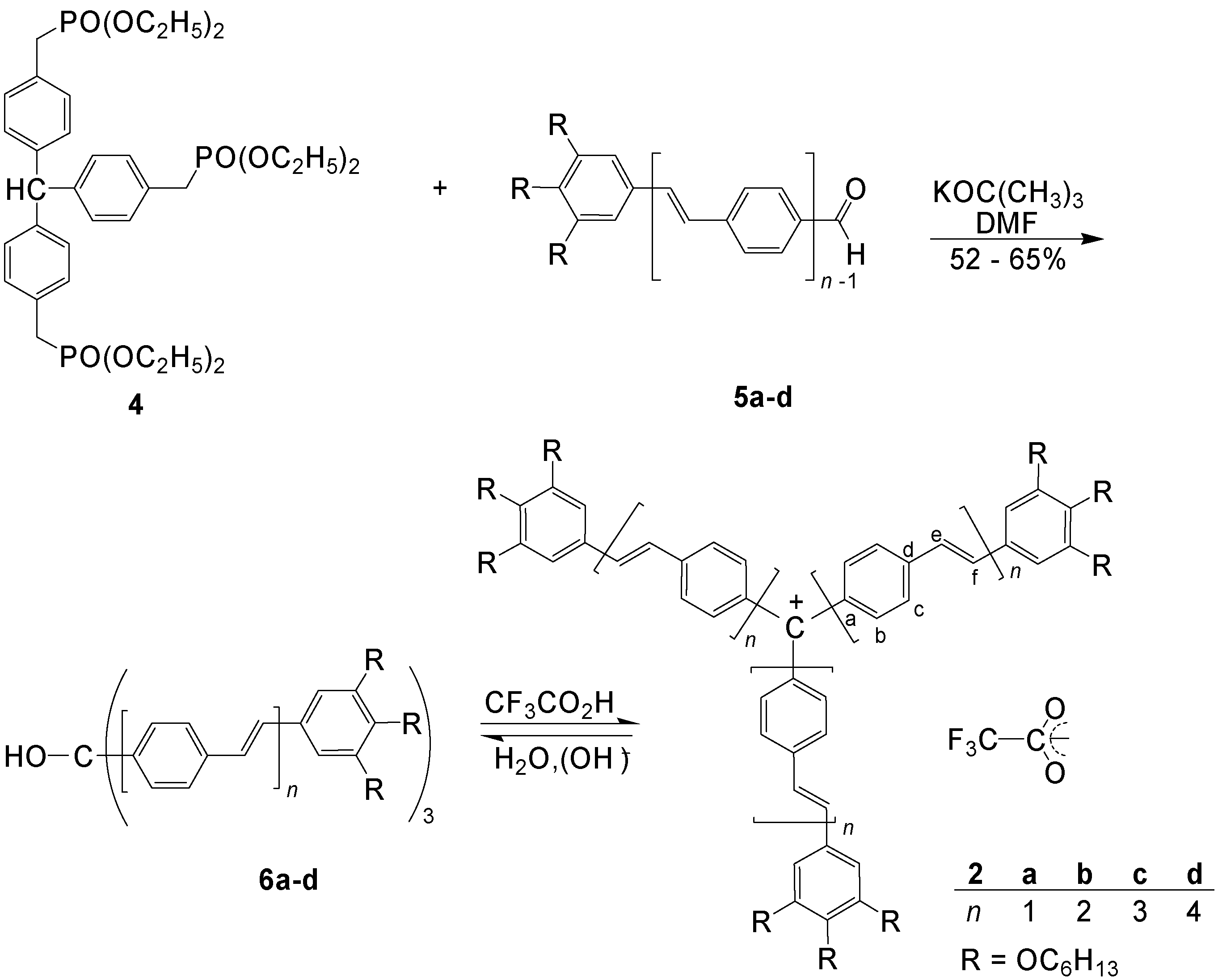
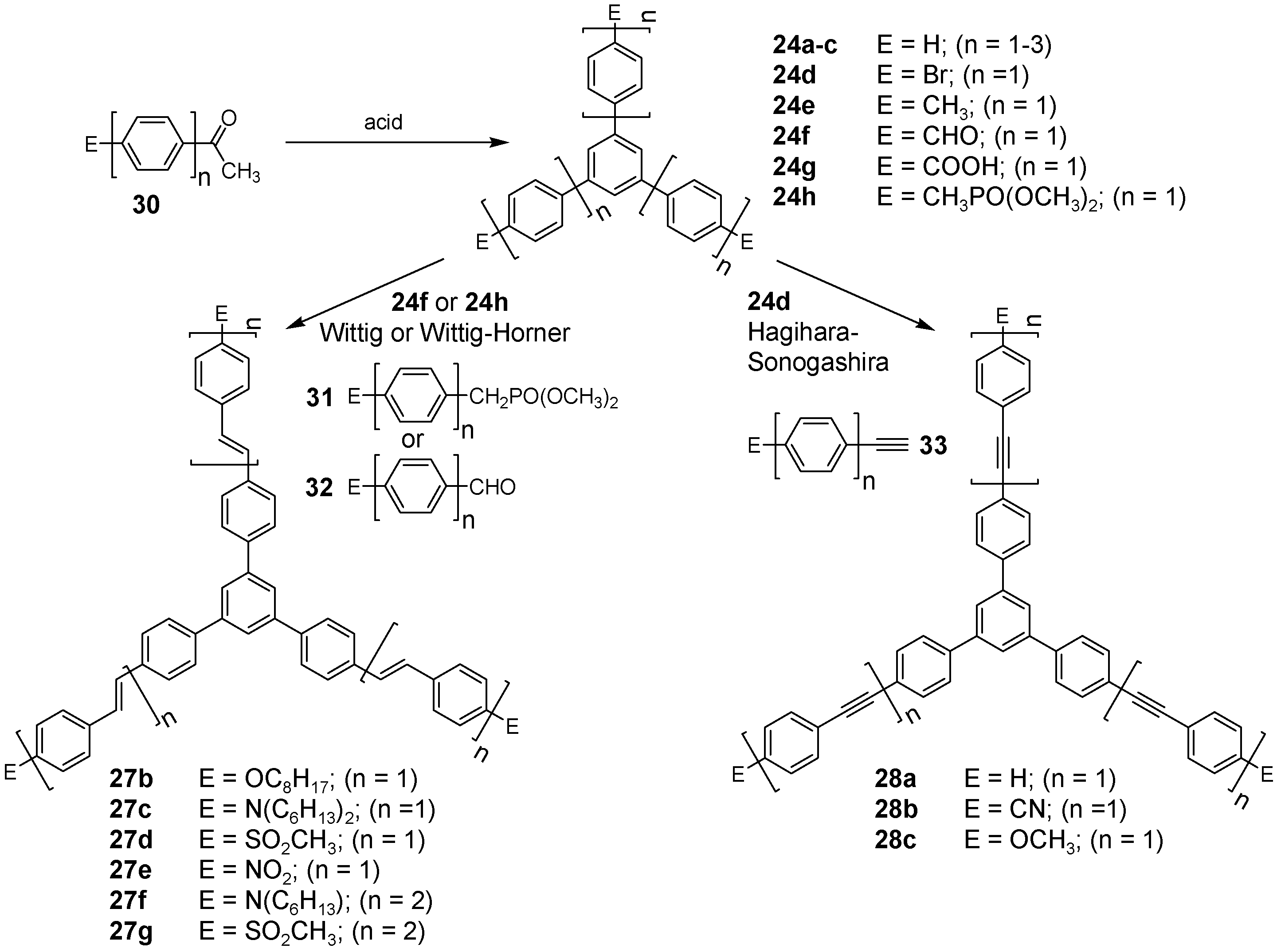
4.1.2. Synthesis
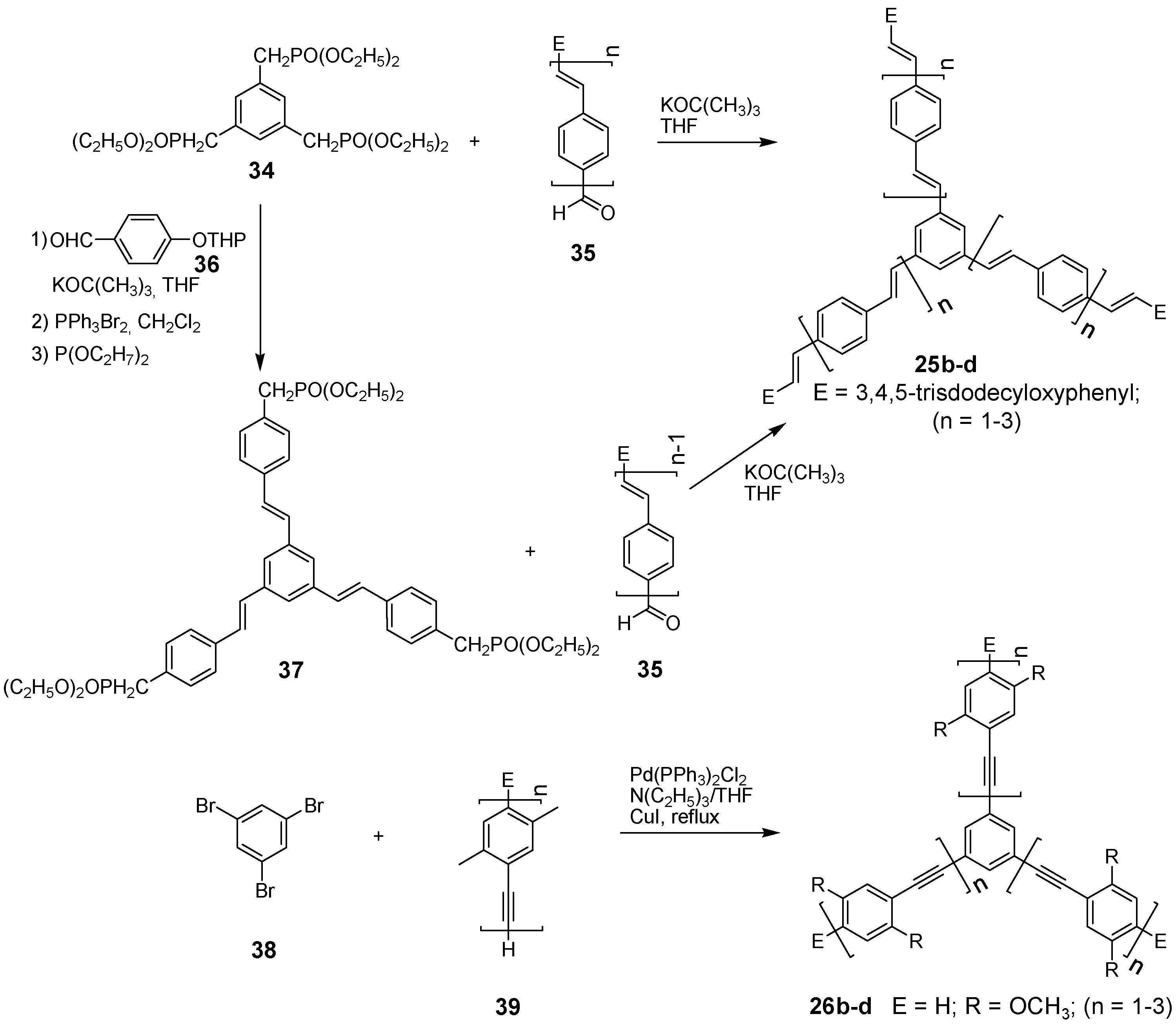
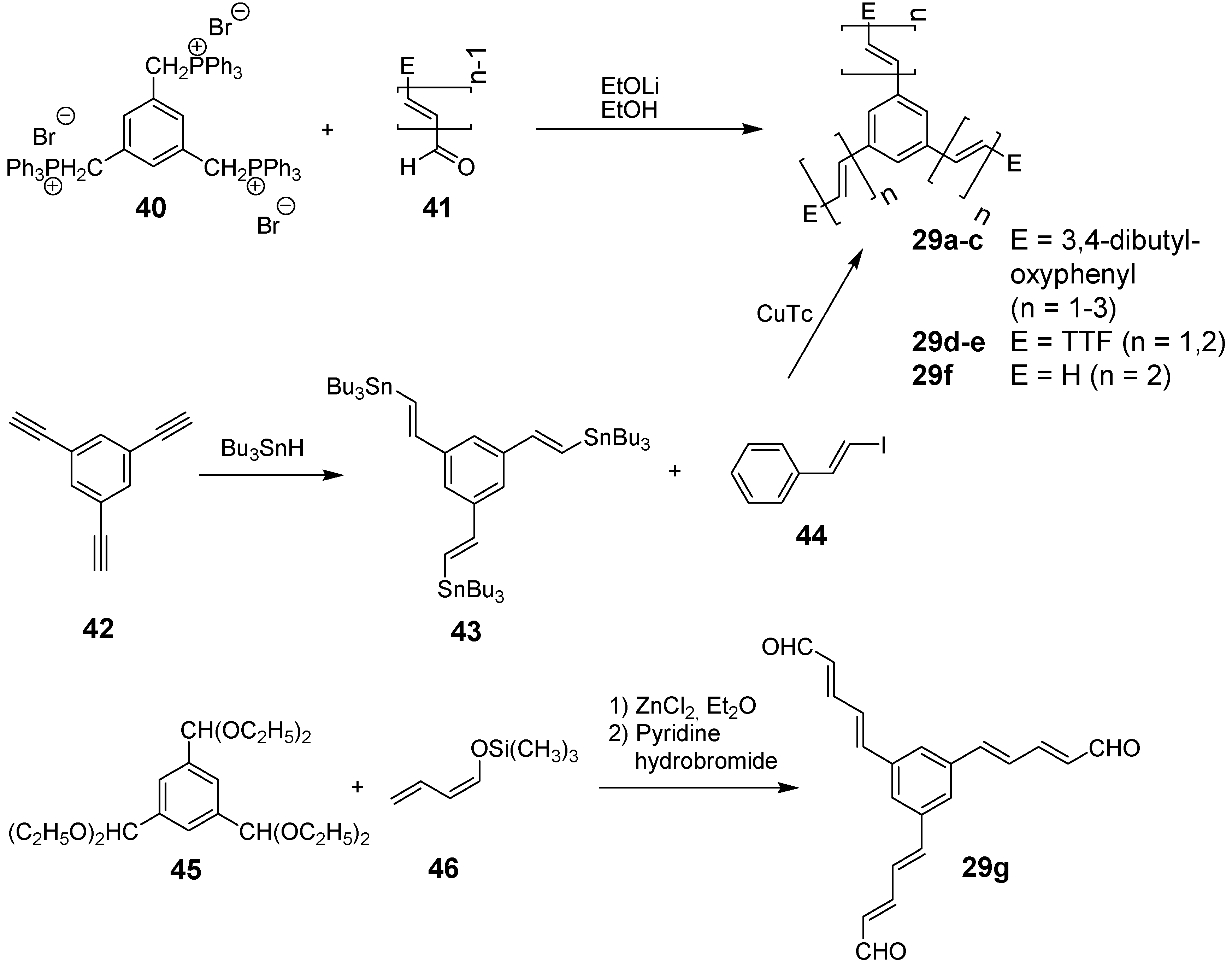
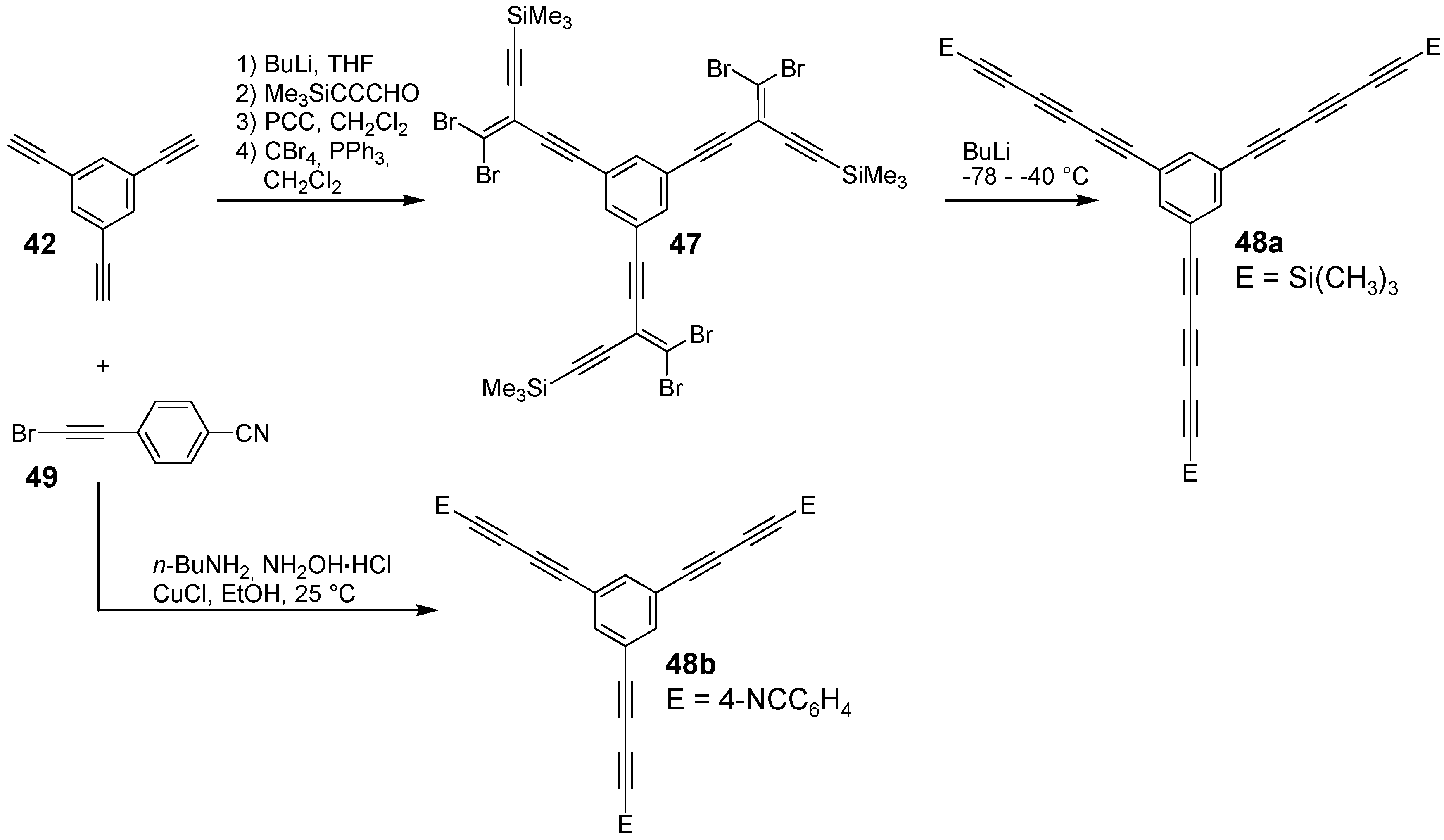
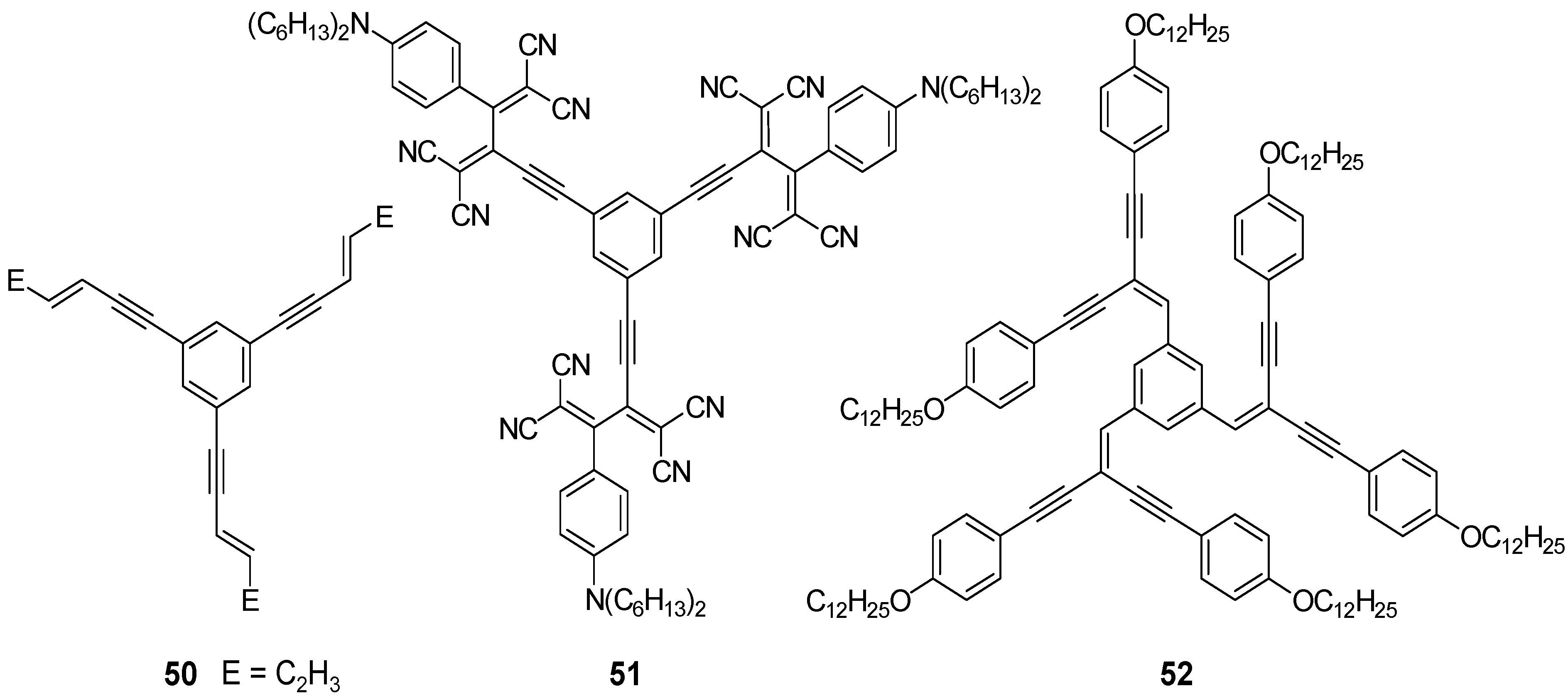
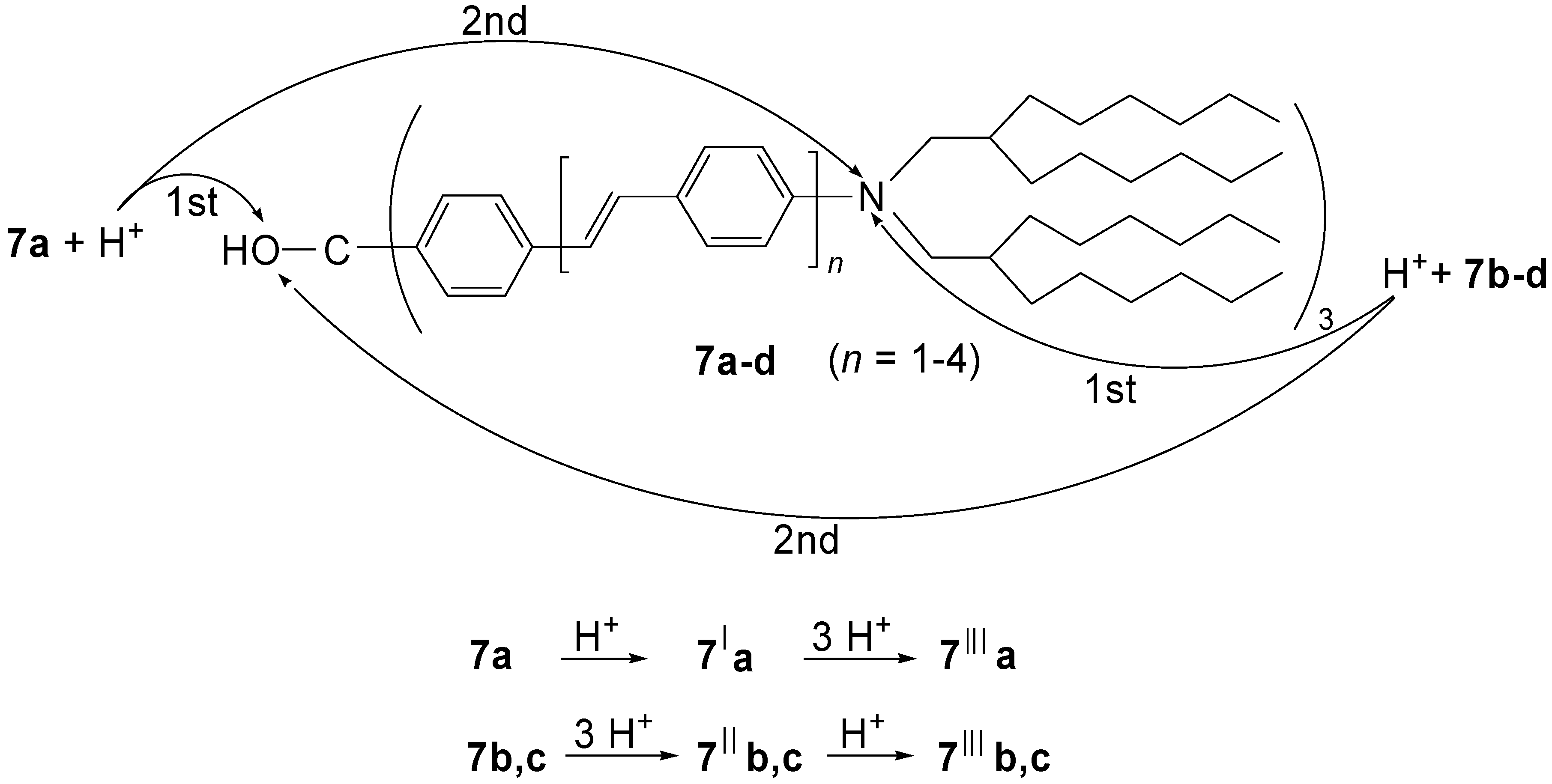
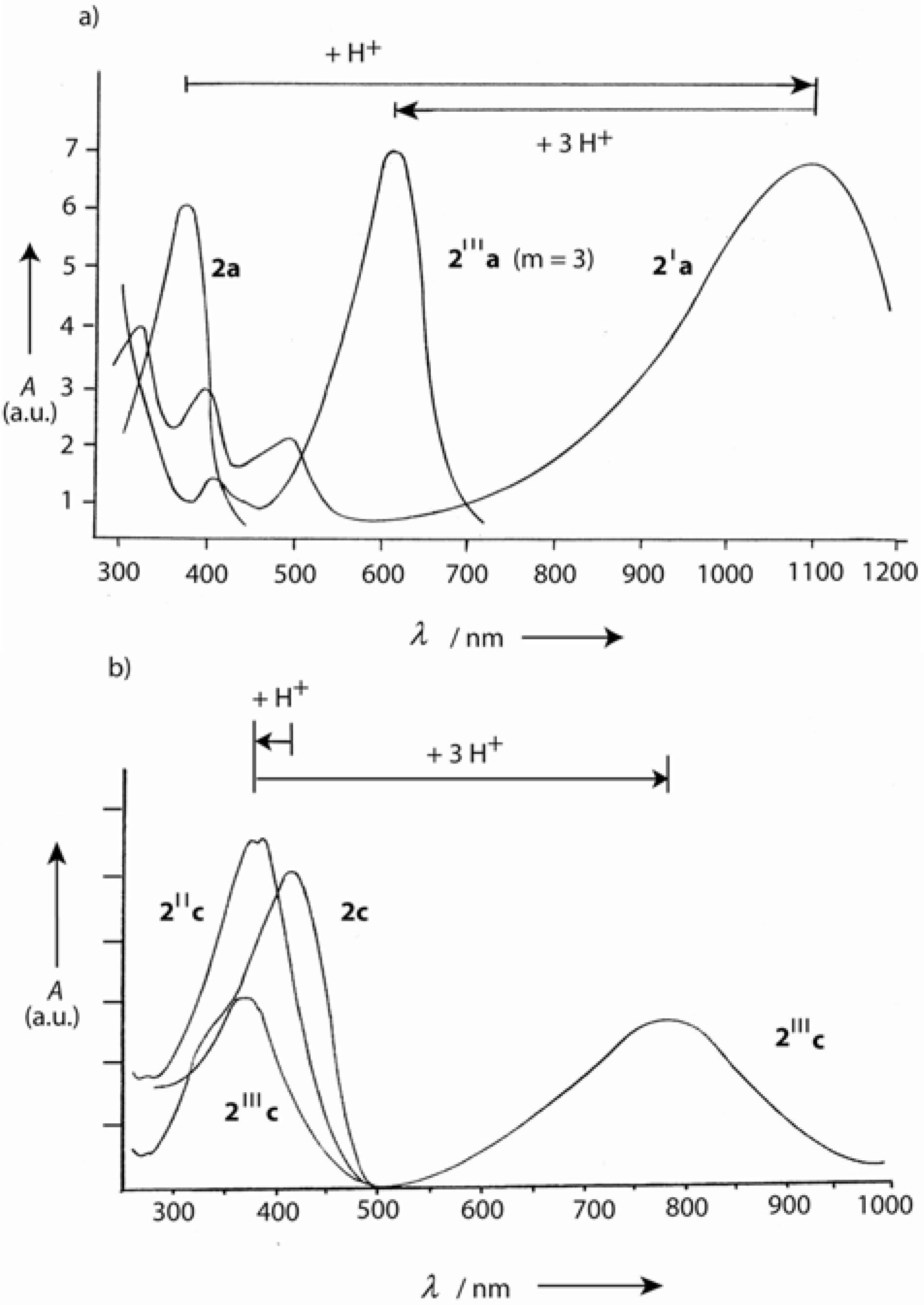
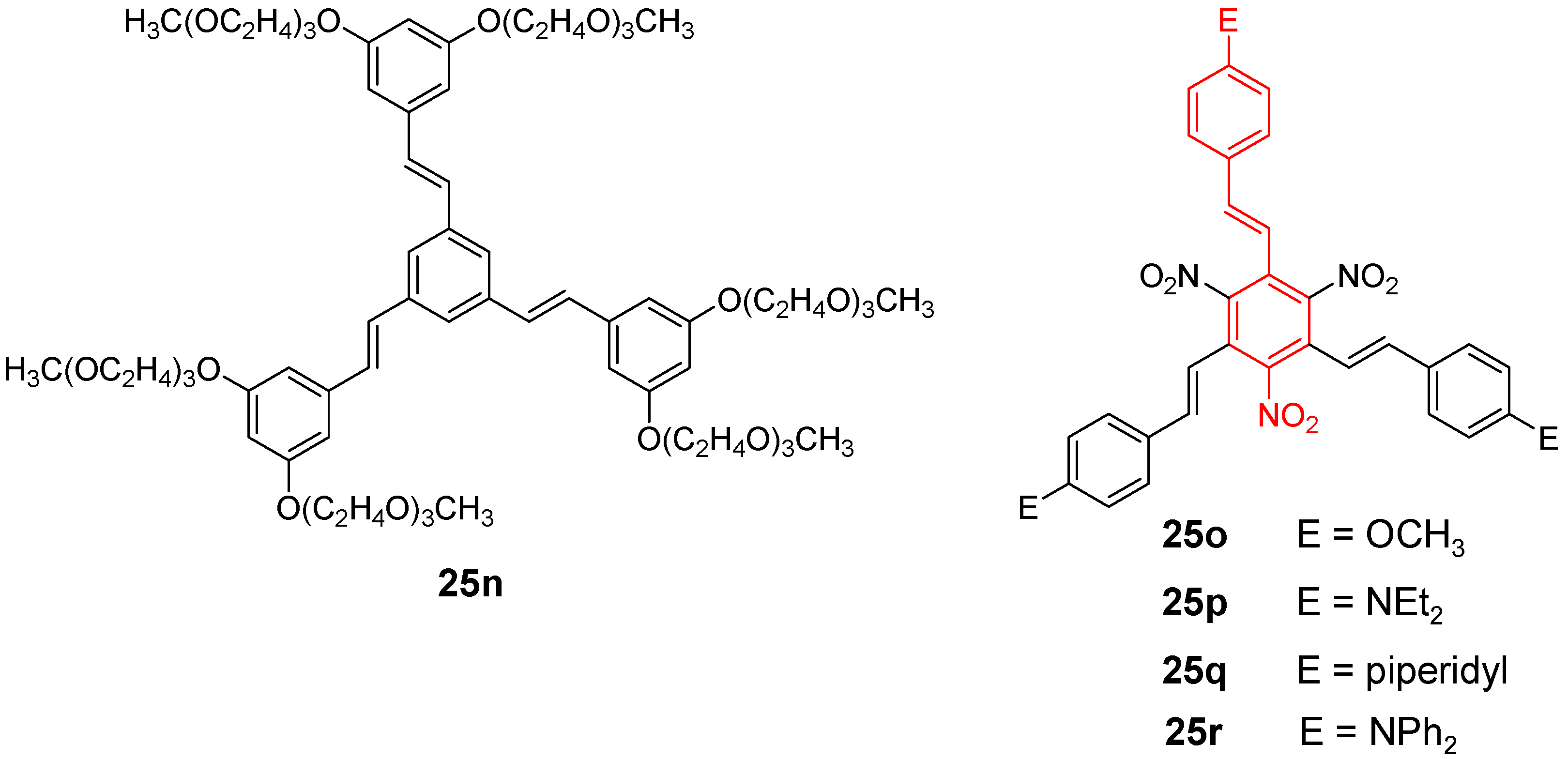
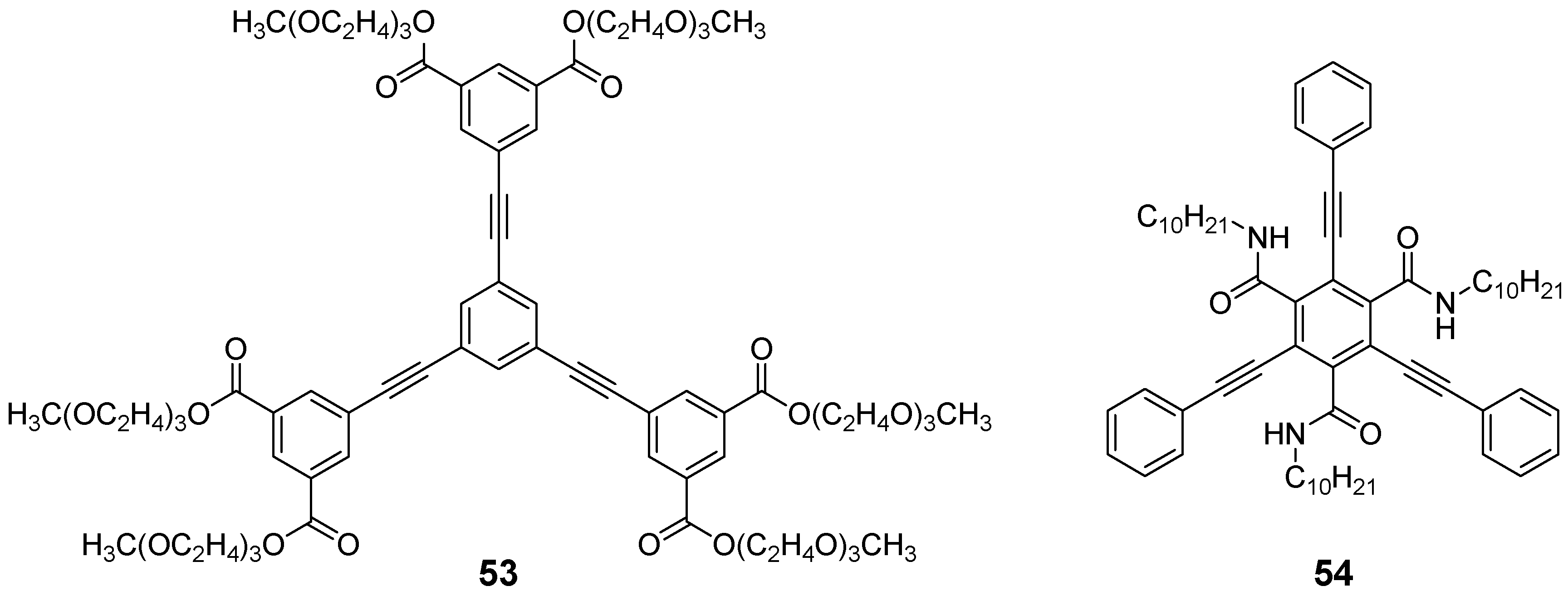
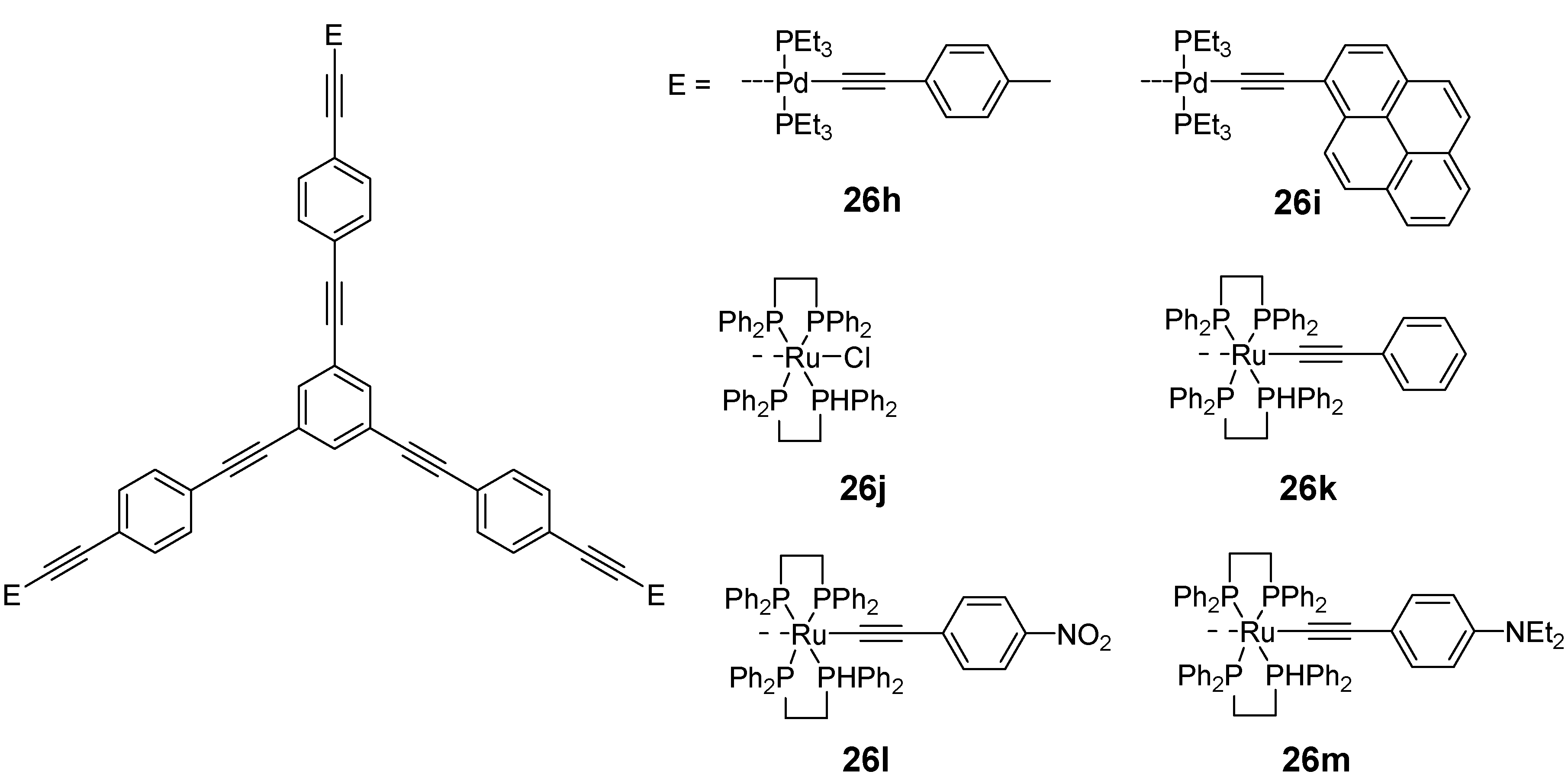
4.1.3. Three-arm Stars with Benzene Centers and Materials Science
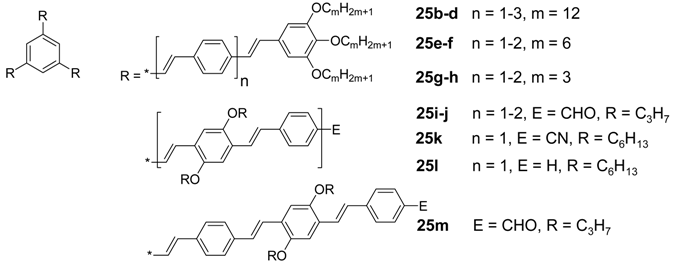 | ||
| Compound | Thermotropic behavior (T [°C] / ΔH [kJ/mol]a | Ref. |
| 25b (n = 1) | Cr 38/39 Colhd 75/10 I | [88,111] |
| 25c (n = 2) | g 21 (Tg) Colhd 108/4 I | [89] |
| 25d (n = 3) | g 55 (Tg) Colhd 199/3 I | [69] |
| 25e (n = 1) | g -15 (Tg) Colhd 74/4 I | [111] |
| 25f (n = 2) | g 21 (Tg) LD 129/3 I | [89] |
| 25g (n = 1) | Cr 189/45 I | [111] |
| 25h (n = 2) | Cr 216/40 I | [89] |
| 25i (n = 1) | g 140 (Tg) ND 260 Ib | [113] |
| 25j (n = 2) | g 246 (Tg) ND 296 Ib | [113] |
| 25k (n = 1) | Cr 209/40 ND 232/1 I | [112] |
| 25l (n = 1) | g 2 (Tg) ND 114/0.2 ND’ 126/0.4 I | [112] |
| 25m | g 140 (Tg) N 226 Ib | [113] |

4.2. Four-arm Systems-Tetrasubstituted Benzene (C-5)
4.2.1. Structure and Conjugation
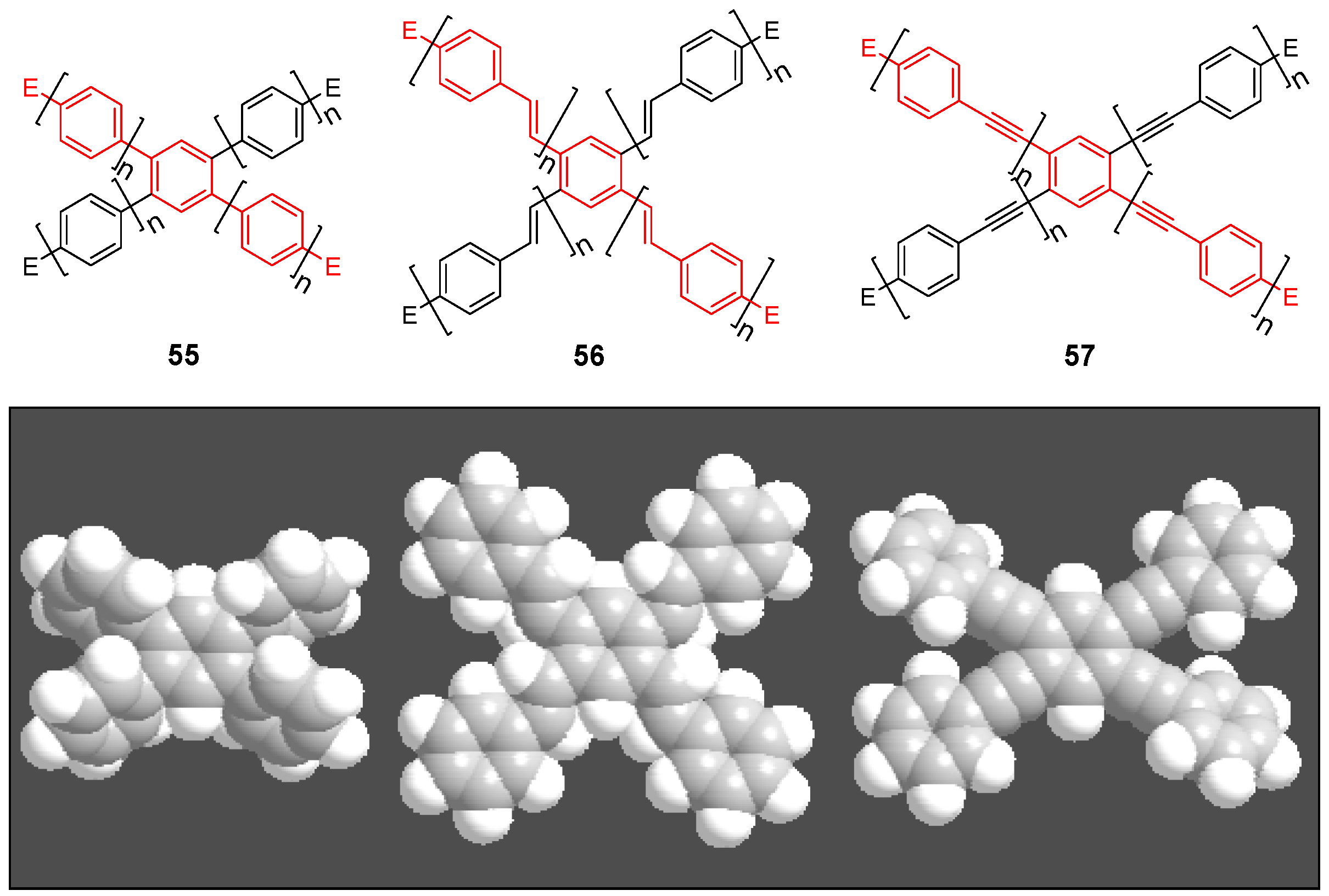
| Compounda | Substituents E, R Solvent, T (K) | Absorption λmax [nm] (ε [Lcm-1mol-1]) | Emission λmax [nm] (Quantum yield Ф) | Ref. | |
| 55b (n = 2) | star | H (CHCl3) | 276 (-) | 410 | [158] |
| linear | H (CHCl3) | 309 (log4,8) | 386 (0.90) | [159] | |
| 56a (n = 1) | star | H (Toluene, 293 K) (Toluene 77K) | 337/370 (S) 350/365(S)/375(S) | 450/472 442/463 | [64] |
| linear | H (Hexane, rt) | 350 | 417 | [160] | |
| 57a | star | H CHCl3 | 315(134,900)/350 | 391 (0.57) | [143] [159] |
| linear | H CHCl3 | 328 (38,900) | 348 (0.83) | [159] | |
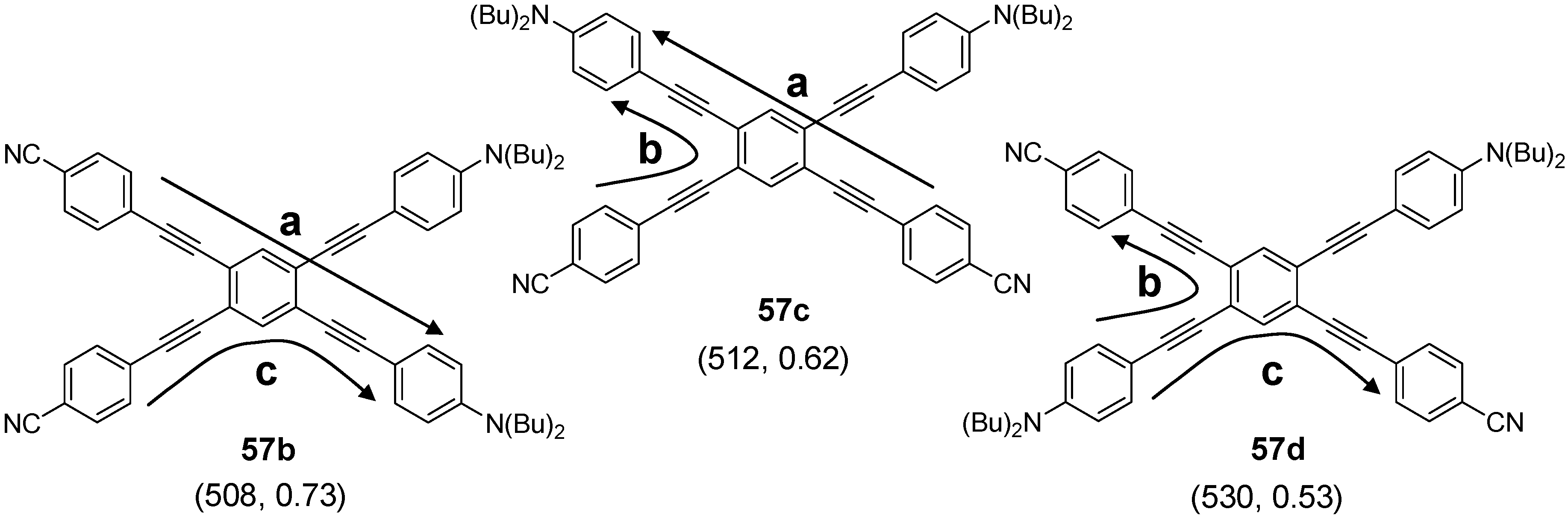
4.2.2. Synthesis
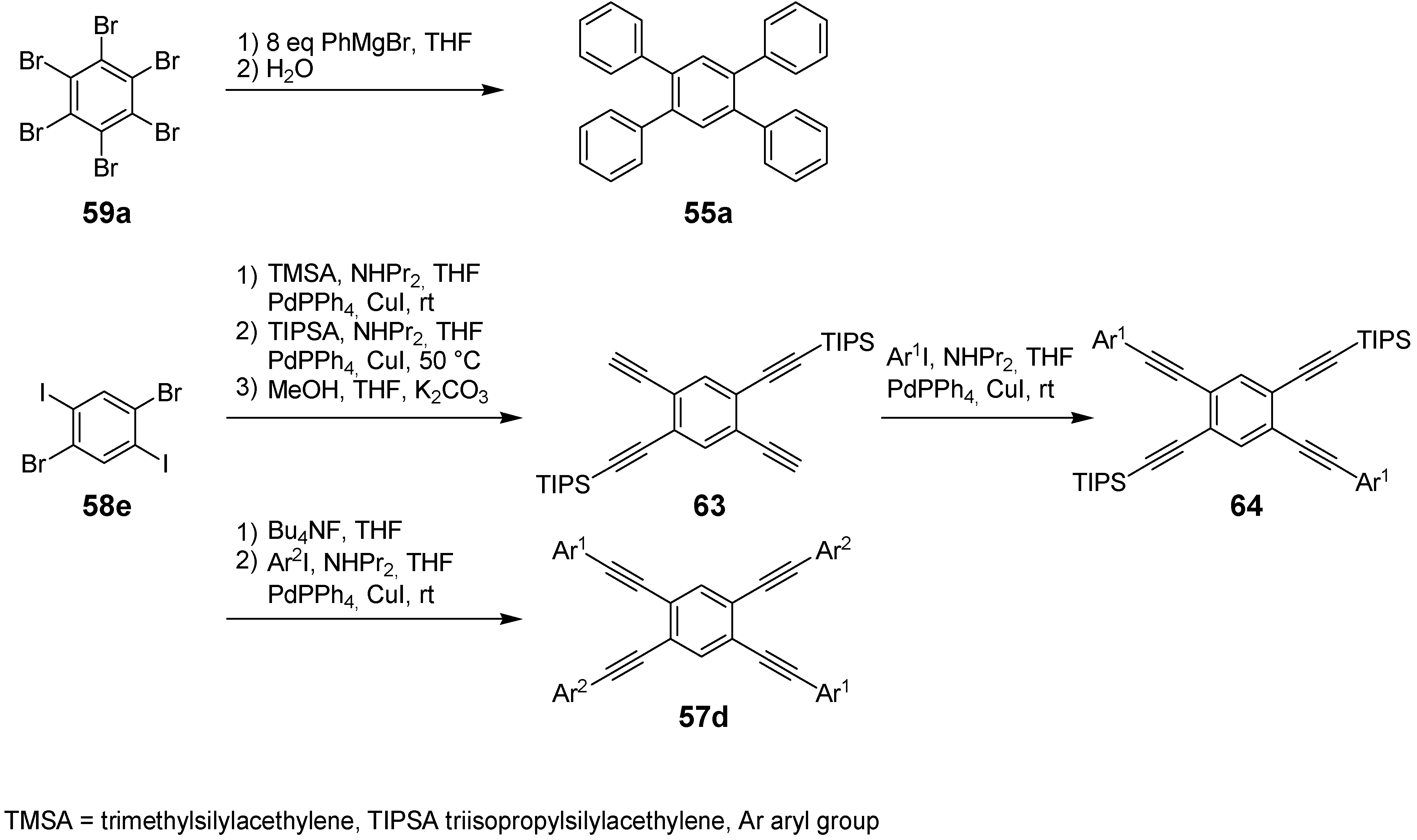
4.2.3. Four-arm Stars with Benzene Centers and Materials Science
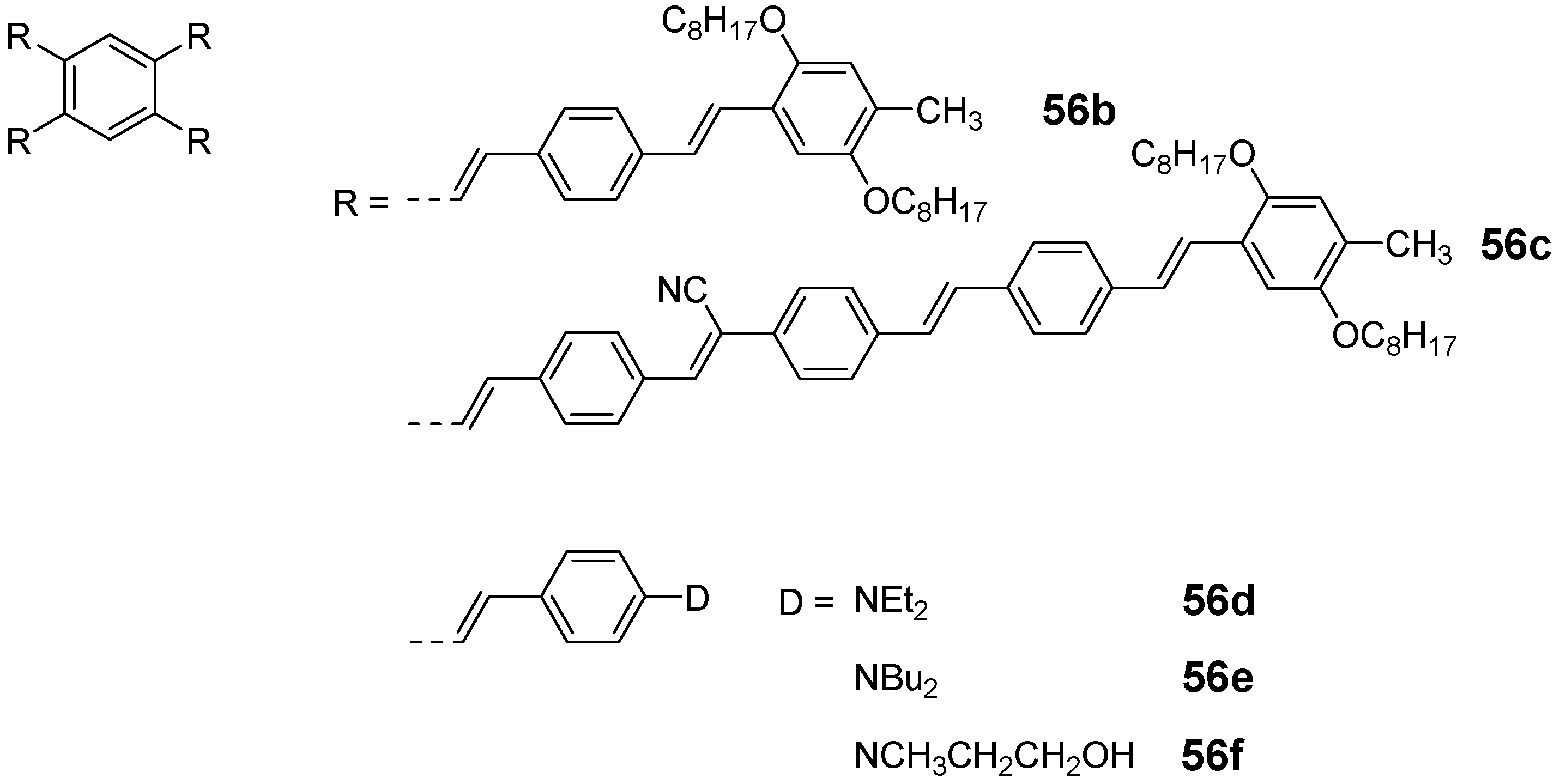
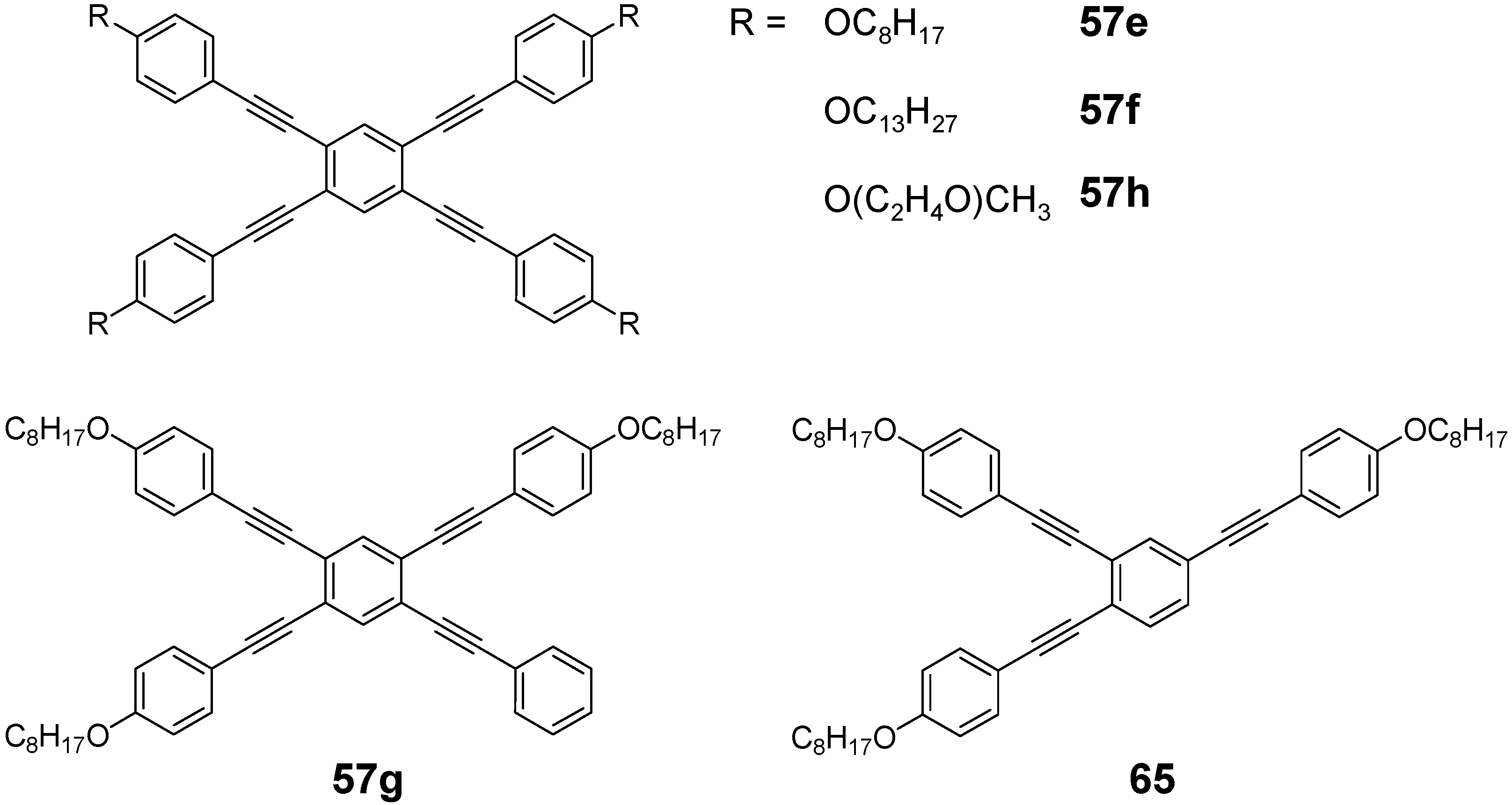
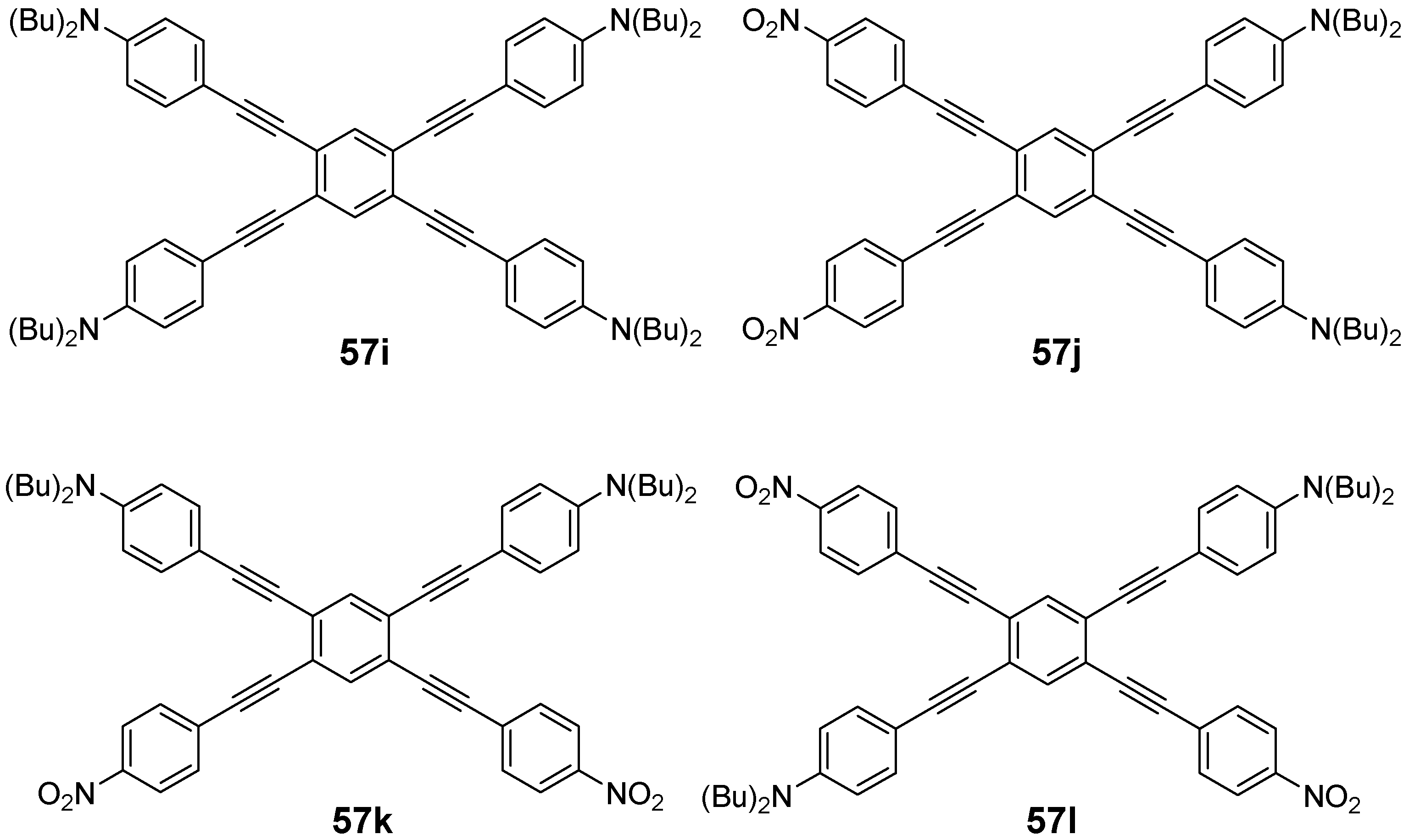
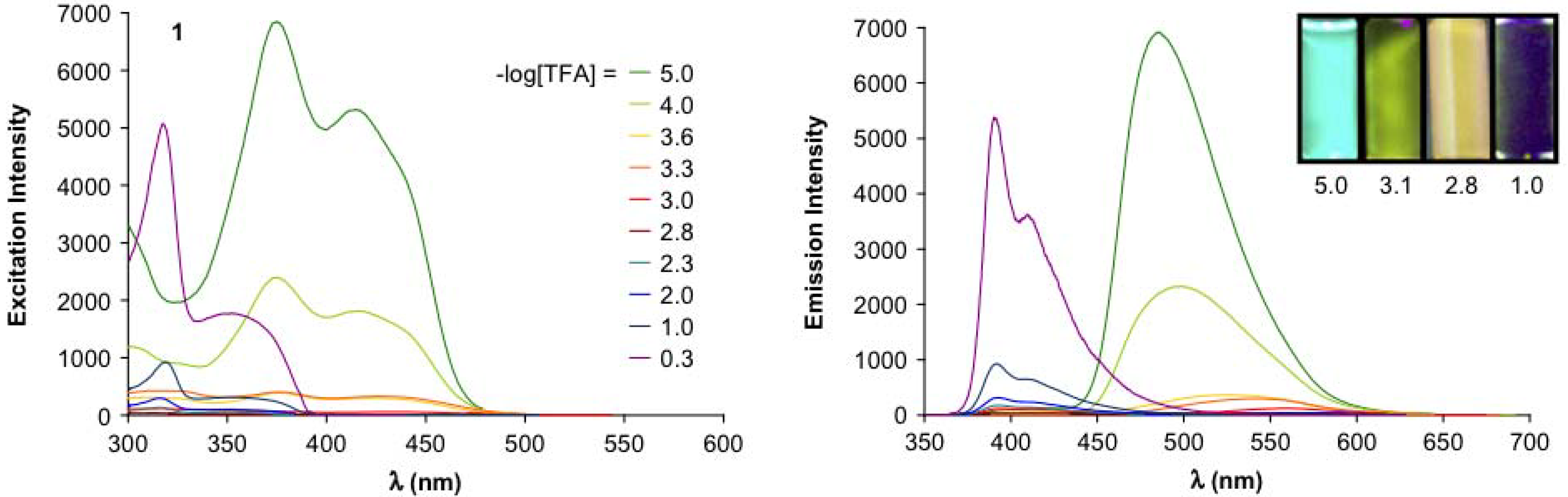
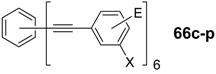
4.3. Six-arm Systems-Hexasubstituted Benzenes (C-6)
4.3.1. Structure and Conjugation of Parent Systems
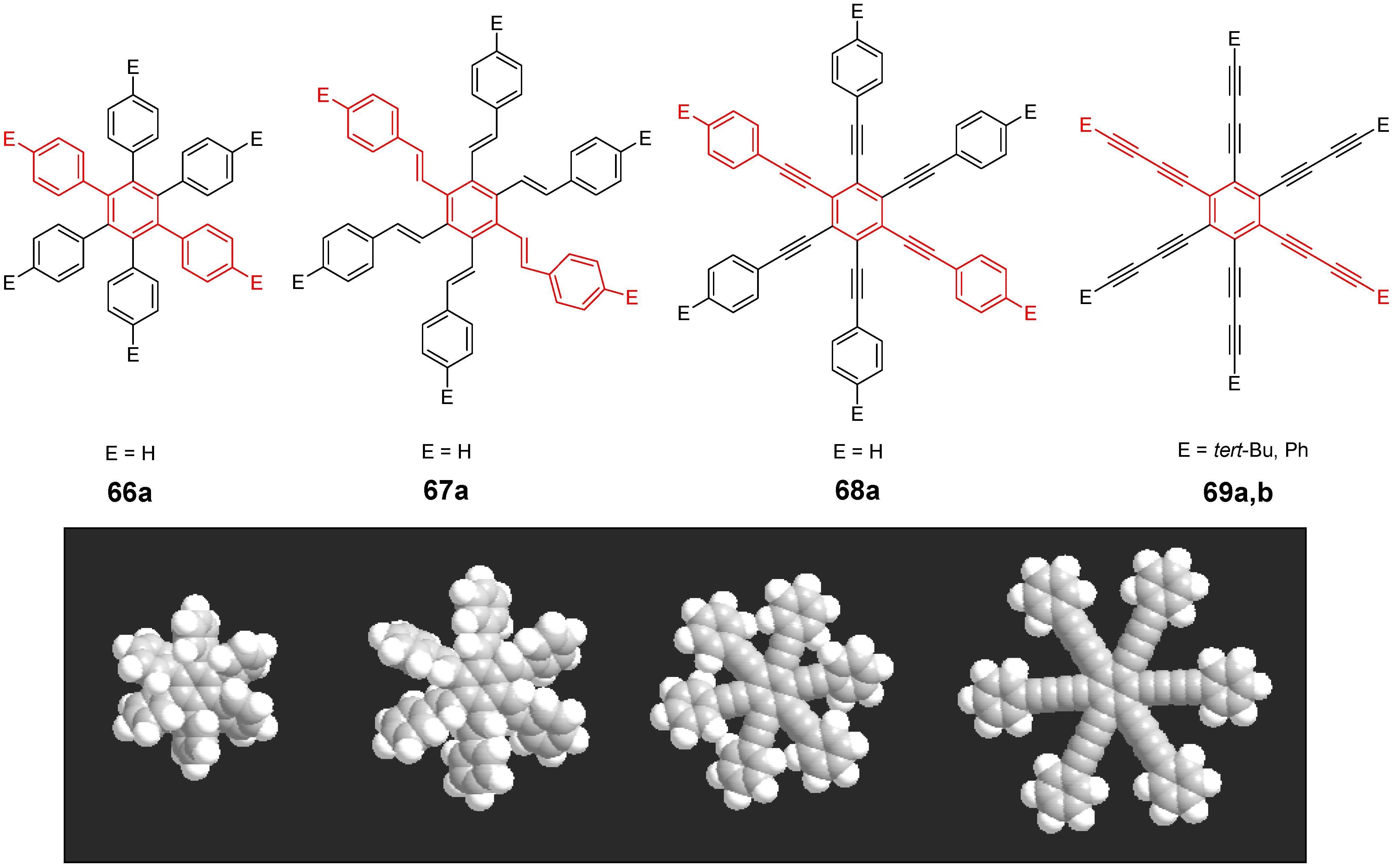
| Compounda | Substituents E | Absorption λmax [nm] | Emission λmax [nm] | References | |
| 66a | star | H | 249 | 337 | [197] |
| linear | H | 280 | 342 | [159] | |
| 67b | star | 4-dodecyloxy | 342 | - | [198] |
| linear | 369 | - | |||
| 68a | star | H | 349 | 449 | [143,159] |
| linear | H | 328 | 348 | [159] | |
| 69c | star | 4-tert-butylphenyl | 385/415 | - | [199] |
| linear | phenyl | 335/360 | - | [200] | |
4.3.2. Synthesis of the Parent Systems
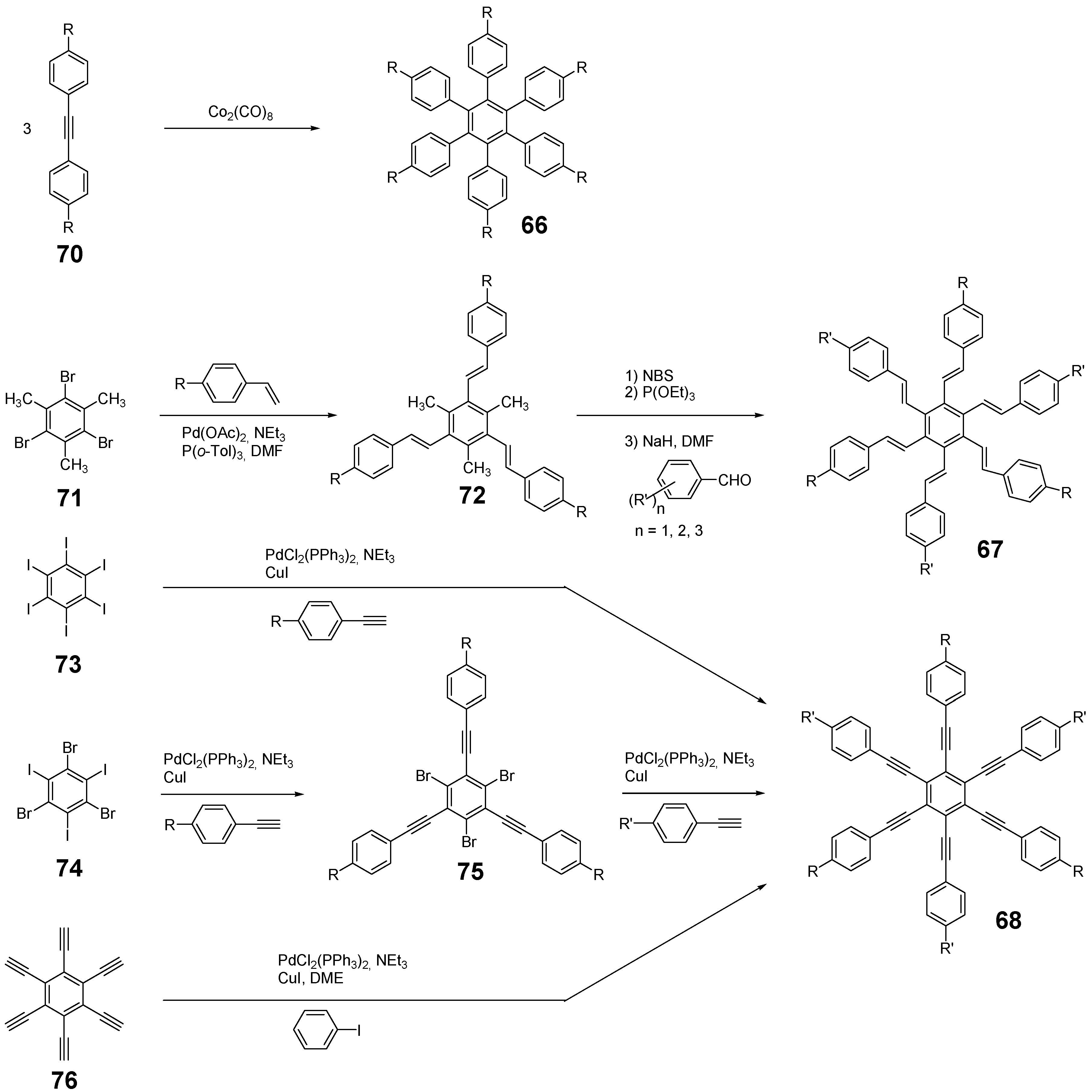
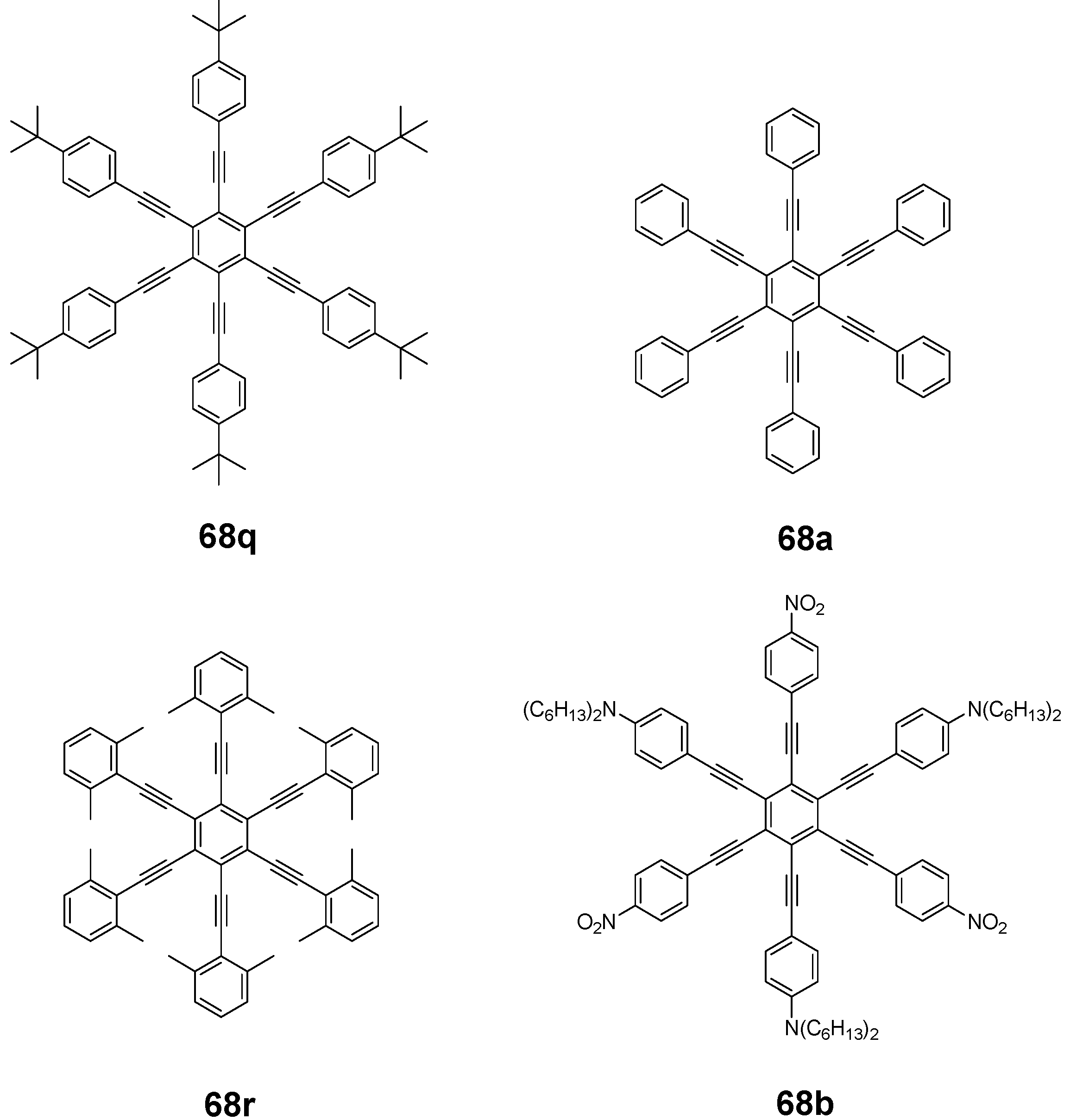
4.3.3. Hexaarm Stars with Benzene Centers and Materials Science
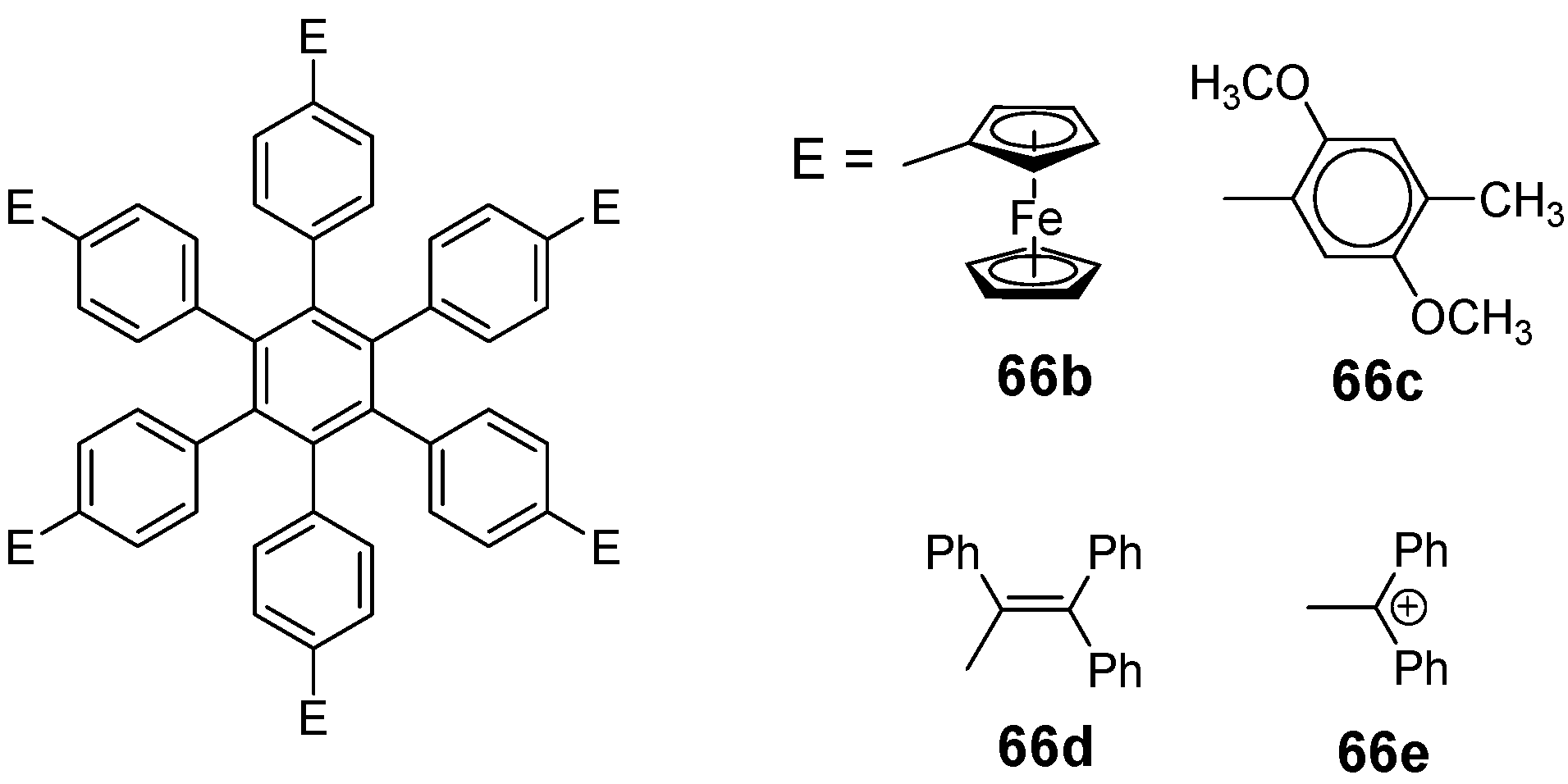

 | |||||
| Ea | X | Transition Temperatures /°Cb | Ea | X | Transition Temperatures /°Cb |
| n-pentyl | H | Cr 170 ND 185 I | hexyloxy | H | Cr 144 ND 216 I |
| n-hexyl | H | Cr 124 ND 142 I | heptyloxy | H | Cr 109 ND 193 I |
| n-heptyl | H | Cr 98 ND 131 I | meta hexyloxy | H | Cr 87 I |
| n-octyl | H | Cr 80 ND 96 I | ortho hexyloxy | H | Cr 63 I |
| n-nonyl | H | Cr 67 ND 83 I | octyloxy | CH3 | Cr 95 ND 176 I |
| n-decyl | H | Cr 71 (ND 54) I | 3,7-dimethyl-octyloxy | H | Cr 80 ND 124 I |
| n-dodecyl | H | no LC | 3,7-dimethyl-octyloxy | CH3 | Cr 71 ND 147-160 I |
5. Compounds with Heterocyclic Cores
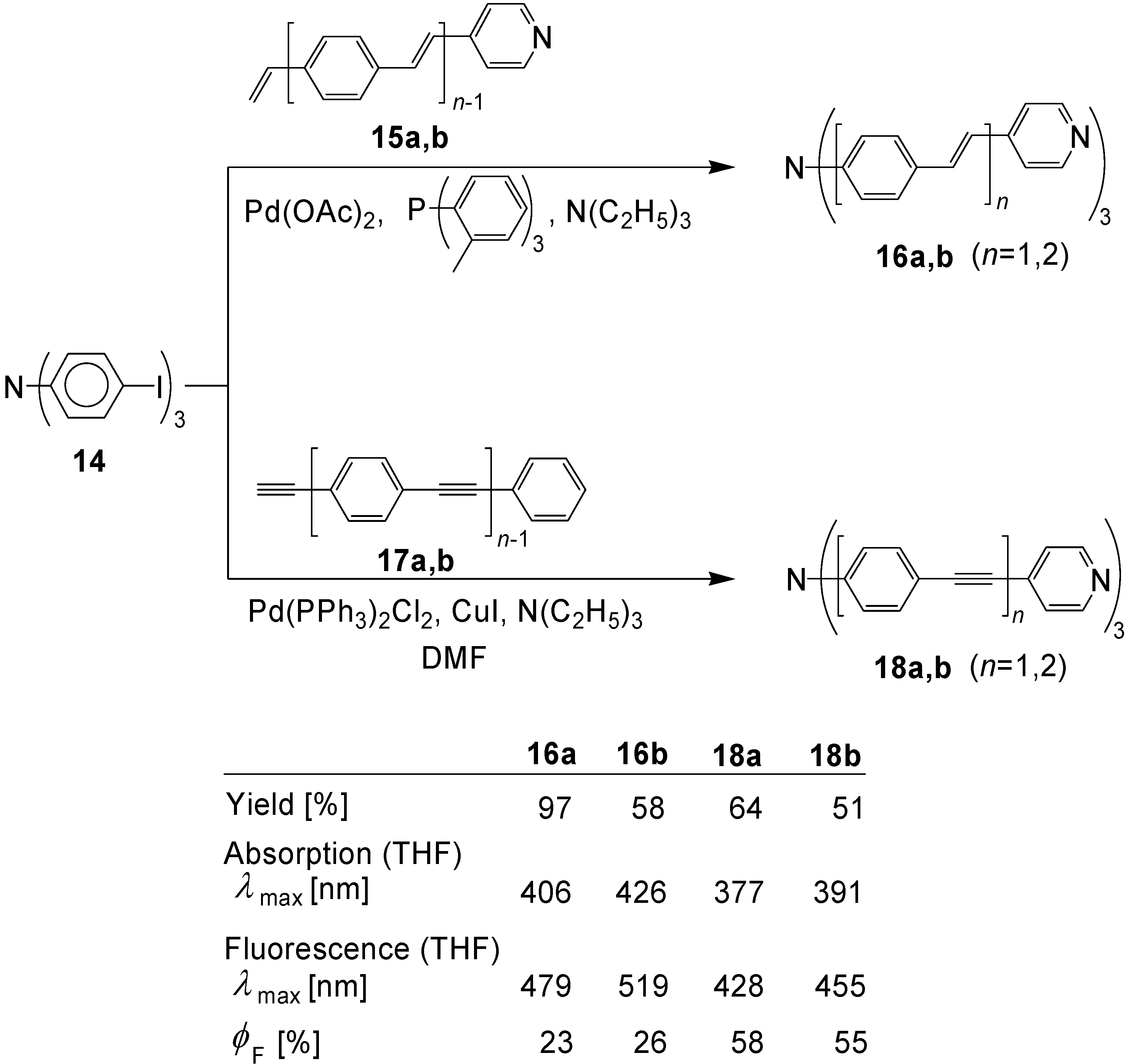
5.1. Pyridine-based Stars (C-7-A-6, C-7-A-7):
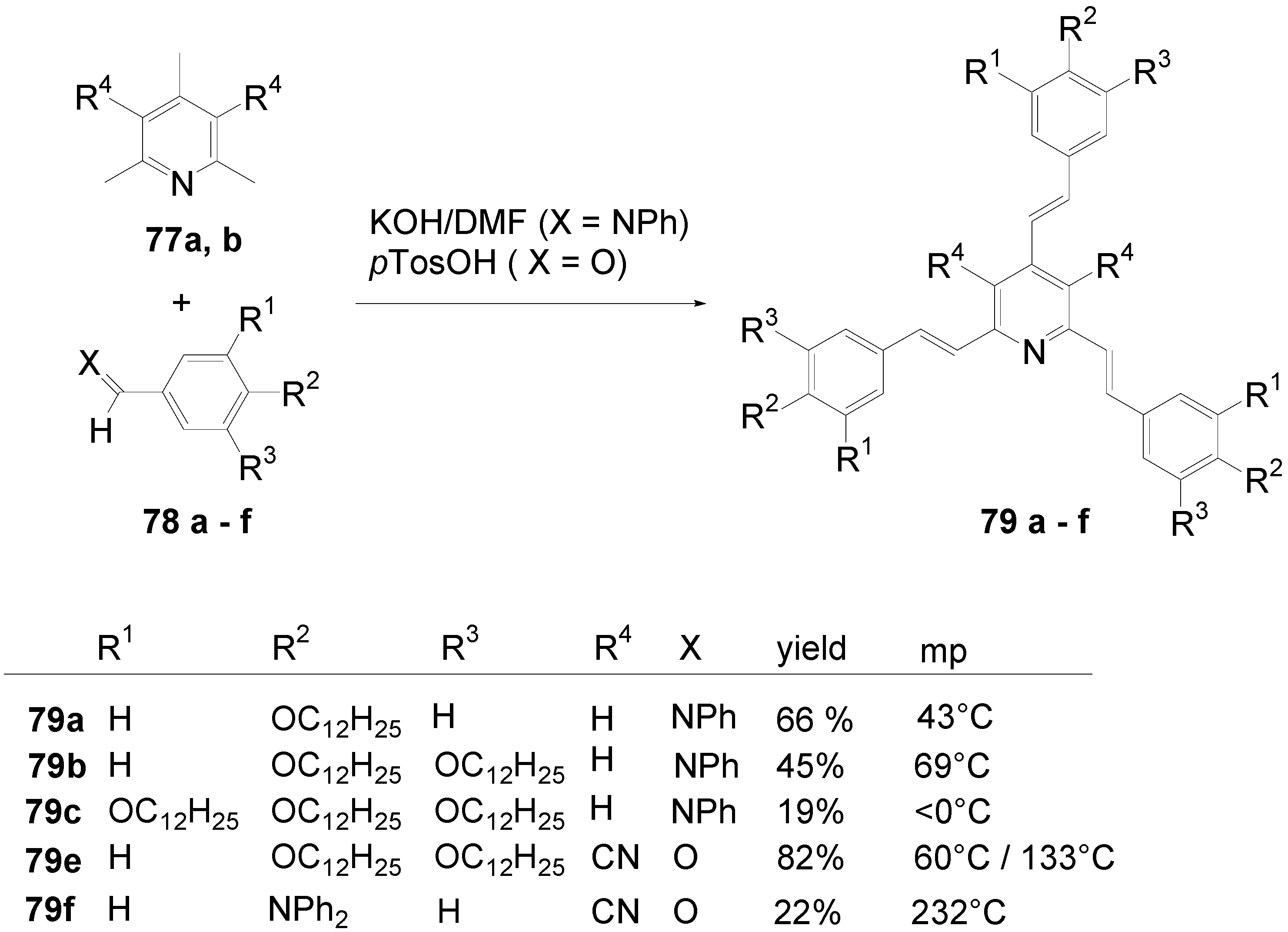
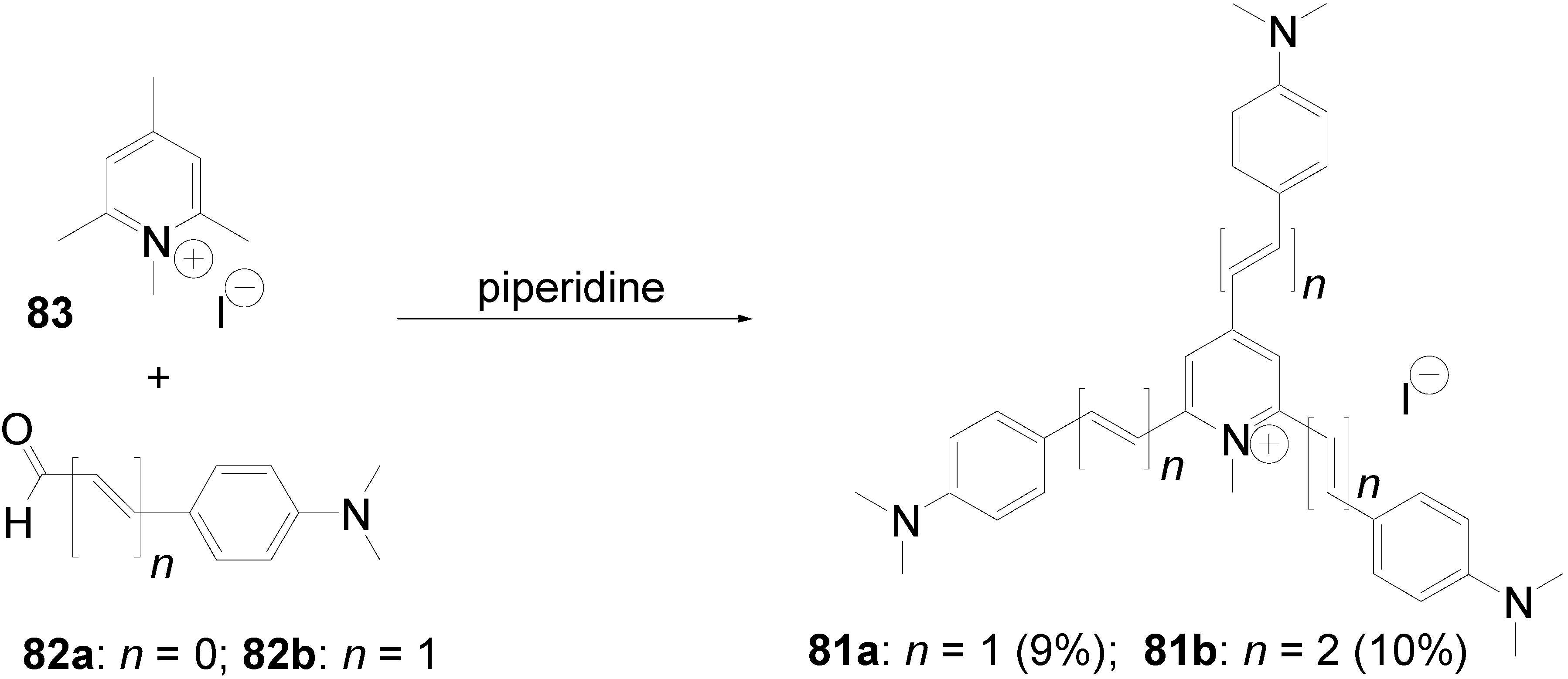
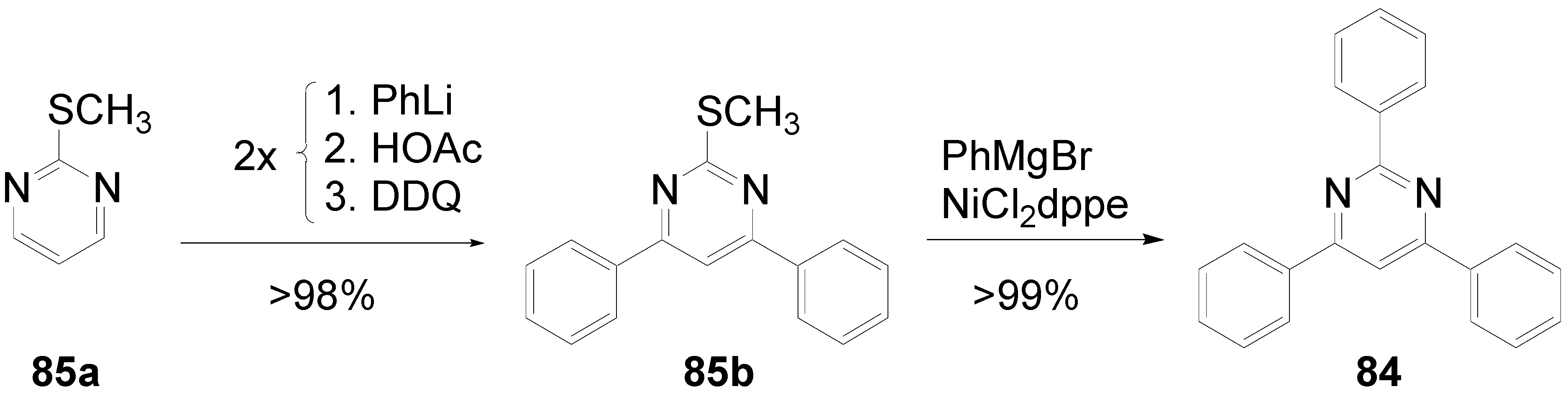
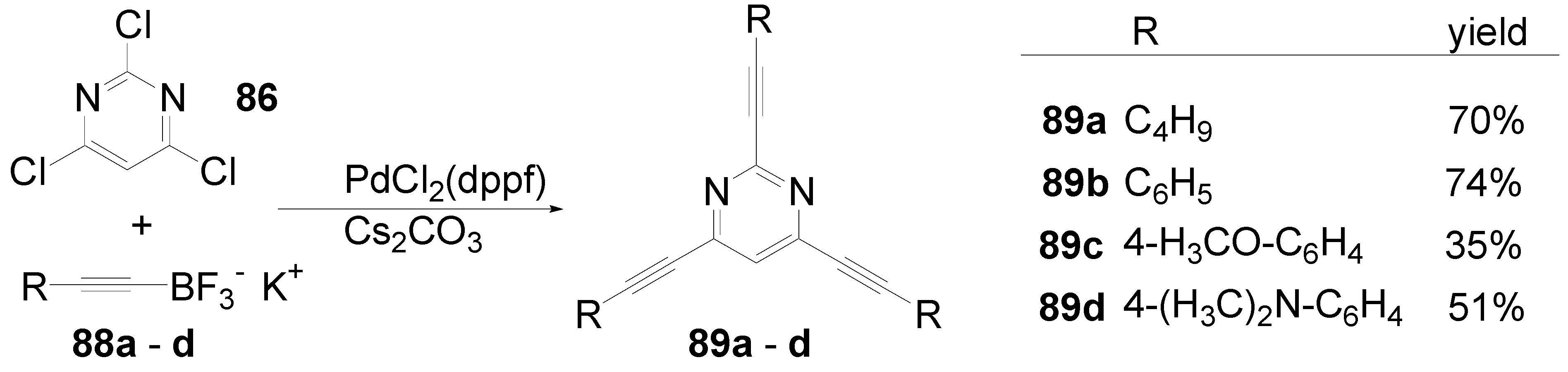
5.2. Stars with a Pyrimidine Core

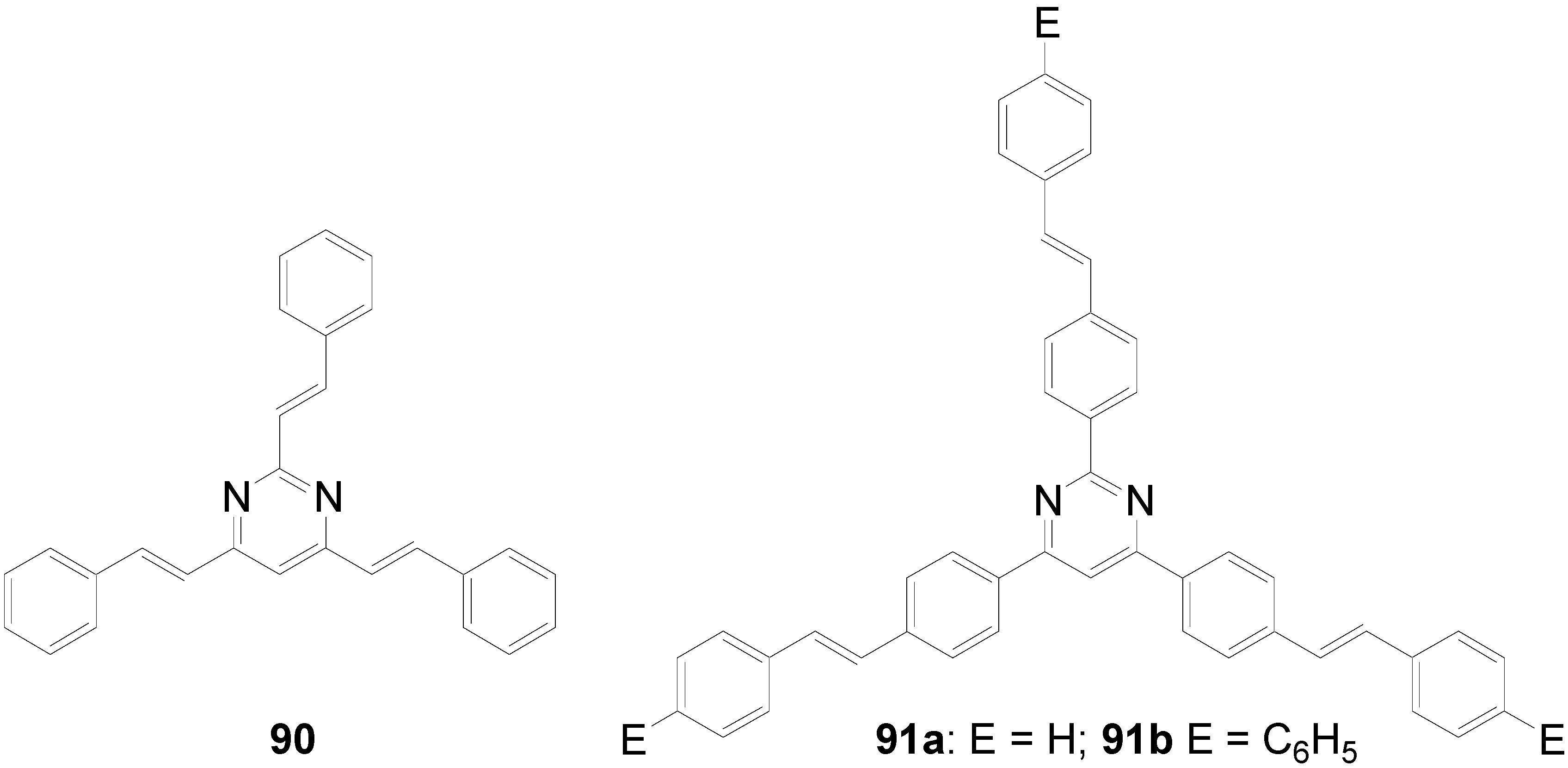
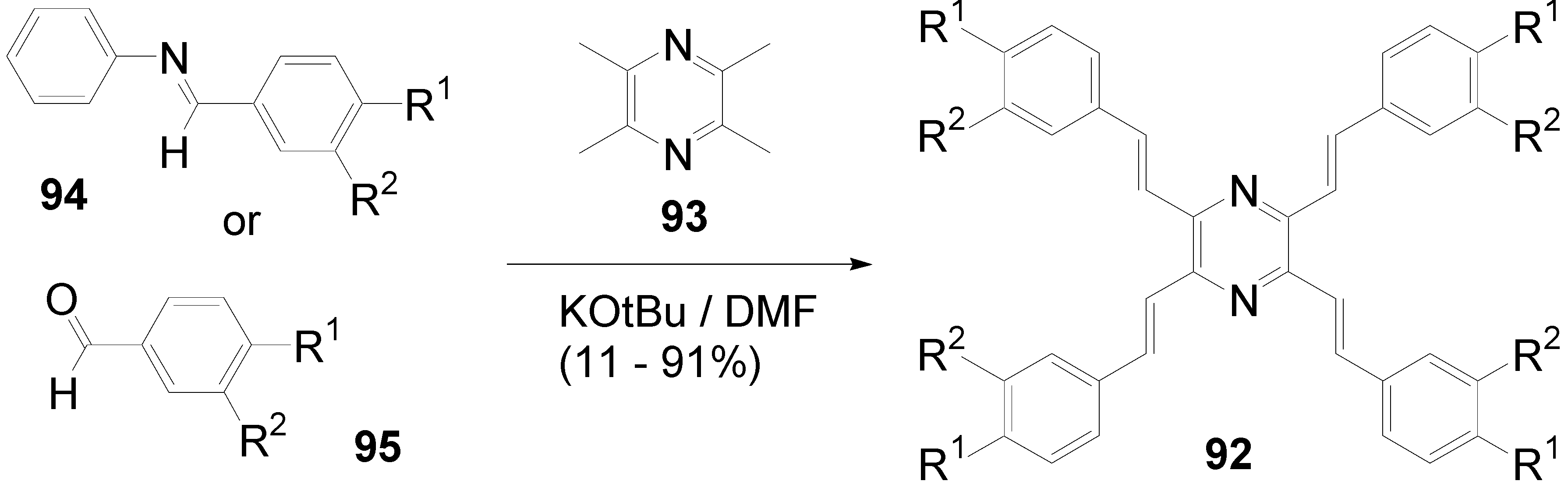
5.3. Pyrazine as Core (C-10-A-6)
| Compound | Transition T ΔH (cal/g) | Transition T ΔH (cal/g) |
| 92d: 3,4-dihexyloxy | Cryst → Col 102 °C (14.0) | Col → i 210 °C (0.4) |
| 92e: 3,4-didecyloxy | Cryst → Col 101 °C (19.3) | Col → i 173 °C (1.5) |
| 92f: 3,4-didodecyloxy | Cryst → Col 95 °C (18.8) | Col → i 172 °C (0.7) |
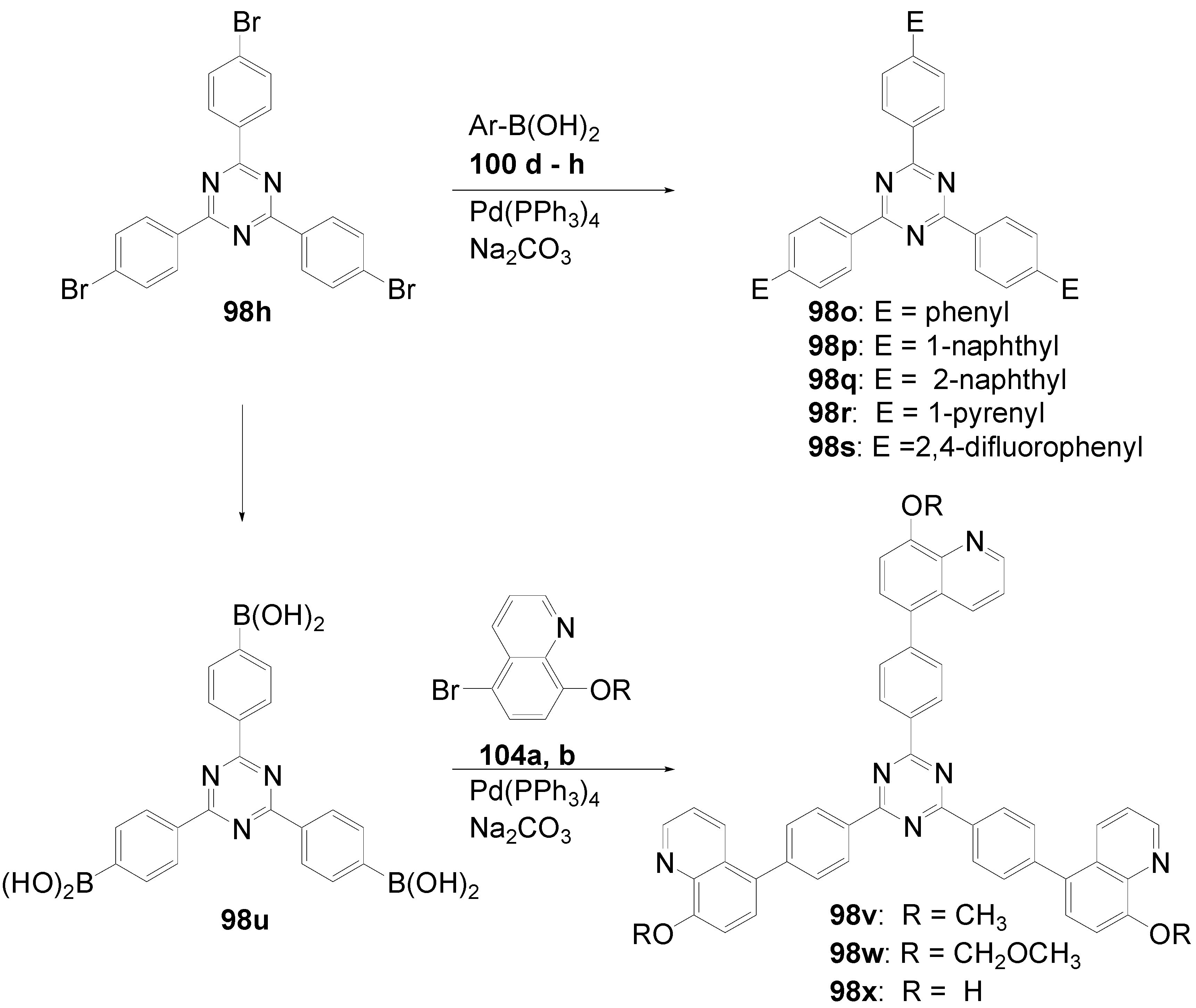
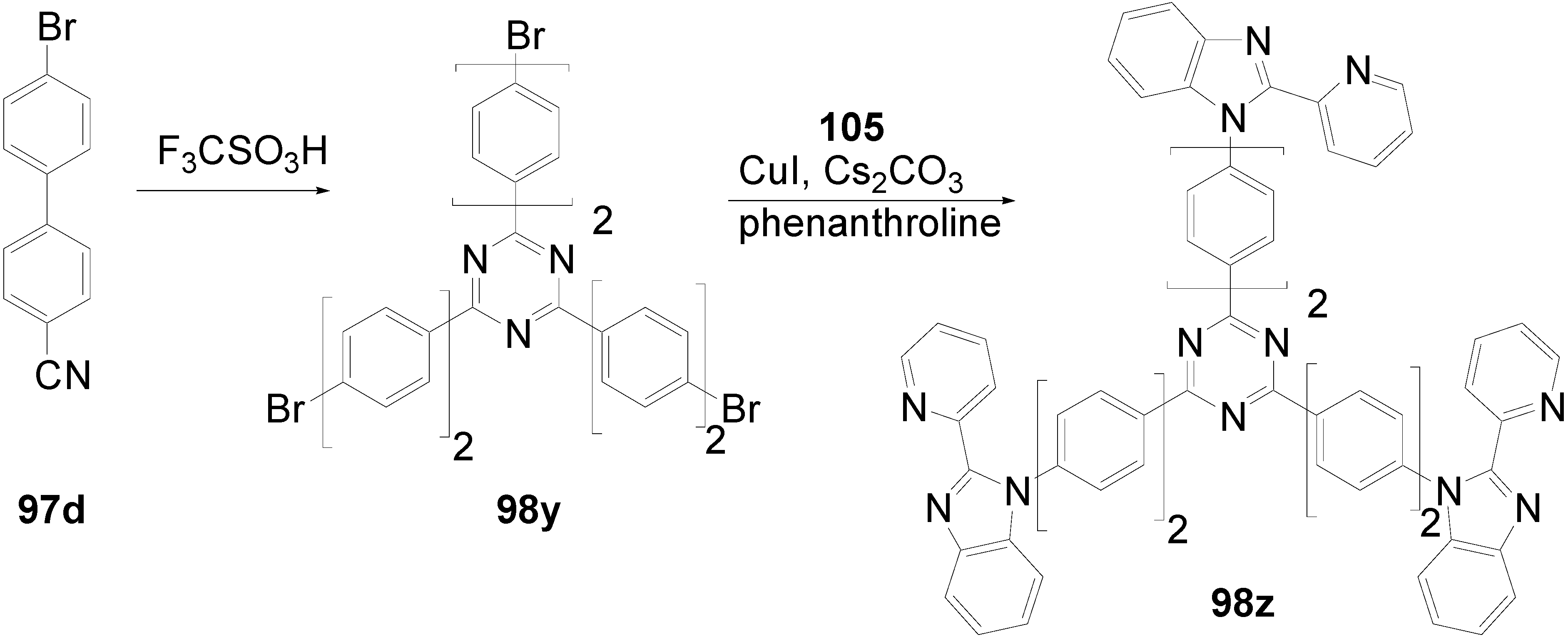
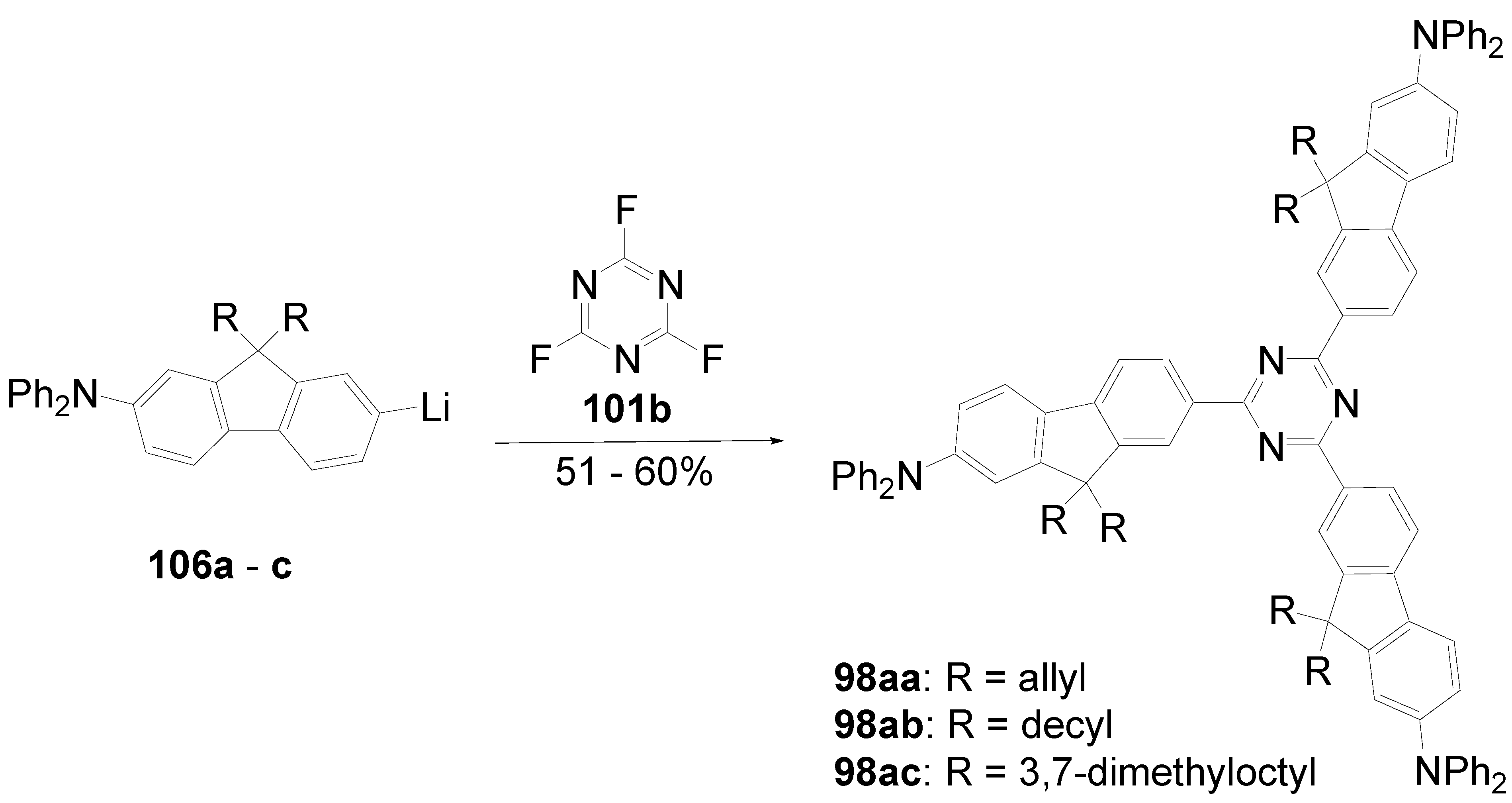
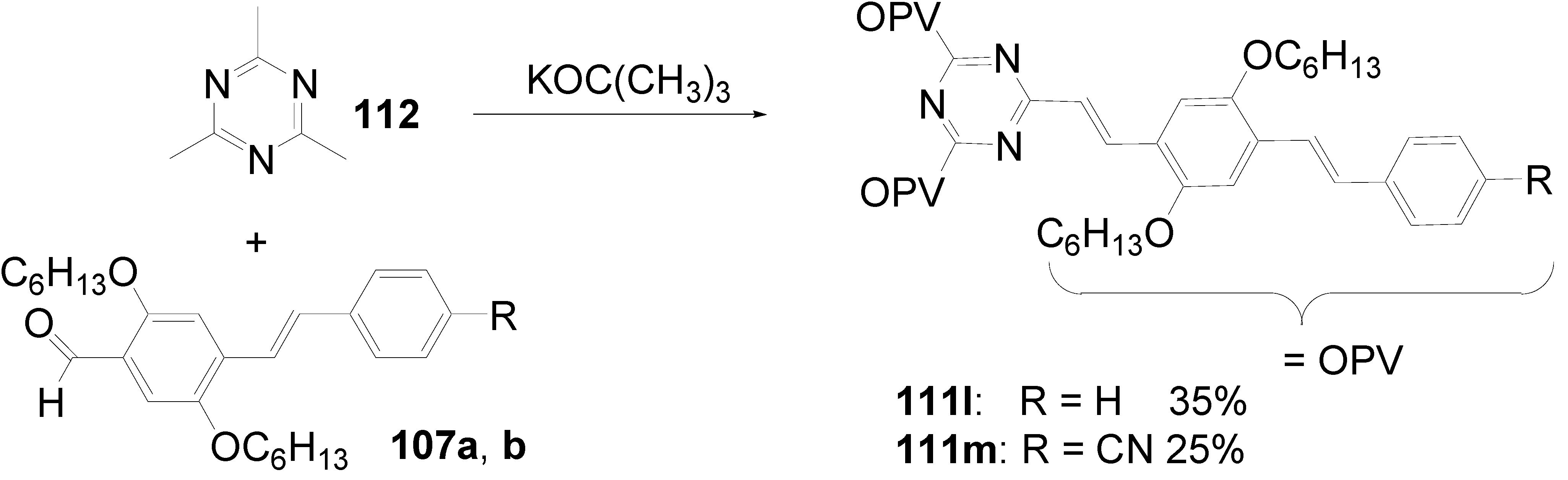
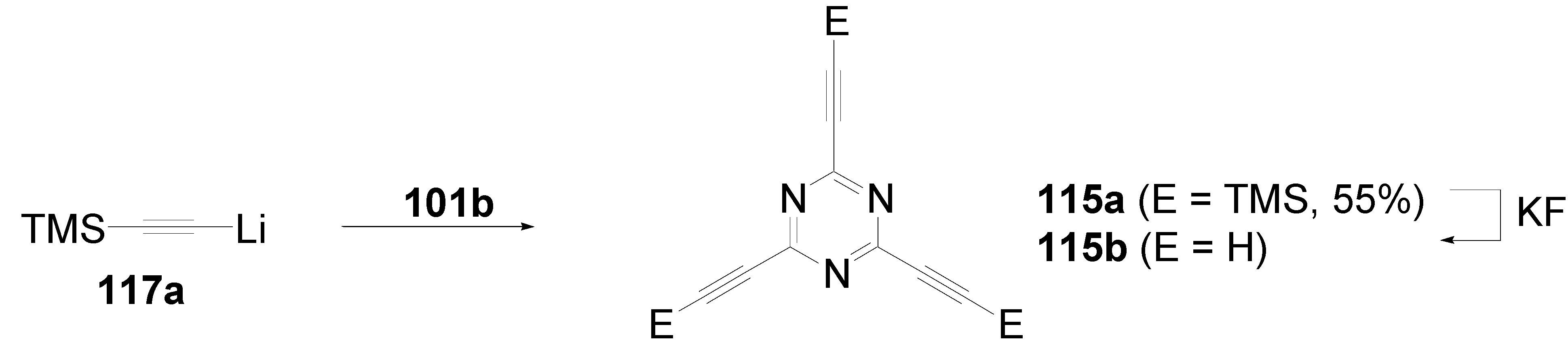
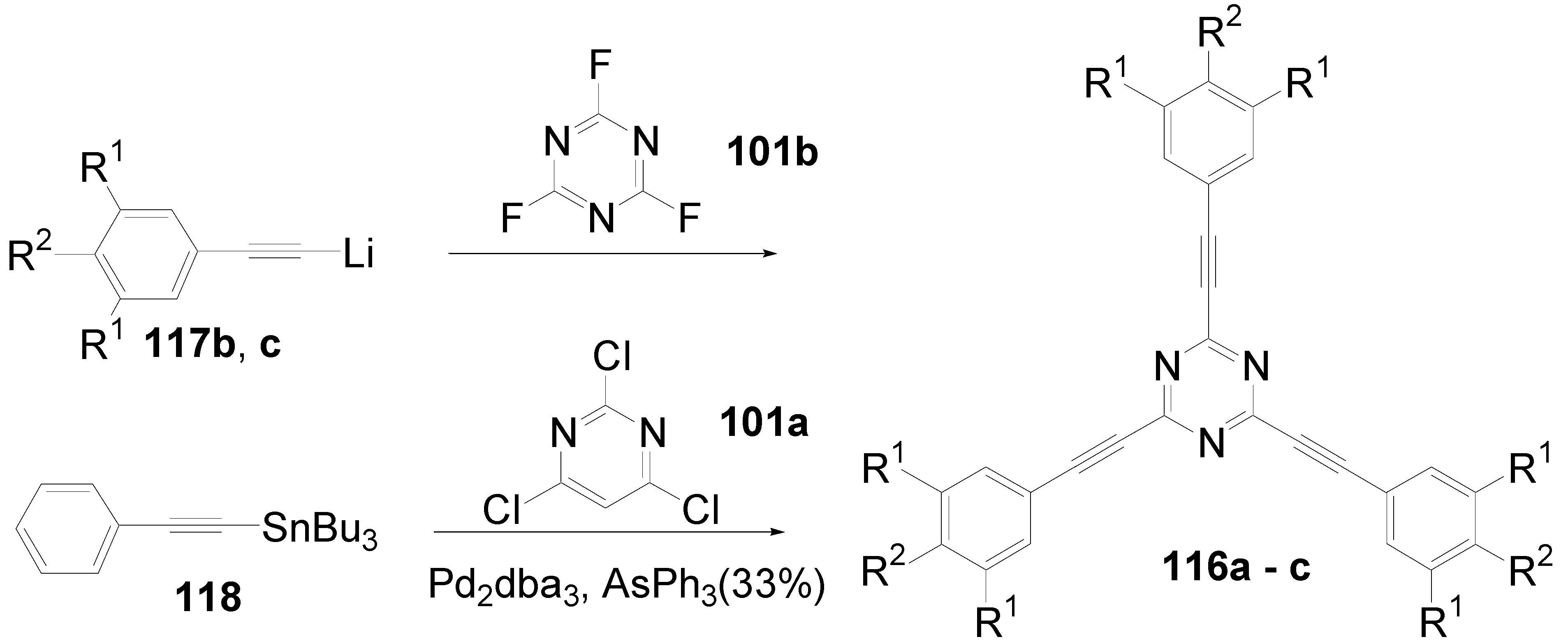
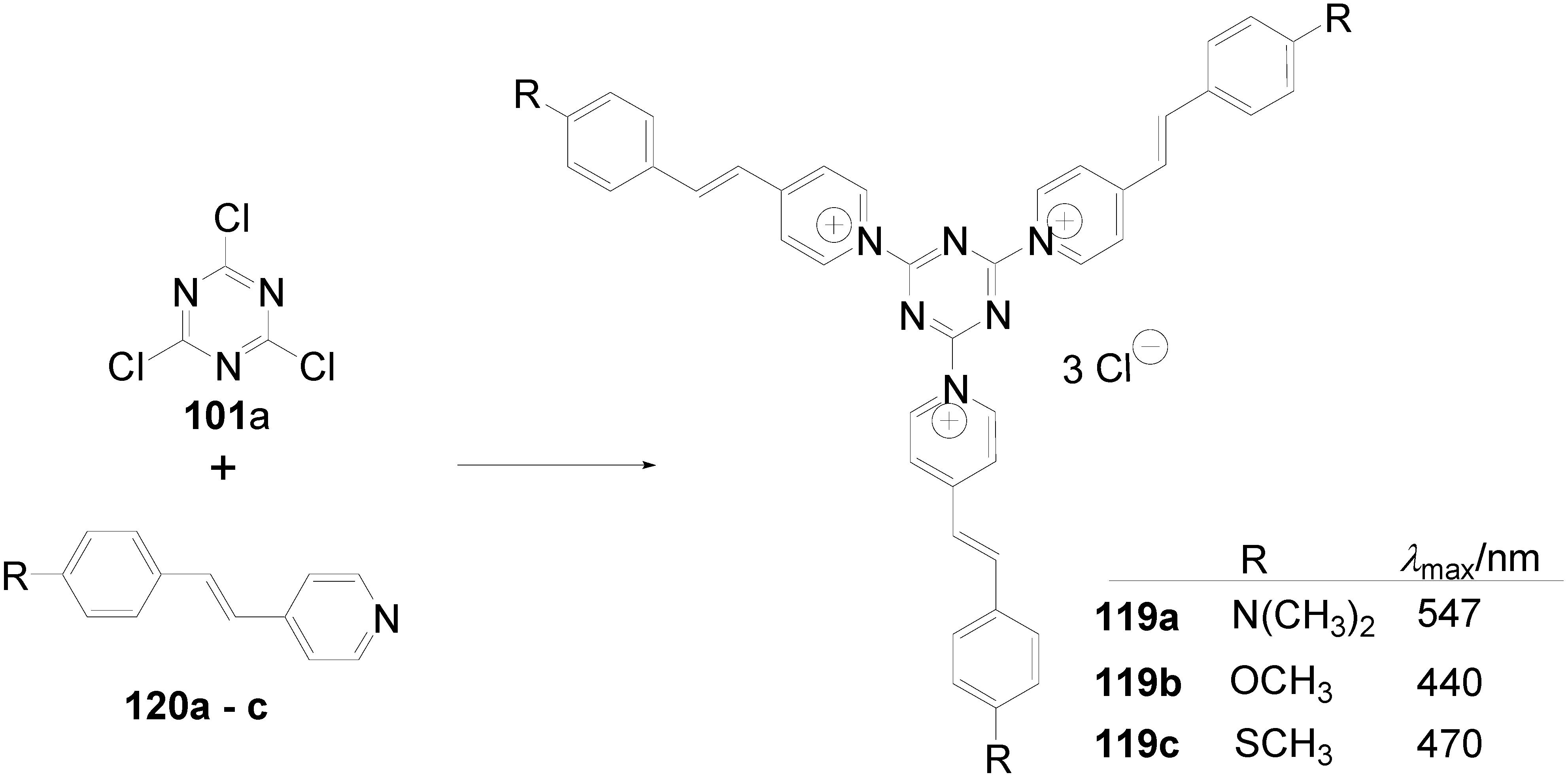
5.4. 1,3,5-Triazine as Core
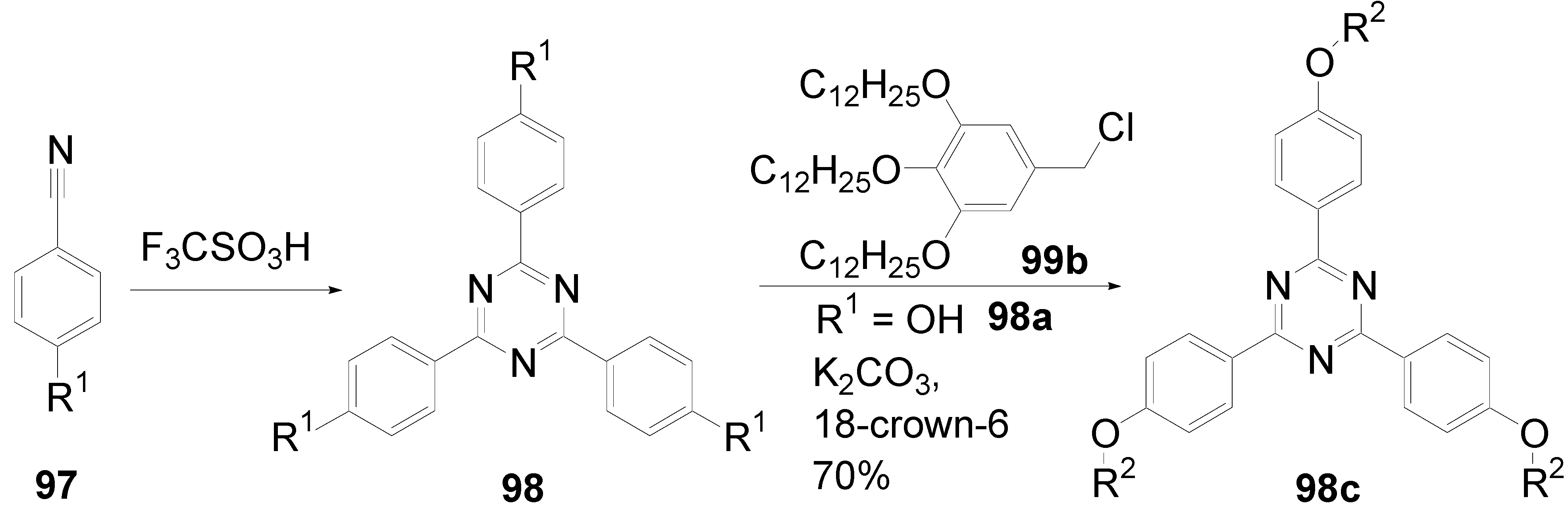
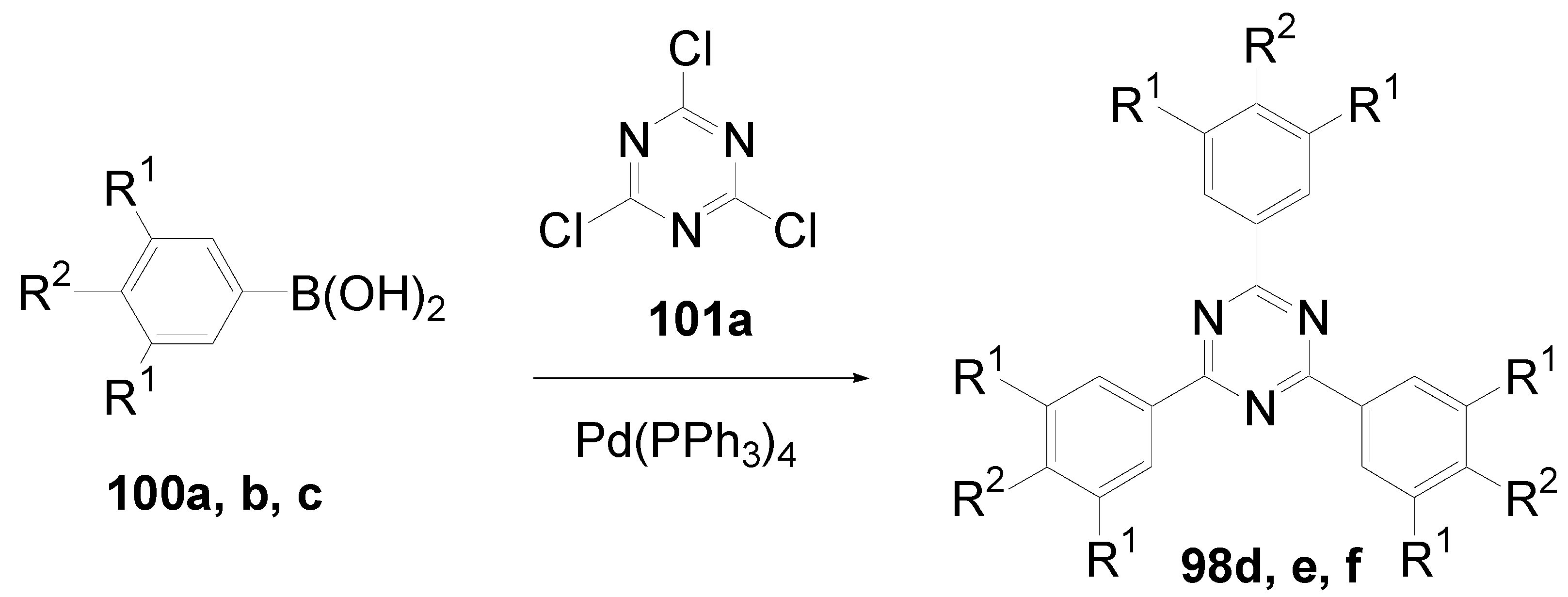
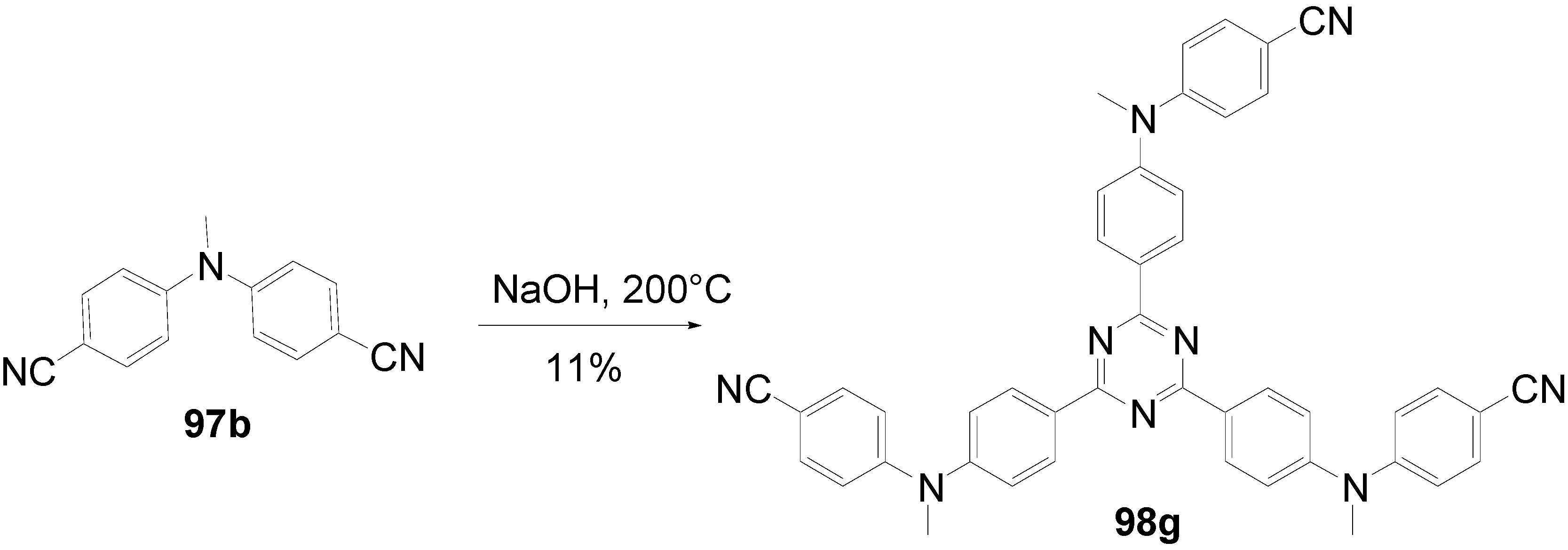
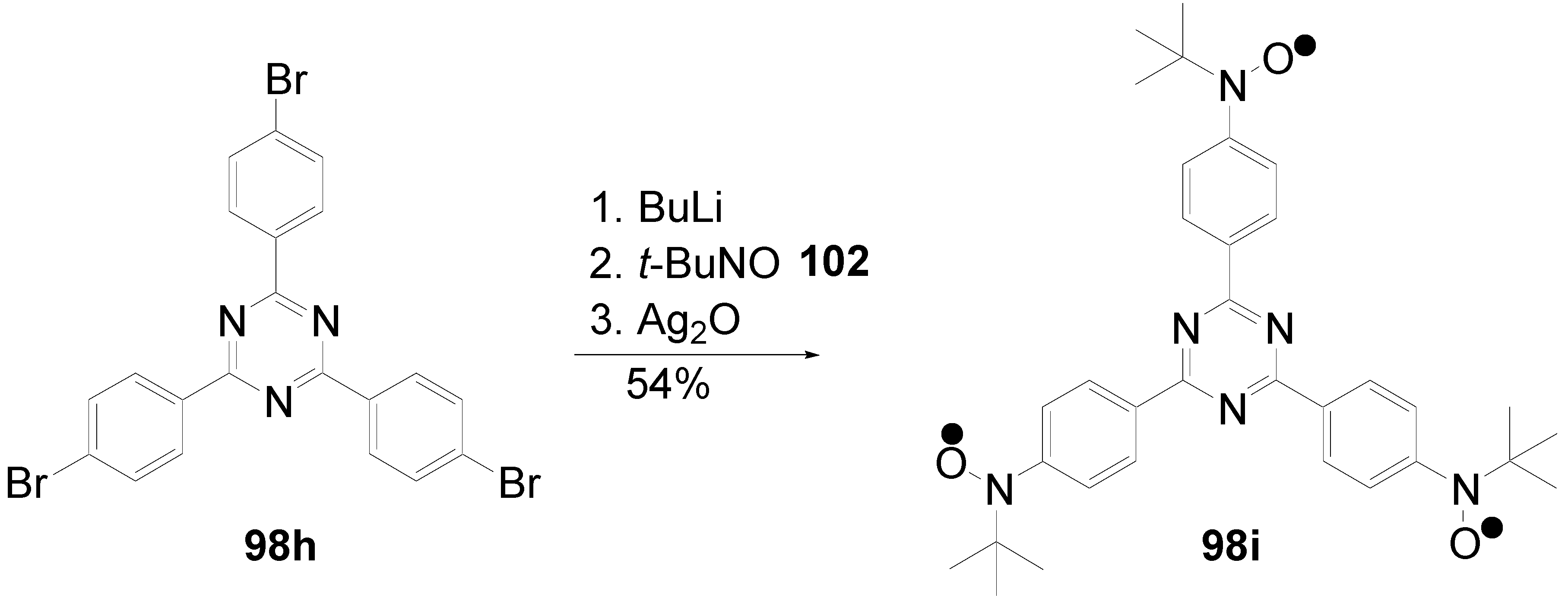
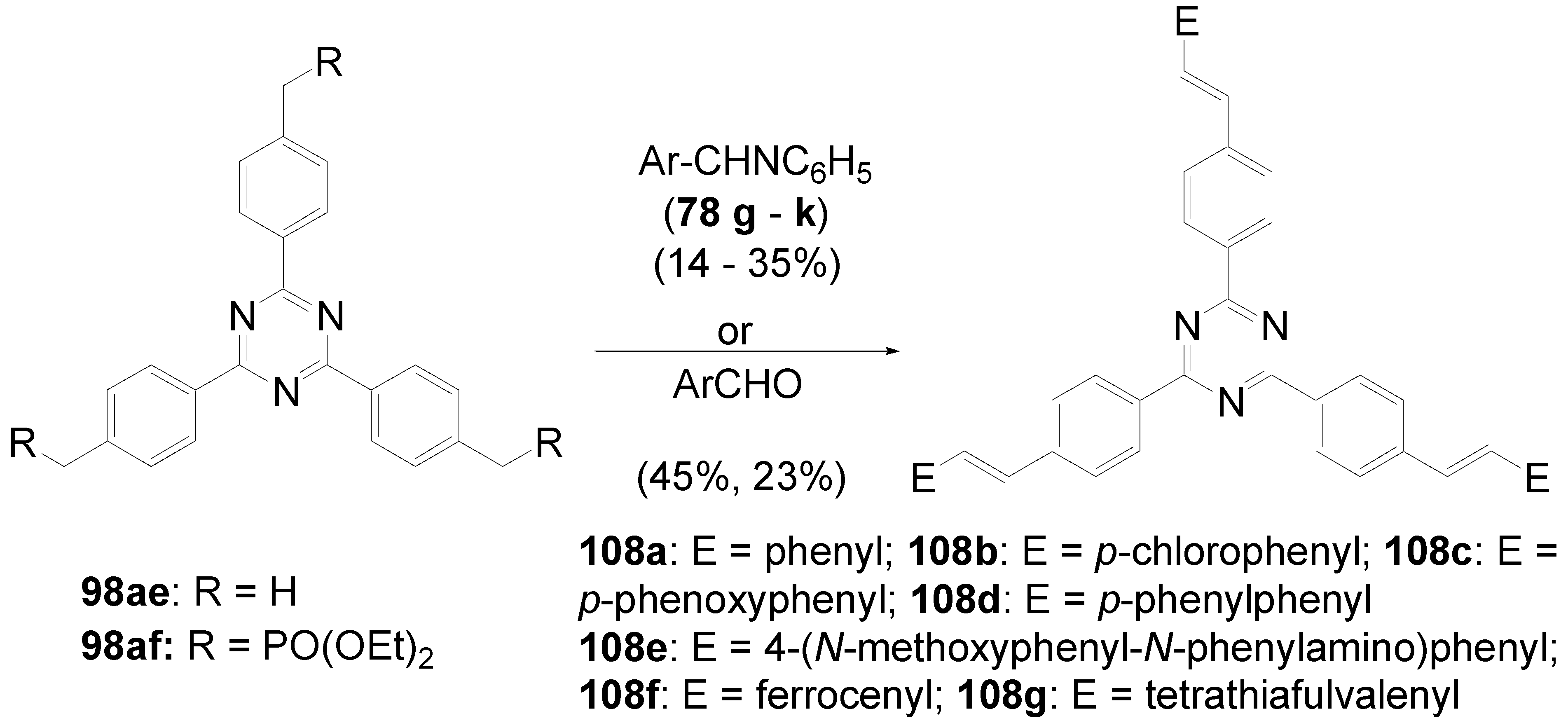
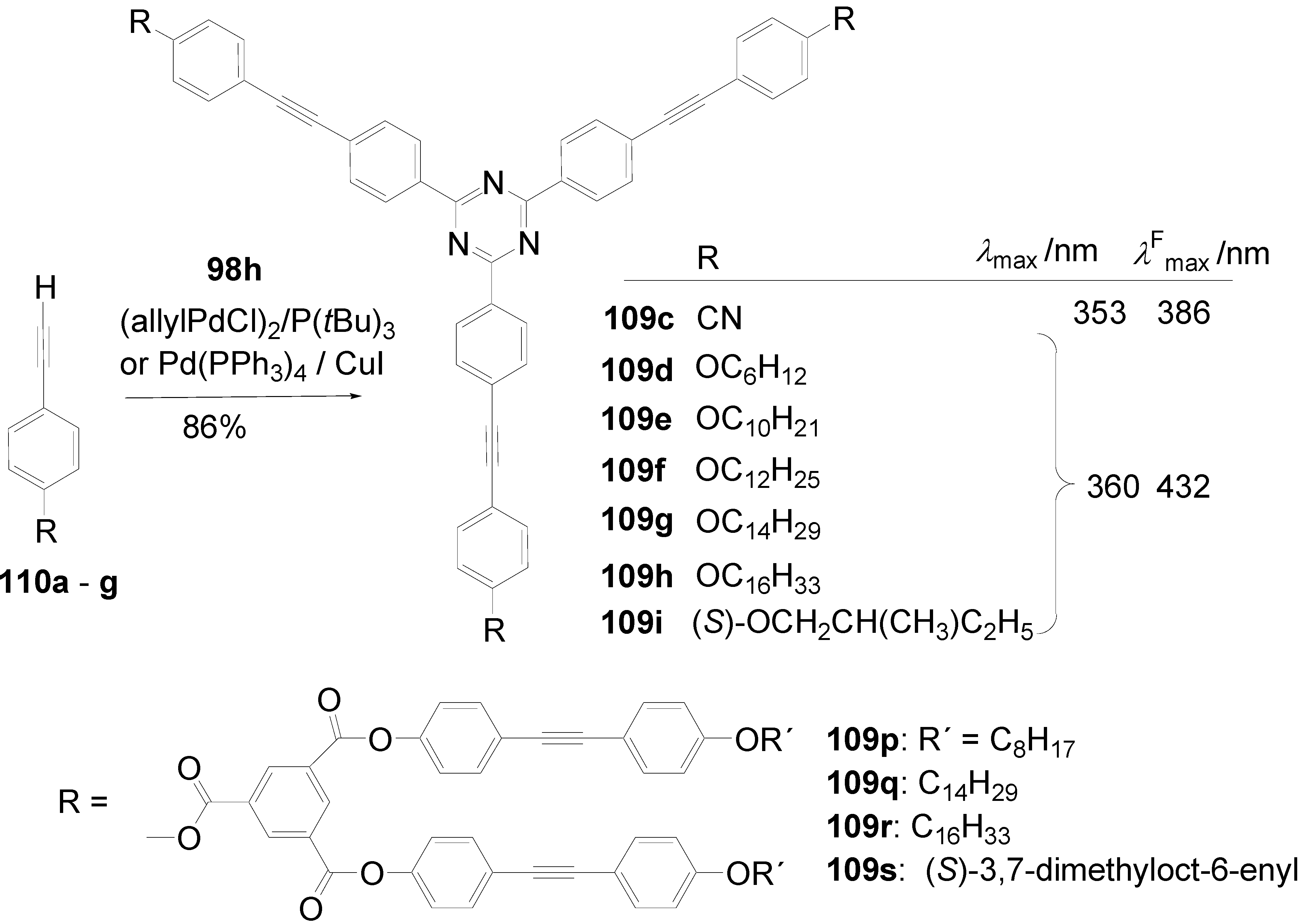
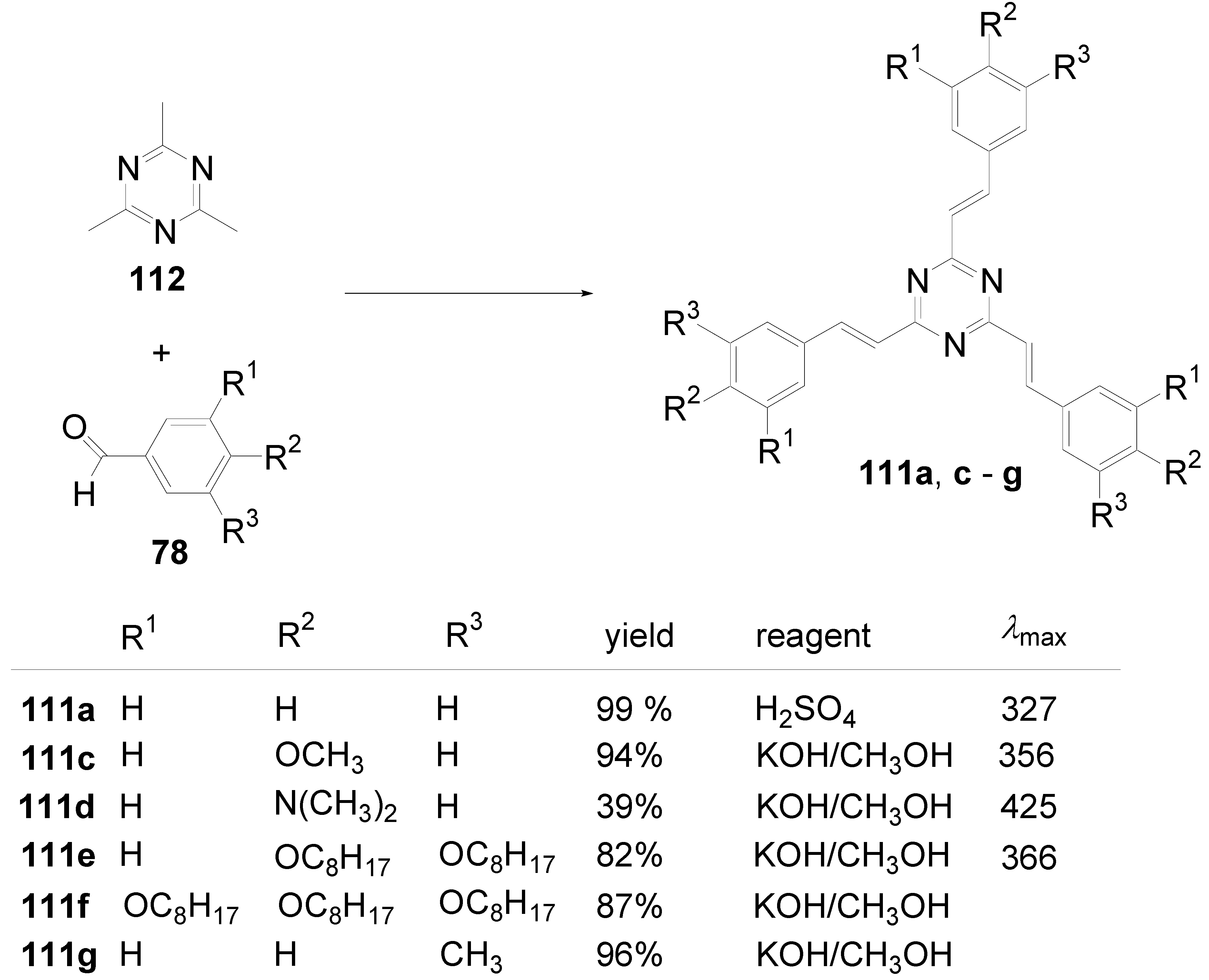
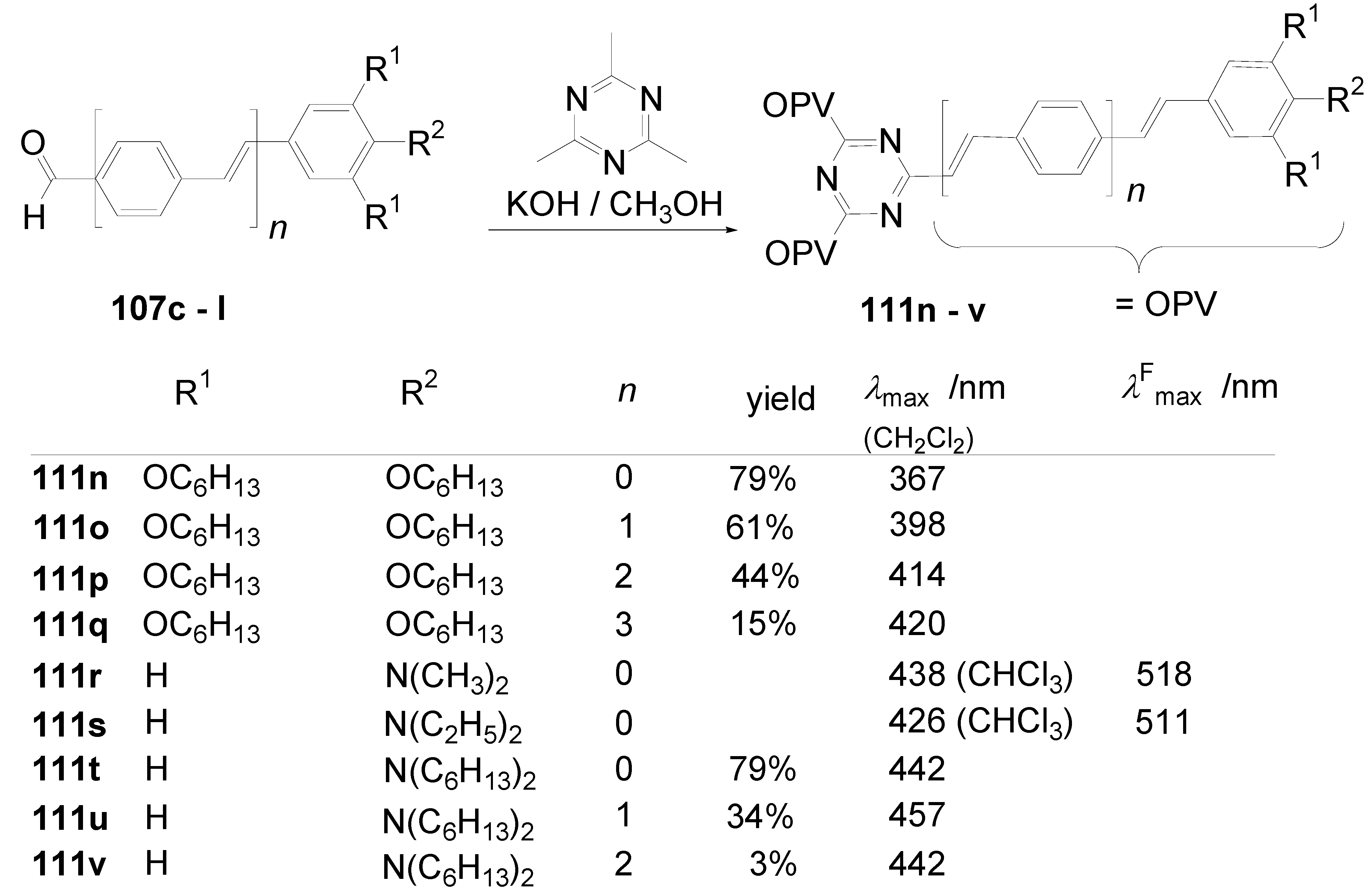
- a)
- ΔE (n+1) < ΔE (n) monotonous bathochromic shift
- b)
- ΔE (n+1) > ΔE (n) monotonous hypsochromic shift
- c)
- ΔE (n+1) ≈ ΔE (n) borderline case between a) and b)
- a)
- ΔE goes through a minimum for a certain n
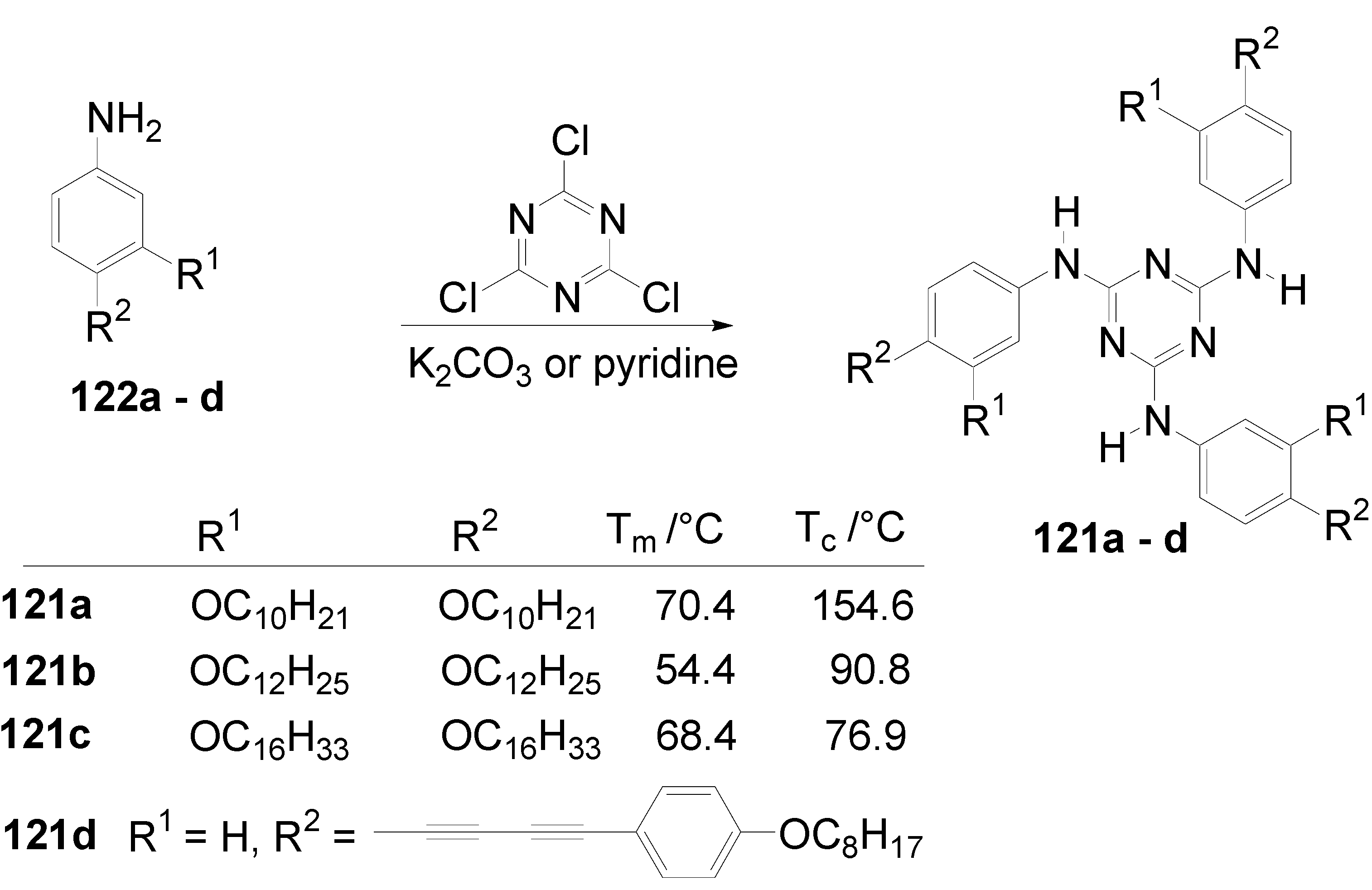
| Star | 2. compound | Tm /°C | Tc /°C | Structure |
| 121a | 70.4 | 154.6 | Colhd | |
| 121b | 54.4 | 90.8 | Dhd | |
| 121c | 68.4 | 76.9 | Dro | |
| 121a | 123a | 32.4 | 64.8 | Colhd |
| 121a | 123b | 34.5 | 75.1 | Colhd |
| 121a | 123c | 18.7 | 73.6 | Colhd |
| 121a | 123d | 62.2 | 88.7 | Colrd |
| 121a | 124 | 68.1 | 110.4 | SmA |
| 121b | 124 | 68.6 | 115.9 | SmA |
| 121b | 125 | 93.7 | 147.2 | Colr |
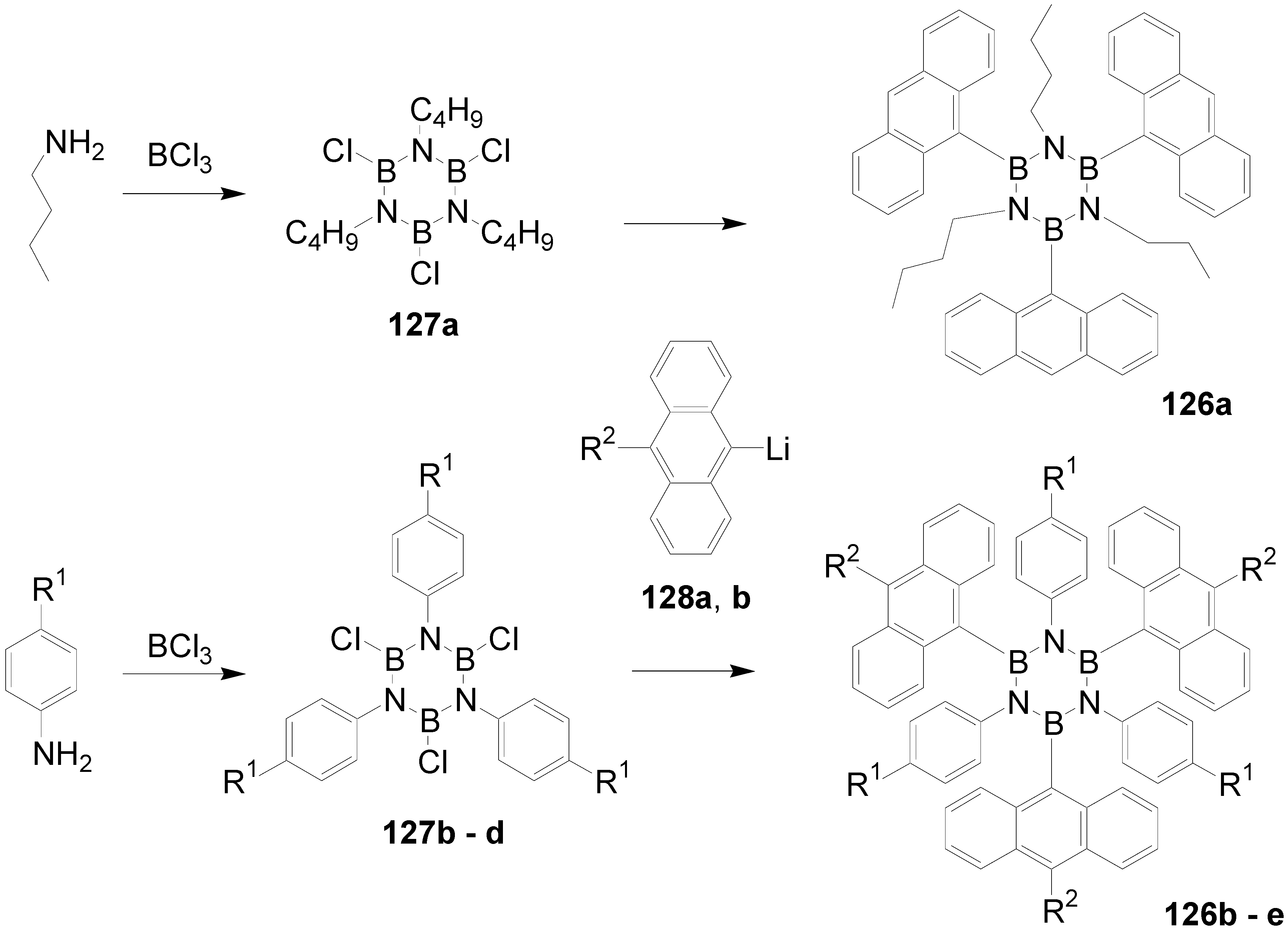
5.5. Borazine as a Core
6. Condensed Ring Systems as Cores
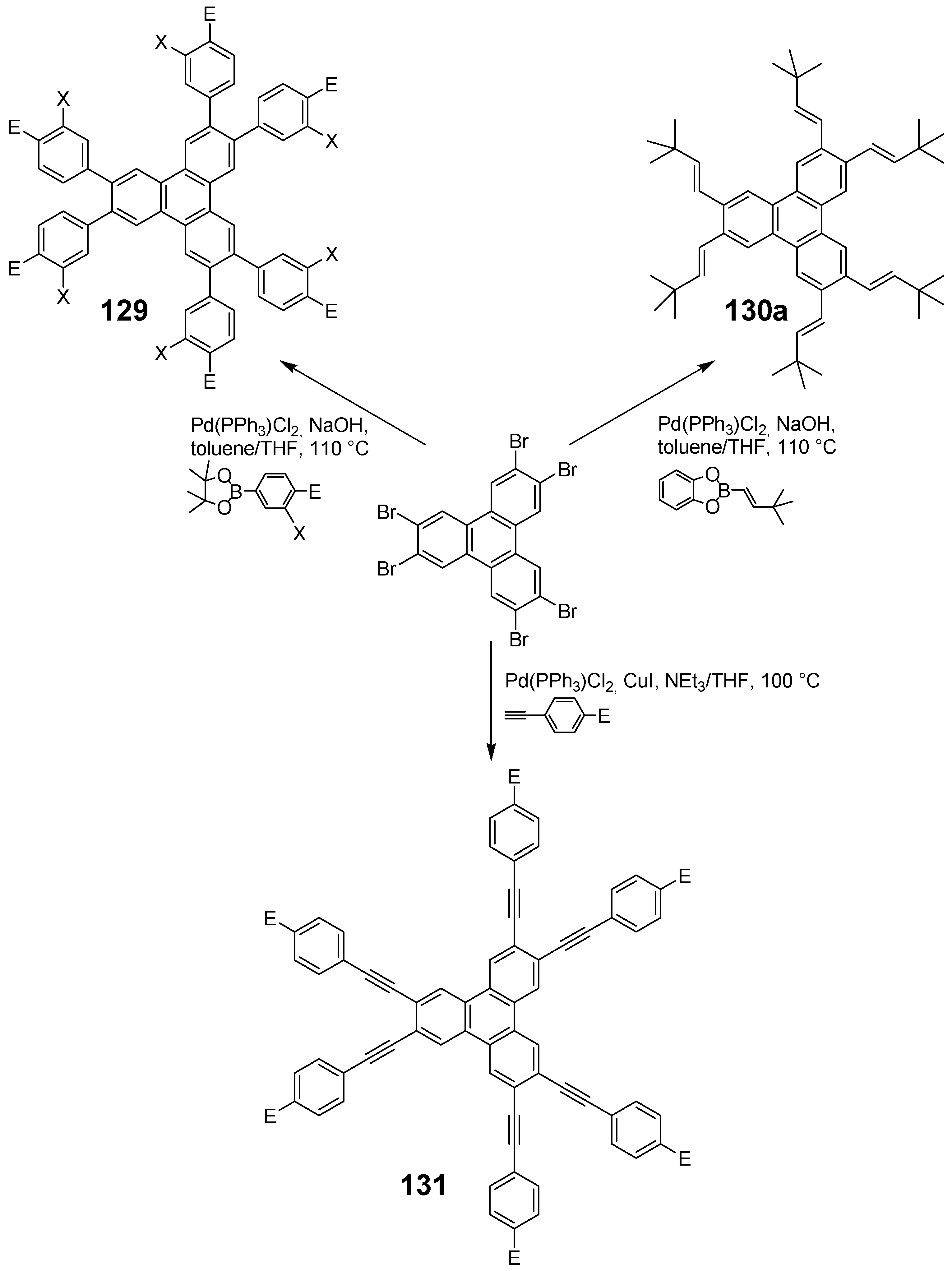
6.1. Triphenylene Star Compounds (C-12)
6.1.1. Structures and Synthesis

6.1.2. Triphenylene Derivatives and Materials Science
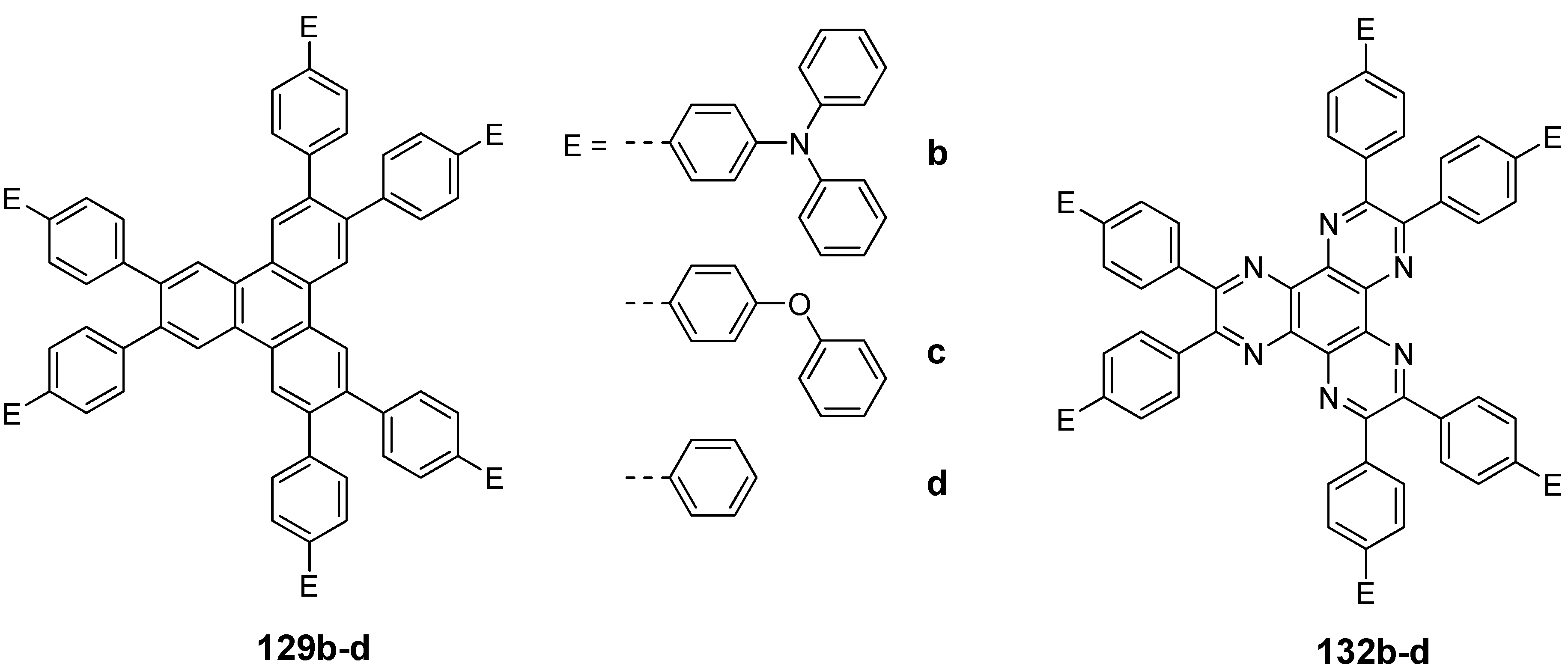
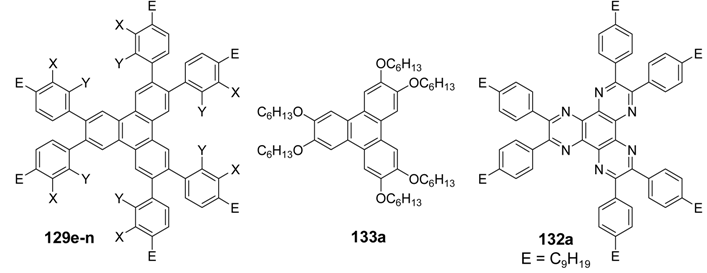 | |||||
| Compound | E | X | Y | phase transitions [°C] | reference |
| 129e | OC6H13 | OC6H13 | H | Cr 111 Col 126 I Cr 65 Colh 135 I | [335,336] |
| 129f | OC8H17 | OC8H17 | H | Cr 85 Col 104 I | [335] |
| 129g | OC10H21 | OC10H21 | H | Cr 74 Col 103 I | [335] |
| 129h | OC12H25 | OC12H25 | H | Cr 47 Col 101 I | [335] |
| 129i | OC6H13 | H | H | Cr 153 I | [332] |
| 129j | OC11H23 | H | H | Cr 66 I | [332] |
| 129k | H | OC6H13 | H | Cr 11 I | [337] |
| 129l | H | H | OC6H13 | Cr 81 I | [337] |
| 129m | C9H19 | H | H | Cr 59 I | [332] |
| 129n | C12H25 | H | H | Cr 37 I | [332] |
| 129m + 133a 1 :1 | Cr 66 Colh 155 I | [336] | |||
| 132a | C9H19 | Cr 81 I | [336,338] | ||
| 132a + 133a 1:1 | Colh 240 I | [336,338] | |||
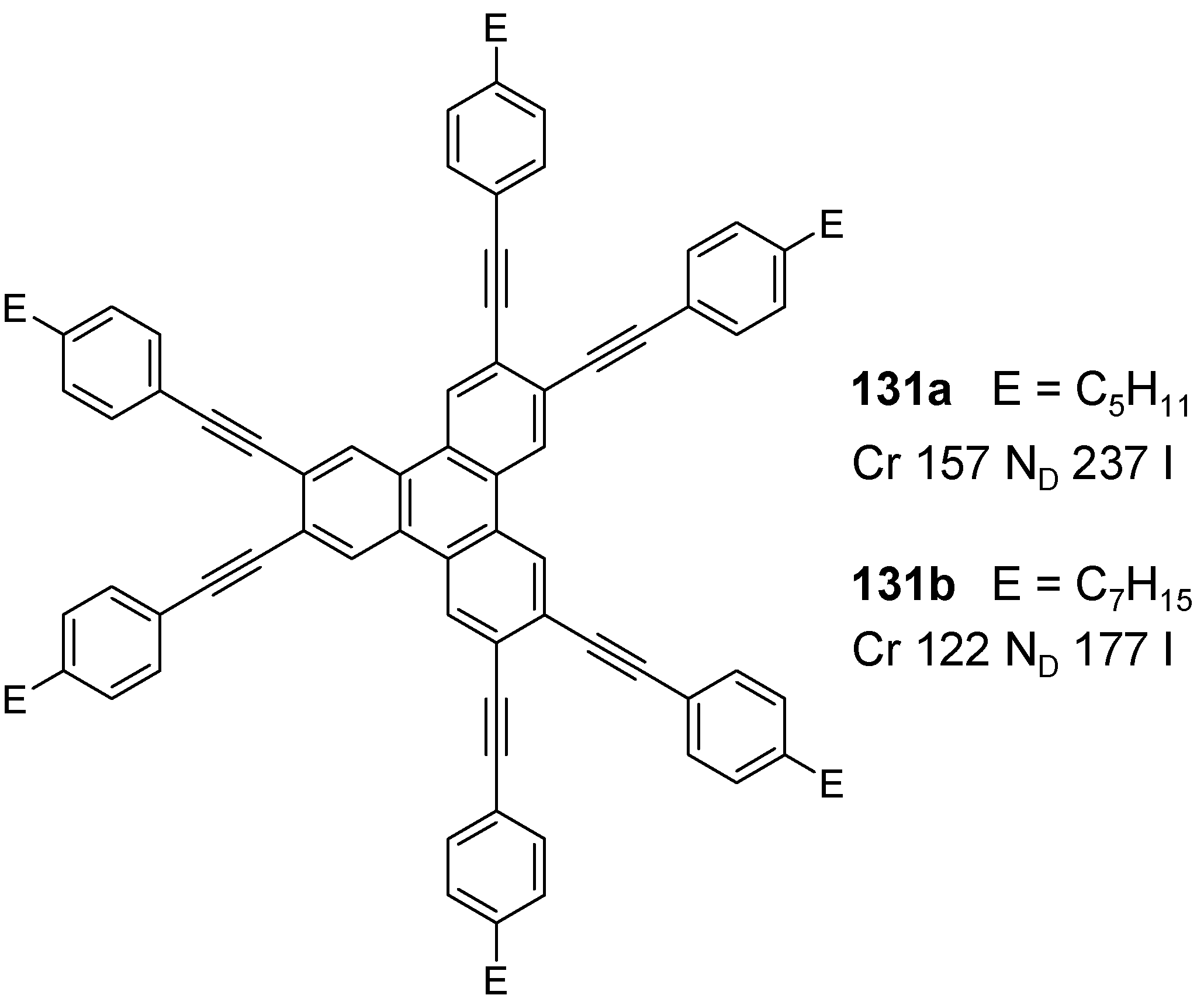
6.2. Hexaazatriphenylenec (C-13-A-3 and C-13-A-6)
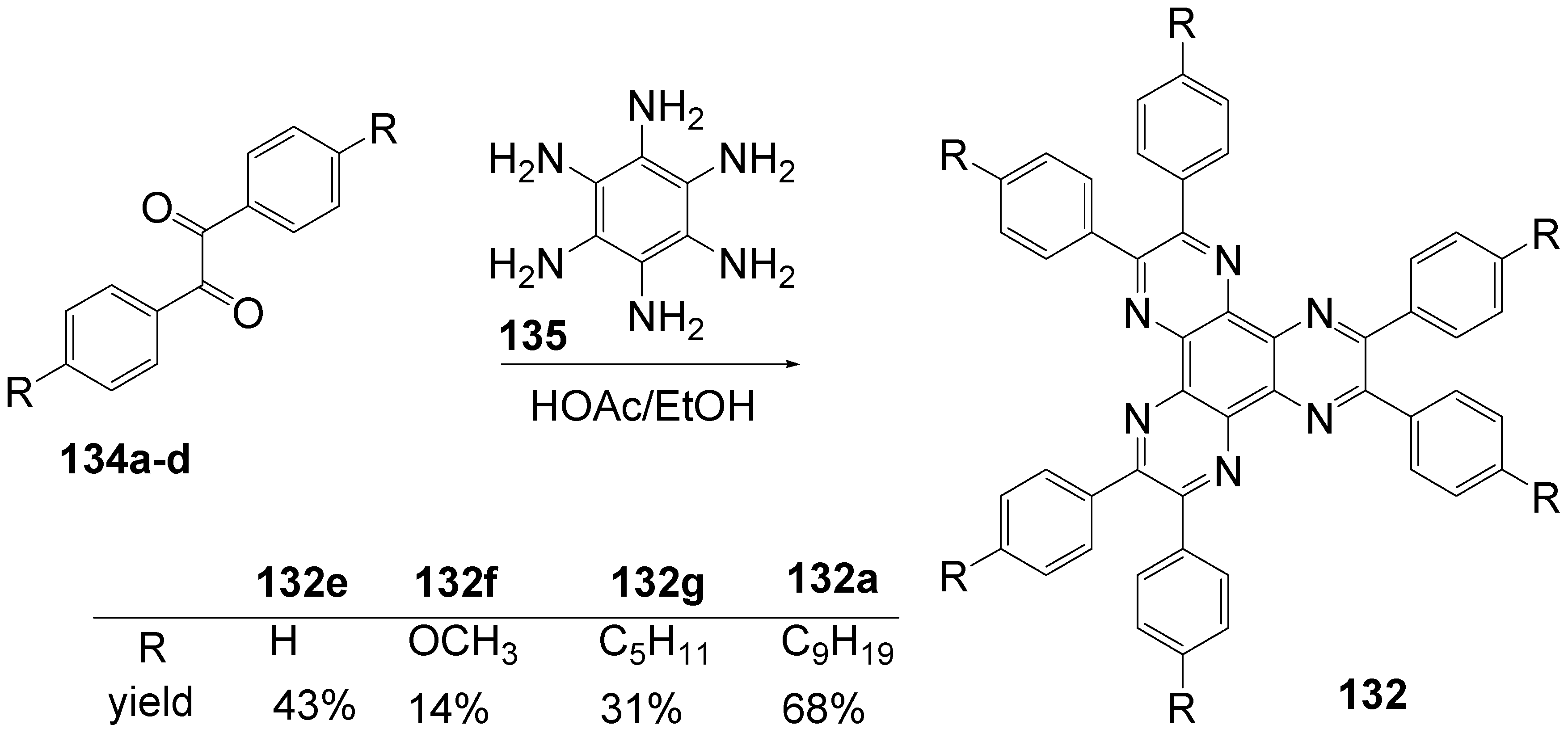
| R = | λmax /nm (toluene) | ε / l/mol°cm | λFmax / nm(toluene) | Φ | λFmax / nm (CH2Cl2) | Φ | |
| 132e | H | 366 | 21.000 | 419 | 0.01 | 424 | 0.03 |
| 132k | -N(C6H5)2 | 453 | 98.000 | 502 | 0.95 | 563 | 0.75 |
| 132d | -C6H5 | 392 | 68.000 | 438 | 0.43 | 449 | 0.50 |
| 132b | -C6H4- N(C6H5)2 | 417 | 129.000 | 501 | 0.96 | 624 | 0.27 |
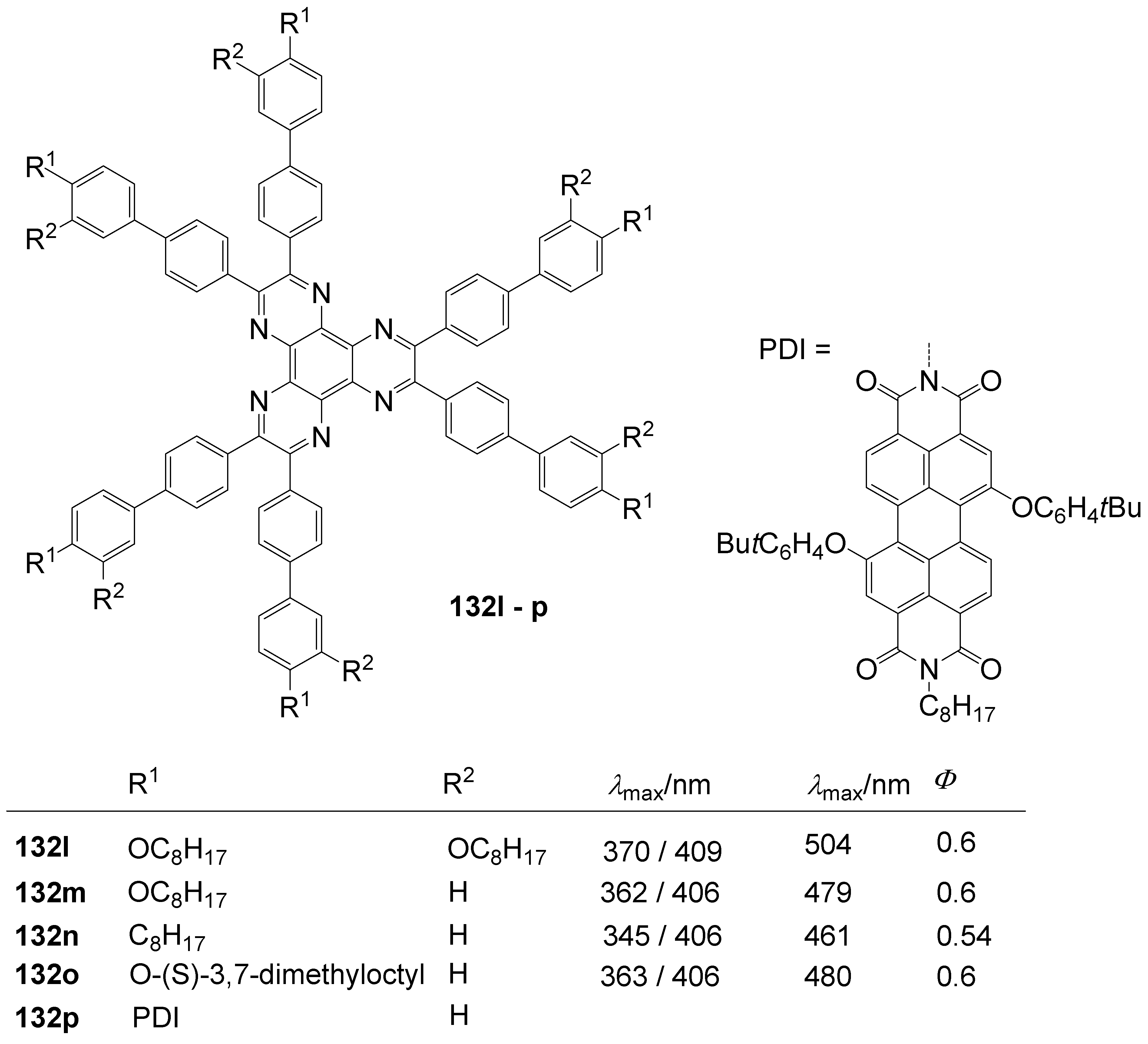
6.3. Triazatruxenes (C-14-A-3 and C-14-A-8)
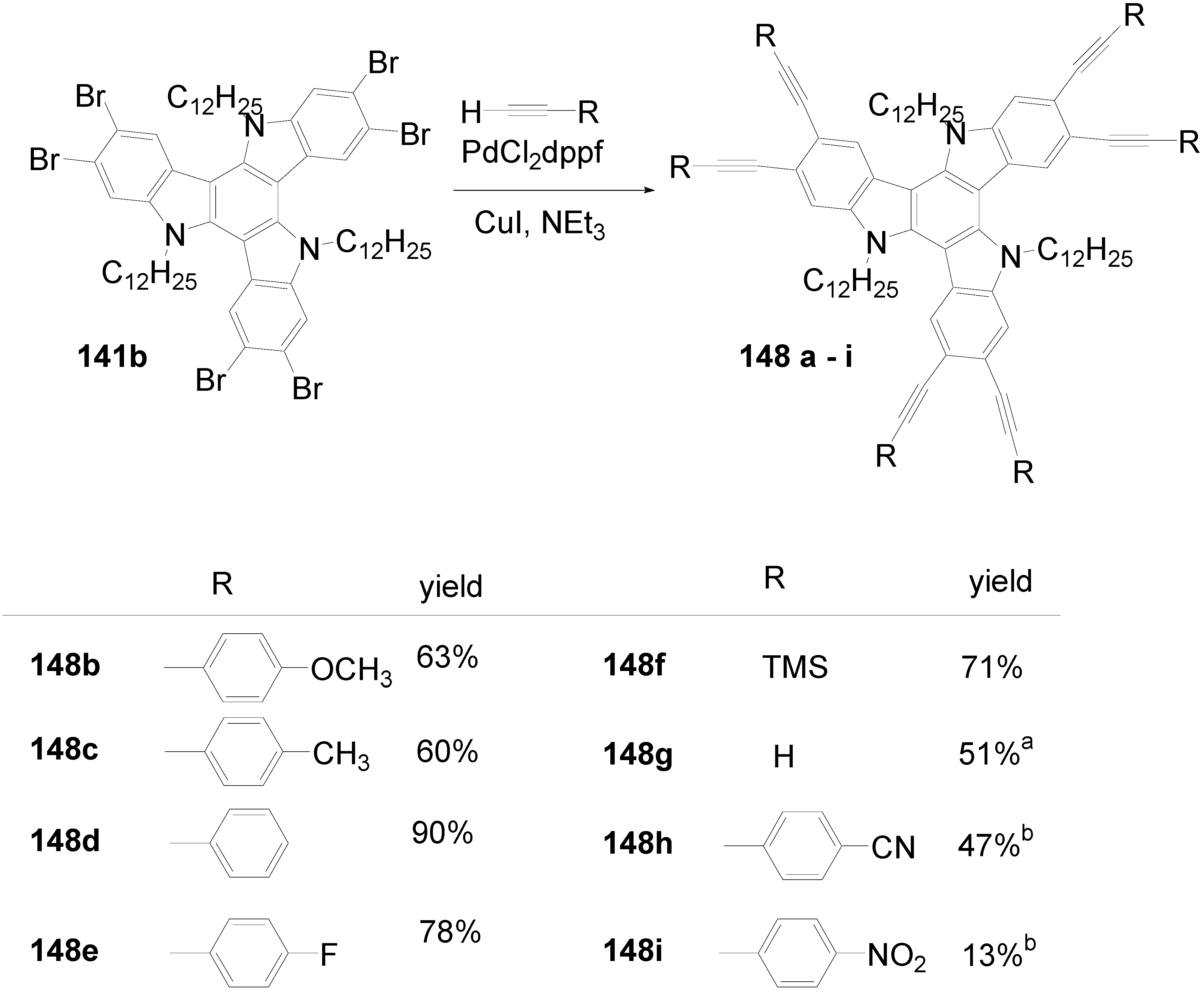
6.4. Tristriazolotriazines (C-15-A-3 and C-15-A-9)
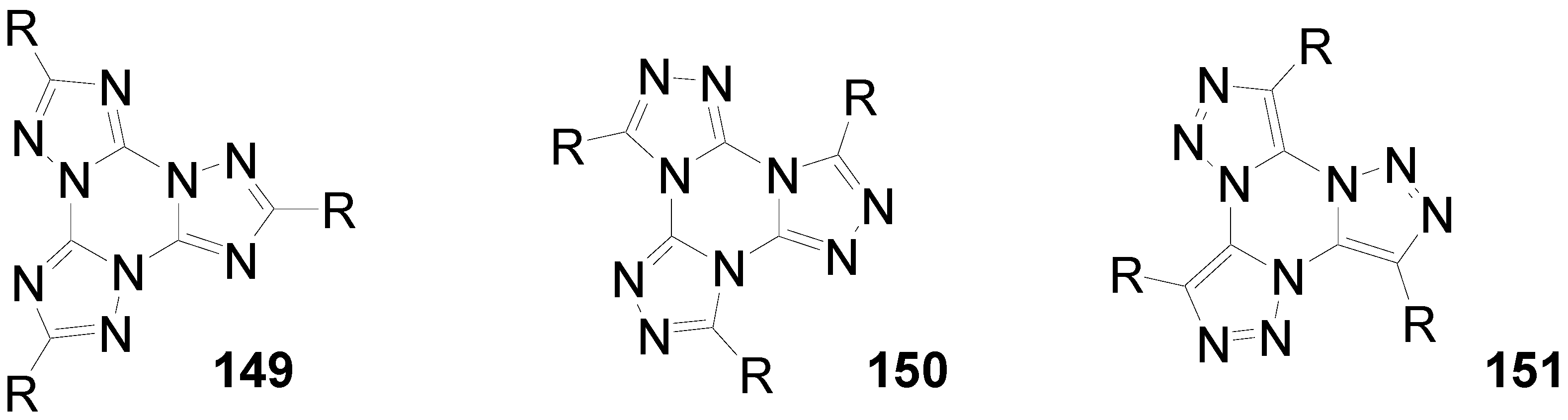
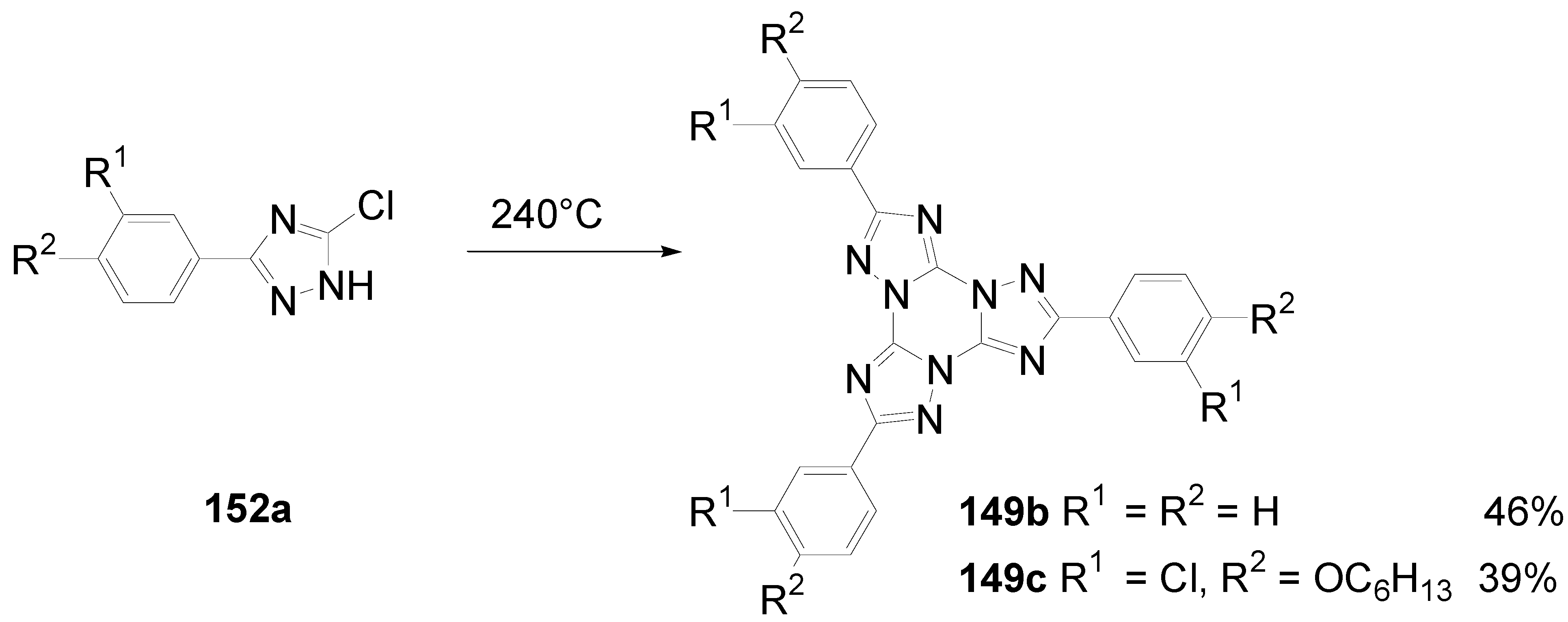
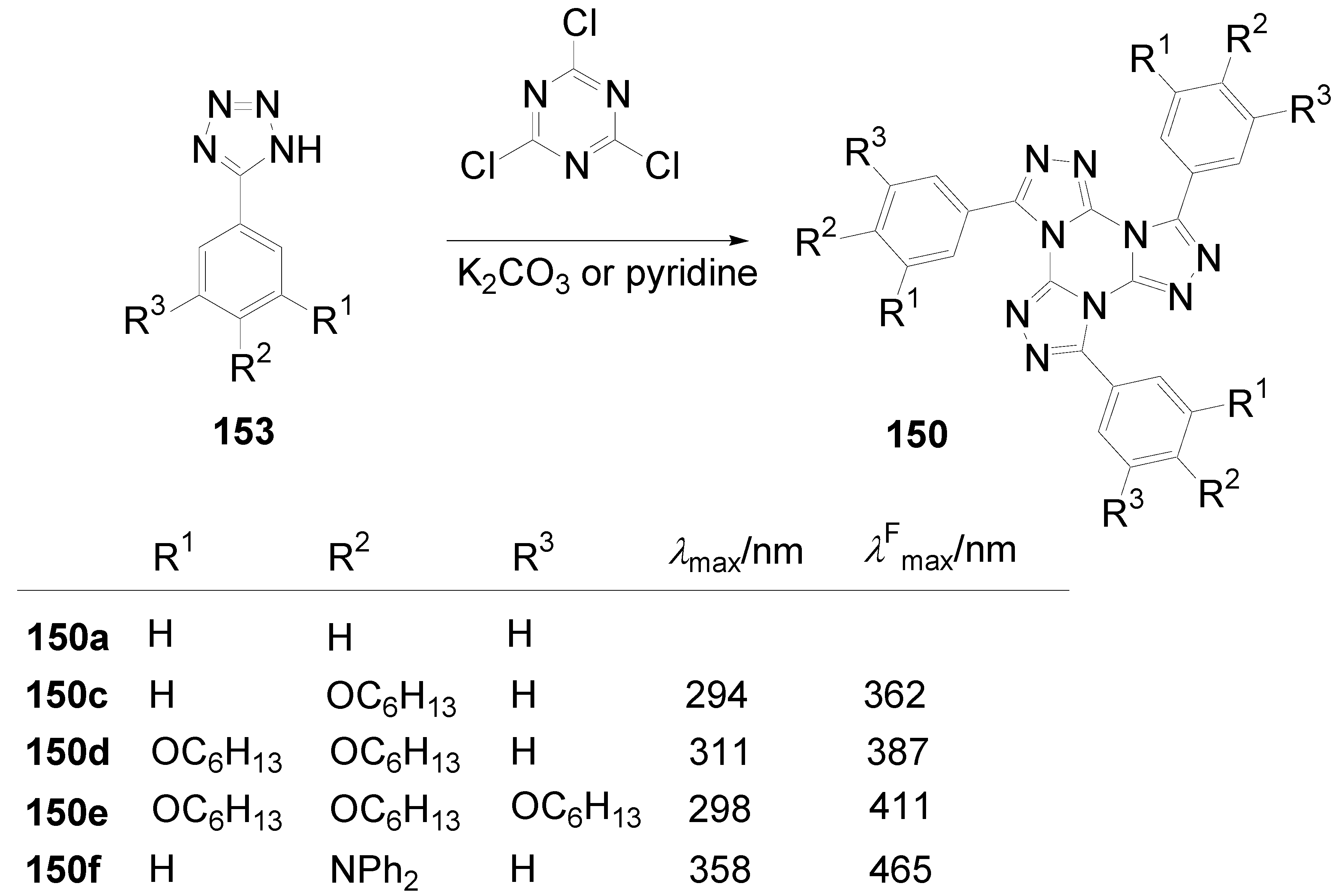
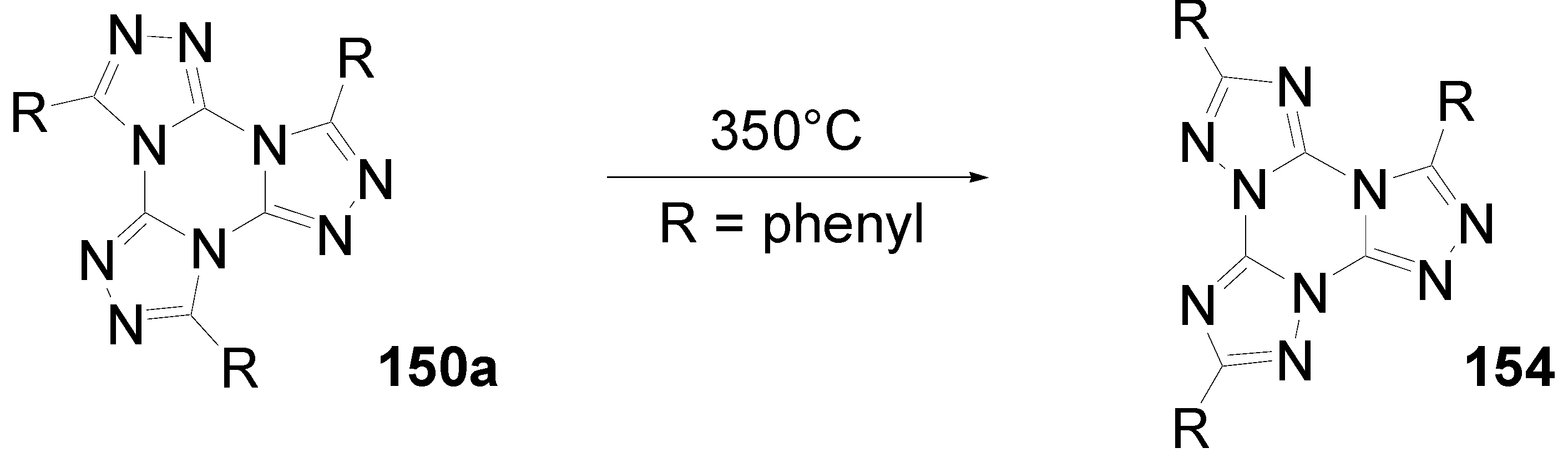
7. Summary and Conclusion
References and Notes
- Tour, J.M. Conjugated macromolecules of precise length and constitution. Organic synthesis for the construction of nanoarchitectures. Chem. Rev. 1996, 96, 537–554. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.S. Shape-persistent molecular architectures of nanoscale dimension. Acc. Chem. Res. 1997, 30, 402–413. [Google Scholar] [CrossRef]
- Roncali, J. Synthetic principles for band gap control in linear π-conjugated systems. Chem. Rev. 1997, 97, 173–206. [Google Scholar] [CrossRef] [PubMed]
- Müllen, K.; Wegner, G. Electronic Materials: The Oligomer Approach; Wiley-VCH: Weinheim, Germany, 1998. [Google Scholar]
- Swager, T.M. The molecular wire approach to sensory signal amplification. Acc. Chem. Res. 1998, 31, 201–207. [Google Scholar] [CrossRef]
- Martin, R.E.; Diederich, F. Linear monodisperse π-conjugated oligomers: Model compounds for polymers and more. Angew. Chem. Int. Ed. 1999, 38, 1350–1377. [Google Scholar] [CrossRef]
- Schwab, P.F.H.; Levin, M.D.; Michl, J. Molecularrods. Simple axial rods. J. Chem. Rev. 1999, 99, 1863–1933. [Google Scholar] [CrossRef]
- Diederich, F.; Gobbi, L. Cyclic and linear acetylenic molecular scaffolding. Top. Curr. Chem. 1999, 201, 43–79. [Google Scholar]
- Scherf, U. Oligo- and polyacrylenes, oligo- and polyarylenevinylenes. Top. Curr. Chem. 1999, 201, 163–222. [Google Scholar]
- Roncali, J. Oligothienylenevinylenes as a new class of multinanometer linear π-conjugated systems for micro- and nanoelectronics. Acc. Chem. Res. 2000, 33, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Segura, J.L.; Martin, N. Functionalized oligoarylenes as building blocks for new organic materials. J. Mater. Chem. 2000, 10, 2403–2435. [Google Scholar] [CrossRef]
- Szafert, S.; Gladysz, J.A. Carbon in one dimension: Structural analysis of higher conjugated polyynes. Chem. Rev. 2003, 103, 4175–4206. [Google Scholar] [CrossRef] [PubMed]
- Babudri, F.; Farinola, G.M.; Naso, F. Synthesis of conjugated oligomers and polymers: The organometallic way. J. Mater. Chem. 2004, 14, 11–34. [Google Scholar] [CrossRef]
- Meier, H. Conjugated oligomers with terminal donor-acceptor substitution. Angew. Chem. Int. Ed. 2005, 44, 2482–2506. [Google Scholar] [CrossRef]
- Grimsdale, A.C.; Müllen, K. The chemistry of organic nanomaterials. Angew. Chem. Int. Ed. 2005, 44, 5592–5629. [Google Scholar] [CrossRef]
- Meier, H. Defined-length carbon-rich conjugated oligomers. In Carbon-Rich Compounds; Haley, M.M., Tykwinski, R.R., Eds.; Wiley-VCH: Weinheim, Germany, 2006; pp. 476–528. [Google Scholar]
- Meier, H. Oligo- and poly(2,5-thienylene-ethynylene)s. In Targets in Heterocyclic Systems; Attanasi, O.A., Spinelli, D., Eds.; Societa Chimica Italiana: Urbino, Italy, 2008; pp. 390–413. [Google Scholar]
- Lehmann, M. Star mesogens (hekates) − tailor-made molecular for programming supramolecular functionality. Chem. Eur. J. 2009, 15, 3638–3651. [Google Scholar] [CrossRef] [PubMed]
- Zettler, F.; Hausen, H.D.; Hess, H. Die Kristall- und Molekülstruktur des Triphenylborans. J. Organomet. Chem. 1974, 72, 157–162. [Google Scholar] [CrossRef]
- Zollinger, H. Color Chemistry, 3rd ed.; Wiley-VCH: Weinheim, Germany, 2003; pp. 101–122. [Google Scholar]
- Sobolev, A.N.; Belsky, V.K.; Romm, I.P.; Chernikowa, N.Y.; Guryanova, E.N. Structural investigation of the triaryl derivatives of the group V elements. IX Structure of triphenylamine, C18H15N. Acta Crystallogr. Sect. C: Cryst. Struct. Commun. 1985, 41, 967–971. [Google Scholar] [CrossRef]
- Meier, H.; Schollmeyer, D.; University of Mainz, Mainz, Germany. Unpublished work. 2010.
- Dewar, M.J.S. A molecular orbital theory of organic chemistry. II. The structure of mesomeric systems. J. Am. Chem. Soc. 1952, 74, 3345–3350. [Google Scholar] [CrossRef]
- Kim, S.; Oehlhof, A.; Beile, B.; Meier, H. Donor-acceptor-substituted oligo(1,4-phenylene)s. Helv. Chim. Acta 2009, 92, 1023–1033. [Google Scholar] [CrossRef]
- Van Averbeke, B.; Belgonne, D. Conformational effects on excitation transport along conjugated polymer chains. J. Phys. Chem. A. 2009, 113, 2677–2682. [Google Scholar] [CrossRef] [PubMed]
- Magyar, R.J.; Tretiak, S.; Gao, Y.; Wang, H.-L.; Shreve, A.P. Chem. Phys. Lett. 2005, 401, 149–156.
- Li, N.; Jia, K.; Wang, S.; Xia, A. Theoretical study of spectroscopic properties of dimethoxy-p-phenylene-ethynylene oligomers: Planarization of the conjugated backbone. J. Phys. Chem. A. 2007, 111, 9393–9398. [Google Scholar] [CrossRef] [PubMed]
- Meier, H.; Stalmach, U.; Kolshorn, H. Effective conjugation length and UV/Vis spectra of oligomers. Acta Polymer. 1997, 48, 379–384. [Google Scholar] [CrossRef]
- Mühling, B. Dissertation, Mainz, 2004.
- Gierschner, J.; Cornil, J.; Egelhaaf, H.-J. Optical band gaps of π-conjugated organic materials at the polymer limit: Experiment and theory. Adv. Mater. 2007, 19, 173–191. [Google Scholar] [CrossRef]
- Bednarz, M.; Reineker, P.; Mena-Osteritz, E.; Bäuerle, P. Theoretical study of the size confinement effect in linear π-conjugated oligomers. Chem. Phys. 2007, 342, 191–200. [Google Scholar] [CrossRef]
- Meier, H.; University of Mainz, Mainz, Germany. Unpublished work. 2010.
- Morton, A.A.; Emerson, W.S. Condensations by sodium. XI. Trimethoxytrixenylcarbinol and comparison of colors of some carbonium salts in this series. J. Am. Chem. Soc. 1938, 60, 284–285. [Google Scholar] [CrossRef]
- See also: Ryu, J.-H.; Kim, H.-J.; Huang, E.L.; Lee, M. Self-assembling molecular dumbbells: From nanohelices to nanocapsules triggered by guest intercalation. Angew. Chem. Int. Ed. 2006, 45, 5304–5307. [Google Scholar] [CrossRef]
- Theilacker, W.; Berger, W.; Popper, P. Untersuchungen zur Farbstofftheorie: Triarylmethanfarbstoffe der Biphenylreihe, II. Chem. Ber. 1956, 89, 970–983. [Google Scholar] [CrossRef]
- Hellwinkel, D.; Fritsch, H. Phenylvinylog erweiterte Triphenylmethylium-Systeme. Chem. Ber. 1989, 122, 2351–2360. [Google Scholar] [CrossRef]
- Arbez-Gindre, C.; Screttas, C.G.; Fiorini, C.; Schmidt, C.; Nunzi, J.-M. Organolithium reagents bearing nonlinear optical chromophores. Synthesis of triarylmethane dyes. Tetrahedron Lett. 1999, 40, 7413–7416. [Google Scholar] [CrossRef]
- Sengupta, S.; Sodhukhan, S.K. Trivinylogs of Crystal Violet: Synthesis and absorption properties of new near-IR dyes. J. Chem. Soc., Perkin Trans 1 2000, 4332–4334. [Google Scholar]
- Sengupta, S. An octupolar near-IR dye with triphenylamine donors: Preparation and absorption properties. Tetrahedron Lett. 2003, 44, 307–310. [Google Scholar] [CrossRef]
- Villalonga-Barber, C.; Heele, B.R.; Kovac, V.; Micha-Screttas, M.; Screttas, C.G. New stable, isolable triarylmethyl based dyes absorbing in the near infrared. J. Organometal. Chem. 2006, 691, 2785–2792. [Google Scholar] [CrossRef]
- Meier, H.; Kim, S. Methylium ions with OPV chains − new NIR dyes. Eur. J. Org. Chem. 2001, 1163–1167. [Google Scholar] [CrossRef]
- Kim, S.; Kalbitz, H.; Hillmann, S.; Meier, H. Tris[oligo(1,4-phenylenevinylene)]methylium dyes. Eur. J. Org. Chem. 2009, 1976–1983. [Google Scholar] [CrossRef]
- Jamaguchi, S.; Shirazaka, T.; Tamao, K. Tridurylboranes extended by three arylethynyl groups as a new family of boron-based π-electron systems. Org. Lett. 2000, 2, 4129–4132. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.-L.; Song, D.; Wang, S. Blue luminescent three-coordinate organoboron compounds with a 2,2′-dipyridylamino functional group. J. Org. Chem. 2003, 68, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.-L.; Moran, M.J.; Yuan, Y.-Y.; Lu, Z.H.; Wang, S. (1-Naphthyl)phenylamino functionalized three-coordinate organoboron compounds: Syntheses, structures, and applications in OLEDs. J. Mater. Chem. 2005, 15, 3326–3333. [Google Scholar] [CrossRef]
- Bhaskar, A.; Ramakrishna, G.; Lu, Z.; Twieg, R.; Hales, J.M.; Hagan, D.J.; Van Stryland, E.; Goodson, T., III. Investigation of two-photon absorption properties in branched alkene and alkyne chromophores. J. Am. Chem. Soc. 2006, 128, 11840–11849. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.-X.; Tao, X.-T.; Sun, Y.-H.; Wang, C.-K.; Xu, G.-B.; Yang, J.-X.; Ren, Y.; Zhao, X.; Wu, Y.-Z.; Yu, X.-Q.; Jiang, M.-H. Synthesis and nonlinear optical properties of novel multi-branched two-photon polymerization initiators. J. Mater. Chem. 2004, 14, 2995–3000. [Google Scholar] [CrossRef]
- Horner, L.; Hoffmann, H.; Wippel, H. G. Phosphororganische Verbindungen XII. Phosphinoxyde als Olefinierungsreagenzien. Chem. Ber. 1958, 91, 61–63. [Google Scholar] [CrossRef]
- Yoo, J.; Yang, S.K.; Jeong, M.-Y.; Anh, H.C.; Jeon, S.-J.; Cho, B.R. Bis-1,4-(p-diarylaminostyryl)-2,5-dicyanobenzene derivatives with large two-photon absorption cross- sections. Org. Lett. 2003, 5, 645–648. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.J.; Kim, D.Y.; Kim, C.H.; Jeong, M.-Y.; Lee, S.K.; Jeon, S.-J.; Cho, B.R. Triphenylamine derivatives with large two-photon cross-sections. Org. Lett. 2004, 6, 1389–1392. [Google Scholar] [CrossRef] [PubMed]
- Le Droumaguet, C.; Mongin, O.; Werts, M.H.V.; Blanchard-Desce, M. Towards “smart” multiphoton fluorophores: Strongly solvatochromic probes for two-photon sensing of micropolarity. Chem. Commun. 2005, 2802–2804. [Google Scholar] [CrossRef]
- Katan, C.; Terenziani, F.; Mongin, O.; Werts, M.H.V.; Porrès, L.; Pons, T.; Mertz, J.; Tretiak, S.; Blanchard-Desce, M. Effects of (multi)branching of dipolar chromophores on photophysical properties and two-photon absorption. J. Phys. Chem. A 2005, 109, 3024–3037. [Google Scholar] [CrossRef] [PubMed]
- Lim, B.; Huang, J.-T.; Kim, J.Y.; Ghim, J.; Vak, D.; Noh, Y.-Y.; Lee, S.-H.; Lee, K.; Heeger, A.J.; Kim, D.-Y. Synthesis of a new cross-linkable perfluorcyclobutane-based hole-transport material. Org. Lett. 2006, 8, 4703–4706. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, H.; Ohnishi, K.; Shirota, Y.; Suita, Y. Tri(p-terphenyl-4-yl)amine as a novel blue-emitting material for organic electroluminescent devices. Synthetic Metals 1997, 91, 243–245. [Google Scholar] [CrossRef]
- Kinoshita, M.; Shirota, Y. 1,3-Bis[5-(dimesitylboryl)thiophen-2-yl]benzene as novel family of electron-transporting hole blockers for organic electroluminescent devices. Chem. Lett. 2001, 30, 614–615. [Google Scholar] [CrossRef]
- Kinoshita, M.; Kita, H.; Shirota, Y. A novel family of boron-containing hole-blocking amorphous molecular materials for blue- and blue-violet-emitting organic electroluminescent devices. Adv. Funct. Mater. 2002, 12, 780–786. [Google Scholar] [CrossRef]
- Lin, Y.; Williams, D.E. 1,3,5-Triphenylbenzene. Acta Crystallogr. B 1975, B 31, 318–320. [Google Scholar] [CrossRef]
- Weber, E.; Hecker, M.; Koepp, E.; Orlia, W.; Czugler, M. New trigonal lattice hosts: stoicheiometric crystal inclusions of laterally trisubstituted benzenes-X-ray crystal structure of 1,3,5-tris-(4- carboxyphenyl)benzene dimethylformamide. J. Chem. Soc. Perk. Trans. 2 1988, 7, 1251–1257. [Google Scholar] [CrossRef]
- Vergadou, V.; Pistolis, G.; Michaelides, A.; Varvounis, G.; Siskos, M.; Boukos, N.; Skoulika, S. Self-organization of four symmetric tri-phenylbenzene derivatives. Cryst. Growth Des. 2006, 6, 2486–2492. [Google Scholar] [CrossRef]
- Meier, H.; Karpouk, E.; Lehmann, M.; Schollmeyer, D.; Enkelmann, V. Guest-host systems of 1,3,5-tristyrylbenzenes. Z. Naturforsch. B 2003, 58, 775–781. [Google Scholar]
- Ponzini, F.; Zagha, R.; Hardcastle, K.; Siegel, J. S. Phenyl/pentafluorophenyl interactions and the generation of ordered mixed crystals: sym-triphenethynylbenzene and sym-tris(perfluorophenethynyl)benzene. Angew. Chem. Int. Ed. 2000, 39, 2323–2325. [Google Scholar] [CrossRef]
- Schmidt, B.; Rinke, M.; Güsten, H. Photophysical properties of 1,3,5-tris-(p-oligophenylene)benzenes. J. Photochem. Photobiol. A 1989, 49, 131–135. [Google Scholar] [CrossRef]
- Juriew, J.; Skorochodowa, T.; Merkuschew, J.; Winter, W.; Meier, H. A simple route to a new type of cyclophane. Angew. Chem. Int. Ed. 1981, 20, 269–270. [Google Scholar] [CrossRef]
- Meier, H.; Zertani, R.; Noller, K.; Oelkrug, D.; Krabichler, G. Investigations on the fluorescence of styryl-substituted benzenes. Chem. Ber. 1986, 119, 1716–1724. [Google Scholar] [CrossRef]
- Dyck, R.H.; McClure, D.S. Ultraviolet spectra of stilbene, p-monohalogen stilbenes and azobenzene and trans to cis photoisomerisation process. J. Chem. Phys. 1962, 36, 2326–2345. [Google Scholar] [CrossRef]
- Gase, R. Untersuchungen über die Ultraviolett-Spektren von Stilben und Tolanderivaten bei 77 °K. Z. Phys. Chem.-Leipzig 1968, 238, 233–240. [Google Scholar]
- Yamaguchi, Y.; Ochi, T.; Miyamura, S.; Tanaka, T.; Kobayashi, S.; Wakamiya, T.; Matsubara, Y.; Yoshida, Z. Rigid molecular architectures that comprise a 1,3,5-trisubstituted benzene core and three oligoaryleneethynylene arms: light-emitting characteristics and π-conjugation between the arms. J. Am. Chem. Soc. 2006, 128, 4504–4505. [Google Scholar] [CrossRef] [PubMed]
- Khotina, I.A.; Izumrudov, V.; Tchebotareva, N.; Rusanov, A.L. Aromatic symmetric cyclotrimers and poly(arylenevinylene)s with branched aromatic substituents in vinylene groups. Synthesis and optical properties. Macromol. Chem. Phys. 2001, 202, 2360–2366. [Google Scholar] [CrossRef]
- Lehmann, M. Synthese und Struktur-Eigenschafts-Beziehungen von Stilbenoiden Dendrimeren und Sternverbindungen. Dissertation, Mainz, 1999. [Google Scholar]
- Siegrist, A.E.; Liechti, P.; Meyer, H.R.; Weber, K. Anil synthesis 3. Synthesis of styryl derivatives from methyl substituted carbocyclic aromatic compounds. Helv. Chim. Acta 1969, 52, 2521–2554. [Google Scholar] [CrossRef]
- Accorsi, G.; Armaroli, N.; Eckert, J.F.; Nierengarten, J. Functionalization of [60]fullerene with new light-collecting oligophenylenevinylene-terminated dendritic wedges. Tetrahedron Lett. 2002, 43, 65–68. [Google Scholar] [CrossRef]
- Derkowska, B.; Mulatier, J.; Fuks, I.; Sahraoui, B.; Phu, X.; Andraud, C. Third-order optical nonlinearities in new octupolar molecules and their dipolar subunits. J. Op. Soc. Am. B 2001, 18, 610–616. [Google Scholar] [CrossRef]
- Winter, W.; Langjahr, U.; Meier, H.; Merkuschew, J.; Juriew, J. Photochemistry of 1,3,5-tristyrylbenzene. Chem. Ber. 1984, 117, 2452–2463. [Google Scholar] [CrossRef]
- Meier, H.; Lehmann, M. Stilbenoid Dendrimers-Preparation and Properties. In Encyclopedia of Nanoscience and Nanotechnology; Nalva, H.S., Ed.; American Scientific Publishers: Stevensons Ranch, CA, USA, 2004; Volume 10, pp. 95–106. [Google Scholar]
- Peruga, A.; Mata, J.; Sainz, D.; Peris, E. Facile synthesis of bidimensional ferrocenyl-based branched oligomers by palladium-catalyzed coupling reactions. J. Organomet. Chem. 2001, 637-639, 191–197. [Google Scholar] [CrossRef]
- González, A.; Segura, J.L.; Martı́n, N. New planar and soluble tris-tetrathiafulvalene derivatives with threefold-symmetry. Tetrahedron Lett. 2000, 41, 3083–3086. [Google Scholar] [CrossRef]
- Fukutome, H.; Takahashi, A.; Ozaki, M. Design of conjugated polymers with polaronic ferromagnetism. Chem. Phys. Lett. 1987, 133, 34–38. [Google Scholar] [CrossRef]
- Kaisako, D.A.; Chang, W.H.; Dougherty, D.A. Novel magnetic properties of a doped organic polymer-a possible prototype for a polaronic ferromagnet. J. Am. Chem. Soc. 1991, 113, 2764–2766. [Google Scholar] [CrossRef]
- Lehmann, M.; Meier, H.; Gherghel, L.; Kolb, U.; Chemnitz University of Technology, Chemnitz, Germany. Unpublished work. 2010.
- Baumgarten, M.; Yuksel, T.; Caparros, D.; Karabunarliev, S. Novel high spin molecules and intermolecular aggregates. Mol. Cryst. Liq. Cryst. A 1999, 335, 443–452. [Google Scholar] [CrossRef]
- Wirth, H.; Kern, W.; Schmitz, E. Synthese und Eigenschaften von verzweigten oligophenylenen, die sich vom 1,3,5-Triphenylbenzol ableiten.14. Makromol. Chem. 1963, 68, 69–99. [Google Scholar] [CrossRef]
- Kotha, S.; Kashinath, D.; Lahiri, K.; Sunoj, R.B. Synthesis of C3-symmetric nano-sized polyaromatic compounds by trimerization and Suzuki-Miyaura cross-coupling reactions. Eur. J. Org. Chem. 2004, 4003–4013. [Google Scholar] [CrossRef]
- Brunel, J.; Ledoux, I.; Zyss, J.; Blanchard-Desce, M. Propeller-shaped molecules with giant off-resonance optical nonlinearities. Chem. Commun. 2001, 923–924. [Google Scholar] [CrossRef]
- Li, J.; Ambroise, A.; Yang, S.I.; Diers, J.R.; Seth, J.; Wack, C.R. Template-directed synthesis, excited-state photodynamics, and electronic communication in a hexameric wheel of porphyrins. J. Am. Chem. Soc. 1999, 121, 8927–8940. [Google Scholar] [CrossRef]
- Kiang, Y.H.; Gardner, G.B.; Lee, S.; Xu, Z.; Lobkovsky, E.B. Variable pore size, variable chemical functionality, and an example of reactivity reactivity within porous phenylacetylene silver salts. J. Am. Chem. Soc. 1999, 121, 8204–8215. [Google Scholar] [CrossRef]
- Meier, H. The photochemistry of stilbenoid compounds and their role in materials technology. Angew. Chem. Int. Ed. 1992, 31, 1399–1420. [Google Scholar] [CrossRef]
- Jung, I.; Lee, T.; Kang, S.O.; Ko, J. Hydrostannation of diyne and triyne p-electron bridges: Efficient Stille cross-coupling of 1,3,5-tris[(E)-2-(tributylstannyl)vinyl]benzene. Synthesis 2005, 986–992. [Google Scholar]
- Meier, H.; Lehmann, M. Stilbenoide Dendrimere. Angew. Chem. 1998, 110, 666–669. [Google Scholar] [CrossRef]
- Lehmann, M.; Schartel, B.; Hennecke, M.; Meier, H. Dendrimers consisting of stilbene or distyrylbenzene building blocks synthesis and stability. Tetrahedron 1999, 55, 13377–13394. [Google Scholar] [CrossRef]
- Jandke, M.; Strohriegl, P.; Berleb, S.; Werner, E.; Brutting, W. Phenylquinoxaline polymers and low molar mass glasses as electron-transport materials in organic light-emitting diodes. Macromolecules 1998, 31, 6434–6443. [Google Scholar] [CrossRef]
- Briel, O.; Fehn, A.; Beck, W. Hydrocarbon bridged metal complexes XLV. Dinuclear polyene-bridged Fischer carbene complexes and a star-shaped benzene-bridged tris(ferrocenyl-decapentaenyl) compound. J. Organometal. Chem. 1999, 578, 247–251. [Google Scholar]
- Andraud, C.; Zabulon, T.; Collet, A.; Zyss, J. Nonlinear optical properties of polyenoctupoles: a multipolar tensorial quantum analysis. Chem. Phys. 1999, 245, 243–261. [Google Scholar] [CrossRef]
- The 3,4-alkoxyphenyl substituted derivatives were presumably also prepared via this method. In the cited references of Andraud et al. (reference 74) no description of the final threefold coupling steps could be found.
- Eisler, S.; Tykwinski, R.R. Migrating alkynes in vinylidene carbenoids: An unprecedented route to polyynes. J. Am. Chem. Soc. 2000, 122, 10736–10737. [Google Scholar] [CrossRef]
- Eisler, S.; Chahal, N.; McDonald, R. Alkyne migration in alkylidene carbenoid species: A new method of polyyne synthesis. Chem. Eur. J. 2003, 9, 2542–2550. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.A.; Zweifel, G. A Convenient synthesis of 1-trimethylsilyl-1,3-diynes and their conversion into terminal 1,3-diynes. Synthesis 1983, 128–130. [Google Scholar] [CrossRef]
- Rubin, Y.; Parker, T.C.; Khan, S.I.; Holliman, C.L. Precursors to endohedral metal fullerene complexes: Synthesis and X-ray structure of a flexible acetylenic cyclophane C60H18. J. Am. Chem. Soc. 1996, 118, 5308–5309. [Google Scholar] [CrossRef]
- Chin, C.S.; Kim, M.; Lee, H.; Noh, S.; Ok, K.M. Regio-and stereoselective C−C bond formation between alkynes: Synthesis of linear dienynes from alkynes. Organometallics 2002, 21, 4785–4793. [Google Scholar] [CrossRef]
- Kaafarani, B.R.; Wex, B.; Wang, F.; Catanescu, O.; Chien, L.C. Synthesis of highly fluorescent Y-enyne dendrimers with four and six arms. J. Org. Chem. 2003, 68, 5377–5380. [Google Scholar] [CrossRef] [PubMed]
- Lindentan, S.V.; Struchkov, Y.T.; Shklouer, V.E.; Khotina, I.A.; Teplyakou, M.M.; Korshak, V.V. Structural study of compounds modelling elementary polymer units, 3. Molecular and crystal structure of 1,3,5-tris[4‘-(C-o-carboranyl)biphenyl-4-yl]benzene. Makromol. Chem. 1987, 188, 1511–1519. [Google Scholar] [CrossRef]
- Zeng, F.W.; Zimmerman, S.C.; Kolotuchin, S.V.; Reichert, D.E.C.; Ma, Y.G. Supramolecular polymer chemistry: design, synthesis, characterization, and kinetics, thermodynamics, and fidelity of formation of self-assembled dendrimers. Tetrahedron 2002, 58, 825–843. [Google Scholar] [CrossRef]
- Sun, D.; Ke, Y.; Mattox, T.M.; Parkin, S.; Zhou, H. Stability and porosity enhancement through concurrent ligand extension and secondary building unit stabilization. Inorg. Chem. 2006, 45, 7566–7568. [Google Scholar] [CrossRef] [PubMed]
- Schmittel, M.; He, B.; Fan, J.; Bats, J.; Engeser, M.; Schlosser, M.; Deiseroth, H. Cap for Copper(I) ions! Metallosupramolecular solid and solution state structures on the basis of the dynamic tetrahedral [Cu(phenAr2)(py)2]+ motif. Inorg. Chem. 2009, 48, 8192–8200. [Google Scholar] [CrossRef] [PubMed]
- Belser, P.; Vonzelewsky, A.; Frank, M.; Seel, C.; Vögtle, F.; DeCola, L.; Barigelletti, F.; Balzani, V. Supramolecular Ru and or Os complexes of tris(bipyridine) bridging ligands-synthesis, absorption-spectra, luminescence properties, electrochemical-behavior, intercomponent energy and electron transfer. J. Am. Chem. Soc. 1993, 115, 4076–4086. [Google Scholar] [CrossRef]
- Kotha, S.; Shah, V.R. Synthesis of C3-symmetric and C4-symmetric amino acid derivatives via Suzuki–Miyaura cross-coupling reaction. Amino Acids 2008, 35, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.P.; Tao, Y.; D'iorio, M.; Li, Y.N.; Ding, J.F.; Day, M. Pure deep blue light-emitting diodes from alternating fluorene/carbazole copolymers by using suitable hole-blocking materials. Macromolecules 2004, 37, 2442–2449. [Google Scholar] [CrossRef]
- Beavington, R.; Frampton, M.; Lupton, J.; Burn, P.; Samuel, I. The Effect of core delocalization on intermolecular interactions in conjugated dendrimers. Adv. Funct. Mater. 2003, 13, 211–218. [Google Scholar] [CrossRef]
- Brunel, J.; Mongin, O.; Jutand, A.; Ledoux, I.; Zyss, J.; Blanchard-Desce, M. Propeller-shaped octupolar molecules derived from triphenylbenzene for nonlinear optics: Synthesis and optical studies. Chem. Mater. 2003, 15, 4139–4148. [Google Scholar] [CrossRef]
- Simpson, C.; Mattersteig, G.; Martin, K.; Gherghel, L.; Bauer, R.; Rader, H.; Müllen, K. Nanosized molecular propellers by cyclodehydrogenation of polyphenylene dendrimers. J. Am. Chem. Soc. 2004, 126, 3139–3147. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.; Willi, T.; DiPietro, V.; Hedrick, J.; Miller, H.; Hedrick, J. First example of a nanoporous high-temperature polymer thermoset: Eluding transition-time-temperature constraints associated with organic thermosets. Adv. Mater. 2004, 16, 1525–1529. [Google Scholar] [CrossRef]
- Meier, H.; Lehmann, M.; Kolb, U. Stilbenoid dendrimers. Chem. Eur. J. 2000, 6, 2462–2469. [Google Scholar] [CrossRef] [PubMed]
- Meier, H.; Lehmann, M.; Holst, H.C.; Schwöppe, D. Star-shaped conjugated compounds forming nematic discotic systems. Tetrahedron 2004, 60, 6881–6888. [Google Scholar] [CrossRef]
- Lehmann, M. Synthese und Struktur-Eigenschafts-Beziehungen von Stilbenoiden Sternstrukturen. Diplomarbeit, Mainz, 1995. [Google Scholar]
- Lehmann, M.; Rumpf, N.; Chemnitz University of Technology, Chemnitz, Germany. Unpublished work. 2010.
- Lehmann, M.; Fischbach, I.; Spiess, H.W.; Meier, H. Photochemistry and mobility of stilbenoid dendrimers in their neat phases. J. Am. Chem. Soc. 2004, 126, 772–784. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.W.; Dai, L. Luminescent amphiphilic dendrimers with oligo(p-phenylene vinylene) core branches and oligo(ethylene oxide) terminal chains: syntheses and stimuli-responsive properties. J. Mater. Chem. 2007, 17, 364–371. [Google Scholar] [CrossRef]
- Stuhr-Hansen, N.; Christensen, J.B.; Harrit, N.; Bjørnholm, T. Novel synthesis of protected thiol end-capped stilbenes and oligo(phenylenevinylene)s (OPVs). J. Org. Chem. 2003, 68, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.R.; Lee, S.J.; Lee, S.H.; Son, K.H.; Kim, Y.H.; Doo, J.; Lee, G.J.; Kang, T.I.; Lee, Y.K.; Cho, M.; Jeon, S. Octupolar Crystals for Nonlinear Optics: 1,3,5-Trinitro-2,4,6-tris(styryl)benzene Derivatives. Chem. Mater. 2001, 13, 1438–1440. [Google Scholar] [CrossRef]
- Godt, A.; Franzen, C.; Veit, S.; Enkelmann, V.; Pannier, M. EPR probes with well-defined, long distances between two or three unpaired electrons. J. Org. Chem. 2000, 65, 7575–7582. [Google Scholar] [CrossRef] [PubMed]
- Polyhach, Y.; Godt, A.; Bauer, C.; Jeschke, G. Spin pair geometry revealed by high-field DEER in the presence of conformational distributions. J. Mag. Res. 2007, 185, 118–129. [Google Scholar] [CrossRef]
- Jeschke, G.; Sajid, M.; Schulte, M.; Godt, A. Three-spin correlations in double electron–electron resonance. Phys. Chem. Chem. Phys. 2009, 11, 6580–6591. [Google Scholar] [CrossRef] [PubMed]
- Kiang, Y.H.; Gardner, G.B.; Lee, S.; Xu, Z.T. Porous siloxane linked phenylacetylene nitrile silver salts from solid state dimerization and low polymerization. J. Am. Chem. Soc. 2000, 122, 6871–6883. [Google Scholar] [CrossRef]
- Xu, Z.; Lee, S.; Kiang, Y.H.; Mallik, A.B.; Tsomaia, N. A cross-linked large channel organic coordination solid. Adv. Mater. 2001, 13, 637–641. [Google Scholar] [CrossRef]
- Mu, Z.; Shu, L.; Fuchs, H.; Mayor, M.; Chi, L. Two dimensional chiral networks emerging from the Aryl−F··· H hydrogen-bond-driven self-assembly of partially fluorinated rigid molecular structures. J. Am. Chem. Soc. 2008, 130, 10840–10841. [Google Scholar] [CrossRef] [PubMed]
- An, D.L.; Chen, Q.; Zhang, Z.Y.; Yan, H.; Fang, J.K. Selective tandem inter/intramolecular eglington coupling for chiral cyclophyne synthesis. Synth. Commun. 2009, 39, 3092–3100. [Google Scholar] [CrossRef]
- Rucareanu, S.; Mongin, O.; Schuwey, A.; Hoyler, N.; Gossauer, A.; Amrein, W.; Hediger, H.U. Supramolecular assemblies between macrocyclic porphyrin hexamers and star-shaped porphyrin arrays. J. Org. Chem. 2001, 66, 4973–4988. [Google Scholar] [CrossRef] [PubMed]
- Brousseau, L.C.; Novak, J.P.; Marinakos, S.M.; Feldheim, D.L. Assembly of phenylacetylene-bridged gold nanocluster dimers and trimers. Adv. Mater. 1999, 11, 447–449. [Google Scholar] [CrossRef]
- Novak, J.P.; Feldheim, D.L. Assembly of phenylacetylene-bridged silver and gold nanoparticle arrays. J. Am. Chem. Soc. 2000, 122, 3979–3980. [Google Scholar] [CrossRef]
- Morin, J.F.; Sasaki, T.; Shirai, Y.; Guerrero, J.M.; Tour, J.M. Synthetic routes toward carborane-wheeled nanocars. J. Org. Chem. 2007, 72, 9481–9490. [Google Scholar] [CrossRef] [PubMed]
- Kayser, B.; Altman, J.; Beck, W. Alkyne bridged alpha-amino acids by palladium mediated coupling of alkynes with N-t-Boc-4-iodophenylalanine methyl ester. Tetrahedron 1997, 53, 2475–2484. [Google Scholar] [CrossRef]
- Kayser, B.; Altman, J.; Noth, H.; Knizek, J.; Beck, W. Metal complexes of biologically important ligands. CVIII. Metal complexes of alkyne-bridged alpha-amino acids. Eur. J. Inorg. Chem. 1998, 1791–1798. [Google Scholar] [CrossRef]
- Beletskaya, I.; Chuchuryukin, A.; van Koten, G.; Dijkstra, H.; van Klink, G.; Kashin, A.; Nefedov, S.; Eremenko, I. Synthesis and catalytic properties of di-and trinuclear palladium complexes with PCP-pincer ligands. Russ. J. Org. Chem. 2003, 39, 1268–1281. [Google Scholar] [CrossRef]
- Pesak, D.J.; Moore, J. Columnar liquid crystals from shape-persistent dendritic molecules. Angew. Chem. Int. Ed. 1997, 36, 1636–1639. [Google Scholar] [CrossRef]
- Bushey, M.; Nguyen, T.; Nuckolls, C. Synthesis, self-assembly, and switching of one-dimensional nanostructures from new crowded aromatics. J. Am. Chem. Soc. 2003, 125, 8264–8269. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.; Martel, R.; Avouris, P.; Bushey, M.; Brus, L.; Nuckolls, C. Molecular interactions in one-dimensional organic nanostructures. J. Am. Chem. Soc. 2004, 126, 5234–5242. [Google Scholar] [CrossRef] [PubMed]
- Mongin, O.; Papamicael, C.; Hoyler, N.; Gossauer, A. Modular synthesis of benzene-centered porphyrin trimers and a dendritic porphyrin hexamer. J. Org. Chem. 1998, 63, 5568–5580. [Google Scholar] [CrossRef]
- Brodard, P.; Matzinger, S.; Vauthey, E.; Mongin, O.; Papamicael, C.; Gossauer, A. Investigations of electronic energy transfer dynamics in multiporphyrin arrays. J. Phys. Chem. A 1999, 103, 5858–5870. [Google Scholar] [CrossRef]
- Morandeira, A.; Vauthey, E.; Schuwey, A.; Gossauer, A. Ultrafast excited state dynamics of tri- and hexaporphyrin arrays. J. Phys. Chem. A 2004, 108, 5741–5751. [Google Scholar] [CrossRef]
- Rodríguez, J.G.; Tejedor, J.L. Carbon dendron nano-chains with π-extended conjugation based on end-capped N,N-dimethylamino in linear 1,4-phenylethynyl or in 1,5-naphthylethynyl subunits: fluorescence analysis. Tetrahedron 2005, 61, 2047–2054. [Google Scholar] [CrossRef]
- Tao, C.H.; Zhu, N.; Yam, V.W. Room-temperature phosphorescence and energy transfer in luminescent multinuclear platinum(II) complexes of branched alkynyls. Chem. Eur. J. 2005, 11, 1647–1657. [Google Scholar] [CrossRef] [PubMed]
- Shirai, Y.; Zhao, Y.; Cheng, L.; Tour, J.M. Facile synthesis of multifullerene-OPE hybrids via in situ ethynylation. Org. Lett. 2004, 6, 2129–2132. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Shirai, Y.; Slepkov, A.D.; Cheng, L.; Alemany, L.B.; Sasaki, T.; Hegmann, F.A.; Tour, J.M. Synthesis, spectroscopic and nonlinear optical properties of multiple [60]fullerene-oligo(p-phenylene ethynylene) hybrids. Chem. Eur. J. 2005, 11, 3643–3658. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Yasuda, S.; Sakaguchi, T.; Miya, M. The third-order optical non-linearity of the phenylethynyl-substituted benzene system. J. Chem. Soc. Chem. Commun. 1995, 55–56. [Google Scholar] [CrossRef]
- Cifuentes, M.P.; Powell, C.E.; Morrall, J.P.; McDonagh, A.M.; Lucas, N.T.; Humphrey, M.G.; Samoc, M.; Houbrechts, S.; Asselberghs, I.; Clays, K.; Persoons, A.; Isoshima, T. Electrochemical, spectroelectrochemical, and molecular quadratic and cubic nonlinear optical properties of alkynylruthenium dendrimers. J. Am. Chem. Soc. 2006, 128, 10819–10832. [Google Scholar] [CrossRef] [PubMed]
- Cifuentes, M.P.; Powell, C.E.; Humphrey, M.G.; Heath, G.A.; Samoc, M.; Luther-Davies, B. Organometallic complexes for nonlinear optics. 24. Reversible electrochemical switching of nonlinear absorption. J. Phys. Chem. A 2001, 105, 9625–9627. [Google Scholar] [CrossRef]
- Tao, C.H.; Yang, H.; Zhu, N.; Yam, V.W.W.; Xu, S.J. Branched luminescent multinuclear platinum (II) alkynyl complexes: candidates for efficient two-photon induced luminescence. Organometallics 2008, 27, 5453–5458. [Google Scholar] [CrossRef]
- Novak, J.P.; Brousseau, L.C., III; Vance, F.W.; Johnson, R.C. Nonlinear optical properties of molecularly bridged gold nanoparticle arrays. J. Am. Chem. Soc. 2000, 122, 12029–12030. [Google Scholar] [CrossRef]
- Tour, J.M.; Kozaki, M.; Seminario, J.M. Molecular scale electronics: a synthetic/computational approach to digital computing. J. Am. Chem. Soc. 1998, 120, 8486–8493. [Google Scholar] [CrossRef]
- Fuks-Janczarek, I.; Kityk, I.; Berdowski, J.; Sahraoui, B.; Andraud, C. Physical origin of the third-order nonlinear optical properties of polyenes. J. Modern Opt. 2005, 52, 1933–1945. [Google Scholar] [CrossRef]
- Liu, C.; Speyer, J.; Ovchinnikov, I.; Neuhauser, D. Nonlinear signal mixing in a three-terminal molecular wire. J. Chem. Phys. 2007, 126, 024705. [Google Scholar] [CrossRef] [PubMed]
- Kivala, M.; Boudon, C.; Gisselbrecht, J.; Seiler, P.; Gross, M.; Diederich, F. Charge-transfer chromophores by cycloaddition-retro-electrocyclization: Multivalent systems and cascade reactions. Angew. Chem. Int. Ed. 2007, 46, 6357–6360. [Google Scholar] [CrossRef]
- Kivala, M.; Stanoeva, T.; Michinobu, T.; Frank, B.; Gescheidt, G.; Diederich, F. One-electron-reduced and -oxidized stages of donor-substituted 1,1,4,4-tetracyanobuta-1,3-dienes of different molecular architectures. Chem. Eur. J. 2008, 14, 7638–7647. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, E.; Maris, T.; Arseneault, P.; Maly, K.; Wuest, J. Structural features in crystals of derivatives of benzene with multiple contiguous phenyl substituents. Cryst. Growth Des. 2010, 10, 648–657. [Google Scholar] [CrossRef]
- Li, Z.; Wong, M.; Tao, Y. Two-dimensional oligoarylenes: synthesis and structure–properties relationships. Tetrahedron 2005, 61, 5277–5285. [Google Scholar] [CrossRef]
- Samori, S.; Tojo, S.; Fujitsuka, M.; Spitler, E.L.; Haley, M.M.; Majima, T. Donor-acceptor-substituted tetrakis(phenylethynyl)benzenes as emissive molecules during pulse radiolysis in benzene. J. Org. Chem. 2007, 72, 2785–2793. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.J.; Marsden, J.A.; Haley, M.M. Synthesis and optical properties of donor-acceptor tetrakis(phenylethynyl)benzenes. Synlett 2004, 165–168. [Google Scholar]
- Marsden, J.A.; Miller, J.J.; Shirtcliff, L.D.; Haley, M.M. Structure-property relationships of donor/acceptor-functionalized tetrakis(phenylethynyl)benzenes and bis(dehydrobenzoannuleno) benzenes. J. Am. Chem. Soc. 2005, 127, 2464–2476. [Google Scholar] [CrossRef] [PubMed]
- Ahna, K.; Ryua, G.Y.; Younb, S.; Shin, D. The conjugation effects on the luminescence properties of oligophenylenes for the OLED. Mater. Sci Eng. 2004, C24, 163–165. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Matsubara, Y.; Ochi, T.; Wakamiya, T.; Yoshida, Z. How the pi conjugation length affects the fluorescence emission efficiency. J. Am. Chem. Soc. 2008, 130, 13867–13869. [Google Scholar] [CrossRef] [PubMed]
- Drehfahl, G.; Kühmstedt, R.; Oswald, H.; Hörhold, H. Oligomere als Modelle für Poly-p-xylyliden. Makro. Chem. 1970, 131, 89–103. [Google Scholar] [CrossRef]
- Chaitanya, G.K.; Thomas, A.; Sinu, C.R.; Francis, B.; Subhashchandran, K.P.; Ramakrishna, K.; Bhanuprakash, K. Insight into the electron delocalization in phenylacetylenes and phenylvinylenes: An NBO analysis. Indian J. Chem. A 2008, 47, 1171–1180. [Google Scholar]
- Ojima, J.; Kakumi, H.; Kitatani, K.; Wada, K.; Ejiri, E.; Nakada, T. Tricyclic system containing large rings. The synthesis of fully-conjugated [12](1,3)[12](4,6)cyclophanpolyenepolyynes and related compounds. Can. J. Chem. 1985, 63, 2885–2891. [Google Scholar] [CrossRef]
- Schnatter, W.F.; Almarsson, Ö.; Bruice, T.C. The synthesis of a porphyrin with a hydrocarbon encapsulated face. Tetrahedron 1991, 47, 8687–8700. [Google Scholar] [CrossRef]
- Meier, H.; Fetten, M. A new synthetic route to tribenzo[a,e,i][12]annulenes. Tetrahedron Lett. 2000, 41, 1525–1538. [Google Scholar]
- Gerold, J.; Holzenkamp, U.; Meier, H. Bis-, tris-, and tetrakis(squaraines) linked by stilbenoid scaffolds. Eur. J. Org. Chem. 2001, 2757–2763. [Google Scholar] [CrossRef]
- Haley, M.M.; Brand, S.C.; Pak, J.J. Carbon Networks Based on Dehydrobenzoannulenes: Synthesis of Graphdiyne Substructures. Angew. Chem. Int. Ed. 1997, 36, 835–838. [Google Scholar] [CrossRef]
- Harada, K.; Hart, H.; Du, C.J.F. Reaction of aryl Grignard-reagents with hexahalobenzenes-novel arenes via a multiple aryne sequence. J. Org. Chem. 1985, 50, 5524–5528. [Google Scholar] [CrossRef]
- Wan, W.B.; Brand, S.C.; Pak, J.J.; Haley, M.M. Synthesis of expanded graphdiyne substructures. Chem. Eur. J. 2000, 6, 2044–2052. [Google Scholar] [CrossRef] [PubMed]
- Marsden, J.A.; Haley, M.M. Carbon networks based on dehydrobenzoannulenes. 5. Extension of two-dimensional conjugation in graphdiyne nanoarchitectures. J. Org. Chem. 2005, 70, 10213–10225. [Google Scholar] [CrossRef] [PubMed]
- Spitler, E.L.; Haley, M.M. Synthesis and structure-property relationships of donor/acceptor-functionalized bis(dehydrobenzo[18]annuleno)benzenes. Org Biomol. Chem. 2008, 6, 1569–1576. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.; Sun, Y.; Xu, Z.; Zeller, M.; Hunter, A.D. Structural regularity and diversity in hybrids of aromatic thioethers and BiBr3: from discrete complexes to layers and 3D nets. Dalton Trans. 2009, 5083–5093. [Google Scholar] [CrossRef]
- Gadzikwa, T.; Farha, O.K.; Malliakas, C.D.; Kanatzidis, M.G.; Hupp, J.T.; Nguyen, S.T. Selective bifunctional modification of a non-catenated metal-organic framework material via "click" chemistry. J. Am. Chem. Soc. 2009, 131, 13613–13615. [Google Scholar] [CrossRef] [PubMed]
- Mulfort, K.L.; Farha, O.K.; Stern, C.L.; Sarjeant, A.A.; Hupp, J.T. Post-synthesis alkoxide formation within metal-organic framework materials: a strategy for incorporating highly coordinatively unsaturated metal ions. J. Am. Chem. Soc. 2009, 131, 3866–3868. [Google Scholar] [CrossRef] [PubMed]
- Farha, O.K.; Mulfort, K.L.; Hupp, J.T. An example of node-based postassembly elaboration of a hydrogen-sorbing, metal-organic framework material. Inorg. Chem. 2008, 47, 10223–10225. [Google Scholar] [CrossRef] [PubMed]
- Bae, Y.; Farha, O.; Hupp, J.; Snurr, R.Q. Enhancement of CO2/N2 selectivity in a metal-organic framework by cavity modification. J. Mater Chem. 2009, 19, 2131–2134. [Google Scholar] [CrossRef]
- Shultz, A.M.; Farha, O.K.; Hupp, J.T.; Nguyen, S.T. A catalytically active, permanently microporous MOF with metalloporphyrin struts. J. Am. Chem. Soc. 2009, 131, 4204–4205. [Google Scholar] [CrossRef] [PubMed]
- Maly, K.E.; Gagnon, E.; Maris, T.; Wuest, J.D. Engineering hydrogen-bonded molecular crystals built from derivatives of hexaphenylbenzene and related compounds. J. Am. Chem. Soc. 2007, 129, 4306–4322. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.H.; Tong, K.; Wong, M.; So, S.K. Novel fluorine-containing X-branched oligophenylenes: structure–hole blocking property relationships. J. Mater. Chem. 2006, 765–772. [Google Scholar] [CrossRef]
- Fratiloiu, S.; Senthilkumar, K.; Grozema, F.C.; Christian-Pandya, H.; Niazimbetova, Z.I.; Bhandari, Y.J.; Galvin, M.E.; Siebbeles, L.D.A. Two-dimensional charge delocalization in X-shaped phenylenevinylene oligomers. Chem. Mater. 2006, 18, 2118–2129. [Google Scholar] [CrossRef]
- Niazimbetova, Z.I.; Menon, A.; Galvin, M.E.; Evans, D.H. Synthesis and electrochemical investigation of oligo(arylenevinylenes) intended for the preparation of two-dimensional polymer networks. J. Electroanal. Chem. 2002, 529, 43–50. [Google Scholar] [CrossRef]
- Casado, J.; Ortiz, R.; López Navarrete, J.; Ito, S.; Morita, N. A practical spectroscopic and theoretical approach to study the electrochromism in molecular-based materials: The case of a family of dendrimerlike poly(6-azulenylethenyl)benzenes. J. Phys. Chem. B 2004, 108, 18463–18471. [Google Scholar] [CrossRef]
- Suna, Y.; Zhaoa, K.; Wanga, C.; Luo, Y.; Ren, Y.; Tao, X.; Jiang, M. Two-photon absorption properties of multi-branched bis-(styryl)benzene based organic chromophores. J. Mol. Struc. Theochem. 2004, 682, 185–189. [Google Scholar] [CrossRef]
- Ren, Y.; Xin, Q.; Tao, X.T.; Wang, L.; Yu, X.Q.; Yang, J.X.; Jiang, M.H. Novel multi-branched organic compounds with enhanced two-photon absorption benefiting from the strong electronic coupling. Chem. Phys. Lett. 2005, 414, 253–258. [Google Scholar] [CrossRef]
- Wang, L.; Tao, X.; Yang, J.; Xu, G.; Ren, Y.; Liu, Y.; Yan, Y.; Liu, Z.; Jiang, M. Synthesis and properties of a new two-photon absorbing chromophore. Synth. Met. 2006, 156, 141–145. [Google Scholar] [CrossRef]
- Rumi, M.; Pond, S.J.K.; Meyer-Friedrichsen, T.; Zhang, Q.; Bishop, M.; Zhang, Y.; Barlow, S.; Marder, S.R.; Perry, J.W. Tetrastyrylarene derivatives: Comparison of one- and two-photon spectroscopic properties with distyrylarene analogues. J. Phys. Chem. C 2008, 112, 8061–8071. [Google Scholar] [CrossRef]
- Lim, I.S.; Vaiana, C.; Zhang, Z.; Zhang, Y.; An, D.; Zhong, C. X-shaped rigid arylethynes to mediate the assembly of nanoparticles. J. Am. Chem. Soc. 2007, 129, 5368–5369. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.; Chen, H.; Kuo, C.; Wang, B.; Chiu, H. Design and investigation of calamitic liquid crystals with low aspect-ratios: rigid y-shaped 1,2,4-tris(4-alkoxyphenylethynyl)benzenes. J. Mater. Chem. 2005, 15, 14854–14861. [Google Scholar] [CrossRef]
- Norbert, W.D.J.A.; Goodby, J.W.; Hird, M.; Toyne, K.J. The synthesis and mesomorphic behaviour of the 1,2,4,5-tetra-substituted benzenes with (4-tridecyloxyphenyl)ethynyl and (4-tridecyloxyphenyl)carbonyloxy substituents. Liq. Cryst. 1997, 22, 631–642. [Google Scholar] [CrossRef]
- García, F.; Fernández, G.; Sánchez, L. Modulated morphology in the self-organization of a rectangular amphiphile. Chem. Eur. J. 2009, 15, 6740–6747. [Google Scholar] [CrossRef] [PubMed]
- Spitler, E.L.; Haley, M.M. Dynamic proton-induced two-stage emission switching in donor-functionalized bis(dehydrobenzo[n]annuleno)benzenes and 1,2,4,5-tetrakis(phenylethynyl) benzene. Tetrahedron 2008, 64, 11469–11474. [Google Scholar] [CrossRef]
- Slepkov, A.D.; Hegmann, F.A.; Tykwinski, R.R.; Kamada, K.; Ohta, K.; Marsden, J.A.; Spitler, E.L.; Miller, J.J.; Haley, M.M. Two-photon absorption in two-dimensional conjugated quadrupolar chromophores. Opt. Lett. 2006, 31, 3315–3317. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Feng, J.; Ren, A.; Sun, C. Theoretical study of two-photon absorption properties for donor/acceptor-functionalized tetrakis(phenylethynyl)benzenes and bis(dehydrobenzoannuleno) benzenes. Opt. Mater. 2007, 29, 955–962. [Google Scholar] [CrossRef]
- Dalton, G.T.; Cifuentes, M.P.; Petrie, S.; Stranger, R.; Humphrey, M.G.; Samoc, M. Independent switching of cubic nonlinear optical properties in a ruthenium alkynyl cruciform complex by employing protic and electrochemical stimuli. J. Am. Chem. Soc. 2007, 129, 11882–11883. [Google Scholar] [CrossRef] [PubMed]
- Bart, J. The crystal structure of a modification of hexaphenylbenzene. Acta Cryst. B 1968, 24, 1277–1287. [Google Scholar] [CrossRef]
- Traber, B.; Wolff, J.; Rominger, F.; Oeser, T.; Gleiter, R.; Goebel, M.; Wortmann, R. Hexasubstituted donor-acceptor benzenes as nonlinear optically active molecules with multiple charge-transfer transitions. Chem. Eur. J. 2004, 10, 1227–1238. [Google Scholar] [CrossRef] [PubMed]
- Boese, R.; Green, J.; Mittendorf, J.; Mohler, D.; Vollhardt, K. Die ersten Hexabutadiinylbenzolderivate: Synthesen und Strukturen. Angew. Chem. 1992, 104, 1643–1645. [Google Scholar] [CrossRef]
- Berlman, I.; Wirth, H.; Steingraber, O. Systematics of the electronic spectra of the p-oligophenylenes and their substituted analogs. J. Phys. Chem. 1971, 75, 318–325. [Google Scholar] [CrossRef]
- Meier, H.; Holst, H. Alkoxy-substituted hexastyrylbenzenes. Adv. Synth. Cat. 2003, 345, 1005–1011. [Google Scholar] [CrossRef]
- Wan, W.; Haley, M. Carbon networks based on dehydrobenzoannulenes. 4. Synthesis of “star” and “trefoil” graphdiyne substructures via sixfold cross-coupling of hexaiodobenzene. J. Org. Chem. 2001, 66, 3893–3901. [Google Scholar] [CrossRef] [PubMed]
- Anand, S.; Varnavski, O.; Marsden, J.; Haley, M.; Schlegel, H.; Goodson, T. Optical Excitations in Carbon Architectures Based on Dodecadehydrotribenzo (18) annulene. J. Phys. Chem. A 2006, 110, 1305–1318. [Google Scholar] [CrossRef] [PubMed]
- Marguet, S.; Germain, A.; Millie, P. Electronic states of multichromophoric phenylethynylbenzene derivatives: exciton theory and CS-INDO-CIPSI calculations. Chem. Phys. 1996, 208, 351–373. [Google Scholar] [CrossRef]
- Kobayashi, K.; Kobayashi, N. Synthesis and self-association, absorption, and fluorescence properties of differentially functionalized hexakis(p-substituted-phenylethynyl)benzenes. J. Org. Chem. 2004, 69, 2487–2497. [Google Scholar] [CrossRef] [PubMed]
- Meier, H.; Mühling, B.; Gerold, J.; Jacob, D.; Oehlhof, A. Push-pull oligomers with 2, 2-dicyanovinyl groups as electron acceptors. Eur. J. Org. Chem. 2007, 625–631. [Google Scholar] [CrossRef]
- Rathore, R.; Burns, C.; Abdelwahed, S. Hopping of a single hole in hexakis[4-(1,1,2-triphenylethenyl)phenyl]benzene cation radical through the hexaphenylbenzene propeller. Org. Lett. 2004, 6, 1689–1692. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Watson, M.; Zhang, L.; Wang, Z.; Müllen, K. Hexakis (4-iodophenyl)-peri-hexabenzocoronene-A versatile building block for highly ordered discotic liquid crystalline materials. J. Am. Chem. Soc 2004, 126, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Takase, M.; Ismael, R.; Murakami, R.; Ikeda, M.; Kim, D.; Shinmori, H.; Furuta, H.; Osuka, A. Efficient synthesis of benzene-centered cyclic porphyrin hexamers. Tetrahedron Lett. 2002, 43, 5157–5159. [Google Scholar] [CrossRef]
- Tao, W.; Nesbitt, S.; Heck, R. Palladium-catalyzed alkenylation and alkynylation of polyhaloarenes. J. Org. Chem. 1990, 55, 63–69. [Google Scholar] [CrossRef]
- Stulgies, B.; Prinz, P.; Magull, J.; Rauch, K.; Meindl, K.; Rühl, S.; de Meijere, A. Six-and eightfold palladium-catalyzed cross-coupling reactions of hexa-and octabromoarenes. Chem. Eur. J. 2005, 11, 308–320. [Google Scholar] [CrossRef]
- Meier, H.; Hanold, N.; Kalbitz, H. Synthesis of hexastyrylbenzenes. Synthesis 1997, 276–278. [Google Scholar] [CrossRef]
- Ebert, M.; Jungbauer, D.A.; Kleppinger, R.; Kohne, B.; Praefke, K. Structural and dynamic properties of a new type of discotic nematic compounds. Liq. Cryst. 1989, 4, 53–67. [Google Scholar] [CrossRef]
- Praefke, K.; Kohne, B.; Singer, D. Liquid crystalline compounds 52. Hexaalkynyltriphenylene: A new type of nematic-discotic hydrocarbon. Angew. Chem. Int. Ed. 1990, 29, 177–179. [Google Scholar] [CrossRef]
- Nierle, J.; Barth, D.; Kuck, D. Pentakis(phenylethynyl) benzene and hexakis(phenylethynyl) benzene: A revision concerning two far too similar prototype hydrocarbons. Eur. J. Org. Chem. 2004, 867–872. [Google Scholar] [CrossRef]
- Feng, X.; Pisula, W.; Kudernac, T.; Wu, D.; Zhi, L.; De Feyter, S.; Müllen, K. Controlled self-assembly of C3-symmetric hexa-peri-hexabenzocoronenes with alternating hydrophilic and hydrophobic substituents in solution, in the bulk, and on a surface. J. Am. Chem. Soc. 2009, 131, 4439–4448. [Google Scholar] [CrossRef] [PubMed]
- Chebny, V.; Gwengo, C.; Gardinier, J.; Rathore, R. Synthesis and electronic properties of iso-alkyl substituted hexa-peri-hexabenzocoronenes (HBC’s) from a versatile new HBC synthon, hexakis (4-acetylphenyl) benzene. Tetrahedron Lett. 2008, 49, 4869–4872. [Google Scholar] [CrossRef]
- Wu, J.; Watson, M.; Müllen, K. The versatile synthesis and self-assembly of star-type hexabenzocoronenes. Angew. Chem. Int. Ed. 2003, 42, 5329–5333. [Google Scholar] [CrossRef]
- Lee, M.; Kim, J.W.; Peleshanko, S.; Larson, K. Amphiphilic hairy disks with branched hydrophilic tails and a hexa-peri-hexabenzocoronene core. J. Am. Chem. Soc. 2002, 124, 9121–9128. [Google Scholar] [CrossRef] [PubMed]
- Wiesler, U.; Berresheim, A.; Morgenroth, F.; Lieser, G.; Müllen, K. Divergent synthesis of polyphenylene dendrimers: The role of core and branching reagents upon size and shape. Macromolecules 2001, 34, 187–199. [Google Scholar]
- Song, K.H.; Kang, S.O.; Ko, J. Template synthesis of a huge macrocycle by olefin metathesis using easily accessible [Pt(PEt(3))(2)] templates. Chem. Eur. J. 2007, 13, 5129–5134. [Google Scholar] [CrossRef] [PubMed]
- Mössinger, D.; Hornung, J.; Lei, S.; De Feyter, S.; Höger, S. Molecularly defined shape-persistent 2D oligomers: the covalent-template approach to molecular spoked wheels. Angew. Chem. Int. Ed. 2007, 46, 6802–6806. [Google Scholar] [CrossRef]
- Lu, Y.; Suzuki, T.; Zhang, W.; Moore, J.; Mariñas, B. Nanofiltration membranes based on rigid star amphiphiles. Chem. Mater. 2007, 19, 3194–3204. [Google Scholar] [CrossRef]
- Suzuki, T.; Lu, Y.; Zhang, W.; Moore, J.; Mariñas, B. Performance characterization of nanofiltration membranes based on rigid star amphiphiles. Environ. Sci. Technol. 2007, 41, 6246–6252. [Google Scholar] [CrossRef] [PubMed]
- Takase, M.; Nakajima, A.; Takeuchi, T. Synthesis of an extended hexagonal molecule as a highly symmetrical ligand. Tetrahedron Lett. 2005, 46, 1739–1742. [Google Scholar] [CrossRef]
- Cho, H.S.; Rhee, H.; Song, J.K.; Min, C.K.; Takase, M.; Aratani, N.; Cho, S.; Osuka, A.; Joo, T.; Kim, D. Excitation energy transport processes of porphyrin monomer, dimer, cyclic trimer, and hexamer probed by ultrafast fluorescence anisotropy decay. J. Am. Chem. Soc. 2003, 125, 5849–5860. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Fechtenkötter, A.; Müllen, K. Star-like substituted hexaarylbenzenes: synthesis and mesomorphic properties. J. Mater. Chem. 2001, 11, 1634–1641. [Google Scholar] [CrossRef]
- Eshdat, L.; Hoffman, R.E.; Fechtenkötter, A.; Müllen, K.; Rabinovitz, M. Stereodynamics and characterization of the hexa(4-n-dodecylbiphenylyl)benzene hexaanion that includes a twisted benzene core. Chem. Eur. J. 2003, 9, 1844–1851. [Google Scholar] [CrossRef] [PubMed]
- Chebny, V.; Dhar, D.; Lindeman, S.; Rathore, R. Simultaneous ejection of six electrons at a constant potential by hexakis(4-ferrocenylphenyl)benzene. Org. Lett. 2006, 8, 5041–5044. [Google Scholar] [CrossRef] [PubMed]
- Rathore, R.; Burns, C.; Deselnicu, M. Multiple-electron transfer in a single step. Design and synthesis of highly charged cation-radical salts. Org. Lett. 2001, 3, 2887–2890. [Google Scholar] [CrossRef] [PubMed]
- Rathore, R.; Burns, C.; Guzei, I. Synthesis and isolation of polytrityl cations by utilizing hexaphenylbenzene and tetraphenylmethane scaffolds. J. Org. Chem. 2004, 69, 1524–1530. [Google Scholar] [CrossRef] [PubMed]
- Keegstra, M.; Müllen, K.; De Feyter, S.; De Schryver, F. Hexaterphenylyl-and hexaquaterphenylylbenzene: The behavior of chromophores and electrophores in a restricted space. Angew. Chem. Int. Ed. 1996, 35, 774–776. [Google Scholar] [CrossRef]
- Tomović, Z.; van Dongen, J.; George, S.J.; Xu, H.; Pisula, W.; Leclère, P.; Smulders, M.M.; De Feyter, S.; Meijer, E.W.; Schenning, A.P. Star-shaped oligo(p-phenylenevinylene) substituted hexaarylbenzene: purity, stability, and chiral self-assembly. J. Am. Chem. Soc. 2007, 129, 16190–16196. [Google Scholar] [CrossRef] [PubMed]
- Constable, E.; Gusmeroli, D.; Housecroft, C.; Neuburger, M.; Schaffner, S. Cobalt decorated metallostars and metallodendrimers: Synthetic strategies and spectroscopic correlations. Polyhedron 2006, 25, 421–428. [Google Scholar] [CrossRef]
- Constable, E.; Eich, O.; Housecroft, C. Metallostars and metallodendrimers based upon hexaphenylbenzene cores. Inorg. Chem. Commun. 1999, 2, 431–433. [Google Scholar] [CrossRef]
- Constable, E.; Eich, O.; Fenske, D.; Housecroft, C.; Johnston, L. Metallostars: High-nuclearity linearly developed nanostructures containing multiple cluster motifs. Chem. Eur. J. 2000, 6, 4364–4370. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Lin, Y.; Lin, P.; Chen, Y. Star-shaped molecules containing polyalkynyl groups with metal moieties on benzene and triphenylene cores. Eur. J. Org. Chem. 2006, 4510–4518. [Google Scholar] [CrossRef]
- Kayser, B.; Altman, J.; Beck, W. Benzene-bridged hexaalkynylphenylalanines and first-generation dendrimers thereof. Chem. Eur. J. 1999, 5, 754–758. [Google Scholar] [CrossRef]
- Hamaoui, B.E.; Zhi, L.; Wu, J.; Li, J.; Lucas, N.T.; Tomovic, Z.E.; Kolb, U.; Müllen, K. Solid-state pyrolysis of polyphenylene-metal complexes: A facile approach toward carbon nanoparticles. Adv. Funct. Mater. 2007, 17, 1179–1187. [Google Scholar] [CrossRef]
- Kumar, S.; Varshney, S.; Chauhan, D. Room-temperature discotic nematic liquid crystals. Mol. Cryst. Liq. Cryst. 2003, 396, 241–250. [Google Scholar] [CrossRef]
- Shih, T.; Lee, J. Stable, low-temperature discotic nematic superstructures by incorporating a laterally substituted sidearm in hexakis(phenylethynyl)benzene discogens. Adv. Funct. Mater. 2007, 17, 1896–1902. [Google Scholar] [CrossRef]
- Nair, G.; Shankar Rao, D.; Krishna Prasad, S.; Chandrasekhar, S.; Kumar, S. Electrooptic and viewing angle characteristics of a display device employing a discotic nematic liquid crystal. Mol. Cryst. Liq. Cryst. 2003, 397, 245–252. [Google Scholar] [CrossRef]
- Kamada, K.; Antonov, L.; Yamada, S.; Ohta, K.; Yoshimura, T.; Tahara, K.; Inaba, A.; Sonoda, M.; Tobe, Y. Two-photon absorption properties of dehydrobenzo [12] annulenes and hexakis (phenylethynyl) benzenes: Effect of edge-linkage. ChemPhysChem 2007, 8, 2671–2677. [Google Scholar] [CrossRef] [PubMed]
- Selsby, R.G.; Pennance, P.; Barnhard, K.I. Application of the PPP Method to the Calculation of Ionization Potentials and Electron Affinities of Conjugated Organic Molecules. Int. J. Quant. Chem. 1990, 37, 539–546. [Google Scholar] [CrossRef]
- Lifka, T.; Oehlhof, A.; Meier, H. Dodecyloxy-Substituted 2,4,6-Tris(styryl)pyridines. J. Heterocycl. Chem. 2008, 45, 935–937. [Google Scholar] [CrossRef]
- Bléger, D.; Kreher, D.; Mathevet, F.; Attias, A.-J.; Schull, G.; Huard, A.; Duillard, L.; Forini-Debuischert, C.; Charra, F. Surface Noncovalent Bonding for Rational Design of Hierarchical Molecular Self-Assemblies. Angew. Chem. Int. Ed. 2007, 46, 7404–7407. [Google Scholar] [CrossRef]
- Shigeo, S.; Shimizu, S.; Takano, H. Preparation of styrylpyridine derivatives as electroluminescent materials (Nippon Pharma Dev. Inst, Japan). JKXXAF JP 07285937 A 19951031, 1995. CAN 124:175851. [Google Scholar]
- Attias, A.-J.; Cavalli, C.; Donnio, B.; Guillon, D.; Hapiot, P.; Malthête, J. Columnar Mesophase from a New Disclike Mesogen Based on a 3,5-Dicyano-2,4,6-tristyrylpyridine Core. Chem. Mater. 2002, 14, 375–384. [Google Scholar] [CrossRef]
- Attias, A.-J.; Cavalli, C.; Donnio, B.; Guillon, D.; Hapiot, P.; Malthête, J. Self-assembled Columnar Mesophase from a New Disclike Polar Mesogen Based on a 3,5-Dicyano-2,4,6-Tristyrylpyridine Core. Mol. Cryst. Liq. Cryst. 2004, 415, 169–177. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.; Shao, P.; Liang, Y.; Wang, H.; Qin, J.; Gong, Q. Synthesis and properties of new two-thopton absorption chromophores containing 3,5-dicyano-2,4,6-tristyrylpyridine as the core. New. J. Chem. 2005, 29, 792–797. [Google Scholar] [CrossRef]
- Siegrist, A.E. Über eine neue Synthese zur Darstellung heterocyclisch substituierter Stilbenverbindungen, die Anil-Synthese. Helv. Chim. Acta 1967, 50, 906–957. [Google Scholar] [CrossRef]
- Clavreul, R.; Bloch, B.; Brigodiot, M.; Marechal, E. Synthesis and Curing of poly(styrylpyridine)s. Structural study on models and on the resin, 1 Thermal behaviour of low-molecular-weight styrylpyridines. Makromol. Chem. 1987, 188, 47–65. [Google Scholar] [CrossRef]
- Matsui, M.; Kawamura, S.; Shibata, K.; Muramatsu, H. Synthesis and Characterization of Mono-, Bis-, and Trissubstituted Pyridinium and Pyrylium Dyes. Bull. Chem. Soc. Jpn. 1992, 65, 71–74. [Google Scholar] [CrossRef]
- Wu, D.-G.; Huang, C.-H.; Gan, L.-B.; Zhang, W.; Zheng, J.; Luo, H.X.; Li, N.Q. Light-Induced Electron Transfer on a Trichromophore Dye Modified ITO Electrode. J. Phys. Chem. B 1999, 103, 4377–4381. [Google Scholar] [CrossRef]
- Abbotto, A.; Beverina, L.; Bozio, R.; Facchetti, A.; Ferrante, C.; Pagani, G.A.; Pedron, G.; Signorini, R. Novel heteroaromatic-based multi-branched dyes with enhanced two-photon absorption activity. Chem. Commun. 2003, 2144–2145. [Google Scholar] [CrossRef]
- Tobe, Y.; Nakanishi, H.; Sonoda, M.; Wakabayashi, T.; Achiba, Y. Pyridine analogue of macrocyclic polyyne C58H4N2 as a precursor to diazafullerene. Chem. Commun. 1999, 1625–1626. [Google Scholar]
- Berthelot, M.; Laurence, C.; Safar, M.; Besseau, F. Hydrogen-bond basicity pKHB scale of six-membered aromatic N-heterocycles. J. Chem. Soc. Perkin Trans. 2 1998, 283–290. [Google Scholar] [CrossRef]
- Achelle, S.; Ramondenc, Y.; Marsais, F.; Plé, N. Star- and banana-shaped oligomers with a pyrimidine core: Synthesis and Light-Emitting Properties. Eur. J. Org. Chem. 2008, 3129–3140. [Google Scholar] [CrossRef]
- Garcia Martinez, A.; Herrera Fernandez, A.; Moreno Jimenez, F.; Garcia Fraile, A.; Subramanian, L.R.; Hanack, M. On the mechanism of the reaction between ketones and trifluoromethanesulfonic anhydride. An improved and convenient method for the preparation of pyrimidines and condensed pyrimidines. J. Org. Chem. 1992, 57, 1627–1630. [Google Scholar] [CrossRef]
- Müller, T.J.J.; Braun, R.; Ansorge, M. A Novel Three-Component One-Pot Pyrimidine Synthesis Based upon a Coupling−Isomerization Sequence. Org. Lett. 2000, 2, 1967–1970. [Google Scholar] [CrossRef] [PubMed]
- Asahina, Y.; Kuroda, E. Über eine Bildungsweise der substituierten Pyridine. Chem. Ber. 1914, 47, 1815–1820. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Lai, C.K.; Chang, Y.-C.; Liu, K.-T. Formation of hexagonal columnar phases by heterocyclic pyrimidine derivatives. Liq. Cryst. 2002, 29, 237–242. [Google Scholar] [CrossRef]
- Itami, K.; Yamazaki, D.; Yoshida, J.-I. Pyrimidine-Core Extended π-Systems: General Synthesis and Interesting Fluorescent Properties. J. Am. Chem. Soc. 2004, 126, 15396–15397. [Google Scholar] [CrossRef] [PubMed]
- Schomaker, J.M.; Delia, T.J. Arylation of Halogenated Pyrimidines via a Suzuki Coupling Reaction. J. Org. Chem. 2001, 66, 7125–7128. [Google Scholar] [CrossRef] [PubMed]
- Molander, G.A.; Katona, B.W.; Machrouhi, F. Development of the Suzuki-Miyaura Cross-coupling reaction: use of air-stable Potassium alkynyltrifluoroborates in aryl alkynylations. J. Org. Chem. 2002, 67, 8416–8423. [Google Scholar] [CrossRef] [PubMed]
- Achelle, S.; Ramondenc, Y.; Dupas, G.; Plé, N. Bis- and tris(arylethynyl)pyrimidine oligomers: synthesis and light-emitting properties. Tetrahedron 2008, 64, 2783–2791. [Google Scholar]
- Yamanaka, H.; Ogawa, S.; Konno, S. Studies on Pyrimidine Derivatives. XVIII. Reaction of Active Methyl Groups on Pyrimidine N-Oxides. Chem. Pharm. Bull. 1980, 28, 1526–1533. [Google Scholar] [CrossRef]
- Bowman, A. Some Pyrimidine derivatives. J. Chem. Soc. 1937, 494–495. [Google Scholar]
- Braun, E.; Meyer, V. Ueber die Aldine. Chem. Ber. 1888, 21, 1269–1282. [Google Scholar] [CrossRef]
- Behun, J.D.; Kann, P.T. Thermal and oxidation stability of high-temperature functional fluids. Prepr. Am. Chem. Soc. 1963, 8, C117–C136. [Google Scholar]
- Electrophotographic material containing organic photoconductive compounds Gevaert Photo-Produkten N.V. BE 595696 19610201, 1961. CAN 58: 14918.
- Yabe, M.; Fugono, M. Electron transport material for organic electroluminescent device. (Mitsubishi Chemical Corp. Japan). Jpn. Kokai Tokkyo Koho JP 2005243266 A 20050908, 2005. CAN 143:275313. [Google Scholar]
- Takahashi, T.; Satake, K. Syntheses of heterocyclic compounds of nitrogen. LXXIX. Photosensitizing dyes. Yakugaku Zasshi 1952, 8, 1188–1192, CAN 47:44597. [Google Scholar]
- Nohara, M.; Saigo, K.; Ohashi, Y.; Hasegawa, M. Organic electroluminescent properties of tri- and tetrastyrylpyrazine derivatives. Trans. Mater. Res. Soc. Jpn. 1994, 15A, 389–391. [Google Scholar]
- Zerban, G. Styrylsubstituierte Aromaten mit langen Alkoxyseitenketten. PhD thesis, University of Mainz, Germany, 1991. [Google Scholar]
- Schmitt, V. Synthese und Charakterisierung aza-analoger Oligo(phenylenvinylene)e mit umgebungsabhängiger UV-vis-Absorption und Fluoreszenz. PhD thesis, University of Mainz, Germany, 2009. [Google Scholar]
- Collette, J.C.; Harper, A.W. Properties and chemical environment effects of alkylamino styryl pyrazine two-photon fluorophores. Proc. SPIE 2003, 5212, 184–192. [Google Scholar]
- Rumi, M.; Pond, S.J.K.; Zhang, Q.; Bishop, M.; Zhang, Y.; Barlow, S.; Marder, S.R.; Perry, J.W. Two-photon absorption in cross-shaped chromophores with phenylene-vinylene backbones. Proc. SPIE 2008, 6891, 689104:1–689104:12. [Google Scholar]
- Hayami, S.; Inoue, K. Structure and Magnetic Property of the Organic Triradical with Triazine Skeleton; 2,4,6-Tris{p-(N-oxy-N-tert-butylamino)phenyl}triazine. Chem. Lett. 1999, 545–546. [Google Scholar] [CrossRef]
- Kotha, S.; Kashinath, D.; Kumar, S. Synthesis of liquid crystalline materials based on 1,3,5-triphenylbenzene and 2,4,6-triphenyl-1,3,5-s-triazine. Tetrahedron Lett. 2008, 49, 5419–5423. [Google Scholar] [CrossRef]
- Murase, T.; Fujita, M. Highly Blue Luminescent Triazine-Amine conjugated Oligomers. J. Org. Chem. 2005, 70, 9269–9278. [Google Scholar] [CrossRef] [PubMed]
- Anderson, H.L.; Anderson, S.; Sanders, J.K.M. Ligand binding by butadiyne-linked porphyrin dimers, trimers and tetramers. J. Chem. Soc. Perkin Trans. 1 1995, 2231–2245. [Google Scholar] [CrossRef]
- Wortmann, R.; Glania, C.; Krämer, P.; Matschiner, R.; Wolff, J.J.; Kraft, S.; Treptow, B.; Barbu, E.; Längle, D.; Görlitz, G. Nondipolar Structures with Threefold Symmetry for Nonlinear Optics. Chem. Eur. J. 1997, 3, 1765–1773. [Google Scholar] [CrossRef]
- Lee, H.; Kim, D.; Lee, H.-K.; Qiu, W.; Oh, N.-K.; Zin, W.-C.; Kim, K. Discotic liquid crystalline materials for potential nonlinear optical applications: synthesis and liquid crystalline behavior of 1,3,5-triphenyl-2,4,6-triazine derivatives containing achiral and chiral alkyl chains in the periphery. Tetrahedron Lett. 2004, 45, 1019–1022. [Google Scholar] [CrossRef]
- Lee, C.-H.; Yamamoto, T. A New Class of Star-Shaped Discotic Liquid Crystal Containing a 2,4,6-Triphenyl-1,3,5-triazine Unit as a Core. Bull Chem. Soc. Jpn. 2002, 75, 615–618. [Google Scholar] [CrossRef]
- Lee, C.-H.; Yamamoto, T. Synthesis and characterization of a new class of liquid-crystalline, highly luminescent molecules containing a 2,4,6-triphenyl-1,3,5-triazine unit. Tetrahedron Lett. 2001, 42, 3993–3996. [Google Scholar] [CrossRef]
- Ranganathan, A.; Heisen, B.C.; Dix, I.; Meyer, F. A triazine—based three-directional rigid-rod tecton forms a novel 1D channel structure. Chem. Comm. 2007, 3637–3639. [Google Scholar] [CrossRef]
- Lee, C.-H.; Yamamoto, T. Synthesis of Liquid Crystalline, Highly Luminescent π-Conjugated 1.3.5-Triazine Derivatives by Palladium-Catalyzed Cross-Coupling Reaction. Mol. Cryst. Liq. Cryst. 2002, 378, 13–21. [Google Scholar] [CrossRef]
- Ray, P.C.; Das, P.K. First-order hyperpolarizabilities of octupolar aromatic molecules: symmetrically substituted triazines. Chem. Phys. Lett. 1995, 244, 153–156. [Google Scholar] [CrossRef]
- Park, G.; Cho, B.R. First hyperpolarizabilities of triazine derivatives. Ab initio studies and Hammett correlation. J. Phys. Org. Chem. 2004, 17, 169–173. [Google Scholar] [CrossRef]
- Huang, W.-H.; Jia, W.-L.; Wang, S. 7-Azaindolyl- and indolyl-fuctionalized starburst molecules with a 1,3,5-triazine or a benzene core-Synthesis and luminescence. Can. J Chem. 2006, 84, 477–485. [Google Scholar] [CrossRef]
- Pang, J.; Tao, Y.; Freiberg, S.; Yang, X.-P.; D´Iorio, M.; Wang, S. Syntheses, structures, and electroluminescence of new blue luminescent star-shaped compounds based on 1,3,5-triazine and 1,3,5-trisubstituted benzene. J. Mater. Chem. 2002, 12, 206–212. [Google Scholar] [CrossRef]
- Kusukawa, T.; Fujita, M. Self-assembled M6L4-Type Coordination Nanocage with 2,2´-Bipyridine Ancillary Ligands. Facile Crystallization and X-ray Analysis of Shape-Selective Enclathration of Neutral Guests in the Cage. J. Am. Chem. Soc. 2002, 124, 13576–13582. [Google Scholar] [CrossRef] [PubMed]
- Oyamada, T.; Yoshizaki, H.; Sasabe, H.; Adachi, C. Efficient Electron Injection Characteristics of Triazine Derivatives for Transparent OLEDs (TOLEDs). Chem. Lett. 2004, 33, 1034–1035. [Google Scholar] [CrossRef]
- Fujita, M.; Oka, H.; Ogura, K. Palladium(0)/LiCl Promoted Cross-Coupling Reaction of (4-Pyridyl)stannanes and Aromatic Bromides: Easy access to Poly(4-pyridyl)-Substituted Aromatics. Tetrahedron Lett. 1995, 36, 5247–5250. [Google Scholar] [CrossRef]
- Ishi-i, T.; Yaguma, K.; Thiemann, T.; Yashima, M.; Ueno, K.; Mataka, S. High Electron Drift Mobility in an Amorphous Film of 2,4,6-Tris[4-(1-naphthyl]phenyl]-1,3,5-triazine. Chem. Lett. 2004, 33, 1244–1245. [Google Scholar] [CrossRef]
- Cui, Y.; Wang, S. Diboron and Triboron Compounds Based on Linear and Star-Shaped Conjugated Ligands with 8-Hydroxyquinolate Functionality: Impact of Intermolecular Interaction and Boron Coordination on Luminescence. J. Org. Chem. 2006, 71, 6485–6496. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.-L.; Hu, Y.-F.; Gao, J.; Wang, S. Linear and star-shaped polynuclear Ru(II) complexes of 2-(2´-pyridyl)benzimidazolyl derivatives: synthesis, photophysical properties and red light-emitting devices. Dalton Trans. 2006, 1721–1728. [Google Scholar] [CrossRef]
- Kannan, R.; He, G. S.; Lin, T.-C.; Prasad, P.N.; Vaia, R.A.; Tan, L.-S. Towards Highly Active Two-Photon Absorbing Liquids. Synthesis and Characterization of 1,3,5-Triazine-Based Octupolar Molecules. Chem. Mater. 2004, 16, 185–194. [Google Scholar] [CrossRef]
- Rogers, J.E.; Slagle, J.E.; Mclean, D.G.; Sutherland, R.L.; Sankaran, B.; Kannan, R.; Tan, L.-S.; Fleitz, P.A. Understanding the One-Photon Photophysical Properties of a two-Photon Absorbing Chromophore. J. Phys. Chem. A 2004, 108, 5514–5520. [Google Scholar] [CrossRef]
- Chen, H.-F.; Yang, S.-J.; Tsai, Z.-H.; Hung, W.-Y.; Wang, T.-C.; Wong, K.-T. 1,3,5-Triazine derivatives as new electron transport-type host materials for highly green phosphorescent OLEDs. J. Mater. Chem. 2009, 19, 8112–8118. [Google Scholar] [CrossRef]
- Chérioux, F.; Maillotte, H.; Audebert, P.; Zyss, J. Synthesis and characterisation of an octupolar polymer and new molecular octupoles with off-resonant third order optical nonlinearities. Chem. Comm. 1999, 2083–2084. [Google Scholar] [CrossRef]
- Leriche, P.; Piron, F.; Ripaud, E.; Frère, P.; Allain, M.; Roncali, J. Star-shaped triazine-thiophene conjugated systems. Tetrahedron Lett. 2009, 50, 5673–5676. [Google Scholar] [CrossRef]
- Meng, F.; Li, B.; Qian, S.; Chen, K.; Tian, H. Enhanced Two-Photon Properties of Tri-branched Styryl Derivatives Based on 1,3,5-Triazine. Chem. Lett. 2004, 33, 470–471. [Google Scholar] [CrossRef]
- Li, B.; Tong, R.; Zhu, R.; Meng, F.; Tian, H.; Qian, S. The Ultrafast Dynamics and Nonlinear Optical Properties of Tribranched Styryl Derivatives based on 1,3,5-Triazine. J. Phys. Chem. B 2005, 109, 10705–10710. [Google Scholar] [CrossRef] [PubMed]
- García, A.; Insuasti, B.; Herranz, M.A.; Martínez-Àlvarez, R.; Martín, N. New Building Block for C3 Symmetry Molecules: Synthesis of s-Triazine-Based Redox-active Chromophores. Org. Lett. 2009, 11, 5398–5401. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, C.; Ulrich, H.; Kreutzberger, A. Triazine. IV. Phenyltriazine. Chem. Ber. 1953, 86, 181–186. [Google Scholar] [CrossRef]
- Elias, H.-G.; Greth, E. Triazin-Polymere 1. Poly(vinylenphenylen)triazine. Makromol. Chem. 1969, 123, 203–222. [Google Scholar] [CrossRef]
- Meier, H.; Holst, H.C.; Oehlhof, A. Star-shaped Compounds Having 1,3,5-triazine Cores. Eur. J. Org. Chem. 2003, 4173–4180. [Google Scholar]
- Holst, H.C.; Pakula, T.; Meier, H. Liquid crystals in the series of 2,4,6-tristyryl-1,3,5-triazines. Tetrahedron 2004, 60, 6765–6775. [Google Scholar] [CrossRef]
- Meier, H.; Karpouk, E.; Holst, H.C. Star-shaped Push-Pull Compounds Having 1,3,5-Triazine Cores. Eur. J. Org. Chem. 2006, 2609–2617. [Google Scholar] [CrossRef]
- Zheng, X.; Wong, N.-B.; Li, W.-K.; Tian, A. Theoretical Studies on the Nonlinear Optical Properties of Octupolar Tri-s-triazines. J. Chem. Theory Comput. 2006, 2, 808–814. [Google Scholar] [CrossRef]
- Cui, Y.Z.; Fang, Q.; Xue, G.; Yu, W.T. The Structure and Nonlinear Optical Properties of Octupolar Compound: 2,4,6-Tristyryl-s-triazine. Chinese Chem. Lett. 2003, 14, 856–859. [Google Scholar]
- Cui, Y.Z.; Fang, Q.; Juang, Z.L.; Xue, G.; Xu, G.B.; Yu, W.T. Frequency up-conversion of s-triazine derivatives via two-photon absorption and second-harmonic generation. J. Mater. Chem. 2004, 14, 2443–2449. [Google Scholar] [CrossRef]
- Cho, B.R.; Son, K.H.; Lee, S.H.; Song, Y.-S.; Lee, Y.-K.; Jeon, S.-J.; Choi, J.H.; Lee, H.; Cho, M. Two-Photon Absorption properties of 1,3,5-Tricyano-2,4,6-tris(styryl)benzene derivatives. J. Am. Chem. Soc. 2001, 123, 10039–10045. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Cui, Y.Z.; Fang, Q.; Xue, G.; Xu, G.B.; Yu, W.T. Synthesis and Two-Photon Absorption Properties of s-Triazine Derivatives. Chin. Chem. Lett. 2005, 16, 739–742. [Google Scholar]
- Cui, Y.Z.; Fang, Q.; Xue, G.; Xu, G.B.; Yin, L.; Yu, W.T. Cooperative Enhancement of Two-Photon Absorption of Multibranched Compounds with Vinylenes Attaching to the s-Triazine Core. Chem. Lett. 2005, 34, 644–645. [Google Scholar] [CrossRef]
- Kouvetakis, J.; Grotjahn, D.; Becker, P.; Moore, S.; Dupon, R. Synthesis of Ethynyl-Substituted Precursors to Carbon-Nitrogen-Sulfur Extended Structures: Reaction of C3N3F3 and C2N2SCl2 with Alkali-Metal (Trimethylsilyl)acetylides. Chem. Mater. 1994, 6, 636–639. [Google Scholar] [CrossRef]
- Faust, R.; Göbelt, B. First 2,3-Dialkynyl-1,4-diazabutadienes by Pd-catalyzed Cross-Coupling of Bis(imidoyl chlorides) and Alkyknylstannanes. Tetrahedron Lett. 1997, 38, 8017–8020. [Google Scholar] [CrossRef]
- Ohkita, M.; Kawano, M.; Suzuki, T.; Tsuji, T. Supramolecular Graphyne: a C(sp)-H....N hydrogen-bonded unique network structure of 2,4,6-triethynyl-1,3,5-triazine. Chem. Commun. 2002, 3054–3055. [Google Scholar] [CrossRef]
- Pieterse, K.; Lauritsen, A.; Schenning, A.P.H.J.; Vekemans, J.A.J.M.; Meijer, E.W. Symmetrical Electron-Deficient Materials Incorporating Azaheterocycles. Chem. Eur. J. 2003, 9, 5597–5604. [Google Scholar] [CrossRef] [PubMed]
- Cherioux, F.; Audebert, P.; Hapiot, P. New Star-Shaped Molecules with Extended Electronic Delocalization. Chem. Mater. 1998, 10, 1984–1989. [Google Scholar] [CrossRef]
- Brasselet, S.; Chérioux, F.; Audebert, P.; Zyss, J. New Octupolar Star-Shaped Structures for Quadratic Nonlinear Optics. Chem. Mater. 1999, 11, 1915–1920. [Google Scholar] [CrossRef]
- Goldmann, D.; Janietz, D.; Festag, R.; Schmidt, C.; Wendorff, J.H. New disc-shaped triarylamino-1,3,5-triazines with heteroaromatic central cores. Liq. Cryst. 1996, 21, 619–623. [Google Scholar] [CrossRef]
- Goldmann, D.; Dietel, R.; Janietz, D.; Schmidt, C.; Wendorff, J.H. Sheet-shaped mesogens based on 1,3,5-triazines: variation of columnar mesophases through intermolecular hydrogen bonding. Liq. Cryst. 1998, 24, 407–411. [Google Scholar] [CrossRef]
- Goldmann, D.; Janietz, D.; Schmidt, C.; Wendorff, J.H. Induktion lamellarer mesomorpher Strukturen in columnare Phasen bildenden 1,3,5-Triazinen durch Charge-Transfer-Wechselwirkungen mit Elektronenakzeptoren. Angew. Chem. 2000, 112, 1922–1925, Angew. Chem. Int. Ed. 2000, 39, 1851-1854. [Google Scholar]
- Janietz, D.; Goldmann, D.; Schmidt, C.; Wendorff, J.H. Control of mesomorphic structures of 1,3,5-triazines through molecular shape and intramolecular functionalization. Mol. Cryst. Liq. Cryst. A 1999, 332, 2651–2658. [Google Scholar]
- Lee, C.J.; Lee, S.J.; Chang, J.Y. Synthesis of a polymerizable discotic liquid crystalline compound with a 1,3,5-triazine core. Tetrahedron Lett. 2002, 43, 3863–3866. [Google Scholar] [CrossRef]
- Lee, S.J.; Chang, J.Y. Molecular ordering of photoreactive nonmesogenic 1,3,5-triazine compounds into columnar mesophases by charge transfer interaction. Tetrahedron Lett. 2003, 44, 7493–7497. [Google Scholar] [CrossRef]
- Wakamiya, A.; Die, T.; Yamaguchi, S. Toward π-Conjugated Molecule Bundles: Synthesis of a Series of B,B´,B´´-Trianthryl-N,N´N´´-triarylborazines and the bundle effect of their properties. J. Am. Chem. Soc. 2005, 127, 14859–14866. [Google Scholar] [CrossRef]
- Sergeyev, S.; Pisula, W.; Geerts, Y.H. Discotic liquid crystals: A new generation of organic semiconductors. Chem. Soc. Rev. 2007, 36, 1902–1929. [Google Scholar] [CrossRef] [PubMed]
- Cammidge, A.N.; Bushby, R.J. Discotic Liquid Crystals- Synthesis and Structural Features. In Handbook of Liquid Crystals; Demus, D., Goodby, J.W., Gray, G.W., Spiess, H.W., Vill, V., Eds.; VCH: Weinheim, Germany, 1998; Volume 2b, pp. 693–748. [Google Scholar]
- Andresen, T.L.; Krebs, F.C.; Thorup, N.; Bechgaard, K. Crystal structures of 2,3,6,7,10,11-oxytriphenylenes. Implications for columnar discotic mesophases. Chem. Mater. 2000, 12, 2428–2433. [Google Scholar] [CrossRef]
- Perez, D.; Guitian, E. Selected strategies for the synthesis of triphenylenes. Chem. Soc. Rev. 2004, 33, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Boden, N.; Bushby, R.J.; Headdock, G.; Lozman, O.R.; Wood, A. Syntheses of new 'large core' discogens based on the triphenylene, azatriphenylene and hexabenztrinaphthylene nuclei. Liq. Cryst. 2001, 28, 139–144. [Google Scholar] [CrossRef]
- Huang, G.; Tsang, C.; Xu, Z.; Li, K.; Zeller, M.; Hunter, A.D.; Chui, S.S.; Che, C. Flexible Thioether-Ag(I) Interactions for Assembling Large Organic Ligands into Crystalline Networks. Cryst. Growth Des. 2009, 9, 1444–1451. [Google Scholar] [CrossRef]
- Ishi-i, T.; Tashiro, H.; Kuwahara, R.; Mataka, S.; Yoshihara, T.; Tobita, S. An efficient energy transfer found in triphenylene/hexaazatriphenylene system with electronic complimentarity and structural similarity. Chem. Lett. 2006, 35, 158–159. [Google Scholar] [CrossRef]
- Yatabe, T.; Harbison, M.A.; Brand, J.D.; Wagner, M.; Müllen, K.; Samori, P.; Rabe, J.P. Extended triphenylenes: synthesis, mesomorphic properties and molecularly resolved scanning tunneling microscopy images of hexakis(dialkoxyphenyl)triphenylenes and dodeca(alkoxy) tris(triphenylenylene)s. J. Mater. Chem. 2000, 10, 1519–1525. [Google Scholar] [CrossRef]
- Arikainen, E.O.; Boden, N.; Bushby, R.J.; Lozman, O.R.; Vinter, J.G.; Wood, A. Complimentary polytopic interactions. Angew. Chem. Int. Ed. 2000, 39, 2333–2336. [Google Scholar] [CrossRef]
- Bushby, R.J.; Fisher, J.; Lozman, O.R.; Lange, S.; Lydon, J.E.; McLaren, S.R. The stability of columns comprising alternating triphenylene and hexaphenyltriphenylene molecules: variations in the structure of the hexaphenyltriphenylene component. Liq. Cryst. 2006, 33, 653–664. [Google Scholar] [CrossRef]
- Boden, N.; Bushby, R.J.; Cooke, G.; Lozman, O.R.; Lu, Z.B. CPI: A recipe for improving applicable properties of discotic liquid crystals. J. Am. Chem. Soc. 2001, 123, 7915–7916. [Google Scholar] [CrossRef] [PubMed]
- Boden, N.; Bushby, R.J.; Liu, Q.Y.; Lozman, O.R. CPI (complementary polytopic interaction) stabilised liquid crystal compounds formed by esters of 2-hydroxy-3,6,7,10,11-pentakis(hexyloxy)triphenylene. J. Mater. Chem. 2001, 11, 1612–1617. [Google Scholar] [CrossRef]
- Boden, N.; Bushby, R.J.; Lozman, O.R. A comparison of CPI and charge-transfer two-component columnar phases. Mol. Cryst. Liq. Cryst. 2004, 411, 1387–1396. [Google Scholar] [CrossRef]
- Lozman, O.R.; Bushby, R.J.; Vinter, J.G. Complementary polytopic interactions (CPI) as revealed by molecular modelling using the XED force field. J. Chem. Soc. Perk. Trans. 2 2001, 1446–1452. [Google Scholar] [CrossRef]
- Kreouzis, T.; Scott, K.; Donovan, K.J.; Boden, N.; Bushby, R.J.; Lozman, O.R.; Liu, Q. Enhanced electronic transport properties in complementary binary discotic liquid crystal systems. Chem. Phys. 2000, 262, 489–497. [Google Scholar] [CrossRef]
- Wegewijs, B.R.; Siebbeles, L.D.A.; Boden, N.; Bushby, R.J.; Movaghar, B.; Lozman, O.R.; Liu, Q.; Pecchia, A.; Mason, L.A. Charge-carrier mobilities in binary mixtures of discotic triphenylene derivatives as a function of temperature. Phys. Rev. B 2002, 65, 245112. [Google Scholar] [CrossRef]
- Bushby, R.J.; Donovan, K.J.; Kreouzis, T.; Lozman, O.R. Molecular engineering of triphenylene-based discotic liquid crystal conductors. Opto-Electron Rev. 2005, 13, 269–279. [Google Scholar]
- Boden, N.; Bushby, R.J.; Lozman, O.R.; Lu, Z.B.; McNeill, A.; Movaghar, B. Enhanced conduction in the discotic mesophase. Mol. Cryst. Liq. Cryst. 2004, 410, 541–549. [Google Scholar] [CrossRef]
- Bushby, R.J.; Evans, S.D.; Lozman, O.R.; McNeill, A.; Movaghar, B. Enhanced charge conduction in discotic liquid crystals. J. Mater. Chem. 2001, 11, 1982–1984. [Google Scholar] [CrossRef]
- Kreouzis, T.; Donovan, K.J.; Boden, N.; Bushby, R.J.; Lozman, O.R.; Liu, Q. Temperature-independent hole mobility in discotic liquid crystals. J. Chem. Phys. 2001, 114, 1797–1802. [Google Scholar] [CrossRef]
- Donovan, K.J.; Kreouzis, T.; Scott, K.; Bunning, J.C.; Bushby, R.J.; Boden, N.; Lozman, O.R.; Movaghar, B. Molecular engineering the phototransport properties of discotic liquid crystals. Mol. Cryst. Liq. Cryst. 2003, 396, 91–112. [Google Scholar] [CrossRef]
- An, Z.S.; Yu, J.S.; Jones, S.C.; Barlow, S.; Yoo, S.; Domercq, B.; Prins, P.; Siebbeles, L.D.A.; Kippelen, B.; Marder, S.R. High electron mobility in room-temperature discotic liquid-crystalline perylene diimides. Adv. Mater. 2005, 17, 2580–2583. [Google Scholar] [CrossRef]
- Feng, X.; Marcon, V.; Pisula, W.; Hansen, M.R.; Kirkpatrick, J.; Grozema, F.; Andrienko, D.; Kremer, K.; Muellen, K. Towards high charge-carrier mobilities by rational design of the shape and periphery of discotics. Nat. Mater. 2009, 8, 421–426. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.B.; Feng, J.K.; Ren, A.M.; Zhou, X. A comparative study of the two-photon absorption properties of new three-branched molecules-Sumanene and triphenylene derivatives. Chem. Phys. 2006, 322, 269–278. [Google Scholar] [CrossRef]
- Praefke, K.; Singer, D.; Kohne, B. New inductions of columnar mesophases in nematic multiyne mesogens. Liq. Cryst. 1993, 13, 445–454. [Google Scholar] [CrossRef]
- Ishi-i, T.; Murakami, K.-I.; Imai, Y.; Mataka, S. Light-Harvesting and Energy-Transfer System Based on Self-Assembling Perylene Diimide-Appended Hexaazatriphenylene. Org. Lett. 2005, 7, 3175–3178. [Google Scholar] [CrossRef] [PubMed]
- Aumiller, W.D.; Dalton, C.R.; Czarnik, A.W. Preparatively Useful Oxidation Reactions of Hexamethylhexaazatriphenylene. J. Org. Chem. 1995, 60, 728–729. [Google Scholar] [CrossRef]
- Kohne, B.; Praefcke, K. Eine neue und einfache Synthese des Dipyrazino[2,3-f: 2´,3´-h]-chinoxalin-Ringsystems. Liebigs Ann. Chem. 1985, 522–528. [Google Scholar] [CrossRef]
- Secondo, P.; Fages, F. Design and Synthesis of Bismacrocyclic Hexaazatriphenylene Derivatives. Org. Lett. 2006, 8, 1311–1314. [Google Scholar] [CrossRef] [PubMed]
- Nasielski, J.; Moucheron, C.; Verhoeven, C.; Nasielski-Hinkens, R. 2,3-Diphenyl- and 2,3,6,7-tetraphenyl-hexa-azatriphenylene, ligands for transition metals. Tetrahedron Lett. 1990, 31, 2573–2576. [Google Scholar] [CrossRef]
- Baxter, P.N.W.; Lehn, J.-M.; Kneisel, B.O.; Baum, G.; Fenske, D. The Designed Self-Assembly of Multicomponent and Multicompartmental cyclindrical Nanoarchitectures. Chem. Eur. J. 1999, 5, 113–120. [Google Scholar] [CrossRef]
- Baxter, P.; Lehn, J.-M.; DeCian, A.; Fischer, J. Selbstorganisation von Multikomponenten-systemen: spontane Bildung eines zylinderförmigen Komplexes aus fünf Liganden und sechs Metall-Ionen. Angew. Chem. 1993, 105, 92–95. [Google Scholar] [CrossRef]
- Bushby, R.J.; Lozman, O.R.; Mason, L.A.; Taylor, N.; Kumar, S. Cyclic voltammetry studies pf discotic liquid crystals. Mol. Cryst. Liq. Cryst. 2004, 410, 171–181. [Google Scholar] [CrossRef]
- Ishi-i, T.; Hirayama, T.; Murakami, K.-I.; Tashiro, H.; Thiemann, T.; Kubo, K.; Mori, A.; Yamasaki, S.; Akao, T.; Tsuboyama, A.; Mukaide, T.; Ueno, K.; Mataka, S. Combination of an Aromatic Core and Aromatic Side Chains Which Constitutes Discotic Liquid Crystal and Organogel Supramoleculat Assemblies. Langmuir 2005, 21, 1261–1268. [Google Scholar] [CrossRef] [PubMed]
- Ishi-i, T.; Tashiro, H.; Kuwahara, R.; Mataka, S.; Yoshihara, T.; Tobita, S. An Efficient Energy Transfer Found in Triphenylene/Hexaazatriphenylene System with Electronic Complimentary and Structural Similarity. Chem. Lett. 2006, 35, 158–159. [Google Scholar] [CrossRef]
- Hirayama, T.; Yamasaki, S.; Ameku, H.; Ishi-i, T.; Thiemann, T.; Mataka, S. Dyes Pigm. 2005, 67, 105–110.
- Ishi-i, T.; Murakami, K.-I.; Imai, Y.; Mataka, S. Self-Assembled Fluorescent Hexaazatriphenylenes That Act as a Light-Harvesting Antenna. J. Org. Chem. 2006, 71, 5752–5760. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.R.; Lee, S.K.; Kim, K.A.; Son, K.N.; Kang, T.I.; Jeon, S.J. First Hyperpolarizabilities of Hexaazatriphenylene Derivatives: Octupolar Nonlinear Optical Molecules. Tetrahedron Lett. 1998, 39, 9205–9208. [Google Scholar] [CrossRef]
- Gao, B.; Liu, Y.; Geng, Y.; Cheng, Y.; Wang, L.; Jing, X.; Wang, F. Starburst substituted hexaazatriphenylene compounds: synthesis, photophysical and electrochemical properties. Tetrahedron Lett. 2009, 50, 1649–1652. [Google Scholar] [CrossRef]
- Robertson, N.; Parsons, S.; MacLean, E.J.; Coxall, R.A.; Mount, A.R. Preparation, X-ray structure and properties of a hexabrominated symmetric indole trimer and its TCNQ adduct: a new route to functional molecular materials. J. Mater. Chem. 2000, 10, 2043–2047. [Google Scholar] [CrossRef]
- Luo, J.; Zhao, B.; Shao, J.; Lim, K.A.; Chan, H.S.O.; Chi, C. Room-temperature discotic liquid crystals based on oligothiophenes-attached and fused triazatruxenes. J. Mater. Chem. 2009, 19, 8327–8334. [Google Scholar] [CrossRef]
- Gómez-Lor, B.; Alonso, B.; Omenat, A.; Serrano, J.L. Electroactive C3 symmetric discotic liquid-crystalline triindoles. Chem. Commun. 2006, 5012–5014. [Google Scholar] [CrossRef]
- Hiyoshi, H.; Kumagai, H.; Ooi, H.; Sonoda, T.; Mataka, S. Donor-acceptor type symmetric cyclic triindoles: synthesis and properties. Heterocycles 2007, 72, 231–238. [Google Scholar] [CrossRef]
- Lai, W.-Y.; Chen, Q.-Q.; He, Q.-Y; Fan, Q.-L.; Huang, W. Microwave-enhanced multiple Suzuki couplings toward highly luminescent starburst monodisperse macromolecules. Chem. Comm. 2006, 1959–1961. [Google Scholar] [CrossRef]
- Lai, W.-Y.; He, Q.-Y; Zhu, R.; Chen, Q.-Q.; Huang, W. Kinked Star-Shaped Fluorene/Triazatruxene Co-oligomer Hybrids with Enhanced Functional Properties for High-Performance, Solution-Processed, Blue Organic Light-Emitting Diodes. Adv. Funct. Mater. 2008, 18, 265–276. [Google Scholar] [CrossRef]
- Lai, W.-Y.; Zhu, R.; Fan, Q.-L.; Hou, L.-T.; Cao, Y.; Huang, W. Monodisperse Six-Armed Triazatruxenes: Microwave-enhanced Synthesis and Highly Efficient Pure-Deep-Blue Electroluminescence. Macromolecules 2006, 39, 3707–3709. [Google Scholar] [CrossRef]
- Levermore, P.A.; Xia, R.; Lai, W.; Wang, X.-H.; Huang, W.; Bradley, D.D.C. Deep-blue emittig triazatruxene core/oligo-fluorene branch dendrimers for electroluminescence and optical gain applications. J. Phys. D Appl. Phys. 2007, 40, 1896–1901. [Google Scholar] [CrossRef]
- Zhu, R.; Lai, W.-Y.; Wang, H.-Y.; Yu, N.; Peng, B.; Huang, W.; Hou, L.-T.; Peng, J.-B.; Cao, Y. Monodisperse star-shaped compound and its blend in uncapped polyfluorene matrices as the active materials for high-performance pure blue light-emitting devices. Appl. Phys. Lett. 2007, 90, 141909, 1–3. [Google Scholar] [CrossRef]
- Feng, G.-L.; Lai, W.-Y.; Ji, S.-J.; Huang, W. Synthesis of novel star-shaped carbazole-functionalized triazatruxenes. Tetrahedron Lett. 2006, 47, 7089–7092. [Google Scholar]
- Lai, W.-Y.; He, Q.-Y.; Chen, D.-Y.; Huang, W. Synthesis and Characterization of Starburst 9-Phenylcarbazole/Triazatruxene Hybrids. Chem. Lett. 2008, 37, 986–987. [Google Scholar] [CrossRef]
- García-Frutos, E.M.; Gómez-Lor, B. Synthesis and Self-Association Properties of Functionalized C3-Symmetric Hexakis(p-substituted-phenylethynyl)triindoles. J. Am. Chem. Soc. 2008, 130, 9173–9177. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, K.A.; Ehrhart, O. Melamazin aus Hydrazinsalz und Dicyandiamid. Chem. Ber. 1911, 44, 2713–2717. [Google Scholar] [CrossRef]
- Kaiser, D.W.; Peters, G.A.; Wystrach, V.P.; Dicyandiamide, V. Structures of guanazo- and pyroguanazoles, and reaction of dicyandiamide with 3-amino-5-substituted-4-H-1,2,4-triazoles. J. Org. Chem. 1953, 18, 1610–1615. [Google Scholar] [CrossRef]
- Huisgen, R.; Sturm, H.J.; Seidel, M.; Ringöffnungen der Azole, V. Weitere Reaktionen der Tetrazole mit elektrophilen Agenzien. Chem. Ber. 1961, 94, 1555–1562. [Google Scholar] [CrossRef]
- Tartakovsky, V.A.; Frumkin, A.E.; Churakov, A.M.; Strelenko, Yu. A. New Approaches to the synthesis of tris[1,2,4]triazolo[1,3,5]triazines. Russ. Chem. Bull. Int. Ed. 2005, 54, 719–725. [Google Scholar] [CrossRef]
- Glang, S. Donorsubstituierte Tristriazolotriazine-Diskotische Flüssigkristalle und octupolare Fluoreszenzfarbstoffe. PhD thesis, University of Mainz, Germany, 2009. [Google Scholar]
- Cristiano, R.; Gallardo, H.; Bortoluzzi, A.J.; Bechtold, I.H.; Campos, C.E.M.; Longo, R.L. Tristriazolotriazines: a core for luminescent discotic liquid crystals. Chem. Commun. 2008, 5134–5136. [Google Scholar] [CrossRef]
- Kratochwil, M. Synthese und Eigenschaften von zwei- und dreifach Triazolo-anellierten Azinen. Diploma thesis, University of Mainz, Germany, 2009. [Google Scholar]
- Glang, S.; Schmitt, V.; Detert, H. Tristriazolotriazines-A novel heteroaromatic core for discotic liquid crystals. Proc. 36th German Topical Meeting on Liquid Crystals 2008, 125–128. [Google Scholar]
- Kanibolotsky, A.L.; Berridge, R.; Skabara, P.J.; Perepichka, I.F.; Bradley, D.D.C.; Koeberg, M. Synthesis and properties of monodisperse oligofluorene-functionalized truxenes: highly fluorescent star-shaped architectures. J. Am. Chem. Soc. 2004, 126, 13695–13702. [Google Scholar] [CrossRef] [PubMed]
- Roncali, J.; Leriche, P.; Cravino, A. From one- to three-dimensional organic semiconductors: in search of the organic silicon. Adv. Mat. 2007, 19, 2045–2060. [Google Scholar] [CrossRef]
- Garcia, F.; Sanchez, L. Dendronized triangular oligo(phenylene-ethynylene) amphiphiles: nanofibrillar self-assembly and dye encapsulation. Chem. Eur. J. 2010, 16, 2955–2964. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Detert, H.; Lehmann, M.; Meier, H. Star-Shaped Conjugated Systems. Materials 2010, 3, 3218-3330. https://doi.org/10.3390/ma3053218
Detert H, Lehmann M, Meier H. Star-Shaped Conjugated Systems. Materials. 2010; 3(5):3218-3330. https://doi.org/10.3390/ma3053218
Chicago/Turabian StyleDetert, Heiner, Matthias Lehmann, and Herbert Meier. 2010. "Star-Shaped Conjugated Systems" Materials 3, no. 5: 3218-3330. https://doi.org/10.3390/ma3053218
APA StyleDetert, H., Lehmann, M., & Meier, H. (2010). Star-Shaped Conjugated Systems. Materials, 3(5), 3218-3330. https://doi.org/10.3390/ma3053218




