Abstract
The use of coke with high reactivity in the ironmaking blast furnace (BF) has yet to be explored, and a thorough understanding is still required to clarify the effect of coke reactivity on the BF gas utilization efficiency. In this paper, a one-dimensional kinetic model of the BF is presented and the accuracy of the model is verified. The model is then applied to analyze the effect of coke reactivity on the gas utilization efficiency of the BF. The results show that, under the operating conditions considered, the height of indirect reduction region and the starting temperature of coke solution loss reaction decrease with the increase of coke reactivity. Moreover, coke reactivity is first, directly proportional to gas utilization efficiency, and then, inversely proportional to it. In addition, high-reactivity coke may not improve gas utilization efficiency in case of high H2 content. Both, lowly and highly reactive coke need to be combined with highly reducible iron ore to maximize the gas utilization efficiency. Nevertheless, only appropriately reactive coke can combine with lowly reducible iron ore to obtain an optimal gas utilization efficiency. Hence, it is necessary to select coke with appropriate reactivity, in accordance with iron-ore reducibility, instead of blindly pursuing high-reactivity coke in actual operation.
1. Introduction
The blast furnace (BF) is the most widely employed reactor for iron production since the iron and steel begin to be used on a large scale. In the ironmaking process, the gas utilization efficiency is an important index since it can reflect the overall performance of the BF, which is related to the utilization of energy. In the BF, the reducing gas (mainly carbon monoxide, i.e., CO) is utilized in the thermal reserve zone (TRZ), where the indirect reduction of iron oxides (mainly wustite, i.e., FeO) occurs and the rate of coke solution loss reaction is negligible. It has been widely accepted that the TRZ temperature is nearly constant and can be taken as the temperature at the TRZ bottom where the coke solution loss reaction starts to take place with a discernable rate, consuming the charged coke and producing CO. In principle, the gas utilization efficiency can be promoted by increasing the reducibility of iron ore, due to a higher reduction rate. This explains why iron ore with a high reducibility is desirable in practice. By both theoretical analysis and experimental investigations, it has been shown that the TRZ temperature also plays a role in determining the gas utilization efficiency because the exothermic CO reduction of FeO in the TRZ is favored with a lower temperature [1,2,3,4,5,6,7,8,9]. The starting temperature of coke solution loss reaction, which is linked to the TRZ temperature, depends on the coke reactivity. Moreover, it has been reported that a higher reactivity can lead to a lower starting temperature of coke solution loss reaction, thus, a lower TRZ temperature. In order to improve the gas utilization efficiency and reduce the fuel rate of the BF, some investigators, therefore, suggested the application of high-reactivity coke and then put it into practice [1,2,3,4,5,6,7,10]. In some instances, however, the gas utilization efficiency appears to decrease when coke with a higher reactivity is charged into the BF. From a perspective of kinetics, a decreased TRZ temperature lowers the gas-solid reaction rate constant, as well as the diffusion coefficient of gas species although it favors the exothermic reduction reaction thermodynamically [1,8]. Therefore, a thorough understanding is still required to clarify the effect of coke reactivity on gas utilization efficiency of the BF. This is one of the motivations behind the current work.
In this paper, a one-dimensional kinetic model of the BF is presented and validated by comparing the reported results from the literature [11]. The model is further employed to conduct a series of simulations for investigating the effect of coke reactivity on the BF process. After that, the combination of coke reactivity and iron-ore reducibility is discussed.
2. Model Description
2.1. Process Variables and Chemical Reactions
It is necessary to simplify the BF process when a mathematical model based on mass and energy conservation is built since the ironmaking process is considerably complicated. In the current model, the inner state of a BF is described by nine process variables along the axial direction of the BF, including (1) temperature of gas, (2) temperature of solid phases, (3) reduction degree of iron ores, (4) decomposition degree of flux (mainly limestone and dolomite), (5) solution loss degree of coke, (6) CO content of gas, (7) CO2 content of gas, (8) H2 content of gas, and (9) pressure of gas. The profiles of these variables can be obtained by solving a series of differential and algebraic equations that govern the heat and mass transfers (as well as chemical reactions) between the countercurrent gas and solid flows. The chemical reactions considered in the current model are shown in Table 1.

Table 1.
Chemical reactions considered in the model.
2.2. Simplifications and Assumptions
All the reactions listed in Table 1 do not take place in every part of the BF. Hence, for simulating the BF process conveniently, the calculation domain covering the area from stockline to the tuyere level is divided into two zones: The stack zone and the bosh zone, which are characterized by the reactions taking place within them. Figure 1 illustrates the two zones and the reactions in these zones. The stack zone is the region from the stockline to the level of the solid temperature reaching 1623 K, which is the pre-specified melting temperature, namely, at the lower end of the stack zone the iron ore starts to melt. Within this zone, the reducing gas reacts with iron ore as it descends to produce magnetite and wustite and iron, as well as water and gas react with each other, and the flux decomposes into basic oxide and carbon dioxide. In addition, the iron-ore softening area, where coke begins to undergo the reactions (3) and (4) and the direct reduction of FeO starts to occur in the form of coupling reactions (1-c), (2-c), (3) and (4), is also included in the stack zone. Note that the starting temperature of reaction (3) is set as the solid temperature when the reaction degree of coke is around 10−3. Hence, in this model the TRZ, as well as the indirect reduction region end when the reaction degree of coke exceeds 10−3.
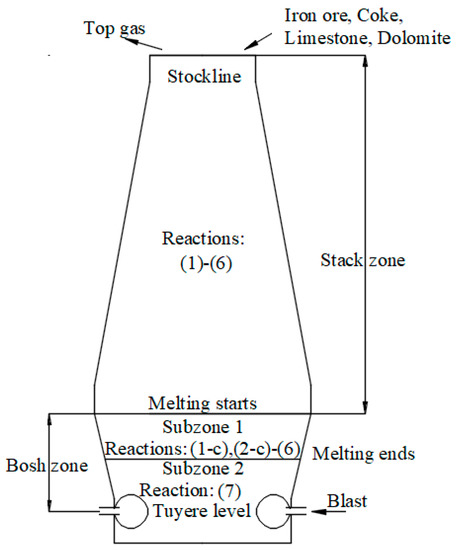
Figure 1.
Schematic diagram of zones in blast furnace.
When the temperature of solids exceeds the melting point of the iron ore, the bosh zone starts, and it ends at tuyere level. There are two parts of this zone, namely, subzone 1: Melting region and subzone 2: Dripping region. Within the subzone 1, the chemical reactions taken into consideration are the same as in the softening area, except for melting of solid FeO. When the melting process becomes complete, the dripping region begins. The difference between these two subzones is that only direct reduction of liquid FeO (reaction (7)) is assumed to take place in subzone 2 because of the incalculable kinetics and heat transfer of this reaction during melting.
The combustion in the so-called combustion zone is not treated in the model directly. The composition and temperature of the gas at the tuyere level are applied to subsequent model validation, which will be illustrated in the following section.
The primary heat transfer function in these zones is the exchange of heat between the gas and solids. The axial and radial conduction of heat in the gas and solids is neglected. Therefore, the important energy terms are the sensible heat of the gas and solids, heat transfer according to axial convection, the heat produced or used in the chemical reactions, and the heat losses to the cooling system and to the wall along the axis of the furnace. Note, the heat losses to the cooling system and to the wall are calculated by some correction factors because that different cooling areas of the furnace need different factors, as well as interior heat transfer phenomena is uncertain factor and difficult to measure.
After simplifications, some assumptions need to be made for simulation. Through the study of the blast furnace by Yagi and co-authors [12,13,14] and the measurements on the working furnaces, the findings reveal that: (1) The mass flow, temperature and composition of the gas and solid phases are non-uniform distribution along the radial direction, and (2) the burden materials present a layered structure. Therefore, the following assumptions are made for the model:
- The BF stays a steady state.
- Radial distribution of process variables is ignored.
- Each solid particle is assumed to be homogeneous in temperature and all the burden particles are well mixed.
- The size and physical properties of each burden component are maintained as the cell moves down in the shaft and the movement is a cylindrical type.
- The void fraction in the furnace packed bed is constant.
- The mass and heat transfer in a phase along the axis of furnace is not considered.
2.3. Governing Equations
The governing equations for mass, heat balance are expressed in the following form. Solid material balances: Through conducting the mass balance for the solid phases, the following three ordinary differential equations (ODEs) are obtained.
Reduction degree of the iron ore:
Reaction degree of the coke,
The decomposition degree of the flux,
where fP = fractional reduction of iron-bearing materials, fC = fraction of carbon in coke reacted, fF = fractional decomposition of flux, nj = number of moles of species j in the packed bed (mol/m3), subscript 0 is original burden conditions for component in question of the material in the cell (assuming no reaction), ε = void fraction of bed, Az = cross sectional area of shaft at elevation z (m2), Fs = volumetric flow rate of solids (m3(particles)/s), Rk = reaction rate of the k-th reaction (mol/(m3·s)), j represents solid phases.
Gas components’ material balances: Through conducting the mass balance for the gas phase, the following three ODEs are obtained.
CO content in the gas:
CO2 content in the gas:
H2 content in the gas:
H2O content in the gas,
where F = volumetric rate of gas flow (m3/s), C = molar concentration of gas stream (mol/m3), Yi = volume fraction of component i in gas phase, subscript CZ is gas property at combustion zone entering the bosh, RMC = rate of evaporation of moisture (mol/s), WCi = mass flow rate of component i in the coke (kg/s), MH2O = molecular weight of component H2O, Gg = molar flow rate of gas (mol/s), i represents: CO, CO2, H2, H2O, N2.
Solid and gas energy balances: Through conducting the heat balance for the gas and solid phases in a differential volume of the furnace, two ODEs are shown in Equations (8) and (9).
Gas temperature:
Solid phase temperature:
where Cpg = heat capacity of gas mixture (J/(mol·K)), Cs = average heat capacity of solid phases (J/(mol·K)), Qgs = heat transfer from gas to solid phases (J/(m·s)), Qwg = heat losses assigned to gas phase (J/(m·s)), Qws = heat losses assigned to solid phases (J/(m·s)), ρs = molar density of solid phases (mol/m3), M is molecular weight with a subscript, νk,i = stoichiometric coefficient of gas component i in the k-th reaction, νk,j = stoichiometric coefficient of component j of solid phases in the k-th reaction, αi = the proportion of reaction heat gained by gas phase, ΔHk = heat of k-th reaction (J/mol), Tg = temperature of bulk gas (K), Ts = temperature of solid phases (K).
The gas pressure is described by the Ergun’s equation as follows [11]:
where P = pressure of gas (Pa), dp = average diameter of solid particles (m), μg = viscosity of gas (Pa·s), ρ = density of gas (kg/m3).
The heat capacity data for gas phases are obtained in a reliable source [15]. Table 2 shows the kinetic parameters of gasification reactions of coke in two sets of data which are comparison objects for model validation from Bethlehem Steel Corporation’s L-Furnace and Number 7 furnace of Algoma Steel Corporation [11]:

Table 2.
Values of frequency factor in the simulation.
2.4. Reaction Rates
In this model, reaction rates of indirect reduction of iron ore by CO and H2, R1 and R2, are calculated by the equations derived from one-interface, unreacted shrinking-core (URC) model [16,17,18]. The process of iron ore reduction in URC model has been summarized to three limiting rate control parts: Gas film resistance, diffusion resistance through the porous production layer and chemical reaction resistance at the reaction interface. The kinetics parameters, such as the frequency factors and reaction rate constants for six reductions, are calculated by the equations from the literature [11,16,17].
Coke is gasified in the reactions (3) and (4) in the stack zone and subzone 1. Carbon dioxide and water vapor flow through the porous coke particle and the reactions (3) and (4) occur in the vacancy of the particle instead of at the interface. Meanwhile, it is supposed that this reaction is irreversible and first-order. The kinetic equations of coke reactions for a single particle and the corresponding kinetic parameters are from the liable sources [11,19]. Assuming the rates of these reactions, R3 and R4, are very small and can be set as zero in the indirect reduction region, and are equal to R1 and R2, respectively, in the softening area and subzone 1 [20].
The unreacted core model with one interface also applies to calculating the flux decomposition reaction rate, R5 [11,21]. The rate equations of water gas shift reaction, R6, and direct reaction, R7, are determined by the equation proposed by related researchers [11,12].
2.5. Computational Algorithm
To solve the governing equations faces a boundary-value problem, thus, it is necessary to determine the boundary conditions. The values of 9 process variables mentioned above at the stockline of the BF serve as the upper boundary conditions which are listed in Table 3, among them the temperature and components of gas are preset, reaction degrees are all zero and H2O is not listed because it is not measured in practice but can be calculated by Equation (7). The chemical composition of iron ore is listed in Table 4. The lower boundary conditions are the components and temperature of gas at tuyere level from the literature [11], as shown in Table 5 Other parameters in Table 3 which are also from the two sets of data mentioned above are employed as input data. The ratio of ore, fluxes and coke charged in two furnaces can be calculated from the data in Table 3, which are 182:1:52 and 44:1:12 respectively.

Table 3.
Operating parameters of two reference BFs.

Table 4.
Chemical composition of iron ore.

Table 5.
Gas composition and temperature at the tuyere level of two reference BFs.
After all upper and lower boundary conditions are determined, the process variables of the BF are calculated through solving the nonlinear ODEs in Section 2.3. The calculation process is that inputting the initial operating data at the stock line and tuyere level first, then the ODEs are integrated from the stockline to the tuyere level. However, the input gas information at the stockline is missing as mentioned, while gas data at the tuyere level is known. Therefore, this leads to a two-point boundary problem. The nonlinear ODEs under the two-point boundary conditions is solved by means of shooting method. To be specific, this method is to assume the unknown gas data in the upper boundary conditions, and then integrate the ODEs to get the values of Tg and Yi at the tuyere level through employing the fourth-order Runge-Kutta approach [22]. To determine the input gas data in the upper boundary conditions, a multi-variable least-square optimization scheme is used with the objective of minimizing the difference between the calculated Tg and Yi at the tuyere level and the corresponding given lower boundary values. The loop of the calculation ends if the difference is minimal, otherwise the integration cycle will run again, moreover, the assumed gas data in the upper boundary conditions will be re-estimated on the occasion. Once the loop ends, the preset values in the final iteration, namely, the upper boundary values of Tg and Yi turn into what this model wants, meanwhile, other process variables are obtained.
2.6. Model Validation
The validation of this model is carried out through comparing the top gas composition and temperature in the literature [11] with the calculation results as shown in Table 6. The small error values in the table are noted between them, reflecting the high accuracy of the model.

Table 6.
Comparisons between the calculated and measured parameters of two reference BFs.
3. Results and Discussion
3.1. Baseline Case
The operation data are from Algoma’s No.7 furnace. The calculated gas utilization efficiency is 0.535. The direct reduction is not discussed, since the focus of current work is on the gas utilization. The distributions of the volume fraction of CO and H2, as well as solid temperature and indirect reduction degree in the indirect region are shown in Figure 2. As the CO reaction rate of FeO reduction is low at the beginning, the increase in volume fraction of CO along the axis of furnace becomes slow, which corresponds to the sudden decrease in the slope of curve. As for reduction with H2, when FeO reduction begins, the change in the slope of H2 volume fraction curve is not obvious resulting from the high reduction reaction rate. Due to low H2 volume fraction in this simulation, the indirect reduction degree is mainly affected by CO reduction. Hence, in this case, the slope of indirect reduction degree curve also suddenly decreases when FeO reduction starts. The reason for the slope of solid temperature curve does not change in the initial stage of FeO reduction is that the exothermic heat of this reaction is insufficient to change the trend of curve. The result of the indirect reduction region height is 18.3 m and the starting temperature of coke solution loss reaction is 1147 K in the baseline case.
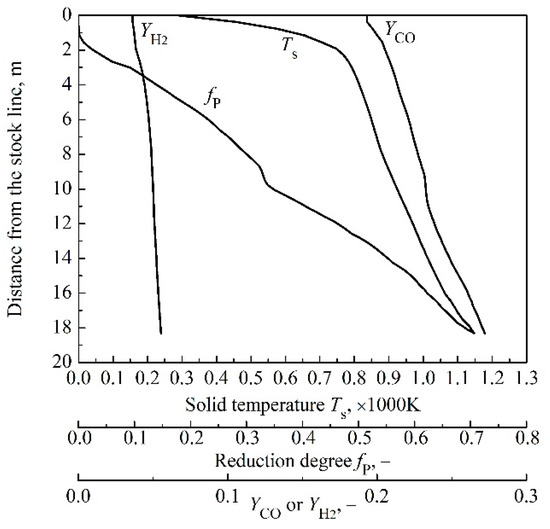
Figure 2.
Baseline case results: Volume fraction of CO, volume fraction of H2, solid temperature, ore reduction degree in indirect reduction region.
3.2. Influence of Coke Reactivity
In reality, many kinetic parameters, such as porosity, reaction rate constant and effective diffusivity and so on, can impact the iron-ore reducibility and coke reactivity. Therefore, these parameters should be simplified through a modified reaction rate expression to reflect the reducibility of iron ore and reactivity of coke for simulation. In this paper, the simplification has been conducted to set a series of reaction rate factors to represent the changes of the reactivity of coke and reducibility of iron ore. They are known as the reaction rate factors α and β to modify the overall reaction rates (R’ = αR and R’ = βR). For the baseline case, α = 1 and β = 1 are assumed for coke reactions and reduction reactions of iron ore.
The relationship between the gas utilization efficiency and coke reactivity is shown in Figure 3. Because that the thermodynamic force of the reduction of FeO with CO is improved with a higher coke reactivity, the gas utilization efficiency should have been increased from the thermodynamic aspect. However, as can be seen in the figure, the utilization efficiency of gas increases at the beginning, but decreases later when the reactivity of coke increases continuously. The reason is that a higher decrease in the temperature of TRZ deteriorates the environment of reduction kinetics, which in turn, reduces the utilization efficiency of gas. To be specific, due to the decrease in TRZ temperature, the reduction rate slows down and the lower edge of the TRZ moves up, which decreases the height of indirect reduction region and the time of indirect reduction. As a result, the gas utilization efficiency increases first and then decreases.
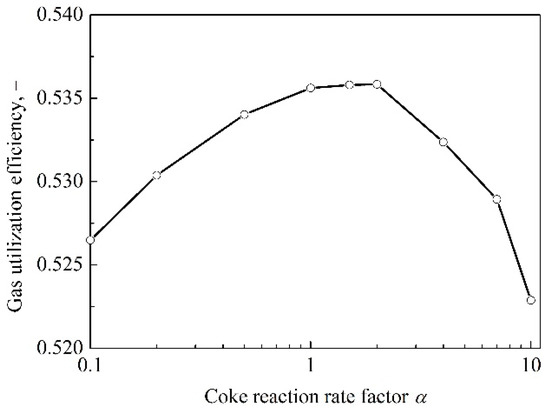
Figure 3.
The relationship between the gas utilization efficiency and coke reactivity.
In order to further analyze the reasons for the change of relationship between gas utilization efficiency and coke reactivity, the chemical energy utilization of gas for different coke reactivity and the relationship between driving force control of reduction reaction and coke reactivity in the indirect region are discussed through the concept of surplus gas reduction potential. The surplus gas reduction potential are calculated by difference between the reduction potential and the equilibrium reduction potential at corresponding temperature. The reduction of Fe2O3 is ignored in the discussion because Fe2O3 is quickly reduced to Fe3O4 and the temperature has little influence on the surplus reduction potential of this reaction. Since both the reduction of Fe3O4 and reduction of FeO with H2 are endothermic reactions, the decrease in temperature increases equilibrium volume fraction of reducing gas and decreases reduction potential of surplus reducing gas. However, for the reduction of FeO with CO, the influence of the temperature decrease on the equilibrium volume fraction and the reduction potential of surplus gas is opposite because this reaction is exothermic.
In order to correspond with the discussion on thermodynamics and kinetics of indirect reduction in the above analysis, the coke reaction rate factors, α = 0.1, 2 and 10, are selected. The distributions of the volume fraction of CO and H2, as well as solid temperature and indirect reduction degree in the indirect reduction region are described in Figure 4, respectively. The results show that as the coke reactivity increases, the starting temperature of coke solution loss reaction decreases from 1258 K to 1002 K and the height of indirect reduction region decreases from 21.3 m to 15.2 m. Moreover, increasing the coke reactivity decreases the volume fraction of H2 and the solid temperature as shown in Figure 4b,c. For the third reaction rate factor, the YCO and fP are least, meanwhile, the YCO and fP for the first reaction rate factor are greater compared with those for the second one until the reduction of FeO begins, and then the comparison results are opposite, as shown in Figure 4a,d. The change of slope of CO volume fraction curves and indirect reduction degree curves is caused by FeO reduction. The reason has been explained in Section 3.1.
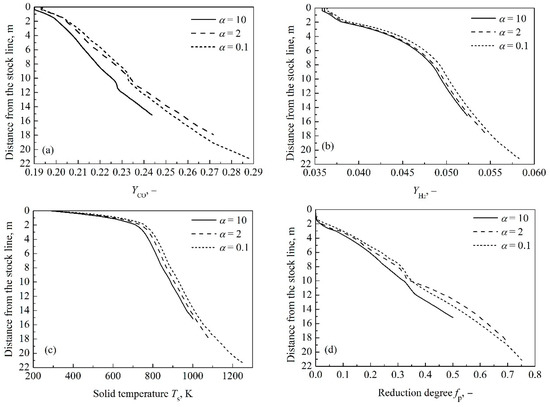
Figure 4.
Effect of coke reactivity on volume fraction distributions of CO (a) and H2 (b), temperature distribution (c) and reduction degree distribution (d) in indirect reduction region.
Figure 5 shows the reduction potential of surplus reducing gas for different coke reactivity. It can be seen from this figure that the reduction potential of surplus reducing gas decreases during the transformation from Fe3O4 to FeO with the increment of coke reactivity. This result implies that during this reduction stage, the surplus thermodynamic condition of reducing gas is weakened and the chemical energy utilization is increased, which indicate kinetics controls the reduction of Fe3O4. As shown in Figure 5a, as the coke reactivity increases, the reduction potential of surplus CO in FeO reduction increases first and then decreases. In other words, the order of the reduction potential of surplus CO under three coke reaction rate factors is as follows: Δφ2 > Δφ0.1 > Δφ10, Δφ represents the reduction potential of surplus CO, the number of subscript is the coke reaction rate factor. As shown in Figure 4a, the volume fraction of CO during FeO reduction increases when α changes from 0.1 to 2, which induces higher Δφ. It illustrates that the surplus thermodynamic condition of reducing gas is enhanced and the utilization of chemical energy is reduced with the increase of coke reactivity, and the reduction is controlled by thermodynamics. When α increases from 2 to 10, the equilibrium volume fraction of CO decreases and the volume fraction of CO decreases in the stage of FeO reduction, as shown in Figure 4a, finally resulting in that Δφ2 > Δφ10. It indicates the chemical energy utilization of gas increases, but also implies the kinetics becomes a dominant role in the reduction. According to Figure 5b, as the coke becomes higher reactive, the reduction potential of surplus H2 in FeO reduction decreases, which means the chemical energy utilization of gas increases and kinetics dominates the reduction.
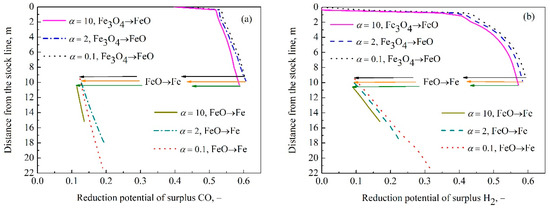
Figure 5.
The reduction potential of surplus CO (a) and H2 (b) under different coke reactivity in indirect reduction region.
According to the rate formulas introduced in Section 2.4, the reduction rate distributions under different coke reactivity in the indirect reduction region are obtained as shown in Figure 6. The average reduction rates of Fe3O4 are calculated for a better comparison. The results show that with the increase of coke reactivity, the average CO reduction rate decreases from 57.3 mol/s to 39.3 mol/s and H2 reduction rate decreases from 14.4 mol/s to 8.1 mol/s. It indicates that the temperature plays a great influence on the reaction rate and kinetics controls the reduction. As for the CO reduction of FeO in Figure 6a, when α increases from 0.1 to 2, the reaction rate increases at the same height because of Δφ2 > Δφ0.1, which indicates that the reduction potential has a greater impact on the reaction rate. It means the thermodynamics plays the leading role in the reduction. When α raises from 2 to 10, the leading role becomes kinetics because the reaction rate decreases at the same height which illustrates the temperature has a great effect on the reaction rate. It can be seen from Figure 6b, the reduction of FeO with H2 is controlled by kinetics because the reaction rate decreases at the same height as the coke reactivity increases, which illustrates temperature acts a key role in reduction.
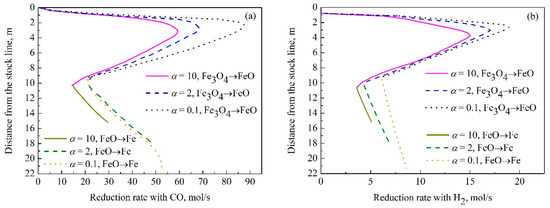
Figure 6.
Distributions of reduction rate with CO (a) and H2 (b) under different coke reactivity in indirect reduction region.
It is also concluded from the above analysis that the gas utilization efficiency of BF may not increase with the increase of coke reactivity if H2 content increases. Therefore, the improvement of gas utilization efficiency cannot be achieved by applying high-reactivity coke in the scenario that volume fraction of H2 is high.
An indirect reduction is mainly affected by CO reduction as mentioned, so the relationship between reduction degree and coke reactivity shown in Figure 4d is similar to that between CO reduction rate and coke reactivity. The reason lies in the fact that volumetric rate of solids descent is fixed in this model, which indicates that the reaction time is the same within the same height. This leads to a conclusion that the greater the reaction rate, the greater the degree of reduction in the same height. Therefore, it can be inferred from Figure 4d that the reduction process is controlled by thermodynamics first, and then by kinetics.
3.3. Influence of Iron Ore Reducibility
As mentioned above, iron ore reducibility plays an important role in the gas utilization efficiency. Therefore, it is necessary to study the effect of iron ore reducibility on the gas utilization efficiency. In Figure 7, it can be seen that the effect of iron ore reducibility on the gas utilization efficiency is consistent with common sense, namely, it is a positive effect. When the reactivity of coke remains constant, i.e., α = 1, with the improvement of iron ore reducibility, the gas utilization efficiency increases. However, it can also be observed that the increment of the gas utilization efficiency is limited when β is larger than 1. The reason lies in the fact that the gas utilization efficiency is also affected by other factors, such as the height of indirect reduction region, which remains unchanged when the coke reactivity remains stable.
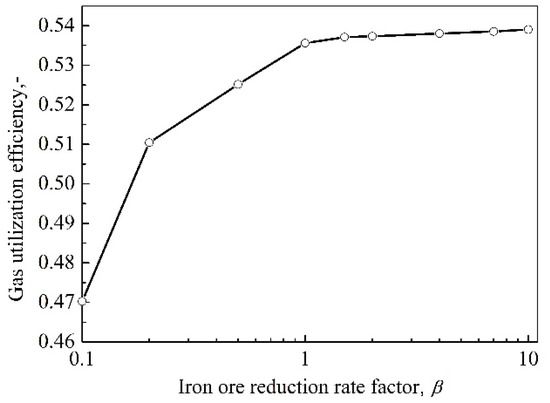
Figure 7.
The relationship between the gas utilization efficiency and iron ore reducibility.
The effects of coke reactivity and iron ore reducibility on the gas utilization efficiency at top of the BF were discussed above. The results demonstrate that simply increasing coke reactivity does not necessarily improve the gas utilization efficiency of the BF, so it is necessary to discuss the combination of coke and iron ore in order to obtain the maximum gas utilization efficiency. The results are shown in Figure 8, taking β = 0.1, 1, 10 as an example.
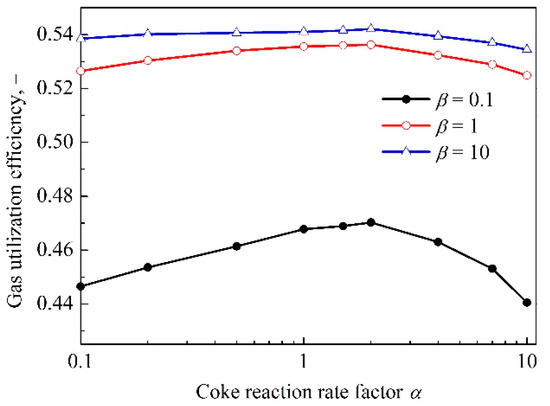
Figure 8.
The relationship between the gas utilization efficiency and coke reactivity under different iron ore reducibility.
In Figure 8, as a whole, the gas utilization efficiency decreases after increasing with the increase of coke reactivity. The reason has been illustrated above and is not repeated here. Moreover, overall, the curves of the gas utilization efficiency are convex and they become more gentle with the increment of reducibility of iron ore under the current operating conditions. This can be explained by the reason that when the iron ore reducibility increases to a certain extent, the deterioration of kinetic reduction condition, caused by the decrease in the TRZ temperature and height of indirect reduction region will be alleviated. In addition, it can be observed that although the combination of highly reducible iron ore and both highly and lowly reactive coke can get high gas utilization efficiency [3,10,23], the lowly reducible iron ore cannot be ignored, either from the actual energy situation or from the economic point of view. For example, as shown in Figure 8, lowly reducible iron ore, i.e., β = 0.1, combining with coke with reactivity of 2, i.e., α = 2, can obtain optimal gas utilization efficiency.
4. Conclusions
A mathematical model of the BF has been presented, and the calculated temperature and components of gas at the top of the BF agree quite well with the reported values from literature. Further, the model has been used to gain a thorough understanding of the effects of coke reactivity and iron-ore reducibility on the gas utilization efficiency of the BF. Under the current operating conditions, the following main conclusions can be drawn.
- The height of indirect reduction region decreases from 21.3 m to 15.2 m and the starting temperature of coke solution loss reaction decreases from 1258 K to 1002 K with the increase of coke reactivity.
- The utilization efficiency of gas at top of the furnace increases first and then decreases with the increment in the coke reactivity.
- As the coke reactivity increases, the gas utilization efficiency may not increase in case of high H2 content.
- The reducibility of iron ore is proportional to the gas utilization efficiency.
- Either lowly or highly reactive coke needs to combine with highly reducible iron ore to make sure that the indirect reduction region is fully utilized and the gas utilization efficiency of the furnace is optimal.
- High gas utilization efficiency can be achieved by using lowly reducible iron ore with the appropriately reactive coke.
Author Contributions
Conceptualization, F.M. and Z.Z.; methodology, F.M. and Z.Z.; software, F.M. and L.S.; validation, F.M. and L.S.; writing-original draft preparation, F.M.; writing-review and editing, Z.Z. and L.S.; supervision, Z.Z. and L.S.; project administration, Z.Z. and L.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by National Natural Science Foundation of China (No. 51574064).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Nomura, S.; Matsuzaki, S.; Naito, M.; Ayukawa, H.; Koizumi, S.; Abe, T.; Kitaguchi, H.; Tahara, T. Improvement in blast furnace reaction efficiency through the use of catalyst-doped highly reactive coke. Shinnittetsu Giho 2006, 384, 100. [Google Scholar]
- Shimoyama, I. Research prospect on coke reactivity aiming at low-RAR blast furnace operation. Tetsu Hagané 2010, 96, 25–33. [Google Scholar] [CrossRef]
- Kasai, A.; Matsui, Y. Lowering of thermal reserve zone temperature in blast furnace by adjoining carbonaceous material and iron ore. ISIJ Int. 2004, 44, 2073–2078. [Google Scholar] [CrossRef]
- Nomura, S.; Ayukawa, H.; Kitaguchi, H.; Tahara, T.; Matsuzaki, S.; Naito, M.; Koizumi, S.; Ogata, Y.; Nakayama, T.; Abe, T. Improvement in blast furnace reaction efficiency through the use of highly reactive calcium rich coke. ISIJ Int. 2005, 45, 316–324. [Google Scholar] [CrossRef]
- Naito, M.; Okamoto, A.; Yamaguchi, K.; Yamaguchi, T.; Inoue, Y. Improvement of blast furnace reaction efficiency by the temperature control of thermal reserve zone. Shinnittetsu Giho 2006, 384, 95. [Google Scholar]
- Natsui, S.; Shibasaki, R.; Kon, T.; Ueda, S.; Inoue, R.; Ariyama, T. Effect of high reactivity coke for mixed charge in ore layer on reaction behavior of each particle in blast furnace. ISIJ Int. 2013, 53, 1770–1778. [Google Scholar] [CrossRef]
- Nomura, S.; Higuchi, K.; Kunitomo, K.; Naito, M. Reaction behavior of formed iron coke and its effect on decreasing thermal reserve zone temperature in blast furnace. ISIJ Int. 2010, 50, 1388–1395. [Google Scholar] [CrossRef]
- Babich, A.; Senk, D.; Gudenau, H.W. Effect of coke reactivity and nut coke on blast furnace operation. Ironmak Steelmak 2009, 36, 222–229. [Google Scholar] [CrossRef]
- Ueda, S.; Yanagiya, K.; Watanabe, K.; Murakami, T.; Inoue, R.; Ariyama, T. Reaction model and reduction behavior of carbon iron ore composite in blast furnace. ISIJ Int. 2009, 49, 827–836. [Google Scholar] [CrossRef]
- Naito, M.; Okamoto, A.; Yamaguchi, K.; Yamaguchi, T.; Inoue, Y. Improvement of blast furnace reaction efficiency by use of high reactivity coke. Tetsu Hagané 2001, 87, 357–364. [Google Scholar] [CrossRef]
- Acevedo, U. Mathematical Modelling and Control of the Blast Furnace Process. Ph.D. Thesis, Perdue University, Michigan, USA, 1981. [Google Scholar]
- Yagi, J.; Muchi, I. Improved mathematical model for estimating process variables in blast furnace. Trans. ISIJ 1970, 10, 181–187. [Google Scholar] [CrossRef]
- Muchi, I.; Yagi, J.; Tamura, K.; Moriyama, A. Reaction kinetics in the blast furnace. J. Jpn. Inst. Met. Mater. 1966, 30, 826–831. [Google Scholar] [CrossRef]
- Yagi, J.; Sasaki, K.; Muchi, I. Theoretical investigation on the blast furnace operations with the aid of mathematical model. Tetsu Hagané 1968, 54, 1019–1031. [Google Scholar] [CrossRef][Green Version]
- Smith, J.M.; Van, N.H.C. Introduction to Chemical Engineering Thermodynamics, 3rd ed.; McGraw-Hill: New York, NY, USA, 1975; p. 107. [Google Scholar]
- Tsay, Q.T.; Ray, W.H.; Szekely, J. The modeling of hematite reduction with hydrogen plus carbon monoxide mixtures: Part I. The behavior of single pellets. AIChE J. 1976, 22, 1064–1072. [Google Scholar] [CrossRef]
- McKewan, W.M. Reduction kinetics of magnetite in hydrogen at high pressures. Trans. Metall. Soc. AIME 1962, 224, 387–393. [Google Scholar]
- Murayama, T.; Ono, Y.; Kawai, Y. Step-wise reduction of hematite pellets with CO-CO2 gas mixtures. Tetsu Hagané 1977, 63, 1099–1107. [Google Scholar] [CrossRef][Green Version]
- Miyasaka, N.; Kondo, S.I. The rate of cokes gasification by gas consisting of CO2, H2O, CO, H2 and N2. Tetsu Hagané 1968, 54, 1427–1431. [Google Scholar] [CrossRef]
- Bi, X.G.; Krister, T.; Olle, W. Simulation of the blast furnace process by a mathematical model. ISIJ Int. 1992, 32, 470–480. [Google Scholar] [CrossRef]
- Asaki, Z.; Fukunaka, Y.; Nagase, T.; Kondo, Y. Thermal decomposition of limestone in a fluidized bed. Metall. Trans. 1974, 5, 381–390. [Google Scholar] [CrossRef]
- Kosti, A.A.; Anastassi, Z.A.; Simos, T.E. An optimized explicit runge-kutta method with increased phase-lag order for the numerical solution of the schrödinger equation and related problems. J. Math. Chem. 2010, 47, 315. [Google Scholar] [CrossRef]
- Kasai, A.; Toyota, H.; Nozawa, K.; Kitayama, S. Reduction of reducing agent rate in blast furnace operation by carbon composite iron ore hot briquette. ISIJ Int. 2011, 51, 1333–1335. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).