Ethosome-Based Colloidal Systems for Transdermal Delivery: The Role of Biosurfactant in Enhancing Stability and Efficacy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
- P-x8T-M(1+4) denotes a sample prepared with the following composition: 10% (w/w) of propylene glycol, 0.625% (w/w) of Tween 80, 30% (w/w) of alcohol mixture (ethanol + n-propanol) and 2.5% (w/w) of phospholipid; the preparation method used was mixing.
- H-yE-xMix-1 denotes a sample prepared with the following composition: 10% (w/w) of ethylene glycol, 0.625% (w/w) of surfactant mixture, 30% (w/w) of ethanol and 2.5% (w/w) of phospholipid, the preparation method used was homogenization.
2.2. Preparation of Ethosomes
2.3. Optimization of Preparation of Ethosomes
2.3.1. Optimization of Phospholipid Weight Concentration
2.3.2. Optimization of Alcohol Type
2.3.3. Optimization of Glycol Type and Its Concentration
2.3.4. Optimization of Surfactant Presence
2.4. Stability Testing
2.4.1. Zeta Potential (ZP)
2.4.2. Hydrodynamic Diameter (Dh) and PDI Values
2.4.3. Statistical Analyses
2.5. SEM
3. Results and Discussion
3.1. Optimization of Phospholipid Weight Concentration

3.2. Optimization of Alcohol Type

3.3. Optimization of Glycol Type and Its Concentration

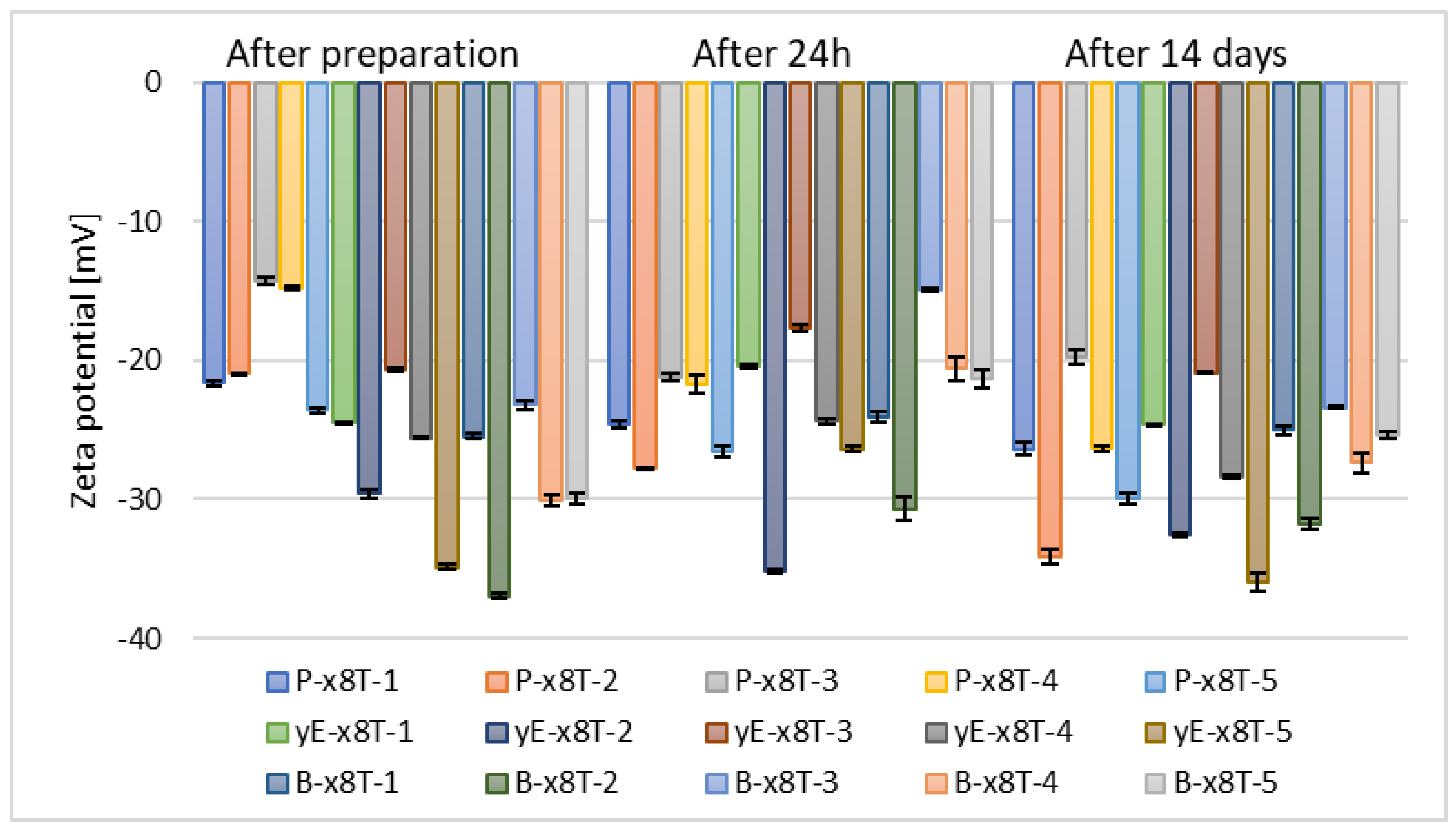

3.4. Optimization of Surfactant Presence

| Directly After Preparation | After 24 h in the Refrigerator | After 14 Days in the Refrigerator | ||||
|---|---|---|---|---|---|---|
| Formulation | Zeta Potential [mV] | ±SD [mV] | Zeta Potential [mV] | ±SD [mV] | Zeta Potential [mV] | ±SD [mV] |
| yE-x8T-1 | −25.70 | 0.44 | −24.97 | 0.74 | −27.90 | 0.52 |
| yE-y8T-1 | −26.27 | 0.15 | −24.17 | 0.49 | −29.70 | 0.17 |
| yE-z8T-1 | −26.50 | 0.10 | −25.23 | 0.15 | −30.13 | 0.31 |
| yE-xML-1 | −26.07 | 0.25 | −25.50 | 0.20 | −26.73 | 0.15 |
| yE-yML-1 | −32.77 | 0.12 | −26.30 | 0.23 | −21.40 | 0.17 |
| yE-zML-1 | −31.63 | 0.21 | −26.40 | 0.26 | −32.20 | 0.61 |
| H-yE-x8T-1 | −26.60 | 0.10 | −21.73 | 0.21 | −27.83 | 0.32 |
| H-yE-y8T-1 | −35.33 | 0.38 | −29.97 | 0.55 | −29.20 | 0.26 |
| H-yE-z8T-1 | −26.80 | 0.10 | −30.87 | 0.12 | −28.97 | 0.25 |
| H-yE-xML-1 | −19.43 | 0.91 | −29.20 | 0.10 | −30.00 | 0.70 |
| H-yE-yML-1 | −35.37 | 0.15 | −19.10 | 0.17 | −26.53 | 0.25 |
| H-yE-zML-1 | −27.27 | 0.12 | −26.43 | 0.21 | −28.33 | 0.21 |


| Directly After Preparation | After 24 h in the Refrigerator | After 14 Days in the Refrigerator | ||||
|---|---|---|---|---|---|---|
| Formulation | Zeta Potential [mV] | ±SD [mV] | Zeta Potential [mV] | ±SD [mV] | Zeta Potential [mV] | ±SD [mV] |
| yE-xMix-1 | −28.30 | 0.10 | −20.50 | 0.10 | −24.53 | 0.25 |
| yE-yMix-1 | −28.47 | 0.40 | −26.57 | 0.31 | −23.73 | 0.06 |
| yE-zMix-1 | −27.87 | 0.06 | −28.57 | 0.12 | −27.20 | 0.10 |
| H-yE-xMix-1 | −20.67 | 0.21 | −17.27 | 0.42 | −23.47 | 0.40 |
| H-yE-yMix-1 | −30.77 | 0.25 | −31.40 | 0.46 | −37.97 | 0.35 |
| H-yE-zMix-1 | −28.37 | 0.21 | −27.33 | 0.06 | −30.03 | 0.06 |


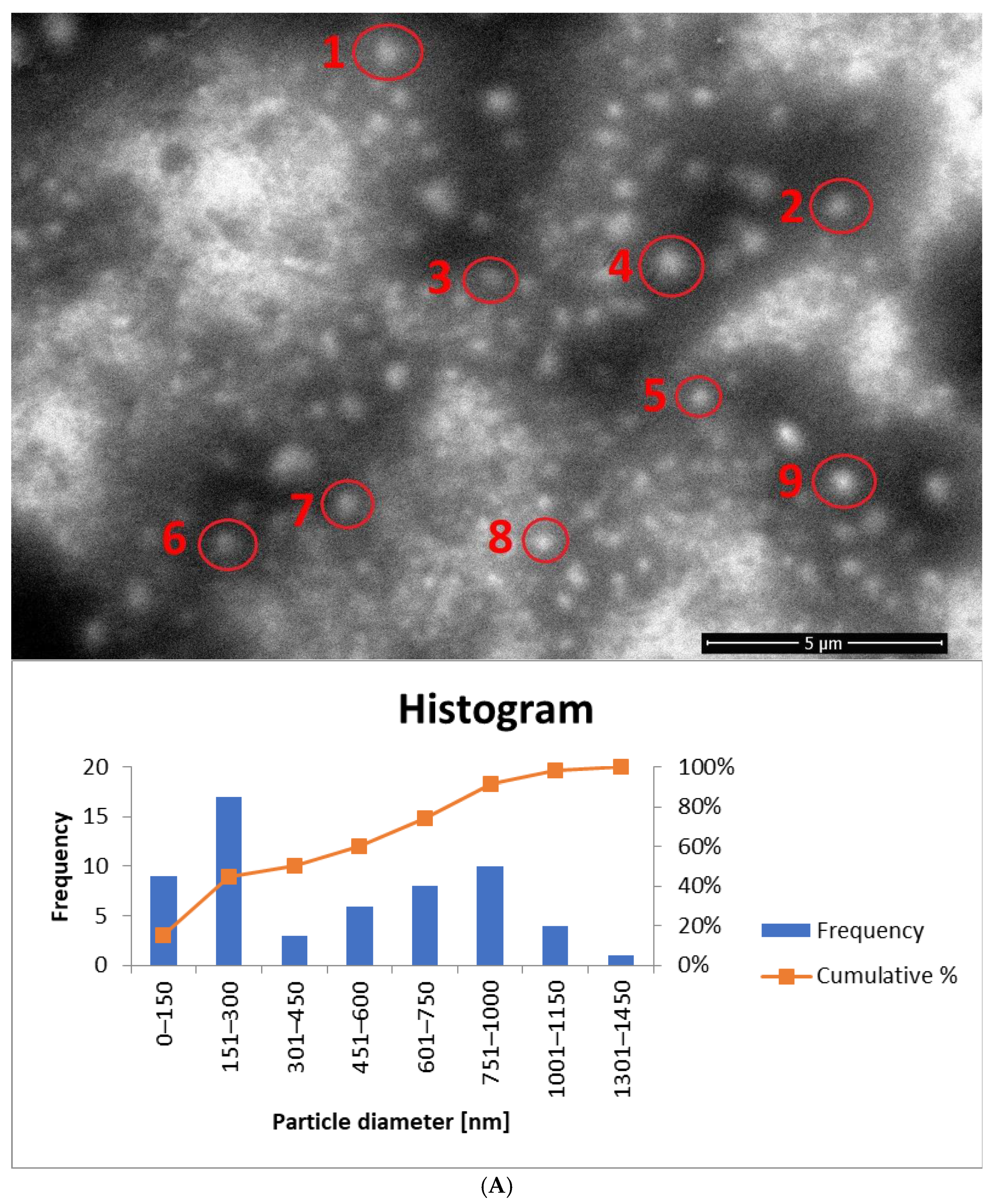
3.5. SEM Measurements





4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ZP | Zeta potential |
| PDI | Polydispersity index |
| Dh | Hydrodynamic diameter |
| SEM | Scanning electron microscopy |
| TDDS | Transdermal drug delivery system |
| CQA | Critical quality attribute |
| ELS | Electrophoretic light scattering |
| DLS | Dynamic light scattering |
| PALS | Phase analysis light scattering |
References
- Sparr, E.; Björklund, S.; Pham, Q.D.; Mojumdar, E.H.; Stenqvist, B.; Gunnarsson, M.; Topgaard, D. The Stratum Corneum Barrier—From Molecular Scale to Macroscopic Properties. Curr. Opin. Colloid. Interface Sci. 2023, 67, 101725. [Google Scholar] [CrossRef]
- Neubert, R.H.H. Mechanisms of Penetration and Diffusion of Drugs and Cosmetic Actives across the Human Stratum Corneum. Eur. J. Pharm. Biopharm. 2024, 202, 114394. [Google Scholar] [CrossRef]
- Gu, Y.; Bian, Q.; Zhou, Y.; Huang, Q.; Gao, J. Hair Follicle-Targeting Drug Delivery Strategies for the Management of Hair Follicle-Associated Disorders. Asian J. Pharm. Sci. 2022, 17, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Supe, S.; Takudage, P. Methods for Evaluating Penetration of Drug into the Skin: A Review. Skin. Res. Technol. 2021, 27, 299–308. [Google Scholar] [CrossRef]
- Nsairat, H.; Khater, D.; Sayed, U.; Odeh, F.; Al Bawab, A.; Alshaer, W. Liposomes: Structure, Composition, Types, and Clinical Applications. Heliyon 2022, 8, e09394. [Google Scholar] [CrossRef] [PubMed]
- Emanet, M.; Ciofani, G. Ethosomes as Promising Transdermal Delivery Systems of Natural-Derived Active Compounds. Adv. Nanobiomed Res. 2023, 3, 202300020. [Google Scholar] [CrossRef]
- Musielak, E.; Krajka-Kuźniak, V. Liposomes and Ethosomes: Comparative Potential in Enhancing Skin Permeability for Therapeutic and Cosmetic Applications. Cosmetics 2024, 11, 191. [Google Scholar] [CrossRef]
- Paiva-Santos, A.C.; Silva, A.L.; Guerra, C.; Peixoto, D.; Pereira-Silva, M.; Zeinali, M.; Mascarenhas-Melo, F.; Castro, R.; Veiga, F. Ethosomes as Nanocarriers for the Development of Skin Delivery Formulations. Pharm. Res. 2021, 38, 947–970. [Google Scholar] [CrossRef]
- Zhan, B.; Wang, J.; Li, H.; Xiao, K.; Fang, X.; Shi, Y.; Jia, Y. Ethosomes: A Promising Drug Delivery Platform for Transdermal Application. Chemistry 2024, 6, 993–1019. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Y.; Liu, X.; He, X.; Zhang, J.; Zhou, J.; Qiao, Y.; Wu, H.; Sun, F.; Zhou, Q. Optimal Development of Apoptotic Cells-Mimicking Liposomes Targeting Macrophages. J. Nanobiotechnology 2024, 22, 501. [Google Scholar] [CrossRef]
- Kocas, M.; Yamashita, F.; Comoglu, T.; Zhang, Q. Enhancing Intracellular Uptake of Ivermectin through Liposomal Encapsulation. AAPS PharmSciTech 2025, 26, 123. [Google Scholar] [CrossRef] [PubMed]
- Huanbutta, K.; Rattanachitthawat, N.; Luangpraditkun, K.; Sriamornsak, P.; Puri, V.; Singh, I.; Sangnim, T. Development and Evaluation of Ethosomes Loaded with Zingiber Zerumbet Linn Rhizome Extract for Antifungal Skin Infection in Deep Layer Skin. Pharmaceutics 2022, 14, 2765. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Gautam, G. Formulation, Development and Characterization of Ethosomes of Atorvastatin. Int. J. Pharm. Investig. 2020, 10, 156–159. [Google Scholar] [CrossRef]
- Niu, X.-Q.; Zhang, D.-P.; Bian, Q.; Feng, X.-F.; Li, H.; Rao, Y.-F.; Shen, Y.-M.; Geng, F.-N.; Yuan, A.-R.; Ying, X.-Y.; et al. Mechanism Investigation of Ethosomes Transdermal Permeation. Int. J. Pharm. X 2019, 1, 100027. [Google Scholar] [CrossRef]
- Kesimer, M.; Gupta, R. Physical Characterization and Profiling of Airway Epithelial Derived Exosomes Using Light Scattering. Methods 2015, 87, 59–63. [Google Scholar] [CrossRef]
- Muslim, R.K.; Maraie, N.K. Preparation and Evaluation of Nano-Binary Ethosomal Dispersion for Flufenamic Acid. Mater. Today Proc. 2022, 57, 354–361. [Google Scholar] [CrossRef]
- Aljohani, A.A.; Alanazi, M.A.; Munahhi, L.A.; Hamroon, J.D.; Mortagi, Y.; Qushawy, M.; Soliman, G.M. Binary Ethosomes for the Enhanced Topical Delivery and Antifungal Efficacy of Ketoconazole. OpenNano 2023, 11, 100145. [Google Scholar] [CrossRef]
- Elsayed, M.M.A.; Abdallah, O.Y.; Naggar, V.F.; Khalafallah, N.M. Lipid Vesicles for Skin Delivery of Drugs: Reviewing Three Decades of Research. Int. J. Pharm. 2007, 332, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chowdary, P.; Padmakumar, A.; Rengan, A.K. Exploring the Potential of Transethosomes in Therapeutic Delivery: A Comprehensive Review. MedComm-Biomater. Appl. 2023, 2, e59. [Google Scholar] [CrossRef]
- López-Pinto, J.M.; González-Rodríguez, M.L.; Rabasco, A.M. Effect of Cholesterol and Ethanol on Dermal Delivery from DPPC Liposomes. Int. J. Pharm. 2005, 298, 1–12. [Google Scholar] [CrossRef]
- Abdulbaqi, I.M.; Darwis, Y.; Abdul Karim Khan, N.; Abou Assi, R.; Ali Khan, A. Ethosomal Nanocarriers: The Impact of Constituents and Formulation Techniques on Ethosomal Properties, in Vivo Studies, and Clinical Trials. Int. J. Nanomed. 2016, 11, 2279–2304. [Google Scholar] [CrossRef]
- Touitou, E.; Dayan, N.; Bergelson, L.; Godin, B.; Eliaz, M. Ethosomes—Novel Vesicular Carriers for Enhanced Delivery: Characterization and Skin Penetration Properties. J. Control. Release 2000, 65, 403–418. [Google Scholar] [CrossRef]
- Chauhan, N.; Vasava, P.; Khan, S.L.; Siddiqui, F.A.; Islam, F.; Chopra, H.; Emran, T. Bin Ethosomes: A Novel Drug Carrier. Ann. Med. Surg. 2022, 82, 104595. [Google Scholar] [CrossRef]
- Verma, P.; Pathak, K. Therapeutic and Cosmeceutical Potential of Ethosomes: An Overview. J. Adv. Pharm. Technol. Res. 2010, 1, 274. [Google Scholar] [CrossRef] [PubMed]
- Waqar, M.A.; Zaman, M.; Hameed, H.; Munir, M. Ethosomes: A Novel Approach for The Delivery of Drug. Int. J. Pharm. Integr. Heal. Sci. 2023, 4, 31–46. [Google Scholar] [CrossRef]
- Mgbechidinma, C.L.; Akan, O.D.; Zhang, C.; Huang, M.; Linus, N.; Zhu, H.; Wakil, S.M. Integration of Green Economy Concepts for Sustainable Biosurfactant Production—A Review. Bioresour. Technol. 2022, 364, 128021. [Google Scholar] [CrossRef] [PubMed]
- Nagtode, V.S.; Cardoza, C.; Yasin, H.K.A.; Mali, S.N.; Tambe, S.M.; Roy, P.; Singh, K.; Goel, A.; Amin, P.D.; Thorat, B.R.; et al. Green Surfactants (Biosurfactants): A Petroleum-Free Substitute for Sustainability─Comparison, Applications, Market, and Future Prospects. ACS Omega 2023, 8, 11674–11699. [Google Scholar] [CrossRef] [PubMed]
- Sharma, N.; Ahlawat, Y.K.; Sharma, A.J.; Chamoli, N.; Thakur, M.; Sharma, A.; Mehmood, S.; Malik, A.; Ahmed, M.; Punia, H.; et al. A Comprehensive Review on Microbial Production and Significant Applications of Multifunctional Biomolecules: Biosurfactants. Biodegradation 2025, 36, 26. [Google Scholar] [CrossRef]
- Mirasoft® SL L60 (Syensqo). Available online: https://www.syensqo.com/en/product/mirasoft-sl-l60 (accessed on 14 November 2025).
- Acharya, S.R.; Padmanabha, R.V.; Acharya, N. Optimization of Size Controlled Poly (Lactide-Co-Glycolic Acid) Nanoparticles Using Quality by Design Concept. Asian J. Pharm. 2015, 9, 152. [Google Scholar] [CrossRef]
- Sivadasan, D.; Madkhali, O.A. The Design Features, Quality by Design Approach, Characterization, Therapeutic Applications, and Clinical Considerations of Transdermal Drug Delivery Systems—A Comprehensive Review. Pharmaceuticals 2024, 17, 1346. [Google Scholar] [CrossRef]
- Varenne, F.; Hillaireau, H.; Bataille, J.; Smadja, C.; Barratt, G.; Vauthier, C. Application of Validated Protocols to Characterize Size and Zeta Potential of Dispersed Materials Using Light Scattering Methods. Colloids Surf. A Physicochem. Eng. Asp. 2019, 560, 418–425. [Google Scholar] [CrossRef]
- Takechi-Haraya, Y.; Ohgita, T.; Demizu, Y.; Saito, H.; Izutsu, K.; Sakai-Kato, K. Current Status and Challenges of Analytical Methods for Evaluation of Size and Surface Modification of Nanoparticle-Based Drug Formulations. AAPS PharmSciTech 2022, 23, 150. [Google Scholar] [CrossRef] [PubMed]
- Caputo, F.; Clogston, J.; Calzolai, L.; Rösslein, M.; Prina-Mello, A. Measuring Particle Size Distribution of Nanoparticle Enabled Medicinal Products, the Joint View of EUNCL and NCI-NCL. A Step by Step Approach Combining Orthogonal Measurements with Increasing Complexity. J. Control. Release 2019, 299, 31–43. [Google Scholar] [CrossRef]
- Stetefeld, J.; McKenna, S.A.; Patel, T.R. Dynamic Light Scattering: A Practical Guide and Applications in Biomedical Sciences. Biophys. Rev. 2016, 8, 409–427. [Google Scholar] [CrossRef] [PubMed]
- Öztürk, K.; Kaplan, M.; Çalış, S. Effects of Nanoparticle Size, Shape, and Zeta Potential on Drug Delivery. Int. J. Pharm. 2024, 666, 124799. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wu, L.; Wu, D.; Shi, D.; Wang, T.; Zhu, X. Mechanism of Transdermal Permeation Promotion of Lipophilic Drugs by Ethosomes. Int. J. Nanomed. 2017, 12, 3357–3364. [Google Scholar] [CrossRef]
- Al-Ameri, A.A.F.; Al-Gawhari, F.J. Formulation Development of Meloxicam Binary Ethosomal Hydrogel for Topical Delivery: In Vitro and In Vivo Assessment. Pharmaceutics 2024, 16, 898. [Google Scholar] [CrossRef]
- Limsuwan, T.; Boonme, P.; Khongkow, P.; Amnuaikit, T. Ethosomes of Phenylethyl Resorcinol as Vesicular Delivery System for Skin Lightening Applications. Biomed. Res. Int. 2017, 2017, 8310979. [Google Scholar] [CrossRef]
- Garg, B.J.; Garg, N.K.; Beg, S.; Singh, B.; Katare, O.P. Nanosized Ethosomes-Based Hydrogel Formulations of Methoxsalen for Enhanced Topical Delivery against Vitiligo: Formulation Optimization, in Vitro Evaluation and Preclinical Assessment. J. Drug Target. 2016, 24, 233–246. [Google Scholar] [CrossRef]
- Sudhakar, K.; Mishra, V.; Jain, S.; Rompicherla, N.C.; Malviya, N.; Tambuwala, M.M. Development and Evaluation of the Effect of Ethanol and Surfactant in Vesicular Carriers on Lamivudine Permeation through the Skin. Int. J. Pharm. 2021, 610, 121226. [Google Scholar] [CrossRef]
- Yang, Y.-M.; Wu, K.-C.; Huang, Z.-L.; Chang, C.-H. On the Stability of Liposomes and Catansomes in Aqueous Alcohol Solutions. Langmuir 2008, 24, 1695–1700. [Google Scholar] [CrossRef]
- Hmingthansanga, V.; Singh, N.; Banerjee, S.; Manickam, S.; Velayutham, R.; Natesan, S. Improved Topical Drug Delivery: Role of Permeation Enhancers and Advanced Approaches. Pharmaceutics 2022, 14, 2818. [Google Scholar] [CrossRef] [PubMed]
- Hafidi, Z.; El Achouri, M. The Effect of Polar Head and Chain Length on the Physicochemical Properties of Micellization and Adsorption of Amino Alcohol-Based Surfactants. J. Surfactants Deterg. 2019, 22, 663–672. [Google Scholar] [CrossRef]
- Dickey, A.N.; Faller, R. How Alcohol Chain-Length and Concentration Modulate Hydrogen Bond Formation in a Lipid Bilayer. Biophys. J. 2007, 92, 2366–2376. [Google Scholar] [CrossRef]
- Reeves, M.D.; Schawel, A.K.; Wang, W.; Dea, P. Effects of Butanol Isomers on Dipalmitoylphosphatidylcholine Bilayer Membranes. Biophys. Chem. 2007, 128, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Heard, C.M. Ethanol and Other Alcohols: Old Enhancers, Alternative Perspectives. In Percutaneous Penetration Enhancers Chemical Methods in Penetration Enhancement; Springer: Berlin/Heidelberg, Germany, 2015; pp. 151–172. [Google Scholar]
- Haq, A.; Michniak-Kohn, B. Effects of Solvents and Penetration Enhancers on Transdermal Delivery of Thymoquinone: Permeability and Skin Deposition Study. Drug Deliv. 2018, 25, 1943–1949. [Google Scholar] [CrossRef]
- Zhang, M.; Zhuang, X.; Li, S.; Wang, Y.; Zhang, X.; Li, J.; Wu, D. Designed Fabrication of Phloretin-Loaded Propylene Glycol Binary Ethosomes: Stability, Skin Permeability and Antioxidant Activity. Molecules 2023, 29, 66. [Google Scholar] [CrossRef]
- Li, W.-Z.; Hao, X.-L.; Zhao, N.; Han, W.-X.; Zhai, X.-F.; Zhao, Q.; Wang, Y.-E.; Zhou, Y.-Q.; Cheng, Y.-C.; Yue, Y.-H.; et al. Propylene Glycol-Embodying Deformable Liposomes as a Novel Drug Delivery Carrier for Vaginal Fibrauretine Delivery Applications. J. Control. Release 2016, 226, 107–114. [Google Scholar] [CrossRef]
- Ahad, A.; Al-Saleh, A.A.; Al-Mohizea, A.M.; Al-Jenoobi, F.I.; Raish, M.; Yassin, A.E.B.; Alam, M.A. Formulation and Characterization of Phospholipon 90 G and Tween 80 Based Transfersomes for Transdermal Delivery of Eprosartan Mesylate. Pharm. Dev. Technol. 2018, 23, 787–793. [Google Scholar] [CrossRef]
- Zaki, R.M.; Alfadhel, M.M.; Alossaimi, M.A.; Elsawaf, L.A.; Devanathadesikan Seshadri, V.; Almurshedi, A.S.; Yusif, R.M.; Said, M. Central Composite Optimization of Glycerosomes for the Enhanced Oral Bioavailability and Brain Delivery of Quetiapine Fumarate. Pharmaceuticals 2022, 15, 940. [Google Scholar] [CrossRef]
- Ramteke, S.; Barupal, A.; Gupta, V. Preparation and Characterization of Ethosomes for Topical Delivery of Aceclofenac. Indian. J. Pharm. Sci. 2010, 72, 582. [Google Scholar] [CrossRef]
- Manca, M.L.; Zaru, M.; Manconi, M.; Lai, F.; Valenti, D.; Sinico, C.; Fadda, A.M. Glycerosomes: A New Tool for Effective Dermal and Transdermal Drug Delivery. Int. J. Pharm. 2013, 455, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Malviya, N.; Prabakaran, A.; Alexander, A. Comparative Study on Ethosomes and Transferosomes for Enhancing Skin Permeability of Sinapic Acid. J. Mol. Liq. 2023, 383, 122098. [Google Scholar] [CrossRef]
- Ghafourian, T.; Nokhodchi, A.; Kaialy, W. Surfactants as Penetration Enhancers for Dermal and Transdermal Drug Delivery. In Percutaneous Penetration Enhancers Chemical Methods in Penetration Enhancement; Springer: Berlin/Heidelberg, Germany, 2015; pp. 207–230. [Google Scholar]
- Seenivasan, R.; Halagali, P.; Nayak, D.; Tippavajhala, V.K. Transethosomes: A Comprehensive Review of Ultra-Deformable Vesicular Systems for Enhanced Transdermal Drug Delivery. AAPS PharmSciTech 2025, 26, 41. [Google Scholar] [CrossRef]
- Mohd, H.; Dopierała, K.; Zidar, A.; Virani, A.; Michniak-Kohn, B. Effect of Edge Activator Combinations in Transethosomal Formulations for Skin Delivery of Thymoquinone via Langmuir Technique. Sci. Pharm. 2024, 92, 29. [Google Scholar] [CrossRef]
- Cho, W.Y.; Ng, J.F.; Yap, W.H.; Goh, B.H. Sophorolipids—Bio-Based Antimicrobial Formulating Agents for Applications in Food and Health. Molecules 2022, 27, 5556. [Google Scholar] [CrossRef]
- Schneider, M.; Stracke, F.; Hansen, S.; Schaefer, U.F. Nanoparticles and Their Interactions with the Dermal Barrier. Dermatoendocrinology 2009, 1, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Leong, M.Y.; Kong, Y.L.; Burgess, K.; Wong, W.F.; Sethi, G.; Looi, C.Y. Recent Development of Nanomaterials for Transdermal Drug Delivery. Biomedicines 2023, 11, 1124. [Google Scholar] [CrossRef]
- Kang, Y.; Zhang, S.; Wang, G.; Yan, Z.; Wu, G.; Tang, L.; Wang, W. Nanocarrier-Based Transdermal Drug Delivery Systems for Dermatological Therapy. Pharmaceutics 2024, 16, 1384. [Google Scholar] [CrossRef]
- Sakran, W.; Abdel-Rashid, R.S.; Saleh, F.; Abdel-Monem, R. Ethosomal Gel for Rectal Transmucosal Delivery of Domperidone: Design of Experiment, in Vitro, and in Vivo Evaluation. Drug Deliv. 2022, 29, 1477–1491. [Google Scholar] [CrossRef]
- Priya, S.; Desai, V.M.; Singhvi, G. Surface Modification of Lipid-Based Nanocarriers: A Potential Approach to Enhance Targeted Drug Delivery. ACS Omega 2023, 8, 74–86. [Google Scholar] [CrossRef] [PubMed]
- El Kayal, M. Optimization of the colloidal properties of different vesicular systems aiming to encapsulate (-)-epigallocatechin-3-gallate. Farmacia 2020, 68, 97–110. [Google Scholar] [CrossRef]
- Sharma, N.; Madan, P.; Lin, S. Effect of Process and Formulation Variables on the Preparation of Parenteral Paclitaxel-Loaded Biodegradable Polymeric Nanoparticles: A Co-Surfactant Study. Asian J. Pharm. Sci. 2016, 11, 404–416. [Google Scholar] [CrossRef]



| Parameter | Liposomes | Ethosomes | Egzosomes |
|---|---|---|---|
| hydrodynamic diameter (Dh) | Approx. 50 nm–200 nm; depends on ingredients and method of preparation | Approx. 120 nm–400 nm; depends on ingredients | Approx. 80 nm–340 nm |
| polydispersity index (PDI) | <0.3 | Approx. 0.15–0.35 | Approx. 0.2–0.4 |
| zeta potential (ZP) | Approx. −5–(−)40 mV | Approx. −30–(−)60 mV | >(−)30 mV |
| Symbol | Meaning |
|---|---|
| 1 | Ethyl alcohol |
| 2 | n-Butyl alcohol |
| 3 | Isopropyl alcohol |
| 4 | n-Propyl alcohol |
| 5 | tert-Butyl alcohol |
| K | 5% (w/w) phospholipid |
| F | Phospholipid + respective alcohol |
| P | 10% (w/w) Propylene glycol |
| B | 10% (w/w) 1,3-Butanediol |
| xE | 5% (w/w) Ethylene glycol |
| yE | 10% (w/w) Ethylene glycol |
| zE | 15% (w/w) Ethylene glycol |
| M(1+2) | Ethyl alcohol + n-butyl alcohol |
| M(1+3) | Ethyl alcohol + isopropyl alcohol |
| M(1+4) | Ethyl alcohol + n-propyl alcohol |
| M(1+5) | Ethyl alcohol + tert-butyl alcohol |
| x8T | 0.625% (w/w) Tween 80 |
| y8T | 1.00% (w/w) Tween 80 |
| z8T | 1.25% (w/w) Tween 80 |
| xML | 0.625% (w/w) Mirasoft® SL L60 |
| yML | 1.00% (w/w) Mirasoft® SL L60 |
| zML | 1.25% (w/w) Mirasoft® SL L60 |
| xMix | 0.625% (w/w) surfactant mixture |
| yMix | 1.00% (w/w) surfactant mixture |
| zMix | 1.25% (w/w) surfactant mixture |
| H | Homogenization: 3 min; 15,000 rpm |
| Sample | DLS (Dh) [nm] | ±SD [nm] | SEM (Particle Diameter) [nm] | ±SD [nm] |
|---|---|---|---|---|
| yE-z8T-1 | 124 | 1 | 315 | 131 |
| H-yE-z8T-1 | 97 | 1 | 451 | 533 |
| yE-zML-1 | 1165 | 56 | 275 | 144 |
| H-yE-zML-1 | 433 | 3 | 513 | 92 |
| yE-xMix-1 | 254 | 9 | 509 | 335 |
| H-yE-xMix-1 | 157 | 1 | 215 | 64 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chudzińska-Skorupinska, J.; Wawrzyńczak, A.; Feliczak-Guzik, A. Ethosome-Based Colloidal Systems for Transdermal Delivery: The Role of Biosurfactant in Enhancing Stability and Efficacy. Materials 2025, 18, 5355. https://doi.org/10.3390/ma18235355
Chudzińska-Skorupinska J, Wawrzyńczak A, Feliczak-Guzik A. Ethosome-Based Colloidal Systems for Transdermal Delivery: The Role of Biosurfactant in Enhancing Stability and Efficacy. Materials. 2025; 18(23):5355. https://doi.org/10.3390/ma18235355
Chicago/Turabian StyleChudzińska-Skorupinska, Jagoda, Agata Wawrzyńczak, and Agnieszka Feliczak-Guzik. 2025. "Ethosome-Based Colloidal Systems for Transdermal Delivery: The Role of Biosurfactant in Enhancing Stability and Efficacy" Materials 18, no. 23: 5355. https://doi.org/10.3390/ma18235355
APA StyleChudzińska-Skorupinska, J., Wawrzyńczak, A., & Feliczak-Guzik, A. (2025). Ethosome-Based Colloidal Systems for Transdermal Delivery: The Role of Biosurfactant in Enhancing Stability and Efficacy. Materials, 18(23), 5355. https://doi.org/10.3390/ma18235355








