Application of Ozonation-Biodegradation Hybrid System for Polycyclic Aromatic Hydrocarbons Degradation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Chemicals
2.1.2. Bacterial Strains
2.2. Methods
2.2.1. Culture Preparation
2.2.2. Biodegradation Tests
2.2.3. Total Organic Carbon and Chemical Oxygen Demand
2.2.4. Gas Chromatographic Analyses
2.2.5. Microbial Activity Measurements
2.2.6. Statistical Analyses and Initial Reaction Rates
3. Results
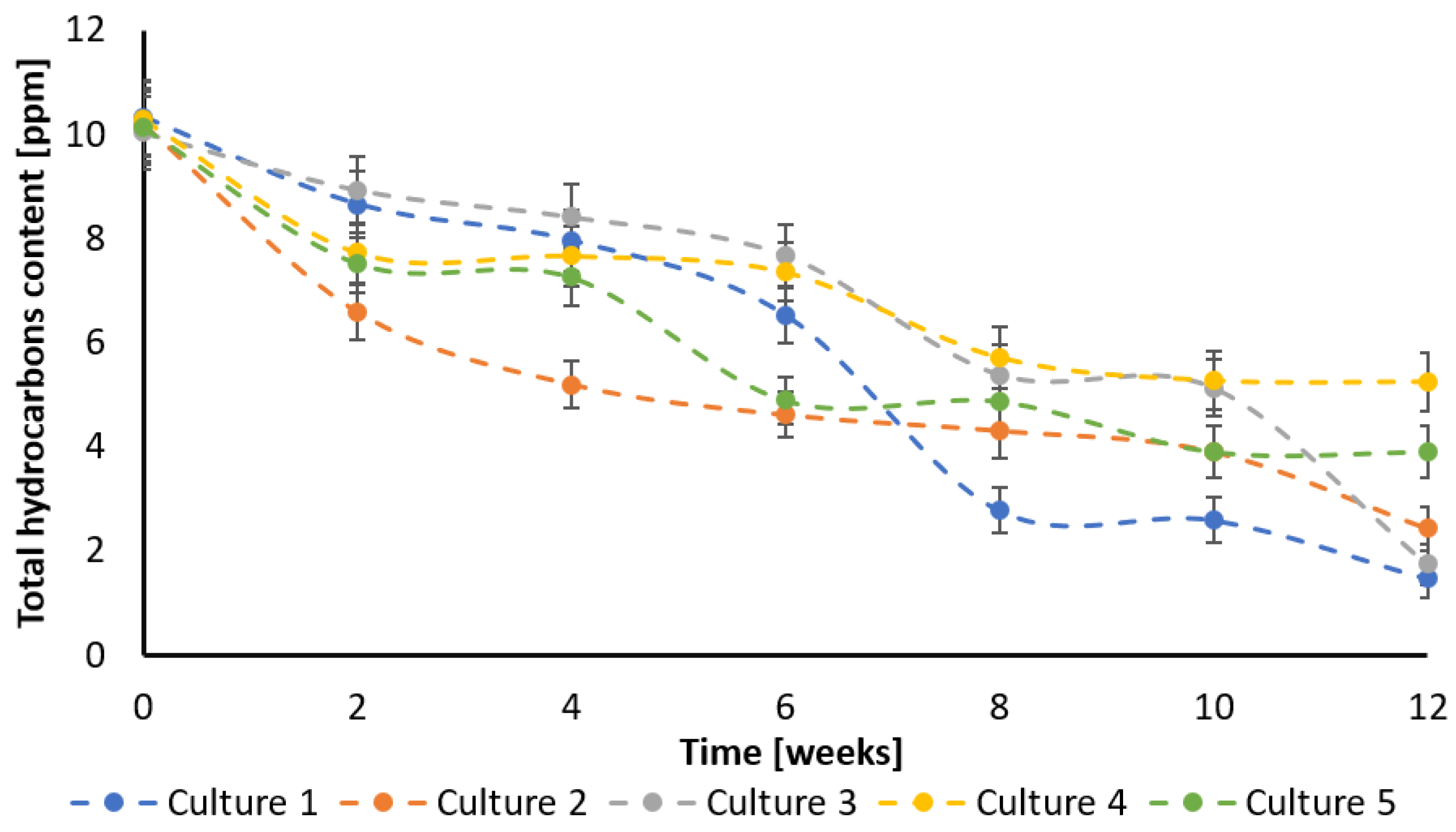
3.1. Creosote Hydrocarbons Degradation
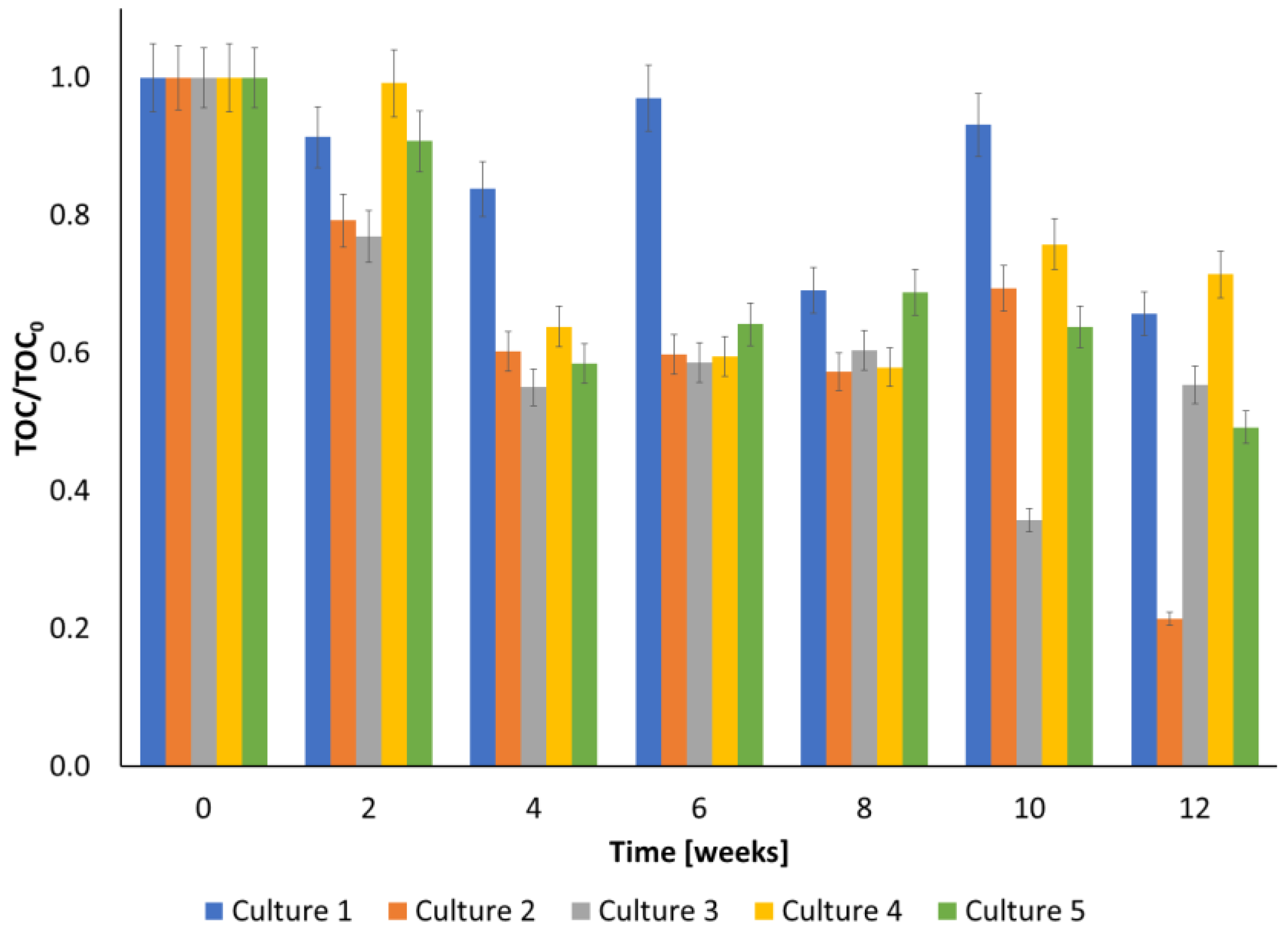
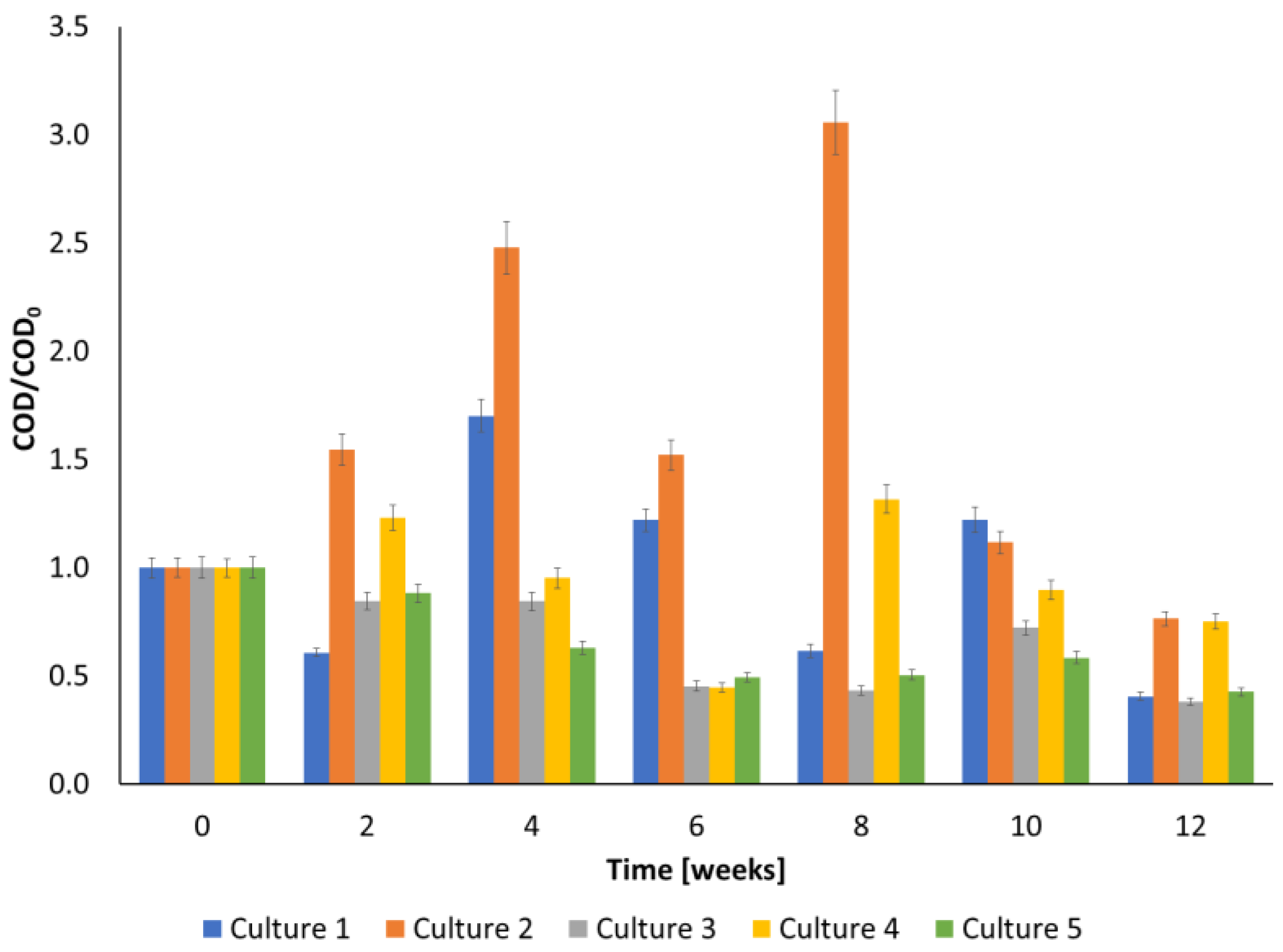
3.2. TOC and COD during Biodegradation
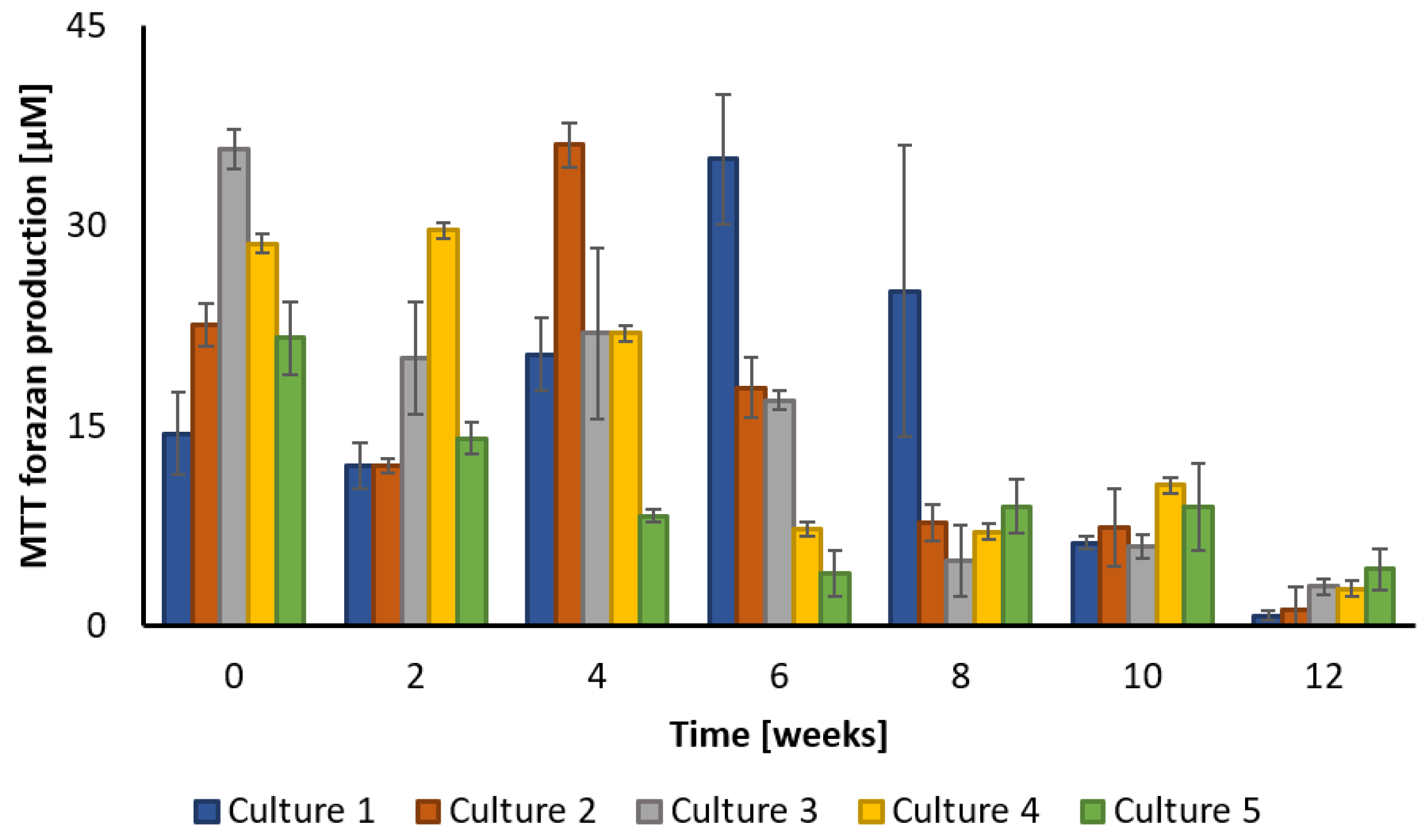
3.3. Changes in Microbial Activity during Biodegradation
4. Discussion
| Medium | Time of Study | CO3 | COD Reduction (bio + O3) | Microorganisms | BTP Reduction | PYR Reduction | PHE Reduction | FLU Reduction | THC Reduction | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| coking wastewater | 12 h | 30 mg L−1 h−1 | 48.5% | Comamonadaceae, Paracoccus, Comamonas, Corynebacterium, Truepera microbes | - | - | - | - | - | [49] |
| soil | 9 weeks | 2 g/h | - | - | ~61% after 7 days bio + 3 h O3 | ~66% after 7 days bio + 3 h O3 | - | - | ~100% | [35] |
| soil | bio: 4 weeks, O3: 2 days | 12 mg/day | - | - | 23.2% (bio + O3); 73.5% (O3 + bio) | 10.5% (bio + O3); 40.8% (O3 + bio) | 46.5% (bio + O3); 71.6% (O3 + bio) | ~100% | - | [42] |
| soil | 12 days | 20–790 mg L−1 | - | Pseudomonas sp. | - | - | - | - | 90% | [53] |
| simulated creosote wastewater | 12 weeks | O31: 0.76; O32: 6.63 mg L−1 | 23.4% (O31 + bio); 62.0% (O32 + bio); 24.8% (bio + O31); 57.3% (bio + O32) | Pseudomonas sp. MChB, Pseudomonas sp. OS4, Raoultella planticola SA2, Achromobacter sp. KW1, Rahnella aquatilis DA2 | 83% (bio); 89% (O31 + bio); 59% (O32 + bio); 43% (bio + O31); 51% (bio + O32) | 89% (bio); 91% (O31 + bio); 76% (O32 + bio); 92% (bio + O31); 87% (bio+ O32) | 96% (bio); 97% (O31 + bio); 43% (O32 + bio); 86% (bio + O31); 36% (bio + O32) | 82% (bio), 88% (O31 + bio); 62% (O32 + bio); 43% (bio + O31); 52% (bio+ O32) | 86% (bio), 76% (O31 + bio); 83% (O32 + bio); 49% (bio + O31); 61% (bio+ O32) | this work |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Patel, A.K.; Singhania, R.R.; Albarico, F.P.J.B.; Pandey, A.; Chen, C.-W.; Dong, C.-D. Organic Wastes Bioremediation and Its Changing Prospects. Sci. Total Environ. 2022, 824, 153889. [Google Scholar] [CrossRef]
- Malik, B.; Pirzadah, T.B.; Hakeem, K.R. Phytoremediation of Persistent Organic Pollutants (POPs). In Phytoremediation; Elsevier: Amsterdam, The Netherlands, 2022; pp. 415–436. ISBN 978-0-323-89874-4. [Google Scholar]
- Gutierrez-Urbano, I.; Villen-Guzman, M.; Perez-Recuerda, R.; Rodriguez-Maroto, J.M. Removal of Polycyclic Aromatic Hydrocarbons (PAHs) in Conventional Drinking Water Treatment Processes. J. Contam. Hydrol. 2021, 243, 103888. [Google Scholar] [CrossRef]
- Liu, S.-H.; Zeng, G.-M.; Niu, Q.-Y.; Liu, Y.; Zhou, L.; Jiang, L.-H.; Tan, X.; Xu, P.; Zhang, C.; Cheng, M. Bioremediation Mechanisms of Combined Pollution of PAHs and Heavy Metals by Bacteria and Fungi: A Mini Review. Bioresour. Technol. 2017, 224, 25–33. [Google Scholar] [CrossRef]
- Tauler, M.; Vila, J.; Nieto, J.M.; Grifoll, M. Key High Molecular Weight PAH-Degrading Bacteria in a Soil Consortium Enriched Using a Sand-in-Liquid Microcosm System. Appl. Microbiol. Biotechnol. 2016, 100, 3321–3336. [Google Scholar] [CrossRef]
- Smułek, W.; Sydow, M.; Zabielska-Matejuk, J.; Kaczorek, E. Bacteria Involved in Biodegradation of Creosote PAH—A Case Study of Long-Term Contaminated Industrial Area. Ecotoxicol. Environ. Saf. 2020, 187, 109843. [Google Scholar] [CrossRef]
- Behera, B.K.; Das, A.; Sarkar, D.J.; Weerathunge, P.; Parida, P.K.; Das, B.K.; Thavamani, P.; Ramanathan, R.; Bansal, V. Polycyclic Aromatic Hydrocarbons (PAHs) in Inland Aquatic Ecosystems: Perils and Remedies through Biosensors and Bioremediation. Environ. Pollut. 2018, 241, 212–233. [Google Scholar] [CrossRef]
- Smułek, W.; Pacholak, A.; Kaczorek, E. Modification of the Bacterial Cell Wall—Is the Bioavailability Important in Creosote Biodegradation? Processes 2020, 8, 147. [Google Scholar] [CrossRef]
- Ali, S.S.; Jiao, H.; Mustafa, A.M.; Koutra, E.; El-Sapagh, S.; Kornaros, M.; Elsamahy, T.; Khalil, M.; Bulgariu, L.; Sun, J. Construction of a Novel Microbial Consortium Valued for the Effective Degradation and Detoxification of Creosote-Treated Sawdust along with Enhanced Methane Production. J. Hazard. Mater. 2021, 418, 126091. [Google Scholar] [CrossRef]
- Haritash, A.K.; Kaushik, C.P. Biodegradation Aspects of Polycyclic Aromatic Hydrocarbons (PAHs): A Review. J. Hazard. Mater. 2009, 169, 1–15. [Google Scholar] [CrossRef]
- Madrid, F.; Rubio-Bellido, M.; Villaverde, J.; Peña, A.; Morillo, E. Natural and Assisted Dissipation of Polycyclic Aromatic Hydrocarbons in a Long-Term Co-Contaminated Soil with Creosote and Potentially Toxic Elements. Sci. Total Environ. 2019, 660, 705–714. [Google Scholar] [CrossRef]
- Volkoff, S.J.; Rodriguez, D.L.; Singleton, D.R.; McCumber, A.W.; Aitken, M.D.; Stewart, J.R.; Gunsch, C.K. Identifying Bioaugmentation Candidates for Bioremediation of Polycyclic Aromatic Hydrocarbons in Contaminated Estuarine Sediment of the Elizabeth River, VA, USA. Appl. Microbiol. Biotechnol. 2022, 106, 1715–1727. [Google Scholar] [CrossRef]
- Wang, F.; Dong, W.; Wang, H.; Zhao, Y.; Zhao, Z.; Huang, J.; Zhou, T.; Wu, Z.; Li, W. Enhanced Bioremediation of Sediment Contaminated with Polycyclic Aromatic Hydrocarbons by Combined Stimulation with Sodium Acetate/Phthalic Acid. Chemosphere 2022, 291, 132770. [Google Scholar] [CrossRef]
- Ng, Y.J.; Lim, H.R.; Khoo, K.S.; Chew, K.W.; Chan, D.J.C.; Bilal, M.; Munawaroh, H.S.H.; Show, P.L. Recent Advances of Biosurfactant for Waste and Pollution Bioremediation: Substitutions of Petroleum-Based Surfactants. Environ. Res. 2022, 212, 113126. [Google Scholar] [CrossRef]
- Gou, Y.; Ma, J.; Yang, S.; Song, Y. Insights into the Effects of Fenton Oxidation on PAH Removal and Indigenous Bacteria in Aged Subsurface Soil. Environ. Pollut. 2022, 298, 118872. [Google Scholar] [CrossRef]
- Gomes, J.; Costa, R.; Quinta-Ferreira, R.M.; Martins, R.C. Application of Ozonation for Pharmaceuticals and Personal Care Products Removal from Water. Sci. Total Environ. 2017, 586, 265–283. [Google Scholar] [CrossRef]
- Mathon, B.; Coquery, M.; Liu, Z.; Penru, Y.; Guillon, A.; Esperanza, M.; Miège, C.; Choubert, J.-M. Ozonation of 47 Organic Micropollutants in Secondary Treated Municipal Effluents: Direct and Indirect Kinetic Reaction Rates and Modelling. Chemosphere 2021, 262, 127969. [Google Scholar] [CrossRef]
- Olak-Kucharczyk, M.; Ledakowicz, S. Decomposition of Phenylphenol Isomers by UVC-Enhanced Ozonation Process. Ozone Sci. Eng. 2017, 39, 333–342. [Google Scholar] [CrossRef]
- Bilińska, L.; Gmurek, M.; Ledakowicz, S. Comparison between Industrial and Simulated Textile Wastewater Treatment by AOPs—Biodegradability, Toxicity and Cost Assessment. Chem. Eng. J. 2016, 306, 550–559. [Google Scholar] [CrossRef]
- Sakshi; Singh, S.K.; Haritash, A.K. Polycyclic Aromatic Hydrocarbons: Soil Pollution and Remediation. Int. J. Environ. Sci. Technol. 2019, 16, 6489–6512. [Google Scholar] [CrossRef]
- Li, W.; Zhu, N.; Shen, Y.; Yuan, H. Towards Efficient Elimination of Polycyclic Aromatic Hydrocarbons (PAHs) from Waste Activated Sludge by Ozonation. Environ. Res. 2021, 195, 110783. [Google Scholar] [CrossRef]
- Ji, H.; Gong, Y.; Duan, J.; Zhao, D.; Liu, W. Degradation of Petroleum Hydrocarbons in Seawater by Simulated Surface-Level Atmospheric Ozone: Reaction Kinetics and Effect of Oil Dispersant. Mar. Pollut. Bull. 2018, 135, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Yavuz, B.M.; Delgado, A.G.; Januszewski, B.; Zuo, Y.; Westerhoff, P.; Krajmalnik-Brown, R.; Rittmann, B.E. Multicycle Ozonation+Bioremediation for Soils Containing Residual Petroleum. Environ. Eng. Sci. 2019, 36, 1443–1451. [Google Scholar] [CrossRef]
- Paździor, K.; Wrębiak, J.; Klepacz-Smółka, A.; Gmurek, M.; Bilińska, L.; Kos, L.; Sójka-Ledakowicz, J.; Ledakowicz, S. Influence of Ozonation and Biodegradation on Toxicity of Industrial Textile Wastewater. J. Environ. Manag. 2017, 195, 166–173. [Google Scholar] [CrossRef]
- El-taliawy, H.; Casas, M.E.; Bester, K. Removal of Ozonation Products of Pharmaceuticals in Laboratory Moving Bed Biofilm Reactors (MBBRs). J. Hazard. Mater. 2018, 347, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, A.; Rai, B.N.; Singh, R.S.; Jaiswal, R.P. A Comprehensive Review on the Integration of Advanced Oxidation Processes with Biodegradation for the Treatment of Textile Wastewater Containing Azo Dyes. Rev. Chem. Eng. 2021, 38, 617–639. [Google Scholar] [CrossRef]
- Tang, K.; Spiliotopoulou, A.; Chhetri, R.K.; Ooi, G.T.H.; Kaarsholm, K.M.S.; Sundmark, K.; Florian, B.; Kragelund, C.; Bester, K.; Andersen, H.R. Removal of Pharmaceuticals, Toxicity and Natural Fluorescence through the Ozonation of Biologically-Treated Hospital Wastewater, with Further Polishing via a Suspended Biofilm. Chem. Eng. J. 2019, 359, 321–330. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Y.; Gamal El-Din, M. Integrated Mild Ozonation with Biofiltration Can Effectively Enhance the Removal of Naphthenic Acids from Hydrocarbon-Contaminated Water. Sci. Total Environ. 2019, 678, 197–206. [Google Scholar] [CrossRef]
- Ulucan-Altuntas, K.; Ilhan, F. Enhancing Biodegradability of Textile Wastewater by Ozonation Processes: Optimization with Response Surface Methodology. Ozone Sci. Eng. 2018, 40, 465–472. [Google Scholar] [CrossRef]
- Marcelino, R.B.P.; Leão, M.M.D.; Lago, R.M.; Amorim, C.C. Multistage Ozone and Biological Treatment System for Real Wastewater Containing Antibiotics. J. Environ. Manag. 2017, 195, 110–116. [Google Scholar] [CrossRef]
- de Wilt, A.; van Gijn, K.; Verhoek, T.; Vergnes, A.; Hoek, M.; Rijnaarts, H.; Langenhoff, A. Enhanced Pharmaceutical Removal from Water in a Three Step Bio-Ozone-Bio Process. Water Res. 2018, 138, 97–105. [Google Scholar] [CrossRef]
- Bar Oz, Y.; Mamane, H.; Menashe, O.; Cohen-Yaniv, V.; Kumar, R.; Iasur Kruh, L.; Kurzbaum, E. Treatment of Olive Mill Wastewater Using Ozonation Followed by an Encapsulated Acclimated Biomass. J. Environ. Chem. Eng. 2018, 6, 5014–5023. [Google Scholar] [CrossRef]
- Phan, L.T.; Schaar, H.; Saracevic, E.; Krampe, J.; Kreuzinger, N. Effect of Ozonation on the Biodegradability of Urban Wastewater Treatment Plant Effluent. Sci. Total Environ. 2022, 812, 152466. [Google Scholar] [CrossRef]
- Bernal-Martinez, A.; Carrère, H.; Patureau, D.; Delgenès, J.-P. Ozone Pre-Treatment as Improver of PAH Removal during Anaerobic Digestion of Urban Sludge. Chemosphere 2007, 68, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Derudi, M.; Venturini, G.; Lombardi, G.; Nano, G.; Rota, R. Biodegradation Combined with Ozone for the Remediation of Contaminated Soils. Eur. J. Soil Biol. 2007, 43, 297–303. [Google Scholar] [CrossRef]
- Sakulthaew, C.; Comfort, S.; Chokejaroenrat, C.; Harris, C.; Li, X. A Combined Chemical and Biological Approach to Transforming and Mineralizing PAHs in Runoff Water. Chemosphere 2014, 117, 1–9. [Google Scholar] [CrossRef]
- Kulik, N.; Goi, A.; Trapido, M.; Tuhkanen, T. Degradation of Polycyclic Aromatic Hydrocarbons by Combined Chemical Pre-Oxidation and Bioremediation in Creosote Contaminated Soil. J. Environ. Manag. 2006, 78, 382–391. [Google Scholar] [CrossRef]
- Zdarta, A.; Smułek, W.; Bielan, Z.; Zdarta, J.; Nguyen, L.N.; Zgoła-Grześkowiak, A.; Nghiem, L.D.; Jesionowski, T.; Kaczorek, E. Significance of the Presence of Antibiotics on the Microbial Consortium in Wastewater—The Case of Nitrofurantoin and Furazolidone. Bioresour. Technol. 2021, 339, 125577. [Google Scholar] [CrossRef] [PubMed]
- HACH LANGE 1 Working Procedure LCK386 TOC Total Organic Carbon. Available online: https://uk.hach.com/toc-cuvette-test-purging-method-30-300-mg-l-c-25-tests/product-downloads?id=26370291468 (accessed on 21 March 2023).
- HACH LANGE 2 Working Procedure: LCK314 COD Chemical Oxygen Demand. Available online: https://uk.hach.com/cod-cuvette-test-15-150-mg-l-o-25-tests/product-downloads?id=26370291408 (accessed on 21 March 2023).
- Pacholak, A.; Smułek, W.; Zgoła-Grześkowiak, A.; Kaczorek, E. Nitrofurantoin—Microbial Degradation and Interactions with Environmental Bacterial Strains. Int. J. Environ. Res. Public Health 2019, 16, 1526. [Google Scholar] [CrossRef]
- Nam, G.; Mohamed, M.M.; Jung, J. Enhanced Degradation of Benzo[a]Pyrene and Toxicity Reduction by Microbubble Ozonation. Environ. Technol. 2021, 42, 1853–1860. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Wei, H.; Wu, Z.; Zhao, Q.; Jiang, X. Enhanced Biodegradation of Poorly Available Polycyclic Aromatic Hydrocarbons by Easily Available One. Int. Biodeterior. Biodegrad. 2013, 84, 72–78. [Google Scholar] [CrossRef]
- Hoigne, J. Chemistry of Aqueous Ozone and Transformation of Pollutants by Ozonation and Advanced Oxidation Processes. In The Handbook of Environmental Chemistry, Quality and Treatment of Drinking Water II; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- Baghal Asghari, F.; Dehghani, M.H.; Dehghanzadeh, R.; Farajzadeh, D.; Shanehbandi, D.; Mahvi, A.H.; Yaghmaeian, K.; Rajabi, A. Performance Evaluation of Ozonation for Removal of Antibiotic-Resistant Escherichia Coli and Pseudomonas Aeruginosa and Genes from Hospital Wastewater. Sci. Rep. 2021, 11, 24519. [Google Scholar] [CrossRef]
- Sommaggio, L.R.D.; Oliveira, F.A.; Malvestiti, J.A.; Mazzeo, D.E.C.; Levy, C.E.; Dantas, R.F.; Marin-Morales, M.A. Assessment of Phytotoxic Potential and Pathogenic Bacteria Removal from Secondary Effluents during Ozonation and UV/H2O2. J. Environ. Manag. 2022, 318, 115522. [Google Scholar] [CrossRef]
- Hong, P.K.A.; Chao, J.-C. A Polar−Nonpolar, Acetic Acid/Heptane, Solvent Medium for Degradation of Pyrene by Ozone. Ind. Eng. Chem. Res. 2004, 43, 7710–7715. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, W.; Yang, J.; Cao, H. Removal of Phenanthrene from Contaminated Soil by Ozonation Process. China Pet. Process. Petrochem. Technol. 2020, 22, 73–79. [Google Scholar]
- Cui, B.; Fu, S.; Hao, X.; Zhou, D. Synergistic Effects of Simultaneous Coupling Ozonation and Biodegradation for Coking Wastewater Treatment: Advances in COD Removal, Toxic Elimination, and Microbial Regulation. Chemosphere 2023, 318, 137956. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Wang, X.; Dong, S.; Fu, S.; Zhou, D.; Rittmann, B.E. Towards a Simultaneous Combination of Ozonation and Biodegradation for Enhancing Tetracycline Decomposition and Toxicity Elimination. Bioresour. Technol. 2020, 304, 123009. [Google Scholar] [CrossRef] [PubMed]
- Nika, M.-C.; Aalizadeh, R.; Thomaidis, N.S. Non-Target Trend Analysis for the Identification of Transformation Products during Ozonation Experiments of Citalopram and Four of Its Biodegradation Products. J. Hazard. Mater. 2021, 419, 126401. [Google Scholar] [CrossRef] [PubMed]
- Caniani, D.; Caivano, M.; Mazzone, G.; Masi, S.; Mancini, I.M. Effect of Site-Specific Conditions and Operating Parameters on the Removal Efficiency of Petroleum-Originating Pollutants by Using Ozonation. Sci. Total Environ. 2021, 800, 149393. [Google Scholar] [CrossRef]
- Haapea, P.; Tuhkanen, T. Integrated Treatment of PAH Contaminated Soil by Soil Washing, Ozonation and Biological Treatment. J. Hazard. Mater. 2006, 136, 244–250. [Google Scholar] [CrossRef] [PubMed]
| Culture | Concentration of Ozone, mg L−1 | Volume of Synthetic Wastewater, mL | Volume of Bacteria Inoculum before Adding Ozone, mL | Volume of Creosote, mL | Volume of Ozonated Water Addition, mL | Volume of Bacteria Inoculum after Adding Ozone, mL | Final Volume, mL |
|---|---|---|---|---|---|---|---|
| 1 | - | 4 | 20 | 0.4 | - | - | 420.4 |
| 2 | 0.76 ± 0.17 | 4 | - | 0.4 | 20 | 20 | 420.4 |
| 3 | 6.63 ± 0.68 | 4 | - | 0.4 | 200 | 20 | 420.4 |
| 4 | 0.76 ± 0.17 | 4 | 20 | 0.4 | 20 | - | 420.4 |
| 5 | 6.63 ± 0.68 | 4 | 20 | 0.4 | 200 | - | 420.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olak-Kucharczyk, M.; Festinger, N.; Smułek, W. Application of Ozonation-Biodegradation Hybrid System for Polycyclic Aromatic Hydrocarbons Degradation. Int. J. Environ. Res. Public Health 2023, 20, 5347. https://doi.org/10.3390/ijerph20075347
Olak-Kucharczyk M, Festinger N, Smułek W. Application of Ozonation-Biodegradation Hybrid System for Polycyclic Aromatic Hydrocarbons Degradation. International Journal of Environmental Research and Public Health. 2023; 20(7):5347. https://doi.org/10.3390/ijerph20075347
Chicago/Turabian StyleOlak-Kucharczyk, Magdalena, Natalia Festinger, and Wojciech Smułek. 2023. "Application of Ozonation-Biodegradation Hybrid System for Polycyclic Aromatic Hydrocarbons Degradation" International Journal of Environmental Research and Public Health 20, no. 7: 5347. https://doi.org/10.3390/ijerph20075347
APA StyleOlak-Kucharczyk, M., Festinger, N., & Smułek, W. (2023). Application of Ozonation-Biodegradation Hybrid System for Polycyclic Aromatic Hydrocarbons Degradation. International Journal of Environmental Research and Public Health, 20(7), 5347. https://doi.org/10.3390/ijerph20075347







