Abstract
The worldwide, extraordinary outbreak of coronavirus pandemic (i.e., COVID-19) and other emerging viral expansions have drawn particular interest to the design and development of novel antiviral, and viricidal, agents, with a broad-spectrum of antiviral activity. The current indispensable challenge lies in the development of universal virus repudiation systems that are reusable, and capable of inactivating pathogens, thus reducing risk of infection and transmission. In this review, science-based methods, mechanisms, and procedures, which are implemented in obtaining resultant antiviral coated substrates, used in the destruction of the strains of the different viruses, are reviewed. The constituent antiviral members are classified into a few broad groups, such as polymeric materials, metal ions/metal oxides, and functional nanomaterials, based on the type of materials used at the virus contamination sites. The action mode against enveloped viruses was depicted to vindicate the antiviral mechanism. We also disclose hypothesized strategies for development of a universal and reusable virus deactivation system against the emerging COVID-19. In the surge of the current, alarming scenario of SARS-CoV-2 infections, there is a great necessity for developing highly-innovative antiviral agents to work against the viruses. We hypothesize that some of the antiviral coatings discussed here could exert an inhibitive effect on COVID-19, indicated by the results that the coatings succeeded in obtaining against other enveloped viruses. Consequently, the coatings need to be tested and authenticated, to fabricate a wide range of coated antiviral products such as masks, gowns, surgical drapes, textiles, high-touch surfaces, and other personal protective equipment, aimed at extrication from the COVID-19 pandemic.
1. Introduction
The presence of different microorganisms in nature may sometimes cause a detrimental impact on human health [1]. Specifically, viruses have always been regarded as increasing hazards by impairing health, as human contact with these microbes from the environment can lead to extreme illnesses and other ailments [2]. For example, tropical and subtropical countries survived an outbreak of dengue virus, known to cause the severe form of dengue hemorrhagic fever/dengue shock syndrome (DHS/DSS) [3,4]. Since the emergence of the Spanish flu outbreak (1918), influenza viral pandemics are known to appear within the interval of every 10 to 15 years [5]. Characterized by their variations in pathogenicity, the most virulent type, A influenza viruses (H1N1 and H5N1) (2009), are known to cause serious human pandemics via common transmission from animals to humans and vice versa [6,7]. Lethal varieties of coronavirus, such as severe acute respiratory syndrome-related coronavirus (SARSr-CoV) and middle east respiratory syndrome-related coronavirus (MERS-CoV) are known to cause SARS (2003) and MERS (2014) outbreaks, respectively. These coronavirus related infections were reported in several countries of North America, South America, Europe, and Asia [8,9]. Recently, an Ebola hemorrhagic fever (EHF) (2014) outbreak severely affected the living species of Africa [10,11]. In late December 2019, the emergence of a novel pneumonia drew animated attention around the world. Visualizing the chronological order, the ingenious agent that was responsible for causing the novel pneumonia has been identified as a novel coronavirus (nCoV or SARS-CoV-2) [12,13]. The outbreak of a novel coronavirus disease (COVID-19) has created a devastating challenge to the human health of various sections in the world [14]. It has caused negative social effects and massive economic damage, on a global scale. Coronaviruses contain spherical even-shaped virions, a type of enveloped RNA virus initially causing respiratory unevenness, and further leading to extreme flu [15,16]. An increased concern has arisen in the recent past with respect to a growing number of new, more virulent and pathologic viruses, such as those associated with SARS and, more recently COVID-19 [17,18].
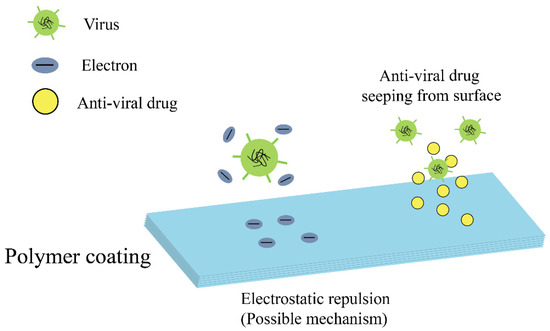
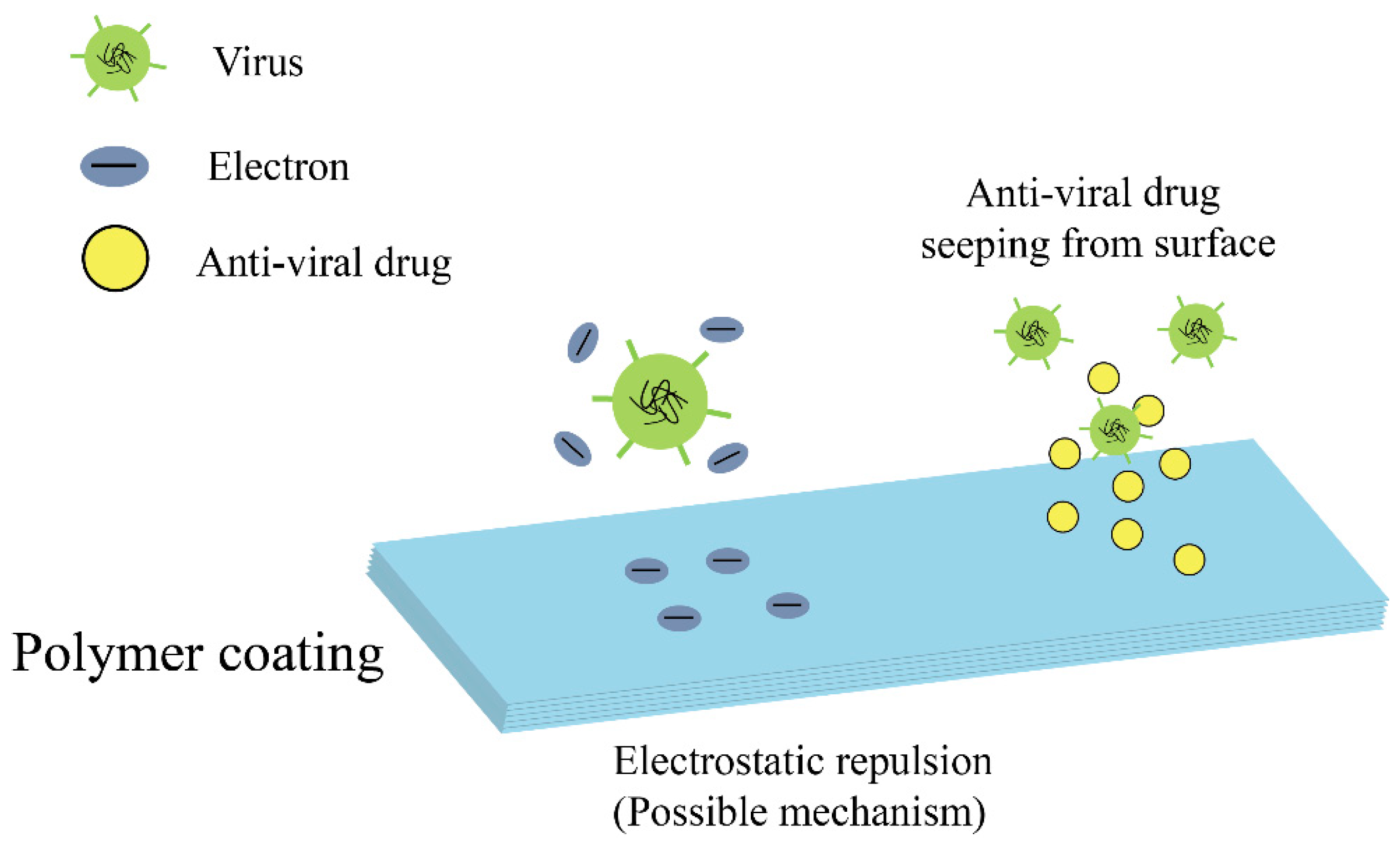
Microorganisms constitute both bacteria and viruses [19]. Bacterial cells and viruses primarily differ from each other in terms of their size and mode of infection. More importantly, viruses reproduce by infecting a host cell and then multiplying in great numbers, causing serious illness, while the bacteria usually restricts its growth in a localized area, causing a local infection by creating an impact on a specific part of the human body [20,21]. Thus, the bacterial infections are easier to target using novel antimicrobials than the viruses. Most of the antimicrobial coatings so far developed and commercialized are antibacterial, but there are very few reports on commercialized antiviral coatings. Hence, it is highly desirable to search for potential antiviral and viricidal elements (materials and coatings) to design personal protective equipment (PPE), hygienic implements, and other devices to fight against the rise of viral pandemics and virus-associated fatal risks [22]. This review visualizes the techniques and methods that are involved in the design and development of different antiviral coatings, aiming to inspire strategies for development of coatings that are supposed to enhance antiviral efficiency, eliciting their potential application in the inhibition of COVID 19 like pandemics.
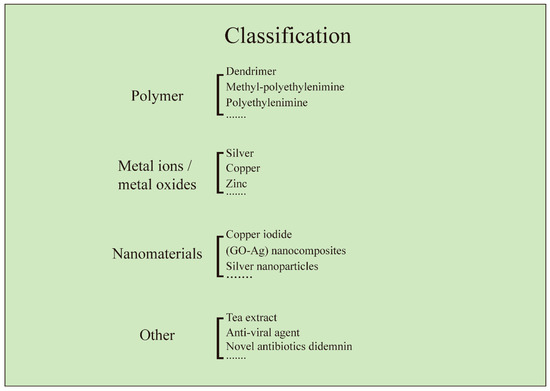
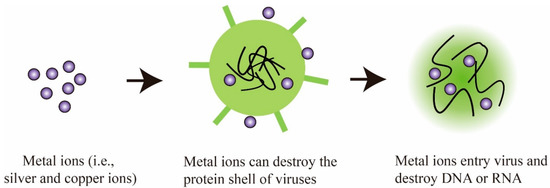
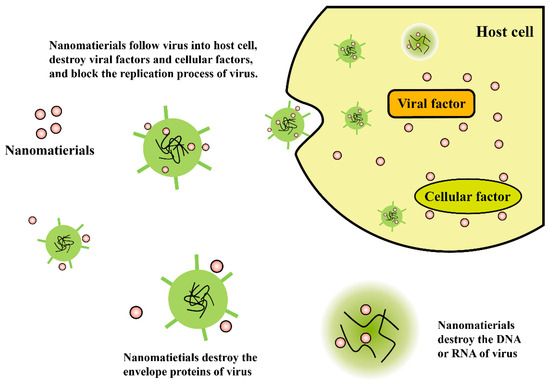
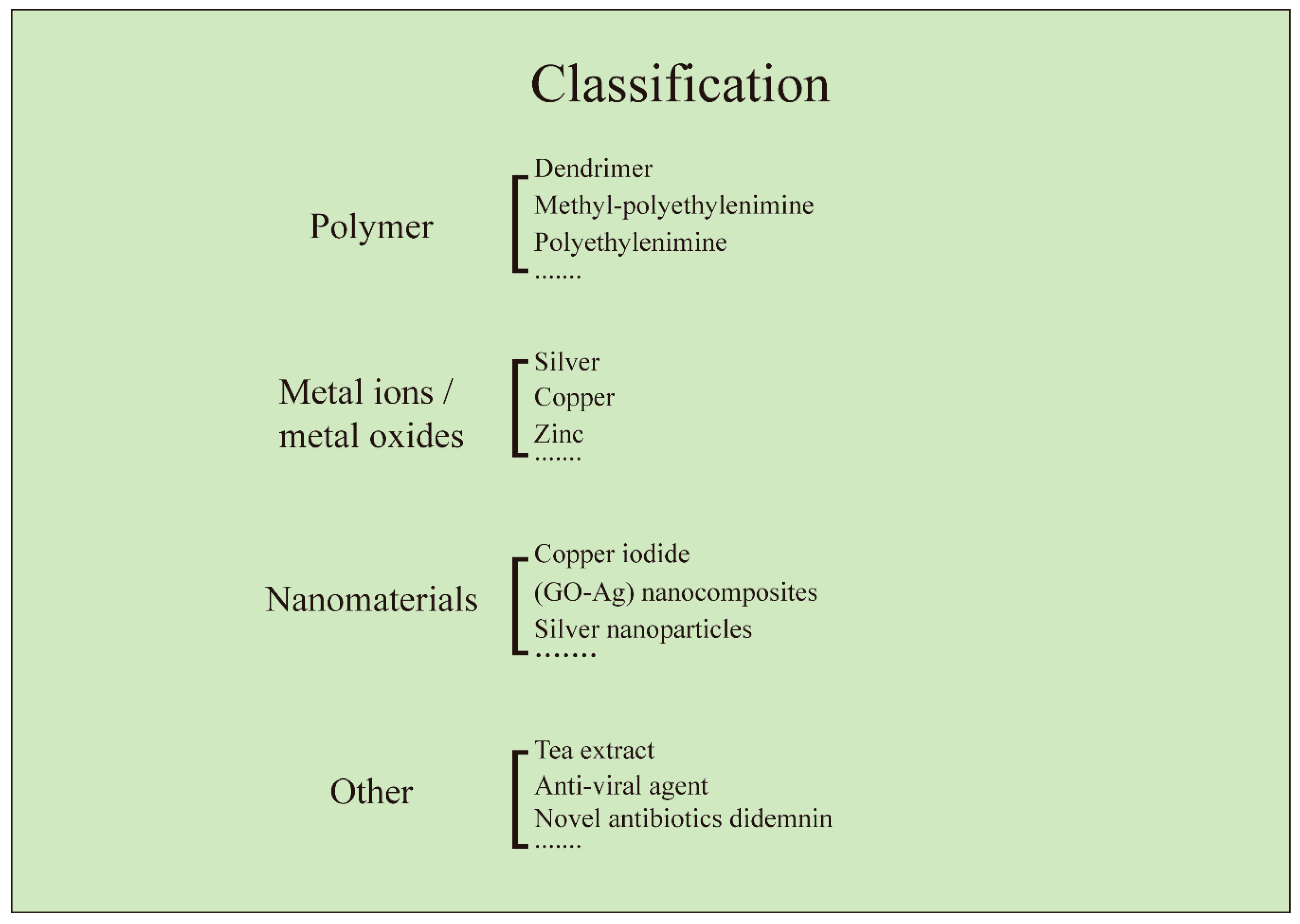
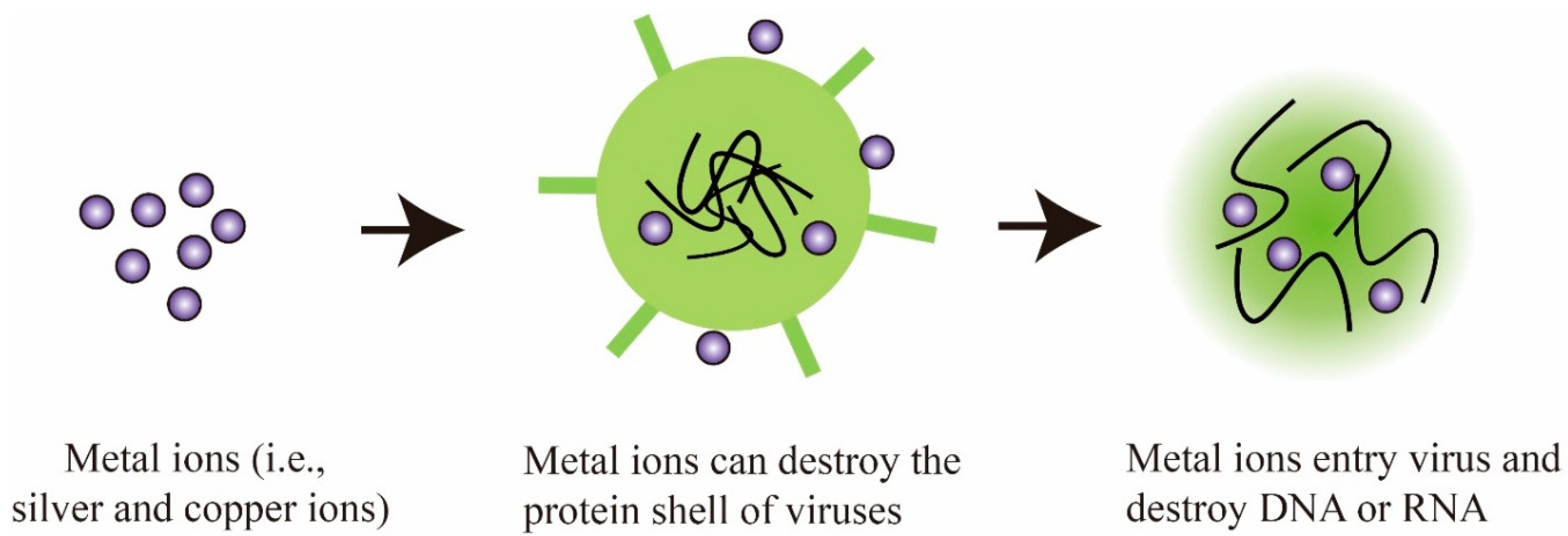
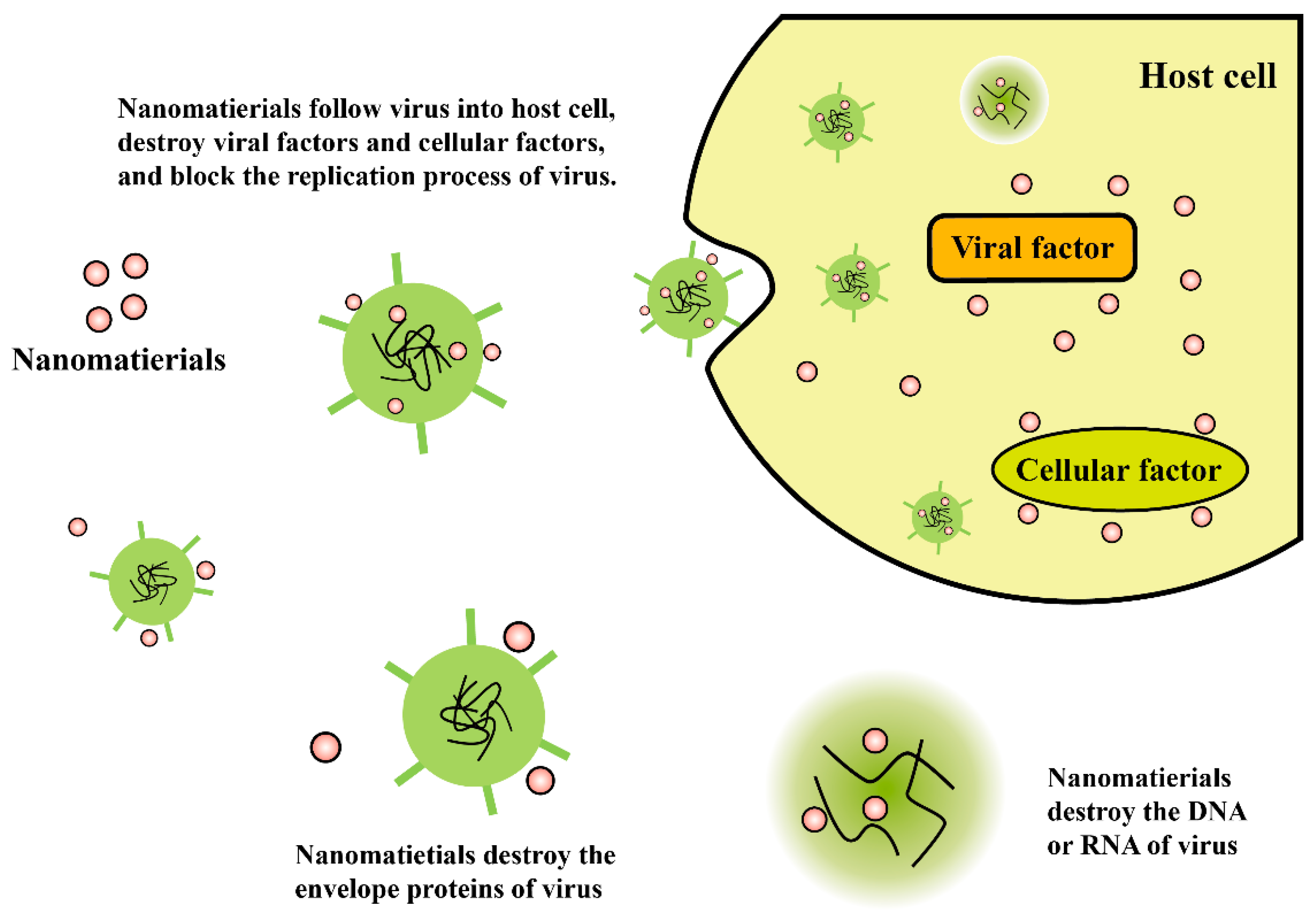
Members of antiviral coatings have been divided into three major groups (antiviral polymers, metal ions/metal oxides, and functional nanomaterials), based on the type of materials used at the contaminated sites. The methods for the treatment of virus affected substrates for preventing the virus deposition over the surfaces, using antiviral and viricidal coatings are discussed. The potential antiviral and viricidal coating technologies implemented, for design and development of a wide range of commercialized antiviral products, such as personal protective equipment (PPE), medical instrument, appliances, and hygienic implements, are discussed. Antiviral products are designed with the concept of modifying the surface, with any of the antiviral and viricidal coating compositions, using the most promising surface modification technologies [23,24,25,26,27]. Both antiviral and viricidal compositions and surface modification technologies play a major role in the destruction of viruses, by providing a thin film over the surface to retain its antiviral activity. The current review also highlights some formulations and applications of antiviral products on the basis of their antiviral compositions and activities.
4. Conclusions and Future Perspectives
In this review, specific coating materials that inactivate viruses have been discussed. Various strategies involved in the development of antiviral and viricidal coatings, like modifying the surface of a substrate via antiviral polymers, incorporation of metal ions/oxides, and functional nanoparticles were discussed. The antiviral efficacies of the developed coatings were detailed, and their possible and promising applications were further correlated with emerging viral pandemics, like COVID-19. There are a few areas that may need special attention to improve the existing technologies to fight against the current pandemic. 1. Processing of nano-sized metal particles and focusing their utility either in their original form or mixing them with polymers, to prepare novel functional coatings. 2. Chemical modification of the polymers to prepare a highly effective antiviral formulation. 3. The functional modification of the antiviral or viricidal nanoparticles, using other chemical moieties for better integration within a coating composition. In addition, immaculate procedures have to be identified in processing the antiviral coating materials. Based on recent progress, both inorganic polymer-based, and nanostructured, coating materials mentioned in this review have demonstrated the properties of superhydrophobicity, photo-induced superhydrophilicity, and excellent surface topography of the coated surfaces. An important problem that needs considerable attention is the long-term persistence of the virus particles on the surface layer of face masks, putting them at higher risk level during their usage and disposal. Hence, the manufacturing of optimized face masks by the application of a metal ions, consisting of nanoparticles on the surface of the filtering layer, could be considered as a viable approach for instantaneous elimination of viruses. Moreover, self-cleaning coatings on the filtering layer of the masks, could be applied to avoid the attachment of infectious microdroplets on face masks. The current coronavirus responsible for COVID-19 is transmitted not only through droplets, but also via various surfaces that can convey the virus from one person to another. Furthermore, research shows that the virus remains viable on various surfaces for extended periods of time, for days and even longer. Therefore, there is a clear need for durable anti-viral coatings that can be sprayed or painted on surfaces, just like paint or varnish, and that will prevent viral transmission. Finally, this review attempts to summarize and improvise the extension of present techniques, with certain modifications, for the prevention of COVID-19, and further inspire future antiviral strategies. Apart from viable functional coatings, conductive nanocoating (photo-thermally or electrically) of materials/metals onto surfaces, via robust and sterile methods, such as non-thermal plasma process, could be used in the future, not only for sterilizing surfaces, but to deliver nanomaterials-driven anti-infective surfaces for prolonged/reusable applications.
Author Contributions
R.P., X.Z. and M.D. wrote the paper. H.S.N., Y.Z. and S.R. designed the manuscript layout. H.S.N., Y.Z., X.P. and V.T. revised the paper and added significant discussions. S.R. and S.J. provided added critical comments on which H.S.N., Y.Z. and V.T. have worked further and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Startup Research Grant (SRG/2019/001504) awarded to HSN from department of science and technology (DST), government of India. The authors gratefully acknowledge the support from Guangdong medical university Scientific Research Foundation (4SG19003Ga) awarded to YZ. This work is also supported by Guangdong basic and applied basic research foundation (2019A1515111112) awarded to YZ.
Conflicts of Interest
All the authors declare no conflict of interest.
References
- Howell, A.B.; D’Souza, D.H. The Pomegranate: Effects on Bacteria and Viruses That Influence Human Health. Evid.-Based Complementary Altern. Med. 2013, 2013, 606212. [Google Scholar] [CrossRef] [PubMed]
- Daszak, P.; Cunningham, A.A.; Hyatt, A.D. Emerging Infectious Diseases of Wildlife—Threats to Biodiversity and Human Health. Science 2000, 287, 443. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, S. Dengue shock. J. Emergencies Trauma Shock 2011, 4, 120. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.J.; Parker, R.A.; Strilms, R.L. Association between respiratory syncytial virus outbreaks and lower respiratory tract deaths of infants and young children. J. Infect. Dis. 1990, 161, 640–646. [Google Scholar] [CrossRef]
- Hsieh, Y.-C.; Wu, T.-Z.; Liu, D.-P.; Shao, P.-L.; Chang, L.-Y.; Lu, C.-Y.; Lee, C.-Y.; Huang, F.-Y.; Huang, L.-M. Influenza pandemics: Past, present and future. J. Formos. Med. Assoc. 2006, 105, 1–6. [Google Scholar] [CrossRef]
- Neumann, G.; Chen, H.; Gao, G.F.; Shu, Y.; Kawaoka, Y. H5N1 influenza viruses: Outbreaks and biological properties. Cell Res. 2010, 20, 51–61. [Google Scholar] [CrossRef]
- Cheung, T.K.W.; Poon, L.L.M. Biology of influenza a virus. Ann. N. Y. Acad. Sci. 2007, 1102, 1–25. [Google Scholar] [CrossRef]
- Zhong, N.; Zheng, B.; Li, Y.; Poon, L.; Xie, Z.; Chan, K.; Li, P.; Tan, S.; Chang, Q.; Xie, J.P. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People’s Republic of China, in February, 2003. Lancet 2003, 362, 1353–1358. [Google Scholar] [CrossRef]
- Abdel-Moneim, A.S. Middle East respiratory syndrome coronavirus (MERS-CoV): Evidence and speculations. Springer 2014, 159, 1575–1584. [Google Scholar] [CrossRef]
- Muyembe-Tamfum, J.-J.; Mulangu, S.; Masumu, J.; Kayembe, J.; Kemp, A.; Paweska, J.T. Ebola virus outbreaks in Africa: Past and present. Onderstepoort J. Vet. Res. 2012, 79, 6–13. [Google Scholar] [CrossRef]
- Safari, S.; Baratloo, A.; Rouhipour, A.; Ghelichkhani, P.; Yousefifard, M.J.E. Ebola hemorrhagic fever as a public health emergency of international concern; A review article. Arch. Acad. Emerg. Med. 2015, 3, 3. [Google Scholar]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Wang, W.; Tang, J.; Wei, F. Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan, China. J. Med. Virol. 2020, 92, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. (Lond. Engl.) 2020, 78, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.L.; Wang, Y.M.; Li, X.W.; Ren, L.L.; Zhao, J.P.; Hu, Y.; Zhang, L.; Fan, G.H.; Xu, J.Y.; Gu, X.Y.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, B.; Li, Q.; Wen, L.; Zhang, R. Clinical Features of 69 Cases with Coronavirus Disease 2019 in Wuhan, China. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Phua, J.; Weng, L.; Ling, L.; Egi, M.; Lim, C.-M.; Divatia, J.V.; Shrestha, B.R.; Arabi, Y.M.; Ng, J.; Gomersall, C.D.; et al. Intensive care management of coronavirus disease 2019 (COVID-19): Challenges and recommendations. Lancet Respiratory Medicine 2020, 8, 506–517. [Google Scholar] [CrossRef]
- Prompetchara, E.; Kettoy, C.; Palaga, T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020, 38, 1–9. [Google Scholar] [CrossRef]
- West, S.A.; Griffin, A.S.; Gardner, A.; Diggle, S.P. Social evolution theory for microorganisms. Nat. Rev. Microbiol. 2006, 4, 597–607. [Google Scholar] [CrossRef]
- Durmus Tekir, S.; Cakir, T.; Ulgen, K. Infection Strategies of Bacterial and Viral Pathogens through Pathogen–Human Protein–Protein Interactions. Front. Microbiol. 2012, 3, 46. [Google Scholar] [CrossRef]
- Kaufmann, S.H.; Dorhoi, A.; Hotchkiss, R.S.; Bartenschlager, R. Host-directed therapies for bacterial and viral infections. Nat. Rev. Drug Discov. 2018, 17, 35. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Fan, C.H.; Li, M.; Nie, H.L.; Wang, F.B.; Wang, H.; Wang, R.; Xia, J.B.; Zheng, X.; Zuo, X.L.; et al. COVID-19: A Call for Physical Scientists and Engineers. ACS Nano 2020, 14, 3747–3754. [Google Scholar] [CrossRef] [PubMed]
- Haldar, J.; Weight, A.K.; Klibanov, A.M. Preparation, application and testing of permanent antibacterial and antiviral coatings. Nat. Protoc. 2007, 2, 2412. [Google Scholar] [CrossRef]
- Fabbri, P.; Messori, M. Surface Modification of Polymers: Chemical, Physical, and Biological Routes. Modif. Polym. Prop. 2017, 109–130. [Google Scholar] [CrossRef]
- Ahirwar, H.; Zhou, Y.; Mahapatra, C.; Ramakrishna, S.; Kumar, P.; Nanda, H.S. Materials for Orthopedic Bioimplants: Modulating Degradation and Surface Modification Using Integrated Nanomaterials. Coatings 2020, 10, 264. [Google Scholar] [CrossRef]
- Nanda, H.S. Surface modification of promising cerium oxide nanoparticles for nanomedicine applications. RSC Adv. 2016, 6, 111889–111894. [Google Scholar] [CrossRef]
- Nanda, H.S. Preparation and Biocompatible Surface Modification of Redox Altered Cerium Oxide Nanoparticle Promising for Nanobiology and Medicine. Bioengineering 2016, 3, 28. [Google Scholar] [CrossRef]
- Le, T.T.; Andreadakis, Z.; Kumar, A.; Rom?n, R.G.; Tollefsen, S.; Saville, M.; Mayhew, S. The COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020, 19, 305–306. [Google Scholar] [CrossRef]
- Lurie, N.; Saville, M.; Hatchett, R.; Halton, J. Developing Covid-19 Vaccines at Pandemic Speed. N. Engl. J. Med. 2020, 382, 1969–1973. [Google Scholar] [CrossRef]
- Lara, H.H.; Garza-Trevino, E.N.; Ixtepan-Turrent, L.; Singh, D.K. Silver nanoparticles are broad-spectrum bactericidal and virucidal compounds. J. Nanobiotechnol. 2011, 9, 30. [Google Scholar] [CrossRef]
- Fujimori, Y.; Sato, T.; Hayata, T.; Nagao, T.; Nakayama, M.; Nakayama, T.; Sugamata, R.; Suzuki, K. Novel Antiviral Characteristics of Nanosized Copper(I) Iodide Particles Showing Inactivation Activity against 2009 Pandemic H1N1 Influenza Virus. Appl. Environ. Microbiol. 2012, 78, 951–955. [Google Scholar] [CrossRef]
- Galdiero, S.; Falanga, A.; Vitiello, M.; Cantisani, M.; Marra, V.; Galdiero, M. Silver Nanoparticles as Potential Antiviral Agents. Molecules 2011, 16, 8894–8918. [Google Scholar] [CrossRef] [PubMed]
- Lara, H.H.; Ixtepan-Turrent, L.; Garza-Treviño, E.N.; Rodriguez-Padilla, C. PVP-coated silver nanoparticles block the transmission of cell-free and cell-associated HIV-1 in human cervical culture. J. Nanobiotechnol. 2010, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Papp, I.; Sieben, C.; Ludwig, K.; Roskamp, M.; Boettcher, C.; Schlecht, S.; Herrmann, A.; Haag, R. Inhibition of Influenza Virus Infection by Multivalent Sialic-Acid-Functionalized Gold Nanoparticles. Small 2010, 6, 2900–2906. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Bonilla, A.; Fernández-García, M. Polymeric materials with antimicrobial activity. Prog. Polym. Sci. 2012, 37, 281–339. [Google Scholar] [CrossRef]
- Modak, S.M.; Sampath, L. Antiviral Glove. U.S. Patent 5,133,090, 28 July 1992. [Google Scholar]
- Yao, L. Thermoplastic Particles which Comprise an Antiviral or Antimicrobial Agent. U.S. Patent 10/408,095, 15 January 2004. [Google Scholar]
- Snyder Jr, D.E. Anti-Bacterial/Anti-Viral Coatings, Coating Process and Parameters Thereof. U.S. Patent 5,968,538, 19 October, 1999. [Google Scholar]
- Swanson, M.J. Virus Inactivating Coatings. U.S. Patent 5,783,502, 21 July 1998. [Google Scholar]
- Adachi, T.; Latthe, S.S.; Gosavi, S.W.; Roy, N.; Suzuki, N.; Ikari, H.; Kato, K.; Katsumata, K.-i.; Nakata, K.; Furudate, M. Photocatalytic, superhydrophilic, self-cleaning TiO2 coating on cheap, light-weight, flexible polycarbonate substrates. Appl. Surf. Sci 2018, 458, 917–923. [Google Scholar] [CrossRef]
- Hill, B.R.; Watson Sr, T.F.; Triplett, B.L. Antimicrobial Microporous Coating. U.S. Patent 11/726,121, 18 October 2007. [Google Scholar]
- Gabbay, J. Antimicrobial and Antiviral Polymeric Materials and a Process for Preparing the Same. U.S. Patent 10/890,936, 9 December 2004. [Google Scholar]
- Matsushita, M.; Otsuki, K.; Takakuwa, H.; Tsunekuni, R. Antiviral Substance, Antiviral Fiber, and Antiviral Fiber Structure. U.S. Patent 12/679,211, 28 October 2010. [Google Scholar]
- Haldar, J.; Chen, J.; Tumpey, T.M.; Gubareva, L.V.; Klibanov, A.M. Hydrophobic polycationic coatings inactivate wild-type and zanamivir- and/or oseltamivir-resistant human and avian influenza viruses. Biotechnol. Lett. 2008, 30, 475–479. [Google Scholar] [CrossRef]
- Larson, A.M. Antiviral Polymeric Drugs and Surface Coatings; Massachusetts Institute of Technology: Cambridge, MA, USA, 2013. [Google Scholar]
- Lieleg, O.; Lieleg, C.; Bloom, J.; Buck, C.B.; Ribbeck, K. Mucin Biopolymers As Broad-Spectrum Antiviral Agents. Biomacromolecules 2012, 13, 1724–1732. [Google Scholar] [CrossRef]
- Matthews, B.R.; Holan, G. Antiviral Dendrimers. U.S. Patent 6,190,650, 20 February 2001. [Google Scholar]
- Tavakoli, A.; Ataei-Pirkooh, A.; Mm Sadeghi, G.; Bokharaei-Salim, F.; Sahrapour, P.; Kiani, S.J.; Moghoofei, M.; Farahmand, M.; Javanmard, D.; Monavari, S.H. Polyethylene glycol-coated zinc oxide nanoparticle: An efficient nanoweapon to fight against herpes simplex virus type 1. Nanomedicine 2018, 13, 2675–2690. [Google Scholar] [CrossRef]
- Poggio, C.; Colombo, M.; Arciola, C.R.; Greggi, T.; Scribante, A.; Dagna, A.J.M. Copper-Alloy Surfaces and Cleaning Regimens against the Spread of SARS-CoV-2 in Dentistry and Orthopedics. From Fomites to Anti-Infective Nanocoatings. Materials 2020, 13, 3244. [Google Scholar] [CrossRef]
- Thurman, R.B.; Gerba, C.P. The molecular mechanisms of copper and silver ion disinfection of bacteria and viruses. CRC Crit. Rev. Environ. Control 1988, 18, 295–315. [Google Scholar] [CrossRef]
- Hodek, J.; Zajicova, V.; Lovetinska-Slamborova, I.; Stibor, I.; Muellerova, J.; Weber, J. Protective hybrid coating containing silver, copper and zinc cations effective against human immunodeficiency virus and other enveloped viruses. BMC Microbiol. 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Miki, S.; Ueda, T.; Yamashina, D.; Kinugawa, K. Coating Agent Composition and Antibacterial/Antiviral Member. U.S. Patent 10,131,797, 20 November 2018. [Google Scholar]
- Pallenberg, A.J.; Marschner, T.M.; Barnhart, D.M. Phenanthroline complexes of the d10 metals nickel(0), zinc(II) and silver(I)—comparison to copper(I) species. Polyhedron 1997, 16, 2711–2719. [Google Scholar] [CrossRef]
- Zeedan, G.; EL-Razik, K.; Allam, A.; Abdalhamed, A.; Zeina, H.A.A. Evaluations of potential antiviral effects of green zinc oxide and silver nanoparticles against bovine herpesvirus-1. Adv. Anim. Vet. Sci. 2020, 8, 433–443. [Google Scholar] [CrossRef]
- Bright, K.R.; Sicairos-Ruelas, E.E.; Gundy, P.M.; Gerba, C.P. Assessment of the Antiviral Properties of Zeolites Containing Metal Ions. Food Environ. Virol. 2008, 1, 37. [Google Scholar] [CrossRef]
- Rai, M.; Deshmukh, S.D.; Ingle, A.P.; Gupta, I.R.; Galdiero, M.; Galdiero, S. Metal nanoparticles: The protective nanoshield against virus infection. Crit. Rev. Microbiol. 2016, 42, 46–56. [Google Scholar] [CrossRef]
- Ueda, T.; Yamashina, D.; Kinugawa, K. Copper Complex Titanium Oxide Dispersion Liquid, Coating Agent Composition, and Antibacterial/Antiviral Member. U.S. Patent 9,585,385, 7 March 2017. [Google Scholar]
- Charan, N.; Lavanya, N.; Praveen, B.; Praveen, A.; Sridevi, A.; Narasimha, G. Antiviral activity of antimony and arsenic oxides. Der. Pharma. Chem. 2012, 4, 687–689. [Google Scholar]
- Kelly, S.R.; Gbadamosi, K.A.; Seger, G.E.; Biedermann, K.A.; Morgan, J.M.; Swaile, D.F. Antiviral Compositions for Tissue Paper. U.S. Patent 6,475,501, 5 November 2002. [Google Scholar]
- Hei, R.D.; Smith, K.R.; Laugen, P.D.; Kennedy, S.P. Antimicrobial and Antiviral Compositions Containing an Oxidizing Species. U.S. Patent 6,436,445, 20 August 2002. [Google Scholar]
- Hei, R.D.; Richter, F.L.; Reinhardt, D.J.; Leafblad, B.R. Antimicrobial and Antiviral Compositions Containing an Oxidizing Species. U.S. Patent 6,855,328, 15 February 2005. [Google Scholar]
- Gabbay, J. Antimicrobial and Antiviral Polymeric Materials. U.S. Patent 7,169,402, 30 January 2007. [Google Scholar]
- Oka, H.; Tomioka, T.; Tomita, K.; Hoshino, K.; Nishino, A.; Ueda, S. Method of Disinfecting an Object and Antiviral Disinfection Liquid Therefor. U.S. Patent 5,645,846, 8 July 1997. [Google Scholar]
- Trogolo, J.A. Antiviral Methods. U.S. Patent 11/726,121, 18 October 2007. [Google Scholar]
- Zhou, J.; Hu, Z.; Zabihi, F.; Chen, Z.; Zhu, M. Progress and Perspective of Antiviral Protective Material. Adv. Fiber Mater. 2020, 2, 123–139. [Google Scholar] [CrossRef]
- Borkow, G.; Zhou, S.S.; Page, T.; Gabbay, J. A Novel Anti-Influenza Copper Oxide Containing Respiratory Face Mask. PLoS ONE 2010, 5. [Google Scholar] [CrossRef]
- Park, D.; Larson, A.M.; Klibanov, A.M.; Wang, Y. Antiviral and Antibacterial Polyurethanes of Various Modalities. Appl. Biochem. Biotechnol. 2013, 169, 1134–1146. [Google Scholar] [CrossRef]
- Park, D. Antimicrobial Polyurethane Materials and Methods of Forming and Using Same. U.S. Patent 9,949,484, 24 April 2018. [Google Scholar]
- Bignozzi, C.A.; Dissette, V.; Corallini, A.; Carra, G.; Della Valle, R. Functional Nanomaterials with Antibacterial and Antiviral Activity. U.S. Patent 8,158,137, 17 April 2012. [Google Scholar]
- Broglie, J.J.; Alston, B.; Yang, C.; Ma, L.; Adcock, A.F.; Chen, W.; Yang, L. Antiviral Activity of Gold/Copper Sulfide Core/Shell Nanoparticles against Human Norovirus Virus-Like Particles. PLoS ONE 2015, 10, e0141050. [Google Scholar] [CrossRef] [PubMed]
- Vincent, M.; Duval, R.E.; Hartemann, P.; Engels-Deutsch, M. Contact killing and antimicrobial properties of copper. J. Appl. Microbiol. 2018, 124, 1032–1046. [Google Scholar] [CrossRef] [PubMed]
- Cagno, V.; Andreozzi, P.; D’Alicarnasso, M.; Silva, P.J.; Mueller, M.; Galloux, M.; Le Goffic, R.; Jones, S.T.; Vallino, M.; Hodek, J.; et al. Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nat. Mater. 2018, 17, 195. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, S.; Ingle, A.; Gade, A.; Rai, M.; Falanga, A.; Incoronato, N.; Russo, L.; Galdiero, S.; Galdiero, M. Antiviral activity of mycosynthesized silver nanoparticles against herpes simplex virus and human parainfluenza virus type 3. Int. J. Nanomed. 2013, 8, 4303–4314. [Google Scholar] [CrossRef]
- Hang, X.; Peng, H.; Song, H.; Qi, Z.; Miao, X.; Xu, W. Antiviral activity of cuprous oxide nanoparticles against Hepatitis C Virus in vitro. J. Virol. Methods 2015, 222, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Masarwa, M.; Cohen, H.; Meyerstein, D.; Hickman, D.L.; Bakac, A.; Espenson, J.H. Reactions of low-valent transition-metal complexes with hydrogen peroxide. Are they "Fenton-like" or not? 1. The case of Cu+aq and Cr2+aq. J. Am. Chem. Soc. 1988, 110, 4293–4297. [Google Scholar] [CrossRef]
- Khandelwal, N.; Kaur, G.; Kumar, N.; Tiwari, A. Application of silver nanoparticles in viral inhibition: A new hope for antivirals. Dig. J. Nanomater. Biostruct. 2014, 9, 175–186. [Google Scholar]
- Ghosh, S.K. Anti-Viral Surface Coating to Prevent Spread of Novel Coronavirus (COVID-19) Through Touch; Nova Surface-Care Centre Pvt: Maharashtra, India, 2020. [Google Scholar]
- Lysenko, V.; Lozovski, V.; Lokshyn, M.; Gomeniuk, Y.V.; Dorovskih, A.; Rusinchuk, N.; Pankivska, Y.; Povnitsa, O.; Zagorodnya, S.; Tertykh, V.; et al. Nanoparticles as antiviral agents against adenoviruses. Adv. Nat. Sci. Nanosci. Nanotechnol. 2018, 9, 025021. [Google Scholar] [CrossRef]
- Mori, Y.; Ono, T.; Miyahira, Y.; Vinh Quang, N.; Matsui, T.; Ishihara, M. Antiviral activity of silver nanoparticle/chitosan composites against H1N1 influenza A virus. Nanoscale Res. Lett. 2013, 8. [Google Scholar] [CrossRef]
- Zhong, H.; Zhu, Z.; Lin, J.; Cheung, C.F.; Lu, V.L.; Yan, F.; Chan, C.-Y.; Li, G. Reusable and recyclable graphene masks with outstanding superhydrophobic and photothermal performances. ACS Nano 2020, 14, 6213–6221. [Google Scholar] [CrossRef]
- Chen, Y.-N.; Hsueh, Y.-H.; Hsieh, C.-T.; Tzou, D.-Y.; Chang, P.-L. Antiviral activity of graphene–silver nanocomposites against non-enveloped and enveloped viruses. Int. J. Environ. Res. Public Health 2016, 13, 430. [Google Scholar] [CrossRef] [PubMed]
- Ueda, T.; Kinugawa, K.; Yamashina, D. Cuprous Oxide Particle Dispersion Liquid, Coating Agent Composition, and Antibacterial/Antiviral Member. U.S. Patent 9,414,585, 16 August 2016. [Google Scholar]
- Watanabe, Y.; Shuto, Y.; Itoda, Y. Antibacterial/Antiviral Coating Material, and Method for Forming Antibacterial/Antiviral Coating Film. U.S. Patent 9,943,081, 17 April 2018. [Google Scholar]
- Otsuki, K.; Ito, T.; Murase, T.; Ito, H.; Wakabayashi, K.; Yakura, M.; Yamana, H. Antiviral Agent, and Fabric and Antiviral Member Supporting Antiviral Agent. U.S. Patent 10/567,986, Feb. 12, 2009. [Google Scholar]
- Sugiura, K. Antiviral Agent, Coating Composition, Resin Composition and Antiviral Product. U.S. Patent 16/078,534, 14 February 2019. [Google Scholar]
- Rossett, H. Fluid Compositions that can Form a Coating Having Antiviral Properties. U.S. Patent 13/899,902, 5 June 2014. [Google Scholar]
- Rolf, D. Inhalation Antiviral Patch. U.S. Patent 10/458,078, 15 April 2004. [Google Scholar]
- Rinehart, K.L., Jr. Didemnins A, B, C, and derivatives thereof, as antiviral agents. U.S. Patent 4,493,796, 15 January 1985. [Google Scholar]
- Nashimoto, K.; Tashiro, Y.; Kosaka, Y.; Hara, Y. Antiviral Filter Air Cleaner Impregnated with Tea Extract. U.S. Patent 5,747,053, 5 May 1998. [Google Scholar]
- Praditya, D.; Kirchhoff, L.; Brüning, J.; Rachmawati, H.; Steinmann, J.; Steinmann, E. Anti-infective properties of the golden spice curcumin. Front. Microbiol. 2019, 10, 912. [Google Scholar] [CrossRef] [PubMed]
- Revathi, T.; Thambidurai, S. Synthesis of chitosan incorporated neem seed extract (Azadirachta indica) for medical textiles. Int. J. Biol. Macromol. 2017, 104, 1890–1896. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.K.; Akl, E.A.; Duda, S.; Solo, K.; Yaacoub, S.; Schunemann, H.J. Physical distancing, face masks, and eye protection to prevent person-to-person transmission of SARS-CoV-2 and COVID-19: A systematic review and meta-analysis. Lancet (Lond. Engl.) 2020. [Google Scholar] [CrossRef]
- Odeberg, J.; Wirsén, A.; Norberg, Å.; Frie, J.; Printz, G.; Lagercrantz, H.; Gudmundsson, G.H.; Agerberth, B.; Jonsson, B. A novel cysteine-linked antibacterial surface coating significantly inhibits bacterial colonization of nasal silicone prongs in a phase one pre-clinical trial. Mater. Sci. Eng. C 2018, 93, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Wen, S.H. Antiviral and Antibacterial Respirator Mask. U.S. Patent 6,681,765, 27 January 2004. [Google Scholar]
- Morse, S.S.; Garwin, R.L.; Olsiewski, P.J. Public health—Next flu pandemic: What to do until the vaccine arrives? Science 2006, 314, 929. [Google Scholar] [CrossRef][Green Version]
- Tong, H.W.; Kwok, S.K.C.; Kwok, H.C. Protective Masks with Coating Comprising Different Electrospun Fibers Interweaved with Each Other, Formulations Forming the Same, and Method of Producing Thereof. U.S. Patent 10,201,198, 12 February 2019. [Google Scholar]
- Tebyetekerwa, M.; Xu, Z.; Yang, S.; Ramakrishna, S. Electrospun Nanofibers-Based Face Masks. SpringerLink 2020. [Google Scholar] [CrossRef]
- Fox, P.S.; Pedersen, D.E.; Rolando, J.J.; Staub, R.K. Compositions Having a High Antiviral Efficacy. U.S. Patent 8,034,844, 11 October 2011. [Google Scholar]
- Torkelson, A.A.; da Silva, A.K.; Love, D.C.; Kim, J.Y.; Alper, J.P.; Coox, B.; Dahm, J.; Kozodoy, P.; Maboudian, R.; Nelson, K.L. Investigation of quaternary ammonium silane-coated sand filter for the removal of bacteria and viruses from drinking water. J. Appl. Microbiol. 2012, 113, 1196–1207. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).