Improvement of the Quality of Recycled Concrete Aggregate Subjected to Chemical Treatments: A Review
Abstract
:1. Introduction
2. Chemistry of Acid Treatment in RCA
3. RCA Properties
3.1. Water Absorption
3.2. Determination of Mortar Loss
3.3. Bulk Density
3.4. Microscopic Analysis of the RCA
4. Properties of Concrete with Treated RCA
4.1. Fresh-State Properties and Density
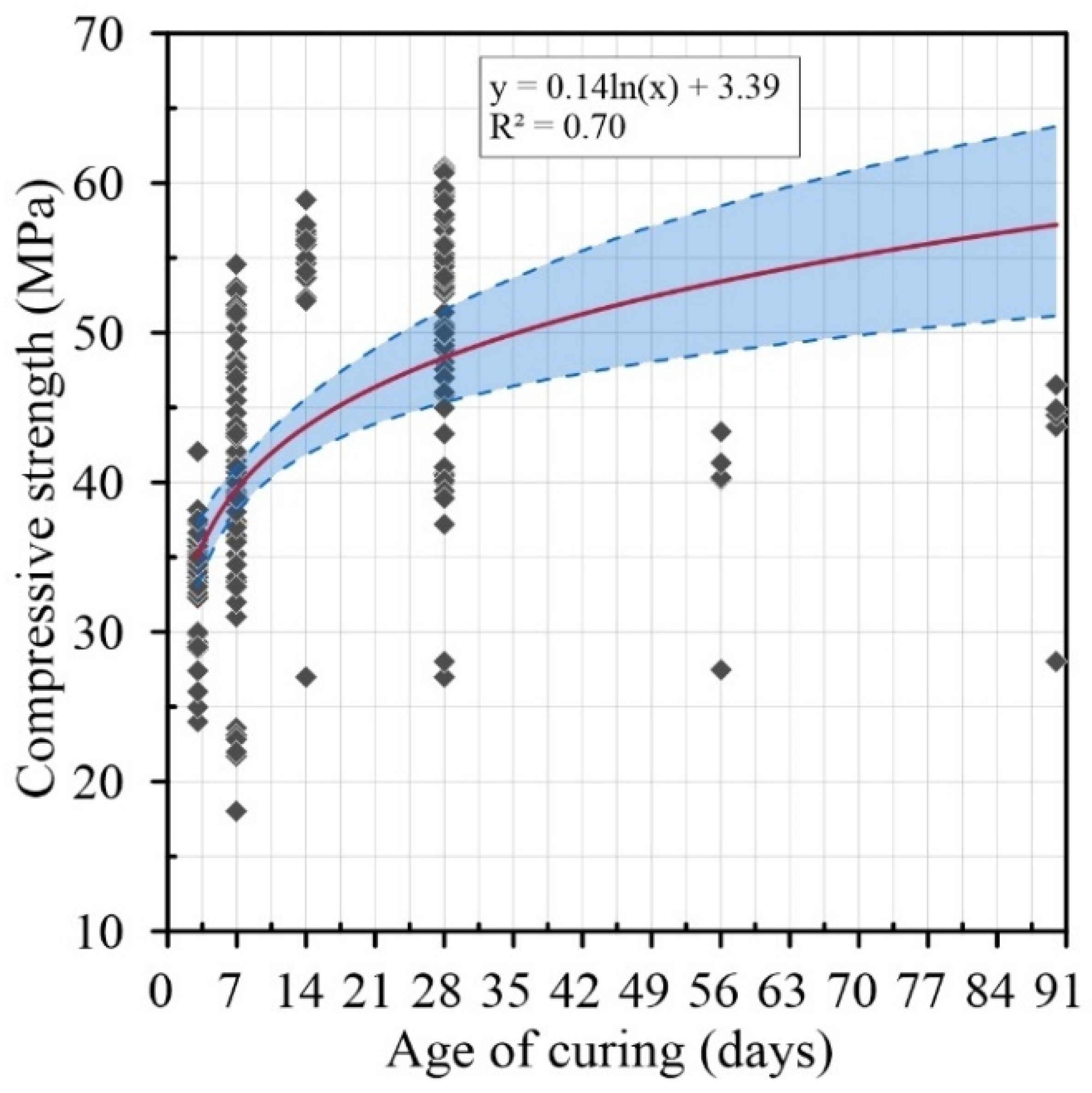
4.2. Compressive Strength
4.3. Tensile Strength
4.4. Modulus of Elasticity
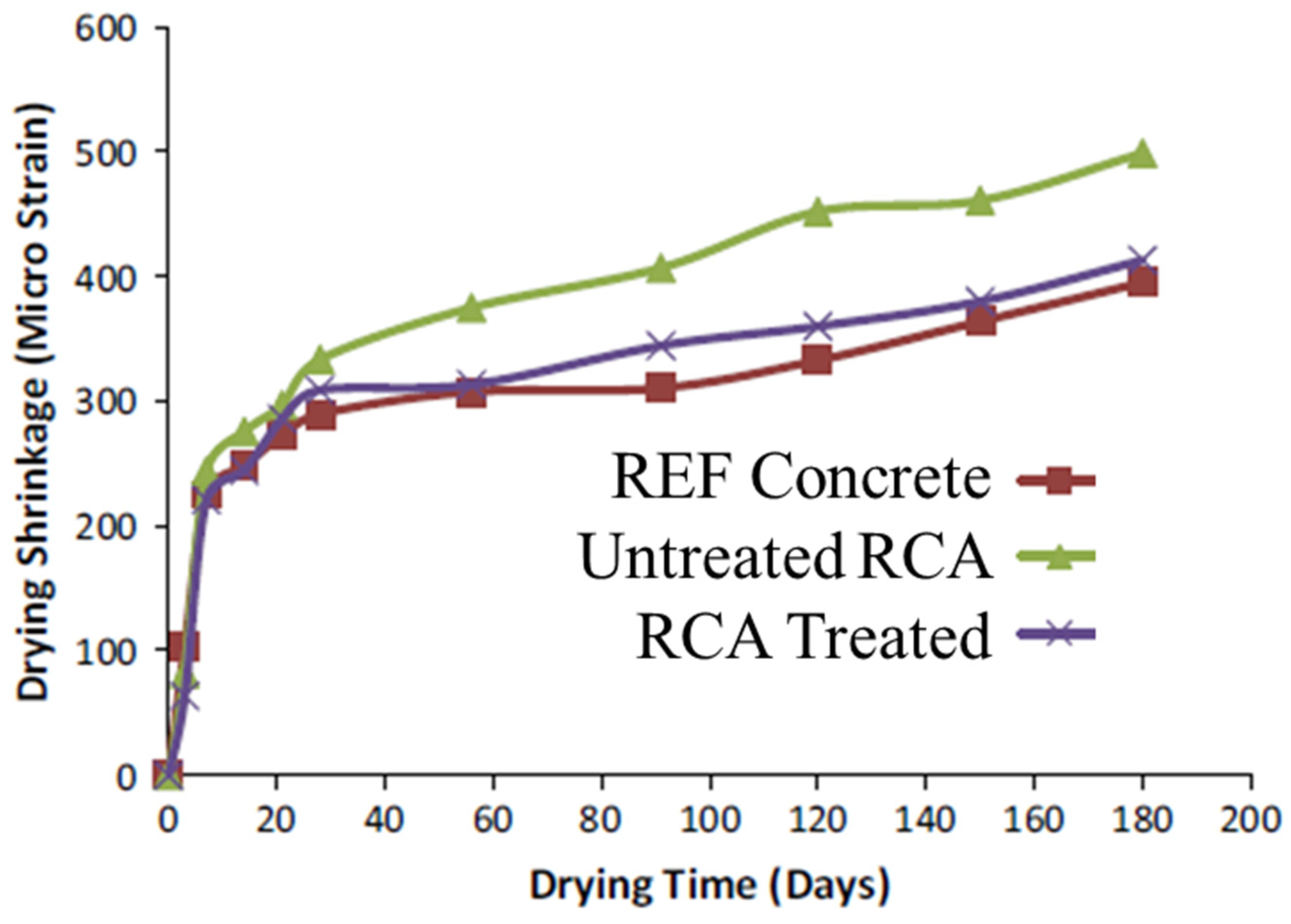
4.5. Shrinkage
4.6. Chloride Ion Penetrability and Carbonation Resistance
4.7. Interfacial Zone between Cement Paste and RCA
5. Statistical Analysis
Statistical Analysis of RCA Properties
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Poon, C.S.; Shui, Z.; Lam, L. Effect of microstructure of ITZ on compressive strength of concrete prepared with recycled aggregates. Constr. Build. Mater. 2004, 18, 461–468. [Google Scholar] [CrossRef]
- Sidorova, A.; Vazquez-Ramonich, E.; Barra-Bizinotto, M.; Roa-Rovira, J.J.; Jimenez-Pique, E. Study of the recycled aggregates nature’s influence on the aggregate–cement paste interface and ITZ. Constr. Build. Mater. 2014, 68, 677–684. [Google Scholar] [CrossRef]
- Evangelista, L.; De Brito, J. Concrete with fine recycled aggregates: A review. Eur. J. Environ. Civ. Eng. 2014, 18, 129–172. [Google Scholar] [CrossRef]
- Silva, R.; Neves, R.; De Brito, J.; Dhir, R. Carbonation behaviour of recycled aggregate concrete. Cem. Concr. Compos. 2015, 62, 22–32. [Google Scholar] [CrossRef] [Green Version]
- Abbas, A.; Fathifazl, G.; Isgor, O.B.; Razaqpur, A.G.; Fournier, B.; Foo, S. Proposed method for determining the residual mortar content of recycled concrete aggregates. J. ASTM Int. 2007, 5, 1–12. [Google Scholar]
- Akbarnezhad, A.; Ong, K.; Zhang, M.; Tam, C. Acid treatment technique for determining the mortar content of recycled concrete aggregates. J. Test. Eval. 2013, 41, 441–450. [Google Scholar] [CrossRef]
- Akbarnezhad, A.; Ong, K.; Zhang, M.; Tam, C.; Foo, T. Microwave-assisted beneficiation of recycled concrete aggregates. Constr. Build. Mater. 2011, 25, 3469–3479. [Google Scholar] [CrossRef]
- Al-Bayati, H.K.A.; Das, P.K.; Tighe, S.L.; Baaj, H. Evaluation of various treatment methods for enhancing the physical and morphological properties of coarse recycled concrete aggregate. Constr. Build. Mater. 2016, 112, 284–298. [Google Scholar] [CrossRef]
- Ismail, S.; Ramli, M. Engineering properties of treated recycled concrete aggregate (RCA) for structural applications. Constr. Build. Mater. 2013, 44, 464–476. [Google Scholar] [CrossRef]
- Ismail, S.; Ramli, M. Mechanical strength and drying shrinkage properties of concrete containing treated coarse recycled concrete aggregates. Constr. Build. Mater. 2014, 68, 726–739. [Google Scholar] [CrossRef]
- Kim, Y.; Hanif, A.; Kazmi, S.M.S.; Munir, M.J.; Park, C. Properties enhancement of recycled aggregate concrete through pretreatment of coarse aggregates—Comparative assessment of assorted techniques. J. Clean. Prod. 2018, 191, 339–349. [Google Scholar] [CrossRef]
- Pandurangan, K.; Dayanithy, A.; Prakash, S.O. Influence of treatment methods on the bond strength of recycled aggregate concrete. Constr. Build. Mater. 2016, 120, 212–221. [Google Scholar] [CrossRef]
- Purushothaman, R.; Amirthavalli, R.R.; Karan, L. Influence of treatment methods on the strength and performance characteristics of recycled aggregate concrete. J. Mater. Civ. Eng. 2014, 27, 04014168. [Google Scholar] [CrossRef]
- Saravanakumar, P.; Abhiram, K.; Manoj, B. Properties of treated recycled aggregates and its influence on concrete strength characteristics. Constr. Build. Mater. 2016, 111, 611–617. [Google Scholar] [CrossRef]
- Tam, V.W.Y.; Tam, C.M.; Le, K.N. Removal of cement mortar remains from recycled aggregate using pre-soaking approaches. Resour. Conserv. Recycl. 2007, 50, 82–101. [Google Scholar] [CrossRef] [Green Version]
- Shima, H.; Tateyashiki, H.; Matsuhashi, R.; Yoshida, Y. An advanced concrete recycling technology and its applicability assessment through input-output analysis. J. Adv. Concr. Technol. 2005, 3, 53–67. [Google Scholar] [CrossRef] [Green Version]
- Yonezawa, T.; Kamiyama, Y.; Yanagibashi, K.; Kojima, M.; Arakawa, K. A study on a technology for producing high quality recycled coarse aggregate. Zairyo 2001, 50, 835–842. [Google Scholar]
- Ogawa, H.; Nawa, T. Improving the quality of recycled fine aggregate by selective removal of brittle defects. J. Adv. Concr. Technol. 2012, 10, 395–410. [Google Scholar] [CrossRef] [Green Version]
- Sui, Y.; Mueller, A. Development of thermo-mechanical treatment for recycling of used concrete. Mater. Struct. 2012, 45, 1487–1495. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.; Qian, X.; Chen, P.; Xu, Y.; Guo, J. An environmentally friendly method to improve the quality of recycled concrete aggregates. Constr. Build. Mater. 2017, 144, 432–441. [Google Scholar] [CrossRef] [Green Version]
- Kou, S.-C.; Poon, C.-S. Properties of concrete prepared with PVA-impregnated recycled concrete aggregates. Cem. Concr. Compos. 2010, 32, 649–654. [Google Scholar] [CrossRef]
- Dhir, B. Biotechnological tools for remediation of acid mine drainage (removal of metals from wastewater and leachate). In Bio-Geotechnologies for Mine Site Rehabilitation; Elsevier: Amsterdam, The Netherlands, 2018; pp. 67–82. [Google Scholar]
- Beddoe, R.E.; Dorner, H.W. Modelling acid attack on concrete: Part I. The essential mechanisms. Cem. Concr. Res 2005, 35, 2333–2339. [Google Scholar] [CrossRef]
- Scrivener, K.; Young, J.F. Mechanisms of Chemical Degradation of Cement-Based Systems; CRC Press: Boca Raton, FL, USA, 2014; pp. 4–21. [Google Scholar]
- Hewlett, P.; Liska, M. Lea’s Chemistry of Cement and Concrete; Butterworth-Heinemann: Oxford, UK, 2019. [Google Scholar]
- Kurdowski, W. Cement and Concrete Chemistry; Springer Science & Business: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Mehta, P.K.; Monteiro, P.J. Concrete Microstructure, Properties and Materials; McGraw Hill: New York, NY, USA, 2017. [Google Scholar]
- Mindess, S.; Young, F.; Darwin, D. Concrete, 2nd ed.; Technical Documents; Prentice Hall, Pearson Education, Inc.: Upper Saddle River, NJ, USA, 2003. [Google Scholar]
- Sarkar, S.; Mahadevan, S.; Meeussen, J.; Van der Sloot, H.; Kosson, D. Numerical simulation of cementitious materials degradation under external sulfate attack. Cem. Concr. Compos. 2010, 32, 241–252. [Google Scholar] [CrossRef] [Green Version]
- Clifton, J.R.; Pommersheim, J. Sulfate attack of cementitious materials: Volumetric relations and expansions. NIST IR 1994, 5390, 4–9. [Google Scholar]
- Brown, P.; Taylor, H. The role of ettringite in external sulfate attack. In Materials Science of Concrete: Sulfate Attack Mechanisms; The American Ceramic Society: Columbus, OH, USA, 1999; pp. 73–98. [Google Scholar]
- Allahverdi, A.; SKVARA, F. Acidic corrosion of hydrated cement based materials. Part 1.: Mechanism of the phenomenon. Ceramics 2000, 44, 114–120. [Google Scholar]
- Dyer, T. Concrete Durability; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Ramaswamy, K.; Santhanam, M. Influence of Mineralogical Nature of Aggregates on Acid Resistance of Mortar. In Proceedings of the Sixth International Conference on Durability of Concrete Structures, Leeds, UK, 18–20 July 2018. [Google Scholar]
- Alexander, M.; Fourie, C. Performance of sewer pipe concrete mixtures with portland and calcium aluminate cements subject to mineral and biogenic acid attack. Mater. Struct. 2011, 44, 313–330. [Google Scholar] [CrossRef]
- Verian, K.P.; Ashraf, W.; Cao, Y. Properties of recycled concrete aggregate and their influence in new concrete production. Resour. Conserv. Recycl. 2018, 133, 30–49. [Google Scholar] [CrossRef]
- De Brito, J.; Saikia, N. Recycled Aggregate in Concrete: Use of Industrial, Construction and Demolition Waste; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- de Brito, J.; Silva, R. Current status on the use of recycled aggregates in concrete: Where do we go from here? RILEM Tech. Lett. 2016, 1, 1–5. [Google Scholar] [CrossRef]
- Silva, R.; De Brito, J.; Dhir, R. The influence of the use of recycled aggregates on the compressive strength of concrete: A review. Eur. J. Environ. Civ. Eng. 2015, 19, 825–849. [Google Scholar] [CrossRef]
- Allahverdi, A.; ŠKVÁRA, F. Acidic corrosion of hydrated cement based materials. Ceram. Silikáty 2000, 44, 152–160. [Google Scholar]
- Pavlik, V.; Unčík, S. The rate of corrosion of hardened cement pastes and mortars with additive of silica fume in acids. Cem. Concr. Res. 1997, 27, 1731–1745. [Google Scholar] [CrossRef]
- Amorim, P.; De Brito, J.; Evangelista, L. Concrete made with coarse concrete aggregate: Influence of curing on durability. ACI Mater. J. 2012, 109, 195–204. [Google Scholar]
- Ferreira, L.; De Brito, J.; Barra, M. Influence of the pre-saturation of recycled coarse concrete aggregates on concrete properties. Mag. Concr. Res. 2011, 63, 617–627. [Google Scholar] [CrossRef]
- Silva, R.V.; De Brito, J.; Dhir, R. Properties and composition of recycled aggregates from construction and demolition waste suitable for concrete production. Constr. Build. Mater. 2014, 65, 201–217. [Google Scholar] [CrossRef]
- Belin, P.; Habert, G.; Thiery, M.; Roussel, N. Cement paste content and water absorption of recycled concrete coarse aggregates. Mater. Struct. 2014, 47, 1451–1465. [Google Scholar] [CrossRef]
- Cui, H.; Shi, X.; Memon, S.A.; Xing, F.; Tang, W. Experimental study on the influence of water absorption of recycled coarse aggregates on properties of the resulting concretes. J. Mater. Civ. Eng. 2014, 27, 04014138. [Google Scholar] [CrossRef]
- Barbudo, A.; de Brito, J.; Evangelista, L.; Bravo, M.; Agrela, F. Influence of water-reducing admixtures on the mechanical performance of recycled concrete. J. Clean. Prod. 2013, 59, 93–98. [Google Scholar] [CrossRef]
- Movassaghi, R. Durability of Reinforced Concrete Incorporating Recycled Concrete as Aggregate (RCA). Master’s Thesis, University of Waterloo, Waterloo, ON, Canada, 2006. [Google Scholar]
- Tang, A.J.; De Jesus, R.; Cunanan, A. Microstructure and mechanical properties of concrete with treated recycled concrete aggregates. Int. J. GEOMATE 2019, 16, 21–27. [Google Scholar]
- De Juan, M.S.; Gutiérrez, P.A. Study on the influence of attached mortar content on the properties of recycled concrete aggregate. Constr. Build. Mater. 2009, 23, 872–877. [Google Scholar] [CrossRef]
- Abbas, A.; Fathifazl, G.; Fournier, B.; Isgor, O.; Zavadil, R.; Razaqpur, A.; Foo, S. Quantification of the residual mortar content in recycled concrete aggregates by image analysis. Mater. Charact. 2009, 60, 716–728. [Google Scholar] [CrossRef]
- Shi, C.; Stegemann, J. Acid corrosion resistance of different cementing materials. Cem. Concr. Res. 2000, 30, 803–808. [Google Scholar] [CrossRef]
- Pavlik, V. Corrosion of hardened cement paste by acetic and nitric acids Part III: Influence of water/cement ratio. Cem. Concr. Res. 1996, 26, 475–490. [Google Scholar] [CrossRef]
- Tam, V.W.; Tam, C.M. Parameters for assessing recycled aggregate and their correlation. Waste Manag. Res. 2009, 27, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Pedro, D.; De Brito, J.; Evangelista, L. Influence of the use of recycled concrete aggregates from different sources on structural concrete. Constr. Build. Mater. 2014, 71, 141–151. [Google Scholar] [CrossRef]
- Silva, R.; De Brito, J.; Dhir, R. Fresh-state performance of recycled aggregate concrete: A review. Constr. Build. Mater. 2018, 178, 19–31. [Google Scholar] [CrossRef]
- Butler, L.; West, J.; Tighe, S. The effect of recycled concrete aggregate properties on the bond strength between RCA concrete and steel reinforcement. Cem. Concr. Res 2011, 41, 1037–1049. [Google Scholar] [CrossRef]
- ACI 211.1-91; Standard Practice for Selecting Proportions for Normal, Heavyweight, and Mass Concrete, No. 9. ACI Committee: Farmington Hills, MI, USA, 2002; pp. 120–121.
- Fathifazl, G.; Abbas, A.; Razaqpur, A.G.; Isgor, O.B.; Fournier, B.; Foo, S. New mixture proportioning method for concrete made with coarse recycled concrete aggregate. J. Mater. Civ. Eng. 2009, 21, 601–611. [Google Scholar] [CrossRef]
- Kim, H.-S.; Kim, B.; Kim, K.-S.; Kim, J.-M. Quality improvement of recycled aggregates using the acid treatment method and the strength characteristics of the resulting mortar. J. Mater. Cycles Waste Manag. 2017, 19, 968–976. [Google Scholar] [CrossRef]
- Brown, T.; LeMay, H.; Bursten, B.B.; Burdge, J.R. Química: A Ciência Central; Pearson: London, UK, 2007. [Google Scholar]
- Mehta, P.K. Mechanism of sulfate attack on portland cement concrete—Another look. Cem. Concr. Res. 1983, 13, 401–406. [Google Scholar] [CrossRef]
- Neville, A.M. Properties of Concrete; Longman: London, UK, 1995; Volume 4. [Google Scholar]
- Zhou, F.; Lydon, F.; Barr, B. Effect of coarse aggregate on elastic modulus and compressive strength of high performance concrete. Cem. Concr. Res 1995, 25, 177–186. [Google Scholar] [CrossRef]
- Kheder, G.; Al-Windawi, S. Variation in mechanical properties of natural and recycled aggregate concrete as related to the strength of their binding mortar. Mater. Struct. 2005, 38, 701–709. [Google Scholar] [CrossRef]
- Beshr, H.; Almusallam, A.; Maslehuddin, M. Effect of coarse aggregate quality on the mechanical properties of high strength concrete. Constr. Build. Mater. 2003, 17, 97–103. [Google Scholar] [CrossRef]
- Adam, I.; Sakata, K.; Ayano, T. Influence of coarse aggregate on the shrinkage of normal and high-strength concretes. Okayama Univ. Fac. Environ. Sci. Eng. Res. Rep. 2001, 6, 41–45. [Google Scholar]
- Xiao, J.; Li, W.; Sun, Z.; Lange, D.A.; Shah, S.P. Properties of interfacial transition zones in recycled aggregate concrete tested by nanoindentation. Cem. Concr. Compos. 2013, 37, 276–292. [Google Scholar] [CrossRef]
- Walpole, R.E.; Myers, R.H.; Myers, S.L.; Ye, K. Probability and Statistics for Engineers and Scientists; Macmillan: New York, NY, USA, 1993; Volume 5. [Google Scholar]
- Brown, J.D. Advanced Statistics for the Behavioral Sciences; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Meyer, R.; Krueger, D.D. A Minitab Guide to Statistics; Prentice Hall PTR: Hoboken, NJ, USA, 2001. [Google Scholar]
- Barbudo, A.; Agrela, F.; Ayuso, J.; Jiménez, J.; Poon, C. Statistical analysis of recycled aggregates derived from different sources for sub-base applications. Constr. Build. Mater. 2012, 28, 129–138. [Google Scholar] [CrossRef]
- Çakır, Ö. Experimental analysis of properties of recycled coarse aggregate (RCA) concrete with mineral additives. Constr. Build. Mater. 2014, 68, 17–25. [Google Scholar] [CrossRef]
- Tam, V.W.; Soomro, M.; Evangelista, A.C.J. A review of recycled aggregate in concrete applications (2000–2017). Constr. Build. Mater. 2018, 172, 272–292. [Google Scholar] [CrossRef]
- Shi, C.; Li, Y.; Zhang, J.; Li, W.; Chong, L.; Xie, Z. Performance enhancement of recycled concrete aggregate—A review. J. Clean. Prod. 2016, 112, 466–472. [Google Scholar] [CrossRef]
- Shaban, W.M.; Yang, J.; Su, H.; Mo, K.H.; Li, L.; Xie, J. Quality Improvement Techniques for Recycled Concrete Aggregate: A review. J. Adv. Concr. Technol. 2019, 17, 151–167. [Google Scholar] [CrossRef] [Green Version]
- Makul, N. A review on methods to improve the quality of recycled concrete aggregates. J. Sustain. Cem. -Based Mater. 2021, 10, 65–91. [Google Scholar] [CrossRef]





| Measured Parameters | Technique | References |
|---|---|---|
| Water absorption | Immersion of RCA in acidic HCl, H2SO4, and H3PO4 | Tam et al. [15] |
| Water absorption, mortar content, and bulk density | Immersion of RCA in acidic H2SO4 | Akbarnezhad et al. [6,7] |
| Water absorption, mortar content, bulk density, and microscopic analysis of the RCA | Immersion of RCA in acidic HCl | Ismail and Ramli [9,10] |
| Water absorption | Immersion of RCA in acidic HCl and H2SO4 | Purushothaman et al. [13] |
| Water absorption | Immersion of RCA in acidic HNO3 | Pandurangan [6,12] |
| Water absorption, mortar content, and bulk density | Immersion of RCA in acidic HCl, H2SO4 and HNO3 | Saravankumar et al. [14] |
| Water absorption, mortar content, and microscopic analysis of the RCA | Immersion of RCA in acidic HCl and C2H4O2 | Al-Bayati et al. [8] |
| Water absorption and mortar content | Immersion of RCA in acidic HCl and Na2SO4 | Kim et al. [11] |
| Mortar content | Immersion of RCA in acidic HCl | Juan and Gutierrez [50] |
| Mortar content | Immersion of RCA in sodium sulfate (Na2SO4), magnesium sulfate (MgSO4), and magnesium chloride (MgCl2) | Abbas et al. [5,51] |
| Water absorption, bulk density, and specific gravity | Immersion of RCA in acidic H2SO4 | Tang et al. [49] |
| Water absorption and apparent density | Immersion of RCA in acidic CH3COOH | Want et al. [20] |
| Parameter | Concrete Mix | References |
|---|---|---|
| Density | Concrete with RCA treated with HCl and Na2SO4 | Al-Bayati et al. [8] |
| Workability of concrete, density compressive strength, tensile strength, UPV, modulus of elasticity (E), and shrinkage | Concrete with RCA treated with HCl | Ismail and Ramli [9,10] |
| Workability of concrete and compressive strength | Concrete with RCA treated with HNO3 | Pandurangan et al. [12] |
| Compressive strength and modulus of elasticity (E) | Concrete with RCA treated with HCl, H2SO4, and H3PO4 | Purushothaman et al. [13] |
| Compressive strength | Concrete with RCA treated with HCl, H2SO4, and HNO3 | Saravanakumar et al. [14] |
| Compressive strength, tensile strength, and modulus of elasticity (E) | Concrete with RCA treated with HCl, H2SO4, and H3PO4 | Tam et al. [15] |
| Compressive strength, tensile strength, and modulus of elasticity (E) | Concrete with RCA treated with HCl and H2SO4 | Wang et al. [20] |
| Compressive strength | Concrete with RCA treated with HNO3 | Pandurangan et al. [12] |
| Compressive strength, chloride ion penetrability, and carbonation resistance | Concrete with RCA treated with HCl and Na2SO4 | Kim et al. [11] |
| Compressive strength, flexural strength, and modulus of elasticity (E) | Mortar with RCA treated with HCl and H2SO4 | Kim et al. [60] |
| Workability of concrete | Concrete with RCA treated with HCl and HNO3 | Butler et al. [57] |
| Molarity (M) | Time (days) | Size of Aggregate (mm) | Water Absorption (%) | |
|---|---|---|---|---|
| Molarity (M) | 1 | - | - | - |
| p-Value | 1 | - | - | - |
| Time | −0.094 | 1 | - | - |
| p-Value | 0.633 | 1 | - | - |
| Size of aggregate (mm) | 0.077 | −0.007 | 1 | - |
| p-Value | 0.708 | 0.975 | 1 | - |
| Water absorption (%) | −0.140 | 0.235 | −0.211 | 1 |
| p-Value | 0.478 | 0.230 | 0.300 | 1 |
| Mortar loss (%) | 0.400 | −0.170 | −0.368 | −0.632 |
| p-Value | 0.050 | 0.415 | 0.077 | 0.000 |
| Molarity (M) | Time (days) | Water Absorption (%) | |
|---|---|---|---|
| Molarity (M) | 1 | - | - |
| p-Value | 1 | - | - |
| Time | −0.300 | 1 | - |
| p-Value | 0.022 | 1 | - |
| Water Absorption (%) | 0.055 | 0.135 | 1 |
| p-Value | 0.889 | 0.730 | 1 |
| Mortar loss (%) | 0.563 | −0.170 | −0.992 |
| p-Value | 0.000 | 0.015 | 0.000 |
| Aggregate Class | A | B | C | D | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| I | II | III | I | II | III | I | II | III | ||
| Maximum water absorption (%) | 1.5 | 2.5 | 3.5 | 5 | 6.5 | 8.5 | 10.5 | 13 | 15 | No limit |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Forero, J.A.; Brito, J.d.; Evangelista, L.; Pereira, C. Improvement of the Quality of Recycled Concrete Aggregate Subjected to Chemical Treatments: A Review. Materials 2022, 15, 2740. https://doi.org/10.3390/ma15082740
Forero JA, Brito Jd, Evangelista L, Pereira C. Improvement of the Quality of Recycled Concrete Aggregate Subjected to Chemical Treatments: A Review. Materials. 2022; 15(8):2740. https://doi.org/10.3390/ma15082740
Chicago/Turabian StyleForero, Javier A., Jorge de Brito, Luís Evangelista, and Cláudio Pereira. 2022. "Improvement of the Quality of Recycled Concrete Aggregate Subjected to Chemical Treatments: A Review" Materials 15, no. 8: 2740. https://doi.org/10.3390/ma15082740
APA StyleForero, J. A., Brito, J. d., Evangelista, L., & Pereira, C. (2022). Improvement of the Quality of Recycled Concrete Aggregate Subjected to Chemical Treatments: A Review. Materials, 15(8), 2740. https://doi.org/10.3390/ma15082740








