Organic Synthesis of New Secosteroids from Fucosterol, Its Intestinal Absorption by Caco-2 Cells, and Simulation of the Biological Activities of Vitamin D
Abstract
1. Introduction
2. Results
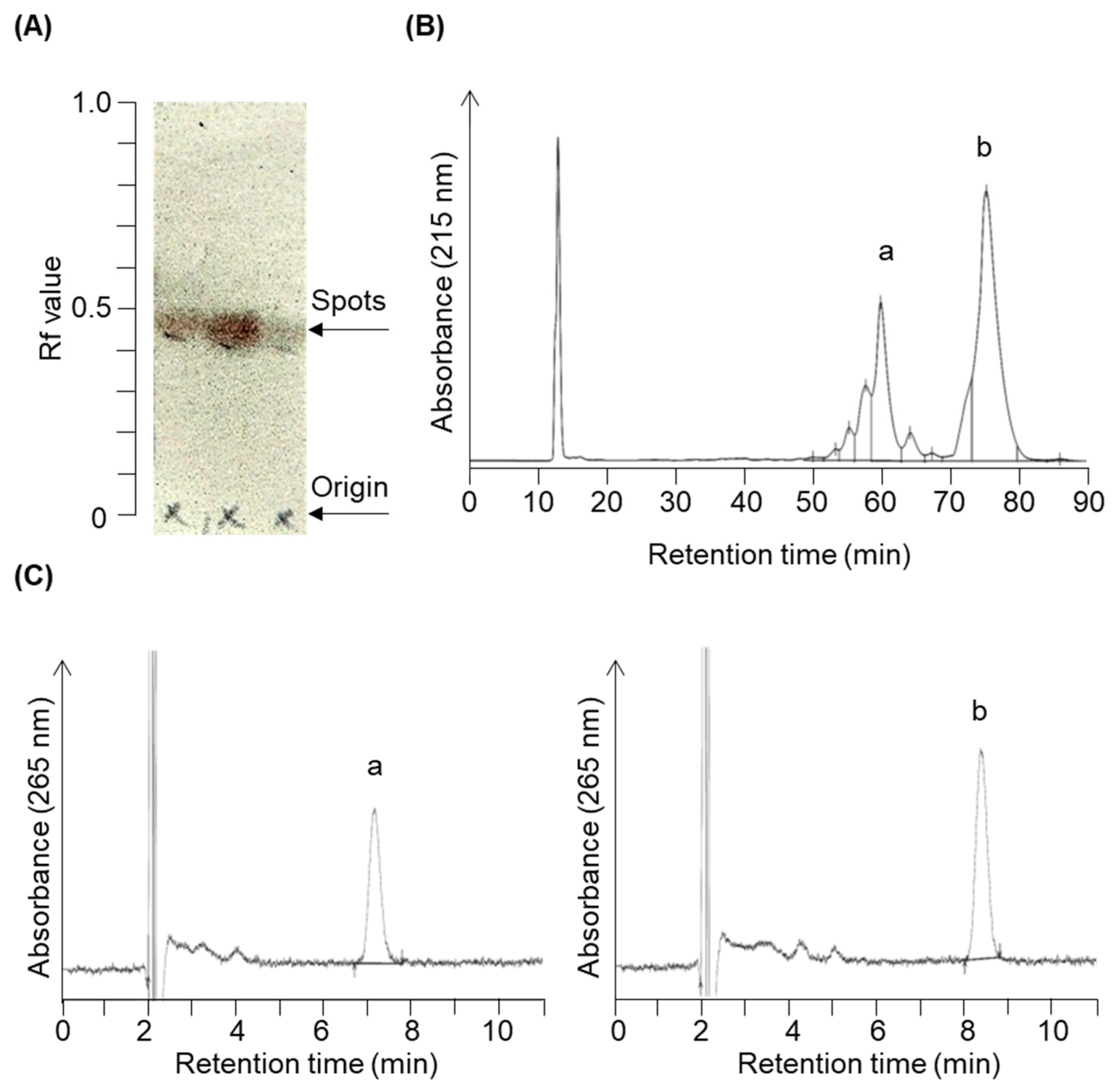
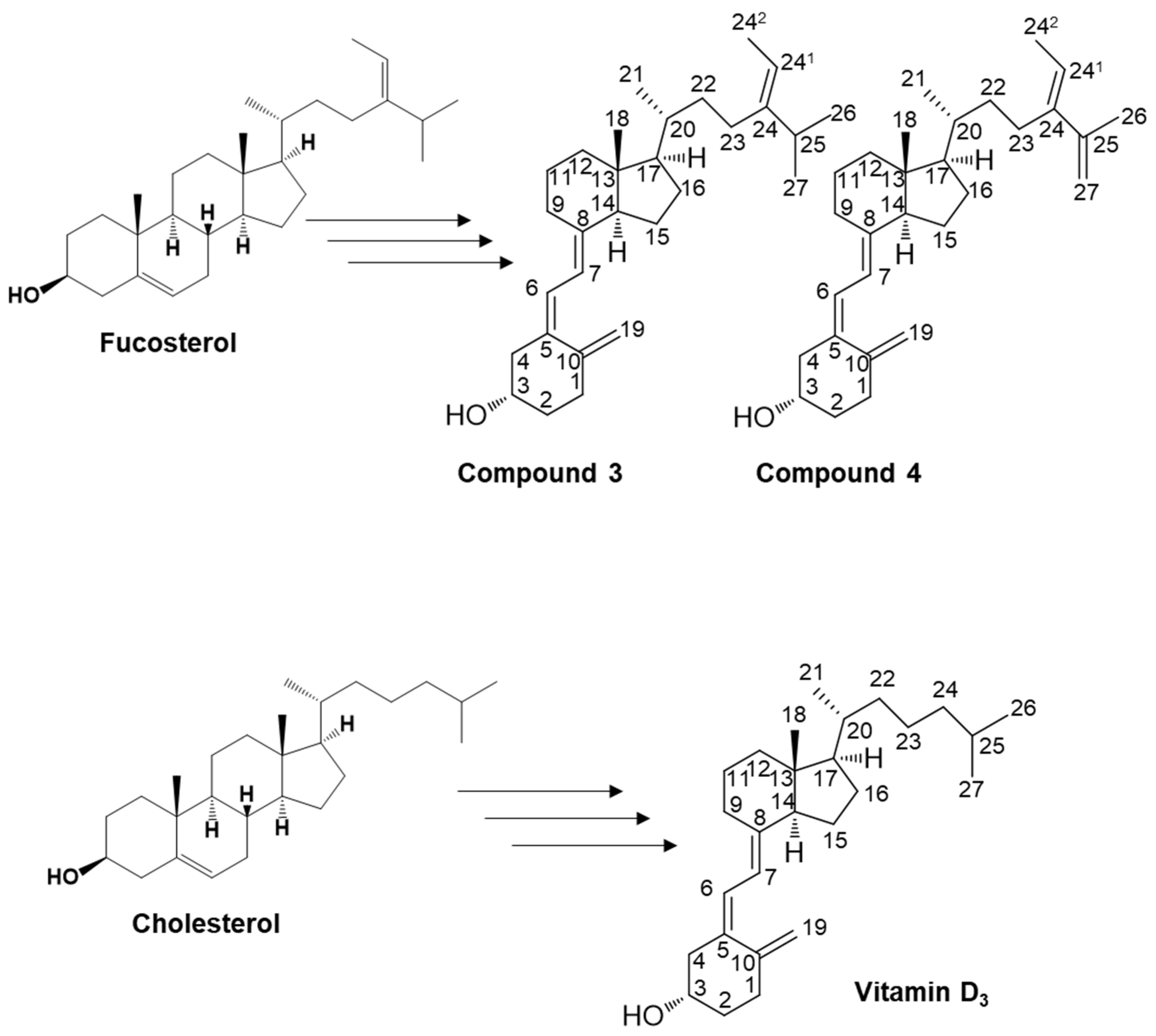
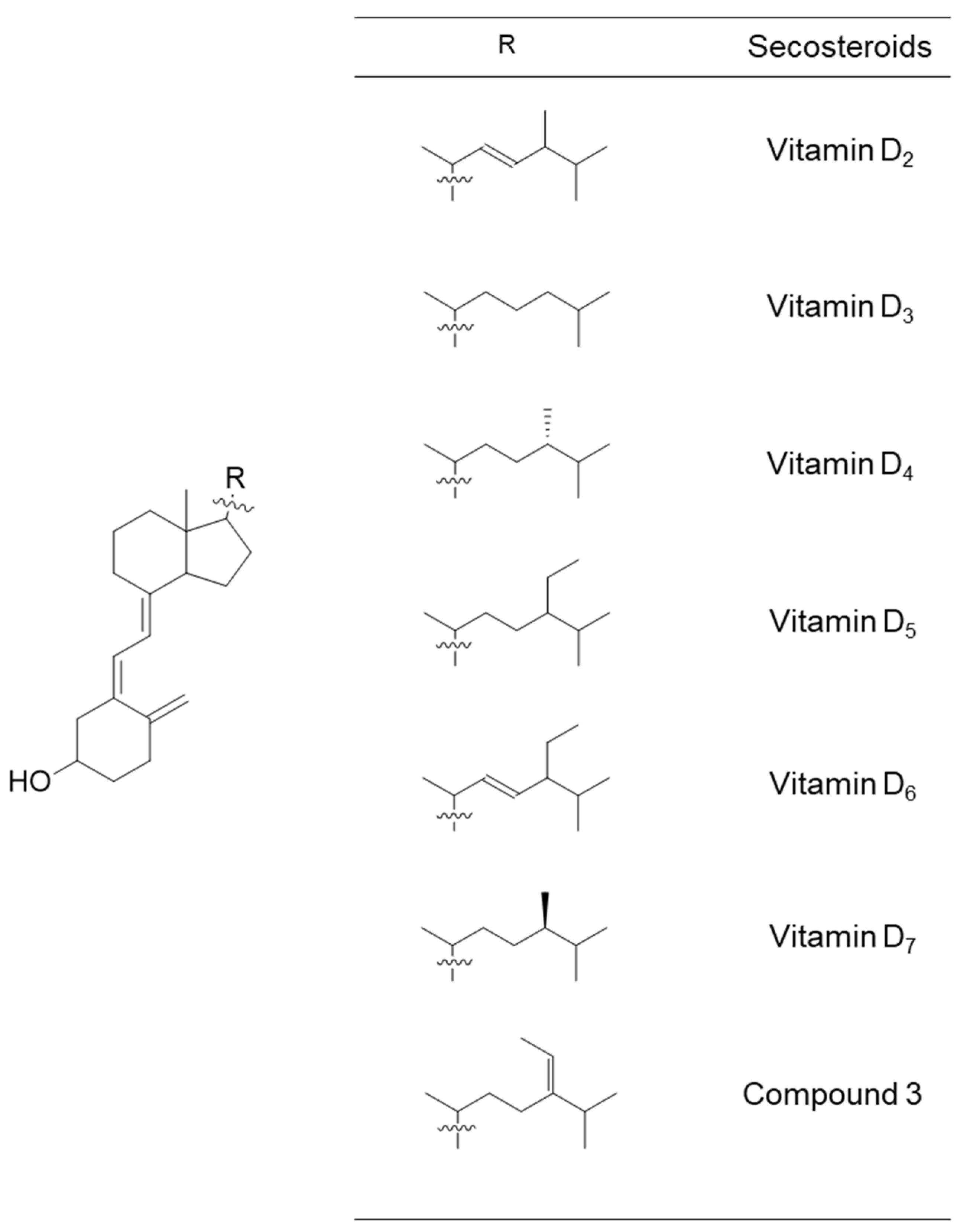
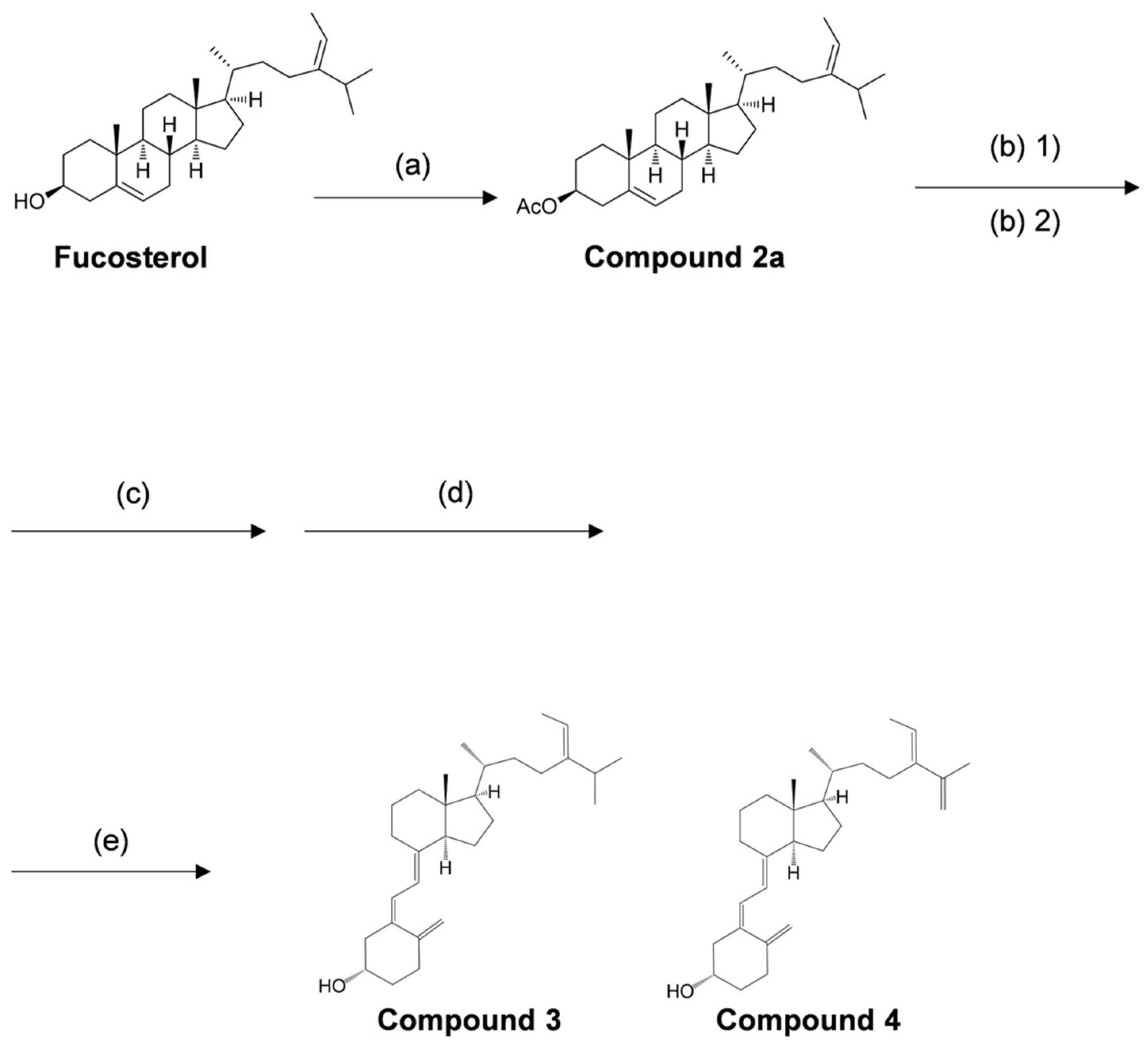
2.1. Organic Synthesis
2.2. Cell Biochemistry
2.2.1. Levels of Micellar Secosteroid Remaining
2.2.2. Uptake of Secosteroid by Differentiated Caco-2 Cells
2.2.3. Transport of Secosteroid by Differentiated Caco-2 Cells in Tight Monolayer Configuration
2.3. Estimating the Biological Activity of Secosteroids
2.3.1. PASS Online Simulation Part 1
2.3.2. PASS Online Simulation Part 2
2.3.3. PASS Online Simulation Part 3
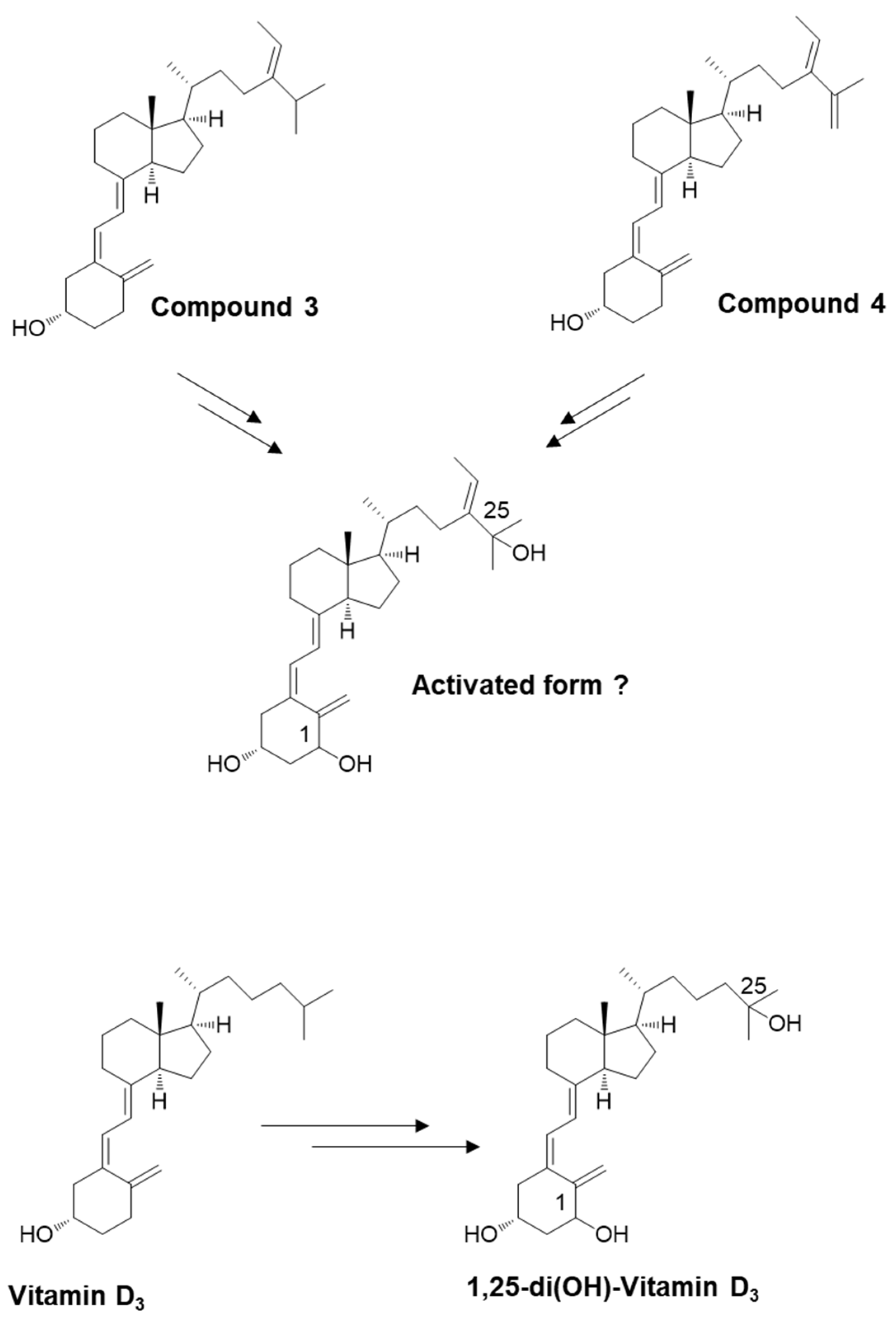
3. Discussion
4. Materials and Methods
4.1. Organic Synthesis
4.1.1. Reagents and Conditions for Organic Synthesis
4.1.2. Organic Synthesis of New Secosteroids
4.2. Cell Biochemistry
4.2.1. Materials
4.2.2. Cells and Culture
4.2.3. Human Intestinal Cell Model
4.2.4. Mixed Micelles Preparation
4.2.5. Evaluation of Secosteroid Intestinal Absorption (Uptake and Transport)
- (i):
- Initially, 20 µL of 0.2 mM trolox/ethyl alcohol was added to the collected basal-side medium (approximately 2.5 mL). Then, the obtained mixture (2.2 mL) was combined with the internal standard (0.2 mL). Subsequently, ethyl alcohol (2.0 mL), acetic ether (2.2 mL), and normal hexane (2.2 mL) were added, with the solution shaken using a Vortex mixer after each addition. The upper phase of the two-layered solution was collected, and the bottom phase underwent the same procedure with the addition of acetic ether and normal hexane, followed by shaking. The upper phases were combined, and the phase was dried using a centrifugal evaporator. The resulting extract was dissolved in 200 µL of dichloromethane/methyl alcohol/water (38:152:10, v/v/v), with an aliquot (60 µL) submitted to HPLC condition 2, described below.In the event of any disruption to the tight monolayer due to an experimental mishap, the target component (in this case, secosteroid) could leak from the apical to the basolateral side. Phenol red in the apical-side medium was used to indicate such leakage. After incubation, phenol red concentration in the collected basolateral medium (not present initially) was measured at 560 nm under alkaline conditions. The Tecan Infinite F50R microplate reader (Tecan Group Ltd., Männedorf, Switzerland) was used for this measurement.
- (ii):
- The differentiated Caco-2 cells, forming the tight monolayers, were rinsed twice with Hank’s balanced salt solution. These cells were gathered in glass tubes by gently detaching them from the polycarbonate membrane of the insert wells using the back of a spatula. The purpose was to measure the cellular uptake of secosteroids. The cells were homogenized in 2.0 mL of PBS containing 20 µL of 0.2 mM trolox/ethyl alcohol using a probe-type sonicator (Ultra S, VP-5S; Taitec, Saitama, Japan). A portion (1.8 mL) of the cell homogenate suspension was mixed with 0.2 mL of the internal standard. Following this, 1.6 mL of ethyl alcohol, 1.8 mL of acetic ether, and 1.8 mL of normal hexane were introduced. Subsequent extraction and drying steps were performed as described in i). The resulting extract was dissolved in 200 µL of dichloromethane/methyl alcohol/water (38:152:10, v/v/v). This solution’s aliquot (20 µL) was submitted for HPLC under condition 2, described below.The protein content of the cells was assessed using the DC protein assay kit (Bio-Rad Laboratories, Hercules, CA, USA). An aliquot (50 µL) of the cell homogenate suspension was diluted fourfold in PBS and applied to the kit. The final reaction solution’s absorbance was measured at 750 nm using the same microplate reader mentioned earlier. Cellular uptake of secosteroid was normalized based on this protein level.
- (iii):
- The reduction in micellar secosteroid due to aggregation might have influenced secosteroid uptake from mixed micelles. This is because only micellar secosteroids could be assimilated by Caco-2 cells, as explained in our prior report [49].
4.2.6. HPLC Analysis (HPLC Condition 2)
4.2.7. Statistical Analysis
4.3. Online Simulation for Estimating Biological Activities of Secosteroids
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Angus, T.C.; Askew, F.A.; Bourdillon, R.B.; Bruce, H.M.; Callow, R.K.; Fischmann, C.; Philpot, J.S.L.; Webster, T.A. A crystalline antirachitic substance. Proc. R. Soc. London Ser. B Contain. Pap. A Biol. Character 1931, 108, 340–359. [Google Scholar]
- Windaus, A.; Linsert, O.; Lüttringhaus, A.; Weidlich, G. Über das krystallisierte vitamin D2. Eur. J. Org. Chem. 1932, 492, 226–241. [Google Scholar] [CrossRef]
- Zeng, R.; Li, Y.; Shen, S.; Qiu, X.; Chang, C.L.; Koplin, J.J.; Perrett, K.P.; Dharmage, S.C.; Lodge, C.J.; Lowe, A.J. Is antenatal or early-life vitamin D associated with eczema or food allergy in childhood? A systematic review. Clin. Exp. Allergy 2023, 53, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.L.; Pham, T.T.M.; Chen, Y.C.; Chang, J.S.; Chao, J.C.; Bai, C.H. Effects of climate, sun exposure, and dietary intake on vitamin D concentrations in pregnant women: A population-based study. Nutrients 2023, 15, 1182. [Google Scholar] [CrossRef]
- Eller, A.B.P.; Ejzenberg, D.; Monteleone, P.A.A.; Soares, J.M., Jr.; Baracat, E.C. Vitamin D and in vitro fertilization: A systematic review. J. Assist. Reprod. Genet. 2023, 40, 735–743. [Google Scholar] [CrossRef]
- Grzesiak, M.; Tchurzyk, M.; Socha, M.; Sechman, A.; Hrabia, A. An overview of the current known and unknown roles of vitamin D3 in the female reproductive system: Lessons from farm animals, birds, and fish. Int. J. Mol. Sci. 2022, 23, 14137. [Google Scholar] [CrossRef]
- Calagna, G.; Catinella, V.; Polito, S.; Schiattarella, A.; De Franciscis, P.; D’Antonio, F.; Calì, G.; Perino, A.; Cucinella, G. Vitamin D and male reproduction: Updated evidence based on literature review. Nutrients 2022, 14, 3278. [Google Scholar] [CrossRef]
- Uchiyama, M.; Mizukami, S.; Arima, K.; Nishimura, T.; Tomita, Y.; Abe, Y.; Tanaka, N.; Honda, Y.; Goto, H.; Hasegawa, M.; et al. Association between serum 25-hydroxyvitamin D and physical performance measures in middle-aged and old Japanese men and women: The Unzen study. PLoS ONE 2021, 16, e0261639. [Google Scholar] [CrossRef]
- Lee, J.; Bae, E.H.; Kim, S.W.; Chung, W.; Kim, Y.H.; Oh, Y.K.; Kim, Y.S.; Oh, K.H.; Park, S.K. The association between vitamin D deficiency and risk of renal event: Results from the Korean cohort study for outcomes in patients with chronic kidney disease (KNOW-CKD). Front. Med. 2023, 10, 1017459. [Google Scholar] [CrossRef]
- De Menezes-Júnior, L.A.A.; Sabião, T.D.S.; de Moura, S.S.; Batista, A.P.; de Menezes, M.C.; Carraro, J.C.C.; Andrade, A.C.S.; Machado-Coelho, G.L.L.; Meireles, A.L. Influence of sunlight on the association between 25-hydroxyvitamin D levels and sleep quality in Brazilian adults: A population-based study. Nutrition 2023, 110, 112008. [Google Scholar] [CrossRef]
- Rosso, C.; Fera, N.; Murugan, N.J.; Voutsadakis, I.A. Vitamin D levels in newly diagnosed breast cancer patients according to tumor sub-types. J. Diet. Suppl. 2022, 20, 926–938. [Google Scholar] [CrossRef] [PubMed]
- Hysaj, O.; Karavasiloglou, N.; Limam, M.; Wanner, M.; Korol, D.; Rohrmann, S. Is season of diagnosis a predictor of cancer survival? Results from the Zurich cancer registry. Nutrients 2022, 14, 4291. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Deng, L.; Huangfu, Y.; Zhou, Y.; Wang, P.; Shen, L. Adequate vitamin D level associated with reduced risk of sporadic colorectal cancer. Front. Nutr. 2023, 10, 1024849. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, E.M.; Rylander, C.; Brustad, M.; Jensen, T.E. Pre-diagnostic intake of vitamin D and incidence of colorectal cancer by anatomical subsites: The Norwegian women and cancer cohort study (NOWAC). Br. J. Nutr. 2023, 130, 1047–1055. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Liu, B.; Bao, W.; Rong, S. Association between serum 25-hydroxyvitamin D concentrations and respiratory infection among United States adults. J. Nutr. 2023, 153, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Wannamethee, S.G.; Welsh, P.; Papacosta, O.; Lennon, L.; Whincup, P. Vitamin D deficiency, impaired lung function and total and respiratory mortality in a cohort of older men: Cross-sectional and prospective findings from the British regional heart study. BMJ Open 2021, 11, e051560. [Google Scholar] [CrossRef]
- Tamara, L.; Kartasasmita, C.B.; Alam, A.; Gurnida, D.A. Effects of Vitamin D supplementation on resolution of fever and cough in children with pulmonary tuberculosis: A randomized double-blind controlled trial in Indonesia. J. Glob. Health 2022, 12, 04015. [Google Scholar] [CrossRef]
- Acen, E.L.; Biraro, I.A.; Bbuye, M.; Kateete, D.P.; Joloba, M.L.; Worodria, W. Hypovitaminosis D among newly diagnosed pulmonary TB patients and their household contacts in Uganda. Sci. Rep. 2022, 12, 5296. [Google Scholar] [CrossRef]
- Minter, M.; Augustin, H.; van Odijk, J.; Vanfleteren, L.E.G.W. Gender differences in vitamin D status and determinants of vitamin D insufficiency in patients with chronic obstructive pulmonary disease. Nutrients 2023, 15, 426. [Google Scholar] [CrossRef]
- Hung, K.C.; Wu, J.Y.; Illias, A.M.; Chiu, C.C.; Chang, Y.J.; Liao, S.W.; Wang, K.F.; Chen, I.W.; Sun, C.K. Association of a low vitamin D status with risk of post-stroke depression: A meta-analysis and systematic review. Front. Nutr. 2023, 10, 1142035. [Google Scholar] [CrossRef]
- Wu, Z.; Hu, H.; Wang, C.; Rao, J.; Wu, J.; Shi, Y.; Fu, Y.; Cheng, X.; Li, P. Sleep patterns modify the association between vitamin D status and coronary heart disease: Results from NHANES 2005–2008. J. Nutr. 2023, 153, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Hagău, A.C.; Pușcaș, A.; Togănel, R.; Muntean, I. Is hypovitaminosis D a risk factor for heart failure? Life 2023, 13, 372. [Google Scholar] [CrossRef] [PubMed]
- Rashidmayvan, M.; Sahebi, R.; Avan, A.; Sharifan, P.; Esmaily, H.; Afshari, A.; Nattagh-Eshtivani, E.; Najar Sedghdoust, F.; Aghasizadeh, M.; Ferns, G.A.; et al. Double blind control trial of vitamin D fortified milk on the expression of lncRNAs and adiponectin for patients with metabolic syndrome. Diabetol. Metab. Syndr. 2023, 15, 9. [Google Scholar] [CrossRef] [PubMed]
- Koda, S.; Wada, K.; Yamakawa, M.; Nakashima, Y.; Hayashi, M.; Takeda, N.; Yasuda, K.; Nagata, C. Associations of plasma 25-hydroxy vitamin D and dietary vitamin D intake with insulin resistance in healthy Japanese women. J. Nutr. Sci. Vitaminol. 2023, 69, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, X.; Yang, Y.; Zhao, J.; Yu, Y. Correlation analysis of serum vitamin D levels and postoperative cognitive disorder in elderly patients with gastrointestinal tumor. Front. Psychiatry 2022, 13, 893309. [Google Scholar] [CrossRef]
- Ghahremani, M.; Smith, E.E.; Chen, H.Y.; Creese, B.; Goodarzi, Z.; Ismail, Z. Vitamin D supplementation and incident dementia: Effects of sex, APOE, and baseline cognitive status. Alzheimer’s Dement. 2023, 15, e12404. [Google Scholar] [CrossRef]
- Chen, W.Y.; Huang, M.C.; Chiu, C.C.; Cheng, Y.C.; Kuo, C.J.; Chen, P.Y.; Kuo, P.H. The interactions between vitamin D and neurofilament light chain levels on cognitive domains in bipolar disorder. BJPsych Open 2022, 8, e207. [Google Scholar] [CrossRef]
- Luo, C.W.; Chen, S.P.; Chiang, C.Y.; Wu, W.J.; Chen, C.J.; Chen, W.Y.; Kuan, Y.H. Association between ultraviolet B exposure levels and depression in Taiwanese adults: A nested case-control study. Int. J. Environ. Res. Public Health 2022, 19, 6846. [Google Scholar] [CrossRef]
- Seiler, N.; Tsiglopoulos, J.; Keem, M.; Das, S.; Waterdrinker, A. Prevalence of vitamin D deficiency among psychiatric inpatients: A systematic review. Int. J. Psychiatry Clin. Pract. 2022, 26, 330–336. [Google Scholar] [CrossRef]
- Zech, L.D.; Scherf-Clavel, M.; Daniels, C.; Schwab, M.; Deckert, J.; Unterecker, S.; Herr, A.S. Patients with higher vitamin D levels show stronger improvement of self-reported depressive symptoms in psychogeriatric day-care setting. J. Neural Transm. 2021, 128, 1233–1238. [Google Scholar] [CrossRef]
- Van Helmond, N.; Brobyn, T.L.; LaRiccia, P.J.; Cafaro, T.; Hunter, K.; Roy, S.; Bandomer, B.; Ng, K.Q.; Goldstein, H.; Mitrev, L.V.; et al. Vitamin D3 supplementation at 5000 IU daily for the prevention of influenza-like illness in healthcare workers: A pragmatic randomized clinical trial. Nutrients 2022, 15, 180. [Google Scholar] [CrossRef] [PubMed]
- Hakola, L.; Oikarinen, M.; Niinistö, S.; Cuthbertson, D.; Lehtonen, J.; Puustinen, L.; Sioofy-Khojine, A.B.; Honkanen, J.; Knip, M.; Krischer, J.P.; et al. Serum 25-hydroxyvitamin D and fatty acids in relation to the risk of microbial infections in children: The TRIGR Divia study. Clin. Nutr. 2022, 41, 2729–2739. [Google Scholar] [CrossRef] [PubMed]
- Topan, A.; Lupse, M.; Calin, M.; Jianu, C.; Leucuta, D.C.; Briciu, V. 25 Hydroxyvitamin D serum concentration and COVID-19 severity and outcome-a retrospective survey in a Romanian hospital. Nutrients 2023, 15, 1227. [Google Scholar] [CrossRef] [PubMed]
- Vásquez-Procopio, J.; Torres-Torres, J.; Borboa-Olivares, H.; Sosa, S.E.Y.; Martínez-Portilla, R.J.; Solis-Paredes, M.; Tolentino-Dolores, M.C.; Helguera-Repetto, A.C.; Marrufo-Gallegos, K.C.; Missirlis, F.; et al. Association between 25-OH vitamin D deficiency and COVID-19 severity in pregnant women. Int. J. Mol. Sci. 2022, 23, 15188. [Google Scholar] [CrossRef] [PubMed]
- Merzon, E.; Tworowski, D.; Gorohovski, A.; Vinker, S.; Golan Cohen, A.; Green, I.; Frenkel-Morgenstern, M. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: An Israeli population-based study. FEBS J. 2020, 287, 3693–3702. [Google Scholar] [CrossRef]
- Wepner, F.; Scheuer, R.; Schuetz-Wieser, B.; Machacek, P.; Pieler-Bruha, E.; Cross, H.S.; Hahne, J.; Friedrich, M. Effects of vitamin D on patients with fibromyalgia syndrome: A randomized placebo-controlled trial. Pain 2014, 155, 261–268. [Google Scholar] [CrossRef]
- Shaw, A.C.; Goldstein, D.R.; Montgomery, R.R. Age-dependent dysregulation of innate immunity. Nat. Rev. Immunol. 2013, 13, 875–887. [Google Scholar] [CrossRef]
- Zhou, A.; Hyppönen, E. Vitamin D deficiency and C-reactive protein: A bidirectional Mendelian randomization study. Int. J. Epidemiol. 2023, 52, 260–271. [Google Scholar] [CrossRef]
- Zong, W.; Patel, A.; Chang, V.; Mitchel, E.B.; Stoner, N.; Baldassano, R.N.; Albenberg, L. Clinical and lifestyle patterns in Asian children with inflammatory bowel disease in the U.S. PLoS ONE 2023, 18, e0281949. [Google Scholar] [CrossRef]
- Yamade, I.; Inoue, T.; Hamada, H.; Sudou, S.; Otsubo, M.; Sawada, M.; Nakayama, T.; Hatayama, H. Ineffectiveness of antenatal guidance intervention for vitamin D insufficiency and deficiency in pregnant women in Kyoto, Japan. J. Obstet. Gynaecol. Res. 2021, 47, 3540–3550. [Google Scholar] [CrossRef]
- Miyamoto, H.; Kawakami, D.; Hanafusa, N.; Nakanishi, T.; Miyasaka, M.; Furutani, Y.; Ikeda, Y.; Ito, K.; Kato, T.; Yokoyama, K.; et al. Determination of a serum 25-hydroxyvitamin D reference ranges in Japanese adults using fully automated liquid chromatography-tandem mass spectrometry. J. Nutr. 2023, 153, 1253–1264. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, M.; Hirai, C.; Nakajima, H. The solar exposure time required for vitamin D3 synthesis in the human body estimated by numerical simulation and observation in Japan. J. Nutr. Sci. Vitaminol. 2013, 59, 257–263. [Google Scholar] [CrossRef]
- MacLaughlin, J.; Holick, M.F. Aging decreases the capacity of human skin to produce vitamin D3. J. Clin. Investig. 1985, 76, 1536–1538. [Google Scholar] [CrossRef] [PubMed]
- Neale, R.E.; Lucas, R.M.; Byrne, S.N.; Hollestein, L.; Rhodes, L.E.; Yazar, S.; Young, A.R.; Berwick, M.; Ireland, R.A.; Olsen, C.M. The effects of exposure to solar radiation on human health. Photochem. Photobiol. Sci. 2023, 22, 1011–1047. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.M.; Horst, R.L.; Koszewski, N.J.; Simon, R.R. Vitamin D4 in mushrooms. PLoS ONE 2012, 7, e40702. [Google Scholar] [CrossRef]
- Prema, T.P.; Raghuramulu, N. Vitamin D3 and its metabolites in the tomato plant. Phytochemistry 1996, 42, 617–620. [Google Scholar] [CrossRef]
- Aburjaia, T.; Al-Khalil, S.; Abuirjeie, M. Vitamin D3 and its metabolites in tomato, potato, egg plant and zucchini leaves. Phytochemistry 1998, 49, 2497–2499. [Google Scholar] [CrossRef]
- Komba, S.; Kotake-Nara, E.; Tsuzuki, W. Simultaneous synthesis of vitamins D2, D4, D5, D6, and D7 from commercially available phytosterol, β-sitosterol, and identification of each vitamin D by HSQC NMR. Metabolites 2019, 9, 107. [Google Scholar] [CrossRef]
- Kotake-Nara, E.; Komba, S.; Hase, M. Uptake of vitamins D2, D3, D4, D5, D6, and D7 solubilized in mixed micelles by human intestinal cells, Caco-2, an enhancing effect of lysophosphatidylcholine on the cellular uptake, and estimation of vitamins D’ biological activities. Nutrients 2021, 13, 1126. [Google Scholar] [CrossRef]
- Windaus, A.; Trautmann, G. Über das krystallisierte vitamin D4. Hoppe Seylers Z. Physiol. Chem. 1937, 247, 185–188. [Google Scholar] [CrossRef]
- Wunderlich, W. Über das 7-Dehydro-sitosterin. Hoppe Seylers Z. Physiol. Chem. 1936, 241, 116–124. [Google Scholar] [CrossRef]
- Linsert, O. Über das 7-Dehydro-stigmasterin. Hoppe Seylers Z. Physiol. Chem. 1936, 241, 125–128. [Google Scholar] [CrossRef]
- Ruigh, W.L. 7-Dehydrocampesterol, a new provitamin D. J. Am. Chem. Soc. 1942, 64, 1900–1902. [Google Scholar] [CrossRef]
- Wohl, A. Bromierung ungesättigter Verbindungen mit N-Brom-acetamid, ein Beitrag zur Lehre vom Verlauf chemischer Vorgänge. Eur. J. Inorg. Chem. 1919, 52, 51–63. [Google Scholar] [CrossRef]
- Ziegler, K.; Schenck, G.; Krockow, E.W.; Siebert, A.; Wenz, A.; Weber, H. Die Synthese des Cantharidins. European J. Org. Chem. 1942, 551, 1–79. [Google Scholar] [CrossRef]
- Djerassi, C. Brominations with N-bromosuccinimide and related compounds; The Wohl-Ziegler reaction. Chem. Rev. 1948, 43, 271–317. [Google Scholar] [CrossRef]
- Walter, W.R.; Deutsch, H.R.; Bowman, M.I.; Joffe, I.B. New methods of preparation of 2-methy cyclohexen-1-one1. J. Am. Chem. Soc. 1950, 72, 5759–5760. [Google Scholar]
- Yoshiyama-Yanagawa, T.; Enya, S.; Shimada-Niwa, Y.; Yaguchi, S.; Haramoto, Y.; Matsuya, T.; Shiomi, K.; Sasakura, Y.; Takahashi, S.; Asashima, M.; et al. The conserved Rieske oxygenase DAF-36/Neverland is a novel cholesterol-metabolizing enzyme. J. Biol. Chem. 2011, 286, 25756–25762. [Google Scholar] [CrossRef]
- Kotake-Nara, E.; Nagao, A. Absorption and metabolism of xanthophylls. Mar. Drugs 2011, 9, 1024–1037. [Google Scholar] [CrossRef]
- Compassi, S.; Werder, M.; Weber, F.E.; Boffelli, D.; Hauser, H.; Schulthess, G. Comparison of cholesterol and sitosterol uptake in different brush border membrane models. Biochemistry 1997, 36, 6643–6652. [Google Scholar] [CrossRef]
- Kotake-Nara, E.; Yonekura, L.; Nagao, A. Lysoglyceroglycolipids improve the intestinal absorption of micellar fucoxanthin by Caco-2 cells. J. Oleo Sci. 2015, 64, 1207–1211. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.D. Clinical counterpoint: Vitamin D: New actions, new analogs, new therapeutic potential. Endocr. Rev. 1992, 13, 765–784. [Google Scholar]
- Reichrath, J.; Perez, A.; Müller, S.M.; Chen, T.C.; Kerber, A.; Bahmer, F.A.; Holick, M.F. Topical calcitriol (1,25-dihydroxyvitamin D3) treatment of psoriasis: An immunohistological evaluation. Acta Derm. Venereol. 1997, 77, 268–272. [Google Scholar] [CrossRef]
- Egan, K.M.; Sosman, J.A.; Blot, W.J. Sunlight and reduced risk of cancer: Is the real story vitamin D? J. Natl. Cancer Inst. 2005, 97, 161–163. [Google Scholar] [CrossRef]
- Giovannucci, E.; Liu, Y.; Rimm, E.B.; Hollis, B.W.; Fuchs, C.S.; Stampfer, M.J.; Willett, W.C. Prospective study of predictors of vitamin D status and cancer incidence and mortality in men. J. Natl. Cancer Inst. 2006, 98, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B. Vitamin D status: Ready for guiding prostate cancer diagnosis and treatment? Clin. Cancer Res. 2014, 20, 2241–2243. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schwartz, G.G.; Hulka, B.S. Is vitamin D deficiency a risk factor for prostate cancer? (Hypothesis). Anticancer Res. 1990, 10, 1307–1311. [Google Scholar] [PubMed]
- Schwartz, G.G. Vitamin D and intervention trials in prostate cancer: From theory to therapy. Ann. Epidemiol. 2009, 19, 96–102. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 1980, 9, 227–231. [Google Scholar] [CrossRef]
- Garland, C.F.; Garland, F.C. Do sunlight and vitamin D reduce the likelihood of colon cancer? Int. J. Epidemiol. 2006, 35, 217–220. [Google Scholar] [CrossRef]
- Miyaura, C.; Abe, E.; Kuribayashi, T.; Tanaka, H.; Konno, K.; Nishii, Y.; Suda, T. 1 Alpha,25-dihydroxyvitamin D3 induces differentiation of human myeloid leukemia cells. Biochem. Biophys. Res. Commun. 1981, 102, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Abe, E.; Miyaura, C.; Kuribayashi, T.; Konno, K.; Nishii, Y.; Suda, T. 1 Alpha,25-Dihydroxycholecalciferol and a human myeloid leukaemia cell line (HL-60). Biochem. J. 1982, 204, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa, N.; Nakagawa, K.; Kawamoto, Y.; Tachibana, Y.; Hayashi, T.; Ozono, K.; Okano, T. Biological activity profiles of 1alpha,25-dihydroxyvitamin D2, D3, D4, D7, and 24-epi-1alpha,25-dihydroxyvitamin D2. Biol. Pharm. Bull. 1999, 22, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Gross, C.; Stamey, T.; Hancock, S.; Feldman, D. Treatment of early recurrent prostate cancer with 1,25-dihydroxyvitamin D3 (calcitriol). J. Urol. 1998, 159, 2035–2039. [Google Scholar] [CrossRef]
- Smith, D.C.; Johnson, C.S.; Freeman, C.C.; Muindi, J.; Wilson, J.W.; Trump, D.L. A phase I trial of calcitriol (1,25-dihydroxycholecalciferol) in patients with advanced malignancy. Clin. Cancer Res. 1999, 5, 1339–1345. [Google Scholar]
- Lazzaro, G.; Agadir, A.; Qing, W.; Poria, M.; Mehta, R.R.; Moriarty, R.M.; Das Gupta, T.K.; Zhang, X.K.; Mehta, R.G. Induction of differentiation by 1alpha-hydroxyvitamin D(5) in T47D human breast cancer cells and its interaction with vitamin D receptors. Eur. J. Cancer 2000, 36, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.G.; Moriarty, R.M.; Mehta, R.R.; Penmasta, R.; Lazzaro, G.; Constantinou, A.; Guo, L. Prevention of preneoplastic mammary lesion development by a novel vitamin D analogue, 1alpha-hydroxyvitamin D5. J. Natl. Cancer Inst. 1997, 89, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.; Hawthorne, M.; Uselding, L.; Albinescu, D.; Moriarty, R.; Christov, K.; Mehta, R. Prevention of N-methyl-N-nitrosourea-induced mammary carcinogenesis in rats by 1-hydroxyvitamin D(5). J. Natl. Cancer Inst. 2000, 92, 1836–1840. [Google Scholar] [CrossRef]
- Mehta, R.R.; Bratescu, L.; Graves, J.M.; Green, A.; Mehta, R.G. Differentiation of human breast carcinoma cells by a novel vitamin D analog: 1alpha-hydroxyvitamin D5. Int. J. Oncol. 2000, 16, 65–73. [Google Scholar] [CrossRef]
- Mehta, R.G.; Hussain, E.A.; Mehta, R.R.; Das Gupta, T.K. Chemoprevention of mammary carcinogenesis by 1alpha-hydroxyvitamin D5, a synthetic analog of vitamin D. Mutat. Res. 2003, 523–524, 253–264. [Google Scholar] [CrossRef]
- Bohannon, B.M.; Jowais, J.J.; Nyberg, L.; Olivier-Meo, V.; Corradi, V.; Tieleman, D.P.; Liin, S.I.; Larsson, H.P. Mechanistic insights into robust cardiac IKs potassium channel activation by aromatic polyunsaturated fatty acid analogues. eLife 2023, 12, e85773. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.T.; Quayle, J.M. Physiological roles and properties of potassium channels in arterial smooth muscle. Am. J. Physiol. 1995, 268, C799–C822. [Google Scholar] [CrossRef]
- Koh, S.D.; Ward, S.M.; Dick, G.M.; Epperson, A.; Bonner, H.P.; Sanders, K.M.; Horowitz, B.; Kenyon, J.L. Contribution of delayed rectifier potassium currents to the electrical activity of murine colonic smooth muscle. J. Physiol. 1999, 515, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Casanova, V.; Sousa, F.H.; Stevens, C.; Barlow, P.G. Antiviral therapeutic approaches for human rhinovirus infections. Future Virol. 2018, 13, 505–518. [Google Scholar] [CrossRef] [PubMed]
- Schögler, A.; Muster, R.J.; Kieninger, E.; Casaulta, C.; Tapparel, C.; Jung, A.; Moeller, A.; Geiser, T.; Regamey, N.; Alves, M.P. Vitamin D represses rhinovirus replication in cystic fibrosis cells by inducing LL-37. Eur. Respir. J. 2016, 47, 520–530. [Google Scholar] [CrossRef]
- Telcian, A.G.; Zdrenghea, M.T.; Edwards, M.R.; Laza-Stanca, V.; Mallia, P.; Johnston, S.L.; Stanciu, L.A. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antiviral Res. 2017, 137, 93–101. [Google Scholar] [CrossRef]
- Mostafa, B.E.-D.; Taha, M.S.; Abdel Hamid, T.; Omran, A.; Lotfi, N. Evaluation of vitamin D levels in allergic fungal sinusitis, chronic rhinosinusitis, and chronic rhinosinusitis with polyposis. Int. Forum Allergy Rhinol. 2016, 6, 185–190. [Google Scholar] [CrossRef]
- Lim, J.H.; Ravikumar, S.; Wang, Y.M.; Thamboo, T.P.; Ong, L.; Chen, J.; Goh, J.G.; Tay, S.H.; Chengchen, L.; Win, M.S.; et al. Bimodal influence of vitamin D in host response to systemic candida infection-Vitamin D dose matters. J. Infect. Dis. 2015, 212, 635–644. [Google Scholar] [CrossRef]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef]
- Goldberg, P. Multiple sclerosis: Vitamin D and calcium as environmental determinants of prevalence. (A viewpoint) part 1: Sunlight, dietary factors and epidemiology. Int. J. Environ. Stud. 1974, 6, 19–27. [Google Scholar] [CrossRef]
- Munger, K.L.; Levin, L.I.; Hollis, B.W.; Howard, N.S.; Ascherio, A. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA 2006, 296, 2832–2838. [Google Scholar] [CrossRef] [PubMed]
- Afzal, S.; Bojesen, S.E.; Nordestgaard, B.G. Reduced 25-hydroxyvitamin D and risk of Alzheimer’s disease and vascular dementia. Alzheimer’s Dement. 2014, 10, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Mizwicki, M.T.; Menegaz, D.; Zhang, J.; Barrientos-Durán, A.; Tse, S.; Cashman, J.R.; Griffin, P.R.; Fiala, M. Genomic and nongenomic signaling induced by 1α,25(OH)2-vitamin D3 promotes the recovery of amyloid-β phagocytosis by Alzheimer’s disease macrophages. J. Alzheimer’s Dis. 2012, 29, 51–62. [Google Scholar] [CrossRef]
- Feng, L.; Cheah, I.K.; Ng, M.M.; Li, J.; Chan, S.M.; Lim, S.L.; Mahendran, R.; Kua, E.H.; Halliwell, B. The association between mushroom consumption and mild cognitive impairment: A community-based cross-sectional study in Singapore. J. Alzheimer’s Dis. 2019, 68, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Soltesz, G.; Patterson, C.C.; Dahlquist, G. Worldwide childhood type 1 diabetes incidence—What can we learn from epidemiology? Pediatr. Diabetes 2007, 8, 6–14. [Google Scholar] [CrossRef]
- Munger, K.L.; Levin, L.I.; Massa, J.; Horst, R.; Orban, T.; Ascherio, A. Preclinical serum 25-hydroxyvitamin D levels and risk of type 1 diabetes in a cohort of US military personnel. Am. J. Epidemiol. 2013, 177, 411–419. [Google Scholar] [CrossRef]
- Deng, X.; Song, Y.; Manson, J.E.; Signorello, L.B.; Zhang, S.M.; Shrubsole, M.J.; Ness, R.M.; Seidner, D.L.; Dai, Q. Magnesium, vitamin D status and mortality: Results from US National Health and Nutrition Examination Survey (NHANES) 2001 to 2006 and NHANES III. BMC Med. 2013, 11, 187. [Google Scholar] [CrossRef]
- Omura, T.; Sato, R. A new cytochrome in liver microsomes. J. Biol. Chem. 1962, 237, 1375–1376. [Google Scholar] [CrossRef]
- Thomas, R.L.; Jiang, L.; Adams, J.S.; Xu, Z.Z.; Shen, J.; Janssen, S.; Ackermann, G.; Vanderschueren, D.; Pauwels, S.; Knight, R.; et al. Vitamin D metabolites and the gut microbiome in older men. Nat. Commun. 2020, 11, 5997. [Google Scholar] [CrossRef]
- Silvestro, D.; Villette, C.; Delecolle, J.; Olsen, C.E.; Motawia, M.S.; Geoffroy, P.; Miesch, M.; Jensen, P.E.; Heintz, D.; Schaller, H. Vitamin D 5 in Arabidopsis thaliana. Sci. Rep. 2018, 8, 16348. [Google Scholar] [CrossRef]
- Seckbach, J.; Ikan, R. Sterols and chloroplast structure of Cyanidium caldarium. Plant Physiol. 1972, 49, 457–459. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Karmakar, T.; Chakraborty, D.P. 7-Dehydrositosterol from Rauwolfia serpentina. Phytochemistry 1983, 22, 608–609. [Google Scholar] [CrossRef]
- Abuzenadah, A.M.; Al-Sayes, F.; Mahafujul Alam, S.S.; Hoque, M.; Karim, S.; Hussain, I.M.R.; Tabrez, S. Identification of potential poly (ADP-ribose) polymerase-1 inhibitors derived from Rauwolfia serpentina: Possible implication in cancer therapy. Evid. Based Complement. Alternat. Med. 2022, 2022, 3787162. [Google Scholar] [CrossRef] [PubMed]
- Korn, E.D.; Von Brand, T.; Tobie, E.J. The sterols of Trypanosoma cruzi and Crithidia fasciculata. Comp. Biochem. Physiol. 1969, 30, 601–610. [Google Scholar] [CrossRef]
- Smith, F.R.; Korn, E.D. 7-Dehydrostigmasterol and ergosterol: The major sterols of an amoeba. J. Lipid Res. 1968, 9, 405–408. [Google Scholar] [CrossRef]
- De Rosa, S.; Milone, A.; Popov, S. Sterol composition of the sponge Fasciospongia cavernosa, from the Adriatic, Aegean and Tyrrhenian seas. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1999, 123, 235–239. [Google Scholar] [CrossRef]
- Sica, D.; Piccialli, V.; Pronzato, R. Δ5,7Sterols from the sponges Ircinia pipetta and Dysidea avara identification of cholesta-5,7,24-trien-3B-OL. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1987, 88, 293–296. [Google Scholar] [CrossRef]
- Dini, A.; Sica, D.; Boniforti, L. Two new Δ5,7-sterols from two spongiidae sponges. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1984, 78, 741–744. [Google Scholar] [CrossRef]
- Teshima, S.; Kanazawa, A.; Yamada, I.; Tanaka, Y. Sterols of the spongeTethya amamensis: Occurrence of (24E)-24-ethylidenecholesta-5,7-dienol, (24E)-24-propylidenecholesta-5,7-dienol, and (24Z)-24-propylidenecholesta-5,7-dienol. Lipids 1983, 18, 193–197. [Google Scholar] [CrossRef]
- Popplestone, C.R.; Unrau, A.M. Major sterols of Achlya bisexualis. Phytochemistry 1973, 12, 1131–1133. [Google Scholar] [CrossRef]
- Nes, W.R.; Malya, P.A.; Mallory, F.B.; Ferguson, K.A.; Landrey, J.R.; Conner, R.L. Conformational analysis of enzyme-substrate complex in the dehydrogenation of sterols by Tetrahymena pyriformis. J. Biol. Chem. 1971, 246, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Yonekura, L.; Tsuzuki, W.; Nagao, A. Acyl moieties modulate the effects of phospholipids on beta-carotene uptake by Caco-2 cells. Lipids 2006, 41, 629–636. [Google Scholar] [CrossRef]
- Reboul, E.; Goncalves, A.; Comera, C.; Bott, R.; Nowicki, M.; Landrier, J.F.; Jourdheuil-Rahmani, D.; Dufour, C.; Collet, X.; Borel, P. Vitamin D intestinal absorption is not a simple passive diffusion: Evidences for involvement of cholesterol transporters. Mol. Nutr. Food Res. 2011, 55, 691–702. [Google Scholar] [CrossRef] [PubMed]
- Lagunin, A.; Stepanchikova, A.; Filimonov, D.; Poroikov, V. PASS: Prediction of activity spectra for biologically active substances. Bioinformatics 2000, 16, 747–748. [Google Scholar] [CrossRef] [PubMed]


| Vitamin D2 | Vitamin D3 | Vitamin D4/D7 * | Vitamin D5 | Vitamin D6 | Compound 3 ** | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Activity | Pa 1 | Pi 2 | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi |
| Anti-osteoporotic | 0.982 a | 0.003 | 0.973 b | 0.003 | 0.969 | 0.003 | 0.965 | 0.003 | 0.970 | 0.003 | 0.940 | 0.003 |
| Bone diseases treatment | 0.981 a | 0.003 | 0.976 b | 0.003 | 0.968 | 0.003 | 0.966 | 0.003 | 0.966 | 0.003 | 0.938 | 0.003 |
| Vitamin | 0.977 b | 0.000 | 0.975 | 0.000 | 0.936 | 0.000 | 0.978 a | 0.000 | 0.976 | 0.000 | 0.907 | 0.000 |
| Hyperparathyroidism treatment | 0.946 a | 0.000 | 0.933 b | 0.000 | 0.883 | 0.000 | 0.882 | 0.000 | 0.901 | 0.000 | 0.846 | 0.000 |
| Calcium regulator | 0.902 a | 0.001 | 0.876 | 0.002 | 0.869 | 0.002 | 0.870 | 0.002 | 0.880 b | 0.001 | 0.821 | 0.002 |
| Vitamin D-like | 0.869 a | 0.000 | 0.757 | 0.000 | 0.578 | 0.000 | 0.791 | 0.000 | 0.844 b | 0.000 | 0.570 | 0.000 |
| Vitamin D receptor agonist | 0.816 a | 0.000 | 0.693 | 0.000 | 0.680 | 0.000 | 0.686 | 0.000 | 0.738 b | 0.000 | 0.542 | 0.000 |
| Vitamin D2 | Vitamin D3 | Vitamin D4/D7 | Vitamin D5 | Vitamin D6 | Compound 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Activity | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi |
| Antipsoriatic | 0.987 b | 0.002 | 0.976 | 0.002 | 0.959 | 0.002 | 0.962 | 0.002 | 0.992 a | 0.001 | 0.938 | 0.002 |
| Dermatologic | 0.985 b | 0.002 | 0.975 | 0.003 | 0.963 | 0.003 | 0.966 | 0.003 | 0.988 a | 0.002 | 0.950 | 0.003 |
| Anti-eczematic | 0.948 | 0.003 | 0.953 a | 0.002 | 0.947 | 0.003 | 0.948 | 0.003 | 0.946 | 0.003 | 0.949 b | 0.003 |
| Antipruritic | 0.800 | 0.004 | 0.834 a | 0.002 | 0.824 | 0.003 | 0.825 b | 0.003 | 0.791 | 0.004 | 0.821 | 0.003 |
| Antineoplastic | 0.925 b | 0.005 | 0.886 | 0.005 | 0.861 | 0.006 | 0.873 | 0.005 | 0.936 a | 0.004 | 0.909 | 0.005 |
| Adenomatous polyposis treatment | 0.840 | 0.002 | 0.894 | 0.001 | 0.905 b | 0.001 | 0.916 a | 0.001 | 0.852 | 0.002 | 0.877 | 0.001 |
| Apoptosis agonist | 0.782 a | 0.009 | 0.742 | 0.011 | 0.642 | 0.021 | 0.665 | 0.019 | 0.765 b | 0.010 | 0.718 | 0.013 |
| Antileukemic | 0.609 a | 0.008 | 0.599 | 0.009 | 0.591 | 0.009 | 0.601 b | 0.009 | 0.558 | 0.010 | 0.597 | 0.009 |
| Chemopreventive | 0.861 | 0.003 | 0.873 | 0.003 | 0.844 | 0.003 | 0.874 b | 0.003 | 0.868 | 0.003 | 0.917 a | 0.002 |
| Vitamin D2 | Vitamin D3 | Vitamin D4/D7 | Vitamin D5 | Vitamin D6 | Compound 3 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Activity | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi | Pa | Pi |
| Respiratory analeptic | 0.828 | 0.007 | 0.962 a | 0.003 | 0.931 | 0.004 | 0.961 b | 0.003 | 0.843 | 0.006 | 0.940 | 0.004 |
| Analeptic | 0.714 | 0.009 | 0.924 a | 0.003 | 0.840 | 0.004 | 0.877 b | 0.004 | 0.738 | 0.008 | 0.853 | 0.004 |
| Anti-inflammatory | 0.700 | 0.016 | 0.747 a | 0.010 | 0.660 | 0.021 | 0.699 | 0.016 | 0.688 | 0.017 | 0.736 b | 0.012 |
| Antidiabetic (type 1) | 0.598 | 0.002 | 0.824 a | 0.001 | 0.630 | 0.002 | 0.635 b | 0.002 | 0.566 | 0.002 | 0.592 | 0.002 |
| Polarization stimulant | - * | - | - | - | - | - | - | - | - | - | 0.870 a | 0.001 |
| Multiple sclerosis treatment | 0.631 a | 0.005 | - | - | 0.296 | 0.059 | 0.312 | 0.050 | 0.380 b | 0.029 | - | - |
| Anti-parkinsonian, rigidity relieving | 0.626 a | 0.004 | 0.445 | 0.013 | 0.439 | 0.013 | 0.439 | 0.013 | 0.595 b | 0.004 | 0.379 | 0.026 |
| Anti-viral (Rhinovirus) | 0.480 | 0.032 | 0.549 b | 0.012 | 0.538 | 0.014 | 0.538 | 0.014 | 0.477 | 0.034 | 0.591 a | 0.007 |
| Anti-fungal | 0.445 | 0.040 | 0.521 | 0.027 | 0.549 | 0.024 | 0.606 b | 0.018 | 0.474 | 0.035 | 0.628 a | 0.016 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komba, S.; Hase, M.; Kotake-Nara, E. Organic Synthesis of New Secosteroids from Fucosterol, Its Intestinal Absorption by Caco-2 Cells, and Simulation of the Biological Activities of Vitamin D. Mar. Drugs 2023, 21, 540. https://doi.org/10.3390/md21100540
Komba S, Hase M, Kotake-Nara E. Organic Synthesis of New Secosteroids from Fucosterol, Its Intestinal Absorption by Caco-2 Cells, and Simulation of the Biological Activities of Vitamin D. Marine Drugs. 2023; 21(10):540. https://doi.org/10.3390/md21100540
Chicago/Turabian StyleKomba, Shiro, Megumi Hase, and Eiichi Kotake-Nara. 2023. "Organic Synthesis of New Secosteroids from Fucosterol, Its Intestinal Absorption by Caco-2 Cells, and Simulation of the Biological Activities of Vitamin D" Marine Drugs 21, no. 10: 540. https://doi.org/10.3390/md21100540
APA StyleKomba, S., Hase, M., & Kotake-Nara, E. (2023). Organic Synthesis of New Secosteroids from Fucosterol, Its Intestinal Absorption by Caco-2 Cells, and Simulation of the Biological Activities of Vitamin D. Marine Drugs, 21(10), 540. https://doi.org/10.3390/md21100540








