A Multicentre, Randomised, Controlled Trial of a Combined Clinical Treatment for First-Episode Psychosis
Abstract
:1. Introduction
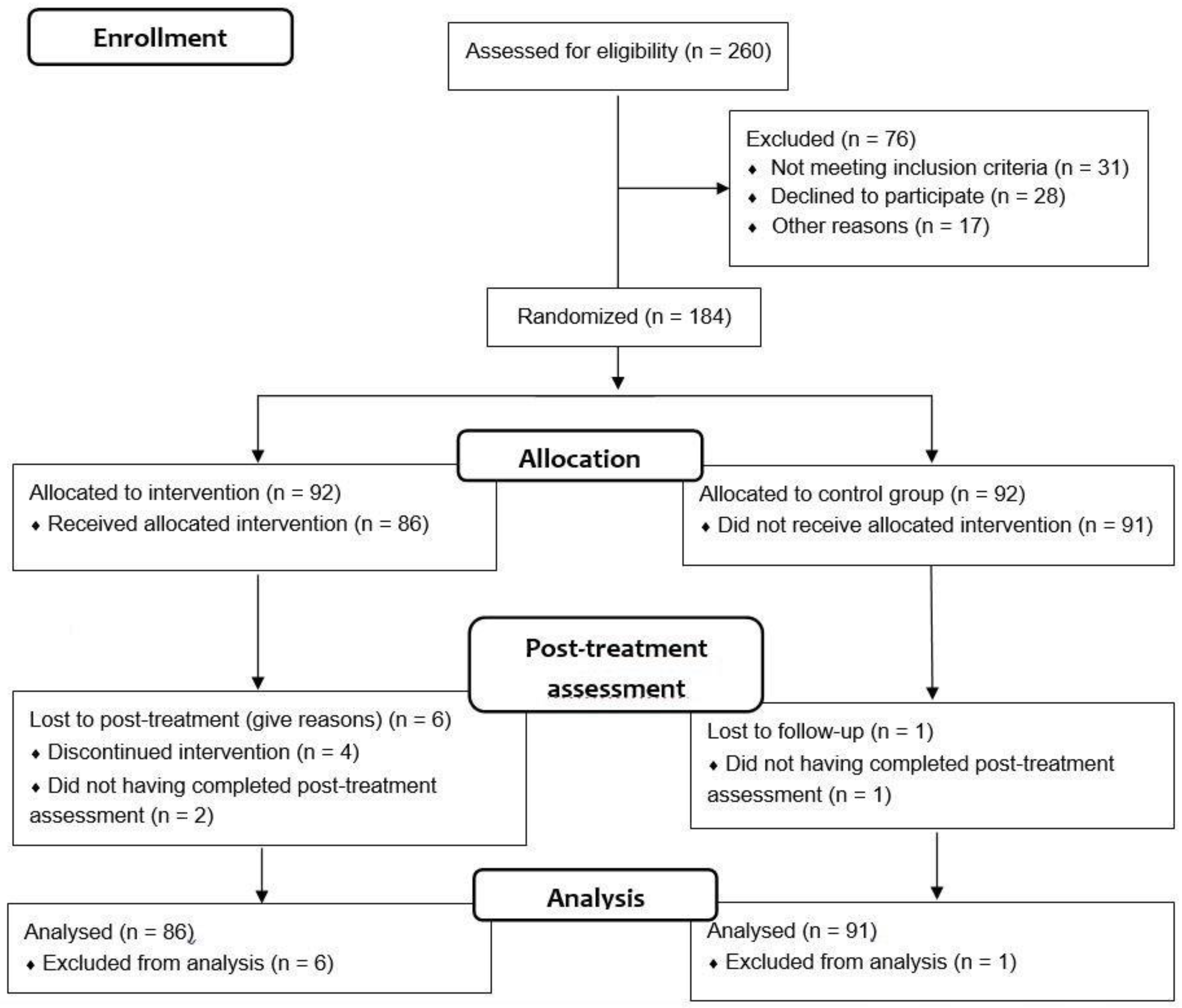
2. Materials and Methods
2.1. Participants
2.2. Instruments
2.2.1. Primary Outcome
2.2.2. Secondary Outcomes
2.3. Procedure
2.4. Intervention Programme
- What is a first episode of psychosis?
- Challenge and importance of insight into vulnerability.
- Symptom recognition.
- Prevention of relapses: protective and risk factors.
- Detection of prodromes.
- What can I do if I perceive that the symptoms are emerging again?
- Treatment adherence.
- Healthy lifestyles: sleep and sexuality.
- Healthy lifestyles: substance use.
- Anxiety management techniques (I).
- Anxiety management techniques (II).
- Social skills: assertiveness techniques.
- Problem-solving techniques.
- Final doubts and farewell.
2.5. Statistical Analyses
3. Results
3.1. Sociodemographic and Clinical Baseline Characteristics of the Sample
3.2. Clinical and Functional Outcomes of FEP Patients
3.3. Influence of Clinical Improvements on Functional Outcomes
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Di Capite, S.; Upthegrove, R.; Mallikarjun, P. The relapse rate and predictors of relapse in patients with first-episode psychosis following discontinuation of antipsychotic medication. Early Interv. Psychiatry 2018, 12, 893–899. [Google Scholar] [CrossRef] [Green Version]
- Albert, N.; Weibell, M.A. The outcome of early intervention in first episode psychosis. Int. Rev. Psychiatry 2019, 31, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Lykissas, M.G.; Batistatou, A.K.; Charalabopoulos, K.A.; Beris, A.E. The role of neurotrophins in axonal growth, guidance, and regeneration. Curr. Neurovasc. Res. 2007, 4, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bueno, B.; Bioque, M.; Mac-Dowell, K.S.; Barcones, M.F.; Martínez-Cengotitabengoa, M.; Pina-Camacho, L.; Rodríguez-Jiménez, R.; Sáiz, P.A.; Castro, C.; Lafuente, A.; et al. Pro-/anti-inflammatory dysregulation in patients with first episode of psychosis: Toward an integrative inflammatory hypothesis of schizophrenia. Schizophr. Bull. 2014, 40, 376–387. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Pinto, A.; Mosquera, F.; Palomino, A.; Alberich, S.; Gutierrez, A.; Haidar, K.; Vega, K.; Barbeito, S.; Matute, C. Increase in brain-derived neurotrophic factor in first episode psychotic patients after treatment with atypical antipsychotics. Int. Clin. Psychopharmacol. 2010, 25, 241–245. [Google Scholar] [CrossRef]
- Martínez-Cengotitabengoa, M.; MacDowell, K.S.; Alberich, S.; Diaz, F.J.; Garcia-Bueno, B.; Rodriguez-Jimenez, R.; Bioque, M.; Berrocoso, E.; Parellada, M.; Lobo, A.; et al. BDNF and NGF Signalling in Early Phases of Psychosis: Relationship With Inflammation and Response to Antipsychotics After 1 Year. Schizophr. Bull. 2016, 42, 142–151. [Google Scholar] [CrossRef] [Green Version]
- Sanada, K.; Zorrilla, I.; Iwata, Y.; Bermúdez-Ampudia, C.; Graff-Guerrero, A.; Martínez-Cengotitabengoa, M.; González-Pinto, A. The Efficacy of Non-Pharmacological Interventions on Brain-Derived Neurotrophic Factor in Schizophrenia: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2016, 17, 1766. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, B.S.; Molendijk, M.L.; Köhler, C.A.; Soares, J.C.; Leite, C.M.G.S.; Machado-Vieira, R.; Ribeiro, T.L.; Silva, J.C.; Sales, P.M.G.; Quevedo, J.; et al. Peripheral brain-derived neurotrophic factor (BDNF) as a biomarker in bipolar disorder: A meta-analysis of 52 studies. BMC Med. 2015, 13, 289. [Google Scholar] [CrossRef] [Green Version]
- Ruiz de Azua, S.; Matute, C.; Stertz, L.; Mosquera, F.; Palomino, A.; de La Rosa, I.; Barbeito, S.; Vega, P.; Kapczinski, F.; González-Pinto, A. Plasma brain-derived neurotrophic factor levels, learning capacity and cognition in patients with first episode psychosis. BMC Psychiatry 2013, 13, 27. [Google Scholar] [CrossRef] [Green Version]
- Palomino, A.; Vallejo-Illarramendi, A.; González-Pinto, A.M.; Aldama, A.; González-Gómez, C.; Mosquera, F.; González-García, G.; Matute, C. Decreased levels of plasma BDNF in first-episode schizophrenia and bipolar disorder patients. Schizophr. Res. 2006, 86, 321–322. [Google Scholar] [CrossRef]
- Fusar-Poli, P.; McGorry, P.D.; Kane, J.M. Improving outcomes of first-episode psychosis: An overview. World Psychiatry 2017, 16, 251–265. [Google Scholar] [CrossRef]
- Mattai, A.K.; Hill, J.L.; Lenroot, R.K. Treatment of early-onset schizophrenia. Curr. Opin. Psychiatry 2010, 23, 304–310. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE). Psychosis and Schizophrenia in Adults; National Clinical Guideline Number 178; NICE: London, UK, 2014. [Google Scholar]
- Early Psychosis Guidelines Working Group. Australian Clinical Guidelines for Early Psychosis; Orygen The National Centre of Excellence in Youth Mental Health: Melbourne, Australia, 2016. [Google Scholar]
- Galletly, C.; Castle, D.; Dark, F.; Humberstone, V.; Jablensky, A.; Killackey, E.; Kulkarni, J.; McGorry, P.; Nielssen, O.; Tran, N. Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the management of schizophrenia and related disorders. Aust. N. Z. J. Psychiatry 2016, 50, 410–472. [Google Scholar] [CrossRef] [Green Version]
- Scottish Intercollegiate Guidelines Network (SIGN). Management of Schizophrenia; National Clinical Guideline131; SIGN: Edinburgh, UK, 2013. [Google Scholar]
- Guideline Writing Group; Systematic Review Group; Committee on Practice Guideline. The American Psychiatric Association Practice Guideline for the Treatment of Patients with Schizophrenia, 3th ed.; American Psychiatric Association: Washington, DC, USA, 2021. [Google Scholar]
- Ruiz de Azúa, S.; González-Pinto, A.; Vega, P.; Gutíerrez-Fraile, M.; Asua, J. Revisión de Los Tratamientos Psicológicos en Primeros Episodios Psicóticos; Plan de Calidad para el Sistema Nacional de Salud del Ministerio de Sanidad y Política Social Servicio de Evaluación de Tecnologías Sanitarias del País Vasco; Informes de Evaluación de Tecnologías Sanitarias; OSTEBA Nº 2007/08; Servicio Central de Publicaciones del Gobierno Vasco: Vitoria-Gasteiz, Spain, 2009. [Google Scholar]
- Vega, P.; Gonzaléz-Pinto, A.; Ruiz de Azúa, S.; Barbeito, S.; Rico, R. Evaluación de la Efectividad de la Intervención Psicológica en Los Primeros Episodios Psicóticos Mediante Tratamiento Multidisciplinar; Ministerio de Sanidad, Servicios Sociales e Igualdad. Servicio de Evaluación de Tecnologías Sanitarias del País Vasco; Informes de Evaluación de Tecnologías Sanitarias; OSTEBA Servicio Central de Publicaciones del Gobierno Vasco: Vitoria-Gasteiz, Spain, 2014. [Google Scholar]
- Breitborde, N.J.; Bell, E.K.; Dawley, D.; Woolverton, C.; Ceaser, A.; Waters, A.C.; Dawson, S.C.; Bismark, A.W.; Polsinelli, A.J.; Bartolomeo, L.; et al. The Early Psychosis Intervention Center (EPICENTER): Development and six-month outcomes of an American first-episode psychosis clinical service. BMC Psychiatry 2015, 15, 266. [Google Scholar] [CrossRef] [Green Version]
- Correll, C.U.; Galling, B.; Pawar, A.; Krivko, A.; Bonetto, C.; Ruggeri, M.; Craig, T.J.; Nordentoft, M.; Srihari, V.H.; Guloksuz, S.; et al. Comparison of Early Intervention Services vs Treatment as Usual for Early-Phase Psychosis: A Systematic Review, Meta-analysis, and Meta-regression. JAMA Psychiatry 2018, 75, 555–565. [Google Scholar] [CrossRef]
- Fowler, D.; Hodgekins, J.; French, P.; Marshall, M.; Freemantle, N.; McCrone, P.; Everard, L.; Lavis, A.; Jones, P.B.; Amos, T.; et al. Social recovery therapy in combination with early intervention services for enhancement of social recovery in patients with first-episode psychosis (SUPEREDEN3): A single-blind, randomised controlled trial. Lancet Psychiatry 2018, 5, 41–50. [Google Scholar] [CrossRef] [Green Version]
- Lasalvia, A.; Bonetto, C.; Lenzi, J.; Rucci, P.; Iozzino, L.; Cellini, M.; Comacchio, C.; Cristofalo, D.; D’Agostino, A.; de Girolamo, G.; et al. Predictors and moderators of treatment outcome in patients receiving multi-element psychosocial intervention for early psychosis: Results from the GET UP pragmatic cluster randomised controlled trial. Br. J. Psychiatry 2017, 210, 342–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lutgens, D.; Iyer, S.; Joober, R.; Brown, T.G.; Norman, R.; Latimer, E.; Schmitz, N.; Baki, A.A.; Abadi, S.; Malla, A. A five-year randomized parallel and blinded clinical trial of an extended specialized early intervention vs. regular care in the early phase of psychotic disorders: Study protocol. BMC Psychiatry 2015, 15, 22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- North, C.S.; Simic, Z.; Burruss, J. Design, Implementation, and Assessment of a Public Comprehensive Specialty Care Program for Early Psychosis. J. Psychiatr Pract. 2019, 25, 91–102. [Google Scholar] [CrossRef]
- Ruggeri, M.; Bonetto, C.; Lasalvia, S.; Fioritti, A.; de Girolamo, G.; Santonastaso, P.; Pileggi, F.; Neri, G.; Ghigi, D.; Giubilini, F.; et al. Feasibility and Effectiveness of a Multi-Element Psychosocial Intervention for First-Episode Psychosis: Results from the Cluster-Randomized Controlled GET UP PIANO Trial in a Catchment Area of 10 Million Inhabitants. Schizophr. Bull. 2015, 41, 1192–1203. [Google Scholar] [CrossRef]
- Rosenheck, R.; Leslie, D.; Sint, K.; Lin, H.; Robinson, D.G.; Schooler, N.R.; Mueser, K.T.; Penn, D.L.; Addington, J.; Brunette, M.F.; et al. Cost-effectiveness of comprehensive, integrated care for first episode psychosis in the NIMH RAISE Early Treatment Program. Schizophr. Bull. 2016, 42, 896–906. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Secher, R.G.; Hjorthøj, C.R.; Austin, S.F.; Thorup, A.; Jeppesen, P.; Mors, O.; Nordentoft, M. Ten-Year Follow-up of the OPUS Specialized Early Intervention Trial for Patients with a First Episode of Psychosis. Schizophr. Bull. 2015, 41, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Barbeito, S.; Vega, P.; Ruiz de Azua, S.; Balanza- Martinez, V.; Colom, F.; Lorente, E.; Luengo, A.; Cerrillo, E.; Crespo, J.M.; González-Pinto, A. Integrated treatment of first episode psychosis with online training (e-learning): Study protocol for a randomised controlled trial. Trials 2014, 15, 416. [Google Scholar] [CrossRef] [Green Version]
- McFarlane, W.R.; Levin, B.; Travis, L.; Lucas, F.L.; Lynch, S.; Verdi, M.; Williams, D.; Adelsheim, S.; Calkins, R.; Carter, C.S.; et al. Clinical and functional outcomes after 2 years in the Early Detection and Intervention for the Prevention of Psychosis multisite effectiveness trial. Schizophr. Bull. 2015, 41, 30–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, S.H.; Thornicroft, G.; Coffey, M.; Dunn, G. A brief mental health outcome scale-reliability and validity of the Global Assessment of Functioning (GAF). Br. J. Psychiatry 1995, 166, 654–659. [Google Scholar] [CrossRef]
- American Psychiatric Association (APA). Diagnostic and Statistical Manual of Mental Disorders (DSM IV-TR), 4th ed.; text rev.; American Psychiatric Press: Washington, DC, USA, 2000. [Google Scholar]
- Rosa, A.R.; Sánchez-Moreno, J.; Martıínez-Aran, A.; Salamero, M.; Torrent, C.; Reinares, M.; Comes, M.; Colom, F.; Van Riel, W.; Ayuso-Mateos, J.L.; et al. Validity and reliability of the Functioning Assessment Short Test (FAST) in bipolar disorder. Clin. Pract. Epidemio. Ment. Health 2007, 3, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González-Ortega, I.; Rosa, A.; Alberich, S.; Barbeito, S.; Vega, P.; Echeburúa, E.; Vieta, E.; González-Pinto, A. Validation and use of the functioning assessment short test in first psychotic episodes. J. Nerv. Ment. Dis. 2010, 198, 836–840. [Google Scholar] [CrossRef] [Green Version]
- First, M.B.; Spitzer, R.; Gibbon, M. Structured Clinical Interview for DSM-IV. Axis I Disorders; American Psychiatric Press Inc.: Washington, DC, USA, 1997. [Google Scholar]
- Guy, W. ECDEU Assessment Manual for Psychopharmacology-Revised; Department of Health, Education and Welfare: Rockville, MD, USA, 1976. [Google Scholar]
- Kay, S.R.; Fiszbein, A.; Opler, L.A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 1987, 13, 261–276. [Google Scholar] [CrossRef]
- Peralta, V.; Cuesta, M. Validación de la escala de los síndromes positivo y negativo (PANNS) en una muestra de esquizofrénicos españoles. Actas. Luso. Esp. Neurol. Psiquiatr. 1994, 22, 171–177. [Google Scholar]
- Guillén-Riquelme, A.; Buela-Casal, G. Actualización psicométrica y funcionamiento diferencial de los ítems en el State Trait Anxiety Inventory (STAI). Psicothema 2011, 23, 510–515. [Google Scholar]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.; Vagg, P.R.; Jacobs, G.A. Manual for the State-Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Hamilton, M. A rating scale for depression. J. Neurol. Neurosurg. Psychiatry 1960, 23, 56–62. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ramos-Brieva, J.A.; Cordero, A. Validación de la versión castellana de la escala Hamilton para la depresión. Actas. Luso. Esp. Neurol. Psiquiatr. Cienc. Afines. 1986, 14, 324–334. [Google Scholar]
- Amador, X.F.; Strauss, D.H.; Yale, S.A.; Flaum, M.M.; Endicott, J.; Gorman, J.M. Assessment of insight in psychosis. Am. J. Psychiatry 1993, 150, 873–879. [Google Scholar] [CrossRef] [Green Version]
- Ruiz, A.; Pousa, E.; Duñó, R.; Crosas, J.; Cuppa, S.; García, C. Spanish adaptation of the Scale to Assess Unawareness of Mental Disorder (SUMD). Actas. Esp. Psiquiatr. 2008, 36, 111–1198. [Google Scholar]
- Morisky, D.E.; Green, L.W.; Levine, D.M. Concurrent and predictive validity of a self-reported measure of medication adherence. Med. Care 1986, 24, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Val, A.; Amoró, G.; Martínez, P.; Fernández, M.L.; León, M. Descriptive study of patient compliance in pharmacologic antihypertensive treatment and validation of the Morisky and Green test. Aten. Primaria 1992, 10, 767–770. (In Spanish) [Google Scholar]
- Iorfino, F.; Hermens, D.F.; Cross, S.P.; Zmicerevska, N.; Nichles, A.; Badcock, C.A.; Groot, J.; Scott, E.M.; Hickie, I.B. Delineating the trajectories of social and occupational functioning of young people attending early intervention mental health services in Australia: A longitudinal study. BMJ Open. 2018, 8, e020678. [Google Scholar] [CrossRef] [Green Version]
- Bozzatello, P.; Bellino, S.; Rocca, P. Predictive Factors of Treatment Resistance in First Episode of Psychosis: A Systematic Review. Front. Psychiatry 2019, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Demjaha, A.; Lappin, J.M.; Stahl, D.; Patel, M.X.; MacCabe, J.H.; Howes, O.D.; Heslin, M.; Reininghaus, U.A.; Donoghue, K.; Lomas, B.; et al. Antipsychotic treatment resistance in first-episode psychosis: Prevalence, subtypes and predictors. Psychol. Med. 2017, 47, 1981–1989. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wunderink, L.; Bebber, J.V.; Sytema, S.; Boonstra, N.; Meijer, R.R.; Wigman, J.T.W. Negative symptoms predict high relapse rates and both predict less favorable functional outcome in first episode psychosis, independent of treatment strategy. Schizophr. Res. 2020, 216, 192–199. [Google Scholar] [CrossRef]
- Lutgens, D.; Gariepy, G.; Malla, A. Psychological and psychosocial interventions for negative symptoms in psychosis: Systematic review and meta-analysis. Br. J. Psychiatry 2017, 210, 324–332. [Google Scholar] [CrossRef] [PubMed]
- McGinty, J.; Upthegrove, R. Depressive symptoms during first episode psychosis and functional outcome: A systematic review and meta-analysis. Schizophr. Res. 2020, 218, 14–27. [Google Scholar] [CrossRef] [PubMed]
- González-Ortega, I.; Alberich, S.; Echeburúa, E.; Aizpuru, F.; Millán, E.; Vieta, E.; Matute, C.; González-Pinto, A. Subclinical Depressive Symptoms and Continued Cannabis Use: Predictors of Negative Outcomes in First Episode Psychosis. PLoS ONE 2015, 10, e0123707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raghavan, V.; Mohan, G.; Gopal, S.; Ramamurthy, M.; Rangaswamy, T. Medication adherence in first-episode psychosis and its association with psychopathology. Indian. J. Psychiatry 2019, 61, 342–346. [Google Scholar] [CrossRef]
- Tan, C.; Abdin, E.; Liang, W.; Poon, L.Y.; Poon, N.Y.; Verma, S. Medication adherence in first-episode psychosis patients in Singapore. Early Interv. Psychiatry 2018, 13, 780–788. [Google Scholar] [CrossRef]
- Randall, J.; Chateau, D.; Bolton, J.M.; Smith, M.; Katz, L.; Burland, E.; Taylor, C.; Nickel, N.C.; Enns, J.; Katz, A.; et al. Increasing medication adherence and income assistance access for first-episode psychosis patients. PLoS ONE 2017, 12, e0179089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, J.G.; Roh, D.; Kim, C.H. Clinical Psychopharmacology and Neuroscience: The Official Scientific Journal of the Korean College of Neuropsychopharmacology. Eur. PMC 2019, 17, 273–278. [Google Scholar] [CrossRef]
- Reinares, M.; Martinez-Aran, A.; Vieta, E. Psychotherapy for Bipolar Disorders: An Integrative Approach; Cambridge University Press: Cambridge, UK, 2020. [Google Scholar] [CrossRef]
| Variable | Total (n = 177) | CBT + TAU | TAU | t/χ2 | p | |
|---|---|---|---|---|---|---|
| Sex | Women | 72 (40.7%) | 39 (45.3%) | 33 (36.3%) | χ2 = 1.512 | 0.219 |
| Age | 28.53 ± 8.97 | 27.74 ± 7.66 | 29.27 ± 10.03 | t = −1.136 | 0.258 | |
| Occupation | Employed | 69 (39.2%) | 31 (35.7%) | 39 (42.5%) | χ2 = 0.863 | |
| Unemployed | 57 (32.2%) | 30 (34.5%) | 27 (29.9%) | 0.650 | ||
| Student | 51 (28.7%) | 26 (29.8%) | 25 (27.6%) | |||
| Education level | Primary school | 6 (3.4%) | 1 (1.4%) | 5 (5.3%) | χ2 = 2.784 | 0.249 |
| Secondary school | 128 (72.1%) | 67 (77.5%) | 61 (67.1%) | |||
| College | 43 (24.5%) | 18 (21.1%) | 25 (27.6%) | |||
| Civil status | Single | 146 (82.5%) | 72 (83.3%) | 74 (81.6%) | χ2 = 0.136 | |
| Married | 23 (12.9%) | 10 (11.9%) | 13 (13.8%) | 0.934 | ||
| Divorced | 8 (4.7%) | 4 (4.8%) | 4 (4.6%) | |||
| Diagnosis | Non-specified psychotic disorder | 118 (66.7%) | 58 (67.4%) | 60 (65.9%) | χ2 = 2.500 | 0.776 |
| Bipolar disorder with psychotic symptoms | 23 (13%) | 13 (15.1%) | 10 (11%) | |||
| Schizophreniform disorder | 17 (9.6%) | 6 (7%) | 11 (12.1%) | |||
| Brief psychotic disorder | 13 (7.3%) | 6 (7%) | 7 (7.7%) | |||
| Major depressive disorder with psychotic symptoms | 3 (1.7%) | 1 (1.2%) | 2 (2.2%) | |||
| Delusional disorder | 3 (1.7%) | 2 (2.3%) | 1 (1.1%) | |||
| Treatment | Antipsychotics | 171 (97.2% | 85 (98.8%) | 86 (95.6%) | χ2 = 1.716 | 0.190 |
| Benzodiazepines | 77 (43.8%9 | 35 (40.7%) | 42 (46.7%) | χ2 = 0.637 | 0.425 | |
| Mood stabilisers | 24 (13.6%) | 12 (14%) | 12 (13.3%) | χ2 = 0.014 | 0.905 | |
| Antidepressants | 20 (11.4%) | 11 (12.8%) | 9 (10%) | χ2 = 0.340 | 0.560 | |
| FAST | 31.61 ± 15.81 | 33,09 ± 15,519 | 30,16 ± 16,053 | t = −1.218 | 0.225 | |
| GAF | 58.54 ± 11.0 | 59.63 ± 9.99 | 56.78 ± 11.82 | t = 1.133 | 0.259 | |
| CGI-S | 7.64 ± 5.75 | 7.74 ± 5.94 | 7.55 ± 5.60 | t = 0.224 | 0.823 | |
| CGI-I | 8.65 ± 7.77 | 9.06 ± 8.10 | 8.26 ± 7.46 | t = 0.677 | 0.499 | |
| PANSS P | 14.16 ± 6.95 | 13.40 ± 6.46 | 15.36 ± 7.71 | t = −1.960 | 0.052 | |
| PANSS N | 13.69 ± 6.27 | 14.48 ± 6.96 | 13.19 ± 5.83 | t = 1.635 | 0.104 | |
| PANSS PG | 29.19 ± 7.98 | 29.64 ± 8.68 | 28.77 ± 7.2 | t = 0.886 | 0.377 | |
| HDRS | 11.75 ± 7.40 | 12.46 ± 8.22 | 11.07 ± 6.51 | t = 1.241 | 0.219 | |
| STAI-State | 24.82 ± 10.90 | 23.87 ± 10.96 | 26.14 ± 10.59 | t = −0.892 | 0.374 | |
| SUMD | 4.94 ± 3.18 | 5.50 ± 3.05 | 5.47 ± 3.95 | t = −0.054 | 0.957 | |
| MMAS | 97 (54.8%) | 52 (60.0%) | 45 (49.4%) | χ2 = 1.940 | 0.164 | |
| BDNF (pg/Ml) | 6.95 ± 5.77 | 7.66 ± 6.79 | 6.49 ± 5.05 | t = −0.839 | 0.404 |
| Group | CBT + TAU | TAU | ||||
|---|---|---|---|---|---|---|
| Variable | Pre | Post | t (p) | Pre | Post | t (p) |
| FAST | 33.43 ± 15.531 | 15.15 ± 16.425 | 7.638 (≤0.001) | 30.96 ± 20.05 | 20.05 ± 15.221 | 6.109 (≤0.001) |
| GAF | 61.24 ± 10.62 | 76.03 ± 13.60 | −7.669 (≤0.001) | 56.92 ± 12.03 | 69.58 ± 13.91 | −7.310 (0.001) |
| CGI-S | 6.74 ± 5.43 | 6.09 ± 6.61 | 1.519 (0.133) | 7.16 ± 5.41 | 6.99 ± 6.68 | 0.380 (0.705) |
| CGI-I | 8.08 ± 7.81 | 7.07 ± 7.57 | 2.131 (0.037) | 7.48 ± 7.03 | 6.25 ± 6.09 | 3.033 (0.003) |
| Panns P | 13.34 ± 6.44 | 8.47 ± 3.22 | 6.365 (≤0.001) | 15.39 ± 7.66 | 10.39 ± 4.90 | 5.705 (≤0.01) |
| Panns N | 14.78 ± 7.49 | 10.55 ± 5.71 | 5.394 (≤0.001) | 13.41 ± 6.29 | 11.39 ± 5.43 | 3.787 (≤0.001) |
| Panns G | 30.17 ± 10.33 | 21.35 ± 7.60 | 6.738 (≤0.001) | 28.81 ± 7.29 | 23.33 ± 6.67 | 6.861 (≤0.001) |
| HDRS | 12.31 ± 8.31 | 5.24 ± 5.58 | 6.434 (≤0.001) | 11.30 ± 6.28 | 6.65 ± 4.96 | 8.066 (≤0.001) |
| STAI-State | 24.11 ± 10.91 | 19.05 ± 9.18 | 3.192 (0.002) | 25.97 ± 11.30 | 21.28 ± 7.93 | 3.309 (0.002) |
| SUMD | 4.67 ± 3.05 | 3.07 ± 2.88 | 6.067 (≤0.001) | 4.64 ± 2.97 | 3.93 ± 3.03 | 2.697 (0.008) |
| MMAS (good) 1 | 51 (60.0%) | 55 (72.4%) | p = 0.052 1 | 38 (43.7%) | 36 (45.6%) | p = 0.690 1 |
| BDNF (pg/Ml) | 7.66 ± 6.79 | 9.76 ± 6.78 | −1.760; (0.090) | 6.49 ± 5005 | 6.95 ± 4.39 | −0.675 (0.503) |
| Variable | CBT + TAU Δ | TAU Δ | F | p | |
|---|---|---|---|---|---|
| FAST | −18.28 ± 20.73 | −10.91 ± 15.88 | 5.468 | 0.021 | 0.04 |
| GAF | 14.79 ± 16.70 | 12.66 ± 15.78 | 6.269 | 0.013 | 0.04 |
| CGI-S | −0.65 ± 3.66 | −0.17 ± 3.98 | 0.605 | 0.438 | 0.00 |
| CGI-I | −1.01 ± 3.98 | −1.24 ± 3.62 | 0.379 | 0.539 | 0.00 |
| Panns P | −4.87 ± 6.67 | −5.00 ± 7.94 | 6.214 | 0.014 | 0.04 |
| Panns N | −4.22 ± 6.83 | −2.02 ± 4.84 | 4.008 | 0.047 | 0.03 |
| Panns G | −8.83 ± 11.34 | −5.48 ± 7.19 | 4.626 | 0.033 | 0.03 |
| HDRS | −7.07 ± 9.51 | −4.65 ± 5.16 | 4.078 | 0.045 | 0.03 |
| STAI State | −5.05 ± 11.95 | −4.69 ± 11.69 | 1.494 | 0.224 | 0.01 |
| SUMD | −1.60 ± 2.28 | −0.71 ± 2.43 | 6.564 | 0.011 | 0.04 |
| MMAS (good) 1 | 12.4% | 1.9% | 1.002 | 0.007 | 2.723 |
| BDNF (pg/Ml) | 2.10 ± 6.30 | 0.46 ± 4.55 | 3.923 | 0.050 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Ortega, I.; Vega, P.; Echeburúa, E.; Alberich, S.; Fernández-Sevillano, J.; Barbeito, S.; Balanzá-Martínez, V.; Vieta, E.; Lorente-Rovira, E.; Luengo, A.; et al. A Multicentre, Randomised, Controlled Trial of a Combined Clinical Treatment for First-Episode Psychosis. Int. J. Environ. Res. Public Health 2021, 18, 7239. https://doi.org/10.3390/ijerph18147239
González-Ortega I, Vega P, Echeburúa E, Alberich S, Fernández-Sevillano J, Barbeito S, Balanzá-Martínez V, Vieta E, Lorente-Rovira E, Luengo A, et al. A Multicentre, Randomised, Controlled Trial of a Combined Clinical Treatment for First-Episode Psychosis. International Journal of Environmental Research and Public Health. 2021; 18(14):7239. https://doi.org/10.3390/ijerph18147239
Chicago/Turabian StyleGonzález-Ortega, Itxaso, Patricia Vega, Enrique Echeburúa, Susana Alberich, Jessica Fernández-Sevillano, Sara Barbeito, Vicent Balanzá-Martínez, Eduard Vieta, Esther Lorente-Rovira, Ana Luengo, and et al. 2021. "A Multicentre, Randomised, Controlled Trial of a Combined Clinical Treatment for First-Episode Psychosis" International Journal of Environmental Research and Public Health 18, no. 14: 7239. https://doi.org/10.3390/ijerph18147239
APA StyleGonzález-Ortega, I., Vega, P., Echeburúa, E., Alberich, S., Fernández-Sevillano, J., Barbeito, S., Balanzá-Martínez, V., Vieta, E., Lorente-Rovira, E., Luengo, A., Cerrillo, E., Crespo, J. M., Matute, C., & González-Pinto, A. (2021). A Multicentre, Randomised, Controlled Trial of a Combined Clinical Treatment for First-Episode Psychosis. International Journal of Environmental Research and Public Health, 18(14), 7239. https://doi.org/10.3390/ijerph18147239








