Oral Cancer Pain Includes Thermal Allodynia That May Be Attenuated by Chronic Alcohol Consumption
Abstract
1. Introduction
2. Results
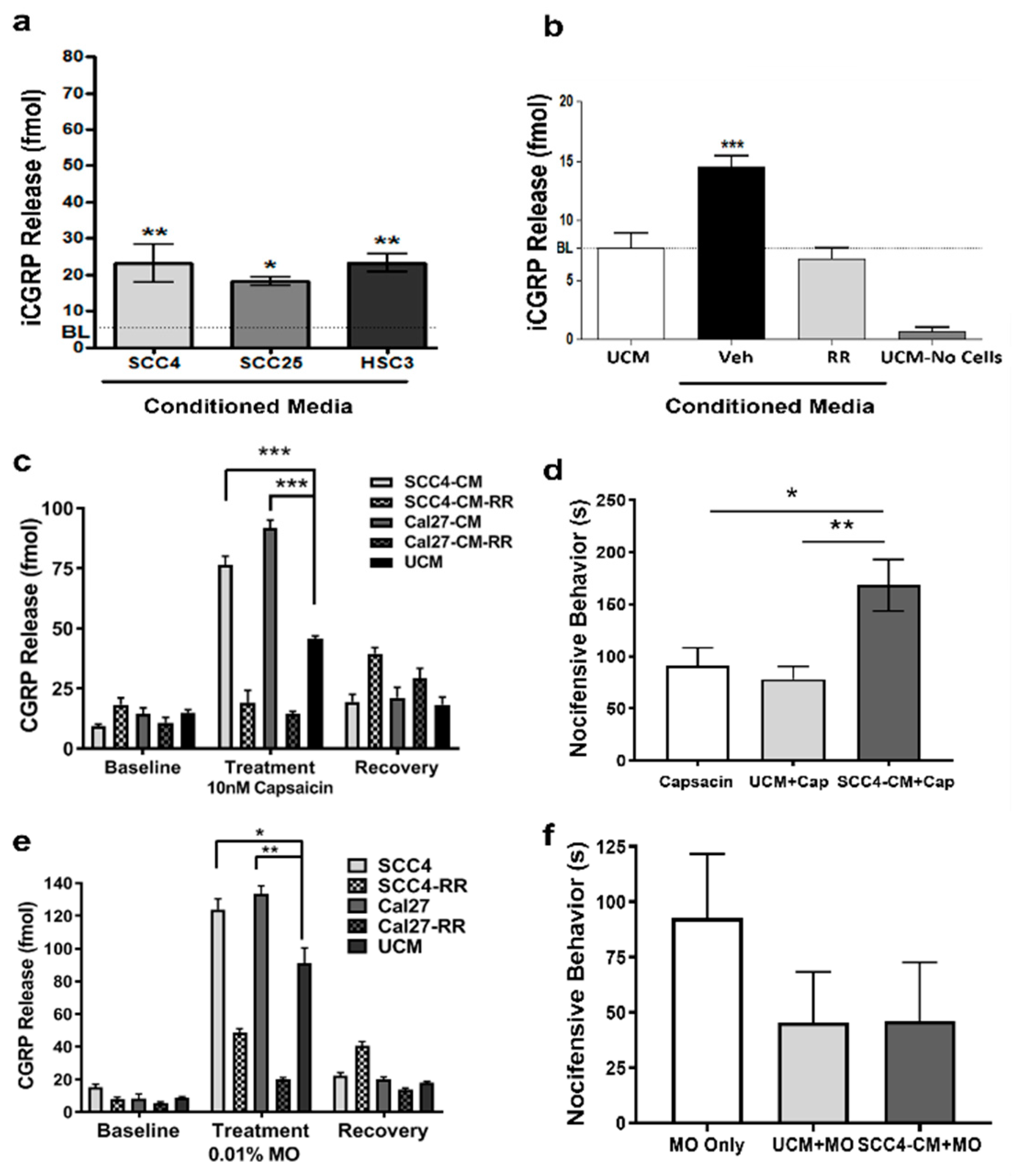
2.1. OSCC-Secreted Factors Activate TRPA1 and TRPV1 Channels
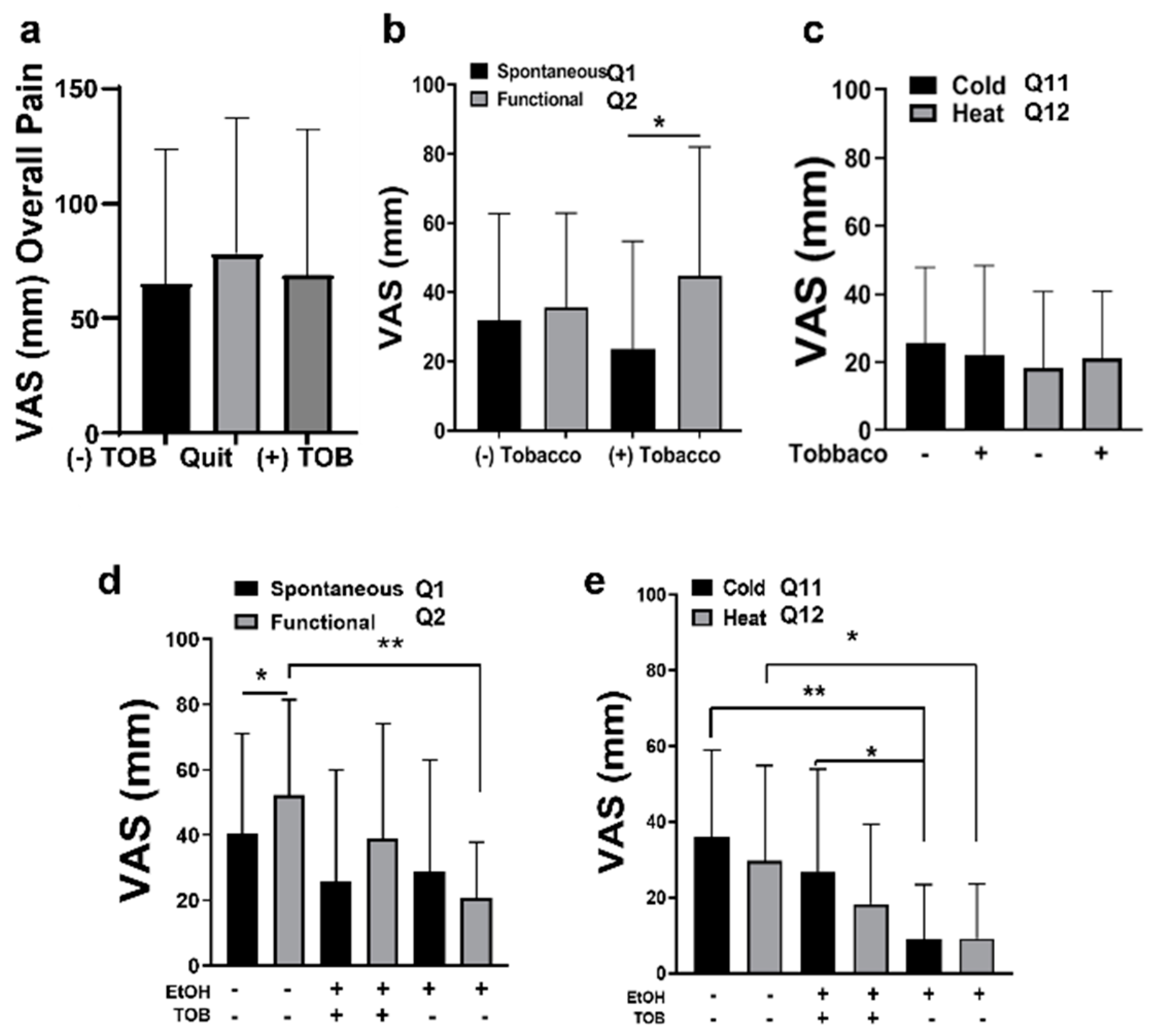
2.2. Advanced OSCC Patients Who Do Not Consume Alcohol Experience More Functional Pain
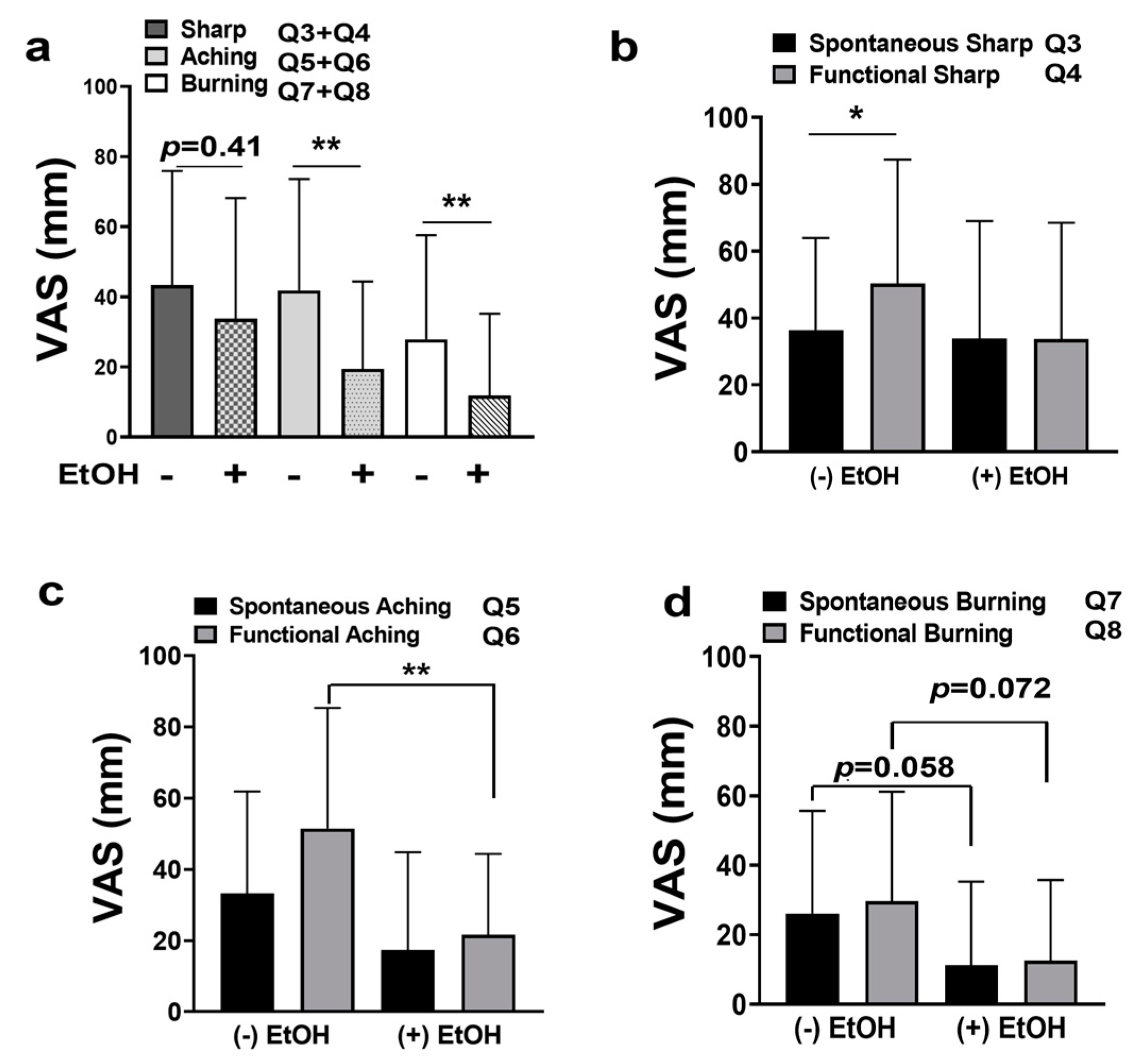
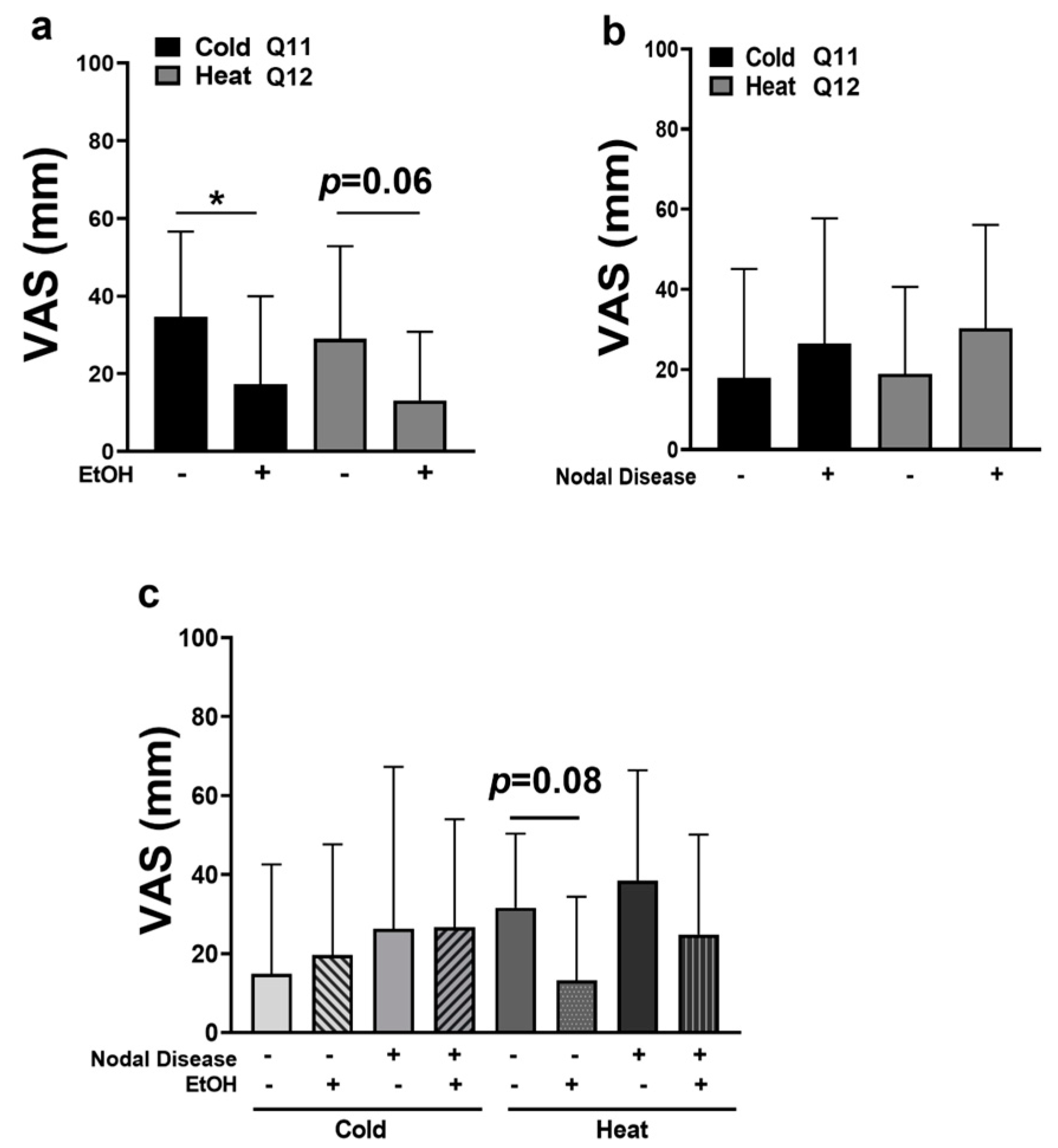
2.3. OSCC Pain Is Attenuated by Alcohol Consumption
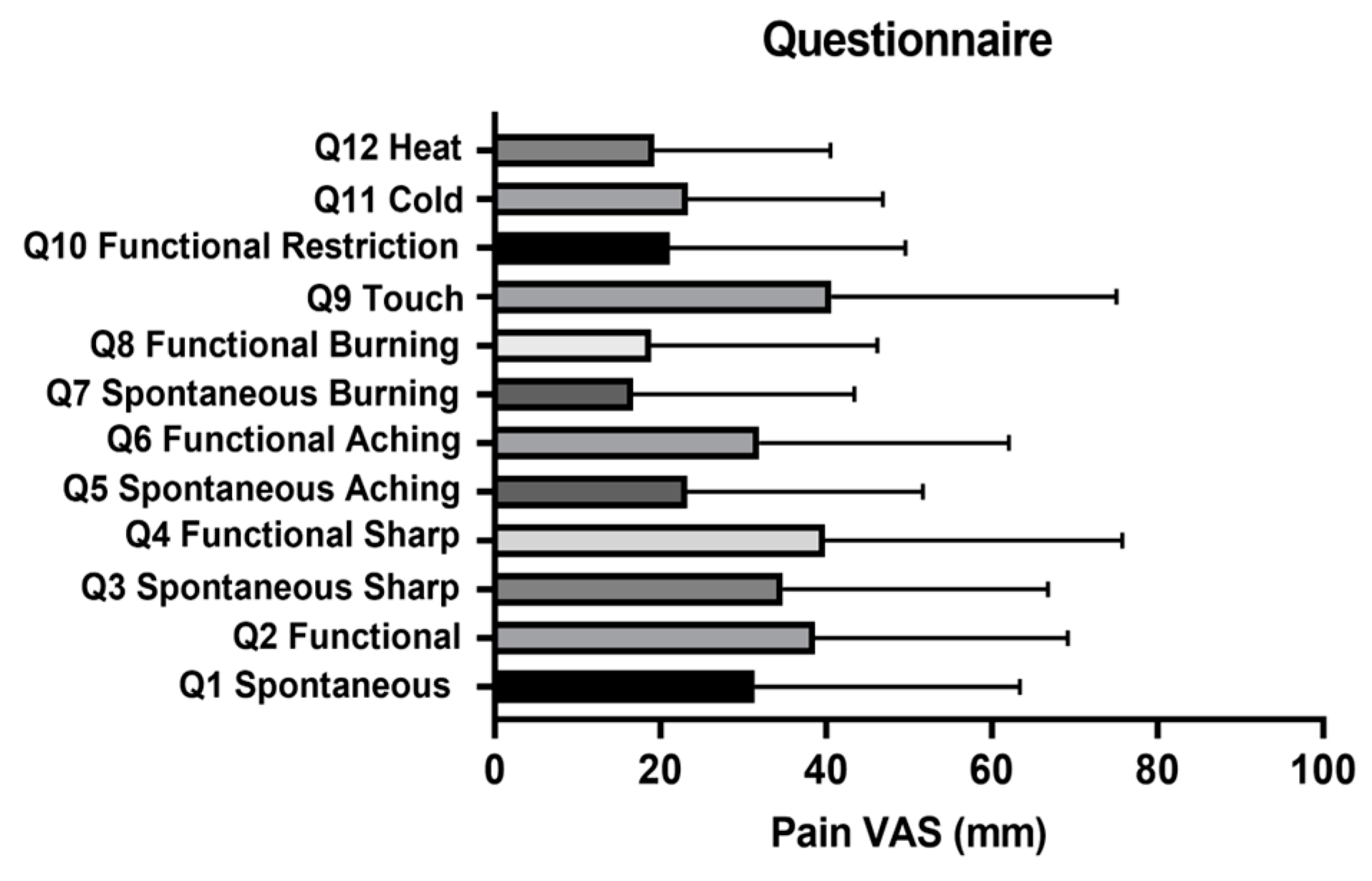
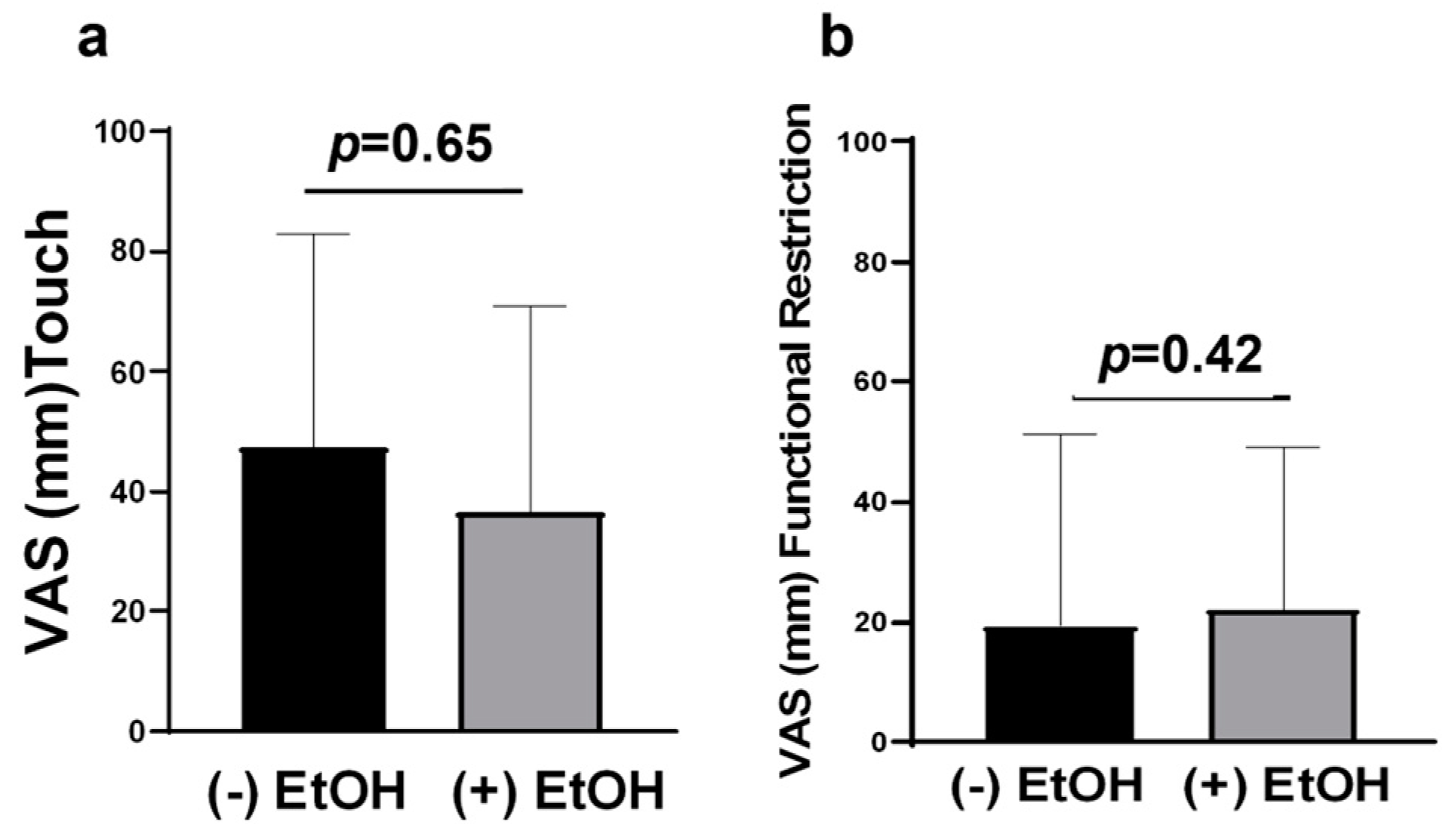
2.4. OSCC Patients Experience Thermal Allodynia
2.5. Tobacco
3. Discussion
4. Materials and Methods
4.1. Human OSCC Cell Lines
4.2. Reagents
4.3. Rat Trigeminal Ganglia (TG) Primary Culture
4.4. CGRP Release Assay and iCGRP Radioimmunoassay (RIA)
4.5. Eye-Wipe Testing
4.6. Expanded UCFS Oral Cancer Pain Questionnaire
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mercadante, S.; Casuccio, A.; Pumo, S.; Fulfaro, F. Opioid responsiveness-primary diagnosis relationship in advanced cancer patients followed at home. J. Pain Symptom Manag. 2000, 20, 27–34. [Google Scholar] [CrossRef]
- Mercadante, S.; Masedu, F.; Valenti, M.; Aielli, F. Breakthrough pain in patients with head & neck cancer. A secondary analysis of IOPS MS study. Oral Oncol. 2019, 95, 87–90. [Google Scholar] [CrossRef]
- Lam, D.K.; Schmidt, B.L. Orofacial pain onset predicts transition to head and neck cancer. Pain 2011, 152, 1206–1209. [Google Scholar] [CrossRef]
- Cuffari, L.; Tesseroli de Siqueira, J.T.; Nemr, K.; Rapaport, A. Pain complaint as the first symptom of oral cancer: A descriptive study. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2006, 102, 56–61. [Google Scholar] [CrossRef]
- Pickering, V.; Jay Gupta, R.; Quang, P.; Jordan, R.C.; Schmidt, B.L. Effect of peripheral endothelin-1 concentration on carcinoma-induced pain in mice. Eur. J. Pain 2008, 12, 293–300. [Google Scholar] [CrossRef]
- Chodroff, L.; Bendele, M.; Valenzuela, V.; Henry, M.; Ruparel, S. Express: BDNF Signaling Contributes to Oral Cancer Pain in a Preclinical Orthotopic Rodent Model. Mol. Pain 2016, 12, 1744806916666841. [Google Scholar] [CrossRef]
- Lam, D.K.; Dang, D.; Zhang, J.; Dolan, J.C.; Schmidt, B.L. Novel animal models of acute and chronic cancer pain: A pivotal role for PAR2. J. Neurosci. 2012, 32, 14178–14183. [Google Scholar] [CrossRef]
- Schmidt, B.L.; Pickering, V.; Liu, S.; Quang, P.; Dolan, J.; Connelly, S.T.; Jordan, R.C. Peripheral endothelin A receptor antagonism attenuates carcinoma-induced pain. Eur. J. Pain 2007, 11, 406–414. [Google Scholar] [CrossRef]
- Quang, P.N.; Schmidt, B.L. Peripheral endothelin B receptor agonist-induced antinociception involves endogenous opioids in mice. Pain 2010, 149, 254–262. [Google Scholar] [CrossRef]
- Constantin, C.E.; Mair, N.; Sailer, C.A.; Andratsch, M.; Xu, Z.Z.; Blumer, M.J.; Scherbakov, N.; Davis, J.B.; Bluethmann, H.; Ji, R.R.; et al. Endogenous tumor necrosis factor alpha (TNFalpha) requires TNF receptor type 2 to generate heat hyperalgesia in a mouse cancer model. J. Neurosci. 2008, 28, 5072–5081. [Google Scholar] [CrossRef]
- Tu, N.H.; Jensen, D.D.; Anderson, B.M.; Chen, E.; Jimenez-Vargas, N.N.; Scheff, N.N.; Inoue, K.; Tran, H.D.; Dolan, J.C.; Meek, T.A.; et al. Legumain Induces Oral Cancer Pain by Biased Agonism of Protease-Activated Receptor-2. J. Neurosci. 2021, 41, 193–210. [Google Scholar] [CrossRef]
- Dubeykovskaya, Z.A.; Tu, N.H.; Garcia, P.D.R.; Schmidt, B.L.; Albertson, D.G. Oral Cancer Cells Release Vesicles that Cause Pain. Adv. Biol. 2022, 6, e2200073. [Google Scholar] [CrossRef]
- Connelly, S.T.; Schmidt, B.L. Evaluation of pain in patients with oral squamous cell carcinoma. J. Pain 2004, 5, 505–510. [Google Scholar] [CrossRef]
- Kolokythas, A.; Connelly, S.T.; Schmidt, B.L. Validation of the University of California San Francisco Oral Cancer Pain Questionnaire. J. Pain 2007, 8, 950–953. [Google Scholar] [CrossRef]
- Gonzalez-Ramirez, R.; Chen, Y.; Liedtke, W.B.; Morales-Lazaro, S.L. TRP Channels and Pain. In Neurobiology of TRP Channels; Emir, T.L.R., Ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 125–147. [Google Scholar] [CrossRef]
- Koltzenburg, M.; Lundberg, L.E.R.; Torebjork, E.H. Dynamic and static components of mechanical hyperalgesia in human hairy skin. Pain 1992, 51, 207–219. [Google Scholar] [CrossRef]
- Namer, B.; Seifert, F.; Handwerker, H.O.; Maihofner, C. TRPA1 and TRPM8 activation in humans: Effects of cinnamaldehyde and menthol. Neuroreport 2005, 16, 955–959. [Google Scholar] [CrossRef]
- Reyes-Gibby, C.C.; Anderson, K.O.; Merriman, K.W.; Todd, K.H.; Shete, S.S.; Hanna, E.Y. Survival patterns in squamous cell carcinoma of the head and neck: Pain as an independent prognostic factor for survival. J. Pain 2014, 15, 1015–1022. [Google Scholar] [CrossRef]
- Mercadante, S.; Dardanoni, G.; Salvaggio, L.; Armata, M.G.; Agnello, A. Monitoring of opioid therapy in advanced cancer pain patients. J. Pain Symptom Manag. 1997, 13, 204–212. [Google Scholar] [CrossRef]
- Scheff, N.N.; Bhattacharya, A.; Dowse, E.; Dang, R.X.; Dolan, J.C.; Wang, S.; Kim, H.; Albertson, D.G.; Schmidt, B.L. Neutrophil-Mediated Endogenous Analgesia Contributes to Sex Differences in Oral Cancer Pain. Front. Integr. Neurosci. 2018, 12, 52. [Google Scholar] [CrossRef]
- Brand, G.; Jacquot, L. Sensitization and desensitization to allyl isothiocyanate (mustard oil) in the nasal cavity. Chem. Senses 2002, 27, 593–598. [Google Scholar] [CrossRef]
- Prescott, J.; Swain-Campbell, N. Responses to repeated oral irritation by capsaicin, cinnamaldehyde and ethanol in PROP tasters and non-tasters. Chem. Senses 2000, 25, 239–246. [Google Scholar] [CrossRef]
- Albin, K.C.; Carstens, M.I.; Carstens, E. Modulation of oral heat and cold pain by irritant chemicals. Chem. Senses 2008, 33, 3–15. [Google Scholar] [CrossRef]
- Ruparel, S.; Bendele, M.; Wallace, A.; Green, D. Released lipids regulate transient receptor potential channel (TRP)-dependent oral cancer pain. Mol. Pain 2015, 11, 30. [Google Scholar] [CrossRef]
- Pullen, R.L. Management of Alcohol-Induced Peripheral Neuropathy. Nurs. Made Incred. Easy 2019, 17, 28–36. [Google Scholar] [CrossRef]
- Glendinning, J.I.; Simons, Y.M.; Youngentob, L.; Youngentob, S.L. Fetal ethanol exposure attenuates aversive oral effects of TrpV1, but not TrpA1 agonists in rats. Exp. Biol. Med. 2012, 237, 236–240. [Google Scholar] [CrossRef]
- Lautner, M.A.; Ruparel, S.B.; Patil, M.J.; Hargreaves, K.M. In vitro sarcoma cells release a lipophilic substance that activates the pain transduction system via TRPV1. Ann. Surg. Oncol. 2011, 18, 866–871. [Google Scholar] [CrossRef]
- Zanoni, D.K.; Patel, S.G.; Shah, J.P. Changes in the 8th Edition of the American Joint Committee on Cancer (AJCC) Staging of Head and Neck Cancer: Rationale and Implications. Curr. Oncol. Rep. 2019, 21, 52. [Google Scholar] [CrossRef]

| Patient | TNM | Nodal Disease | Location | Sex | Age | Race | EtOH | Tobacco |
|---|---|---|---|---|---|---|---|---|
| 1 | T 2 N1 M0 | Yes | Tongue | M | 47 | Non-Hispanic White | Yes | Yes |
| 2 | T 4 N2 M0 | Yes | BOT & OP | M | 57 | Non-Hispanic White | Yes | Quit |
| 3 | T2 N1 M0 | Yes | FOM | M | 62 | Non-Hispanic White | Yes | Quit |
| 4 | T4 N1 M0 | Yes | Buccal Mucosa | M | 44 | Hispanic White | Yes | Yes |
| 5 | T2N1M0 | Yes | BOT | M | 46 | Hispanic White | Yes | Yes |
| 6 | T2 N1 M0 | Yes | Max. alveolar ridge, Palate, BOT | M | 53 | Non-Hispanic White | Yes | Yes |
| 7 | T2 N2 M0 | Yes | Tongue | M | 63 | Non-Hispanic White | Yes | Quit |
| 8 | T3N0M0 | No | Soft Palate | M | 73 | Hispanic White | Yes | Yes |
| 9 | T4 N0 M0 | No | Mand. alveolar ridge | M | 58 | Hispanic White | Yes | No |
| 10 | T2 N0 M0 | No | Tongue | M | 52 | Hispanic White | Yes | No |
| 11 | T4 N0 M0 | No | Tongue and FOM | F | 81 | Hispanic White | Yes | Quit |
| 12 | T1 N0 M0 | No | Tongue | F | 49 | Non-Hispanic White | Yes | Yes |
| 13 | T1 N0 M0 | No | Tongue | M | 62 | Non-Hispanic White | Yes | Yes |
| 14 | SCC In Situ | No | Tongue | M | 53 | Hispanic White | Yes | Yes |
| 15 | T2 N0 M0 | No | Buccal Mucosa | M | 67 | Non-Hispanic White | Yes | Quit |
| 16 | T2 N0 M0 | No | Tongue | F | 79 | Hispanic White | Yes | Quit |
| 17 | T1 N0 M0 | No | FOM | F | 75 | Non-Hispanic White | Yes | Quit |
| 18 | TxN1M0 | Yes | Tongue | F | 67 | Non-Hispanic White | No | No |
| 19 | T1 N2 M0 | Yes | Palate | M | 58 | Non-Hispanic White | No | Yes |
| 20 | T4 N1 M0 | Yes | Buccal mucosa | F | 81 | Hispanic White | No | No |
| 21 | T1 N1 M0 | Yes | Tongue | F | 50 | Non-Hispanic White | No | No |
| 22 | T1 N0 M0 | No | Tongue | F | 42 | Hispanic White | No | No |
| 23 | T1 N0 M0 | No | Tongue | F | 77 | Hispanic White | No | No |
| 24 | Invasive SCC | No | Max alveolar ridge | F | 78 | Non-Hispanic White | No | No |
| 25 | T4 N0 M0 | No | Tongue | M | Hispanic White | No | No | |
| 26 | T4 N0 M0 | No | Mandible | F | 93 | Non-Hispanic White | No | No |
| 27 | T2 N0 M0 | No | Tongue | F | 57 | Non-Hispanic White | No | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonzales, C.B.; De La Chapa, J.J.; Patwardhan, A.M.; Hargreaves, K.M. Oral Cancer Pain Includes Thermal Allodynia That May Be Attenuated by Chronic Alcohol Consumption. Pharmaceuticals 2023, 16, 518. https://doi.org/10.3390/ph16040518
Gonzales CB, De La Chapa JJ, Patwardhan AM, Hargreaves KM. Oral Cancer Pain Includes Thermal Allodynia That May Be Attenuated by Chronic Alcohol Consumption. Pharmaceuticals. 2023; 16(4):518. https://doi.org/10.3390/ph16040518
Chicago/Turabian StyleGonzales, Cara B., Jorge J. De La Chapa, Amol M. Patwardhan, and Kenneth M. Hargreaves. 2023. "Oral Cancer Pain Includes Thermal Allodynia That May Be Attenuated by Chronic Alcohol Consumption" Pharmaceuticals 16, no. 4: 518. https://doi.org/10.3390/ph16040518
APA StyleGonzales, C. B., De La Chapa, J. J., Patwardhan, A. M., & Hargreaves, K. M. (2023). Oral Cancer Pain Includes Thermal Allodynia That May Be Attenuated by Chronic Alcohol Consumption. Pharmaceuticals, 16(4), 518. https://doi.org/10.3390/ph16040518





