Abstract
This paper presents the results of a study of the effects of the lanthanide ions Ce3+, Pr3+, Nd3+ and Sm3+ on the electronic structure and antioxidant and biological (antimicrobial and cytotoxic) properties of p-coumaric acid (p-CAH2). Structural studies were conducted via spectroscopic methods (FTIR, ATR, UV). Thermal degradation studies of the complexes were performed. The results are presented in the form of TG, DTG and DSC curves. Antioxidant properties were determined via activity tests against DPPH, ABTS and OH radicals. The reducing ability was tested via CUPRAC assays. Minimum inhibitory concentrations (MICs) of the ligand and lanthanide complexes were determined on E. coli, B. subtilis and C. albicans microorganisms. The antimicrobial activity was also determined using the MTT assay. The results were presented as the relative cell viability of C. albicans, P. aeruginosa, E. coli and S. aureus compared to controls and expressed as percentages. In the obtained complexes in the solid phase, lanthanide ions coordinate three ligands in a bidentate chelating coordination mode through the carboxyl group of the acid. Spectroscopic analysis showed that lanthanide ions increase the aromaticity of the pi electron system of the ligand. Thermal analysis showed that the complexes are hydrated and have a higher thermal stability than the ligand. The products of thermal decomposition of the complexes are lanthanide oxides. In the aqueous phase, the metal combines with the ligand in a 1:1 molar ratio. Antioxidant activity tests showed that the complexes have a similar ability to remove free radicals. ABTS and DPPH tests showed that the complexes have twice the ability to neutralise radicals than the ligand, and a much higher ability to remove the hydroxyl radical. The abilities of the complexes and the free ligand to reduce Cu2+ ions in the CUPRAC test are at a similar level. Lanthanide complexes of p-coumaric acid are characterised by a higher antimicrobial capacity than the free ligand against Escherichia coli bacteria, Bacillus subtilis and Candida albicans fungi.
1. Introduction
Lanthanide complexes with ligands of natural origin are characterised by increased antimicrobial and antioxidant activities compared to the ligands or inorganic salts of these metals. In a study by Zheng et al., 3,5-dimethoxybenzoic acid complexes with terbium, dysprosium, erbium and ytterbium were shown to have higher antimicrobial potential against Escherichia coli, Staphylococcus aureus and Candida albicans than the ligand, which had a low effect on bacteria, as did lanthanide chlorides [1]. Zhao et al. (2017) analysed the effect of complexation with Eu(III), Gd(III), Tb(III), Ho(III) and Er(III) ions on the antimicrobial activity of 2-bromo-5-methoxybenzoic acid [2]. The ligand showed no activity against Candida albicans, Escherichia coli and Staphylococcus aureus strains, while the complexes showed enhanced antimicrobial activity, particularly strong against Candida albicans.
In antimicrobial studies of europium, dysprosium and gadolinium complexes with caffeic and p-coumaric acid (p-CAH2), complexes showed a higher activity against E. coli, B. subtilis and C. albicans than the ligand or metal chloride [3].
Complexes of erbium, terbium, dysprosium and holmium with 3-bromo-5-iodobenzoic acid show moderate antimicrobial activity against Candida albicans and Staphylococcus aureus and high activity against Escherichia coli [4]. Samarium and terbium complexes with aminobenzoic acid and 2-amino-5-chlorobenzoic acid show higher antimicrobial activity against S. aureus (G+) and E. coli (G) w bacteria and the fungus C. albicans with reference to free ligands [5].
Lanthanide complexes with heteroaromatic ligands (pyridine, pyrazine derivatives) [6,7] and complexes with flavone compounds [8,9,10] or with Schiff bases [11,12,13,14] also show antimicrobial activity.
The increase in antimicrobial activity of the complexed ligand is caused by a decrease in the polarity of the lanthanide ions during complexation due to the transfer of ligand electrons to the free metal orbitals and an increase in the delocalisation of the pi electron system of the ligand. This results in an increase in lipophilicity and the ability to penetrate the lipid membranes of bacterial cells [2,15].
The formation of lanthanide complexes with organic ligands also alters the antioxidant activity. Complexes of La(III), Sm(III), Eu(III), Gd(III), Tb(III) and Dy(III) with salicylaldehyde-2-picolinoylhydrazone (Schiff base) show a higher antioxidant activity in the DPPH assay than the ligand [16]. The same was observed for N,N’-bis(1-naphthalenediamine)-o-phenylenediamine complexes with the lanthanides Dy(III), Tb(III), La(III), Pr(III), Er(III), Sm(III), Nd(III) and Gd(III). The antiproliferative activity tested by the DPPH assay was higher in the complexes than in the ligand [17].
The reason for the higher antioxidant activity of the tested lanthanide(III) complexes compared to the free ligand is the increase in the polarity of the -OH groups. The H atom then exhibits a greater ionisation capacity in the complex than in the free ligand, accelerating the HAT reaction [17].
The antioxidant activity of ligands can also decrease when complexes are formed with lanthanides (also other metals). Complexation of cerium, lanthanum and neodymium with aminoorotic acid results in a decrease in the antioxidant activity of the ligand [18]. Also, in the case of complexes of europium, dysprosium and gadolinium with caffeic acid, a reduced activity of the complexes was observed compared to the ligand [3]. In this case, this was due to the formation of the complex by metal binding to the hydroxyl groups of the aromatic ring of the ligand, which were responsible for the anti-radical properties of caffeic acid. Also, complexation of quercetin with metal ions may reduce its antioxidant activity. The terbium (III) complex with quercetin shows a lower antiradical activity than the free quercetin ligand. The complex is formed by metal attachment to the ligand via hydroxyl groups, causing a decrease in antioxidant activity [19].
Studies have shown that compounds from the phenolic acid group can exhibit higher antioxidant activity when complexed with metals. An example of such a compound is p-CAH2, which exhibits a high antioxidant potential [20,21]. Our previous studies have shown that complexes of this acid with transition metals, as well as with some lanthanides, have higher antimicrobial and antioxidant activities than the ligand [3,22]. This paper presents the results of a study of the structure and biological properties of Ce(III), Pr(III), Nd(III) and Sm(III) complexes with p-CAH2.
2. Materials and Methods
2.1. Synthesis
Different amounts of p-CAH2 acid (0.001 mol) were dissolved in 10 cm3 of a 0.1 mol/dm3 aqueous sodium hydroxide solution. The mixtures were incubated in a shaker at 50 °C for 1 h. To the clear solution was added an aqueous solution of lanthanide chloride (cerium, neodymium, praseodymium, samarium) at a concentration of 0.1 mol/dm3 in a stoichiometric amount of 3:1 (ligand/metal), and the whole mixture was stirred in a shaker for 2 h at 50 °C. The turbid solutions were left for 48 h to precipitate. The resulting complexes were drained using filters and then washed to elute residual chlorides. The resulting precipitates were dried at room temperature in a desiccator for 72 h and then stored in airtight vessels. The synthesis was carried out three times. The obtained sediments were studied by measuring ATR spectra. The repeatability of the synthesis of the obtained substances was determined, both for the freshly precipitated sediments and for the sediment collected after 48 h. It was not possible to obtain the substance in a crystalline form useful for examining its structure using the X-ray diffraction method.
2.2. Elemental and Thermal Analysis
Elemental analysis was carried out on a Perkin Elmer Series Analyser II CHNS (Waltcham, MA, USA). The percentage of carbon and hydrogen in the samples was determined. Measurements were taken three times for each sample and the average results were calculated. Thermal analysis was performed on an STA 600 Frontier thermal analyser (Perkin Elmer, Waltham, MA, USA). Aliquots of samples (approximately 5 mg) were annealed in a ceramic crucible over a temperature range of 40–900 °C in an air atmosphere at a heating rate of 10 °C/min. Thermal decomposition products were analysed based on recorded TG, DTG and DSC curves.
2.3. Spectroscopic Study
FTIR spectra were recorded for samples prepared as pressed KBr pellets (1 mg sample per 200 mg KBr) and via the ATR multi-reflectance technique. Spectra were recorded on a Bruker alpha instrument (Billerica, MA, USA) in the range 4000–400 cm−1 (number of scans 32, resolution 1 cm−1). Raman spectra were recorded for solid-phase samples on a MultiRam instrument from Bruker (Billerica, MA, USA) (measuring resolution 1 cm−1, number of scans 124, laser power of 200 mW). Ultraviolet (UV) spectra were recorded on an HACH 2000 DR instrument in the range of 200–400 nm, with a resolution of 0.1 nm. UV spectra were recorded in aqueous solutions at a concentration of 5 × 10−4 mol/dm3. Measurements were performed for complexes with a molar ratio of 3:1 (ligand/metal). Solutions were obtained by dissolving solid substances in an aqueous solution. The composition of the complexes in aqueous solutions was examined spectrophotometrically (UV) at pH = 7.4.
Spectroscopic and thermal characterisation of the tested complexes was performed in the solid phase. Antioxidant and microbiological tests were performed in aqueous solutions. Before preparing appropriate solutions for testing, analyses of the composition of the complexes in aqueous solutions were performed. The results of these analyses were used to prepare complexes for testing in solutions with an appropriate composition (molar ratio of metal to ligand).
2.4. Antioxidant Study
The antioxidant properties of the tested compounds were determined using DPPH (reaction with the 2,2-Diphenyl-1-picrylhydrazylradical), ABTS (reaction with the 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid radical) and CUPRAC tests (based on the reduction of Cu2+ to Cu1+). The assays were performed in accordance with the methodology described in the literature [23,24]. Absorbance measurements were performed using a UV-VIS Carry 5000 spectrophotometer Agilent (Santa Clara, CA, USA). Solutions of Ce(III), Pr(III), Nd(III) and Sm(III) complexes with p-CAH2 with a metal–ligand composition of 1:1 were prepared in accordance with the results of previous studies on the composition of the complexes in aqueous solution. First, 10% methanolic solutions of p-CAH2 (0.05 and 0.005 mol/L) were added to aqueous sodium hydroxide solution (0.1 mol/L) in a molar ratio of 1:1. Then, aqueous solutions of Ce(III), Pr(III), Nd(III) and Sm(III) chlorides (0.01 mol/L) were prepared and mixed with sodium salts of the ligand (1:1 molar ratio). The final concentration of metal complexes for the determination of DPPH was in the range of 2.00–20.00 mmol/L, while for the ABTS test it was 1.0–16.0 µmol/L and for the CUPRAC test it was 31.25 µmol/L. All experiments were performed with five replicates in three independent experiments and values are expressed as means ± SD.
The antioxidant activity towards the hydroxyl radical was determined spectrophotometrically. The radical was produced by the reaction of Fe2+ ions (FeSO4) with H2O2. The radical was then quenched by reaction with salicylic acid, resulting in the coloured compound 2,3-dihydroxybenzoic acid (2,3DHBA) with an absorbance maximum at λ = 510 nm. In the reaction with antioxidant compounds, the amount of the resulting coloured 2,3DHBA acid decreased, which was the basis for the spectrophotometric determination of the quenching capacity of the OH radical. The antioxidant activity was determined for a series of solutions of the tested complexes at a concentration of 0.3–1 mmol/dm3.
2.5. Minimal Inhibitory Concentration Test
Microorganism commonly found in the environment were selected for this study. They are typically used in basic tests aimed at examining the antimicrobial properties of chemical compounds. A series of dilutions of the tested compounds in sterile Mueller–Hinton agar (Oxoid) were made in order to determine the MIC values. In the next step, the appropriate inoculum of microorganisms was added to each agar medium and incubated at 37 °C for 24 h. C. albicans (PCM 2566-FY), E. coli (PCM 2857) and B. subtilis (PCM 2850) came from the Polish Collection of Microorganisms (Wroclaw, Poland). The microorganisms were grown overnight and then were resuspended in physiological saline to obtain an optical density at 600 nm (OD600) of 0.60, corresponding to 5.0 × 108 CFU/ml. The tested compounds were dissolved in DMSO solution (Sigma-Aldrich). The agar plates with DMSO were the negative controls, while the plates with gentamicin (Sigma-Aldrich, 345815) were the positive control for bacteria, and fluconazole agar plates (Sigma-Aldrich, F-031) were a positive control for fungi. The lowest concentration without visible bacterial growth was determined as the MIC (minimal inhibitory concentration).
2.6. Cell Viability of Tested Microorganisms (MTT Test)
Staphylococcus aureus (ATCC 25923), Escherichia coli (ATCC 25922), Pseudomonas aeruginosa (ATCC 27853) and Candida albicans (ATCC 10231) were obtained from the ATCC (American Type Culture Collection (Manassas, VA, USA)). The studied strains of microorganisms were cultured in MH II (Mueller–Hinton II broth) for 18 h at 37 °C (P. aeruginosa, E. coli and S. aureus) and at 26 °C for C. albicans. Then, cultures were diluted in fresh MH II to obtain 108 CFU/mL (colony forming units per mL). The inoculum with antimicrobial activities of 106 CFU/mL for the suspension of P. aeruginosa, E. coli and S. aureus cells and 104 CFU/mL for C. albicans cells was used.
The preparation of serial two-fold dilutions of the studied p-CAH2 and Ce(III), Pr(III), Nd(III) and Sm(III) complexes was carried out according to Jabłońska-Trypuć et al. [25]. The final concentrations of tested compounds in each well were 2.5 mM, 1.25 mM, 0.63 mM, 0.31 mM, 0.16 mM, 0.08 mM, 0.04 mM, 0.02 mM and 0.01 mM. The study was carried out in aqueous solutions of complexes with a molar ratio of ligand to metal of 1:1.
The antimicrobial activity of p-CAH2 and complexes against C. albicans, P. aeruginosa, E. coli and S. aureus was determined using an MTT assay as described in [26]. The antimicrobial activity of the tested compound was expressed as the relative cell viability of C. albicans, P. aeruginosa, E. coli and S. aureus in comparison to the control and presented as a percentage. The determination of antimicrobial activity in all samples was performed in triplicate.
2.7. Statistical Analysis
The differences between the obtained means for the relative cell viability of C. albicans, P. aeruginosa, E. coli and S. aureus were evaluated by a one-way analysis of variance (ANOVA) and significant differences were estimated by Dunnett’s test at p < 0.05, p < 0.01 and p < 0.001 using STATISTICA.
3. Results and Discussion
3.1. Elemental and Thermal Analysis
Table 1 shows the results of the elemental analysis of p-CAH2 complexes with Ce(III), Pr(III), Nd (III) and Sm(III). The results showed that, in the solid state, these metals form complexes with p-CAH2 in a molar ratio of 1:3 (metal/ligand). The complexes (Ce(III), Pr(III) and Sm(III) cerium, praseodymium and samarium) were hydrated and contained six water molecules each, while the Nd(III) complex contained five water molecules.

Table 1.
Results of elemental analysis of p-CAH2 complexes with Ce(III), Pr(III), Nd(III) and Sm(III).
The degree of hydration of the obtained complexes was confirmed via thermal analysis of the complexes studied. The thermal analysis made it possible to characterise the type of water present in the molecules. Each of the complexes contained three hydration water molecules and three coordination water molecules. Figure 1 shows the thermal decomposition curves (TG, DTG and DSC) of the p-CAH2 complexes with Ce(III), Pr(III), Nd(III) and Sm(III). Table 2 shows the results of the thermal analysis of the studied complexes, i.e., the temperatures of the individual decomposition steps (dehydration, thermal decomposition) and the decomposition products. The first stage of the thermal decomposition of the studied complexes is dehydration. This process occurs at temperatures between 60 °C and 130 °C. In this stage, the complexes lose three hydration water molecules each. The next step in the thermal decomposition of the complexes is the loss of coordination water. This process occurs in the temperature range of 180 to 240 °C. After the loss of water, further heating of the complexes leads to their degradation. All the complexes studied are characterised by a similar thermal stability.
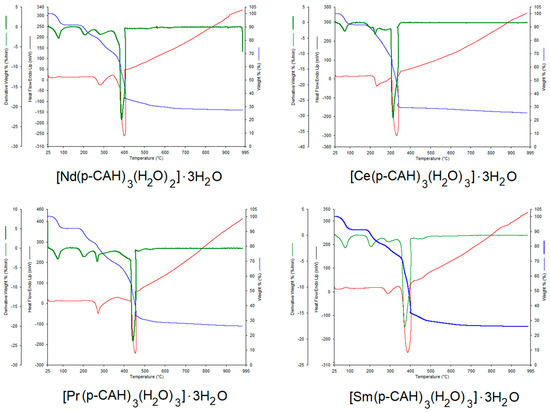
Figure 1.
TG, DTG and DSC curves for lanthanide complexes: [Ce(p-CAH)3(H2O)3]·3H2O, [Nd(p-CAH)3(H2O)2]·3H2O, [Pr(p-CAH)3(H2O)3]·3H2O and [Sm(p-CAH)3(H2O)3]·3H2O.

Table 2.
Thermal decomposition parameters (TG, DTG and DSC) of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III).
The degradation process of the lanthanide p-coumarates starts immediately after the dehydration process, i.e., at a temperature of about 240 °C. The least thermally stable complex is samarium p-coumarate. Decomposition of this compound starts at 220 °C. The complexes studied undergo thermal decomposition to metal oxides (Ce2O3, Pr2O3, Nd2O3, Sm2O3). The thermal degradation of lanthanide complexes occurs in several stages, with the individual steps not clearly observed in the degradation curves. One of the first steps in the thermal degradation is probably the decarboxylation of the compounds under investigation, with decomposition of the aromatic ring occurring in a further step. The final products are obtained at temperatures of about 360–400 °C. These products are lanthanide oxides, containing small residual amounts of organic carbon, which undergo slow afterburning to a temperature of about 800 °C (increase in the DSC curve). This is followed by stabilisation of the process—pure lanthanide oxides are formed. The dehydration process of the complexes is endothermic. However, the peaks related to the process of hydration water detachment and coordination were practically invisible in the DSC curves. High-intensity peaks related to the exothermic process of thermal decomposition of the decomposed complexes were observed in the DSC curves. This is related to the combustion of organic carbon in the process of thermal decomposition in an oxygen atmosphere.
3.2. IR and Raman Spectra
Figure 2 shows the spectra recorded in the infrared (FTIR KBr) for p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III). The spectra cover the spectral range 600–1800 cm−1. Selected bands from the vibrations of the carboxyl group/carboxylate anion and the vibration bands of the aromatic system are marked. Table 3 summarises the wavenumbers of the bands present in the FTIR spectra of the studied compounds recorded in a KBr matrix via the ATR and Raman techniques, together with the assignments according to the numbering of normal vibrations given by Versanyi [27]. After complexation of p-CAH2, vibrational bands originating from the carboxylate anion are observed in the spectra of the complexes. These are asymmetric stretching vibrations denoted as νasCOO− present at 1514–1513 cm−1 (IR KBr), 1510 cm−1 (ATR) and 1519–1517 cm−1 (Raman) and symmetric νsCOO− present in the range of 1409–1405 cm−1 (IR KBr), 1399–1397 cm−1 (ATR) and 1400–1396 cm−1 (Raman).
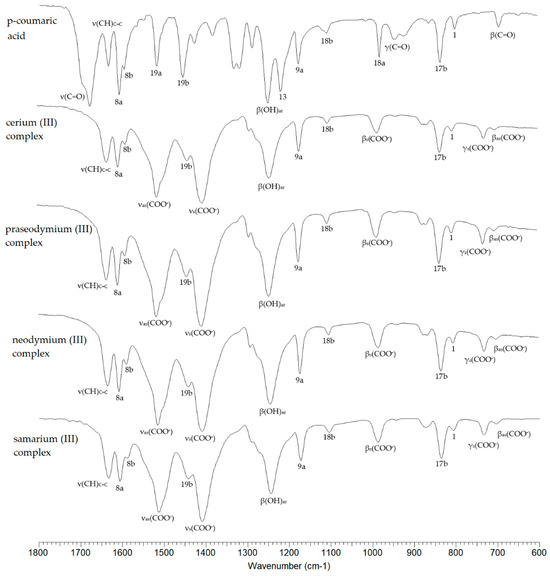
Figure 2.
FTIRKBr spectra of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III).
The difference in the values of the wavenumbers of the asymmetric and symmetric stretching vibrations of the carboxylate anion (Δν) can indicate the type of metal–ligand coordination in the complex according to the criteria described by Nakamoto [28]. In the complexes studied, this difference is 104–109 cm−1, which is much lower than the value (Δν) for the sodium salt [29]. This indicates a type of bidentate chelating coordination (Figure 3).
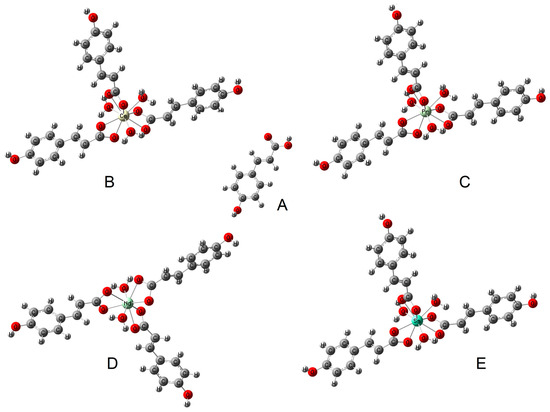
Figure 3.
The type of coordination occurring in the studied complexes: (B) cerium complex, (C) praseodymium complex, (D) neodymium complex, (E) samarium complex (based on FTIR study) and (A) structure of ligand (p-coumaric acid).
Also present in the spectra of lanthanide complexes of p-CAH2 are bands associated with symmetric and asymmetric bending vibrations in the ring plane (βsCOO− and βasCOO− vibrations) and bending vibrations outside the ring plane (γCOO−). Metal complexation of aromatic carboxylic acids affects the π-electron arrangement of the aromatic ring of the ligand, which consequently affects the reactivity of the chemical compound and its biological activity. Studies have shown that alkali metals destabilise the electron arrangement of the aromatic ring [30], while 3d transition metals and 4f metals can stabilise the electron arrangement of the ligand. Electron system stabilisation is related to electron transfer to the electron system of the metal. This effect can be observed by comparing the infrared and Raman spectra of the ligand and the complex. In 3d transition metal and lanthanide complexes, a shift in the bands towards higher wavenumbers and an increase in band intensities are observed.
In the case of the spectra of Ce(III), Pr(III), Nd(III) and Sm(III) complexes, an increase in the wavenumbers of most of the bands of the aromatic system relative to the ligand was observed. Here, we mention the bands numbered 20b, 8a, 17a, 10a, 17b, 1. The bands numbered 9a and 18b do not change position in the spectra of the complexes relative to the spectrum of the ligand. A few bands in the spectra of the complexes are shifted towards higher wavenumber values relative to their position in the spectrum of p-CAH2 (7b, 8b, 19b). The change in the position of the bands confirms that in the case of complexation of p-CAH2 with lanthanide ions (Ce(III), Pr(III), Nd(III) and Sm(III)), an increase in the stability of the electron arrangement of the aromatic ring of the ligand is observed. An increase in the aromaticity of the ring reduces its reactivity in substitution reactions. At the same time, a change in the electron charge distribution of the aromatic ring will affect the electron density distribution of the hydroxyl group directly attached to the aromatic system, which will affect the antioxidant activity of lanthanide complexes with p-CAH2.


Table 3.
Wavenumbers of the bands present in the FTIR spectra of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) recorded via KBr matrix, ATR and Raman techniques.
Table 3.
Wavenumbers of the bands present in the FTIR spectra of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) recorded via KBr matrix, ATR and Raman techniques.
| p-CAH2 [29] | [Ce(p-CAH)3(H2O)3]·3H2O | [Pr(p-CAH)3(H2O)3]·3H2O | [Nd(p-CAH)3(H2O)2]·3H2O | [Pr(p-CAH)3(H2O)3]·3H2O | Assignment | No. [27] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| IRKBr | IRATR | Raman | IRKBr | IRATR | Raman | IRKBr | IRATR | Raman | IRKBr | IRATR | Raman | IRKBr | IRATR | Raman | ||
| 3383 s | 3375 w | - | 3394 m | - | - | 3422 m | - | - | 3378 m | - | - | 3357 m | - | - | ν(OH)ar | - |
| - | - | - | 3256 m | - | - | 3251 m | - | - | 3251 m | - | - | 3256 m | - | - | ν(OH)ar | - |
| 3026 w | - | 3025 vw | 3024 w | - | - | 3027 w | - | 3024 vw | 3025 w | - | - | 3027 w | - | - | ν(CH) + ν(CH)C=C | 20b |
| 2959 m | - | - | 2955 w | - | - | 2952 vw | - | - | 3952 vw | - | - | 2955 vw | - | - | ν(CH + ν(CH)C=C | 7b |
| 2839–2513 m | - | - | ν(OH) | - | ||||||||||||
| 1672 vs | 1667 vs | - | - | - | - | - | - | - | - | - | - | - | - | - | ν(C=O) | - |
| 1628 s | 1626 m | 1636 m | 1633 s | 1633 m | 1635 s | 1633 s | 1633 m | 1636 s | 1633 s | 1633 s | 1636 s | 1633 s | 1633 m | 1633 s | ν(CC)C=C | - |
| 1601 vs | 1600 s | 1606 vs | 1606 s | 1603 m | 1607 vs | 1606 s | 1603 m | 1608 vs | 1606 s | 1605 s | 1607 vs | 1606 s | 1605 m | 1605 vs | ν(CC) | 8a |
| 1591 s | 1589 s | 1593 m | 1589 m | - | - | 1589 m | - | - | 1589 m | - | - | 1588 m | - | - | ν(CC) | 8b |
| 1512 s | 1510 m | 1519 w | - | - | 1547 w | - | - | - | - | - | 1547 w | - | - | - | ν(CC) | 19a |
| - | - | - | 1514 vs | 1510 s | 1517 w | 1514 vs | 1510 vs | 1517 w | 1514 vs | 1510 vs | 1519 w | 1513 vs | 1510 vs | 1518 w | νas(COO) | - |
| 1449 vs | 1446 s | 1448 w | 1440 m | 1440 m | 1435 w | 1440 m | 1443 m | 1433 w | 1440 s | 1441 s | 1435 w | 1442 m | 1439 m | 1439 w | ν(CC) | 19b |
| 1422 s | 1422 m | - | - | - | - | - | - | - | - | - | - | - | - | - | β(CH)C=C | - |
| - | - | - | 1405 vs | 1397 vs | 1397 w | 1405 vs | 1398 vs | 1400 w | 1406 vs | 1399 vs | 1396 w | 1409 vs | 1399 vs | 1400 w | νs(COO-) | - |
| 1379 m | 1376 m | - | β(CH)C=C + β(OH)ar | 14 | ||||||||||||
| 1314 s | 1306 w | 1322 vw | - | 1321 vw | - | - | 1320 w | - | - | 1321 vw | - | - | 1323 vw | β(CH)C=C + β(OH)ar | 3 | |
| 1283 m | 1309 s | 1282 w | 1292 m | - | 1292 vw | 1292 w | - | 1293 vw | 1292 m | - | 1291 vw | 1289 sh | - | 1293 vw | ν(C-OH) | |
| 1244 vs | 1241 vs | 1260 m | 1244 s | 1239 s | 1249 m | 1244 s | 1239 s | 1249 m | 1244 s | 1241 vs | 1250 m | 1244 s | 1239 s | 1251 m | β(OH) | - |
| 1215 s | 1211 s | 1212 w | - | - | 1204 w | - | - | 1204 w | - | - | 1202 w | - | - | 1205 w | β(CH) | 13 |
| 1173 s | 1171 s | 1171 w | 1173 m | 1171 m | 1171 m | 1173 m | 1171 m | 1171 w | 1173 m | 1171 s | 1171 w | 1173 m | 1171 m | 1170 m | β(CH) | 9a |
| 1105 m | 1103 m | - | 1105 w | 1106 w | - | 1105 w | 1109 w | - | 1105 w | 1106 s | - | 1105 w | 1106 m | - | β(CH) | 18b |
| 1013 w | 1012 w | - | β(CH) | 18a | ||||||||||||
| 986 m | 986 w | 984 vw | 986 w | 986 m | 985 vw | 984 m | 986 s | 987 vw | 987 w | 986 m | 980 w | βs(COO) | ||||
| 978 s | 977 s | 977 vw | ν(CCO) | |||||||||||||
| 941 m | 937 m | 952 vw | 943 w | - | - | - | - | - | 942 w | 942 s | - | 943 vw | - | - | γ(CH) | 17a |
| 920 s | 912 m | - | - | - | - | - | - | - | γ(CO) | - | ||||||
| - | - | - | 877 m | 873 w | - | 877 w | 873 w | - | 875 w | 873 s | - | 873 w | 870 m | 867 w | γ(CH) | 5 |
| 860 w | - | 864 w | 867 m | - | 867 vw | 867 w | - | 866 vw | 867 w | - | 867 vw | - | - | - | γ(CH) | 10a |
| 833 s | 827 s | 837 w | 836 s | 834 m | 837 vw | 836 m | 834 m | - | 834 m | 834 s | - | 836 m | 834 s | - | γ(CH) | 17b |
| 799 m | 797 m | 800 w | 806 m | 805 w | 806 vw | 806 w | 807 m | 806 vw | 806 m | - | 807 vw | 806 w | 807 m | 806 w | α(CCC) | 1 |
| - | - | - | 731 m | 728 m | - | 731 m | 728 m | - | 731 m | 731 s | - | 732 w | 732 m | 732 vw | γs(COO) | - |
| 704 m | - | - | 704 w | 704 m | - | 703 w | 700 s | - | 704 vw | 703 m | 706 vw | βas(COO) | - | |||
| 692 m | 690 m | - | - | - | - | - | - | - | - | - | - | - | - | β(CO) | - | |
| 646 w | 645 w | 645 vw | 643 w | - | 642 vw | - | - | 643 vw | 642 w | - | 643 vw | 643 vw | 630 m | 642 vw | φ(CC) | 4 |
3.3. UV Spectra
Figure 4 shows the UV spectra of aqueous solutions of p-CAH2 and its complexes with Ce(III), Pr(III), Nd (III) and Sm(III). In the UV spectrum of p-CAH2 recorded in aqueous solution at a concentration of 5 × 10−5 mol/dm3 in the range 200–450 nm, one band is observed, the absorption maximum of which is located at a wavelength of λmax = 285.5 nm. This band is responsible for the electron transition in the ligand’s π→π* aromatic ring. In the spectra of light lanthanide complexes with p-CAH2, this band undergoes a hypsochromic shift. For the cerium complex, this band is located at λmax = 285.0 nm; for samarium, λmax = 276.0 nm; for praseodymium, λmax = 275.5 nm; and for the neodymium complex, λmax = 273.5 nm. Analysis of the UV spectra showed that there is a shift in the absorption maxima of the π→π* band in the spectra of the complexes relative to the band present in the spectrum of the ligand. A change in the intensity of these bands in individual complexes was also observed (generally, an increase in intensity). This proves the influence of the lanthanide ion on the aromatic system of the ligand (changes in the position of the π→π*band), as well as on the change in the distribution of the electron density of the hydroxyl group substituted on the aromatic ring. In turn, this affects the ability of hydroxyl group to react with free radicals. Light lanthanide ions are known to have similar physicochemical properties; therefore, they have a similar effect on the electron system of the ligand. Therefore, the antioxidant activity of the complexes is at a similar level. The UV spectra of solutions of lanthanide complexes with different metal/ligand molar ratios were also recorded. This was important because antioxidant and microbiological tests were performed on these compounds in solutions.
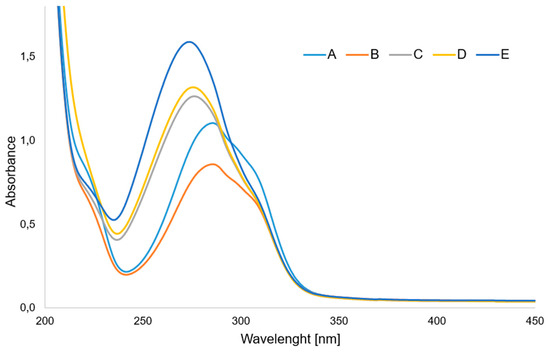
Figure 4.
UV spectra of aqueous solution of (A) p-CAH2 and its complexes with (B) Ce(III), (C) Sm(III), (D) Pr(III) and (E) Nd(III). (Molar ratio of ligand to metal—3:1).
Figure 5 shows the dependence of the absorbance of lanthanide complexes with p-CAH2 on the composition of the complex in aqueous solutions. The course of the curves indicates that at concentrations of 5 × 10−5 mol/dm3, metal–ligand complexes are formed in a molar ratio of 1:1. This study was carried out in aqueous solutions of tris HCl buffer with pH = 7.4.
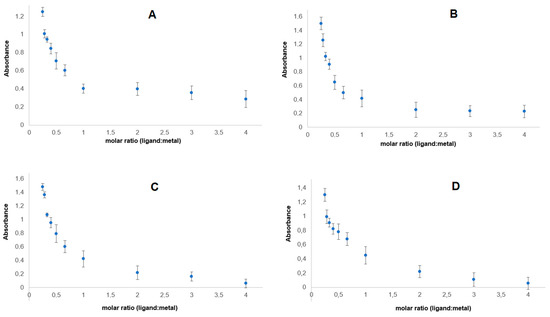
Figure 5.
Dependence of absorbance on the composition of lanthanide complexes of p-CAH2 with (A) Ce(III), (B) Nd(III), (C) Pr(III) and (D) Sm(III).
3.4. Antioxidant Properties
The antioxidant properties of Ce(III), Nd(III), Pr(III) and Sm(III) complexes of p-CAH2 were studied by means of four different spectroscopic assays. The final concentration of metal complexes for the determination of DPPH was in the range of 2.00–20.00 mmol/L, while for the ABTS test it was 1.0–16.0 µmol/L and for the CUPRAC test it was 31.25 µmol/L. The molar ratio of metal to ligand was 1:1, which is consistent with the results obtained in Job’s study. The results of the tests carried out are shown in Figure 6.
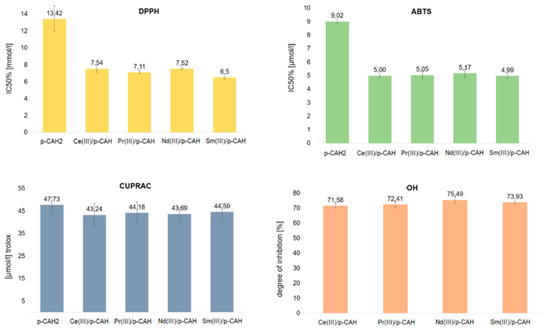
Figure 6.
Antioxidant activity of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) determined by DPPH, ABTS, CUPRAC and OH assays. (Molar ratio of ligand to metal—1:1).
All complexes of p-CAH2 showed higher antioxidant properties than the free ligand. DPPH and ABTS assays are related to mixed HAT (hydrogen atom transfer) and SET (single-electron transfer) mechanisms that are generally called SPLET (sequential proton loss–electron transfer) or SET-PT (single-electron transfer–proton transfer) mechanisms [31]. In these tests, the tested complexes were characterised by a nearly two times higher antioxidant activity than the free ligand. However, it should be noted that the activity of individual complexes remains at a fairly similar level. The CUPRAC methods rely on the reduction of metal ions Cu2+ in the presence of substances with antioxidant properties.
The measurement of the reducing capacity of a given chemical compound was carried out spectrophotometrically for the complex compounds formed with reduced ions. The antioxidant activity in the CUPRAC assay is expressed as Trolox equivalents. In the CUPRAC assay, the Ce(III), Nd(III), Pr(III) and Sm(III) complexes with p-CAH2 were shown to have a lower reducing capacity than the free ligand. However, the decrease in the reducing capacity of the complexes with respect to the ligand was small and was within the statistical error limit.
The Cu2+ cation reduction method (CUPRAC) and the reaction with the hydroxyl radical are based on different mechanisms of antiradical activity. In the case of the reduction of Cu2+ cations, no significant difference was noted between the effects of the ligand and the complexes.
Studies of antioxidant activity against the hydroxyl radical OH have shown that lanthanide complexes with p-CAH2 at a concentration of 0.5 mmol inhibit the action of the radical by approximately 70% (Table 4). The highest degree of inhibition is shown by the neodymium complex (75.5%) and the lowest by the cerium complex (71.6%). In the case of p-CAH2, no OH radical inhibitory activity was observed in the range of complex concentrations tested. p-CAH2 exhibits a significantly lower antioxidant activity against the OH radical than that of the tested complexes. In the range of the tested concentrations of lanthanide complexes with inhibitory effects on OH radicals, p-CAH2 did not show such properties.

Table 4.
Antioxidant activity of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) determined by DPPH, ABTS, CUPRAC and OH assays. (Molar ratio of ligand to metal—1:1).
3.5. Antimicrobial Properties
Table 5 shows minimal inhibitory concentration (MIC) of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III), as well as the minimal inhibitory concentration (MIC) values for antibiotics against selected microorganisms. C. albicans shows the greatest resistance to both p-CAH2 and its complexes with metals ions in comparison to Gram-positive and Gram-negative bacteria. It has been shown that p-CAH2 has a strong limiting effect on the growth of pathogenic fungi and stimulates numerous defence mechanisms in host cells [32]. However, C. albicans is known to be a pathogen that forms multimicrobial biofilms with various bacteria. These biofilms are formed by communities of microorganisms belonging to different kingdoms. These microorganisms interact with each other through various molecular mechanisms [33]. For this reason, this yeast very often shows a high resistance to various antifungal substances, which makes it difficult to treat fungal infections. Complexes of p-CAH2 with metal ions have a much stronger fungicidal and bactericidal effect. Complexes of metal ions with various organic compounds have been widely considered as potential antifungal and antibacterial drugs for some time. Metal complexes with amino acids have been shown to have an antibacterial effect against both Gram-positive and Gram-negative bacteria and against fungal pathogens [34]. In addition, metal nanoparticles are used as carriers of antifungal drugs to increase their biocompatibility and effectiveness [35]. As a result of the conducted analyses, it was shown that metal ions significantly increase the growth-inhibiting properties of p-CAH2 in relation to yeast, as well as Gram-positive and Gram-negative bacteria.

Table 5.
Minimal inhibitory concentration (MIC) of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) for selected strains of microorganisms [mmol/L].
3.6. Cell Viability of Tested Microorganisms
To determine the effect of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) on E. coli, C. albicans, S. aureus and P. aeruginosa, an MTT assay was used. As presented in Figure 7, in the case of C. albicans, it was observed that p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) caused an insignificant decrease in microorganism growth with an increase in the concentration of the studied acids, where the highest value was approximately 20%. The highest inhibition for C. albicans was observed after samarium and neodymium complex application in the concentration range of 0.63 to 2.5 mM (Figure 7).
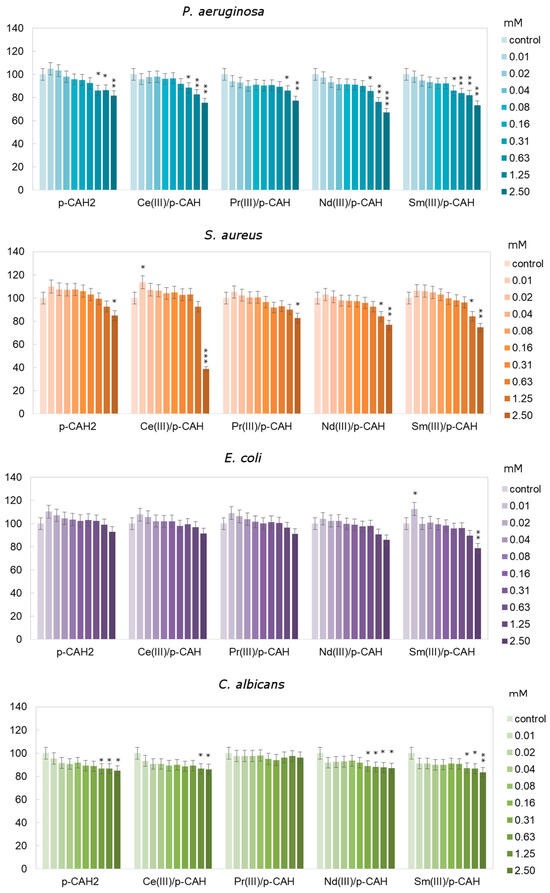
Figure 7.
Different relative cell viability of C. albicans, P. aeruginosa, E. coli and S. aureus after 24 h depending on the concentration of p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) (*—p < 0.05, **—p < 0.01 and ***—p < 0.001 indicate significant differences between treatments and control, which were evaluated via Dunnett’s test; error bars represent standard deviation ± SD). (Molar ratio of ligand to metal—1:1).
For p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III), the average relative viability of P. aeruginosa was approximately between 1 and 18% compared to untreated cells. After the application of acid complexes at concentrations of 1.25 to 2.5 mM, a significant decrease in cell viability of approximately of 19 to 31% was observed compared to the control cells.
The result obtained for S. aureus showed that p-CAH2 and its complexes with Ce(III), Pr(III), Nd(III) and Sm(III) caused inhibition of the growth of the tested bacteria with increasing concentrations of the analysed acid. It was observed that the highest level of inhibition (>50%) was obtained for 2.5 mM of the cerium complex compared to the control cells. Moreover, the application of p-CAH2 and the cerium complex significantly increased the number of S. aureus in the concentration range of 0.01 to 0.31 mM by approximately 7%.
After application of the complexes on E. coli at the lowest concentrations (from 0.01 to 0.16 mM), an increase in relative cell viability of 1 to 12% was observed. At concentrations of 0.31 to 2.5 mM, there was a decrease in the relative number of viable cells of 1 to 22% compared to the untreated control cells (Figure 7).
4. Conclusions
P-coumaric acid is a compound of natural origin found in many plants (fruits, vegetables, herbs). It is characterised by high antioxidant and antimicrobial potentials. Complexing the acid with metal ions may change its activity. Using lanthanide ions in paracoumaric acid complexes stabilises the aromatic system in the ligand, which was observed by analysing the infrared and Raman spectra of the tested compounds. Changing the electronic charge distribution in the aromatic ring increases the antioxidant activity by activating the hydroxyl group, which participates in radical quenching processes. In solid-phase lanthanide complexes (cerium, samarium, praseodymium and neodymium), the central atom is attached to two molecules of p-coumaric acid through its carboxyl groups. Thermal analysis showed that the complexes were hydrated and had a higher thermal stability than the ligand. Studies have shown that complexation of p-coumaric acid changes its biological activities. Lanthanide complexes (praseodymium, neodymium, samarium and cerium) showed almost twice the antioxidant activity in tests with ABTS and DPPH radicals. In studies of antioxidant activity with the hydroxyl radical, the complexes showed a much stronger radical inhibiting effect than the uncomplexed ligand. Complexing p-coumaric acid with lanthanide ions also affects its antimicrobial properties. Complexes of p-CAH2 with metal ions have a much stronger fungicidal and bactericidal effect. Lanthanide complexes with biologically derived ligands are of increasing interest due to their many beneficial properties. There are many reports in the literature showing that these compounds have high antimicrobial, antioxidant and even anticancer potentials. The results of the analyses carried out in this work and in our other works have shown that complexes of lanthanides with phenolic acids are compounds that could be potentially used as biologically active agents. Complexing lanthanide ions with aromatic acids stabilises the aromatic system, thereby influencing the reactivity and biological activity of these compounds.
Author Contributions
Conceptualisation, G.Ś. and M.K; methodology, G.Ś. and M.K.; investigation, E.G., M.K., G.Ś., R.Ś., A.C., A.P., P.M., U.W. and E.W., curation G.Ś., N.K., E.G., M.K., A.C., A.P., E.W. and U.W.; writing—original draft preparation, G.Ś., N.K., E.G., A.P., E.W. and U.W.; writing—review and editing, W.L., G.Ś., R.Ś. and W.P.; visualisation, E.G., G.Ś., U.W., E.W. and A.P.; supervision, G.Ś.; project administration, M.K. and G.Ś.; funding acquisition, W.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Science Centre (Poland), grant no. 2018/31/B/NZ7/03083.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Zheng, J.; Ren, S.; Ren, N.; Zhang, J.; Zhang, D.; Wang, S. Synthesis, thermodynamic properties and antibacterial activities oflanthanide complexes with 3,5-dimethoxybenzoic acid and1,10-phenanthroline. Thermochim. Acta 2013, 572, 101–106. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhu, M.; Zhang, J. A series of novel lanthanide complexes with 2-bromine-5-methoxybenzoic acid and 2,20-bipyridine: Syntheses, crystal structures, and luminescent properties. J. Mol. Struct. 2017, 1149, 171–182. [Google Scholar] [CrossRef]
- Świderski, G.; Kalinowska, M.; Gołębiewska, E.; Świsłocka, R.; Lewandowski, W.; Kowalczyk, N.; Naumowicz, M.; Cudowski, A.; Pietryczuk, A.; Nalewajko-Sieliwoniuk, E.; et al. Structures, Antioxidant Properties, and Antimicrobial Properties of Eu(III), Gd(III), and Dy(III) Caffeinates and p-Coumarates. Molecules 2023, 28, 6506. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Ren, N.; Zhang, J.J.; Zhang, C.Y. Preparation, thermodynamic property and antimicrobial activity of some rare-earth (III) complexes with 3-bromo-5-iodobenzoic acid and 1, 10-phenanthroline. Thermochim. Acta 2013, 570, 51–58. [Google Scholar] [CrossRef]
- Essawy, A.A.; Afifi, M.A.; Moustafa, H.; El-Medani, S.M. DFT calculations, spectroscopic, thermal analysis and biological activity of Sm (III) and Tb (III) complexes with 2-aminobenzoic and 2-amino-5-chloro-benzoic acids. Spectrochim. Acta 2014, 131, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Xu, Y.; Cheng, N.; Zhou, X.; Shi, Y.; He, Q. Synthesis, characterization, and biological studies of lanthanide complexes with 2, 6-pyridine dicarboxylic acid and α-picolinic acid. J. Coord. Chem. 2010, 63, 2360–2369. [Google Scholar] [CrossRef]
- Premkumar, T.; Govindarajan, S. Antimicrobial study on trivalent lighter rare-earth complexes of 2-pyrazinecarboxylate with hydrazinium cation. World J. Microbiol. Biotech. 2006, 22, 1105–1108. [Google Scholar] [CrossRef]
- Selvaraj, S.; Krishnaswamy, S.; Devashya, V.; Sethuraman, S.; Krishnan, U.M. Flavonoid–metal ion complexes: A novel class of therapeutic agents. Med. Res. Rev. 2014, 34, 677–702. [Google Scholar] [CrossRef]
- Kopacz, M.; Woznicka, E.; Gruszecka, J. Antibacterial activity of morin and its complexes with La(III), Gd(III) and Lu(III) ions. Acta Pol. Pharm. 2005, 62, 65–67. [Google Scholar]
- Jayaprakash, S.; Ramesh, S.; Karthikeyan, A.; Murugappan, S.; Sidharthan, P.; Selvaraj, S. Transition Metal Coordination Complexes of Flavonoids: A Class of Better Pharmacological Active Molecules to Develop New Drugs. Anticancer Agents Med. Chem. 2023, 23, 417–431. [Google Scholar]
- Alghool, S.; Zoromba, M.S.; Abd El-Halim, H.F. Lanthanide amino acid Schiff base complexes: Synthesis, spectroscopic characterization, physical properties and in vitro antimicrobial studies. J. Rare Earths 2013, 31, 715–721. [Google Scholar] [CrossRef]
- Kaczmarek, M.T.; Zabiszak, M.; Nowak, M.; Jastrzab, R. Lanthanides: Schiff base complexes, applications in cancer diagnosis, therapy, and antibacterial activity. Coord. Chem. Rev. 2018, 370, 42–54. [Google Scholar] [CrossRef]
- Catalano, A.; Sinicropi, M.S.; Iacopetta, D.; Ceramella, J.; Mariconda, A.; Rosano, C.; Longo, P. A review on the advancements in the field of metal complexes with Schiff bases as antiproliferative agents. Appl. Sci. 2021, 11, 6027. [Google Scholar] [CrossRef]
- Alghool, S.; Abd El-Halim, H.F.; Abd El-sadek, M.S.; Yahia, I.S.; Wahab, L.A. Synthesis, thermal characterization, and antimicrobial activity of lanthanum, cerium, and thorium complexes of amino acid Schiff base ligand. J. Therm. Anal. Calorim. 2013, 112, 671–681. [Google Scholar] [CrossRef]
- Shiju, C.; Arish, D.; Kumaresan, S. Synthesis, characterization, cytotoxicity, DNAcleavage, and antimicrobial activity of lanthanide(III) complexes of a Schiff base ligand derived from glycylglycine and 4-nitrobenzaldehyde. Arab. J. Chem. 2017, 10, 2584–2591. [Google Scholar] [CrossRef]
- Taha, Z.A.; Hijazi, A.K.; Al Momani, W.M. Lanthanide complexes of the tridentate Schiff base ligand salicylaldehyde-2-picolinoylhydrazone: Synthesis, characterization, photophysical properties, biological activities and catalytic oxidation of aniline. J. Mol. Struct. 2020, 1220, 128712. [Google Scholar] [CrossRef]
- Taha, Z.A.; Ajlouni, A.M.; Momani, W.A.; Al-Ghzawi, A. Syntheses, characterization, biological activities and photophysical properties of lanthanides complexes with a tetradentate Schiff base ligand. Spectrochim. Acta Part A 2011, 81, 570–577. [Google Scholar] [CrossRef] [PubMed]
- Kostova, I.; Traykova, M.; Rastogi, V.K. New lanthanide complexes with antioxidant activity. Med. Chem. 2008, 4, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Shaghaghi, M.; Rashtbari, S.; Vejdani, S.; Dehghan, G.; Jouyban, A.; Yekta, R. Exploring the interactions of a Tb (III)–quercetin complex with serum albumins (HSA and BSA): Spectroscopic and molecular docking studies. Luminescence 2020, 35, 512–524. [Google Scholar] [CrossRef]
- Boz, H. p-Coumaric acid in cereals: Presence, antioxidant and antimicrobial effects. Inter. J. Food Sci. Technol. 2015, 50, 2323–2328. [Google Scholar] [CrossRef]
- Pei, K.; Ou, J.; Huang, J.; Ou, S. p-Coumaric acid and its conjugates: Dietary sources, pharmacokinetic properties and biological activities. J. Sci. Food Agric. 2016, 96, 2952–2962. [Google Scholar] [CrossRef] [PubMed]
- Laderiere, B.; Champagne, P.; Kalinowska, M.; Kowczyk, M.; Lewandowski, W. Mn(II), Cu(II), Zn(II), Cd(II) and Ca(II) p-coumarate: FT-IR, FT-Raman, 1H and 13C NMR, thermogravimetric and microbiological studies. In Proceedings of the Colloquim Spectroscopicum Internationale XXXVII, Buzios, Brazil, 28 August–2 September 2011. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E. Novel total antioxidant capacity index for dietary polyphenols and vitamins C and E, using their cupric ion reducing capability in the presence of neocuproine: CUPRAC method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef] [PubMed]
- Jabłońska-Trypuć, A.; Wydro, U.; Serra-Majem, L.; Wołejko, E.; Butarewicz, A. The Analysis of Bifenox and Dichlobenil Toxicity in Selected Microorganisms and Human Cancer Cells. Int. J. Environ. Res. Public Health 2019, 16, 4137. [Google Scholar] [CrossRef] [PubMed]
- Jabłońska-Trypuć, A.; Wydro, U.; Wołejko, E.; Świderski, G.; Lewandowski, W. Biological Activity of New Cichoric Acid–Metal Complexes in Bacterial Strains, Yeast-Like Fungi, and Human Cell Cultures In Vitro. Nutrients 2020, 12, 154. [Google Scholar] [CrossRef]
- Varsányi, G. Vibrational Spectra of Benzene Derivatives; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Nakamoto, K.; McCarthy, P.J. Spectroscopy and Structure of Metal Chelate Compounds; John Wiley and Sons: New York, NY, USA, 1968. [Google Scholar]
- Świsłocka, R.; Kowczyk-Sadowy, M.; Kalinowska, M.; Lewandowski, W. Spectroscopic (FT-IR, FT-Raman, 1H and 13C NMR) and theoretical studies of p-coumaric acid and alkali metal p-coumarates. Spectroscopy 2012, 27, 35–48. [Google Scholar] [CrossRef]
- Lewandowski, W.; Kalinowska, M.; Lewandowska, H. The influence of metals on the electronic system of biologically important ligands. Spectroscopic study of benzoates, salicylates, nicotinates and isoorotates. Review. J. Inorg. Biochem. 2005, 99, 1407–1423. [Google Scholar] [CrossRef] [PubMed]
- Santos-Sánchez, N.F.; Salas-Coronado, R.; Villanueva-Cañongo, C.; Hernández-Carlos, B. Antioxidant compounds and their antioxidant mechanism. Antioxidants 2019, 10, 1–29. [Google Scholar]
- Yuan, S.; Li, W.; Li, Q.; Wang, L.; Cao, J.; Jiang, W. Defense responses, induced by p-coumaric acid and methyl p-coumarate, of Jujube (Ziziphus jujuba Mill.) fruit against black spot rot caused by Alternaria alternata. J. Agric. Food Chem. 2019, 67, 2801. [Google Scholar] [CrossRef]
- Pohl, C.H. Recent advances and opportunities in the study of Candida albicans polymicrobial biofilms. Front. Cell. Infect. Microbiol. 2022, 12, 836379. [Google Scholar] [CrossRef]
- Chohan, Z.H.; Arif, M.; Akhtar, M.A.; Supuran, C.T. Metal-Based antibacterial and antifungal agents: Synthesis, characterization, and in vitro biological evaluation of Co(II), Cu(II), Ni(II), and Zn(II) complexes with amino acid-derived compoun. Bioinorg. Chem. Appl. 2006, 2006, 83131. [Google Scholar] [CrossRef] [PubMed]
- Carmo, P.H.F.; Garcia, M.T.; Figueiredo-Godoi, L.M.A.; Lage, A.C.P.; Silva, N.S.; Junqueira, J.C. Metal nanoparticles to combat Candida albicans infections: An Update. Microorganisms 2023, 11, 138. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).