Abstract
The present study aimed to perform a systematic critical appraisal of the methodological quality of systematic reviews (SRs) on the effect of autologous platelet concentrates (APCs) in the treatment of periodontal intraosseous defects and to provide a synthesis of the main clinical findings available. An electronic and hand search was performed up to February 2020; 14 systematic reviews of randomized controlled trials (RCTs), of which 11 were meta-analyses, were included. Only one SR fully satisfied all 11 items of the AMSTAR (“A Measurement Tool to Assess Systematic Reviews”) checklist for methodological quality evaluation, 3 SRs were classified of high quality, 8 of medium quality, and 2 of low quality. There is some evidence on the beneficial additive effect of APCs in the surgical treatment of intraosseous defects when used alone or in combination with bone grafts. APCs did not show any advantage when used together with guided tissue regeneration (GTR) or enamel matrix derivative (EMD). Undertaking SRs which adhere to rigorous standards and protocols is strongly recommended. There are increasing data on the positive adjunctive effect of APCs in the surgical treatment of intraosseous defects but, due to the heterogeneity of the available primary studies, the quality of evidence remains rather low and further long-term well-designed RCTs are encouraged.
1. Introduction
Autologous platelet concentrates (APCs) have been applied since ’90 [1,2,3], as an autologous source of growth and differentiating factors to enhance the healing and regeneration of soft and hard tissues in different fields of oral surgery, including the treatment of periodontal intraosseous defects. APCs include different preparations, each with specific characteristics, that have been used in similar clinical applications. Platelet-rich plasma (PRP) and plasma rich in growth factors (PRGF) are considered as a “first generation” platelet concentrates. PRP was the first APC proposed in oral surgery [1,2], and is obtained by double centrifugation. Conversely, PRGF, first introduced by Anitua in 1999 [3], requires single centrifugation and, with respect to PRP, does not contain leukocytes and requires a smaller blood volume. Both PRP and PRGF need anticoagulants before centrifugation, and heterologous activators to trigger polymerization, which occurs rapidly, but also produces a rapid release of a large amount of growth factors, which tends to decrease considerably within a few days. Platelet-rich fibrin (PRF) is considered as a “second-generation” APC, obtained from 100% autologous sources. Its preparation requires just a single centrifugation stage in which polymerization occurs naturally, without the need for activators. In addition, PRF is characterized by a strong fibrous structure consisting of a three-dimensional fibrin scaffold and shows a sustained release of growth factors for several days [4,5,6]. Based on their leukocyte and fibrin content, APCs have been classified into four categories: P-PRP (pure platelet-rich plasma, no leukocytes, includes PRGF), L-PRP (leukocyte and platelet-rich plasma), P-PRF (pure platelet-rich fibrin, no leukocytes) and L-PRF (leukocyte and platelet-rich fibrin) [6].
The efficacy of platelet concentrates in periodontal regeneration is matter of debate in literature. In the last few years, several systematic reviews, with or without meta-analysis, examined the effect of APCs in the treatment of intraosseous periodontal defects, with heterogeneous findings.
Some authors considered exclusively PRP, exclusively PRF, or both. Some authors focused on the use of the APC alone, others included also bone grafts and barrier membranes or enamel matrix derivative (EMD). In some cases, different periodontal surgical procedures were included, on both soft and hard tissues, in others, the authors specifically focused on the intraosseous defects. Some authors reported beneficial effects of APCs in terms of clinical attachment level (CAL) gain and probing depth (PD) reduction, whereas others limited this effect only to the intraosseous defect fill.
The available reviews on the use of APCs in periodontal surgery were very heterogeneous and when the effects of treatment are not clear, it is difficult to provide clinical indications to practitioners.
Systematic reviews (SRs) of randomized controlled trials are considered the best source of evidence to support clinical decisions on interventions [7,8,9]. The way a systematic review is conducted can change the findings and recommendations. A critical assessment of available SRs may be essential to identify possible causes of heterogeneity and methodological problems of both reviews and primary studies, in order to guide future research to address specific research questions. The quality of SR can be critically and reproducibly assessed using specific grading instruments [10,11]. “A Measurement Tool to Assess Systematic Reviews” checklist (AMSTAR), for instance, was specifically developed for grading the quality of the reviews [12]. Only a few studies, to date, have been published in the periodontal field on the assessment of SRs’ quality using these established guidelines.
The aim of this meta-review, therefore, was to perform a systematic critical appraisal of the methodological quality of systematic reviews on the effect of autologous platelet concentrates in the treatment of periodontal intraosseous defects, and to provide a synthesis of the main clinical findings and recommendations deriving from the examined SRs.
2. Materials and Methods
2.1. Research Question
This critical appraisal of SRs was conducted and is reported, based on the Preferred Reporting Items for Systematic Reviews (PRISMA) statement (www.prismastatement.org) [13]. The concept of the study was registered in the PROSPERO International Prospective Register of Systematic Reviews (CRD42020178492). The research question addressed was the following: have the SRs, about the effect of APCs on the regeneration of periodontal intraosseous defects, been undertaken following a high methodological quality?
2.2. Literature Search
An extensive literature search up to March 2020 was conducted in the MEDLINE Database (via PubMed, and Books), Embase, and the Cochrane Database of Systematic Reviews (CDSR) using the search strategy depicted in Table 1. A hand search was also conducted on the major international journal of periodontics. Grey literature (including documents not controlled by commercial publishing organizations, such as internal reports, working papers, newsletters [14]) was also searched (https://www.greylit.org/; http://www.opengrey.eu/). The reference lists of all original research and review articles identified to be relevant to the subject were scanned for possible additional studies.

Table 1.
Literature search strategy.
2.3. Inclusion and Exclusion Criteria
The inclusion criteria set were as follows:
- SRs and meta-analyses of either randomized controlled trials or controlled clinical trials on the effect of APCs on the treatment of periodontal intraosseous defects;
- SRs that evaluated the effect of any type of APC, either alone or in conjunction with other bio-materials or procedures, compared with a non-APC control.
The exclusion criteria were as follows:
- Narrative reviews;
- SRs including trials on non-intraosseous periodontal defects (or in which such data could not be extracted);
- SRs of in vitro or animal studies.
Only articles in English or Italian were included. No publication date restriction was applied.
2.4. Review Selection Process
Two reviewers (R.R. and L.L.) independently scanned the literature to identify the eligible articles, and in case of disagreements on the selection process, a consensus was reached through discussion. In the first stage, the titles and abstracts of potential papers researched were assessed, and papers that did not meet the inclusion criteria were discarded. In the second stage, the full text of selected papers was assessed and texts that did not meet the inclusion criteria were excluded with reason.
2.5. Data Extraction
Two independent reviewers (A.G. and G.C.) extracted data from the selected studies. Only data directly related to the regeneration of periodontal intraosseous defects were retrieved. Data were obtained exclusively from the meta-analyses (effect size) and not from the primary study reports. Disagreement was solved by discussion between the 2 authors to reach a consensus. For necessary missing data, the authors of the studies were contacted.
2.6. Methodological Assessment of the Systematic Reviews (SRs)
The methodological quality of the SRs included was assessed using the AMSTAR checklist. The methodological assessment was made by two reviewers (A.G. and G.C.) independently, after appropriate calibration. Any disagreement was solved by discussion. AMSTAR is a validated checklist comprising 11 items addressing important aspects of an SR. This tool addresses specific criteria when conducting systematic reviews, e.g., search strategy, inclusion and exclusion criteria, assessment of the methodological quality of trials included in the review. Each of the checklist items was scored with “2” (the assessed criterion was explicitly met in the SR), “1” (the criterion was not completely met), “0” (the criterion was not met), “CA” (cannot answer, the item is relevant but not described by the authors) or “NA” (not applicable, the item is not relevant, e.g., meta-analysis was not possible or was not attempted by the authors). The sum of all scores gives a total AMSTAR score. For each review, the total score could range from 0 (none of the criteria met) to 22 (all the 11 criteria met). For each item, the total score could range from 0 (none of the reviews satisfied the question) to 2x (number of included reviews). There is no guideline to classify the studies based on AMSTAR score so it has been suggested that a final score ranging from 15 to 22 corresponds to a high-quality SR; 8–14 corresponds to a medium-quality SR; 7 or less corresponds to a low-quality SR.
3. Results
3.1. Study Selection
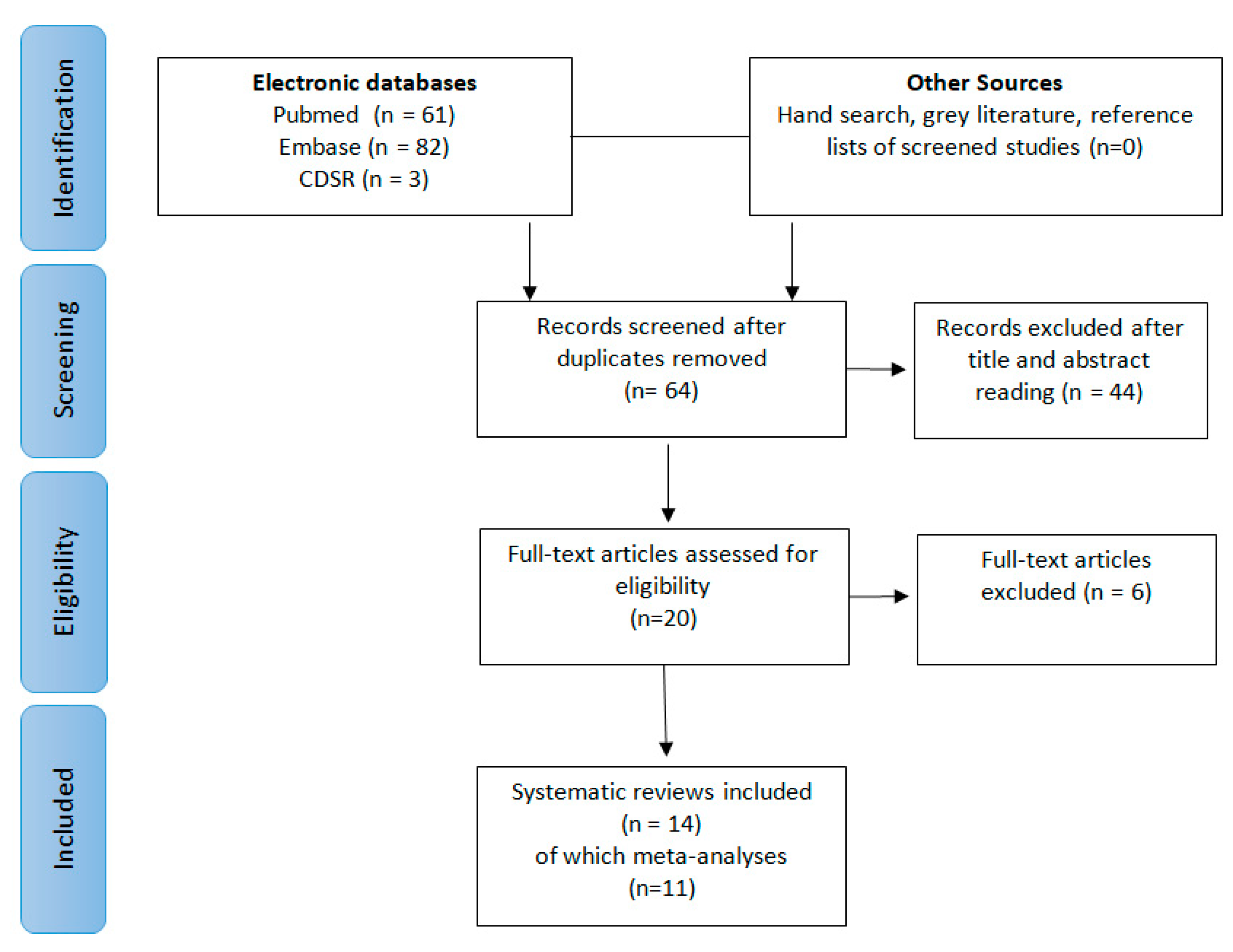
The search in the electronic databases initially generated 64 potential papers. The other sources generated no additional paper. After duplicate removal and abstract assessment, 20 SRs were selected. Six articles were excluded after full-text reading because not systematically conducted [15], or because they pooled together different types of periodontal defects [16,17,18,19,20] (Table 2). Finally, 14 SR, 11 of which MA, were included for this critical assessment. The literature search process is depicted in Figure 1.

Table 2.
Excluded articles with reason of exclusion.
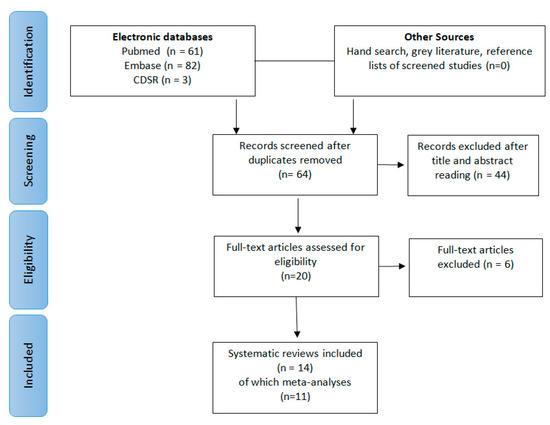
Figure 1.
Preferred Reporting Items for Systematic Reviews (PRISMA) flow diagram of the study selection process.
3.2. Characteristics of the SRs Included
Table 3 and Table 4 describes in detail the features of the SRs included. Four SR included only studies on PRP [21,22,23,24] (one of them included a study on “platelet pellet” that the authors considered as a PRP preparation) [21], five on PRF only [25,26,27,28,29] and five on platelet concentrates irrespective of their type (PRP/PRF and, in one case, PRP/PRF/PRGF) [22,30,31,32,33]. All the SRs included exclusively randomized controlled trials (RCT) on the use of APCs in the treatment of intraosseous defects. Four of them included also studies on other surgical applications of APCs (e.g., furcation defects, periodontal plastic surgery, alveolar socket preservation sinus elevation, etc.) [23,27,28,30], but in these SRs it was possible to clearly extract data about intraosseous defects. In all SRs the only difference between control and test groups was the presence of the APC. Three SRs included RCTs on APC alone as intervention group [25,27,29,33]. Six SRs included studies on both APCs used alone and added to other biomaterials [22,26,28,30,31,34]. Three SRs [21,23,24] included only studies in which PRP was added to biomaterials. In one SR [32] the studies included were exclusively on APCs (PRP and PRF) added to demineralized freeze-dried bone allograft (DFDBA). The follow-up of the studies included in the selected SRs ranged from 1 month in one study [26] to 5 years in another study [34], whereas most of the SRs included 6- or 9- to 12-month follow-up studies. All 11 MA were done on CAL gain, 8 on PD reduction [22,24,25,27,29,32,33,34], 5 on gingival marginal level change [25,29,32,33,34], 3 on intraosseous defect reduction [25,29,33], 4 on bone fill [22,27,29,32], 1 on bone reduction [32]. 4 MA performed sub-group analyses basing on the type of APC used (PRP/PRF) [32], the adjunct of a membrane (guided tissue regeneration (GTR)/no-GTR) [23,24,30], the experimental design (parallel groups/split-mouth design) [22,23,24,30], the follow-up period tested (3–6 months/9–12 months) [22]. Furthermore, one SR [22] performed separate meta-analyses based on the biomaterial or technique adjunctive to the APC (GTR/bone grafting (BG)/EMD/none), and another performed univariate meta-regression analyses of potential sources of heterogeneity (GTR, design, type of control) [24].

Table 3.
Characteristics of the included studies: search details.

Table 4.
Characteristics of the included studies: main findings.
The overall effect size of the different outcomes is reported in Table 4. A direct comparison among the different SRs limited to CAL gain overall measurements is reported in Table 5.

Table 5.
Overall effect size for CAL gain.
3.3. Methodological Quality
The SRs were scored from 7 to 22 leading to a mean AMSTAR score of 12.6 ± 4.2 (standard deviation) corresponding to overall medium quality (Table 6). Only one SR fully satisfied all 11 items reaching an AMSTAR score of 22 [22]. Three SRs [21,31,34] were classified of high quality. All the others were of medium quality, excepting two [26,28] classified as low quality SRs. Only one review [22] completely met the first item (“a priori” design).

Table 6.
Quality assessment of the systematic reviews included following the AMSTAR (“A Measurement Tool to Assess Systematic Reviews”) checklist.
Most of the SRs (12/14) were conducted by two reviewers independently with consensus procedures for disagreements (item #2); 9 of 14 SRs used at least two electronic sources and one supplementary source (item #3), whereas only five explicitly search for “grey literature” (item #4).
Most of the SRs (8/14) clearly reported a list of the excluded studies (item #5) and all SRs reported the main characteristics of included studies (item #6), although only 5 completely met the criterion.
All SRs, excepting one [28], evaluated and documented the methodological quality of the included studies (item #7), although five SRs did not completely meet the criterion (score 1). In six SRs the results of the methodological rigor and scientific quality were considered in the analysis and the conclusions of the reviews and explicitly stated in formulating recommendations (item #8).
Six of the 10 MA used a test to ensure whether the studies were combinable and to assess their homogeneity (i.e., chi-squared test for homogeneity, or I2) and considered such aspects for methodological considerations (e.g., if heterogeneity exists, a random effects model should be used) (item #9). Similarly, in eight MA the publication bias was assessed by statistical test (e.g., Egger regression test) and/or graphical aids (e.g., funnel plot) (item #9), although two of them did not completely meet the criterion.
Finally, all the articles reported about potential sources of conflict of interest for the SR itself, but only two of them duly acknowledged the source of funding or support or the conflict of interest for each of the included studies.
3.4. Summary of Findings
Based on the results obtained from the systematic reviews reaching the highest quality scores, it can be concluded that there is some evidence for the beneficial additive effect of APCs in the surgical treatment of intraosseous defects when used alone or in combination with bone grafts, although the quality of evidence for such findings is low. By contrast, there is no evidence of any advantage when APCs are used together with GTR or EMD.
4. Discussion
With the widespread availability of scientific information, it may be difficult for clinicians to correctly interpret results and find evidence about clinical questions to guide clinical practice. This also applies to systematic reviews, the number of which has exponentially increased in recent years [35]. Systematic reviews of RCTs stay at the top of the evidence pyramid of scientific literature, but they need to be conducted following a very precise and well-defined methodology in order to be reliable and lead to consensus, recommendations, and clinical practice guidelines. For this reason, there is a need for a systematic critical appraisal of the methodological quality of systematic reviews on the basis of specific evaluation tools such as the AMSTAR [12].
In particular, the present AMSTAR-based assessment of SRs on the effect of APCs in the treatment of periodontal intraosseous defects revealed that some methodological aspects of the reviews could be improved. Out of 14 reviews assessed, in fact, only five [21,22,27,31,34] completely met (score 2) more than 50% of the AMSTAR criteria. Following this evaluation tool, several limitations of the SRs assessed were evidenced.
For example, almost all the SRs included did not fully meet an “a priori” design criterion. The execution of an SR adhering to acknowledged standards and guidelines, such as the PRISMA statement or the Methodological Expectations of Cochrane Intervention Reviews (MECIR) manual, is always strongly recommended. Only 5 of the 14 SRs included [22,24,27,29,33] referred to one of these guidelines. The use of a specialized framework, such as the PICO model [36] allows practitioners to formulate a well-focused research question, facilitating the literature search process to identify relevant evidence. PICO stands for Patient Problem (or Population), Intervention, Comparison (or Control), and Outcome, and its use is warmly recommended to correctly carry out an SR. Among the analyzed SRs, only 5 reported a PICO-based research question [22,27,28,31,34]. There are several public databases in which SRs can be registered, such as the International Prospective Register of Systematic Reviews of the National Institute for Health Research (PROSPERO), or the Cochrane Collaboration. Only two of the included SRs [22,33] referred to a registered protocol, and for one of them [33] the authors referred to ongoing submission. Systematic reviews should be registered at the protocol stage to enable comparison with already registered protocols, avoiding duplication, and to allow a post hoc comparison between the final publication and the planned protocol.
Most SRs performed a comprehensive literature search strategy with the use of supplementary sources of articles, such as a manual search in the main journals of periodontics, with analysis of the reference lists of the screened articles. However, extensive use of language limits, and limited use of grey literature sources, including the registers of clinical trials (e.g., http://www.clinicaltrials.gov), was found, which can imply a risk of publication bias [37,38,39].
Furthermore, only in five [21,22,24,28,32,34] of the included SRs a reproducible string of key-terms with Boolean operators was provided and only in two of them, specific strings for each database used were reported [22,27].
Finally, although most of the reviews make available a list of the studies excluded, it was limited only to the last phase of the review process, which is the full-text reading.
All these aspects, strongly limit the reproducibility of the review process in all its steps.
A complete report of the main characteristics of the included studies is of paramount importance to provide readers with an accurate report of the primary studies. All the analyzed SRs reported tables with such characteristics, however in more than half of SRs (those with score 1 of item #6 in Table 5) important details about participants (number, age, gender), operative protocol (preparation protocols of APCs) or results (mean values and standard deviation of baseline and follow-up values, and relative changes) were lacking.
Another aspect of central importance in carrying out an SR is the quality assessment of primary studies and its use to discuss results and make recommendations. It is usually performed using specific quality scoring tools or checklists, such as the Jadad scale [40], or the risk of bias tool of the Cochrane Collaboration [41]. All the analyzed SRs performed a methodological assessment of the primary trials. However, in four of them, the quality score was not reported for each study, and in one case [26], the authors used a minimum quality score as an “a priori” inclusion criterion, instead of performing such evaluation on all the included studies. Furthermore, only in one case [22] was the quality of evidence ranked based on specifically developed tools (i.e., Grading of Recommendations Assessment, Development and Evaluation, GRADE) [42]. Such aspects are of paramount importance to correctly interpret and weigh the validity of the results of each study [43], although the evaluation of each quality scoring tool is subjective. Despite calibration procedures and multiple assessment protocols, indeed, some discrepancies among different SRs can be found, which may influence the final interpretation of the results reported by the selected studies. Also when the scientific quality was correctly evaluated, we found that, in some cases, such evaluation was not explicitly used to analyze the results and to formulate scientific recommendations. In six of the analyzed SRs (score 1, item #8), for instance, the limits of the primary studies analyzed, and the relative recommendations referred to aspects not included in the quality assessment tool used. These elements regard statistical aspects (e.g., sample size calculation, appropriate statistical methods, analysis of confounding variables, etc.), or protocol aspects (e.g., appropriate follow-up duration, choice of the experimental groups, selection criteria, etc.). Probably, their evaluation could allow a more comprehensive “a priori” assessment of SRs’ scientific quality. Anyway, most of the SRs recommended performing better-designed RCT, with particular attention to methodological aspects such as randomization and allocation concealment.
Eleven of the 14 SRs undertook a MA of the main outcome measurements. MA is an element of paramount importance in an SR, allowing the treatment effect and its precision to be quantified. Nevertheless, if some methodological inaccuracy exists (e.g., heterogeneous trials pooled together), results and conclusions may be misleading. In some of the SRs analyzed, there were issues about the way the statistical aspects of the meta-analysis were described and presented. In particular, the evaluation of heterogeneity, as well as publication bias among studies, was not extensively reported and/or commented on [23,25,30,31,34]. In other cases, some discrepancy was found between figures and text [25], some data were missing, i.e., effect size [34] or significance values [25,32]. In one SR [34] the meta-analysis included only one study [44], with an inappropriate control group.
Interestingly, some SRs have performed sub-group [22,23,24,30,32] and meta-regression [24] analyses. However, the few primary studies available in some cases may considerably limit these attempts.
Finally, the reporting of potential sources of conflict of interest for each of the included studies remains one of the less met items in the quality assessment of the analyzed SRs.
Looking at the clinical findings from the SRs included, their conclusions are quite homogeneous. The evidence supporting the use of APCs in the treatment of periodontal intraosseous defects and its quality has been growing in the last few years but they are still limited.
As shown in Table 5, the standardized mean difference (SDM) of CAL gain for APCs alone (PRF, PRP, PRGF) ranged from 0.39 (95% confidence interval (CI), 0.35, 0.43, p < 0.00001) ([33] 16 studies), to 1.47 mm, 95% CI 1.11 to 1.82 mm; p < 0.00001 ([22], 12 studies). When added to a bone graft it was 0.72 mm, 95% CI 0.43 to 1.00 mm; p < 0.00001 ([22], 12 studies). A not significant overall effect, on the contrary, was reported for APCs added to GTR or to EMD [22,23,24,30,31]. Probably, as suggested by the authors, the reason can be found in the significantly high contribution of the membrane or EMD in the test group that may overwhelm and mask the positive influence of APC on the healing of the periodontal wound.
Overall, the authors agree on the beneficial effect of APC alone (mainly PRF) or in combination with different types of bone grafts (mainly PRP), but the clinical significance of the obtained results has been described as of limited value. This aspect was highlighted, in particular, by one SR [33], in which the authors followed a different approach for data analysis and interpretation, evaluating the “actual quantitative mean gains” (AQMG) for the main outcomes, intending to give more insight into clinical significance of conducted studies and a more direct evidential reference to the clinicians. Also calculating AQMG, the adjunctive effect of APCs in terms of clinical outcomes was considered negligible, because it was lower than a visible and perceptible gain value, which should be at least 2 mm [45].
Data are few and heterogeneous to speculate on the superiority of one type of APC compared to another (as well as on the superiority of one type of added graft compared to another). A direct comparison between different APCs was only rarely undertaken [46]. Furthermore, also indirect comparisons made by subgroups meta-analyses have not been attempted in the examined SRs, due to the low number and heterogeneity of available primary studies. Whereas, indeed, a consistent number of studies on PRF alone have been published in the last years, only one study on PRP alone was included in the examined SRs. Similarly, whereas most of studies on PRP included also an adjunctive graft, only very few studies on PRF + grafts are available to date. The reason for such a discrepancy might be the fluid nature of PRP, that limits its mechanical support when used alone, as opposed to the strong mechanical consistency of PRF, that, in turn, can limit its use in combination with grafts.
Although the similar clinical efficacy of PRF, PRP or PRGF in the treatment of intraosseous defects is shown in the few available comparative studies [46,47], some unique biological properties and advantageous procedural aspects of PRF compared to the other APCs have been suggested. PRF, indeed, shows gradual and prolonged release of growth factors, as well as antibacterial and anti-inflammatory effect due to the content of leucocytes [48,49,50,51,52]. Furthermore, PRF requires a single centrifugation, does not need any additive, and is characterized by an enhanced handling due to its more robust and long-lasting fibrin mesh [48,49,50].
Several other variables could affect the results of primary studies. For instance, different protocols exist for producing the same type of platelet concentrate, and they may affect the final composition and the biological properties. Lower centrifugation speed and time may result in higher leukocyte concentration, more even distribution of leukocytes throughout the PRF scaffold, and increased release of growth factors [4,51,53]. The clinical results, moreover, could be influenced by disease characteristics (grading of periodontitis), defect characteristics (number of walls, depth, width), surgical technique (modified Widman flap, Kirkland flap, papilla preservation flaps, single flap approaches).
RCTs analysing all these variables are still lacking and need to be specifically designed and carried out to achieve useful clinical indications. Also the follow-up period must be considered. It has been shown that the bone fill progressively goes on for a long time, reaching the highest level over 36 months [54]. For this reason, a more precise assessment of bone healing and regeneration would require longer follow-up studies.
Another systematic appraisal of SRs on the use of APCs for the treatment of periodontal intraosseous defects was published a few years ago [55], however some differences exist with the present overview. That study was not exclusively focused on intraosseous defects treatment but also on the furcations and gingival recessions treatment. It included 9 SRs on intraosseous defects (up to March 2016), whereas 14 SRs were identified in the present one (up to February 2020). Finally, some of the SRs included in that work were excluded in the present one after full-text reading [16,17,18,56], due to different selection criteria.
5. Conclusions
The methodological quality of the examined SRs was heterogeneous. The execution of SRs which adhere to acknowledged standards and guidelines, i.e., PRISMA or MECIR, and analyze the quality of evidence available in more depth employing specific instruments, e.g., GRADE, are strongly recommended
The results obtained from the systematic reviews reaching the highest quality scores suggest that the evidence on the positive adjunctive effect of APCs in the regeneration of intraosseous defects, alone (mainly PRF) or in combination with bone grafts (mainly PRP), has been increasing in recent years. Conversely, APCs did not show any advantage when used together with GTR or EMD. Due to paucity and heterogeneity of the available primary studies, it is not possible to speculate on the superiority of one type of APC or adjunctive graft compared to the others. The quality of evidence for such findings is still rather low and further long-term, multicentre well-designed RCTs are needed to validate these therapeutic approaches and provide evidence-based clinical recommendations.
Author Contributions
Conceptualization and methodology, A.G., G.C., M.D.F. and M.A.; screening R.R. and L.L.; data analysis A.G. and G.C.; writing—original draft preparation, A.G. and G.C.; writing review and editing, A.G., G.C., R.R., L.L., M.A. and M.D.F.; supervision, M.D.F. and M.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors want to thank Prof. Luigi Guida from the University of Campania “Luigi Vanvitelli”, Naples, Italy, for the precious help in the conceptualization of the review protocol.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Marx, R.E.; Carlson, E.R.; Eichstaedt, R.M.; Schimmele, S.R.; Strauss, J.E.; Georgeff, K.R. Platelet-rich plasma: Growth factor enhancement for bone grafts. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 1998, 85, 638–646. [Google Scholar] [CrossRef]
- Whitman, D.H.; Berry, R.L.; Green, D.M. Platelet gel: An autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. J. Oral Maxillofac. Surg. 1997, 55, 1294–1299. [Google Scholar] [CrossRef]
- Anitua, E. Plasma rich in growth factors: Preliminary results of use in the preparation of future sites for implants. Int. J. Oral Maxillofac. Implant. 1999, 14, 529–535. [Google Scholar]
- Kobayashi, E.; Flückiger, L.; Fujioka-Kobayashi, M.; Sawada, K.; Sculean, A.; Schaller, B.; Miron, R.J. Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin. Oral Investig. 2016, 20, 2353–2360. [Google Scholar] [CrossRef] [PubMed]
- Tsay, R.C.; Vo, J.; Burke, A.; Eisig, S.B.; Lu, H.H.; Landesberg, R. Differential growth factor retention by platelet-rich plasma composites. J. Oral Maxillofac. Surg. 2005, 63, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Dohan Ehrenfest, D.M.; Rasmusson, L.; Albrektsson, T. Classification of platelet concentrates: From pure platelet-rich plasma (P-PRP) to leucocyte- and platelet-rich fibrin (L-PRF). Trends Biotechnol. 2009, 27, 158–167. [Google Scholar] [CrossRef]
- Lau, J.; Ioannidis, J.P.A.; Schmid, C.H. Summing up evidence: One answer is not always enough. Lancet 1998, 351, 123–127. [Google Scholar] [CrossRef]
- Guyatt, G.H.; Haynes, R.B.; Jaeschke, R.Z.; Cook, D.J.; Green, L.; Naylor, C.D.; Wilson, M.C.; Richardson, W.S. Users’ guides to the medical literature: XXV. Evidence-based medicine: Principles for applying the users’ guides to patient care. J. Am. Med. Assoc. 2000, 284, 1290–1296. [Google Scholar] [CrossRef]
- Evans, D. Hierarchy of evidence: A framework for ranking evidence evaluating healthcare interventions. J. Clin. Nurs. 2003, 12, 77–84. [Google Scholar] [CrossRef]
- Moher, D.; Jadad, A.R.; Nichol, G.; Penman, M.; Tugwell, P.; Walsh, S. Assessing the quality of randomized controlled trials: An annotated bibliography of scales and checklists. Control. Clin. Trials 1995, 16, 62–73. [Google Scholar] [CrossRef]
- Afrashtehfar, K.I.; Del Fabbro, M. Clinical performance of zirconia implants: A meta-review. J. Prosthet. Dent. 2020, 123, 419–426. [Google Scholar] [CrossRef]
- Shea, B.J.; Hamel, C.; Wells, G.A.; Bouter, L.M.; Kristjansson, E.; Grimshaw, J.; Henry, D.A.; Boers, M. AMSTAR is a reliable and valid measurement tool to assess the methodological quality of systematic reviews. J. Clin. Epidemiol. 2009, 62, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Swartz, M.K. The PRISMA statement: A guideline for systematic reviews and meta-analyses. J. Pediatr. Health Care 2011, 25, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.; Hillier-Brown, F.C.; Moore, H.J.; Lake, A.A.; Araujo-Soares, V.; White, M.; Summerbell, C. Searching and synthesising “grey literature” and “grey information” in public health: Critical reflections on three case studies. Syst. Rev. 2016, 5, 164. [Google Scholar] [CrossRef] [PubMed]
- Rock, L. Potential of platelet rich fibrin in regenerative periodontal therapy: Literature review. Can. J. Dent. Hyg. 2013, 47, 33–37. [Google Scholar]
- Plachokova, A.S.; Nikolidakis, D.; Mulder, J.; Jansen, J.A.; Creugers, N.H.J. Effect of platelet-rich plasma on bone regeneration in dentistry: A systematic review. Clin. Oral Implant. Res. 2008, 19, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Zapata, M.J.; Martí-Carvajal, A.; Solà, I.; Bolibar, I.; Ángel Expósito, J.; Rodriguez, L.; García, J. Efficacy and safety of the use of autologous plasma rich in platelets for tissue regeneration: A systematic review. Transfusion 2009, 49, 44–56. [Google Scholar] [CrossRef]
- Roselló-Camps, À.; Monje, A.; Lin, G.H.; Khoshkam, V.; Chávez-Gatty, M.; Wang, H.L.; Gargallo-Albiol, J.; Hernandez-Alfaro, F. Platelet-rich plasma for periodontal regeneration in the treatment of intrabony defects: A meta-analysis on prospective clinical trials. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 120, 562–574. [Google Scholar] [CrossRef]
- Verma, U.P.; Yadav, R.K.; Dixit, M.; Gupta, A. Platelet-rich fibrin: A paradigm in periodontal therapy-A systematic review. J. Int. Soc. Prev. Community Dent. 2017, 7, 227–233. [Google Scholar]
- Franchini, M.; Cruciani, M.; Mengoli, C.; Masiello, F.; Marano, G.; D’Aloja, E.; Dell’Aringa, C.; Pati, I.; Veropalumbo, E.; Pupella, S.; et al. The use of platelet-rich plasma in oral surgery: A systematic review and meta-analysis. Blood Transfus. 2019, 17, 357–367. [Google Scholar]
- Kotsovilis, S.; Markou, N.; Pepelassi, E.; Nikolidakis, D. The adjunctive use of platelet-rich plasma in the therapy of periodontal intraosseous defects: A systematic review. J. Periodontal Res. 2010, 45, 428–443. [Google Scholar] [CrossRef] [PubMed]
- Del Fabbro, M.; Karanxha, L.; Panda, S.; Bucchi, C.; Nadathur Doraiswamy, J.; Sankari, M.; Ramamoorthi, S.; Varghese, S.; Taschieri, S. Autologous platelet concentrates for treating periodontal infrabony defects. Cochrane Database Syst. Rev. 2018, 2018, CD011423. [Google Scholar] [CrossRef] [PubMed]
- Del Fabbro, M.; Bortolin, M.; Taschieri, S.; Weinstein, R. Is Platelet Concentrate Advantageous for the Surgical Treatment of Periodontal Diseases? A Systematic Review and Meta-Analysis. J. Periodontol. 2011, 82, 1100–1111. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Yuan, J.; Aisaiti, A.; Liu, Y.; Zhao, J. The effect of platelet-rich plasma on clinical outcomes of the surgical treatment of periodontal intrabony defects: A systematic review and meta-analysis. BMC Oral Health 2016, 16, 71. [Google Scholar] [CrossRef]
- Shah, M.; Deshpande, N.; Bharwani, A.; Nadig, P.; Doshi, V.; Dave, D. Effectiveness of autologous platelet-rich fibrin in the treatment of intra-bony defects: A systematic review and meta-analysis. J. Indian Soc. Periodontol. 2014, 18, 698–704. [Google Scholar]
- Najeeb, S.; Khurshid, Z.; Agwan, M.A.S.; Ansari, S.A.; Zafar, M.S.; Matinlinna, J.P. Regenerative Potential of Platelet Rich Fibrin (PRF) for Curing Intrabony Periodontal Defects: A Systematic Review of Clinical Studies. Tissue Eng. Regen. Med. 2017, 14, 735–742. [Google Scholar] [CrossRef]
- Castro, A.B.; Meschi, N.; Temmerman, A.; Pinto, N.; Lambrechts, P.; Teughels, W.; Quirynen, M. Regenerative potential of leucocyte- and platelet-rich fibrin. Part A: Intra-bony defects, furcation defects and periodontal plastic surgery. A systematic review and meta-analysis. J. Clin. Periodontol. 2017, 44, 67–82. [Google Scholar] [CrossRef]
- Miron, R.J.; Zucchelli, G.; Pikos, M.A.; Salama, M.; Lee, S.; Guillemette, V.; Fujioka-Kobayashi, M.; Bishara, M.; Zhang, Y.; Wang, H.L.; et al. Use of platelet-rich fibrin in regenerative dentistry: A systematic review. Clin. Oral Investig. 2017, 21, 1913–1927. [Google Scholar] [CrossRef]
- Li, A.; Yang, H.; Zhang, J.; Chen, S.; Wang, H.; Gao, Y. Additive effectiveness of autologous platelet-rich fibrin in the treatment of intrabony defects: A PRISMA-compliant meta-analysis. Medicine 2019, 98, e14759. [Google Scholar] [CrossRef]
- Del Fabbro, M.; Ceci, C.; Taschieri, S. Systematic review on the effect of platelet concentrates for the surgical treatment of periodontal defects. Dent. Cadmos 2013, 81, 138–145. [Google Scholar]
- Panda, S.; Doraiswamy, J.; Malaiappan, S.; Varghese, S.S.; Del Fabbro, M. Additive effect of autologous platelet concentrates in treatment of intrabony defects: A systematic review and meta-analysis. J. Investig. Clin. Dent. 2016, 7, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Sun, C.; Huang, S.; Wu, X.; Zhao, Y.; Pan, C.; Wang, H.; Liu, J.; Li, Q.; Kou, Y. Efficacy of Adjunctive Bioactive Materials in the Treatment of Periodontal Intrabony Defects: A Systematic Review and Meta-Analysis. Biomed Res. Int. 2018, 2018, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Baghele, O.K.; Kathole, V.; Tuteja, A.K.; Giri, T. Actual quantitative attachment gain secondary to use of autologous platelet concentrates in the treatment of intrabony defects: A meta-analysis. J. Indian Soc. Periodontol. 2019, 23, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Saleem, M.; Pisani, F.; Zahid, F.M.; Georgakopoulos, I.; Pustina-Krasniqi, T.; Xhajanka, E.; Almasri, M. Adjunctive platelet-rich plasma (PRP) in infrabony regenerative treatment: A systematic review and RCT’s meta-analysis. Stem Cells Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Ioannidis, J.P.A. The Mass Production of Redundant, Misleading, and Conflicted Systematic Reviews and Meta-analyses. Milbank Q. 2016, 94, 485–514. [Google Scholar] [CrossRef]
- Schardt, C.; Adams, M.B.; Owens, T.; Keitz, S.; Fontelo, P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med. Inform. Decis. Mak. 2007, 7, 16. [Google Scholar] [CrossRef]
- Egger, M.; Smith, G.D.; Phillips, A.N. Meta-analysis: Principles and procedures. BMJ 1997, 315, 1533–1537. [Google Scholar] [CrossRef]
- Hopewell, S.; McDonald, S.; Clarke, M.; Egger, M. Grey literature in meta-analyses of randomized trials of health care interventions. Cochrane Database Syst. Rev. 2007, 18, MR000010. [Google Scholar] [CrossRef]
- Song, F.; Parekh, S.; Hooper, L.; Loke, Y.K.; Ryder, J.; Sutton, A.J.; Hing, C.; Kwok, C.S.; Pang, C.; Harvey, I. Dissemination and publication of research findings: An updated review of related biases. Health Technol. Assess. (Rockv). 2010, 14, 1–193. [Google Scholar] [CrossRef]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.M.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Sterne, J.A.C. Chapter 8: Assessing risk of bias in included studies. In Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0; 2011; Available online: http://handbook.cochrane.org (accessed on 16 September 2020).
- Guyatt, G.H.; Oxman, A.D.; Vist, G.E.; Kunz, R.; Falck-Ytter, Y.; Alonso-Coello, P.; Schünemann, H.J. GRADE: An emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008, 336, 924–926. [Google Scholar] [CrossRef] [PubMed]
- Moja, L.P.; Telaro, E.; D’Amico, R.; Moschetti, I.; Coe, L.; Liberati, A. Assessment of methodological quality of primary studies by systematic reviews: Results of the metaquality study cross sectional study. Br. Med. J. 2005, 330, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Camargo, P.M.; Lekovic, V.; Weinlaender, M.; Vasilic, N.; Madzarevic, M.; Kenney, E.B. A reentry study on the use of bovine porous bone mineral, GTR, and platelet-rich plasma in the regenerative treatment of intrabony defects in humans. Int. J. Periodontics Restor. Dent. 2005, 25, 49–59. [Google Scholar]
- Addy, M.; Newcombe, R.G. Statistical versus clinical significance in periodontal research and practice. Periodontology 2000 2005, 39, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Pradeep, A.R.; Rao, N.S.; Agarwal, E.; Bajaj, P.; Kumari, M.; Naik, S.B. Comparative Evaluation of Autologous Platelet-Rich Fibrin and Platelet-Rich Plasma in the Treatment of 3-Wall Intrabony Defects in Chronic Periodontitis: A Randomized Controlled Clinical Trial. J. Periodontol. 2012, 83, 1499–1507. [Google Scholar] [CrossRef]
- Gamal, A.Y.; Abdel Ghaffar, K.A.; Alghezwy, O.A. Crevicular Fluid Growth Factors Release Profile Following the Use of Platelet-Rich Fibrin and Plasma Rich Growth Factors in Treating Periodontal Intrabony Defects: A Randomized Clinical Trial. J. Periodontol. 2016, 87, 654–662. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part I: Technological concepts and evolution. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, e37–e44. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part II: Platelet-related biologic features. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, e45–e50. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part III: Leucocyte activation: A new feature for platelet concentrates? Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2006, 101, e51–e55. [Google Scholar] [CrossRef]
- Ghanaati, S.; Booms, P.; Orlowska, A.; Kubesch, A.; Lorenz, J.; Rutkowski, J.; Les, C.; Sader, R.; Kirkpatrick, C.J.; Choukroun, J. Advanced platelet-rich fibrin: A new concept for cell- Based tissue engineering by means of inflammatory cells. J. Oral Implantol. 2014, 40, 679–689. [Google Scholar] [CrossRef]
- Madurantakam, P.; Yoganarasimha, S.; Hasan, F.K. Characterization of Leukocyte-platelet Rich Fibrin, A Novel Biomaterial. J. Vis. Exp. 2015, 103, e53221. [Google Scholar] [CrossRef] [PubMed]
- Choukroun, J.; Ghanaati, S. Reduction of relative centrifugation force within injectable platelet-rich-fibrin (PRF) concentrates advances patients’ own inflammatory cells, platelets and growth factors: The first introduction to the low speed centrifugation concept. Eur. J. Trauma Emerg. Surg. 2018, 44, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Nevins, M.; Kao, R.T.; McGuire, M.K.; McClain, P.K.; Hinrichs, J.E.; McAllister, B.S.; Reddy, M.S.; Nevins, M.L.; Genco, R.J.; Lynch, S.E.; et al. Platelet-Derived Growth Factor Promotes Periodontal Regeneration in Localized Osseous Defects: 36-Month Extension Results From a Randomized, Controlled, Double-Masked Clinical Trial. J. Periodontol. 2013, 84, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Del Fabbro, M.; Lolato, A.; Panda, S.; Corbella, S.; Satpathy, A.; Das, A.C.; Kumar, M.; Taschieri, S. Methodological Quality Assessment of Systematic Reviews on Autologous Platelet Concentrates for the Treatment of Periodontal Defects. J. Evid. Based Dent. Pract. 2017, 17, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, F.; Ferrari, D.; Herten, M.; Mihatovic, I.; Wieland, M.; Sager, M.; Becker, J. Effects of surface hydrophilicity and microtopography on early stages of soft and hard tissue integration at non-submerged titanium implants: An immunohistochemical study in dogs. J. Periodontol. 2007, 78, 2171–2184. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).