Association Between Blood Benzene Levels and Periodontal Disease in a Nationally Representative Adult U.S. Population
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Eligibility Criteria
- Adults aged ≥ 30 years, consistent with NHANES eligibility for full-mouth periodontal examinations.
- Participants with complete periodontal clinical data, including probing depth (PD) and clinical attachment loss (CAL).
- Participants with measured blood benzene and serum cotinine levels.
- Individuals with available data on key covariates: age, sex, race/ethnicity, education level, occupation, diabetes status.
- Participants with missing data on periodontal status, blood benzene, or cotinine levels.
- Individuals with conditions requiring antibiotic prophylaxis prior to dental exams (excluded by NHANES protocols).
- Participants under 30 years old, since they were not eligible for the FMPE (Full-Mouth Periodontal Examination).
- Subjects with implausible biomarker values or incomplete demographic/medical history data.
2.3. Periodontal Examination
2.4. Definition of the Dependent Variable: Severe Periodontitis
2.5. Description of Independent Variable: Benzene Exposure
2.6. Cotinine: Mediator Variable
2.7. The Potential Confounding Variables
2.8. Statistical Methods
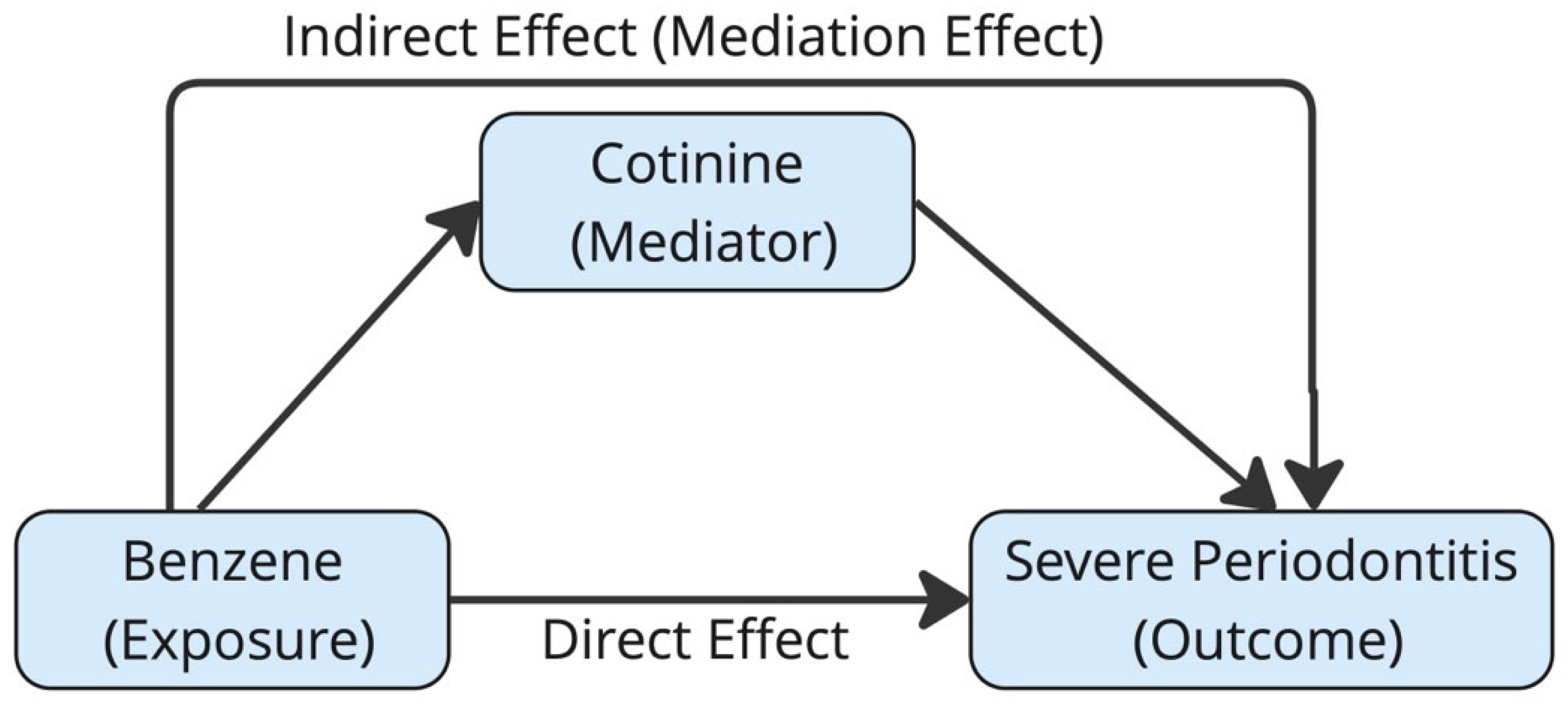
2.9. Structural Equation Modeling (SEM) Mediation Analysis
3. Results
4. Discussion
4.1. Limitation of the Research
4.2. Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Primers 2017, 3, 17038. [Google Scholar] [CrossRef] [PubMed]
- Kassebaum, N.J.; Bernabé, E.; Dahiya, M.; Bhandari, B.; Murray, C.J.; Marcenes, W. Global burden of severe periodontitis in 1990–2010: A systematic review and meta-regression. J. Dent. Res. 2014, 93, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Eke, P.I.; Thornton-Evans, G.O.; Wei, L.; Borgnakke, W.S.; Dye, B.A.; Genco, R.J. Periodontitis in US Adults: National Health and Nutrition Examination Survey 2009–2014. J. Am. Dent. Assoc. 2018, 149, 576–588.e6. [Google Scholar] [CrossRef]
- Nazir, M.A. Prevalence of periodontal disease, its association with systemic diseases and prevention. Int. J. Health Sci. (Qassim) 2017, 11, 72–80. [Google Scholar]
- Daalderop, L.A.; Wieland, B.V.; Tomsin, K.; Reyes, L.; Kramer, B.W.; Vanterpool, S.F.; Been, J.V. Periodontal disease and pregnancy outcomes: Overview of systematic reviews. JDR Clin. Trans. Res. 2018, 3, 10–27. [Google Scholar] [CrossRef]
- Meyle, J.; Chapple, I. Molecular aspects of the pathogenesis of periodontitis. Periodontology 2000 2015, 69, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Silva, N.; Abusleme, L.; Bravo, D.; Dutzan, N.; Garcia-Sesnich, J.; Vernal, R.; Hernandez, M.; Gamonal, J. Host response mechanisms in periodontal diseases. J. Appl. Oral Sci. 2015, 23, 329–355. [Google Scholar] [CrossRef]
- Shaddox, L.M.; Mullersman, A.F.; Huang, H.; Wallet, S.M.; Langaee, T.; Aukhil, I. Epigenetic regulation of inflammation in localized aggressive periodontitis. Clin. Epigenet. 2017, 9, 94. [Google Scholar] [CrossRef]
- Bahadar, H.; Mostafalou, S.; Abdollahi, M. Current understandings and perspectives on non-cancer health effects of benzene: A global concern. Toxicol. Appl. Pharmacol. 2014, 276, 83–94. [Google Scholar] [CrossRef]
- US Environmental Protection Agency. 2017 TRI National Analysis: Hazardous Air Pollutants; US Environmental Protection Agency: Washington, DC, USA, 2019. Available online: https://www.epa.gov/sites/default/files/2020-02/documents/2017_toxics_release_inventory_national_analysis_complete_report.pdf (accessed on 15 April 2023).
- Wallace, L. Environmental exposure to benzene: An update. Environ. Health Perspect. 1996, 104 (Suppl. S6), 1129–1136. [Google Scholar] [CrossRef]
- Weisel, C.P. Benzene exposure: An overview of monitoring methods and their findings. Chem. Biol. Interact. 2010, 184, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Brunnemann, K.D.; Kagan, M.R.; Cox, J.E.; Hoffmann, D. Analysis of 1,3-butadiene and other selected gas-phase components in cigarette mainstream and sidestream smoke by gas chromatography-mass selective detection. Carcinogenesis 1990, 11, 1863–1868. [Google Scholar] [CrossRef] [PubMed]
- Heibati, B.; Pollitt, K.J.; Karimi, A.; Charati, J.Y.; Ducatman, A.; Shokrzadeh, M.; Mohammadyan, M. BTEX exposure assessment and quantitative risk assessment among petroleum product distributors. Ecotoxicol. Environ. Saf. 2017, 144, 445–449. [Google Scholar] [CrossRef]
- Loomis, D.; Guyton, K.Z.; Grosse, Y.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Vilahur, N.; Mattock, H.; Straif, K. Carcinogenicity of benzene. Lancet Oncol. 2017, 18, 1574–1575. [Google Scholar] [CrossRef]
- Fustinoni, S.; Rossella, F.; Campo, L.; Mercadante, R.; Bertazzi, P.A. Urinary BTEX, MTBE and naphthalene as biomarkers to gain environmental exposure profiles of the general population. Sci. Total Environ. 2010, 408, 2840–2849. [Google Scholar] [CrossRef]
- Barreto, G.; Madureira, D.; Capani, F.; Aon-Bertolino, L.; Saraceno, E.; Alvarez-Giraldez, L.D. The role of catechols and free radicals in benzene toxicity: An oxidative DNA damage pathway. Environ. Mol. Mutagen. 2009, 50, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Badham, H.J.; Winn, L.M. In utero exposure to benzene disrupts fetal hematopoietic progenitor cell growth via reactive oxygen species. Toxicol. Sci. 2010, 113, 207–215. [Google Scholar] [CrossRef]
- Leite, F.R.; Nascimento, G.G.; Scheutz, F.; Lopez, R. Effect of smoking on periodontitis: A systematic review and meta-regression. Am. J. Prev. Med. 2018, 54, 831–841. [Google Scholar] [CrossRef]
- He, R.; Zhong, H.; He, C.; Li, H.; Wang, Z.; Zheng, J.; Li, G.; An, T. Individual and mixture effects of BTEX occupational exposure with hematologic and hepatic profiles in petrochemical workers and the metabolic mechanism. J. Environ. Sci. (China) 2025, 154, 163–173. [Google Scholar] [CrossRef]
- Aminoshariae, A.; Kulild, J.; Gutmann, J. The association between smoking and periapical periodontitis: A systematic review. Clin. Oral Investig. 2020, 24, 533–545. [Google Scholar] [CrossRef]
- Hajimiragha, H.; Ewers, U.; Brockhaus, A.; Boettger, A. Levels of benzene and other volatile aromatic compounds in the blood of non-smokers and smokers. Int. Arch. Occup. Environ. Health 1989, 61, 513–518. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, H.; Lin, J.; Liu, J.; Jiang, X.; Peng, L. Association between serum cotinine and volatile organic compounds (VOCs) in adults living with HIV, HBV, or HCV (NHANES 2005–2018). Sci. Rep. 2022, 12, 21769. [Google Scholar] [CrossRef] [PubMed]
- Eke, P.I.; Page, R.C.; Wei, L.; Thornton-Evans, G.; Genco, R.J. Update of the case definitions for population-based surveillance of periodontitis. J. Periodontol. 2012, 83, 1449–1454. [Google Scholar] [CrossRef]
- CDC. NHANES 2013–2014 Laboratory Procedures Manual; VOCs in Blood Section; CDC: Atlanta, GA, USA, 2017. Available online: https://wwwn.cdc.gov/nchs/nhanes/search/datapage.aspx?Component=Laboratory&CycleBeginYear=2013 (accessed on 11 May 2025).
- Esteve-Turrillas, F.A.; Armenta, S.; Garrigues, S.; Pastor, A.; de la Guardia, M. Headspace-mass spectrometry determination of benzene, toluene and the mixture of ethylbenzene and xylene isomers in soil samples using chemometrics. Anal. Chim. Acta 2007, 587, 89–96. [Google Scholar] [CrossRef]
- Smith, M.T. Advances in understanding benzene health effects and susceptibility. Annu. Rev. Public Health 2010, 31, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Xu, M.; Wang, Y.; Wang, L.; Zhou, Y.; Deng, Y.; Yu, K. Air pollution and oral health: An overall insight from genetic causality. Int. Dent. J. 2025, 75, 1979–1987. [Google Scholar] [CrossRef]
- Li, W.; Zong, X.; He, Y.S.; Liu, X.; Zhao, C.; Wang, Y.; Zhang, J.; Pan, H.F. The effect of air pollution exposure on the risk of outpatient visits for periodontitis: A time-series study. Int. J. Environ. Health Res. 2023, 34, 2418–2429. [Google Scholar] [CrossRef] [PubMed]
- Sinjari, B.; Santilli, M.; Di Carlo, P.; Aruffo, E.; Caputi, S. The Correlation between Oral Health and Air Pollution: A Systematic Review. Dent. J. 2024, 12, 215. [Google Scholar] [CrossRef]
- Chapple, I.L.; Matthews, J.B. The role of reactive oxygen and antioxidant species in periodontal tissue destruction. Periodontology 2000 2007, 43, 160–232. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Eisner, M.D.; Lazcano-Ponce, E.; Zielinska-Danch, W.; Koszowski, B.; Sobczak, A.; Havel, C.; Jacob, P.; Benowitz, N.L. Comparison of urine cotinine and the tobacco-specific nitrosamine metabolite 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) and their ratio to discriminate active from passive smoking. Nicotine Tob. Res. Off. J. Soc. Res. Nicotine Tob. 2011, 13, 202–208. [Google Scholar] [CrossRef]
- Brunnemann, K.D.; Kagan, M.R.; Cox, J.E.; Hoffmann, D. Determination of benzene, toluene and 1,3-butadiene in cigarette smoke by GC-MSD. Exp. Pathol. 1989, 37, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, M.; Kuroiwa, Y.; Yokoi, T. INTERINDIVIDUAL DIFFERENCES IN NICOTINE METABOLISM AND GENETIC POLYMORPHISMS OF HUMAN CYP2A6. Drug Metab. Rev. 2002, 34, 865–877. [Google Scholar] [CrossRef] [PubMed]
- Harjumäki, R.; Pridgeon, C.S.; Ingelman-Sundberg, M. CYP2E1 in Alcoholic and Non-Alcoholic Liver Injury. Roles of ROS, Reactive Intermediates and Lipid Overload. Int. J. Mol. Sci. 2021, 22, 8221. [Google Scholar] [CrossRef] [PubMed]
- Tranfo, G.; Pigini, D.; Paci, E.; Marini, F.; Bonanni, R.C. Association of exposure to benzene and smoking with oxidative damage to nucleic acids by means of biological monitoring of general population volunteers. Environ. Sci. Pollut. Res. Int. 2017, 24, 13885–13894. [Google Scholar] [CrossRef]
- Abplanalp, W.; DeJarnett, N.; Riggs, D.W.; Conklin, D.J.; McCracken, J.P.; Srivastava, S.; Xie, Z.; Rai, S.; Bhatnagar, A.; O’Toole, T.E. Benzene exposure is associated with cardiovascular disease risk. PloS ONE 2017, 12, e0183602. [Google Scholar] [CrossRef]
- Monks, T.J.; Butterworth, M.; Lau, S.S. The fate of benzene-oxide. Chem.-Biol. Interact. 2010, 184, 201–206. [Google Scholar] [CrossRef]
- Mwenifumbo, J.C.; Lessov-Schlaggar, C.N.; Zhou, Q.; Krasnow, R.E.; Swan, G.E.; Benowitz, N.L.; Tyndale, R.F. Identification of Novel CYP2A6*1B Variants: The CYP2A6*1B Allele is Associated with Faster In Vivo Nicotine Metabolism. Clin. Pharmacol. Ther. 2008, 83. [Google Scholar] [CrossRef]
- Tian, G.; Longest, P.W. Transient absorption of inhaled vapors into a multilayer mucus-tissue-blood system. Ann. Biomed. Eng. 2010, 38, 517–536. [Google Scholar] [CrossRef]
- Silva, H. Tobacco Use and Periodontal Disease—The Role of Microvascular Dysfunction. Biology 2021, 10, 441. [Google Scholar] [CrossRef]
- Bosshardt, D.D. (2018). The periodontal pocket: Pathogenesis, histopathology and consequences. Periodontology 2000, 76, 43–50. [Google Scholar] [CrossRef]
- Fitzsimmons, T.R.; Sanders, A.E.; Bartold, P.M.; Slade, G.D. Local and systemic biomarkers in gingival crevicular fluid increase odds of periodontitis. J. Clin. Periodontol. 2010, 37, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Cordiano, R.; Papa, V.; Cicero, N.; Spatari, G.; Allegra, A.; Gangemi, S. Effects of Benzene: Hematological and Hypersensitivity Manifestations in Resident Living in Oil Refinery Areas. Toxics 2022, 10, 678. [Google Scholar] [CrossRef] [PubMed]
- Ranjit, S.; Sinha, N.; Kodidela, S.; Kumar, S. Benzo(a)pyrene in Cigarette Smoke Enhances HIV-1 Replication through NF-κB Activation via CYP-Mediated Oxidative Stress Pathway. Sci. Rep. 2018, 8, 10394. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Nishida, N.; Tanaka, M.; Hayashi, N.; Matsuse, R.; Nakayama, K.; Morimoto, K.; Shizukuishi, S. Association between passive and active smoking evaluated by salivary cotinine and periodontitis. J. Clin. Periodontol. 2005, 32, 1041–1046. [Google Scholar] [CrossRef] [PubMed]
- Duque, A.D.; Martínez, P.J.; Giraldo, A.; Gualtero, D.F.; Ardila, C.M.; Contreras, A.; Duarte, S.; Lafaurie, G.I. Accuracy of cotinine serum test to detect the smoking habit and its association with periodontal disease in a multicenter study. Med. Oral Patol. Oral Y Cirugía Bucal 2017, 22, e425–e431. [Google Scholar] [CrossRef]
- Wang, Y.; Andrukhov, O.; Rausch-Fan, X. Oxidative stress and antioxidant system in periodontitis. Front. Physiol. 2017, 8, 910. [Google Scholar] [CrossRef]
- Snyder, R. Leukemia and benzene. Int. J. Environ. Res. Public Health 2012, 9, 2875–2893. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P., 3rd. Nicotine chemistry, metabolism, kinetics and biomarkers. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2009; Volume 192, pp. 29–60. [Google Scholar] [CrossRef]
- Kazor, C.; Taylor, G.W.; Loesche, W.J. The prevalence of BANA-hydrolyzing periodontopathic bacteria in smokers. J. Clin. Periodontol. 1999, 26, 814–821. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Fourth National Report on Human Exposure to Environmental Chemicals: Updated Tables; U.S. Department of Health and Human Services: Washington, DC, USA, 2021; Volume 1.
- Yuan, J.; Murphy, S.E.; Stepanov, I.S.; Wang, R.; Carmella, S.G.; Nelson, H.H.; Hatsukami, D.K.; Hecht, S.S. 2-Phenethyl Isothiocyanate, Glutathione S-transferase M1 and T1 Polymorphisms, and Detoxification of Volatile Organic Carcinogens and Toxicants in Tobacco Smoke. Cancer Prev. Res. 2016, 9, 598–606. [Google Scholar] [CrossRef]
- Katsuragi, H.; Ohtake, M.; Kurasawa, I.; Saito, K. Intracellular production and extracellular release of oxygen radicals by PMNs and oxidative stress on PMNs during phagocytosis of periodontopathic bacteria. Odontology 2003, 91, 13–18. [Google Scholar] [CrossRef]
- Mason, M.R.; Preshaw, P.M.; Nagaraja, H.N.; Dabdoub, S.M.; Rahman, A.; Kumar, P.S. The subgingival microbiome of clinically healthy current and never smokers. ISME J. 2015, 9, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Cogo, K.; Calvi, B.M.; Mariano, F.S.; Franco, G.C.; Gonçalves, R.B.; Groppo, F.C. The effects of nicotine and cotinine on Porphyromonas gingivalis colonisation of epithelial cells. Arch. Oral Biol. 2009, 54, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Sopori, M. Effects of cigarette smoke on the immune system. Nat. Rev. Immunol. 2002, 2, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Rothman, N.; Bassig, B.A.; Zhang, L.; Vermeulen, R.C.; Li, G.; Kemp, T.J.; Hu, W.; Purdue, M.P.; Yin, S.; Rappaport, S.M.; et al. O08-2 Occupational exposure to benzene and alterations in immune/inflammatory markers. Occup. Environ. Med. 2016, 73. [Google Scholar]
- NCHS. National Health and Nutrition Examination Survey. Available online: https://wwwn.cdc.gov/nchs/nhanes/ (accessed on 4 December 2023).
| Covariate | No Periodontitis N = 2930 | Mild Periodontitis N = 614 | Moderate Periodontitis N = 796 | Severe Periodontitis N = 329 | Total N = 4669 | p-Value |
|---|---|---|---|---|---|---|
| Sex | <0.001 | |||||
| male | 1221 (55.10%) | 346 (15.61%) | 420 (18.95%) | 229 (10.33%) | 2216 (100%) | |
| female | 1709 (69.67%) | 268 (10.93%) | 376 (15.33%) | 100 (4.08%) | 2453 (100%) | |
| Age | <0.001 | |||||
| 30–34 | 354 (70.66%) | 93 (18.56%) | 41 (8.18%) | 13 (2.59%) | 501 (100%) | |
| 35–49 | 982 (66.94%) | 247 (16.84%) | 165 (11.25%) | 73 (4.98%) | 1467 (100%) | |
| 50–64 | 817 (56.89%) | 168 (11.70%) | 286 (19.92%) | 165 (11.49%) | 1436 (100%) | |
| 65+ | 777 (61.42%) | 106 (8.38%) | 304 (24.03%) | 78 (6.17%) | 1265 (100%) | |
| Race | 1402 (68.73%) | 190 (9.31%) | 351 (17.21%) | 97 (4.75%) | 2040 (100%) | <0.001 |
| white | 539 (56.86%) | 150 (15.82%) | 153 (16.14%) | 106 (11.18%) | 948 (100%) | |
| Black Hispanic | 574 (56.33%) | 194 (19.04%) | 161 (15.80%) | 90 (8.83%) | 1019 (100%) | |
| Asian | 341 (62.34%) | 69 (12.61%) | 107 (19.56%) | 30 (5.48%) | 547 (100%) | |
| Other | 74 (64.35%) | 11 (9.57%) | 24 (20.87%) | 6 (5.22%) | 115 (100%) | |
| Education | <0.001 | |||||
| <High School | 563 (54.03%) | 174 (16.70%) | 182 (17.47%) | 123 (11.80%) | 1042 (100%) | |
| High School/GED | 593 (56.64%) | 157 (15.00%) | 202 (19.29%) | 95 (9.07%) | 1047 (100%) | |
| College or More | 1772 (68.79%) | 282 (10.95%) | 411 (15.95%) | 111 (4.31%) | 2576 (100%) | |
| Diabetes | 0.927 | |||||
| No | 2524 (63.66%) | 521 (13.14%) | 641 (16.17%) | 279 (7.04%) | 3965 (100%) | |
| Yes | 403 (57.49%) | 93 (13.27%) | 155 (22.11%) | 50 (7.13%) | 701 (100%) | |
| Any disease | 0.072 | |||||
| No | 1626 (63.32%) | 389 (15.15%) | 357 (13.90%) | 196 (7.63%) | 2568 (100%) | |
| Yes | 1294 (62.00%) | 224 (10.73%) | 438 (20.99%) | 131 (6.28%) | 2087 (100%) |
| Measurement Periodontal Status Benzene (ng/mL) | Benzene (ng/mL) (N = 2096) | Cotinine (ng/mL) (N = 4464) | ||||
|---|---|---|---|---|---|---|
| Mean (SD) | Sample Number | p-Value | Mean (SD) | Sample Number | p-Value | |
| No periodontitis | 0.05 (0.1) | 1288 | <0.0001 | 48.8 (126.6) | 2790 | <0.0001 |
| Mild periodontitis | 0.07 (0.1) | 293 | <0.0001 | 76.7 (144.3) | 588 | <0.0001 |
| Moderate periodontitis | 0.07 (0.1) | 364 | <0.0001 | 73.4 (153.9) | 771 | <0.0001 |
| Severe periodontitis | 0.1 (0.1) | 151 | <0.0001 | 124.8 (173.5) | 315 | <0.0001 |
| Total | 2096 | 4464 | ||||
| Composite | ||||
|---|---|---|---|---|
| Covariate | Odds Ratio | Confidence Interval | p Value | |
| Lower | Upper | |||
| Benzene | 2.9 | 1.6 | 5.3 | (0.001) * |
| Age | ||||
| 35 to 49 | 6.8 | 1.1 | 40.8 | (0.037) * |
| 50 to 64 | 15.5 | 2.8 | 86.4 | (0.004) * |
| ≥65 | 2.6 | 298.8 | (0.009) * | |
| Sex (reference: male) | 0.5 | 0.2 | 1.2 | (0.146) |
| Race (reference: non-Hispanic white) | ||||
| Non-Hispanic black | 3.2 | 1.4 | 6.8 | (0.005) * |
| Hispanic | 1.5 | 0.3 | 7.4 | (0.545) |
| Non Hispanic Asian | 1.8 | 0.5 | 6.1 | (0.285) |
| Other | 6.0 | 0.7 | 49 | (0.085) |
| Education (reference: <high school) | ||||
| High school/GED | 0.9 | 0.2 | 2.8 | (0.084) |
| Some college or more | 0.4 | 0.2 | 1.0 | (0.060) |
| Occupation | 1.2 | 0.5 | 2.7 | (0.612) |
| Diabetes | 1.1 | 0.3 | 4.1 | (0.824) |
| Any disease | 0.2 | 0.09 | 0.8 | (0.025) * |
| Path | Estimate | Standard Error | p-Value | 95% CI |
|---|---|---|---|---|
| Benzene > Cotinine | 2.93 | 0.05 | < 0.001 | 2.83–3.02 |
| Benzene > Severe periodontitis | 0.29 | 0.25 | 0.261 | −0.21–0.78 |
| Cotinine > Severe periodontitis | 0.23 | 0.07 | 0.001 | 0.09–0.37 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamoud, B.; Alfailakwi, M.; Aljalahmah, H.; Almael, F.; Alsaeedi, S.; Saleh, K.; Ahmad, B.; Alqaderi, H. Association Between Blood Benzene Levels and Periodontal Disease in a Nationally Representative Adult U.S. Population. Int. J. Environ. Res. Public Health 2025, 22, 853. https://doi.org/10.3390/ijerph22060853
Hamoud B, Alfailakwi M, Aljalahmah H, Almael F, Alsaeedi S, Saleh K, Ahmad B, Alqaderi H. Association Between Blood Benzene Levels and Periodontal Disease in a Nationally Representative Adult U.S. Population. International Journal of Environmental Research and Public Health. 2025; 22(6):853. https://doi.org/10.3390/ijerph22060853
Chicago/Turabian StyleHamoud, Basel, Meshari Alfailakwi, Hessah Aljalahmah, Fatema Almael, Sarah Alsaeedi, Khaled Saleh, Bushra Ahmad, and Hend Alqaderi. 2025. "Association Between Blood Benzene Levels and Periodontal Disease in a Nationally Representative Adult U.S. Population" International Journal of Environmental Research and Public Health 22, no. 6: 853. https://doi.org/10.3390/ijerph22060853
APA StyleHamoud, B., Alfailakwi, M., Aljalahmah, H., Almael, F., Alsaeedi, S., Saleh, K., Ahmad, B., & Alqaderi, H. (2025). Association Between Blood Benzene Levels and Periodontal Disease in a Nationally Representative Adult U.S. Population. International Journal of Environmental Research and Public Health, 22(6), 853. https://doi.org/10.3390/ijerph22060853






