Industrial Winemaking Waste to Sustainable Palladium(II) Recovery: A Green One-Step Synthesis of Activated Carbon from Grape Seeds
Abstract
1. Introduction
2. Materials and Methods
2.1. Activated Carbon Synthesis
2.2. Ash Content and Composition
2.3. Morphology and Surface Area
2.4. X-Ray Diffraction
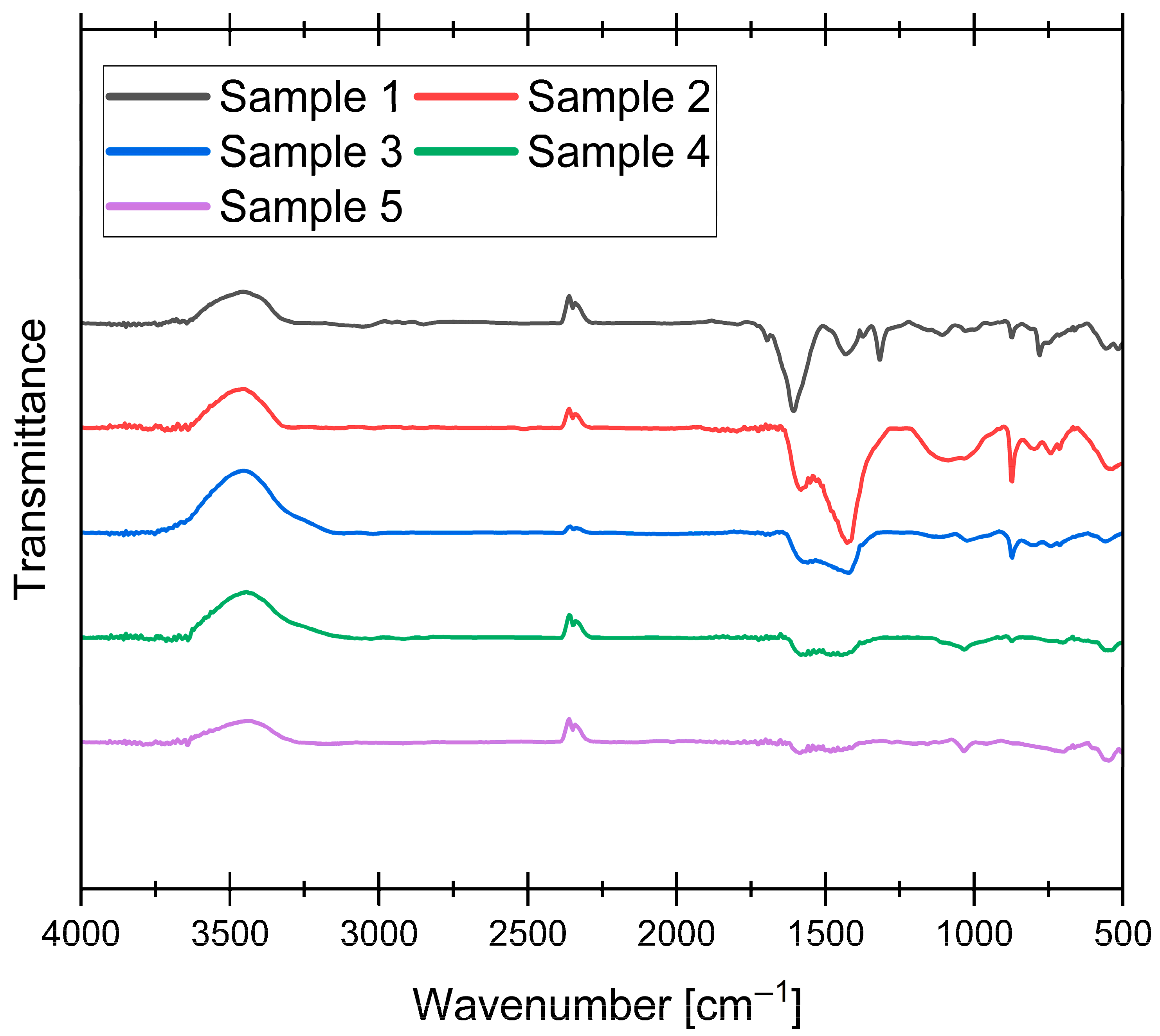
2.5. Functional Groups
2.6. Point of Zero Charge
2.7. Pd(II) Adsorption
2.8. Life Cycle Assessment
3. Results and Discussion
3.1. Ash Content and Composition Analysis
3.2. Morphology and Surface Area Analysis
3.3. XRD Analysis
3.4. Functional Groups Analysis
3.5. PZC Analysis
3.6. Pd(II) Adsorption Test
3.7. LCA Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hosseinzadeh, M.; Petersen, J. Recovery of Pt, Pd, and Rh from spent automotive catalysts through combined chloride leaching and ion exchange: A review. Hydrometallurgy 2024, 228, 106360. [Google Scholar] [CrossRef]
- Hughes, A.E.; Haque, N.; Northey, S.A.; Giddey, S. Platinum Group Metals: A Review of Resources, Production and Usage with a Focus on Catalysts. Resources 2021, 10, 93. [Google Scholar] [CrossRef]
- Afewerki, S.; Franco, A.; Balu, A.M.; Tai, C.-W.; Luque, R.; Córdova, A. Sustainable and recyclable heterogenous palladium catalysts from rice husk-derived biosilicates for Suzuki-Miyaura cross-couplings, aerobic oxidations and stereoselective cascade carbocyclizations. Sci. Rep. 2020, 10, 6407. [Google Scholar] [CrossRef]
- Michałek, T.; Hessel, V.; Wojnicki, M. Production, Recycling and Economy of Palladium: A Critical Review. Materials 2024, 17, 45. [Google Scholar] [CrossRef]
- Wang, M.; Tan, Q.; Chiang, J.F.; Li, J. Recovery of rare and precious metals from urban mines—A review. Front. Environ. Sci. Eng. 2017, 11, 1. [Google Scholar] [CrossRef]
- Alentiev, D.A.; Bermeshev, M.V.; Volkov, A.V.; Petrova, I.V.; Yaroslavtsev, A.B. Palladium Membrane Applications in Hydrogen Energy and Hydrogen-Related Processes. Polymers 2025, 17, 743. [Google Scholar] [CrossRef]
- Jha, M.K.; Gupta, D.; Lee, J.-C.; Kumar, V.; Jeong, J. Solvent extraction of platinum using amine based extractants in different solutions: A review. Hydrometallurgy 2014, 142, 60–69. [Google Scholar] [CrossRef]
- Yamada, M.; Kimura, S.; Gandhi, M.R.; Shibayama, A. Environmentally friendly Pd(II) recovery from spent automotive catalysts using resins impregnated with a pincer-type extractant. Sci. Rep. 2021, 11, 365. [Google Scholar] [CrossRef]
- Wang, Q.; Li, M.; Xi, M.; Zhao, M.; Wang, X.; Chen, X.; Ding, L. Recovery of Ag(I) from Wastewater by Adsorption: Status and Challenges. Toxics 2024, 12, 351. [Google Scholar] [CrossRef] [PubMed]
- Mariana, M.; H.P.S., A.K.; Mistar, E.; Yahya, E.B.; Alfatah, T.; Danish, M.; Amayreh, M. Recent advances in activated carbon modification techniques for enhanced heavy metal adsorption. J. Water Process Eng. 2021, 43, 102221. [Google Scholar] [CrossRef]
- Díez, E.; Gómez, J.; Rodríguez, A.; Bernabé, I.; Sáez, P.; Galán, J. A new mesoporous activated carbon as potential adsorbent for effective indium removal from aqueous solutions. Microporous Mesoporous Mater. 2020, 295, 109984. [Google Scholar] [CrossRef]
- Gu, H.; Bergman, R.; Anderson, N.; Alanya-Rosenbaum, S. Life cycle assessment of activated carbon from woody biomass. Wood Fiber Sci. 2018, 50, 229–243. [Google Scholar] [CrossRef]
- Malini, K.; Selvakumar, D.; Kumar, N. Activated carbon from biomass: Preparation, factors improving basicity and surface properties for enhanced CO2 capture capacity—A review. J. CO2 Util. 2023, 67, 102318. [Google Scholar] [CrossRef]
- Sujiono, E.; Zabrian, D.; Zurnansyah; Mulyati; Zharvan, V.; Samnur; Humairah, N. Fabrication and characterization of coconut shell activated carbon using variation chemical activation for wastewater treatment application. Results Chem. 2022, 4, 100291. [Google Scholar] [CrossRef]
- Michałek, T.; Wojtaszek, K.; Małecki, S.; Kornaus, K.; Wandor, S.; Druciarek, J.; Fitzner, K.; Wojnicki, M. Recovery of Pd(II) Ions from Aqueous Solutions Using Activated Carbon Obtained in a Single-Stage Synthesis from Cherry Seeds. C 2023, 9, 46. [Google Scholar] [CrossRef]
- Menya, E.; Olupot, P.; Storz, H.; Lubwama, M.; Kiros, Y. Production and performance of activated carbon from rice husks for removal of natural organic matter from water: A review. Chem. Eng. Res. Des. 2018, 129, 271–296. [Google Scholar] [CrossRef]
- Tsiapali, O.I.; Ayfantopoulou, E.; Tzourouni, A.; Ofrydopoulou, A.; Letsiou, S.; Tsoupras, A. Unveiling the Utilization of Grape and Winery By-Products in Cosmetics with Health Promoting Properties. Appl. Sci. 2025, 15, 1007. [Google Scholar] [CrossRef]
- Dávila, I.; Robles, E.; Egüés, I.; Labidi, J.; Gullón, P. 2—The Biorefinery Concept for the Industrial Valorization of Grape Processing By-Products. In Handbook of Grape Processing By-Products; Galanakis, C.M., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 29–53. [Google Scholar]
- Đorđević, T.R.; Vujetić, J.C.; Pintać-Šarac, D. Current circular economy aspect in valorization of agro-industrial waste as value-added products. Food Feed. Res. 2024, 51, 37. [Google Scholar] [CrossRef]
- Baylan, N.; Meriçboyu, A.E. Adsorption of lead and copper on bentonite and grapeseed activated carbon in single- and binary-ion systems. Sep. Sci. Technol. 2016, 51, 2360–2368. [Google Scholar] [CrossRef]
- Salman, J.M. Optimization study of preparation Iraqi grape seeds activated carbon for cadmium ion removal from aqueous solution. J. Madenat Alelem Univ. Coll. 2022, 14, 1–7. [Google Scholar]
- Özçimen, D.; Ersoy-Meriçboyu, A. Removal of copper from aqueous solutions by adsorption onto chestnut shell and grapeseed activated carbons. J. Hazard. Mater. 2009, 168, 1118–1125. [Google Scholar] [CrossRef]
- Mabrouk, S.; Fizer, M.; Mariychuk, R.; Dhaouadi, H. Grape seeds waste activated carbon as an adsorber of paracetamol drug residue: Experimental and theoretical approaches. J. Mol. Liq. 2024, 408, 125325. [Google Scholar] [CrossRef]
- Purnomo, C.W.; Castello, D.; Fiori, L. Granular Activated Carbon from Grape Seeds Hydrothermal Char. Appl. Sci. 2018, 8, 331. [Google Scholar] [CrossRef]
- Al Bahri, M.; Calvo, L.; Gilarranz, M.; Rodriguez, J. Activated carbon from grape seeds upon chemical activation with phosphoric acid: Application to the adsorption of diuron from water. Chem. Eng. J. 2012, 203, 348–356. [Google Scholar] [CrossRef]
- Hassani, S.E.A.E.; Driouich, A.; Chaair, H.; Mellouk, H.; Digua, K. Optimization of activated carbon by chemical activation from grape seeds using the response surface methodology. Desalination Water Treat. 2022, 245, 144–157. [Google Scholar] [CrossRef]
- Ani, J.U.; Akpomie, K.G.; Okoro, U.C.; Aneke, L.E.; Onukwuli, O.D.; Ujam, O.T. Potentials of activated carbon produced from biomass materials for sequestration of dyes, heavy metals, and crude oil components from aqueous environment. Appl. Water Sci. 2020, 10, 69. [Google Scholar] [CrossRef]
- Ganesh, K.N.; Zhang, D.; Miller, S.J.; Rossen, K.; Chirik, P.J.; Kozlowski, M.C.; Zimmerman, J.B.; Brooks, B.W.; Savage, P.E.; Allen, D.T.; et al. Green Chemistry: A Framework for a Sustainable Future. Org. Process Res. Dev. 2021, 25, 1455–1459. [Google Scholar] [CrossRef]
- Wojnicki, M.; Socha, R.P.; Pędzich, Z.; Mech, K.; Tokarski, T.; Fitzner, K. Palladium(II) Chloride Complex Ion Recovery from Aqueous Solutions Using Adsorption on Activated Carbon. J. Chem. Eng. Data 2018, 63, 702–711. [Google Scholar] [CrossRef]
- Wojnicki, M.; Podborska, A. The Mechanism of Redox Reaction between Palladium(II) Complex Ions and Potassium Formate in Acidic Aqueous Solution. Arch. Met. Mater. 2017, 62, 737–745. [Google Scholar] [CrossRef]
- Kalam, S.; Abu-Khamsin, S.A.; Kamal, M.S.; Patil, S. Surfactant Adsorption Isotherms: A Review. ACS Omega 2021, 6, 32342–32348. [Google Scholar] [CrossRef]
- Dada, A.O.; Olalekan, A.P.; Olatunya, A.M.; Dada, O.J.I.J.C. Langmuir, Freundlich, Temkin and Dubinin–Radushkevich Isotherms Studies of Equilibrium Sorption of Zn2+ Unto Phosphoric Acid Modified Rice Husk. J. Appl. Chem. 2012, 3, 38–45. [Google Scholar]
- Maulina, S.; Iriansyah, M. Characteristics of activated carbon resulted from pyrolysis of the oil palm fronds powder. IOP Conf. Ser. Mater. Sci. Eng. 2018, 309, 012072. [Google Scholar] [CrossRef]
- Kwiatkowski, M.; Belver, C.; Bedia, J. Effect of synthesis conditions on the porous texture of activated carbons obtained from Tara Rubber by FeCl3 activation. Sci. Rep. 2024, 14, 2266. [Google Scholar] [CrossRef]
- Dudek, K.; Wojtaszek, K.; Żabiński, P. Ni-Doped Activated Carbon from Invasive Plants as a Potential Catalyst. Metals 2024, 14, 790. [Google Scholar] [CrossRef]
- Knappe, D.R.U. Chapter 9—Surface chemistry effects in activated carbon adsorption of industrial pollutants. In Interface Science and Technology; Newcombe, G., Dixon, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2006; pp. 155–177. [Google Scholar]
- Ahmad, A.A.; Alwahbi, A.; Al Khatib, L.A.; Dammag, H. Biomass-Based Activated Carbon. In From Biomass to Biobased Products; Jacob-Lopes, E., Zepka, L.Q., Dias, R.R., Eds.; IntechOpen: Rijeka, Croatia, 2024. [Google Scholar]
- Ioannidou, O.; Zabaniotou, A. Agricultural residues as precursors for activated carbon production—A review. Renew. Sustain. Energy Rev. 2007, 11, 1966–2005. [Google Scholar] [CrossRef]
- Pasternak, T.P.; Steinmacher, D. Plant Growth Regulation in Cell and Tissue Culture In Vitro. Plants 2024, 13, 327. [Google Scholar] [CrossRef]
- Xu, W.; Liu, J.; Sun, K.; Liu, Y.; Chen, C.; Wang, A.; Sun, H. Effect of activation temperature on properties of H3PO4-activated carbon. BioResources 2021, 16, 4007–4020. [Google Scholar] [CrossRef]
- Siemak, J.; Wróbel, R.J.; Pęksiński, J.; Michalkiewicz, B. Investigation of CO2 Adsorption on Avocado Stone-Derived Activated Carbon Obtained through NaOH Treatment. Materials 2023, 16, 4390. [Google Scholar] [CrossRef]
- Liou, T.-H. Development of mesoporous structure and high adsorption capacity of biomass-based activated carbon by phosphoric acid and zinc chloride activation. Chem. Eng. J. 2010, 158, 129–142. [Google Scholar] [CrossRef]
- Kennedy, L.J.; Vijaya, J.J.; Sekaran, G. Effect of Two-Stage Process on the Preparation and Characterization of Porous Carbon Composite from Rice Husk by Phosphoric Acid Activation. Ind. Eng. Chem. Res. 2004, 43, 1832–1838. [Google Scholar] [CrossRef]
- Hanifa, M.; Sharma, U.; Thapliyal, P.C.; Singh, L.P. Effects of temperature, pH and calcium-to-carbonate ratio on the calcium carbonate polymorphs. Discov. Civ. Eng. 2025, 2, 147. [Google Scholar] [CrossRef]
- Karunadasa, K.S.; Manoratne, C.; Pitawala, H.; Rajapakse, R. Thermal decomposition of calcium carbonate (calcite polymorph) as examined by in-situ high-temperature X-ray powder diffraction. J. Phys. Chem. Solids 2019, 134, 21–28. [Google Scholar] [CrossRef]
- Mokti, N.; Borhan, A.; Zaine, S.N.A.; Zaid, H.F.M. Development of Rubber Seed Shell–Activated Carbon Using Impregnated Pyridinium-Based Ionic Liquid for Enhanced CO2 Adsorption. Processes 2021, 9, 1161. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R.; Freitas, M.M.A.; Órfão, J.J.M. Modification of the surface chemistry of activated carbons. Carbon 1999, 37, 1379–1389. [Google Scholar] [CrossRef]
- Sevilla, M.; Fuertes, A. The production of carbon materials by hydrothermal carbonization of cellulose. Carbon 2009, 47, 2281–2289. [Google Scholar] [CrossRef]
- Li, L.; Yao, X.; Li, H.; Liu, Z.; Ma, W.; Liang, X. Thermal Stability of Oxygen-Containing Functional Groups on Activated Carbon Surfaces in a Thermal Oxidative Environment. J. Chem. Eng. Jpn. 2014, 47, 21–27. [Google Scholar] [CrossRef]
- Świątkowski, A.; Grajek, H.; Pakuła, M.; Biniak, S.; Witkiewicz, Z. Voltammetric studies of the gradual thermal decomposition of activated carbon surface oxygen complexes. Colloids Surf. A Physicochem. Eng. Asp. 2002, 208, 313–320. [Google Scholar] [CrossRef]
- Menéndez, J.A.; Phillips, J.; Xia, B.; Radovic, L.R. On the Modification and Characterization of Chemical Surface Properties of Activated Carbon: In the Search of Carbons with Stable Basic Properties. Langmuir 1996, 12, 4404–4410. [Google Scholar] [CrossRef]
- Charmas, B.; Zięzio, M.; Jedynak, K. Assessment of the Porous Structure and Surface Chemistry of Activated Biocarbons Used for Methylene Blue Adsorption. Molecules 2023, 28, 4922. [Google Scholar] [CrossRef] [PubMed]
- Banik, C.; Lawrinenko, M.; Bakshi, S.; Laird, D.A. Impact of Pyrolysis Temperature and Feedstock on Surface Charge and Functional Group Chemistry of Biochars. J. Environ. Qual. 2018, 47, 452–461. [Google Scholar] [CrossRef]
- Araya, M.; Rivas, J.; Sepúlveda, G.; Espinoza-González, C.; Lira, S.; Meynard, A.; Blanco, E.; Escalona, N.; Ginocchio, R.; Garrido-Ramírez, E.; et al. Effect of Pyrolysis Temperature on Copper Aqueous Removal Capability of Biochar Derived from the Kelp Macrocystis pyrifera. Appl. Sci. 2021, 11, 9223. [Google Scholar] [CrossRef]
- Al-Wabel, M.I.; Al-Omran, A.; El-Naggar, A.H.; Nadeem, M.; Usman, A.R. Pyrolysis temperature induced changes in characteristics and chemical composition of biochar produced from conocarpus wastes. Bioresour. Technol. 2013, 131, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Jabłońska, B.; Jabłoński, P.; Gęga, J. Kinetics and Thermodynamics of Pb(II), Zn(II), and Cd(II) Adsorption from Aqueous Solutions onto Activated Biochar Obtained from Tobacco Waste. Materials 2025, 18, 2324. [Google Scholar] [CrossRef]
- Di Natale, F.; Orefice, M.; La Motta, F.; Erto, A.; Lancia, A. Unveiling the potentialities of activated carbon in recovering palladium from model leaching solutions. Sep. Purif. Technol. 2017, 174, 183–193. [Google Scholar] [CrossRef]
- Arampatzidou, A.C.; Deliyanni, E.A. Comparison of activation media and pyrolysis temperature for activated carbons development by pyrolysis of potato peels for effective adsorption of endocrine disruptor bisphenol-A. J. Colloid Interface Sci. 2016, 466, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Iida, T.; Amano, Y.; Machida, M.; Imazeki, F. Effect of surface property of activated carbon on adsorption of nitrate ion. Chem. Pharm. Bull. 2013, 61, 1173–1177. [Google Scholar] [CrossRef]
- Kasaini, H.; Goto, M.; Furusaki, S. Selective Separation of Pd(II), Rh(III), and Ru(III) Ions from a Mixed Chloride Solution Using Activated Carbon Pellets. Sep. Sci. Technol. 2000, 35, 1307–1327. [Google Scholar] [CrossRef]
- Wojnicki, M. Kinetic studies of the sorption process of Pd(II) chloride complex ions from diluted aqua solutions using commercially available activated carbon. React. Kinet. Mech. Catal. 2017, 122, 177–192. [Google Scholar] [CrossRef]
- Wojnicki, M. Spectrophotometric Analysis of the Kinetic of Pd(II) Chloride Complex Ions Sorption Process from Diluted Aqua Solutions Using Commercially Available Activated Carbon. Arch. Met. Mater. 2017, 62, 2405–2411. [Google Scholar] [CrossRef]
- Veses, A.; Sanahuja-Parejo, O.; Navarro, M.; López, J.; Murillo, R.; Callén, M.; García, T. From laboratory scale to pilot plant: Evaluation of the catalytic co-pyrolysis of grape seeds and polystyrene wastes with CaO. Catalysis Today 2021, 379, 87–95. [Google Scholar] [CrossRef]
- Bayer, P.; Heuer, E.; Karl, U.; Finkel, M. Economical and ecological comparison of granular activated carbon (GAC) adsorber refill strategies. Water Res. 2005, 39, 1719–1728. [Google Scholar] [CrossRef]
- Muñoz, I.; Peral, J.; Ayllón, J.A.; Malato, S.; Martin, M.J.; Perrot, J.Y.; Vincent, M.; Domènech, X. Life-Cycle Assessment of a Coupled Advanced Oxidation-Biological Process for Wastewater Treatment: Comparison with Granular Activated Carbon Adsorption. Environ. Eng. Sci. 2007, 24, 638–651. [Google Scholar] [CrossRef]
- Hjaila, K.; Baccar, R.; Sarrà, M.; Gasol, C.; Blánquez, P. Environmental impact associated with activated carbon preparation from olive-waste cake via life cycle assessment. J. Environ. Manag. 2013, 130, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Arena, N.; Lee, J.; Clift, R. Life Cycle Assessment of activated carbon production from coconut shells. J. Clean. Prod. 2016, 125, 68–77. [Google Scholar] [CrossRef]









| Sample Number | AC Mass [g] | Ash Mass [g] | Ash Content [%] |
|---|---|---|---|
| 1 | 1.002 | 0.047 | 4.66 |
| 2 | 0.801 | 0.046 | 5.80 |
| 3 | 0.709 | 0.038 | 5.34 |
| 4 | 0.681 | 0.037 | 5.39 |
| 5 | 0.711 | 0.040 | 5.68 |
| Element [%] | Sample Number | Mean ± SD | ||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | ||
| Ca | 57.60 | 60.90 | 57.90 | 59.40 | 62.10 | 59.58 ± 1.72 |
| P | 23.90 | 20.10 | 23.80 | 21.70 | 20.60 | 22.02 ± 1.77 |
| K | 8.90 | 9.60 | 8.40 | 9.00 | 8.00 | 8.78 ± 0.61 |
| Mg | 5.40 | 5.00 | 6.10 | 5.60 | 5.30 | 5.48 ± 0.41 |
| S | 2.70 | 2.80 | 2.20 | 2.70 | 2.40 | 2.56 ± 0.25 |
| Fe | 1.10 | 1.20 | 1.20 | 1.10 | 1.20 | 1.16 ± 0.05 |
| Cu | 0.13 | 0.13 | 0.15 | 0.13 | 0.13 | 0.13 ± 0.01 |
| Zn | 0.10 | 0.10 | 0.10 | 0.20 | 0.10 | 0.12 ± 0.04 |
| Mn | 0.12 | 0.13 | 0.11 | 0.12 | 0.12 | 0.12 ± 0.01 |
| Cr | 0.03 | 0.02 | 0.02 | 0.02 | 0.02 | 0.022 ± 0.004 |
| Ti | 0.02 | 0.02 | 0.02 | 0.03 | 0.03 | 0.024 ± 0.005 |
| Points | Elemental Content (at.%) | |||||||
|---|---|---|---|---|---|---|---|---|
| C | N | O | Mg | Si | P | K | Ca | |
| 001 | 66.31 | 19.24 | 10.26 | 0.41 | 0.07 | 1.26 | 0.72 | 1.73 |
| 002 | 84.31 | 6.31 | 6.00 | 0.60 | 0.18 | 1.75 | 0.46 | 0.39 |
| 003 | 56.05 | 15.60 | 26.88 | 0.11 | 0.06 | 0.24 | 0.08 | 0.98 |
| 004 | 54.76 | 0.00 | 41.05 | 0.00 | 0.10 | 0.09 | 0.05 | 3.95 |
| 005 | 58.08 | 6.12 | 31.36 | 0.03 | 0.05 | 0.09 | 0.05 | 4.22 |
| 006 | 79.14 | 4.64 | 14.36 | 0.16 | 0.00 | 0.26 | 0.23 | 1.21 |
| 007 | 63.54 | 19.56 | 12.34 | 0.11 | 0.73 | 0.47 | 2.82 | 0.43 |
| 008 | 81.46 | 4.49 | 10.86 | 0.53 | 0.36 | 1.53 | 0.44 | 0.33 |
| 009 | 83.65 | 4.78 | 11.19 | 0.00 | 0.11 | 0.06 | 0.18 | 0.03 |
| 010 | 66.15 | 3.72 | 17.91 | 0.43 | 1.10 | 1.38 | 2.91 | 6.40 |
| 011 | 38.78 | 0.00 | 13.71 | 0.09 | 0.25 | 1.13 | 0.57 | 45.47 |
| 012 | 64.83 | 0.00 | 21.50 | 0.13 | 0.09 | 1.11 | 7.80 | 4.54 |
| 013 | 63.26 | 0.00 | 23.82 | 0.62 | 0.16 | 4.47 | 0.36 | 7.31 |
| 014 | 79.71 | 0.00 | 15.68 | 0.92 | 0.21 | 1.50 | 0.42 | 1.56 |
| 015 | 56.03 | 0.00 | 26.19 | 0.55 | 0.18 | 6.75 | 0.31 | 9.99 |
| Temperature [°C] | Freundlich Isotherm | Temkin Isotherm | Experimental Maximum Adsorption Capacity | ||||
|---|---|---|---|---|---|---|---|
| KF [(mg/g) (L/mg)1/n] | n | R2 | AT [L/g] | bT [kJ/mol] | R2 | qe, Max [mg/g] | |
| Sample 1 | |||||||
| 30 | 0.0086 | 0.693 | 0.999 | 0.0761 | 0.822 | 0.873 | 7.76 |
| 40 | 0.0468 | 0.842 | 0.986 | 0.0932 | 0.591 | 0.937 | 11.27 |
| 50 | 0.0860 | 0.863 | 0.995 | 0.1019 | 0.417 | 0.954 | 16.20 |
| Sample 2 | |||||||
| 30 | 0.0277 | 0.891 | 0.955 | 0.0926 | 1.183 | 0.977 | 5.29 |
| 40 | 0.0729 | 1.009 | 0.975 | 0.1050 | 0.920 | 0.989 | 7.52 |
| 50 | 0.1061 | 1.046 | 0.932 | 0.1180 | 0.832 | 0.998 | 8.49 |
| Sample 3 | |||||||
| 30 | 0.0517 | 1.034 | 0.957 | 0.1064 | 1.378 | 0.978 | 4.69 |
| 40 | 0.0862 | 1.052 | 0.994 | 0.1011 | 0.875 | 0.916 | 8.47 |
| 50 | 0.1819 | 1.139 | 1.000 | 0.1171 | 0.653 | 0.937 | 11.92 |
| Sample 4 | |||||||
| 30 | 0.0698 | 1.162 | 0.971 | 0.1128 | 1.704 | 0.968 | 3.98 |
| 40 | 0.0876 | 1.065 | 0.993 | 0.1069 | 0.984 | 0.940 | 7.26 |
| 50 | 0.1754 | 1.141 | 0.986 | 0.1261 | 0.674 | 0.932 | 10.49 |
| Sample 5 | |||||||
| 30 | 0.1344 | 1.349 | 0.997 | 0.1288 | 1.590 | 0.936 | 4.73 |
| 40 | 0.1525 | 1.199 | 0.987 | 0.1224 | 0.962 | 0.942 | 7.79 |
| 50 | 0.2516 | 1.229 | 0.996 | 0.1317 | 0.684 | 0.931 | 11.45 |
| qe, Max [mg/g] | AC Origin | AC Activation | Medium | AC Dose [g/L] | C0 of Pd(II) [mg/L] | Temperature [°C] | Reference |
|---|---|---|---|---|---|---|---|
| 16.2 | Grape seeds | None | 0.1 M HCl | 1.67 | 106.42 | 50 | This study |
| 15.6 | Cherry seeds | None | 0.1 M HCl | 1.67 | 212.84 | 50 | [15] |
| 67.0 | Coconut shell | Steam | 0.1 M HCl | 1.67 | 239.45 | 50 | [29] |
| 27.0 | Peat | Steam | 2 M HCl | 7.50 | 225.00 | 25 | [60] |
| 51.6 | Bituminous coal | Steam | 0.1 M HCl | 0.61 | 50.00 | 20 | [57] |
| 111.3 | Wood charcoal | Steam | 0.1 M HCl | 1.60 | 266.05 | 21 | [61] |
| 81.1 | Coconut shell | Not specified | 0.1 M HCl | 1.66 | 42.57 | 21 | [62] |
| 41.39 | Coconut shell | Steam | 0.1 M HCl | 0.70 | 50.00 | 20 | [57] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Michałek, T.; Mańka, M.; Wojnicki, M. Industrial Winemaking Waste to Sustainable Palladium(II) Recovery: A Green One-Step Synthesis of Activated Carbon from Grape Seeds. Materials 2026, 19, 107. https://doi.org/10.3390/ma19010107
Michałek T, Mańka M, Wojnicki M. Industrial Winemaking Waste to Sustainable Palladium(II) Recovery: A Green One-Step Synthesis of Activated Carbon from Grape Seeds. Materials. 2026; 19(1):107. https://doi.org/10.3390/ma19010107
Chicago/Turabian StyleMichałek, Tomasz, Maciej Mańka, and Marek Wojnicki. 2026. "Industrial Winemaking Waste to Sustainable Palladium(II) Recovery: A Green One-Step Synthesis of Activated Carbon from Grape Seeds" Materials 19, no. 1: 107. https://doi.org/10.3390/ma19010107
APA StyleMichałek, T., Mańka, M., & Wojnicki, M. (2026). Industrial Winemaking Waste to Sustainable Palladium(II) Recovery: A Green One-Step Synthesis of Activated Carbon from Grape Seeds. Materials, 19(1), 107. https://doi.org/10.3390/ma19010107









