Insights into the Modification of Carbonous Felt as an Electrode for Vanadium Redox Flow Batteries
Abstract
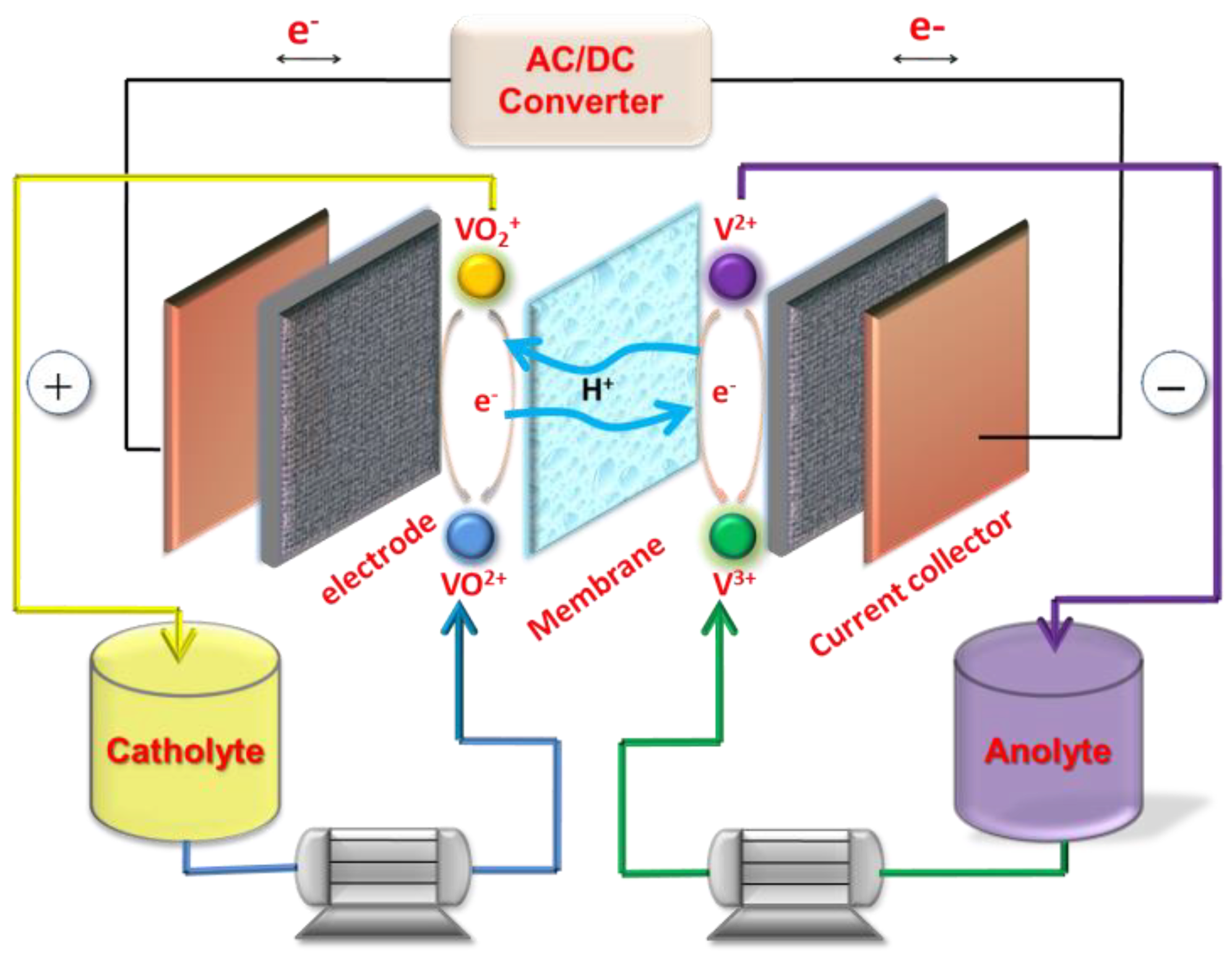
1. Introduction
2. Literature Review of Modification of Carbonous Felt
2.1. Surface Treatment or Structure Rebuilding
2.2. Metallic Modification
2.2.1. Single Metal Oxide Doping
2.2.2. Transition Metal Carbides, Nitrides, and Carbonitrides
2.3. Nonmetal Doping
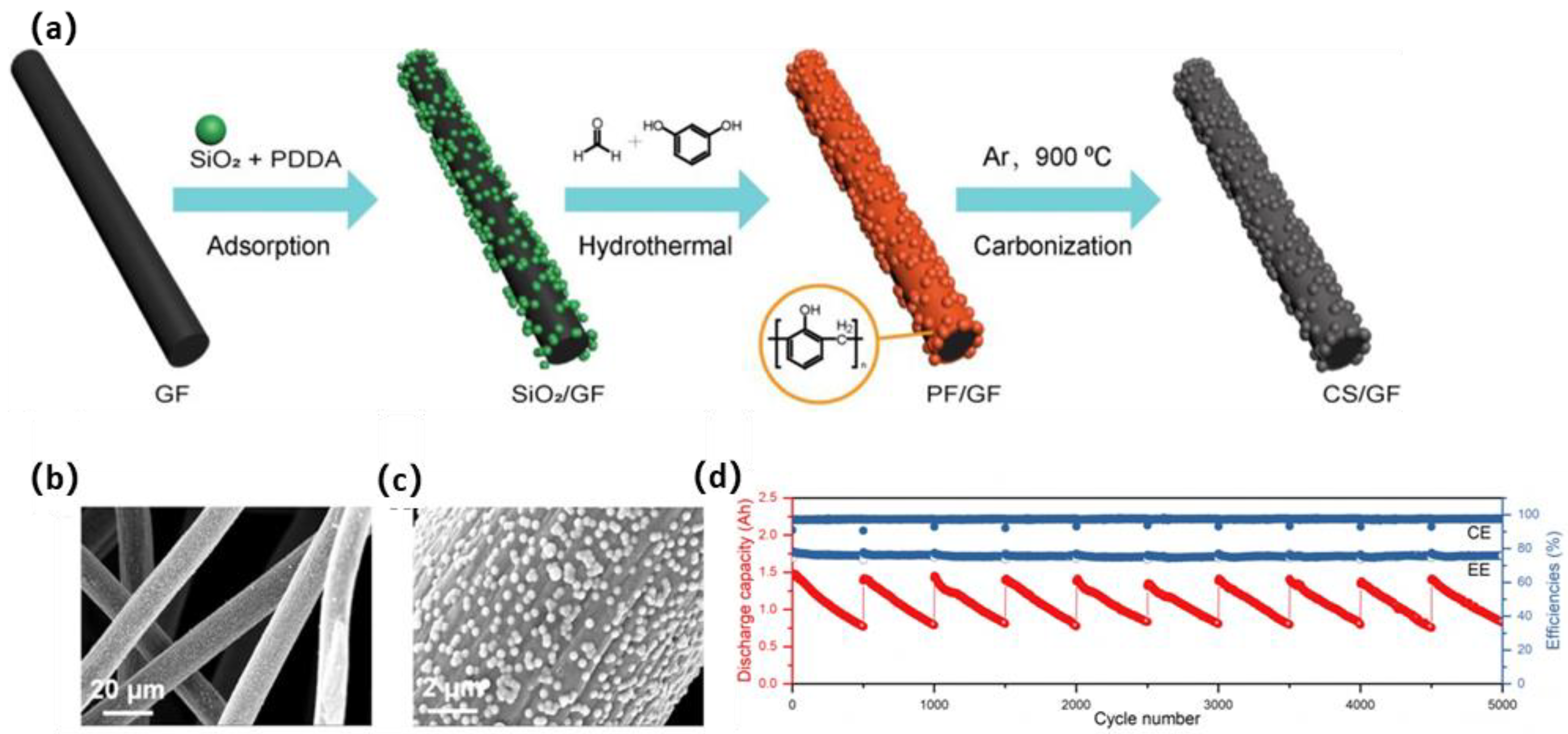
2.4. Carbonous Nanomaterial Modification
3. Conclusions and Perspectives
- Traditional surface treatments as well as novel intrinsic treatments all belong to the category of morphology-retaining methods, transforming the hydrophobic surfaces of bare felts into hydrophilic ones by the attaching various oxygen functional groups to the surfaces of the electrodes. The enhanced wettability makes it more feasible for the vanadium ion to migrate and obtain access to the active sites of the modified electrodes. Certain –OH or –COOH functional groups are widely regarded as offering active reaction sites for the vanadium redox reaction, enhancing the reactivity and wettability of the carbon electrodes. However, these new surface modification methods rely on the development of specific equipment or complex operations, restricting their large-scale application.
- Metallic or metal oxide nanoparticles attached to the carbon-based electrode via modification significantly improve the electrical conductivity and the electro-active surface, which ensures low polarization and enhanced reversibility in vanadium redox reactions. In particular, to address the issue of HER on the precious metal catalysts, various metal oxides have been presented to abate the overpotential in the negative half-cell. However, the functional and exact mechanisms of metal oxides or their superiority over others in most of the relevant studies remain ambiguous. The questions of how to ensure uniformity of the coating and reduce the costs of integrating oxygen functional groups on the surfaces of electrodes are worth intensive investigation. Furthermore, it must be noted that the deposited electrocatalyst can be exfoliated from the carbonous felt electrode under the flushing of the circulated acid electrolyte, degrading the electrode’s cycling stability and durability. Notably, novel nanosized metallic alloys with good corrosion resistance, a high surface area, and excellent electrical conductivity can be proposed as a possible candidate to generate additional catalytic activity.
- Heteroatom doping, including O, P, PO3, F, and SO3, is an effective approach that introduces functional groups into the matrix, regulating the surface electronic structure and generating catalytic sites and functional groups. With respect to heteroatom doping, diverse dopants such as P, B, and S, etc., have been also shown to display excellent electrochemical performance in VRFB applications. In particular, N groups are very attractive since they can synchronously improve the hydrophilicity and electrocatalytic activity, as well as the electrical conductivity. Significantly, dual- or even triple-doped carbon nanomaterials, such as B- or N-doped graphene and P-, S-, or N-doped carbon, easily controlling the electronic states of carbon materials, have been considered and will represent the predominant development direction in the future. Nonetheless, a more efficient and environmentally friendly heteroatom doping method is essential for industrial production.
- The surface area of the electrode could be enlarged by etching or decorating well-tailored carbon-based nanomaterials of different dimensions on the matrix of the electrode, which can provide a large surface area, rich surface functional groups, and superb electrical conductivity comparable to that of a metallic catalyst. Most surface moieties could serve as dominant active sites for redox reactions and promote the adsorption and desorption processes of vanadium ions, resulting in accelerated V2+/V3+ and VO2+/VO2+ reaction kinetics and high mass transport kinetics. Patterned microporous electrodes possessing a specific surface area and permeability would be promising. Researchers are expected to devote more efforts to developing novel functional carbon materials based on CNTs to promote the V2+/V3+ and VO2+/VO2+ redox reaction kinetics, although achieving their application without any performance deterioration will be a major challenge regarding long-term operation in practical VRFB systems.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Trovò, A.; Rugna, M.; Poli, N.; Guarnieri, M. Prospects for industrial vanadium flow batteries. Ceram. Int. 2023; accepted. [Google Scholar] [CrossRef]
- Dunn, B.; Kamath, H.; Tarascon, J.M. Electrical energy storage for the grid: A battery of choices. Science 2011, 334, 928–935. [Google Scholar] [CrossRef]
- Ding, C.; Zhang, H.M.; Li, X.F.; Liu, T.; Xing, F. Vanadium flow battery for energy storage: Prospects and challenges. J. Phys. Chem. Lett. 2013, 4, 1281–1294. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Huang, J.; Feng, J.-K. Research progress in preparation of electrolyte for all-vanadium redox flow battery. J. Ind. Eng. Chem. 2023, 118, 33–43. [Google Scholar] [CrossRef]
- Lv, Y.; Han, C.; Zhu, Y.; Zhang, T.; Yao, S.; He, Z.; Dai, L.; Wang, L. Recent advances in metals and metal oxides as catalysts for vanadium redox flow battery: Properties, structures, and perspectives. J. Mater. Sci. Technol. 2021, 75, 96–109. [Google Scholar] [CrossRef]
- Wang, Z.; Ren, J.; Sun, J.; Guo, Z.; Wei, L.; Fan, X.; Zhao, T. Characterizations and selections of electrodes with optimal performance for large-scale vanadium redox flow batteries through lab-scale experiments. J. Power Sources 2022, 549, 232094. [Google Scholar] [CrossRef]
- Xu, Z.; Jing, M.; Liu, J.; Yan, C.; Fan, X. Advanced dual-gradient carbon nanofibers/graphite felt composite electrode for the next-generation vanadium flow battery. J. Mater. Sci. Technol. 2023, 136, 32–42. [Google Scholar] [CrossRef]
- Cheng, D.; Zhu, W.; Gao, J.; Li, J.; Yang, Y.; Dai, L.; Liu, Y.; Wang, L.; He, Z. High-graphitization, large-surface area, and porous carbon nanofiber: A superior bi-functional electrode for vanadium redox flow battery. Appl. Surf. Sci. 2022, 599, 153919. [Google Scholar] [CrossRef]
- Park, M.; Ryu, J.; Cho, J. Nanostructured electrocatalysts for all-vanadium redox flow batteries. Chem.-Asian J. 2015, 10, 2096–2110. [Google Scholar] [CrossRef]
- Liu, T.; Li, X.; Zhang, H.; Chen, J. Progress on the electrode materials towards vanadium flow batteries (VFBs) with improved power density. J. Energy Chem. 2018, 27, 1292–1303. [Google Scholar] [CrossRef]
- Ulaganathan, M.; Aravindan, V.; Yan, Q.; Madhavi, S.; Skyllas-Kazacos, M.; Lim, T.M. Recent advancements in all-vanadium redox flow batteries. Adv. Mater. Interfaces 2016, 3, 1500309. [Google Scholar] [CrossRef]
- Sun, B.; Skyllas-Kazacos, M. Modification of graphite electrode materials for vanadium redox flow battery application—I. Thermal treatment. Electrochim. Acta 1992, 37, 1253–1260. [Google Scholar] [CrossRef]
- Sun, B.; Skyllas-Kazacos, M. Chemical modification of graphite electrode materials for vanadium redox flow battery application—Part II. Acid treatments. Electrochim. Acta 1992, 37, 2459–2465. [Google Scholar] [CrossRef]
- Cho, Y.I.; Park, S.J.; Hwang, H.J.; Lee, J.G.; Jeon, Y.K.; Chu, Y.H.; Shul, Y.G. Effects of microwave treatment on carbon electrode for vanadium redox flow battery. ChemElectroChem 2015, 2, 872–876. [Google Scholar] [CrossRef]
- He, Z.X.; Jiang, Y.Q.; Zhou, H.Z.; Cheng, G.; Meng, W.; Wang, L.; Dai, L. Graphite felt electrode modified by square wave potential pulse for vanadium redox flow battery. Int. J. Energy Res. 2017, 41, 439–447. [Google Scholar] [CrossRef]
- Estevez, L.; Reed, D.; Nie, Z.; Schwarz, A.M.; Nandasiri, M.I.; Kizewski, J.P.; Wang, W.; Thomsen, E.; Liu, J.; Zhang, J.G.; et al. Tunable oxygen functional groups as electrocatalysts on graphite felt surfaces for all-vanadium flow batteries. ChemSusChem 2016, 9, 1455–1461. [Google Scholar] [CrossRef]
- He, Z.X.; Jiang, Y.Q.; Meng, W.; Jiang, F.Y.; Zhou, H.Z.; Li, Y.H.; Zhu, J.; Wang, L.; Dai, L. HF/H2O2 treated graphite felt as the positive electrode for vanadium redox flow battery. Appl. Surf. Sci. 2017, 423, 111–118. [Google Scholar] [CrossRef]
- Kim, K.J.; Kim, Y.J.; Kim, J.H.; Park, M.S. The effects of surface modification on carbon felt electrodes for use in vanadium redox flow batteries. Mater. Chem. Phys. 2011, 131, 547–553. [Google Scholar] [CrossRef]
- Park, S.M.; Kim, H. Porous electrodes with lower impedance for vanadium redox flow batteries. Korean Chem. Eng. Res. 2015, 53, 638–645. [Google Scholar] [CrossRef]
- Chang, Y.C.; Chen, J.Y.; Kabtamu, D.M.; Lin, G.Y.; Hsu, N.Y.; Chou, Y.S.; Wei, H.J.; Wang, C.H. High efficiency of CO2 -activated graphite felt as electrode for vanadium redox flow battery application. J. Power Sources 2017, 364, 1–8. [Google Scholar] [CrossRef]
- Kabtamu, D.M.; Chen, J.Y.; Chang, Y.C.; Wang, C.H. Water-activated graphite felt as a high-performance electrode for vanadium redox flow batteries. J. Power Sources 2017, 341, 270–279. [Google Scholar] [CrossRef]
- Park, J.J.; Park, J.H.; Park, O.O.; Yang, J.H. Highly porous graphenated graphite felt electrodes with catalytic defects for high-performance vanadium redox flow batteries produced via NiO/Ni redox reactions. Carbon 2016, 110, 17–26. [Google Scholar] [CrossRef]
- Liu, Y.C.; Shen, Y.; Yu, L.H.; Liu, L.; Liang, F.; Qiu, X.P.; Xi, J.Y. Holey-engineered electrodes for advanced vanadium flow batteries. Nano Energy 2018, 43, 55–62. [Google Scholar] [CrossRef]
- Jiang, H.R.; Shyy, W.; Wu, M.C.; Zhang, R.H.; Zhao, T.S. A bi-porous graphite felt electrode with enhanced surface area and catalytic activity for vanadium redox flow batteries. Appl. Energy 2019, 233–234, 105–113. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, H.; Chu, Y.; Tong, S. Roughened graphite felt electrode with enhanced electrochemical activity for vanadium redox flow batteries. Int. J. Electrochem. Sci. 2019, 14, 5408–5421. [Google Scholar]
- Ghimire, P.C.; Schweiss, R.; Scherer, G.G.; Lim, T.M.; Wai, N.; Bhattarai, A.; Yan, Q.Y. Optimization of thermal oxidation of electrodes for the performance enhancement in all-vanadium redox flow betteries. Carbon 2019, 155, 176–185. [Google Scholar] [CrossRef]
- Gautam, R.K.; Kapoor, M.; Verma, A. Tactical surface modification of a 3D graphite felt as an electrode of vanadium redox flow batteries with enhanced electrolyte utilization and fast reaction kinetics. Energy Fuels 2020, 34, 5060–5071. [Google Scholar] [CrossRef]
- Zhang, Z.Y.; Xi, J.Y.; Zhou, H.P.; Qiu, X.P. KOH etched graphite felt with improved wettability and activity for vanadium flow batteries. Electrochim. Acta 2016, 218, 15–23. [Google Scholar] [CrossRef]
- Hassan, A.; Tzedakis, T. Enhancement of the electrochemical activity of a commercial graphite felt for vanadium redox flow battery (VRFB), by chemical treatment with acidic solution of K2Cr2O7. J. Energy Storage 2019, 26, 100967. [Google Scholar] [CrossRef]
- Jiang, H.R.; Shyy, W.; Ren, Y.X.; Zhang, R.H.; Zhao, T.S. A room-temperature activated graphite felt as the cost-effective, highly active and stable electrode for vanadium redox flow batteries. Appl. Energy 2019, 233–234, 544–553. [Google Scholar] [CrossRef]
- Abbas, S.; Lee, H.; Hwang, J.; Mehmood, A.; Shin, H.; Mehboob, S.; Lee, J.; Ha, H.Y. A novel approach for forming carbon nanorods on the surface of carbon felt electrode by catalytic etching for high-performance vanadium redox flow battery. Carbon 2018, 128, 31–37. [Google Scholar] [CrossRef]
- Zhou, X.L.; Zhang, X.Y.; Lv, Y.H.; Lin, L.Y.; Wu, Q.X. Nano-catalytic layer engraved carbon felt via copper oxide etching for vanadium redox flow batteries. Carbon 2019, 153, 674–681. [Google Scholar] [CrossRef]
- Zhang, L.; Yue, J.P.; Deng, Q.; Ling, W.; Zhou, C.J.; Zeng, X.X.; Zhou, C.S.; Wu, X.W.; Wu, Y.P. Preparation of a porous graphite felt electrode for advance vanadium redox flow batteries. RSC Adv. 2020, 10, 13374–13378. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Li, Y.S.; He, Y.L. Achieving gradient-pore-oriented graphite felt for vanadium redox flow batteries: Meeting improved electrochemical activity and enhanced mass transport from nano- to micro-scale. J. Mater. Chem. A 2019, 7, 10962–10970. [Google Scholar] [CrossRef]
- Wu, X.; Xie, Z.; Zhou, H.; Xiong, Z.A.; Yin, X.; Tang, H.; Ma, Q.; Liao, J. Designing high efficiency graphite felt electrode via HNO3 vapor activation towards stable vanadium redox flow battery. Electrochim. Acta 2023, 440, 141728. [Google Scholar] [CrossRef]
- Di Blasi, A.; Di Blasi, O.; Briguglio, N.; Aricò, A.S.; Sebastián, D.; Lázaro, M.J.; Monforte, G.; Antonucci, V. Investigation of several graphite-based electrodes for vanadium redox flow cell. J. Power Sources 2013, 227, 15–23. [Google Scholar] [CrossRef]
- Rudolph, S.; Schröder, U.; Bayanov, I.M.; Hage, D. Measurement, simulation and in situ regeneration of energy efficiency in vanadium redox flow battery. J. Electroanal. Chem. 2014, 728, 72–80. [Google Scholar] [CrossRef]
- Rychcik, M.; Skyllas-Kazacos, M. Evaluation of electrode materials for vanadium redox cell. J. Power Sources 1987, 19, 45–54. [Google Scholar] [CrossRef]
- Wei, L.; Zhao, T.S.; Zeng, L.; Zhou, X.L.; Zeng, Y.K. Copper nanoparticle-deposited graphite felt electrodes for all vanadium redox flow batteries. Appl. Energy 2016, 180, 386–391. [Google Scholar] [CrossRef]
- Wen, Y.; Neville, T.P.; Jorge Sobrido, A.; Shearing, P.R.; Brett, D.J.L.; Jervis, R. Bismuth concentration influenced competition between electrochemical reactions in the all-vanadium redox flow battery. J. Power Sources 2023, 566, 232861. [Google Scholar] [CrossRef]
- González, Z.; Sánchez, A.; Blanco, C.; Granda, M.; Menéndez, R.; Santamaría, R. Enhanced performance of a Bi-modified graphite felt as the positive electrode of a vanadium redox flow battery. Electrochem. Commun. 2011, 13, 1379–1382. [Google Scholar] [CrossRef]
- Li, B.; Gu, M.; Nie, Z.M.; Shao, Y.Y.; Luo, Q.T.; Wei, X.L.; Li, X.L.; Xiao, J.; Wang, C.M.; Sprenkle, V.; et al. Bismuth nanoparticle decorating graphite felt as a high-performance electrode for an all-vanadium redox flow battery. Nano Lett. 2013, 13, 1330–1335. [Google Scholar] [CrossRef] [PubMed]
- Suárez, D.J.; González, Z.; Blanco, C.; Granda, M.; Menéndez, R.; Santamaría, R. Graphite felt modified with bismuth nanoparticles as negative electrode in a vanadium redox flow battery. ChemSusChem 2014, 7, 914–918. [Google Scholar] [CrossRef] [PubMed]
- Fetyan, A.; El-Nagar, G.A.; Derr, I.; Kubella, P.; Dau, H.; Roth, C. A neodymium oxide nanoparticle-doped carbon felt as promising electrode for vanadium redox flow batteries. Electrochim. Acta 2018, 268, 59–65. [Google Scholar] [CrossRef]
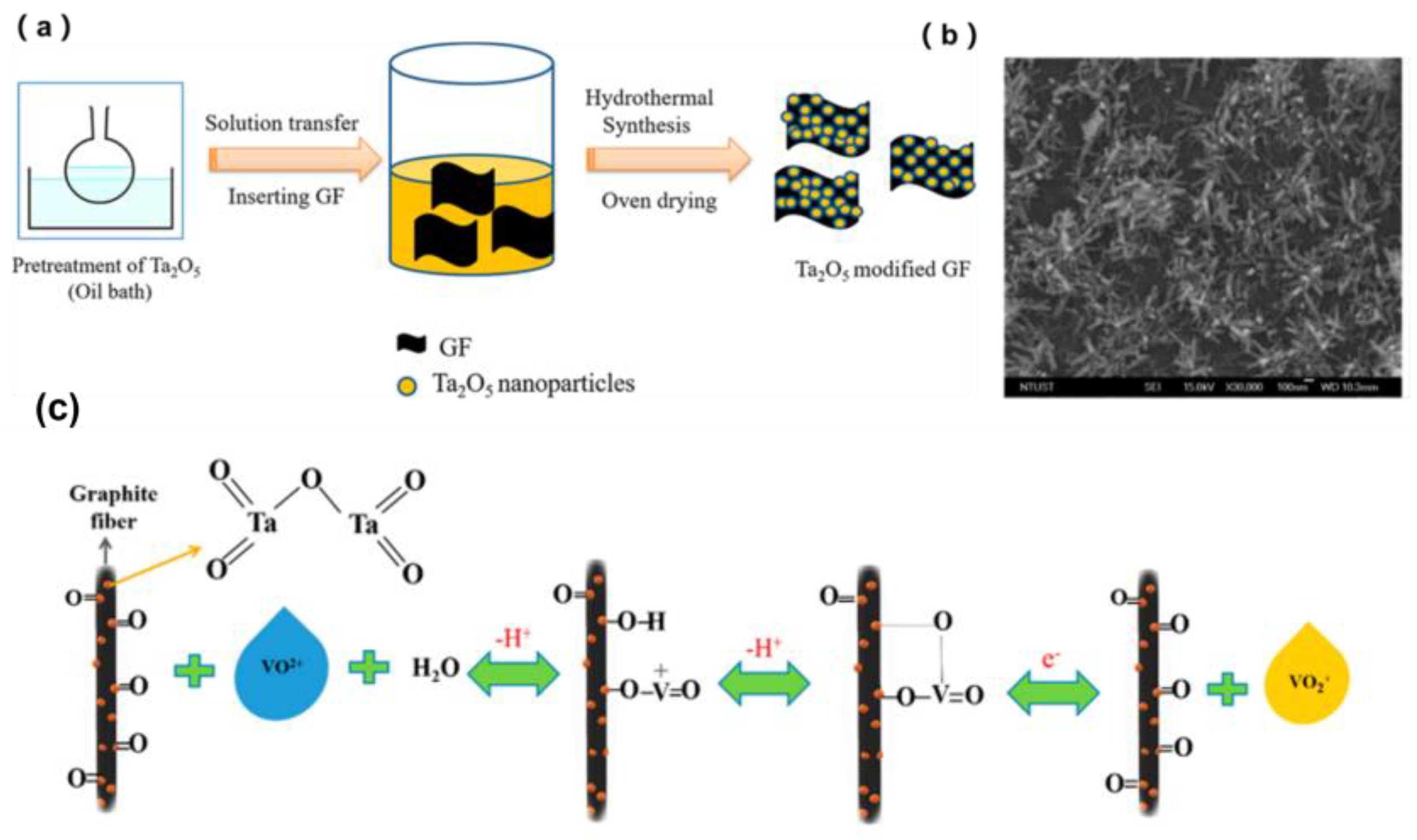
- Bayeh, A.W.; Kabtamu, D.M.; Chang, Y.C.; Chen, G.C.; Chen, H.Y.; Lin, G.Y.; Liu, T.R.; Wondimu, T.H.; Wang, K.C.; Wang, C.H. Ta2O5-nanoparticle-modified graphite felt as a high-performance electrode for a vanadium redox flow battery. ACS Sustain. Chem. Eng. 2018, 6, 3019–3028. [Google Scholar] [CrossRef]
- Mehboob, S.; Ali, G.; Shin, H.J.; Hwang, J.; Abbas, S.; Chung, K.Y.; Ha, H.Y. Enhancing the performance of all-vanadium redox flow batteries by decorating carbon felt electrodes with SnO2 nanoparticles. Appl. Energy 2018, 229, 910–921. [Google Scholar] [CrossRef]
- Kim, K.J.; Park, M.S.; Kim, Y.J.; Kim, J.H.; Dou, S.X.; Skyllas-Kazacos, M. A technology review of electrodes and reaction mechanisms in vanadium redox flow batteries. J. Mater. Chem. A 2015, 3, 16913–16933. [Google Scholar] [CrossRef]
- Shen, Y.; Xu, H.F.; Xu, P.C.; Wu, X.X.; Dong, Y.M.; Lu, L. Electrochemical catalytic activity of tungsten trioxide- modified graphite felt toward VO2+/VO2+ redox reaction. Electrochim. Acta 2014, 132, 37–41. [Google Scholar] [CrossRef]
- Thu Pham, H.T.; Jo, C.; Lee, J.; Kwon, Y. MoO2 nanocrystals interconnected on mesocellular carbon foam as a powerful catalyst for vanadium redox flow battery. RSC Adv. 2016, 6, 17574–17582. [Google Scholar] [CrossRef]
- Zhou, H.P.; Xi, J.Y.; Li, Z.H.; Zhang, Z.Y.; Yu, L.H.; Liu, L.; Qiu, X.P.; Chen, L.Q. CeO2decorated graphite felt as a high-performance electrode for vanadium redox flow batteries. RSC Adv. 2014, 4, 61912–61918. [Google Scholar] [CrossRef]
- Zhou, H.; Shen, Y.; Xi, J.; Qiu, X.; Chen, L. ZrO2-nanoparticle-modified graphite felt: Bifunctional effects on vanadium flow batteries. ACS Appl. Mater. Interfaces 2016, 8, 15369–15378. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.X.; Xu, H.F.; Lu, L.; Zhao, H.; Fu, J.; Shen, Y.; Xu, P.C.; Dong, Y.M. PbO2-modified graphite felt as the positive electrode for an all-vanadium redox flow battery. J. Power Sources 2014, 250, 274–278. [Google Scholar] [CrossRef]
- Ma, Q.; Deng, Q.; Sheng, H.; Ling, W.; Wang, H.R.; Jiao, H.W.; Wu, X.W.; Zhou, W.X.; Zeng, X.X.; Yin, Y.X.; et al. High electro-catalytic graphite felt/MnO2 composite electrodes for vanadium redox flow batteries. Sci. China Chem. 2018, 61, 732–738. [Google Scholar] [CrossRef]
- Kim, K.J.; Park, M.S.; Kim, J.H.; Hwang, U.; Lee, N.J.; Jeong, G.; Kim, Y.J. Novel catalytic effects of Mn3O4 for all vanadium redox flow batteries. Chem. Commun. 2012, 48, 5455–5457. [Google Scholar] [CrossRef]
- He, Z.X.; Dai, L.; Liu, S.Q.; Wang, L.; Li, C.C. Mn3O4 anchored on carbon nanotubes as an electrode reaction catalyst of V(IV)/V(V) couple for vanadium redox flow batteries. Electrochim. Acta 2015, 176, 1434–1440. [Google Scholar] [CrossRef]
- Xiang, Y.; Daoud, W.A. Cr2O3-modified graphite felt as a novel positive electrode for vanadium redox flow battery. Electrochim. Acta 2018, 290, 176–184. [Google Scholar] [CrossRef]
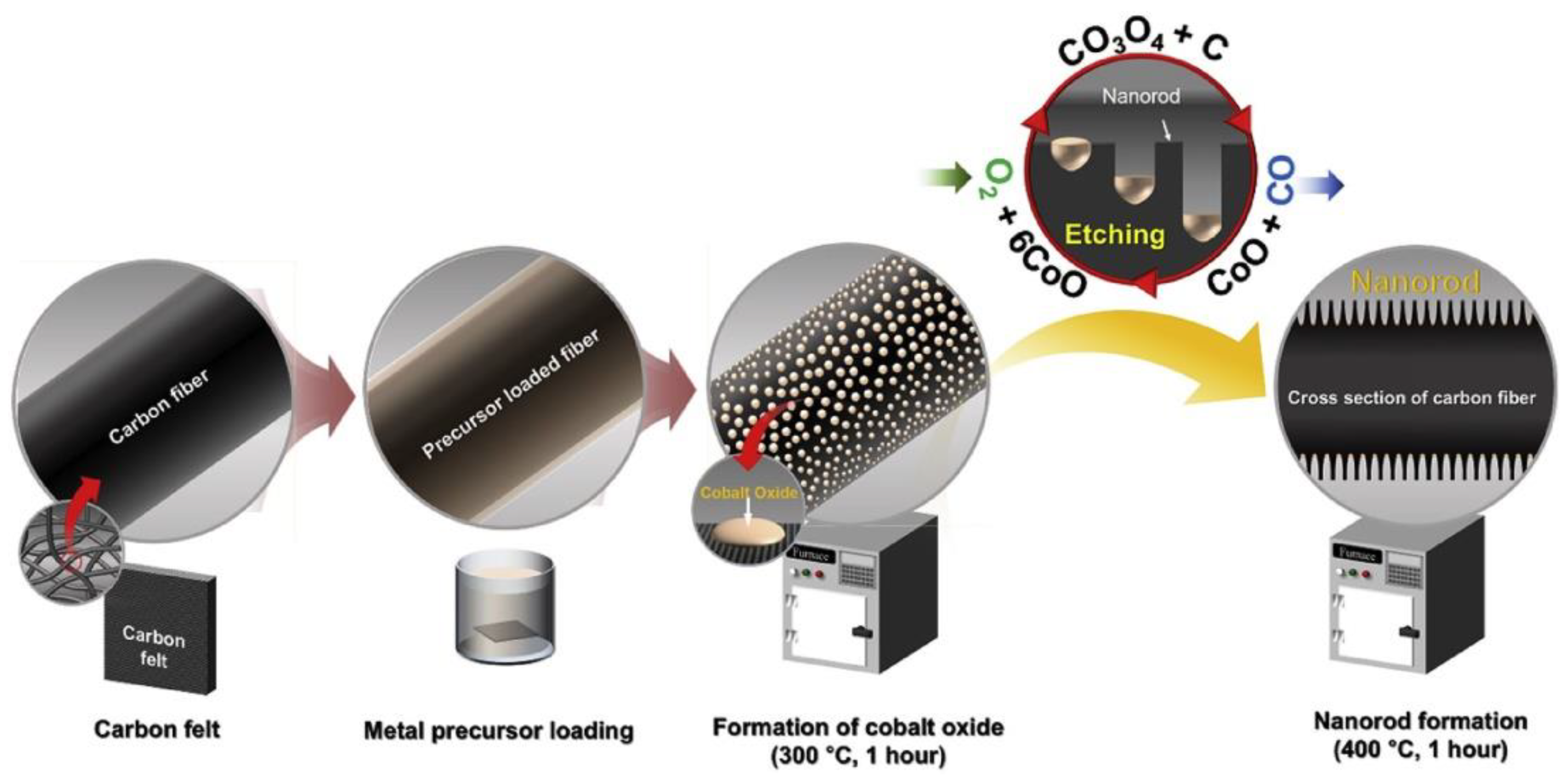
- Xiang, Y.; Daoud, W.A. Investigation of an advanced catalytic effect of cobalt oxide modification on graphite felt as the positive electrode of the vanadium redox flow battery. J. Power Sources 2019, 416, 175–183. [Google Scholar] [CrossRef]
- Yun, N.; Park, J.J.; Park, O.O.; Lee, K.B.; Yang, J.H. Electrocatalytic effect of NiO nanoparticles evenly distributed on a graphite felt electrode for vanadium redox flow batteries. Electrochim. Acta 2018, 278, 226–235. [Google Scholar] [CrossRef]
- Li, B.; Gu, M.; Nie, Z.; Wei, X.; Wang, C.; Sprenkle, V.; Wang, W. Nanorod niobium oxide as powerful catalysts for an all vanadium redox flow battery. Nano Lett. 2014, 14, 158–165. [Google Scholar] [CrossRef]
- Kazacos, M.S. A Rotating Ring-Disk Electrode Study of Soluble Lead (IV) Species in Sulfuric Acid Solution. J. Electrochem. Soc. 1981, 128, 817–820. [Google Scholar] [CrossRef]
- Irwin, M.D.; Buchholz, D.B.; Hains, A.W.; Chang, R.P.H.; Marks, T.J. P-type semiconducting nickel oxide as an efficiency-enhancing anode interfacial layer in polymer bulk-heterojunction solar cells. Proc. Natl. Acad. Sci. USA 2008, 105, 2783–2787. [Google Scholar] [CrossRef]
- Ismail, A.A.; Faisal, M.; Harraz, F.A.; Al-Hajry, A.; Al-Sehemi, A.G. Synthesis of mesoporous sulfur-doped Ta2O5 nanocomposites and their photocatalytic activities. J. Colloid Interface Sci. 2016, 471, 145–154. [Google Scholar] [CrossRef] [PubMed]
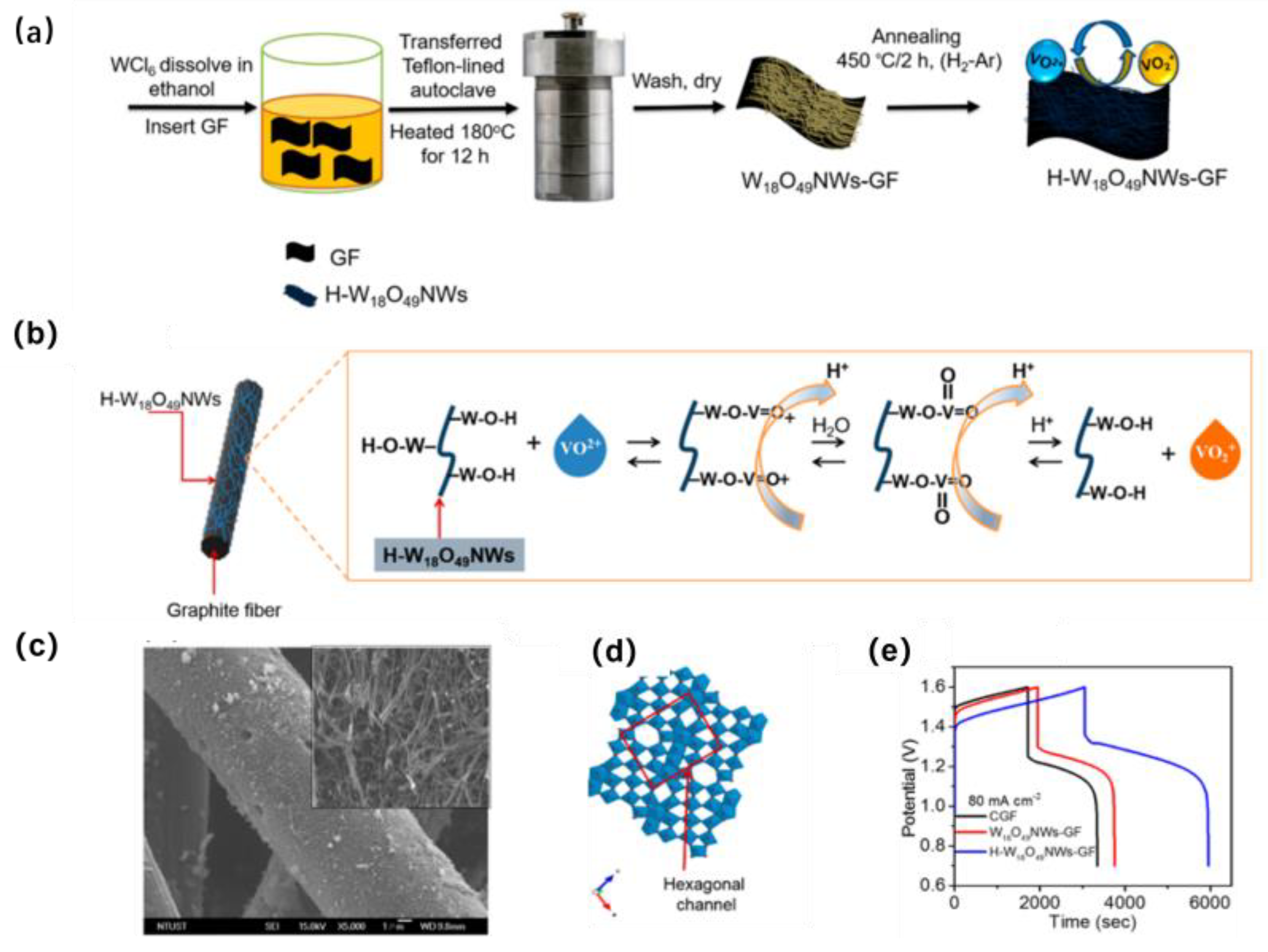
- Bayeh, A.W.; Kabtamu, D.M.; Chang, Y.C.; Chen, G.C.; Chen, H.Y.; Liu, T.R.; Wondimu, T.H.; Wang, K.C.; Wang, C.H. Hydrogen-treated defect-rich W18O9 nanowire-modified graphite felt as high-performance electrode for vanadium redox flow battery. ACS Appl. Energy Mater. 2019, 2, 2541–2551. [Google Scholar] [CrossRef]
- Vazquez-Galvan, J.; Flox, C.; Fabrega, C.; Ventosa, E.; Parra, A.; Andreu, T.; Morante, J.R. Hydrogen-treated rutile tio2 shell in graphite-core structure as a negative electrode for high-performance vanadium redox flow batteries. ChemSusChem 2017, 10, 2089–2098. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Galván, J.; Flox, C.; Jervis, J.R.; Jorge, A.B.; Shearing, P.R.; Morante, J.R. High-power nitrided TiO2 carbon felt as the negative electrode for all-vanadium redox flow batteries. Carbon 2019, 148, 91–104. [Google Scholar] [CrossRef]
- Wei, L.; Zhao, T.S.; Zeng, L.; Zeng, Y.K.; Jiang, H.R. Highly catalytic and stabilized titanium nitride nanowire array-decorated graphite felt electrodes for all vanadium redox flow batteries. J. Power Sources 2017, 341, 318–326. [Google Scholar] [CrossRef]
- Ghimire, P.C.; Schweiss, R.; Scherer, G.G.; Wai, N.; Lim, T.M.; Bhattarai, A.; Nguyen, T.D.; Yan, Q.Y. Titanium carbide-decorated graphite felt as high performance negative electrode in vanadium redox flow batteries. J. Mater. Chem. A 2018, 6, 6625–6632. [Google Scholar] [CrossRef]
- Oyama, S.T. Preparation and catalytic properties of transition metal carbides and nitrides. Catal. Today 1992, 15, 179–200. [Google Scholar] [CrossRef]
- Hasnat Hossain, M.; Abdullah, N.; Rahman, S.; Amran Mohd Radzi, M. MXene-based electrolyte for vanadium-bromide redox flow battery for green energy. Mater. Today Proc. 2023, accepted. [Google Scholar] [CrossRef]
- Wei, L.; Xiong, C.; Jiang, H.R.; Fan, X.Z.; Zhao, T.S. Highly catalytic hollow Ti3C2Tx MXene spheres decorated graphite felt electrode for vanadium redox flow batteries. Energy Storage Mater. 2020, 25, 885–892. [Google Scholar] [CrossRef]
- Kabtamu, D.M.; Chen, J.Y.; Chang, Y.C.; Wang, C.H. Electrocatalytic activity of Nb-doped hexagonal WO3 nanowire-modified graphite felt as a positive electrode for vanadium redox flow batteries. J. Mater. Chem. A 2016, 4, 11472–11480. [Google Scholar] [CrossRef]
- Kabtamu, D.M.; Bayeh, A.W.; Chiang, T.C.; Chang, Y.C.; Lin, G.Y.; Wondimu, T.H.; Su, S.K.; Wang, C.H. TiNb2O7 nanoparticle-decorated graphite felt as a high-performance electrode for vanadium redox flow batteries. Appl. Surf. Sci. 2018, 462, 73–80. [Google Scholar] [CrossRef]
- Xiang, Y.; Daoud, W.A. Binary NiCoO2-modified graphite felt as an advanced positive electrode for vanadium redox flow batteries. J. Mater. Chem. A 2019, 7, 5589–5600. [Google Scholar] [CrossRef]
- You, D.; Lou, J.; Li, X.; Zhou, Y.; Sun, X.; Wang, X. Investigation of advanced catalytic effect of Co3O4 nanosheets modified carbon felts as vanadium flow battery electrodes. J. Power Sources 2021, 494, 229775. [Google Scholar] [CrossRef]
- Chen, T.W.; Anushya, G.; Chen, S.M.; Kalimuthu, P.; Mariyappan, V.; Gajendran, P.; Ramachandran, R. Recent advances in nanoscale based electrocatalysts for metal-air battery, fuel cell and water-splitting applications: An overview. Mater. 2022, 15, 458. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.Y.; Wang, X.Q.; Engelhard, M.; Wang, C.M.; Dai, S.; Liu, J.; Yang, Z.G.; Lin, Y.H. Nitrogen-doped mesoporous carbon for energy storage in vanadium redox flow batteries. J. Power Sources 2010, 195, 4375–4379. [Google Scholar] [CrossRef]
- Park, S.; Kim, H. Fabrication of nitrogen-doped graphite felts as positive electrodes using polypyrrole as a coating agent in vanadium redox flow batteries. J. Mater. Chem. A 2015, 3, 12276–12283. [Google Scholar] [CrossRef]
- Lee, H.J.; Kim, H. Graphite felt coated with dopamine-derived nitrogen-doped carbon as a positive electrode for a vanadium redox flow battery. J. Electrochem. Soc. 2015, 162, A1675–A1681. [Google Scholar] [CrossRef]
- Youn, C.; Song, S.A.; Kim, K.; Woo, J.Y.; Chang, Y.W.; Lim, S.N. Effect of nitrogen functionalization of graphite felt electrode by ultrasonication on the electrochemical performance of vanadium redox flow battery. Mater. Chem. Phys. 2019, 237, 121873. [Google Scholar] [CrossRef]
- Chen, J.Z.; Liao, W.Y.; Hsieh, W.Y.; Hsu, C.C.; Chen, Y.S. All-vanadium redox flow batteries with graphite felt electrodes treated by atmospheric pressure plasma jets. J. Power Sources 2015, 274, 894–898. [Google Scholar] [CrossRef]
- Dixon, D.; Babu, D.J.; Bhaskar, A.; Bruns, H.M.; Schneider, J.J.; Scheiba, F.; Ehrenberg, H. Tuning the performance of vanadium redox flow batteries by modifying the structural defects of the carbon felt electrode. Beilstein J. Nanotechnol. 2019, 10, 1698–1706. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Chung, D.J.; Park, H.I.; Ansari, M.Z.; Song, T.; Kim, H. Direct nitradated graphite felt as an electrode material for the vanadium redox flow battery. Bull. Korean Chem. Soc. 2018, 39, 281–286. [Google Scholar] [CrossRef]
- He, Z.Q.; Zhou, X.J.; Zhang, Y.; Jiang, F.J.; Yu, Q.C. Low-temperature nitrogen-doping of graphite felt electrode for vanadium redox flow batteries. J. Electrochem. Soc. 2019, 166, A2336–A2340. [Google Scholar] [CrossRef]
- He, Z.X.; Shi, L.; Shen, J.X.; He, Z.; Liu, S.Q. Effects of nitrogen doping on the electrochemical performance of graphite felts for vanadium redox flow batteries. Int. J. Energy Res. 2015, 39, 709–716. [Google Scholar] [CrossRef]
- Yoon, S.J.; Kim, S.; Kim, D.K.; So, S.; Hong, Y.T.; Hempelmann, R. Ionic liquid derived nitrogen-doped graphite felt electrodes for vanadium redox flow batteries. Carbon 2020, 166, 131–137. [Google Scholar] [CrossRef]
- Yoon, S.J.; Kim, S.; Kim, D.K.; Yu, D.M.; Hempelmann, R.; Hong, Y.T.; So, S. Nitrogen-doping through two-step pyrolysis of polyacrylonitrile on graphite felts for vanadium redox flow batteries. Energy Fuels 2020, 34, 5052–5059. [Google Scholar] [CrossRef]
- Yu, L.H.; Lin, F.; Xu, L.; Xi, J.Y. P-doped electrode for vanadium flow battery with high-rate capability and all-climate adaptability. J. Energy Chem. 2019, 35, 55–59. [Google Scholar] [CrossRef]
- Ryu, J.; Park, M.; Cho, J. Catalytic effects of b/n-co-doped porous carbon incorporated with ketjenblack nanoparticles for all-vanadium redox flow batteries. J. Electrochem. Soc. 2015, 163, A5144–A5149. [Google Scholar] [CrossRef]
- Shi, L.; Liu, S.Q.; He, Z.; Yuan, H.; Shen, J.X. Synthesis of boron and nitrogen co-doped carbon nanofiber as efficient metal-free electrocatalyst for the VO2+/VO2+ Redox Reaction. Electrochim. Acta 2015, 178, 748–757. [Google Scholar] [CrossRef]
- Jiang, H.R.; Shyy, W.; Zeng, L.; Zhang, R.H.; Zhao, T.S. Highly efficient and ultra-stable boron-doped graphite felt electrodes for vanadium redox flow batteries. J. Mater. Chem. A 2018, 6, 13244–13253. [Google Scholar] [CrossRef]
- Chung, Y.J.; Noh, C.; Kwon, Y. Role of borate functionalized carbon nanotube catalyst for the performance improvement of vanadium redox flow battery. J. Power Sources 2019, 438, 227063. [Google Scholar] [CrossRef]
- Ma, Q.; Zeng, X.X.; Zhou, C.J.; Deng, Q.; Wang, P.F.; Zuo, T.T.; Zhang, X.D.; Yin, Y.X.; Wu, X.W.; Chai, L.Y.; et al. Designing high-performance composite electrodes for vanadium redox flow batteries: Experimental and computational investigation. ACS Appl. Mater. Interfaces 2018, 10, 22381–22388. [Google Scholar] [CrossRef] [PubMed]
- Sheng, H.; Ma, Q.; Yu, J.G.; Zhang, X.D.; Zhang, W.; Yin, Y.X.; Wu, X.W.; Zeng, X.X.; Guo, Y.G. Robust electrodes with maximized spatial catalysis for vanadium redox flow batteries. ACS Appl. Mater. Interfaces 2018, 10, 38922–38927. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ma, Q.; Hu, J.P.; Liu, J.; Deng, Q.; Ning, P.; Zhou, C.S.; Wu, X.W.; Wu, Y.P. Enhancing the catalytic kinetics of electrodes by using a multidimensional carbon network for applications in vanadium redox flow batteries. ChemElectroChem 2020, 7, 1023–1028. [Google Scholar] [CrossRef]
- Yang, D.S.; Han, J.H.; Jeon, J.W.; Lee, J.Y.; Kim, D.G.; Seo, D.H.; Kim, B.G.; Kim, T.H.; Hong, Y.T. Multimodal porous and nitrogen-functionalized electrode based on graphite felt modified with carbonized porous polymer skin layer for all-vanadium redox flow battery. Mater. Today Energy 2019, 11, 159–165. [Google Scholar] [CrossRef]
- Su, J.C.; Zhao, Y.; Xi, J.Y. Phosphorus-doped carbon nitride as powerful electrocatalyst for high-power vanadium flow battery. Electrochim. Acta 2018, 286, 22–28. [Google Scholar] [CrossRef]
- Kim, J.; Lim, H.; Jyoung, J.Y.; Lee, E.S.; Yi, J.S.; Lee, D. High electrocatalytic performance of N and O atomic co-functionalized carbon electrodes for vanadium redox flow battery. Carbon 2017, 111, 592–601. [Google Scholar] [CrossRef]
- Kim, J.; Lim, H.; Jyoung, J.Y.; Lee, E.S.; Yi, J.S.; Lee, D. Effects of doping methods and kinetic relevance of N and O atomic co-functionalization on carbon electrode for V(IV)/V(V) redox reactions in vanadium redox flow battery. Electrochim. Acta 2017, 245, 724–733. [Google Scholar] [CrossRef]
- Lee, M.E.; Jin, H.J.; Yun, Y.S. Synergistic catalytic effects of oxygen and nitrogen functional groups on active carbon electrodes for all-vanadium redox flow batteries. RSC Adv. 2017, 7, 43227–43232. [Google Scholar] [CrossRef]
- Lee, H.J.; Kil, D.; Kim, H. Synthesis of activated graphite felt using consecutive post-treatments for vanadium redox flow batteries. J. Electrochem. Soc. 2016, 163, A2586–A2591. [Google Scholar] [CrossRef]
- Kim, S.C.; Lim, H.; Kim, H.; Yi, J.S.; Lee, D. Nitrogen and oxygen dual-doping on carbon electrodes by urea thermolysis and its electrocatalytic significance for vanadium redox flow battery. Electrochim. Acta 2020, 348, 136286. [Google Scholar] [CrossRef]
- Kim, K.J.; Lee, H.S.; Kim, J.; Park, M.S.; Kim, J.H.; Kim, Y.J.; Skyllas-Kazacos, M. Superior electrocatalytic activity of a robust carbon-felt electrode with oxygen-rich phosphate groups for all-vanadium redox flow batteries. ChemSusChem 2016, 9, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.W.; Deng, Q.; Peng, C.; Zeng, X.X.; Wu, A.J.; Zhou, C.J.; Ma, Q.; Yin, Y.X.; Lu, X.Y.; Guo, Y.G. Unveiling the role of heteroatom gradient-distributed carbon fibers for vanadium redox flow batteries with long service life. ACS Appl. Mater. Interfaces 2019, 11, 11451–11458. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Ling, W.; Sheng, H.; Zhou, Y.; Wu, X.; Zeng, X.-X.; Wu, X.; Guo, Y.-G. Heteroatom-doped electrodes for all-vanadium redox flow batteries with ultralong lifespan. J. Mater. Chem. A 2018, 6, 41–44. [Google Scholar] [CrossRef]
- Pasala, V.; Ramavath, J.N.; He, C.; Ramani, V.K.; Ramanujam, K. N- and P-co-doped graphite felt electrode for improving positive electrode chemistry of the vanadium redox flow battery. ChemistrySelect 2018, 3, 8678–8687. [Google Scholar] [CrossRef]
- Shah, A.B.; Wu, Y.H.; Joo, Y.L. Direct addition of sulfur and nitrogen functional groups to graphite felt electrodes for improving all-vanadium redox flow battery performance. Electrochim. Acta 2019, 297, 905–915. [Google Scholar] [CrossRef]
- Ji, J.; Noh, C.; Shin, M.; Oh, S.; Chung, Y.; Kwon, Y.; Kim, D.-H. Vanadium redox flow batteries using new mesoporous nitrogen-doped carbon coated graphite felt electrode. Appl. Surf. Sci. 2023, 611, 155665. [Google Scholar] [CrossRef]
- Kang, M.G.; Ahn, W.; Kang, J.; Song, S.A.; Kim, K.; Woo, J.Y.; Jeong, Y.-C.; Koo, B.; Jung, D.S.; Lim, S.N. Superior electrocatalytic negative electrode with tailored nitrogen functional group for vanadium redox flow battery. J. Energy Chem. 2023, 78, 148–157. [Google Scholar] [CrossRef]
- Wang, S.Y.; Zhao, X.S.; Cochell, T.; Manthiram, A. Nitrogen-Doped Carbon Nanotube/Graphite Felts as Advanced Electrode Materials for Vanadium Redox Flow Batteries. J. Phys. Chem. Lett. 2012, 3, 2164–2167. [Google Scholar] [CrossRef]
- Jin, J.; Fu, X.; Liu, Q.; Liu, Y.; Wei, Z.; Niu, K.; Zhang, J. Identifying the active site in nitrogen-doped graphene for the VO2+/VO2+ redox reaction. ACS Nano 2013, 7, 4764–4773. [Google Scholar] [CrossRef]
- Schnucklake, M.; Kuecken, S.; Fetyan, A.; Schmidt, J.; Thomas, A.; Roth, C. Salt-templated porous carbon–carbon composite electrodes for application in vanadium redox flow batteries. J. Mater. Chem. A 2017, 5, 25193–25199. [Google Scholar] [CrossRef]
- Yang, S.L.; Peng, L.; Huang, P.P.; Wang, X.S.; Sun, Y.B.; Cao, C.Y.; Song, W.G. Nitrogen, phosphorus, and sulfur co-doped hollow carbon shell as superior metal-free catalyst for selective oxidation of aromatic alkanes. Angew. Chem. Int. Ed. 2016, 55, 4016–4020. [Google Scholar] [CrossRef]
- Wu, Z.S.; Winter, A.; Chen, L.; Sun, Y.; Turchanin, A.; Feng, X.; Mullen, K. Three-dimensional nitrogen and boron co-doped graphene for high-performance all-solid-state supercapacitors. Adv. Mater. 2012, 24, 5130–5135. [Google Scholar] [CrossRef] [PubMed]
- Tareen, A.K.; Khan, K.; Iqbal, M.; Zhang, Y.; Long, J.; Nazeer, F.; Mahmood, A.; Mahmood, N.; Shi, Z.; Ma, C.; et al. Recent advances in novel graphene: New horizons in renewable energy storage technologies. J. Mater. Chem. C 2022, 10, 11472–11531. [Google Scholar] [CrossRef]
- Kim, H.; Paick, J.; Yi, J.S.; Lee, D. Kinetic relevancy of surface defects and heteroatom functionalities of carbon electrodes for the vanadium redox reactions in flow batteries. J. Power Sources 2023, 557, 232612. [Google Scholar] [CrossRef]
- Inagaki, M.; Toyoda, M.; Soneda, Y.; Morishita, T. Nitrogen-doped carbon materials. Carbon 2018, 132, 104–140. [Google Scholar] [CrossRef]
- Wei, L.; Zhao, T.S.; Zhao, G.; An, L.; Zeng, L. A high-performance carbon nanoparticle-decorated graphite felt electrode for vanadium redox flow batteries. Appl. Energy 2016, 176, 74–79. [Google Scholar] [CrossRef]
- Schmidt, C.N.; Cao, G.Z. Properties of mesoporous carbon modified carbon felt for anode of all-vanadium redox flow battery. Sci. China Mater. 2016, 59, 1037–1050. [Google Scholar] [CrossRef]
- Yang, H.T.; Fan, C.L.; Zhu, Q.S. Activated charcoal modified graphite felts using for positive electrodes of vanadium redox flow battery. J. Electrochem. Energy Convers. Storage 2017, 14, 041004. [Google Scholar] [CrossRef]
- Faraji, S.; Ani, F.N. The development supercapacitor from activated carbon by electroless plating—A review. Renew. Sustain. Energy Rev. 2015, 42, 823–834. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, L.; Shen, Y.; Wu, L.T.; Yu, L.H.; Liang, F.; Xi, J.Y. Carbon dots promoted vanadium flow batteries for all-climate energy storage. Chem. Commun. 2017, 53, 7565–7568. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Hung, C.H.; Wang, S.P.; Chiang, I.L. Graphite felt with vapor grown carbon fibers as electrodes for vanadium redox flow batteries. Rare Met. 2011, 30, 1–4. [Google Scholar] [CrossRef]
- Park, M.; Jung, Y.J.; Kim, J.; Lee, H.; Cho, J. Synergistic effect of carbon nanofiber/nanotube composite catalyst on carbon felt electrode for high-performance all-vanadium redox flow battery. Nano Lett. 2013, 13, 4833–4839. [Google Scholar] [CrossRef]
- Huang, J.S.; Liu, Y.; You, T.Y. Carbon nanofiber based electrochemical biosensors: A review. Anal. Methods 2010, 2, 202. [Google Scholar] [CrossRef]
- Jelyani, M.Z.; Rashid-Nadimi, S.; Asghari, S. Treated carbon felt as electrode material in vanadium redox flow batteries: A study of the use of carbon nanotubes as electrocatalyst. J. Solid State Electrochem. 2016, 21, 69–79. [Google Scholar] [CrossRef]
- Abbas, A.; Eng, X.E.; Ee, N.; Saleem, F.; Wu, D.; Chen, W.; Handayani, M.; Tabish, T.A.; Wai, N.; Lim, T.M. Development of reduced graphene oxide from biowaste as an electrode material for vanadium redox flow battery. J. Energy Storage 2021, 41, 102848. [Google Scholar] [CrossRef]
- Fu, H.; Bao, X.; He, M.; Xu, J.; Miao, Z.; Ding, M.; Liu, J.; Jia, C. Defect-rich graphene skin modified carbon felt as a highly enhanced electrode for vanadium redox flow batteries. J. Power Sources 2023, 556, 232443. [Google Scholar] [CrossRef]
- Di Blasi, O.; Briguglio, N.; Busacca, C.; Ferraro, M.; Antonucci, V.; Di Blasi, A. Electrochemical investigation of thermically treated graphene oxides as electrode materials for vanadium redox flow battery. Appl. Energy 2015, 147, 74–81. [Google Scholar] [CrossRef]
- González, Z.; Flox, C.; Blanco, C.; Granda, M.; Morante, J.R.; Menéndez, R.; Santamaría, R. Outstanding electrochemical performance of a graphene-modified graphite felt for vanadium redox flow battery application. J. Power Sources 2017, 338, 155–162. [Google Scholar] [CrossRef]
- Deng, Q.; Huang, P.; Zhou, W.X.; Ma, Q.; Zhou, N.; Xie, H.; Ling, W.; Zhou, C.J.; Yin, Y.X.; Wu, X.W.; et al. A high-performance composite electrode for vanadium redox flow batteries. Adv. Energy Mater. 2017, 7, 1700461. [Google Scholar] [CrossRef]
- Moghim, M.H.; Eqra, R.; Babaiee, M.; Zarei-Jelyani, M.; Loghavi, M.M. Role of reduced graphene oxide as nano-electrocatalyst in carbon felt electrode of vanadium redox flow battery. J. Electroanal. Chem. 2017, 789, 67–75. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, H.R.; Ma, Q.; Wu, A.J.; Zhang, W.; Zhang, C.X.; Chen, Z.H.; Zeng, X.X.; Wu, X.W.; Wu, Y.P. Carbon sheet-decorated graphite felt electrode with high catalytic activity for vanadium redox flow batteries. Carbon 2019, 148, 9–15. [Google Scholar] [CrossRef]
- Wu, L.T.; Shen, Y.; Yu, L.H.; Xi, J.Y.; Qiu, X.P. Boosting vanadium flow battery performance by nitrogen-doped carbon nanospheres electrocatalyst. Nano Energy 2016, 28, 19–28. [Google Scholar] [CrossRef]
- Zhao, Y.; Yu, L.H.; Qiu, X.P.; Xi, J.Y. Carbon layer-confined sphere/fiber hierarchical electrodes for efficient and durable vanadium flow batteries. J. Power Sources 2018, 402, 453–459. [Google Scholar] [CrossRef]
- He, Z.X.; Jiang, Y.Q.; Wei, Y.L.; Zhao, C.; Jiang, F.Y.; Li, L.; Zhou, H.Z.; Meng, W.; Wang, L.; Dai, L. N,P co-doped carbon microsphere as superior electrocatalyst for VO2+/VO2+ redox reaction. Electrochim. Acta 2018, 259, 122–130. [Google Scholar] [CrossRef]
- Opar, D.O.; Nankya, R.; Lee, J.; Jung, H. Three-dimensional mesoporous graphene-modified carbon felt for high-performance vanadium redox flow batteries. Electrochim. Acta 2020, 330, 135276. [Google Scholar] [CrossRef]
- Lee, J.; Nankya, R.; Kim, A.; Jung, H. Fine-tuning the pore size of mesoporous graphene in a few nanometer-scale by controlling the interaction between graphite oxide sheets. Electrochim. Acta 2018, 290, 496–505. [Google Scholar] [CrossRef]
- Park, S.; Kim, J.; Kwon, K. A review on biomass-derived N-doped carbons as electrocatalysts in electrochemical energy applications. Chem. Eng. J. 2022, 446, 137116. [Google Scholar] [CrossRef]
- Park, M.; Ryu, J.; Kim, Y.; Cho, J. Corn protein-derived nitrogen-doped carbon materials with oxygen-rich functional groups: A highly efficient electrocatalyst for all-vanadium redox flow batteries. Energy Environ. Sci. 2014, 7, 3727–3735. [Google Scholar] [CrossRef]
- Aziz, M.A.; Hossain, S.I.; Shanmugam, S. Hierarchical oxygen rich-carbon nanorods: Efficient and durable electrode for all-vanadium redox flow batteries. J. Power Sources 2020, 445, 227329. [Google Scholar] [CrossRef]
- Mahanta, V.; Raja, M.; Kothandaraman, R. Activated carbon from sugarcane bagasse as a potential positive electrode catalyst for vanadium redox flow battery. Mater. Lett. 2019, 247, 63–66. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.A.; Wu, X.W.; Yuan, X.H.; Hu, J.P.; Zhou, Q.M.; Liu, Z.H.; Wu, Y.P. Porous carbon derived from disposable shaddock peel as an excellent catalyst toward VO2+/VO2+ couple for vanadium redox battery. J. Power Sources 2015, 299, 301–308. [Google Scholar] [CrossRef]
- Jiang, Y.Q.; Li, Y.H.; Zhu, J.; He, Z.X.; Meng, W.; Zhou, H.Z.; Wang, L.; Dai, L. Fungi-derived, functionalized, and wettability-improved porous carbon materials: An excellent electrocatalyst toward VO2+/VO2+ redox reaction for vanadium redox flow battery. J. Electrochem. Soc. 2018, 165, A1813–A1821. [Google Scholar] [CrossRef]
- Jiang, Y.Q.; Cheng, G.; He, Z.X.; Chen, J.; Li, Y.H.; Zhu, J.; Meng, W.; Zhou, H.Z.; Dai, L.; Wang, L. Biomass-derived porous graphitic carbon with excellent electrocatalytic performances for vanadium redox reactions. J. Electrochem. Soc. 2019, 166, A3918–A3926. [Google Scholar] [CrossRef]
- Lv, Y.; Li, Y.; Han, C.; Chen, J.; He, Z.; Zhu, J.; Dai, L.; Meng, W.; Wang, L. Application of porous biomass carbon materials in vanadium redox flow battery. J. Colloid Interface Sci. 2020, 566, 434–443. [Google Scholar] [CrossRef]
- Cheng, D.X.; Tian, M.R.; Wang, B.Y.; Zhang, J.L.; Chen, J.F.; Feng, X.J.; He, Z.X.; Dai, L.; Wang, L. One-step activation of high-graphitization N-doped porous biomass carbon as advanced catalyst for vanadium redox flow battery. J. Colloid Interface Sci. 2020, 572, 216–226. [Google Scholar] [CrossRef]
- Hu, Z.; Miao, Z.; Xu, Z.; Zhu, X.; Zhong, F.; Ding, M.; Wang, J.; Xie, X.; Jia, C.; Liu, J. Carbon felt electrode modified by lotus seed shells for high-performance vanadium redox flow battery. Chem. Eng. J. 2022, 450, 138377. [Google Scholar] [CrossRef]
- Lee, M.E.; Jang, D.; Lee, S.; Yoo, J.; Choi, J.; Jin, H.-J.; Lee, S.; Cho, S.Y. Silk protein-derived carbon fabric as an electrode with high electro-catalytic activity for all-vanadium redox flow batteries. Appl. Surf. Sci. 2021, 567, 150810. [Google Scholar] [CrossRef]




| Modification Method | Modification Reagent | Electrode Size | Electrode | Current Density/mA·cm−2 | EE | Ref. |
|---|---|---|---|---|---|---|
| Microwave | Ar and O2 | 3 × 3 cm2 | P and N | 40 | 85% | [14] |
| Square wave potential pulse | NaOH | 3 × 3 × 0.6 cm3 | / | 30 | 87.0% | [15] |
| Hydrothermal method | HF/H2O2 | 3 × 3 cm2 | P | 50 | 75.7% | [17] |
| Heating | Air | 5.4 × 6 × 0.3 cm3 | / | 40 | 75% | [18] |
| Heating | CO2 | 5 × 5 cm2 | / | 50(80) | 84.15% (~78%) | [20] |
| Heating | Water vapor | 5 × 5 × 0.65 cm3 | P and N | 80(50) | 78.12% (83.10%) | [21] |
| Heating and etching | Ni(NO3)2·6H2O HNO3 and HCl | 16 × 0.3 cm3 | / | 150 | ~71% (VE = 72.8%) | [22] |
| Hydrothermal method and heating and etching | N2/ HCl | 5 × 5 × 0.5 cm3 | / | 300(50) | 57.3% (~75%) | [23] |
| Heating | C4H6O4·Co·4H2O/Air | 25 cm2 | / | 50 | 87.3% | [31] |
| Heating and etching | FeCl3/Air/HCl | 2 × 2 cm2 | / | 200 (500) | 87.02% (73.23%) | [30] |
| Heating | K2FeO4 | 2 × 2 cm2 | / | 200 (500) | 79.74% (63.41%) | [34] |
| Vapor treatment | HNO3 | 2 × 2 cm2 × 4.2 mm | P and N | 250 | 71.86% | [35] |
| Precursor | Electrode Modified | Electrode Size | Electrode | Current Density/mA·cm−2 | EE | Ref. |
|---|---|---|---|---|---|---|
| CuSO4 | Cu-GF | 4.7 × 0.15 cm3 | / | 300 (200) | 80.1% (84%) | [39] |
| α-PbO2 and β-PbO2 | PbO2-GF | 3 × 4 × 0.3 cm3 | P | 80 | 78.1% | [52] |
| Cr(NO3)3 | Cr2O3-GF | 2 × 2 × 0.3 cm3 | P and N | 150 | 67.6% | [56] |
| Co(NO3)2 | CoO-GF | 2 × 2 × 0.3 cm3 | P and N | 150 | 69.4% | [57] |
| Ni(NO3)2 | NiO-GF | 16 × 0.3 cm3 | P and N | 125 | 74.5% | [58] |
| Ta2O5 | Ta2O5-GF | 5 × 5 cm2 | P and N | 80 | 73.73% | [45] |
| SnCl4 | SnO2-CF | 5 × 5 cm2 | / | 150 | 77.3% | [46] |
| WCl6 | H-W18O49NWs-GF | 5 × 5 × 0.5 cm3 | / | 160 | 66.1% | [63] |
| Na2WO4 and NbCl5 | Nb/h-WO3 NWs-GF | 5 cm2 | / | 160 | 65.83% | [71] |
| Niobium ethoxide and titanium isopropoxide | TiNb2O7-GF | 5 × 5 × 0.65 cm3 | / | 160 | 70.32% | [72] |
| Co(NO3)2 and NiCl2 | NiCoO2-GF | 2 × 2 × 0.3 cm3 | P and N | 150 | 72.5% | [73] |
| Titanium butoxide and H2 | TiO2:H-GF | 4 × 0.5 cm3 | N | 150 | 66.1% | [64] |
| Titanium butoxide and NH3 | TiO2:N-CF | 4 × 0.5 cm3 | N | 150 | 71% | [65] |
| TiCl4 and NH3 | TiN-GF | 4.7 × 0.15 cm3 | N | 300 (200) | 77.4% (82.8%) | [66] |
| Ti3C2Tx | Ti3C2Tx-GF | 1.95 × 3 × 0.6 cm3 | N | 200 (300) | 81.3% (75%) | [70] |
| TiF4 | TiC-GF | 5 × 4 cm2 | N | 100 | ~74% | [67] |
| C4H6CoO4⋅4H2O | Co3O4-CF | 3.3 × 3.3 cm2 | P and N | 160 300 | 71.4% (55.8%) | [74] |
| Source of Heteroatom | Electrode Modified | Electrode Size | Side Modified Electrode Used | Current Density/mA·cm−2 | EE | Ref. |
|---|---|---|---|---|---|---|
| Pyrrole | N-GF | 3 × 3 × 0.3 cm3 | / | 150 | 74.2% | [77] |
| Dopamine | N-GF | 3 × 3 cm2 | P and N | 150 | 75.8% | [78] |
| Dopamine | N-GF | 3.3 × 3.3 × 0.42 cm3 | / | 150 | 75.5% | [79] |
| Urea | N/C-GF | 2 × 2 cm2 | / | 200 | 64.1% | [92] |
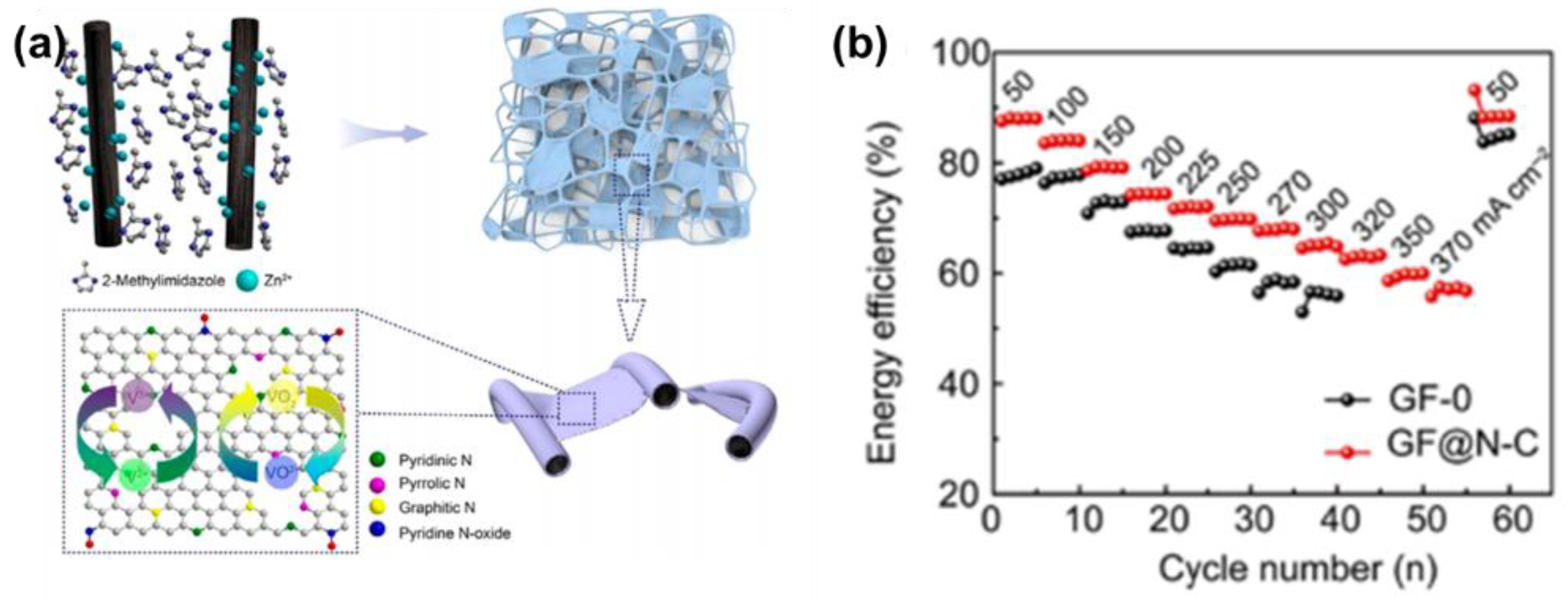
| Zn(NO3)2 and 2-methylimidazole | N/C-GF | 2 × 2 × 20.5 cm3 | / | 200 (370) | 74.3% (56.9%) | [93] |
| p-Phenylene-diamine and phytic acid | N/C-GF | 4 cm2 | / | 200 | 65.4% | [94] |
| PIM | N/C-GF | 3 × 3 cm2 | / | 150 | ~71.5% | [95] |
| NH3 and O2 | N/O-GF | 5 × 3 × 0.5 cm3 | P and N | 110 | ~73% | [97] |
| Urea; NH3 and O2 | N/O-GF | 5 × 3 × 0.5 cm3 | P and N | 80 | ~71% | [98] |
| Urea | N-GF | 5 × 8 × 0.6 cm3 | / | 150 | 81.32% | [83] |
| N2 | N-GF | 3.2 × 3.2 × 0.6 cm3 | P and N | 80 | 76.8% | [80] |
| NH3 | N-CF | 4 × 5cm2 | P | 100 | 85% | [82] |
| N2 plasma | N-CF | 5 × 5cm2 | / | 64 | ~70% | [81] |
| HEDP | P-GF | 5 × 5cm2 | / | 150 (400) | ~79% (~52%) | [87] |
| NH4PF6 | P-O-CF | 5 × 5cm2 | P and N | 120 | ~75.5% | [102] |
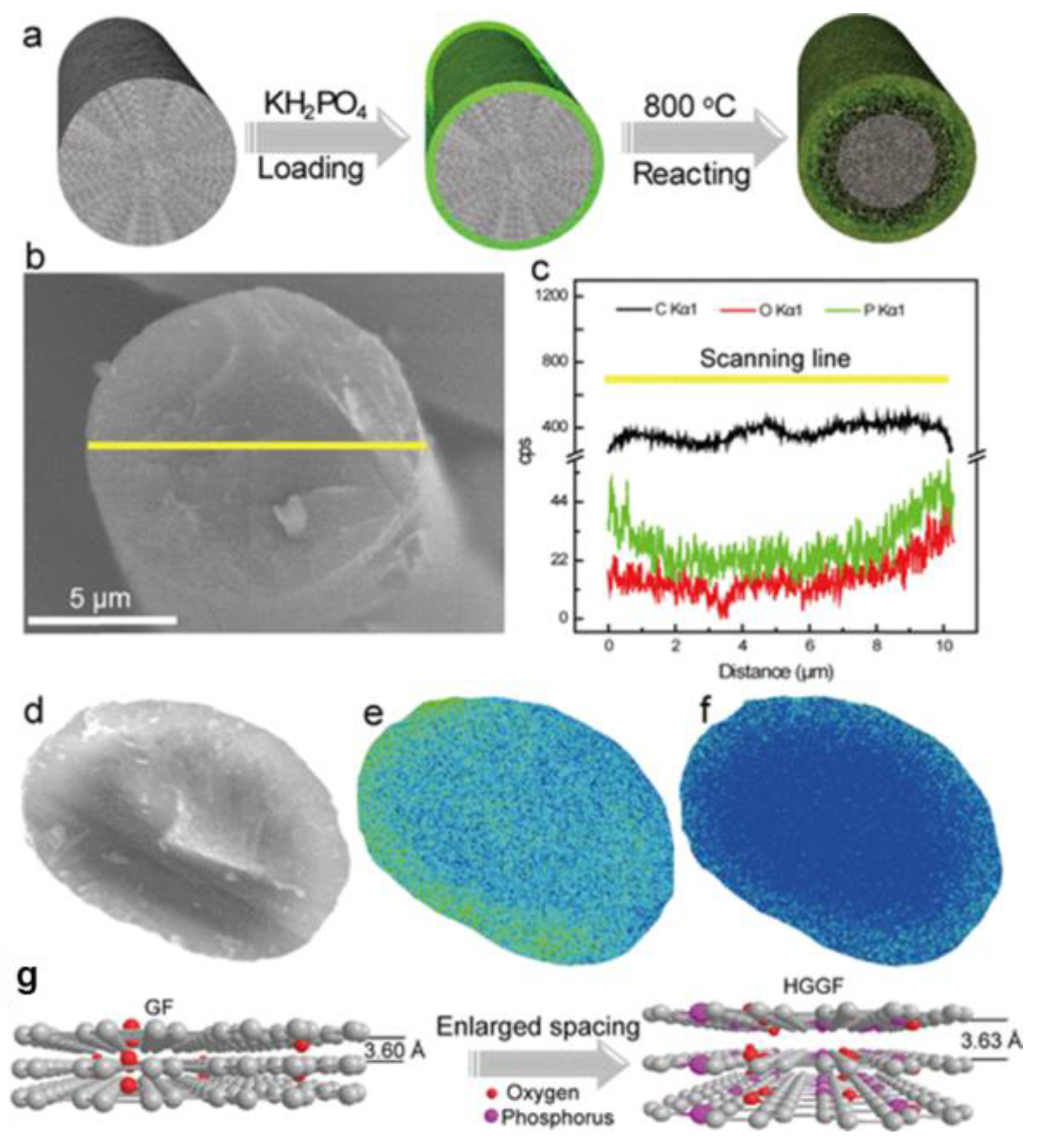
| KH2PO4 | HGGF | / | / | 150 (300) | 73.34% (60%) | [103] |
| Microcrystalline cellulose and (NH4)2HPO4 | N-P-GF | 16 cm2 | N (cathode) | 70 | ~74% | [105] |
| Precursors | Electrode Modified | Electrode Size | Side Modified Electrode Used | Current Density /mA·cm−2 | EE | Ref. |
|---|---|---|---|---|---|---|
| CNP | N-CNP-GF | 1.8 × 2.6 × 0.6 cm3 | / | 200 | 72.7% | [117] |
| Sucrose | Charcoal-GF | 3 × 3 × 0.3 cm3 | P | 100 | 81.7% | [119] |
| Ethylenediamine | N-CNT-GF | 5 cm2 | / | 10 | 77% | [109] |
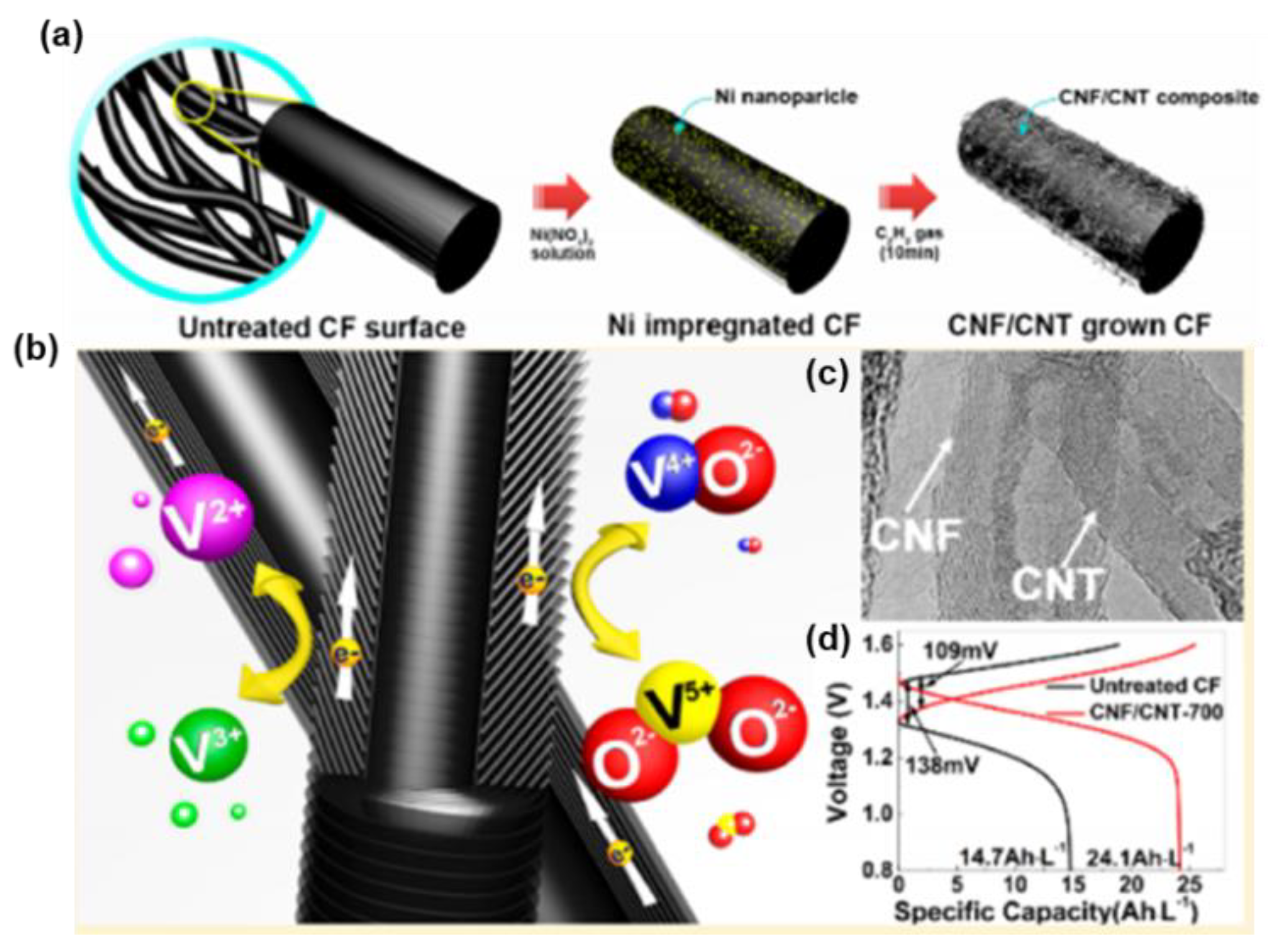
| C2H2 | CNF/CNT-CF | 5 cm2 | / | 100 | 65.6% | [123] |
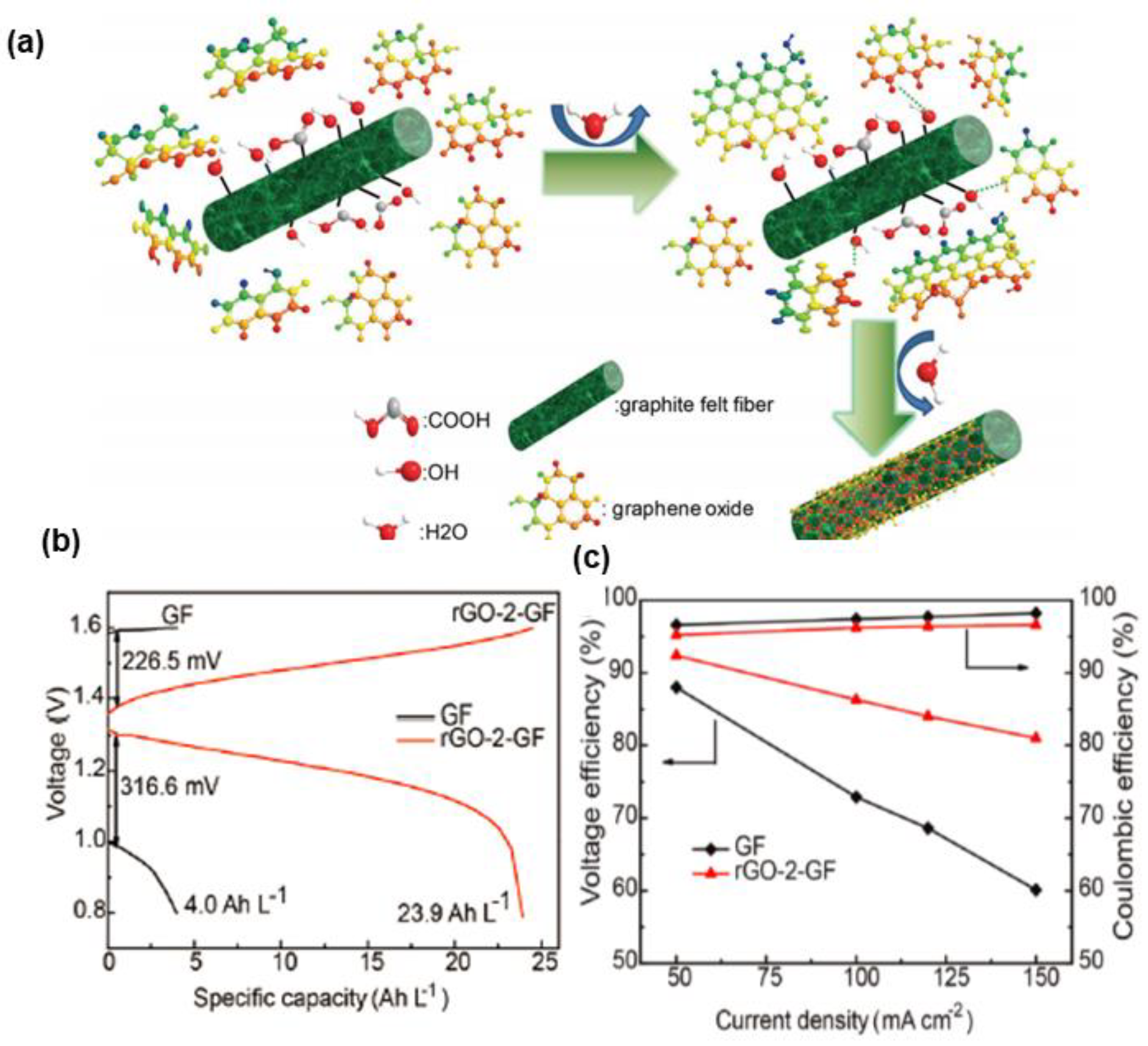
| GO | G-GF | 4 cm2 | P | 25 | 95.8% | [129] |
| GO | rGO-GF | 2 × 2 × 0.5cm3 | / | 300 | 60% | [130] |
| Phytic acid and urea | GF@CS | 2 × 2cm2 | / | 150 (300) | 74.79% (~54%) | [132] |
| Dopamine | N-CNS-GF | 5 × 5 × 0.5 cm3 | / | 300 | 53% | [133] |
| Glucose and (NH4)2HPO4 | N/P-CMS-GF | 3 × 3 cm2 | P | 100 | 67.1% | [135] |
| PF and SiO2 nanospheres | CS-GF | 5 × 5 × 0.5 cm3 | / | 150 (300) | 75.5% (63.1%) | [134] |
| GO and triblock copolymer | MG-CF | 6 × 0.42 cm3 | / | 200 | 53.3% | [136] |
| Corn protein and CB | N-CB-CF | 5 cm2 | P and N | 150 | 68.6% | [139] |
| Corn protein and PAN | N-CNRs-CF | 9 × 0.3 cm3 | P and N | 160 | ~79.5% | [140] |
| SCB | SCB-GF | 5 × 5 × 0.6 cm3 | / | 100 | ~72% | [141] |
| Modification Methods | Main Advantages | Main Drawbacks |
|---|---|---|
| Surface treatment or structure rebuilding | Increased specific surface area, enriched oxygen functional groups, high electrochemical activity, enhanced wettability, reduced polarization, and prominent energy efficiency. | Corrosion of electrode caused by excessive surface oxidation; oxygen functional groups may not be stable enough during cycling. |
| Metallic modification | Improved electrical conductivity, lower polarization, and better kinetic reversibility and electrochemical performance. | Highly energy-/time-consuming and environmentally unfriendly, high cost of production, and relatively weak adhesion between the grown metal species and the carbonous matrix. |
| Nonmetal doping | Improved electron distribution and local bonding environment on the surface of carbon-based materials, hydrophilicity and electrocatalytic activity, as well as improved electrical conductivity; reasonable cost and satisfactory cycle stability. | Unsatisfactory battery durability; more efficient and environmentally friendly heteroatom doping techniques should be explored. |
| Carbonous nanomaterial modification | Enlarged surface area of electrode, rich surface functional groups, superb electrical conductivity, promotion of adsorption and desorption processes of vanadium ions, accelerated reaction kinetics, and high mass transport kinetics. | 0D or 1D carbonous nanomaterials: insufficient contact area and unconnected nanomaterials. 2D carbonous nanomaterials: irreversible stacking and the agglomeration of nanosheets due to strong π–π interaction, resulting in decreased active surface area. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, C.; Shen, Z.; Zhu, Y.; Cheng, Y. Insights into the Modification of Carbonous Felt as an Electrode for Vanadium Redox Flow Batteries. Materials 2023, 16, 3811. https://doi.org/10.3390/ma16103811
Ding C, Shen Z, Zhu Y, Cheng Y. Insights into the Modification of Carbonous Felt as an Electrode for Vanadium Redox Flow Batteries. Materials. 2023; 16(10):3811. https://doi.org/10.3390/ma16103811
Chicago/Turabian StyleDing, Cong, Zhefei Shen, Ying Zhu, and Yuanhui Cheng. 2023. "Insights into the Modification of Carbonous Felt as an Electrode for Vanadium Redox Flow Batteries" Materials 16, no. 10: 3811. https://doi.org/10.3390/ma16103811
APA StyleDing, C., Shen, Z., Zhu, Y., & Cheng, Y. (2023). Insights into the Modification of Carbonous Felt as an Electrode for Vanadium Redox Flow Batteries. Materials, 16(10), 3811. https://doi.org/10.3390/ma16103811







