Summary
A 62-year-old patient was admitted to our hospital because of severe fatigue, headache, arthralgias, myalgias and an unsteady gait. She presented with a reduced general condition and splinter haemorrhages of the fingers. Due to a known history of asthma, signs of eosinophilia, sinusitis, endomyocardial thrombotic apposition in both chambers and neurological deterioration, the diagnosis of multisystem vasculitis (Churg-Strauss syndrome) was made. The symptoms resolved after steroid therapy. Churg-Strauss syndrome is a rare necrotising systemic inflammatory vasculitis of the small and medium-size vessels. Typically, the disease has three distinct stages, which can possibly involve several organic systems (e.g., pulmonary, cardial, renal and nervous system). The treatment for Churg-Strauss syndrome includes steroids and/or other immunosuppressive drugs. After introduction of steroid therapy, the prognosis of the disease improves significantly, but heart failure remains as a major determinant of mortality.
Case report
A 62-year-old female with chest pain was referred for acute coronary syndrome. ECG at admission showed new ECG-changes (T-inversion, ST-segment depression) and laboratory testing revealed a positive troponin. The patient underwent coronary angiography, which ruled out a significant coronary artery disease. Later on, the patient also complained about general fatigue, dizziness and of having an unsteady gait for several weeks. Moreover, a few days before admission, she had noticed diffuse myalgia, arthralgia and cephalgia, whereas fever, night sweats, as well as gastrointestinal signs and symptoms were denied. The physical examination showed an afebrile patient with normal vital signs, specifically with a blood pressure of 142/80 mm Hg and a regular heart rate of 56 bpm. She was, however, somnolent with reduced psychomotor functions and a general weakness, yet without focal neurological signs. In addition, the examination of the upper extremities revealed splinter haemorrhages. The patient’s past medical history included asthma, which was treated with 50 µg salmeterol and 250 µg fluticasone via inhaler per day (Seretide Diskus®) and 100 µg salbutamole via inhaler on demand.
Laboratory tests showed an elevated NTpro-BNP level (4280 ng/l) and the white blood cell count revealed transient hypereosinophilia (Table 1). Immunological testing revealed unspecifically elevated ANA of 1:80. The antineutrophil cytoplasmatic antibodies (ANCA) were negative. Only immunoglobulin E (IgE) was positive (187 kU/l). Echocardiography suggested endomyocardial fibrosis with involvement of both chambers (Figure 1). A mural apposition thrombus could be detected on the apical wall segment and the inferior midventricular wall segment with hypokinesia. Left ventricular ejection fraction was preserved (60%). Mitral inflow pattern and pulmonary vein flow variables suggested first grade diastolic dysfunction. The right ventricle showed only apical involvement. A small pericardial effusion without any haemodynamic relevance was noted. The mitral valve leaflets were normal, but the chordae tendineae were thickened.
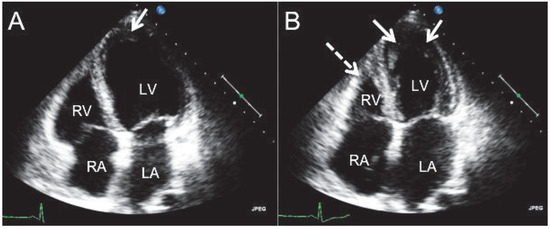
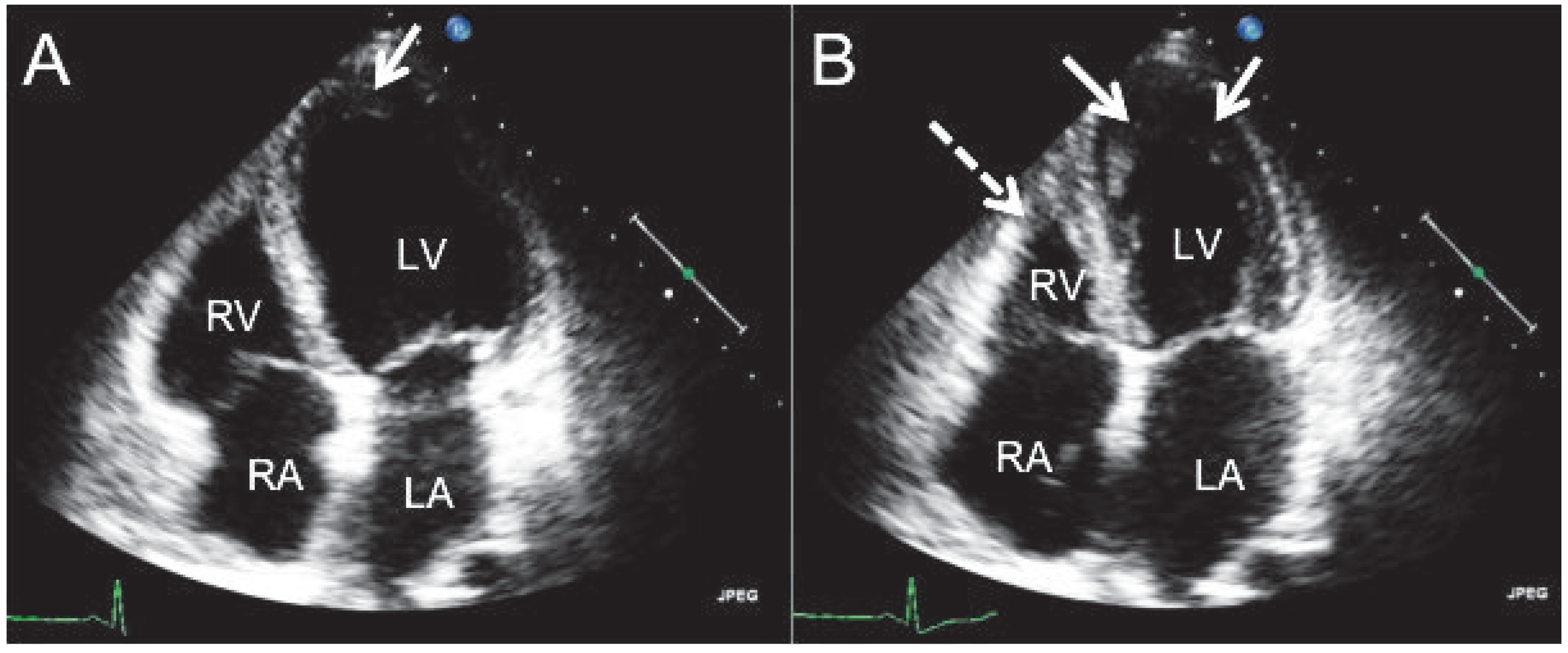
Figure 1.
The transthoracic apical 4-chamber view shows endomyocardial fibrosis in enddiastole (A) and endsystole (B). The apposition is on the apical wall segment and the inferior midventricular wall segment of the left ventricle (arrows). The right ventricle shows apical involvement (broken arrow). RA = right atrium; RV = right ventricle; LA = left atrium; LV = left ventricle.
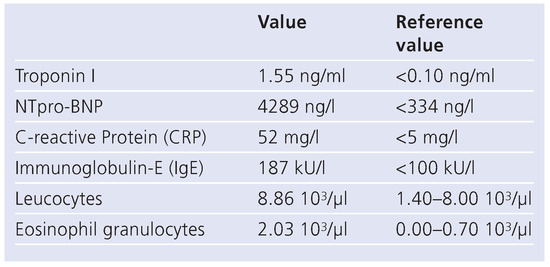
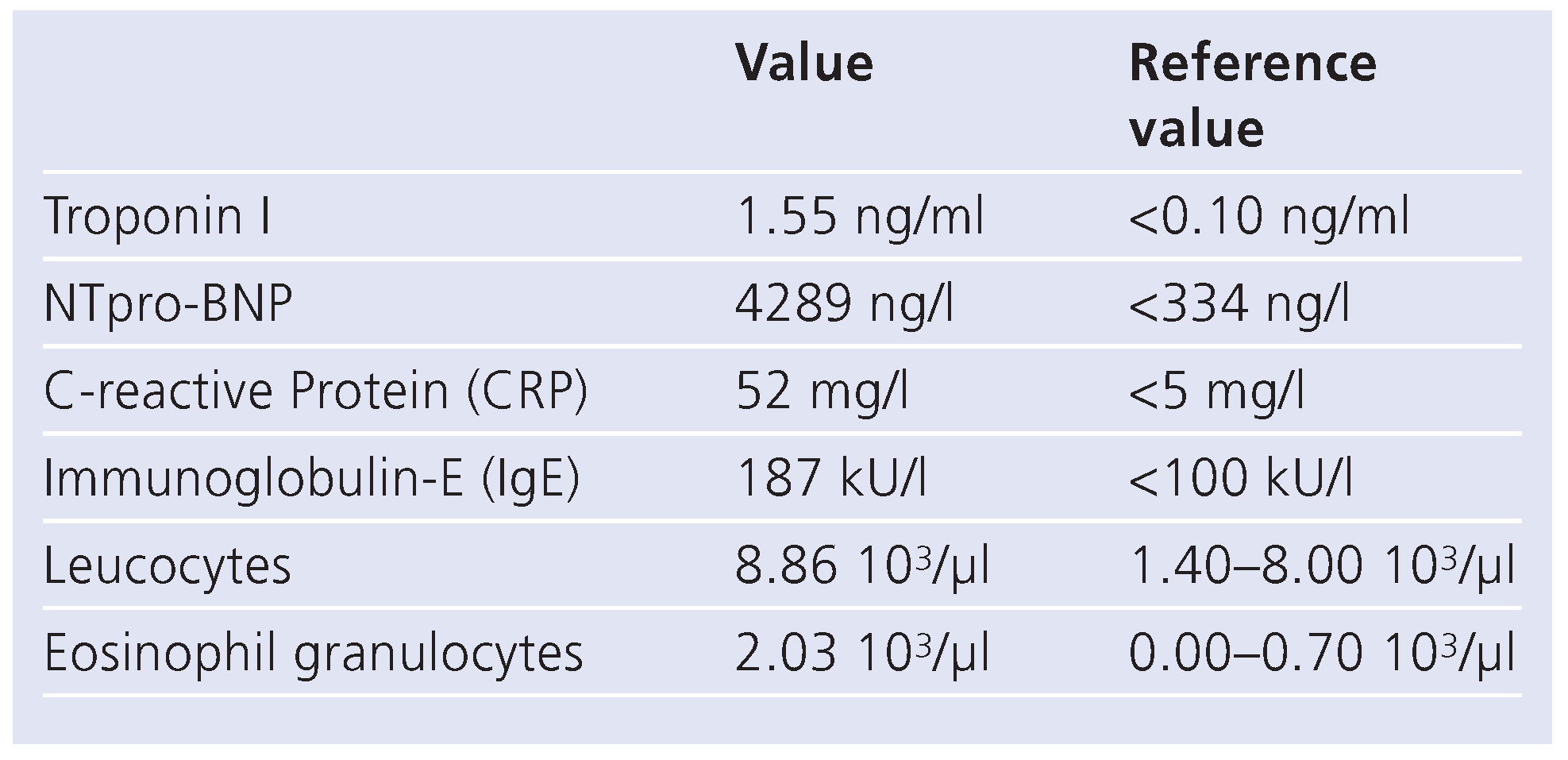
Table 1.
Laboratory results.
Table 1.
Laboratory results.
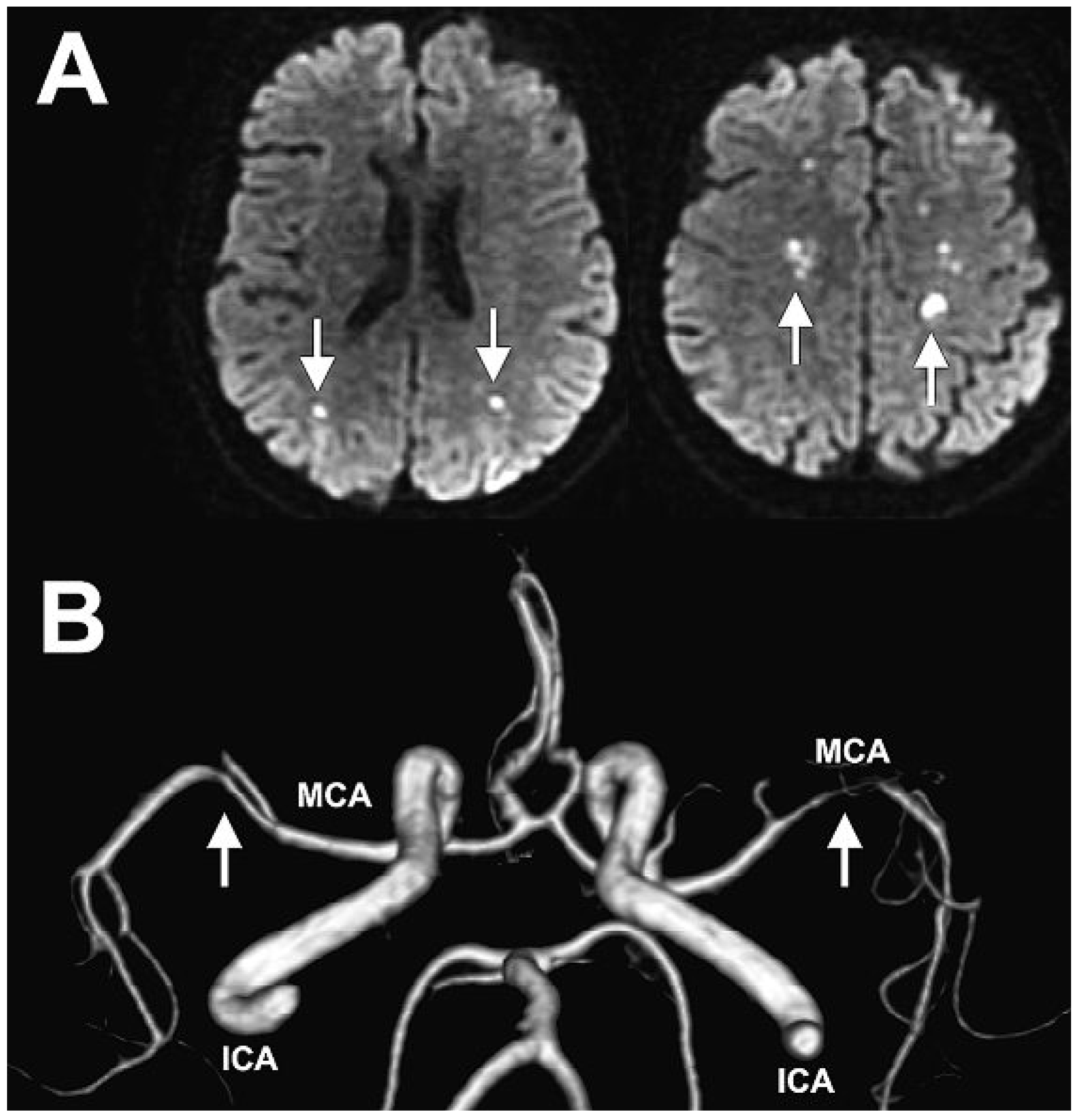
A CT-scan of the brain was performed for further evaluation of the neurological symptoms and showed pansinusitis and multifocal vascular lesions in the brain. The diffusion-weighted MRI sequences revealed multiple small acute brain infarcts (Figure 2A) and the MR angiography showed extensive stenosis of both middle cerebral arteries (Figure 2B).
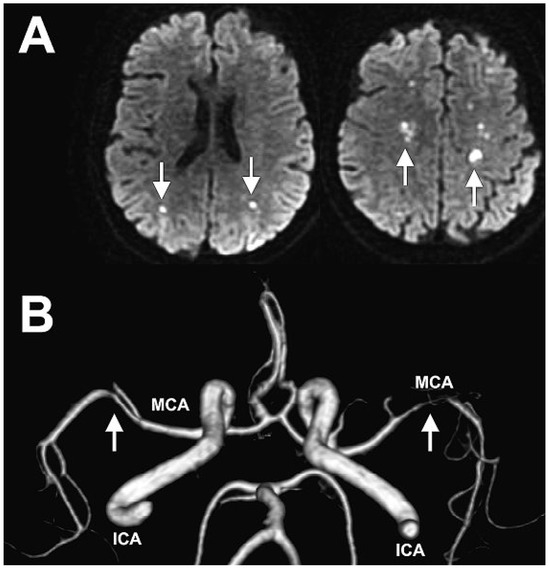
Figure 2.
A Multiple small acute brain infarcts (white arrows) on diffusion-weighted imaging(DWI) sequences. B MR angiography showing extensive stenosis of both middle cerebral arteries(white arrows). ICI = internal carotid artery; MCA = middle cerebral artery.
Given the eosinophilia, sinusitis, endomyocardial fibrosis and neurologic disease, we made the diagnosis of multisystem vasculitis or Churg-Strauss-like syndrome and commenced steroid therapy (initial solumedrol 1 g i.v. for 3 days, then prednison 1 mg/kg p.o.). The eosinophilia, however, decreased before the steroid therapy was initiated. After the start of the immunosuppressive therapy, symptoms reduced and the patient was discharged from hospital.
Discussion
Jacob Churg and Lotte Strauss from the Division of Pathology at Mount Sinai Hospital in New York were the first to publish a series of 13 autopsy cases with the clinical symptoms of severe asthma, fever and hypereosinophilia, accompanied by vascular embarrassment in various organ systems, in 1951 [1]. The Churg-Strauss syndrome is characterised by pulmonary and systemic small-vessel necrotising vasculitis, eosinophilia and tissue infiltration by eosinophils, occurring in individuals with asthma and often allergic rhinitis or sinus polyposis [2]. The disease is rare with an annual incidence ranging between 0.5 and 6.8 per million inhabitants [3,4], according to location and the classification criteria used, and a prevalence of between 10.7–13 per million inhabitants [5,6]. In autopsy studies, cardiac involvement has been reported in up to 60% of patients with Churg-Strauss syndrome [1] and appeared to be the major reason of death in up to 48% of cases [1,7]. The frequency of cardiac involvement according to the literature varies between 15 and 56% and reached 39% in the largest series of 112 patients reported by Sable et al. [8]. However, many different types of cardiac involvement have been described, including endomyocardial fibrosis, eosinophilic myocarditis (13–25%) leading to heart failure (17%), coronaritis, isolated tachycardia, conduction abnormalities or other arrhythmias, valvular heart disease and pericarditis (8–35%), as well as intracavitary thrombi [9,10]. While coronary arteries may be involved in the disease process, in the present patient epicardial coronary artery involvement was excluded by angiography. Rat heart cell models have demonstrated deleterious effects of eosinophilic cationic protein and eosinophil major basic protein on plasma membranes and mitochondrial respiration [10,11]. Intracardiac thrombosis might be promoted through the loss of endothelial integrity as well as substances with prothrombotic qualities released during the degranulation process [10,11]. Reliable noninvasive investigations able to detect coronary involvement are not yet standardised, but cardiac magnetic resonance (CMR) imaging may become a novel diagnostic modality [12]. Indeed, CMR may reveal subclinical lesions, inflammatory infiltration of the myocardium and pericardial effusion [9]. Major advantages are the improved visualisation of thrombi and different T1-weighted relaxation properties of the myocardium and thrombi, which allows easier distinction [10,13]. Studies comparing the transthoracic echocardiography (TTE) with CMR showed an advantage for the latter in terms of thrombus detection with a sensitivity of 88% versus 23% for TTE, and a specificity of 99% versus 96% for TTE [14]. Delayed enhancement on CMR has proven useful in identifying the presence and extent of myocardial scar and areas of inflammation, especially in the case of early vasculitis or myocarditis [15,16].
In patients presenting with heart failure, Churg-Strauss syndrome as well as other systemic vasculitides (e.g., Giant cell arteritis) and hypersensitivity reactions (e.g., hypersensitivity vasculitis) are important differential diagnoses to be considered. A histological diagnosis on endomyocardial biopsy may guide therapy and may lead to improved prognosis [17].
Skin lesions, as seen in our patient and occurring in 40–75% of patients, and palpable (necrotic) purpura on the lower extremities are the most frequent skin manifestations [2]. Other important manifestations of the disease include digestive tract symptoms and kidney disease. Neurological involvement is also very common. In case series, up to 50–78% of patients present with peripheral neuropathy, essentially mononeuritis multiplex [2] and although not very common, CNS manifestations were reported in up to 39% of cases in the series by Reid and colleagues [18].
Pathophysiologically, Churg-Strauss syndrome is characterised by activated circulating eosinophilies and a nonspecific elevation of immunoglobulin E (IgE) levels in 75% of patients [2]. Antineurophil cytoplasmatic antibodies (ANCA) can be detected in 38–50% of the patients, whereas 75–81% of these patients have autoantibodies with a perinuclear immunofluorescentlabelling pattern, most frequently (i.e., in 92–100% of the patients) specific to myeloperoxidase (MPO) [2]. Lanham and colleagues [19] divided the clinical course of the disease into three, usually successive phases: the prodromic phase (phase 1; asthma and often allergic manifestations), followed by phase 2, resulting from eosinophilic infiltration into tissues, particularly the lung, myocardium and/or gastrointestinal tract with or without granulomas, and the systemic phase (or phase 3), with the development of necrotising vasculitis, usually occurring after 3–4 years of asthma, and affecting the skin, peripheral nerves and kidneys in particular [2]. Retrospective studies suggest that patients with ANCA-positive Churg-Strauss syndrome differ clinically significantly from those without ANCAs [2]. While ANCA-negative patients have higher rates of cardiac involvement, pericarditis and cardiomyopathy, pleural effusion, fever and livedo, ANCA-positive patients have more renal involvement, sinusitis, peripheral neuropathy and purpura [2]. Most importantly, mortality rates and relapse rates at 3–5 years were similar regardless of ANCA status at diagnosis [2]. Inhaled allergens, vaccinations, desensitisation and/or infections (parasitic or bacterial) have been implicated in triggering some cases of Churg-Strauss syndrome, but it has not been possible to identify any common antigen yet [2]. However, drugs such as macrolides, carbamazepine and quinine, as well as leukotriene receptor antagonists (zafirlukast, montelukast or pranlukast) have been associated with the disease [2]. Indeed, leukotriene modifiers usually provide the opportunity for substantial tapering or withdrawing of corticosteroids, which in turn may unmask an underlying and previously incomplete disease and allow it to develop fully [2]. The prognosis of Churg-Strauss syndrome has improved dramatically since the introduction of corticosteroids and, when indicated, immunosuppressants. Remission is usually achieved rapidly in more than 80%, while relapses may occur in 15–43% of patients [2]. The overall 5-year survival rate is about 90%, while 75% of the deaths are directly attributable to vasculitis, with cardiac involvement being the primary cause. Poor prognostic factors are proteinuria (>1 g/d), renal functional impairment (creatinine >140 µmol/l), cardiomyopathy, gastrointestinal tract involvement and central nervous system involvement.
Therapy is usually initiated with high doses of corticosteroids (prednisone 1 mg/kg/d) or methylprednisolone (15 mg/kg intravenously, for 1–3 days) for more severe forms. As soon as patients improve clinically and inflammatory markers return to normal, corticosteroid tapering can be initiated, but it is often impossible to discontinue corticosteroids due to residual asthma, leaving most patients with maintenance dosages of 10 mg/d and/or inhaled corticosteroids [2].
References
- Churg, J.; Strauss, L. Allergic granulomatosis, allergic angiitis, and 0periarteritis nodosa. Am J Pathol. 1951, 27, 277–301. [Google Scholar] [PubMed]
- Pagnoux, C.; Guilpain, P.; Guillevin, L. Churg-Strauss syndrome. Curr Opin Rheumatol. 2007, 19, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Watts, R.A.; Lane, S.; Scott, D.G. What is known about the epidemiology of the vasculitides? Best Pract Res Clin Rheumatol. 2005, 19, 191–207. [Google Scholar] [CrossRef]
- Martin, R.M.; Wilton, L.V.; Mann, R.D. Prevalence of Churg-Strauss syndrome, vasculitis, eosinophilia and associated conditions: retrospective analysis of 58 prescription-event monitoring cohort studies. Pharmacoepidemiol Drug Saf. 1999, 8, 179–189. [Google Scholar] [PubMed]
- Mahr, A.; Guillevin, L.; Poissonnet, M.; Ayme, S. Prevalences of polyarteritis nodosa, microscopic polyangiitis, Wegener’s granulomatosis, and Churg-Strauss syndrome in a French urban multiethnic population in 2000: a capture-recapture estimate. Arthritis Rheum. 2004, 51, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Haugeberg, G.; Bie, R.; Bendvold, A.; Larsen, A.S.; Johnsen, V. Primary vasculitis in a Norwegian community hospital: a retrospective study. Clin Rheumatol. 1998, 17, 364–368. [Google Scholar] [CrossRef]
- Hellemans, S.; Dens, J.; Knockaert, D. Coronary involvement in the Churg-Strauss syndrome. Heart. 1997, 77, 576–578. [Google Scholar] [CrossRef][Green Version]
- Sable-Fourtassou, R.; Cohen, P.; Mahr, A.; et al. Antineutrophil cytoplasmic antibodies and the Churg-Strauss syndrome. Ann Intern Med. 2005, 143, 632–638. [Google Scholar] [CrossRef]
- Vinit, J.; Bielefeld, P.; Muller, G.; et al. Heart involvement in ChurgStrauss syndrome: Retrospective study in French Burgundy population in past 10years. Eur J Intern Med. 2010, 21, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Bhagirath, K.M.; Paulson, K.; Ahmadie, R.; Bhalla, R.S.; Robinson, D.; Jassal, D.S. Clinical utility of cardiac magnetic resonance imaging in ChurgStrauss syndrome: case report and review of the literature. Rheumatol Int. 2009, 29, 445–449. [Google Scholar] [CrossRef] [PubMed]
- Spry, C.J.; Take, M.; Tai, P.C. Eosinophilic disorders affecting the myocardium and endocardium: a review. Heart Vessels Suppl. 1985, 1, 240–242. [Google Scholar] [CrossRef] [PubMed]
- Smedema, J.P.; van Paassen, P.; van Kroonenburgh, M.J.; Snoep, G.; Crijns, H.J.; Tervaert, J.W. Cardiac involvement of Churg Strauss syndrome demonstrated by magnetic resonance imaging. Clin Exp Rheumatol. 2004, 22 (Suppl. 36), S75–S78. [Google Scholar] [PubMed]
- Terasaki, F.; Hayashi, T.; Hirota, Y.; et al. Evolution to dilated cardiomyopathy from acute eosinophilic pancarditis in Churg-Strauss syndrome. Heart Vessels. 1997, 12, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Lipke, C.; Katoh, M.; Franke, A.; Krombach, G.; Buecker, A.; Kuhl, H.P. The value of non-contrast harmonic transthoracic echocardiography for the detection of left ventricular thrombi in patients with cardiomyopathy: comparison with contrast-enhanced magnetic resonance imaging. Int J Cardiovasc Imaging. 2007, 23, 479–487. [Google Scholar] [CrossRef] [PubMed]
- De Cobelli, F.; Pieroni, M.; Esposito, A.; et al. Delayed gadolinium-enhanced cardiac magnetic resonance in patients with chronic myocarditis presenting with heart failure or recurrent arrhythmias. J Am Coll Cardiol. 2006, 47, 1649–1654. [Google Scholar] [CrossRef] [PubMed]
- Elliott, M.D.; Kim, R.J. Late gadolinium cardiovascular magnetic resonance in the assessment of myocardial viability. Coron Artery Dis. 2005, 16, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.T.; Baughman, K.L.; Feldman, A.M.; et al. The role of endomyocardial biopsy in the management of cardiovascular disease: a scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology Endorsed by the Heart Failure Society of America and the Heart Failure Association of the European Society of Cardiology. Eur Heart J. 2007, 28, 3076–3093. [Google Scholar] [PubMed]
- Reid, A.J.; Harrison, B.D.; Watts, R.A.; Watkin, S.W.; McCann, B.G.; Scott, D.G. Churg-Strauss syndrome in a district hospital. QJM. 1998, 91, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Lanham, J.G.; Elkon, K.B.; Pusey, C.D.; Hughes, G.R. Systemic vasculitis with asthma and eosinophilia: a clinical approach to the Churg-Strauss syndrome. Medicine (Baltimore). 1984, 63, 65–81. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the author. Attribution - Non-Commercial - NoDerivatives 4.0.